Abstract
Introduction
Few studies have explored the impact of the COVID-19 pandemic on the eating behaviors, dietary quality, and changes in weight of postoperative bariatric surgery patients.
Methods
A cross-sectional survey on eating behaviors and attitudes toward food was emailed or given to patients who had bariatric surgery before March 2020. Patient charts were reviewed for weight measures.
Results
Seventy-five (71.43%) patients experienced weight recurrence with an average increase in body mass index (BMI) of 2.83 kg/m2 (SD: 2.19). The majority of patients reported no symptoms of binge eating (n = 81, 77.14%) with 16 (15.24%) qualifying for loss of control eating (LOCE). LOCE was significantly associated with grazing behavior (p = 0.04), emotional over-eating (p = 0.001), and food responsiveness (p = 0.002). LOCE was negatively associated with dietary quality (p = 0.0009) and satiety responsiveness (p = 0.01). Grazing behavior was significantly associated with emotional over-eating (p < 0.0001) and food responsiveness (p < 0.0001) as well as negatively associated with dietary quality (p < 0.0001). Slow eating was negatively associated with grazing (p = 0.01), emotional over-eating (p = 0.003), and food responsiveness (p < 0.0001). When included in a regression model controlling for age and sex, emotional over-eating was a significant predictor of weight recurrence (β = 0.25; p = 0.04).
Conclusion
Our results suggest that maladaptive eating behaviors contributed to LOCE and poor dietary quality during the COVID-19 pandemic; however, slow eating may be protective against grazing, emotional over-eating, and food responsiveness.
Graphical abstract

Supplementary Information
The online version contains supplementary material available at 10.1007/s11695-023-06460-2.
Keywords: Bariatric surgery, Surgical weight loss, Eating behaviors, Dietary quality, Loss of control eating, Weight recurrence, Adult eating behavior questionnaire, AEBQ
Introduction
COVID-19 was declared a worldwide pandemic by the World Health Organization in March 2020, and almost every aspect of daily life was disrupted. Orders for social distancing and quarantine were critical to slow down disease spread, but came at the expense and limitation of other liberties such as physical activity, in-person socialization, and engagement in normal daily activities. An international survey examined the effect of stay-at-home orders during COVID-19 in 1074 adults and found that there was increased consumption of unhealthy food, impulsive eating, and snacking [1]. In patients with a current or past history of obesity, recent studies have examined the psychological impact of the COVID-19 pandemic and found increased psychological distress associated with changes in dietary compliance, including increased frequency of snacking and impulsive eating [2–4]. Patients have reported difficulty in achieving weight loss goals and increased frequency of stress eating [3, 5].
In the context of the bariatric patient population, emotional dysregulation resulting in maladaptive eating behaviors can lead to undesirable long-term postoperative outcomes, such as weight recurrence (WR) [6–8]. Poor dietary quality has been reported in postoperative bariatric patients, and patients reported some degree of return of maladaptive behaviors, such as smoking or drinking more alcohol than usual [5]. Maladaptive eating behaviors have been associated with less weight loss and/or more WR in the postoperative bariatric population, and up to 40% of postoperative bariatric patients have been reported to develop loss of control eating (LOCE) by 2 years after postoperatively [9, 10].
Most of the current literature suggests that the pandemic has had detrimental effects on postoperative weight loss [11]. Conceição et al. (2021) conducted a cross-sectional study in Portugal of postoperative bariatric patients and found that the pandemic was associated with less total weight loss and larger WR at 3 years after surgery compared to patients whose postoperative course was completed before the pandemic [12]. Another cross-sectional study with 75 postoperative bariatric patients found that during the pandemic, higher reported levels of emotional distress during quarantine were associated with higher consumption of energy-dense micronutrient-poor foods, which was associated with higher odds of maintaining or increasing their body mass indices (BMI) [13].
There is a dearth of literature on the impact of maladaptive eating behaviors in the bariatric population in the context of the COVID-19 pandemic within a US population. The purpose of this study was to describe the pandemic’s impact on eating behaviors such as binge eating, grazing, LOCE, its effects on dietary quality, and the resulting changes in post-surgical weight of bariatric surgery patients.
Methods
Participants and Procedures
This cross-sectional study was conducted at a single academic medical center from October to December 2021 using REDCap [14]. The survey was emailed and distributed in clinic to those who underwent weight loss surgery prior to March 2020. Topics included demographics, eating behaviors, and dietary quality with additional variables abstracted from the electronic health record (EHR). Inclusion criteria were as follows: patient of the institution, adult (age 18 or older), able to access the internet, and underwent surgery prior to March 2020. Exclusion criteria included patients under 18 as well as those who had surgery after March of 2020 or never had bariatric surgery. Respondents were instructed to reflect on their dietary quality and eating behaviors since the start of the COVID-19 lockdown orders in March of 2020. Those who offered completed responses were entered into a raffle for one of four $50.00 gift cards.
Data Collection Instruments and Measures
Dietary Quality
Dietary quality was measured using the Rapid Eating Assessment for Participants (Short Version; REAP-S) [15], which has previously been used in samples including persons with obesity and those presenting for bariatric surgery [16, 17]. The REAP-S is a 16-item instrument that is designed to assess the quality of an individual’s diet by accounting for the frequency with which individuals consume certain foods or engage in certain behaviors on a 3-point scale. The first 13 of these items are included in the total REAP-S score with higher numbers indicating better dietary quality and a total score range of 13 to 39.
Eating Behaviors
Eating behaviors were evaluated using the Adult Eating Behaviors Questionnaire (AEBQ), which has been previously validated in a pre-bariatric patient sample [18, 19]. The AEBQ has 35 items and contains 8 subscales including hunger, food responsiveness, emotional overeating, enjoyment of food, satiety responsiveness, emotional undereating, food fussiness, and slowness in eating. Higher scores for the subscale indicated a stronger presence of the eating behavior. Pre-surgical AEBQ responses were also abstracted from the patient’s intake evaluation at the beginning of the program. Additionally, two items from the Weight and Lifestyle Inventory (WALI) were used to assess for binge eating along with LOCE [20]. These items are consistent with the Diagnostic and Statistical Manual of Mental Disorders—5th edition for binge-eating disorder (BED) diagnostic criteria [21]. The sum of these two items resulted in a 3-point categorical scale of no binge eating, binge eating, and LOCE. Lastly, a single item was presented to each respondent with a definition of “grazing.” Respondents were asked to identify how frequently they engaged in this style of eating using a 4-point ordinal scale of never to always.
Sociodemographic and Weight Data
A number of items were included at the end of the questionnaire to capture sociodemographic information including age, sex, race, ethnicity, education, alcohol use, and tobacco use. Additionally, weight and height data for participants were abstracted from the EHR to avoid the potential for recall bias. Each patient’s highest recorded weight, lowest recorded weight since surgery, pre-pandemic weight (measured at or within 2 months of March 2020), and their current weight at the time of survey completion were recorded along with their height to calculate BMI. All weights were measured in kilograms (kg) and BMI in kg/m2.
Statistical Analyses
Statistical analyses were conducted using SAS Version 9.4 (SAS Institute Inc., Cary, NC). Missing data was minimal with ranges of 0.00 to 8.57%. Variables with missing data were excluded from analyses. Summary statistics were used to characterize the sample along with the instrument scores. Pre-surgical AEBQ scores were compared to current scores using paired means Student’s T-test to compare scores for those respondents who completed both time points. A linear multivariable stepwise regression model was built predicting the amount of WR from the patient’s lowest recorded weight. All continuous predictor variables were standardized using the sample standard deviation (SD). The significance levels for stay and entry were set to 0.15. Due to limits for the sample size, only the AEBQ subscales were included in the stepwise model and controlled for age and sex. A correlation matrix was also built using Spearman’s correlation coefficients with rho values from 0 to 0.29 considered poor, values from 0.30 to 0.60 considered fair, and values from 0.61 to 0.80 considered strong [22].
Results
Across all recruitment methods, 124 participants completed part or all of the survey. A total response rate could not be calculated as the total number of participants who were recruited in clinic and saw the institutional recruitment boards could not be ascertained. Of those 124 individuals, 6 (4.84%) declined to participate after reading the study summary, 12 (9.68%) responses were incomplete and deemed to be a withdrawal of consent, and 1 (0.81%) was a duplicate response. The total number of complete responses included in these analyses is 105 (84.68%) with sample characteristics presented in Table 1. Informed consent was obtained from all individual participants included in the study.
Table 1.
Sample characteristics
| Variable | (N = 105) |
|---|---|
| Ageμ | 47.95 (12.48) [25, 78] |
| Highest BMIμ | 48.48 (8.11) [35.22, 70.83] |
| Lowest BMIμ | 34.37 (7.62) [19.85, 61.38] |
| March BMIμ (n = 39) | 34.96 (8.11) [22.85, 61.38] |
| Current BMIμ | 36.61 (8.29) [25.40, 69.53] |
| Change in BMI from highestμ | − 11.87 (6.29) [− 33.08, 0.82] |
| Change in BMI from Marchμ | 0.30 (3.08) [− 5.61, 8.15] |
| %EWL from highest (in kg) | 66.94 (25.79) [− 3.34, 98.13] |
| %TWL from highest (in kg) | 24.25 (11.53) [− 1.65, 49.21] |
| Average amount of BMI recurrence (n = 75)μ | 2.83 (2.19) [0.07, 11.70] |
| Average amount of BMI recurrence since March 2020 (N = 22)μ | 2.43 (1.77) [0.14, 8.15] |
| Average time since surgery (months) | 40.33 (20.90) [16, 200] |
| Sex | |
| Male | 22 (21.36) |
| Female | 81 (78.64) |
| Race | |
| Black/African American | 2 (1.90) |
| White/Caucasian | 101 (96.19) |
| Other/decline to answer | 2 (1.90) |
| Ethnicity | |
| Hispanic/Latino | 1 (0.98) |
| Non-Hispanic/Latino | 98 (96.08) |
| Decline to answer | 3 (2.94) |
| Education | |
| High school diploma/ GED | 30 (28.57) |
| Associate’s, some college, or trade | 38 (36.19) |
| Bachelor’s degree | 22 (20.95) |
| Graduate | 14 (13.33) |
| Decline to answer | 1 (0.94) |
| Alcohol use | |
| No Alcohol | 38 (36.54) |
| 1 drink or less per month | 32 (30.77) |
| 2–3 drinks per month | 15 (14.42) |
| 1–2 drinks per week | 9 (8.65) |
| 3–6 drinks per week | 5 (4.81) |
| 1 drink per day | - |
| More than 1 drink per day | 5 (4.81) |
| Smoking | |
| Current | 8 (7.62) |
| Former | 29 (27.62) |
| Never | 68 (64.76) |
BMI, body mass index (kg/m2); %EWL, percent excess weight loss; %TWL, percent total weight loss; GED, graduate educational development
Data are presented as frequencies (N) and percents (%) unless indicated with (μ), which are presented as means and standard deviations. %EWL was calculated taking the difference between the patient’s highest weight and current weight over the calculated excess body weight using a reference of BMI equal to 25. %BWL was calculated taking the difference between the patient’s highest weight and current weight over their highest weight
Of these, the majority of the sample were female (n = 81, 78.64%), White/Caucasian (n = 101, 96.19%), and non-Hispanic/Latino (n = 98; 96.08%). There was an average reduction of total BMI of − 11.87 kg/m2 (SD: 6.29) from the highest recorded weight. Of those who responded, 75 (71.43%) experienced WR with an average increase in BMI of 2.83 kg/m2 (SD: 2.19) and a total range from 0.07 to 11.70 kg/m2. In total, 39 (37.14%) had a recorded weight at or around March 2020. Of these individuals, 22 (56.41%) experienced WR with an average increase in BMI of 2.43 kg/m2 (SD: 1.77) and a range of 0.14 to 8.15 kg/m2. The average time since surgery was 40.33 (SD: 20.90) months with a range of 16 to 200 months.
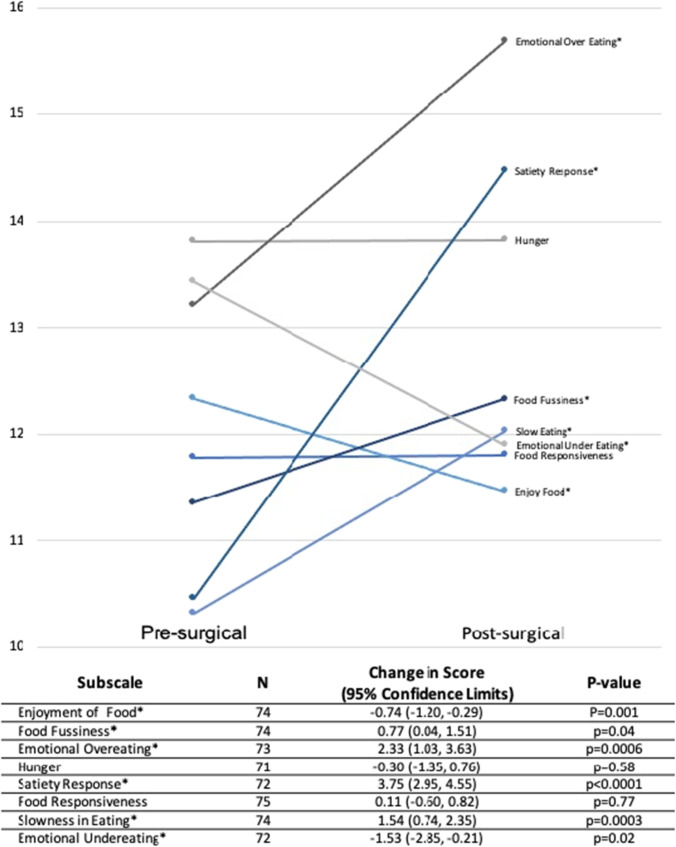
In terms of instrument scores (Table 2), the majority of patients reported no symptoms of binge eating (n = 81; 77.14%) with 16 (15.24%) qualifying for LOCE. An equal number of respondents (n = 43; 40.95%) indicated that they engage in grazing eating behaviors “sometimes” or “often.” Mean (SD) dietary quality scores were fair at 28.65 (4.32). Some of the highest mean subscales scores for the AEBQ were emotional overeating (15.69 [5.38]), satiety responsiveness (14.48 [2.98]), and hunger (13.82 [3.63]). Figure 1 shows the average change in AEBQ subscale scores from the patients’ presurgical assessments. Paired mean analyses indicated that satiety responsiveness had the highest mean increase of 3.75 with a 95% confidence limits (95% CL) of [2.95, 4.55]; p < 0.0001. Emotional overeating and slowness in eating scores also significantly increased by 2.33 [1.03, 3.63] points (p < 0.0001) and 1.54 [0.74,2.35] (p = 0.0003), respectively.
Table 2.
Eating behavior instrument scores
| N(%) | |
|---|---|
| Binge eating (n = 105) | |
| None | 81 (77.14) |
| Binge Eating | 8 (7.62) |
| LOCE | 16 (15.24) |
| Grazing (n = 105) | |
| Never | 8 (7.62) |
| Sometimes | 43 (40.95) |
| Often | 43 (40.95) |
| Always | 11 (10.48) |
|
Mean (SD); Total possible score range |
|
| REAPS (n = 96) | 28.65 (4.32); [13–39] |
| AEBQ | |
| Enjoyment of food (n = 103) | 11.47 (2.33); [3–15] |
| Food fussiness (n = 103) | 12.33 (4.53); [5–25] |
| Emotional overeating (n = 104) | 15.69 (5.38); [5–25] |
| Hunger (n = 100) | 13.82 (3.63); [5–25] |
| Satiety response (n = 104) | 14.48 (2.98); [4–20] |
| Food responsiveness (n = 104) | 11.81 (3.76); [4–20] |
| Slow eating (n = 104) | 12.03 (3.71); [4–20] |
| Emotional undereating (n = 102) | 11.90 (4.56); [5–25] |
LOCE, loss of control eating; SD, standard deviation; REAP-S, rapid eating assessment for participants—short version; AEBQ, adult eating behavior questionnaire
Fig. 1.
Plotted mean scores represent all pre- and post-questionnaire responses. Paired T-test was conducted for those who completed both assessments. *Indicates statistically significant change in scores from pre-surgical assessments p < 0.05
Table 3 shows the results of the stepwise linear regression model including the eight AEBQ subscales and WR predictions. After controlling for age and sex, only the emotional overeating subscale remained in the model with a parameter estimate of 1.69 and a p-value of 0.01 with a significant model p-value of 0.02. Thus, emotional overeating was significantly associated with WR after controlling for age and sex with an increase of 1.69 kg for every increase in SD of the emotional overeating score.
Table 3.
Stepwise multivariable linear regression model predicting weight recurrence (n = 91)
| Variable | Parameter estimate (SE) | p-value |
|---|---|---|
| Age | − 0.73 (0.66) | 0.28 |
| Sex | − 3.13 (1.65) | 0.06 |
| Emotional overeating | 1.69 (0.65) | 0.01* |
Full model included all subscales of the Adult Eating Behavior Questionnaire and controlled for age and sex. Continuous independent variables were standardized using the sample standard deviation for that specific variable. Model was significant at p = 0.02
SE, standard error
*p < 0.05
Univariable analyses are presented in Table 4 and demonstrated that without controlling for other variables, WR was most strongly correlated with emotional overeating, though this association was non-significant. In addition, emotional overeating was positively correlated with hunger (r = 0.27), binge eating (r = 0.34), grazing (r = 0.41), enjoyment of food (r = 0.44), and food responsiveness (r = 0.51) as well as negatively correlated with slow eating (r = − 0.28), dietary quality (r = − 0.35), satiety responsiveness (r = − 0.37), and emotional undereating (r = − 0.37). Satiety responsiveness was also negatively correlated with food responsiveness (r = − 0.50) and positively associated with slow eating (r = 0.42). Food responsiveness was also positively associated with hunger (r = 0.38), grazing (r = 0.52), and enjoyment of food (r = 0.67).
Table 4.
Spearman Correlation Matrix
| Binge eating | Grazing | REAP-S | Enjoyment of food | Food fussiness | Emotional overeating | Hunger | Satiety response | Food responsiveness | Slowness in eating | Emotional undereating | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Weight recurrence^ | 0.07 | 0.08 | − 0.04 | 0.09 | − 0.04 | 0.17 | − 0.12 | − 0.06 | 0.08 | 0.02 | − 0.13 |
| Binge eating | 0.21* | − 0.36** | 0.27** | − 0.03 | 0.34** | 0.16 | − 0.22* | 0.29** | − 0.15 | − 0.08 | |
| Grazing | − 0.49*** | 0.34** | 0.05 | 0.41*** | 0.15 | − 0.33** | 0.52*** | − 0.23* | − 0.12 | ||
| REAP-S | − 0.23* | − 0.25* | − 0.35** | − 0.16 | 0.15 | − 0.33** | 0.11 | 0.08 | |||
| Enjoyment of food | 0.01 | 0.44*** | 0.30** | − 0.41*** | 0.67*** | − 0.26** | − 0.17 | ||||
| Food fussiness | 0.19 | 0.23* | 0.02 | − 0.02 | − 0.09 | − 0.01 | |||||
| Emotional overeating | 0.27** | − 0.37*** | 0.51*** | − 0.28** | − 0.37** | ||||||
| Hunger | − 0.11 | 0.38*** | − 0.16 | 0.19 | |||||||
| Satiety response | − 0.50*** | 0.42*** | 0.31** | ||||||||
| Food responsiveness | − 0.38*** | − 0.15 | |||||||||
| Slow eating | 0.32** |
REAP-S, rapid eating assessment for participants—short form
^Measured in kg from the lowest recorded weight
*p < 0.05; **p < 0.01; ***p < 0.0001
Discussion
Our study found that approximately 15% of postoperative patients reported clinical symptoms of LOCE—a central component of BED. Previous studies have estimated the prevalence of eating disorders to be about 7.83% among post-surgical bariatric patients [23]. The relationship between maladaptive eating disorders and postoperative WR has been established in the pre-pandemic context, and LOCE is a negative prognostic indicator for long-term weight outcomes [5–10]. However, the literature is rather heterogenous in regards to the type of bariatric surgery performed, various types of assessment, differing definitions of weight outcomes (BMI, % excess weight loss, WR), and variable definitions of LOCE particularly in self-reported assessment measures [10, 24]. Our study sought to examine these relationships in the context of the COVID-19 pandemic.
In line with the available literature, our results suggest that maladaptive eating behaviors may have contributed to LOCE and poor dietary quality in a subset of the bariatric patient population during the COVID-19 pandemic [25]. In our study, LOCE was significantly associated with grazing behavior, emotional over-eating, and food responsiveness. Both LOCE and grazing behavior were negatively associated with dietary quality, and LOCE was also negatively associated with satiety responsiveness. It has been established that consuming foods with higher micronutrient density is associated with long-term weight loss maintenance [26]. These factors may have contributed to the WR experienced by more than half of the postoperative bariatric patients in our study, as 75 (71.43%) patients experienced WR and 81 (77.14%) experienced binge eating with 16 (15.24%) of those experiencing LOCE. These issues present opportunities for clinicians and bariatric programs to counsel patients regarding triggers for LOCE in both pre-operative and postoperative settings as a form of preventative medicine, particularly during the COVID-19 pandemic in which LOCE may be exacerbated.
Furthermore, our cohort displayed more “eating off-set” traits according to the AEBQ than “eating onset” traits [19]. “Eating off-set traits” include satiety response, emotional under-eating, food fussiness, and slowness in eating, while “eating onset” traits include hunger, food responsiveness, emotional over-eating, and enjoyment of food [27]. Patients reported increased satiety response, food fussiness, and slow eating in the peri-COVID time frame compared to the pre-surgical time frame, which may be due to the ramifications of bariatric surgery, such as the restrictive component likely leading to earlier satiety. However, despite increases in these “eating off-set” traits, emotional over-eating was still significantly associated with WR after controlling for age and sex. Hunger and food responsiveness remained the same over time while food enjoyment experienced by our patients in this study decreased with time. Clinicians may find it beneficial to counsel patients on triggers for emotional overeating and its relationship with WR.
Most notably, our study found that slow eating was negatively associated with grazing, emotional overeating, and food responsiveness. Consequently, eating meals slowly may be protective against these maladaptive eating behaviors that may encourage WR. Slow eating strategies have been described in literature as a way to maximize satiation, reduce energy intake within meals, and aid with body weight regulation [28, 29]. However, there is limited literature on slow eating strategies in postoperative bariatric patients. These results highlight the clinical and practical importance to encourage slow eating for postoperative bariatric patients in efforts to reduce maladaptive eating behaviors even outside of the context of the COVID-19 pandemic. Additionally, our patients experienced increased satiety response over time, which is also beneficial in conjunction with eating meals slowly.
Limitations
Limitations of this study include a small sample size. The limited sample size, single center, and demographics of the respondents may limit the generalizability of these findings. Furthermore, the directionality of these associations is not definitive given the cross-sectional design of the study. Additionally, as this study was issued by the surgical weight loss program where these participants sought care and was self-reported, respondents may engage in impression management, resulting in response bias. Lastly, interpretations of definitions and the assessment of grazing and binge-eating behaviors may not be consistent as the “objectively large” criterion may not be uniform across all bariatric patients [30]. One factor that was not accounted for was type of bariatric surgery, which may or may not impact eating behaviors in the context of the pandemic.
Conclusions
These findings highlight that significant social adversity (such as that experienced during the COVID-19 pandemic) can have a negative impact on eating behaviors of postoperative bariatric patients. Clinicians should be aware of these stressors and counsel patients to utilize adaptive eating behaviors to maintain weight loss from bariatric surgery. In order to assess better the relationship between LOCE and WR in bariatric surgery patients, future studies should be designed to include pre-operative data on the presence of existing or a prior history of eating behavior disorders.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
This research was presented at the June 2022 Association for Metabolic and Bariatric Surgery (ASMBS) annual conference in Dallas, TX.
Funding
MB was supported by the Health Resources and Services Administration (HRSA) of the US Department of Health and Human Services (HHS) as part of an award totaling $473,925 with 15% financed with non-governmental sources. The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement, by HRSA, HHS, or the US government. For more information, please visit HRSA.gov.
Data Availability
Data are not available due to regulatory restrictions.
Declarations
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of Interest
AHu has no conflict of interest. AHarvey has no conflict of interest. AMR has received honoraria from Intuitive Surgical, Medtronic, Ethicon Endosurgery, and WL Gore. All other authors have no financial disclosures. AR has no conflict of interest. MB has no conflict of interest.
Footnotes
Key Points
• The majority of patients experienced weight recurrence.
• Loss of control eating was significantly associated with emotional over-eating.
• When included in a regression model controlling for age and sex, emotional over-eating was a significant predictor of weight recurrence.
• Slow eating may be protective against grazing, emotional over-eating and food responsiveness.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ammar A, Brach M, Trabelsi K, et al. Effects of COVID-19 home confinement on eating behaviour and physical activity: results of the ECLB-COVID19 international online survey. Nutrients. 2020;12(6):1583. doi: 10.3390/nu12061583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sisto A, Vicinanza F, Tuccinardi D, et al. The psychological impact of COVID-19 pandemic on patients included in a bariatric surgery program. Eat Weight Disord. 2021;26(6):1737–1747. doi: 10.1007/s40519-020-00988-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Almandoz JP, Xie L, Schellinger JN, et al. Impact of COVID-19 stay-at-home orders on weight-related behaviours among patients with obesity. Clin Obes. 2020;10(5):e12386. doi: 10.1111/cob.12386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sockalingam S, Leung SE, Cassin SE. The impact of coronavirus disease 2019 on bariatric surgery: redefining psychosocial care. Obesity (Silver Spring) 2020;28(6):1010–1012. doi: 10.1002/oby.22836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Athanasiadis DI, Hernandez E, Hilgendorf W, et al. How are bariatric patients coping during the coronavirus disease 2019 (COVID-19) pandemic? Analysis of factors known to cause weight regain among postoperative bariatric patients. Surg Obes Relat Dis. 2021;17(4):756–764. doi: 10.1016/j.soard.2020.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Taube-Schiff M, Van Exan J, Tanaka R, et al. Attachment style and emotional eating in bariatric surgery candidates: the mediating role of difficulties in emotion regulation. Eat Behav. 2015;18:36–40. doi: 10.1016/j.eatbeh.2015.03.011. [DOI] [PubMed] [Google Scholar]
- 7.Shakory S, Van Exan J, Mills JS, et al. Binge eating in bariatric surgery candidates: the role of insecure attachment and emotion regulation. Appetite. 2015;91:69–75. doi: 10.1016/j.appet.2015.03.026. [DOI] [PubMed] [Google Scholar]
- 8.Nasirzadeh Y, Kantarovich K, Wnuk S, et al. Binge eating, loss of control over eating, emotional eating, and night eating after bariatric surgery: results from the Toronto Bari-PSYCH cohort study. Obes Surg. 2018;28(7):2032–2039. doi: 10.1007/s11695-018-3137-8. [DOI] [PubMed] [Google Scholar]
- 9.Conceição EM, Mitchell JE, Pinto-Bastos A, et al. Stability of problematic eating behaviors and weight loss trajectories after bariatric surgery: a longitudinal observational study. Surg Obes Relat Dis. 2017;13(6):1063–1070. doi: 10.1016/j.soard.2016.12.006. [DOI] [PubMed] [Google Scholar]
- 10.Meany G, Conceição E, Mitchell JE. Binge eating, binge eating disorder and loss of control eating: effects on weight outcomes after bariatric surgery. Eur Eat Disord Rev. 2014;22(2):87–91. doi: 10.1002/erv.2273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Barranquero AG, Cimpean S, Raglione D, et al. Impact of the COVID-19 pandemic and lockdown on gastric bypass results at 1-year follow-up. Obes Surg. 2021;31(10):4511–4518. doi: 10.1007/s11695-021-05640-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Conceição E, de Lourdes M, Ramalho S, et al. Eating behaviors and weight outcomes in bariatric surgery patients amidst COVID-19. Surg Obes Relat Dis. 2021;17(6):1165–1174. doi: 10.1016/j.soard.2021.02.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Durão C, Vaz C, de Oliveira VN, et al. Confinement during the COVID-19 pandemic after metabolic and bariatric surgery-associations between emotional distress, energy-dense foods, and body mass index. Obes Surg. 2021;31(10):4452–4460. doi: 10.1007/s11695-021-05608-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap): a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Segal-Isaacson CJ, Wylie-Rosett J, Gans KM. Validation of a short dietary assessment questionnaire: the rapid eating and activity assessment for participants short version (REAP-S) Diabetes Educ. 2004;30(5):774–781. doi: 10.1177/014572170403000512. [DOI] [PubMed] [Google Scholar]
- 16.Al-Nimr RI, Wright KCS, Aquila CL, et al. Intensive nutrition counseling as part of a multi-component weight loss intervention improves diet quality and anthropometrics in older adults with obesity. Clin Nutr ESPEN. 2020;40:293–299. doi: 10.1016/j.clnesp.2020.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lin D, Zickgraf H, Butt M, et al. Food insecurity is linked to poorer dietary quality in prebariatric surgery patients. Surg Obes Relat Dis. 2021;17(2):263–270. doi: 10.1016/j.soard.2020.10.013. [DOI] [PubMed] [Google Scholar]
- 18.Hunot C, Fildes A, Croker H, et al. Appetitive traits and relationships with BMI in adults: development of the Adult Eating Behaviour Questionnaire. Appetite. 2016;105:356–363. doi: 10.1016/j.appet.2016.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zickgraf HF, Rigby A. The Adult Eating Behaviour Questionnaire in a bariatric surgery-seeking sample: factor structure, convergent validity, and associations with BMI. Eur Eat Disord Rev. 2019;27(1):97–104. doi: 10.1002/erv.2628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wadden TA, Foster GD. Weight and Lifestyle Inventory (WALI) Surg Obes Relat Dis. 2006;2(2):180–199. doi: 10.1016/j.soard.2006.03.017. [DOI] [PubMed] [Google Scholar]
- 21.American Psychiatric Association (2022) Diagnostic and statistical manual of mental disorders, 5th edn., text rev. 10.1176/appi.books.9780890425787
- 22.Akoglu H. User’s guide to correlation coefficients. Turk J Emerg Med. 2018;18(3):91–93. doi: 10.1016/j.tjem.2018.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Taba JV, Suzuki MO, Nascimento FSD, et al. The development of feeding and eating disorders after bariatric surgery: a systematic review and meta-analysis. Nutrients. 2021;13(7):2396. doi: 10.3390/nu13072396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ivezaj V, Carr MM, Brode C, et al. Disordered eating following bariatric surgery: a review of measurement and conceptual considerations. Surg Obes Relat Dis. 2021;17(8):1510–1520. doi: 10.1016/j.soard.2021.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Devlin MJ, King WC, Kalarchian MA, et al. Eating pathology and experience and weight loss in a prospective study of bariatric surgery patients: 3-year follow-up. Int J Eat Disord. 2016;49(12):1058–1067. doi: 10.1002/eat.22578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pascual RW, Phelan S, La Frano MR, et al. Diet quality and micronutrient intake among long-term weight loss maintainers. Nutrients. 2019;11(12):3046. doi: 10.3390/nu11123046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hunot-Alexander C, Arellano-Gómez LP, Smith AD, et al. Examining the validity and consistency of the Adult Eating Behaviour Questionnaire-Español (AEBQ-Esp) and its relationship to BMI in a Mexican population. Eat Weight Disord. 2022;27:651–663. doi: 10.1007/s40519-021-01201-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Andrade AM, Greene GW, Melanson KJ. Eating slowly led to decreases in energy intake within meals in healthy women. J Am Diet Assoc. 2008;108(7):1186–1191. doi: 10.1016/j.jada.2008.04.026. [DOI] [PubMed] [Google Scholar]
- 29.Simonson AP, Davis KK, Barone Gibbs B, et al. Comparison of mindful and slow eating strategies on acute energy intake. Obes Sci Pract. 2020;6(6):668–676. doi: 10.1002/osp4.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mitchell JE, King WC, Courcoulas A, et al. Eating behavior and eating disorders in adults before bariatric surgery. Int J Eat Disord. 2015;48(2):215–222. doi: 10.1002/eat.22275. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are not available due to regulatory restrictions.