Abstract
Gastric cancer is one of the most common cancers in Korea and the world. Since 2004, this is the 4th gastric cancer guideline published in Korea which is the revised version of previous evidence-based approach in 2018. Current guideline is a collaborative work of the interdisciplinary working group including experts in the field of gastric surgery, gastroenterology, endoscopy, medical oncology, abdominal radiology, pathology, nuclear medicine, radiation oncology and guideline development methodology. Total of 33 key questions were updated or proposed after a collaborative review by the working group and 40 statements were developed according to the systematic review using the MEDLINE, Embase, Cochrane Library and KoreaMed database. The level of evidence and the grading of recommendations were categorized according to the Grading of Recommendations, Assessment, Development and Evaluation proposition. Evidence level, benefit, harm, and clinical applicability was considered as the significant factors for recommendation. The working group reviewed recommendations and discussed for consensus. In the earlier part, general consideration discusses screening, diagnosis and staging of endoscopy, pathology, radiology, and nuclear medicine. Flowchart is depicted with statements which is supported by meta-analysis and references. Since clinical trial and systematic review was not suitable for postoperative oncologic and nutritional follow-up, working group agreed to conduct a nationwide survey investigating the clinical practice of all tertiary or general hospitals in Korea. The purpose of this survey was to provide baseline information on follow up. Herein we present a multidisciplinary-evidence based gastric cancer guideline.
Keywords: Stomach neoplasms, Chemotherapy, Endoscopy, Surgery, Guidelines
INTRODUCTION
Background
Gastric cancer is one of the most common cancers in Korea and the world, which is ranks 5th in incidence and was the 4th leading cause of death among all solid cancers, excluding nonmelanoma skin cancer, globally in 2020 [1]. In Korea, new cases of gastric cancer (26,662) ranked 4th (10.8%), followed by thyroid cancer (11.8%), lung cancer (11.7%), and colon cancer (11.2%), with small differences in 2020, according to the report of the Korea Central Cancer Registry [2,3,4]. Early detection through national and public screening programs and advancements in treatment resulted in the proportion of surgically treated early gastric cancer (EGC) to increase from 28.6% in 1995 to 63.6% in 2019, and the 5-year survival increased from 43.9% (1993–1995) to 77.5% (2015–2019) [5]. Environmental factors, local dietary factors, socioeconomic factors, and Helicobacter pylori infections are considered important in the development of gastric cancer [6,7,8,9].
Chronology
Since 2004, 3 guidelines have been published and this is the 4th gastric cancer guideline published in Korea which is the revised version of previous evidence-based approach in 2018 [10,11,12]. It is the third guideline directed by the Korean gastric cancer association and prepared as a designated project assignment (No. 1020440) under the Research and Development Program for Cancer Control, conducted by the Ministry of Health and Welfare, South Korea. This is a collaborative work of the interdisciplinary working group that was nominated by the Korean Society of Medical Oncology, the Korean Society of Gastroenterology, the Korean College of Helicobacter and Upper Gastrointestinal Research, the Korean Society of Gastrointestinal Endoscopy, the Korean Society of Pathologists, the Korean Society of Abdominal Radiology, the Korean Society of Radiation Oncology, the Korean Society of Nuclear Medicine and the Korean Gastric Cancer Association along with the participation of experts in guideline development methodology (National Evidence-based Healthcare Collaborating Agency).
Methodology
After a collaborative review by the working group, key questions (KQs) were either updated or proposed (de novo). For the updated KQs, published literature was systematically searched using the MEDLINE, Embase, Cochrane Library and KoreaMed database, between January 2018 and December 2021, followed by a previous systematic search [10]. For de Novo KQ, a comprehensive search was performed up to December 2021, and starting date was not limited. Manual-searching was also performed for complementary results, and some literature published in 2022 and unpublished results from completed studies were also included. Screening and selection were performed by 2 reviewers. Criteria for selection and exclusion were predefined by the KQs. Initial screening of the articles was performed by title and abstract, and secondary screening was done by full-text review. Each panel independently selected the articles and compared the results for inconsistency. When disagreements occurred during the review process, a final agreement was reached through consensus with the involvement of a third review panel.
For quality assessments, the Cochrane Risk of Bias 2.0 (ROB) was used for randomized controlled trials (RCTs), the Risk of Bias for Nonrandomized Studies (RoBANS) was used for non-RCTs, the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) was used for diagnostic studies, and AMSTAR 2 was used for systematic reviews/meta-analyses.
In this revised edition, the level of evidence and grading of recommendation were redefined based on the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) methodology review [13]. The level of evidence was classified into 4 levels (Table 1) and the recommendation grading was categorized into 5 levels following the GRADE methodology (Table 2). We considered evidence level, benefit, harm, and clinical applicability as recommendation factors. The development working group reviewed the draft simultaneously and discussed for consensus.
Table 1. Level of evidence (Grading of Recommendations, Assessment, Development and Evaluation approach).
| Level | Definition |
|---|---|
| High | We are very confident that the true effect lies close to that of the estimated effect. |
| Moderate | We are moderately confident in the effect estimate. The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
| Low | Our confidence in the effect estimate is limited. The true effect may be substantially different from the estimated effect. |
| Very low | We have very little confidence in the estimated effect. The true effect is likely to be substantially different from the estimated effect. |
Table 2. Grading of recommendations.
| Grade | Definition |
|---|---|
| Strong for | The benefit of the intervention is greater than the harm, with high or moderate levels of evidence. The intervention can be strongly recommended in most clinical practice. |
| Conditional for | The benefit and harm of the intervention may vary depending on the clinical situation or patient/social value. The intervention is recommended conditionally according to the clinical situation. |
| Conditional against | The benefit and harm of the intervention may vary depending on the clinical situation or patient/social values. The intervention may not be recommended in clinical practice. |
| Strong against | The harm of the intervention is greater than the benefit, with high or moderate levels of evidence. The intervention should not be recommended in clinical practice. |
| Investigational | It is not possible to determine the recommendation direction owing to a lack of evidence or a discrepancy in results. Thus, further evidence is needed. |
Meta-analysis output and forest plots were computed from Review Manager (RevMan; Cochrane, London, UK) software. Evidence tables were summarized according to KQs, and the evidence-to-decision table was applied using GRADEpro (https://gradepro.org) software.
STATEMENT LIST
GENERAL CONSIDERATIONS
Endoscopy
Screening
Korea has shown the highest age-standardized incidence rates of gastric cancer worldwide, but the ratio of gastric cancer–related mortality to cancer incidence is much lower than that of other countries [14]. The Korean National Cancer Screening Program (KNCSP) for gastric cancer seems to have played a pivotal role in increasing the number of curable cancers by early detection and eventually improving overall survival (OS) [15]. The KNCSP for gastric cancer, launched in Korea in 2002, invites any Korean individual who is 40 years old or older to undergo endoscopy or upper gastrointestinal series (UGIS) every 2 years. A recent study showed that the screening group had a 41% decreased hazard ratio (HR) for gastric cancer mortality compared with the nonscreening group [16]. However, the reduction in gastric cancer mortality was only significant in the group that received endoscopic screening and was not in the group that received UGIS [17].
Diagnosis and classification of EGC
In the Japanese classification of gastric carcinoma, superficial gastric carcinoma is categorized according to morphologic features; polypoid lesions are classified as type I (protruding), flat lesions as type II (superficial), and ulcerated lesions as type III (excavated) [18]. Type II lesions are subdivided into 3 groups according to the elevation or depression of the lesion compared to the surrounding mucosa: IIa (superficial elevated), IIb (superficial flat) and IIc (superficial depressed). Tumors elevated by more than 3 mm are classified as type I [18].
Staging by endoscopic ultrasound (EUS)
EUS can be helpful for assessing the depth of local tumor invasion (T stage) as well as regional LN metastasis [19]. According to the results of the Cochrane review, the summary for sensitivity and specificity of EUS in discriminating T1 and T2 (superficial) vs. T3 and T4 (advanced) gastric carcinomas were 86% (95% confidence interval [CI], 81% to 90%) and 90 (95% CI, 87% to 93%), respectively [20] (Table 3). For the diagnostic capacity of EUS to distinguish T1 vs. T2 tumors, a meta-analysis of 46 studies (n=2,742) showed that the summary sensitivity and specificity were 85% (95% CI, 78% to 91%) and 90% (95% CI, 85% to 93%), respectively. For the capacity of EUS to distinguish between T1a (mucosal) vs. T1b (submucosal) cancers, the meta-analysis of 20 studies (n=3,321) showed that sensitivity and specificity were 87% (95% CI, 81% to 92%) and 75% (95% CI, 62% to 84%), respectively. Finally, for the metastatic involvement of LNs (N-stage), the meta-analysis of 44 studies (n=3,573) showed that sensitivity and specificity were 83% (95% CI, 79% to 87%) and 67% (95% CI, 61% to 72%), respectively. However, the heterogeneity between studies was high, reflecting that the diagnostic accuracy of EUS depends on the operator.
Table 3. Diagnostic accuracy of endoscopic ultrasound (Cochrane review).
| Test | Study No. | Patient No. | Sensitivity (%) | Specificity (%) |
|---|---|---|---|---|
| T1a vs. T1b | 20 | 3,321 | 87 (81 to 92) | 75 (62 to 84) |
| T1 vs. T2 | 46 | 2,742 | 85 (78 to 91) | 90 (85 to 93) |
| T1–2 vs. T3–4 | 50 | 4,397 | 86 (81 to 90) | 90 (87 to 93) |
| N− vs. N+ | 44 | 3,573 | 83 (79 to 87) | 67 (61 to 72) |
Values are presented as number of percentage (95% confidence interval).
Radiology
UGIS
The UGIS has been used for screening and evaluation for postoperative complications in gastric cancer. Recently, the percentage of participants of the KNCSP who undergo UGIS for screening of gastric cancer has decreased [21,22]. Studies comparing UGIS and endoscopy using the KNCSP database reported that UGIS showed lower detection sensitivity and disadvantage in long-term survival compared to endoscopy [17,23]. Although its role as screening method has been reduced, UGIS is still a valuable tool where endoscopy is not available or when the examinee cannot tolerate endoscopy.
CT
CT has been widely used to detect and diagnose gastric cancers, to determine the optimal treatment method via accurate staging (cTNM) and to identify therapeutic effects after surgery or anticancer treatments. MDCT with multiple parallel rows of X-ray detectors in the craniocaudal direction (z-direction) enables various high-quality MPR imaging. After the introduction of MDCT, the accuracy of gastric cancer staging and the detection of EGCs or small metastatic lesions have improved. Although isolated lung metastasis is not common in gastric cancer, chest CT can be helpful in case of esophageal involvement in GEJ cancer [24,25,26,27].
Protocol
An MDCT unit with 16 or more channels is recommended to acquire isotropic imaging with less than 1.25-mm collimation [28]. The patient needs to fast for at least 6 hours. Optimal gastric distension is critical for successful CT gastrography. Stomach distension is achieved using a negative contrast agent (effervescent gas-producing agent) or a neutral contrast agent (water). Anti-peristaltic drugs can reduce motion artifacts. Patient positioning is determined according to the location of the suspected lesion and the choice of oral contrast (e.g., supine/prone, right decubitus/left posterior oblique). Obtaining images from appropriate positions helps to evaluate the entire stomach in its distended state. Portal venous phase images usually provide information on the depth of the tumor, regional LN metastasis, and distant metastasis. Arterial phase images are useful to detect abnormal gastric wall enhancement and assess possible anatomic variation in the surgically relevant vasculature, such as replacing left hepatic artery arising from the left gastric artery.
KQ 1: Is acquisition of additional MPR images better than axial images alone in terms of T and N staging accuracy for gastric cancer patients?
Statement 1: Acquisition of MPR images in addition to axial images should be performed for gastric cancer staging using MDCT (evidence: low, recommendation: strong for).
The staging accuracy of MDCT has been reported to be 67.1% to 89.1% (median, 78.6%) for T staging and 49.3% to 79.5% (median, 68.8%) for N staging [29,30,31,32,33,34,35,36,37,38,39,40,41,42]. MDCT, which allows faster scanning with thinner slice thicknesses, can generate excellent reformation images, such as MPR images, CT gastrography, or virtual gastroscopy. In a meta-analysis, acquisition of MPR images in addition to axial images improved staging accuracy, especially in T staging (accuracy difference [95% CI], 0.10 [0.02 to 0.18] for T staging [P=0.01] and 0.04 [−0.04 to 0.13] for N staging [P=0.33]) [30,32,33] (Fig. 1). The 3D reformation images, such as CT gastrography or virtual gastroscopy, can improve the detection rate of EGCs, possibly allowing for more accurate T staging [29,30,39]. Ability to detect peritoneal metastases, it has been reported that MDCT has high specificity 57.1% to 100% (median, 96.5%) but low sensitivity 25.0% to 90.0% (median, 57.6%) [43,44,45,46,47,48].
Fig. 1. Forest plot comparing staging accuracy between axial plane plus MPR vs. axial plane only in multidetector row computed tomography.
MPR = multiplanar reformation; CI = confidence interval.
Magnetic resonance imaging (MRI)
Evaluation of liver metastases is one of the most potent applications of MRI in gastric cancer. Many studies on gastrointestinal malignancies, especially colorectal cancer, have shown that liver-specific contrast-enhanced MRI with diffusion-weighted imaging (DWI) is the most sensitive imaging method for the diagnosis of liver metastases [49]. Although no study has been restricted to gastric cancer patients, liver-specific contrast agent-enhanced MRI with DWI is expected to also be useful in diagnosing liver metastases in gastric cancer given its high contrast resolution. A meta-analysis showed the applicability of MRI in the evaluation of T stage and peritoneal metastases [50,51]. However, further investigation is needed to confirm these results due to the small number of patients included in these analyses.
FDG PET/CT
FDG PET/CT can reflect the degree of glucose uptake and metabolism in many cancer lesions [52]. FDG PET/CT can also provide good evidence to differentiate cancer recurrence from inflammatory and postoperative changes [53,54]. The degree of FDG uptake is known to be related to the biological characteristics of cancer cells, and the possibility of false negative results should be considered. High FDG uptake has been shown to be correlated with tumor hypoxia, increased Ki-67 index, and aggressive biological features, whereas low FDG uptake has been correlated with small tumor size, diffuse type Lauren classification, mucin predominant pathology, and HER2 negative expression in gastric cancer [55,56,57].
Diagnostic accuracy of FDG PET/CT for staging
KQ 2: Is additional FDG PET/CT helpful for accurate diagnosis in detecting LN and distant metastases during staging work-up for gastric cancer patients?
Statement 2: FDG PET/CT can be considered an additional supplementary diagnostic tool during staging workup (evidence: moderate, recommendation: conditional for).
A total of 20 studies were reviewed, 19 studies with 2,195 patients were included in the meta-analysis of the diagnostic ability of FDG PET/CT for detecting LN metastasis [44,58,59,60,61,62,63,64,65,66,67,68,69] or distant metastasis [64,66,70,71,72,73,74,75] in gastric cancer patients during staging. The pooled sensitivity and specificity of FDG PET/CT for detecting LN metastasis were 45% (95% CI, 34% to 57%) and 87% (95% CI, 80% to 92%), respectively. In the evaluation of distant metastasis, the pooled sensitivity and specificity were 61% (95% CI, 42% to 78%) and 97% (95% CI, 82% to 99%), respectively.
One possible factor for the low sensitivity of FDG PET/CT in detecting LN or distant metastasis could be the inclusion of diffuse (Lauren classification) or signet ring cell type cancers, which generally have lower FDG uptake. However, FDG PET/CT has a higher tendency for accurate diagnosis in intestinal-type pathology.
FDG PET/CT showed high specificity in detecting LN metastasis and distant metastasis and can be considered a supplementary diagnostic tool with diagnostic CT for staging the work-up of gastric cancer.
Diagnostic accuracy of FDG PET/CT for cancer recurrence
KQ 3: Can PET/CT be more accurate in diagnosing recurrence when recurrence is suspicious for gastric cancer patients who underwent curative surgery?
Statement 3: FDG PET/CT can be considered for the differential diagnosis of suspected recurrence in patients with gastric cancer after curative surgery (evidence: low, recommendation: conditional for).
A total of 13 studies with 1,567 patients were included in the meta-analysis [76,77,78,79,80,81,82,83,84,85,86,87]. The pooled sensitivity and specificity of FDG PET/CT for detecting the recurrence of gastric cancer were 81% (95% CI, 71% to 88%) and 88% (95% CI, 80% to 93%), respectively, with an area under the summarized receiver operating characteristic curve (AUC) of 0.91 (95% CI, 0.89 to 0.93). Of the 13 studies, 5 studies with 438 patients compared the diagnostic ability in detecting recurrence between FDG PET/CT and contrast-enhanced CT [76,78,80,86,88].
In the meta-analysis of these 5 studies, FDG PET/CT showed a pooled sensitivity of 72% (95% CI, 50% to 87%) and specificity of 89% (95% CI, 69% to 97%) with an AUC of 0.88 (95% CI, 0.85 to 0.90), whereas contrast-enhanced CT revealed a pooled sensitivity of 88% (95% CI, 74% to 95%) and specificity of 83% (95% CI, 65% to 93%) with an AUC of 0.92 (95% CI, 0.90 to 0.94). There was no statistically significant difference in diagnostic accuracy between FDG PET/CT and contrast-enhanced CT (P>0.05).
While FDG PET/CT showed higher sensitivity for detecting bone metastasis than contrast-enhanced CT, contrast-enhanced CT showed higher sensitivity for detecting peritoneal metastasis than FDG PET/CT. Because of the high specificity, PET/CT could be helpful for the differential diagnosis of equivocal lesions on contrast-enhanced CT.
Regarding recurrence, 2 studies assessed the diagnostic value of FDG PET/CT for detecting recurrence in 29 patients with elevated levels of serum tumor markers and negative results on conventional radiological imaging [83]. Among these 29 patients, FDG PET/CT detected cancer recurrence in 17 patients (59%). FDG PET/CT could be useful for detecting recurrence in patients who showed equivocal results on contrast-enhanced CT and elevated serum tumor marker levels but negative findings on conventional imaging.
Pathology
Preparation of the specimens
For resected gastric cancer specimens, the stomach is opened along the greater curvature, unless the tumor is located on the greater curvature (in which case, it is opened along the lesser curvature). For endoscopic mucosal resection (EMR)/ESD specimens, the specimen is spread out with the mucosal side up and pinned on a flat board. The proximal and distal directions are marked for orientation.
Specimen fixation
After completing the preparation process, the specimens are immediately immersed in 10% buffered formalin solution (as quickly as possible). The volume of fixative solution should be more than ten times that of the specimen [89]. Proper fixation time (between 24 and 48 hours) at average room temperature is recommended for additional immunohistochemical or genomic evaluation [18,90].
Macroscopic types
Superficial gastric cancer can be subclassified into 5 categories. Protruding (EGC type I), superficial elevated (EGC type IIa), superficial flat (EGC type IIb), superficial depressed (EGC type IIc), and excavated (EGC type III) [18].
Based on Borrmann’s classification, the gross type of AGC can be divided into polypoid (type 1), ulcerofungating (type 2), ulceroinfiltrative (type 3), diffuse infiltrative (type 4) and unclassifiable (type 5) [91,92].
Inspection and sectioning of the specimens
For resected specimens, the location, size (maximum diameter), number, macroscopic types, appearance of the tumor and length of the closest proximal and distal resection margins should be measured and recorded. The deepest part of the tumor invasion should be noted. It is also necessary to assess whether there are findings other than the tumor lesion, such as congestion, hemorrhage, ulcer, and perforation. For EMR/ESD samples, all specimens are collected and embedded in blocks. The lateral and basal resection margins should be marked with ink, which helps with proper evaluation of the margins.
For sectioning, EMR/ESD specimens should be sectioned serially at 2-mm intervals parallel to a line that includes the closest lateral margin of the specimen. If the lesion is grossly AGC, at least 4 representative sections should be taken, including the deepest part of the tumor invasion. If the lesion is grossly EGC, grid mapping should be performed at a width of 4 to 5 mm. If there is suspicion of resection margin involvement with the tumor lesion, additional sections should be taken. In postchemotherapy gastrectomy specimens, representative sections are sufficient if the lesion is grossly obvious. However, the entire tumor bed must be microscopically examined when there are no residual cancer cells in the representative section, residual lesion is small or grossly inconspicuous. For multiple tumors or lesions with unusual configurations, suitable sectioning should be implemented for proper evaluation on a case-by-case basis.
Histologic classification
The World Health Organization (WHO) classification system of digestive tumors, 5th edition, is used for the pathologic classification of gastric carcinoma [93,94]. In addition, the Lauren classification can be applied in resected specimens, including ESD specimens [95].
A. WHO classification
a. Tubular adenocarcinoma
Tubular adenocarcinoma is the most common histologic subtype of gastric carcinoma and is characterized by irregularly distended, fused, or branching tubules of various sizes. Tumors with solid structures and rare tubule formation, corresponding to “poorly 1 (solid type): por1” in the Japanese Gastric Cancer Association classification, are included in this group [18]. Prominent intraluminal mucus and inflammatory debris can be observed.
b. Papillary adenocarcinoma
This relatively rare subtype usually shows an exophytic growth pattern and papillary tumor structure with a central fibrovascular core with columnar or cuboidal tumor cells. The tumor is classified as papillary adenocarcinoma when more than 50% of the tumor area shows papillary structures [96]. Papillary adenocarcinoma is frequently associated with liver metastasis, a higher rate of LN involvement and poor outcome [96,97,98].
c. Poorly cohesive carcinoma (PCC), including signet-ring cell carcinoma (SRCC)
PCCs are composed of poorly cohesive neoplastic cells that are isolated or form small aggregates without gland formation. This type includes SRCC and nonsignet-ring cell variants (PCC-NOS). SRCC is diagnosed when the tumor cells were predominantly or exclusively an SRC component [94]. Recent studies have revealed that the clinical behavior may differ in SRCC and PCC-NOS, with a relatively poor prognosis of PCC-NOS compared to SRCC and different mutational profiles between SRCC and PCC-NOS [99,100,101].
d. Mucinous adenocarcinoma
This subtype is defined by malignant epithelial cells and extracellular mucin pools, which comprise more than 50% of the tumor volume. The tumor cells can show glandular architecture and irregular cell clusters, with occasional single scattered tumor cells, including floating SRCs. Mucinous adenocarcinoma tends to be diagnosed at a more advanced stage, which correlates with deeper invasion depth and poorer survival outcomes compared with nonmucinous gastric cancer [102,103].
e. Mixed adenocarcinoma
This type of tumor refers to carcinomas having a discrete mixture of both glandular (tubular/papillary) and signet ring/poorly cohesive components. It is recommended that any distinct histological component be reported. Recent data suggest that patients with mixed adenocarcinomas have a poorer clinical outcome than those with a pure subtype of carcinoma, especially in EGC [104,105,106]. However, there are still no clear diagnostic criteria for the minimum ratio of glandular to signet ring/poorly cohesive components for the definition of mixed adenocarcinoma.
f. Other histological subtypes
According to the WHO classification, other rare subtypes include gastric (adeno)carcinoma with lymphoid stroma, hepatoid adenocarcinoma, micropapillary adenocarcinoma, gastric adenocarcinoma of fundic-gland type, mucoepidermoid carcinoma, Paneth cell carcinoma and parietal cell carcinoma.
B. Grading
The grading of adenocarcinoma can be applied to tubular and papillary carcinomas but not to other subtypes. Well-differentiated adenocarcinoma is composed of a tumor with well-formed glands, whereas poorly differentiated adenocarcinoma shows poorly formed glands or no luminal structures (solid cluster). Although the WHO classification recommends a 2-tier grading system, low grade (well or moderately differentiated) vs. high grade (poorly differentiated), considering that most pathologists and clinicians are more familiar with a 3-tier grading system, we have agreed to use the current 3-tier grading system (well/moderately/poorly differentiated) to avoid confusion.
C. Lauren classification
The Lauren classification divides gastric cancers into intestinal, diffuse, and mixed types [95]. According to the recent WHO classification, well or moderately differentiated papillary and tubular adenocarcinomas are classified as intestinal type, whereas PCCs, including SRCC, are classified as diffuse type. In addition, poorly differentiated adenocarcinomas forming solid areas are classified as indeterminate type. Mucinous adenocarcinoma can be classified as intestinal, diffuse or indeterminate according to the differentiation of the main tumor components [94]. The mixed type is used for tumors containing approximately equal proportions of intestinal and diffuse components.
Addendum: To determine the feasibility of EMR/ESD specimens in gastric cancer, many studies use the 2-tier categories (differentiated or undifferentiated types) of the Japanese guidelines [107]. In this classification, tumors with solid structures correspond to the undifferentiated type. To avoid confusion with undifferentiated carcinoma in the WHO classification, it is not recommended to use the term ‘differentiated/undifferentiated type’ in pathology reports.
Tumor size
Tumor size describes the largest dimension (cm) of the tumor.
Depth of invasion
In the staging of gastric cancer, the pT category is determined according to the depth of invasion of the tumor. Tumors with invasion beyond the proper muscle layer are classified as AGC, and tumors with mucosal or submucosal layer invasion are classified as EGC. The submucosal invasion depth is divided into the upper third (sm1), middle third (sm2), and lower third (sm3). When the proper muscle layer is lost at the ulcer site and there is a tumor in that area, it is considered subserosal invasion. Even if there is no tumor cell invasion of the muscle, if the tumor invades below the soft imaginary line connecting the proper muscle layers, it is staged as proper muscle invasion.
For endoscopic resection specimens, submucosal invasion depth is measured from the lowest surface of the muscularis mucosa. When the muscularis mucosa is lost in the area of deepest invasion, the invasion depth is measured from the virtual line that smoothly connects adjacent normal layers.
Lymph node
A sufficient number of regional LN dissections and pathological evaluations are important to ensure the proper diagnosis of stage N. The pathologic assessment should contain the number of nodes and the number of positive nodes. It is necessary to assess at least 16 local nodes to evaluate N3a staging; however, some studies suggest that it is desirable to remove/assess 30 or more nodes [108,109].
A tumor deposit is defined as a discrete tumor nodule within the lymphatic drainage zone of primary carcinoma without identifiable LN tissue, blood vessels or neural structures [108]. Tumor deposits, in which metastatic tumor lesions in the subserosal fat separate from adjacent primary gastric cancer are observed without evidence of LN tissue, are considered to be local LN metastases.
Resection margin
In gastric cancer, the proximal and distal margin status are described, and where applicable, the circumferential margin status is additionally described in GEJ cancer. The safety margin describes the distance between the resection margin and the tumor. If the distance of the safety margin in gross description is different from that of microscopic observation, the findings of microscopic observation are described.
For mucosal resection margins of endoscopic resection specimens, the direction close to the resection margin and the distance from the resection margin are described. The deep resection margin is also measured at the nearest point from the tumor and described.
Lymphatic invasion, vascular invasion, and perineural invasion
The presence or absence of lymphovascular invasion and perineural invasion should be described. For endoscopic resection specimens, it is recommended to separately mention lymphatic invasion and vascular invasion. The use of immunohistochemical staining (D2-40) could be helpful in identifying lymphatic invasion.
Regression grade
For the grading of primary tumor regression after neoadjuvant therapy, the modified Ryan system is recommended [110] (Table 4).
Table 4. Regression grade.
| Grade | Definition |
|---|---|
| Grade 0 | Complete response (no viable cancer cells) |
| Grade 1 | Near-complete response (single cells or rare small groups of cancer cells) |
| Grade 2 | Partial response (residual cancer with evident tumor regression, but more than single cells or rare small groups of cancer cells) |
| Grade 3 | Poor or no response (extensive residual cancer with no evident tumor regression) |
Peritoneal washing
Positive cancer cells in peritoneal washing cytology are classified as metastatic disease (pM1). There is evidence that positive cancer cells in the washing cytology of AGC patients are correlated with poor prognosis. Peritoneal washing cytology could be helpful in the staging of AGC.
Biomarkers
A. HER2
Because HER2 positivity is an indication for anti-HER2 targeted therapy, the HER2 status should be evaluated before systemic therapy and re-evaluated for recurrent and metastatic lesions. IHC tests should first be performed for the evaluation of HER2 status [111,112]. IHC 3+ is considered positive for HER2 overexpression, while IHC 0–1+ is considered negative [113]. IHC 2+ is regarded as an equivocal finding and should be followed by ISH tests. The area with the strongest IHC intensity should be selected and stained for HER2 and chromosome enumeration probe (CEP) 17. The criterion for HER2 amplification was a HER2:CEP17 ratio of ≥2. If CEP17 polysomy is present and the ratio is <2, an average HER2 signal of >6 is interpreted as a positive finding. IHC 3+ or IHC 2+ and ISH positivity are considered HER2-positive.
B. Microsatellite instability (MSI)
MSI status can be assessed by either polymerase chain reaction (PCR) or IHC for the 4 DNA mismatch repair (MMR) proteins [114]. Instability is examined by PCR of a representative panel of microsatellites [115]. The grade of the instability is determined by the numbers of unstable microsatellites: MSI-high (MSI-H), MSI-low (MSI-L), or microsatellite stable (MSS) [116]. In the IHC method, IHC staining is performed for the 4 MMR proteins MLH1, MSH2, PMS2, and MSH6 [117]. When the expression of any of the MMR proteins is lost, the case is considered MMR deficient (dMMR).
MSI-H/dMMR gastric cancer is classified as a separate subtype in the molecular classifications of gastric cancer and shows elevated mutation rates (high tumor mutation burden) and distinctive patterns of methylation [118]. This subtype has unique clinical characteristics, including distal location, high frequency of intestinal-type histology, lower stage, and favorable prognosis. In the palliative setting, MSI-H/dMMR is well known predictive biomarker to identify patients with gastric cancer who are most likely to benefit from immune checkpoint inhibitor (ICI) therapy [119].
C. Epstein–Barr virus (EBV)
The presence of the EBV genome can be examined by ISH to EBV-encoded RNA [120,121]. When signals in the tumor cell nuclei are observed, the case is considered EBV-positive. EBV-positive gastric cancer is classified as a separate subtype in the molecular classification of gastric cancer and shows hypermethylation different from that of the MSI subtype [118]. This subtype is distinct in its proximal location, relation to poorly differentiated histology, lower stage, and good prognosis.
D. PD-L1 IHC
The PD-L1 interpretation method and cutoff value depend on the antibody clones and the predefined settings of approved clinical trials. However, most anti-programmed cell death protein 1 (PD-1)/PD-L1 therapies require the CPS interpretation system [122,123]. The CPS enumerator includes the number of PD-L1-stained tumor cells showing partial or complete membrane staining intensity and the number of PD-L1-stained mononuclear immune cells (lymphocytes and macrophages) within tumor nests and adjacent stroma.
There have been 2 different PD-L1 assays coupled to clinical trials for gastric cancer patients. The PD-L1 IHC 22C3 pharmDx assay uses CPS ≥1 as a criterion for PD-L1 positivity, and the 28-8 pharmDx assay uses a cutoff of CPS ≥5 [124,125].
For reliable PD-L1 interpretation, different cutoff values should be applied depending on the antibody used. It is also recommended to re-evaluate PD-L1 staining in cases of recurrent or metastatic tumors.
E. Next-generation sequencing (NGS)
Biomarkers associated with advanced gastric cancer management include HER2, MSI, PD-L1, tumor mutational burden (TMB) status, and NTRK gene fusion according to recent National Comprehensive Cancer Network (NCCN) guidelines [111]. For biomarker testing, IHC, ISH, or target PCR methods should be preferentially considered; however, validated NGS assay performed in an appropriate environment could be used for the identification of the biomarkers mentioned above. Additionally, there are some targets that could be tested by NGS assay and have shown promising clinical results in AGC, such as FGFR2 amplification, epidermal growth factor receptor (EGFR) amplification, MET amplification, and alteration of homologous recombination deficiency-related genes [126,127,128,129].
TMB, which can be quantified by NGS assays, has been suggested as a potent biomarker for first line pembrolizumab-based therapy in patients with AGC [130]. While whole-exome sequencing is considered the gold standard for TMB, recent targeted gene panels also provide fairly accurate quantification of TMB [131]. However, the lack of cutoffs and different quantification methods across different panels is one of the main limitations to adopting TMB as a biomarker in clinical practice.
For accurate and reliable NGS assays, tissue preparation is one of the most important factors [132]. Most targeted NGS assays require total DNA and RNA amounts ranging from 10 to 300 ng, which can be obtained from both formalin-fixed, paraffin-embedded tissue and cytology specimens. In addition, a sufficient tumor fraction of the sample (surface area >10%–20% and 5 mm2, respectively) could also affect reliable NGS results.
For further detailed information about the pathology for gastric cancer, please refer to the Guideline for Standardized Pathology Report for Gastric Cancer, first edition and the upcoming revised version [133,134].
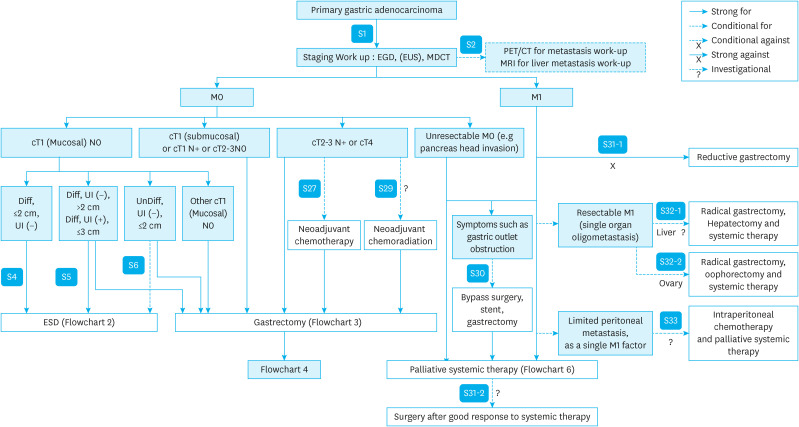
OVERALL TREATMENT ALGORITHM (Flowchart 1)
Flowchart 1. Overall treatment algorithm.
EGD = esophagogastroduodenoscopy; EUS = endoscopic ultrasound; MDCT = multidetector row computed tomography; PET = positron emission tomography; CT = computed tomography; MRI = magnetic resonance imaging; Diff = well or moderately differentiated; UI = ulcer lesion; UnDiff = poorly differentiated/poorly cohesive (including signet-ring cell); ESD = endoscopic submucosal dissection.
ENDOSCOPIC TREATMENT (Flowchart 2)
Flowchart 2. Endoscopic treatment.
Diff = well or moderately differentiated; UI = ulcer lesion; UnDiff = poorly differentiated/poorly cohesive (including signet-ring cell); ESD = endoscopic submucosal dissection; APC = argon plasma coagulation.
-
KQ 4: Can endoscopic resection for EGC that meets classical absolute indications result in comparable survival to that of gastrectomy?
Statement 4: Endoscopic resection is recommended for well or moderately differentiated tubular or papillary EGCs meeting the following endoscopic findings: endoscopically estimated tumor size ≤2 cm, endoscopically mucosal cancer, and no ulcer in the tumor (evidence: moderate, recommendation: strong for).
ESD has been used as a minimally invasive therapy modality for EGC since the early 2000s in Korea [135,136]. The data in Korean National Health Insurance Service System showed 23,828 cases of ESD for EGC between November 2011 and December 2014 [136]. Previous studies have suggested that ESD could be considered as the first-line therapy modality for mucosal confined EGC with well or moderately differentiated tubular adenocarcinoma or papillary adenocarcinoma, with a tumor size ≤2 cm, and without ulcer in the tumor (classical absolute indication) because these findings indicated that the lesions had a very low risk of LN metastasis [137] and ESD allows high rates of en bloc curative resection with low adverse event rates [135,138,139,140,141]. A large retrospective Japanese study including 5,265 patients who had undergone gastrectomy with LN dissection for EGC showed that none of the 1,230 well differentiated intramucosal cancers with diameters less than 30 mm were associated with metastases, and none of the 929 lesions without ulceration were associated with LN metastasis regardless of tumor size [137]. For classical absolute indications, the en bloc resection rate was 97.1%–99%, the curative resection rate was 91.5%–96.4%, and the local recurrence rate was 0.2%–1.8% [138].
Studies comparing survival data between ESD and gastrectomy for the classical absolute indication have rarely been conducted. In most studies, ESD cases were mixed with classical absolute and expanded indications and were not separately analyzed.
In Korean retrospective cohort studies, when the patients met the classical absolute indications, the 5-year OS rates (ESD, 93.6%–96.4% vs. gastrectomy, 94.2%–97.2%) and 10-year OS rates (ESD, 81.9% vs. gastrectomy 84.9%) did not differ between treatment methods [139,140,141].
A small Korean study including 35 endoscopic resections and 20 gastrectomies with same settings, showed no difference in OS (months) (93.4±3.2 [endoscopic resection], 85.8±5.5 [gastrectomy]) or disease-free survival (DFS) (months) (89.7±3.6 [endoscopic resection], 90.4±3.5 [gastrectomy]) [142]. Similar results were reported in a Japanese study, where patients were divided in different age groups (<65 years, ≥65 years). When the cases met the classical absolute indications, there were no significant difference in OS between endoscopic resection and gastrectomy in all age groups [143].
The 5-year metachronous recurrence rates were higher after endoscopic resection (5.8%–10.9%), compared to gastrectomy (0.9%–1.1%) [139,140,141]. Close endoscopic surveillance should be performed after ESD for early detection of metachronous cancer.
After endoscopic resection, preservation of the stomach may be associated with higher incidence of metachronous cancer, however, better QOL, shorter hospital stay, lower costs and lower treatment-related complication rates may be more anticipated compared to gastrectomy [139,140,141,142,144].
KQ 5: Is there any difference in survival rate between ESD and surgery in the treatment of well or moderately differentiated, tubular or papillary, EGC meets the following endoscopic findings: endoscopically estimated tumor size >2 cm, endoscopically mucosal cancer, and no ulcer in the tumor or endoscopically estimated tumor size ≤3 cm, endoscopically mucosal cancer, and ulcer in the tumor?
Statement 5: ESD as well as gastrectomy with LN dissection can be indicated for well or moderately differentiated tubular or papillary EGCs meeting the following endoscopic findings: endoscopically estimated tumor size >2 cm, endoscopically mucosal cancer, and no ulcer in the tumor, or endoscopically estimated tumor size ≤3 cm, endoscopically mucosal cancer, and ulcer in the tumor (evidence: moderate, recommendation: strong for).
Endoscopic resection for EGC is limited in that LN dissection cannot be included during the procedure. Therefore, in order to achieve curative resection with comparable survival to that of surgery by endoscopic resection, early cancers with very low risk of LN metastasis should be carefully selected. The clinically acceptable threshold of LN metastasis might be equivalent to the context of perioperative mortality following radical gastrectomy (0.1%–0.3% in high-volume centers in Korea and Japan) [145,146,147]. In addition, with endoscopic resection, it is technically feasible to achieve en bloc resection which is important to avoid remnant tumors or local recurrence after the procedure.
When the following criteria were met in the pathologic review of endoscopic resection specimens, the extragastric recurrence (nodal or distant metastasis) rate after endoscopic resection was between 0% and 0.21%, which was comparable to that of radical gastrectomy: well or moderately differentiated tubular adenocarcinoma or papillary adenocarcinoma, en bloc resection, negative lateral resection margins, negative vertical resection margin, no lymphovascular invasion, and 1) tumor size >2 cm, mucosal cancer, and no ulcer in the tumor or 2) tumor size ≤3 cm, mucosal cancer, and ulcer in the tumor [148,149,150]. The OS was also comparable between patients undergoing endoscopic resection and those treated with radical surgery (93.3%–96.4%. vs. 92.0%–97.2%) [139,144,151,152,153,154,155,156,157,158,159,160,161].
Although a number of retrospective cohort studies support ESD, no prospective trial has compared the outcomes with those of standard operations based on these criteria where concerns for node metastases may still be present [149,162,163,164]. Thus, gastrectomy with LND may also be considered a valid treatment option, especially in cases of ESD with technical difficulty or where periodic endoscopic follow-up may not be feasible or affordable.
KQ 6: Is there any difference in the survival rate between surgery and ESD for poorly differentiated tubular or poorly cohesive (including signet-ring cell) EGCs meeting the following endoscopic findings: endoscopically estimated tumor size ≤2 cm, endoscopically mucosal cancer, and no ulcer in the tumor?
Statement 6: Endoscopic resection could be cautiously considered for poorly differentiated tubular or poorly cohesive (including signet-ring cell) EGCs meeting the following endoscopic findings after sufficient discussion: endoscopically estimated tumor size ≤2 cm, endoscopically mucosal cancer, and no ulcer in the tumor (evidence: low, recommendation: conditional for).
EGCs with poorly differentiated tubular and PCC (including SRCC) are associated with a higher risk of LN metastasis than well and moderately differentiated tubular EGCs. Thus, endoscopic resection can be considered very cautiously.
In previous Japanese Gastric Cancer guidelines, through a literature review of previous studies, endoscopic resection could be considered in poorly differentiated tubular adenocarcinoma or PCC (including SRCC) histologic confirmation from forceps biopsy specimens, endoscopically estimated tumor size ≤2 cm, endoscopically mucosal cancer, and no ulcer in the tumor [107]. When the criteria were fulfilled, the risk of LN metastasis was reported to be 0%–2.3% [165,166,167].
Under the mentioned endoscopic findings, endoscopic resection could be considered for initial treatment. However, when risk factors for node metastasis (tumor size >2 cm, submucosal invasion, ulcer in the tumor, and lymphovascular invasion) are confirmed in pathologic reports, additional gastrectomy may be required [168].
In this guideline, we reviewed papers published after the previous edition. To date, there has been no prospective RCT comparing the long-term OS of endoscopic resection with that of gastrectomy with LN dissection, the standard treatment for these indications [169]. According to retrospective studies, there was no difference between gastrectomy and endoscopic resection in terms of OS, but endoscopic resection had a higher local recurrence rate in terms of recurrence-free survival (RFS), which is consistent with the findings of previous studies [144,170,171]. In a prospective, single-arm, phase III observational study in Japan (JCOG 1009/1010), the curative resection rate of the endoscopic resection group in undifferentiated EGC was 71% (195/275), and during the median follow-up period of 69.9 months, the 5-year OS rate was 99.3% (95% CI, 97.1% to 99.8%) and 5-year RFS rate was 98.9% (95% CI, 96.5% to 99.6%) [172]. In Korea, a study on Comparison of Endoscopic Resection And Surgery for Early Gastric Cancer with undifferentiated histological type: a multicenter randomized controlled trial (ERASE-GC trial, NCT04890171), is under way; the results of this study should be followed-up.
To date, the standard treatment for these criteria has been gastrectomy with LN dissection. Only retrospective cohort studies support these criteria for endoscopic resection, and the results of prospective trials are still lacking. A significant portion of these criteria estimated by pre-endoscopic resection work-up is confirmed to be out of criteria by the pathologic examination of endoscopic resection specimens. Thus, standard operation (gastrectomy with LN dissection) can also be considered for cases meeting these criteria. Therefore, it is advisable to decide on a treatment method after sufficient discussion with the patient about the possibility of LN metastasis and complications of the endoscopic procedure and surgery.
KQ 7: When the results of endoscopic resection for EGC do not meet the criteria for curative resection, can additional surgery improve survival outcome compared to observation?
Statement 7: Additional surgery is recommended when the result of endoscopic resection for EGC does not meet the criteria for curative resection or when there is lymphovascular invasion or positive vertical margin (evidence: low, recommendation: strong for).
The results of endoscopic resection of EGC could be revealed as being beyond the criteria for curative resection based on pathological evaluation of resected specimens. Resected tumor characteristics that do not meet the following criteria are considered noncurative: 1) differentiated type (well or moderately differentiated tubular or papillary adenocarcinoma) mucosal cancer of any size without ulcer, 2) differentiated type mucosal cancer measuring ≤3 cm with ulcer, 3) differentiated type cancer with minute submucosal invasion (invasion depth ≤500 µm) measuring ≤3 cm, or 4) undifferentiated type (poorly differentiated tubular adenocarcinoma or PCC) mucosal cancer measuring ≤2 cm without ulcer. Lymphovascular invasion and positive vertical margins are also important conditions that require additional surgical treatment.
As a result of a literature search for reinforcement of the up-to-date guidelines, a total of 17 studies that compared additional surgery and observation were included in the final table of evidence [149,163,173,174,175,176,177,178,179,180,181,182,183,184,185]. Most studies appeared to have a high risk of bias in terms of participant comparability. Patients who did not undergo surgery were older and tended to have a higher incidence of comorbidities than those who underwent additional curative surgery [149,163,173,174,175,176,178,181,183,185]. In addition, there was a significant difference in tumor-related characteristics [149,163,173,175,176,177,179,180,182,183,184].
The 5-year OS rate in 15 studies was significantly higher in the surgery group than in the observation group [149,163,175,176,180,181,183,185]. Regarding disease-specific survival in 12 studies, all except one study showed a survival benefit for additional surgery [163,182,183], although this difference was not statistically significant in several studies [173,175,176,178,180,184,185]. In a study that performed propensity score matching analysis, it was also found that 5-year overall and disease-specific survival rates were significantly higher in the surgery group than in the observation group (91.0% and 99.0% in the surgery group and 75.5% and 96.8% in the observation group) [180].
Among patients who underwent additional surgery after noncurative endoscopic resection, LN metastasis was found in 2.0%–20.0% of patients [149,163,173,175,176,177,179,180,181,182,183,184,185]. Given the high incidence of LN metastasis and survival benefit associated with curative surgery, additional gastrectomy with LN dissection is recommended when the result of endoscopic resection for EGC does not meet the criteria for curative resection.
The survival benefit of additional surgery in older patients (>75 years) is controversial [164,178,179,183,186]. In addition, curative surgery may not be feasible in some patients because of underlying diseases or poor general conditions. In these patients, observation with regular follow-up could be a valid option when they give informed consent after receiving an explanation of the risk of recurrence.
KQ 8: When the results of endoscopic resection for EGC have only a positive horizontal margin and meet all other criteria for curative resection, are re-ESD or APC, or close observation acceptable options in terms of recurrence, mortality and survival rate compared to gastrectomy?
Statement 8: After endoscopic resection in EGC, endoscopic treatment such as ESD and APC could be considered for EGCs that have only positive lateral margins and meet all other criteria for curative resection (evidence: low, recommendation: conditional for).
It has been reported that there is little risk of LN metastasis with en bloc resection, when only the lateral margin is positive and other criteria for complete resection are met. In the case of differentiated-type EGC with lateral margin positivity after ESD, when only close observation was performed, the 5-year local recurrence rate was 11.9% and there was no gastric cancer related mortality [187]. Therefore, close observation, endoscopic treatment (ESD or APC) and gastrectomy are considered as possible treatment options in these cases.
Seven retrospective studies compared the recurrence rate of endoscopic treatments including re-ESD and APC, with gastrectomy or close observation [188,189,190,191,192,193,194]. The mean follow-up period for the 6 studies was 60 months, and these studies included both differentiated and undifferentiated cancers. Local recurrence rates were 0% (95% CI, 0% to 0.02%; 0/163) in the gastrectomy group, 1.9% (95% CI, 0.5% to 6.9%; 2/101) in the re-ESD group, 13.4% (95% CI, 7.2% to 23.6%; 9/67) in the APC group, and 23.5% (95% CI, 17.4% to 30.1%; 35/149) in the observation group. Overall, endoscopic treatments (including both re-ESD and APC) significantly lowered the recurrence rate compared to close observation (relative risk [RR], 0.22; 95% CI, 0.06 to 0.86; P=0.03) in the meta-analysis (Fig. 2A). The recurrence rate of the endoscopic treatment group was significantly higher than that of the gastrectomy group (RR, 6.45; 95% CI, 1.17 to 35.52; P=0.03) (Fig. 2B), and in the gastrectomy group, local residual cancer was found in 64.7% (95% CI, 56.8% to 71.9%; 97/150) and the LN metastasis rate was 0.6% (95% CI, 0.1% to 1.9%; 1/150). However, all local recurrence cases can be successfully managed with further endoscopic treatment or surgery. Gastric cancer mortality was reported in 3 studies, and there was no gastric cancer-related death in the endoscopic treatment group or the observation and gastrectomy group [188,189,192]. Thus, considering QOL after endoscopic treatment and mortality related to gastrectomy, endoscopic treatment could be considered in patients with a positive lateral margin after ESD in EGC. Considering the recurrence rate of endoscopic treatment (5.8%; 95% CI, 2.29% to 9.21%; 10/174), close follow-up after endoscopic treatment is necessary. Although no deaths related to gastric cancer were reported in any of the 3 groups, the study population of each study was not large, and the baseline characteristics were different due to the observational study design. Further research is needed to compare the mortality and survival outcomes of close observation, endoscopic treatment, and gastrectomy in a large population.
Fig. 2. Forest plot for comparison of local recurrence. (A) Risk of local recurrence in endoscopic treatment group vs. follow-up without therapy group. (B) Risk of local recurrence in endoscopic treatment group vs. gastrectomy.
Tx = treatment; CI = confidence interval.
There were 3 retrospective studies comparing gastrectomy and close observation in patients with a positive lateral margin after ESD in differentiated type EGC [188,189,192]. The local recurrence rate of the gastrectomy group (0%; 95% CI, 0% to 0.1%; 0/44) was significantly lower than that of the close observation group (19.6%; 95% CI, 12.9% to 28.6%; 19/97), but cancer-related mortality was zero in both groups. All local recurrence cases in the observation group can also be managed with endoscopic treatment or surgery. In patients who underwent gastrectomy, local residual cancer was found in 51.6% (95% CI, 34.8% to 68.0%; 16/31), but the LN metastasis rate was 0% (95% CI, 0% to 0.1%; 0/44). In particular, long-term follow-up studies showed that a cancer-positive lateral margin length longer than 6 mm was significantly associated with local recurrence [187]. Therefore, close observation could be considered a selective treatment option in cases of positive lateral margins in differentiated-type EGC. Recently, a retrospective study comparing gastrectomy and nonsurgical treatments (endoscopic treatment [6/52] and close observation [46/52]) in undifferentiated-type EGC has also been published [194,195]. The local recurrence rate was 0% in the surgical group and 21.2% (11/52) in the nonsurgical group, and the 5-year survival rate was 87.8% in the nonsurgical group, lower than the 95.0% in the surgical group, but without statistical significance. Therefore, close observation may be considered in elderly patients or patients with high morbidity in undifferentiated-type EGC, but further studies are needed.
KQ 9: Can H. pylori eradication prevent metachronous gastric cancer in patients who are successfully treated by endoscopic resection for EGC with H. pylori?
Statement 9: H. pylori eradication is recommended for the prevention of metachronous gastric cancer in patients successfully treated by endoscopic resection of EGC with H. pylori infection (evidence: moderate, recommendation: strong for).
H. pylori was proposed as the first-class carcinogen for gastric cancer by the 1999 WHO. H. pylori infects approximately 50% of the world’s population. Eradication provided a significant benefit for asymptomatic infected individuals (pooled incidence rate ratio, 0.62; 95% CI, 0.49 to 0.79) [196]. Therefore, eradication of H. pylori is considered an important strategy to prevent gastric cancer.
In addition, eradication of H. pylori would be an important issue for the prevention of metachronous gastric cancer in patients successfully treated by endoscopic resection of EGC with H. pylori. We identified 3 RCTs that observed metachronous gastric cancer and precancerous lesions in both the H. pylori eradication treatment and noneradication treatment groups in patients successfully treated by endoscopic resection for EGC with H. pylori [197,198]. The median follow-up periods were 3–5 years. The incidence rate of metachronous gastric cancer and precancerous lesions during the follow-up was 4.80% (41/856) in the H. pylori treatment group and 9.75% (87/892) in the non-H. pylori treatment group. The risk of metachronous gastric cancer and precancerous lesions among patients who underwent H. pylori eradication treatment was significantly lower than that among patients who did not undergo eradication treatment (HR, 0.45; 95% CI, 0.31 to 0.66). In addition, H. pylori eradication treatment had benefits for the risk of metachronous gastric cancer based on the study by Fukase et al. [197], which included only metachronous gastric cancer (HR 0.34; 95% CI 0.16-0.73) (Fig. 3). Therefore, H. pylori eradication is helpful for the prevention of metachronous gastric cancer in patients successfully treated by endoscopic resection of EGC with H. pylori.
Fig. 3. Forest plot for a comparison of the risk of metachronous gastric cancer between Helicobacter pylori eradication (experimental) vs. no treatment (control).
SE = standard error; IV = interval variable; CI = confidence interval.
SURGICAL TREATMENT (Flowchart 3, 4, 5)
Flowchart 3. Approach and extent of gastrectomy.
DG = distal gastrectomy; TG = total gastrectomy; PPG = pylorus-preserving gastrectomy; PG = proximal gastrectomy; LND = lymph node dissection; LN = lymph node.
Flowchart 4. Treatment plans after gastrectomy.
LN = lymph node; XELOX = capecitabine and oxaliplatin.
*To obtain negative margin, single or combinations of various methods including intraoperative frozen section, perioperative gastroscopy, various preoperative clipping or dyeing, fluorescence imaging technique, ultrasonography, and simple X-ray, etc. can be applied.
†Preferred in pStage II with LN+ or pStage III.
Flowchart 5. Treatment guidelines in gastroesophageal junction adenocarcinoma.
ESD = endoscopic submucosal dissection; TG = total gastrectomy; PG = proximal gastrectomy; LND = lymph node dissection.
KQ 10: Is RY and BI reconstruction better than BII reconstruction following DG in gastric cancer regarding functional or nutritional outcomes?
Statement 10: There are no differences in functional outcomes, or nutritional outcomes (weight loss, albumin) between BI, BII, and RY reconstruction methods after DG. Each reconstruction method has advantages and disadvantages, and surgeons may make case-specific decisions (evidence: high, recommendation: conditional for).
Functional and nutritional outcomes may differ according to the various reconstruction methods including BI, BII, and RY [199].
Well-designed studies comparing each reconstruction method are rare. In our meta-analysis with a limited number of studies, BI showed advantages in operation time (P<0.01), hospital stay (P<0.01), and bile reflux (P<0.03) over BII [200,201]. There was no difference in complications (P=0.10). BI was more favorable than RY in terms of operation time (P<0.01), complications (P=0.01) and hospital stay (P<0.01) [200,202,203,204,205]. Other merits of BI include decreased iron deficiency anemia, preservation of the continuity of the alimentary tract, no risk of Petersen hernia, less small bowel adhesion and easier access to the duodenum and biliary tract in cases of biliary diseases [206,207,208,209] (Fig. 4).
Fig. 4. Forest plots comparing reconstruction methods. (A) Operation time. (B) Complications. (C) Hospital stay. (D) Bile reflux. (E) Esophageal reflux.
SD = standard deviation; IV = interval variable; CI = confidence interval.
RY showed advantages in preventing bile reflux and esophageal reflux under endoscopic findings [200,201,202,203,204,205,210]. However, to date, evidence regarding whether endoscopic bile reflux directly leads to superior QOL or the prevention of metachronous cancer is insufficient. In a retrospective series, RY with an increased length of limbs after gastrectomy showed favorable metabolic effects for gastric cancer patients with type II diabetes [208,211].
There was no difference in QOL (P=0.290–0.994) or nutritional aspects (weight loss, albumin) among the 3 reconstruction methods [200,212]. There is insufficient evidence to show differences in survival outcomes among reconstruction methods [213].
Taken together, the working group in the guideline could not recommend a specific reconstruction method as the best option for all cases due to the different advantages/disadvantages of each reconstruction method. We recommend that surgeons choose the best option according to the characteristics of the cancer and patients.
KQ 11: Can intraoperative evaluation of tumor margin, reresection or reoperation show improved outcome in margin positivity and survival outcome for gastric cancer patients who undergo gastrectomy?
Statement 11-1: Various efforts to achieve negative margins are recommended for better survival outcomes in EGC patients. Reresection or reoperation should be considered when patient condition is favorable and technically feasible (evidence: low, recommendation: strong for).
The impact of microscopically positive margin status was different according to the pathologic status of the cancer [214,215]. In our review, positive resection margins showed inferior survival outcomes compared to negative margins in pathologic T1 cancers ([68.6% vs. 97.4%, P<0.0001], [66.7% vs. 93.1%, P<0.04]) and T2 cancers ([21.5% vs. 55.2%, P<0.001], [8% vs. 64%, P<0.001]) [215,216,217].
There is a debate regarding whether securing an adequate length of margin may influence oncologic outcome in EGC [214,218,219,220]. However, most literature agrees that obtaining negative margin regardless of margin length shows better survival [219,221,222]. To obtain negative margin, single or combinations of various methods including intraoperative frozen section, perioperative gastroscopy, various preoperative clipping or dyeing, fluorescence imaging technique, ultrasonography, and simple X-ray, etc., were introduced [223,224,225,226,227,228,229,230,231].
In EGC, when pathologic results reveal tumor involvement of the resection margin, additional surgery to obtain R0 resection showed a survival benefit in several studies [221,222,232]. Therefore, development of this guideline made a consensus to recommend additional surgery when the patient condition is favorable and additional surgery is technically feasible.
There were also reports showing that R1 resection does not always lead to recurrence, partly due to lack of blood supply on the remnant transection line, discrepancy of true surgical margin from the use of surgical stapler or patients’ immunity [214,216]. Watchful observation with frequent follow-up might be cautiously considered when the extent of the involved margin is minimal, or the anticipated risk of reoperation is high. However, further investigations are required to clarify the indications for no additional surgery.
Statement 11-2: Efforts should be made to obtain negative margins in advanced or infiltrative gastric cancer surgery. If the final postoperative pathologic margin shows involvement of the margin, reoperation to achieve R0 should be chosen cautiously, considering the possibility of limited survival benefits and the risk of postoperative complications in advanced-stage cancer (evidence: low, recommendation: conditional for).
In previous reports, various macroscopic margin lengths (3–8 cm) were recommended to secure pathologic negative resection margins in advanced or infiltrative cancer [230,233,234]. Intraoperative frozen section showed improved accuracy compared to macroscopic margin prediction to secure R0 resection [227,230,235,236,237]. The aforementioned methods to achieve negative margins can also benefit advanced cancer cases to obtain secure margins and tumor localization.
Unlike early-stage gastric cancer, many studies showed that a positive microscopic margin had no prognostic impact when staging was ≥ T3 or ≥ N2 or ≥ IIIa (American Joint Committee on Cancer [AJCC] 7th) [215,218,219,221,238,239,240,241,242]. In these situations, achievement of a negative margin showed limited survival benefits.
Therefore, utilizing various methods including intraoperative frozen section is advisable to achieve R0 resection in advanced cancer. However, in advanced diseases (≥ pT3 or ≥ pN2 or ≥ Stage IIIa (AJCC 7th) with R1 resection, reoperation should be decided cautiously considering pathologic stage, patient status, risk of postoperative complications and risk of delayed systemic therapy.
KQ 12: Can PG with DTR show better outcome than TG in terms of short-term surgical outcomes, nutritional status, QOL, and survival rate for EGC in the upper third of the stomach?
Statement 12: PG with DTR as well as TG can be considered for EGC in the upper third of the stomach in terms of less vitamin B12 deficiency and similar survival and reflux symptoms compared to TG (evidence: low, recommendation: conditional for).
TG has been a standard treatment for upper gastric cancer. Gastric cancer in the upper third of the stomach-has limited node metastasis to the lower part of the stomach, which makes PG acceptable and oncologically safe [243,244]. However, reconstruction has been a hurdle for PG due to the high incidence of reflux esophagitis and anastomosis stricture in esophagogastrostomy.
DTR was recently reported for its feasibility under laparoscopic settings. In our systematic review of retrospective studies, there were significantly fewer patients with vitamin B12 deficiency in the PG-DTR group than in the TG group (RR, 0.30; 95% CI, 0.23 to 0.40; P<0.01) [245,246,247]. Weight loss after surgery did not differ between the groups (RR, −4.89; 95% CI, −11.75 to 1.97; P=0.16) [247,248]. There were no differences in reflux symptoms (RR, 1.28; 95% CI, 0.33 to 4.93) [246,249,250]. Complications were reported less frequently in the PG-DTR group (RR, 0.61; 95% CI, 0.45 to 0.83; P=0.002) (Fig. 5).
Fig. 5. Forest plots for comparison between proximal gastrectomy with double tract reconstruction vs. total gastrectomy in retrospective studies. (A) Vitamin B12 deficiency. (B) Weight loss. (C) Early complications. (D) Reflux symptom.
PG = proximal gastrectomy; DTR = double tract reconstruction; TG = total gastrectomy; IV = interval variable; CI = confidence interval.
The Korean Laparoendoscopic Gastrointestinal Surgery Study Group conducted a prospective RCT on LPG-DTR vs. LTG (KLASS-05) and recently reported early results. The cumulative amount of intramuscular vitamin B12 supplementation required for patients during 2 postoperative years was significantly lower in the PG-DTR group than in the TG group (0.6±2.0 mg vs. 3.4±4.1 mg, P<0.001). The proportion of patients who required vitamin B12 supplementation was also significantly lower in the PG-DTR group (14.7% vs. 58.0%, P<0.001). However, the hemoglobin change after surgery, which was the co-first outcome, was not significantly different between the groups (5.6%±7.4% vs. 6.9%±8.3%, P=0.349). The Visick score for reflux symptoms at 2 weeks postoperatively (P=0.793) and postoperative complications were not different between the groups (23.5% vs. 17.4%, P=0.373) [251].
In the KLASS-05, the 2-year OS rates and DFS rates of the PG-DTR and TG groups were 98.5% vs. 100%, (P=0.330) and 98.5% vs. 97.1%, respectively (P=0.540) [251]. Regarding long-term QOL, the PG-DTR group showed better scores on the physical functioning (P=0.029) and social functioning (P=0.031) scales (European Organization for Research and Treatment of Cancer QOL Questionnaire [EORTC QLQ-C30]).
Recently, other methods such as side overlap esophagogastrostomy, double flap technique (DFT) reconstruction are being investigated for better functional outcomes [252,253,254,255]. Some studies show that DFT has better outcomes than TG in terms of morbidity, postoperative hospital stay, reflux esophagitis and postoperative nutritional status [252]. However, laparoscopic PG-DFT requires a more complex intracorporeal suturing technique and longer duration of surgery [253]. Further investigation is required for higher level of evidence [255].
KQ 13: Can PPG show improved outcomes than DG in terms of nutritional status, QOL, complications, and survival outcomes for patients with middle third gastric cancer?
Statement 13: For EGC located ≥5 cm proximal from the pylorus, PPG as well as DG could be performed. PPG has the benefits of less gallstone formation and protein preservation; however, delayed gastric emptying should be considered when making decisions (evidence: moderate, recommendation: conditional for).
PPG preserves the pylorus and distal antrum to prevent the rapid transit of food into the duodenum and the reflux of duodenal contents. Consequently, the postoperative incidence of dumping syndrome and reflux gastritis has been expected to show benefits in nutrition and QOL compared to DG.
Recently, a prospective RCT on laparoscopic PPG vs. laparoscopic DG (KLASS-04) was conducted and the results were reported [256,257].
In KLASS-04, there were no differences in survival outcomes or complications between the PPG group and the DG group. There was no difference in the incidence of dumping syndrome one year after surgery (13.2% vs. 15.8%, P=0.62). Reflux esophagitis (17.8% vs. 6.3%, P<0.01) and delayed gastric emptying (16.3% vs. 4.0%, P<0.01) were more frequent in the PPG group than in the DG group 3 years after surgery. However, bile reflux (13.2% vs. 24.4%, P=0.02) and the incidence of gallstone formation (2.3% vs. 8.7% P=0.03) were lower in the PPG group than in the DG group.
Although there was no difference in body weight change after surgery, the total protein level was preserved after PPG compared to DG (P<0.01). Regarding QOL after surgery, there was no difference between groups in terms of the EORTC QLQ-C30 and EORTC-QLQ-Gastric Cancer Module (STO22) questionnaires.
In our meta-analysis, most studies on PPG were retrospective. There were no differences in survival outcomes or postoperative complications [257,258,259,260,261,262]. The PPG group showed lower incidence of postoperative dumping syndrome and reflux [260,262,263]. Some studies reported reduced development of gallstones after PPG, probably due to preservation of the hepatic branch of the vagus nerve [258,260]. However, PPG was related to more frequent delayed gastric emptying than DG [258,260,261,262,263].
Regarding nutritional status, the decreases in serum protein and albumin from postoperative months 1 to 6 and abdominal fat area at postoperative year 1 were lower in the PPG group than in the DG group, and the PPG group showed more improvement in hemoglobin than the DG group [258,261,262,264]
PPG tended to present better improvement of QOL and fewer symptoms than DG with BI at 2 years after surgery [264]. The PPG group had a better physical functioning score (86.7 vs. 90.0, P=0.032) but also greater pain and reflux than the DG group (median score 8.3 vs. 16.7 in pain, 11.1 vs. 11.1 in reflux, P=0.034 and 0.001, respectively) at least 2 years after surgery [262].
In summary, PPG showed benefits in gallstone formation, bile reflux and preservation of serum total protein, while survival, postoperative complications and QOL were similar in comparison to DG in KLASS-04. Observational studies reported additional possible benefits in nutritional and functional aspects of PPG. For EGC located ≥5 cm proximal from the pylorus, PPG could be performed in this regard; however, delayed gastric emptying should be considered when making decisions.
KQ 14: Can splenectomy for prophylactic LN dissection of the splenic hilum provide better survival and complication outcomes than radical TG without splenectomy in advanced gastric cancer?
Statement 14: Prophylactic splenectomy for splenic hilar LN dissection is not recommended in curative resection for advanced gastric cancer in the proximal stomach without greater curvature invasion (evidence: high, recommendation: strong against).
The standard surgical procedure for proximal-third gastric carcinoma TG with proper LN dissection. Therapeutic splenectomy may be necessary if the tumor directly invades the spleen or if LN metastasis around the splenic hilum is suspected. However, there is debate regarding whether splenic resection with LN dissection of the splenic hilum for cancer should be performed in the absence of direct invasion of the spleen, splenic hilum, or greater curvature of the stomach.
Three prospective RCTs evaluated the survival advantage of prophylactic splenectomy in proximal-third gastric carcinoma [252,265,266]. Our meta-analysis showed no difference in survival (HR, 0.91; 95% CI, 0.68 to 1.21; P=0.51) but significantly fewer postoperative complications in the nonsplenectomy group (HR, 0.42; 95% CI, 0.31 to 0.59, P<0.01) (Fig. 6).
Fig. 6. Forest plot for a comparison between no splenectomy (experimental) vs. splenectomy (control). (A) Survival. (B) Complications.
CI = confidence interval.
Studies in our meta-analysis did not include advanced cancer with gross involvement to the greater curvature or to gastrosplenic ligament, where the metastasis rate of LN#10 is relatively high and splenic hilar dissection with splenectomy is required to accomplish standard treatment [267,268,269].
In any circumstance, splenectomy increases postoperative complications and mortality rates. To overcome this, an operative technique for LN#10 dissection around the splenic hilum without splenectomy has been reported; however, its oncologic outcome is still under investigation [270,271,272].
KQ 15: Can PG without LN dissection at the distal stomach be recommended to treat advanced adenocarcinoma invading the GEJ compared to TG with standard LN dissection?
Statement 15: PG may be performed in advanced gastric cancer with adenocarcinoma histology located in the GEJ (Siewert II/III) without serosal invasion, due to low rate of LN metastasis to the distal part of the stomach (evidence: low, recommendation: conditional for).
TG is the standard treatment for AGC in the upper part of the stomach [10,111,234]. Some studies have raised suspicion that removing the entire stomach along with the perigastric tissues and LNs may be unnecessary in selected cases [243,273,274].
In our meta-analysis, 5 retrospective studies that investigated the LN mapping of proximal gastric cancer after TG were included [273,274,275,276,277]. In the pooled data, the distal LN station #4d, #5 and #6 (distal LN) metastasis rates were analyzed. The metastasis rates in pT2 cancer were very low: #4d (0/359), #5 (1/425) and #6 (0/359) (distal LN). The risk ratio of metastasis in the distal LN for pT3 was 1.82 (CI, 0.77 to 4.29) compared to pT2 (P=0.17), and the risk ratio for pT4 was 9.89 (CI, 4.66 to 20.95) compared to pT2 (P<0.01) (Fig. 7).
Fig. 7. Forest plot, LN metastasis rates of distal stomach according to the depth of tumor. (A) LN metastasis rate comparison T3 vs. T2 (P=0.17). (B) LN metastasis rate comparison T4 vs. T2 (P<0.01).
LN = lymph node; CI = confidence interval.
In one retrospective study in Korea, 873 patients were reviewed [243]. In their multivariable analysis, a GEJ to tumor epicenter distance longer than 30 mm, tumor size >70 mm, macroscopic Bormann type IV tumor or serosa invasion were risk factors for LN metastasis to the distal stomach. In patients without any risk factors from above, the LN metastasis rates at stations 4d, 5, and 6 were 0.0%, 0.4%, and 0.4%, respectively, and the therapeutic value index (TVI) of LN #4d, #5, and #6 were low; 0, 0.4, and 0.4), respectively (TVI is calculated by incidence of LN metastasis and 5-year survival after removing corresponding LN and can be used for evaluating necessity of dissection of specific LN stations) [268].
From these results, PG without dissection of LN stations #4d, #5, and #6 could be considered in selected cases of advanced gastric cancer with adenocarcinoma histology located in the GEJ (Siewert II/III) without serosal invasion. However, more data are required to determine the detailed indications for PG and to evaluate the clinical outcomes of PG.
KQ 16: Can additional lower mediastinal LN dissection improve oncologic outcome for adenocarcinoma invading GEJ?
Statement 16-1: Lower mediastinal LN dissection could be performed to remove possible metastatic LNs for advanced cancer invading the GEJ (evidence: low, recommendation: conditional for).
The definition and extent of surgery around the GEJ have not been solidly established. The most frequently used classification is the Siewert classification, which defines GEJ carcinoma as a tumor with an epicenter within 5 cm proximal and distal of the anatomical cardia and categorizes it into 3 types: type 1 (lower esophageal cancer), type 2 (true GEJ cancer), and type 3 (subcardial cancer) [278]. In Japan, GEJ carcinoma is defined as a tumor according to the Japanese classification system, regardless of histological type, when its epicenter is located within 2 cm proximal or distal to the GEJ [18].
It is generally acknowledged that Siewert type I and type III carcinomas are usually treated as esophageal and gastric tumors, respectively [279,280]. Siewert type II adenocarcinoma, located 1 cm above to 2 cm below the GEJ, represents adenocarcinoma that arises from the epithelium of the cardia or short segments of intestinal metaplasia mostly in Western countries. There has been considerable controversy about whether Siewert type II carcinoma is esophageal or gastric cancer and about the extent of LN dissection.
In a retrospective analysis conducted in Korea, 672 patients who underwent radical TG with lymphadenectomy without lower mediastinal LN dissection for GEJ carcinoma type II, type III, or upper third of the stomach were reviewed [281]. They suggested that lower mediastinal LN dissection will not be essential for early-stage cancer based on excellent survival regardless of the location (93.2% vs. 96.7% vs. 98.7% for Siewert type II, III, and upper-third gastric cancer, P=0.158). However, for advanced cancer, the survival was worse in Siewert type II than that in Siewert type III cancer (47.9% vs. 75.4% vs. 71.8% in Siewert type II, III, and upper-third gastric cancer, P<0.001), which result implies the necessity of mediastinal LN dissection.
On the other hand, another retrospective analysis in Korea that reviewed 125 type II and 338 type III GEJ cancer patients demonstrated that there was no increase of recurrence in the mediastinal LNs without complete mediastinal LN dissection, regardless of the type. From this result, they suggested that TH approach without complete mediastinal LN dissection can be acceptable, at least for frequent types of GEJ cancer in Korea [282].
A prospective nationwide multicenter study in Japan reviewed the frequency of LN metastasis of GEJ tumors with cT2-T4 stages and recommended lower mediastinal LN dissection (especially station 110) if the length of esophageal invasion was more than 2 cm [26].
Taken together, lower mediastinal LN dissection seems to be not essential for early GEJ cancers. For advanced cancer, lower mediastinal LN dissection may be needed to sufficiently remove possible metastatic LNs in case esophageal involvement is more than 2 cm; however, further data in terms of local recurrence and survival gain remain to be needed.
Statement 16-2: The TH approach rather than the TT approach is recommended to acquire negative resection margin and perform lower mediastinal LN dissection in resectable adenocarcinoma invading the GEJ (evidence: moderate, recommendation: strong for).
Two RCTs on optimal surgical approach for GEJ adenocarcinoma (Siewert type II, III) compared the surgical and oncological outcomes of TH and TT approaches (one left thoracoabdominal approach and one right thoracotomy). Our meta-analysis of RCTs demonstrated comparable in-hospital mortality (P=0.10) and anastomosis leakage (P=0.58) between the TT and TH approaches, but a higher incidence of pulmonary complication in the TT approach (P<0.0001) (Fig. 8). Although the Japanese RCT (JCOG9502) comparing the left thoracoabdominal and TH approaches was to be stopped after the interim analysis, the 5-year OS of TH approach were not inferior to that of the TT approach (HR, 0.90; CI, 0.73 to 1.10; P=0.31) in 2 RCTs [283,284].
Fig. 8. Forest plots for comparisons between the TH abdominal approach vs. TT approach. TT approaches in the observational studies included. In-hospital mortality: (A) RCTs; (B) Observational studies. Pulmonary complications: (C) RCTs; (D) Observational studies. Five-year survival: (E) RCTs; (F) Observational studies.
TH = transhiatal; TT = transthoracic; CI = confidence interval; RCT = randomized controlled trial.
In our meta-analysis with observational studies including right and left thoracotomy and right thoracoscopic approaches, the TT approach was associated with a higher incidence of pulmonary complications (P=0.0002), a higher in-hospital mortality rate (P=0.02) and similar anastomosis leakage (P=0.57) when compared to the TH approach. The TH approach was not inferior to the TT approach in 5-year OS (HR, 0.80; CI, 0.59 to 1.11; P=0.18) [26,285,286,287,288,289,290,291].
Regarding the high surgical complications of the TT approach and no difference in 5-year OS, the TH approach is recommended rather than the TT approach for resectable adenocarcinoma invading the GEJ.
KQ 17: Can D1+ dissection show comparable survival outcome for EGC (cT1N0) patients compared to D2 dissection?
Statement 17: D1+ dissection can be performed during surgery for EGC (cT1N0) patients in terms of survival (evidence: low, recommendation: Strong for).
D2 dissection was regarded as the standard LN dissection based on the long-term survival data of the Dutch trial [292]. However, application of standard D2 dissection has been questioned for EGC, especially in laparoscopic surgery. There have been no studies comparing D2 dissection and less dissection in EGC patients, but D1+ dissection has been widely accepted by surgeons in Korea and Japan considering the spatial information about LN metastasis, TVI calculated by the frequency of LN metastasis and 5-year survival rate after removing each LN station, and the Maruyama index, which evaluates the adequacy of LN dissection [268,293,294].
An Italian study suggested D2 dissection considering significant LN metastasis [295]. Others reported 10-year survival rates of 95% and 87.5% after standard D2 and D1 dissection, respectively, in EGC patients without statistical significance (P=0.80) [296]. In a report from Japan, the 5- and 10-year survival rates were 97% and 91% in patients after standard D2 dissection and 98% and 91% after modified D2 (D1+) dissection. There were no cases with metastasis to second-tier LNs in patients with cT1N0 or cT1N1 disease [297].
Although evidence for comparing D1+ and D2 dissection is insufficient, we referred to the excellent survival results of the RCTs for EGC that compared laparoscopic and open gastrectomy, in which less than D2 dissection was performed, and strongly suggested that D1+ dissection can be performed for EGC [298,299,300].
KQ 18: Can SNNS be considered a treatment option compared to conventional LG regarding survival, nutritional outcomes and QOL?
Statement 18: SNNS implemented by well-designed protocols and follow-up plans could be considered as a treatment option for cT1N0 and ≤3 cm gastric cancers in terms of better nutritional outcomes and QOL. Treatment decisions should be made after sufficient discussion with the patient regarding the possibility of metachronous cancer and rescue surgery (evidence: moderate, recommendation: conditional for).
To date, numerous feasibility studies for the sentinel node concept have shown a high detection rate and acceptable sensitivity of sentinel node mapping [301,302]. In previous meta-analyses, pooled detection rate and sensitivity rate were 93.7%–97% and 80.8%–89%, respectively. Clinical T1 tumors, dual tracers, submucosal injection, and IHC examinations were associated with higher sensitivity [301,302].
The oncologic safety and clinical benefits of SNNS have been evaluated only in few studies so far [303,304,305,306,307,308]. In these studies, SNNS was performed on small sized early lesion less than 3 cm in diameter. Two case-control studies and one RCT compared 5-year OS rates, and there was no significant difference between the SNNS and conventional LG groups [304,307,308]. In the RCT (SENORITA trial), 3-year DFS, the primary end point, did not show non-inferiority in SNNS compared to LG due to a higher incidence of metachronous cancer (91.8% vs. 95.5%, SNNS vs. LG) [308].
However, 5-year DFS, OS, and disease-specific survival, which were secondary end points, were not different after rescue surgery in cases of recurrence/metachronous gastric cancer; 88.9% vs. 80.7%, 97.3% vs. 88.3%, and 99.2% vs. 99.3% in SNNS vs LG, respectively (P=0.056, 0.74, and 0.959) [308].
Regarding nutritional outcomes, SNNS had less body weight loss and higher hemoglobin level than LG [303,305,308]. QOL was assessed using various tools in each study, and SNNS had better QOL in some subscales [303,305,306,308] (Fig. 9).
Fig. 9. Forest plots for comparison between the sentinel node navigation surgery vs. conventional surgery in observational studies. (A) Overall survival. (B) Body weight: percentages compared to preoperative body weight.
SE = standard error; SNNS = sentinel node navigation surgery; SD = standard deviation; IV = interval variable; CI = confidence interval.
There are still controversies over technical issue for sentinel node such as type of tracer, detection method, and practical pathological examination method. It should be noted that the SENORITA trial had a fastidious protocol including dual tracer composed of radioactive isotope (Technetium-99 m) and indocyanine green, sentinel basin dissection instead of pick-up biopsy, intraoperative frozen examination for sentinel LNs with 2 mm-interval cutting and 4-direction resection margins, and cytokeratin IHC for permanent histological evaluation. Therefore, SNNS should be performed under a strict protocol including indication criteria, detection method, and follow-up plan, and treatment decision should be made after sufficient discussion with the patient regarding the possibility of metachronous cancer and rescue surgery. Under these conditions, SNNS could be a treatment option for EGC in aspects of better nutritional outcomes and QOL.
KQ 19: Can LDG show comparable surgical and survival outcomes compared to ODG in the treatment of early or locally advanced gastric cancer?
Statement 19-1: LDG is recommended for c-Stage I gastric cancer in terms of better short-term surgical outcomes and comparable long-term survival compared to ODG (evidence: high, recommendation: strong for).
Since the first clinical trial was reported in the early 2000s several pivotal trials comparing LDG and ODG for early or locally advanced gastric cancer have been published thus far [309].
Three prospective RCTs (KLASS-01, COACT 0301, JCOG0912) were conducted to evaluate the noninferiority of LDG for EGC [147,310,311]. Our meta-analysis demonstrated longer operation times of laparoscopic surgery (P<0.01) but better surgical outcomes, such as less operative blood loss (P<0.001), fewer postoperative complications (P<0.001), or reduced hospital stay (P<0.001), compared with ODG (Fig. 10). The long-term survival in LDG was not inferior to that of patients in the ODG group in all 3 trials (HR, 0.90; CI, 0.69 to 1.16; P=0.42) [298,299,310].
Fig. 10. Forest plots for comparisons between laparoscopic and open (conventional) distal gastrectomy in c-Stage I gastric cancer. (A)Overall survival. (B)Complications. (C) Intraoperative blood loss.
SE = standard error; SD = standard deviation; IV = interval variable; CI = confidence interval.
Statement 19-2: LDG as well as ODG can be recommended for locally advanced gastric cancers for comparable survival outcomes (evidence: high, recommendation: strong for).
For AGC, 2 RCTs (KLASS-02, CLASS-01) were included in the meta-analysis [312,313]. In both trials, preoperatively diagnosed AGC cases of cT2-T4a were enrolled. The KLASS-02 trials included cases with no LN metastasis or limited metastasis to the left gastric artery or perigastric LNs, while the CLASS-01 trial recruited all cN0-3 cases.
The KLASS-02 study reported a lower complication rate in the laparoscopic group than in the open group (6.5% vs. 11.0%, P=0.01). The 5-year OS of LDG (88.9%) was not inferior to that of ODG (88.7%, P=0.30) [314,315] (Fig. 11).
Fig. 11. Forest plots for comparisons between laparoscopic and open (conventional) distal gastrectomy in cT2-4a gastric cancer. (A) Overall survival. (B) Complications. (C) Intraoperative blood loss. (D) Operation time. (E) Hospital stay.
SE = standard error; SD = standard deviation; IV = interval variable; CI = confidence interval.
The CLASS-01 study reported no significant difference in postoperative complications between LDG and ODG (15.2% vs. 12.9%, P=0.285) and noninferior 5-year OS (LDG, 72.6% vs. ODG, 76.3%, P=0.19) [316].
In our meta-analysis, LDG showed shorter hospital time (P=0.02), less intraoperative blood loss (P<0.01), and similar postoperative complications (P=0.27) but a longer operation time (P<0.01). For long-term outcomes, LDG was not inferior to ODG in the 5-year survival rate (HR, 1.12; CI, 0.92 to 1.36; P=0.26).
LDG can be considered an optimal treatment option for AGC as well as ODG. However, since far advanced cases, such as cT4b cancer where multivisceral resection may be needed, were excluded from the studies, the application of LDG should not be interpreted beyond indications [317].
KQ 20: Can RG show better surgical, survival and economical outcome than LG for treating gastric cancer?
Statement 20: RG can be considered a treatment option for gastric cancer in terms of noninferior survival and fewer complications than LG. However, disadvantages such as additional cost and longer operation time should also be considered for patient shared decision-making (evidence: moderate, recommendation: conditional for).
RG has some technical advantages compared to LG, including surgeon-controlled vision, tremor filter and ergonomic wrist motion instruments [318,319]. Two RCTs and 8 retrospective studies were included in our systematic review to investigate RG vs. LG.
In our meta-analysis of 2 RCTs, RG was associated with fewer postoperative complications than LG (RR, 0.49; 95% CI, 0.31 to 0.78; P=0.003). The incidence of pancreatic fistula was not different in the RCT (P=0.20) and non-RCT (P=0.58) analyses. The reoperation rate (P=0.25) and hospital stay (P=0.11) showed no difference. RG was associated with a longer operation time (mean difference [MD], 47.04 minutes; 95% CI, 30.67 to 63.41; P<0.01) compared to LG (Fig. 12).
Fig. 12. Forest plots for comparisons between daVinci™ robot gastrectomy vs. laparoscopic gastrectomy. (A) Complications (RCTs). (B) Pancreatic fistula (RCTs and observational studies). (C) Overall survival (observational studies). (D) Operation time (observational studies).
SE = standard error; SD = standard deviation; IV = interval variable; CI = confidence interval; RCT = randomized controlled trial.
In retrospective studies, there were no differences in either the 5-year OS (HR, 0.84; 95% CI, 0.57 to 1.24; P=0.38) or the 5-year RFS rate (HR, 0.98; 95% CI, 0.71 to 1.34, P=0.88) between the groups.
In selected reports, the additional total hospital cost for RG ranged from $3,000 to $5,000 compared to LG [319,320].
Based on the current evidence, the guideline task force team decided the recommendation as “conditional for” because of noninferior perioperative and survival outcomes and fewer postoperative complications of RG compared to LG. Further investigation is required to identify the potential benefit of RG to justify longer operation time and higher cost, which should be discussed with the patient in the aspect of decision-making.
KQ 21: In patients with advanced gastric cancer, can PO show comparable survival, recurrence rates and complication rates compared to total omentectomy (TO)?
Statement 21: PO could be considered for advanced gastric cancer patients (evidence: low, recommendation: conditional for).
TO is regarded as a mandatory treatment for AGC without high-level evidence [107]. However, TO during LG is difficult and time-consuming [321]. PO is now widely adopted as a safe procedure for EGC based on excellent survival outcomes in randomized clinical trials in which PO was performed in the majority of cases; thus, we focused on its applicability to AGC in our meta-analysis [147,300].
In our meta-analysis, 5 retrospective studies were included to compare PO vs. TO [322,323,324,325]. Among all retrospective studies, 5 studies for meta-analysis regarding survival results were selected when propensity matching was performed to minimize selection bias. PO was not inferior to TO in relapse-free survival (RFS) (HR, 0.89; CI, 0.74 to 1.07; P=0.20) or OS (HR, 0.82; CI 0.67 to 1.00; P=0.06]. There was no difference in overall complications (P=0.10) or serious complications (P=0.92) between the procedures (Fig. 13).
Fig. 13. Forest plots for comparisons between PO (experimental) vs. TO (control) in advanced gastric cancer in observational studies. (A) Overall survival (propensity score matched). (B) Relapse-free survival (propensity score matched). (C) Complications.
PO = partial omentectomy; TO = total omentectomy; SE = standard error; IV = interval variable; CI = confidence interval.
Seven previous meta-analyses studied the oncologic feasibility of PO [326,327,328,329,330]. All the meta-analyses showed that PO was not inferior to TO regarding OS and relapse-free survival. Moreover, PO required less operation time and involved lower blood loss.
Although a significant proportion of patients with serosal invasion (T4a) were included in the studies, the working group of guidelines expressed concern about the possibly insufficient extent of resection in locally far advanced cancer, including cT4a cases and recommended cautious consideration in such cases while waiting for more confirmative results.
KQ 22: Can UDCA treatment reduce gallstone formation in patients after gastrectomy for gastric cancer treatment?
Statement 22: Administration of UDCA for one year can be recommended to reduce gallstone formation after gastrectomy (evidence: moderate, recommendation: conditional for).
Gallstone formation is known as one of the long-term complications following gastrectomy [207]. Denervation of the vagus nerve, obesity, rapid weight loss and TG precipitate the incidence and severity of gallstone [206,331,332]. Since prophylactic UDCA showed positive effect on reducing gallstones after bariatric surgery, UDCA can be as effective in gastric cancer patients.
One RCT studied the use of prophylactic UDCA after gastrectomy in patients with gastric cancer [208]. Patients were randomized into 3 groups: placebo, 300 mg or 600 mg group, and UDCA was administered as allocated for one year. Compared to placebo, 300 mg (odds ratio [OR], 0.27; 95% CI, 0.12 to 0.62; P<0.002) and 600 mg (OR, 0.20; 95% CI, 0.08 to 0.50; P<0.001) UDCA showed decreased gallstone formation. Daily 300 mg seems to be sufficient because the protective effect did not differ between 300 mg and 600 mg.
Considering that there are some risk factors for gallstone formation and that the PPG or DG with preservation of the hepatic branch of vagus nerve rarely experiences gallstone formation, further studies may answer to question of who may especially benefit from UDCA prophylaxis, as well as long-term effect over 1 year postoperatively.
SYSTEMIC THERAPY
Adjuvant chemotherapy
KQ 23: Could adjuvant chemotherapy improve survival compared to surgery only in patients with pathological stage II or III disease who undergo curative gastrectomy?
Statement 23: Adjuvant chemotherapy (S-1 or XELOX) is recommended in patients with pathological stage II or III gastric cancer (evidence: high, recommendation: strong for).
Surgical resection with D2 LN dissection is the standard treatment for gastric cancer. However, the prognosis is usually poor in AGC [333,334]. Two randomized phase III trials conducted in Asia showed significant survival benefits for adjuvant chemotherapy over observation after curative gastrectomy with D2 LN dissection in patients with gastric cancer [335,336]. In the adjuvant chemotherapy trial of S-1 for gastric cancer (ACTS-GC) in Japan, 1,059 patients with stage II (excluding T1) or III gastric cancer (by Japanese classification, 2nd English edition) who underwent D2 gastrectomy received adjuvant S-1 for 1 year or were observed after surgery [336,337]. The 3-year DFS rates were 72.2% in the S-1 group and 59.6% in the surgery-only group (HR, 0.62; 95% CI, 0.50 to 0.77; P<0.001), and the 3-year OS rates were 80.1% and 70.1%, respectively (HR, 0.68; 95% CI, 0.52 to 0.87; P=0.003). In the capecitabine and oxaliplatin study in stomach cancer (CLASSIC) conducted in South Korea, China, and Taiwan, 1,035 patients with stage II–IIIB gastric cancer (by AJCC 6th edition [338]) who underwent D2 gastrectomy received either XELOX for 6 months or were observed [335]. The 3-year DFS rates were 74% in the chemotherapy and surgery groups and 59% in the surgery-only group (HR, 0.56; 95% CI, 0.44 to 0.72; P<0.001). The 5-year follow-up data in these 2 studies confirmed these findings [339,340]. In our present meta-analysis, adjuvant chemotherapy significantly improved OS and DFS compared to surgery alone (OS: HR, 0.66, 95% CI, 0.56 to 0.78; DFS: HR, 0.62, 95% CI, 0.54 to 0.72; P<0.001) (Fig. 14). Based on these results, both chemotherapy regimens (S-1 or XELOX) are currently accepted as standard treatments in patients with pathological stage II or III gastric cancer after curative gastrectomy. It should be noted that there is currently no evidence to support the use of adjuvant chemotherapy for patients who fall into the category of stage IB by the AJCC 6th edition but stage IIA by the AJCC 7th and 8th editions (pT1N2M0 and pT3N0M0) [341].
Fig. 14. Forest plots for comparisons between adjuvant chemotherapy vs. surgery only and doublet vs. S1 monotherapy. (A) Overall survival for adjuvant chemotherapy (experimental) vs. surgery only (control). (B) Disease-free survival for adjuvant chemotherapy (experimental) vs. surgery only (control). (C) Disease-free survival for oral fluoropyrimidine-based doublet (experimental) vs. S1 monotherapy (control).
SE = standard error; IV = interval variable; CI = confidence interval.
Although the survival benefit of adjuvant S-1 for 1 year in gastric cancer patients was demonstrated, the optimal duration of adjuvant S-1 for gastric cancer was unclear. In the randomized phase III noninferiority trial (OPAS-1) in Japan, 590 patients with stage II (excluding T1N2–3 and T3N0) gastric cancer (by Japanese classification, 3rd English edition) who underwent gastrectomy with D2 LN dissection (with D1 plus dissection being allowed for clinical stage IA) received 8 courses (12 months) or 4 courses (6 months) of S-1 [18,342]. At the first planned interim analysis, this study was terminated early because the HR for DFS of the 4-course group compared with the 8-course group exceeded 1.37 and met the prespecified criteria for early termination. The 3-year DFS rate was 93.1% for the 8-course group and 89.8% for the 4-course group (HR, 1.84; 95% CI, 0.93 to 3.6; noninferiority margin for HR, 1.37), and the 3-year OS rates were 96.1% and 92.6%, respectively (HR, 3.37; 95% CI, 1.23 to 9.19). Therefore, S-1 for 1 year remains the standard adjuvant treatment for pathological stage II gastric cancer.
Despite the benefit of adjuvant S-1 for gastric cancer, as shown through the ACTS-GC trial, there was a question about a lack of efficacy in advanced stages [336,340]. In the randomized phase III trial (JACCRO GC-07), adjuvant chemotherapy of the S-1 plus docetaxel group was associated with survival benefits compared to the S-1 group in patients with stage III gastric cancer (by Japanese classification, 3rd English edition) who underwent D2 gastrectomy [18,343]. The 3-year DFS rates were 67.7% in the S-1 plus docetaxel group and 57.4% in the S-1 group (HR, 0.72; 95% CI, 0.59 to 0.87; P<0.001), and the 3-year OS rates were 77.7% and 71.2%, respectively (HR, 0.74; 95% CI, 0.60 to 0.92; P=0.008). The randomized phase III trial (ARTIST-2) for stage II or III gastric cancer with positive LNs (by AJCC 7th edition [344]) also showed the superiority of S-1 plus oxaliplatin (SOX; 74.3%) to S-1 monotherapy (64.8%) for the 3-year DFS rate (HR, 0.69; 95% CI, 0.41 to 0.99; P=0.042) [345]. When comparing the SOX and S-1 arms, the 3-year DFS rates were 74.3% and 64.8%, respectively (HR, 0.69; 95% CI, 0.41 to 0.99; P=0.042) [345]. In the present meta-analysis including JACCRO GC-07 and ARTIST-2, adjuvant oral pyrimidine-based doublet regimens improved DFS compared to S-1 monotherapy (HR, 0.71; 95% CI, 0.59 to 0.85, P=0.0001). Furthermore, the subgroup analysis of the CLASSIC trial [335] showed that the efficacy of XELOX was maintained even in a more advanced stage, which was not observed in the ACTS-GC trial. According to these results, oral pyrimidine-based doublet regimens can be a more favorable treatment option than S-1 alone for pathological stage II with positive LN or stage III gastric cancer.
Palliative systemic therapy (Flowchart 6)
Flowchart 6. Treatment guideline for palliative systemic therapy.
HER2 = human epidermal growth factor receptor 2; PD-L1 = programmed cell death-ligand 1; CPS = combined positive score; FOLFOX = 5-fluorouracil, leucovorin, and oxaliplatin; XELOX = capecitabine and oxaliplatin; FP = 5-fluorouracil and cisplatin; XP = capecitabine plus cisplatin; MSI-H = microsatellite instability-high; dMMR = mismatch repair deficient.
KQ 24: Could palliative first-line systemic therapy improve survival in patients with locally advanced unresectable or metastatic gastric cancer?
Statement 24-1: Palliative first-line platinum/fluoropyrimidine-based chemotherapy is recommended in patients with locally advanced unresectable or metastatic gastric cancer (evidence: moderate, recommendation: strong for).
Chemotherapy vs. best supportive care
Several cytotoxic chemotherapeutic agents, including fluoropyrimidine, platinum, taxane, anthracycline, and irinotecan, have been shown to be active against AGC. In our present meta-analysis, chemotherapy significantly improved OS compared to best supportive care (HR, 0.39; 95% CI, 0.28 to 0.52, P<0.001) (Fig. 15). In a Cochrane meta-analysis, the efficacy of combination chemotherapy showed a statistically significant survival benefit over single-agent chemotherapy (HR, 0.82; 95% CI, 0.75 to 0.9; median survival, 8.3 vs. 6.7 months) [346].
Fig. 15. Forest plots for comparisons between palliative first-line chemotherapy (experimental) vs. best supportive care (control). (A) Overall survival. (B) Progression-free survival.
SE = standard error; IV = interval variable; CI = confidence interval.
Fluoropyrimidines: oral vs. intravenous
With oral fluoropyrimidines, patients can avoid complications and inconveniences associated with venous access for infusional 5-FU. Randomized phase III studies have demonstrated that the oral fluoropyrimidines capecitabine [347,348,349] and S-1 [350,351] are noninferior to infusional 5-FU. Therefore, oral fluoropyrimidines (capecitabine or S-1) are safe and have been widely used as an alternative to infusional 5-FU for combinations with platinum compounds in patients with AGC. Recently, TAS-118 was developed as a new oral drug containing S-1 and leucovorin. The SOLAR Phase III trial demonstrated clinically significant improvement in OS with manageable toxicity when TAS-118 plus oxaliplatin was administered compared to S-1 plus cisplatin in Asian patients with AGC (median OS, 16 vs. 15.1 months; HR, 0.83; 95% CI, 0.69 to 0.99; P=0.039) [352].
Platinum (cisplatin vs. oxaliplatin)
Regarding avoidance of cisplatin-associated toxicity, several studies [347,353,354,355] have shown noninferior or better efficacy and less toxicity with oxaliplatin than with cisplatin in patients with AGC. Therefore, oxaliplatin was preferred over cisplatin with a favorable safety profile.
Triplet vs. doublet combination
The triplet regimens have been investigated, and it remains unclear whether triplet or doublet regimens are better. The phase III V325 trial showed that docetaxel, cisplatin, and FU improved survival outcomes compared with cisplatin and FU (median OS, 9.2 vs. 8.6 months) but also markedly increased severe toxicities, including hematological and gastrointestinal toxicity [356]. The JCOG1013 phase III trial showed that modified docetaxel, S-1, and cisplatin did not improve OS but increased toxicity, such as neutropenia and leukopenia, compared to S-1 and cisplatin in Japanese patients with AGC [357]. Triplet combinations can be considered in selected patients.
Statement 24-2: Palliative first-line trastuzumab combined with capecitabine or FU plus cisplatin is recommended in patients with HER2 IHC 3+ or IHC 2+ and ISH-positive advanced gastric cancer (evidence: high, recommendation: strong for).
The Trastuzumab for Gastric Cancer (ToGA) phase III trial demonstrated the efficacy of trastuzumab, which is a monoclonal antibody against HER2, plus fluoropyrimidine/cisplatin as the first-line treatment in patients with HER2-positive AGC [112]. The OS was improved with the addition of trastuzumab to capecitabine or FU/cisplatin (median, 13.8 vs. 11.1 months; HR, 0.74; 95% CI, 0.60 to 0.91; P<0.01). The survival benefit from trastuzumab was more pronounced in patients with IHC 3+ or IHC 2+ and FISH+ (median OS, 16 vs. 11.8 months; HR, 0.65; 95% CI, 0.51 to 0.83; P<0.01).
In terms of dual blockade of HER2, the JACOB phase III trial evaluated the efficacy of pertuzumab (which is a monoclonal antibody interfering with HER2 heterodimerization with other EGFR family members), plus trastuzumab and chemotherapy as first-line therapy compared to trastuzumab and chemotherapy [358]. Although PFS increased with the addition of pertuzumab to trastuzumab plus fluoropyrimidine/cisplatin (median, 8.5 vs. 7.2 months; HR, 0.73; 95% CI, 0.62 to 0.85; P<0.01), there was no statistically significant improvement in OS for the primary endpoint in patients with HER2-positive AGC (median, 17.5 vs. 14.2; HR, 0.84; 95% CI, 0.71 to 1.00, P=0.057). Lapatinib is a small-molecule tyrosine kinase inhibitor of EGFR and HER2. In the LOGiC phase III trial, lapatinib plus capecitabine and oxaliplatin as first-line therapy did not significantly improve OS (median, 12.2 vs. 10.5 months; HR, 0.91; 95% CI, 0.73 to 1.12; P=0.349) compared to capecitabine and oxaliplatin in HER2-amplified AGC. Therefore, trastuzumab plus capecitabine or FU/cisplatin is recommended in patients with HER2-positive AGC [359].
Several targets, including EGFR [360,361], vascular endothelial growth factor/receptor-2 (VEGF/R2) [362,363], hepatocyte growth factor receptor/MET [364,365,366], and matrix metalloproteinase [367], have been evaluated as first-line treatments for AGC. However, none of these targeting agents demonstrated significant OS benefits in phase III trials.
Statement 24-3: Palliative first-line nivolumab combined with capecitabine or FU plus oxaliplatin (XELOX or FOLFOX) is recommended in patients with PD-L1 CPS ≥5 and HER2-negative advanced gastric cancer (evidence: high, recommendation: strong for).
Nivolumab is a PD-1 inhibitor and the first successful ICI used in combination with platinum/fluoropyrimidine chemotherapy as first-line treatment in AGC. The CheckMate 649 global phase III trial demonstrated significant improvement in OS with the addition of nivolumab to capecitabine or FU/oxaliplatin in patients with PD-L1 CPS ≥5 and HER2-negative AGC (median OS, 14.1 vs. 11.1 months; HR, 0.71; 95% CI, 0.59 to 0.86; P<0.01) [125]. PD-L1 IHC was performed using a Dako PD-L1 IHC 28-8 pharmDx assay (Dako, Santa Clara, CA, USA). In the ATTRACTION-4 phase III trial, nivolumab plus capecitabine or S-1/oxaliplatin as the first-line treatment significantly improved PFS (10.5 vs. 8.3 months; HR, 0.68; 95% CI, 0.51 to 0.90; P<0.01) but not OS in Asian patients with HER2-negative AGC regardless of PD-L1 status [368]. In the KEYNOTE-062 global phase III trial, pembrolizumab, another PD-1 inhibitor, plus chemotherapy (FU or capecitabine and cisplatin) was not superior to chemotherapy for OS in patients with PD-L1 CPS ≥1 and HER2-negative AGC (median OS, 12.5 vs. 11.1 months; HR, 0.85; 95% CI, 0.70 to 1.03; P=0.05) [369]. PD-L1 IHC was performed using a PD-L1 IHC 22C3 pharmDx assay (Agilent Technologies, Carpinteria, CA, USA). In our meta-analysis including CheckMate 649, ATTRACTION-4, and KEYNOTE-062, palliative first-line systemic therapy with anti-PD1 improved OS compared to chemotherapy alone (HR, 0.82; 95% CI, 0.74 to 0.92, P=0.004) (Fig. 16). The KEYNOTE-859 phase III trial evaluating pembrolizumab in combination with chemotherapy (FU/cisplatin or capecitabine/oxaliplatin) as first-line treatment for patients with HER2-negative AGC is ongoing [367]. The KEYNOTE-859 trial will use a different chemotherapy backbone than the KEYNOTE-062 trial and a modified statistical design to better identify the underlying clinical benefit. Based on the aforementioned results, nivolumab plus XELOX or FOLFOX is currently approved as the first-line treatment in Korea.
Fig. 16. Forest plots for comparisons between palliative first-line chemotherapy with immune checkpoint inhibitor (experimental) vs. chemotherapy (control). (A) Overall survival. (B) Progression-free survival.
ICI = immune checkpoint inhibitor; SE = standard error; IV = interval variable; CI = confidence interval.
There are some issues according to different PD-L1 IHC assays and a lack of evidence for the addition of ICIs for low PD-L1-expressing AGC. A recent analysis of pivotal trials of ICI with chemotherapy showed that there were no significant benefits from nivolumab on OS in patients with PD-L1 CPS 1-4 from the CheckMate 649 trial [370]. Therefore, nivolumab combined with XELOX or FOLFOX is recommended in patients with PD-L1 CPS ≥5 and HER2-negative AGC. Meanwhile, the KEYNOTE-811 phase III trial evaluating pembrolizumab plus trastuzumab and chemotherapy for HER2-positive AGC is ongoing with promising tumor reduction (objective response rate, 74.4% vs. 51.9%; P<0.01) [367]. The dual blockade of PD-1/PD-L1 and HER2 needs to be further investigated in the future.
KQ 25: Could palliative second-line systemic therapy improve survival in patients with locally advanced unresectable or metastatic gastric cancer who progress after or fail palliative first-line systemic therapy?
Statement 25: Palliative second-line systemic therapy is recommended in patients with locally advanced unresectable or metastatic gastric cancer. Ramucirumab plus paclitaxel is preferentially recommended, but other agents could also be considered (evidence: high, recommendation: strong for).
RCTs and previous meta-analyses have demonstrated the survival benefit of second-line palliative chemotherapy (with irinotecan or taxanes) compared to best supportive care alone for patients with locally advanced unresectable or metastatic gastric cancer [371,372,373,374]. In the present meta-analysis, second-line systemic therapy significantly improved OS compared to best supportive care (HR, 0.69; 95% CI, 0.59 to 0.82, P<0.001) (Fig. 17). Weekly paclitaxel treatment was associated with similar survival outcomes to biweekly irinotecan treatment in previous phase III trials [375,376]. Meanwhile, ramucirumab monotherapy, involving a monoclonal antibody that targets VEGFR2, significantly improved OS and PFS compared to placebo in the REGARD trial [377]. Furthermore, the addition of ramucirumab to weekly paclitaxel significantly prolonged OS (median, 9.6 vs. 7.4 months; HR, 0.807; 95% CI, 0.678 to 0.962; P=0.017) and PFS (median, 4.4 vs. 2.9 months, HR, 0.635; 95% CI, 0.536 to 0.752; P<0.0001) compared to paclitaxel plus placebo in the RAINBOW trial [378].
Fig. 17. Forest plots for comparisons between palliative second-line systemic therapy (experimental) vs. best supportive care or placebo (control). (A) Overall survival. (B) Progression-free survival.
SE = standard error; IV = interval variable; CI = confidence interval.
Based on previous trials, ramucirumab in combination with paclitaxel is recommended as the most preferred second-line treatment. Other agents, including irinotecan, docetaxel, paclitaxel, or ramucirumab monotherapy, can also be considered second-line options if not previously administered in the first-line treatment.
Pembrolizumab, an anti-PD-1 antibody, failed to provide a significant survival benefit compared to paclitaxel [379,380,381], whereas it was efficacious in patients with solid tumors characterized as MSI-H, dMMR, or TMB-H (≥10 mutations/megabase) [382,383]. In Korea, pembrolizumab was approved in patients with several inoperable or metastatic solid tumors, including gastric cancer with MSI-H or dMMR, who have progressed following prior treatment and who have no satisfactory alternative treatment options.
Trastuzumab deruxtecan, a HER2-directed antibody and topoisomerase inhibitor conjugate, was recently approved by the Food and Drug Administration for the treatment of patients with HER2-positive gastric or GEJ adenocarcinoma who have received a prior trastuzumab-based regimen. The phase 2 trial of trastuzumab deruxtecan as second-line treatment provided clinical evidence only in the Western population [384]. Therefore, further phase 3 trials comparing the survival outcomes of trastuzumab deruxtecan to ramucirumab plus paclitaxel, the current standard second-line treatment, are needed in Asian patients with HER2-positive gastric cancer.
KQ 26: Could palliative third-line systemic therapy improve survival in patients with locally advanced unresectable or metastatic gastric cancer who progress after 2 previous palliative systemic therapies?
Statement 26: Palliative third-line systemic therapy is recommended in patients with locally advanced unresectable or metastatic gastric cancer (evidence: high, recommendation: strong for).
For patients with preserved performance status who have disease progression after second-line systemic therapy, palliative third-line systemic therapy is recommended. In our present meta-analysis, third-line systemic therapy significantly improved OS compared to best supportive care (HR, 0.68; 95% CI, 0.60 to 0.77, P<0.001) (Fig. 18).
Fig. 18. Forest plots for comparisons between palliative third-line systemic therapy (experimental) vs. best supportive care (control). (A) Overall survival. (B) Progression-free survival.
SE = standard error; IV = interval variable; CI = confidence interval.
Cytotoxic agents can be recommended as palliative third-line therapy. Docetaxel and irinotecan, as a randomized phase III trial (median OS, 5.3 vs. 3.8 months; HR, 0.66; 95% CI, 0.49 to 0.89; P=0.007) [371]. Several phase II and retrospective studies investigating taxane- or irinotecan-based chemotherapy as third-line treatment also showed consistent results [385,386,387]. In a randomized phase III trial, trifluridine/tipiracil significantly improved OS vs. placebo (median OS, 5.7 vs. 3.6 months; HR, 0.69; 95% CI, 0.56 to 0.85; P=0.00058) in AGC patients who had received at least 2 previous systemic treatments [388].
Nivolumab showed a survival benefit over placebo in heavily treated patients who received 2 or more previous systemic therapies in a randomized phase 3 ATTRACTION-2 trial (median OS, 5.3 vs. 4.1 months; HR, 0.63; 95% CI, 0.51 to 0.78; P<0.0001) [389]. Two-year updated data from the ATTRACTION-2 trial confirmed the long-term survival benefit of nivolumab regardless of PD-L1 expression status [390]. Based on the ATTRACTION-2 trial, nivolumab is recommended as a palliative third-line therapy for AGC patients who are naïve to ICI therapy.
In the phase 2 DESTINY-Gastric01 trial conducted in Japan and South Korea, trastuzumab deruxtecan was compared to the physician’s choice of irinotecan or paclitaxel in patients with HER2-positive gastric cancer who received at least 2 previous palliative systemic treatments including trastuzumab [391]. In this trial, trastuzumab deruxtecan was associated with significant improvements in the objective response rate (51% vs. 14%, P<0.001) and OS (median OS, 12.5 vs. 8.4 months; HR, 0.59; 95% CI, 0.39 to 0.88; P=0.01) compared to the physician’s choice of irinotecan or paclitaxel. The positive results from the DESTINY-Gastric01 trial led to approval for prescription of trastuzumab deruxtecan in patients with HER2-positive gastric or GEJ adenocarcinoma who received a prior trastuzumab-based regimen as second-line or later treatment in the US and third-line or later treatment in Korea.
Neoadjuvant chemotherapy (NCT)
KQ 27: Could NCT as part of perioperative chemotherapy improve survival in patients with resectable locally advanced gastric cancer compared to upfront surgery followed by adjuvant chemotherapy?
Statement 27: NCT as part of perioperative chemotherapy can be considered for patients with resectable locally advanced gastric cancer (evidence: high, recommendation: conditional for).
Adjuvant chemotherapy following D2 gastrectomy has been the standard treatment for pathological stage II or III gastric cancer in Asia. However, survival outcomes with adjuvant chemotherapy are still disappointing, especially for those with stage III disease. Moreover, adjuvant chemotherapy is often delayed following surgical resection due to surgical morbidities, and chemotherapy after gastrectomy is associated with more frequent adverse events. In this regard, NCT may be considered for the advantages of intensifying chemotherapy and commencing chemotherapy at an earlier time when patients are more medically fit.
Three prospective randomized phase 3 clinical trials have evaluated the clinical benefit of NCT as part of perioperative chemotherapy in Asia. The PRODIGY study performed in Korea investigated whether neoadjuvant docetaxel, oxaliplatin, and S-1 (DOS) followed by surgery and adjuvant S-1 could improve outcomes vs. upfront surgery followed by adjuvant S-1 in locally AGC patients with clinical T2/3 N+ or cT4 N-any disease [392]. NCT led to a higher rate of complete resection (R0 resection) than upfront surgery (95% vs. 84%, P<0.001). Neoadjuvant DOS followed by surgery and adjuvant S-1 significantly improved PFS compared to upfront surgery followed by adjuvant S-1 (adjusted HR, 0.70; 95% CI, 0.52 to 0.95; stratified log-rank P=0.023). The RESOLVE study evaluated perioperative SOX vs. upfront surgery followed by adjuvant CapOx [393]. Perioperative SOX significantly improved DFS compared with adjuvant XELOX in patients (HR, 0.77; 95% CI, 0.61 to 0.97; P=0.027). The JCOG0501 study evaluated the efficacy of neoadjuvant S-1 plus cisplatin followed by gastrectomy and adjuvant chemotherapy vs. upfront surgery and adjuvant S-1 in patients with Borrmann type 4 or large (≥8 cm) type 3 gastric cancer [394]. However, NCT with S-1 plus cisplatin failed to demonstrate a survival benefit. In our meta-analysis of Asian neoadjuvant trials, NCT showed clinical benefit compared to upfront surgery (DFS: HR, 0.79; 95% CI, 0.68 to 0.91) (Fig. 19).
Fig. 19. Forest plot for a comparison of disease-free survival between neoadjuvant chemotherapy as part of perioperative chemotherapy (experimental) vs. adjuvant chemotherapy only (control).
SE = standard error; IV = interval variable; CI = confidence interval.
On the other hand, it should be noted that the efficacy and safety of perioperative chemotherapy regimens used in Western countries including FU plus leucovorin, oxaliplatin, and docetaxel (FLOT) have never been reported in Asian populations.
Because NCT is commenced based on clinical radiological staging, which can be frequently inaccurate, inadvertent inclusion of early-stage disease may be an issue. In the exploratory analysis of the PRODIGY study, radiological criteria involving cT4 disease exhibited a lower percentage of pathologic stage I disease (5%) while preserving sensitivity for pathologic stage III disease (80.1%) [395]. Accordingly, RR reduction by NCT was most prominent in patients meeting this cT4N-any criterion, suggesting that patients meeting this criterion may be preferentially considered for NCT.
Considering its clinical benefit for DFS, NCT as part of perioperative chemotherapy can be considered one of the viable therapeutic options for patients with resectable locally advanced gastric cancer in Korea. The clinical decision to proceed with NCT should be made based on a careful discussion considering various factors, including the clinical stage as well as potential advantages and limitations of NCT over upfront surgery (either followed by adjuvant chemotherapy or not according to the pathological stage). A multidisciplinary team approach is recommended to make such treatment decisions. Long-term follow-up studies reporting OS outcomes with NCT, which may further help to make clinical decisions, are awaited.
RADIATION THERAPY
Adjuvant radiation therapy
KQ 28: Could adjuvant CRT improve treatment outcomes compared to adjuvant chemotherapy alone in patients with pathological stage II or III disease who have undergone curative gastrectomy?
Statement 28: Adjuvant CRT is not usually recommended in patients with pathological stage II or III gastric cancer who have undergone curative gastrectomy (evidence: high, recommendation: conditional against).
A total of 6 RCTs were included in the present meta-analysis, including 2 recent RCTs published after the Korean Practice Guideline for Gastric Cancer 2018: An Evidence-based, Multidisciplinary Approach [345,396,397,398,399]. The target volume of radiation therapy was generally similar in these trials, including the tumor bed, anastomotic site and/or stump, and regional LN stations.
In the meta-analysis, the addition of adjuvant CRT reduced locoregional recurrence compared to chemotherapy alone (HR, 0.62; 95% CI, 0.48 to 0.81; P=0.0004) with no significant difference in grade 3 or higher toxicities (HR, 0.85; 95% CI, 0.63 to 1.13; P=0.26). Adjuvant CRT showed superior outcomes compared to adjuvant chemotherapy alone in terms of DFS (HR, 0.85; 95% CI, 0.713 to 0.98; P=0.03). However, when compared to platinum-based combination chemotherapy, there was no benefit in terms of DFS (HR, 0.91; 95% CI, 0.78 to 1.07, P=0.24) and OS (HR, 1.03; 95% CI, 0.87 to 1.23; P=0.70) (Fig. 20).
Fig. 20. Forest plots for comparison between adjuvant concurrent CRT vs. adjuvant platinum-based combination CA. (A) Overall survival. (B) Disease-free survival. (C) Locoregional recurrence.
CRT = chemoradiation therapy; CA = chemotherapy alone; SE = standard error; IV = interval variable; CI = confidence interval.
Based on these studies, the addition of adjuvant CRT is not usually recommended in gastric cancer patients after complete resection with D2 lymphadenectomy. Further prospective trials should be focus on identifying potential candidates who might benefit from adjuvant CRT.
Neoadjuvant radiation therapy
KQ 29: Could neoadjuvant CRT improve treatment outcomes compared to NCT alone in patients with locally advanced gastric cancer?
Statement 29: The evidence for adding radiation to NCT is not conclusive in patients with locally advanced gastric cancer (evidence: moderate, recommendation: investigational).
Neoadjuvant chemoradiation (NCRT) is mainly studied for cancer of the esophagus, GEJ, and/or gastric cardia, where obtaining a complete R0 resection is challenging, and thus, there is a higher probability of locoregional relapse. The MAGIC trial showed that perioperative chemotherapy significantly improved OS over surgery alone for distal esophageal and gastric cardia adenocarcinoma [400]. Studies have focused on determining whether adding radiation therapy to NCT would have a benefit for gastric cancer. Two RCTs have been conducted, and 1 RCT is ongoing to compare the outcomes of NCRT vs. NCT alone in resectable cancer of the GEJ or stomach [401,402,403,404,405].
Final treatment outcomes were reported in the POET trial and the NeoRes [401,403]. In the pooled analysis, the pathologic complete remission rate (23.6% in NCRT vs. 6.3% in NCT) and pathologic N0 rate (69.9% in NCRT vs. 45.7% in NCT) were significantly improved in the NCRT group. Local PFS was reported only in the POET trial and showed a significant improvement with NCRT in the long-term follow-up data (HR, 0.37; 95% CI, 0.16 to 0.85). However, the improved pathologic responses did not lead to a significant improvement in OS (HR, 0.85; 95% CI, 0.63 to 1.15). In addition, there were no significant differences in the R0 resection rate (74% in NCRT vs. 66% in NCT). PFS was reported only in the NeoRes trial, and there was no significant difference in the 3-year PFS rate (44% in NCRT vs. 44% in NCT). An interim analysis of the TOPGEAR study showed no significant difference in adverse events or surgical complications [402]. In the pooled analysis of the TOPGEAR and NeoRes studies, there was no significant difference in severe gastrointestinal toxicity (15.3% in NCRT vs. 13.2% in NCT) (Fig. 21).
Fig. 21. Forest plots for comparisons between NCRT compared to NCT. (A) Overall survival. (B) Pathologic complete response. (C) Pathologic complete nodal regression. (D) R0 resection.
NCRT = neoadjuvant chemoradiation; NCT = neoadjuvant chemotherapy; SE = standard error; CI = confidence interval.
Notably, the mentioned studies were performed mainly in patients with esophageal and/or GEJ cancer. GEJ cancer is common in Western countries, and most studies evaluating the efficacy of NCRT for gastric cancer (mainly GEJ cancer) have also been performed in Western populations. In addition, in the NeoRes trial, some patients with squamous cell carcinoma at the esophagus were included. Evidence is still insufficient, and further prospective studies including Asian populations and nonjunction cancers are needed for better evidence.
TREATMENT FOR FAR ADVANCED GASTRIC CANCER
KQ 30: Can endoscopic stent insertion as a palliative therapy improve oral intake with comparable complication rate for malignant GOO compared to surgical bypass?
Statement 30: In patients with GOO caused by unresectable gastric cancer, either ES or surgical GJ for palliative treatment can be performed. The decision should be based on a multidisciplinary assessment of patients’ performance status, projected clinical course, and preferences (evidence: low, recommendation: conditional for).
In patients with advanced or metastatic gastric cancer, GOO commonly appears as a symptom, causing nausea, vomiting, dehydration, and malnutrition and leading to marked deteriorations in patients’ QOL. Given that radical surgery is not indicated in patients with incurable gastric cancer, palliative treatment is required to relieve symptoms of GOO and restore the ability to tolerate an oral diet.
Surgical GJ and ES are palliative treatments for GOO caused by unresectable gastric cancer. GJ is the standard palliative treatment for GOO and adequately relieves symptoms of GOO. However, early major complications and procedure-related mortality have been reported to be substantial [406,407]. ES is increasingly being performed for malignant GOO. With a shorter procedure time, faster resumption of oral intake, and shorter duration of hospital stay than GJ, ES presents an effective and less invasive therapeutic option for the palliative treatment of GOO [406,407]. Nevertheless, compared with GJ, a higher rate of complications, reintervention, and recurrent obstruction has also been reported [406,407].
In this clinical guideline, we compared the outcomes of ES with GJ by conducting a meta-analysis. Our literature search identified 1,637 articles, and a total of 15 studies were finally selected through the literature selection process [186,408,409,410,411,412,413,414,415,416,417,418,419,420]. These studies were included in the final table of evidence, i.e., 12 observational studies in addition to 3 RCTs. Of the 15 studies, 5 were Korean [186,413,416,417,418]. A total of 818 patients received ES, and 468 patients underwent GJ. The overall certainty for outcome results reported by the RCTs (low) and the observational studies (very low) was downgraded because of the small number of events and because of bias due to confounding and selection of participants. Regarding procedure outcomes, technical success (OR, 1.33; 95% CI, 0.37 to 4.73; P=0.66) and clinical success (OR, 0.68; 95% CI, 0.41 to 1.13; P=0.14) were not significantly different between the ES and GJ groups (Fig. 22A and B). There was no significant difference in procedure-related mortality (OR, 0.64; 95% CI, 0.26 to 1.63; P=0.35) (Fig. 22C). Regarding postoperative outcomes, patients in the ES group had faster resumption of oral intake (MD, −3.94 days; 95% CI, −4.01 to −3.88; P<0.001) and a shorter duration of hospital stay (MD, −6.56 days; 95% CI, −7.20 to −5.92; P<0.001) (Fig. 23A and B). The rate of minor complications was not significantly different between the 2 groups (OR, 0.52; 95% CI, 0.25 to 1.10; P=0.09) (Fig. 23C). However, the rates of major complications (OR, 1.81; 95% CI, 1.10 to 2.96; P=0.02) and reintervention (OR, 3.83; 95% CI, 2.40 to 6.12; P<0.001) were significantly higher in the ES group than in the GJ group (Fig. 23D and E). Moreover, ES was significantly associated with a shorter patency duration (MD, −4.97 months; 95% CI, −6.42 to −3.51; P<0.001) (Fig. 23F). However, OS was not significantly different between the ES and GJ groups (MD, 0.12 months; 95% CI, −0.48 to 0.72, P=0.69) (Fig. 23G).
Fig. 22. Forest plot results of meta-analysis of procedure outcomes. (A) Technical success. (B) Clinical success. (C) Procedure related mortality.
CI = confidence interval.
Fig. 23. Forest plot results of meta-analysis of postoperative outcomes. (A) Resumption of oral intake. (B) Duration of hospital stay. (C) Minor complications. (D) Major complications. (E) Re-intervention. (F) Patency duration. (G) Overall survival.
SD = standard deviation; IV = interval variable; CI = confidence interval.
Both GJ and ES are effective palliative treatments for GOO caused by unresectable gastric cancer. Our results suggest that ES may be associated with more favorable results in patients who are poor surgical candidates with relatively short life expectancy and those who place a high value on resumption of oral diet and being discharged early, while GJ is preferable in patients with more prolonged prognosis and good performance status. Stent insertion by radiologic intervention showed similar efficacy to endoscopic stent insertion in a single-center study [418]. When available, radiologic stent insertion can also be considered for GOO.
KQ 31: Can surgery plus chemotherapy improve survival outcomes for stage IV gastric cancer patients compared to chemotherapy only?
Statement 31-1: Reduction gastrectomy (or upfront debulking gastrectomy without systemic LN dissection) should not be considered as initial treatment options for stage IV gastric cancer patients who are susceptible to chemotherapy (evidence: high, recommendation: strong against).
The REGATTA trial, the only phase III RCT comparing gastrectomy with D1 dissection followed by chemotherapy vs. chemotherapy alone, focused on pure reduction surgery without metastasectomy [418]. Gastric cancer patients with a single noncurable factor were enrolled. Reduction surgery showed no survival benefit compared to chemotherapy alone, and the trial was terminated after the first interim analysis owing to no benefit in the surgery group (HR, 1.08; 95% CI, 0.74 to 1.58; P=0.06). Based on these findings, it was concluded that in patients with metastatic gastric cancer, reductive gastrectomy cannot be justified.
Statement 31-2: In stage IV gastric cancer patients with limited metastasis, conversion surgery might be considered as a treatment option for those with a good response to chemotherapy (evidence: low, recommendation: investigational).
Three retrospective studies and one prospective study were included in our meta-analysis. The data regarding OS showed better survival in stage IV gastric cancer patients who underwent systemic chemotherapy (SC) followed by radical surgery than in patients who received chemotherapy alone (HR, 0.58; 95% CI, 0.42 to 0.80; P<0.01) (Fig. 24).
Fig. 24. Forest plot for a comparison of overall survival between surgery after chemotherapy vs. chemotherapy alone in metastatic gastric cancer.
SE = standard error; IV = interval variable; CI = confidence interval.
In a large retrospective review, patients with stage IV cancer who were responsive to chemotherapy and underwent R0 resection following chemotherapy experienced better survival than patients in the R1 and R2 resection groups [421]. In one prospective nonrandomized trial, surgery after chemotherapy, especially in R0 resection, was associated with a survival benefit for gastric cancer patients with limited distant metastasis [422]. However, since collected studies are retrospective and thus may be influenced by selection bias in nature, evidence is limited for stronger recommendations. A prospective multicenter randomized trial, RENAISSANCE (AIO-FLOT5), is being conducted to elucidate the effects of surgical resection after chemotherapy on the survival and QOL of patients with limited metastatic adenocarcinoma of the stomach and esophagogastric junction, and its results are anticipated for a higher level of evidence [422].
Despite possible benefits from the meta-analysis, the role of conversion surgery and detailed indications are inconclusive as of now because of inevitable selection bias in the observational studies comparing surgery and systemic therapy for stage IV gastric cancer as well as uncertainty regarding whether the development of new systemic therapy will increase or decrease the clinical meaning of conversion surgery.
KQ 32: Can radical gastrectomy with local treatment plus SC improve survival outcomes compared to chemotherapy alone for gastric cancer patients with single organ oligometastasis?
Statement 32-1: Radical gastrectomy, metastasectomy and perioperative chemotherapy may be considered for selected gastric cancer patients with oligometastases in the liver (evidence: very low, recommendation: investigational).
Traditionally, oligometastasis has been defined as an intermediate state between localized and widespread systemic disease with the presence of fewer than 5 metastases [423]. However, the definition is still not clear, and oligometastasis is generally defined as fewer than 3 or 5 metastatic lesions involving 1 or 2 organs [424,425].
For hepatic oligometastasis in gastric cancer, 2 retrospective studies were included in our meta-analysis (Fig. 25) [426,427]. The meta-analysis showed that radical gastrectomy with hepatectomy plus SC provided a survival benefit compared to chemotherapy alone (HR, 0.27; 95% CI, 0.12 to 0.62; P<0.001). Local treatments, such as transarterial chemoembolization (TACE), radiofrequency ablation, and hepatic arterial infusion, have also been reported to provide potential survival benefits [428,429,430,431]. However, because most of the related studies were single-arm studies or compared with hepatectomy, they could not be included in the meta-analysis. Liu et al. [431] retrospectively compared radical gastrectomy with TACE plus SC with chemotherapy alone. Although all types of liver metastasis (H1, H2, H3) were included in this study, the median OS was 14 and 8 months, respectively, between the surgery and chemotherapy groups (P<0.001).
Fig. 25. Forest plot for comparison of overall survival between (hepatectomy and gastrectomy with chemotherapy) vs. (chemotherapy only) in gastric cancer with oligometastasis confined to liver from observational studies.
SE = standard error; IV = interval variable; CI = confidence interval.
There is some optimistic evidence that resection of liver oligometastasis may provide survival benefit. However, because of the small sample size and retrospective nature, the evidence of the included study is weak. Therefore, candidates for liver resection should be selected cautiously. Further evidence is required for generalization.
Statement 32-2: Radical gastrectomy, oophorectomy and perioperative chemotherapy could be considered for selected gastric cancer patients with oligometastases in the ovary (evidence: very low, recommendation: conditional for).
For ovarian metastasis, 3 retrospective studies were analyzed in the meta-analysis, and there was better survival in the metastasectomy group (HR, 0.45; 95% CI, 0.34 to 0.59; P<0.001) [432,433,434] (Fig. 26). Cheong et al. [435] reported that Krukenberg tumors were frequently accompanied by peritoneal dissemination with a significantly worse prognosis (HR, 1.74; 95% CI, 1.28 to 2.36; P<0.001), and only when curative resection was obtained was the median OS time longer in the resection group than in the nonresection group (17 vs. 3 months, P<0.001).
Fig. 26. Forest plot for comparison of overall survival between (oophorectomy and gastrectomy with chemotherapy) vs. (chemotherapy only) in gastric cancer with oligometastasis confined to ovary in observational studies.
SE = standard error; IV = interval variable; CI = confidence interval.
Regarding para-aortic LNs, only 3 prospective nonrandomized studies evaluated the response rate of preoperative chemotherapy and the efficacy of subsequent D2 LND plus para-aortic LN dissection; they did not show favorable survival outcomes [436,437,438].
KQ 33: Can additional IP chemotherapy improve survival outcome for gastric cancer patients with peritoneal carcinomatosis compared to SC alone?
Statement 33: For gastric cancer patients with peritoneal carcinomatosis, additional IP chemotherapy should be applied for investigational purposes (evidence: low, recommendation: investigational).
Peritoneal metastasis is known to be less responsive to SC and offer worse prognosis than hematogenous or lymphatic metastasis (mean survival time [MST]: 5.2–18 months) [439,440]. One of the causes is the limited delivery of anticancer drugs to the peritoneum due to the peritoneum-plasma barrier. For direct application to cancer cells on the peritoneum, IP infusion using anticancer drugs with lower absorption and systemic toxicity, such as paclitaxel and docetaxel, has been studied in various cancers. It is considered effective and safe in treating peritoneal cancer dissemination from ovarian cancer [441,442,443,444,445,446,447].
In gastric cancer patients with peritoneal metastasis, phase 1 and 2 studies showed that patients in the IP (paclitaxel + docetaxel) plus SC group had improved survival compared to patients in the SC alone group in terms of MST (24.6 vs. 15.1 months) and 1-year survival time (78% vs. 70.4%) [440].
However, the phase 3 trial of the IP plus SC study conducted by Ishigami et al. [448] did not show significant improvement in survival outcome compared to SC alone (HR, 0.72; 95% CI, 0.49 to 1.04; stratified log-rank P=0.080). In their study, the authors claim that patient withdrawal and protocol violation caused the true effect of IP therapy to be underestimated.
Currently, phase 1, 2, and 3 studies are underway in Korea. Further investigation is required for recommendation, and until trial outcome data become available, IP chemotherapy should only be applied for investigational purposes.
FOLLOW UP AND NUTRITIONAL CONSIDERATIONS
Oncologic follow-up
Patients are regularly followed-up after curative gastrectomy for gastric cancer. The primary goal of regular follow-up is early detection of recurrence or secondary cancer followed by timely treatment. Other important goals are to manage postgastrectomy symptoms, receive nutritional support, and improve QOL. However, there is a lack of high-level evidence on which examination or how often the examination should be performed. Although the NCCN, Japanese, and Chinese guidelines for gastric cancer recommend some follow-up schedules, they were based on expert opinions [89,92,111]. Because lack of evidence, we conducted a nationwide survey targeting all tertiary or general hospitals. The purpose of presenting this survey was only to provide baseline information regarding current practices and not to recommend, force, or limit practices. We hope to encourage further discussion and study on this issue.
A total of 71 representative clinicians from each hospital responded to the questionnaire via e-mail. Table 5 shows the main intervals (months) for the physical examination, blood test, tumor markers, abdomen pelvis CT, chest X-ray, and endoscopy. For patients with pathological stage I tumors, physical examination, and blood tests, including tumor markers, were mainly conducted every 6 months for 3 years and then every 6–12 months until 5 years postoperatively. Abdomen pelvis CT and chest X-ray were mainly checked every 6 months for 2 years, 6 or 12 months in the third year, and then annually until 5 years postoperatively. For patients with pathological stage II or III tumors, physical examination, and blood tests, including tumor markers, were mainly conducted every 3 months for 1 year postoperatively and then every 6 months until 5 years postoperatively. Abdomen pelvis CT and chest X-ray were mainly performed every 3 or 6 months for 1 year postoperatively, every 6 months in the second and third years, and then every 6 or 12 months until 5 years postoperatively. Esophagogastroduodenoscopy (EGD) was conducted once or twice within 1 year and then annually until 5 years postoperatively regardless of stage. After 5 years, annual EGD was recommended for all patients. In addition, a few hospitals checked chest CT as a routine examination annually during the follow-up period.
Table 5. Investigations of oncologic follow up period in 71 hospitals.
| Stage | Examinations | Within 1 yr | 1–2 yr | 2–3 yr | 3–5 yr | After 5 yr |
|---|---|---|---|---|---|---|
| Stage I | Physical examination, blood test, tumor makers | 6 mo (65%) | 12 mo (10%) | 12 mo (40%) | 12 mo (60%) | 24 mo (5%) |
| 3 mo (34%) | 6 mo (80%) | 6 mo (58%) | 6 mo (38%) | 12 mo (35%) | ||
| 3 mo (10%) | 3 mo (2%) | 3 mo (2%) | None (60%) | |||
| Abdomen pelvis CT, chest X-ray | 12 mo (58%) | 12 mo (78%) | 12 mo (90%) | 12 mo (95%) | 24 mo (5%) | |
| 6 mo (40%) | 6 mo (22%) | 6 mo (10%) | 6 mo (5%) | 12 mo (35%) | ||
| 3 mo (2%) | None (60%) | |||||
| Endoscopy | 12 mo (10%) | 12 mo (78%) | 12 mo (90%) | 12 mo (95%) | 24 mo (5%) | |
| 6 mo (80%) | 6 mo (22%) | 6 mo (10%) | 6 mo (5%) | 12 mo (35%) | ||
| 3 mo (10%) | None (60%) | |||||
| Stage II/III | Physical examination, blood test, tumor makers | 6 mo (25%) | 6 mo (70%) | 12 mo (5%) | 12 mo (20%) | 24 mo (2%) |
| 3 mo (65%) | 4 mo (10%) | 6 mo (70%) | 6 mo (75%) | 12 mo (56%) | ||
| Etc. (4, 2, 1 mo) (10%) | 3 mo (20%) | 4 mo (10%) | 3 mo (5%) | 6 mo (2%) | ||
| 3 mo (15%) | None (40%) | |||||
| Abdomen pelvis CT, chest X-ray | 12 mo (5%) | 12 mo (5%) | 12 mo (10%) | 12 mo (10%) | 24 mo (5%) | |
| 6 mo (65%) | 6 mo (65%) | 6 mo (80%) | 6 mo (80%) | 12 mo (35%) | ||
| 3 mo (30%) | 4 mo (5%) | 4 mo (5%) | 4 mo (5%) | None (60%) | ||
| 3 mo (25%) | 3 mo (5%) | 3 mo (5%) | ||||
| Endoscopy | 12 mo (60%) | 12 mo (80%) | 12 mo (85%) | 12 mo (85%) | 12 mo (85%) | |
| 6 mo (40%) | 6 mo (20%) | 6 mo (15%) | 6 mo (10%) | None (10%) |
Korea numbers in the parenthesis are proportions of the response from the participants.
CT = computed tomography.
Nutritional follow-up
Gastrectomy can be accompanied by not only short-term but also long-term nutritional deterioration. Therefore, nutritional status must be monitored after surgery, and nutritional supplements may be given accordingly. Table 6 shows the main interval (months) for body weight, nutritional parameters, anemia study, and bone related study.
Table 6. Investigations of nutritional follow up period.
| Resection type | Examinations | Within 1 yr | 1–2 yr | 2–3 yr | 3–5 yr | After 5 yr |
|---|---|---|---|---|---|---|
| Total gastrectomy | Body weight | 6 mo (40%) | 12 mo (5%) | 12 mo (20%) | 12 mo (60%) | 12 mo (50%) |
| 3 mo (50%) | 6 mo (80%) | 6 mo (80%) | 6 mo (38%) | None (50%) | ||
| 1–2 mo (10%) | 3 mo (15%) | 3 mo (2%) | ||||
| Nutritional parameters (total protein, albumin, total cholesterol) | 6 mo (40%) | 6 mo (80%) | 12 mo (15%) | 12 mo (40%) | 12 mo (50%) | |
| 3 mo (60%) | 3 mo (20%) | 6 mo (80%) | 6 mo (60%) | None (50%) | ||
| 3 mo (5%) | ||||||
| Anemia study (hemoglobin, iron, ferritin, vitamin B12, folate) | 6 mo (50%) | 6 mo (70%) | 12 mo (50%) | 12 mo (60%) | 12 mo (50%) | |
| 3 mo (50%) | 3 mo (25%) | 6 mo (50%) | 6 mo (40%) | 6 mo (10%) | ||
| None (40%) | ||||||
| Bone related | 6 mo (40%) | 6 mo (60%) | 12 mo (20%) | 12 mo (40%) | 12 mo (40%) | |
| 3 mo (40%) | 3 mo (20%) | 6 mo (60%) | 6 mo (40%) | 6 mo (10%) | ||
| None (20%) | None (20%) | None (20%) | None (20%) | None (50%) | ||
| Partial gastrectomy | Body weight | 6 mo (40%) | 12 mo (5%) | 12 mo (20%) | 12 mo (60%) | 12 mo (50%) |
| 3 mo (60%) | 6 mo (80%) | 6 mo (80%) | 6 mo (38%) | None (50%) | ||
| 3 mo (15%) | 3 mo (2%) | |||||
| Nutritional parameters (total protein, albumin, total cholesterol) | 6 mo (40%) | 6 mo (80%) | 12 mo (15%) | 12 mo (40%) | 12 mo (50%) | |
| 3 mo (60%) | 3 mo (20%) | 6 mo (80%) | 6 mo (60%) | None (50%) | ||
| 3 mo (5%) | ||||||
| Anemia study (hemoglobin, iron, ferritin, vitamin B12, folate) | 6 mo (40%) | 6 mo (80%) | 12 mo (40%) | 12 mo (40%) | 24 mo (5%) | |
| 3 mo (50%) | 3 mo (20%) | 6 mo (30%) | 6 mo (30%) | 12 mo (35%) | ||
| 1–2 mo (10%) | None (30%) | None (30%) | None (60%) | |||
| Bone related | 6 mo (40%) | 6 mo (60%) | 12 mo (20%) | 12 mo (40%) | 12 mo (40%) | |
| 3 mo (40%) | 3 mo (20%) | 6 mo (60%) | 6 mo (40%) | 6 mo (10%) | ||
| None (20%) | None (20%) | None (20%) | None (20%) | None (50%) |
Numbers in the parenthesis are proportions of the response from the participants.
Patients lose a significant amount of body weight after gastric cancer surgery. Lower preoperative body mass index (BMI), female sex, and TG or PG were significant risk factors for malnutrition (BMI <18.5 kg/m2) 6 months after surgery [449]. Postoperative sarcopenia could serve as a prognostic factor for survival in gastric cancer patients [449]. Some have suggested that postoperative oral nutritional supplementation could improve nutritional outcomes in high-risk patients, but concrete evidence is still lacking [450,451].
Iron deficiency is one of the most common nutritional problems after gastric cancer surgery, and the incidence gradually increases with time after gastrectomy. The prevalence of iron deficiency at 3 years was reported to be 64.8% and 90.5% after DG and TG, respectively, and overt anemia was observed in 31.9% of patients after gastric cancer surgery at 3 years [452]. Female sex and TG have been consistently identified as independent risk factors for iron deficiency in the literature [452,453,454]. Oral iron supplementation should be given in patients with iron deficiency to correct anemia and replenish body reserve. Intravenous iron can be used when oral preparations are not tolerated or are ineffective. Intravenous iron (ferric carboxymaltose) was proven effective in managing isovolemic anemia that occurred within a week after radical gastrectomy and significantly reduced the need for additional treatments for anemia [455]. Transfusion is reserved for patients with an urgent need or risks of cardiovascular decompensation [456].
Vitamin B12 deficiency can occur from the reduction in intrinsic factors and gastric acidity after gastric cancer surgery. The cumulative incidence was reported at 100% at 4 years after TG with a median time to deficiency of 15 months, while a significantly lower rate of 15.7% was seen after DG [452]. Elderly patients with low preoperative vitamin B12 levels can be predisposed to vitamin B12 deficiency even after DG. Prolonged vitamin B12 deficiency is associated with anemia and with irreversible neuropathy. Nationwide studies in Korea demonstrated that vitamin B12 deficiency after TG could also be related to the pathogenesis of Alzheimer’s dementia and Parkinson’s disease [457,458]. Therefore, periodic monitoring of serum vitamin B12 levels and adequate supplementation for therapeutic or prophylactic purposes are warranted for patients undergoing gastric cancer surgery. Intramuscular injection of vitamin B12 is generally suggested as the treatment of choice in TG patients deprived of intrinsic factors. At the same time, daily oral vitamin B12 supplementation at a high dosage (1,500 µg once daily) can be an alternative option with similar efficacy [459,460,461].
Postgastrectomy patients can suffer from metabolic bone disorders demonstrating significantly decreased bone mineral density [462,463,464]. A decrease in oral calcium intake and generalized malabsorption induced by rapid gut transit in the early postoperative period and vitamin D deficiency and secondary hyperparathyroidism in the longer term are suggested as common mechanisms underlying bone mineral density impairment after gastric cancer surgery [463,465]. A Korean nationwide cohort study demonstrated that gastric cancer survivors had an elevated risk of osteoporotic fracture (HR, 1.61; 95% CI, 1.53 to 1.70) [466]. Older age, female sex, and marked weight loss (≥20%) were independently associated with an increased risk of osteoporosis [467]. There is currently little evidence on the optimal strategies for monitoring bone health and fracture in patients undergoing gastric cancer surgery. Dual-energy X-ray absorptiometry can be used for quantitative assessment of bone mineral content and screening osteoporosis in gastrectomy patients. Currently, no universal guidelines are available for the prevention or management of metabolic bone disorders related to gastrectomy. Generally, oral calcium and vitamin D supplementation is recommended in populations with an increased risk of osteoporosis. A few recent RCTs demonstrated that alendronate therapy effectively reduced bone loss and bone resorption in gastrectomy patients [468].
According to a nationwide survey in 2022, the postoperative nutritional monitoring schedule was not significantly different between total and partial gastrectomy. During the first year after surgery, the majority of the respondents followed their patients either every 3 months (50%–51%) or every 6 months (41.7%–43.7%). The interval increased to every 6 months (73.6%–80.0%) in the following 2nd and 3rd years and then to either every 6 months (43.7%–48.6%) or 12 months (41.7%–49.3%) up to the 5th year. After 5 years, half of the respondents continued nutritional monitoring mostly every year, while the other half discontinued surveillance. Body weight, hemoglobin, total protein, albumin, and total cholesterol were evaluated every visit in most of the centers. Other commonly monitored nutritional indices included the following: iron (76.6%–81.9%), ferritin (73.2%–81.9%), vitamin B12 (86.1%–93.1%), folate (62.0%–73.6%), and calcium (80.3%–81.6%) levels. Other indices, such as vitamin D (30%), parathyroid hormones (5%), prealbumin (15%), and thiamine (5%), were selectively evaluated by a smaller number of respondents, and micronutrients, such as copper or zinc, were rarely monitored. Annual bone densitometry was utilized to evaluate bone health at approximately 10% of the centers.
MULTIDISCIPLINARY TREATMENT (MDT)
Although treatment plans for gastric cancer patients can be made straightforward in many routine cases, there are also numerous cases requiring multidisciplinary considerations to arrive at the best treatment option. The advantages of MDT may include correct diagnosis, change into better treatment plan, shorter decision-making time and survival benefit [469,470,471,472]. For these reasons, health services in several countries have implemented MDT as the preferred system in cancer treatment [111,469,470,473]. The MDT team in gastric cancer treatment can include surgeons, gastroenterologists, medical and radiation oncologists, radiologists, pathologists, nuclear medicine experts, and other members, such as nutritional services, social workers, nurses, and palliative care specialists [474,475,476,477].
Several studies have shown the advantages of MDT in gastrointestinal malignancies. After the MDT meeting, changes in diagnosis occurred for 18.4%–26.9% of the evaluated patients [478,479], and the treatment plan was changed in 23.0%–76.81% of cancer patients [479,480,481].
From the caregivers’ perspective, MDT meetings may provide an interprofessional opportunity for feedback on various diagnostic imaging, operative findings, and pathologic results, which is beneficial for all parties [482]. MDT meetings can be a good opportunity to record specialists’ opinions on complex cases and improve diagnosis accuracy, treatment quality and accurate communication [482,483].
However, regardless of the potential benefits of MDT, there is little evidence to support its advantage and scarce information about how and for whom MDT activities should be conducted in gastric cancer. Considering cost and time effectiveness, how to select patients and how to organize the MDT meeting can be one of the issues because many patients without substantial comorbidities can follow routine decision-making processes without MDT team discussion [484]. However, the number of cases requiring MDT team discussion may increase according to the development of diverse treatment options and increasing proportions of patients with very old ages and comorbidities. Allum et al. [485] recommended that MDT team activities should also involve discussing treatment decisions with patients. However, there is no evidence that the treatment discussion with patients is better than the conference type of MDT discussion. All reports regarding the benefit of MDT were about professional consensus meetings followed by private interviews of the designated caregiver with the patient [485,486,487,488,489,490,491]. We may wait for further research on which MDT discussion type is better in terms of treatment outcome and cost effectiveness.
ACKNOWLEDGMENTS
We especially express our sincere gratitude to the advisory committee: Jun Haeng Lee, Keun Won Ryu, and Sun Young Rha for counseling; Hwa Kyung Byun for contributing as an active investigator in radiation oncology; Keun-Wook Lee for internal review; Chang Hee Cho for contribution as librarian; and Jie Hye Kim for communication and secretarial contribution.
Footnotes
Endorsements: The present guidelines were endorsed by the Korean Society of Medical Oncology, the Korean Society of Gastroenterology, the Korean College of Helicobacter and Upper Gastrointestinal Research, the Korean Society of Gastrointestinal Endoscopy, the Korean Society of Pathologists, the Korean Society of Abdominal Radiology, the Korean Society of Radiation Oncology, the Korean Society of Nuclear Medicine, and the Korean Gastric Cancer Association.
Funding: This work was supported by The Korean Gastric Cancer Association and The Korean Cancer Management Guideline Network (KCGN), a research project supported by the Korean Ministry of Health and Welfare, 2021 commissioned task to the National Cancer Center (Grant number; NCC-2112570-1, NCC-2112570-2, NCC-2112570-3).
Conflict of Interest: Seong-Ho Kong has received research funding from Stryker Co., Ltd. and Medtronic Inc. as the principal investigator and is the CEO of VITCAL, Co., Ltd. No potential conflict of interest relevant to this article was reported.
References
- 1.International Agency for Research on Cancer. Cancer fact sheet stomach: Globocan 2020 [Internet] Geneva: World Health Organization; 2022. Available from: https://gco.iarc.fr/today/data/factsheets/cancers/7-Stomach-fact-sheet.pdf. [Google Scholar]
- 2.Hong S, Won YJ, Lee JJ, Jung KW, Kong HJ, Im JS, et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2018. Cancer Res Treat. 2021;53:301–315. doi: 10.4143/crt.2021.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.National Cancer Center, Korea Central Cancer Registry. Korean Cancer Report 2020. Goyang: National Cancer Center; 2022. [Google Scholar]
- 4.Park SH, Kang MJ, Yun EH, Jung KW. Epidemiology of gastric cancer in Korea: trends in incidence and survival based on Korea Central Cancer Registry Data (1999–2019) J Gastric Cancer. 2022;22:160–168. doi: 10.5230/jgc.2022.22.e21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Information Committee of the Korean Gastric Cancer Association. Korean Gastric Cancer Association-led nationwide survey on surgically treated gastric cancers in 2019. J Gastric Cancer. 2021;21:221–235. doi: 10.5230/jgc.2021.21.e27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.World Cancer Research Fund Network. Diet, nutrition, physical activity and stomach cancer [Internet] London: World Cancer Research Fund Network; 2018. Available from: https://www.wcrf.org/wp-content/uploads/2021/02/stomach-cancer-report.pdf. [Google Scholar]
- 7.Cavaleiro-Pinto M, Peleteiro B, Lunet N, Barros H. Helicobacter pylori infection and gastric cardia cancer: systematic review and meta-analysis. Cancer Causes Control. 2011;22:375–387. doi: 10.1007/s10552-010-9707-2. [DOI] [PubMed] [Google Scholar]
- 8.Dalmartello M, Turati F, Zhang ZF, Lunet N, Rota M, Bonzi R, et al. Allium vegetables intake and the risk of gastric cancer in the Stomach cancer Pooling (StoP) Project. Br J Cancer. 2022;126:1755–1764. doi: 10.1038/s41416-022-01750-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cai H, Sobue T, Kitamura T, Ishihara J, Nanri A, Mizoue T, et al. Low-carbohydrate diet and risk of cancer incidence: The Japan Public Health Center-based prospective study. Cancer Sci. 2022;113:744–755. doi: 10.1111/cas.15215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Guideline Committee of the Korean Gastric Cancer Association (KGCA), Development Working Group & Review Panel. Korean practice guideline for gastric cancer 2018: an evidence-based, multi-disciplinary approach. J Gastric Cancer. 2019;19:1–48. doi: 10.5230/jgc.2019.19.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee JH, Kim JG, Jung HK, Kim JH, Jeong WK, Jeon TJ, et al. Clinical practice guidelines for gastric cancer in Korea: an evidence-based approach. J Gastric Cancer. 2014;14:87–104. doi: 10.5230/jgc.2014.14.2.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.The Korean Gastric Cancer Association. Korean guideline for gastric cancer. J Korean Gastric Cancer Assoc. 2004;4:286–293. [Google Scholar]
- 13.GRADE Working Group. Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach [Internet] GRADE Working Group; 2013. Available from: https://gdt.gradepro.org/app/handbook/handbook.html. [Google Scholar]
- 14.Suh YS, Yang HK. Screening and early detection of gastric cancer: east versus west. Surg Clin North Am. 2015;95:1053–1066. doi: 10.1016/j.suc.2015.05.012. [DOI] [PubMed] [Google Scholar]
- 15.Ahn HS, Lee HJ, Yoo MW, Jeong SH, Park DJ, Kim HH, et al. Changes in clinicopathological features and survival after gastrectomy for gastric cancer over a 20-year period. Br J Surg. 2011;98:255–260. doi: 10.1002/bjs.7310. [DOI] [PubMed] [Google Scholar]
- 16.Suh YS, Lee J, Woo H, Shin D, Kong SH, Lee HJ, et al. National cancer screening program for gastric cancer in Korea: nationwide treatment benefit and cost. Cancer. 2020;126:1929–1939. doi: 10.1002/cncr.32753. [DOI] [PubMed] [Google Scholar]
- 17.Jun JK, Choi KS, Lee HY, Suh M, Park B, Song SH, et al. Effectiveness of the Korean national cancer screening program in reducing gastric cancer mortality. Gastroenterology. 2017;152:1319–1328.e7. doi: 10.1053/j.gastro.2017.01.029. [DOI] [PubMed] [Google Scholar]
- 18.Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14:101–112. doi: 10.1007/s10120-011-0041-5. [DOI] [PubMed] [Google Scholar]
- 19.Hayes T, Smyth E, Riddell A, Allum W. Staging in esophageal and gastric cancers. Hematol Oncol Clin North Am. 2017;31:427–440. doi: 10.1016/j.hoc.2017.02.002. [DOI] [PubMed] [Google Scholar]
- 20.Mocellin S, Pasquali S. Diagnostic accuracy of endoscopic ultrasonography (EUS) for the preoperative locoregional staging of primary gastric cancer. Cochrane Database Syst Rev. 2015;2015:CD009944. doi: 10.1002/14651858.CD009944.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee S, Jun JK, Suh M, Park B, Noh DK, Jung KW, et al. Gastric cancer screening uptake trends in Korea: results for the National Cancer Screening Program from 2002 to 2011: a prospective cross-sectional study. Medicine (Baltimore) 2015;94:e533. doi: 10.1097/MD.0000000000000533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ryu JE, Choi E, Lee K, Jun JK, Suh M, Jung KW, et al. Trends in the performance of the Korean National Cancer Screening Program for gastric cancer from 2007 to 2016. Cancer Res Treat. 2022;54:842–849. doi: 10.4143/crt.2021.482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Luu XQ, Lee K, Jun JK, Suh M, Jung KW, Choi KS. Effect of gastric cancer screening on long-term survival of gastric cancer patients: results of Korean national cancer screening program. J Gastroenterol. 2022;57:464–475. doi: 10.1007/s00535-022-01878-4. [DOI] [PubMed] [Google Scholar]
- 24.Chong CS, Ng CW, Shabbir A, Kono K, So JB. Computed tomography of the thorax for gastric cancer staging: is it necessary? Scand J Surg. 2015;104:244–247. doi: 10.1177/1457496915571400. [DOI] [PubMed] [Google Scholar]
- 25.Chen AH, Chan WH, Lee YH, Tseng JH, Yeh TS, Chiu CT, et al. Routine chest CT for staging of gastric cancer. Br J Surg. 2019;106:1197–1203. doi: 10.1002/bjs.11186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kurokawa Y, Takeuchi H, Doki Y, Mine S, Terashima M, Yasuda T, et al. Mapping of lymph node metastasis from esophagogastric junction tumors: a prospective nationwide multicenter study. Ann Surg. 2021;274:120–127. doi: 10.1097/SLA.0000000000003499. [DOI] [PubMed] [Google Scholar]
- 27.Shen L, Shan YS, Hu HM, Price TJ, Sirohi B, Yeh KH, et al. Management of gastric cancer in Asia: resource-stratified guidelines. Lancet Oncol. 2013;14:e535–e547. doi: 10.1016/S1470-2045(13)70436-4. [DOI] [PubMed] [Google Scholar]
- 28.Choi JI, Joo I, Lee JM. State-of-the-art preoperative staging of gastric cancer by MDCT and magnetic resonance imaging. World J Gastroenterol. 2014;20:4546–4557. doi: 10.3748/wjg.v20.i16.4546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bhandari S, Shim CS, Kim JH, Jung IS, Cho JY, Lee JS, et al. Usefulness of three-dimensional, multidetector row CT (virtual gastroscopy and multiplanar reconstruction) in the evaluation of gastric cancer: a comparison with conventional endoscopy, EUS, and histopathology. Gastrointest Endosc. 2004;59:619–626. doi: 10.1016/s0016-5107(04)00169-5. [DOI] [PubMed] [Google Scholar]
- 30.Kim HJ, Kim AY, Oh ST, Kim JS, Kim KW, Kim PN, et al. Gastric cancer staging at multi-detector row CT gastrography: comparison of transverse and volumetric CT scanning. Radiology. 2005;236:879–885. doi: 10.1148/radiol.2363041101. [DOI] [PubMed] [Google Scholar]
- 31.Shimizu K, Ito K, Matsunaga N, Shimizu A, Kawakami Y. Diagnosis of gastric cancer with MDCT using the water-filling method and multiplanar reconstruction: CT-histologic correlation. AJR Am J Roentgenol. 2005;185:1152–1158. doi: 10.2214/AJR.04.0651. [DOI] [PubMed] [Google Scholar]
- 32.Hur J, Park MS, Lee JH, Lim JS, Yu JS, Hong YJ, et al. Diagnostic accuracy of multidetector row computed tomography in T- and N staging of gastric cancer with histopathologic correlation. J Comput Assist Tomogr. 2006;30:372–377. doi: 10.1097/00004728-200605000-00005. [DOI] [PubMed] [Google Scholar]
- 33.Chen CY, Hsu JS, Wu DC, Kang WY, Hsieh JS, Jaw TS, et al. Gastric cancer: preoperative local staging with 3D multi-detector row CT--correlation with surgical and histopathologic results. Radiology. 2007;242:472–482. doi: 10.1148/radiol.2422051557. [DOI] [PubMed] [Google Scholar]
- 34.Yang DM, Kim HC, Jin W, Ryu CW, Kang JH, Park CH, et al. 64 multidetector-row computed tomography for preoperative evaluation of gastric cancer: histological correlation. J Comput Assist Tomogr. 2007;31:98–103. doi: 10.1097/01.rct.0000234072.16209.ab. [DOI] [PubMed] [Google Scholar]
- 35.Yan C, Zhu ZG, Yan M, Zhang H, Pan ZL, Chen J, et al. Value of multidetector-row computed tomography in the preoperative T and N staging of gastric carcinoma: a large-scale Chinese study. J Surg Oncol. 2009;100:205–214. doi: 10.1002/jso.21316. [DOI] [PubMed] [Google Scholar]
- 36.Hwang SW, Lee DH, Lee SH, Park YS, Hwang JH, Kim JW, et al. Preoperative staging of gastric cancer by endoscopic ultrasonography and multidetector-row computed tomography. J Gastroenterol Hepatol. 2010;25:512–518. doi: 10.1111/j.1440-1746.2009.06106.x. [DOI] [PubMed] [Google Scholar]
- 37.Moschetta M, Stabile Ianora AA, Anglani A, Marzullo A, Scardapane A, Angelelli G. Preoperative T staging of gastric carcinoma obtained by MDCT vessel probe reconstructions and correlations with histological findings. Eur Radiol. 2010;20:138–145. doi: 10.1007/s00330-009-1482-7. [DOI] [PubMed] [Google Scholar]
- 38.Furukawa K, Miyahara R, Itoh A, Ohmiya N, Hirooka Y, Mori K, et al. Diagnosis of the invasion depth of gastric cancer using MDCT with virtual gastroscopy: comparison with staging with endoscopic ultrasound. AJR Am J Roentgenol. 2011;197:867–875. doi: 10.2214/AJR.10.5872. [DOI] [PubMed] [Google Scholar]
- 39.Kim JW, Shin SS, Heo SH, Choi YD, Lim HS, Park YK, et al. Diagnostic performance of 64-section CT using CT gastrography in preoperative T staging of gastric cancer according to 7th edition of AJCC cancer staging manual. Eur Radiol. 2012;22:654–662. doi: 10.1007/s00330-011-2283-3. [DOI] [PubMed] [Google Scholar]
- 40.Kumano S, Okada M, Shimono T, Kuwabara M, Yagyu Y, Imaoka I, et al. T-staging of gastric cancer of air-filling multidetector-row CT: comparison with hydro-multidetector-row CT. Eur J Radiol. 2012;81:2953–2960. doi: 10.1016/j.ejrad.2011.12.039. [DOI] [PubMed] [Google Scholar]
- 41.Feng XY, Wang W, Luo GY, Wu J, Zhou ZW, Li W, et al. Comparison of endoscopic ultrasonography and multislice spiral computed tomography for the preoperative staging of gastric cancer - results of a single institution study of 610 Chinese patients. PLoS One. 2013;8:e78846. doi: 10.1371/journal.pone.0078846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wani AH, Parry AH, Feroz I, Choh NA. Preoperative staging of gastric cancer using computed tomography and its correlation with histopathology with emphasis on multi-planar reformations and virtual gastroscopy. J Gastrointest Cancer. 2021;52:606–615. doi: 10.1007/s12029-020-00436-6. [DOI] [PubMed] [Google Scholar]
- 43.Burbidge S, Mahady K, Naik K. The role of CT and staging laparoscopy in the staging of gastric cancer. Clin Radiol. 2013;68:251–255. doi: 10.1016/j.crad.2012.07.015. [DOI] [PubMed] [Google Scholar]
- 44.Jeong J, Cho I, Kong E, Chun K, Jang B, Kim T, et al. Evaluation of hybrid PET/CT gastrography in gastric cancer. Nucl Med (Stuttg) 2013;52:107–112. doi: 10.3413/Nukmed-0504-12-05. [DOI] [PubMed] [Google Scholar]
- 45.Kim JE, Lee MW, Moon JW, Song KD, Park Y, Park MJ, et al. CT detection of dry peritoneal seeding in patients with advanced gastric cancer. Hepatogastroenterology. 2012;59:2202–2206. doi: 10.5754/hge11001. [DOI] [PubMed] [Google Scholar]
- 46.Kim SJ, Kim HH, Kim YH, Hwang SH, Lee HS, Park DJ, et al. Peritoneal metastasis: detection with 16- or 64-detector row CT in patients undergoing surgery for gastric cancer. Radiology. 2009;253:407–415. doi: 10.1148/radiol.2532082272. [DOI] [PubMed] [Google Scholar]
- 47.Pan Z, Zhang H, Yan C, Du L, Ding B, Song Q, et al. Determining gastric cancer resectability by dynamic MDCT. Eur Radiol. 2010;20:613–620. doi: 10.1007/s00330-009-1576-2. [DOI] [PubMed] [Google Scholar]
- 48.Perlaza P, Ortín J, Pagès M, Buxó E, Fernández-Esparrach G, Colletti PM, et al. Should 18F-FDG PET/CT be routinely performed in the clinical staging of locally advanced gastric adenocarcinoma? Clin Nucl Med. 2018;43:402–410. doi: 10.1097/RLU.0000000000002028. [DOI] [PubMed] [Google Scholar]
- 49.Mao Y, Chen B, Wang H, Zhang Y, Yi X, Liao W, et al. Diagnostic performance of magnetic resonance imaging for colorectal liver metastasis: a systematic review and meta-analysis. Sci Rep. 2020;10:1969. doi: 10.1038/s41598-020-58855-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Huang Z, Xie DH, Guo L, Hu CH, Fang X, Meng Q, et al. The utility of MRI for pre-operative T and N staging of gastric carcinoma: a systematic review and meta-analysis. Br J Radiol. 2015;88:20140552. doi: 10.1259/bjr.20140552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Laghi A, Bellini D, Rengo M, Accarpio F, Caruso D, Biacchi D, et al. Diagnostic performance of computed tomography and magnetic resonance imaging for detecting peritoneal metastases: systematic review and meta-analysis. Radiol Med (Torino) 2017;122:1–15. doi: 10.1007/s11547-016-0682-x. [DOI] [PubMed] [Google Scholar]
- 52.Upadhyay M, Samal J, Kandpal M, Singh OV, Vivekanandan P. The Warburg effect: insights from the past decade. Pharmacol Ther. 2013;137:318–330. doi: 10.1016/j.pharmthera.2012.11.003. [DOI] [PubMed] [Google Scholar]
- 53.Daamen LA, Groot VP, Goense L, Wessels FJ, Borel Rinkes IH, Intven MP, et al. The diagnostic performance of CT versus FDG PET-CT for the detection of recurrent pancreatic cancer: a systematic review and meta-analysis. Eur J Radiol. 2018;106:128–136. doi: 10.1016/j.ejrad.2018.07.010. [DOI] [PubMed] [Google Scholar]
- 54.Chen Y, Zhang D, Fan K. Diagnostic accuracy of F18-fluorodeoxyglucose positron emission tomography-computed tomography for the detection of non-small cell lung cancer recurrence: a systematic review and meta-analysis. Curr Med Imaging. 2022 doi: 10.2174/1573405618666220802101446. [DOI] [PubMed] [Google Scholar]
- 55.Stahl A, Ott K, Weber WA, Becker K, Link T, Siewert JR, et al. FDG PET imaging of locally advanced gastric carcinomas: correlation with endoscopic and histopathological findings. Eur J Nucl Med Mol Imaging. 2003;30:288–295. doi: 10.1007/s00259-002-1029-5. [DOI] [PubMed] [Google Scholar]
- 56.Yun M. Imaging of gastric cancer metabolism using 18 F-FDG PET/CT. J Gastric Cancer. 2014;14:1–6. doi: 10.5230/jgc.2014.14.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ock CY, Kim TY, Lee KH, Han SW, Im SA, Kim TY, et al. Metabolic landscape of advanced gastric cancer according to HER2 and its prognostic implications. Gastric Cancer. 2016;19:421–430. doi: 10.1007/s10120-015-0504-1. [DOI] [PubMed] [Google Scholar]
- 58.Kim EY, Lee WJ, Choi D, Lee SJ, Choi JY, Kim BT, et al. The value of PET/CT for preoperative staging of advanced gastric cancer: comparison with contrast-enhanced CT. Eur J Radiol. 2011;79:183–188. doi: 10.1016/j.ejrad.2010.02.005. [DOI] [PubMed] [Google Scholar]
- 59.Lee SJ, Lee WW, Yoon HJ, Lee HY, Lee KH, Kim YH, et al. Regional PET/CT after water gastric inflation for evaluating loco-regional disease of gastric cancer. Eur J Radiol. 2013;82:935–942. doi: 10.1016/j.ejrad.2013.01.014. [DOI] [PubMed] [Google Scholar]
- 60.Namikawa T, Okabayshi T, Nogami M, Ogawa Y, Kobayashi M, Hanazaki K. Assessment of (18)F-fluorodeoxyglucose positron emission tomography combined with computed tomography in the preoperative management of patients with gastric cancer. Int J Clin Oncol. 2014;19:649–655. doi: 10.1007/s10147-013-0598-6. [DOI] [PubMed] [Google Scholar]
- 61.Park K, Jang G, Baek S, Song H. Usefulness of combined PET/CT to assess regional lymph node involvement in gastric cancer. Tumori. 2014;100:201–206. doi: 10.1177/030089161410000214. [DOI] [PubMed] [Google Scholar]
- 62.Altini C, Niccoli Asabella A, Di Palo A, Fanelli M, Ferrari C, Moschetta M, et al. 18F-FDG PET/CT role in staging of gastric carcinomas: comparison with conventional contrast enhancement computed tomography. Medicine (Baltimore) 2015;94:e864. doi: 10.1097/MD.0000000000000864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Okumura Y, Aikou S, Onoyama H, Jinbo K, Yamagata Y, Mori K, et al. Evaluation of 18F-FDG uptake for detecting lymph node metastasis of gastric cancer: a prospective pilot study for one-to-one comparison of radiation dose and pathological findings. World J Surg Oncol. 2015;13:327. doi: 10.1186/s12957-015-0743-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lee DH, Kim SH, Joo I, Hur BY, Han JK. Comparison between 18F-FDG PET/MRI and MDCT for the assessment of preoperative staging and resectability of gastric cancer. Eur J Radiol. 2016;85:1085–1091. doi: 10.1016/j.ejrad.2016.03.015. [DOI] [PubMed] [Google Scholar]
- 65.Nakajo M, Kajiya Y, Tani A, Jinguji M, Nakajo M, Yoshiura T. FLT-PET/CT diagnosis of primary and metastatic nodal lesions of gastric cancer: comparison with FDG-PET/CT. Abdom Radiol (NY) 2016;41:1891–1898. doi: 10.1007/s00261-016-0788-6. [DOI] [PubMed] [Google Scholar]
- 66.Lehmann K, Eshmuminov D, Bauerfeind P, Gubler C, Veit-Haibach P, Weber A, et al. 18FDG-PET-CT improves specificity of preoperative lymph-node staging in patients with intestinal but not diffuse-type esophagogastric adenocarcinoma. Eur J Surg Oncol. 2017;43:196–202. doi: 10.1016/j.ejso.2016.08.020. [DOI] [PubMed] [Google Scholar]
- 67.Kudou M, Kosuga T, Kubota T, Okamoto K, Komatsu S, Shoda K, et al. Value of preoperative PET-CT in the prediction of pathological stage of gastric cancer. Ann Surg Oncol. 2018;25:1633–1639. doi: 10.1245/s10434-018-6455-0. [DOI] [PubMed] [Google Scholar]
- 68.Bosch KD, Chicklore S, Cook GJ, Davies AR, Kelly M, Gossage JA, et al. Staging FDG PET-CT changes management in patients with gastric adenocarcinoma who are eligible for radical treatment. Eur J Nucl Med Mol Imaging. 2020;47:759–767. doi: 10.1007/s00259-019-04429-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Jiang D, Chen X, You Z, Wang H, Zhang X, Li X, et al. Comparison of [68 Ga]Ga-FAPI-04 and [18F]-FDG for the detection of primary and metastatic lesions in patients with gastric cancer: a bicentric retrospective study. Eur J Nucl Med Mol Imaging. 2022;49:732–742. doi: 10.1007/s00259-021-05441-w. [DOI] [PubMed] [Google Scholar]
- 70.Roedl JB, Prabhakar HB, Mueller PR, Colen RR, Blake MA. Prediction of metastatic disease and survival in patients with gastric and gastroesophageal junction tumors: the incremental value of PET-CT over PET and the clinical role of primary tumor volume measurements. Acad Radiol. 2009;16:218–226. doi: 10.1016/j.acra.2008.06.004. [DOI] [PubMed] [Google Scholar]
- 71.Chung HW, Lee EJ, Cho YH, Yoon SY, So Y, Kim SY, et al. High FDG uptake in PET/CT predicts worse prognosis in patients with metastatic gastric adenocarcinoma. J Cancer Res Clin Oncol. 2010;136:1929–1935. doi: 10.1007/s00432-010-0852-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Smyth E, Schöder H, Strong VE, Capanu M, Kelsen DP, Coit DG, et al. A prospective evaluation of the utility of 2-deoxy-2-[(18) F]fluoro-D-glucose positron emission tomography and computed tomography in staging locally advanced gastric cancer. Cancer. 2012;118:5481–5488. doi: 10.1002/cncr.27550. [DOI] [PubMed] [Google Scholar]
- 73.Findlay JM, Antonowicz S, Segaran A, El Kafsi J, Zhang A, Bradley KM, et al. Routinely staging gastric cancer with 18F-FDG PET-CT detects additional metastases and predicts early recurrence and death after surgery. Eur Radiol. 2019;29:2490–2498. doi: 10.1007/s00330-018-5904-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Dębiec K, Wydmański J, d’Amico A, Gorczewska I, Krzywon A, Cortez AJ, et al. The application of 18F-FDG-PET/CT in gastric cancerstaging and factors affecting its sensitivity. Hell J Nucl Med. 2021;24:66–74. doi: 10.1967/s002449912308. [DOI] [PubMed] [Google Scholar]
- 75.Gertsen EC, Brenkman HJ, van Hillegersberg R, van Sandick JW, van Berge Henegouwen MI, Gisbertz SS, et al. 18F-fludeoxyglucose-positron emission tomography/computed tomography and laparoscopy for staging of locally advanced gastric cancer: a multicenter prospective Dutch cohort study (PLASTIC) JAMA Surg. 2021;156:e215340. doi: 10.1001/jamasurg.2021.5340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Bilici A, Ustaalioglu BB, Seker M, Kefeli U, Canpolat N, Tekinsoy B, et al. The role of 18F-FDG PET/CT in the assessment of suspected recurrent gastric cancer after initial surgical resection: can the results of FDG PET/CT influence patients’ treatment decision making? Eur J Nucl Med Mol Imaging. 2011;38:64–73. doi: 10.1007/s00259-010-1611-1. [DOI] [PubMed] [Google Scholar]
- 77.Graziosi L, Bugiantella W, Cavazzoni E, Cantarella F, Porcari M, Baffa N, et al. Role of FDG-PET/CT in follow-up of patients treated with resective gastric surgery for tumour. Ann Ital Chir. 2011;82:125–129. [PubMed] [Google Scholar]
- 78.Kim DW, Park SA, Kim CG. Detecting the recurrence of gastric cancer after curative resection: comparison of FDG PET/CT and contrast-enhanced abdominal CT. J Korean Med Sci. 2011;26:875–880. doi: 10.3346/jkms.2011.26.7.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kim SJ, Cho YS, Moon SH, Bae JM, Kim S, Choe YS, et al. Primary tumor 18F-FDG avidity affects the performance of 18F-FDG PET/CT for detecting gastric cancer recurrence. J Nucl Med. 2016;57:544–550. doi: 10.2967/jnumed.115.163295. [DOI] [PubMed] [Google Scholar]
- 80.Lee JE, Hong SP, Ahn DH, Jeon TJ, Kang MK, Kwon CI, et al. The role of 18F-FDG PET/CT in the evaluation of gastric cancer recurrence after curative gastrectomy. Yonsei Med J. 2011;52:81–88. doi: 10.3349/ymj.2011.52.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lee DY, Lee CH, Seo MJ, Lee SH, Ryu JS, Lee JJ. Performance of (18)F-FDG PET/CT as a postoperative surveillance imaging modality for asymptomatic advanced gastric cancer patients. Ann Nucl Med. 2014;28:789–795. doi: 10.1007/s12149-014-0871-4. [DOI] [PubMed] [Google Scholar]
- 82.Lee JW, Lee SM, Son MW, Lee MS. Diagnostic performance of FDG PET/CT for surveillance in asymptomatic gastric cancer patients after curative surgical resection. Eur J Nucl Med Mol Imaging. 2016;43:881–888. doi: 10.1007/s00259-015-3249-5. [DOI] [PubMed] [Google Scholar]
- 83.Nakamoto Y, Togashi K, Kaneta T, Fukuda H, Nakajima K, Kitajima K, et al. Clinical value of whole-body FDG-PET for recurrent gastric cancer: a multicenter study. Jpn J Clin Oncol. 2009;39:297–302. doi: 10.1093/jjco/hyp010. [DOI] [PubMed] [Google Scholar]
- 84.Park MJ, Lee WJ, Lim HK, Park KW, Choi JY, Kim BT. Detecting recurrence of gastric cancer: the value of FDG PET/CT. Abdom Imaging. 2009;34:441–447. doi: 10.1007/s00261-008-9424-4. [DOI] [PubMed] [Google Scholar]
- 85.Sharma P, Singh H, Suman SK, Sharma A, Reddy RM, Thulkar S, et al. 18F-FDG PET-CT for detecting recurrent gastric adenocarcinoma: results from a Non-Oriental Asian population. Nucl Med Commun. 2012;33:960–966. doi: 10.1097/MNM.0b013e328355b694. [DOI] [PubMed] [Google Scholar]
- 86.Sim SH, Kim YJ, Oh DY, Lee SH, Kim DW, Kang WJ, et al. The role of PET/CT in detection of gastric cancer recurrence. BMC Cancer. 2009;9:73. doi: 10.1186/1471-2407-9-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Sun L, Su XH, Guan YS, Pan WM, Luo ZM, Wei JH, et al. Clinical role of 18F-fluorodeoxyglucose positron emission tomography/computed tomography in post-operative follow up of gastric cancer: initial results. World J Gastroenterol. 2008;14:4627–4632. doi: 10.3748/wjg.14.4627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kim JH, Heo SH, Kim JW, Shin SS, Min JJ, Kwon SY, et al. Evaluation of recurrence in gastric carcinoma: Comparison of contrast-enhanced computed tomography and positron emission tomography/computed tomography. World J Gastroenterol. 2017;23:6448–6456. doi: 10.3748/wjg.v23.i35.6448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.National Health Commission of the People’s Republic of China. National guidelines for diagnosis and treatment of gastric cancer 2022 in China (English version) Chin J Cancer Res. 2022;34:207–237. doi: 10.21147/j.issn.1000-9604.2022.03.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Reiner-Concin A. External quality assurance in immunohistochemistry - is it the solution to a complex problem? Breast Care (Basel) 2008;3:78–79. doi: 10.1159/000126738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hu B, El Hajj N, Sittler S, Lammert N, Barnes R, Meloni-Ehrig A. Gastric cancer: classification, histology and application of molecular pathology. J Gastrointest Oncol. 2012;3:251–261. doi: 10.3978/j.issn.2078-6891.2012.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Agnes A, Estrella JS, Badgwell B. The significance of a nineteenth century definition in the era of genomics: linitis plastica. World J Surg Oncol. 2017;15:123. doi: 10.1186/s12957-017-1187-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182–188. doi: 10.1111/his.13975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.International Agency for Research on Cancer. WHO Classification of Tumours of the Digestive System. Lyon: International Agency for Research on Cancer; 2019. [Google Scholar]
- 95.Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. an attempt at a histo-clinical classification. Acta Pathol Microbiol Scand. 1965;64:31–49. doi: 10.1111/apm.1965.64.1.31. [DOI] [PubMed] [Google Scholar]
- 96.Yasuda K, Adachi Y, Shiraishi N, Maeo S, Kitano S. Papillary adenocarcinoma of the stomach. Gastric Cancer. 2000;3:33–38. doi: 10.1007/pl00011687. [DOI] [PubMed] [Google Scholar]
- 97.Huang Q, Zou X. Clinicopathology of early gastric carcinoma: an update for pathologists and gastroenterologists. Gastrointest Tumors. 2017;3:115–124. doi: 10.1159/000456005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Cheng Y, Du M, Zhou X, Guo L, Xu K, Huang J, et al. High-grade papillary early gastric carcinoma with high risk for lymph node metastasis and poor prognosis: a clinicopathologic study of 96 cases among 1136 consecutive radical gastrectomies. Am J Surg Pathol. 2021;45:1661–1668. doi: 10.1097/PAS.0000000000001759. [DOI] [PubMed] [Google Scholar]
- 99.Kim YH, Kim JH, Kim H, Kim H, Lee YC, Lee SK, et al. Is the recent WHO histological classification for gastric cancer helpful for application to endoscopic resection? Gastric Cancer. 2016;19:869–875. doi: 10.1007/s10120-015-0538-4. [DOI] [PubMed] [Google Scholar]
- 100.Kook MC. Risk factors for lymph node metastasis in undifferentiated-type gastric carcinoma. Clin Endosc. 2019;52:15–20. doi: 10.5946/ce.2018.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Garcia-Pelaez J, Barbosa-Matos R, Gullo I, Carneiro F, Oliveira C. Histological and mutational profile of diffuse gastric cancer: current knowledge and future challenges. Mol Oncol. 2021;15:2841–2867. doi: 10.1002/1878-0261.12948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kunisaki C, Akiyama H, Nomura M, Matsuda G, Otsuka Y, Ono HA, et al. Clinicopathologic characteristics and surgical outcomes of mucinous gastric carcinoma. Ann Surg Oncol. 2006;13:836–842. doi: 10.1245/ASO.2006.03.077. [DOI] [PubMed] [Google Scholar]
- 103.Choi JS, Kim MA, Lee HE, Lee HS, Kim WH. Mucinous gastric carcinomas: clinicopathologic and molecular analyses. Cancer. 2009;115:3581–3590. doi: 10.1002/cncr.24422. [DOI] [PubMed] [Google Scholar]
- 104.Komatsu S, Ichikawa D, Miyamae M, Shimizu H, Konishi H, Shiozaki A, et al. Histological mixed-type as an independent prognostic factor in stage I gastric carcinoma. World J Gastroenterol. 2015;21:549–555. doi: 10.3748/wjg.v21.i2.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lee JH, Choi IJ, Han HS, Kim YW, Ryu KW, Yoon HM, et al. Risk of lymph node metastasis in differentiated type mucosal early gastric cancer mixed with minor undifferentiated type histology. Ann Surg Oncol. 2015;22:1813–1819. doi: 10.1245/s10434-014-4167-7. [DOI] [PubMed] [Google Scholar]
- 106.Seo HS, Lee GE, Kang MG, Han KH, Jung ES, Song KY. Mixed histology is a risk factor for lymph node metastasis in early gastric cancer. J Surg Res. 2019;236:271–277. doi: 10.1016/j.jss.2018.11.055. [DOI] [PubMed] [Google Scholar]
- 107.Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2018 (5th edition) Gastric Cancer. 2021;24:1–21. doi: 10.1007/s10120-020-01042-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, et al. AJCC Cancer Staging Manual. Eighth edition. Chicago (IL): Springer; 2017. [DOI] [PubMed] [Google Scholar]
- 109.Liu YY, Fang WL, Wang F, Hsu JT, Tsai CY, Liu KH, et al. Does a higher cutoff value of lymph node retrieval substantially improve survival in patients with advanced gastric cancer?-time to embrace a new digit. Oncologist. 2017;22:97–106. doi: 10.1634/theoncologist.2016-0239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Ryan R, Gibbons D, Hyland JM, Treanor D, White A, Mulcahy HE, et al. Pathological response following long-course neoadjuvant chemoradiotherapy for locally advanced rectal cancer. Histopathology. 2005;47:141–146. doi: 10.1111/j.1365-2559.2005.02176.x. [DOI] [PubMed] [Google Scholar]
- 111.Ajani JA, D’Amico TA, Bentrem DJ, Chao J, Cooke D, Corvera C, et al. Gastric cancer, version 2.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2022;20:167–192. doi: 10.6004/jnccn.2022.0008. [DOI] [PubMed] [Google Scholar]
- 112.Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687–697. doi: 10.1016/S0140-6736(10)61121-X. [DOI] [PubMed] [Google Scholar]
- 113.Bartley AN, Washington MK, Colasacco C, Ventura CB, Ismaila N, Benson AB, 3rd, et al. HER2 testing and clinical decision making in gastroesophageal adenocarcinoma: guideline from the College of American Pathologists, American Society for Clinical Pathology, and the American Society of Clinical Oncology. J Clin Oncol. 2017;35:446–464. doi: 10.1200/JCO.2016.69.4836. [DOI] [PubMed] [Google Scholar]
- 114.Lee HS, Kim WH, Kwak Y, Koh J, Bae JM, Kim KM, et al. Molecular testing for gastrointestinal cancer. J Pathol Transl Med. 2017;51:103–121. doi: 10.4132/jptm.2017.01.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ratti M, Lampis A, Hahne JC, Passalacqua R, Valeri N. Microsatellite instability in gastric cancer: molecular bases, clinical perspectives, and new treatment approaches. Cell Mol Life Sci. 2018;75:4151–4162. doi: 10.1007/s00018-018-2906-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Murphy KM, Zhang S, Geiger T, Hafez MJ, Bacher J, Berg KD, et al. Comparison of the microsatellite instability analysis system and the Bethesda panel for the determination of microsatellite instability in colorectal cancers. J Mol Diagn. 2006;8:305–311. doi: 10.2353/jmoldx.2006.050092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Luchini C, Bibeau F, Ligtenberg MJ, Singh N, Nottegar A, Bosse T, et al. ESMO recommendations on microsatellite instability testing for immunotherapy in cancer, and its relationship with PD-1/PD-L1 expression and tumour mutational burden: a systematic review-based approach. Ann Oncol. 2019;30:1232–1243. doi: 10.1093/annonc/mdz116. [DOI] [PubMed] [Google Scholar]
- 118.The Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202–209. doi: 10.1038/nature13480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Kim ST, Cristescu R, Bass AJ, Kim KM, Odegaard JI, Kim K, et al. Comprehensive molecular characterization of clinical responses to PD-1 inhibition in metastatic gastric cancer. Nat Med. 2018;24:1449–1458. doi: 10.1038/s41591-018-0101-z. [DOI] [PubMed] [Google Scholar]
- 120.Yoon CJ, Chang MS, Kim DH, Kim W, Koo BK, Yun SC, et al. Epstein-Barr virus-encoded miR-BART5-5p upregulates PD-L1 through PIAS3/pSTAT3 modulation, worsening clinical outcomes of PD-L1-positive gastric carcinomas. Gastric Cancer. 2020;23:780–795. doi: 10.1007/s10120-020-01059-3. [DOI] [PubMed] [Google Scholar]
- 121.Lee HS, Chang MS, Yang HK, Lee BL, Kim WH. Epstein-barr virus-positive gastric carcinoma has a distinct protein expression profile in comparison with epstein-barr virus-negative carcinoma. Clin Cancer Res. 2004;10:1698–1705. doi: 10.1158/1078-0432.ccr-1122-3. [DOI] [PubMed] [Google Scholar]
- 122.Kwak Y, Seo AN, Lee HE, Lee HS. Tumor immune response and immunotherapy in gastric cancer. J Pathol Transl Med. 2020;54:20–33. doi: 10.4132/jptm.2019.10.08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.DAKO Agilent Technologies. Interpretation Manual: Gastric or Gastroesophageal Junction Adenocarcinoma. PD-L1 IHC 22C3 PharmDx Interpretation Manual: Gastric or Gastroesophageal Junction Adenocarcinoma. Santa Clara (CA): DAKO Agilent Technologies; 2018. [Google Scholar]
- 124.Fuchs CS, Doi T, Jang RW, Muro K, Satoh T, Machado M, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 Clinical KEYNOTE-059 trial. JAMA Oncol. 2018;4:e180013. doi: 10.1001/jamaoncol.2018.0013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398:27–40. doi: 10.1016/S0140-6736(21)00797-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Catenacci DV, Rasco D, Lee J, Rha SY, Lee KW, Bang YJ, et al. Phase I Escalation and expansion study of bemarituzumab (FPA144) in patients with advanced solid tumors and FGFR2b-selected gastroesophageal adenocarcinoma. J Clin Oncol. 2020;38:2418–2426. doi: 10.1200/JCO.19.01834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Maron SB, Alpert L, Kwak HA, Lomnicki S, Chase L, Xu D, et al. Targeted therapies for targeted populations: anti-EGFR treatment for EGFR-amplified gastroesophageal adenocarcinoma. Cancer Discov. 2018;8:696–713. doi: 10.1158/2159-8290.CD-17-1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Lee J, Kim ST, Kim K, Lee H, Kozarewa I, Mortimer PG, et al. Tumor genomic profiling guides patients with metastatic gastric cancer to targeted treatment: the VIKTORY umbrella trial. Cancer Discov. 2019;9:1388–1405. doi: 10.1158/2159-8290.CD-19-0442. [DOI] [PubMed] [Google Scholar]
- 129.Smyth EC, Cafferkey C, Loehr A, Waddell T, Begum R, Peckitt C, et al. Genomic loss of heterozygosity and survival in the REAL3 trial. Oncotarget. 2018;9:36654–36665. doi: 10.18632/oncotarget.26336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Lee KW, Van Cutsem E, Bang YJ, Fuchs CS, Kudaba I, Garrido M, et al. Association of tumor mutational burden with efficacy of pembrolizumab±chemotherapy as first-line therapy for gastric cancer in the phase III KEYNOTE-062 study. Clin Cancer Res. 2022;28:3489–3498. doi: 10.1158/1078-0432.CCR-22-0121. [DOI] [PubMed] [Google Scholar]
- 131.Fancello L, Gandini S, Pelicci PG, Mazzarella L. Tumor mutational burden quantification from targeted gene panels: major advancements and challenges. J Immunother Cancer. 2019;7:183. doi: 10.1186/s40425-019-0647-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Ascierto PA, Bifulco C, Palmieri G, Peters S, Sidiropoulos N. Preanalytic variables and tissue stewardship for reliable next-generation sequencing (NGS) clinical analysis. J Mol Diagn. 2019;21:756–767. doi: 10.1016/j.jmoldx.2019.05.004. [DOI] [PubMed] [Google Scholar]
- 133.Kim WH, Park CK, Kim YB, Kim YW, Kim HG, Bae HI, et al. A standardized pathology report for gastric cancer. Korean J Pathol. 2005;39:106–113. [Google Scholar]
- 134.Park YS, Kook MC, Kim BH, Lee HS, Kang DW, Gu MJ, et al. A standardized pathology report for gastric cancer: 2nd edition. J Gastric Cancer. 2023;23:107–145. doi: 10.5230/jgc.2023.23.e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Chung IK, Lee JH, Lee SH, Kim SJ, Cho JY, Cho WY, et al. Therapeutic outcomes in 1000 cases of endoscopic submucosal dissection for early gastric neoplasms: Korean ESD Study Group multicenter study. Gastrointest Endosc. 2009;69:1228–1235. doi: 10.1016/j.gie.2008.09.027. [DOI] [PubMed] [Google Scholar]
- 136.Kim SG, Lyu DH, Park CM, Lee NR, Kim J, Cha Y, et al. Current status of endoscopic submucosal dissection for early gastric cancer in Korea: role and benefits. Korean J Intern Med. 2019;34:785–793. doi: 10.3904/kjim.2017.374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Gotoda T, Yanagisawa A, Sasako M, Ono H, Nakanishi Y, Shimoda T, et al. Incidence of lymph node metastasis from early gastric cancer: estimation with a large number of cases at two large centers. Gastric Cancer. 2000;3:219–225. doi: 10.1007/pl00011720. [DOI] [PubMed] [Google Scholar]
- 138.Nishizawa T, Yahagi N. Long-term outcomes of using endoscopic submucosal dissection to treat early gastric cancer. Gut Liver. 2018;12:119–124. doi: 10.5009/gnl17095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Hahn KY, Park CH, Lee YK, Chung H, Park JC, Shin SK, et al. Comparative study between endoscopic submucosal dissection and surgery in patients with early gastric cancer. Surg Endosc. 2018;32:73–86. doi: 10.1007/s00464-017-5640-8. [DOI] [PubMed] [Google Scholar]
- 140.Choi IJ, Lee JH, Kim YI, Kim CG, Cho SJ, Lee JY, et al. Long-term outcome comparison of endoscopic resection and surgery in early gastric cancer meeting the absolute indication for endoscopic resection. Gastrointest Endosc. 2015;81:333–41.e1. doi: 10.1016/j.gie.2014.07.047. [DOI] [PubMed] [Google Scholar]
- 141.Choi KS, Jung HY, Choi KD, Lee GH, Song HJ, Kim DH, et al. EMR versus gastrectomy for intramucosal gastric cancer: comparison of long-term outcomes. Gastrointest Endosc. 2011;73:942–948. doi: 10.1016/j.gie.2010.12.032. [DOI] [PubMed] [Google Scholar]
- 142.Kim DY, Hong SJ, Cho GS, Jeong GA, Kim HK, Han JP, et al. Long-term efficacy of endoscopic submucosal dissection compared with surgery for early gastric cancer: a retrospective cohort study. Gut Liver. 2014;8:519–525. doi: 10.5009/gnl13061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Fukase KM, Suzuki M, Toda H, Okuyama Y, Sakai J. Evaluation of the efficacy of endoscopic treatment for gastric cancer considered in terms of long-term prognosis. Dig Endosc. 1994;6:241–247. [Google Scholar]
- 144.Lee S, Choi KD, Han M, Na HK, Ahn JY, Jung KW, et al. Long-term outcomes of endoscopic submucosal dissection versus surgery in early gastric cancer meeting expanded indication including undifferentiated-type tumors: a criteria-based analysis. Gastric Cancer. 2018;21:490–499. doi: 10.1007/s10120-017-0772-z. [DOI] [PubMed] [Google Scholar]
- 145.Honda M, Hiki N, Kinoshita T, Yabusaki H, Abe T, Nunobe S, et al. Long-term outcomes of laparoscopic versus open surgery for clinical stage I gastric cancer: the LOC-1 study. Ann Surg. 2016;264:214–222. doi: 10.1097/SLA.0000000000001654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Kim HH, Han SU, Kim MC, Hyung WJ, Kim W, Lee HJ, et al. Long-term results of laparoscopic gastrectomy for gastric cancer: a large-scale case-control and case-matched Korean multicenter study. J Clin Oncol. 2014;32:627–633. doi: 10.1200/JCO.2013.48.8551. [DOI] [PubMed] [Google Scholar]
- 147.Kim W, Kim HH, Han SU, Kim MC, Hyung WJ, Ryu SW, et al. Decreased morbidity of laparoscopic distal gastrectomy compared with open distal gastrectomy for stage I gastric cancer: short-term outcomes from a multicenter randomized controlled trial (KLASS-01) Ann Surg. 2016;263:28–35. doi: 10.1097/SLA.0000000000001346. [DOI] [PubMed] [Google Scholar]
- 148.Lee S, Choi KD, Hong SM, Park SH, Gong EJ, Na HK, et al. Pattern of extragastric recurrence and the role of abdominal computed tomography in surveillance after endoscopic resection of early gastric cancer: Korean experiences. Gastric Cancer. 2017;20:843–852. doi: 10.1007/s10120-017-0691-z. [DOI] [PubMed] [Google Scholar]
- 149.Kim ER, Lee H, Min BH, Lee JH, Rhee PL, Kim JJ, et al. Effect of rescue surgery after non-curative endoscopic resection of early gastric cancer. Br J Surg. 2015;102:1394–1401. doi: 10.1002/bjs.9873. [DOI] [PubMed] [Google Scholar]
- 150.Oda I, Oyama T, Abe S, Ohnita K, Kosaka T, Hirasawa K, et al. Preliminary results of multicenter questionnaire study on long-term outcomes of curative endoscopic submucosal dissection for early gastric cancer. Dig Endosc. 2014;26:214–219. doi: 10.1111/den.12141. [DOI] [PubMed] [Google Scholar]
- 151.Kim HJ, Chung H, Shin SK, Kim HI, Park JC, Lee SK, et al. Comparison of long-term clinical outcomes between endoscopic and surgical resection for early-stage adenocarcinoma of the esophagogastric junction. Surg Endosc. 2018;32:3540–3547. doi: 10.1007/s00464-018-6076-5. [DOI] [PubMed] [Google Scholar]
- 152.Chang JY, Shim KN, Tae CH, Lee KE, Lee J, Lee KH, et al. Comparison of clinical outcomes after endoscopic submucosal dissection and surgery in the treatment of early gastric cancer: a single-institute study. Medicine (Baltimore) 2017;96:e7210. doi: 10.1097/MD.0000000000007210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Chiu PW, Teoh AY, To KF, Wong SK, Liu SY, Lam CC, et al. Endoscopic submucosal dissection (ESD) compared with gastrectomy for treatment of early gastric neoplasia: a retrospective cohort study. Surg Endosc. 2012;26:3584–3591. doi: 10.1007/s00464-012-2371-8. [DOI] [PubMed] [Google Scholar]
- 154.Cho JH, Cha SW, Kim HG, Lee TH, Cho JY, Ko WJ, et al. Long-term outcomes of endoscopic submucosal dissection for early gastric cancer: a comparison study to surgery using propensity score-matched analysis. Surg Endosc. 2016;30:3762–3773. doi: 10.1007/s00464-015-4672-1. [DOI] [PubMed] [Google Scholar]
- 155.Feng F, Sun L, Xu G, Cai L, Hong L, Yang J, et al. Is it reasonable to treat early gastric cancer with mucosal infiltration and well differentiation by endoscopic submucosal resection? J Gastrointest Surg. 2015;19:2111–2119. doi: 10.1007/s11605-015-2932-y. [DOI] [PubMed] [Google Scholar]
- 156.Fukunaga S, Nagami Y, Shiba M, Ominami M, Tanigawa T, Yamagami H, et al. Long-term prognosis of expanded-indication differentiated-type early gastric cancer treated with endoscopic submucosal dissection or surgery using propensity score analysis. Gastrointest Endosc. 2017;85:143–152. doi: 10.1016/j.gie.2016.06.049. [DOI] [PubMed] [Google Scholar]
- 157.Gong EJ, Kim DH, Ahn JY, Jung KW, Lee JH, Choi KD, et al. Comparison of long-term outcomes of endoscopic submucosal dissection and surgery for esophagogastric junction adenocarcinoma. Gastric Cancer. 2017;20(Suppl 1):84–91. doi: 10.1007/s10120-016-0679-0. [DOI] [PubMed] [Google Scholar]
- 158.Kim YI, Kim YW, Choi IJ, Kim CG, Lee JY, Cho SJ, et al. Long-term survival after endoscopic resection versus surgery in early gastric cancers. Endoscopy. 2015;47:293–301. doi: 10.1055/s-0034-1391284. [DOI] [PubMed] [Google Scholar]
- 159.Park CH, Lee H, Kim DW, Chung H, Park JC, Shin SK, et al. Clinical safety of endoscopic submucosal dissection compared with surgery in elderly patients with early gastric cancer: a propensity-matched analysis. Gastrointest Endosc. 2014;80:599–609. doi: 10.1016/j.gie.2014.04.042. [DOI] [PubMed] [Google Scholar]
- 160.Pyo JH, Lee H, Min BH, Lee JH, Choi MG, Lee JH, et al. Long-term outcome of endoscopic resection vs. surgery for early gastric cancer: a non-inferiority-matched cohort study. Am J Gastroenterol. 2016;111:240–249. doi: 10.1038/ajg.2015.427. [DOI] [PubMed] [Google Scholar]
- 161.Shin DW, Hwang HY, Jeon SW. Comparison of endoscopic submucosal dissection and surgery for differentiated type early gastric cancer within the expanded criteria. Clin Endosc. 2017;50:170–178. doi: 10.5946/ce.2016.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Eom BW, Kim YI, Kim KH, Yoon HM, Cho SJ, Lee JY, et al. Survival benefit of additional surgery after noncurative endoscopic resection in patients with early gastric cancer. Gastrointest Endosc. 2017;85:155–163.e3. doi: 10.1016/j.gie.2016.07.036. [DOI] [PubMed] [Google Scholar]
- 163.Hatta W, Gotoda T, Oyama T, Kawata N, Takahashi A, Yoshifuku Y, et al. Is radical surgery necessary in all patients who do not meet the curative criteria for endoscopic submucosal dissection in early gastric cancer? A multi-center retrospective study in Japan. J Gastroenterol. 2017;52:175–184. doi: 10.1007/s00535-016-1210-4. [DOI] [PubMed] [Google Scholar]
- 164.Kusano C, Iwasaki M, Kaltenbach T, Conlin A, Oda I, Gotoda T. Should elderly patients undergo additional surgery after non-curative endoscopic resection for early gastric cancer? Long-term comparative outcomes. Am J Gastroenterol. 2011;106:1064–1069. doi: 10.1038/ajg.2011.49. [DOI] [PubMed] [Google Scholar]
- 165.Hirasawa T, Gotoda T, Miyata S, Kato Y, Shimoda T, Taniguchi H, et al. Incidence of lymph node metastasis and the feasibility of endoscopic resection for undifferentiated-type early gastric cancer. Gastric Cancer. 2009;12:148–152. doi: 10.1007/s10120-009-0515-x. [DOI] [PubMed] [Google Scholar]
- 166.Chung JW, Jung HY, Choi KD, Song HJ, Lee GH, Jang SJ, et al. Extended indication of endoscopic resection for mucosal early gastric cancer: analysis of a single center experience. J Gastroenterol Hepatol. 2011;26:884–887. doi: 10.1111/j.1440-1746.2010.06611.x. [DOI] [PubMed] [Google Scholar]
- 167.Oh SY, Lee KG, Suh YS, Kim MA, Kong SH, Lee HJ, et al. Lymph node metastasis in mucosal gastric cancer: reappraisal of expanded indication of endoscopic submucosal dissection. Ann Surg. 2017;265:137–142. doi: 10.1097/SLA.0000000000001649. [DOI] [PubMed] [Google Scholar]
- 168.Abdelfatah MM, Barakat M, Lee H, Kim JJ, Uedo N, Grimm I, et al. The incidence of lymph node metastasis in early gastric cancer according to the expanded criteria in comparison with the absolute criteria of the Japanese Gastric Cancer Association: a systematic review of the literature and meta-analysis. Gastrointest Endosc. 2018;87:338–347. doi: 10.1016/j.gie.2017.09.025. [DOI] [PubMed] [Google Scholar]
- 169.Huh CW, Ma DW, Kim BW, Kim JS, Lee SJ. Endoscopic submucosal dissection versus surgery for undifferentiated-type early gastric cancer: a systematic review and meta-analysis. Clin Endosc. 2021;54:202–210. doi: 10.5946/ce.2020.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Lee GH, Lee E, Park B, Roh J, Lim SG, Shin SJ, et al. Long-term outcomes of endoscopic submucosal dissection and surgery for undifferentiated intramucosal gastric cancer regardless of size. World J Gastroenterol. 2022;28:840–852. doi: 10.3748/wjg.v28.i8.840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Lim JH, Kim J, Kim SG, Chung H. Long-term clinical outcomes of endoscopic vs. surgical resection for early gastric cancer with undifferentiated histology. Surg Endosc. 2019;33:3589–3599. doi: 10.1007/s00464-018-06641-6. [DOI] [PubMed] [Google Scholar]
- 172.Takizawa K, Ono H, Hasuike N, Takashima A, Minashi K, Boku N, et al. A nonrandomized, single-arm confirmatory trial of expanded endoscopic submucosal dissection indication for undifferentiated early gastric cancer: Japan Clinical Oncology Group study (JCOG1009/1010) Gastric Cancer. 2021;24:479–491. doi: 10.1007/s10120-020-01134-9. [DOI] [PubMed] [Google Scholar]
- 173.Choi JY, Jeon SW, Cho KB, Park KS, Kim ES, Park CK, et al. Non-curative endoscopic resection does not always lead to grave outcomes in submucosal invasive early gastric cancer. Surg Endosc. 2015;29:1842–1849. doi: 10.1007/s00464-014-3874-2. [DOI] [PubMed] [Google Scholar]
- 174.Hoteya S, Iizuka T, Kikuchi D, Ogawa O, Mitani T, Matsui A, et al. Clinicopathological outcomes of patients with early gastric cancer after non-curative endoscopic submucosal dissection. Digestion. 2016;93:53–58. doi: 10.1159/000441758. [DOI] [PubMed] [Google Scholar]
- 175.Yamanouchi K, Ogata S, Sakata Y, Tsuruoka N, Shimoda R, Nakayama A, et al. Effect of additional surgery after noncurative endoscopic submucosal dissection for early gastric cancer. Endosc Int Open. 2016;4:E24–E29. doi: 10.1055/s-0034-1393124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Kawata N, Kakushima N, Takizawa K, Tanaka M, Makuuchi R, Tokunaga M, et al. Risk factors for lymph node metastasis and long-term outcomes of patients with early gastric cancer after non-curative endoscopic submucosal dissection. Surg Endosc. 2017;31:1607–1616. doi: 10.1007/s00464-016-5148-7. [DOI] [PubMed] [Google Scholar]
- 177.Kikuchi S, Kuroda S, Nishizaki M, Kagawa T, Kanzaki H, Kawahara Y, et al. Management of early gastric cancer that meet the indication for radical lymph node dissection following endoscopic resection: a retrospective cohort analysis. BMC Surg. 2017;17:72. doi: 10.1186/s12893-017-0268-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Pyo JH, Lee H, Min BH, Lee JH, Kim KM, Yoo H, et al. Comparison of long-term outcomes after non-curative endoscopic resection in older patients with early gastric cancer. Ann Surg Oncol. 2017;24:2624–2631. doi: 10.1245/s10434-017-5888-1. [DOI] [PubMed] [Google Scholar]
- 179.Sumiyoshi T, Kondo H, Fujii R, Minagawa T, Fujie S, Kimura T, et al. Short- and long-term outcomes of endoscopic submucosal dissection for early gastric cancer in elderly patients aged 75 years and older. Gastric Cancer. 2017;20:489–495. doi: 10.1007/s10120-016-0628-y. [DOI] [PubMed] [Google Scholar]
- 180.Suzuki S, Gotoda T, Hatta W, Oyama T, Kawata N, Takahashi A, et al. Survival benefit of additional surgery after non-curative endoscopic submucosal dissection for early gastric cancer: a propensity score matching analysis. Ann Surg Oncol. 2017;24:3353–3360. doi: 10.1245/s10434-017-6039-4. [DOI] [PubMed] [Google Scholar]
- 181.Toya Y, Endo M, Nakamura S, Akasaka R, Kosaka T, Yanai S, et al. Clinical outcomes of non-curative endoscopic submucosal dissection with negative resected margins for gastric cancer. Gastrointest Endosc. 2017;85:1218–1224. doi: 10.1016/j.gie.2016.11.018. [DOI] [PubMed] [Google Scholar]
- 182.Jeon MY, Park JC, Hahn KY, Shin SK, Lee SK, Lee YC. Long-term outcomes after noncurative endoscopic resection of early gastric cancer: the optimal time for additional endoscopic treatment. Gastrointest Endosc. 2018;87:1003–1013.e2. doi: 10.1016/j.gie.2017.10.004. [DOI] [PubMed] [Google Scholar]
- 183.Yano T, Ishido K, Tanabe S, Wada T, Azuma M, Kawanishi N, et al. Long-term outcomes of patients with early gastric cancer found to have lesions for which endoscopic treatment is not indicated on histopathological evaluation after endoscopic submucosal dissection. Surg Endosc. 2018;32:1314–1323. doi: 10.1007/s00464-017-5809-1. [DOI] [PubMed] [Google Scholar]
- 184.Kim HJ, Kim SG, Kim J, Hong H, Lee HJ, Kim MS, et al. Clinical outcomes of early gastric cancer with non-curative resection after pathological evaluation based on the expanded criteria. PLoS One. 2019;14:e0224614. doi: 10.1371/journal.pone.0224614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Iwai N, Dohi O, Naito Y, Inada Y, Inoue K, Okayama T, et al. High-risk comorbidity influences prognosis in early gastric cancer after noncurative endoscopic submucosal dissection: a retrospective study. Dig Dis. 2021;39:96–105. doi: 10.1159/000510115. [DOI] [PubMed] [Google Scholar]
- 186.Min SH, Son SY, Jung DH, Lee CM, Ahn SH, Park DJ, et al. Laparoscopic gastrojejunostomy versus duodenal stenting in unresectable gastric cancer with gastric outlet obstruction. Ann Surg Treat Res. 2017;93:130–136. doi: 10.4174/astr.2017.93.3.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Sekiguchi M, Suzuki H, Oda I, Abe S, Nonaka S, Yoshinaga S, et al. Risk of recurrent gastric cancer after endoscopic resection with a positive lateral margin. Endoscopy. 2014;46:273–278. doi: 10.1055/s-0034-1364938. [DOI] [PubMed] [Google Scholar]
- 188.Hwang JJ, Park KJ, Park YS, Lee HS, Yoon H, Shin CM, et al. A scoring system for patients with a tumor-positive lateral resection margin after endoscopic resection of early gastric cancer. Surg Endosc. 2016;30:2751–2758. doi: 10.1007/s00464-015-4543-9. [DOI] [PubMed] [Google Scholar]
- 189.Kikuchi D, Iizuka T, Hoteya S, Yamada A, Furuhata T, Yamashita S, et al. Safety and efficacy of secondary endoscopic submucosal dissection for residual gastric carcinoma after primary endoscopic submucosal dissection. Digestion. 2012;86:288–293. doi: 10.1159/000342114. [DOI] [PubMed] [Google Scholar]
- 190.Kim HW, Kim JH, Park JC, Jeon MY, Lee YC, Lee SK, et al. Additive endoscopic resection may be sufficient for patients with a positive lateral margin after endoscopic resection of early gastric cancer. Gastrointest Endosc. 2017;86:849–856. doi: 10.1016/j.gie.2017.02.037. [DOI] [PubMed] [Google Scholar]
- 191.Kim TS, Min BH, Min YW, Lee H, Rhee PL, Kim JJ, et al. Long-term outcomes of additional endoscopic treatments for patients with positive lateral margins after endoscopic submucosal dissection for early gastric cancer. Gut Liver. 2022;16:547–554. doi: 10.5009/gnl210203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Oda I, Gotoda T, Sasako M, Sano T, Katai H, Fukagawa T, et al. Treatment strategy after non-curative endoscopic resection of early gastric cancer. Br J Surg. 2008;95:1495–1500. doi: 10.1002/bjs.6305. [DOI] [PubMed] [Google Scholar]
- 193.Lee JH, Lee JH, Kim KM, Kang KJ, Min BH, Kim JJ. Clinicopathological factors of multiple lateral margin involvement after endoscopic submucosal dissection for early gastric cancer. Surg Endosc. 2015;29:3460–3468. doi: 10.1007/s00464-015-4095-z. [DOI] [PubMed] [Google Scholar]
- 194.Yang HJ, Lee WS, Lee BE, Ahn JY, Jang JY, Lim JH, et al. Long-term outcomes of undifferentiated-type early gastric cancer with positive horizontal margins after endoscopic resection. Gut Liver. 2021;15:723–731. doi: 10.5009/gnl20291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Ahn S, Do IG, Sohn JH, Yang HJ, Yoo CH, Kim K. Total length of positive resection margins can predict remnant gastric cancer following endoscopic submucosal dissection. Pathol Res Pract. 2020;216:153183. doi: 10.1016/j.prp.2020.153183. [DOI] [PubMed] [Google Scholar]
- 196.Ford AC, Yuan Y, Forman D, Hunt R, Moayyedi P. Helicobacter pylori eradication for the prevention of gastric neoplasia. Cochrane Database Syst Rev. 2020;7:CD005583. doi: 10.1002/14651858.CD005583.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, et al. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:392–397. doi: 10.1016/S0140-6736(08)61159-9. [DOI] [PubMed] [Google Scholar]
- 198.Choi IJ, Kook MC, Kim YI, Cho SJ, Lee JY, Kim CG, et al. Helicobacter pylori therapy for the prevention of metachronous gastric cancer. N Engl J Med. 2018;378:1085–1095. doi: 10.1056/NEJMoa1708423. [DOI] [PubMed] [Google Scholar]
- 199.Smith JW, Brennan MF. Surgical treatment of gastric cancer. Proximal, mid, and distal stomach. Surg Clin North Am. 1992;72:381–399. doi: 10.1016/s0039-6109(16)45685-9. [DOI] [PubMed] [Google Scholar]
- 200.Lee MS, Ahn SH, Lee JH, Park DJ, Lee HJ, Kim HH, et al. What is the best reconstruction method after distal gastrectomy for gastric cancer? Surg Endosc. 2012;26:1539–1547. doi: 10.1007/s00464-011-2064-8. [DOI] [PubMed] [Google Scholar]
- 201.Ren Z, Wang WX. Comparison of Billroth I, Billroth II, and Roux-en-Y reconstruction after totally laparoscopic distal gastrectomy: a randomized controlled study. Adv Ther. 2019;36:2997–3006. doi: 10.1007/s12325-019-01104-3. [DOI] [PubMed] [Google Scholar]
- 202.Ishikawa M, Kitayama J, Kaizaki S, Nakayama H, Ishigami H, Fujii S, et al. Prospective randomized trial comparing Billroth I and Roux-en-Y procedures after distal gastrectomy for gastric carcinoma. World J Surg. 2005;29:1415–1420. doi: 10.1007/s00268-005-7830-0. [DOI] [PubMed] [Google Scholar]
- 203.Hirao M, Takiguchi S, Imamura H, Yamamoto K, Kurokawa Y, Fujita J, et al. Comparison of Billroth I and Roux-en-Y reconstruction after distal gastrectomy for gastric cancer: one-year postoperative effects assessed by a multi-institutional RCT. Ann Surg Oncol. 2013;20:1591–1597. doi: 10.1245/s10434-012-2704-9. [DOI] [PubMed] [Google Scholar]
- 204.Nakamura M, Nakamori M, Ojima T, Iwahashi M, Horiuchi T, Kobayashi Y, et al. Randomized clinical trial comparing long-term quality of life for Billroth I versus Roux-en-Y reconstruction after distal gastrectomy for gastric cancer. Br J Surg. 2016;103:337–347. doi: 10.1002/bjs.10060. [DOI] [PubMed] [Google Scholar]
- 205.Yang K, Zhang WH, Liu K, Chen XZ, Zhou ZG, Hu JK. Comparison of quality of life between Billroth-I and Roux-en-Y anastomosis after distal gastrectomy for gastric cancer: a randomized controlled trial. Sci Rep. 2017;7:11245. doi: 10.1038/s41598-017-09676-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Kim SY, Bang WJ, Lim H, Lim MS, Kim M, Choi HG. Increased risk of gallstones after gastrectomy: a longitudinal follow-up study using a national sample cohort in Korea. Medicine (Baltimore) 2019;98:e15932. doi: 10.1097/MD.0000000000015932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Park DJ, Kim KH, Park YS, Ahn SH, Park J, Kim HH. Risk factors for gallstone formation after surgery for gastric cancer. J Gastric Cancer. 2016;16:98–104. doi: 10.5230/jgc.2016.16.2.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Kim JH, Huh YJ, Park S, Park YS, Park DJ, Kwon JW, et al. Multicenter results of long-limb bypass reconstruction after gastrectomy in patients with gastric cancer and type II diabetes. Asian J Surg. 2020;43:297–303. doi: 10.1016/j.asjsur.2019.03.018. [DOI] [PubMed] [Google Scholar]
- 209.Toyomasu Y, Ogata K, Suzuki M, Yanoma T, Kimura A, Kogure N, et al. Comparison of the physiological effect of Billroth-I and Roux-en-Y reconstruction following laparoscopic distal gastrectomy. Surg Laparosc Endosc Percutan Tech. 2018;28:328–333. doi: 10.1097/SLE.0000000000000575. [DOI] [PubMed] [Google Scholar]
- 210.So JB, Rao J, Wong AS, Chan YH, Pang NQ, Tay AY, et al. Roux-en-Y or Billroth II reconstruction after radical distal gastrectomy for gastric cancer: a multicenter randomized controlled trial. Ann Surg. 2018;267:236–242. doi: 10.1097/SLA.0000000000002229. [DOI] [PubMed] [Google Scholar]
- 211.Choi YS, Yi JW, Shin WY, Heo Y. Oncometabolic surgery in gastric cancer patients with type 2 diabetes. Sci Rep. 2022;12:11853. doi: 10.1038/s41598-022-15404-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Inokuchi M, Kojima K, Yamada H, Kato K, Hayashi M, Motoyama K, et al. Long-term outcomes of Roux-en-Y and Billroth-I reconstruction after laparoscopic distal gastrectomy. Gastric Cancer. 2013;16:67–73. doi: 10.1007/s10120-012-0154-5. [DOI] [PubMed] [Google Scholar]
- 213.Tanigawa N, Nomura E, Lee SW, Kaminishi M, Sugiyama M, Aikou T, et al. Current state of gastric stump carcinoma in Japan: based on the results of a nationwide survey. World J Surg. 2010;34:1540–1547. doi: 10.1007/s00268-010-0505-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Shin D, Park SS. Clinical importance and surgical decision-making regarding proximal resection margin for gastric cancer. World J Gastrointest Oncol. 2013;5:4–11. doi: 10.4251/wjgo.v5.i1.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Sun Z, Li DM, Wang ZN, Huang BJ, Xu Y, Li K, et al. Prognostic significance of microscopic positive margins for gastric cancer patients with potentially curative resection. Ann Surg Oncol. 2009;16:3028–3037. doi: 10.1245/s10434-009-0624-0. [DOI] [PubMed] [Google Scholar]
- 216.Nagata T, Ichikawa D, Komatsu S, Inoue K, Shiozaki A, Fujiwara H, et al. Prognostic impact of microscopic positive margin in gastric cancer patients. J Surg Oncol. 2011;104:592–597. doi: 10.1002/jso.22022. [DOI] [PubMed] [Google Scholar]
- 217.Morgagni P, La Barba G, Colciago E, Vittimberga G, Ercolani G. Resection line involvement after gastric cancer treatment: handle with care. Updates Surg. 2018;70:213–223. doi: 10.1007/s13304-018-0552-2. [DOI] [PubMed] [Google Scholar]
- 218.Morgagni P, Garcea D, Marrelli D, De Manzoni G, Natalini G, Kurihara H, et al. Resection line involvement after gastric cancer surgery: clinical outcome in nonsurgically retreated patients. World J Surg. 2008;32:2661–2667. doi: 10.1007/s00268-008-9747-x. [DOI] [PubMed] [Google Scholar]
- 219.Cho BC, Jeung HC, Choi HJ, Rha SY, Hyung WJ, Cheong JH, et al. Prognostic impact of resection margin involvement after extended (D2/D3) gastrectomy for advanced gastric cancer: a 15-year experience at a single institute. J Surg Oncol. 2007;95:461–468. doi: 10.1002/jso.20731. [DOI] [PubMed] [Google Scholar]
- 220.Lee CM, Jee YS, Lee JH, Son SY, Ahn SH, Park DJ, et al. Length of negative resection margin does not affect local recurrence and survival in the patients with gastric cancer. World J Gastroenterol. 2014;20:10518–10524. doi: 10.3748/wjg.v20.i30.10518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Cascinu S, Giordani P, Catalano V, Agostinelli R, Catalano G. Resection-line involvement in gastric cancer patients undergoing curative resections: implications for clinical management. Jpn J Clin Oncol. 1999;29:291–293. doi: 10.1093/jjco/29.6.291. [DOI] [PubMed] [Google Scholar]
- 222.Raziee HR, Cardoso R, Seevaratnam R, Mahar A, Helyer L, Law C, et al. Systematic review of the predictors of positive margins in gastric cancer surgery and the effect on survival. Gastric Cancer. 2012;15(Suppl 1):S116–S124. doi: 10.1007/s10120-011-0112-7. [DOI] [PubMed] [Google Scholar]
- 223.Park SH, Lee HJ, Park JH, Kim TH, Son YG, Huh YJ, et al. Clinical significance of intra-operative gastroscopy for tumor localization in totally laparoscopic partial gastrectomy. J Gastrointest Surg. 2021;25:1134–1146. doi: 10.1007/s11605-020-04809-x. [DOI] [PubMed] [Google Scholar]
- 224.Choi Y, Kim KG, Kim JK, Nam KW, Kim HH, Sohn DK. A novel endoscopic fluorescent clip for the localization of gastrointestinal tumors. Surg Endosc. 2011;25:2372–2377. doi: 10.1007/s00464-010-1557-1. [DOI] [PubMed] [Google Scholar]
- 225.Jeong O, Cho SB, Joo YE, Ryu SY, Park YK. Novel technique for intraoperative tumor localization during totally laparoscopic distal gastrectomy: endoscopic autologous blood tattooing. Surg Endosc. 2012;26:1778–1783. doi: 10.1007/s00464-011-2067-5. [DOI] [PubMed] [Google Scholar]
- 226.Jeong SH, Bae K, Ha CY, Lee YJ, Lee OJ, Jung WT, et al. Effectiveness of endoscopic clipping and computed tomography gastroscopy for the preoperative localization of gastric cancer. J Korean Surg Soc. 2013;84:80–87. doi: 10.4174/jkss.2013.84.2.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Shen JG, Cheong JH, Hyung WJ, Kim J, Choi SH, Noh SH. Intraoperative frozen section margin evaluation in gastric cancer of the cardia surgery. Hepatogastroenterology. 2006;53:976–978. [PubMed] [Google Scholar]
- 228.Hyung WJ, Lim JS, Cheong JH, Kim J, Choi SH, Song SY, et al. Intraoperative tumor localization using laparoscopic ultrasonography in laparoscopic-assisted gastrectomy. Surg Endosc. 2005;19:1353–1357. doi: 10.1007/s00464-004-8196-3. [DOI] [PubMed] [Google Scholar]
- 229.Park DJ, Lee HJ, Kim SG, Jung HC, Song IS, Lee KU, et al. Intraoperative gastroscopy for gastric surgery. Surg Endosc. 2005;19:1358–1361. doi: 10.1007/s00464-004-2217-0. [DOI] [PubMed] [Google Scholar]
- 230.Berlth F, Kim WH, Choi JH, Park SH, Kong SH, Lee HJ, et al. Prognostic impact of frozen section investigation and extent of proximal safety margin in gastric cancer resection. Ann Surg. 2020;272:871–878. doi: 10.1097/SLA.0000000000004266. [DOI] [PubMed] [Google Scholar]
- 231.Park JH, Jeong SH, Lee YJ, Kim TH, Kim JM, Kim DH, et al. Safety and efficacy of post-anastomotic intraoperative endoscopy to avoid early anastomotic complications during gastrectomy for gastric cancer. Surg Endosc. 2020;34:5312–5319. doi: 10.1007/s00464-019-07319-3. [DOI] [PubMed] [Google Scholar]
- 232.Wang SY, Yeh CN, Lee HL, Liu YY, Chao TC, Hwang TL, et al. Clinical impact of positive surgical margin status on gastric cancer patients undergoing gastrectomy. Ann Surg Oncol. 2009;16:2738–2743. doi: 10.1245/s10434-009-0616-0. [DOI] [PubMed] [Google Scholar]
- 233.Bozzetti F, Bonfanti G, Bufalino R, Menotti V, Persano S, Andreola S, et al. Adequacy of margins of resection in gastrectomy for cancer. Ann Surg. 1982;196:685–690. doi: 10.1097/00000658-198212001-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2021 (6th edition) Gastric Cancer. 2023;26:1–25. doi: 10.1007/s10120-022-01331-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Squires MH, 3rd, Kooby DA, Pawlik TM, Weber SM, Poultsides G, Schmidt C, et al. Utility of the proximal margin frozen section for resection of gastric adenocarcinoma: a 7-Institution Study of the US Gastric Cancer Collaborative. Ann Surg Oncol. 2014;21:4202–4210. doi: 10.1245/s10434-014-3834-z. [DOI] [PubMed] [Google Scholar]
- 236.Nakanishi K, Morita S, Taniguchi H, Otsuki S, Fukagawa T, Katai H. Diagnostic accuracy and usefulness of intraoperative margin assessment by frozen section in gastric cancer. Ann Surg Oncol. 2019;26:1787–1794. doi: 10.1245/s10434-019-07302-1. [DOI] [PubMed] [Google Scholar]
- 237.Spicer J, Benay C, Lee L, Rousseau M, Andalib A, Kushner Y, et al. Diagnostic accuracy and utility of intraoperative microscopic margin analysis of gastric and esophageal adenocarcinoma. Ann Surg Oncol. 2014;21:2580–2586. doi: 10.1245/s10434-014-3669-7. [DOI] [PubMed] [Google Scholar]
- 238.Kim SH, Karpeh MS, Klimstra DS, Leung D, Brennan MF. Effect of microscopic resection line disease on gastric cancer survival. J Gastrointest Surg. 1999;3:24–33. doi: 10.1016/s1091-255x(99)80004-3. [DOI] [PubMed] [Google Scholar]
- 239.Shen JG, Cheong JH, Hyung WJ, Kim J, Choi SH, Noh SH. Influence of a microscopic positive proximal margin in the treatment of gastric adenocarcinoma of the cardia. World J Gastroenterol. 2006;12:3883–3886. doi: 10.3748/wjg.v12.i24.3883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Bickenbach KA, Gonen M, Strong V, Brennan MF, Coit DG. Association of positive transection margins with gastric cancer survival and local recurrence. Ann Surg Oncol. 2013;20:2663–2668. doi: 10.1245/s10434-013-2950-5. [DOI] [PubMed] [Google Scholar]
- 241.Liang Y, Ding X, Wang X, Wang B, Deng J, Zhang L, et al. Prognostic value of surgical margin status in gastric cancer patients. ANZ J Surg. 2015;85:678–684. doi: 10.1111/ans.12515. [DOI] [PubMed] [Google Scholar]
- 242.Chen JD, Yang XP, Shen JG, Hu WX, Yuan XM, Wang LB. Prognostic improvement of reexcision for positive resection margins in patients with advanced gastric cancer. Eur J Surg Oncol. 2013;39:229–234. doi: 10.1016/j.ejso.2012.08.004. [DOI] [PubMed] [Google Scholar]
- 243.Lee S, Son WJ, Roh YH, Song JH, Park SH, Cho M, et al. Indication of proximal gastrectomy for advanced proximal gastric cancer based on lymph node metastasis at the distal part of the stomach. Ann Surg Open. 2021;2:e107. doi: 10.1097/AS9.0000000000000107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Kong SH, Kim JW, Lee HJ, Kim WH, Lee KU, Yang HK. Reverse double-stapling end-to-end esophagogastrostomy in proximal gastrectomy. Dig Surg. 2010;27:170–174. doi: 10.1159/000264658. [DOI] [PubMed] [Google Scholar]
- 245.Cho M, Son T, Kim HI, Noh SH, Choi S, Seo WJ, et al. Similar hematologic and nutritional outcomes after proximal gastrectomy with double-tract reconstruction in comparison to total gastrectomy for early upper gastric cancer. Surg Endosc. 2019;33:1757–1768. doi: 10.1007/s00464-018-6448-x. [DOI] [PubMed] [Google Scholar]
- 246.Jung DH, Lee Y, Kim DW, Park YS, Ahn SH, Park DJ, et al. Laparoscopic proximal gastrectomy with double tract reconstruction is superior to laparoscopic total gastrectomy for proximal early gastric cancer. Surg Endosc. 2017;31:3961–3969. doi: 10.1007/s00464-017-5429-9. [DOI] [PubMed] [Google Scholar]
- 247.Sugiyama M, Oki E, Ando K, Nakashima Y, Saeki H, Maehara Y. Laparoscopic proximal gastrectomy maintains body weight and skeletal muscle better than total gastrectomy. World J Surg. 2018;42:3270–3276. doi: 10.1007/s00268-018-4625-7. [DOI] [PubMed] [Google Scholar]
- 248.Park JY, Park KB, Kwon OK, Yu W. Comparison of laparoscopic proximal gastrectomy with double-tract reconstruction and laparoscopic total gastrectomy in terms of nutritional status or quality of life in early gastric cancer patients. Eur J Surg Oncol. 2018;44:1963–1970. doi: 10.1016/j.ejso.2018.08.014. [DOI] [PubMed] [Google Scholar]
- 249.Kim DJ, Kim W. Laparoscopy-assisted proximal gastrectomy with double tract anastomosis is beneficial for vitamin B12 and iron absorption. Anticancer Res. 2016;36:4753–4758. doi: 10.21873/anticanres.11031. [DOI] [PubMed] [Google Scholar]
- 250.Ko HJ, Kim KH, Lee SH, Choi CW, Kim SJ, In Choi C, et al. Can proximal gastrectomy with double-tract reconstruction replace total gastrectomy? A propensity score matching analysis. J Gastrointest Surg. 2020;24:516–524. doi: 10.1007/s11605-019-04195-z. [DOI] [PubMed] [Google Scholar]
- 251.Park DJ, Han SU, Hyung WJ, et al. Effect of laparoscopic proximal gastrectomy and double-tract reconstruction vs total gastrectomy on hemoglobin level and vitamin B12 supplementation in upper-third early gastric cancer: a KLASS 05 randomized clinical trial. JAMA Netw Open. 2023;6:e2256004. doi: 10.1001/jamanetworkopen.2022.56004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Hayami M, Hiki N, Nunobe S, Mine S, Ohashi M, Kumagai K, et al. Clinical outcomes and evaluation of laparoscopic proximal gastrectomy with double-flap technique for early gastric cancer in the upper third of the stomach. Ann Surg Oncol. 2017;24:1635–1642. doi: 10.1245/s10434-017-5782-x. [DOI] [PubMed] [Google Scholar]
- 253.Shoji Y, Nunobe S, Ida S, Kumagai K, Ohashi M, Sano T, et al. Surgical outcomes and risk assessment for anastomotic complications after laparoscopic proximal gastrectomy with double-flap technique for upper-third gastric cancer. Gastric Cancer. 2019;22:1036–1043. doi: 10.1007/s10120-019-00940-0. [DOI] [PubMed] [Google Scholar]
- 254.Yamashita Y, Yamamoto A, Tamamori Y, Yoshii M, Nishiguchi Y. Side overlap esophagogastrostomy to prevent reflux after proximal gastrectomy. Gastric Cancer. 2017;20:728–735. doi: 10.1007/s10120-016-0674-5. [DOI] [PubMed] [Google Scholar]
- 255.Yamashita Y, Tatsubayashi T, Okumura K, Miyamoto T, Ueno K. Modified side overlap esophagogastrostomy after laparoscopic proximal gastrectomy. Ann Gastroenterol Surg. 2022;6:594–599. doi: 10.1002/ags3.12549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Lee HJ, Kim YW, Park DJ, Han SU, Ryu KW, Kim HH, et al. Laparoscopic PPG versus DG for middle third EGC; final result of multicenter RCT (KLASS-04); International Gastric Cancer Congress 2022; 2022 Mar 6-9; Houston (TX). Tokyo: International Gastric Cancer Association; 2022. [Google Scholar]
- 257.Park DJ, Kim YW, Yang HK, Ryu KW, Han SU, Kim HH, et al. Short-term outcomes of a multicentre randomized clinical trial comparing laparoscopic pylorus-preserving gastrectomy with laparoscopic distal gastrectomy for gastric cancer (the KLASS-04 trial) Br J Surg. 2021;108:1043–1049. doi: 10.1093/bjs/znab295. [DOI] [PubMed] [Google Scholar]
- 258.Suh YS, Han DS, Kong SH, Kwon S, Shin CI, Kim WH, et al. Laparoscopy-assisted pylorus-preserving gastrectomy is better than laparoscopy-assisted distal gastrectomy for middle-third early gastric cancer. Ann Surg. 2014;259:485–493. doi: 10.1097/SLA.0b013e318294d142. [DOI] [PubMed] [Google Scholar]
- 259.Ikeguchi M, Hatada T, Yamamoto M, Miyake T, Matsunaga T, Fukuda K, et al. Evaluation of a pylorus-preserving gastrectomy for patients preoperatively diagnosed with early gastric cancer located in the middle third of the stomach. Surg Today. 2010;40:228–233. doi: 10.1007/s00595-009-4043-4. [DOI] [PubMed] [Google Scholar]
- 260.Xiao XM, Gaol C, Yin W, Yu WH, Qi F, Liu T. Pylorus-preserving versus distal subtotal gastrectomy for surgical treatment of early gastric cancer: a meta-analysis. Hepatogastroenterology. 2014;61:870–879. [PubMed] [Google Scholar]
- 261.Xia X, Xu J, Zhu C, Cao H, Yu F, Zhao G. Objective evaluation of clinical outcomes of laparoscopy-assisted pylorus-preserving gastrectomy for middle-third early gastric cancer. BMC Cancer. 2019;19:481. doi: 10.1186/s12885-019-5695-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Eom BW, Park B, Yoon HM, Ryu KW, Kim YW. Laparoscopy-assisted pylorus-preserving gastrectomy for early gastric cancer: a retrospective study of long-term functional outcomes and quality of life. World J Gastroenterol. 2019;25:5494–5504. doi: 10.3748/wjg.v25.i36.5494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Nakane Y, Akehira K, Inoue K, Iiyama H, Sato M, Masuya Y, et al. Postoperative evaluation of pylorus-preserving gastrectomy for early gastric cancer. Hepatogastroenterology. 2000;47:590–595. [PubMed] [Google Scholar]
- 264.Huang C, Yu F, Zhao G, Xia X. Postoperative quality of life after laparoscopy-assisted pylorus-preserving gastrectomy compared with laparoscopy-assisted distal gastrectomy for early gastric cancer. J Gastroenterol Hepatol. 2020;35:1712–1719. doi: 10.1111/jgh.14985. [DOI] [PubMed] [Google Scholar]
- 265.Csendes A, Burdiles P, Rojas J, Braghetto I, Diaz JC, Maluenda F. A prospective randomized study comparing D2 total gastrectomy versus D2 total gastrectomy plus splenectomy in 187 patients with gastric carcinoma. Surgery. 2002;131:401–407. doi: 10.1067/msy.2002.121891. [DOI] [PubMed] [Google Scholar]
- 266.Yu W, Choi GS, Chung HY. Randomized clinical trial of splenectomy versus splenic preservation in patients with proximal gastric cancer. Br J Surg. 2006;93:559–563. doi: 10.1002/bjs.5353. [DOI] [PubMed] [Google Scholar]
- 267.Hayashi T, Yoshikawa T, Kamiya A, Date K, Wada T, Otsuki S, et al. Is splenectomy for dissecting splenic hilar lymph nodes justified for scirrhous gastric cancer? Gastric Cancer. 2020;23:922–926. doi: 10.1007/s10120-020-01063-7. [DOI] [PubMed] [Google Scholar]
- 268.Sasako M, McCulloch P, Kinoshita T, Maruyama K. New method to evaluate the therapeutic value of lymph node dissection for gastric cancer. Br J Surg. 1995;82:346–351. doi: 10.1002/bjs.1800820321. [DOI] [PubMed] [Google Scholar]
- 269.Kosuga T, Ichikawa D, Okamoto K, Komatsu S, Shiozaki A, Fujiwara H, et al. Survival benefits from splenic hilar lymph node dissection by splenectomy in gastric cancer patients: relative comparison of the benefits in subgroups of patients. Gastric Cancer. 2011;14:172–177. doi: 10.1007/s10120-011-0028-2. [DOI] [PubMed] [Google Scholar]
- 270.Zheng C, Xu Y, Zhao G, Cai L, Li G, Xu Z, et al. Outcomes of laparoscopic total gastrectomy combined with spleen-preserving hilar lymphadenectomy for locally advanced proximal gastric cancer: a nonrandomized clinical trial. JAMA Netw Open. 2021;4:e2139992. doi: 10.1001/jamanetworkopen.2021.39992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Zheng CH, Xu YC, Zhao G, Cai LS, Li GX, Xu ZK, et al. Safety and feasibility of laparoscopic spleen-preserving No. 10 lymph node dissection for locally advanced upper third gastric cancer: a prospective, multicenter clinical trial. Surg Endosc. 2020;34:5062–5073. doi: 10.1007/s00464-019-07306-8. [DOI] [PubMed] [Google Scholar]
- 272.Jeong O, Kim HG, Ryu SY, Park YK, Jung MR. Adverse prognostic impact of splenectomy on survival in gastric carcinoma patients: regression and propensity score matching analysis of 1074 patients. PLoS One. 2018;13:e0203820. doi: 10.1371/journal.pone.0203820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Yun WG, Lim MH, Kim S, Kim SH, Park JH, Kong SH, et al. Oncologic feasibility of proximal gastrectomy in upper third advanced gastric and esophagogastric junctional cancer. J Gastric Cancer. 2021;21:169–178. doi: 10.5230/jgc.2021.21.e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Yura M, Yoshikawa T, Otsuki S, Yamagata Y, Morita S, Katai H, et al. Oncological safety of proximal gastrectomy for T2/T3 proximal gastric cancer. Gastric Cancer. 2019;22:1029–1035. doi: 10.1007/s10120-019-00938-8. [DOI] [PubMed] [Google Scholar]
- 275.Isozaki H, Okajima K, Yamada S, Nakata E, Nishimura J, Ichinona T, et al. Proximal subtotal gastrectomy for the treatment of carcinoma of the upper third of the stomach: its indications based on lymph node metastasis and perigastric lymphatic flow. Surg Today. 1995;25:21–26. doi: 10.1007/BF00309380. [DOI] [PubMed] [Google Scholar]
- 276.Yang X, Zheng Y, Feng R, Zhu Z, Yan M, Li C. Feasibility of preserving No. 5 and No. 6 lymph nodes in gastrectomy of proximal gastric adenocarcinoma: a retrospective analysis of 395 patients. Front Oncol. 2022;12:810509. doi: 10.3389/fonc.2022.810509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Haruta S, Shinohara H, Hosogi H, Ohkura Y, Kobayashi N, Mizuno A, et al. Proximal gastrectomy with exclusion of no. 3b lesser curvature lymph node dissection could be indicated for patients with advanced upper-third gastric cancer. Gastric Cancer. 2017;20:528–535. doi: 10.1007/s10120-016-0624-2. [DOI] [PubMed] [Google Scholar]
- 278.Siewert JR, Stein HJ. Classification of adenocarcinoma of the oesophagogastric junction. Br J Surg. 1998;85:1457–1459. doi: 10.1046/j.1365-2168.1998.00940.x. [DOI] [PubMed] [Google Scholar]
- 279.Mariette C, Piessen G, Briez N, Gronnier C, Triboulet JP. Oesophagogastric junction adenocarcinoma: which therapeutic approach? Lancet Oncol. 2011;12:296–305. doi: 10.1016/S1470-2045(10)70125-X. [DOI] [PubMed] [Google Scholar]
- 280.Suh YS, Han DS, Kong SH, Lee HJ, Kim YT, Kim WH, et al. Should adenocarcinoma of the esophagogastric junction be classified as esophageal cancer? A comparative analysis according to the seventh AJCC TNM classification. Ann Surg. 2012;255:908–915. doi: 10.1097/SLA.0b013e31824beb95. [DOI] [PubMed] [Google Scholar]
- 281.Lee IS, Ahn JY, Yook JH, Kim BS. Mediastinal lymph node dissection and distal esophagectomy is not essential in early esophagogastric junction adenocarcinoma. World J Surg Oncol. 2017;15:28. doi: 10.1186/s12957-016-1088-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Suh YS, Lee KG, Oh SY, Kong SH, Lee HJ, Kim WH, et al. Recurrence pattern and lymph node metastasis of adenocarcinoma at the esophagogastric junction. Ann Surg Oncol. 2017;24:3631–3639. doi: 10.1245/s10434-017-6011-3. [DOI] [PubMed] [Google Scholar]
- 283.Omloo JM, Lagarde SM, Hulscher JB, Reitsma JB, Fockens P, van Dekken H, et al. Extended transthoracic resection compared with limited transhiatal resection for adenocarcinoma of the mid/distal esophagus: five-year survival of a randomized clinical trial. Ann Surg. 2007;246:992–1000. doi: 10.1097/SLA.0b013e31815c4037. [DOI] [PubMed] [Google Scholar]
- 284.Kurokawa Y, Sasako M, Sano T, Yoshikawa T, Iwasaki Y, Nashimoto A, et al. Ten-year follow-up results of a randomized clinical trial comparing left thoracoabdominal and abdominal transhiatal approaches to total gastrectomy for adenocarcinoma of the oesophagogastric junction or gastric cardia. Br J Surg. 2015;102:341–348. doi: 10.1002/bjs.9764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Blank S, Schmidt T, Heger P, Strowitzki MJ, Sisic L, Heger U, et al. Surgical strategies in true adenocarcinoma of the esophagogastric junction (AEG II): thoracoabdominal or abdominal approach? Gastric Cancer. 2018;21:303–314. doi: 10.1007/s10120-017-0746-1. [DOI] [PubMed] [Google Scholar]
- 286.Mertens AC, Kalff MC, Eshuis WJ, Van Gulik TM, Van Berge Henegouwen MI, Gisbertz SS, et al. Transthoracic versus transhiatal esophagectomy for esophageal cancer: a nationwide propensity score-matched cohort analysis. Ann Surg Oncol. 2021;28:175–183. doi: 10.1245/s10434-020-08760-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Reddavid R, Strignano P, Sofia S, Evangelista A, Deiro G, Cannata G, et al. Transhiatal distal esophagectomy for Siewert type II cardia cancer can be a treatment option in selected patients. Eur J Surg Oncol. 2019;45:1943–1949. doi: 10.1016/j.ejso.2019.04.001. [DOI] [PubMed] [Google Scholar]
- 288.Tosolini C, Reim D, Schirren R, Feith M, Friess H, Novotny AR. Influence of the surgical technique on survival in the treatment of carcinomas of the true cardia (Siewert Type II) - right thoracoabdominal vs. transhiatal-abdominal approach. Eur J Surg Oncol. 2019;45:416–424. doi: 10.1016/j.ejso.2018.09.017. [DOI] [PubMed] [Google Scholar]
- 289.Voron T, Gronnier C, Pasquer A, Thereaux J, Gagniere J, Lebreton G, et al. Adenocarcinoma of the oesophagogastric junction Siewert II: an oesophageal cancer better cured with total gastrectomy. Eur J Surg Oncol. 2019;45:2473–2481. doi: 10.1016/j.ejso.2019.07.022. [DOI] [PubMed] [Google Scholar]
- 290.Yang ZF, Wu DQ, Wang JJ, Feng XY, Zheng JB, Hu WX, et al. Surgical approach for Siewert type II adenocarcinoma of the esophagogastric junction: transthoracic or transabdominal?—a single-center retrospective study. Ann Transl Med. 2018;6:6. doi: 10.21037/atm.2018.10.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Davies AR, Sandhu H, Pillai A, Sinha P, Mattsson F, Forshaw MJ, et al. Surgical resection strategy and the influence of radicality on outcomes in oesophageal cancer. Br J Surg. 2014;101:511–517. doi: 10.1002/bjs.9456. [DOI] [PubMed] [Google Scholar]
- 292.Songun I, Putter H, Kranenbarg EM, Sasako M, van de Velde CJ. Surgical treatment of gastric cancer: 15-year follow-up results of the randomised nationwide Dutch D1D2 trial. Lancet Oncol. 2010;11:439–449. doi: 10.1016/S1470-2045(10)70070-X. [DOI] [PubMed] [Google Scholar]
- 293.Yoo MW, Park DJ, Ahn HS, Jeong SH, Lee HJ, Kim WH, et al. Evaluation of the adequacy of lymph node dissection in pylorus-preserving gastrectomy for early gastric cancer using the maruyama index. World J Surg. 2010;34:291–295. doi: 10.1007/s00268-009-0318-6. [DOI] [PubMed] [Google Scholar]
- 294.Kong SH, Yoo MW, Kim JW, Lee HJ, Kim WH, Lee KU, et al. Validation of limited lymphadenectomy for lower-third gastric cancer based on depth of tumour invasion. Br J Surg. 2011;98:65–72. doi: 10.1002/bjs.7266. [DOI] [PubMed] [Google Scholar]
- 295.Folli S, Morgagni P, Roviello F, De Manzoni G, Marrelli D, Saragoni L, et al. Risk factors for lymph node metastases and their prognostic significance in early gastric cancer (EGC) for the Italian Research Group for Gastric Cancer (IRGGC) Jpn J Clin Oncol. 2001;31:495–499. doi: 10.1093/jjco/hye107. [DOI] [PubMed] [Google Scholar]
- 296.Degiuli M, Calvo F. Survival of early gastric cancer in a specialized European center. Which lymphadenectomy is necessary? World J Surg. 2006;30:2193–2203. doi: 10.1007/s00268-006-0179-1. [DOI] [PubMed] [Google Scholar]
- 297.Shimoyama S, Yasuda H, Mafune K, Kaminishi M. Indications of a minimized scope of lymphadenectomy for submucosal gastric cancer. Ann Surg Oncol. 2002;9:625–631. doi: 10.1007/BF02574477. [DOI] [PubMed] [Google Scholar]
- 298.Kim HH, Han SU, Kim MC, Kim W, Lee HJ, Ryu SW, et al. Effect of laparoscopic distal gastrectomy vs open distal gastrectomy on long-term survival among patients with stage I gastric cancer: the KLASS-01 randomized clinical trial. JAMA Oncol. 2019;5:506–513. doi: 10.1001/jamaoncol.2018.6727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Katai H, Mizusawa J, Katayama H, Morita S, Yamada T, Bando E, et al. Survival outcomes after laparoscopy-assisted distal gastrectomy versus open distal gastrectomy with nodal dissection for clinical stage IA or IB gastric cancer (JCOG0912): a multicentre, non-inferiority, phase 3 randomised controlled trial. Lancet Gastroenterol Hepatol. 2020;5:142–151. doi: 10.1016/S2468-1253(19)30332-2. [DOI] [PubMed] [Google Scholar]
- 300.Kim YW, Yoon HM, Yun YH, Nam BH, Eom BW, Baik YH, et al. Long-term outcomes of laparoscopy-assisted distal gastrectomy for early gastric cancer: result of a randomized controlled trial (COACT 0301) Surg Endosc. 2013;27:4267–4276. doi: 10.1007/s00464-013-3037-x. [DOI] [PubMed] [Google Scholar]
- 301.Wang Z, Dong ZY, Chen JQ, Liu JL. Diagnostic value of sentinel lymph node biopsy in gastric cancer: a meta-analysis. Ann Surg Oncol. 2012;19:1541–1550. doi: 10.1245/s10434-011-2124-2. [DOI] [PubMed] [Google Scholar]
- 302.Huang L, Wei T, Chen J, Zhou D. Feasibility and diagnostic performance of dual-tracer-guided sentinel lymph node biopsy in cT1-2N0M0 gastric cancer: a systematic review and meta-analysis of diagnostic studies. World J Surg Oncol. 2017;15:103. doi: 10.1186/s12957-017-1159-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Yaguchi Y, Tsujimoto H, Kumano I, Takahata R, Matsumoto Y, Yoshida K, et al. Sentinel node navigation surgery attenuates the functional disorders in early gastric cancer. Oncol Rep. 2012;27:643–649. doi: 10.3892/or.2011.1568. [DOI] [PubMed] [Google Scholar]
- 304.Liu N, Niu Z, Niu W, Peng C, Zou X, Sun S, et al. Intraoperative sentinel lymph node mapping guides laparoscopic-assisted distal gastrectomy for distal gastric cancer. Int J Clin Exp Med. 2015;8:5760–5766. [PMC free article] [PubMed] [Google Scholar]
- 305.Okubo K, Arigami T, Matsushita D, Sasaki K, Kijima T, Noda M, et al. Evaluation of postoperative quality of life by PGSAS-45 following local gastrectomy based on the sentinel lymph node concept in early gastric cancer. Gastric Cancer. 2020;23:746–753. doi: 10.1007/s10120-020-01047-7. [DOI] [PubMed] [Google Scholar]
- 306.Youn SI, Son SY, Lee K, Won Y, Min S, Park YS, et al. Quality of life after laparoscopic sentinel node navigation surgery in early gastric cancer: a single-center cohort study. Gastric Cancer. 2021;24:744–751. doi: 10.1007/s10120-020-01145-6. [DOI] [PubMed] [Google Scholar]
- 307.Kinami S, Nakamura N, Miyashita T, Kitakata H, Fushida S, Fujimura T, et al. Life prognosis of sentinel node navigation surgery for early-stage gastric cancer: Outcome of lymphatic basin dissection. World J Gastroenterol. 2021;27:8010–8030. doi: 10.3748/wjg.v27.i46.8010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Kim YW, Min JS, Yoon HM, An JY, Eom BW, Hur H, et al. Laparoscopic sentinel node navigation surgery for stomach preservation in patients with early gastric cancer: a randomized clinical trial. J Clin Oncol. 2022;40:2342–2351. doi: 10.1200/JCO.21.02242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Kitano S, Shiraishi N, Fujii K, Yasuda K, Inomata M, Adachi Y. A randomized controlled trial comparing open vs laparoscopy-assisted distal gastrectomy for the treatment of early gastric cancer: an interim report. Surgery. 2002;131(Suppl):S306–S311. doi: 10.1067/msy.2002.120115. [DOI] [PubMed] [Google Scholar]
- 310.Kim YW, Baik YH, Yun YH, Nam BH, Kim DH, Choi IJ, et al. Improved quality of life outcomes after laparoscopy-assisted distal gastrectomy for early gastric cancer: results of a prospective randomized clinical trial. Ann Surg. 2008;248:721–727. doi: 10.1097/SLA.0b013e318185e62e. [DOI] [PubMed] [Google Scholar]
- 311.Katai H, Mizusawa J, Katayama H, Takagi M, Yoshikawa T, Fukagawa T, et al. Short-term surgical outcomes from a phase III study of laparoscopy-assisted versus open distal gastrectomy with nodal dissection for clinical stage IA/IB gastric cancer: Japan Clinical Oncology Group Study JCOG0912. Gastric Cancer. 2017;20:699–708. doi: 10.1007/s10120-016-0646-9. [DOI] [PubMed] [Google Scholar]
- 312.Lee HJ, Hyung WJ, Yang HK, Han SU, Park YK, An JY, et al. Short-term outcomes of a multicenter randomized controlled trial comparing laparoscopic distal gastrectomy with D2 lymphadenectomy to open distal gastrectomy for locally advanced gastric cancer (KLASS-02-RCT) Ann Surg. 2019;270:983–991. doi: 10.1097/SLA.0000000000003217. [DOI] [PubMed] [Google Scholar]
- 313.Hu Y, Huang C, Sun Y, Su X, Cao H, Hu J, et al. Morbidity and mortality of laparoscopic versus open D2 distal gastrectomy for advanced gastric cancer: a randomized controlled trial. J Clin Oncol. 2016;34:1350–1357. doi: 10.1200/JCO.2015.63.7215. [DOI] [PubMed] [Google Scholar]
- 314.Hyung WJ, Yang HK, Park YK, Lee HJ, An JY, Kim W, et al. Long-term outcomes of laparoscopic distal gastrectomy for locally advanced gastric cancer: the KLASS-02-RCT randomized clinical trial. J Clin Oncol. 2020;38:3304–3313. doi: 10.1200/JCO.20.01210. [DOI] [PubMed] [Google Scholar]
- 315.Son SY, Hur H, Hyung WJ, Park YK, Lee HJ, An JY, et al. Laparoscopic vs open distal gastrectomy for locally advanced gastric cancer: 5-year outcomes of the KLASS-02 randomized clinical trial. JAMA Surg. 2022;157:879–886. doi: 10.1001/jamasurg.2022.2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Huang C, Liu H, Hu Y, Sun Y, Su X, Cao H, et al. Laparoscopic vs open distal gastrectomy for locally advanced gastric cancer: five-year outcomes from the CLASS-01 randomized clinical trial. JAMA Surg. 2022;157:9–17. doi: 10.1001/jamasurg.2021.5104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Han SU, Hur H, Lee HJ, Cho GS, Kim MC, Park YK, et al. Surgeon quality control and standardization of D2 lymphadenectomy for gastric cancer: a prospective multicenter observational study (KLASS-02-QC) Ann Surg. 2021;273:315–324. doi: 10.1097/SLA.0000000000003883. [DOI] [PubMed] [Google Scholar]
- 318.Lee S, Kim JK, Kim YN, Jang DS, Kim YM, Son T, et al. Safety and feasibility of reduced-port robotic distal gastrectomy for gastric cancer: a phase I/II clinical trial. Surg Endosc. 2017;31:4002–4009. doi: 10.1007/s00464-017-5435-y. [DOI] [PubMed] [Google Scholar]
- 319.Eom BW, Yoon HM, Ryu KW, Lee JH, Cho SJ, Lee JY, et al. Comparison of surgical performance and short-term clinical outcomes between laparoscopic and robotic surgery in distal gastric cancer. Eur J Surg Oncol. 2012;38:57–63. doi: 10.1016/j.ejso.2011.09.006. [DOI] [PubMed] [Google Scholar]
- 320.Lu J, Zheng CH, Xu BB, Xie JW, Wang JB, Lin JX, et al. Assessment of robotic versus laparoscopic distal gastrectomy for gastric cancer: a randomized controlled trial. Ann Surg. 2021;273:858–867. doi: 10.1097/SLA.0000000000004466. [DOI] [PubMed] [Google Scholar]
- 321.Kim DJ, Lee JH, Kim W. A comparison of total versus partial omentectomy for advanced gastric cancer in laparoscopic gastrectomy. World J Surg Oncol. 2014;12:64. doi: 10.1186/1477-7819-12-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Hasegawa S, Kunisaki C, Ono H, Oshima T, Fujii S, Taguri M, et al. Omentum-preserving gastrectomy for advanced gastric cancer: a propensity-matched retrospective cohort study. Gastric Cancer. 2013;16:383–388. doi: 10.1007/s10120-012-0198-6. [DOI] [PubMed] [Google Scholar]
- 323.Lee S, Suh YS, Kang SH, Won Y, Park YS, Ahn SH, et al. Should total omentectomy be performed for advanced gastric cancer?: The role of omentectomy during laparoscopic gastrectomy for advanced gastric cancer. Surg Endosc. 2022;36:6937–6948. doi: 10.1007/s00464-022-09039-7. [DOI] [PubMed] [Google Scholar]
- 324.Ri M, Nunobe S, Honda M, Akimoto E, Kinoshita T, Hori S, et al. Gastrectomy with or without omentectomy for cT3-4 gastric cancer: a multicentre cohort study. Br J Surg. 2020;107:1640–1647. doi: 10.1002/bjs.11702. [DOI] [PubMed] [Google Scholar]
- 325.Seo WJ, Choi S, Roh CK, Cho M, Kim YM, Kim HI, et al. Omentum preservation as an oncologically comparable and surgically superior alternative to total omentectomy during radical gastrectomy for T3-T4 gastric cancer. Surgery. 2021;170:610–616. doi: 10.1016/j.surg.2021.02.008. [DOI] [PubMed] [Google Scholar]
- 326.Chai SW, Wang SH, Wang CY, Chen YC, Soong RS, Huang TS. Partial versus total omentectomy in patients with gastric cancer: a systemic review and meta-analysis. Cancers (Basel) 2021;13:13. doi: 10.3390/cancers13194971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 327.Ishizuka M, Shibuya N, Takagi K, Hachiya H, Tago K, Matsumoto T, et al. Omentectomy does not affect the postoperative outcome of patients with locally advanced gastric cancer: a systematic review and meta-analysis. J Surg Res. 2021;264:287–295. doi: 10.1016/j.jss.2021.02.033. [DOI] [PubMed] [Google Scholar]
- 328.Kong M, Chen H, Zhang R, Sheng H, Li L. Overall survival advantage of omentum preservation over omentectomy for advanced gastric cancer: a systematic review and meta-analysis. World J Surg. 2022;46:1952–1961. doi: 10.1007/s00268-022-06562-5. [DOI] [PubMed] [Google Scholar]
- 329.Lin HW, Loh EW, Shen SC, Tam KW. Gastrectomy with or without omentectomy for gastric cancer: a systematic review and meta-analysis. Surgery. 2022;171:1281–1289. doi: 10.1016/j.surg.2021.10.052. [DOI] [PubMed] [Google Scholar]
- 330.Zhu A, Yin G, Liu X, Kong W, Zhang Y, Shan Y, et al. Efficiency of complete omentectomy in patients with resectable gastric cancer: a meta\xe2\x80\x91analysis and systematic review. BMC Gastroenterol. 2021;21:346. doi: 10.1186/s12876-021-01921-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Wang CJ, Kong SH, Park JH, Choi JH, Park SH, Zhu CC, et al. Preservation of hepatic branch of the vagus nerve reduces the risk of gallstone formation after gastrectomy. Gastric Cancer. 2021;24:232–244. doi: 10.1007/s10120-020-01106-z. [DOI] [PubMed] [Google Scholar]
- 332.Lee YW, Kim A, Han M, Yoo MW. Risk factors for gallbladder stone formation after gastric cancer surgery. J Gastric Cancer. 2019;19:417–426. doi: 10.5230/jgc.2019.19.e37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Wu CW, Lo SS, Shen KH, Hsieh MC, Chen JH, Chiang JH, et al. Incidence and factors associated with recurrence patterns after intended curative surgery for gastric cancer. World J Surg. 2003;27:153–158. doi: 10.1007/s00268-002-6279-7. [DOI] [PubMed] [Google Scholar]
- 334.D’Angelica M, Gonen M, Brennan MF, Turnbull AD, Bains M, Karpeh MS. Patterns of initial recurrence in completely resected gastric adenocarcinoma. Ann Surg. 2004;240:808–816. doi: 10.1097/01.sla.0000143245.28656.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Bang YJ, Kim YW, Yang HK, Chung HC, Park YK, Lee KH, et al. Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): a phase 3 open-label, randomised controlled trial. Lancet. 2012;379:315–321. doi: 10.1016/S0140-6736(11)61873-4. [DOI] [PubMed] [Google Scholar]
- 336.Sakuramoto S, Sasako M, Yamaguchi T, Kinoshita T, Fujii M, Nashimoto A, et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med. 2007;357:1810–1820. doi: 10.1056/NEJMoa072252. [DOI] [PubMed] [Google Scholar]
- 337.Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma - 2nd English edition. Gastric Cancer. 1998;1:10–24. doi: 10.1007/s101209800016. [DOI] [PubMed] [Google Scholar]
- 338.Greene FL, Page DL, Fleming ID, Fritz AG, Balch CM, Haller DG, et al. AJCC Cancer Staging Manual. 6th ed. Chicago (IL): Springer; 2002. [Google Scholar]
- 339.Noh SH, Park SR, Yang HK, Chung HC, Chung IJ, Kim SW, et al. Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol. 2014;15:1389–1396. doi: 10.1016/S1470-2045(14)70473-5. [DOI] [PubMed] [Google Scholar]
- 340.Sasako M, Sakuramoto S, Katai H, Kinoshita T, Furukawa H, Yamaguchi T, et al. Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J Clin Oncol. 2011;29:4387–4393. doi: 10.1200/JCO.2011.36.5908. [DOI] [PubMed] [Google Scholar]
- 341.Lee KG, Lee HJ, Oh SY, Yang JY, Ahn HS, Suh YS, et al. Is there any role of adjuvant chemotherapy for T3N0M0 or T1N2M0 gastric cancer patients in stage II in the 7th TNM but stage I in the 6th TNM system? Ann Surg Oncol. 2016;23:1234–1243. doi: 10.1245/s10434-015-4980-7. [DOI] [PubMed] [Google Scholar]
- 342.Yoshikawa T, Terashima M, Mizusawa J, Nunobe S, Nishida Y, Yamada T, et al. Four courses versus eight courses of adjuvant S-1 for patients with stage II gastric cancer (JCOG1104 [OPAS-1]): an open-label, phase 3, non-inferiority, randomised trial. Lancet Gastroenterol Hepatol. 2019;4:208–216. doi: 10.1016/S2468-1253(18)30383-2. [DOI] [PubMed] [Google Scholar]
- 343.Kakeji Y, Yoshida K, Kodera Y, Kochi M, Sano T, Ichikawa W, et al. Three-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 plus docetaxel versus S-1 alone in stage III gastric cancer: JACCRO GC-07. Gastric Cancer. 2022;25:188–196. doi: 10.1007/s10120-021-01224-2. [DOI] [PubMed] [Google Scholar]
- 344.Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC Cancer Staging Manual. 7th ed. Chicago (IL): Springer; 2010. [Google Scholar]
- 345.Park SH, Lim DH, Sohn TS, Lee J, Zang DY, Kim ST, et al. A randomized phase III trial comparing adjuvant single-agent S1, S-1 with oxaliplatin, and postoperative chemoradiation with S-1 and oxaliplatin in patients with node-positive gastric cancer after D2 resection: the ARTIST 2 trial. Ann Oncol. 2021;32:368–374. doi: 10.1016/j.annonc.2020.11.017. [DOI] [PubMed] [Google Scholar]
- 346.Wagner AD, Syn NL, Moehler M, Grothe W, Yong WP, Tai BC, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev. 2017;8:CD004064. doi: 10.1002/14651858.CD004064.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Cunningham D, Starling N, Rao S, Iveson T, Nicolson M, Coxon F, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med. 2008;358:36–46. doi: 10.1056/NEJMoa073149. [DOI] [PubMed] [Google Scholar]
- 348.Kang YK, Kang WK, Shin DB, Chen J, Xiong J, Wang J, et al. Capecitabine/cisplatin versus 5-fluorouracil/cisplatin as first-line therapy in patients with advanced gastric cancer: a randomised phase III noninferiority trial. Ann Oncol. 2009;20:666–673. doi: 10.1093/annonc/mdn717. [DOI] [PubMed] [Google Scholar]
- 349.Okines AF, Norman AR, McCloud P, Kang YK, Cunningham D. Meta-analysis of the REAL-2 and ML17032 trials: evaluating capecitabine-based combination chemotherapy and infused 5-fluorouracil-based combination chemotherapy for the treatment of advanced oesophago-gastric cancer. Ann Oncol. 2009;20:1529–1534. doi: 10.1093/annonc/mdp047. [DOI] [PubMed] [Google Scholar]
- 350.Boku N, Yamamoto S, Fukuda H, Shirao K, Doi T, Sawaki A, et al. Fluorouracil versus combination of irinotecan plus cisplatin versus S-1 in metastatic gastric cancer: a randomised phase 3 study. Lancet Oncol. 2009;10:1063–1069. doi: 10.1016/S1470-2045(09)70259-1. [DOI] [PubMed] [Google Scholar]
- 351.Ajani JA, Rodriguez W, Bodoky G, Moiseyenko V, Lichinitser M, Gorbunova V, et al. Multicenter phase III comparison of cisplatin/S-1 with cisplatin/infusional fluorouracil in advanced gastric or gastroesophageal adenocarcinoma study: the FLAGS trial. J Clin Oncol. 2010;28:1547–1553. doi: 10.1200/JCO.2009.25.4706. [DOI] [PubMed] [Google Scholar]
- 352.Kang YK, Chin K, Chung HC, Kadowaki S, Oh SC, Nakayama N, et al. S-1 plus leucovorin and oxaliplatin versus S-1 plus cisplatin as first-line therapy in patients with advanced gastric cancer (SOLAR): a randomised, open-label, phase 3 trial. Lancet Oncol. 2020;21:1045–1056. doi: 10.1016/S1470-2045(20)30315-6. [DOI] [PubMed] [Google Scholar]
- 353.Al-Batran SE, Hartmann JT, Probst S, Schmalenberg H, Hollerbach S, Hofheinz R, et al. Phase III trial in metastatic gastroesophageal adenocarcinoma with fluorouracil, leucovorin plus either oxaliplatin or cisplatin: a study of the Arbeitsgemeinschaft Internistische Onkologie. J Clin Oncol. 2008;26:1435–1442. doi: 10.1200/JCO.2007.13.9378. [DOI] [PubMed] [Google Scholar]
- 354.Yamada Y, Higuchi K, Nishikawa K, Gotoh M, Fuse N, Sugimoto N, et al. Phase III study comparing oxaliplatin plus S-1 with cisplatin plus S-1 in chemotherapy-naïve patients with advanced gastric cancer. Ann Oncol. 2015;26:141–148. doi: 10.1093/annonc/mdu472. [DOI] [PubMed] [Google Scholar]
- 355.Lee KW, Chung IJ, Ryu MH, Park YI, Nam BH, Oh HS, et al. Multicenter phase III trial of S-1 and cisplatin versus S-1 and oxaliplatin combination chemotherapy for first-line treatment of advanced gastric cancer (SOPP trial) Gastric Cancer. 2021;24:156–167. doi: 10.1007/s10120-020-01101-4. [DOI] [PubMed] [Google Scholar]
- 356.Van Cutsem E, Moiseyenko VM, Tjulandin S, Majlis A, Constenla M, Boni C, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol. 2006;24:4991–4997. doi: 10.1200/JCO.2006.06.8429. [DOI] [PubMed] [Google Scholar]
- 357.Yamada Y, Boku N, Mizusawa J, Iwasa S, Kadowaki S, Nakayama N, et al. Docetaxel plus cisplatin and S-1 versus cisplatin and S-1 in patients with advanced gastric cancer (JCOG1013): an open-label, phase 3, randomised controlled trial. Lancet Gastroenterol Hepatol. 2019;4:501–510. doi: 10.1016/S2468-1253(19)30083-4. [DOI] [PubMed] [Google Scholar]
- 358.Tabernero J, Hoff PM, Shen L, Ohtsu A, Shah MA, Cheng K, et al. Pertuzumab plus trastuzumab and chemotherapy for HER2-positive metastatic gastric or gastro-oesophageal junction cancer (JACOB): final analysis of a double-blind, randomised, placebo-controlled phase 3 study. Lancet Oncol. 2018;19:1372–1384. doi: 10.1016/S1470-2045(18)30481-9. [DOI] [PubMed] [Google Scholar]
- 359.Hecht JR, Bang YJ, Qin SK, Chung HC, Xu JM, Park JO, et al. Lapatinib in combination with capecitabine plus oxaliplatin in human epidermal growth factor receptor 2-positive advanced or metastatic gastric, esophageal, or gastroesophageal adenocarcinoma: TRIO-013/LOGiC--a randomized phase III trial. J Clin Oncol. 2016;34:443–451. doi: 10.1200/JCO.2015.62.6598. [DOI] [PubMed] [Google Scholar]
- 360.Lordick F, Kang YK, Chung HC, Salman P, Oh SC, Bodoky G, et al. Capecitabine and cisplatin with or without cetuximab for patients with previously untreated advanced gastric cancer (EXPAND): a randomised, open-label phase 3 trial. Lancet Oncol. 2013;14:490–499. doi: 10.1016/S1470-2045(13)70102-5. [DOI] [PubMed] [Google Scholar]
- 361.Waddell T, Chau I, Cunningham D, Gonzalez D, Okines AF, Okines C, et al. Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): a randomised, open-label phase 3 trial. Lancet Oncol. 2013;14:481–489. doi: 10.1016/S1470-2045(13)70096-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 362.Ohtsu A, Shah MA, Van Cutsem E, Rha SY, Sawaki A, Park SR, et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol. 2011;29:3968–3976. doi: 10.1200/JCO.2011.36.2236. [DOI] [PubMed] [Google Scholar]
- 363.Fuchs CS, Shitara K, Di Bartolomeo M, Lonardi S, Al-Batran SE, Van Cutsem E, et al. Ramucirumab with cisplatin and fluoropyrimidine as first-line therapy in patients with metastatic gastric or junctional adenocarcinoma (RAINFALL): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20:420–435. doi: 10.1016/S1470-2045(18)30791-5. [DOI] [PubMed] [Google Scholar]
- 364.Catenacci DV, Tebbutt NC, Davidenko I, Murad AM, Al-Batran SE, Ilson DH, et al. Rilotumumab plus epirubicin, cisplatin, and capecitabine as first-line therapy in advanced MET-positive gastric or gastro-oesophageal junction cancer (RILOMET-1): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2017;18:1467–1482. doi: 10.1016/S1470-2045(17)30566-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 365.Doi T, Kang YK, Muro K, Jiang Y, Jain RK, Lizambri R. A phase 3, multicenter, randomized, double-blind, placebo-controlled study of rilotumumab in combination with cisplatin and capecitabine (CX) as first-line therapy for Asian patients (pts) with advanced MET-positive gastric or gastroesophageal junction (G/GEJ) adenocarcinoma: The RILOMET-2 trial. J Clin Oncol. 2015;33(3 Suppl):TPS226–TPS226. [Google Scholar]
- 366.Shah MA, Bang YJ, Lordick F, Alsina M, Chen M, Hack SP, et al. Effect of fluorouracil, leucovorin, and oxaliplatin with or without onartuzumab in HER2-negative, MET-positive gastroesophageal adenocarcinoma: the METGastric randomized clinical trial. JAMA Oncol. 2017;3:620–627. doi: 10.1001/jamaoncol.2016.5580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 367.Janjigian YY, Kawazoe A, Yañez P, Li N, Lonardi S, Kolesnik O, et al. The KEYNOTE-811 trial of dual PD-1 and HER2 blockade in HER2-positive gastric cancer. Nature. 2021;600:727–730. doi: 10.1038/s41586-021-04161-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Kang YK, Chen LT, Ryu MH, Oh DY, Oh SC, Chung HC, et al. Nivolumab plus chemotherapy versus placebo plus chemotherapy in patients with HER2-negative, untreated, unresectable advanced or recurrent gastric or gastro-oesophageal junction cancer (ATTRACTION-4): a randomised, multicentre, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2022;23:234–247. doi: 10.1016/S1470-2045(21)00692-6. [DOI] [PubMed] [Google Scholar]
- 369.Shitara K, Van Cutsem E, Bang YJ, Fuchs C, Wyrwicz L, Lee KW, et al. Efficacy and safety of pembrolizumab or pembrolizumab plus chemotherapy vs chemotherapy alone for patients with first-line, advanced gastric cancer: the KEYNOTE-062 phase 3 randomized clinical trial. JAMA Oncol. 2020;6:1571–1580. doi: 10.1001/jamaoncol.2020.3370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 370.Zhao JJ, Yap DW, Chan YH, Tan BK, Teo CB, Syn NL, et al. Low programmed death-ligand 1-expressing subgroup outcomes of first-line immune checkpoint inhibitors in gastric or esophageal adenocarcinoma. J Clin Oncol. 2022;40:392–402. doi: 10.1200/JCO.21.01862. [DOI] [PubMed] [Google Scholar]
- 371.Kang JH, Lee SI, Lim DH, Park KW, Oh SY, Kwon HC, et al. Salvage chemotherapy for pretreated gastric cancer: a randomized phase III trial comparing chemotherapy plus best supportive care with best supportive care alone. J Clin Oncol. 2012;30:1513–1518. doi: 10.1200/JCO.2011.39.4585. [DOI] [PubMed] [Google Scholar]
- 372.Thuss-Patience PC, Kretzschmar A, Bichev D, Deist T, Hinke A, Breithaupt K, et al. Survival advantage for irinotecan versus best supportive care as second-line chemotherapy in gastric cancer--a randomised phase III study of the Arbeitsgemeinschaft Internistische Onkologie (AIO) Eur J Cancer. 2011;47:2306–2314. doi: 10.1016/j.ejca.2011.06.002. [DOI] [PubMed] [Google Scholar]
- 373.Ford HE, Marshall A, Bridgewater JA, Janowitz T, Coxon FY, Wadsley J, et al. Docetaxel versus active symptom control for refractory oesophagogastric adenocarcinoma (COUGAR-02): an open-label, phase 3 randomised controlled trial. Lancet Oncol. 2014;15:78–86. doi: 10.1016/S1470-2045(13)70549-7. [DOI] [PubMed] [Google Scholar]
- 374.Kim HS, Kim HJ, Kim SY, Kim TY, Lee KW, Baek SK, et al. Second-line chemotherapy versus supportive cancer treatment in advanced gastric cancer: a meta-analysis. Ann Oncol. 2013;24:2850–2854. doi: 10.1093/annonc/mdt351. [DOI] [PubMed] [Google Scholar]
- 375.Hironaka S, Ueda S, Yasui H, Nishina T, Tsuda M, Tsumura T, et al. Randomized, open-label, phase III study comparing irinotecan with paclitaxel in patients with advanced gastric cancer without severe peritoneal metastasis after failure of prior combination chemotherapy using fluoropyrimidine plus platinum: WJOG 4007 trial. J Clin Oncol. 2013;31:4438–4444. doi: 10.1200/JCO.2012.48.5805. [DOI] [PubMed] [Google Scholar]
- 376.Lee KW, Maeng CH, Kim TY, Zang DY, Kim YH, Hwang IG, et al. A phase III study to compare the efficacy and safety of paclitaxel versus irinotecan in patients with metastatic or recurrent gastric cancer who failed in first-line therapy (KCSG ST10-01) Oncologist. 2019;24:18–e24. doi: 10.1634/theoncologist.2018-0142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 377.Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2014;383:31–39. doi: 10.1016/S0140-6736(13)61719-5. [DOI] [PubMed] [Google Scholar]
- 378.Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014;15:1224–1235. doi: 10.1016/S1470-2045(14)70420-6. [DOI] [PubMed] [Google Scholar]
- 379.Chung HC, Kang YK, Chen Z, Bai Y, Wan Ishak WZ, Shim BY, et al. Pembrolizumab versus paclitaxel for previously treated advanced gastric or gastroesophageal junction cancer (KEYNOTE-063): a randomized, open-label, phase 3 trial in Asian patients. Cancer. 2022;128:995–1003. doi: 10.1002/cncr.34019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 380.Fuchs CS, Özgüroğlu M, Bang YJ, Di Bartolomeo M, Mandala M, Ryu MH, et al. Pembrolizumab versus paclitaxel for previously treated PD-L1-positive advanced gastric or gastroesophageal junction cancer: 2-year update of the randomized phase 3 KEYNOTE-061 trial. Gastric Cancer. 2022;25:197–206. doi: 10.1007/s10120-021-01227-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Shitara K, Özgüroğlu M, Bang YJ, Di Bartolomeo M, Mandalà M, Ryu MH, et al. Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial. Lancet. 2018;392:123–133. doi: 10.1016/S0140-6736(18)31257-1. [DOI] [PubMed] [Google Scholar]
- 382.Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357:409–413. doi: 10.1126/science.aan6733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Marabelle A, Le DT, Ascierto PA, Di Giacomo AM, De Jesus-Acosta A, Delord JP, et al. Efficacy of pembrolizumab in patients with noncolorectal high microsatellite instability/mismatch repair-deficient cancer: results from the phase II KEYNOTE-158 study. J Clin Oncol. 2020;38:1–10. doi: 10.1200/JCO.19.02105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Cutsem EV. Primary analysis of a phase II single-arm trial of trastuzumab deruxtecan (T-DXd) in western patients (Pts) with HER2-positive (HER2+) unresectable or metastatic gastric or gastroesophageal junction (GEJ) cancer who progressed on or after a trastuzumab-containing regimen. Ann Oncol. 2021;32:S1283–S1346. [Google Scholar]
- 385.Lee MJ, Hwang IG, Jang JS, Choi JH, Park BB, Chang MH, et al. Outcomes of third-line docetaxel-based chemotherapy in advanced gastric cancer who failed previous oxaliplatin-based and irinotecan-based chemotherapies. Cancer Res Treat. 2012;44:235–241. doi: 10.4143/crt.2012.44.4.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 386.Fanotto V, Uccello M, Pecora I, Rimassa L, Leone F, Rosati G, et al. Outcomes of advanced gastric cancer patients treated with at least three lines of systemic chemotherapy. Oncologist. 2017;22:1463–1469. doi: 10.1634/theoncologist.2017-0158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Choi IS, Choi M, Lee JH, Kim JH, Suh KJ, Lee JY, et al. Treatment patterns and outcomes in patients with metastatic gastric cancer receiving third-line chemotherapy: a population-based outcomes study. PLoS One. 2018;13:e0198544. doi: 10.1371/journal.pone.0198544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Shitara K, Doi T, Dvorkin M, Mansoor W, Arkenau HT, Prokharau A, et al. Trifluridine/tipiracil versus placebo in patients with heavily pretreated metastatic gastric cancer (TAGS): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2018;19:1437–1448. doi: 10.1016/S1470-2045(18)30739-3. [DOI] [PubMed] [Google Scholar]
- 389.Kang YK, Boku N, Satoh T, Ryu MH, Chao Y, Kato K, et al. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;390:2461–2471. doi: 10.1016/S0140-6736(17)31827-5. [DOI] [PubMed] [Google Scholar]
- 390.Chen LT, Satoh T, Ryu MH, Chao Y, Kato K, Chung HC, et al. A phase 3 study of nivolumab in previously treated advanced gastric or gastroesophageal junction cancer (ATTRACTION-2): 2-year update data. Gastric Cancer. 2020;23:510–519. doi: 10.1007/s10120-019-01034-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Shitara K, Bang YJ, Iwasa S, Sugimoto N, Ryu MH, Sakai D, et al. Trastuzumab deruxtecan in previously treated HER2-positive gastric cancer. N Engl J Med. 2020;382:2419–2430. doi: 10.1056/NEJMoa2004413. [DOI] [PubMed] [Google Scholar]
- 392.Kang YK, Yook JH, Park YK, Lee JS, Kim YW, Kim JY, et al. PRODIGY: a phase III study of neoadjuvant docetaxel, oxaliplatin, and S-1 plus surgery and adjuvant S-1 versus surgery and adjuvant S-1 for resectable advanced gastric cancer. J Clin Oncol. 2021;39:2903–2913. doi: 10.1200/JCO.20.02914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 393.Zhang X, Liang H, Li Z, Xue Y, Wang Y, Zhou Z, et al. RESOLVE study group. Perioperative or postoperative adjuvant oxaliplatin with S-1 versus adjuvant oxaliplatin with capecitabine in patients with locally advanced gastric or gastro-oesophageal junction adenocarcinoma undergoing D2 gastrectomy (RESOLVE): an open-label, superiority and non-inferiority, phase 3 randomised controlled trial. Lancet Oncol. 2021;22:1081–1092. doi: 10.1016/S1470-2045(21)00297-7. [DOI] [PubMed] [Google Scholar]
- 394.Iwasaki Y, Terashima M, Mizusawa J, Katayama H, Nakamura K, Katai H, et al. Gastrectomy with or without neoadjuvant S-1 plus cisplatin for type 4 or large type 3 gastric cancer (JCOG0501): an open-label, phase 3, randomized controlled trial. Gastric Cancer. 2021;24:492–502. doi: 10.1007/s10120-020-01136-7. [DOI] [PubMed] [Google Scholar]
- 395.Kim HD, Lee JS, Yook JH, Ryu MH, Park YK, Kim JY, et al. Radiological criteria for selecting candidates for neoadjuvant chemotherapy for gastric cancer: an exploratory analysis from the PRODIGY study. Gastric Cancer. 2022;25:170–179. doi: 10.1007/s10120-021-01243-z. [DOI] [PubMed] [Google Scholar]
- 396.Kwon HC, Kim MC, Kim KH, Jang JS, Oh SY, Kim SH, et al. Adjuvant chemoradiation versus chemotherapy in completely resected advanced gastric cancer with D2 nodal dissection. Asia Pac J Clin Oncol. 2010;6:278–285. doi: 10.1111/j.1743-7563.2010.01331.x. [DOI] [PubMed] [Google Scholar]
- 397.Kim TH, Park SR, Ryu KW, Kim YW, Bae JM, Lee JH, et al. Phase 3 trial of postoperative chemotherapy alone versus chemoradiation therapy in stage III-IV gastric cancer treated with R0 gastrectomy and D2 lymph node dissection. Int J Radiat Oncol Biol Phys. 2012;84:e585–e592. doi: 10.1016/j.ijrobp.2012.07.2378. [DOI] [PubMed] [Google Scholar]
- 398.Zhu WG, Xua DF, Pu J, Zong CD, Li T, Tao GZ, et al. A randomized, controlled, multicenter study comparing intensity-modulated radiotherapy plus concurrent chemotherapy with chemotherapy alone in gastric cancer patients with D2 resection. Radiother Oncol. 2012;104:361–366. doi: 10.1016/j.radonc.2012.08.024. [DOI] [PubMed] [Google Scholar]
- 399.Cats A, Jansen EP, van Grieken NC, Sikorska K, Lind P, Nordsmark M, et al. Chemotherapy versus chemoradiotherapy after surgery and preoperative chemotherapy for resectable gastric cancer (CRITICS): an international, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19:616–628. doi: 10.1016/S1470-2045(18)30132-3. [DOI] [PubMed] [Google Scholar]
- 400.Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJ, Nicolson M, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355:11–20. doi: 10.1056/NEJMoa055531. [DOI] [PubMed] [Google Scholar]
- 401.Klevebro F, Alexandersson von Döbeln G, Wang N, Johnsen G, Jacobsen AB, Friesland S, et al. A randomized clinical trial of neoadjuvant chemotherapy versus neoadjuvant chemoradiotherapy for cancer of the oesophagus or gastro-oesophageal junction. Ann Oncol. 2016;27:660–667. doi: 10.1093/annonc/mdw010. [DOI] [PubMed] [Google Scholar]
- 402.Leong T, Smithers BM, Haustermans K, Michael M, Gebski V, Miller D, et al. TOPGEAR: a randomized, phase III trial of perioperative ECF chemotherapy with or without preoperative chemoradiation for resectable gastric cancer: interim results from an international, intergroup trial of the AGITG, TROG, EORTC and CCTG. Ann Surg Oncol. 2017;24:2252–2258. doi: 10.1245/s10434-017-5830-6. [DOI] [PubMed] [Google Scholar]
- 403.Stahl M, Walz MK, Riera-Knorrenschild J, Stuschke M, Sandermann A, Bitzer M, et al. Preoperative chemotherapy versus chemoradiotherapy in locally advanced adenocarcinomas of the oesophagogastric junction (POET): long-term results of a controlled randomised trial. Eur J Cancer. 2017;81:183–190. doi: 10.1016/j.ejca.2017.04.027. [DOI] [PubMed] [Google Scholar]
- 404.Stahl M, Walz MK, Stuschke M, Lehmann N, Meyer HJ, Riera-Knorrenschild J, et al. Phase III comparison of preoperative chemotherapy compared with chemoradiotherapy in patients with locally advanced adenocarcinoma of the esophagogastric junction. J Clin Oncol. 2009;27:851–856. doi: 10.1200/JCO.2008.17.0506. [DOI] [PubMed] [Google Scholar]
- 405.von Döbeln GA, Klevebro F, Jacobsen AB, Johannessen HO, Nielsen NH, Johnsen G, et al. Neoadjuvant chemotherapy versus neoadjuvant chemoradiotherapy for cancer of the esophagus or gastroesophageal junction: long-term results of a randomized clinical trial. Dis Esophagus. 2019;32:32. doi: 10.1093/dote/doy078. [DOI] [PubMed] [Google Scholar]
- 406.Hong J, Chen Y, Li J, Hu P, Chen P, Du N, et al. Comparison of gastrojejunostomy to endoscopic stenting for gastric outlet obstruction: an updated systematic review and meta-analysis. Am J Surg. 2022;223:1067–1078. doi: 10.1016/j.amjsurg.2021.10.038. [DOI] [PubMed] [Google Scholar]
- 407.Bian SB, Shen WS, Xi HQ, Wei B, Chen L. Palliative therapy for gastric outlet obstruction caused by unresectable gastric cancer: a meta-analysis comparison of gastrojejunostomy with endoscopic stenting. Chin Med J (Engl) 2016;129:1113–1121. doi: 10.4103/0366-6999.180530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Alonso-Lárraga JO, Alvaro-Villegas JC, Sobrino-Cossío S, Hernández-Guerrero A, de-la-Mora-Levy G, Figueroa-Barojas P. Self-expanding metal stents versus antrectomy for the palliative treatment of obstructive adenocarcinoma of the gastric antrum. Rev Esp Enferm Dig. 2012;104:185–189. doi: 10.4321/s1130-01082012000400003. [DOI] [PubMed] [Google Scholar]
- 409.Fiori E, Crocetti D, Sapienza P, Cirocchi R, Sterpetti AV, Miccini M, et al. Palliative surgery or metallic stent positioning for advanced gastric cancer: differences in QOL. Medicina (Kaunas) 2021;57:57. doi: 10.3390/medicina57050428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.Fiori E, Lamazza A, Volpino P, Burza A, Paparelli C, Cavallaro G, et al. Palliative management of malignant antro-pyloric strictures. Gastroenterostomy vs. endoscopic stenting. A randomized prospective trial. Anticancer Res. 2004;24:269–271. [PubMed] [Google Scholar]
- 411.Fiori E, Lamazza A, Demasi E, Decesare A, Schillaci A, Sterpetti AV. Endoscopic stenting for gastric outlet obstruction in patients with unresectable antro pyloric cancer. Systematic review of the literature and final results of a prospective study. The point of view of a surgical group. Am J Surg. 2013;206:210–217. doi: 10.1016/j.amjsurg.2012.08.018. [DOI] [PubMed] [Google Scholar]
- 412.Haga Y, Hiki N, Kinoshita T, Ojima T, Nabeya Y, Kuwabara S, et al. Treatment option of endoscopic stent insertion or gastrojejunostomy for gastric outlet obstruction due to gastric cancer: a propensity score-matched analysis. Gastric Cancer. 2020;23:667–676. doi: 10.1007/s10120-020-01040-0. [DOI] [PubMed] [Google Scholar]
- 413.Jang SH, Lee H, Min BH, Kim SM, Kim HS, Carriere KC, et al. Palliative gastrojejunostomy versus endoscopic stent placement for gastric outlet obstruction in patients with unresectable gastric cancer: a propensity score-matched analysis. Surg Endosc. 2017;31:4217–4223. doi: 10.1007/s00464-017-5480-6. [DOI] [PubMed] [Google Scholar]
- 414.Keränen I, Kylänpää L, Udd M, Louhimo J, Lepistö A, Halttunen J, et al. Gastric outlet obstruction in gastric cancer: a comparison of three palliative methods. J Surg Oncol. 2013;108:537–541. doi: 10.1002/jso.23442. [DOI] [PubMed] [Google Scholar]
- 415.Maetani I, Akatsuka S, Ikeda M, Tada T, Ukita T, Nakamura Y, et al. Self-expandable metallic stent placement for palliation in gastric outlet obstructions caused by gastric cancer: a comparison with surgical gastrojejunostomy. J Gastroenterol. 2005;40:932–937. doi: 10.1007/s00535-005-1651-7. [DOI] [PubMed] [Google Scholar]
- 416.No JH, Kim SW, Lim CH, Kim JS, Cho YK, Park JM, et al. Long-term outcome of palliative therapy for gastric outlet obstruction caused by unresectable gastric cancer in patients with good performance status: endoscopic stenting versus surgery. Gastrointest Endosc. 2013;78:55–62. doi: 10.1016/j.gie.2013.01.041. [DOI] [PubMed] [Google Scholar]
- 417.Park CH, Park JC, Kim EH, Chung H, An JY, Kim HI, et al. Impact of carcinomatosis and ascites status on long-term outcomes of palliative treatment for patients with gastric outlet obstruction caused by unresectable gastric cancer: stent placement versus palliative gastrojejunostomy. Gastrointest Endosc. 2015;81:321–332. doi: 10.1016/j.gie.2014.06.024. [DOI] [PubMed] [Google Scholar]
- 418.Fujitani K, Yang HK, Mizusawa J, Kim YW, Terashima M, Han SU, et al. Gastrectomy plus chemotherapy versus chemotherapy alone for advanced gastric cancer with a single non-curable factor (REGATTA): a phase 3, randomised controlled trial. Lancet Oncol. 2016;17:309–318. doi: 10.1016/S1470-2045(15)00553-7. [DOI] [PubMed] [Google Scholar]
- 419.Shimura T, Kataoka H, Sasaki M, Yamada T, Hayashi K, Togawa S, et al. Feasibility of self-expandable metallic stent plus chemotherapy for metastatic gastric cancer with pyloric stenosis. J Gastroenterol Hepatol. 2009;24:1358–1364. doi: 10.1111/j.1440-1746.2009.05857.x. [DOI] [PubMed] [Google Scholar]
- 420.Wang C, Zhang X, Lin S, Yang C, Zhou B, Mi Y, et al. Superiority of laparoscopic gastrojejunostomy combined with multimodality therapy for gastric outlet obstruction caused by advanced gastric cancer. Front Oncol. 2022;12:814283. doi: 10.3389/fonc.2022.814283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 421.Yoshida K, Yasufuku I, Terashima M, Young Rha S, Moon Bae J, Li G, et al. International retrospective cohort study of conversion therapy for stage IV gastric cancer 1 (CONVO-GC-1) Ann Gastroenterol Surg. 2021;6:227–240. doi: 10.1002/ags3.12515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Al-Batran SE, Goetze TO, Mueller DW, Vogel A, Winkler M, Lorenzen S, et al. The RENAISSANCE (AIO-FLOT5) trial: effect of chemotherapy alone vs. chemotherapy followed by surgical resection on survival and quality of life in patients with limited-metastatic adenocarcinoma of the stomach or esophagogastric junction - a phase III trial of the German AIO/CAO-V/CAOGI. BMC Cancer. 2017;17:893. doi: 10.1186/s12885-017-3918-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Hellman S, Weichselbaum RR. Oligometastases. J Clin Oncol. 1995;13:8–10. doi: 10.1200/JCO.1995.13.1.8. [DOI] [PubMed] [Google Scholar]
- 424.Lavacchi D, Giommoni E, Cianchi F, Antonuzzo L. Treatment strategy for oligometastatic gastric cancer: brief considerations. J Gastrointest Cancer. 2022 doi: 10.1007/s12029-021-00792-x. [DOI] [PubMed] [Google Scholar]
- 425.Jin P, Ji X, Tian Y. Surgical management of oligometastatic disease in gastric cancer. Clin Res Hepatol Gastroenterol. 2020;44:638–645. doi: 10.1016/j.clinre.2020.02.003. [DOI] [PubMed] [Google Scholar]
- 426.Chen S, Li YF, Feng XY, Zhou ZW, Yuan XH, Chen YB. Significance of palliative gastrectomy for late-stage gastric cancer patients. J Surg Oncol. 2012;106:862–871. doi: 10.1002/jso.23158. [DOI] [PubMed] [Google Scholar]
- 427.Miki Y, Fujitani K, Hirao M, Kurokawa Y, Mano M, Tsujie M, et al. Significance of surgical treatment of liver metastases from gastric cancer. Anticancer Res. 2012;32:665–670. [PubMed] [Google Scholar]
- 428.Tang K, Zhang B, Dong L, Wang L, Tang Z. Radiofrequency ablation versus traditional liver resection and chemotherapy for liver metastases from gastric cancer. J Int Med Res. 2020;48:300060520940509. doi: 10.1177/0300060520940509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 429.Hwang JE, Kim SH, Jin J, Hong JY, Kim MJ, Jung SH, et al. Combination of percutaneous radiofrequency ablation and systemic chemotherapy are effective treatment modalities for metachronous liver metastases from gastric cancer. Clin Exp Metastasis. 2014;31:25–32. doi: 10.1007/s10585-013-9606-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Kim HO, Hwang SI, Hong HP, Yoo CH. Radiofrequency ablation for metachronous hepatic metastases from gastric cancer. Surg Laparosc Endosc Percutan Tech. 2009;19:208–212. doi: 10.1097/SLE.0b013e3181a033d7. [DOI] [PubMed] [Google Scholar]
- 431.Liu SF, Lu CR, Cheng HD, Xi HQ, Cui JX, Li JY, et al. Comparison of therapeutic efficacy between gastrectomy with transarterial chemoembolization plus systemic chemotherapy and systemic chemotherapy alone in gastric cancer with synchronous liver metastasis. Chin Med J (Engl) 2015;128:2194–2201. doi: 10.4103/0366-6999.162497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 432.Cho JH, Lim JY, Choi AR, Choi SM, Kim JW, Choi SH, et al. Comparison of surgery plus chemotherapy and palliative chemotherapy alone for advanced gastric cancer with Krukenberg tumor. Cancer Res Treat. 2015;47:697–705. doi: 10.4143/crt.2013.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 433.Lu LC, Shao YY, Hsu CH, Hsu C, Cheng WF, Lin YL, et al. Metastasectomy of Krukenberg tumors may be associated with survival benefits in patients with metastatic gastric cancer. Anticancer Res. 2012;32:3397–3401. [PubMed] [Google Scholar]
- 434.Yu P, Huang L, Cheng G, Yang L, Dai G, Ying J, et al. Treatment strategy and prognostic factors for Krukenberg tumors of gastric origin: report of a 10-year single-center experience from China. Oncotarget. 2017;8:82558–82570. doi: 10.18632/oncotarget.19759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Cheong JH, Hyung WJ, Chen J, Kim J, Choi SH, Noh SH. Survival benefit of metastasectomy for Krukenberg tumors from gastric cancer. Gynecol Oncol. 2004;94:477–482. doi: 10.1016/j.ygyno.2004.05.007. [DOI] [PubMed] [Google Scholar]
- 436.Yoshikawa T, Sasako M, Yamamoto S, Sano T, Imamura H, Fujitani K, et al. Phase II study of neoadjuvant chemotherapy and extended surgery for locally advanced gastric cancer. Br J Surg. 2009;96:1015–1022. doi: 10.1002/bjs.6665. [DOI] [PubMed] [Google Scholar]
- 437.Tsuburaya A, Mizusawa J, Tanaka Y, Fukushima N, Nashimoto A, Sasako M, et al. Neoadjuvant chemotherapy with S-1 and cisplatin followed by D2 gastrectomy with para-aortic lymph node dissection for gastric cancer with extensive lymph node metastasis. Br J Surg. 2014;101:653–660. doi: 10.1002/bjs.9484. [DOI] [PubMed] [Google Scholar]
- 438.Ito S, Sano T, Mizusawa J, Takahari D, Katayama H, Katai H, et al. A phase II study of preoperative chemotherapy with docetaxel, cisplatin, and S-1 followed by gastrectomy with D2 plus para-aortic lymph node dissection for gastric cancer with extensive lymph node metastasis: JCOG1002. Gastric Cancer. 2017;20:322–331. doi: 10.1007/s10120-016-0619-z. [DOI] [PubMed] [Google Scholar]
- 439.Chau I, Norman AR, Cunningham D, Waters JS, Oates J, Ross PJ. Multivariate prognostic factor analysis in locally advanced and metastatic esophago-gastric cancer--pooled analysis from three multicenter, randomized, controlled trials using individual patient data. J Clin Oncol. 2004;22:2395–2403. doi: 10.1200/JCO.2004.08.154. [DOI] [PubMed] [Google Scholar]
- 440.Kitayama J, Ishigami H, Yamaguchi H, Sakuma Y, Horie H, Hosoya Y, et al. Treatment of patients with peritoneal metastases from gastric cancer. Ann Gastroenterol Surg. 2018;2:116–123. doi: 10.1002/ags3.12060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.Padmakumar S, Parayath N, Leslie F, Nair SV, Menon D, Amiji MM. Intraperitoneal chemotherapy for ovarian cancer using sustained-release implantable devices. Expert Opin Drug Deliv. 2018;15:481–494. doi: 10.1080/17425247.2018.1446938. [DOI] [PubMed] [Google Scholar]
- 442.Fujiwara Y, Takiguchi S, Nakajima K, Miyata H, Yamasaki M, Kurokawa Y, et al. Intraperitoneal docetaxel combined with S-1 for advanced gastric cancer with peritoneal dissemination. J Surg Oncol. 2012;105:38–42. doi: 10.1002/jso.22057. [DOI] [PubMed] [Google Scholar]
- 443.Ishigami H, Kitayama J, Kaisaki S, Hidemura A, Kato M, Otani K, et al. Phase II study of weekly intravenous and intraperitoneal paclitaxel combined with S-1 for advanced gastric cancer with peritoneal metastasis. Ann Oncol. 2010;21:67–70. doi: 10.1093/annonc/mdp260. [DOI] [PubMed] [Google Scholar]
- 444.Fushida S, Kinoshita J, Kaji M, Hirono Y, Goda F, Yagi Y, et al. Phase I/II study of intraperitoneal docetaxel plus S-1 for the gastric cancer patients with peritoneal carcinomatosis. Cancer Chemother Pharmacol. 2013;71:1265–1272. doi: 10.1007/s00280-013-2122-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Armstrong DK, Bundy B, Wenzel L, Huang HQ, Baergen R, Lele S, et al. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med. 2006;354:34–43. doi: 10.1056/NEJMoa052985. [DOI] [PubMed] [Google Scholar]
- 446.Yamaguchi H, Kitayama J, Ishigami H, Emoto S, Yamashita H, Watanabe T. A phase 2 trial of intravenous and intraperitoneal paclitaxel combined with S-1 for treatment of gastric cancer with macroscopic peritoneal metastasis. Cancer. 2013;119:3354–3358. doi: 10.1002/cncr.28204. [DOI] [PubMed] [Google Scholar]
- 447.Armstrong DK, Alvarez RD, Backes FJ, Bakkum-Gamez JN, Barroilhet L, Behbakht K, et al. NCCN guidelines® insights: ovarian cancer, version 3.2022. J Natl Compr Canc Netw. 2022;20:972–980. doi: 10.6004/jnccn.2022.0047. [DOI] [PubMed] [Google Scholar]
- 448.Ishigami H, Fujiwara Y, Fukushima R, Nashimoto A, Yabusaki H, Imano M, et al. Phase III trial comparing intraperitoneal and intravenous paclitaxel plus S-1 versus cisplatin plus S-1 in patients with gastric cancer with peritoneal metastasis: PHOENIX-GC trial. J Clin Oncol. 2018;36:1922–1929. doi: 10.1200/JCO.2018.77.8613. [DOI] [PubMed] [Google Scholar]
- 449.Lee JK, Park YS, Lee K, Youn SI, Won Y, Min SH, et al. Prognostic significance of surgery-induced sarcopenia in the survival of gastric cancer patients: a sex-specific analysis. J Cachexia Sarcopenia Muscle. 2021;12:1897–1907. doi: 10.1002/jcsm.12793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Meng Q, Tan S, Jiang Y, Han J, Xi Q, Zhuang Q, et al. Post-discharge oral nutritional supplements with dietary advice in patients at nutritional risk after surgery for gastric cancer: a randomized clinical trial. Clin Nutr. 2021;40:40–46. doi: 10.1016/j.clnu.2020.04.043. [DOI] [PubMed] [Google Scholar]
- 451.Miyazaki Y, Omori T, Fujitani K, Fujita J, Kawabata R, Imamura H, et al. Oral nutritional supplements versus a regular diet alone for body weight loss after gastrectomy: a phase 3, multicenter, open-label randomized controlled trial. Gastric Cancer. 2021;24:1150–1159. doi: 10.1007/s10120-021-01188-3. [DOI] [PubMed] [Google Scholar]
- 452.Hu Y, Kim HI, Hyung WJ, Song KJ, Lee JH, Kim YM, et al. Vitamin B(12) deficiency after gastrectomy for gastric cancer: an analysis of clinical patterns and risk factors. Ann Surg. 2013;258:970–975. doi: 10.1097/SLA.0000000000000214. [DOI] [PubMed] [Google Scholar]
- 453.Lim CH, Kim SW, Kim WC, Kim JS, Cho YK, Park JM, et al. Anemia after gastrectomy for early gastric cancer: long-term follow-up observational study. World J Gastroenterol. 2012;18:6114–6119. doi: 10.3748/wjg.v18.i42.6114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.Jun JH, Yoo JE, Lee JA, Kim YS, Sunwoo S, Kim BS, et al. Anemia after gastrectomy in long-term survivors of gastric cancer: a retrospective cohort study. Int J Surg. 2016;28:162–168. doi: 10.1016/j.ijsu.2016.02.084. [DOI] [PubMed] [Google Scholar]
- 455.Kim YW, Bae JM, Park YK, Yang HK, Yu W, Yook JH, et al. Effect of intravenous ferric carboxymaltose on hemoglobin response among patients with acute isovolemic anemia following gastrectomy: the FAIRY randomized clinical trial. JAMA. 2017;317:2097–2104. doi: 10.1001/jama.2017.5703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 456.Snook J, Bhala N, Beales IL, Cannings D, Kightley C, Logan RP, et al. British Society of Gastroenterology guidelines for the management of iron deficiency anaemia in adults. Gut. 2021;70:2030–2051. doi: 10.1136/gutjnl-2021-325210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 457.Choi YJ, Choi IY, Jang W, Jeong SM, Park S, Han K, et al. Gastrectomy, vitamin B12 supplementation and the risk of Parkinson’s disease: a nationwide cohort study. Parkinsonism Relat Disord. 2021;83:15–21. doi: 10.1016/j.parkreldis.2020.12.026. [DOI] [PubMed] [Google Scholar]
- 458.Choi YJ, Shin DW, Jang W, Lee DH, Jeong SM, Park S, et al. Risk of dementia in gastric cancer survivors who underwent gastrectomy: a nationwide study in Korea. Ann Surg Oncol. 2019;26:4229–4237. doi: 10.1245/s10434-019-07913-8. [DOI] [PubMed] [Google Scholar]
- 459.Adachi S, Kawamoto T, Otsuka M, Todoroki T, Fukao K. Enteral vitamin B12 supplements reverse postgastrectomy B12 deficiency. Ann Surg. 2000;232:199–201. doi: 10.1097/00000658-200008000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Kim HI, Hyung WJ, Song KJ, Choi SH, Kim CB, Noh SH. Oral vitamin B12 replacement: an effective treatment for vitamin B12 deficiency after total gastrectomy in gastric cancer patients. Ann Surg Oncol. 2011;18:3711–3717. doi: 10.1245/s10434-011-1764-6. [DOI] [PubMed] [Google Scholar]
- 461.Namikawa T, Maeda M, Yokota K, Iwabu J, Munekage M, Uemura S, et al. Enteral vitamin B12 supplementation is effective for improving anemia in patients who underwent total gastrectomy. Oncology. 2021;99:225–233. doi: 10.1159/000513888. [DOI] [PubMed] [Google Scholar]
- 462.Atsumi Y, Rino Y, Wada H, Kitani Y, Ozawa Y, Aoyama T, et al. Changes in bone metabolism after gastric cancer surgery in male patients: a prospective observational study. Gastric Cancer. 2019;22:237–243. doi: 10.1007/s10120-018-0835-9. [DOI] [PubMed] [Google Scholar]
- 463.Oh HJ, Yoon BH, Ha YC, Suh DC, Lee SM, Koo KH, et al. The change of bone mineral density and bone metabolism after gastrectomy for gastric cancer: a meta-analysis. Osteoporos Int. 2020;31:267–275. doi: 10.1007/s00198-019-05220-2. [DOI] [PubMed] [Google Scholar]
- 464.Rino Y, Takanashi Y, Yamamoto Y, Inagaki D, Kawamoto M, Harada H, et al. Bone disorder and vitamin D after gastric cancer surgery. Hepatogastroenterology. 2007;54:1596–1600. [PubMed] [Google Scholar]
- 465.Baek KH, Jeon HM, Lee SS, Lim DJ, Oh KW, Lee WY, et al. Short-term changes in bone and mineral metabolism following gastrectomy in gastric cancer patients. Bone. 2008;42:61–67. doi: 10.1016/j.bone.2007.08.027. [DOI] [PubMed] [Google Scholar]
- 466.Shin DW, Suh B, Lim H, Suh YS, Choi YJ, Jeong SM, et al. Increased risk of osteoporotic fracture in postgastrectomy gastric cancer survivors compared with matched controls: a nationwide cohort study in Korea. Am J Gastroenterol. 2019;114:1735–1743. doi: 10.14309/ajg.0000000000000436. [DOI] [PubMed] [Google Scholar]
- 467.Yoo SH, Lee JA, Kang SY, Kim YS, Sunwoo S, Kim BS, et al. Risk of osteoporosis after gastrectomy in long-term gastric cancer survivors. Gastric Cancer. 2018;21:720–727. doi: 10.1007/s10120-017-0777-7. [DOI] [PubMed] [Google Scholar]
- 468.Ha J, Lee JM, Lim Y, Kim MK, Kwon HS, Song KH, et al. Effect of bisphosphonate on the prevention of bone loss in patients with gastric cancer after gastrectomy: a randomized controlled trial. Bone. 2020;130:115138. doi: 10.1016/j.bone.2019.115138. [DOI] [PubMed] [Google Scholar]
- 469.Lamb B, Green JS, Vincent C, Sevdalis N. Decision making in surgical oncology. Surg Oncol. 2011;20:163–168. doi: 10.1016/j.suronc.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 470.Stephens MR, Lewis WG, Brewster AE, Lord I, Blackshaw GR, Hodzovic I, et al. Multidisciplinary team management is associated with improved outcomes after surgery for esophageal cancer. Dis Esophagus. 2006;19:164–171. doi: 10.1111/j.1442-2050.2006.00559.x. [DOI] [PubMed] [Google Scholar]
- 472.Xiang YY, Deng CC, Liu HY, Kuo ZC, Zhang CH, He YL. The prognostic effect of multidisciplinary team intervention in patients with advanced gastric cancer. Curr Oncol. 2022;29:1201–1212. doi: 10.3390/curroncol29020102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 472.Di L, Wu H, Zhu R, Li Y, Wu X, Xie R, et al. Multi-disciplinary team for early gastric cancer diagnosis improves the detection rate of early gastric cancer. BMC Gastroenterol. 2017;17:147. doi: 10.1186/s12876-017-0711-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Yuan Z, Weng S, Ye C, Hu H, Zhang S, Yuan Y. CSCO guidelines for colorectal cancer version 2022: updates and discussions. Chin J Cancer Res. 2022;34:67–70. doi: 10.21147/j.issn.1000-9604.2022.02.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 474.Brar SS, Mahar AL, Helyer LK, Swallow C, Law C, Paszat L, et al. Processes of care in the multidisciplinary treatment of gastric cancer: results of a RAND/UCLA expert panel. JAMA Surg. 2014;149:18–25. doi: 10.1001/jamasurg.2013.3959. [DOI] [PubMed] [Google Scholar]
- 475.Smyth EC, Verheij M, Allum W, Cunningham D, Cervantes A, Arnold D, et al. Gastric cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27(Suppl 5):v38–v49. doi: 10.1093/annonc/mdw350. [DOI] [PubMed] [Google Scholar]
- 476.Van Cutsem E, Sagaert X, Topal B, Haustermans K, Prenen H. Gastric cancer. Lancet. 2016;388:2654–2664. doi: 10.1016/S0140-6736(16)30354-3. [DOI] [PubMed] [Google Scholar]
- 477.Waddell T, Verheij M, Allum W, Cunningham D, Cervantes A, Arnold D, et al. Gastric cancer: ESMO-ESSO-ESTRO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24(Suppl 6):vi57–vi63. doi: 10.1093/annonc/mdt344. [DOI] [PubMed] [Google Scholar]
- 478.Basta YL, Baur OL, van Dieren S, Klinkenbijl JH, Fockens P, Tytgat KM. Is there a benefit of multidisciplinary cancer team meetings for patients with gastrointestinal malignancies? Ann Surg Oncol. 2016;23:2430–2437. doi: 10.1245/s10434-016-5178-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Basta YL, Bolle S, Fockens P, Tytgat KM. The value of multidisciplinary team meetings for patients with gastrointestinal malignancies: a systematic review. Ann Surg Oncol. 2017;24:2669–2678. doi: 10.1245/s10434-017-5833-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Du CZ, Li J, Cai Y, Sun YS, Xue WC, Gu J. Effect of multidisciplinary team treatment on outcomes of patients with gastrointestinal malignancy. World J Gastroenterol. 2011;17:2013–2018. doi: 10.3748/wjg.v17.i15.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 481.Oxenberg J, Papenfuss W, Esemuede I, Attwood K, Simunovic M, Kuvshinoff B, et al. Multidisciplinary cancer conferences for gastrointestinal malignancies result in measureable treatment changes: a prospective study of 149 consecutive patients. Ann Surg Oncol. 2015;22:1533–1539. doi: 10.1245/s10434-014-4163-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 482.Lamb BW, Sevdalis N, Arora S, Pinto A, Vincent C, Green JS. Teamwork and team decision-making at multidisciplinary cancer conferences: barriers, facilitators, and opportunities for improvement. World J Surg. 2011;35:1970–1976. doi: 10.1007/s00268-011-1152-1. [DOI] [PubMed] [Google Scholar]
- 483.Lamb BW, Jalil RT, Sevdalis N, Vincent C, Green JS. Strategies to improve the efficiency and utility of multidisciplinary team meetings in urology cancer care: a survey study. BMC Health Serv Res. 2014;14:377. doi: 10.1186/1472-6963-14-377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 484.Chinai N, Bintcliffe F, Armstrong EM, Teape J, Jones BM, Hosie KB. Does every patient need to be discussed at a multidisciplinary team meeting? Clin Radiol. 2013;68:780–784. doi: 10.1016/j.crad.2013.02.011. [DOI] [PubMed] [Google Scholar]
- 485.Allum WH, Blazeby JM, Griffin SM, Cunningham D, Jankowski JA, Wong R, et al. Guidelines for the management of oesophageal and gastric cancer. Gut. 2011;60:1449–1472. doi: 10.1136/gut.2010.228254. [DOI] [PubMed] [Google Scholar]
- 486.Kunkler IH, Prescott RJ, Lee RJ, Brebner JA, Cairns JA, Fielding RG, et al. TELEMAM: a cluster randomised trial to assess the use of telemedicine in multi-disciplinary breast cancer decision making. Eur J Cancer. 2007;43:2506–2514. doi: 10.1016/j.ejca.2007.08.026. [DOI] [PubMed] [Google Scholar]
- 487.Sundi D, Cohen JE, Cole AP, Neuman BP, Cooper J, Faisal FA, et al. Establishment of a new prostate cancer multidisciplinary clinic: format and initial experience. Prostate. 2015;75:191–199. doi: 10.1002/pros.22904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 488.Kelly SL, Jackson JE, Hickey BE, Szallasi FG, Bond CA. Multidisciplinary clinic care improves adherence to best practice in head and neck cancer. Am J Otolaryngol. 2013;34:57–60. doi: 10.1016/j.amjoto.2012.08.010. [DOI] [PubMed] [Google Scholar]
- 489.Pawlik TM, Laheru D, Hruban RH, Coleman J, Wolfgang CL, Campbell K, et al. Evaluating the impact of a single-day multidisciplinary clinic on the management of pancreatic cancer. Ann Surg Oncol. 2008;15:2081–2088. doi: 10.1245/s10434-008-9929-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 490.Santoso JT, Schwertner B, Coleman RL, Hannigan EV. Tumor board in gynecologic oncology. Int J Gynecol Cancer. 2004;14:206–209. doi: 10.1111/j.1048-891X.2004.014200.x. [DOI] [PubMed] [Google Scholar]
- 491.Borneman T, Koczywas M, Cristea M, Reckamp K, Sun V, Ferrell B. An interdisciplinary care approach for integration of palliative care in lung cancer. Clin Lung Cancer. 2008;9:352–360. doi: 10.3816/CLC.2008.n.051. [DOI] [PubMed] [Google Scholar]