Abstract
The Global Initiative for Asthma (GINA) was established in 1993 by the World Health Organization and the US National Heart Lung and Blood Institute to improve asthma awareness, prevention and management worldwide. GINA develops and publishes evidence-based, annually updated resources for clinicians. GINA guidance is adopted by national asthma guidelines in many countries, adapted to fit local healthcare systems, practices, and resource availability. GINA is independent of industry, funded by the sale and licensing of its materials. This review summarizes key practical guidance for primary care from the 2022 GINA strategy report. It provides guidance on confirming the diagnosis of asthma using spirometry or peak expiratory flow. GINA recommends that all adults, adolescents and most children with asthma should receive inhaled corticosteroid (ICS)-containing therapy to reduce the risk of severe exacerbations, either taken regularly, or (for adults and adolescents with “mild” asthma) as combination ICS–formoterol taken as needed for symptom relief. For patients with moderate–severe asthma, the preferred regimen is maintenance-and-reliever therapy (MART) with ICS–formoterol. Asthma treatment is not “one size fits all”; GINA recommends individualized assessment, adjustment, and review of treatment. As many patients with difficult-to-treat or severe asthma are not referred early for specialist review, we provide updated guidance for primary care on diagnosis, further investigation, optimization and treatment of severe asthma across secondary and tertiary care. While the GINA strategy has global relevance, we recognize that there are special considerations for its adoption in low- and middle-income countries, particularly the current poor access to inhaled medications.
Subject terms: Asthma, Therapeutics
Introduction
Asthma affects more than a quarter of a billion people worldwide, is the most common chronic condition in childhood, and is responsible for over 1000 deaths a day, of which the majority are preventable1–4.
The Global Initiative for Asthma (GINA) was established by the World Health Organization and the US National Heart Lung and Blood institute in 1993 to improve asthma awareness, prevention, and management worldwide. GINA is independent of industry, funded by the sale and licensing of its evidence-based, annually updated reports and figures. The GINA methodology is published on its website (https://ginasthma.org/about-us/methodology).
The GINA report is a global evidence-based strategy that can be adapted for local health systems and local medicine availability. Many countries have their own national asthma guidelines, with many of these based on GINA5. However, most national guidelines are updated only infrequently, so they may not reflect current best evidence. In recent years, some countries have conducted partial updates of their asthma guidelines, by undertaking a detailed review of evidence for a limited number of clinical questions, but this process often takes several years. By contrast, the GINA strategy is updated every year based on a twice-yearly cumulative review of new evidence. Hence, even when national asthma guidelines are available, the GINA report may provide a useful resource for clinicians (both primary care and specialists) to be aware of the most recent evidence, and to understand how it can be integrated into holistic asthma care. However, when assessing and treating patients, health professionals are strongly advised to use their own professional judgment, and to take into account local and national regulations and guidelines, and the needs of the individual patient.
While the GINA strategy report is intended to have global relevance, there are particular considerations for asthma management in low- and middle-income countries6,7. Of particular concern is the widespread lack of access to affordable diagnostic tools and inhaled medications, which contributes substantially to the heavy burden of asthma mortality and morbidity seen in these countries.
At the most fundamental level, patients in many areas do not have access even to low-dose inhaled corticosteroids (ICS), which are the cornerstone of care for asthma patients of all severity.
GINA collaborates with and strongly supports the call by the International Union against Tuberculosis and Lung Diseases for a World Health Assembly Resolution on universal access to affordable and effective asthma care, as a step towards addressing these needs8.
GINA is also a partner organization in a program launched in March 2006 by the World Health Organization (WHO) and the Global Alliance against Chronic Respiratory Diseases (GARD). Through the work of GINA, and in co-operation with GARD and with the International Union Against Tuberculosis and Lung Diseases, substantial progress toward better care for all patients with asthma globally should be achieved in the next decade.
To achieve this, GINA believes that the safest and most effective approach to asthma treatment in adolescents and adults, which also avoids the consequences of starting treatment with short-acting beta2 agonists (SABA) alone, depends on access to ICS–formoterol across all asthma severity levels. With budesonide-formoterol now on the WHO essential medicines list9, the fundamental changes to treatment of mild asthma first included in the ground-breaking 2019 GINA report10 may provide a feasible solution to reduce the risk of severe exacerbations with very low dose treatment.
In this review we discuss four key concepts for asthma management in primary care: diagnosis, long-term treatment, assessment of control, and management of severe asthma. We provide the background to the latest (May 2022) update of the GINA strategy report11, with a focus on changes (Table 1) and selected recommendations that are particularly pertinent to primary care practitioners, and their rationale. The full strategy documents, podcasts, educational materials, and summary booklets are available on the GINA website (https://ginasthma.org).
Table 1.
Summary of changes in the 2022 GINA Strategy Report of particular relevance to primary care.
| Topic or section | Changes |
|---|---|
| Diagnosis of asthma |
Diagnostic testing is different depending on whether the patient is already on controller treatment or is treatment-naive or taking SABA alone (see Tables 2 and 3). Detail has been included about diagnosis and management of asthma in low-resource settings |
| Assessment of symptom control |
When assessing symptom control, record how often the patient is using their reliever inhaler (ICS–formoterol or SABA). For patients prescribed a SABA reliever, use of SABA more than two days a week should prompt review of their adherence and inhaler technique with their maintenance controller treatment. This criterion does not apply to patients using an ICS–formoterol reliever, as it is providing additional controller treatment along with the symptom relief. Dispensing of three or more SABA canisters a year (more than average 1.5 puffs/day) is associated with increased risk of severe exacerbations, and may be associated with increased risk of asthma death |
| Definition of mild asthma | GINA suggests that the term ‘mild asthma’ should generally be avoided in clinical practice where possible, because patients often assume that it means they do not need any controller treatment. However, if the term is used, explain to the patient that patients with apparently mild asthma can still have severe attacks, and that using ICS-containing treatment, especially with ICS–formoterol reliever, will markedly reduce this risk |
| GINA treatment figure for adults and adolescents | The rationale for showing two treatment tracks has been reinforced: Track 1, with as-needed ICS–formoterol as reliever across treatment steps, is preferred based on evidence for lower risk of exacerbations and similar or better symptom control compared with using SABA as reliever |
| Treatment figure for children 6–11 years | The figure has been updated to explain the “other controller options” and new Step 5 options for this age group |
| Adding LAMA to ICS-LABA for adults and adolescents (Step 5) | Patients with exacerbations despite ICS-LABA should receive at least medium dose ICS-LABA before considering add-on LAMA |
| Difficult-to-treat and severe asthma in adults and adolescents |
The GINA Guide and decision tree for assessment and management of difficult-to-treat and severe asthma in adults and adolescents has been revised and enlarged. Additional investigations have been suggested for patients with difficult-to-treat asthma and blood eosinophils ≥300/μL, including investigating for non-asthma causes such as Strongyloides, which is often asymptomatic. New biologic treatment options have been approved for severe asthma and are available in many countries, so referral to a specialist is recommended if asthma is poorly controlled despite Step 4 treatment |
| Maintenance oral corticosteroids—consider only as last resort | Because of the risk of serious long-term adverse effects, maintenance OCS should be considered only as a last resort in any age group |
| Written asthma action plans (handwritten, printed, digital, or pictorial) | Give patients documented instructions about how to change their medications when their asthma worsens, and when to seek medical advice. Verbal instructions are often forgotten |
| Management of wheezing episodes in pre-school children | In children ≤5 years with intermittent viral wheezing and no or few interval respiratory symptoms, consideration of intermittent short-course ICS has been added to the treatment figure. It should be considered only if the physician is confident that it will be used appropriately, because of the risk of side effects |
| Management of acute asthma in healthcare settings | After an Emergency Department visit or hospitalization, make sure patients are returned to as-needed (rather than regular) reliever use. For patients using ICS–formoterol as their reliever, make sure that they switch back to this after any acute healthcare presentation |
Modified with permission from ref. 11.
ICS inhaled corticosteroid, LABA long-acting beta2 agonist, LAMA long-acting muscarinic antagonist, SABA short-acting beta2 agonist, OCS oral corticosteroid.
Diagnosis of asthma
It is critical to confirm the diagnosis of asthma
Primary care clinicians are consulted by patients with many hundreds of different medical conditions in any year. Every day, they are faced with the challenge of quickly arriving at an accurate diagnosis in limited time, and often with limited access to specialized investigations.
In order to ensure diagnosis of asthma is considered as early as possible, clinicians should maintain a high index of suspicion when patients present with respiratory symptoms12.
Over- and under-diagnosis of asthma are common and are usually due to the lack of objective lung function testing which can demonstrate variable expiratory airflow limitation that will support the diagnosis of asthma and help to exclude other causes13,14. For continuity of care, it is important to ensure that the diagnosis is recorded in each patient’s medical record, detailing the basis for the diagnosis, including objective measurements of variable airflow obstruction and airway inflammation, if available. These details are often lacking in the medical records of children15 and adults treated for asthma16,17.
Medical records should also contain details of treatment prescribed, education given to help patients understand the chronic nature of their disease, and provision of a personal written action plan to enable them to change their treatment and seek assistance when needed.
Diagnosing asthma in adults, adolescents and children aged 6–11 years
Confirm the diagnosis of asthma before starting controller treatment, if possible
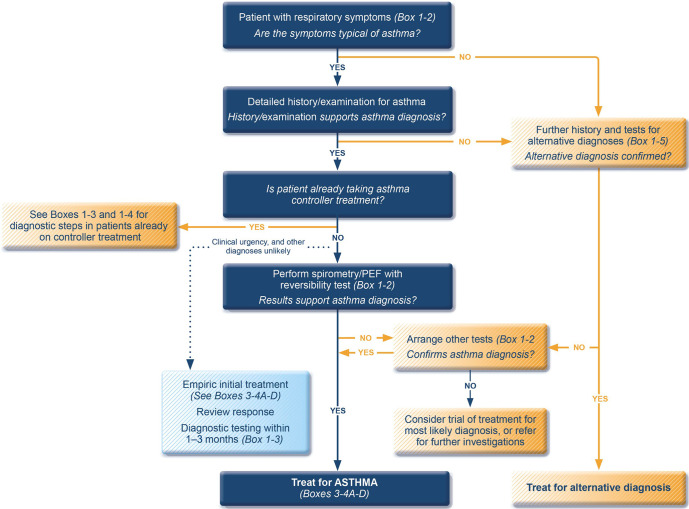
There is no single test for confirming the diagnosis of asthma. First, a clinical diagnosis starts with a history of respiratory symptoms (such as cough, wheeze, difficulty breathing and/or shortness of breath) that typically vary over time and intensity (Table 2 and Fig. 1). Symptoms of asthma are often worse at night and in the early morning, and may be triggered by factors such as viral infections, allergen exposure, exercise, strong smells, cigarette smoke, exhaust fumes and laughter. When taking a history, it may be helpful to show patients or carers a video depicting typical symptoms, such as the one developed by Wellington School of Medical and Health Sciences, University of Otago, New Zealand, available from the Global Asthma Network website (http://globalasthmanetwork.org/surveillance/manual/Asthma_AVQ3.1.mp4). Physical examination may be entirely normal.
Table 2.
Diagnostic criteria for asthma in adults, adolescents, and children 6–11 years.
| 1. HISTORY OF VARIABLE RESPIRATORY SYMPTOMS | |
| Feature | Symptoms or features that support the diagnosis of asthma |
|
Wheeze, shortness of breath, chest tightness and cough (Descriptors may vary between cultures and by age) |
• More than one type of respiratory symptom (in adults, isolated cough is seldom due to asthma) • Symptoms occur variably over time and vary in intensity • Symptoms are often worse at night or on waking • Symptoms are often triggered by exercise, laughter, allergens, cold air • Symptoms often appear or worsen with viral infections |
| 2. CONFIRMED VARIABLE EXPIRATORY AIRFLOW LIMITATION | |
| Feature | Considerations, definitions, criteria |
| 2.1 Documented* expiratory airflow limitation | At a time when FEV1 is reduced, confirm that FEV1/FVC is reduced compared with the lower limit of normal (it is usually >0.75–0.80 in adults, >0.90 in children) |
| AND | |
| 2.2 Documented* excessive variability in lung function* (one or more of the following): | The greater the variations, or the more occasions excess variation is seen, the more confident the diagnosis. If initially negative, tests can be repeated during symptoms or in the early morning. |
| • Positive bronchodilator (BD) responsiveness (reversibility) test |
Adults: increase in FEV1 of >12% and >200 mL (greater confidence if increase is >15% and >400 mL). Children: increase in FEV1 by >12% predicted Measure change 10–15 min after 200–400 mcg salbutamol (albuterol) or equivalent, compared with pre-BD readings. Positive test more likely if BD withheld before test: SABA ≥ 4 h, twice-daily LABA 24 h, once-daily LABA 36 h |
| • Excessive variability in twice-daily PEF over 2 weeks |
Adults: average daily diurnal PEF variability >10%a Children: average daily diurnal PEF variability >13%a |
| • Significant increase in lung function after 4 weeks of anti-inflammatory treatment | Adults: increase in FEV1 by >12% and >200 mL (or PEF2 by >20%) from baseline after 4 weeks of treatment, outside respiratory infections |
| • Positive exercise challenge test |
Adults: fall in FEV1 of >10% and >200 mL from baseline Children: fall in FEV1 of >12% predicted, or PEF >15% |
| • Positive bronchial challenge test (usually only for adults) | Fall in FEV1 from baseline of ≥20% with standard doses of methacholine, or ≥15% with standardized hyperventilation, hypertonic saline or mannitol challenge |
| • Excessive variation in lung function between visits (good specificity but poor sensitivity) |
Adults: variation in FEV1 of >12% and >200 mL between visits, outside of respiratory infections Children: variation in FEV1 of >12% in FEV1 or >15% in PEFb between visits (may include respiratory infections) |
Source: Box 1–2 in GINA 2022. Reproduced with permission from ref. 11.
BD bronchodilator (SABA or rapid-acting LABA), FEV1 forced expiratory volume in 1 s, ICS inhaled corticosteroid, LABA long-acting beta2 agonist, PEF peak expiratory flow (highest of three readings), SABA short-acting beta2 agonist.
aDaily diurnal PEF variability is calculated from twice daily PEF as (day’s highest minus day’s lowest) divided by (mean of day’s highest and lowest), averaged over 1 week.
bUse the same PEF meter each time, as PEF may vary by up to 20% between different meters.
Fig. 1. The GINA diagnostic flowchart 2022.
PEF peak expiratory flow. Source: Box 1–1 in GINA report 2022. Box numbers within the figure refer to the GINA 2022 report. Reproduced with permission from ref. 11.
Variable expiratory airflow limitation is the other cardinal feature of untreated asthma. In a patient with a history suggestive of asthma, the diagnosis of asthma is supported by an increase in forced expiratory volume in 1 s (FEV1) recorded by spirometry 15 min after administration of bronchodilator: in adults/adolescents, by an increase of more than 200 mL and 12% from the pre-bronchodilator (baseline) FEV1; in children, by an increase from baseline of more than 12% of the predicted FEV1 value.
Since asthma is a variable condition, bronchodilator reversibility (also called responsiveness) may or may not be present at the time of initial lung function testing. If it is not documented on spirometry at an initial attempt, the test should be repeated at one or more later visits, preferably when the patient is symptomatic and bronchodilator medicines have been withheld. Otherwise, an alternative test may be conducted (as below and in Table 2).
Spirometry is not always accessible in primary care. An alternative method is to instruct the patient to record peak expiratory flow (PEF) each morning and evening over a 2-week period in a diary or using an electronic peak flow meter. PEF should be measured three times on each occasion, and only the highest reading used. Diurnal PEF variability is calculated as each day’s highest minus the day’s lowest reading, divided by the mean of the day’s highest and lowest, then these results are averaged over one week. Excessive diurnal PEF variability is defined as a mean variability of >10% in PEFs in adults or >13% variability in children. When measuring PEF, the same meter should be used for all readings, as variation between different PEF meters may be as large as 20%.
In people with suspected asthma who have normal expiratory airflow and no significant reversibility, a bronchoprovocation test (e.g., methacholine or mannitol) can reveal airway hyperresponsiveness, supporting a diagnosis of asthma. Bronchodilators must be withheld before challenge testing.
Variable expiratory airflow limitation should preferably be demonstrated before initiating asthma controller treatment, except in situations of clinical urgency, as it becomes harder to confirm the diagnosis once controller treatment has been started (Table 3). However, the diagnosis of asthma can also be confirmed if there is a clinically significant improvement in FEV1 (by >12% and >200 mL) or in PEF by >20% after 4 weeks of inhaled corticosteroid (ICS) treatment.
Table 3.
Steps for confirming the diagnosis of asthma in a patient already taking controller treatment.
| Current status | Steps to confirm the diagnosis of asthma |
|---|---|
| Variable respiratory symptoms and variable airflow limitation | Diagnosis of asthma is confirmed. Assess the level of asthma control (Box 2–2) and review controller treatment (Box 3–5). |
| Variable respiratory symptoms but no variable airflow limitation |
Consider repeating spirometry after withholding BD (4 h for SABA, 24 h for twice-daily ICS-LABA, 36 h for once-daily ICS-LABA) or during symptoms. Check between-visit variability of FEV1, and bronchodilator responsiveness. If still normal, consider other diagnoses (Box 1–5). If FEV1 is >70% predicted: consider stepping down controller treatment (see Box 1–5) and reassess in 2–4 weeks, then consider bronchial provocation test or repeating BD responsiveness. If FEV1 is <70% predicted: consider stepping up controller treatment for 3 months (Box 3–5), then reassess symptoms and lung function. If no response, resume previous treatment and refer patient for diagnosis and investigation. |
| Few respiratory symptoms, normal lung function, and no variable airflow limitation |
Consider repeating BD responsiveness test again after withholding BD as above or during symptoms. If normal, consider alternative diagnoses (Box 1–5). Consider stepping down controller treatment (see Box 1–5): • If symptoms emerge and lung function falls: asthma is confirmed. Step up controller treatment to previous lowest effective dose. • If no change in symptoms or lung function at lowest controller step: consider ceasing controller, and monitor patient closely for at least 12 months (Box 3–7). |
| Persistent shortness of breath and persistent airflow limitation | Consider stepping up controller treatment for 3 months (Box 3–5), then reassess symptoms and lung function. If no response, resume previous treatment and refer patient for diagnosis and investigation. Consider asthma–COPD overlap (Chapter 5). |
“Variable airflow limitation” refers to expiratory airflow. GINA recommendations for confirming the diagnosis in those already started on controller treatment. Source: Box 1–3 in GINA 2022. Box and chapter numbers refer to the GINA 2022 report. Reproduced with permission from ref. 11.
BD bronchodilator, COPD chronic obstructive pulmonary disease, FEV1 forced expiratory volume in 1 s, ICS inhaled corticosteroid, LABA long-acting beta2 agonist, SABA short-acting beta2 agonist.
A history or family history of allergic rhinitis or atopic dermatitis, or the presence of atopy (demonstrated by either a positive skin prick test or specific IgE to one or more aeroallergens) increases the chance that a patient with respiratory symptoms has allergic asthma, but these features are not specific for asthma, and asthma may be non-allergic.
Evidence of Type 2 inflammation (for example, fractional exhaled nitric oxide [FeNO] >25 ppb or blood eosinophils >300/μL) is found in some types of asthma, but also in several non-asthma conditions such as allergic rhinitis and eosinophilic bronchitis. Therefore, the presence or absence of these biomarkers cannot confirm or exclude a diagnosis of asthma, particularly if measured after starting ICS treatment. However, in patients with severe asthma, FeNO and blood eosinophils are useful to select and guide treatment.
If symptoms persist or are more typical of an alternative diagnosis, or if the patient experiences no benefit after commencement of controller therapy, the diagnosis should be reviewed, and alternative causes of the symptoms should be considered (Fig. 2).
Fig. 2. Investigating poor symptom control and/or exacerbations despite treatment.

ICS inhaled corticosteroid, NSAID nonsteroidal anti-inflammatory drug, GERD gastro-esophageal reflux disease. Reproduced with permission from ref. 11.
Consider occupational asthma in patients presenting with adult-onset asthma
Occupational asthma should be considered in anyone newly presenting in adulthood with symptoms suggestive of asthma, particularly if there is improvement when away from work. If occupational asthma is suspected, early referral to a specialist (if available) is important, to assist with assessment of the person’s work environment and confirm the diagnosis. Exposure to the sensitizing agent should cease if at all possible, because ongoing exposure to even low levels can lead to severe problems. The specialist may be able to assist with negotiation with employers to reduce/cease exposure and, where relevant, with recommendations for compensation in accordance with applicable local employment laws. Patients with adult-onset asthma should also be asked about exposure to sensitizers or irritants in non-work locations, e.g., use of cleaning agents at home, or hobbies such as woodworking.
Persistent airflow obstruction may develop over time—so it is important to differentiate asthma from chronic obstructive pulmonary disease (COPD)
The history and pattern of symptoms and past records can help to distinguish asthma with persistent airflow limitation from COPD. Asthma and COPD may co-exist in the same patient, particularly in smokers and the elderly.
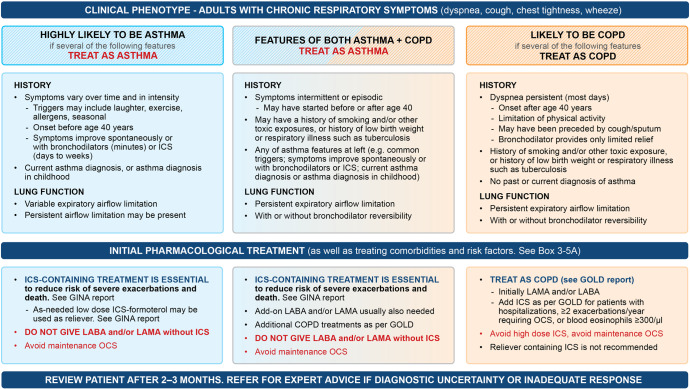
It is important to recognize features of asthma in these patients because anti-inflammatory treatment with ICS is essential in asthma (whether or not there are also features of COPD such as persistent airflow limitation) to prevent severe flare-ups (severe exacerbations) and reduce the risk of asthma-related death. Figure 3 summarizes features that are useful in distinguishing asthma from COPD.
Fig. 3. Approach to initial treatment in patients with asthma and/or COPD.
GOLD Global Initiative for Obstructive Lung Disease, ICS inhaled corticosteroid, LABA long-acting β2 agonist; LAMA long-acting muscarinic antagonist. A summary of differentiating and diagnostic features in people with Asthma, COPD and Asthma + COPD. Source: Box 5–2 in GINA 2022. Reproduced with permission from ref. 11.
Diagnosing asthma in children aged 5 years and under
It can be challenging to make the diagnosis of asthma in some children aged ≤5 years. Recurrent wheezing is very common in this age group, including in children without asthma, typically with viral upper respiratory tract infections. Routine assessment of airflow limitation or bronchodilator responsiveness in this age group is difficult and is not practical in primary care.
Asthma diagnosis in children aged ≤5 years can be based on symptom patterns, the presence of risk factors, therapeutic response to controller treatment, and exclusion of alternative diagnoses
A diagnosis of asthma in young children with a history of wheezing is more likely if they have wheezing or coughing that occurs with exercise, laughing or crying, or in the absence of an apparent respiratory infection, a history of other allergic disease (eczema, food allergy, or allergic rhinitis), atopy or asthma in first-degree relatives, clinical improvement during 2–3 months of controller treatment, and worsening after cessation.
The following questions can be used to elicit features suggestive of asthma in young children and features that help support the diagnosis:
Does your child have wheezing? Wheezing is a high-pitched noise that comes from the chest and not the throat. Use of a video questionnaire, or asking a parent to record an episode on a smartphone if available can help to confirm the presence of wheeze and differentiate from upper airway abnormalities.
Does your child wake up at night because of coughing, wheezing, or difficult breathing, heavy breathing, or breathlessness?
Does your child have to stop running, or play less hard, because of coughing, wheezing or difficult breathing, heavy breathing, or shortness of breath?
Does your child cough, wheeze or get difficult breathing, heavy breathing, or shortness of breath when laughing, crying, playing with animals, or when exposed to strong smells or smoke?
Has your child ever had eczema, or been diagnosed with allergy to foods?
Has anyone in your close family had asthma, hay fever, food allergy, eczema, or any other disease with breathing problems?
In preschool children with wheeze, phenotypes have been proposed based on short-term symptom patterns18 or on symptom pattern trends over time19–21, but these have not proved to be clinically useful or accurate in predicting asthma in later childhood.
Long-term treatment of asthma
All patients diagnosed with asthma should be treated with ICS-containing medication
GINA recommends that all adults, adolescents and children over 5 years with a diagnosis of asthma should be treated with regular or (for mild asthma) as-needed ICS-containing treatment to control symptoms and prevent flare-ups (also called exacerbations or “attacks”), and that they should be reviewed within three months after initiating and/or changing treatment. In children ≤5 years, ICS treatment is recommended if asthma is likely and the child has uncontrolled symptoms and/or ≥3 wheezing episodes/year; a trial of ICS is also recommended if the diagnosis is uncertain and symptoms occur more than every 6–8 weeks.
GINA recommends against treating asthma with SABA alone, without ICS
GINA no longer recommends treatment of asthma with SABA alone (without ICS) in adults, adolescents and children >5 years (Figs. 4–6) because of the risk of severe asthma flare-ups (severe exacerbations) requiring emergency department presentation or hospitalization, and asthma-related death. These risks are markedly reduced by ICS-containing therapy22,23. Treating with ICS also substantially reduces the need for courses of oral corticosteroids, thereby reducing the cumulative risk of long-term adverse effects such as osteoporosis and cataract from even occasional courses of oral corticosteroids24. A further reason to avoid treating asthma with SABA alone is because their quick symptom relief may instill a false sense of security in patients, who may incorrectly assume that these medicines alone are a sufficient treatment for asthma. In addition, regular use of SABA (e.g., 2–4 times daily for as little as 1–2 weeks) increases airway hyperresponsiveness and airway inflammation25,26, and overuse of SABA (indicated by dispensing of ≥3 200-dose canisters in a year, or daily use), is associated with an increased risk of severe exacerbations and death, even in patients also taking ICS27–29.
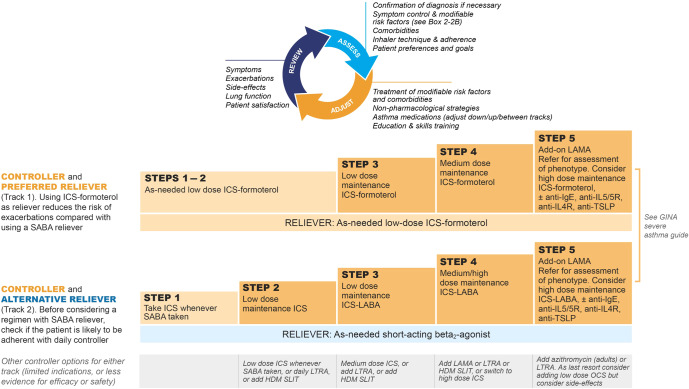
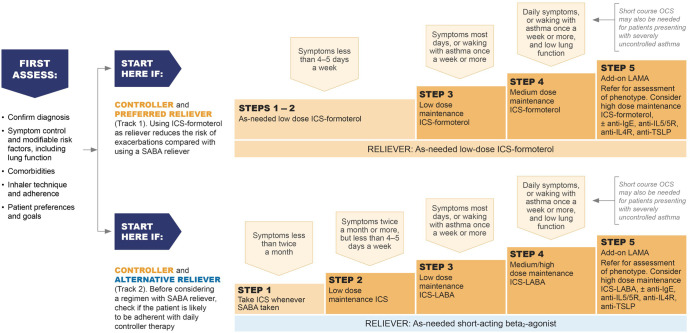
Fig. 4. Two-track options for personalized management of asthma for adults and adolescents, to control symptoms and minimize future risk.
HDM house dust mite, ICS inhaled corticosteroid, LABA long-acting beta2 agonist, LAMA long-acting muscarinic antagonist, LTRA leukotriene receptor antagonist, OCS oral corticosteroids, SABA short-acting beta2 agonist, SLIT sublingual immunotherapy. Box number refers to the GINA 2022 report. Before starting, stepping up or down or switching between tracks, patients should be assessed using the “assess, adjust, review” cycle shown at the top of the figure. Refer to the GINA report for more information about Step 5 options, including biologic therapies for patients with severe asthma. Source: Box 3–5A in GINA report 2022. Reproduced with permission from ref. 11.
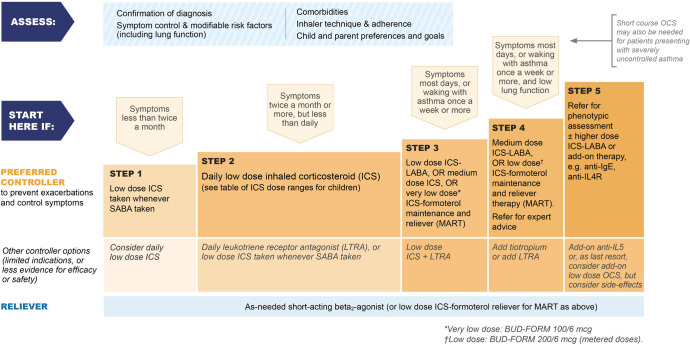
Fig. 6. Initial medications for children aged 6–11 years diagnosed with asthma.
BUD-FORM budesonide–formoterol, ICS inhaled corticosteroid, LABA long-acting beta2 agonist, LTRA leukotriene receptor antagonist, MART maintenance and reliever therapy with ICS–formoterol, OCS oral corticosteroids, SABA short-acting beta2 agonist. Initial treatment for children aged 6–11 years diagnosed with asthma, with guidance on initial levels of medication for each treatment track based on symptoms and lung function where appropriate. Source: Box 3–4Di in GINA report 2022. Refer to the GINA report for other treatment components, including treatment of modifiable risk factors and comorbidities, non-pharmacologic strategies, and education and skills training. Reproduced with permission from ref. 11.
Compared with as-needed SABA, as-needed low-dose ICS–formoterol for symptom relief reduces the risk of severe asthma flare-ups (severe exacerbations) across all levels of asthma severity—either as-needed only in mild asthma or in addition to maintenance ICS–formoterol
GINA’s current recommendations for the pharmacotherapy of asthma in adults and adolescents are shown in two ‘tracks’ (Figs. 4–5). There is strong evidence favoring the Track 1 option, in which low-dose ICS–formoterol is the reliever across all treatment steps, compared with Track 2, in which SABA is the reliever23,30–39.
Fig. 5. Initial medications for adults and adolescents diagnosed with asthma.
ICS inhaled corticosteroid, LABA long-acting beta2 agonist, LAMA long-acting muscarinic antagonist, MART maintenance and reliever therapy with ICS–formoterol, OCS oral corticosteroids, SABA short-acting beta2 agonist. Initial medications for adults and adolescents diagnosed with asthma, with guidance on initial levels of medication for each treatment track based on symptoms and lung function where appropriate. Refer to the GINA report for other treatment components, including treatment of modifiable risk factors and comorbidities, non-pharmacologic strategies, and education and skills training. Source: Box 3.4Bi in GINA report 2022. Reproduced with permission from ref. 11.
This recommendation is based on multiple studies demonstrating that combination low-dose ICS and formoterol, taken as-needed for relief of asthma symptoms (either as-needed only in mild asthma, or in addition to maintenance ICS–formoterol), is a more effective and safer reliever than as-needed SABA.
In patients with mild asthma, as-needed ICS–formoterol reduces the risk of severe flare-ups by 60–64% compared with as-needed SABA36,37. Compared with low-dose maintenance ICS plus as-needed SABA, the risk of severe exacerbations is similar35–38. In a Cochrane systematic review and meta-analysis (n = 9565), patients with mild asthma treated with as-needed ICS–formoterol had a 55% reduction in severe exacerbations and 65% lower emergency department visits or hospitalizations compared with SABA alone. In addition, those treated with as-needed ICS–formoterol had 37% lower risk of emergency department visits or hospitalizations than with daily ICS plus as-needed SABA23. In some of these studies, there were small differences in lung function (FEV1) and symptom control assessed by Asthma Control Questionnaire (ACQ-5) score that favored daily maintenance ICS over as-needed-only low-dose ICS–formoterol. These differences were not clinically important, and may reflect that adherence with maintenance ICS was much higher than is usually achievable in clinical practice. The average daily dose of ICS was much lower with as-needed ICS–formoterol compared with daily ICS plus as-needed SABA.
Further, in patients with moderate-to-severe asthma (Steps 3 and 4, Figs. 4–5), use of ICS–formoterol as both maintenance and reliever therapy (MART) in Track 1 reduces the risk of severe flare-ups (severe exacerbations), compared with taking the same or higher dose of ICS or a combination of ICS and a long-acting beta2 agonist (LABA) plus SABA reliever30,31. In Steps 3 and 4, symptom control and lung function with MART are the same or better compared with use of a SABA reliever.
Although Track 1 is preferred because of the significant reduction in severe exacerbations, Track 2 (with SABA as reliever) is an alternative option if ICS–formoterol is not available or if patients have no risk factors for exacerbations and have good adherence with regular controller therapy. However, before prescribing Track 2 therapy with a SABA reliever, the clinician should assess whether the patient is likely to continue to be adherent with daily controller treatment, as otherwise they will be taking SABA alone, with an increased risk of severe exacerbations.
At the time of publishing, over 45 countries have licensed ICS–formoterol for as-needed use in mild asthma and over 120 countries have licensed prescription of MART in moderate-to-severe asthma (personal communications). Detailed practical advice on the implementation of MART in clinical practice has recently been published40,41, including downloadable resources (ICS–formoterol dosing and SMART action plan).
GINA asthma treatment is not “one size fits all”
Because asthma is a chronic condition prone to flare-ups, GINA emphasizes that patients need regular review, assessment and adjustment. This involves assessment of asthma control, individual risk factors and comorbidities, with review and optimization of treatment, including careful attention to adherence and inhaler technique, and provision of individualized self-management education including a written/pictorial action plan.
Management of co-morbid conditions that may worsen asthma control, increase the risk of severe flare-ups and/or complicate treatment should be optimized. These comorbidities include obesity, chronic rhinosinusitis, obstructive sleep apnea, gastro-esophageal reflux disease, and mental health problems (Fig. 7).
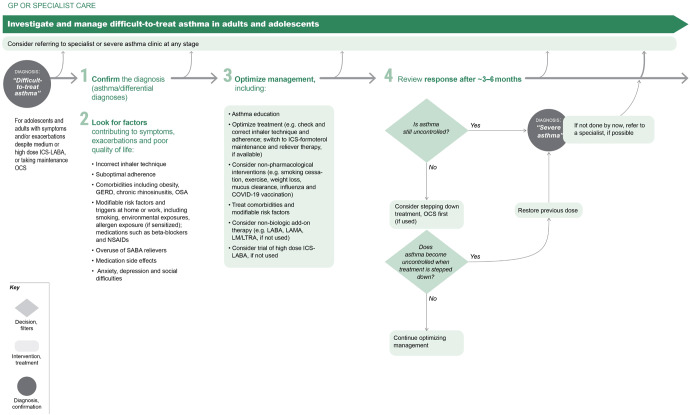
Fig. 7. Investigating and managing difficult-to-treat asthma in adult and adolescent patients.
ICS inhaled corticosteroid, LABA long-acting beta2 agonist, LAMA long-acting muscarinic antagonist, LTRA leukotriene receptor antagonists, SABA short-acting beta2 agoinst, OCS oral corticosteroid. The GINA strategy includes a decision tree about the management of difficult-to-treat and severe asthma spanning primary through tertiary care. The section of the flow diagram applicable to generalists in primary and secondary care is shown here. Source: Box 3–16A in GINA report 2022. Reproduced with permission from ref. 11.
Treatment should be reviewed after any flare-ups or changes in treatment (Fig. 2). The components of these assessments are summarized in the personalized asthma management cycle (Assess, Adjust, Review) shown at the top of Fig. 4, which guides clinicians in personalized asthma review and adjustment of treatment. This approach emphasizes the principle that asthma treatment is not ‘one size fits all’.
Management of each patient’s individual risk factors and comorbidities may include both pharmacologic and non-pharmacologic strategies. Non-pharmacologic strategies may include smoking cessation advice, breathing exercises, weight reduction, avoiding air pollution and allergens, appropriate immunizations as well as strategies for dealing with emotional stress. In addition, it is essential to ensure patients can use their prescribed inhaler correctly with reinforcement of approved local videos (e.g., https://www.nationalasthma.org.au/living-with-asthma/how-to-videos). In patients with severe asthma, assessment of inflammatory biomarkers (blood eosinophils and/or FeNO) is important for guiding selection and adjustment of asthma treatment.
A written or pictorial action plan on the management of asthma exacerbations should be provided to every patient. The action plan should be appropriate for the patient’s level of literacy and health literacy, and their treatment regimen. Examples of action plans, including for patients using ICS–formoterol reliever as in GINA Track 1, are available at https://www.nationalasthma.org.au/health-professionals/asthma-action-plans/asthma-action-plan-library. The risk of adverse effects of medications can be reduced by optimizing inhaler technique and adherence, stepping down ICS dose when asthma has been well-controlled for 2–3 months, by referring patients for specialist review (if available) if asthma is not well controlled despite medium or high dose ICS-LABA, and by identifying patients with SABA overuse who may be potentially switched to GINA Track 1 with an ICS–formoterol reliever.
Figures 5 and 6 summarize the GINA options for initial asthma medications in adults, adolescents and children 6–11 years newly diagnosed with asthma. Once treatment has been initiated, ongoing medication decisions are based on the same personalized cycle, in which treatment is stepped up and down according to the patient’s needs within a track, using the same reliever. Treatment can also be switched between tracks according to patient needs and preferences. Before any step-up (Fig. 2), it is essential to check adherence to treatment, inhaled technique, relevant comorbidities and risk factors, and environmental factors affecting asthma (Supplementary Fig. 1).
Assessment of asthma control in two domains: symptoms and risk factors
GINA defines asthma control in two domains: (i) current symptom control and (ii) risk factors for future poor asthma outcomes
People with asthma should be assessed regularly, including after flare-ups. Unfortunately, in many cases, asthma is managed as though it were an acute illness; patients are treated for flare-ups and then sent home without follow-up42–46.
Patient-reported tools for assessing asthma symptom control (e.g., Asthma Control Questionnaire, Asthma Control Test, Childhood Asthma Control Test) reflect only the past 1–4 weeks, and therefore provide only a snapshot of recent symptoms, not overall asthma control.
Poor symptom control is associated with an increased risk of asthma flare-ups. However, people with good symptom control or seemingly mild asthma can still be at risk of severe flare-ups (severe exacerbations)47, and even death48. GINA therefore recommends that asthma control should be assessed in two domains: (i) current symptom control and (ii) risk factors for future poor asthma outcomes, particularly exacerbations (Supplementary Fig. 1).
Supplementary Fig. 1 summarizes how to assess symptom control and provides a list of modifiable risk factors for exacerbations that are independent of the level of symptom control. This means that even if someone has no current or recent symptoms at the time of assessment, they may still be at risk of asthma flare-ups. Treatment of modifiable risk factors may include, for example, correcting inhaler technique, reducing exposure to tobacco smoke, strategies for weight reduction, allergen immunotherapy and/or allergen avoidance in sensitized patients, and arranging mental health support.
Table 4 summarizes specific questions to be addressed when assessing asthma control in children 6–11 years.
Table 4.
Specific questions to ask when assessing children 6–11 years with asthma.
| Asthma symptom control | |
| Day symptoms | Ask: How often does the child have cough, wheeze, dyspnea or heavy breathing (number of times per week or day)? What triggers the symptoms? How are they handled? |
| Night symptoms | Cough, awakenings, tiredness during the day? (If the only symptom is cough, consider other diagnoses such as rhinitis or gastroesophageal reflux disease). |
| Reliever use | How often is reliever medication used? (check date on inhaler or last prescription) Distinguish between pre-exercise use (sports) and use for relief of symptoms. |
| Level of activity | What sports/hobbies/interests does the child have, at school and in their spare time? How does the child’s level of activity compare with their peers or siblings? How many days is the child absent from school? Try to get an accurate picture of the child’s day from the child without interruption from the parent/carer. |
| Risk factors for adverse outcomes | |
| Exacerbations | Ask: How do viral infections affect the child’s asthma? Do symptoms interfere with school or sports? How long do the symptoms last? How many episodes have occurred since their last medical review? Any urgent doctor/emergency department visits? Is there a written action plan? Risk factors for exacerbations include a history of exacerbations, poor symptom control, poor adherence and poverty, and persistent bronchodilator reversibility even if the child has few symptoms. |
| Lung function | Check curves and technique. Main focus is on FEV1 and FEV1/FVC ratio. Plot these values as percent predicted to see trends over time. |
| Side-effects | Check the child’s height at least yearly, as poorly controlled asthma can affect growth, and growth velocity may be lower in the first 1–2 years of ICS treatment. Ask about frequency and dose of ICS and OCS. |
| Treatment factors | |
| Inhaler technique | Ask the child to show how they use their inhaler. Compare with a device-specific checklist. |
| Adherence | Is there any controller medication in the home at present? On how many days does the child use their controller in a week (e.g. 0, 2, 4, 7 days)? Is it easier to remember to use it in the morning or evening? Where is inhaler kept – is it in plain view to reduce forgetting? Check date on inhaler. |
| Goals/concerns | Does the child or their parent/carer have any concerns about their asthma (e.g. fear of medication, side-effects, interference with activity)? What are the child’s/parent’s/carer’s goals for treatment? |
| Comorbidities | |
| Allergic rhinitis | Itching, sneezing, nasal obstruction? Can the child breathe through their nose? What medications are being taken for nasal symptoms? |
| Eczema | Sleep disturbance, topical corticosteroids? |
| Food allergy | Is the child allergic to any foods? (confirmed food allergy is a risk factor for asthma-related death) |
| Obesity | Check age-adjusted BMI. Ask about diet and physical activity. |
| Other investigations (if needed) | |
| 2-week diary | If no clear assessment can be made based on the above questions, ask the child or parent/carer to keep a daily diary of asthma symptoms, reliever use and peak expiratory flow (best of three) for 2 weeks (Appendix Chapter 4). |
| Exercise challenge (laboratory) | Provides information about airway hyperresponsiveness and fitness (Box 1–2). Only undertake a challenge if it is otherwise difficult to assess asthma control. |
Source: Box 2–3 in GINA 2022. Box and appendix numbers refer to GINA 2022 report. Reproduced with permission from ref. 11.
FEV1 forced expiratory volume over 1 s, FVC forced vital capacity, ICS inhaled corticosteroid, OCS oral corticosteroid.
Difficult-to-treat and severe asthma
Refer people with severe asthma to a respiratory specialist, if possible
Difficult-to-treat asthma is defined as asthma that is uncontrolled despite prescribing of medium- or high-dose ICS with a second controller (usually a LABA) or with maintenance oral corticosteroids, or that requires high-dose ICS to maintain good asthma control. For many such patients, their asthma can be well controlled by optimizing care, including identifying and addressing modifiable risk factors listed in Figs. 2 and 7 and Supplementary Fig. 1. Poor adherence and incorrect inhaler technique are particularly common contributors to poor asthma control.
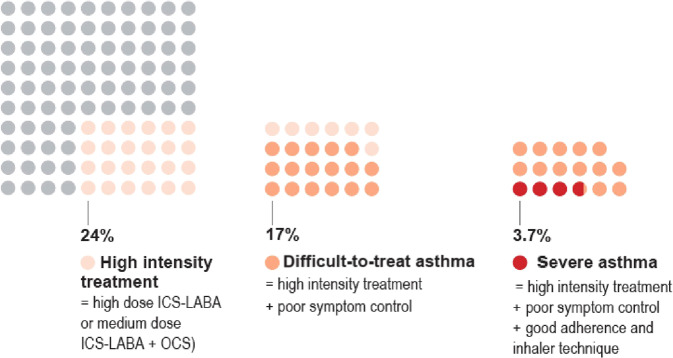
Severe asthma is a subset of difficult-to-treat asthma (Fig. 8). Severe asthma is defined as asthma that is uncontrolled despite adherence with optimized high-dose ICS-LABA treatment with correct inhaler technique and management of contributory factors such as comorbidities and environment exposures, or that worsens when the dose of ICS-LABA is reduced.
Fig. 8. Proportion of adults with difficult-to-treat or severe asthma.
Severe asthma is a subset of those with “difficult-to-treat” asthma. Source: Box 3–15 in GINA report 2022, data from Hekking et al.49. Reproduced with permission from ref. 11.
Based on a study in the Netherlands, about 3–4% of people with asthma are estimated to have severe asthma49, but many more patients have difficult-to-treat asthma, that could be improved by referral for specialist assessment and treatment50. While a small proportion of people with asthma have severe disease, they contribute towards a disproportionately high level of morbidity, mortality and healthcare costs51,52.
While most people with asthma can be managed in primary care, it can be challenging to identify those at risk of poor outcomes, and especially those with severe asthma. This difficulty is partly due to the nature of primary care, where large numbers of patients present with many different and often previously undiagnosed medical conditions, there can be severe time pressures, resources may be limited, and at follow-up a patient may see different healthcare professionals with varying levels of expertise or training about asthma. Furthermore, medical records may be poor or incomplete, making it difficult to form a perspective of long-term control and efficacy of treatments, and specifically correctly identifying those that may benefit from specialist referral.
When asthma is poorly controlled despite medium or high dose ICS-LABA, the patient should be reassessed. This involves first ensuring that the diagnosis of asthma has been confirmed and relevant comorbidities and risk factors managed, that ICS have been prescribed, and that asthma treatment has been optimized; that is, that the patient is collecting and using the medication and that they are satisfied with53 and are able to use their inhaler correctly54,55.
Figure 7 shows other factors and interventions that can also be considered in primary care. If asthma remains uncontrolled, there are several reasons why these people should be referred (if possible) for expert assessment, advice and/or provision of medication, and for guidance on ongoing primary care management. In addition to confirming the diagnosis, specialist asthma services have knowledge of, and access to, newer and specific treatment including the latest range of biologic treatments (monoclonal antibodies for severe asthma). They may also have access to specialist nursing, pharmacists, counseling and psychology expertise and the facilities to provide long-term follow-up and access to consistent support from liaison nurses.
While some primary care clinics may have such expertise and resources, most do not. A number of UK coronial inquests on asthma deaths in children concluded that lack of access to continuity of care contributed to these deaths42–44.
The section of the GINA 2022 report on severe asthma diagnosis and management spans the roles of clinicians ranging from primary to tertiary care. Figure 7 summarizes the initial approach to these patients in a primary care setting. The full severe asthma recommendations (including for biologic therapy) as well as a summary booklet are also available on the GINA website.
While most patients’ asthma can be managed in primary care, specialist opinion and treatment is strongly recommended (where available) in some situations:
when the diagnosis is difficult; specialists will have access to more sophisticated investigations and resources for confirming or excluding a diagnosis of asthma;
when there is failure to control symptoms despite adequate therapy, good adherence and good inhaler technique;
when severe asthma is suspected, for characterization of phenotype and for consideration of biologic therapy, depending on availability. For example, primary care physicians should consider referral for patients taking maintenance oral corticosteroids and those who have had two or more courses of oral corticosteroids for acute exacerbations in the previous year, and those who have poorly controlled asthma despite step 4 treatment;
when symptoms suggest complications or comorbidities such as aspirin-exacerbated respiratory disease, allergic bronchopulmonary aspergillosis;
when occupational asthma is suspected;
when a patient has a history of a life-threatening asthma attack, or has confirmed or suspected food allergy as well as asthma.
Conclusion
In summary, the GINA strategy emphasizes that asthma should be considered in the differential diagnosis of anyone presenting with respiratory symptoms, particularly if recurrent and varying in severity. Where possible, the diagnosis of asthma should be confirmed with lung function testing before initiating controller treatment. Asthma control should be assessed in two domains: current symptom control and risk factors for future asthma flare-ups (exacerbations), which include having had a flare-up in the previous 12 months. Asthma treatment for all patients should include ICS: either regularly or (in mild asthma) as needed whenever symptom reliever is taken.
Optimization of asthma treatment includes education and skills training for inhaler technique and adherence, and provision of a written/pictorial asthma action plan. Failure to successfully optimize care in people with severe or difficult-to-treat asthma should prompt careful reassessment—if available, by a specialist with appropriate facilities for diagnosis and interdisciplinary treatment. Collaboration between primary care doctors and respiratory physicians is a key factor in effective asthma management.
Supplementary information
Acknowledgements
The authors thank Rebecca Decker, GINA Executive Director, and Kristi Rurey, GINA Project Manager, for supporting and coordinating the GINA Science Committee and Board, Kate Chisnall for assistance with graphics, and Jennifer Harman for editorial assistance.
Author contributions
All authors have been involved in decisions when updating the 2022 published GINA strategy report. All authors were involved in the design and oversight of this manuscript. M.L.L. and H.K.R. had a lead role in drafting and writing the manuscript and all authors were involved in contributing to and agreeing the final submitted document.
Data availability
No datasets were generated or analyzed during the current study.
Competing interests
J.D. declares no competing interests. The other authors report the following conflicting interests (including financial and non-financial): M.L.L. has received consultancy fees from Clement Clarke International, Boehringer Ingelheim, AstraZeneca, GSK, Orion, TEVA pharmaceuticals, Menarini, NSHI, Chiesi Pharmaceuticals, Novartis, Aspire, Respiri Ltd (Australia), and Smart Respiratory Products Ltd, speaker fees from TEVA, Novartis, Orion, AstraZeneca, NAPP, Chiesi, and NSHI, and honoraria from ADMIT Group - Consorzio Ferrara Ricerche. M.L.L. is Editor Emeritus of npj Primary Care Respiratory Medicine, but was not involved in the journal’s review of, or decisions related to, this manuscript. L.B.B. has received grants from NIH/NIAID and NHLBI, personal fees from GlaxoSmithKline Genentech/Novartis, DBV Technologies, Teva, Boehringer Ingelheim, AstraZeneca, WebMD/Medscape, Sanofi, Regeneron, Vectura, Circassia, Kinaset, Vertex, and OM Pharma, and royalties from Elsevier outside the submitted work. E.B. has received speaker honoraria from AstraZeneca, Boehringer Ingelheim, Cipla, Chiesi, Hikma pharma, Menarini, Novartis, Sanofi Genzyme, and Regeneron, and consulting fees from AstraZeneca, Sanofi Genzyme, Regeneron, and Novartis. L.-P.B. has received research grants for participation in multicenter studies from Amgen, AstraZeneca, GlaxoSmithKline, Merck, Novartis, Sanofi-Regeneron, and BioHaven, research funding for projects introduced by the investigator from AstraZeneca, GlaxoSmithKline, and Merck, consulting and advisory board fees from Astra Zeneca, Novartis, GlaxoSmithKline, Merck, and Sanofi-Regeneron, lecture fees from AstraZeneca, Covis, Cipla, GlaxoSmithKline, Novartis and Merck, and Sanofi, and non-profit grants to produce educational materials from AstraZeneca, Covis, GlaxoSmithKline, Merck, and Novartis. C.B. has received consultancy fees or grants (paid to Institution) from GSK, AZ, BI, Novartis, Chiesi, Genentech, Roche, Sanofi, Regeneron, Mologic, and 4DPharma. G.B. has received speaker fees and advisory board fees from Astra Zeneca, Boehringer-Ingelheim, Chiesi, GSK, Merck Sharp & Dohme, Novartis, and Sanofi Regeneron. R.B. has received consulting fees or honoraria for lectures from ALK, AstraZeneca, Berlin-Chemie, Boehringer-Ingelheim, Chiesi, Cipla, GSK, Novartis, Roche, Sanofi, and TEVA, and grants to Mainz University Hospital for research or clinical trials, or both from Boehringer Ingelheim, GSK, Novartis, and Roche. A.A.C. has received consultancy and speaker fees from Abdi-Ibrahim, AstraZeneca, Boehringer-Ingelheim, Chiesi, Eurofarma, Glennmark, GSK, Novartis, and Sanofi. A.C.C. is a member of the Planning Group of the Global Alliance against Chronic Respiratory Diseases (GARD)/WHO, and the Executive Director of ProAR Foundation – Brazil. L.D. has received grants from the European Union Horizon 2020 research and innovation program (LIFECYCLE, grant agreement No 733206, 2016; EUCAN-Connect grant agreement No 824989; ATHLETE, grant agreement No 874583) and Stichting Vrienden van Sophia, and speaker fee (paid to institute) from Hong Kong University. L.D. is Vice chair of the Dutch Pediatric Respiratory Society. L.F. has received personal fees from Novartis, Astra Zeneca, and Sanofi, outside the submitted work. H.I. has received speaker and advisory board fees from Astellas, AstraZeneca, Boehringer-Ingelheim, Fukuda-Denshi, GSK, Kracie, Kyorin, Novartis, Omron, Pfizer, and Sanofi, and research grants and support (paid to university) from Asahi-Kasei Pharma, AstraZeneca, Boehringer-Ingelheim, Chugai, GlaxoSmithKline, Kyorin, Otsuka, Teijin, Taiho, and Ono. F.W.S.K. has received funding for conducting multi-center research on asthma from the Hong Kong Thoracic Society (which was supported by AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, and Novartis) with all fees paid directly to the institution. J.A.K. has received consulting fees from GlaxoSmithKline, AstraZeneca, and BData, Inc., and grants and contract fees from Regeneron (COVID-19 clinical trial), Sergey Bring Family Foundation (COVID-19 cohort study), Patient-Centered Outcomes Research Institute (research studies in asthma, COPD, insomnia), National Institutes of Health (research studies in asthma, COPD, acute COVID-19, and post-acute sequelae of SARS-CoV-2), and American Lung Association (asthma and COPD research). K.M. has received advisory board fees from AstraZeneca. P.M.P. has received consultancy and speaker fees from AstraZeneca, GSK, Novartis, Boehringer Ingelheim, Novartis, and Sanofi. A.S. has advised AstraZeneca on an unremunerated basis and has received a research grant from GSK (COVID-19 therapeutics). A.S. is editor emeritus of npj PCRM but was not involved in the journal’s review of, or decisions related to, this manuscript. A.Y. has received consultancy and speaker honoraria from Astra-Zeneca, Novartis, GSK, Chiesi, DEVA, and Abdi İbrahim. A.Y. is Chair of GARD. A.Y. is an editor for npj Primary Care Respiratory Medicine but was not involved in the journal’s review of, or decisions related to, this manuscript. H.K.R. has received honoraria for participation in advisory boards for AstraZeneca, Chiesi, GlaxoSmithKline, Novartis, and Sanofi-Genzyme; and from AstraZeneca, Boehringer Ingelheim, Chiesi, Getz, GlaxoSmithKline, Sanofi, and Teva Pharmaceuticals for independent medical educational presentations; she has received independent research funding from AstraZeneca, GlaxoSmithKline, and Novartis; and consulting fees from AstraZeneca and Novartis. L.P.B. (Chair), E.B., G.B., A.A.C., H.I., J.K., M.L.L., H.K.R., and A.Y. are members of Global Initiative for Asthma (GINA) Board of Directors. L.B., E.B., L.P.B., C.B., G.B., R.B., J.D., L.D., L.F., H.I., F.K., K.M., P.M.P., H.K.R. (Chair), A.S., and A.Y. are members of the GINA Science Committee. L.P.B., A.A.C., H.I., M.L.L. (Chair), and A.Y. are members of GINA Dissemination and Implementation Task Group.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s41533-023-00330-1.
References
- 1.Levy ML. The national review of asthma deaths: what did we learn and what needs to change? Breathe. 2015;11:15–24. doi: 10.1183/20734735.008914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Royal College of Physicians. Why Asthma Still Kills. The National Review of Asthma Deaths (NRAD) Confidential Enquiry Report (RCP, 2014).
- 3.Anagnostou K, Harrison B, Iles R, Nasser S. Risk factors for childhood asthma deaths from the UK Eastern Region Confidential Enquiry 2001–2006. Prim. Care Respir. J. 2012;21:71–77. doi: 10.4104/pcrj.2011.00097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.British Thoracic Association. Death from asthma in two regions of England. British Thoracic Association. Br. Med. J. 1982;285:1251–1255. [PMC free article] [PubMed] [Google Scholar]
- 5.Global Asthma Network. The Global Asthma Report 2022. Int. J. Tuberc. Lung Dis. 2022;26:S1–S102. [Google Scholar]
- 6.Mortimer K, Reddel HK, Pitrez PM, Bateman ED. Asthma management in low- and middle-income countries: case for change. Eur. Respir. J. 2022;60:2103179. doi: 10.1183/13993003.03179-2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meghji J, et al. Improving lung health in low-income and middle-income countries: from challenges to solutions. Lancet. 2021;397:928–940. doi: 10.1016/S0140-6736(21)00458-X. [DOI] [PubMed] [Google Scholar]
- 8.Stolbrink M, et al. Improving access to affordable quality-assured inhaled medicines in low- and middle-income countries. Int. J. Tuberc. Lung Dis. 2022;26:1023–1032. doi: 10.5588/ijtld.22.0270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.World Health Organization. Model List of Essential Medicines. 22nd list (2021) (WHO, 2021).
- 10.Reddel HK, et al. GINA 2019: a fundamental change in asthma management: treatment of asthma with short-acting bronchodilators alone is no longer recommended for adults and adolescents. Eur. Respir. J. 2019;53:1901046. doi: 10.1183/13993003.01046-2019. [DOI] [PubMed] [Google Scholar]
- 11.Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention, 2022 (GINA, 2022).
- 12.Global Asthma Network. The Global Asthma Report 2018 (Global Asthma Network, 2018).
- 13.Aaron SD, Boulet LP, Reddel HK, Gershon AS. Underdiagnosis and overdiagnosis of asthma. Am. J. Respir. Crit. Care Med. 2018;198:1012–1020. doi: 10.1164/rccm.201804-0682CI. [DOI] [PubMed] [Google Scholar]
- 14.Aaron SD, et al. Overdiagnosis of asthma in obese and nonobese adults. CMAJ. 2008;179:1121–1131. doi: 10.1503/cmaj.081332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lo DK, et al. Lung function and asthma control in school-age children managed in UK primary care: a cohort study. Thorax. 2020;75:101–107. doi: 10.1136/thoraxjnl-2019-213068. [DOI] [PubMed] [Google Scholar]
- 16.van Schayck CP, van Der Heijden FM, van Den Boom G, Tirimanna PR, van Herwaarden CL. Underdiagnosis of asthma: is the doctor or the patient to blame? The DIMCA project. Thorax. 2000;55:562–565. doi: 10.1136/thorax.55.7.562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Adams RJ, et al. Underdiagnosed asthma in South Australia. Thorax. 2003;58:846–850. doi: 10.1136/thorax.58.10.846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brand PL, et al. Definition, assessment and treatment of wheezing disorders in preschool children: an evidence-based approach [European Respiratory Society Task Force] Eur. Respir. J. 2008;32:1096–1110. doi: 10.1183/09031936.00002108. [DOI] [PubMed] [Google Scholar]
- 19.Martinez FD, et al. Asthma and wheezing in the first six years of life. The Group Health Medical Associates. N. Engl. J. Med. 1995;332:133–138. doi: 10.1056/NEJM199501193320301. [DOI] [PubMed] [Google Scholar]
- 20.Belgrave DCM, et al. Joint modeling of parentally reported and physician-confirmed wheeze identifies children with persistent troublesome wheezing. J. Allergy Clin. Immunol. 2013;132:575.e2–583.e2. doi: 10.1016/j.jaci.2013.05.041. [DOI] [PubMed] [Google Scholar]
- 21.Savenije OE, Kerkhof M, Koppelman GH, Postma DS. Predicting who will have asthma at school age among preschool children. J. Allergy Clin. Immunol. 2012;130:325–331. doi: 10.1016/j.jaci.2012.05.007. [DOI] [PubMed] [Google Scholar]
- 22.Suissa S, Ernst P, Benayoun S, Baltzan M, Cai B. Low-dose inhaled corticosteroids and the prevention of death from asthma. N. Engl. J. Med. 2000;343:332–336. doi: 10.1056/NEJM200008033430504. [DOI] [PubMed] [Google Scholar]
- 23.Crossingham I, et al. Combination fixed-dose beta agonist and steroid inhaler as required for adults or children with mild asthma. Cochrane Database Syst. Rev. 2021;5:CD013518. doi: 10.1002/14651858.CD013518.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Price DB, et al. Adverse outcomes from initiation of systemic corticosteroids for asthma: long-term observational study. J. Asthma Allergy. 2018;11:193–204. doi: 10.2147/JAA.S176026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Inman MD, O’Byrne PM. The effect of regular inhaled albuterol on exercise-induced bronchoconstriction. Am. J. Respir. Crit. Care Med. 1996;153:65–69. doi: 10.1164/ajrccm.153.1.8542164. [DOI] [PubMed] [Google Scholar]
- 26.Gauvreau GM, Jordana M, Watson RM, Cockroft DW, O’Byrne PM. Effect of regular inhaled albuterol on allergen-induced late responses and sputum eosinophils in asthmatic subjects. Am. J. Respir. Crit. Care Med. 1997;156:1738–1745. doi: 10.1164/ajrccm.156.6.96-08042. [DOI] [PubMed] [Google Scholar]
- 27.Suissa S, Blais L, Ernst P. Patterns of increasing beta-agonist use and the risk of fatal or near-fatal asthma. Eur. Respir. J. 1994;7:1602–1609. doi: 10.1183/09031936.94.07091602. [DOI] [PubMed] [Google Scholar]
- 28.Bloom CI, et al. Asthma-related health outcomes associated with short-acting β(2)-agonist inhaler use: an observational UK study as part of the SABINA Global Program. Adv. Ther. 2020;37:4190–4208. doi: 10.1007/s12325-020-01444-5. [DOI] [PubMed] [Google Scholar]
- 29.Nwaru BI, et al. Overuse of short-acting β2-agonists in asthma is associated with increased risk of exacerbation and mortality: a nationwide cohort study of the global SABINA programme. Eur. Respir. J. 2020;55:1901872. doi: 10.1183/13993003.01872-2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sobieraj DM, et al. Association of inhaled corticosteroids and long-acting β-agonists as controller and quick relief therapy with exacerbations and symptom control in persistent asthma: a systematic review and meta-analysis. JAMA. 2018;319:1485–1496. doi: 10.1001/jama.2018.2769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cates, C. J. & Karner, C. Combination formoterol and budesonide as maintenance and reliever therapy versus current best practice (including inhaled steroid maintenance), for chronic asthma in adults and children. Cochrane Database Syst. Rev. CD007313 (2013). [DOI] [PMC free article] [PubMed]
- 32.Jenkins CR, Bateman ED, Sears MR, O’Byrne PM. What have we learnt about asthma control from trials of budesonide/formoterol as maintenance and reliever? Respirology. 2020;25:804–815. doi: 10.1111/resp.13804. [DOI] [PubMed] [Google Scholar]
- 33.Bisgaard H, et al. Budesonide/formoterol maintenance plus reliever therapy: a new strategy in pediatric asthma. Chest. 2006;130:1733–1743. doi: 10.1378/chest.130.6.1733. [DOI] [PubMed] [Google Scholar]
- 34.Bateman ED, et al. Positioning as-needed budesonide-formoterol for mild asthma: effect of prestudy treatment in pooled analysis of SYGMA 1 and 2. Ann. Am. Thorac. Soc. 2021;18:2007–2017. doi: 10.1513/AnnalsATS.202011-1386OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bateman ED, et al. As-needed budesonide-formoterol versus maintenance budesonide in mild asthma. N. Engl. J. Med. 2018;378:1877–1887. doi: 10.1056/NEJMoa1715275. [DOI] [PubMed] [Google Scholar]
- 36.O’Byrne PM, et al. Inhaled combined budesonide–formoterol as needed in mild asthma. N. Engl. J. Med. 2018;378:1865–1876. doi: 10.1056/NEJMoa1715274. [DOI] [PubMed] [Google Scholar]
- 37.Beasley R, et al. Controlled trial of budesonide-formoterol as needed for mild asthma. N. Engl. J. Med. 2019;380:2020–2030. doi: 10.1056/NEJMoa1901963. [DOI] [PubMed] [Google Scholar]
- 38.Hardy J, et al. Budesonide-formoterol reliever therapy versus maintenance budesonide plus terbutaline reliever therapy in adults with mild to moderate asthma (PRACTICAL): a 52-week, open-label, multicentre, superiority, randomised controlled trial. Lancet. 2019;394:919–928. doi: 10.1016/S0140-6736(19)31948-8. [DOI] [PubMed] [Google Scholar]
- 39.FitzGerald JM, et al. Safety of as-needed budesonide-formoterol in mild asthma: data from the two phase III SYGMA studies. Drug Saf. 2021;44:467–478. doi: 10.1007/s40264-020-01041-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Reddel HK, Bateman ED, Schatz M, Krishnan JA, Cloutier MM. A practical guide to implementing SMART in asthma management. J. Allergy Clin. Immunol. Pract. 2022;10:S31–s38. doi: 10.1016/j.jaip.2021.10.011. [DOI] [PubMed] [Google Scholar]
- 41.Reddel, H. K. et al. Global Initiative for Asthma (GINA) Strategy 2021 – Executive summary and rationale for key changes. Eur. Respir. J. 59, 2102730 (2021). [DOI] [PMC free article] [PubMed]
- 42.Terrence Carney. Regulation 28 statement in the matter of Tamara Mills (deceased). (2015).
- 43.Dr Shirley Radcliffe. Regulation 28 Statement in the matter of Sophie Holman (deceased). (2019).
- 44.Dr Shirley Radcliffe. Regulation 28 Statement in the matter of Michael Uriely (deceased). (2017).
- 45.Chan M, et al. Assessment of variation in care following hospital discharge for children with acute asthma. J. Asthma Allergy. 2021;14:797–808. doi: 10.2147/JAA.S311721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Royal College of Physicians. National Asthma and Chronic Obstructive Pulmonary Disease Audit Programme (NACAP) Children and Young People Asthma Audit 2019/20 (Royal College of Physicians, 2021).
- 47.Reddel HK, et al. Should recommendations about starting inhaled corticosteroid treatment for mild asthma be based on symptom frequency: a post-hoc efficacy analysis of the START study. Lancet. 2017;389:157–166. doi: 10.1016/S0140-6736(16)31399-X. [DOI] [PubMed] [Google Scholar]
- 48.Dusser D, et al. Mild asthma: an expert review on epidemiology, clinical characteristics and treatment recommendations. Allergy. 2007;62:591–604. doi: 10.1111/j.1398-9995.2007.01394.x. [DOI] [PubMed] [Google Scholar]
- 49.Hekking PP, et al. The prevalence of severe refractory asthma. J. Allergy Clin. Immunol. 2015;135:896–902. doi: 10.1016/j.jaci.2014.08.042. [DOI] [PubMed] [Google Scholar]
- 50.Bloom CI, Walker S, Quint JK. Inadequate specialist care referrals for high-risk asthma patients in the UK: an adult population-based cohort 2006–2017. J. Asthma. 2021;58:19–25. doi: 10.1080/02770903.2019.1672723. [DOI] [PubMed] [Google Scholar]
- 51.O’Neill S, et al. The cost of treating severe refractory asthma in the UK: an economic analysis from the British Thoracic Society Difficult Asthma Registry. Thorax. 2015;70:376–378. doi: 10.1136/thoraxjnl-2013-204114. [DOI] [PubMed] [Google Scholar]
- 52.Sadatsafavi M, et al. Direct health care costs associated with asthma in British Columbia. Can. Respir. J. 2010;17:74–80. doi: 10.1155/2010/361071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Plaza V, et al. Impact of patient satisfaction with his or her inhaler on adherence and asthma control. Allergy Asthma Proc. 2018;39:437–444. doi: 10.2500/aap.2018.39.4183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Melani AS, et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir. Med. 2011;105:930–938. doi: 10.1016/j.rmed.2011.01.005. [DOI] [PubMed] [Google Scholar]
- 55.Price DB, et al. Inhaler errors in the CRITIKAL study: type, frequency, and association with asthma outcomes. J. Allergy Clin. Immunol. Pract. 2017;5:1071.e9–1081.e9. doi: 10.1016/j.jaip.2017.01.004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analyzed during the current study.