Abstract
The NAD(P)HX repair system is a metabolite damage repair mechanism responsible for restoration of NADH and NADPH after their inactivation by hydration. Deficiency in either of its two enzymes, NAD(P)HX dehydratase (NAXD) or NAD(P)HX epimerase (NAXE), causes a fatal neurometabolic disorder characterized by decompensations precipitated by inflammatory stress. Clinical findings include rapidly progressive muscle weakness, ataxia, ophthalmoplegia, and motor and cognitive regression, while neuroimaging abnormalities are subtle or nonspecific, making a clinical diagnosis challenging. During stress, nonenzymatic conversion of NAD(P)H to NAD(P)HX increases, and in the absence of repair, NAD(P)H is depleted, and NAD(P)HX accumulates, leading to decompensation; however, the contribution of each to the metabolic derangement is not established. Herein, we summarize the clinical knowledge of NAXE deficiency from 30 cases and lessons learned about disease pathogenesis from cell cultures and model organisms and describe a metabolomics signature obtained by untargeted metabolomics analysis in one case at the time of crisis and after initiation of treatment. Overall, biochemical findings support a model of acute depletion of NAD+, signs of mitochondrial dysfunction, and altered lipidomics. These findings are further substantiated by untargeted metabolomics six months post-crisis showing that niacin supplementation reverses primary metabolomic abnormalities concurrent with improved clinical status.
Keywords: NAD(P)HX epimeras, NAXE, PEBEL1, Fever induced encephalopathy, Neurometabolic disorders, Mitochondrial dysfunction, Pellagra, NAD+
1. Introduction
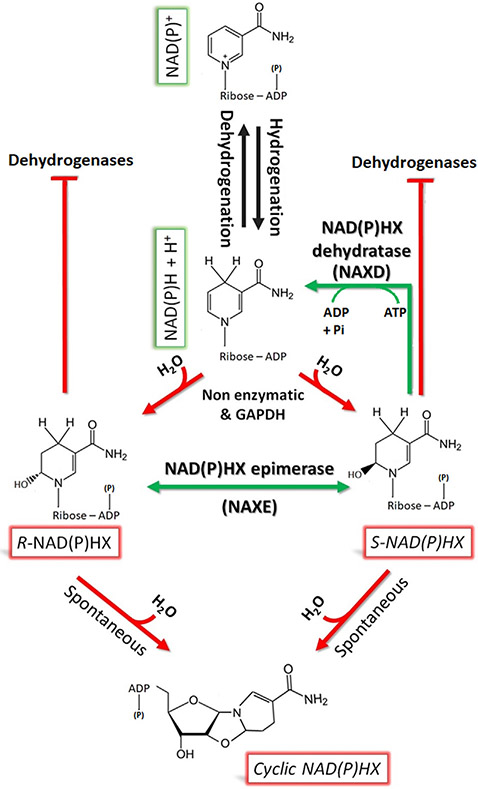
The human gene, NAXE, encodes an enzyme with two vastly different functions. Within the cell, NAXE is an epimerase, interconverting the S and R enantiomers of hydrated reduced nicotinamide adenine dinucleotide (phosphate) [S- and R- NAD(P)HX] [1]. Outside of the cell, it interacts with apolipoprotein A-I (APOA1) [2] to promote cholesterol flux from endothelial cells and macrophages [3] into high density lipoprotein particles (HDL) [4], resulting in lipid raft disassembly [5,6] and activation of Notch1 signaling [7]. Because of its interaction with APOA1, NAXE is also known as APOA1 binding protein (AIBP). Its epimerase activity is part of a two-enzyme NAD(P)HX repair system comprised of the epimerase and an enantiomer-specific dehydratase (NAXD), which together repair inactive NAD(P)HX back to NAD(P)H. NAD(P)H is converted into NAD(P)HX either nonenzymatically in an acidic and hyperthermic environment or as a side product of glyceraldehyde-3-phosphate dehydrogenase (Fig. 1) [8]. NAD(P)HX is thought to be cytotoxic via the inhibition of several dehydrogenases [9,10]. The NAD(P)HX repair system is ubiquitously expressed in all tissues and is highly conserved among prokaryotes, archaea, and eukaryotes [11].
Fig. 1.
The generation of NAD(P)HX and its repair by NAXD and NAXE. The formation of NAD(P)HX, a hydrated form of NAD(P)H, occurs either nonenzymatically from NAD(P)H or from side reactions of other enzymes (e.g., glyceraldehyde-3-phosphate dehydrogenase, GAPDH), and is accelerated by acidic or hyperthermic conditions. NAD(P)HX can inhibit cellular dehydrogenases (red arrows) and can further cyclize into a toxic metabolite that is thought to inhibit the mitochondrial respiratory chain [13]. The repair of NAD(P)HX is performed by its cofactor repair system, comprised of NAXD and NAXE (green arrows).
NAXE is located at 1q21.2-1q22. Sub-cellular localization studies showed two intracellular isoforms for NAXE targeted to the mitochondria and cytosol, respectively [1]. Biallelic loss of function variants in NAXE cause severe progressive infantile encephalopathy with brain edema, leukoencephalopathy, and skin lesions resembling pellagra (PEBEL1, MIM #617186). A total of 30 cases are described thus far [12-20], harboring 19 distinct variants. The course of the disease fluctuates from initially mild myopathy to encephalomyopathic crises, including motor and cognitive regression, quadriparesis, and respiratory and visual symptoms at time of febrile illness, which can be followed by a recovery, yet eventually a terminal febrile crisis of progressive encephalopathy, brain edema, and respiratory failure ensues in the majority of patients. Currently, there is no established therapy; however, encouraging results have been reported with niacin and nicotinamide treatment [14,16,20]. Nonetheless, significant gaps in our knowledge remain regarding the metabolic more than a single process associated with acute illness, and the optimal treatment for its reversal.
Herein, we summarize the clinical knowledge of this disorder based on 30 cases (including a case encountered in our clinic), discuss clinical and imaging findings, and review in vitro and in vivo biochemical information from patients including metabolomics analysis during crisis and following treatment, in an attempt to shed light on the pathomechanisms underlying this disorder and to provide a framework for research and therapeutic approaches.
2. Clinical features for NAXE deficiency
Clinical findings from review of 30 reported cases with NAXE deficiency (Table 1) describe a primarily pediatric population (ranging in age from newborn to 22 years) presenting with a progressive sequelae of symptoms that include ataxia (80%), cognitive regression (70%), ocular abnormalities (60%, including nystagmus, strabismus, anisocoria, and visual disturbances), movement disorders (50%, including tremor, myoclonus, and hyperkinesia), hypotonia (50%), dysarthria and/or dysphagia (50%), and motor regression (45%). In addition, often seen are pellagra-like cutaneous lesions (26%), epilepsy (22%), spasticity, and behavioral changes, including psychosis, confusion, and apathy [12-20] (Table 1). The overall prognosis is dismal, with death or ventilator dependence and bedridden status ensuing within 5 years from diagnosis in 90% of reported cases; however, two cases of late-onset disease and two cases of early onset disease (including one reported case herein) with prolonged survival and less morbidity have been described [14] (Table 1).
Table 1.
Summary of all reported NAXE variants and associated cases of PEBEL1. Molecularly confirmed probands and their siblings (confirmed or unconfirmed), grouped under the pathogenic NAXE variants, are shown. Reported clinical findings, treatments, and outcomes are shown. Bolded are patients that were alive at assessment 1 year or more after diagnosis.
| Case | Genetics of NAXE transcript: NM_144772.2, protein: NP_658985.2 |
SexA | Age | Outcome | Neurologic phenotype | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variant 1 | Δ product | Variant 2 | Δ product | Hypotonia | Loss of motor milestones |
Ataxia | Movement d/o |
Bulbar | Seizures | ||||
| 1 | c.281C>A | p.Ala94Asp | c.281C>A | p.Ala94Asp | ♀ | 6–12 | Death at 1-3y | + | + | ||||
| 2 | ♀ | m | + | + | |||||||||
| 3 | ♀ | + | + | ||||||||||
| 4 | ♂ | + | + | ||||||||||
| 5 | ♂ | Death at 10 yC | + | + | |||||||||
| 6 | c.177C>AD | p.Tyr59* | c.177C>AD | p.Tyr59* | ♂ | 20 m | Death at 21 m | + | + | Quadriparesis, torticollis | |||
| 7 | ♀ | 19 m | Death at 24 m | + | + | Quadriparesis | |||||||
| 8 | c.196C>TD | p.Gln66 | c.516 + 1G>AD | ♀ | 15 m | Death at 24 m | + | Tremor | |||||
| 9 | c.804_807 delinsAD | p.Lys270del | c.804_807 delinsAD | p.Lys270del | ♂ | 16 m | Death at 18 m | + | |||||
| 10 | c.804_807 delinsAD | p.Lys270del | c.368A>T | p.Asp123Val | 1 ♂ | 2 y | Alive at 3 y with residual ataxia E | + | + | + | Torticollis | ||
| 11 | c.653A>T | p.Asp218Val | c.743delC | p.Ala248Glufs*26 | ♂ | 16 m | Death at 29 m | + | + | Dysarthria | + | ||
| 12 | ? | 8 m | Death at 24 m | ||||||||||
| 13 | c.653A>T | p.Asp218Val | c.743_744delCA | p.Ala248Thrfs | ♂F | 8 m | Death at 24 m | + | + | ||||
| 14 | ♂F | ? | ? | + | + | ||||||||
| 15 | c.757G>AD | p.Gly253Ser | c.665-1G>AD | ♂ | 3 y | Death at 6 y | + | Recurrent falls | Dysarthria, dysphagia | ||||
| 16 | c.757G>AD | p.Gly253Ser | c.665-1G>AD,G | ♀ | 20 y | Death at 22 y | + | Myoclonus | |||||
| 17 | c.757G>AD | p.Gly253Ser | c.665-1G>AD | ♀ | 22 y | Alive at 29 y, nonambulatory E | Myoclonus, perioral dyskinesia, spasticity | Dysarthria | + | ||||
| 18 | c.733A>C | p.Lys245Gln | c.733A>C | p.Lys245Gln | ♀ | 20 m | Alive at 5.5 y with unsteady gateH1,I | + | + | + | Quadriparesis, tremor | Dysarthria | + |
| 19 | c.733A>C | p.Lys245Gln | c.733A>C | p.Lys245Gln | ♀F | 4 y | Death at 4 y | + | + | ||||
| 20 | c.652G>A | p.Asp218Asn | c.652G>A | p.Asp218Asn | ♂ | 3 d | Death at 4–6 m | + | + | ||||
| 21 | ? | ? | + | + | |||||||||
| 22 | ? | ? | + | + | |||||||||
| 23 | c.640A>G | p.Ile214Val | c.640A>G | p.Ile214Val | ♂ | 7y | Not reported | ||||||
| 24 | ♀ | ? | diseased, age not reported | ||||||||||
| 25 | c.386G>C | p.Arg129Pro | c.641 T>G | p.Ile214Ser | ♂ | 2 y | Death at 3 y | + | + | + | Quadriparesis, torticollis | Dysarthria | + |
| 26 | c.255A>T | p.Glu85Asp | c.361G>A | p.Gly121Arg | ♂ | 2 y | Death at 3 y | + | + | ||||
| 27 | c.255A>T | p.Glu85Asp | c.361G>A | p.Gly121Arg | ♂ | 2y | Alive at 4 y with mild myopathy H2 | Mild | + | ||||
| 28 | c.565G>A | p.Gly189Ser | c.565G>A | p.Gly189Ser | ♂ | 1 y | Death by 3 y | + | + | + | UE hyperkinesia | ||
| 29 | ♂ | 1 y | Death by 20 m | + | + | + | |||||||
| 30 | c.368A>T | p.Asp123Val | c.733A>C | p.Lys245Gln | ♀F | 2.5 y | Alive at 21 y, ventilatordependent and bedridden | + | |||||
Abbreviations: ND = not determined; m = month(s); y = year(s); Nl = normal; R = regression; T2HI = T2 hyperintensities; DR = diffusion restriction on diffusion weighted imaging; Abn = Abnormal; PR = pigmentary retinopathy; GDD = global developmental delays; HTN = hypertension; skin = Pellagra-like erythematous desquamating rash involving the neck face and torso also described as Lyell-like (toxic epidermal necrolysis) in some reports; WM = white matter; FTT = failure to thrive; UE = upper extremities; FSGS = focal segmental glomerulosclerosis; CK = creatine kinase.
Number includes family members presenting with a similar phenotype even in the absence of molecular confirmation.
Increased lactate in cerebrospinal fluid (CSF) or an abnormal lactate peak on MR spectroscopy.
Patient was bedridden and ventilator-dependent since diagnosis.
No protein expression in fibroblasts.
Treated with niacin (80–200 mg/day).
Information obtained from a retrospective whole exome sequencing study; phenotypic characterization is limited to the human phenotype ontology's (HPO) standardized annotation.
Suspected to be triggered by alcohol and tetrahydrocannabinol consumption.
Treated with nicotinamide, 30 mg/day; along with thiamine 60 mg/day, riboflavin 3 mg/day, pyridoxine 3 mg/day, and coenzyme Q10.
Treated with nicotinamide; and coenzyme Q10, thiamine and riboflavin (doses not reported).
Suffered two crises, recovered from the first one after 2 months of rehabilitation with residual ataxia and hypotonia.
The patient is a product of consanguineous union. Brain malformations in this case, not seen in others, may be related to other genetic variations.
Clinical symptoms prior to decompensation can be minimal including ataxia, hypotonia, and/or psychosis [13,14]. Triggers include febrile gastroenteritis and upper respiratory infection (URI) for the vast majority of cases, but alcohol and tetrahydrocannabinol consumption were suspected to trigger a crisis in one late-onset case [14]. In addition, only minimal or subtle findings on brain imaging have been reported early in the course of the crisis, such as cerebral edema or atrophy [13,16], and even normal findings in the two cases of late-onset disease [14]. During crisis, typical imaging findings have included T2 hyperintensities in the striatum, subcortical, periventricular and cerebellar areas, with diffusion restriction only occasionally reported [20]. The brainstem and spine may also be involved with signs of atrophy and myelopathy. One case reported involvement of the frontal area [12], one the temporal area [17], and one case with ventriculomegaly as the only brain MRI finding [14]. Hyperintense signals have been reported to resolve after recovery, with post-crisis findings showing atrophy [18,20]. One case showed leukodystrophy with significant brain malformations including lissencephaly and cortical dysplasia [14]; however, the patient was the product of a consanguineous union, increasing the likelihood of multi-locus pathogenic variation responsible for these divergent migration defects and white matter abnormalities [21,22] not seen in other cases. In a case encountered in our clinic, MRI of the brain performed on the day of admission was initially reported as normal, noticing the subtle caudate signal abnormalities only in retrospect after an additional MRI on day 3 (Supplementary Fig. 1). These findings are within the spectrum of expected MRI findings associated with NAXE deficiency when compared to other reported cases (Table 1). The paucity of symptoms and subtle imaging findings at the early stages of a crisis emphasize the need for additional diagnostic modalities to promptly diagnose and treat NAXE deficiency prior to irreversible tissue damage.
3. Current treatment modalities
Treatment with glucocorticoids is often given to patients with idiopathic, presumed inflammatory/autoimmune encephalopathy and was reportedly trialed in several NAXE cases, owing to the nonspecific presentation. The common regimen of “steroid pulse”, i.e. 30 mg/kg, were tried in several reports and did not lead to improvement of symptoms (including the case encountered in our clinic) [13,20]. One case reported the resolution of acute ataxia at the age of 16 months after a 3 week steroid use [13]. However, it is unclear whether the corticosteroid or the supportive care improved the patient's ataxia. Moreover; thirteen months later, the patient experienced a fatal crisis, presenting more typically with dysarthria, nystagmus, and hypotonia, and quickly developed brain edema.
Based on reports in all surviving cases, rehabilitative care was the only consistent treatment provided to the patient. We hypothesize that rehabilitation in a closely monitored environment allowed replenishment of the intracellular NAD+ pool by sustaining anabolism, leading to some degree of reversion of cognitive and motor regression. However, this treatment strategy cannot be considered preventative of further episodes of decompensation, as recurrence was demonstrated in few surviving cases [13,20].
Supplementation with niacin has been utilized in two cases of NAXE deficiency. A female with a late-onset presentation, surviving 7 years post-diagnosis and non-ambulatory, exhibited significant cognitive improvement after niacin supplementation [14]. Her dose was escalated from 40 to 80 mg/day. The second case (encountered in our clinic, for which metabolomics data are presented below) highlights a near-return to baseline in a 2-year-old presenting with ataxia, intermittent esotropia, motor regression, and encephalopathy in which 200 mg/day of niacin was used after diagnosis. Nicotinamide supplementation has been reported in two cases [16,20], with a reported dose of 30 mg/day in one of the cases [20]. The daily recommended dose for niacin equivalents (nicotinic acid or nicotinamide) is 6–16 mg/day, depending on age [23]. Niacin and its derivatives were provided with the expectation that NAXE deficiency would lead to NAD (P)HX accumulation and depletion of intracellular NAD(P)H pools; however, no biochemical evidence for the depletion nor the efficacy of repletion have been established.
None of the niacin-treated cases experienced crisis recurrence. Only one case of a NAXE deficient patient admitted for febrile-illness while on niacin-derivative treatment is known (patient encountered in our clinic, described herein). During the admission, the patient did not experience metabolic decompensation but was admitted for rehydration therapy (see Supplementary File for detailed presentation of this case). These findings may indicate that chronic supplementation can prevent acute depletion at time of inflammatory stress, but further data are needed to support this proposition. Furthermore, it is not currently known if there is a chronic, low-grade depletion of intracellular NAD+ pool, limiting our data for niacin supplementation as a prophylactic measure (see below). Nonetheless, as high dose niacin leads to its own conversion by CYP2E into nicotinamide-N-oxide, which is excreted in the urine [24], the clinical and metabolic results are encouraging.
4. Metabolic profiling of NAXE deficiency
None of the published cases reported abnormal laboratory findings capable of serving as disease-specific biomarkers. Acylcarnitine profile, plasma amino acids, and urine organic acid analyses were nondiagnostic in all instances. In 14 cases reporting CSF findings, lactate was elevated in 10, up to only 2 to 3-fold above the upper limit of normal [13,18], like the case presented here, and was normal in 4 cases. In virtually all cases, diagnosis was made by exome sequencing (ES), including three cases in a cohort of patients suspected to have a general mitochondrial disorder or Leigh syndrome [17,19]. Untargeted metabolomics profiling (Global MAPS, Baylor Genetics, Inc.) was conducted for the patient encountered in our clinic at time of acute crisis (Fig. 2., See Supplementary File for detailed case presentation and Methods). Results show absence of N1-methylnicotinamide (MNA) and adenosine-5′-diphosphoribose (ADPR), low levels of N1-methyl-2-pyridone-5-carboxamide (2PY, Z-score − 7.5), and mild reduction in nicotinamide (Z-score − 1.5). Additionally, elevations of branched-chain amino acid (BCAA) degradation metabolites were found, isovalerylglycine (Z-score + 2.6), 2-hydroxy-3-methylvalerate (Z-score + 2.4), isobutyrylglycine (Z-score + 2.2), along with the tricarboxylic acid (TCA) cycle fumarate (Z-score + 2.2). All these abnormalities normalized with niacin treatment, as shown with a second metabolomics analysis performed 6-months post crisis.
Fig. 2.
Plasma untargeted metabolomics during an acute crisis and 6 months post-niacin therapy. A. Alterations in analytes within the NAD+ catabolic pathways. In red, taken at time of crisis, there is absence of N1-methylnicotinamide (MNA) and adenosine-diphosphoribose (ADPR), and low N1-methyl-2-pyridone-5-carboxamide (2PY) (Z-score − 7.5). NAD+ is metabolized to nicotinamide (NAM) and ADPR by several intracellular enzymes; the former is further methylated by nicotinamide-N-methyltransferase (NNMT) into MNA, which is oxidized to 2PY (see Fig. 3 for pathway). Significantly low levels of these analytes strongly indicate low level of available NAD+. Levels of NAD+/NADH in biofluids are not accurately analyzed in clinical metabolomics of biofluids. NAM itself is only modestly reduced (Z-score − 1.5) during crisis (red), see discussion above. Relative analyte levels 6 months post-crisis (green) show repletion while on niacin supplementation. B-D: Heat map visualization of altered metabolite Z-scores (+1.5>Z-score < −1.5) (B) amino acids and their metabolites; (C) lysophospholipids, phosphatidylcholines, phosphatidylethanolamines, plasmalogens; and, (D) fatty acids and acylcarnitines are shown. Red shades represent higher Z-scores, blue – lower Z-scores. The Z-scores illustrate whether a given metabolite is significantly altered relative to standardized references (see Methods in the Supplementary File). In general, a Z-score greater than +1.75 or lower than −1.75 is considered significantly different from the control. B. Alterations in amino acids during crisis and relative normalization after 6 months of niacin therapy. Isovalerylglycine (Z-score + 2.6), 2-hydroxy-3-methylvalerate (Z-score + 2.4), isobutyrylglycine (Z-score + 2.2), and arginine (Z-score + 2.0), elevated at time of crisis, are associated with mitochondrial dysfunction and normalized post-treatment. In addition, elevation of asparagine (Z-score + 2) and reduction of hydroxyasparagine (Z score − 2.7) may indicate asparaginyl hydroxylase inhibition, which can be seen during hypoxic stress [51-53]. Elevated 2-pyrrolidinone (Z-score + 2.4) and guanidinobutanoate (Z-score + 2.4) can indicate inhibition of succinic semialdehyde dehydrogenase (SSADH), which utilizes NAD(P)+ as a substrate (the more pathognomonic metabolite, γ-hydroxybutyrate, is not detected in our untargeted assay). All these derivatives were normalized with long-term niacin supplementation. C. and D. Heat maps of lipid alterations show accumulation of several 18-carbon diacylglycerols and 16- and 18-carbon phosphatidylcholines, and depletion of lysophospholipids and several medium chain-fatty acids and acylcarnitines, with a reversion of these perturbations post-crisis. See text for further discussion.
The absence of both MNA and ADPR during crisis (normal plasma concentrations are ~100 nM and ~ 5–100 nM, respectively) [25-27] is striking, considering the nanomolar sensitivity of the metabolomics profile. Additionally, 2PY, while detected, was significantly reduced (Z-score − 7.5), making all three catabolites of NAD+ possible biomarkers for repair system deficiency. ADPR is the degradation product of NAD+ produced by cyclic ADP-ribose hydrolase 1 (CD38) and cyclic ADP-ribose hydrolase 2 (CD157), both expressed in adaptive immune cells [24]. ADPR is a potent activator of TRPM2, a calcium-permeable cation channel involved in autophagy, endothelial permeability, and inflammation [28]. Interestingly, MNA is the methylation product of nicotinamide, with S-adenosylmethionine (SAM) serving as the methyl donor; MNA is further oxidized into 2PY (Fig. 3). The patient's metabolomics profiling showed a milder reduction of nicotinamide (Z-score − 1.5) in comparison to its degradation products, which could be explained by a secondary SAM deficiency or nicotinamide N-methyltransferase (NNMT) dysfunction. Furthermore, supplementation with MNA was associated with antithrombotic and anti-inflammatory activity, reversal of inflammatory-mediated endothelial dysfunction [29,30], improved nitric oxide synthesis [31], and inhibition of free fatty acid-induced oxidative stress [32]. Elevations of BCAA and TCA cycle metabolites recorded during initial crisis, which improved with treatment, also serve as nonspecific markers for mitochondrial dysfunction [33] or hepatic disease [34].
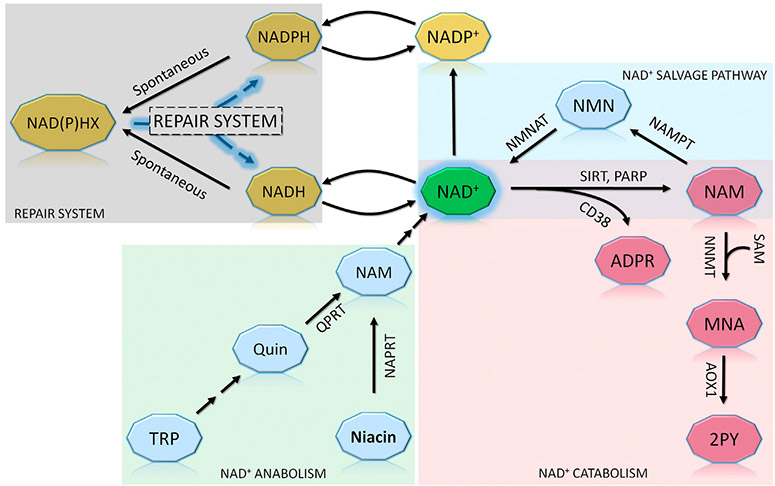
Fig. 3.
Selected pathways of NAD+ metabolism. NAD+ is generated and consumed by multiple cellular pathways. Left, in black shading, the repair system restores spontaneously generated NAD(P)HX back to NAD(P)H. NAD+ is utilized by several enzymes, including members of the sirtuin family (SIRT), poly-ADP-ribose polymerase (PARP), and cyclic ADP ribose hydrolase (CD38). The product of SIRT and PARP, nicotinamide (NAM), can be recycled back to NAD+ by the salvage pathway (green shading, top right) or is further converted into MNA by NNMT and the co-factor SAM. MNA is then oxidized by aldehyde oxidase (AOX1) into 2PY. This degradation is highlighted by the red shading, lower right. The de novo (kynurenine) NAD+ biosynthesis pathway is indicated in the bottom left. Abbreviations: NMN = nicotinamide mononucleotide; NAMPT = nicotinamide phosphoribosyltransferase; NMNAT = nicotinamide mononucleotide adenylyl transferases. TRP = tryptophan; Quin = quinolinic acid; NAM = nicotinamide. ADPR = adenosine-5′-diphosphoribose; MNA = N1-methylnicotinamide; SAM = S-adenosylmethionine; AOX1 = aldehyde oxidase; 2PY = N1-methyl-2-pyridone-5-carboxamide.
Overall, untargeted metabolomics data show that the depletion of NAD+ catabolic products was reverted with niacin supplementation, supporting a model of NAD+ depletion, contributing to the metabolic derangement. Along with the elevations of NAD+ derivatives in post-crisis metabolomics, these results are encouraging in providing indirect evidence that supplementation can still achieve an excess NAD+ storage to avert decompensation during a febrile crisis in repair system deficiency (Fig. 2A-C).
| Case | Ophthalmologic phenotype | Cognitive phenotype | MRI | Lactate in CSF or MRSB |
Skin | Other | PMID (ref) |
||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Nystagmus | Ptosis | Alignment | Regression | Psych | Cerebrum | Cerebellum and spine |
|||||
| 1 | + | Periventricular & frontal areas T2HI | Dentate T2HI | Normal | No | 27122014 (12) |
|||||
| 2 | + | ||||||||||
| 3 | + | ||||||||||
| 4 | + | ||||||||||
| 5 | + | Yes | |||||||||
| 6 | + | Edema, WM T2HI, myelopathy | ↑ | Yes | 27616477 (13) |
||||||
| 7 | + | WM T2HI, myelopathy, brain atrophy | ↑ | Yes | |||||||
| 8 | + | + | Cortical and subcortical WM, striatum, & brain stem T2HI | Edema, myelopa | ↑ | Yes | FTT | 27616477 (13) |
|||
| 9 | + | + | Vertical gaze palsy | + | Edema (CT) | ↑ | No | 27616477 (13) |
|||
| 10 | + | Esotropia | Caudate T2HI | ↑ (mild) | No | HTN | Current | ||||
| 11 | + | + | Edema, progressive atrophy | Diffuse T2HI | ↑ | Yes | 27616477 (13) |
||||
| 12 | Brain atrophy (CT) | ↑ | No | ||||||||
| 13 | Brain edema | ND | 27290639 (19) |
||||||||
| 14 | Brain edema | ND | |||||||||
| 15 | Diplopia | + | Ventriculomegaly | ND | No | 31745726(14) | |||||
| 16 | Hallucinations, anxiety, catatoniaG | Normal | ND | No | Nausea, headache, vertigo | 31745726 (14) |
|||||
| 17 | Strabismus, visual disturbances | + | Confusion | Normal | Normal | No | Headache | 31745726 (14) |
|||
| 18 | Esotropia, anisocoria | + | T2HI + DR in middle cerebellar peduncles; | Normal | No | 34678889 (20) |
|||||
| 19 | ND | 31745726 (14) |
|||||||||
| 20 | Strabismus | Abn signal brain stem & peri-ventricular WM | Cerebellar atrophy | ND | Thrombocytosis | 31745726 (14) |
|||||
| 21 | Strabismus | ND | |||||||||
| 22 | Strabismus | ND | |||||||||
| 23 | GDD | Leukodystrophy, lissencephaly, cortical dysplasia, pachygyria, ventriculomegalyJ | ↑ | Pigmented retinopathy, elevated CK | 31745726 (14) |
||||||
| 24 | GDD | ND | |||||||||
| 25 | + | Normal | Yes | 31758406 (15) |
|||||||
| 26 | ND | 30022751 (16) |
|||||||||
| 27 | Strabismus | Brain atrophy | ND | 30022751 (16) |
|||||||
| 28 | Abn eye movement | + | Temporal T2HI, basal ganglia DR | ↑ | No | 34120322 (18) |
|||||
| 29 | + | + | Brain edema, leukoencephalopathy | ↑ | |||||||
| 30 | Medial temporal T2HI | Diffuse T2HI, cerebellar atrophy | ND | FSGS, scoliosis | 32020600 (17) |
||||||
On the contrary, nicotinamide, participating in the NAD+ salvage pathway, and quinolinic acid, a derivative of the de novo NAD+ synthesis pathway, are both reduced to a lesser degree (Z-scores of −1.5 and −0.73, respectively), which may point to an ongoing diversion of metabolic pathways towards NAD+ synthesis, yet with a net loss of NAD+ flux due to repair system dysfunction (Fig. 3). Ideally, analysis of cells or tissues can provide a more comprehensive analysis of the NAD+ metabolome in the intracellular environment; however, direct measurement of NAD+ and a subset of associated metabolites in biofluids is not reliable [35]. Furthermore, some components of the NAD+ metabolome (including NAD (P)+, NAD(P)H, NAD(P)HX) are unstable, heat labile, and/or rapidly metabolized, requiring special handling immediately after sample collection (e.g. immediate centrifugation and rapid-freezing). Thus, samples collected in either hospitals or in outpatient clinics may have wide variability in the detection of the more sensitive molecules, limiting analysis.
In addition to its anti-inflammatory role discussed above, MNA was also shown to function as a myokine that enhances the utilization of energy [36], which may explain, in part, the common findings of ataxia, hypotonia, dysphagia, and dysarthria at presentation. Remarkably, niacin supplementation alone was linked to reduced spasticity in one case of NAXE deficiency after crisis rehabilitation [14]. The timeframe of NAD+ derangement with respect to development of symptoms is not known; however, the milder symptoms developed in the patient encountered in our clinic during the second febrile illness provide reassurance of chronic supplementation and adequate NAD+ storage to mitigate a crisis. Nonetheless, the role of acute high-dose niacin during a crisis is not known. Abundance of NAD+ could translate into increased NAD(P)HX levels during crisis; yet, no clinical data support worsening outcomes under supplementation.
Elevations in isovalerylglycine and 2-hydroxy-3-methylvalerate (both degradation products of the BCAA isoleucine), along with elevation of fumarate, a TCA intermediate, and mild elevations in orotate (Z-score + 2.2) and arginine (Z-score + 2), both ‘urea cycle dysfunction’ metabolites, can co-occur secondary to mitochondrial dysfunction. These alterations, although nonspecific, are commonly found in inborn errors of metabolism involving dysfunction of mitochondrial enzymes [37,38]. We hypothesize that the mitochondrial isoforms of NAXE and NAXD play a pivotal role in NAD(P)HX repair, while the cytosolic component may be secondary in its importance. Cases of repair system deficiency predicted to affect only the mitochondrial isoform of NAXD and resulting in fatal encephalopathic crises has been previously reported (cases 3 and 5 from Van Bergen et al [39] and the case reported by Borna et al [40]).
5. Genetic characteristics of NAXE
NAXE has 6 exons encoding a single polypeptide chain which homodimerizes post-translation. It has two alternative start sites in exon 1, targeting NAXE to either the mitochondria or cytosol, and a YjeF N-terminal domain spanning exons 2–6 [1,18]. Most reported pathogenic variants (86%) fall within the YjeF N-terminal domain of NAXE. Two reported variants occur within the first exon downstream to the mitochondrial pre-sequence tag and were observed in the homozygote state (one missense and one nonsense). Missense variants are most commonly observed (63%); however, nonsense (10%), splice site (11%), and deletion-insertions (delins) (16%) are reported (Table 1).
Evidence for pathogenicity is available for six variants, showing loss of protein expression in vitro (NM_144772.2 and NP_658985.2): two each in homozygous cases c.804_807delinsA (p.Lys270del) and c.177C>A (p.Tyr59*), and four variants in two compound heterozygous cases [13,14]: c.196C>T (p.Gln66*) with c.516 + 1G>A and c.757G>A (p.Gly253Ser) with c.665-1G>A. The second splicing variant (c.665-1G>A) demonstrated aberrant splicing and reduced total NAXE reference mRNA (Table 1) [14].
NAXD, the NAD(P)HX dehydratase partner of NAXE in the intracellular repair system, localizes to the mitochondrion, cytosol, and endoplasmic reticulum (ER) [1]. The lack of an ER-bound NAXE isoform is surprising; however, the contribution of compartment-specific repair system components to NAD+ homeostasis is currently not known.
6. Model organisms for NAXE deficiency
The repair system constituted by NAXE and NAXD is highly conserved from E. coli, (yjeF) coding for a bifunctional repair enzyme), yeast (YNL200C and YKL151C), and Arabidopsis thaliana (At5g49970 and At5g19150), to mouse (Naxe and Naxd) and human [10,11]. Knockouts of either enzyme did not result in an abnormal phenotype or life cycle/span in Arabidopsis, yeast or even mice (fed with standard chow) [10,41,42]; however, NAD(P)HX accumulated in yeast and Arabidopsis with NAXD (YKL151C or At5g19150) knockout [8,10,41], as seen in NAXD and NAXE deficient human fibroblasts [13,39]. Notwithstanding, NAD+ depletion was documented in yeast [41], a finding which was not recapitulated in vitro in heat-stressed human cells [13]. In vivo biochemical data presented here does support systemic NAD+ depletion. The difference can perhaps be explained by the differences in the imposed stress on the cells: cultured cells were heated to 40° C for 24 h, while patient cells were exposed to systemic fever and other inflammation-related stressors. Previous studies also reported that yeast serine synthesis is inhibited with the accumulation of R- and S-NADHX (secondary to YKL151C [dehydratase] deficiency), leading to decreased levels of serine [41], an interesting finding not reproduced in human cells or in our metabolomics studies. The role of NAD(P)HX in metabolic derangement has come under question, given the limited buildup of NAD(P)HX in fibroblasts with NAXE deficiency [43]. Alternatively, NAD(P)HX buildup may diminish the NAD+/NADH pool, as seen by our metabolomics studies, and could limit substrate availability for the NAD+-consuming enzymes, SIRT1/3/6, which are key immunoregulators [24], among other NAD+-dependent metabolic pathways.
Interestingly, Arabidopsis At5g49970 (NAXE) has a C-terminus with pyridoxine/pyridoxamine phosphate oxidase activity, functioning in the pyridoxine salvage pathway [10]. This enzyme clusters with pyridoxine processing enzymes in E. coli and is co-expressed with such enzymes in yeast and Arabidopsis [43]. Furthermore, inactivating missense variants in Arabidopsis NAXE resulted in decreased levels of pyridoxal-5′-phosphate. No parallel co-expression is known in humans, and metabolomics data did not show decreased levels of pyridoxal during acute crisis, yet a consideration for B6 supplementation should be given, which was the rationale behind providing pyridoxine to the patient encountered in our clinic.
7. Lipid alterations and NAXE moonlighting
NAXE also encodes a secreted isoform which binds the APOA1-HDL complex, destabilizes lipid rafts of endothelial cells, and causes cholesterol efflux from macrophages to the surrounding endothelium. Overexpression of NAXE leads to reduced atherosclerosis and plasma lipids in high-cholesterol, high-fat diet-fed mice with defective low-density lipoprotein (Ldl) receptor [44]. A possible mechanism comes from the observation that NAXE knockout reduced autophagy in macrophages exposed to oxidized LDL particles, possibly by abrogated ubiquitination of mitofusins 1 and 2, normally carried out with the assistance of the mitochondrial isoform of NAXE [45].
Lipid profile alterations can be seen in the untargeted metabolomics data during crisis, including several phospholipids, diacylglycerols, saturated and unsaturated fatty acids, and plasmalogens, owing to the bifunctional nature of the NAXE protein and including requirements for NAD+ and NADPH in fatty acid utilization in the cytosol and in lipid beta-oxidation and synthesis in the peroxisomes, (Fig. 2C-D). Of prominence, the glycerophosphocholines (GPC), 1-stearoyl-2-oleoyl-GPC (18:0/18:1) and 1-linoleoyl-2-linolenoyl-GPC (18:2/18:3), and the glycerophosphoethanolamine (GPE), 1,2-dioleoyl-GPE (18:1/18:1), were enriched with a Z-score of +1.7 — +1.8 during the crisis. Similarly, several diacylglycerols (linoleoyl-arachidonoyl-glycerol (18:2/20:4), and oleoyl-arachidonoyl-glycerol (18:1/20:4)) were increased, as well, with Z-scores of +2.3–2.6. In contrast, the lysophospholipid 2-stearoyl-GPC (18:0) and unsaturated long chain fatty acids (docosadienoate, arachidonate, myristoleate, 10-heptadecenoate, palmitoleate) were reduced to Z-scores of −2 (Fig. 2D), supporting either an impairment of fatty acid transport and/or synthesis. Lastly, the plasmalogen 1-palmitoyl-2-linoleoyl-GPC (O-16:0/18:2) was mildly reduced to Z-score of −1.5. Notwithstanding the alterations that were normalized with treatment, several alterations remained persistent despite niacin supplementation. Examples include several fatty acid-cholines, such as arachidonoylcholine (Z-score of +2.8 during crisis and + 2.6 post-crisis), palmitoylcholine (Z-score + 1.9 and + 1.8), and linoleoylcholine (Z score of +1.8 and + 1.8); a certain ceramide species (d18:1/20:0, d16:1/22:0, d20:1/18:0) (Z-score of −3.1 at time of crisis and − 2.0 post-crisis); and the lysophospholipid 1-adrenoyl-GPE (Z score of −2.4 at crisis and − 2.9 post-crisis). Moreover, the plasmalogens 2-docosahexaenoyl-GPE/GPC (P-16:0 (or 18:0)/22:6) were increased to Z score of +2.5 to +3.2 post-crisis while exhibiting Z-score ≤ +1.5 during the crisis.
The lipid profile is determined genetically and is heavily dependent on nutritional status, diet, and medications [46-48]. Interestingly, lipidomic alterations are an apparent sensitive marker in neuroinflammatory conditions [49] and critical illness in general [50]. However, we further suspect that alterations in the lipidome reflect, in part, the bifunctionality of NAXE—first, as an apolipoprotein binding protein, inherently unresponsive to niacin supplementation and second, as an epimerase, responsive to niacin treatment.
Plasma metabolomics of Naxe−/− mice revealed a similar effect of Naxe deficiency on lipid metabolism: decreased cholesterol, increased bile acid precursors, decreased levels of phosphatidylcholines, phosphatidylinositol, plasmalogens, and sphingomyelins, and an increase in mono- and di-acylglycerols [42]. However, while male mice showed reduced nicotinamide, nicotinamide-N-oxide, MNA, and spermidine, consistent with the findings in humans, these changes were not observed in female mice. It should be noted, however, that the mice were stressed only by 6 h of fasting. While significant alterations in sphingomyelins were not observed in the human samples, other changes showed fluctuations similar to the mice lipidomic profile (elevations in diacylglycerols and reductions in plasmalogens), while other alterations were in the opposite direction (reduced levels of unsaturated long chain fatty acids, GPC, and GPE). These alterations are difficult to ascribe to specific perturbations in metabolic pathways, and additional data points are needed to create a more comprehensive repair system deficiency lipidomic profile, but attention should be drawn towards such alterations that probably involve changes in lipid and cholesterol metabolism and do not merely reflect dietary variations. More research is required to perhaps elucidate a connection between these alterations and neuroinflammatory biomarkers.
8. Conclusions
NAXE is an essential component of the NAD(P)HX repair system in humans, as reflected by the severe neurometabolic and neuroinflammatory condition, PEBEL1, associated with its deficiency. While the co-occurrence of pellagra-like skin lesions, encephalopathy, and brain edema triggered by febrile illness may strongly suggest PEBEL1, further case identification has demonstrated a broader phenotypic spectrum, including cases with nonspecific clinical and neuroimaging findings [14]. Biochemical data ascertained by the treatabolome and presented here shed light on the derangement of NAD+ homeostasis during crises and may indicate a depletion of the NAD+ pool, which is further supported by the identification of biomarkers (ADPR, MNA, 2PY) for rapid case identification. Metabolomic profiling of NAXE deficiency showed a complex picture of the breadth of abnormalities, derived from reduced cellular NAD+/NADH/NAD(P)H but also associated with NAD(P)HX epimerization and binding of apolipoproteins. Supplementation with niacin appears to replete NAD+ stores, suggesting niacin as a preventative measure to alleviate crises. However, longer periods of follow-up are needed to provide more information on the natural history of the disorder in undulating cases and treated patients experiencing febrile illnesses.
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ymgme.2022.04.003.
Supplementary Material
Acknowledgements
Sincere appreciation is extended to the patient and his family for their willingness to participate in this clinical study. All procedures, laboratory tests, and imaging were obtained as part of medical management of the patient.
Funding
D.G.C. was supported by the National Institute of Neurological Disorders and Stroke - Brain Disorders and Development Training Grant (T32 NS043124–19) and an MDA Development Grant (873841). S.H.E. acknowledges support from the National Cancer Institute (R01CA211176).
Abbreviations:
- 2PY
N1-methyl-2-pyridone-5-carboxamide
- ADPR
adenosine-5′-diphosphoribose
- AIBP
apolipoprotein A-I binding protein (NAXE)
- AOX1
aldehyde oxidase
- APOA1
apolipoprotein A-I
- CBC
complete blood count
- CD38
cyclic ADP ribose hydrolase
- CMA
chromosomal microarray analysis
- CRP
C-reactive protein
- CSF
cerebrospinal fluids
- ER
endoplasmic reticulum
- ES
exome sequencing
- ESR
erythrocyte sedimentation rate
- GAPDH
glyceraldehyde-3-phosphate dehydrogenase
- GPC
glycerophosphocholine
- GPE
glycerophosphoethanolamine
- HDL
high density lipoprotein particles
- LDL
low-density lipoprotein
- MNA
N1-methylnicotinamide
- NAD
nicotinamide adenine dinucleotide
- NADH
reduced nicotinamide adenine dinucleotide
- NADHX
hydrated reduced nicotinamide adenine dinucleotide
- NADPHX
hydrated reduced nicotinamide adenine dinucleotide phosphate
- NADP
nicotinamide adenine dinucleotide phosphate
- NAMPT
nicotinamide phosphoribosyltransferase
- NAXD
NAD(P)HX dehydratase
- NAXE
NAD(P)HX epimerase
- NAM
Nicotinamide
- NMDA
N-methyl-d-aspartate
- NMNAT
nicotinamide mononucleotide adenylyl transferases
- NNMT
nicotinamide-N-methyltransferase
- MRI
magnetic resonance imaging
- PARP
poly-ADP-ribose polymerase
- PEBEL1
encephalopathy, progressive, early-onset, with brain edema and/or leukoencephalopathy, 1
- SAM
S-adenosylmethionine
- SIRT
sirtuins
- T4
thyroxine hormone
- TSH
thyroid stimulating hormone
- URI
upper respiratory infection
Footnotes
Competing interests
The Department of Molecular and Human Genetics at Baylor College of Medicine receives revenue from clinical genetic testing conducted at Baylor Genetics (BG) Laboratories. All authors declare no conflict of interest related to this study.
References
- [1].Marbaix AY, Tyteca D, Niehaus TD, Hanson AD, Linster CL, Van Schaftingen E, Occurrence and subcellular distribution of the NADPHX repair system in mammals, Biochem. J 460 (2014) 49–58, 10.1042/bj20131482. [DOI] [PubMed] [Google Scholar]
- [2].Ritter M, Buechler C, Boettcher A, Barlage S, Schmitz-Madry A, Orsó E, et al. , Cloning and characterization of a novel apolipoprotein A-I binding protein, AI-BP, secreted by cells of the kidney proximal tubules in response to HDL or ApoA-I, Genomics 79 (2002) 693–702, 10.1006/geno.2002.6761. [DOI] [PubMed] [Google Scholar]
- [3].Choi SH, Wallace AM, Schneider DA, Burg E, Kim J, Alekseeva E, et al. , AIBP augments cholesterol efflux from alveolar macrophages to surfactant and reduces acute lung inflammation, JCI Insight 3 (2018), 10.1172/jci.insight.120519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Fang L, Choi SH, Baek JS, Liu C, Almazan F, Ulrich F, et al. , Control of angiogenesis by AIBP-mediated cholesterol efflux, Nature 498 (2013) 118–122, 10.1038/nature12166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Dubrovsky L, Ward A, Choi SH, Pushkarsky T, Brichacek B, Vanpouille C, et al. , Inhibition of HIV replication by apolipoprotein A-I binding protein targeting the lipid rafts, mBio 11 (2020), 10.1128/mBio.02956-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Low H, Mukhamedova N, Capettini L, Xia Y, Carmichael I, Cody SH, et al. , Cholesterol efflux-independent modification of lipid rafts by AIBP (apolipoprotein A-I binding protein), Arterioscler. Thromb. Vasc. Biol 40 (2020) 2346–2359, 10.1161/atvbaha.120.315037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Gu Q, Yang X, Lv J, Zhang J, Xia B, Kim JD, et al. , AIBP-mediated cholesterol efflux instructs hematopoietic stem and progenitor cell fate, Science (New York, N.Y.) 363 (2019) 1085–1088, 10.1126/science.aav1749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Colinas M, Shaw HV, Loubéry S, Kaufmann M, Moulin M, Fitzpatrick TB, A pathway for repair of NAD(P)H in plants, J. Biol. Chem 289 (2014) 14692–14706, 10.1074/jbc.M114.556092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Yoshida A, Dave V, Inhibition of NADP-dependent dehydrogenases by modified products of NADPH, Arch. Biochem. Biophys 169 (1975) 298–303, 10.1016/0003-9861(75)90344-6. [DOI] [PubMed] [Google Scholar]
- [10].Niehaus TD, Richardson LG, Gidda SK, ElBadawi-Sidhu M, Meissen JK, Mullen RT, et al. , Plants utilize a highly conserved system for repair of NADH and NADPH hydrates, Plant Physiol. 165 (2014) 52–61, 10.1104/pp.114.236539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Marbaix AY, Noël G, Detroux AM, Vertommen D, Van Schaftingen E, Linster CL, Extremely conserved ATP- or ADP-dependent enzymatic system for nicotinamide nucleotide repair, J. Biol. Chem 286 (2011) 41246–41252, 10.1074/jbc.C111.310847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Spiegel R, Shaag A, Shalev S, Elpeleg O, Homozygous mutation in the APOA1BP is associated with a lethal infantile leukoencephalopathy, Neurogenetics 17 (2016) 187–190, 10.1007/s10048-016-0483-3. [DOI] [PubMed] [Google Scholar]
- [13].Kremer LS, Danhauser K, Herebian D, Petkovic Ramadža D, Piekutowska-Abramczuk D, Seibt A, et al. , NAXE mutations disrupt the cellular NAD(P)HX repair system and cause a lethal Neurometabolic disorder of early childhood, Am. J. Hum. Genet 99 (2016) 894–902, 10.1016/j.ajhg.2016.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Trinh J, Imhoff S, Dulovic-Mahlow M, Kandaswamy KK, Tadic V, Schafer J, et al. , Novel NAXE variants as a cause for neurometabolic disorder: implications for treatment, J. Neurol 267 (2020) 770–782, 10.1007/s00415-019-09640-2. [DOI] [PubMed] [Google Scholar]
- [15].Incecik F, Ceylaner S, Early-onset progressive encephalopathy associated with NAXE gene variants: a case report of a Turkish child, Acta Neurol. Belg 120 (2020) 733–735, 10.1007/s13760-019-01242-z. [DOI] [PubMed] [Google Scholar]
- [16].Yu D, Zhao FM, Cai XT, Zhou H, Cheng Y, Clinical and genetic features of early-onset progressive encephalopathy associated with NAXE gene mutations, Zhongguo dang dai er ke za zhi 20 (2018), 10.7499/j.issn.1008-8830.2018.07.002 524–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Lee JS, Yoo T, Lee M, Lee Y, Jeon E, Kim SY, et al. , Genetic heterogeneity in Leigh syndrome: highlighting treatable and novel genetic causes, Clin. Genet 97 (2020) 586–594, 10.1111/cge.13713. [DOI] [PubMed] [Google Scholar]
- [18].Mohammadi P, Heidari M, Ashrafi MR, Mahdieh N, Garshasbi M, A novel homozygous missense variant in the NAXE gene in an Iranian family with progressive encephalopathy with brain edema and leukoencephalopathy, Acta Neurol. Belg (2021), 10.1007/s13760-021-01717-y. [DOI] [PubMed] [Google Scholar]
- [19].Pronicka E, Piekutowska-Abramczuk D, Ciara E, Trubicka J, Rokicki D, Karkuciñska-Więckowska A, et al. , New perspective in diagnostics of mitochondrial disorders: two years’ experience with whole-exome sequencing at a national paediatric Centre, J. Transl. Med 14 (2016) 174, 10.1186/s12967-016-0930-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Chiu LW, Lin SS, Chen CH, Lin CH, Lee NC, Hong SY, et al. , NAXE gene mutation-related progressive encephalopathy: a case report and literature review, Medicine (Baltimore) 100 (2021), e27548. 10.1097/md.0000000000027548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Mitani T, Isikay S, Gezdirici A, Gulec EY, Punetha J, Fatih JM, et al. , High prevalence of multilocus pathogenic variation in neurodevelopmental disorders in the Turkish population, Am. J. Hum. Genet 108 (2021) 1981–2005, 10.1016/j.ajhg.2021.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Herman I, Jolly A, Du H, Dawood M, Abdel-Salam GMH, Marafi D, et al. , Quantitative dissection of multilocus pathogenic variation in an Egyptian infant with severe neurodevelopmental disorder resulting from multiple molecular diagnoses, Am. J. Med. Genet. A (2021), 10.1002/ajmg.a.62565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Institute of Medicine Standing Committee on the Scientific Evaluation of Dietary Reference, I., its Panel on Folate, O. B. V. & Choline, Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B(6), Folate, Vitamin B(12), Pantothenic Acid, Biotin, and Choline, National Academies Press, US, 1998 1998. [PubMed]
- [24].Xie N, Zhang L, Gao W, Huang C, Huber PE, Zhou X, et al. , NAD(+) metabolism: pathophysiologic mechanisms and therapeutic potential, Signal Transduct. Target. Ther 5 (2020) 227, 10.1038/s41392-020-00311-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Somogyi A, Siebert D, Bochner F, Determination of endogenous concentrations of N1-methylnicotinamide in human plasma and urine by high-performance liquid chromatography, Anal. Biochem 187 (1990) 160–165, 10.1016/0003-2697(90)90435-c. [DOI] [PubMed] [Google Scholar]
- [26].Grant R, Berg J, Mestayer R, Braidy N, Bennett J, Broom S, et al. , A pilot study investigating changes in the human plasma and urine NAD+ metabolome during a 6 hour intravenous infusion of NAD, Front. Aging Neurosci 11 (2019) 257, 10.3389/fnagi.2019.00257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Clement J, Wong M, Poljak A, Sachdev P, Braidy N, The plasma NAD(+) metabolome is dysregulated in “normal” aging, Rejuvenation Res. 22 (2019) 121–130, 10.1089/rej.2018.2077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Zielińska W, Zabrzyński J, Gagat M, Grzanka A, The role of TRPM2 in endothelial function and dysfunction, Int. J. Mol. Sci 22 (2021), 10.3390/ijms22147635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Kolodziejczyk AM, Brzezinka GD, Khurana K, Targosz-Korecka M, Szymonski M, Nanomechanical sensing of the endothelial cell response to anti-inflammatory action of 1-methylnicotinamide chloride, Int. J. Nanomedicine 8 (2013) 2757–2767, 10.2147/ijn.s46936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Bartuś M, Łomnicka M, Kostogrys RB, Kaźmierczak P, Watała C, Słominska EM, et al. , 1-Methylnicotinamide (MNA) prevents endothelial dysfunction in hypertriglyceridemic and diabetic rats, Pharmacol. Rep 60 (2008) 127–138. [PubMed] [Google Scholar]
- [31].Domagala TB, Szeffler A, Dobrucki LW, Dropinski J, Polanski S, Leszczynska-Wiloch M, et al. , Nitric oxide production and endothelium-dependent vasorelaxation ameliorated by N1-methylnicotinamide in human blood vessels, Hypertension (Dallas, Tex. : 1979) 59 (2012) 825–832, 10.1161/hypertensionaha.111.183210. [DOI] [PubMed] [Google Scholar]
- [32].Tanaka Y, Kume S, Araki H, Nakazawa J, Chin-Kanasaki M, Araki S, et al. , 1-Methylnicotinamide ameliorates lipotoxicity-induced oxidative stress and cell death in kidney proximal tubular cells, Free Radic. Biol. Med 89 (2015) 831–841, 10.1016/j.freeradbiomed.2015.10.414. [DOI] [PubMed] [Google Scholar]
- [33].Clarke C, Xiao R, Place E, Zhang Z, Sondheimer N, Bennett M, et al. , Mitochondrial respiratory chain disease discrimination by retrospective cohort analysis of blood metabolites, Mol. Genet. Metab 110 (2013) 145–152, 10.1016/j.ymgme.2013.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Burrage LC, Nagamani SC, Campeau PM, Lee BH, Branched-chain amino acid metabolism: from rare Mendelian diseases to more common disorders, Hum. Mol. Genet 23 (2014) R1–R8, 10.1093/hmg/ddu123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Braidy N, Villalva MD, Grant R, NADomics: measuring NAD(+) and related metabolites using liquid chromatography mass spectrometry, Life (Basel, Switzerland) 11 (2021), 10.3390/life11060512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Ström K, Morales-Alamo D, Ottosson F, Edlund A, Hjort L, Jörgensen SW, et al. , N(1)-methylnicotinamide is a signalling molecule produced in skeletal muscle coordinating energy metabolism, Sci. Rep 8 (2018) 3016, 10.1038/s41598-018-21099-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].McInnes J, Mitochondrial-associated metabolic disorders: foundations, pathologies and recent progress, Nutr. Metab 10 (2013) 63, 10.1186/1743-7075-10-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Lee B, Scaglia F, Inborn Errors of Metabolism: From Neonatal Screening to Metabolic Pathways, Oxford Univ. Press, 2015. [Google Scholar]
- [39].Van Bergen NJ, Guo Y, Rankin J, Paczia N, Becker-Kettern J, Kremer LS, et al. , NAD (P)HX dehydratase (NAXD) deficiency: a novel neurodegenerative disorder exacerbated by febrile illnesses, Brain 142 (2019) 50–58, 10.1093/brain/awy310. [DOI] [PubMed] [Google Scholar]
- [40].Borna NN, Kishita Y, Abe J, Furukawa T, Ogawa-Tominaga M, Fushimi T, et al. , NAD(P)HX dehydratase protein-truncating mutations are associated with neurodevelopmental disorder exacerbated by acute illness, Brain 143 (2020), e54. 10.1093/brain/awaa130. [DOI] [PubMed] [Google Scholar]
- [41].Becker-Kettern J, Paczia N, Conrotte JF, Zhu C, Fiehn O, Jung PP, et al. , NAD(P)HX repair deficiency causes central metabolic perturbations in yeast and human cells, FEBS J. 285 (2018) 3376–3401, 10.1111/febs.14631. [DOI] [PubMed] [Google Scholar]
- [42].Kim JD, Zhu L, Sun Q, Fang L, Systemic metabolite profiling reveals sexual dimorphism of AIBP control of metabolism in mice, PLoS One 16 (2021), e0248964. 10.1371/journal.pone.0248964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Niehaus TD, Elbadawi-Sidhu M, Huang L, Prunetti L, Gregory JF 3rd, de Crécy-Lagard V, et al. , Evidence that the metabolite repair enzyme NAD(P)HX epimerase has a moonlighting function, Biosci. Rep 38 (2018), 10.1042/bsr20180223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Schneider DA, Choi SH, Agatisa-Boyle C, Zhu L, Kim J, Pattison J, et al. , AIBP protects against metabolic abnormalities and atherosclerosis, J. Lipid Res 59 (2018) 854–863, 10.1194/jlr.M083618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Choi SH, Agatisa-Boyle C, Gonen A, Kim A, Kim J, Alekseeva E, et al. , Intracellular AIBP (apolipoprotein A-I binding protein) regulates oxidized LDL (Low-density lipoprotein)-induced mitophagy in macrophages, Arterioscler. Thromb. Vasc. Biol 41 (2021) e82–e96, 10.1161/atvbaha.120.315485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Tabassum R, Rämö JT, Ripatti P, Koskela JT, Kurki M, Karjalainen J, et al. , Genetic architecture of human plasma lipidome and its link to cardiovascular disease, Nat. Commun 10 (2019) 4329, 10.1038/s41467-019-11954-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Kien CL, Bunn JY, Poynter ME, Stevens R, Bain J, Ikayeva O, et al. , A lipidomics analysis of the relationship between dietary fatty acid composition and insulin sensitivity in young adults, Diabetes 62 (2013) 1054–1063, 10.2337/db12-0363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Pati S, Krishna S, Lee JH, Ross MK, de La Serre CB, Harn DA Jr., et al. , Effects of high-fat diet and age on the blood lipidome and circulating endocannabinoids of female C57BL/6 mice, Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1863 (2018) 26–39, 10.1016/j.bbalip.2017.09.011. [DOI] [PubMed] [Google Scholar]
- [49].Puris E, Kouřil Š, Najdekr L, Loppi S, Korhonen P, Kanninen KM, et al. , Metabolomic and lipidomic changes triggered by lipopolysaccharide-induced systemic inflammation in transgenic APdE9 mice, Sci. Rep 11 (2021) 13076, 10.1038/s41598-021-92602-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Wu J, Cyr A, Gruen D, Lovelace T, Benos P, Chen T, et al. , Lipidomic signatures align with inflammatory patterns and outcomes in critical illness, Res. Square (2021), 10.21203/rs.3.rs-106579/v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Volkova YL, Pickel C, Jucht AE, Wenger RH, Scholz CC, The asparagine hydroxylase FIH - a unique oxygen sensor, Antioxid. Redox Signal (2022), 10.1089/ars.2022.0003. [DOI] [PubMed] [Google Scholar]
- [52].Lando D, Peet DJ, Gorman JJ, Whelan DA, Whitelaw ML, Bruick RK, FIH-1 is an asparaginyl hydroxylase enzyme that regulates the transcriptional activity of hypoxia-inducible factor, Genes Dev. 16 (2002) 1466–1471, 10.1101/gad.991402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Wu Y, Li Z, McDonough MA, Schofield CJ, Zhang X, Inhibition of the oxygen-sensing asparaginyl hydroxylase factor inhibiting hypoxia-inducible factor: a potential hypoxia response modulating strategy, J. Med. Chem 64 (2021) 7189–7209, 10.1021/acs.jmedchem.1c00415. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.