Abstract
Myelodysplastic syndromes (MDS) are clonal hematopoietic disorders, representing high risk of progression to acute myeloid leukaemia, and frequently associated to somatic mutations, notably in the epigenetic regulator TET2. Natural Killer (NK) cells play a role in the anti-leukemic immune response via their cytolytic activity. Here we show that patients with MDS clones harbouring mutations in the TET2 gene are characterised by phenotypic defects in their circulating NK cells. Remarkably, NK cells and MDS clones from the same patient share the TET2 genotype, and the NK cells are characterised by increased methylation of genomic DNA and reduced expression of Killer Immunoglobulin-like receptors (KIR), perforin, and TNF-α. In vitro inhibition of TET2 in NK cells of healthy donors reduces their cytotoxicity, supporting its critical role in NK cell function. Conversely, NK cells from patients treated with azacytidine (#NCT02985190; https://clinicaltrials.gov/) show increased KIR and cytolytic protein expression, and IFN-γ production. Altogether, our findings show that, in addition to their oncogenic consequences in the myeloid cell subsets, TET2 mutations contribute to repressing NK-cell function in MDS patients.
Subject terms: NK cells, Myelodysplastic syndrome, Translational immunology, Immunosurveillance
Myelodysplastic syndromes are characterised by clonal haematopoiesis, with the affected cells often harbouring mutations in the TET2 gene, an important regulator of DNA methylation state. Here authors show that the same mutations are also found in NK cells, perturbing their DNA methylation pattern and cytolytic function.
Introduction
Myelodysplastic Syndromes (MDS) are clonal myeloid malignancies characterized by ineffective hematopoiesis and bone marrow (BM) failure leading to blood cytopenias and a high risk of transformation to Acute Myeloid Leukemia (AML)1. MDS patients with a high risk of AML progression (“high-risk MDS” according to the international prognostic scoring system) are treated with hypomethylating agents (HMA) including azacitidine (AZA) and decitabine (DAC) which can increase overall survival and reduce AML progression2,3. Mutations in TET2 and IDH1/2 genes, involved in the epigenetic regulation of gene transcription, are common genetic alterations found in MDS1. Whereas TET2 encodes for a methylcytosine dioxygenase catalyzing the oxidation of the methyl group of 5-methycytosine (5mC) to 5-hydroxymethylcytosine (5-hmC), the isocitrate dehydrogenases (IDH) 1 and 2 control the production of α-ketoglutarate (αKG), a metabolite required for TET2 activity4. Most TET2 mutations described are loss of function and are generally mutually exclusive with IDH1/2 mutations. By contrast, IDH1/2 mutations in MDS lead to a new enzymatic activity that generates 2-hydroxyglutarate (2HG) in place of αKG and inhibits TET2 activity5. Therefore, TET2 and IDH1/2 mutations lead to hypermethylation of the genomic DNA in myeloid cells6. MDS patients carrying mutations in TET2 are more likely to respond to HMA treatment7, but mechanisms of action of HMA on MDS cells are still unclear and could involve the BM microenvironment, including immune (lymphocytes, myeloid-derived suppressive cells)8 and non-immune partners (mesenchymal stromal cells)9.
Natural Killer (NK) lymphocytes are key players in the anti-leukemic response. NK cell targets malignant cells by direct cytotoxicity through the release of perforin, granzymes, engagement of death receptors, or secreting cytokines like IFN-γ and TNF-α10,11. Their function is regulated by a balance between inhibitory (KIR, NKG2A) and activating (NKp30, NKp46, DNAM-1, NKG2D) receptors. We and others have shown defects in NK cell phenotype and function in AML and MDS patients12–15, but mechanisms leading to deficient NK cells are still unclear. Moreover, whether the enhanced sensitivity to HMA treatment of MDS patients with TET2/IDHMUT relies on the restoration of NK-cell activity is unknown.
In this study, we show MDS TET2/IDH mutations are carried by NK cells and associated with defects in phenotype and function. Those perturbations rely on genome-wide hypermethylation of DNA, leading to the reduced expression of key molecules for NK cell function. Finally, HMA treatment normalizes the expression of NK cell-related genes in vitro as well as in vivo. Altogether, our results established that TET2 regulates NK-cell biology, and suggest that HMA treatments in TET2/IDH mutated patients could also be beneficial by restoring the functionality of NK cells.
Results
MDS/CMML patients with TET2/IDH mutations display a distinct NK cell phenotype
Blood NK cells from 33 MDS/CMML patients (myelodysplastic syndromes and chronic myelomonocytic leukemia, respectively, Supplementary Data 1) and 23 HD were phenotyped by flow cytometry. Surface expression of maturation markers (CD69, CD57, KLRG1), activating (NKp30, NKp46, DNAM-1, NKG2D) and inhibitory (CD96, KIR2D, CD85j) receptors was investigated (Supplementary Table 1). Small perturbations were observed on circulating NK cells of MDS/CMML individuals compared with HD in the expression of some activating (DNAM-1, NKp46) and inhibitory (CD96) receptors. Surprisingly, an almost 2-fold reduction of the killer immunoglobulin-like receptors (KIR) expression was found in MDS patients’ NK cells compared to HD (31.2% and 54.8%, respectively, p < 0.01, Supplementary Table 1).
We asked whether patients with mutations affecting DNA methylation (i.e., mutations in TET2, IDH1, or IDH2) could exhibit specific NK-cell phenotypes (Fig. 1, Supplementary Table 1). Interestingly, a deep loss in the expression of KIRs with 2 Ig-like extracellular domains (KIR2D) was observed in patients with TET2/IDH mutations compared to TET2/IDHWT on blood (n = 19 vs. 13, median 11.9% vs. 56.5%, p < 0.001, Fig. 1a) and BM NK cells (n = 7 vs. 12, median 24.2% vs. 45.9% p < 0.05, Fig. 1b). In parallel, the inhibitory receptor NKG2A was significantly increased on peripheral NK cells in TET2/IDHMUT patients compared to TET2/IDHWT (n = 15 vs. 11, 86.3% vs. 78.7%, p = 0.025, Fig. 1A, Supplementary Table 2), underlying the balance between KIRs and NKG2A expression16. Of note, the KIR-positive T-cell subset was not affected by the TET2/IDH mutational status in patients (Supplementary Fig. 1). Importantly, censuring the few patients treated with either drug to stimulate the erythro-thrombopoiesis (EPO n = 4 out of 63 patients, TPO n = 1) or immunomodulating molecules (lenalidomide n = 4, glucocorticoid n = 4) did not modify our observations demonstrating that treatments were not at the origin of the decrease in KIR expression (Supplementary Fig. 2).
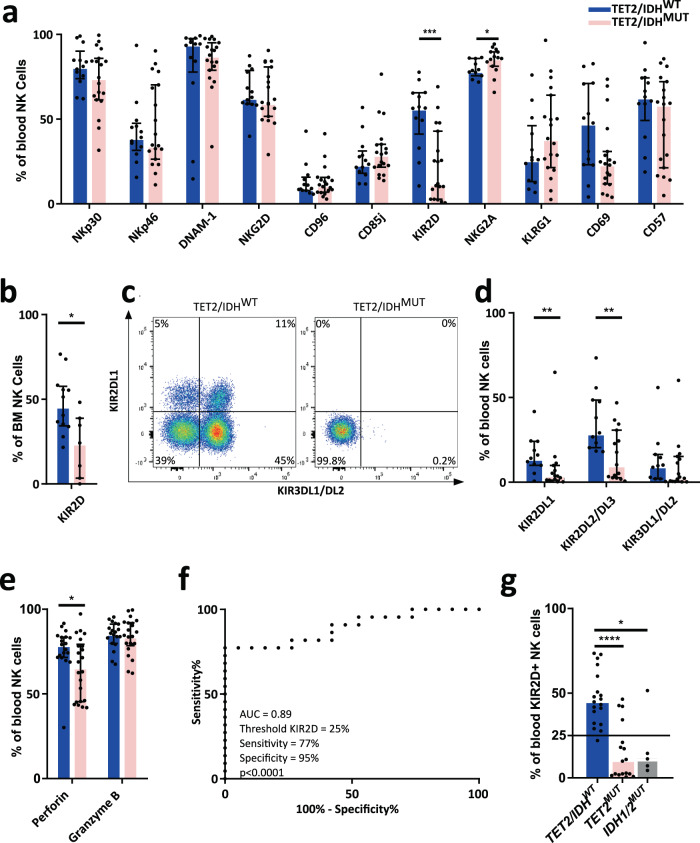
Fig. 1. TET2/IDH mutations in MDS/CMML patients lead to the reduction of KIR and Perforin expression in NK cells.
a Percentages of blood NK cells expressing activating (NKp30, NKp46, DNAM-1, NKG2D) and inhibitory (CD96, CD85j, KIR2D, NKG2A) receptors, and maturation/activation markers (KLRG1, CD69, CD57) were measured by flow cytometry in TET2/IDHWT (n = 13, n = 11 for NKG2A) and TET2/IDHMUT (n = 19, n = 15 for NKG2A) MDS/CMML patients. Statistics were calculated with the nonparametric Mann–Whitney test, two-sided, KIR2D ***p = 0.0005, NKG2A *p = 0.0246. b Percentages of KIR2D+ BM NK cells measured by flow cytometry in TET2/IDHWT (n = 12) and TET2/IDHMUT (n = 7) MDS/CMML patients. Statistics were calculated with the nonparametric Mann–Whitney test two-sided *p = 0.013. c and d Specific expression of KIR2DL1, KIR2DL2/DL3, and KIR3DL1/DL2 in blood NK cells detected by flow cytometry in TET2/IDHWT (n = 11) and TET2/IDHMUT (n = 15) MDS/CMML patients. One representative example is shown in c. Statistics were calculated with the nonparametric Mann–Whitney test, KIR2DL1 **p = 0.0092, two-sided KIR2DL2/DL3 **p = 0.0077. e Intracellular perforin and granzyme B expression in blood NK cells measured by flow cytometry in TET2/IDHWT (n = 19) and TET2/IDHMUT (n = 22) MDS/CMML patients. Statistics were calculated with the nonparametric Mann–Whitney test *p = 0.0337. f Receiver operating characteristic (ROC) curve depicting the relationship of true TET2 mutation presence (sensitivity) and false TET2 mutation presence (100%-specificity) for a KIR2D expression threshold at 25% in blood NK cells (p < 0.0001) quantified by flow cytometry in TET2/IDHWT (n = 19) and TET2MUT (n = 17) MDS/CMML patients. g KIR2D expression on blood NK cells of the TET2/IDHWT (n = 19), TET2MUT (n = 17) and IDHMUT (n = 5) patients showed in the ROC curve. The horizontal black bar indicates the threshold at 25% KIR2D+ NK cells. Statistics were calculated with the nonparametric Mann–Whitney test, TET2/IDHWT vs. TET2MUT: ****p < 0.0001; TET2/IDHWT vs. IDHMUT: *p = 0.0152. For all the analysis, data are presented as medians and interquartile ranges. Source data are provided as a Source Data file.
KIRs are encoded in a 15 genes locus with high variability in gene content across individuals17. To investigate if TET2/IDH mutations could differentially affect KIR genes expression, the KIR locus was genotyped for 22 MDS/CMML patients, and KIR surface expression was analyzed for genes highly present with KIR2DL1, KIR2DL2/DL3, and KIR3DL1/DL2 specific mAbs (Fig. 1c, d, Supplementary Fig. 3 and Table 3). We confirmed a specific reduction of KIR2DL1 (3.6% vs. 14.1%, p < 0.01) and KIR2DL2/DL3 (10.3% vs. 29.1%, p < 0.01) on NK cells of TET2/IDHMUT patients compared to WT (n = 15 vs. n = 11, respectively) (Fig. 1c, d, Supplementary Table 2). By contrast, KIR3DL1/DL2 expression was not significantly modified by the TET2/IDH mutation status (Fig. 1d), despite the strong reduction observed in some patients (Fig. 1c, d).
We next quantified perforin and granzyme B expression in NK cells of MDS/CMML patients as surrogate markers of NK-cell cytolytic capacity. We observed a reduced percentage of perforin-positive NK cells in TET2/IDHMUT patients (p < 0.05), whereas granzyme B expression was not affected (Fig. 1e). This loss of intracellular perforin was correlated with reduced KIR (KIR2D and KIR3DL1/DS1) expression at the NK-cell surface in TET2/IDHMUT patients (r = 0.65, p = 0.001; Supplementary Fig. 4). Indeed, the loss of perforin expression was mainly observed in KIR− NK cells whereas KIR+ NK-cells were not affected (Supplementary Fig. 5), suggesting a co-regulation of KIR and perforin expressions.
Then, we reasoned that the TET2 function may be the main parameter modifying the NK cell phenotype. Therefore, we analyzed the performance of KIR2D expression as a classifier of patients with or without TET2 mutations by calculating a receiver operation characteristic (ROC) curve using KIR2D expression on circulating NK cells in TET2/IDHWT (n = 19) and TET2MUT (n = 17) patients. A maximum area under the curve (AUC) equal to 0.89 predicted that a threshold of 25% of KIR2D+ NK cells allowed classifying mutated and not-mutated patients with a specificity of 95% and a sensitivity of 77% (p < 0.0001, Fig. 1f). Studying KIR2D expression separately in TET2MUT and IDH1/2MUT patients, we observed that 4 out of 5 patients mutated for either IDH1 or IDH2 showed a comparable decrease of KIR2D expression than TET2MUT individuals (Fig. 1g). This observation confirmed that IDH1/2 mutations could phenocopy TET2 mutations at least when considering NK cell phenotype.
Altogether, our results unraveled a strong relationship between the presence of mutations in TET2 and IDH1/2 genes in MDS/CMML patients and the alterations in NK-cell phenotype. To gain further insights into the molecular mechanisms accounting for this loss of NK cell function, we focused our study on TET2 mutated patients, excluding the IDH mutated patients from our cohort, and interrogated whether NK and MDS cells could share TET2 mutations.
TET2 mutations observed in MDS/CMML patients at diagnosis can be found in blood NK-cells
All MDS/CMML patients recruited in this study were analyzed at diagnosis of the disease for the presence of somatic mutations in 80 genes usually found mutated in myeloid malignancies (Supplementary Table 4). As expected, TET2MUT and TET2/IDHWT patients could show mutations in additional genes frequently observed in MDS/CMML1, including SRSF2 (29% and 3%, respectively), SF3B1 (18% and 20%, respectively) or ASXL1 (18% and 17%, respectively) (Supplementary Fig. 6 and Supplementary Table 5). Of note, TET2MUT patients presented a mean of 2 mutations in TET2 (range 1–4 mutations).
Thus, we analyzed whether MDS-associated mutations were present in NK cells. PBMC of 10 MDS patients diagnosed with mutations in TET2 among other genes were sorted for NK cells. Nine out of 10 patients showed some mutations in NK cells shared with the blood or BM white mononuclear cell (WMC) samples at diagnosis, including in TET2 and other genes (Supplementary Table 6). The variant allele frequencies (VAF) for TET2 mutations were not significantly different in NK cells (median = 11%, range 0–54%) compared to WMC (median = 32%, range 2–49%, Fig. 2a). By contrast, consistent with their unchanged phenotype previously observed, MDS-associated mutations were rarely found in T cells (Supplementary Table 6, Supplementary Fig. 7A), and with a VAF below 7% when present (Fig. 2a). Importantly, whereas 7 patients presented a strict concordance in the mutation profiles between analyses on WMC and NK cells (Fig. 2b and Supplementary Table 6), 3 patients (MUT19, MUT22, MUT31) showed mutations in NK cells unobserved in WMC (Fig. 2b).
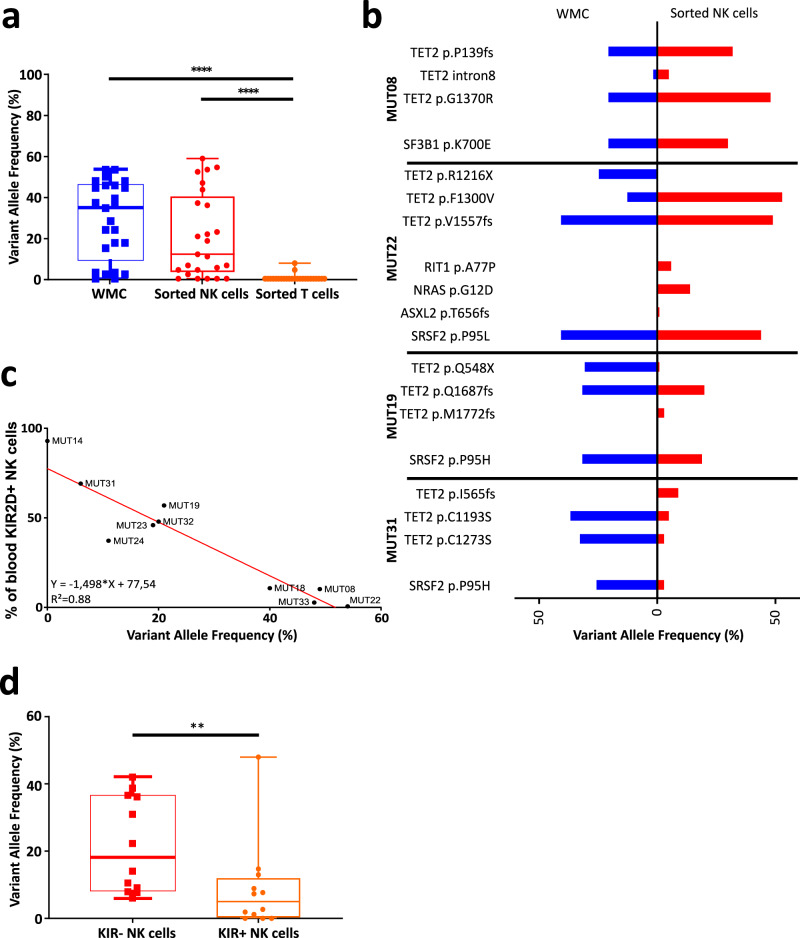
Fig. 2. TET2 mutations identified in bulk cells of MDS patients at diagnosis are also observed in NK cells and correlate with KIR2D expression.
a The percentage of Variant Allele Frequency (VAF) for the mutations of TET2 observed in the white mononuclear cells (WMC) and in sorted NK/T cells at diagnosis of TET2MUT MDS/CMML patients (n = 10) was evaluated by NGS analysis (with a range from 1 to 4 mutations per patient). Data are represented as box-and-whisker plots (minimum VAF, 25% percentile, median, 75% percentile, and maximum VAF respectively for WMC: 0%, 11.25%, 32.5%, 42.5%, 49%; for NK cells: 0%, 3.5%, 14%, 35.5%, 54%; and for T cells: 0%, 0%, 0%, 0%, 7%). Nonparametric two-sided Wilcoxon matched-pairs signed rank test was used to determine statistical significance. ****p < 0.0001. b VAF (%) of the different mutations detected in the WMC at diagnosis (blue) and in sorted NK cells (red) in 4 patients (MUT08, MUT22, MUT19, MUT31; see Supplementary Table 6 for more information). c Correlation curve between the percentage of KIR2D+ NK cells and the TET2 VAF (%) in blood NK cells from the MDS/CMML patients analyzed in (a). Linear regression was calculated, r = 0.88, p < 0.0001. d The VAF percentage in the WMC and in sorted KIR+ and KIR− NK cells at diagnosis from TET2MUT MDS/CMML patients (n = 5, range from 1 to 4 mutations per patient) was evaluated by NGS analysis. Data are represented as box-and-whisker plots (minimum VAF, 25% percentile, median, 75% percentile, and maximum VAF respectively for KIR− NK cells: 6.1%, 8.3%, 18.2%, 36.5%, 42.1%; and KIR+ NK cells: 0%, 1.2%, 7.3%, 13%, 48%). Statistics were calculated with a two-sided nonparametric Wilcoxon matched-pairs signed rank test. *p = 0.0186. Source data are provided as a Source Data file.
Furthermore, we observed a strong correlation between the expression of KIR2D receptor and the VAF of TET2 mutations in NK cells (Fig. 2c, R2 = 0.88, p < 0.0001). In the 10 patients analyzed by NGS, those with a percentage of KIR2D+ NK-cells below 25% showed a TET2 mutation VAF higher than 20% in the WMC at diagnosis and higher or equal to 34% in the NK-cell subset. To confirm this observation, we selected 5 patients with sufficient amounts of KIR2D+ and KIR2D− NK cells and performed an NGS analysis on cells sorted according to the expression of the KIR2D and KIR3DL1/DL2 (hereafter designed has KIR+ or KIR− NK cells). Importantly, 4 out of 5 patients showed a high percentage of KIR2D+ cells, superior to the threshold of 25% we have calculated previously (Fig. 1F). As expected, patients showed higher TET2MUT VAF in the KIR− (median = 18.2%, range 6.1–42.1%) than the KIR+ cells (median = 5%, range 0–48%, p < 0.01, Fig. 2d, Supplementary Fig. 7B). In addition, KIR− NK cell subset showed TET2 mutations leading to non-functional TET2 proteins in all patients tested. In detail, patients MUT08, MUT19, and MUT23 presented frameshift mutations, leading to premature stop-codon, with VAFs ranging from 22% to 37% in KIR− cells, as compared to a range from 2% to 15% in the KIR+ cells. Patients MUT24 and MUT32 showed missense mutations in the TET2 catalytic domain, with VAFs ranging from 11% to 39% in the KIR− cells as compared to 1% to 7% in the KIR+ cells. Moreover, two patients (MUT19 and MUT23) showed a shared nonsense mutation at position 548 only present in the KIR− NK cells. Mutations in other genes were also enriched in the KIR− population (Supplementary Table 6). Altogether, these observations suggested that TET2 mutations can be found in NK cells and are related to reduced KIR expression in TET2MUT MDS/CMML patients.
TET2 directly binds to and regulates KIR locus expression in NK cells
To evaluate the role of TET2 in regulating KIR expression, we first analyzed the type and the positioning of the TET2 mutations found in MDS/CMML patients with a reduced percentage of KIR+ NK cells (below 25%) at diagnosis (Fig. 3a). This analysis revealed that all but one patient showed either nonsense or frameshift mutations leading to a premature stop-codon and the absence of a functional TET2 protein, extending the observation made in KIR− NK cells (Supplementary Table 6). The only exception was the patient MUT30 who presented a missense mutation p.R1261H (c.G3782A substitution). This mutation has already been described in the catalog of somatic mutations in cancer (COSMIC) database18 with a functional consequence prediction (Functional Analysis through Hidden Markov Models (FATHMM)) score equal to 0.9919, forerunning a highly significant functional impact on the TET2 protein.
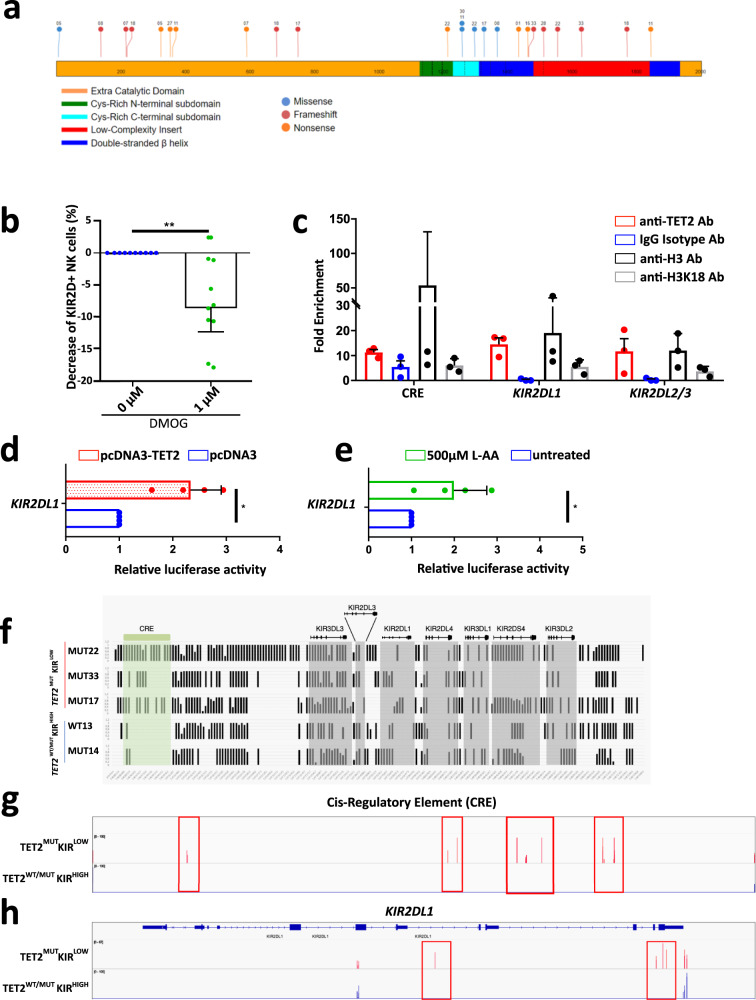
Fig. 3. TET2 regulates KIR expression on NK cells through its binding onto and regulating methylation of the KIR locus.
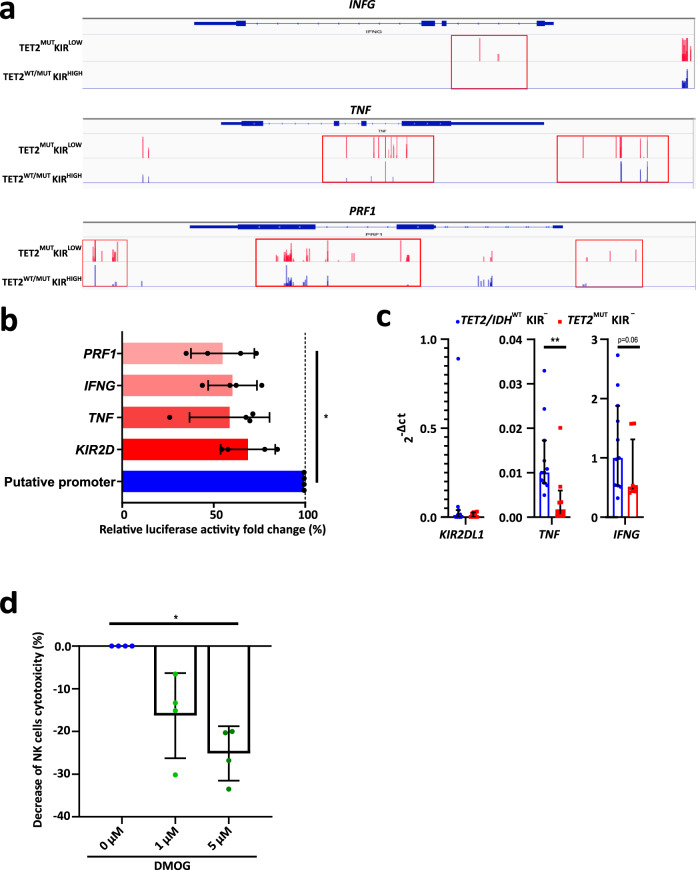
a TET2 mutational landscape in the 13 TET2MUT patients with KIR2D expression below 25% established with the St Jude protein paint software (https://proteinpaint.stjude.org/). Mutations were classified as Missense (blue), Frameshift (red), and Nonsense (orange). Numbers associated with each mutation designed patients (Supplementary Data 1). b Percentage of KIR2D+ NK cells after 5 days of in vitro treatment with 1 µM DMOG (n = 10). DMSO alone was used as a control. One dot represents one PBMC sample. Nonparametric two-sided Wilcoxon matched-pairs signed rank test was used to determine statistical significance. **p = 0.0059. Data are presented as median and interquartile range. c Fold enrichment of sequences specific for the Cis-regulatory element (CRE), KIR2DL1 promoter, and KIR2DL2/3 promoter analyzed in sorted NK cells by ChIP-qPCR with TET2, H3 or H3K18 specific mAbs or IgG isotype control. Means ± SD is shown (n = 3). Each dot represents one independent experiment. d Luciferase activity in HEK293T cells co-transfected with a TET2 full-length plasmid or an empty plasmid as control, and the luciferase-reporter construct containing the region (−147 + 60) of the KIR2DL1 promoter. Means ± SD is shown (n = 4). Each dot represents one independent experiment. Nonparametric two-sided Wilcoxon matched-pairs signed rank test was used to determine statistical significance. *p = 0.0286. e Jurkat cells transfected with the KIR2DL1 promoter-luciferase reporter plasmid were treated with 500 µM of L-AA for 16 h and analyzed by detecting luminescence signal. Means ± SD is shown (n = 4). Each dot represents one independent experiment. Nonparametric two-sided Wilcoxon matched-pairs signed rank test was used to determine statistical significance. *p = 0.0286. f RRBS DNA methylation profiles of the extended KIR locus of NK cells with low or high KIR2D expression. Each graph represents the DNA methylation profile of sorted NK cells from blood samples of 5 patients; vertical bars represent the percentage of DNA methylation at the CpG position. CRE and KIR genes were highlighted in green and gray respectively. g and h IGV (Integrative Genomics Viewer) view of CpG read signals corresponding to DNA methylation in NK cells, based on the high (TET2WT/MUTKIRHIGH, in blue bars) and low (TET2MUTKIRLOW, in red bars) KIR expression. Genome profiles at the CRE region (g) and the KIR2DL1 gene (h) loci showed variation in the DNA methylation pattern between the two groups of patient NK cells. Red peaks/boxes show significant differences in the DNA methylation levels at particular CpG positions of these specific loci. Source data are provided as a Source Data file.
Therefore, we asked whether the inhibition of the enzymatic function of TET2 in NK cells could also lead to a significant reduction of the KIR2D protein expression. We treated NK cells from healthy individuals with dimethyloxalyl glycine (DMOG), an α-ketoglutarate (α-KG) analog which inhibits α-KG-dependent enzymes including TET220. In line with our results in TET2MUT MDS patients, TET2 inhibition in DMOG treatment significantly reduced the percentage of KIR2D+ NK cells (p < 0.001, Fig. 3b), suggesting a role for TET2 in modulating the KIR expression on NK cells or the KIR2D+ subset proliferation.
We then sought to determine whether TET2 had a direct impact on KIR gene expression. We performed chromatin immunoprecipitation (ChIP) followed by real-time qPCR analysis on sorted NK cells from control PBMC with a TET2-specific monoclonal antibody (mAb) and evaluated the presence of the proximal promoter regions of KIR2DL1 and KIR2DL2/3 genes. We also tested TET2 binding site onto a predicted cis-regulatory element (CRE) localized upstream of the KIR locus (chr19:54,688,519–54.702.287)21. We found that tested KIR promoters and CRE sequences were enriched in TET2-specific ChIP fragments (Fig. 3c).
To substantiate the role of TET2 in driving KIR expression, we generated a luciferase reporter vector containing 210 (−147; +63) nucleotides of the predicted KIR2DL1 promoter associated with the TET2 ChIP assay experiment. Co-transfection of this reporter with a TET2 overexpression vector into the HEK293T cell line is associated with an increase of luciferase activity increased by 2.3-fold compared to the empty vector (Fig. 3d). To corroborate this data, the TET2 expressing Jurkat lymphoid cell line was transfected with the KIR2DL1-luciferase reporter vector and then treated with ascorbic acid (L-AA), a catalyst of the TET2 hydroxylation activity of 5-methylcytosine in DNA and an already-known enhancer of the KIR locus demethylation22,23. We observed a significant increase of the luciferase signal by 2-fold in the treated condition compared to the control, demonstrating that L-AA enhanced TET2 binding and activity onto the KIR promoter sequence (Fig. 3e).
Based on these observations, we anticipated that NK cells with low KIR expression and carrying TET2 mutations could show perturbations in the methylation profile of the KIR locus. Therefore, we performed reduced representation bisulfite sequencing (RRBS) analysis on NK-cells sorted from TET2WT and TET2MUT MDS/CMML patients (n = 1 and n = 4 respectively, Fig. 3f–h) to assess their DNA methylation pattern. Three TET2MUT MDS/CMML patients were chosen based on a KIR2D expression on NK cells below 25%, and a TET2 mutation VAF superior to 40% in the cell bulk at diagnosis (TET2MUT KIRLOW patients: MUT22, MUT27, MUT33). One TET2MUT patient was chosen because of a KIR2D expression on NK cells at 90% (TET2MUT KIRHIGH patient: MUT14) and one TET2WT patient (TET2WT KIRHIGH patient: WT13) was used as controls.
The analysis of the CpG islands in the CRE sequence and the KIR2DL1 gene showed increased methylation in the three TET2MUT KIRLOW patients compared to the TET2MUT KIRHIGH and the TET2WT ones (Fig. 3g, h).
Altogether, these findings demonstrate the targeting of the KIR locus by TET2 and strongly suggest that loss of TET2 leads to the hypermethylation of the KIR locus in NK cells.
TET2 mutations lead to hypermethylation of key genes for cytotoxicity and cytokine release by NK cell
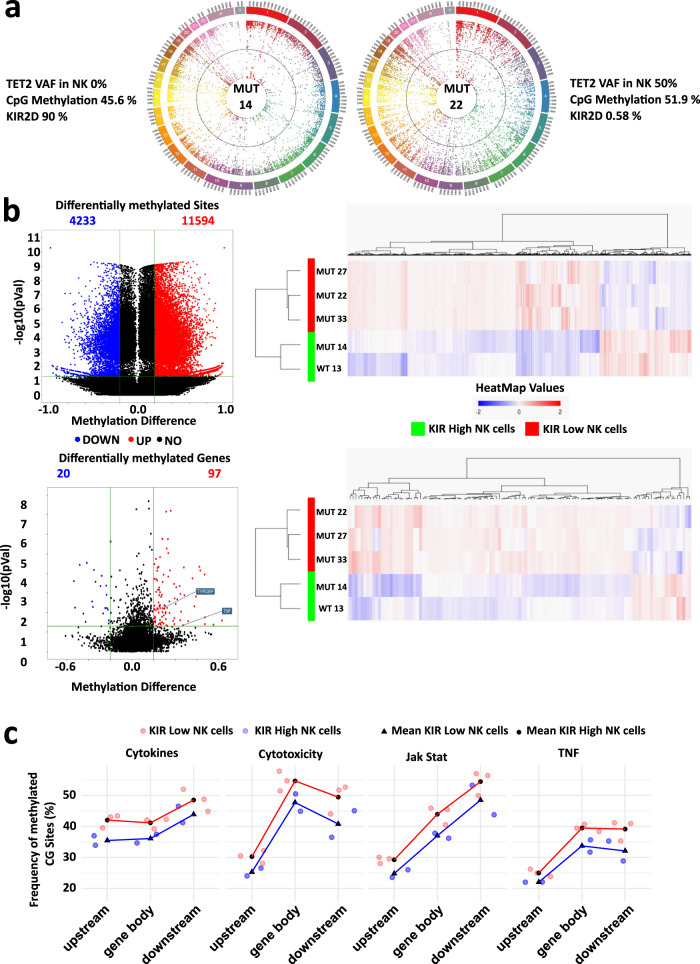
The analysis of DNA methylation in TET2-deficient mouse models showed widespread DNA hypermethylation, including in enhancer elements24. RRBS analyses of NK cells from TET2MUT KIRLOW MDS/CMML patients confirmed that genomic DNA was more methylated in NK cells with a high VAF of TET2MUT and a reduced KIR2D cell surface expression compared to TET2WT NK cells (Fig. 4a). We then asked whether TET2 mutations in NK cells could interfere with the methylation and expression of genes relevant for NK cell function. RRBS analyses of NK cells from TET2MUT KIRLOW MDS/CMML patients showed significant hypermethylation compared to KIRHIGH patients with 11,594 hypermethylated CpG sites among the 15,827 differentially methylated sites (methylation difference ≥20%, p ≤ 0.05) (Fig. 4b, upper panel, Supplementary Data 2). Among the 97 genes found hypermethylated in TET2MUTKIRLOW patients compared to KIRHIGH patients (Fig. 4b, lower panel, Supplementary Data 3), methylation of TYROBP (coding for DAP12) and TNF was predicted to significantly inhibit the NK cell-mediated cytotoxicity pathway as defined by the KEGG database (p < 0.05, KEGG pathway hsa04650)25. Reports suggested variations in methylation by TET2 of the CpG loci according to their position regarding the gene body and the respective regulatory elements24,26,27. We analyzed the percentage of 5hmC in the gene bodies and 10 kb upstream or downstream on the whole genome26. Then, the mean of methylation has been calculated for genes involved in pathways of interest for NK cell biology. Percentage of 5hmC trends up in TET2MUTKIRLOW NK cells compared to KIRHIGH samples in coding and flanking regions of genes implicated in the cytokine–cytokine receptor interactions (KEGG pathway reference hsa04060), the JAK-STAT signaling pathway (hsa04630), the TNF signaling pathway (hsa04668) and the NK cell-mediated cytotoxicity (hsa04650) (Fig. 4c), indicating a decrease of these cell functions. Interestingly, the increase of methylation affected the gene bodies as well as their flanking regions, suggesting a differential role for TET2 in NK cells as compared to myeloid cells in which TET2 mainly regulates enhancers24,27.
Fig. 4. Loss of TET2 leads to DNA hypermethylation and decreases key gene expression for NK cell function.
a Circos plots showing whole-genome CpG methylation status in patient MUT14 characterized by the absence of TET2 mutations in NK cells and a high expression of KIR2D, and in patient MUT22 with a VAF of 50% for the TET2 mutation [NM_001127208:exon11:c,4669_4672del:p.V1557fs] and a very low expression of KIR2D in NK cells. b Volcano plots and heatmaps showed the overall increase of global DNA methylation levels reported after RRBS analysis, in the TET2MUTKIRLOW (n = 3) vs. TET2WT/MUTKIRHIGH (n = 2) NK cells. Heatmaps depicted supervised clustering of the significantly modified sited genes between patients’ subgroups. Blue dots/bars show the hypomethylated CpG/genes whereas red dots/bars show the hypermethylated ones (methylation difference ≥20%, unadjusted p-value ≤ 0.05). Top panel shows the differentially methylated CpG sites. Bottom panel shows the differentially methylated genes. c GO-enrichment analysis on the differentially methylated genes in the TET2MUTKIRLOW and TET2WT/MUTKIRHIGH NK cells. Percentages of methylated CpG sites were calculated in gene bodies and 10 kb upstream or downstream of the gene of interest in TET2MUTKIRLOW and TET2WT/MUTKIRHIGH NK cells (in red and blue, respectively) and aggregated across all genes of a given KEGG pathway for each sample. Pathways of interest shown are the cytokine–cytokine receptor interactions (KEGG reference hsa04060), the JAK-STAT signaling pathway (hsa04630), the TNF signaling pathway (hsa04668), and the NK cell-mediated cytotoxicity pathway (hsa04650). Source data are provided as a Source Data file.
We then looked at the methylation of IFNG, TNF, and PRF1, three genes determinant in NK cell anti-leukemic activity28. RRBS analysis showed hypermethylated CpG islands in the three loci in the TET2MUTKIRLOW patients compared to TET2MUTKIRHIGH or the TET2WT patients, suggesting a reduced transcription of these genes (Fig. 5a).
Fig. 5. Hypermethylation of particular CpG sites represses NK cell gene expression and function.
a Methylation profiles, established after RRBS analyses, depicted with IVG at the IFNG (upper panel), TNF (middle panel) and PRF1 (lower panel) loci, were shown in NK cells from patients segregated on the high (TET2WT/MUTKIRHIGH, in blue bars) and low (TET2MUTKIRLOW, in red bars) KIR expression. Red peaks/boxes show significant differences in the DNA methylation levels of these specific loci. b The regulatory activity of specific CpG sites was analyzed in the HEK293T cell line transfected with a luciferase-reporter plasmid including methylated or non-methylated genes’ regulatory regions. Relative luciferase activities of the in vitro methylated regions were compared to their non-methylated counterpart (FC = Methylated/Putative Promoter). Means ± SD is shown (n = 4). Data were analyzed using the one-way Friedman test followed by a Dunn’s test. *p = 0.0433. c Quantification of the KIR2DL1, TNF, and IFNG transcripts by RT-qPCR on KIR2D− NK cells sorted from TET2/IDHWT (n = 11, in blue) and TET2MUT (n = 10, in red) patients. Medians and interquartile are shown. Statistics were calculated with the two-sided Mann–Whitney test, **p = 0.0036. d Fold changes of NK cells killing activity against the NK-sensitive cell line K562 previously treated for 5 days with 1 µM DMOG (n = 10). Each dot represents one independent experiment. DMSO alone was used as a control. Means ± SD is shown. Data were analyzed using the one-way Friedman test followed by a Dunn’s test. *p = 0.0417. Source data are provided as a Source Data file.
To evaluate the functional relevance of DNA methylation in controlling the regulatory regions of these genes, a luciferase reporter assay was performed in the HEK293T cell line (Fig. 5b). Regions of interest, showing differences in their methylation profile between TET2MUTKIRLOW and TET2MUTKIRHIGH NK cells, were cloned into a CpG-free vector upstream of the luciferase gene, without minimal promoter to determine the functional activity of the promoter. The constructs were then methylated in vitro specifically onto the insert sequences. Methylation resulted in significantly decreased luciferase activity compared to the unmodified construct, suggesting that the promoter function of the region of interest is dampened upon DNA methylation (Fig. 5b). In parallel, the decreased expression of TNF and INFG in presence of TET2 mutations was confirmed by qRT-PCR in KIR2D− NK cell sorted from TET2/IDHWT and TET2MUT patients (Fig. 5c).
Altogether, these results showed that key molecules for NK-cell function were under the control of the TET2 demethylation pathway and that their transcription was reduced in TET2MUT NK cells. Consequently, we asked whether TET2 inhibition could impair NK cell function. NK cells from HD were treated in vitro with DMOG and then co-cultured with the K562 cell line. As expected, K562 killing by DMOG-treated NK-cells was significantly reduced demonstrating that the NK-cell cytotoxicity pathway was, at least in part, under the control of TET2 (p < 0.05, Fig. 5d).
Hypomethylating agents rescue KIR expression on MDS NK cells
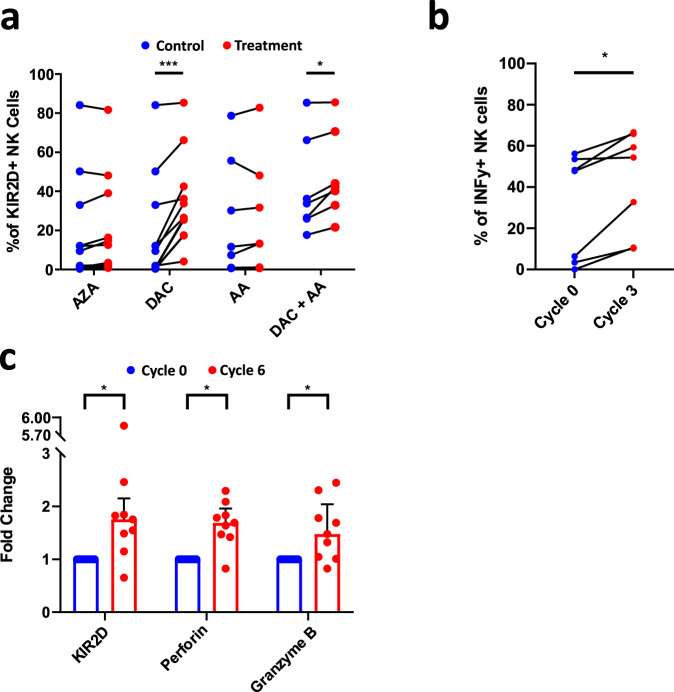
To evaluate whether demethylation could restore KIR expression on NK cells, PBMC from TET2MUT patients were cultured in vitro in presence of IL-2 and with or without AZA or DAC for 5 days. Then, KIR2D expression on NK cells was assessed by flow cytometry (Fig. 6a). Whereas AZA showed only a trend, DAC significantly restored the KIR expression on treated NK cells (p = 0.07 and p < 0.001, respectively). Further, l-AA was used either alone or in combination with DAC to treat PBMC of TET2MUT MDS/CMML patients. l-AA associated with DAC significantly increased KIR expression in NK cells compared with DAC alone (p < 0.05). Of note, NK cells from HD and TET2WT patients also increased KIR expression in response to HMA with or without AA (Supplementary Fig. 8).
Fig. 6. Hypomethylating agents normalize the NK cell phenotype of MDS patients.
a Evaluation of KIR2D surface expression on NK cells of TET2/IDHMUT patients after treatment with azacitidine (AZA, n = 12), decitabine (DAC, n = 12), acid ascorbic (AA, n = 7), and DAC+AA (n = 7). Nonparametric two-sided Wilcoxon matched-pairs signed rank test was used to determine statistical significance, DAC: ***p = 0.001, DAC+AA: *p = 0.0156. b NK cells were isolated from patients’ PBMC before and after 3 cycles of treatment with AZA, and cultured overnight at 100U/ml of IL-2. Subsequently, cells were cultured with PMA-Ionomycin for 6 h. The frequency of responding cells in terms of IFN-γ was assessed by flow cytometry (n = 7). c KIR2D, perforin, and granzyme B expression were measured by flow cytometry in the blood of MDS patients (n = 9) before (in blue) and after (in red) 6 cycles of AZA treatment. Nonparametric two-sided Wilcoxon matched-pairs signed rank test was used to determine statistical significance, KIR2D: *p = 0.0195, Perforin: *p = 0.0273, Granzyme B: *p = 0.0273. Data are presented as medians and interquartile ranges. Source data are provided as a Source Data file.
To confirm in vivo our observations, NK cells from MDS patients recruited in the clinical trial NCT02985190 were analyzed for their capacity to produce IFN-γ and to restore their phenotype, before and after 3 or 6 cycles of treatment with AZA. NCT02985190 clinical trial is a phase II study of efficacy and tolerance of AZA in MDS patients with concomitant systemic auto-immune and inflammatory disorders (SAID), for which the primary outcome measure was the overall response rate of MDS and SAID29. Functional experiments showed an increase in the production of INF-γ by NK cells stimulated in vitro by a mixture of PMA and ionomycin in patients after 3 cycles of treatment with AZA (n = 6, Fig. 6b). Moreover, KIR2D, perforin, and granzyme B expressions, quantified by flow-cytometry, were significantly increased in this group of patients after 6 cycles of treatment (n = 9, Fig. 6c). Importantly, despite the absence of significance due to the small number of patients, this effect of AZA on NK cell phenotype and function was similar in TET2WT and TET2MUT patients.
Taken as a whole, these findings showed that demethylation mediated by TET2 is pivotal in regulating NK cell biology, including the expression of KIR and functional proteins, and, more importantly, that treatment by HMA could normalize the hypermethylated profile harbored by NK cells in MDS patients.
Discussion
In this work, we described the epigenetic, phenotypic, and functional consequences of TET2 mutations for NK cells in MDS patients. We could demonstrate that mutations observed in MDS cancer cells, including in TET2 and IDH1/2 genes, are also generally observed in NK cells. Consequently, TET2 mutations in NK cells are associated with perturbations in methylation of the genomic DNA inducing a reduced expression of KIRs, a locus on which TET2 directly binds, and of other key molecules for NK cell function including perforin and TNF-α. The reduced KIR expression and cytotoxicity of NK cells of healthy donors after TET2 inhibition confirmed its requirement for an efficient physiological NK cell function. Finally, NK cell phenotype can be restored in vitro and in vivo after treatment with ascorbic acid and HMA.
The fact that NK cell defects observed in MDS can be partly due to the oncogenic mutations at the origin of the disease could suggest that both events (i.e. mutations in MDS cells and NK cells) may synergize to sustain the emergence of myeloid malignancies. Abnormalities in NK cell phenotype and function could reduce the antitumor immuno-surveillance efficacy and allow the survival of potential pre-leukemic progenitors. Notably, NK-cell perturbations in MDS patients including reduction of activating NK receptors, such as NKp30, DNAM-1, and NKG2D, and impaired cytotoxicity, have already been reported12,15,30. KIR genotype has also been associated with clinical outcomes31. Our data suggest that these observations should be reinterpreted based on patient mutation profiles, especially for mutations associated with TET2 or IDH1/2 genes. In line with this comment, dissecting the specific role of TET2 in NK cells by contrast to other mutated genes commonly found in MDS or clonal hematopoiesis will require further investigations, which could be decisive in our comprehension of the disease and its treatments. Whether NK-cell function could be associated with a particular mutation landscape may have important consequences in developing NK-cell-based immunotherapeutic intervention. In that context, using autologous NK cells or attempting to rescue NK cell function in MDS patients may be impeded. Combining such therapeutic strategies with the use of HMA could provide a relevant option.
Loss-of-function somatic mutations in TET2 are strongly associated with hematological malignancies, including myeloid, T- and B-cell malignancies and chronic lymphoproliferative disorders of NK cells (CLPD-NK)32–34. Germline mutations in TET2 have been also associated with lymphoma35,36. Here, we found that TET2 mutation in NK cells from MDS patients, with or without additional mutated genes, does not seem to affect NK cells from the oncogenic point of view (i.e. NK cells were not leukemic) but alters their phenotype and function. This raises the question of the requirement of other mutated genes, in addition to TET2, in order to induce malignant hemopathies and how these mutation profiles can be specific to lymphoid or myeloid malignancies.
Intriguingly, we noticed that TET2 mutations in NK cells trended to increase methylation together on the gene body and its flanking regions, at least for genes involved in NK cell function. TET2 has been reported as preferentially targeting regulatory enhancers in hematopoietic and myeloid cells, notably to facilitate the recruitment of transcription factors24,27. However, gene bodies and other genomic regions can be also, although in a less efficient manner, targeted by TET224. In addition, our results could suggest that key genes for NK cell function, including KIR and PRF1, are regulated in a coordinated manner by TET2 and more generally DNA methylation. If it is confirmed, this hypothesis would point out TET2 as a master regulator in NK cell development and education. It remains to be investigated whether and how genomic DNA methylation by TET2 is differentially regulated in NK cells, compared to myeloid subsets, due to differences in chromatin modification and accessibility or in the timing of TET2 expression during NK-cell lymphopoiesis.
From the mechanistic point of view, our results are reminiscent of the work by Wu et al. describing the role of the ascorbic acid in promoting KIR locus demethylation through the mobilization of Runx3, TET2, and TET322. Interestingly, it has been recently shown that TET2KO HSC treated with ascorbic acid could increase the 5hmC levels suggesting a role for TET proteins other than TET2 in this rescuing26. Herein, we showed that the addition of ascorbic acid to NK cells allows the restoration of KIR expression. Altogether, these data support the hypothesis that TET3 could compensate for TET2 mutations in NK cells after treatment with ascorbic acid.
Clonal hematopoiesis (CH) is frequently presented as a precursor state for hematological malignancies, upstream of a continuum including MDS and then AML37. Arends et al. recently demonstrated that TET2 mutations observed in individuals with CH could be found not only in myeloid lineage but also in B- and NK-cell38. Our results support the finding that CH infuses into the complete tree of hematopoiesis, including lymphopoiesis, and could also participate in reduced NK cell immunity in the elderly. It remains to discover the mechanisms by which TET2 mutations alter the production of new lymphocytes in the elderly without significantly reducing the lymphocyte number, and with which consequences for the immune response. High-throughput experimental strategies, including multi-omics approaches combining RNAseq and ATACseq at the single cell level, applied not only to patients’ samples but also to TET2-edited HSC26 or iPSCs36, may help to solve this question.
Altogether, our findings showed that TET2 is a key epigenetic regulator of NK cells and that TET2 mutations associated with MDS can alter their phenotype and function. In that context, HMA treatments could normalize the hypermethylated profile harbored by NK cells in MDS patients and may participate in the emergence of NK cell-mediated anti-tumor immunosurveillance.
Methods
Patient cohorts and samples
The first cohort was composed of 20 sternal bone marrow (BM) aspiration and 58 peripheral blood mononuclear cell (PBMC) samples that were collected from 63 MDS/CMML patients. Patients’ samples were obtained with written informed consent in the Hematology Departments of the Saint Louis Hospital, Paris, France (Supplementary Data 1 and Table 7). MDS and CMML diseases were defined according to the World Health Organization 2016 criteria39 and classified according to the Revised International Prognostic Scoring System (IPSS-R)40. Low-Risk (LR) disease was defined as IPSS-R very-low, low or intermediate whereas High-Risk (HR) disease was designated as IPSS-R high and very-high. Disease evolution was assessed according to the 2006 International Working Group criteria41. Among the 63 patients recruited, a few received treatments consisting of either Darbepoetin (EPO, n = 4) or Eltrombopag (TPO, n = 1) without obvious effect on immune cells, or the immunomodulating drugs Lenalidomide (n = 4) or the glucocorticoid Prednisone (n = 4). The second cohort was composed of MDS patients recruited in clinical trial #NCT02985190 (sponsored by Groupe Francophone des Myélodysplasies [GFM]) and repeatedly treated with azacitidine at 75 mg/m2/J subcutaneously daily for 7 days every 4 weeks29 (Supplementary Table 8). In addition, PBMC samples from 23 healthy individuals (HD) were collected through the EFS (Etablissement Français du Sang) (median age = 46 [27–66]). The study was approved by the Ethical Board Ile-de-France X and conducted in accordance with Helsinki’s declaration. Recruited patients gave their written informed consent for participating.
PBMCs were purified from BM and blood samples by using a ficoll-based gradient density method13 and stored in liquid nitrogen to be further analyzed.
Patient KIR genotyping
Generic KIR typing was performed using a KIR multiplex PCR-SSP method as previously described42. The presence or absence of KIR2DL1, 2DL2, 2DL3, 2DL5, 3DL1, 2DS1, 2DS2, 2DS3, 2DS4/1D, 2DS5, and 3DS1 genes were assigned. KIR genotypes were determined based on the presence or the absence of activating KIR. Thus, an AA KIR haplotype was defined by the presence of only KIR2DS4 or its deleted variant (1D) as activating KIR gene, and a B+ (AB, BB) KIR haplotype by the presence of several activating KIR genes. Centromeric (AA, AB, BB) and telomeric (AA, AB, BB) KIR motifs were defined considering KIR2DL2/3/S2 and KIR3DL1/S1/2DS1/2DS4 genes for centromeric and telomeric motifs, respectively43 (Supplementary Table 3).
Cell lines
HEK293T and Jurkat cell lines (ATCC) were maintained either with DMEM or RPMI1640 medium containing 10% of FBS, penicillin/streptomycin antibiotics, and l-glutamine.
For the measurement of target lysis, K562 cells were first cultured overnight in RPMI1640 without phenol red (Gibco) enriched with 10% FBS, non-essential amino acids (NEAA, Gibco), l-glutamine (Life Technologies) and Sodium Pyruvate (Eurobio). Prior killing assay, K562 cells were diluted at 1 × 106/mL in a culture medium supplemented with 2.5 mM Probenecid (Invitrogen) and stained with calcein-AM (Invitrogen) for 30 min. Then, the excess calcein was washed out.
For in vitro cell treatment, PBMCs were resuspended in RPMI 1640 supplemented with 10% human serum (EFS, Etablissement Français du Sang), 1% Penicillin–Streptomycin (Dutscher), l-glutamine (Life Technologies), sodium pyruvate (EuroBio) and HEPES (EuroBio).
Flow cytometry analysis
Frozen sternal BM aspiration and PBMCs were stained for 30 min on ice13, with the fixable viability dye eFluor 506 and monoclonal antibodies (mAb) listed in Supplementary Table 9. Of note, mAb used in this work to detect KIR molecules on the cell surface was able to identify KIR2DL1, KIR2DL2/DL3, KIR2DL4, KIR3DL1/DL2, KIR3DL1/DS1, KIR2DL5, and KIR2DS4 molecules. Cells were fixed in PBS with 2% of paraformaldehyde (PFA), acquired on a BD Fortessa X20 (flow cytometry core facility of Saint Louis Research Institute, Paris, France) (Supplementary Fig. 9). For intracellular staining, Foxp3 Transcription factor buffer set (eBioscence 00-5523-00) was used and data were acquired on a BD Canto II or Cytek Aurora flow cytometer. Data were analyzed using FlowJo v10.7 software or Cytek SpectroFlo v3.0.3.
NK and T-cells isolation for mutational analysis and reduced representation bisulfite sequencing (RRBS)
After thawing, PBMCs were first labeled in PBS for 30 min at 4 °C with the fixable viability dye eFluor 506 (eBioscience) and then stained with antibodies specific for surface markers to distinguish NK cell subsets (at the whole or based on the surface expression of KIR2D and KIR3DL1/DL2 molecules) and T-cells, and directly sorted using BD Aria III system (Supplementary Table 9 and Supplementary Fig. 10). Dry pellets have been stored after cells sorting for DNA extraction. DNA was purified according to the manufacturer (DNeasy KIT, Qiagen) and quantified by Qubit (Thermo Fisher) for NGS and RRBS studies. Of note, clinical evaluation of MDS mutations was performed in the hematology laboratory, in St. Louis Hospital, Paris, on WMC obtained using gradient density separation on either BM or blood samples.
A panel of 80 genes commonly analyzed in myeloid malignancies (Supplementary Table 4) was examined using next-generation sequencing on an Illumina platform (Illumina, San Diego, CA, USA). Libraries were prepared from 200 ng of DNA using a custom probes panel for the capture of all coding sequences (SureSelectXT Target Enrichment System, Agilent, Santa Clara, CA, USA). Sequencing data were analyzed for variant calling using an in-house pipeline. High-probability pathogenic mutations were retained based on the available databases for SNPs, somatic mutations in cancer, prediction algorithms, and frequencies of variant read. Overall, a mean coverage of 851 reads/amplicon was achieved allowing reliable detection of low-burden mutations. Lolliplot has been realized using the St. Jude protein paint software available online at: https://proteinpaint.stjude.org/ (Fig. 3a)44. Major domains of TET2 protein were determined based on the work by Hu et al. 45. The mutational landscape was established using cBioPortal Cancer Genomics Portal OncoPrinter software available online at (https://www.cbioportal.org/) (Supplementary Fig. 6).
RRBS was performed by Diagenode (Seraing, Belgium) on dry samples of NK- and T-cells sorted from five MDS patients according to the manufacturer’s procedure. RRBS data are available on the Gene Expression Omnibus platform under the accession code GSE183020. 100 ng of purified genomic DNA was used, including pre-RRBS quality control, enzymatic digestion, and bisulfite conversion. Sequencing was performed in paired-end mode 50 bp (PE50), quality control of sequencing reads was performed using FastQC and reads alignment against human reference genome hg38/GRCh38 using Bismark v0.20.0 (available at https://www.bioinformatics.babraham.ac.uk/projects/bismark/) according to Diagenode protocol. Differential gene and site methylation analyses were performed by Genoplice Technology (Paris Biotech Santé, Paris, France). To assess the frequency of methylated CpG sites across all genes of a pathway of interest, gene lists were extracted from the KEGG pathway website25. The position of each CpG site was calculated relative to the transcript start and end positions as follows:
Considering 10 kb upstream and downstream sites. Next, the frequency of methylation was calculated and aggregated across all genes of a given pathway for each sample.
Of note, all sequencing data were analyzed using the human reference genome hg38/GRCh38.
Chromatin Immuno-Precipitation (ChIP)-qPCR assay
ChIP assay was performed with the Active Motif magnetic kit following the manufactory protocol. Briefly, after sonication, the sheared chromatin was incubated overnight with TET2 (Diagenode, C15410255), control H3 (Abcam, AB1791), and H3K18 (Abcam, AB1191) specific mAb and the isotype control (Diagenode, C15410206). ChIP-enriched DNA was analyzed by qPCR with SYBR Green Master Mix (Thermofisher). Primers are listed in Supplementary Table 10.
Transfection and Luciferase assay
HEK293T cell lines (ATCC) were transfected by polyethyleneimine (PEI, Sigma) following previous publication46.
The plasmid pcDNA3-TET2 overexpressing the full-length mouse Tet2 gene as the empty pcDNA3 vector was obtained from Addgene (Plasmid #60939 and #45346). All constructs have been verified by DNA sequencing.
One region of the predicted KIR2DL1 promoter (KIRprom −147 + 60, Supplementary Fig. 11) was cloned in the pT109-tkLUC plasmid. 0.6 µg promoter vector together with the 2.7 µg TET2 plasmids were co-transfected in HEK293T cells with 0.06 µg of pRL-CMV Vector (#E2261 Promega) in presence of PEI solution.
In parallel, Jurkat cells (ATCC) were transfected with 0.26 µg of the KIR promoter plasmid and 0.026 µg of a pRL-CMV vector by Nucleofactor Kit V (Lonza). Jurkat transfected cells were then cultured at 1 × 106/mL and treated with 500 µg of l-ascorbic acid (l-AA, Sigma) for 16 h before analysis47. The luciferase activity was measured after 24 h according to the manufacturer’s instructions (Dual-Luciferase Reporter Assay, Promega).
To investigate the role of the methylation in controlling the functional NK role, putative promoter regions, differentially methylated between KIRHIGH and KIRLOW, were cloned into pCpGfree basic vector (Invivogen) for testing promoter activity. To determine the effect of methylation on the activity of the region, in vitro methylation of the constructs was performed with M.SssI (NEB). 0.6 µg promoter vector, “native” (unmethylated) or in vitro methylated, were co-transfected with 0.06 µg of pRL-CMV Vector (#E2261 Promega) in presence of PEI solution. After 24 h the Lucia and Firefly read-out was generated with a CLARIOstarPlus (BMG LABTECH).
Microfluidic multiplex qPCR
Multiplex qPCR analysis (Biomark, Fluidigm) was performed on 100 cells. NK cells as the whole and KIR+ (KIR2D or KIR3DL1/DS1 positive) or KIR− (KIR2D or KIR3DL1/DS1 negative) NK cell subsets were sorted into 5 µL of reverse transcription/pre-amplification mix, prior to multiplex qRT-PCR following manufacturer’s protocol48. Briefly, the mix contained 2X Reaction mix and Superscript III (CellDirect One-Step qRT–PCR kit, Invitrogen) and 0.2X Taqman assay-specific probes (Life technologies). Targeted cDNA pre-amplification was performed for 19 cycles and the pre-amplified product was diluted 1:5 in TE buffer before processing with Dynamic Array protocol according to the manufacturer’s instructions (Fluidigm). All the probes used for this analysis are described in Supplementary Table 11. Mean expressions for RPL27 and GAPDH housekeeping genes were used for signal normalization.
In vitro cell treatment and functional assay
PBMC were treated in cell culture medium (i.e., RPMI 1640 supplemented with 10% fetal bovine serum) with 0.5 or 1 µM of AZA, DAC, dimethyloxaloylglycine (DMOG) (Sigma Aldrich), alone or in combination with 125 µM Ascorbic Acid. Interleukin-2 (IL-2) (Miltenyi Biotec) was added to a final concentration of 100 UI/mL. DMSO condition was used as a control. The treatments were maintained for 5 days with a half change of medium after 48 h (IL2 ± drugs or DMSO). Then, cells were collected and NK cells were analyzed for their phenotype (Supplementary Table 9 and Supplementary Fig. 9).
After treatment with DMOG, PBMCs were collected, enumerated, and co-cultured with calcein-labeled K562 cells in 96-well U bottom plates in a medium supplemented with Probenecid at 10:1 effector:target ratio. The killing was quantified after 4 h of incubation at 37 °C by measuring calcein-release into the supernatant. The specific killing was calculated as below: (Measured fluorescence of K562 + PBMC well—Spontaneous Fluorescence)/(Maximum fluorescence—Spontaneous fluorescence)*100.
For the ex vivo IFN-γ quantification, enriched NK cells from MDS patients enrolled in the NCT02985190 clinical trial29 were cultured with PMA-Ionomycin (SIGMA, 50 and 500 ng/mL, respectively) for 5 h at 37 °C. Brefeldin A (Sigma) was added at a final concentration of 10 µg/ml after 1 h of incubation. The percentage of IFN-γ positive cells was estimated by flow cytometry in the CD3−CD56+ NK cells. Spontaneous release was detected in the absence of target cells.
Statistical analysis
Data are shown as median ± interquartile range, if not otherwise specified. Cytometry analyses were extracted from FlowJo v10.7 software and analyzed with Graph Pad Prism v8.0 software. Unpaired statistical analyses were calculated with the nonparametric Mann–Whitney test. Paired statistics analyses were calculated with the nonparametric Wilcoxon matched-pairs signed rank test. The Kolmogorov–Smirnov D test was used to calculate differences between treated/transfected and control situations. Pearson coefficient (r) was calculated to evaluate significant correlation. The receiver operation characteristic (ROC) curve was realized based on TET2 mutation status and percentage of KIR2D + NK cells. The area under the curve, confidence interval, p-value, and likelihood ratio were calculated. A Friedman test followed by a Dunn’s test was used to calculate differences in experiments with multiple conditions. p < 0.05 was considered significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Supplementary information
Description of Additional Supplementary Files
Acknowledgements
This study was supported by a grant from the French Ministry of Health and the French National Cancer Institute (#PRT-K2017-109), the Cancéropôle Ile-de-France, the Association Laurette Fugain (#ALF 2016-07), the Association Force Hémato (Call for projects 2017), the Ligue contre le Cancer (Ile-de-France committee), the Association Saint-Louis pour la Recherche sur les Leucémies, and the Association pour la Recherche en Immunologie Médicale et Expérimentale (ARIME). The authors want to acknowledge the support of the “Groupe Francophone des Myelodysplasies (GFM)” in the management of the NCT02985190 clinical trial. The authors thank Niclas Setterblad, Christelle Doliger, Sophie Duchez from the IRSL’s Technological Platform, Nolwenn Legrand from the EFS laboratory in Nantes, as well as Pierre De La Grange and Wail Zeitouni from Genosplice for their technical and analytic support.
Source data
Author contributions
M.B., V.B., and N.D. designed, analyzed, and interpreted all experiments, and wrote the manuscript. M.B., V.B., F.G., and E.L. performed experiments and analyzed the data. L.-P.Z. managed the collection of the patients’ samples and clinical information. B.S. analyzed the RRBS data. G.H. and J.V. performed preliminary feasibility experiments. F.R.-L. provided reagents and analyzed data. K.G. and C.R. performed KIR genotyping. L.L., R.K., and E.C. performed next-generation sequencing and analyzed mutation profile data. L.-P.Z., M.S., P.F., and L.A. recruited the patients for the study. A.M., O.F., and P.F. supervised the NCT02985190 clinical trial. A.C., M.E., K.B., A.T., P.F., and L.A. contributed to the scientific orientation of the study and critically reviewed the manuscript. L.A. designed the patients’ cohort suitable for the study. N.D. supervised the study.
Peer review
Peer review information
Nature Communications thanks Ivo Touw, Jörn Walter and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Data availability
Reduced representation bisulfite sequencing (RRBS) data that support the findings of this study have been deposited in the Gene Expression Omnibus data repository with the accession number GSE183020. The in vitro data generated in this study are available in the Supplementary information. Sequences of oligonucleotides used for the KIR genotyping (PCR-SSP) are limited to non-commercial and research use and are available upon request to Dr. K. Gendzekhadze (kgendzek@coh.org), City Of Hope Medical Foundation, 1500 Duarte Rd, Duarte, CA 91010. Source data are provided with this paper as a Source Data file. Source data are provided with this paper.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Maxime Boy, Valeria Bisio.
Supplementary information
The online version contains supplementary material available at 10.1038/s41467-023-36193-w.
References
- 1.Ades L, Itzykson R, Fenaux P. Myelodysplastic syndromes. Lancet. 2014;383:2239–2252. doi: 10.1016/S0140-6736(13)61901-7. [DOI] [PubMed] [Google Scholar]
- 2.Gurion R, et al. 5-azacitidine prolongs overall survival in patients with myelodysplastic syndrome—a systematic review and meta-analysis. Haematologica. 2010;95:303–310. doi: 10.3324/haematol.2009.010611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kantarjian H, et al. Decitabine improves patient outcomes in myelodysplastic syndromes: results of a phase III randomized study. Cancer. 2006;106:1794–1803. doi: 10.1002/cncr.21792. [DOI] [PubMed] [Google Scholar]
- 4.Kunimoto H, Nakajima H. Epigenetic dysregulation of hematopoietic stem cells and preleukemic state. Int. J. Hematol. 2017;106:34–44. doi: 10.1007/s12185-017-2257-6. [DOI] [PubMed] [Google Scholar]
- 5.Figueroa ME, et al. Leukemic IDH1 and IDH2 mutations result in a hypermethylation phenotype, disrupt TET2 function, and impair hematopoietic differentiation. Cancer Cell. 2010;18:553–567. doi: 10.1016/j.ccr.2010.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yamashita M, Dellorusso PV, Olson OC, Passegue E. Dysregulated haematopoietic stem cell behaviour in myeloid leukaemogenesis. Nat. Rev. Cancer. 2020;20:365–382. doi: 10.1038/s41568-020-0260-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Itzykson R, et al. Impact of TET2 mutations on response rate to azacitidine in myelodysplastic syndromes and low blast count acute myeloid leukemias. Leukemia. 2011;25:1147–1152. doi: 10.1038/leu.2011.71. [DOI] [PubMed] [Google Scholar]
- 8.Costantini B, et al. The effects of 5-azacytidine on the function and number of regulatory T cells and T-effectors in myelodysplastic syndrome. Haematologica. 2013;98:1196–1205. doi: 10.3324/haematol.2012.074823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang J, Xiao Z. Mesenchymal stem cells in pathogenesis of myelodysplastic syndromes. Stem Cell Investig. 2014;1:16. doi: 10.3978/j.issn.2306-9759.2014.08.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cooper MA, Fehniger TA, Caligiuri MA. The biology of human natural killer-cell subsets. Trends Immunol. 2001;22:633–640. doi: 10.1016/s1471-4906(01)02060-9. [DOI] [PubMed] [Google Scholar]
- 11.Morvan MG, Lanier LL. NK cells and cancer: you can teach innate cells new tricks. Nat. Rev. Cancer. 2016;16:7–19. doi: 10.1038/nrc.2015.5. [DOI] [PubMed] [Google Scholar]
- 12.Epling-Burnette PK, et al. Reduced natural killer (NK) function associated with high-risk myelodysplastic syndrome (MDS) and reduced expression of activating NK receptors. Blood. 2007;109:4816–4824. doi: 10.1182/blood-2006-07-035519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Khaznadar Z, et al. Defective NK cells in acute myeloid leukemia patients at diagnosis are associated with blast transcriptional signatures of immune evasion. J. Immunol. 2015;195:2580–2590. doi: 10.4049/jimmunol.1500262. [DOI] [PubMed] [Google Scholar]
- 14.Costello RT, et al. Defective expression and function of natural killer cell-triggering receptors in patients with acute myeloid leukemia. Blood. 2002;99:3661–3667. doi: 10.1182/blood.v99.10.3661. [DOI] [PubMed] [Google Scholar]
- 15.Kiladjian JJ, et al. Cytolytic function and survival of natural killer cells are severely altered in myelodysplastic syndromes. Leukemia. 2006;20:463–470. doi: 10.1038/sj.leu.2404080. [DOI] [PubMed] [Google Scholar]
- 16.Bjorkstrom NK, et al. Expression patterns of NKG2A, KIR, and CD57 define a process of CD56dim NK-cell differentiation uncoupled from NK-cell education. Blood. 2010;116:3853–3864. doi: 10.1182/blood-2010-04-281675. [DOI] [PubMed] [Google Scholar]
- 17.Amorim LM, et al. High-resolution characterization of KIR genes in a large North American Cohort reveals novel details of structural and sequence diversity. Front. Immunol. 2021;12:674778. doi: 10.3389/fimmu.2021.674778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tate JG, et al. COSMIC: the Catalogue Of Somatic Mutations In Cancer. Nucleic Acids Res. 2019;47:D941–D947. doi: 10.1093/nar/gky1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shihab HA, et al. An integrative approach to predicting the functional effects of non-coding and coding sequence variation. Bioinformatics. 2015;31:1536–1543. doi: 10.1093/bioinformatics/btv009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang D, An X, Li Z, Zhang S. Role of gene promoter methylation regulated by TETs and DNMTs in the overexpression of HLA-G in MCF-7 cells. Exp. Ther. Med. 2019;17:4709–4714. doi: 10.3892/etm.2019.7481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature. 2012;489:57–74. doi: 10.1038/nature11247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wu CY, et al. Ascorbic acid promotes KIR demethylation during early NK cell differentiation. J. Immunol. 2020;205:1513–1523. doi: 10.4049/jimmunol.2000212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Minor EA, Court BL, Young JI, Wang G. Ascorbate induces ten-eleven translocation (Tet) methylcytosine dioxygenase-mediated generation of 5-hydroxymethylcytosine. J. Biol. Chem. 2013;288:13669–13674. doi: 10.1074/jbc.C113.464800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rasmussen KD, et al. Loss of TET2 in hematopoietic cells leads to DNA hypermethylation of active enhancers and induction of leukemogenesis. Genes Dev. 2015;29:910–922. doi: 10.1101/gad.260174.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kanehisa M, Goto S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000;28:27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nakauchi Y, et al. The cell type-specific 5hmC landscape and dynamics of healthy human hematopoiesis and TET2-mutant preleukemia. Blood Cancer Discov. 2022;3:346–367. doi: 10.1158/2643-3230.BCD-21-0143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tulstrup M, et al. TET2 mutations are associated with hypermethylation at key regulatory enhancers in normal and malignant hematopoiesis. Nat. Commun. 2021;12:6061. doi: 10.1038/s41467-021-26093-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dulphy N, et al. Underground adaptation to a hostile environment: acute myeloid leukemia vs. natural killer cells. Front. Immunol. 2016;7:94. doi: 10.3389/fimmu.2016.00094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mekinian A, et al. A Phase II prospective trial of azacitidine in steroid-dependent or refractory systemic autoimmune/inflammatory disorders and VEXAS syndrome associated with MDS and CMML. Leukemia. 2022;36:2739–2742. doi: 10.1038/s41375-022-01698-8. [DOI] [PubMed] [Google Scholar]
- 30.Carlsten M, et al. Reduced DNAM-1 expression on bone marrow NK cells associated with impaired killing of CD34+ blasts in myelodysplastic syndrome. Leukemia. 2010;24:1607–1616. doi: 10.1038/leu.2010.149. [DOI] [PubMed] [Google Scholar]
- 31.Stringaris K, et al. KIR gene haplotype: an independent predictor of clinical outcome in MDS patients. Blood. 2016;128:2819–2823. doi: 10.1182/blood-2016-05-713099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lio CJ, Yuita H, Rao A. Dysregulation of the TET family of epigenetic regulators in lymphoid and myeloid malignancies. Blood. 2019;134:1487–1497. doi: 10.1182/blood.2019791475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pastoret C, et al. Linking the KIR phenotype with STAT3 and TET2 mutations to identify chronic lymphoproliferative disorders of NK cells. Blood. 2021;137:3237–3250. doi: 10.1182/blood.2020006721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Olson TL, et al. Frequent somatic TET2 mutations in chronic NK-LGL leukemia with distinct patterns of cytopenias. Blood. 2021;138:662–673. doi: 10.1182/blood.2020005831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kaasinen E, et al. Impact of constitutional TET2 haploinsufficiency on molecular and clinical phenotype in humans. Nat. Commun. 2019;10:1252. doi: 10.1038/s41467-019-09198-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stremenova Spegarova J, et al. Germline TET2 loss of function causes childhood immunodeficiency and lymphoma. Blood. 2020;136:1055–1066. doi: 10.1182/blood.2020005844. [DOI] [PubMed] [Google Scholar]
- 37.Steensma DP, et al. Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. Blood. 2015;126:9–16. doi: 10.1182/blood-2015-03-631747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Arends CM, et al. Hematopoietic lineage distribution and evolutionary dynamics of clonal hematopoiesis. Leukemia. 2018;32:1908–1919. doi: 10.1038/s41375-018-0047-7. [DOI] [PubMed] [Google Scholar]
- 39.Hong M, He G. The 2016 revision to the World Health Organization Classification of myelodysplastic syndromes. J. Transl. Int. Med. 2017;5:139–143. doi: 10.1515/jtim-2017-0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Greenberg PL, et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood. 2012;120:2454–2465. doi: 10.1182/blood-2012-03-420489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cheson BD, et al. Clinical application and proposal for modification of the International Working Group (IWG) response criteria in myelodysplasia. Blood. 2006;108:419–425. doi: 10.1182/blood-2005-10-4149. [DOI] [PubMed] [Google Scholar]
- 42.Sun JY, et al. Development of a multiplex PCR-SSP method for Killer-cell immunoglobulin-like receptor genotyping. Tissue Antigens. 2004;64:462–468. doi: 10.1111/j.1399-0039.2004.00303.x. [DOI] [PubMed] [Google Scholar]
- 43.Cooley S, et al. Donor selection for natural killer cell receptor genes leads to superior survival after unrelated transplantation for acute myelogenous leukemia. Blood. 2010;116:2411–2419. doi: 10.1182/blood-2010-05-283051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhou X, et al. Exploring genomic alteration in pediatric cancer using ProteinPaint. Nat. Genet. 2016;48:4–6. doi: 10.1038/ng.3466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hu L, et al. Crystal structure of TET2–DNA complex: insight into TET-mediated 5mC oxidation. Cell. 2013;155:1545–1555. doi: 10.1016/j.cell.2013.11.020. [DOI] [PubMed] [Google Scholar]
- 46.Longo PA, Kavran JM, Kim MS, Leahy DJ. Single cell cloning of a stable mammalian cell line. Methods Enzymol. 2014;536:165–172. doi: 10.1016/B978-0-12-420070-8.00014-3. [DOI] [PubMed] [Google Scholar]
- 47.Cimmino L, et al. Restoration of TET2 function blocks aberrant self-renewal and leukemia progression. Cell. 2017;170:1079–1095.e1020. doi: 10.1016/j.cell.2017.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Thai LH, et al. BAFF and CD4(+) T cells are major survival factors for long-lived splenic plasma cells in a B-cell-depletion context. Blood. 2018;131:1545–1555. doi: 10.1182/blood-2017-06-789578. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Description of Additional Supplementary Files
Data Availability Statement
Reduced representation bisulfite sequencing (RRBS) data that support the findings of this study have been deposited in the Gene Expression Omnibus data repository with the accession number GSE183020. The in vitro data generated in this study are available in the Supplementary information. Sequences of oligonucleotides used for the KIR genotyping (PCR-SSP) are limited to non-commercial and research use and are available upon request to Dr. K. Gendzekhadze (kgendzek@coh.org), City Of Hope Medical Foundation, 1500 Duarte Rd, Duarte, CA 91010. Source data are provided with this paper as a Source Data file. Source data are provided with this paper.