Abstract
Objectives:
Data on triage practices of children admitted to Princess Marina Hospital in Gaborone, Botswana is limited. The inpatient triage, assessment, and treatment score was developed for low resource settings to predict mortality in children. We assess its performance among children admitted to Princess Marina Hospital and their demographic, clinical, and risk factors for death.
Methods:
This was a secondary data analysis of a prospective cohort study comprising 299 children ages 1 month to 13 years admitted June to September 2018. Descriptive statistics, bivariate analysis, and multivariate logistic regression were used. Sensitivity and specificity data were generated for the inpatient triage, assessment, and treatment score.
Results:
Thirteen children died (13/284, 4.6%). Comorbidity (adjusted odds ratio 4.0, p = 0.020) and high inpatient triage, assessment, and treatment score (adjusted odds ratio 5.0, p = 0.017) increased odds of death. The area under the receiver operating characteristic curve was 0.81. Using inpatient triage, assessment, and treatment cutoff of 4, the sensitivity, specificity, and likelihood ratio were 31%, 94%, and 5.0, respectively.
Conclusion:
Implementing the inpatient triage, assessment, and treatment score in low resource settings may improve identification, treatment, and evaluation of the sickest children.
Keywords: Severity of illness, triage, child mortality, global health, pediatrics
Introduction
In 2020, 5.0 million children under 5 years of age died worldwide.1 More than half of deaths within this age group occurred in sub-Saharan Africa, where the leading causes of preventable deaths were due to infection, namely pneumonia, diarrhea, and malaria.1 Pneumonia alone is estimated to account for 15%–18% of all under-five deaths worldwide within the past 5 years; upwards of 90% of these occur in low- and middle-income countries (LMIC).2,3 There are mortality discrepancies across world economies by income with increased all-cause mortality observed in LMIC.4 The highest under-five mortality rate, 76 per 1000 live births, occurs in the World Health Organization (WHO) African Region. Botswana, in sub-Saharan Africa, is considered an upper middle-income country per the World Bank, and it has an under-five mortality rate of 50 per 1000 live births.5
In LMIC, the lack of appropriately triaging, assessing, intervening, and monitoring contributes to increased morbidity and mortality.6 This is important in LMIC where one-third of child deaths occur within 24 hours of presentation to the hospital.7,8 The quality of triage is variable and dependent on healthcare providers’ availability, training, and experience in identifying patients at highest risk of decompensation. Even when training exists, algorithms to promote a uniform triage process are used infrequently.9 Given that catastrophic deterioration has been shown to be preceded by worsening vital signs, early warning scores using vital sign-based scoring to quantify severity of illness (SOI) and trigger intervention have facilitated systematic and effective triage and early treatment of critically ill patients.10
The pediatric early warning score (PEWS) is one of many examples of EWS systems for inpatient use with children11–14 and has been validated in resource-rich settings. Validation of SOI and EWS systems in resource-constrained settings is needed given potential differences in underlying illness, heightened SOI at the time of presentation, fewer providers with specialty experience, and worse ratios of healthcare personnel to patients. The inpatient triage, assessment, and treatment (ITAT) score was the first pediatric SOI scoring system developed for children admitted to resource-constrained hospitals in developing countries (Supplemental Appendix A). The ITAT score was validated in a large Malawian referral hospital with the purpose of identifying hospitalized children at highest risk of death and facilitating urgent clinical reevaluation throughout admission.15
In this manuscript, we assess the ability of the ITAT score to predict mortality among children admitted to Princess Marina Hospital (PMH) in Gaborone, Botswana. We describe mortality in our cohort and examine associated risk factors.
Methods
Study population and location
PMH is the largest tertiary referral hospital in Botswana and main teaching center for the University of Botswana. The patient population comprises patients referred from local clinics, hospitals in southern Botswana and self-referred “walk-ins.” There are more than 530 beds, including 40 pediatric medical and 25 pediatric surgical beds. Pediatric refers to children less than 13 years of age. There are eight intensive care unit beds. Staff includes licensed medical officers (physicians without specialty training), physicians with specialty training in pediatrics or surgery, trainees (interns), and nurses. Each unit is led by a Senior Nurse Matron and has an average nurse-to-patient ratio of 1:4–6 during day shift and 1:5–10 during night shift. A formalized triage protocol, an adaptation of the South African Triage Scale (SATS), has been used by the PMH Accident and Emergency Department since 2010. It is referred to as the PMH Accident and Emergency Scale (PATS).16,17 No scoring systems are used in inpatient wards.
Study design and data collection
This is a secondary data analysis of a prospective cohort study of children 13 years or younger admitted to the medical or surgical pediatric wards of PMH where ITAT score was calculated upon admission, and outcome (survival, death) was recorded at the end of each participant’s admission. Given the observational nature of this study, a specific sample size was not predetermined, and power calculation was not performed. Admissions were screened during daytime hours. Overnight admissions were screened the following morning using admission logs and charts were collected for review. Inclusion criteria included age 13 years or less and hospital admission. Patients greater than 13 years of age or those not requiring admission were excluded. The following data were abstracted from patient medical records and entered into REDCap (Research Electronic Data Capture) hosted by Penn Medicine: demographic and clinical characteristics, antibiotic use, microbiology laboratory results, and in-hospital mortality. Body mass index (BMI; kg/m2) and weight-for-age z-scores were calculated from available anthropometric data.18 Weight-for-age z-scores were based on the WHO Child Growth Charts and Reference 2007, limited to children 10 years old and younger, with values that are 5 standard deviations or more away from the mean set to missing. Condition refers to broad categorization of diagnosis upon admission. Comorbidities included documented preexisting chronic medical conditions. A local research nurse from Botswana explained the study to each caregiver in Setswana (official language of Botswana). Written informed consent was obtained from parent or legal guardian before the study. Vital signs were measured in a standardized way on admission; heart and respiratory rate were manually counted whereas oxygen saturation and temperature were obtained by pulse oximetry and axillary measurements, respectively. ITAT score was calculated from the first set of vital signs documented in the patient’s medical record on admission. Participants lacking key information such as age, admission diagnosis, or vital signs, were not included in the final cohort.
Study tool
The ITAT score is derived from four vital signs: heart rate, respiratory rate, oxygen saturation, and temperature, with age-adjusted ranges established by the PEWS score. Each vital sign is scored from 0 to 2, where 0 reflects normal and higher values reflect greater derangements. Cumulative scores ranged from 0 to 8. A cumulative score of greater than or equal to 4 was defined as high. Furthermore, a high ITAT score was associated with increased odds of death and triggered immediate physician evaluation.
Statistical analysis
Statistical analyses were performed using STATA (StataCorp. 2015. Stata Statistical Software: Release 14. College Station, TX: StataCorp LP). Our primary outcome was in-hospital mortality. Statistical testing between dependent and independent variables was done using Fisher’s exact test where appropriate. Statistical significance was defined as p < 0.05. Independent variables (p < 0.05) in bivariate analysis and clinical relevance were included in binary logistic regression models to calculate odds ratios. A receiver operating characteristic (ROC) curve was plotted using the sensitivity and specificity (1—specificity) for each ITAT score cutoff. Then, the area under the curve (AUC) was calculated. Variables with missing values totaling less than 299 responses are noted both within the text and in each table as some of the data is incomplete despite prospective data collection, given the study tool was a paper document and data collection and recording were performed by hand.
Ethical review
The study was approved by the institutional review boards of PMH, Botswana’s Ministry of Health Research and Development Committee, the University of Botswana, the Children’s Hospital of Philadelphia, and the University of Pennsylvania.
Results
Sociodemographic and clinical characteristics
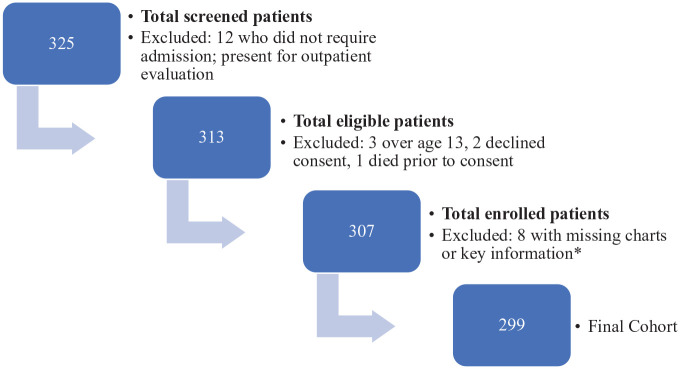
A total of 325 patients were consecutively screened between June and September 2018 resulting in a final cohort of 299 patients (Figure 1). Of 299 participants, 67.8% (202/299) were medical admissions, 44% (131/298) were female, median age was 31.5 months, and 65.9% (182/279) of the participants were less than 5 years of age (Table 1). Of 212 children, 33 (16.7%) had weight-for-age z-scores ⩽2, corresponding to underweight or severely underweight, of which four died. The prevalence of HIV for the entire cohort was 4.4% (10/228). Of children living with HIV, 6/10 (60.0%) were already on antiretroviral (ARV) therapy, 2/10 (20%) were not on ARV, and ARV exposure was unknown for 2/10 (20%) children. There was one newly diagnosed case of HIV. HIV exposure in participants 6 months of age and younger was 28.0% (14/50). Data analysis for HIV exposure was limited to younger participants given heightened risk of seroconversion for children 6 months of age or younger compared to their older counterparts. Infection (of any type) was the most common condition (88/299, 29.4%), with pneumonia (33/88, 37.5%) occurring at higher frequency than gastroenteritis (19/88, 21.6%) and generalized sepsis (17/88, 19.3%). More than a quarter of the cohort had an underlying comorbidity (84/299, 28.1%). Our study details comorbidities including, but not limited to, the following: congenital heart disease, prematurity, genetic and metabolic disease, cytopenias, chronic malnutrition, and HIV.
Figure 1.
Structure of study cohort.
*Charts where key information such as age, admission diagnosis, or vital signs was missing were not included.
Table 1.
Demographic and clinical characteristics of children (⩽13 years) admitted to PMH, June–September 2018 (N = 299)*.
| Variables | N (%) or median Interquartile range (IQR) |
|---|---|
| Age months (n = 279) | 31.5 (8.4, 78.3) |
| Age groups months (n = 279) | |
| 0–1 | 24 (8.6) |
| 2–6 | 34 (12.2) |
| 7–11 | 23 (8.2) |
| 12–24 | 34 (12.2) |
| 25–60 | 69 (24,7) |
| >60 | 95 (34.1) |
| Sex (n = 298) | |
| Male | 167 (56.0) |
| Female | 131 (44.0) |
| Weight, kg (n = 263) | 12.2 (7.6, 18.3) |
| Body Mass Index (BMI) (n = 151) | 16.5 (14.0, 17.6) |
| Weight-for-age z-score (n = 212) | −0.7 (−1.6, 0.3) |
| Location (n = 299) | |
| Medical | 202 (67.6) |
| Surgical | 97 (32.4) |
| HIV-exposed, age 6 months or less (n = 50) | |
| No | 36 (72.0) |
| Yes | 14 (28.0) |
| HIV-positive, age greater than 6 months (n = 228) | |
| No | 218 (95.6) |
| Yes | 10 (4.4) |
| Conditions, by type (n = 299) | |
| Infection† | 88 (29.4) |
| Orthopedic/osteoarticular | 34 (11.4) |
| Surgical, general | 32 (10.7) |
| Neurologic/developmental | 28 (9.4) |
| Oncologic | 27 (9.0) |
| Gastrointestinal | 20 (6.7) |
| Renal | 4 (1.3) |
| Respiratory | 8 (2.7) |
| Cardiovascular | 13 (4.3) |
| Toxic/accidental | 11 (3.7) |
| Surgical, plastics | 10 (3.3) |
| Hematologic | 10 (3.3) |
| Endocrine/metabolic | 9 (3.0) |
| Dermatologic | 5 (1.8) |
| Prescribed antibiotic (n = 298) | |
| No | 146 (49.0) |
| Yes | 152 (51.0) |
| Comorbidity (n = 299) | |
| No | 215 (71.9) |
| Yes | 84 (28.1) |
| ITAT‡ (n = 299) | |
| <4 | (92.3) |
| ⩾4 | 23 (7.7) |
| Outcome (n = 284) | |
| Survival | 271 (95.4) |
| Death | 13 (4.6) |
Missing data were excluded where data does not equal full cohort of 299.
Regardless of primary organ system, infections were grouped together.
ITAT. A score from 0 to 2 was given for each vital sign based on the age-associated degree of abnormality, with higher scores resulting from more abnormal vital signs. A cumulative ITAT score of 4 or higher was associated with increased odds of death.
Inpatient triage, assessment, and treatment score
ITAT scores ranged from 0 to 8 (median 1, IQR 1, 2). There were 23/299 patients (7.7%) with high ITAT score. In sub-analyses with ITAT score, patients with high ITAT score were more likely to be admitted to medical rather than surgical units (21/23 versus 181/276, p = 0.010). High ITAT score was associated with infection (13/23 versus 75/276, p = 0.082).
Outcomes
Outcomes were available for 284/299 (95%) children and included 13 deaths (13/284, 4.6%), details in Table 2, stratified by full cohort and medical admissions only. Patients transferred to another institution were considered survivors (25/284, 8.8%). Outcomes were unknown for 15 participants. The following factors demonstrated statistically significant associations with death independently, when analyzed by entire cohort and limited to medical admissions alone: malnutrition, antibiotic exposure, comorbidity, and high ITAT. Median time from presentation to death was 3 days (IQR 2, 6) and median age of children who died was 15 months (IQR 8, 71) compared to 34 months (IQR 9, 79) for survivors. Six patients who died were HIV-exposed, including two who were HIV-positive. Ten of the 13 patients (77%) died of infection. There was significant overlap in the conditions listed on or around day of death, some patients had multiple infection-related processes. Seven died of generalized sepsis, two of pneumonia, and one of meningitis; however, 7/13 (53.8%) fatalities were diagnosed with pneumonia or respiratory failure during their hospital course. Blood cultures were sent in 10/13 (76.9%) children who died; four yielded positive results (4/10, 40%). Of the positive results, time to positivity varied between 3 and 7 days. With respect to survivors, 92/270 (34.1%) had blood cultures sent with 19/92 (20.7%) yielding a positive result.
Table 2.
Bivariate analysis of factors by outcome for children (⩽13 years) admitted to PMH, June–September 2018 (n = 284)*.
| Variables | Bivariate | |||||
|---|---|---|---|---|---|---|
| All (n = 284) | p-Value | Medical only (n = 202) | p-Value | |||
| Death, N (%) | Survival, N (%) | Death, N (%) | Survival, N (%) | |||
| Age group, months | 0.255 | 0.458 | ||||
| 0–1 | 0 (0) | 23 (9.1) | 0 (0) | 16 (9.1) | ||
| 2–6 | 2 (16.7) | 32 (12.6) | 2 (16.7) | 23 (13.1) | ||
| 7–11 | 3 (25.0) | 19 (7.5) | 3 925.0) | 15 (8.5) | ||
| 12–24 | 2 (16.7) | 28 (11.0) | 2 (16.7) | 24 (13.6) | ||
| 25–60 | 2 (16.7) | 65 (25.6) | 2 (16.7) | 45 (25.6) | ||
| >60 | 3 (25.0) | 87 (34.3) | 3 (25.0) | 53 (30.1) | ||
| Sex | 0.659 | 0.714 | ||||
| Male | 6 (50.0) | 153 (56.5) | 6 (50.0) | 102 (55.4) | ||
| Female | 6 (50.0) | 118 (43.5) | 6 (50.0) | 82 (44.6) | ||
| Location | 0.071 | Deferred | ||||
| Medical | 12 (92.3) | 184 (67.9) | ||||
| Surgical | 1 (7.7) | 87 (32.1) | ||||
| Weight-for-age z-score | 0.037 | 0.038 | ||||
| Less than or equal to −2 | 4 (50.0) | 33 (16.7) | 4 (50.0) | 25 (16.7) | ||
| Greater than −2 | 4 (50.0) | 164 (83.3) | 4 (50.0) | 125 (83.3) | ||
| HIV-exposed, age 6 months or less | 0.077 | 0.124 | ||||
| No | 0 (0) | 35 (74.5) | 0 (0) | 23 (67.7) | ||
| Yes | 2 (100) | 12 (25.5) | 2 (100) | 11 (32.3) | ||
| HIV-positive, age greater than 6 months | 0.071 | 0.110 | ||||
| No | 8 (80) | 198 (96.1) | 8 (80) | 135 (95.1) | ||
| Yes | 2 (20) | 8 (3.9) | 2 (20) | 7 (4.9) | ||
| Conditions, by type | 0.240 | 0.574 | ||||
| Infection† | 8 | 78 | 0 | 75 (40.8) | ||
| Orthopedic/osteoarticular | 0 | 32 | 0 | 0 (0) | ||
| Surgical, general | 0 | 30 | 0 | 4 (2.2) | ||
| Neurologic/developmental | 0 | 27 | 0 | 26 (14.1) | ||
| Oncologic | 1 | 23 | 0 | 23 (12.5) | ||
| Gastrointestinal | 3 | 15 | 0 | 8 (4.4) | ||
| Renal | 0 | 4 | 0 | 2 (1.1) | ||
| Respiratory | 0 | 8 | 0 | 7 (3.8) | ||
| Cardiovascular | 0 | 13 | 0 | 0 (0) | ||
| Toxic/accidental | 0 | 10 | 0 | 4 (2.2) | ||
| Surgical, plastics | 0 | 9 | 0 | 0 (0) | ||
| Hematologic | 1 | 8 | 0 | 8 (4.4) | ||
| Endocrine/metabolic | 0 | 9 | 0 | 9 (4.9) | ||
| Dermatologic | 0 | 5 | 0 | 5 (2.7) | ||
| Prescribed antibiotic | 0.003 | 0.014 | ||||
| No | 1 (7.7) | 137 (50.7) | 1 (8.3) | 83 (45.1) | ||
| Yes | 12 (92.3) | 133 (49.3) | 11 (91.7) | 101 (54.9) | ||
| Comorbidity | 0.008 | 0.027 | ||||
| No | 5 (38.5) | 202 (74.5) | 4 (33.3) | 123 (66.9) | ||
| Yes | 8 (61.5) | 69 (25.5) | 8 (66.7) | 61 (33.2) | ||
| ITAT‡ | 0.012 | 0.028 | ||||
| Low | 9 (69.2) | 253 (93.4) | 8 (66.7) | 167 (90.8) | ||
| High | 4 (33.3) | 18 (6.6) | 4 (33.3) | 17 (9.2) | ||
A total of 284 patients had known outcomes. Missing data were excluded where data does not equal full cohort with known outcomes.
Regardless of primary organ system, infections were grouped together.
A score from 0 to 2 was given for each vital sign based on the age-associated degree of abnormality, with higher scores resulting from more abnormal vital signs. A cumulative ITAT score of 4 or higher was associated with increased odds of death.
Median length of stay was 5 days (IQR 3, 8). All but one of the children who died was treated with antibiotics, whereas 133/270 (49.3%) survivors were treated with antibiotics. The remaining three patients who did not die of infection had the following conditions: leukemia complicated by tumor lysis syndrome, hemophagocytic lymphohistiocytosis, and acute liver failure of unknown etiology.
The proportion of children with high ITAT scores (4/13 versus 18/271, p = 0.012) and single ITAT vital sign of 2 (9/13 versus 52/266, p < 0.001) was significantly greater among fatalities than survivors. Table 3 displays multivariate analysis. In the model adjusting for presence of comorbidity and high ITAT score, both hypothesized to increase odds of death, presence of a comorbidity (adjusted odds ratio (OR) 4.0; 95% CI: 1.2−13.1, p = 0.020) and high ITAT score (adjusted OR 5.0; 95% CI: 1.3−18.4, p = 0.017) were statistically significant predictors of death. The AUC was 0.81 (95% CI: 0.7–0.9). At a predetermined ITAT score cutoff value of 4 as proposed in the original ITAT study, the sensitivity, specificity, and likelihood ratio of death were 31%, 94%, and 5.0, respectively. The positive and negative predictive values were 0.18 and 0.97, respectively (Figure 2).
Table 3.
Multivariate analysis of factors associated with death for children (⩽13 years) admitted to PMH, June–September 2018 (N = 299).
| Variables | Multivariate analysis | |
|---|---|---|
| Odds ratio (95% CI) | p-Value | |
| Age group, months (n = 279) | ||
| 0–1 | 1 | — |
| 2–6 | 1.8 (0.3, 11.3) | 0.525 |
| 7–11 | 4.6 (0.9, 24.5) | 0.075 |
| 12–24 | 2.1 (0.3, 13.0) | 0.438 |
| 25–60 | 0.9 (0.1, 5.5) | 0.902 |
| >60 | Reference | |
| Sex (n = 298) | 0.660 | |
| Male | Reference | |
| Female | 1.3 (0.4, 4.1) | |
| Weight-for-age (n = 205) | 0.035 | |
| Less than or equal to −2 | 5.0 (1.2, 20.9) | |
| Greater than −2 | Reference | |
| Location (n = 299) | 0.098 | |
| Medical | 5.7 (0.7, 44.3) | |
| Surgical | Reference | |
| HIV-positive, age greater than 6 months (n = 228) | 0.036 | |
| No | Reference | |
| Yes | 6.2 (1.1, 34.0) | |
| Prescribed antibiotic (n = 298) | 0.016 | |
| No | Reference | |
| Yes | 12.4 (1.6, 96.4) | |
| Comorbidity (n = 299) | 0.009 | |
| No | Reference | |
| Yes | 4.7 (1.5–14.8) | |
| ITAT* (n = 299) | 0.005 | |
| Low | Reference | |
| High | 6.2 (1.8–22.3) | |
ITAT. A score from 0 to 2 was given for each vital sign based on the age-associated degree of abnormality, with higher scores resulting from more abnormal vital signs. A cumulative ITAT score of 4 or higher was associated with increased odds of death.
Figure 2.
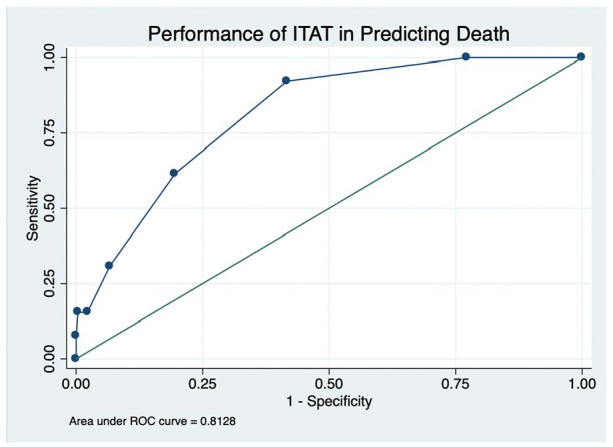
The ROC curve for the ITAT score
At an ITAT cutoff value of 4, the sensitivity, specificity, and likelihood ratio were 31%, 94%, and 5.0, respectively. The positive and negative predictive values were 0.29 and 0.95, respectively.
Discussion
This paper enriches the sparse body of literature on survival outcomes of children admitted to tertiary care facilities in LMIC like PMH. In resource-constrained settings where the child mortality rate is disproportionately higher than that of high-income countries, it is essential to optimize odds of survival over time.19 Systematically implementing effective SOI scores with corresponding escalation and intervention may facilitate achieving this goal by identifying those in immediate need of intervention.
The inpatient pediatric mortality rate at PMH was substantially lower than that of comparable settings in sub-Saharan Africa. Our cohort’s mortality rate of 4.6% mirrored that reported in an independent study from PMH limited to children less than 24 months with pneumonia (4.8%).2 In contrast to mortality rates reported for children at PMH, the cohort from Kamuzu Central Hospital in Lilongwe, Malawi where the ITAT score was validated, reported an inpatient pediatric mortality rate of 7%–9%, where 44% and 59% of deaths occurred within the first 24 and 48 h of admission, respectively.15,20 Time to death was longer in our cohort where 15% and 46% of deaths occurred within the first 24 and 48 h, respectively. Reasons for this may include a less critically ill cohort upon presentation, faster recognition of critically ill children in the Emergency Department, and/or greater availability of resources. Furthermore, malaria is endemic to Lilongwe and not to Gaborone; pediatric mortality due to malaria may account for higher mortality rates at Kamuzu Central Hospital. Lastly, the study population in Malawi only included admissions to the acute care and malnutrition wards, specifically excluding surgical patients who tended to be less ill, potentially reflecting a proportion of elective admissions, who were already receiving specialized care. Our bivariate analysis is divided into total cohort and medical admission only, to demonstrate that medical admissions represented a more critically ill segment of the participants, also evidenced by having a greater proportion of high ITAT than surgical admissions. The factors demonstrating statistically significant associations with death for the entire cohort also did so when limited to medical admissions, supporting our hypothesis that the medical ward represented a more critically ill population.
The patient’s major conditions on day of or surrounding day of death served as surrogate cause of death, which is a common occurrence in clinical settings where diagnostic laboratory and radiological tools that aid in identifying exact cause of death may not be available or pursued.20 Despite the prevalence of infection within the cohort, the use of microbiology results was of limited benefit in affecting clinical management in real-time given lag time between blood culture result and inconsistency in reporting both culture and Gram stain results. For instance, a final organism was reported in only two cases; the speciation of the remaining positive blood cultures was unknown.
A prospective cohort study in a district hospital in Kenya described that less than 50% of dying children had blood cultures, possibly underestimating the true risk of bacteremia.21 Similarly, a study of children admitted to PMH hypothesized that overuse and prolonged duration of antibiotics could be secondary to limited blood culture availability.22 Both studies emphasize how antibiotic use reflects the overwhelming burden of infectious diseases among children at PMH. Increasing antibiotic utilization in Botswana was evaluated through the National Action Plan for antimicrobials in 2016. In addition to underscoring the burden of infectious disease, the plan noted irrational antibiotic use, inconsistent ordering of cultures and drug susceptibility testing, and a lack of antibiograms and antibiotic guidelines.23
Pneumonia is the leading cause of preventable death in children under-five in LMIC, accounting for an estimated 800,000 deaths annually primarily in sub-Saharan Africa and Asia.3 Considering lower respiratory tract infection represents about 40% of infectious causes of death in African children, the incidence of pneumonia-related deaths was higher in our cohort (7/13, 53.8%).24 Hospitalization and death due to pneumonia have declined by nearly 50% in Botswana since the national introduction of vaccines against Haemophilus influenzae type B (Hib) and Streptococcus pneumoniae (PCV13) in 2010 and 2012, respectively.25 Similarly, a reduction in gastroenteritis-related hospitalizations and deaths in the post-rotavirus vaccine period have also been demonstrated in Botswana.26 Although vaccination status was not collected in our study, it may have been informative as a potential predictor of death considering the burden of infectious disease within our population and separately as a marker of adherence to national vaccine campaigns, with respect respiratory and gastrointestinal infection. It is worth noting that our short study period may not have fully captured the true frequency of gastroenteritis within the population which varies by season and country within sub-Saharan Africa.27
Although data on pediatric comorbidity and mortality in LMIC are limited, comorbidity has been demonstrated to increase odds of death, particularly in children with pneumonia.28,29 The presence of any comorbidity increased odds of death within our cohort. While malnutrition, especially co-occurring with infectious causes of death, has been described as a risk factor for death, height and weight were limited in our cohort.30 Of fatalities, three were underweight and one was severely underweight (4/13, 30.8%), slightly below rates in sub-Saharan Africa where about 45% of all child deaths are linked to malnutrition.31 HIV exposure (28.0%) in our cohort paralleled the national prevalence of HIV infection of adults in Botswana. Botswana has the fourth highest prevalence of HIV in the world, where 20.3% of adults aged 15 and 49 years were positive as of 2020.32 A previous study from PMH demonstrated HIV-exposed but negative children with pneumonia had increased risk of in-hospital death.33 In our cohort, there was an association between HIV infection and mortality which aligns with existing data where approximately 50% of deaths in children less than 2 years old in Botswana are HIV-positive or exposed.34 This analysis only reports data on ARV for children living with HIV; we assumed that all exposed children were on prophylactic ARV in line with the long-standing, national “Prevention of Mother-to-Child Transmission” program.35,36 The majority of patients who died were less than 5 years old; around a third were infants. No neonatal deaths occurred in our cohort. This is likely attributed to the structure of PMH where there is a separate newborn nursery and neonatal unit where neonatal deaths were more likely to occur.
The majority of children had low ITAT scores; however, the proportion of high ITAT scores was significantly greater among fatalities than survivors. As in the original validation study, having high ITAT score or a score of 2 for any single vital sign were independent predictors of death within our cohort. The AUC in our study was slightly higher than that reported in the original study. Our study’s sensitivity was lower, specificity was higher, and likelihood ratio was higher. When considering cutoff values for ITAT scores at which to trigger evaluation by a medical provider given increasing risk of death, our study maintained the predetermined cutoff value of 4 established in Olson et al.’s15 original validation of the ITAT score. In our cohort a lower score of 3 compared to 4, would have doubled the sensitivity without substantially changing the specificity or proportion correctly classified. This is meaningful if there are adequate resources available to allow for screening a greater number of patients, recognizing that not all may be at increased risk of deterioration.
The sensitivity at ITAT score 3 or 4 is far lower than demonstrated in more complex triage scores designed for resource-rich settings. For example, PEWS (sensitivity 78%, specificity 95% at cutoff score 5) and bedside PEWS (sensitivity 82%, specificity 93% at cutoff score 8) differ from the ITAT score by incorporating screening criteria such as capillary refill, blood pressure, and mental status in addition to interventions like fluid resuscitation and oxygen therapy.11,12 Despite having reduced sensitivity, the ITAT score’s specificity is comparable to the aforementioned scores. It exceeds that of the triage early warning score (TEWS) with specificity 61%, and of the SATS, which combines TEWS and clinical indicators with 55% specificity.16,37,38 Prioritizing greater specificity in lower-resourced settings may be beneficial when fewer patients with a greater probability of being ill may be screened efficiently. The simplicity of the ITAT score may help bolster its generalizability to the most resource-constrained settings where specific screening methods or interventions may not be available.
Simplicity and ease of application should be prioritized in order to allow for support staff who may lack the medical training to operationalize SOI scores for screening and monitoring. ITAT score does not incorporate medical interventions, clinical signs of distress, or assess mobility, mental status, or trauma—elements of PEWS and TEWS—that may be more challenging for personnel with minimal medical training to evaluate and execute. In fact, the score was originally validated with healthcare workers with minimal medical training who were trained to use the tool. We also relied on healthcare workers to determine ITAT score. This highlights the importance of empowering support staff with varying degrees of medical training to offset the responsibility of serial assessments from more specialized healthcare providers who may be less accessible in the inpatient hospital settings of LMIC.
Limitations
Our data collection was limited to 3 months and may not reflect seasonal variation in presenting conditions. Utilizing a convenience sampling approach rather than power calculation to determine sample size may only represent a segment of potential admission and in turn reduce generalizability. Inconsistent chart documentation resulted in missing values despite daily data collection. We hypothesize this was due to challenges with collecting, recording, and storing data by hand in the absence of electronic record keeping. Furthermore, there is a separate opportunity for incomplete data entry upon manual entry into the online database. We acknowledge the data presented may not accurately reflect the entirety of the pediatric patient population at PMH; however, the key variables such as outcome and ITAT score are well-represented and allow for meaningful analysis and interpretation. Although the ITAT score was designed for continuous patient monitoring after admission, there have been no prospective cohort studies to demonstrate its potential impact on morbidity and mortality. Our findings from research conducted at a tertiary hospital may be less generalizable to a more rural clinical setting with fewer resources.
Conclusion
This analysis confirms the potential generalizability of a simple score like ITAT designed for use in LMIC to predict death in other under resourced settings. Furthermore, this paper presents a general characterization of the pediatric population admitted to the general pediatrics ward of PMH, including but not limited to, their disease, comorbidity, and mortality. Wide, consistent application of the ITAT score with children admitted to PMH may serve to improve early and accurate identification of those sickest to effectively intervene upon and regularly monitor to optimize survival. Additional research must be done to evaluate the feasibility of daily implementation of ITAT scores for children admitted to PMH. Future investigations may have the potential to measure how standardized monitoring with ITAT may definitively reduce mortality.
Supplemental Material
Supplemental material, sj-docx-1-smo-10.1177_20503121221149356 for Severity of illness and mortality among children admitted to a tertiary referral hospital in Botswana: A secondary data analysis of a prospective cohort study by Sheyla Denise Richards, Molly Hayes, Loeto Mazhani, Tonya Arscott-Mills, Unami Mulale, Susan Coffin, Andrew P Steenhoff and Eimear Kitt in SAGE Open Medicine
Acknowledgments
This study was made possible by the staff at Princess Marina Hospital and the Children’s Hospital of Philadelphia’s Global Health Center, through which global health research and mentorship is made possible for pediatric residents. We also wish to acknowledge Kristel Emmer, Kara (Catalyst) Twomey, and Catherine Sayikanmi, for their assistance in data collection.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval: The study was approved by the institutional review boards of Princess Marina Hospital, Botswana’s Ministry of Health Research and Development Committee, the University of Botswana, the Children’s Hospital of Philadelphia, and the University of Pennsylvania (IRB Reference: PMH 5/79(403-1-2018)).
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Melissa Ketunuti Endowment at the Children’s Hospital of Philadelphia. This publication was made possible through core services and support from the Penn Center for AIDS Research, a National Institutes of Health-funded program [grant number P30 AI 045008. SEC receives research support from Merck unrelated to this project. Otherwise, the authors have no pharmaceutical, industrial, or additional funding to disclose.
Informed consent: Written informed consent was obtained from legally authorized representatives before the study.
Data accessibility statement: Data is available upon request.
ORCID iDs: Sheyla Richards  https://orcid.org/0000-0001-8082-1607
https://orcid.org/0000-0001-8082-1607
Andrew P Steenhoff  https://orcid.org/0000-0001-9002-8207
https://orcid.org/0000-0001-9002-8207
Eimear Kitt  https://orcid.org/0000-0003-0142-2726
https://orcid.org/0000-0003-0142-2726
Supplemental material: Supplemental material for this article is available online.
References
- 1. UNICEF. Under-five mortality [Internet], https://data.unicef.org/topic/child-survival/under-five-mortality/ (2020, accessed 7 March 2021).
- 2. Kelly MS, Crotty EJ, Rattan MS, et al. Chest radiographic findings and outcomes of pneumonia among children in Botswana. Pediatr Infect Dis J 2016; 35(3): 257–262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. World Health Organization. Pneumonia [Internet], https://www.who.int/en/news-room/fact-sheets/detail/pneumonia (2019, accessed 2 April 2021).
- 4. World Health Organization. Global Health Observatory data: under-five mortality [Internet], https://www.who.int/gho/child_health/mortality/mortality_under_five_text/en/#:~:text=5.3millionchildrenunderagefivediedin2018.&text=In2018%2Ctheunder-five,deathsper1000livebirths (2020, accessed 10 January 2021).
- 5. World Health Organization. Country statistics global health estimates. Botswana, United Nations: WHO Statistical Profile; p.3 [Internet], https://www.who.int/data/gho/data/countries/country-details/GHO/botswana?countryProfileId=7dac7ff9-b2c1-4ac5-9b71-2ba59ad1636d (2015, accessed 4 August 2020). [Google Scholar]
- 6. Tamburlini G, di Mario S, Maggi RS, et al. Evaluation of guidelines for emergency triage assessment and treatment in developing countries. Arch Dis Child 1999; 81(6): 478–482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Baker T. Critical care in low-income countries. Trop Med Int Health 2009; 14(2): 143–148. [DOI] [PubMed] [Google Scholar]
- 8. Molyneux E, Ahmad S, Robertson A. Improved triage and emergency care for children reduces inpatient mortality in a resource-constrained setting. Bull World Health Organ 2006; 84(4): 314–319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Nolan T, Angos P, Cunha AJLA, et al. Quality of hospital care for seriously ill children in less-developed countries. Lancet 2001; 357(9250): 106–110. [DOI] [PubMed] [Google Scholar]
- 10. Subbe CP, Kruger M, Rutherford P, et al. Validation of a modified early warning score in medical admissions. QJM 2001; 94(10): 521–526. [DOI] [PubMed] [Google Scholar]
- 11. Parshuram CS, Hutchison J, Middaugh K. Development and initial validation of the Bedside Paediatric Early Warning System score. Crit Care 2009; 13(4): 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Duncan H, Hutchison J, Parshuram CS. The pediatric early warning system score: a severity of illness score to predict urgent medical need in hospitalized children. J Crit Care 2006; 21(3): 271–278. [DOI] [PubMed] [Google Scholar]
- 13. Monaghan A. Detecting and managing deterioration in children. Paediatr Nurs 2005; 17(1): 1–4. [DOI] [PubMed] [Google Scholar]
- 14. Haines C, Perrott M, Weir P. Promoting care for acutely ill children: development and evaluation of a Paediatric Early Warning Tool. Intensive Crit Care Nurs 2006; 22(2): 73–81. [DOI] [PubMed] [Google Scholar]
- 15. Olson D, Davis NL, Milazi R, et al. Development of a severity of illness scoring system (inpatient triage, assessment and treatment) for resource-constrained hospitals in developing countries. Trop Med Int Health. 2013; 18(7): 871–878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Twomey M, Wallis LA, Thompson ML, et al. The South African Triage Scale (adult version) provides reliable acuity ratings. Int Emergency Nurs 2012; 20(3): 142–150. [DOI] [PubMed] [Google Scholar]
- 17. Chandra A, Mullan P, Ho-Foster A, et al. Epidemiology of patients presenting to the emergency centre of Princess Marina Hospital in Gaborone, Botswana. Afr J Emerg Med 2014; 4(3): 109–114. [Google Scholar]
- 18. Vidmar SI, Cole TJ, Pan H. Standardizing anthropometric measures in children and adolescents with functions for Egen: update. Stata J 2013; 13: 366–378. [Google Scholar]
- 19. You D, Hug L, Ejdemyr S, et al. Global, regional, and national levels and trends in under-5 mortality between 1990 and 2015, with scenario-based projections to 2030: a systematic analysis by the un Inter-Agency Group for Child Mortality Estimation. The Lancet 2015; 386(10010): 2275–2286. [DOI] [PubMed] [Google Scholar]
- 20. Fitzgerald E, Mlotha-Mitole R, Ciccone EJ, et al. A pediatric death audit in a large referral hospital in Malawi. BMC Pediatr 2018; 18(1): 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Aiken AM, Mturi N, Njuguna P, et al. Risk and causes of paediatric hospital-acquired bacteraemia in Kilifi District Hospital, Kenya: a prospective cohort study. Lancet 2011; 378(9808): 2021–2027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Fisher BT, Meaney PA, Shah SS, et al. Short report: antibiotic use in pediatric patients admitted to a referral hospital in Botswana. Am J Trop Med Hyg 2009; 81(1): 129–131. [PubMed] [Google Scholar]
- 23. Tiroyakgosi C, Matome M, Summers E, et al. Ongoing initiatives to improve the use of antibiotics in Botswana: University of Botswana symposium meeting report. Expert Rev Anti Infect Ther 2018; 16(5): 381–384. [DOI] [PubMed] [Google Scholar]
- 24. Reiner RC, Welgan CA, Casey DC, et al. Identifying residual hotspots and mapping lower respiratory infection morbidity and mortality in African children from 2000 to 2017. Nat Microbiol 2019; 4(12): 2310–2318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Congdon M, Hwanhee H, Young RR, et al. Effect of Haemophilus influenzae type b and 13-valent pneumococcal conjugate vaccines on childhood pneumonia hospitalizations and deaths in Botswana. Clin Infect Dis 2021; 73: e410–e416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Enane LA, Gastañaduy PA, Goldfarb DM, et al. Impact of rotavirus vaccination on hospitalizations and deaths from childhood gastroenteritis in Botswana. Clin Infect Dis 2016; 62: S168–S174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Munjita SM. Current status of norovirus infections in children in sub-Saharan Africa. J Trop Med 2015; 2015: 309648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Caggiano S, Ullmann N, de Vitis E, et al. Factors that negatively affect the prognosis of pediatric community-acquired pneumonia in district hospital in Tanzania. Int J Mol Sci 2017;18(3): 623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Sonego M, Pellegrin MC, Becker G, et al. Risk factors for mortality from acute lower respiratory infections (ALRI) in children under five years of age in low and middle-income countries: a systematic review and meta-analysis of observational studies. PLoS One 2015; 10(1): 1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Black RE, Victora CG, Walker SP, et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013; 382(9890): 427–451. [DOI] [PubMed] [Google Scholar]
- 31. World Health Organization: Regional Office for Africa. Child health [Internet], https://www.afro.who.int/health-topics/child-health (2021, accessed 30 July 2022)
- 32. Central Intelligence Agency. HIV/AIDS – adult prevalence rate [Internet], https://www.cia.gov/the-world-factbook/about/archives/2021/field/hiv-aids-adult-prevalence-rate#:~:text=11.6%25%20(2020%20est (2016, accessed 7 March 2021).
- 33. Kelly MS, Zheng J, Boiditswe S, et al. Investigating mediators of the poor pneumonia outcomes of human immunodeficiency virus – exposed but uninfected children. J Pediatric Infect Dis Soc 2019; 8(1): 13–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Zash R, Souda S, Leidner J, et al. HIV-exposed children account for more than half of 24-month mortality in Botswana. BMC Pediatr 2016; 16(1): 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Kebaabetswe PM. Barriers to participation in the prevention of mother-to-child HIV transmission program in Gaborone, Botswana a qualitative approach. AIDS Care 2007; 19(3): 355–360. [DOI] [PubMed] [Google Scholar]
- 36. Bagcchi S. Prevent mother to child. Lancet 2022; 22(3): 1. [DOI] [PubMed] [Google Scholar]
- 37. Twomey M, Mullan PC, Torrey SB, et al. The Princess Marina Hospital accident and emergency triage scale provides highly reliable triage acuity ratings. Emergency Med J 2012; 29(8): 650–653. [DOI] [PubMed] [Google Scholar]
- 38. Twomey M, Cheema B, Buys H, et al. Vital signs for children at triage: a multicentre validation of the revised South African triage scale (SATS) for children. S Afr Med J 2013; 103(5): 304–308. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-docx-1-smo-10.1177_20503121221149356 for Severity of illness and mortality among children admitted to a tertiary referral hospital in Botswana: A secondary data analysis of a prospective cohort study by Sheyla Denise Richards, Molly Hayes, Loeto Mazhani, Tonya Arscott-Mills, Unami Mulale, Susan Coffin, Andrew P Steenhoff and Eimear Kitt in SAGE Open Medicine