Supplemental Digital Content is Available in the Text.
Key Words: HIV, incidence, HIV testing, health surveys, epidemiology methods
Background:
Estimating HIV incidence is essential to monitoring progress in sub-Saharan African nations toward global epidemic control. One method for incidence estimation is to test nationally representative samples using laboratory-based incidence assays. An alternative method based on reported HIV testing history and the proportion of undiagnosed infections has recently been described.
Methods:
We applied an HIV incidence estimation method which uses history of testing to nationally representative cross-sectional survey data from 12 sub-Saharan African nations with varying country-specific HIV prevalence. We compared these estimates with those derived from laboratory-based incidence assays. Participants were tested for HIV using the national rapid test algorithm and asked about prior HIV testing, date and result of their most recent test, and date of antiretroviral therapy initiation.
Results:
The testing history-based method consistently produced results that are comparable and strongly correlated with estimates produced using a laboratory-based HIV incidence assay (ρ = 0.85). The testing history-based method produced incidence estimates that were more precise compared with the biomarker-based method. The testing history-based method identified sex-, age-, and geographic location-specific differences in incidence that were not detected using the biomarker-based method.
Conclusions:
The testing history-based method estimates are more precise and can produce age-specific and sex-specific incidence estimates that are informative for programmatic decisions. The method also allows for comparisons of the HIV transmission rate and other components of HIV incidence among and within countries. The testing history-based method is a useful tool for estimating and validating HIV incidence from cross-sectional survey data.
INTRODUCTION
Since the start of the HIV epidemic, an estimated 79.3 million individuals have been infected worldwide, with more than 1.5 million new infections in 2020 and more than 36.3 million cumulative deaths.1 In 2014, the Joint United Nations Programme on HIV/AIDS (UNAIDS) set 3 ambitious global targets: (1) to have 90% of all persons living with HIV (PLWH) being aware of their HIV status, (2) to have 90% of those aware of their status receiving antiretroviral therapy (ART), and (3) to have 90% of those on ART achieving viral suppression (90–90–90 goals). The UNAIDS goals were updated to 95–95–95 by 2025. Achievement of these goals was projected to reduce HIV incidence and mortality rates globally by up to 90% by 2030.2 To monitor progress toward achieving these goals, the President's Emergency Plan for AIDS Relief (PEPFAR) and the United States' Centers for Disease Control and Prevention (CDC), and participating ministries of health conducted nationally representative, population-based HIV impact assessment (PHIA) surveys in 12 sub-Saharan African countries between 2015 and 2018.3 Data from these surveys produced estimates of progress toward the 95–95–95 goals; the estimates are used to monitor progress and impact of national HIV treatment and prevention programs.3,4
One key impact measure of the 95–95–95 goals is the incidence of new HIV infections. Incidence is also required for many other important epidemic metrics, such as the incidence/prevalence and incidence/mortality ratios.5 Ideally, incidence is directly measured by determining the number of new cases among a representative population that is followed over time. However, this would be impractical to measure directly in many settings because of the size, cost, and complicated logistics of incidence studies.6 Therefore, HIV incidence is often estimated using one of several methods. The World Health Organization (WHO) has established criteria for estimating HIV incidence using cross-sectional laboratory assays to distinguish recent from long-term infections.7–9 Although widely used as part of cross-sectional household surveys, this biomarker-based method has several limitations, including the requirement for additional laboratory testing, additional logistical complexity, and the large sample requirements needed to generate precise estimates.6 Despite the large sample sizes (typically >10,000 subjects), the small number of recent cases typically identified in such studies is usually insufficient to generate robust subgroup or regional estimates.6 National estimates for subgroups (eg, women and young adults) and subnational regional estimates are of particular interest to HIV prevention and treatment programs for prioritization of specific groups or regions for interventions.
Another limitation is that assay-based incidence estimation requires 2 a priori parameters; these parameters, in turn, require assay calibration and performance evaluation for different specific populations.10 The first parameter is the mean duration of recent infections (MDRI), which is defined as the average length of time that true recently infected PLWH are correctly classified by the assay.11 The second parameter is the false recency rate (FRR), which is the proportion of long-term infections that are incorrectly classified as recent by the assay. These 2 parameters are calibrated from samples of individuals with known time since infection in a reference population. It is assumed that, in FRR and MDRI, this reference population is identical to the population under study, and that the population under study is in a steady state.9,12 FRR or MDRI may be dependent on various local population-specific factors (such as the distribution of HIV subtypes13), which could complicate finding a reference population in which to generate the parameters. In addition, the parameters are heavily dependent on the treatment history of the study population. Individuals taking ART or who have suppressed viral loads (VLs) have elevated rates of false-recent classification, increasing both the FRR and MDRI in the population.14 Since 2015, the WHO has recommended HIV treatment begins immediately after diagnosis without consideration for CD4 cell count or HIV VL,15 resulting in many study populations with high levels of ART coverage. Therefore, the addition of ART and VL testing can improve the accuracy MDRI and FRR estimates by allowing adjustment for those who seem falsely nonrecent due to their ART use or VL.
Given these limitations for cross-sectional laboratory assays, several alternative methods for incidence estimation have been proposed, including modeling national incidence based on surveillance of routine HIV testing programs16 or correlating recency to local population-level viremia dynamics.17 This article evaluates a different method that uses a self-reported history of HIV testing from population-based surveys, referred herein as the testing history-based method.6 This method categorizes the population into 3 groups: uninfected individuals, undiagnosed infections, and diagnosed infections. The incidence rate is defined as the rate at which uninfected individuals enter the undiagnosed infection group because no individual is diagnosed at the moment of infection. Individuals in the undiagnosed infection group then transition to the diagnosed infection group upon receiving a diagnosis. The size of the diagnosed plus undiagnosed infection group (ie, prevalence of HIV) can be directly measured in cross-sectional surveys. Among those with prevalent HIV infection, the size of the undiagnosed infection group is estimated using self-reported HIV status. This method has been validated using data from household surveys conducted in Kenya during 2007 and 2012.6,18
Biomarker-based incidence estimation uses the transition from recent to nonrecent HIV infection to estimate the incidence. Because this recency period is relatively short, biomarker-based incidence estimates are reflective of a short-time window, which may make them more subject to stochastic (ie, random) variation in new infections. In addition, these estimates often have broad confidence intervals because a relatively small number of infections in the recency window are typically observed, even in large samples. By contrast, the testing history method uses the transition from infected but undiagnosed to infected and diagnosed to estimate the incidence (ie, the transition between uninfected and to infected but undiagnosed). Because the average time spent in the undiagnosed group is comparatively longer than the time spent as a recent infection, testing history-based estimates may be less subject to stochastic variation. Incidence estimation based on testing history has an additional advantage in that the required inputs for the testing history method are inexpensive to collect and are already included in many HIV surveys, allowing incidence estimation in the absence of additional laboratory-based incidence assay testing and additional laboratory costs. The testing history-based method also generates estimates of the transmission rate of HIV, defined as the average number of new infections per infected individual per unit time, which allows another means of comparing the HIV epidemic between different populations.
Although the testing history-based method was developed and validated using data from Kenya AIDS Indicator Surveys, it remains unclear whether the method can be generalized to produce robust estimates of incidence across the sub-Saharan African region given the variation in HIV prevalence and incidence among countries. Here, we describe the results of the application of the testing history-based method to generate national, regional, and subgroup HIV incidence estimates for 12 sub-Saharan African countries and to compare these estimates with those estimates generated using a recency biomarker method.
METHODS
PHIA Methods
We used data from PHIA surveys conducted by the US CDC and participating ministries of health in Cameroon, Cote d’Ivoire, Eswatini, Ethiopia, Lesotho, Malawi, Namibia, Rwanda, Tanzania, Uganda, Zambia, and Zimbabwe. The survey design, sampling methodology, and logistics have been previously described.4,19 The PHIA surveys were approved by institutional review boards at CDC, Columbia University, Westat, and in respective countries. Eligible, consenting participants were asked whether they had ever received an HIV test and the month, year, and result of their most recent test. Participants who reported a positive HIV test result were asked the month and year of ART initiation, if applicable. Sociodemographic data about the participants were also recorded. Our analysis applied the 2 different HIV incidence estimation methods to the same PHIA participants from the aforementioned countries and was limited to individuals aged 15–59 years. The number of participants included in our analysis from each PHIA study is summarized in Table 1.
TABLE 1.
Estimated HIV Incidence by Country and Estimate Method, and Percent Difference Between Methods, PHIAs, 2015–2019
| n | Testing History Estimate | Biomarker Estimate | Percent Difference | |||
| HIV Incidence (95% CI) | CIR | HIV Incidence (95% CI) | CIR | |||
| Lesotho 2016 | 16,326 | 1.92 (1.74 to 2.10) | 1.2 | 1.11 (0.68 to 1.52) | 2.2 | −42% |
| Eswatini 2016 | 11,784 | 1.24 (1.00 to 1.48) | 1.5 | 1.14 (0.73 to 1.53) | 2.1 | −8% |
| Zambia 2016 | 27,069 | 0.59 (0.52 to 0.66) | 1.3 | 0.61 (0.40 to 0.81) | 2.0 | 3% |
| Zimbabwe 2015 | 25,309 | 0.55 (0.48 to 0.63) | 1.3 | 0.40 (0.24 to 0.56) | 2.3 | −27% |
| Malawi 2015 | 24,063 | 0.43 (0.38 to 0.49) | 1.3 | 0.37 (0.20 to 0.54) | 2.7 | −14% |
| Tanzania 2016 | 34,503 | 0.36 (0.32 to 0.41) | 1.3 | 0.26 (0.15 to 0.36) | 2.3 | −27% |
| Namibia 2017 | 23,334 | 0.32 (0.26 to 0.38) | 1.5 | 0.37 (0.18 to 0.55) | 3.1 | 16% |
| Uganda 2016 | 31,661 | 0.28 (0.23 to 0.32) | 1.4 | 0.41 (0.25 to 0.56) | 2.2 | 46% |
| Cameroon 2017 | 28,737 | 0.18 (0.15 to 0.22) | 1.5 | 0.24 (0.12 to 0.36) | 3.1 | 33% |
| Rwanda 2018 | 32,468 | 0.07 (0.05 to 0.09) | 1.8 | 0.08 (0.02 to 0.14) | 6.4 | 14% |
| Ethiopia 2017 | 23,385 | 0.07 (0.04 to 0.09) | 2.1 | 0.05 (0 to 0.10) | 15.8 | −29% |
| Cote d'Ivoire 2017 | 22,292 | 0.06 (0.02 to 0.09) | 4.2 | 0.03 (0 to 0.07) | 0.6 | −50% |
Incidence expressed as a percent. 95% confidence interval (CI) generated using the jackknife method. CIR, confidence interval ratio (upper/lower). Percent difference is the biomarker estimate minus testing history, divided by the testing history estimate, multiplied by 100. Negative percent difference indicates that the testing history estimate is larger than the biomarker estimate.
HIV recency testing for confirmed HIV-seropositive participants with plasma samples was conducted using the HIV-1 LAg-Avidity EIA (Sedia Biosciences Corporation, Portland, OR), or if plasma was not available, with DBS using the Maxim HIV-1 LAg DBS EIA (Maxim Biomedical, Bethesda, MD) in a central reference laboratory by laboratorians trained by CDC.20 Country-specific first-line and second-line ARV in DBS samples were detected at the Division of Clinical Pharmacology of the Department of Medicine at the University of Cape Town using qualitative high-performance liquid chromatography and tandem mass spectrometry.21
Testing History Incidence Calculation
Incidence estimates were calculated using the following weighted formula described by Fellows:
where, is the incidence of HIV, P(H) is the proportion of those infected with HIV among the total population, P(U|H) is the proportion of those who are undiagnosed among the total of those with HIV infection, and E(TID) is the estimated time between infection and diagnosis.6 Among study participants, P(H) is directly calculated using individual results of an assay for HIV seropositivity. Those identified through seropositivity as infected individuals who report never having been diagnosed with HIV are considered undiagnosed infections. P(U|H) can be estimated using seroassay results and self-reported diagnosis status for each respondent. Some proportion of respondents will misreport their HIV status; however, this method adjusts for this for by using ART biomarker and HIV VL data on a sample of respondents.6 This method further defines transmission rate, τ, (ie, the number of new infections per unit time per infected individual) as 6. The transmission rate allows a way of comparing populations' burden of new HIV cases that is independent of prevalence. Given this, Incidence Rate, can also be represented as Confidence intervals were generated using the jackknife method.22 An R package which applies this method is available freely online.23
Biomarker-based incidence estimates were generated using a RITA defined as testing recent on the limiting antigen (LAg) avidity enzyme immunoassay with HIV-1 RNA concentration at or greater than 1000 copies/mL and no ART use.24 ART use was defined as the presence of ART biomarkers in specimens.
Biomarker-based incidence estimates were calculated using the formula recommended by the WHO Incidence Working group and Consortium for Evaluation and Performance of Incidence Assays.8 These calculations used the following weighted parameters: MDRI for all nations except Uganda—130 days, MDRI for Uganda—153 days (this is to account for subtypes A and D distribution in the population13), FRR—0.00, and time cutoff—1 year. Biomarker HIV estimates were calculated in SAS 9.4 (SAS Institute Inc., Cary, NC) using a SAS incidence macro available in the PHIA data use manual.25 Confidence intervals were generated using the jackknife variance estimation method.
Linear Regression, Transmission Rate, and Prevalence Analysis
We applied both incidence estimation methods to all 12 data sets to calculate national incidence estimates. We then used median linear regression and the concordance correlation coefficient (ρ) to assess the presence and strength of a correlation between biomarker and testing history estimates. Median linear regression was used to minimize the influence of outliers.26 Although the usual correlation (Pearson) measures the degree to which 2 measures are linearly related, the concordance correlation measures their degree of agreement.27 We also applied each incidence estimation method to specific subgroups based on age category (15–29 and 30–59 years), sex, and geographic location in either an urban or rural community. The Ethiopia 2017 PHIA survey included only participants living in urban areas. All other country surveys were included in each subgroup analysis. We additionally compared the countries' components of the incidence and transmission rate calculations in the testing history-based method, namely the prevalence of HIV, the proportion of PLWH who are undiagnosed, and the estimated time between infection and diagnosis.
RESULTS
HIV Incidence Rate Estimates by Country
The overall HIV incidence estimates for each survey using both the testing history-based and biomarker-based methods are summarized in Table 1. The biomarker-based method incidence estimates ranged from 0.03% in Cote d’Ivoire to 1.14% in Eswatini. In the testing history-based method, these estimates ranged from 0.06% in Cote d’Ivoire to 1.92% in Lesotho. Overall, the testing history-based method produced similar results to the biomarker-based methods (concordance correlation coefficient, ρ = 0.85) (Fig. 1). The largest relative differences between the 2 methods estimates were for Cote d’Ivoire (−50% difference), Lesotho (−42% difference), and Uganda (46% difference).
FIGURE 1.
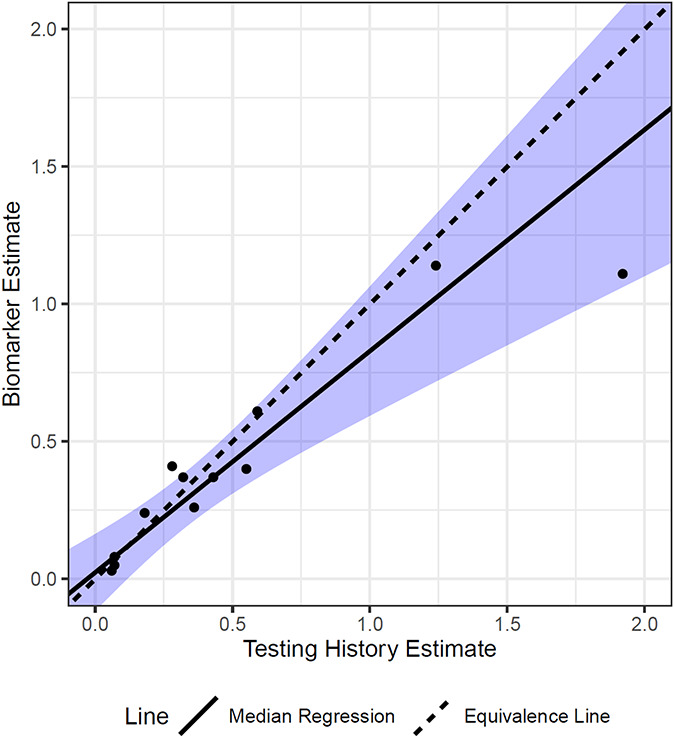
Simple linear regression between methods' estimates of HIV incidence, population-based HIV impact assessments, 2015–2019. Median regression line-of-best-fit. Axis units: %. Equivalence line is a reference line where slope = 1, and X-intercept = 0 and Y-intercept = 0. Confidence limits are shown in shaded blue.
HIV Incidence Estimates by Country, by Age, Sex, and Location
Incidence estimates among women ranged from 0.03% in Cote d’Ivoire and 1.42% in Eswatini using the biomarker-based method, and from 0.11% in Cote d’Ivoire and 2.83% in Lesotho using the testing history-based method (Fig. 2A). Among men, incidence estimates ranged from 1.01% to 1.26% in Lesotho and 0.03% and 0.02% in Cote d’Ivoire using the biomarker-based and testing history-based methods, respectively (Fig. 2B). The testing history method was moderately correlated with the biomarker-based method among women (ρ =0.70) and strongly correlated among men (ρ = 0.94). Analysis with the biomarker-based estimates resulted in a significant sex-specific difference in HIV incidence only in Zambia, but the testing history-based method resulted in significant sex-specific differences in HIV incidence in 8 of 12 countries (Lesotho, Zambia, Zimbabwe, Malawi, Tanzania, Namibia, Uganda, and Cameroon).
FIGURE 2.
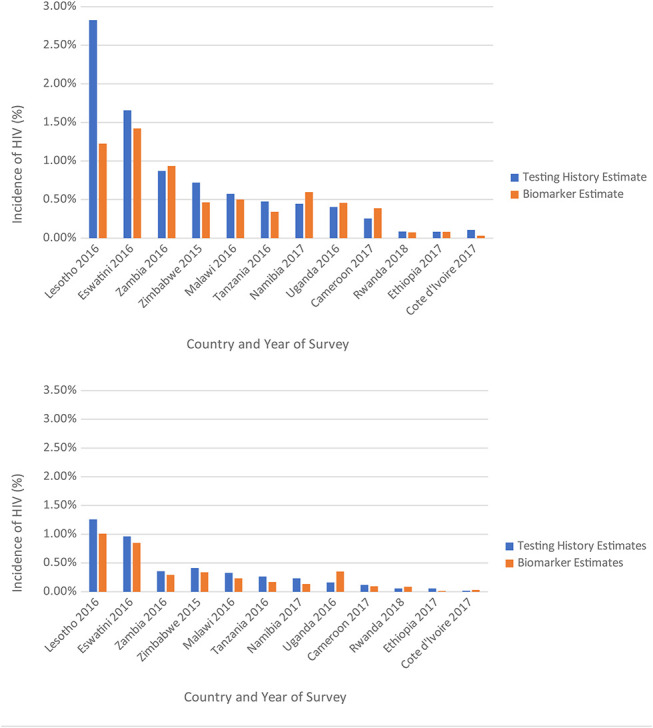
A. Estimated female-specific incidence of HIV by country and estimate method, in 12 sub-Saharan African nations, population-based HIV impact assessments, 2015–2019. B. Estimated male-specific incidence of HIV, by country and estimate method, population-based HIV impact assessments, 2015–2019.
Among participants aged 15–29 years, the age-specific HIV incidence ranged from 0.03% to 0.04% in Ethiopia to 0.98% and 1.40% in Lesotho using the biomarker and testing history-based methods, respectively (see Figure S1A, Supplemental Digital Content, http://links.lww.com/QAI/B989). Among participants aged 15–29 years, the 2 methods' incidence estimates correlated well (ρ = 0.84). Among participants aged 30–59 years, age-specific incidence of HIV ranged from 0.017% to 0.073% in Cote d’Ivoire to 1.29% and 2.88% in Lesotho, using the biomarker-based and testing history-based methods, respectively (see Figure S1B, Supplemental Digital Content, http://links.lww.com/QAI/B989). The testing history-based and biomarker-based age-specific estimates were moderately correlated (ρ =0.56). Analysis with the biomarker-based estimates resulted in age group-specific differences in HIV incidence only in Tanzania, but the testing history-based method resulted in significant (ie, nonoverlapping confidence intervals) age group-specific differences in HIV incidence in 9 of 12 countries (Lesotho, Eswatini, Zambia, Zimbabwe, Malawi, Tanzania, Namibia, Uganda, and Cameroon).
Among those living in urban areas, the estimates of the incidence of HIV derived using biomarker-based and testing history-based method were 0.04% and 0.09% in Cote d’Ivoire, 0.96%% and 1.99% in Eswatini and 1.24% and 2.34% in Lesotho, respectively (see Figure S2A, Supplemental Digital Content, http://links.lww.com/QAI/B989). Among those living in rural areas, estimates of the incidence of HIV by biomarker-based and testing history-based methods were 0.01% and 0.03% in Cote d’Ivoire, and were 0.99% and 1.62% in Lesotho, respectively (see Figure S2B, Supplemental Digital Content, http://links.lww.com/QAI/B989). The testing history method was moderately correlated with the biomarker-based method among urban participants (ρ =0.67) and rural participants (ρ =0.74). Of note, the 95% confidence intervals for the testing history estimate were larger than the biomarker-based estimates for the high-prevalence nations of Eswatini and Lesotho. Analysis with the biomarker-based estimates did not result in significant urban/rural group differences in HIV incidence in any country studied, but the testing history-based estimates suggested urban/rural group differences in HIV incidence in 6 (Lesotho, Zambia, Malawi, Tanzania, Uganda, and Cameroon) of the 11 countries that stratified data by urban/rural status.
Component Analysis of Incidence Estimates and Transmission Rates
Comparing the components of the testing history-based incidence estimates can reveal differences in the state of the HIV epidemic across the region (Table 2). Component analysis revealed that incidence of HIV in countries with the largest incidence rates, such as Eswatini and Lesotho, were driven by their high prevalence of HIV (P(H)). Other countries with lower HIV prevalence, such as Rwanda, Cote d’Ivoire, Ethiopia, and Cameroon, had incidence rates driven by larger estimated time between infection and diagnosis resulting in unsuppressed VL among those undiagnosed individuals. Namibia and Uganda were found to have similar incidence rate estimates to one another; however, component analysis revealed that in Namibia, this was driven by a larger HIV prevalence, while in Uganda this was driven by a larger undiagnosed proportion. The HIV transmission rate (the number of new infections per infected individual per unit time) generated using the testing history-based method is summarized in Table 2. The HIV transmission rate estimates ranged from 1.90% in Namibia to 5.64% in Tanzania.
TABLE 2.
Components of the HIV Transmission Rate and HIV Incidence Derived From the Proportion of PLWH Who are Undiagnosed, Estimated Time Between Infection and Diagnosis, and HIV Prevalence, Population-Based HIV Impact Assessments, 2015–2019
| Transmission Components | Incidence Components | Incidence (%) | |||
| % PLWH Undiagnosed | Time From Infection to Diagnosis | Transmission Rate | HIV Prevalence | ||
| P(U|H)* | E(TID)† | = 100* P(U|H)/E(TID)‡ | P(H) | = *P(H)/(1-P(H))§ | |
| Lesotho 2016 | 12% | 2.34 | 5.11 (5.07 to 5.14) | 0.27 | 1.92 (1.74 to 2.10) |
| Eswatini 2016 | 7% | 2.47 | 3.02 (2.97 to 3.06) | 0.29 | 1.24 (1.00 to 1.48) |
| Zambia 2016 | 16% | 4.03 | 3.97 (3.95 to 4.00) | 0.13 | 0.59 (0.52 to 0.66) |
| Zimbabwe 2015 | 11% | 3.97 | 2.81 (2.78 to 2.83) | 0.16 | 0.55 (0.48 to 0.63) |
| Malawi 2015 | 12% | 3.98 | 2.92 (2.90 to 2.95) | 0.13 | 0.43 (0.38 to 0.49) |
| Tanzania 2016 | 25% | 4.39 | 5.64 (5.59 to 5.68) | 0.06 | 0.36 (0.32 to 0.41) |
| Namibia 2017 | 7% | 3.87 | 1.90 (1.87 to 1.92) | 0.14 | 0.32 (0.26 to 0.38) |
| Uganda 2016 | 15% | 3.60 | 4.26 (4.22 to 4.30) | 0.06 | 0.28 (0.23 to 0.32) |
| Cameroon 2017 | 27% | 5.74 | 4.73 (4.69 to 4.78) | 0.04 | 0.18 (0.15 to 0.22) |
| Rwanda 2018 | 10% | 4.26 | 2.30 (1.67 to 2.35) | 0.03 | 0.07 (0.05 to 0.09) |
| Ethiopia 2017 | 10% | 4.77 | 2.12 (2.07 to 2.93) | 0.03 | 0.07 (0.04 to 0.09) |
| Cote d'Ivoire 2017 | 17% | 7.25 | 2.30 (1.18 to 3.42) | 0.02 | 0.06 (0.02 to 0.09) |
is the proportion of PLWH who are undiagnosed.
E(TID) is the estimated mean time in years between infection and diagnosis.
Transmission rate (annual new cases/100 PLWH), .
Incidence rate, .
95% confidence intervals generated using the jackknife method.
DISCUSSION
Self-reported testing history modeling produced HIV incidence estimates that correlated well (ρ = 0.85) with biomarker-based incidence assay–derived estimates. Across the 12 countries studied, the testing history method produced both estimates relatively larger than the biomarker-based method (varying from 8% to 46% higher in 8 of 12 countries) and estimates relatively smaller than the biomarker-based method (varying from 3% to 47% lower in 4 of 12 countries), indicating no obvious pattern for the differences in HIV incidence. Moreover, the testing history-based method produced estimates that were more precise (ie, narrower confidence intervals and smaller confidence interval ratios, Table 1) than the biomarker-based method. This added precision allowed for the detection of significant differences in HIV incidence by sex, age category, and urban/rural status for many countries that the biomarker-based method would have failed to detect (ranging from 6 to 9 of 12 countries). This added precision is particularly important in studies of key populations such as men who have sex with men, female sex workers, and injection drug users, where sample sizes are usually prohibitively small for the utilization of the biomarker-based incidence estimation method. Increased precision may allow for more localized estimates of the number of new infections and may result in less uncertainty in global HIV modeling. This feature may make this estimation method more useful for national HIV prevention and screening programs wishing to target their interventions and programs to specific subpopulations or regions that have the highest incidence rates of HIV.
Despite the overall correlation of the 2 methods' estimates, there were some countries, notably Cote d’Ivoire, Uganda, and Lesotho, whose incidence estimates had large discrepancies. Further work is needed to differentiate whether this represents a limitation of one of either of the incidence estimation methods due to an underlying component of the population, such as differences in testing history behavior, or timing of ART initiation.
The testing history-based method on average estimated significantly larger incidence rates of HIV among women, those aged 30–59 years, and those who live in rural areas compared with the biomarker-based method, particularly in the countries of Lesotho and Eswatini. This finding may suggest that across the region, there may be important factors that differ by sex, age, and geographic location that may influence testing behavior that contribute to HIV incidence. These factors may also differ by country. Further analysis of these differences across the region and between countries is warranted.
The components used to generate incidence estimates in the testing history-based method may provide additional information useful for evaluating national HIV prevention and screening programs. An additional benefit of the testing history-based method is that it can more directly allow us to attribute differences in HIV incidence to other characteristics of the HIV epidemic in each nation, such as proportion of those living with HIV aware of their diagnosis, testing frequency, and prevalence of HIV. Knowledge of these characteristics may inform nations and programs seeking to curb the incidence of HIV. Although the prevalence of those who are living with HIV may increase in the near term due to reduced AIDS-related mortality, interventions can alter both the proportion of PWLH who are undiagnosed and the time between infection and diagnosis. For example, although the nations of Namibia and Uganda have similar HIV incidence, component analysis reveals that the nations may have different drivers of that incidence. Namibia has a high HIV prevalence but a relatively small proportion of undiagnosed PLWH. Conversely, Uganda has a comparatively small HIV prevalence but also has a relatively large proportion of undiagnosed PLWH, indicating that there may be subgroups that are being missed by the existing screening program, perhaps due to various sociodemographic factors such as limited access to care.
The testing history-based method and our analysis have several key limitations. The method assumes that the disease and total population are in a steady state.6 The impact of this limitation may be mitigated by comparing testing history-based incidence estimates in each country over time as repeated PHIA surveys are conducted; repeated surveys have thus far been conducted in Eswatini, Lesotho, Malawi, Uganda, Zimbabwe, and Zambia. Forecasting modeling efforts, such as UNAIDS′ Spectrum, could also be used to minimize the effect of this limitation. In addition, it assumes that there is no relationship between HIV risk and testing behavior (ie, high-risk groups are not more or less likely to have tested previously). Indeed, those with lower self-perceived risk of HIV report lower rates of uptake of HIV testing.28,29 The method also assumes that self-reported testing history is accurate; misclassification of testing history might introduce information bias, and that bias might be differential among countries and population subgroups. Further investigation into misclassification of self-reported testing history between countries and subgroups is warranted. Perhaps most importantly, the method assumes that those individuals with laboratory-detected evidence of ARV use (ie, those on treatment) misreport their diagnosis status at the same rate as those without detectable evidence of ARV use (ie, those not on treatment). It is likely that this misclassification is differential in different countries or sub-groups. Further sensitivity analysis regarding these assumptions in different regions and subgroups may be needed.
HIV incidence estimates derived from survey participant testing history are highly correlated with, and are more precise than, estimates from the biomarker method across multiple African countries. Although biomarker-based incidence estimates are important to benchmark testing history-based incidence estimate, the testing history-based estimate can complement this benchmark by providing more robust subnational incidence estimates (such as age-, sex-, and geography-specific), which are useful for countries and local HIV prevention programs. The testing history-based method can also be used in other types of surveys (eg, key population surveys) that are not generally large enough to support biomarker-based incidence estimation. Testing history-based incidence estimation is a useful tool to guide program evaluation and to monitor progress toward the UNAIDS goals for HIV epidemic control.
Supplementary Material
ACKNOWLEDGMENTS
The authors thank the Ministries of Health in the countries involved for their leadership in carrying out each of the surveys, the many staff who collected data for both surveys, and the participants of both surveys for sharing personal details of their lives with survey staff, and without whom none of this work would be possible.
Footnotes
Supported by the US President's Emergency Plan for AIDS Relief (PEPFAR) through the Centers for Disease Control and Prevention (CDC) under the terms of cooperative agreements #U2GGH001271 and #U2GGH001226. The findings and conclusions of this document are those of the authors and do not necessarily represent the official position of the funding agencies.
As an inventor of LAg-Avidity EIA, B.S.P. receives royalties from the sale of test kits sold by the manufacturer per US government policy. The remaining authors have no conflicts of interest to disclose.
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal's Web site (www.jaids.com).
Contributor Information
Paul W. Stupp, Email: pws2@cdc.gov.
Ian E. Fellows, Email: ymx4@cdc.gov.
Bharat S. Parekh, Email: bsp1@cdc.gov.
Peter W. Young, Email: fqm1@cdc.gov.
Patrick S. Sullivan, Email: fnf3@cdc.gov.
Andrew C. Voetsch, Email: aav6@cdc.gov.
REFERENCES
- 1.UNAIDS. FACT SHEET 2021 Global HIV Statistics. Geneva, Switzerland: 2021. [Google Scholar]
- 2.UNAIDS. To Help End the AIDS Epidemic. 2014. Available at: http://www.unaids.org/sites/default/files/media_asset/90-90-90_en.pdf. Accessed June 23, 2022. [Google Scholar]
- 3.Porter L, Bello G, Nkambule R, Justman J. HIV general population surveys: shedding light on the status of HIV epidemics and informing future actions. J Acquir Immune Defic Syndromes. 2021;87:2–5. [DOI] [PubMed] [Google Scholar]
- 4.Liljenquist D, Bai G, Anderson GF. Addressing generic-drug market failures — the case for establishing a nonprofit manufacturer. N Engl J Med. 2018;378:1857–1859. [DOI] [PubMed] [Google Scholar]
- 5.Ghys PD, Williams BG, Over M, et al. Epidemiological metrics and benchmarks for a transition in the HIV epidemic. PLoS Med. 2018;15:10026788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fellows IE, Shiraishi RW, Cherutich P, et al. A new method for estimating HIV incidence from a single cross-sectional survey. PLoS One. 2020;15:e02372211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.UNAIDS, World Health Organization (WHO). Monitoring HIV Impact Using Population-Based Surveys. Geneva, Switzerland, 2015. [Google Scholar]
- 8.Kassanjee R, McWalter TA, Bärnighausen T, Welte A. A new general biomarker-based incidence estimator. Epidemiology. 2012;23:721–728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brookmeyer R, Konikoff J, Laeyendecker O, Eshleman SH. Estimation of HIV incidence using multiple biomarkers. Am J Epidemiol. 2013;177:264–272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Joint United Nations Programme on HIV/AIDS (UNAIDS). Recent Infection Testing Algorithm Technical Update. Geneva, Switzerland, 2018. [Google Scholar]
- 11.Duong YT, Qiu M, De AK, et al. Detection of recent HIV-1 infection using a new limiting-antigen avidity assay: potential for HIV-1 incidence estimates and avidity maturation studies. PLoS One. 2012;7:e33328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brookmeyer Ron, Goedert JJ. Censoring in an epidemic with an application to hemophilia-associated AIDS. Biometrics. 1989;45:325–335. [PubMed] [Google Scholar]
- 13.Laeyendecker O, Gray RH, Grabowski MK, et al. Validation of the limiting antigen avidity assay to estimate level and trends in HIV incidence in an A/D epidemic in rakai, Uganda. AIDS Res Hum Retroviruses. 2019;35:364–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kassanjee R, Pilcher CD, Keating SM, et al. Independent assessment of candidate HIV incidence assays on specimens in the CEPHIA repository. AIDS. 2014;28:2439–2449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.World Health Organization, WHO. Progress Report 2016 Prevent HIV, Test and Treat All. Geneva, Switzerland, 2016. [Google Scholar]
- 16.Godin A, Eaton JW, Giguère K, et al. Inferring population HIV incidence trends from surveillance data of recent HIV infection among HIV testing clients. AIDS. 2021;35:2383–2388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Farahani M, Radin E, Saito S, et al. Population viral load, viremia, and recent HIV-1 infections: findings from population-based HIV impact Assessments (PHIAs) in Zimbabwe, Malawi, and Zambia. J Acquir Immune Defic Syndr. 2021;87:S81–S88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Waruiru W, Kim AA, Kimanga DO, et al. The Kenya AIDS indicator survey 2012: rationale, methods, description of participants, and response rates. J Acquir Immune Defic Syndr. 2014;66suppl 1:S3–S12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sachathep K, Radin E, Hladik W, et al. Population-based HIV impact Assessments survey methods, response, and quality in Zimbabwe, Malawi, and Zambia. J Acquir Immune Defic Syndr. 2021;87:S6–S16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Patel HK, Duong YT, Birhanu S, et al. A comprehensive approach to assuring quality of laboratory testing in HIV surveys: lessons learned from the population-based HIV impact assessment project. J Acquir Immune Defic Syndr. 2021;87:S17–S27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Koal T, Burhenne H, Römling R, et al. Quantification of antiretroviral drugs in dried blood spot samples by means of liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom. 2005;19:2995–3001. [DOI] [PubMed] [Google Scholar]
- 22.Hesterberg T, Shao J, Tu D. The Jackknife and Bootstrap. Vol 39; 1997. [Google Scholar]
- 23.Github—Fellstat. Testing History Incidence. New York City, NY: Github; 2020. Available at: https://github.com/fellstat/TestingHistoryIncidence. Accessed June 23, 2022. [Google Scholar]
- 24.Voetsch AC, Duong YT, Stupp P, et al. HIV-1 recent infection testing algorithm with antiretroviral drug detection to improve accuracy of incidence estimates. J Acquir Immune Defic Syndr. 2021;87:S73–S80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.PHIA Collaborating Institutions. PHIA Data Use Manual; 2019. Available at: https://phia-data.icap.columbia.edu/storage/Country/28-09-2021-22-01-17-615390ad6a147.pdf. Accessed June 23, 2022. [Google Scholar]
- 26.Walker E, Kleinbaum D, Kupper L, Muller K. Applied Regression Analysis and Other Multivariable Methods. Vol 31. London, United Kingdom; 1989. [Google Scholar]
- 27.Lin LI. A concordance correlation coefficient to evaluate reproducibility. Biometrics. 1989;45:255–268. Available at: http://www.jstor.org/stable/2532051. Accessed June 23, 2022. [PubMed] [Google Scholar]
- 28.Ajayi AI, Abioye AO, Adeniyi OV, Akpan W. Concerns about contracting HIV, knowing partners' HIV sero-status and discussion of HIV/STI with sexual partners as determinants of uptake of HIV testing. J Biosoc Sci. 2019;51:549–561. [DOI] [PubMed] [Google Scholar]
- 29.De Paoli MM, Manongi R, Klepp KI. Factors influencing acceptability of voluntary counselling and HIV-testing among pregnant women in Northern Tanzania. AIDS Care. 2004;16:411–425. [DOI] [PubMed] [Google Scholar]


