Abstract
The aim of the study was to find the association of various risk factors with permanent hearing impairment in infants. A case–control study was designed on 420 infants with permanent hearing impairment and normal hearing. The case control ratio was 1:1. Alternate sampling method was used for selecting the control group. Review of medical records and parent interview was done to collect the information of risk factors. Family history(adj. OR 7.5; 95% CI 3, 14; P = 0.000), Consanguinity (adj. OR: 4; 95% CI 2,4; P = 0.000), intra uterine infection (adj. OR 18, 95% CI: 2.3–126.5, P = 0.000), post natal infection (adj. OR 3, 95% CI: 1.3–5, P = 0.004), low Apgar score (adj.OR: 4.6, 95% CI: 1.3–15), craniofacial anomaly (OR-4.6, 95% CI: 1.4–9.5, P = 0.005) and low birth weight (adj. OR: 2.3, 95% CI: 1.2–3.8) were significantly associated with hearing impairment. Among the risk factors, intra uterine infection was having highest significant association with permanent hearing impairment. This is followed by family history, low Apgar score, craniofacial anomaly, consanguinity, post natal infection and low birth weight.
Introduction
The prevalence of hearing impairment in infants varies from 0.9 to 10/1000. For babies with risk factors it is 8–13/1000 and in well baby nursery it is 0.9–2/1000 [1, 2]. In neonates, hearing impairment is twenty times more prevalent than other disorders such as phenyl ketonuria and sickle cell anaemia that are routinely screened in a hospital [3]. Studies have documented the incidence of hearing impairment in India to be between 0.05 [4] and 6.8% [5]. It is very well documented [6] that if the sensory deficits among babies remain undetected or untreated, it will lead to a significant handicap, and ultimately affect the quality of life of an individual.
High risk factors serve as red flags to clinicians and help them identify those infants for whom screening for hearing impairment may be considered mandatory. This is useful especially in situations where time and manpower are limited. Joint committee of infant hearing [7–9] has pointed out various risk factors for permanent hearing impairment. However this list was based on studies from developed countries. Many authors had pointed out that risk factors may vary in developing countries due to unique social practices and poor medical facilities [10, 11]. Though some preliminary studies have been done in India earlier [12, 13] in enlisting the risk factors, there is no focused study on analyzing and prioritizing them. Hence the present study aims to study systematically the association between high risk factors and permanent hearing impairment in infants.
Method
Research Design and Study Population
A case-control study with the ratio of 1:1 was designed on 420 infants (6 months–2 years). The study group was infants with permanent hearing impairment and controls were infants with normal hearing. Each group had 210 infants. The sample size was determined based on the prevalence of hearing impairment among infants with risk factors as per Cone wesson et al. [14] for the power of 80%. Infants with history of discharge from ear were excluded from the study.
ABR screening using Beraphone MB11 was done for all the controls to rule out the hearing impairment. Permanent hearing impairment in the study group was confirmed by calibrated diagnostic ABR tests done by audiologists. Hospital records were reviewed and parent interview was done to collect the information of risk factors in control and study group.
Risk Factors Studied
Information was collected regarding the following risk factors:
Family history of hearing impairment
Consanguinity
Craniofacial anomalies/syndromes
Hyperbilirubinemia (> 20 mg/dL)
Prematurity (< 37 weeks),
Low Apgar score (< 8 at 5 min),
Low birth Weight (< 2500 gm),
Post natal infection
Intra uterine infection
Seizures (at least one episode)
Ototoxic drugs
NICU admission (> 5 days)
Mechanical ventilation
Consanguinity was selected in this present study because it is an important risk factor in South India [15]. Institutional ethics committee of Sri Ramachandra University, Chennai has approved this study (IEC- NI/08/MAR/03/11).
Data Analysis
Data were tabulated and statistical analysis was done using SPSS version 14.0 (SPSS Inc, Chicago, IL). We performed univariate analyses (Pearson’s chi-square) to identify the contribution of each risk factor for permanent hearing impairment. The significance of single risk factors was further assessed by multivariate analyses (logistic regression) to know their independent association.
Results
The results are classified as findings of hereditary and acquired risk factors. They are further classified to individual and combined risk factors.
Association of Hereditary Factors
Family history and consanguinity were considered as hereditary risk factors and they were found 122/210 in study group and 37/210 in control group. The combination of family history and consanguinity was analyzed separately. The split up of the categories are shown in Table 1. Both individual and combined factors were higher in the study group. There was only one child with the combined factor of family history and consanguineous parents in the control group as opposed to 21 in the study group.
Table 1.
Percentage of hereditary risk factors
| Risk factors | Study gp (N = 210) % (n) |
Control gp (N = 210) % (n) |
|---|---|---|
| Family history of HI | 18.6% (39) | 2.9% (6) |
| Consanguinity | 39.5% (83) | 14.8% (31) |
| Second degree | 20.5% (43) | 6.2% (13) |
| Third degree | 19% (40) | 8.6% (18) |
| Combined factor of Family history and consanguinity | 10% (21) | 0.5% (1) |
Association of Acquired Factors
Infections
Intra uterine and post natal infections were analyzed (Fig. 1) and the prevalence of both these factors was higher in study group. The prevalence of post natal infection was almost double that of intrauterine infection. There was only one child with intrauterine infection in the control group making it a rare risk factor in normal hearing children; whereas it was present in 8% of the study group.
Fig. 1.
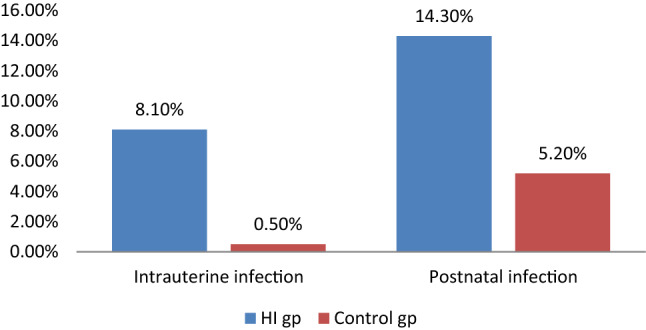
Percentage of intrauterine and postnatal infection
Perinatal risk factors
Perinatal risk factors were hyperbilirubinemia, low birth weight (LBW), preterm birth and low Apgar score. All these risk factors were higher in the study group (Fig. 2) compared to control group. Among them, only hyperbilirubinemia was found to be low prevalent risk factor (> 6%) in the hearing impairment group. LBW and preterm birth were seen almost double in study group. Low Apgar score was present as a rare risk factor with only two infants having in control group while it was seen in 6% of the study group.
Fig. 2.

Percentage of perinatal risk factors
Treatment related risk factors
The prevalence of ototoxicity, mechanical ventilation and NICU admission (> 5 days) were analyzed in the study and control group (Fig. 3). In this the first two factors were not seen in the control group. Absence of these factors in control group may also be due to the unavailability of relevant records to confirm the same. Almost twice the number of babies in the study group was admitted in the NICU beyond 5 days.
Fig. 3.

Percentage of treatment related risk factors
Craniofacial anomaly and seizures
Craniofacial anomaly and seizures was seen in 9.5% and 5.2% of the study group respectively (Fig. 4). The presence of earlier one in the hearing impairment group was four times higher than the control group; whereas both the groups had seizures in equal proportion. Seizures were also found to be a low prevalent risk factor (> 6%).
Fig. 4.

Percentage of craniofacial anomaly and seizures
When the data was subjected to logistic regression analysis (Table 2), family history, Consanguinity, craniofacial anomaly, low Apgar score, low birth weight, intra uterine infection and post natal infection were found to be significantly associated with PHI. Risk factors could be ranked based on risk estimate yielded by adjusted odds ratio. IUI with highest risk estimate was having 17 times the chance of causing PHI. Family history of hearing loss was having eight times the chance of causing PHI. A child with craniofacial anomaly and low Apgar score had six fold chance of having PHI than a child without the risk factors. Infants with postnatal infection and LBW were having two and half times risk of possessing PHI. Other risk factors were not having significant association with PHI.
Table 2.
Multi variate logistic regression analysis of risk factor association
| Sl. no | Risk factors | Adjusted odds ratio | 95% CI | p value | |
|---|---|---|---|---|---|
| Lower | Upper | ||||
| 1 | Family history | 8.1 | 3.2 | 20.6 | 0.000* |
| 2 | Consanguinity | 4.1 | 2.5 | 7 | 0.000* |
| 3 | Craniofacial anomaly | 6.6 | 2.2 | 19.4 | 0.001* |
| 4 | Hyperbilirubinemia | 2.6 | 0.7 | 9.9 | 0.16 |
| 5 | Preterm birth | 1.1 | 0.4 | 3.1 | 0.83 |
| 6 | Low Apgar score | 6.4 | 1.6 | 25.6 | 0.008* |
| 7 | Low birth weight | 2.5 | 1.1 | 6 | 0.034* |
| 8 | Post natal infection | 2.6 | 1.1 | 6.1 | 0.03* |
| 9 | Intra uterine infection | 17.1 | 2.1 | 139 | 0.008* |
| 10 | Seizures | 0.6 | 0.2 | 1.8 | 0.383 |
| 11 | Ototoxic drugs† | undefined | Undefined | 0.999 | |
| 12 | NICU | 0.8 | 0.3 | 2.4 | 0.68 |
| 13 | Mechanical ventilation† | undefined | Undefined | 0.999 | |
*Statistically significant, p = 0.05
†Adjusted Odds ratio couldn’t be found out due to absence of risk factor in control group
Discussion
WHO [16] had reported that two-third of the population with HI are in the developing world which includes India. It also says more than 50% of them have preventable causes. The prevalence and association with hearing impairment determines the importance of risk factors. Both are important indicators in making an action plan of prevention of HI. It is a well-known fact that the importance of risk factors differ from country to country [17]. Table 3 shows the comparative picture of significant risk factors across various countries. Most of the developed countries do not have infections as a significant risk factor for HI except for UK. The developed countries and few developing countries such as Cuba and Oman have successfully reduced the incidence of intrauterine and congenital infections by effective immunization programs [18]. Their governments have taken prompt action against infections due to their indigenous published reports across time. The published reports became a justification for introduction of immunization against such diseases in those countries. This in turn became the primary prevention of hearing impairment.
Table 3.
Significant risk factors of HI across the countries
| USA [14, 22] | UK [23] | Australia [24] | Turkey [25] | Nicaragua [26] | China [27] | Nigeria [28, 29] | India (present study) | |
|---|---|---|---|---|---|---|---|---|
| Family history | Yes | Yes | Yes | Yes | ||||
| Consanguinity | Yes | |||||||
| Infection | Yes | Yes | Yes | Yes | Yes | |||
| Hyperbilirubinemia | Yes | |||||||
| Low Apgar score | Yes | Yes | ||||||
| NICU admission | Yes | Yes | Yes | |||||
| Preterm birth | Yes | |||||||
| Ototoxicity | Yes | Yes | ||||||
| Craniofacial anomaly/syndrome | Yes | Yes | Yes | Yes | Yes | |||
| Mechanical ventilation | Yes | Yes | Yes | |||||
| Low birth weight | Yes | Yes | Yes |
Immunisation against Haemophilus influenza type b and Pneumococcus can prevent the incidence of meningitis and its related complications. The load of H. influenza type b meningitis had been reduced by 98 per cent in developed nations due to expanded immunization programs [19]. In western countries, improved medical care with corticosteroid therapy for children with meningitis has become a standard of care which decreased the incidence of sensorineural hearing loss. It is expected that improved basic community health and child care will subsequently have a similar impact in developing countries as well [20].
In India, the campaign for Rubella vaccination started from 2017. The reason why rubella is not recognized as serious infection for many years is due to several factors such as lack of large scale research evidence of prevalence of rubella infection, methodological issues in testing TORCH infection and higher cost of testing [21]. Given the huge challenges in the prevention of hearing impairment, Government of India had initiated National program of prevention and control of deafness (NPPCD) in 2006. As the name indicates, one of the goals of the program was to prevent avoidable causes of hearing loss and to do early identification & intervention of hearing impairment. It recommends both hospital and community based screening to accomplish these goals. To serve as a baseline for such Government initiatives, there is an urgent need of more and more indigenous studies of risk factors. Only these can form a base for taking active measure in allocation of funds to the hearing health care.
Another risk factor which sets India apart from developed countries is consanguinity. This factor is shown to increase the cases of hearing impairment in isolation and in combination with family history in the present study. Bittles [30] reported consanguinity to be highly prevalent in gulf countries, North African countries and Pakistan. He has also reported this to be highly prevalent in southern India where the present study was done. The link between consanguinity and hearing impairment is well established and physicians generally recommend avoiding these marriages. While there is increasing evidence that consanguinity is decreasing in some countries [31], it is still to be addressed with more seriousness in southern India. Zakzouk et al. [32] speculated that industrialization, increased travel and continuous change in social customs may decrease consanguinity in Asian countries in the coming days.
A recent study in South India [33] revealed around 64% of grandmothers of newborn were aware of the fact that consanguinity may lead to hearing impairment. Their knowledge scores for consanguinity and family history as a cause of hearing impairment was within the top five in the list. When compared with the high prevalence of consanguinity in India, the above data suggest that people may willingly opt for consanguineous marriages for their children even though they are aware of possible risk of consanguinity being associated with hearing impairment. For them it may be a tradeoff between family securities, social compulsion versus risk of having a disabled child.
The other risk factors which are significant in this study are low Apgar score and craniofacial anomaly. In Netherland [34] and Poland [35], authors have linked low Apgar score independently with hearing impairment. In Malaysia [2] and Iran [36], it was not a significant risk factor. Many authors have linked low Apgar score with developmental issues rather than just hearing impairment [10, 22]. In Israel [37], it was identified to be a significant risk factor for conductive hearing loss among infants with low birth weight. In the above discussion it is clear that risk factors vary considerably across various countries though partly this variance could be due to methodological difference.
Conclusion
In summary, intra uterine infection was having highest significant association with permanent hearing impairment. This is followed by family history of hearing loss, low Apgar score, craniofacial anomaly, consanguinity, post natal infection and low birth weight. Appropriate immunization for adolescent girls/expectant mothers, inclusion of rubella and meningitis in National immunization policy, change of consanguineous patterns of marriage, and increased awareness of risk factors by health education is the way forward in prevention and early identification of hearing loss in countries like India.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Prieve B, Dalzell L, Berg A, Bradley M, Cacace A, Campbell D, et al. The New York State universal newborn hearing screening demonstration project: outpatient outcome measures. Ear Hear. 2000;21:104–117. doi: 10.1097/00003446-200004000-00005. [DOI] [PubMed] [Google Scholar]
- 2.Khairi MDM, Din S, Shahid H, Normastura AR. Hearing screening of infants in Neonatal Unit, Hospital Universiti Sains Malaysia using transient evoked otoacoustic emissions. J Laryngol Otol. 2005;119:678–683. doi: 10.1258/0022215054798014. [DOI] [PubMed] [Google Scholar]
- 3.Oghalai JS, Chen L, Brennan ML, Tonini R, Manolidis S. Neonatal hearing loss in the indigent. Laryngoscope. 2002;112:281–286. doi: 10.1097/00005537-200202000-00015. [DOI] [PubMed] [Google Scholar]
- 4.Nagapoornima P, Ramesh A, Rao S, Patricia PL, Gore M, Dominic M. Universal hearing screening. Indian J Pediatr. 2007;74:29–33. doi: 10.1007/s12098-007-0105-z. [DOI] [PubMed] [Google Scholar]
- 5.ICMR, Collaborative study on prevalence and aetiology of hearing impairment. Project report., New Delhi, 1983.
- 6.Yoshinaga-Itano C, Sedey AL, Coulter DK, Mehl AL. Language of early- and later-identified children with hearing loss. Pediatrics. 1998;102:1161–1171. doi: 10.1542/peds.102.5.1161. [DOI] [PubMed] [Google Scholar]
- 7.Joint Committee on Infant Hearing (JCIH) Position statement. ASHA. 1994;36(12):38–41. [PubMed] [Google Scholar]
- 8.Joint Committee on Infant Hearing (JCIH) Year 2000 position statement: principles and guidelines for early hearing detection and intervention programs. Pediatrics. 2000;106:798–817. doi: 10.1542/peds.106.4.798. [DOI] [PubMed] [Google Scholar]
- 9.Joint Committee on Infant Hearing (JCIH) Year 2007 position statement: principles and guidelines for early hearing detection and intervention programs. Pediatrics. 2007;120(4):898–921. doi: 10.1542/peds.2007-2333. [DOI] [PubMed] [Google Scholar]
- 10.Olusanya BO. Making targeted screening for infant hearing loss an effective option in less developed countries. Int J Pediatr Otorhinolaryngol. 2011;75:316–321. doi: 10.1016/j.ijporl.2010.12.002. [DOI] [PubMed] [Google Scholar]
- 11.Krishnan LA, Lafayette W, Donaldson LK. Newborn hearing screening in developing countries: understanding the challenges and complexities of implementation. Perspect Glob Issues Commun Sci Relat Disord. 2012;3(2):54–61. [Google Scholar]
- 12.D’Mello J. High risk register—An economical tool for early identification of hearing loss. Indian J Pediatr. 1995;62:731–735. doi: 10.1007/BF02825129. [DOI] [PubMed] [Google Scholar]
- 13.Bhagya V, Brid SV, Doddamani M. Incidence of hearing loss in infants at risk. Int J Biol Med Res. 2011;2:1102–1105. [Google Scholar]
- 14.Vohr BR, Widen JE, Cone-Wesson B, Sininger YS, Gorga MP, Folsom RC, et al. Identification of neonatal hearing impairment: characteristics of infants in the neonatal intensive care unit and well-baby nursery. Ear Hear. 2000;21:373–382. doi: 10.1097/00003446-200010000-00005. [DOI] [PubMed] [Google Scholar]
- 15.Verma I, Prema A, Puri R. Health effects of consanguinity in pondicherry. Indian Pediatr. 1992;29:685–692. [PubMed] [Google Scholar]
- 16.WHO (2013) Deafness and hearing loss-Fact sheet. http://www.who.int/mediacentre/factsheets/fs300/en/. Accessed 07 Nov 2013
- 17.Parving A. The need for universal neonatal hearing screening —Some aspects of epidemiology and identification. Acta Pediatr. 1999;88:69–72. doi: 10.1111/j.1651-2227.1999.tb01163.x. [DOI] [PubMed] [Google Scholar]
- 18.Robertson SE, Featherstone DA, Gacic-dobo M, Hersh BS. Rubella and congenital rubella syndrome: global update. Rev Panam Salud Publica. 2003;14:306–315. doi: 10.1590/S1020-49892003001000005. [DOI] [PubMed] [Google Scholar]
- 19.Tarabichi MB, Todd C, Khan Z, Yang X, Shehzad B, Tarabichi MM. Deafness in the developing world: the place of cochlear implantation. J Laryngol Otol. 2008;122:877–880. doi: 10.1017/S0022215108002272. [DOI] [PubMed] [Google Scholar]
- 20.Smith R, Bale JF, White KR. Sensorineural hearing loss in children. Lancet. 2005;365:879–890. doi: 10.1016/S0140-6736(05)71047-3. [DOI] [PubMed] [Google Scholar]
- 21.Dewan P, Gupta P. Burden of Congenital Rubella Syndrome (CRS) in India: a systematic review. Indian Pediatr. 2012;49:377–399. doi: 10.1007/s13312-012-0087-4. [DOI] [PubMed] [Google Scholar]
- 22.Cone-Wesson B, Vohr BR, Sininger YS, Widen JE, Folsom RC, Gorga MP, et al. Identification of neonatal hearing impairment: infants with hearing loss. Ear Hear. 2000;21:488–507. doi: 10.1097/00003446-200010000-00012. [DOI] [PubMed] [Google Scholar]
- 23.Wood SA, Davis AC, Sutton GJ. Effectiveness of targeted surveillance to identify moderate to profound permanent childhood hearing impairment in babies with risk factors who pass newborn screening. Int J Audiol. 2013;52:394–399. doi: 10.3109/14992027.2013.769067. [DOI] [PubMed] [Google Scholar]
- 24.Beswick R, Driscoll C, Kei J, Khan A, Glennon S. Which risk factors predict postnatal hearing loss in children? J Am Acad Audiol. 2013;24:205–213. doi: 10.3766/jaaa.24.3.6. [DOI] [PubMed] [Google Scholar]
- 25.Eras Z, Konukseven O, Aksoy HT, Canpolat FE, Genç A, Sakrucu ED, et al. Postnatal risk factors associated with hearing loss among high-risk preterm infants: tertiary center results from Turkey. Eur Arch Oto-Rhino-Laryngol. 2014;271:1485–1490. doi: 10.1007/s00405-013-2653-3. [DOI] [PubMed] [Google Scholar]
- 26.Saunders JE, Vaz S, Greinwald JH, Lai J, Morin L, Mojica K. Prevalence and etiology of hearing loss in rural Nicaraguan children. Laryngoscope. 2007;117:387–398. doi: 10.1097/MLG.0b013e31802e3726. [DOI] [PubMed] [Google Scholar]
- 27.Nie W, Wu H, Qi Y, Lin Q, Xiang L, Li H, et al. A case-control study on high-risk factors for newborn hearing loss in seven cities of Shandong province. J Huazhong Univ Sci Technol. 2007;27:217–220. doi: 10.1007/s11596-007-0230-y. [DOI] [PubMed] [Google Scholar]
- 28.Dunmade A, Segun-Busari S, Olajide T, Ologe F. Profound bilateral sensorineural hearing loss in nigerian children: any shift in etiology? J Deaf Stud Deaf Educ. 2007;12:112–118. doi: 10.1093/deafed/enl019. [DOI] [PubMed] [Google Scholar]
- 29.Olusanya BO, Wirz S, Luxon L. Non-hospital delivery and permanent congenital and early-onset hearing loss in a developing country. Br J Obstet Gynaecol. 2008;115:1419–1427. doi: 10.1111/j.1471-0528.2008.01871.x. [DOI] [PubMed] [Google Scholar]
- 30.Bittles A. Consanguinity in context. 1. Cambridge: Cambridge University Press; 2012. pp. 1–12. [Google Scholar]
- 31.Levi H, Tell L, Cohen T. Sensorineural hearing loss in Jewish children born in Jerusalem. Int J Pediatr Otorhinolaryngol. 2004;68:1245–1250. doi: 10.1016/j.ijporl.2004.04.021. [DOI] [PubMed] [Google Scholar]
- 32.Zakzouk S. Consanguinity and hearing impairment in developing countries: a custom to be discouraged. J Laryngol Otol. 2002;116:811–816. doi: 10.1258/00222150260293628. [DOI] [PubMed] [Google Scholar]
- 33.Rajagopalan R, Selvarajan HG, Rajendran A, Ninan B. Grandmothers’ perspective on hearing loss in children and newborn hearing screening. Indian J Otol. 2014;20:20–23. doi: 10.4103/0971-7749.129803. [DOI] [Google Scholar]
- 34.Coenraad S, Goedegebure A, van Goudoever JB, Hoeve LJ. Risk factors for sensorineural hearing loss in NICU infants compared to normal hearing NICU controls. Int J Pediatr Otorhinolaryngol. 2010;74:999–1002. doi: 10.1016/j.ijporl.2010.05.024. [DOI] [PubMed] [Google Scholar]
- 35.Wroblewska-Seniuk K, Chojnacka K, Pucher B, Szczapa J, Gadzinowski J, Grzegorowski M. The results of newborn hearing screening by means of transient evoked otoacoustic emissions. Int J Pediatr Otorhinolaryngol. 2005;69:1351–1357. doi: 10.1016/j.ijporl.2005.03.023. [DOI] [PubMed] [Google Scholar]
- 36.Fakhim SA, Naderpoor M, Shahidi N, Basharhashemi F, Sakha SH, Alizadeh M. Study of prevalence and causes of hearing loss in high risk neonates admitted to neonatal ward and neonatal intensive care unit. J Int Adv Otol. 2010;6:365–370. [Google Scholar]
- 37.Ari-Even Roth D, Hildesheimer M, Maayan-Metzger A, Muchnik C, Hamburger A, Mazkeret R, et al. Low prevalence of hearing impairment among very low birthweight infants as detected by universal neonatal hearing screening. Arch Dis Child Fetal Neonatal Ed. 2006;91(4):F257–262. doi: 10.1136/adc.2005.074476. [DOI] [PMC free article] [PubMed] [Google Scholar]


