Abstract
Background
Acute coronary syndrome (ACS) and diabetes mellitus (DM) are the leading health risks for the elderly. Triglyceride-glucose (TyG) index is a novel and reliable indicator of insulin resistance (IR). This study aims to explore the relationship between the TyG index and all-cause mortality in oldest-old patients with ACS and DM.
Methods
Seven hundred twenty hospitalized patients with ACS aged ≥ 80 years were enrolled, and 699 patients signed informed consent for the study. During the follow-up period, 37 were lost to follow-up, and the follow-up rate was 94.7%. 231 ACS patients with DM were selected for the study’s analyses. Kaplan–Meier curve, Cox regression model and receiver operating characteristic (ROC) curve were used to analyze the association between the TyG index and all-cause mortality.
Results
The mean age of participants was 81.58 ± 1.93 years, and 32.47% were women. Compared to TyG tertile 1, the Hazard Ratio (HR) [95% confidence interval (CI)] of all-cause mortality was 2.04 (1.09, 3.81) for TyG tertile 3 in the fully adjusted model. For the TyG index per standard deviation (SD) increment, the HR (95% CI) of all-cause mortality was 1.44 (1.13, 1.83). Further, the association between the TyG index and all-cause mortality was dose–response (P for trend = 0.026). ROC curve analyses indicated that the TyG index outperformed FBG and TG in the prediction of mortality risk and improved the prognostic value of the Gensini score combined with LVEF.
Conclusion
The TyG index predicts the risk of all-cause mortality in the oldest-old ACS patients with DM.
Keywords: Triglyceride-glucose index, Oldest-old patients, Diabetes mellitus, Acute coronary syndrome, All-cause mortality
Introduction
With the acceleration of population aging, the population of the elderly is dramatically increasing [1, 2]. DM and coronary heart disease (CHD) are common chronic diseases that jeopardize seniors’ health [3]. The latest data shows that cardiovascular disease remains the leading cause of mortality worldwide [4]. It is well known that DM is one of the traditional risk factors for CHD, which could contribute to its progression and deterioration. ACS, one clinical subtype of CHD, typically represents an emergency and serious condition suggesting an increased risk of adverse cardiovascular outcomes. It is estimated that more than 37% of ACS patients have DM [5]. Compared to ACS patients without DM, those with DM are prone to worse cardiovascular events [6, 7]. In addition, DM is associated with an increased risk of mortality in ACS patients [8, 9].
The condition that insulin target tissues are weakly sensitive to metabolic reactions related to insulin is defined as insulin resistance (IR). IR is a common feature of metabolic syndrome, obesity, and DM, and also one risk factor for cardiovascular disease [10, 11]. The TyG index, calculated from fasting blood glucose and triglyceride, is a new marker of IR [12]. The hyperinsulinemic-euglycemic clamp test, considered the gold standard measure of IR, has been found to correlate well with the TyG index [13, 14]. Moreover, compared with the hyperinsulinemic-euglycemic clamp test, the TyG index has the advantages of simplicity, low price, and non-invasiveness, which is more likely to be suitable for clinical practice [15].
Previous studies have shown that the TyG index is one predictor of the risk of arteriosclerosis, coronary calcification, and DM [16–18]. Furthermore, the TyG index was found to be associated with the risk of adverse cardiovascular events in CHD patients [19, 20]. However, the relationship between the TyG index and all-cause mortality remains unclear. This study was designed to examine the association between the TyG index and all-cause mortality in the oldest-old ACS patients with DM.
Methods
Study design and population
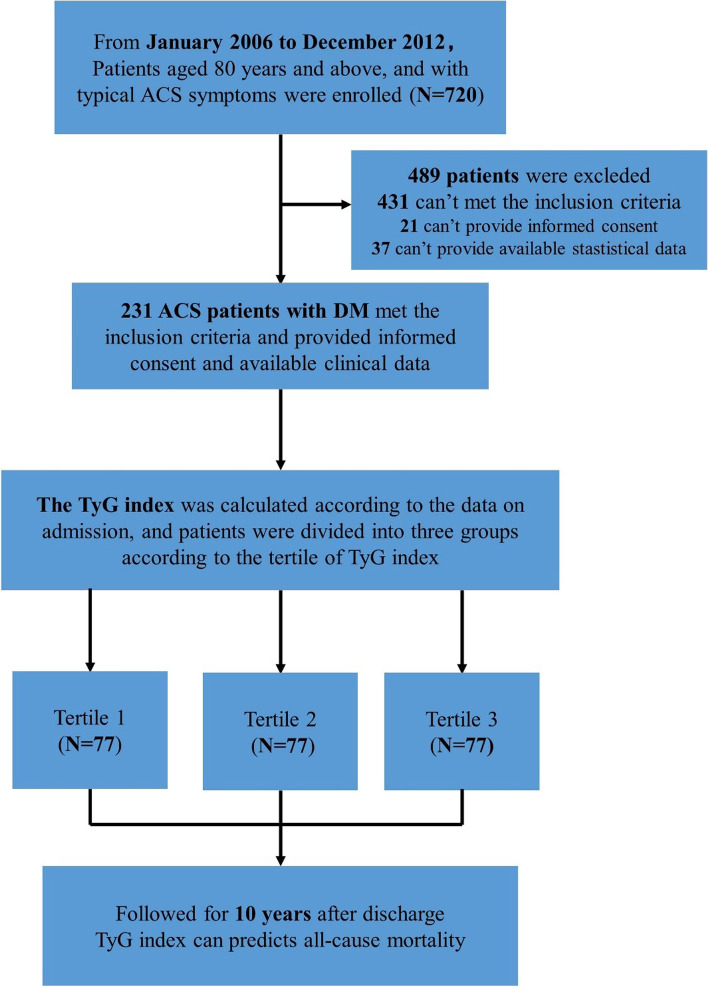
From January 2006 to December 2012, all ACS patients ≥ 80 years old hospitalized in the Department of Cardiology in Chinese PLA General Hospital and who underwent coronary angiography were enrolled. The definition of ACS includes unstable angina pectoris (UAP), non-ST-segment elevation myocardial infarction (NSTEMI), and ST-segment elevation myocardial infarction (STEMI). All patients received standardized treatment based on the coronary angiography results. The exclusion criteria of this study included patients with chronic infectious diseases, types of malignant tumors, rheumatoid arthritis, severe liver insufficiency, pulmonary hypertension, severe valvular heart disease, suspected familial hypertriglyceridemia (triglyceride ≥ 5.65 mmol/L), extreme body mass index (BMI) ≥ 45 kg/m2 and neuropsychiatric disorders. 720 patients were recruited, and 699 signed informed consent. 37 patients were also excluded because of missing follow-up data, and the follow-up rate was 94.7%. The patients with DM were selected for the present study. DM was defined as the previous history of diabetes, fasting blood glucose ≥ 7.0 mmol/L, random blood glucose ≥ 11.1 mmol/L, or oral glucose tolerance test 2-h blood glucose ≥ 11.1 mmol/L. A total of 231 ACS patients with DM were included in the analysis (Fig. 1). This study was approved by the Ethics Service Center of Chinese PLA General Hospital and all methods were performed in accordance with the Helsinki Declaration of Human Rights.
Fig. 1.
Flow chart of the study participants
The sample size needed for the study based on a power calculation of the number of patients required for 80% power at the P = 0.05 level, assuming approximately a 10% loss to follow up. And the number of participants we enrolled is more than the sample size we assessed. Based on the number of participants in the analyses, the Hazard Ratio and the overall probability of event, the power of the research as well as the risk of type II error were calculated.
Data collection
The patients’ general information such as sex, age, height, weight, systolic blood pressure (SBP), diastolic blood pressure (DBP) and smoking history, past medical histories such as hypertension, hyperlipidemia, myocardial infarction (MI), chronic kidney disease (CKD) and stroke, fasting biochemical indicators such as fasting blood glucose (FBG), total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), creatinine and uric acid (UA)], cardiovascular medication history such as aspirin, clopidogrel, statins, beta-blockers, angiotensin-converting enzyme inhibitor (ACEI), angiotensin receptor blocker (ARB)], and left ventricular ejection fraction (LVEF) were collected.
All patients performed coronary intervention. The coronary angiography results were analyzed by the same image analysis software. Coronary culprit vessels, including the left main artery (LM), left anterior descending artery (LAD), left circumflex artery (LCX) and right coronary artery (RCA), were evaluated by the trained professors. Multivessel lesions were defined as the condition that there were two or more culprit vessels. The Gensini score was calculated to estimate the severity of coronary artery stenosis. Based on the coronary angiography results, all patients received standardized and individualized therapeutic strategies, including intensive medicine, percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG).
The TyG index was retrospectively calculated as Ln [fasting TG (mg/dl) × fasting FBG (mg/dl)/2]. Body mass index (BMI) was defined as the body mass (in kilograms) divided by the square of the body height (in meters). The equation of estimated glomerular filtration rate (eGFR) was calculated by the Chinese modified Modification of Diet in Renal Disease: eGFR (mL/min/1.73 m2) = 175 × standardized creatinine (mg/dL) −1.234 × age(year)−0.179 [× 0.79 (if female)]. An eGFR < 60 mL/min/1.73 m2 was considered the criterion for CKD.
Follow-up and endpoint event
The primary endpoint was all-cause mortality. After hospital discharge, all patients were routinely followed up every 12 months. The information regarding adverse events was obtained through telephone contact with patients or their family members and ascertained from a careful review of corresponding medical records. The follow-up time reached up to 10 years. Follow-up time was recorded in months, calculated by the time of the endpoint event minus the time of discharge.
Statistical analysis
The patients were divided into 3 groups (Tertile 1, Tertile 2 and Tertile 3) according to the TyG index tertiles. Continuous data were expressed as mean ± standard deviation (SD) or median (25th to 75th percentile), and analysis of variance (ANOVA) and the Kruskal–Wallis test were used to compare differences in baseline characteristics among the TyG index tertiles according to whether the variables were normally distributed. Categorical data were expressed as n (%), and the Chi-squared test or Fisher’s exact test was used to compare proportions between groups. The correlations between the TyG index and baseline variables were analyzed using Pearson’s correlation test.
Kaplan–Meier curves were used to describe the survival between the TyG index groups and Log-rank tests were performed. A univariate Cox regression analysis was carried out to identify predictors of mortality. The cox proportional hazards model was used to analyze the association between the TyG index and all-cause mortality. In regression analysis, the selection of covariables mainly follows the following principles: 1. the correlated factors screened out by univariable Cox regression analysis; 2. relevant factors found in previous research; 3. risk factors acknowledged widely. We used the Likelihood ratio test to assess the Cox models, and the statistical test of the proportional hazard assumption was performed. Moreover, the Schoenfeld Residuals Test and Kaplan–Meier curve were visually inspected for potential time-variant biases.
Three models were built and potential confounders were adjusted in the models. Model 1 was the unadjusted model. Model 2 was adjusted for age and sex. Model 3 was adjusted for age, sex, BMI, SBP, DBP, LVEF, Gensini score, hypertension, hyperlipidemia, previous MI, previous stroke, CKD, current smoking, TC, LDL-C, HDL-C, eGFR, UA, aspirin, clopidogrel, statin, β-blocker, ACEI/ARB, LM lesion, multivessel lesion and treatment.
The TyG index was treated as three categorical variables for the primary analysis. Trend tests were used to explore the dose–response relationship between the TyG index and all-cause mortality. The TyG index was standardized and put into the regression models to determine the relationship between the TyG per SD increment and mortality. Results were expressed as hazard ratios (HR) and 95% confidence intervals (CI).
Receiver operating characteristic (ROC) curves were used to compare the discrimination capacity of the TyG index, FBG and TG to predict mortality. To compare the discrimination of the three above indicators, harrell C-statistics, continuous net reclassification improvement (cNRI), and integrated discrimination improvement (IDI) were calculated.
The statistical software packages R (http://www.R-project.org, The R Foundation) and Empower Stats (http://www.empowerstats.com, X&Y Solutions, Inc., Boston, MA) were selected to analyze all data. A two-tailed P < 0.05 was regarded as statistically significant.
Results
Baseline characteristics of all participants
Among the 231 oldest-old patients enrolled, most patients were men (67.53%). The average age was 81.58 ± 1.93 years, and the average TyG index was 9.01 ± 0.67. According to the TyG index tertiles, the patients were divided into three groups. The higher group was more likely to be female and to have elevated HR, FBG, TC, TG, and LDL-C. There was no statistical difference in age, BMI, BP, LVEF, smoking history, eGFR, UA, history of past illness, cardiovascular medications, coronary artery lesion and treatment strategy among the TyG index groups (Table 1).
Table 1.
Baseline characteristics of all participants according to TyG index tertiles
| Variables | Total | TyG index | P-value | ||
|---|---|---|---|---|---|
| Tertile 1 | Tertile 2 | Tertile 3 | |||
| N | 231 | 77 | 77 | 77 | |
| General information | |||||
| Age, year | 81.58 ± 1.93 | 81.49 ± 1.69 | 81.77 ± 2.23 | 81.47 ± 1.84 | 0.576 |
| Female, n (%) | 75 (32.47%) | 14 (18.18%) | 28 (36.36%) | 33 (42.86%) | 0.003 |
| Height, cm | 165.41 ± 8.06 | 166.55 ± 8.03 | 165.17 ± 8.40 | 164.51 ± 7.71 | 0.276 |
| Weight, kg | 67.96 ± 11.30 | 68.04 ± 11.89 | 68.12 ± 11.20 | 67.71 ± 10.93 | 0.972 |
| BMI, kg/m2 | 24.78 ± 3.42 | 24.44 ± 3.45 | 24.94 ± 3.62 | 24.96 ± 3.20 | 0.574 |
| SBP, mmHg | 139.19 ± 21.57 | 139.58 ± 18.36 | 143.10 ± 22.03 | 134.90 ± 23.49 | 0.060 |
| DBP, mmHg | 69.77 ± 12.06 | 69.64 ± 10.74 | 70.38 ± 12.31 | 69.31 ± 13.16 | 0.855 |
| LVEF, % | 56.28 ± 9.19 | 58.26 ± 7.82 | 55.87 ± 9.55 | 54.72 ± 9.84 | 0.051 |
| Current smoking, n (%) | 67 (29.00%) | 27 (35.06%) | 21 (27.27%) | 19 (24.68%) | 0.335 |
| Laboratory markers | |||||
| eGFR, mL/min/1.73 m2 | 69.12 ± 20.94 | 72.76 ± 19.90 | 68.75 ± 21.27 | 65.85 ± 21.31 | 0.121 |
| FBG, mmol/L | 8.51 ± 6.06 | 5.92 ± 1.68 | 7.75 ± 2.23 | 11.85 ± 9.21 | < 0.001 |
| UA, umol/L | 354.50 ± 200.08 | 361.12 ± 306.02 | 343.06 ± 97.01 | 359.33 ± 133.73 | 0.828 |
| TC, mmol/L | 4.03 ± 0.99 | 3.69 ± 0.91 | 4.06 ± 0.95 | 4.35 ± 1.01 | < 0.001 |
| TG, mmol/L | 1.50 ± 0.77 | 0.93 ± 0.29 | 1.38 ± 0.32 | 2.21 ± 0.88 | < 0.001 |
| LDL-C, mmol/L | 2.26 ± 0.85 | 2.00 ± 0.73 | 2.33 ± 0.86 | 2.43 ± 0.89 | 0.004 |
| HDL-C, mmol/L | 1.09 ± 0.36 | 1.19 ± 0.47 | 1.09 ± 0.29 | 0.98 ± 0.26 | 0.002 |
| TyG index | 9.01 ± 0.67 | 8.30 ± 0.32 | 8.99 ± 0.16 | 9.75 ± 0.43 | < 0.001 |
| History of Past Illness | |||||
| Hypertension, n (%) | 200 (86.58%) | 68 (88.31%) | 65 (84.42%) | 67 (87.01%) | 0.770 |
| Previous stroke, n (%) | 51 (22.08%) | 21 (27.27%) | 17 (22.08%) | 13 (16.88%) | 0.299 |
| Previous MI, n (%) | 35 (15.15%) | 10 (12.99%) | 15 (19.48%) | 10 (12.99%) | 0.431 |
| Hyperlipidemia, n (%) | 56 (24.24%) | 20 (25.97%) | 16 (20.78%) | 20 (25.97%) | 0.686 |
| CKD, n (%) | 37 (16.02%) | 11 (14.29%) | 11 (14.29%) | 15 (19.48%) | 0.598 |
| Cardiovascular medications | |||||
| Aspirin, n (%) | 227 (98.27%) | 75 (97.40%) | 76 (98.70%) | 76 (98.70%) | 0.775 |
| Clopidogrel, n (%) | 220 (95.24%) | 75 (97.40%) | 72 (93.51%) | 73 (94.81%) | 0.513 |
| Statin, n (%) | 213 (92.21%) | 72 (93.51%) | 68 (88.31%) | 73 (94.81%) | 0.282 |
| β-blocker, n (%) | 154 (66.67%) | 53 (68.83%) | 52 (67.53%) | 49 (63.64%) | 0.776 |
| ACEI/ARB, n (%) | 145 (62.77%) | 49 (63.64%) | 48 (62.34%) | 48 (62.34%) | 0.982 |
| Angiography | |||||
| LAD lesion, n (%) | 195 (84.42%) | 69 (89.61%) | 66 (85.71%) | 60 (77.92%) | 0.126 |
| LCX lesion, n (%) | 150 (64.94%) | 50 (64.94%) | 53 (68.83%) | 47 (61.04%) | 0.598 |
| RCA lesion, n (%) | 169 (73.16%) | 56 (72.73%) | 54 (70.13%) | 59 (76.62%) | 0.658 |
| LM lesion, n (%) | 35 (15.15%) | 9 (11.69%) | 15 (19.48%) | 11 (14.29%) | 0.390 |
| Multivessel lesion, n (%) | 180 (77.92%) | 61 (79.22%) | 61 (79.22%) | 58 (75.32%) | 0.797 |
| Gensini score | 55.44 ± 41.43 | 46.55 ± 29.80 | 58.52 ± 43.33 | 61.25 ± 48.01 | 0.064 |
| Treatment | 0.176 | ||||
| Intensive medication, n (%) | 79 (34.20%) | 31 (40.26%) | 24 (31.17%) | 24 (31.17%) | |
| PCI | 145 (62.77%) | 45 (58.44%) | 48 (62.34%) | 52 (67.53%) | |
| CABG | 7 (3.03%) | 1 (1.30%) | 5 (6.49%) | 1 (1.30%) | |
The TyG index was correlated with clinical variables
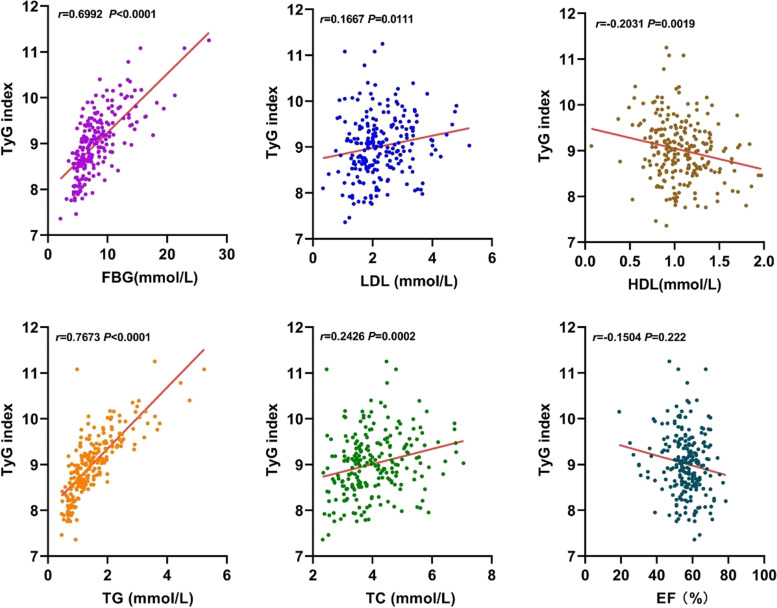
The TyG index was positively correlated with FBG, TC, TG and LDL-C, while negatively correlated with eGFR, LVEF and HDL-C. The TyG index showed no correlation with age, BMI, SBP, DBP, UA and Gensini score. The TyG index had the highest positive and negative correlation with TG and HDL-C, respectively (Fig. 2, Table 2).
Fig. 2.
Correlation between the TyG index and clinical variables
Table 2.
Correlation between TyG index and clinical variables
| Variables | Correlation coefficient, r | 95%CI | P-value |
|---|---|---|---|
| Age | -0.049 | (-0.177, 0.081) | 0.458 |
| BMI | 0.039 | (-0.090, 0.167) | 0.554 |
| SBP | -0.097 | (-0.223, 0.033) | 0.143 |
| DBP | -0.072 | (-0.199, 0.058) | 0.278 |
| eGFR | -0.135 | (-0.259, -0.006) | 0.041 |
| FBG | 0.556 | (0.460, 0.639) | < 0.001 |
| UA | -0.026 | (-0.154, 0.104) | 0.697 |
| LVEF | -0.150 | (-0.274, -0.022) | 0.022 |
| Gensini score | 0.127 | (-0.002, 0.252) | 0.054 |
| TC | 0.242 | (0.117, 0.360) | < 0.001 |
| TG | 0.767 | (0.709, 0.816) | < 0.001 |
| LDL-C | 0.167 | (0.038, 0.289) | 0.011 |
| HDL-C | -0.224 | (-0.343, -0.097) | < 0.001 |
Univariate Cox regression analysis for all-cause mortality
During a median follow-up of 49 months (interquartile range: 36 to 61 months), a total of 86 patients died. Univariate Cox regression analysis was performed to identify the potential predictors of mortality. The all-cause mortality was significantly associated with several factors, the previous stroke, CKD, HR, SBP, DBP, eGFR, FBG, LVEF, Gensini score, and the TyG index included (Table 3).
Table 3.
Univariate Cox regression analysis for the association between baseline variables and all-cause mortality
| Variables | HR (95%CI) | P-value |
|---|---|---|
| Female | 1.07 (0.68, 1.69) | 0.758 |
| Age | 1.11 (0.99, 1.24) | 0.062 |
| BMI | 0.95 (0.90, 1.02) | 0.138 |
| Hypertension | 1.17 (0.61, 2.27) | 0.636 |
| Previous stroke | 1.79 (1.13, 2.83) | 0.012 |
| Previous MI | 1.62 (0.96, 2.72) | 0.071 |
| Hyperlipidemia | 1.02 (0.62, 1.67) | 0.932 |
| CKD | 2.42 (1.49, 3.93) | 0.004 |
| Current smoking | 1.06 (0.66, 1.68) | 0.819 |
| SBP | 0.99 (0.98, 1.00) | 0.01 |
| DBP | 0.97 (0.96, 0.99) | 0.006 |
| Aspirin | 0.33 (0.10, 1.06) | 0.062 |
| Clopidogrel | 0.56 (0.24, 1.29) | 0.174 |
| Statin | 1.12 (0.51, 2.43) | 0.780 |
| β-blocker | 1.06 (0.67, 1.66) | 0.806 |
| ACEI/ARB | 1.17 (0.75, 1.84) | 0.485 |
| eGFR | 0.97 (0.96, 0.98) | < 0.001 |
| FBG | 1.04 (1.02, 1.06) | < 0.001 |
| UA | 1.00 (1.00, 1.00) | 0.537 |
| LVEF | 0.97 (0.95, 0.99) | 0.002 |
| Gensini score | 1.01 (1.00, 1.01) | 0.001 |
| LAD lesion | 0.93 (0.54, 1.60) | 0.797 |
| LCX lesion | 1.28 (0.81, 2.03) | 0.290 |
| RCA lesion, | 1.31 (0.79, 2.18) | 0.300 |
| LM lesion | 0.98 (0.54, 1.77) | 0.951 |
| Multivessel lesion | 1.67 (0.94, 2.97) | 0.079 |
| Treatment | ||
| Intensive medication | 1.0 | |
| PCI | 0.81 (0.52, 1.26) | 0.343 |
| CABG | 0.26 (0.04, 1.90) | 0.185 |
| TC | 1.20 (0.98, 1.48) | 0.081 |
| TG | 1.26 (0.99, 1.60) | 0.057 |
| LDL-C | 1.18 (0.92, 1.51) | 0.204 |
| HDL-C | 0.59 (0.29, 1.19) | 0.142 |
| TyG index | 1.76 (1.28, 2.42) | 0.001 |
| TyG group | ||
| Tertile 1 | 1.0 | |
| Tertile 2 | 1.56 (0.88, 2.78) | 0.129 |
| Tertile 3 | 2.13 (1.23, 3.71) | 0.007 |
Association between the TyG index and all-cause mortality
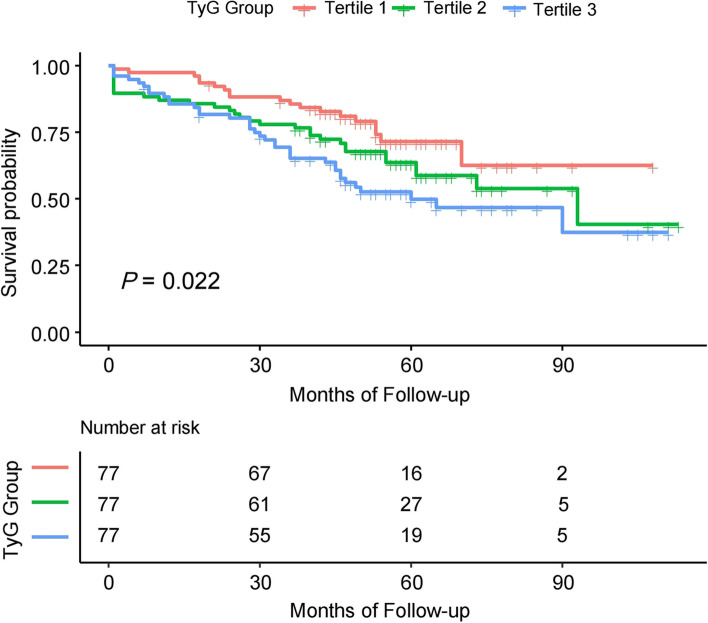
The Cox regression models were built to analyze the association between the TyG index and all-cause mortality. Based on the tertiles, the TyG index was transformed as a categorized variable in the analyses. As shown in Table 4, after fully adjusting for the confounders, the HRs (95% CIs) for all-cause mortality in Tertile 2 and Tertile 3 were 1.62 (0.87, 3.02) and 2.04 (1.09, 3.81), respectively. When the TyG index was entered as a continuous variable into the models, we found a similar result that the HRs (95% CIs) for all-cause mortality was 1.44 (1.13, 1.83) for the TyG index per SD increment. We also conducted the trend test analyses and found that there was a dose–response relationship between the TyG index and all-cause mortality (P for trend = 0.026). Further, the Kaplan–Meier curve showed that the patients in Tertile 3 had the worst survival probability (P = 0.022) (Fig. 3).
Table 4.
Cox regression analysis for the association between TyG index and all-cause mortality
| TyG index | Death, n (%) | Model 1 | Model 2 | Model3 |
|---|---|---|---|---|
| Per SD increase | 86(37.2) | 1.46 (1.18, 1.82) *** | 1.50 (1.21, 1.86) *** | 1.44 (1.13, 1.83) ** |
| TyG Tertiles | ||||
| Tertile 1 | 19(24.7) | 1.0(Ref) | 1.0(Ref) | 1.0(Ref) |
| Tertile 2 | 30(39.0) | 1.56 (0.88, 2.78) | 1.62 (0.90, 2.89) | 1.62 (0.87, 3.02) |
| Tertile 3 | 37(48.1) | 2.13 (1.23, 3.71) ** | 2.18 (1.25, 3.82) ** | 2.04 (1.09, 3.81) * |
| P for trend | 0.007 | 0.006 | 0.026 | |
Model 1: unadjusted
Model 2: adjusted for age and sex
Model 3: adjusted for age, sex, BMI, SBP, DBP, LVEF, Gensini score, hypertension, hyperlipidemia, previous MI, previous stroke, CKD, current smoking, TC, LDL-C, HDL-C, eGFR, UA, aspirin, clopidogrel, statin, β-blocker, ACEI/ARB, LM lesion, multivessel lesion and treatment
*P < 0.05 **P < 0.01 ***P < 0.001
Fig. 3.
Kaplan–Meier survival curves of survival probability according to TyG tertiles
Comparison of the capacity of the TyG index, FBG and TG to predict mortality
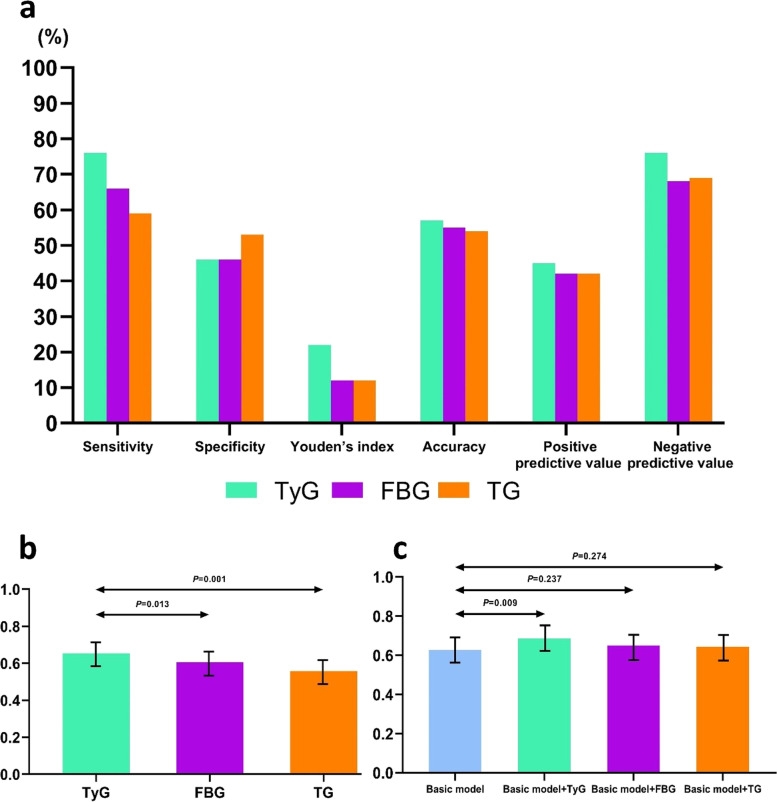
ROC curve was used to compare the discrimination capacity of the TyG index, FBG and TG to predict mortality. The TyG index had the highest sensitivity, accuracy, positive predictive value and negative predictive value, although the specificity of the TyG index was slightly lower. Youden’s index was higher than that of FBG and TG (Fig. 4a). The C-statistics of the TyG index, FBG and TG was 0.653 (0.589–0.717), 0.606 (0.543–0.670), 0.557 (0.493–0.620) respectively. The TyG index was superior to FBG and TG at predicting all-cause mortality, as was seen by the discrimination index values(P < 0.05) (Table 5, Fig. 4b). For mortality risk prediction, considering the effects of traditional risk factors, the basic model on the Gensini score combined with LVEF was constructed for further research. The C-statistics of the Gensini score + LVEF + TyG, the Gensini score + LVEF + FBG, the Gensini score + LVEF + TG was 0.685 (P = 0.009), 0.650 (P = 0.237), 0.643 (P = 0.274), respectively. Only the TyG index provided a significant incremental prognostic value (cNRI = 0.238, IDI = 0.083) (Table 6, Fig. 4c).
Fig. 4.
a Diagnostic performance of TyG, FBG, and TG for all-cause mortality; b C-statistic of TyG, FBG, and TG for prediction of all-cause mortality; c C-statistic of the basic model (Gensini score and LVEF), + TyG, + FBG, and + TG for prediction of all-cause mortality
Table 5.
Comparative analysis of the discrimination of TyG, FBG, and TG for all-cause mortality
| Discrimination ability | TyG | FBG | TG | |||
| Sensitivity, % | 76.2 | 66.0 | 59.3 | |||
| Specificity, % | 46.7 | 46.9 | 53.0 | |||
| Youden’s index, % | 22.9 | 12.3 | 12.9 | |||
| Accuracy, % | 57.7 | 55.3 | 54 | |||
| Positive predictive value, % | 45.9 | 42.8 | 42.4 | |||
| Negative predictive value, % | 76.8 | 68.7 | 69.9 | |||
| C-Statistic (95% CI) | 0.653 (0.589–0.717) | 0.606 (0.543–0.670) | 0.557 (0.493–0.620) | |||
| Comparison | TyG VS. FBG | TyG VS. TG | FBG VS. TG | |||
| Difference | P value | Difference | P value | Difference | P value | |
| C-Statistic | 0.047 | 0.013 | 0.096 | 0.001 | 0.049 | 0.679 |
| cNRI | 0.255 | 0.007 | 0.300 | 0.007 | -0.030 | 0.811 |
| IDI | 0.080 | < 0.001 | 0.091 | < 0.001 | 0.011 | 0.605 |
Table 6.
Model performance after the addition of TyG, FBG, or TG to the basic model (Gensini score and LVEF) for predicting all-cause mortality
| Model | C-Statistic | P value | cNRI | P value | IDI | P value |
|---|---|---|---|---|---|---|
| Gensini score + LVEF | 0.627 (0.563–0.690) | - | - | - | - | - |
| Gensini score + LVEF + TyG | 0.685 (0.624–0.746) | 0.009 | 0.238 | 0.007 | 0.083 | 0.013 |
| Gensini score + LVEF + FBG | 0.650 (0.588–0.713) | 0.237 | -0.077 | 0.399 | 0.005 | 0.764 |
| Gensini score + LVEF + TG | 0.643 (0.581–0.705) | 0.274 | 0.026 | 0.817 | 0.005 | 0.757 |
Discussion
The present study was performed on the oldest-old ACS patients with DM and the follow-up period was as long as ten years. This study, for the first time, investigated the association between the TyG index and all-cause mortality in the population aged ≥ 80 years with ACS and DM. The TyG index was significantly associated with all-cause mortality in the study population regardless of whether the TyG index was used as a continuous or categorical variable. In addition, the relationship between the TyG index and all-cause mortality was dose–response. The TyG index outperformed FBG and TG in the prediction of mortality and further improved the prognostic value on the Gensini score combined with LVEF.
The mortality of coronary heart disease is increasing year by year. As the most severe clinical subtype, ACS has the poorest prognosis. Advanced age and DM are both independent risk factors for CHD, which could cause disease progression and deterioration. In addition, the pace of the population aging worldwide is accelerating, accompanied by the most rapidly growing of the oldest-old (those aged ≥ 80 years) group. It is no doubt that the oldest-old patients with ACS and DM would bring a heavy burden to social medicine and the health system. Accordingly, exploring the predictors associated with the long-term prognosis and early identifying the high-risk patients are effective strategies to deal with this problem.
The TyG index is one surrogate indicator that reflects the condition of IR in the body, which can be calculated from FBG and TG [12]. Studies have shown that the TyG index has a good correlation with traditional IR evaluation markers, such as the hyperinsulinemic-euglycemic clamp test and HOMA-IR [14, 21]. Moreover, compared with the traditional IR evaluation indicators, the TyG index could be calculated by common laboratory markers, which may be more suitable for clinical practice. Previous studies have shown that the TyG index is associated with arterial stiffness [16], coronary artery calcification progression [17, 22], and the incident of DM [22], which are all associated with the incident and progression of cardiovascular disease. In the present study, the TyG index was found to be positively correlated with eGFR, TC, LDL-C and negatively correlated with HDL-C, which are all traditional risk factors for cardiovascular disease. Based on the factors mentioned above, the hypothesis that the TyG index could predict the prognostic risk of cardiovascular disease is rational.
Further, several studies have reported that there is also a significant association between the TyG index and the risk of cardiovascular and cerebrovascular diseases. The TyG index was associated with prognostic risk in both STEMI and NSTEMI populations [23–25]. In these studies, the TyG index was found to be associated with the risk of cardiovascular and cerebrovascular events, but only one study revealed that there was a significant association between the TyG index and the risk of cardiac events and all-cause mortality. In the nondiabetic patients with ACS, the association between TyG index and mortality was not well established. Yang et al. reported that the TyG index was not associated with all-cause mortality in the nondiabetic patients undergoing PCI [26]. In another study based on the nondiabetic patients with NSTE-ACS undergoing PCI, Zhao et al. also revealed that the TyG index had no association with all-cause mortality [27]. However, Şaylık et al. found that the TyG index was associated with the in-hospital and one-year mortality in the elderly nondiabetic patients with STEMI [28]. It was clear that these studies had apparent discrepancies attributed to variations in study population, variable type of the TyG index, time of follow-up, and adjustment for confounders. So, it is need that more studies should be conducted to explore the association between the TyG and mortality in the nondiabetic patients with ACS.
In our study, given that the oldest-old patients with ACS and DM were more susceptible to the adverse outcome, the association between TyG index and all-cause mortality was investigated in this population. Besides, the relationship between the TyG index and all-cause mortality in ACS patients with DM is also not well established. There are merely four studies that have reported the association between the TyG index and all-cause mortality, and the findings of these studies were not well consistent [29–32]. Discrepancies within these studies were likely attributed to variations in the study population, variable type of the TyG index, time of follow-up, and adjustment for confounders. Of the studies mentioned above, the mean age of participants ranged from 60.9 to 66.3 years, and the time of follow-up ranged from 12 to 36 months. Different from previous studies, ACS patients with DM aged 80 years or above were enrolled and the follow-up time ranged from 36 to 113 months. It’s the first study to confirm the association between the TyG index and all-cause mortality in this special population, and also find that the association between the TyG index and mortality was dose–response. To enhance the reliability and validity of the study, we have used several statistical analysis strategies, including continuous and categorical variable types, trend test analyses, subgroup analyses and interaction analyses. Fortunately, the findings are stable and significant in different statistical analysis models. In terms of the study population and data analysis strategies, we think that our study could provide some contributions to this field.
As mentioned above, the TyG index is the product of FBG and TG. Theoretically, the TyG index represents the comprehensive effect of FBG and TG, which should be superior to either FBG or TG. The findings confirm this hypothesis. For mortality risk prediction, compared to FBG and TG, the TyG index has an advantage in the discrimination capacity. Further, the TyG index has a significant improvement in the prediction model based on the Gensini score combined with LVEF, which are the surrogate indicators of the severity of the coronary lesion and myocardial function, respectively.
The research shows that the TyG index is a dependable indicator to predict all-cause mortality in oldest-old patients with ACS and DM. Although the exact pathophysiological mechanism is not clear, we speculate that IR may be the main cause of target organ damage due to the increase of TyG index. First of all, subclinical vascular disease is associated with IR, and vascular functional and structural damage and hyperinsulinemia resulting from IR contribute to the emergence of endothelial cell dysfunction, followed by the genesis of the decrease in bioavailable nitric oxide, which leads to endothelial-dependent vascular dysfunction, including impaired vascular relaxation, inflammation, vascular remodeling, and overt fibrosis [33–37]. Secondly, IR may leads to the intensification of oxidative stress and inflammatory response, disturbance of glucose and lipid metabolism, activation of the renin–angiotensin–aldosterone system (RAAS) and thrombotic activity, and ultimately cell damage, hypertrophy, and fibrosis occur [38–40]. Furthermore, IR is closely associated with microvascular and myocardial damage and poor myocardial perfusion, which leads to various cardiovascular events [17, 29, 41].
Comparisons with other studies and what does the current work add to the existing knowledge
This is the first study among aged 80 years or above patients with ACS and DM. In this special population, the findings of this study have acknowledged the relationship between the TyG index and all-cause mortality and the dose–response relationship between the TyG index and mortality.
Study strengths and limitations
The present study has a number of strengths. The current study has confirmed the TyG index was associated with all-cause mortality in oldest-old ACS patients with DM. And the TyG index outperformed FBG and TG in the prediction of mortality. The TyG index improved the prognostic value on the Gensini score combined with LVEF. There are some limitations in the present study. Firstly, this study is a single-center study, and the sample size is relatively small. There is a certain selection bias, and further multi-center and large-sample studies are needed to verify these findings. Secondly, this study did not conduct a comparative analysis between the TyG index and traditional IR indicators. Thirdly, although many confounding factors have been included in the research analysis, there are still some known factors (dietary habits and nutritional status) or unknown factors that may affect the results that have not been included in the analysis.
Conclusions
The TyG index predicts the risk of all-cause mortality in the oldest-old ACS patients with DM. Particularly, the TyG index is better than FBG and TG in the prediction of mortality risk and improves the prognostic value of the Gensini score combined with LVEF. The TyG index may become a convenient biomarker for screening and predicting all-cause mortality in the oldest-old ACS patients with DM in the future.
.
Acknowledgements
We appreciate the study participants for the information they provided and all the staff involved in this study.
Abbreviations
- ACS
Acute coronary syndrome
- DM
Diabetes mellitus
- IR
Insulin resistance
- CHD
Coronary heart disease
- UAP
Unstable angina pectoris
- NSTEMI
Non-ST-segment elevation myocardial infarction
- STEMI
ST-segment elevation myocardial infarction
- BMI
Body mass index
- HR
Heart rate
- SBP
Systolic blood pressure
- DBP
Diastolic blood pressure
- LVEF
Left ventricular ejection fraction
- eGFR
Estimated glomerular fltration rate
- FBG
Fasting blood glucose
- UA
Uric acid
- TC
Total cholesterol
- TG
Triglyceride
- LDL-C
Low-density lipoprotein-C
- HDL-C
High-density lipoprotein-C
- TyG
Triglyceride glucose
- MI
Myocardial infarction
- CKD
Chronic kidney disease
- ACEI
Angiotensin-converting enzyme inhibitor
- ARB
Angiotensin receptor blocker
- LAD
Left anterior descending artery
- LCX
Left circumfex artery
- RCA
Right coronary artery
- LM
Left main coronary artery
- PCI
Percutaneous coronary intervention
- CABG
Coronary artery bypass grafting
- HR
Hazard ratio
- CI
Confidence interval
- SD
Standard deviation
- ROC
Receiver operating characteristic
- cNRI
Continuous net reclassification improvement
- IDI
Integrated discrimination improvement
Authors’ contributions
The authors’ responsibilities were as follows: JS and BF: data interpretation, and the first draft of the manuscript writing; LF: statistical analysis; YJ, YL, YKS and HNL: data collection; ZHF, XLH and DYL: manuscript revision; DYL and ZHF: study design. All authors: searched the literature, contributed to the revision of the manuscript, and read and approved the final manuscript.
Funding
None.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
The study was performed in accordance with the Declaration of Helsinki and approved by the medical ethics committee of the Chinese PLA General Hospital. All participants provided written informed consent.
Consent for publication
Not applicable.
Competing interests
The author reports no conflicts of interest in this work.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jian Shen, Bin Feng and Li Fan contributed equally to this work.
Contributor Information
Dongyun Li, Email: 1402817523@qq.com.
Zhenhong Fu, Email: fuzhenh@126.com.
References
- 1.Beard JR, Officer A, de Carvalho IA, Sadana R, Pot AM, Michel JP, Lloyd-Sherlock P, Epping-Jordan JE, Peeters G, Mahanani WR, et al. The World report on ageing and health: a policy framework for healthy ageing. Lancet. 2016;387(10033):2145–2154. doi: 10.1016/S0140-6736(15)00516-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Partridge L, Deelen J, Slagboom PE. Facing up to the global challenges of ageing. Nature. 2018;561(7721):45–56. doi: 10.1038/s41586-018-0457-8. [DOI] [PubMed] [Google Scholar]
- 3.Fang EF, Xie C, Schenkel JA, Wu C, Long Q, Cui H, Aman Y, Frank J, Liao J, Zou H, et al. A research agenda for ageing in China in the 21st century (2nd edition): Focusing on basic and translational research, long-term care, policy and social networks. Ageing Res Rev. 2020;64:101174. doi: 10.1016/j.arr.2020.101174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, Baddour LM, Barengo NC, Beaton AZ, Benjamin EJ, Benziger CP, et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J Am Coll Cardiol. 2020;76(25):2982–3021. doi: 10.1016/j.jacc.2020.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhou M, Liu J, Hao Y, Liu J, Huo Y, Smith SC, Ge J, Ma C, Han Y, Fonarow GC, et al. Prevalence and in-hospital outcomes of diabetes among patients with acute coronary syndrome in China: findings from the Improving Care for Cardiovascular Disease in China-Acute Coronary Syndrome Project. Cardiovasc Diabetol. 2018;17(1):147. doi: 10.1186/s12933-018-0793-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ray KK, Colhoun HM, Szarek M, Baccara-Dinet M, Bhatt DL, Bittner VA, Budaj AJ, Diaz R, Goodman SG, Hanotin C, et al. Effects of alirocumab on cardiovascular and metabolic outcomes after acute coronary syndrome in patients with or without diabetes: a prespecified analysis of the ODYSSEY OUTCOMES randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7(8):618–628. doi: 10.1016/S2213-8587(19)30158-5. [DOI] [PubMed] [Google Scholar]
- 7.Preis SR, Hwang S-J, Coady S, Pencina MJ, D'Agostino RB, Savage PJ, Levy D, Fox CS. Trends in all-cause and cardiovascular disease mortality among women and men with and without diabetes mellitus in the Framingham Heart Study, 1950 to 2005. Circulation. 2009;119(13):1728–1735. doi: 10.1161/CIRCULATIONAHA.108.829176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Winzap P, Davies A, Klingenberg R, Obeid S, Roffi M, Mach F, Räber L, Windecker S, Templin C, Nietlispach F, et al. Diabetes and baseline glucose are associated with inflammation, left ventricular function and short- and long-term outcome in acute coronary syndromes: role of the novel biomarker Cyr 61. Cardiovasc Diabetol. 2019;18(1):142. doi: 10.1186/s12933-019-0946-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sethi SS, Akl EG, Farkouh ME. Diabetes mellitus and acute coronary syndrome: lessons from randomized clinical trials. Curr Diab Rep. 2012;12(3):294–304. doi: 10.1007/s11892-012-0272-9. [DOI] [PubMed] [Google Scholar]
- 10.Hill MA, Yang Y, Zhang L, Sun Z, Jia G, Parrish AR, Sowers JR. Insulin resistance, cardiovascular stiffening and cardiovascular disease. Metabolism. 2021;119:154766. doi: 10.1016/j.metabol.2021.154766. [DOI] [PubMed] [Google Scholar]
- 11.Laakso M, Kuusisto J. Insulin resistance and hyperglycaemia in cardiovascular disease development. Nat Rev Endocrinol. 2014;10(5):293–302. doi: 10.1038/nrendo.2014.29. [DOI] [PubMed] [Google Scholar]
- 12.Simental-Mendía LE, Rodríguez-Morán M, Guerrero-Romero F. The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab Syndr Relat Disord. 2008;6(4):299–304. doi: 10.1089/met.2008.0034. [DOI] [PubMed] [Google Scholar]
- 13.DeFronzo RA, Tobin JD, Andres R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Physiol. 1979;237(3):E214–223. doi: 10.1152/ajpendo.1979.237.3.E214. [DOI] [PubMed] [Google Scholar]
- 14.Fiorentino TV, Marini MA, Succurro E, Andreozzi F, Sesti G. Relationships of surrogate indexes of insulin resistance with insulin sensitivity assessed by euglycemic hyperinsulinemic clamp and subclinical vascular damage. BMJ Open Diabetes Res Care. 2019;7(1):e000911. doi: 10.1136/bmjdrc-2019-000911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Su Y, Wang S, Sun J, Zhang Y, Ma S, Li M, Zhang A, Cheng B, Cai S, Bao Q, et al. Triglyceride Glucose Index Associated With Arterial Stiffness in Chinese Community-Dwelling Elderly. Front Cardiovasc Med. 2021;8:737899. doi: 10.3389/fcvm.2021.737899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wu S, Xu L, Wu M, Chen S, Wang Y, Tian Y. Association between triglyceride-glucose index and risk of arterial stiffness: a cohort study. Cardiovasc Diabetol. 2021;20(1):146. doi: 10.1186/s12933-021-01342-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Park K, Ahn CW, Lee SB, Kang S, Nam JS, Lee BK, Kim JH, Park JS. Elevated TyG Index Predicts Progression of Coronary Artery Calcification. Diabetes Care. 2019;42(8):1569–1573. doi: 10.2337/dc18-1920. [DOI] [PubMed] [Google Scholar]
- 18.Xuan X, Hamaguchi M, Cao Q, Okamura T, Hashimoto Y, Obora A, Kojima T, Fukui M, Yuan G, Guo Z, et al. U-shaped association between the triglyceride-glucose index and the risk of incident diabetes in people with normal glycemic level: A population-base longitudinal cohort study. Clin Nutr. 2021;40(4):1555–1561. doi: 10.1016/j.clnu.2021.02.037. [DOI] [PubMed] [Google Scholar]
- 19.Barzegar N, Tohidi M, Hasheminia M, Azizi F, Hadaegh F. The impact of triglyceride-glucose index on incident cardiovascular events during 16 years of follow-up: Tehran Lipid and Glucose Study. Cardiovasc Diabetol. 2020;19(1):155. doi: 10.1186/s12933-020-01121-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ding X, Wang X, Wu J, Zhang M, Cui M. Triglyceride-glucose index and the incidence of atherosclerotic cardiovascular diseases: a meta-analysis of cohort studies. Cardiovasc Diabetol. 2021;20(1):76. doi: 10.1186/s12933-021-01268-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vasques ACJ, Novaes FS. de Oliveira MdS, Souza JRM, Yamanaka A, Pareja JC, Tambascia MA, Saad MJA, Geloneze B: TyG index performs better than HOMA in a Brazilian population: a hyperglycemic clamp validated study. Diabetes Res Clin Pract. 2011;93(3):e98–e100. doi: 10.1016/j.diabres.2011.05.030. [DOI] [PubMed] [Google Scholar]
- 22.Pranata R, Huang I. Irvan, Lim MA, Vania R: The association between triglyceride-glucose index and the incidence of type 2 diabetes mellitus-a systematic review and dose-response meta-analysis of cohort studies. Endocrine. 2021;74(2):254–262. doi: 10.1007/s12020-021-02780-4. [DOI] [PubMed] [Google Scholar]
- 23.Luo E, Wang D, Yan G, Qiao Y, Liu B, Hou J, Tang C. High triglyceride-glucose index is associated with poor prognosis in patients with acute ST-elevation myocardial infarction after percutaneous coronary intervention. Cardiovasc Diabetol. 2019;18(1):150. doi: 10.1186/s12933-019-0957-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mao Q, Zhou D, Li Y, Wang Y, Xu SC, Zhao XH. The Triglyceride-Glucose Index Predicts Coronary Artery Disease Severity and Cardiovascular Outcomes in Patients with Non-ST-Segment Elevation Acute Coronary Syndrome. Dis Markers. 2019;2019:6891537. doi: 10.1155/2019/6891537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhang Y, Ding X, Hua B, Liu Q, Gao H, Chen H, Zhao X-Q, Li W, Li H. High triglyceride-glucose index is associated with adverse cardiovascular outcomes in patients with acute myocardial infarction. Nutr Metab Cardiovasc Dis. 2020;30(12):2351–2362. doi: 10.1016/j.numecd.2020.07.041. [DOI] [PubMed] [Google Scholar]
- 26.Yang J, Tang YD, Zheng Y, Li C, Zhou Q, Gao J, Meng X, Zhang K, Wang W, Shao C. The Impact of the Triglyceride-Glucose Index on Poor Prognosis in NonDiabetic Patients Undergoing Percutaneous Coronary Intervention. Front Endocrinol (Lausanne) 2021;12:710240. doi: 10.3389/fendo.2021.710240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhao Q, Zhang TY, Cheng YJ, Ma Y, Xu YK, Yang JQ, Zhou YJ. Triglyceride-Glucose Index as a Surrogate Marker of Insulin Resistance for Predicting Cardiovascular Outcomes in Nondiabetic Patients with Non-ST-Segment Elevation Acute Coronary Syndrome Undergoing Percutaneous Coronary Intervention. J Atheroscler Thromb. 2021;28(11):1175–1194. doi: 10.5551/jat.59840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Şaylık F, Çınar T, Selçuk M, Tanboğa İH. The predictive value of triglyceride-glucose index for in-hospital and one-year mortality in elderly non-diabetic patients with ST-segment elevation myocardial infarction. J Geriatr Cardiol. 2022;19(8):610–617. doi: 10.11909/j.issn.1671-5411.2022.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ma X, Dong L, Shao Q, Cheng Y, Lv S, Sun Y, Shen H, Wang Z, Zhou Y, Liu X. Triglyceride glucose index for predicting cardiovascular outcomes after percutaneous coronary intervention in patients with type 2 diabetes mellitus and acute coronary syndrome. Cardiovasc Diabetol. 2020;19(1):31. doi: 10.1186/s12933-020-01006-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang L, Cong HL, Zhang JX, Hu YC, Wei A, Zhang YY, Yang H, Ren LB, Qi W, Li WY, et al. Triglyceride-glucose index predicts adverse cardiovascular events in patients with diabetes and acute coronary syndrome. Cardiovasc Diabetol. 2020;19(1):80. doi: 10.1186/s12933-020-01054-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhao Q, Zhang TY, Cheng YJ, Ma Y, Xu YK, Yang JQ, Zhou YJ. Impacts of triglyceride-glucose index on prognosis of patients with type 2 diabetes mellitus and non-ST-segment elevation acute coronary syndrome: results from an observational cohort study in China. Cardiovasc Diabetol. 2020;19(1):108. doi: 10.1186/s12933-020-01086-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang Y, Ding X, Hua B, Liu Q, Gao H, Chen H, Zhao XQ, Li W, Li H. Predictive effect of triglycerideglucose index on clinical events in patients with type 2 diabetes mellitus and acute myocardial infarction: results from an observational cohort study in China. Cardiovasc Diabetol. 2021;20(1):43. doi: 10.1186/s12933-021-01236-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nakagomi A, Sunami Y, Kawasaki Y, Fujisawa T, Kobayashi Y. Sex difference in the association between surrogate markers of insulin resistance and arterial stiffness. J Diabetes Complicat. 2020;34(6):107442. doi: 10.1016/j.jdiacomp.2019.107442. [DOI] [PubMed] [Google Scholar]
- 34.Adeva-Andany MM, Ameneiros-Rodriguez E, Fernandez-Fernandez C, Dominguez-Montero A, Funcasta-Calderon R. Insulin resistance is associated with subclinical vascular disease in humans. World J Diabetes. 2019;10(2):63–77. doi: 10.4239/wjd.v10.i2.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hill MA, Yang Y, Zhang LP, Sun Z, Jia GH, Parrish AR, Sowers JR. Insulin resistance, cardiovascular stiffening and cardiovascular disease. Metabolism. 2021;119:154766. doi: 10.1016/j.metabol.2021.154766. [DOI] [PubMed] [Google Scholar]
- 36.Jia GH, Aroor AR, DeMarco VG, Martinez-Lemus LA, Meininger GA, Sowers JR. Vascular stiffness in insulin resistance and obesity. Front Physiol. 2015;6:231. doi: 10.3389/fphys.2015.00231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hill MA, Jaisser F, Sowers JR. Role of the vascular endothelial sodium channel activation in the genesis of pathologically increased cardiovascular stiffness. Cardiovasc Res. 2022;118(1):130–140. doi: 10.1093/cvr/cvaa326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lambie M, Bonomini M, Davies SJ, Accili D, Arduini A, Zammit V. Insulin resistance in cardiovascular disease, uremia, and peritoneal dialysis. Trends Endocrin Met. 2021;32(9):721–730. doi: 10.1016/j.tem.2021.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jia GH, Hill MA, Sowers JR. Diabetic Cardiomyopathy: An Update of Mechanisms Contributing to This Clinical Entity. Circ Res. 2018;122(4):624–638. doi: 10.1161/CIRCRESAHA.117.311586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Calles-Escandon J, Mirza SA, Sobel BE, Schneider DJ. Induction of hyperinsulinemia combined with hyperglycemia and hypertriglyceridemia increases plasminogen activator inhibitor 1 in blood in normal human subjects. Diabetes. 1998;47(2):290–293. doi: 10.2337/diab.47.2.290. [DOI] [PubMed] [Google Scholar]
- 41.Trifunovic D, Stankovic S, Sobic-Saranovic D, Marinkovic J, Petrovic M, Orlic D, Beleslin B, Banovic M, Vujisic-Tesic B, Petrovic M, et al. Acute insulin resistance in ST-segment elevation myocardial infarction in non-diabetic patients is associated with incomplete myocardial reperfusion and impaired coronary microcirculatory function. Cardiovasc Diabetol. 2014;13:73. doi: 10.1186/1475-2840-13-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.