Abstract
Background
In cattle attempts to evaluate within-herd prevalence of various infectious and parasitic diseases by bulk-tank milk (BTM) testing with ELISA have been made with moderate success. The fact that BTM is composed of variable and unknown volumes of milk from individual lactating animals weakens the relationship between numerical result of the ELISA and the within-herd prevalence. We carried out a laboratory experimental study to evaluate if a pooled milk sample created by mixing an equal volume of individual milk samples from seropositive and seronegative goats, henceforth referred to as an equal-volume milk sample (EVMS), would allow for accurate estimation of within-herd seroprevalence of caprine arthritis-encephalitis (CAE) using 3 different commercial ELISAs. By mixing randomly selected milk samples from seronegative and seropositive goats, 193 EVMS were created – 93 made of seronegative samples and 100 with the proportion of seropositive individual milk samples (EVMS%POS) ranging from 1 to 100%. EVMS%POS could be considered as a proxy for the within-herd seroprevalence. Then, OD of EVMS (ODEVMS) of the 193 EVMS was measured using 3 commercial ELISAs for CAE – 2 indirect and 1 competitive.
Results
The cut-off values of ODEVMS indicating SRLV infection were determined. The regression functions were developed to link ODEVMS with EVMS%POS. A significant monotonic relationship between ODEVMS measured with 2 commercial indirect ELISAs and EVMS%POS was identified. Two regression models developed on this basis described approximately 90% of variability and allowed to estimate EVMS%POS, when it was below 50%. High ODEVMS indicated EVMS%POS of > 50%.
Conclusion
Our study introduces the concept of serological testing of EVMS as a method of detecting SRLV-infected herds and estimating the proportion of strongly seropositive goats. Further field studies are warranted to assess practical benefits of EVMS serological testing.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12917-023-03599-z.
Keywords: Bulk-tank milk, Caprine arthritis-encephalitis, Competitive ELISA, Indirect ELISA, Within-herd seroprevalence
Background
Caprine arthritis-encephalitis (CAE) caused by small ruminant lentivirus (SRLV) is one of the major health problems of goat population worldwide. SRLV infection is lifelong and in a proportion of goats progresses into a symptomatic form, with chronic arthritis being the most common clinical manifestation. Antibodies specific to SRLV are usually produced 4 to 12 weeks post infection, rarely later [1, 2], and remain detectable for life although their levels are known to fluctuate [3–5]. It makes serology the mainstay of CAE diagnostics [6, 7]. Serum testing is considered the optimal diagnostic modality for CAE control programs [8]. Testing of milk samples instead of blood eliminates animal stress as well as reduces costs associated with specimen collection. Therefore, several studies have evaluated diagnostic accuracy of serological milk testing [8–15]. Their results indicate high diagnostic accuracy and high agreement between qualitative results obtained on milk and serum. This makes milk testing a screening method worthy of consideration in dairy goat herds.
Regardless of the disease in question and the biological material used, serological screening of the herd is an expensive procedure as it requires a representative group of animals be properly selected and sampled [16]. This fact has given rise to the idea of serological testing of a bulk tank milk (BTM) sample. This approach is currently used in cattle for screening herds for various viral [17–20], bacterial [21, 22], and parasitic diseases [23–28]. In goats, serological BTM testing has so far been evaluated in terms of CAE, caseous lymphadenitis [29], toxoplasmosis [30], paratuberculosis [31], and Q fever [32, 33]. Estimations of diagnostic accuracy of serological BTM testing vastly depend on the reference standard used. Generally, in the aforementioned studies diagnostic specificity usually outraced sensitivity [29] and higher sensitivities were observed when serological rather than molecular reference standards were used [31]. Serological BTM testing for CAE was shown to be moderately accurate in detecting herds with at least 2% within-herd seroprevalence with the area under ROC curve of 80%. At an optimal cut-off value the procedure was roughly 73% sensitive and 84% specific [29].
The intensity of color reaction in ELISA performed on BTM sample, both raw and corrected optical density (OD), is known to correlate with the within-herd prevalence of infections in lactating individuals. As a consequence, quantitative ELISA results obtained on BTM samples have been used to estimate the within-herd prevalence of infections with bovine viral diarrhea virus (BVDV) [34, 35], bovine herpesvirus type 1 (BHV-1) [36–38], bovine leukemia virus (BLV) [39], and F. hepatica infection in cattle [23, 24], and T. gondii infection in goats [30]. However, the contribution of individual animals to the seroreactivity of a BTM sample depends on the volume of milk they yield and the concentration of antibodies in individual milk samples. As these two variables are not only beyond examiner’s influence but usually also remain unknown, estimations made on BTM samples have been shown to be rather imprecise. The former source of variability can be easily eliminated by creating an artificial milk sample by mixing an equal volume of milk from each animal. A recent study has shown that serological testing of this type of pooled milk samples from only 10 first-lactation cows in a herd yields more accurate result than BTM testing in terms of lungworm diagnosis [40]. This study, however, evaluated qualitative test results. On the other hand, Mazzei et al. [41] showed that the correlation between corrected OD of a pooled milk sample and the share of sheep milk seropositive for maedi-visna disease in this sample was almost perfect (coefficient of determination (R2) = 0.98). However, in this study the second source of variability was also removed by constant diluting a milk sample from the same seropositive sheep in the same seronegative milk sample (made by pooling BTM samples from 3 seronegative herds). Such perfect conditions preclude using the regression formula derived in their article to estimate the within-herd prevalences in field conditions.
Therefore, we carried out a laboratory experimental study to evaluate if testing a pooled milk sample created by mixing an equal volume of individual milk samples selected randomly from seropositive and seronegative goats (henceforth referred to as an equal-volume milk sample, EVMS) using three different commercial ELISAs would accurately estimate the proportion of milk samples coming from SRLV-seropositive goats.
Results
The list of 193 EVMS along with their optical density (ODEVMS) and the proportion of seropositive individual milk samples in EVMS (EVMS%POS) are presented in Table S1.
Negative EVMS
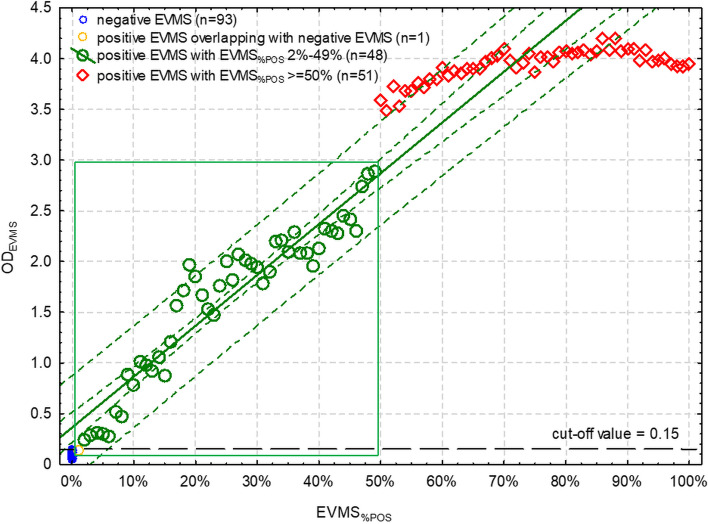
ODEVMS of sp-iELISA ranged from 0.052 to 0.143 with the arithmetic mean (SD) of 0.070 (0.021) and significantly non-normal (p < 0.001) and right-hand skewed distribution (coefficient of skewness [CoS] = 2.07, CI 95%: 1.58 – 2.56). The cut-off value of ODEVMS above which EVMS should be classified as positive was set at 0.15. One positive EVMS (with EVMS%POS = 1%) overlapped with negative EVMS and therefore 99 EVMS with ODEVMS ≥ 0.15 were further investigated for the relationship between ODEVMS and EVMS%POS.
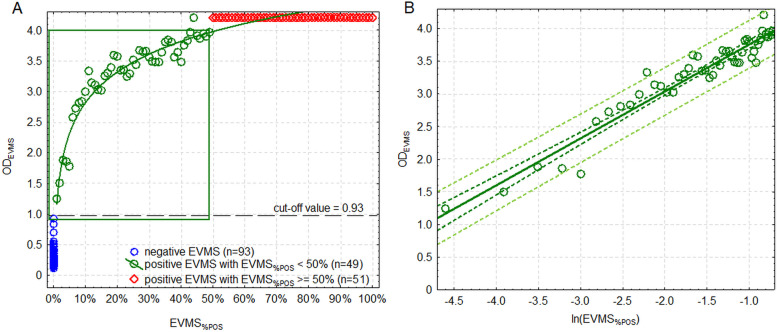
ODEVMS of TM/CA-iELISA ranged from 0.118 to 0.928 with the arithmetic mean (SD) of 0.290 (0.141) and significantly non-normal (p < 0.001) and right-hand skewed distribution (CoS = 2.09, CI 95%: 1.60 – 2.58). The cut-off value of ODEVMS above which EVMS should be classified as positive was set at 0.93. No positive EVMS overlapped with negative EVMS and therefore all 100 EVMS with ODEVMS ≥ 0.93 were further investigated for the relationship between ODEVMS and EVMS%POS.
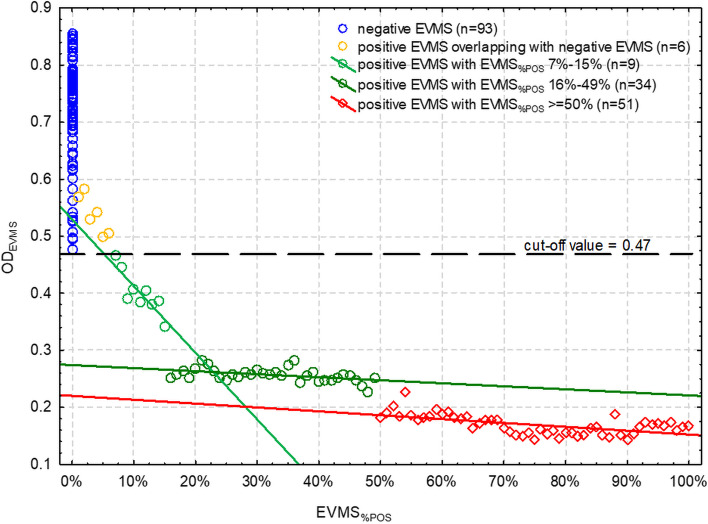
ODEVMS of SU-cELISA ranged from 0.476 to 0.855 with the arithmetic mean (SD) of 0.719 (0.092) and significantly non-normal (p < 0.001) and left-hand skewed distribution (CoS = -1.04, CI 95%: -1.53 – -0.55). The cut-off value of ODEVMS below which EVMS should be classified as positive was set at 0.47. Six positive EVMS (with EVMS%POS from 1 to 6%) overlapped with negative EVMS and therefore 94 EVMS with ODEVMS ≤ 0.47 were further investigated for the relationship between ODEVMS and EVMS%POS.
The aforementioned cut-off values of ODEVMS ensured 100% diagnostic specificity (95% confidence interval [CI 95%]: 96.0% – 100%) of EVMS testing. However, diagnostic sensitivity of EVMS testing certainly was lower and was mainly affected by the analytical sensitivity of the ELISA which was not evaluated in this study. Therefore, EVMS testing yields results with high positive predictive value and positive result is highly trustworthy. However, EVMS testing should never be used for ruling SRLV infection out as the negative predictive value remains unknown. The study showed that low EVMS%POS had ODEVMS overlapping with ODEVMS from negative EVMS.
sp-iELISA on positive EVMS
The scatter plot showed a distinction in ODEVMS between 99 samples with different EVMS%POS (Fig. 1). ODEVMS showed a gradual increase that was linearly linked to increasing EVMS%POS (r = 0.946, CI 95%: 0.905 – 0.970; p < 0.001). This trend could be observed up to the EVMS%POS of 50%, where the linear trend became significantly weaker (r = 0.725, CI 95%: 0.562 – 0.834; p < 0.001). At EVMS%POS ≥ 50% ODEVMS was > 3.0.
Fig. 1.
The relationship between the proportion of individual seropositive milk samples in the equal-volume milk sample (EVMS%POS) and optical density of EVMS (ODEVMS) in the indirect ELISA based on a mixture of synthetic viral peptides (sp-iELISA). The inner green broken line is a 95% confidence interval of the linear regression (green solid line). The outer green broken line is a 95% prediction interval. Green rectangle depicts the range of ODEVMS within which the model allows for estimation of EVMS%POS. Black broken line is a cut-off separating positive and negative EVMS
The EVMS model based on sp-iELISA and applied to ODEVMS between 0.15 and 3.0 fit the data well (F1,46 = 390.8, p < 0.001) and was described by the following equation:
Its parameters are given in Table 1 and R2 was 0.895. Standardized residuals were normally distributed (p = 0.213) and homoscedasticity was retained (p = 0.097).
Table 1.
Parameters of two equal-volume milk sample (EVMS) models based on optical density (OD) measured using two indirect ELISAs
| Variable | Regression coefficient (standard error) | 95% confidence interval of the regression coefficient | p-value |
|---|---|---|---|
| EVMS model based on sp-iELISA | |||
| Intercept | 0.363 (0.074) | 0.0.215 – 0.512 | - |
| EVMS%POS | 5.015 (0.254) | 4.504 – 5.526 | < 0.001 |
| EVMS model based on TM/CA-iELISA | |||
| Intercept | 4.477 (0.055) | 4.367 – 4.587 | - |
| ln(EVMS%POS) | 0.719 (0.029) | 0.661 – 0.778 | < 0.001 |
ELISA used: sp-iELISA – Indirect ELISA based on a mixture of synthetic viral peptides, TM/CA-iELISA – Indirect ELISA based on recombinant transmembrane and capsid proteins
The relationship between ODEVMS measured using sp-iELISA and EVMS%POS in the entire range of possible OD values was as follows:
TM/CA-iELISA on positive EVMS
Also, in terms of this ELISA, the scatter plot showed a distinction in ODEVMS between samples with different EVMS%POS (Fig. 2). No positive EVMS overlapped with negative ones. Then, ODEVMS showed a gradual logarithmic increase that was linked to increasing EVMS%POS (after linearization using a logarithmic transformation of EVMS%POS: r = 0.964, CI 95%: 0.937 – 0.980). The logarithmic relationship could be observed up to the EVMS%POS of 50% where the trend disappeared and the function became constant with ODEVMS > 4.0.
Fig. 2.
A (left): The relationship between the proportion of seropositive individual milk samples in the equal-volume milk sample (EVMS%POS, showed in logarithmic transformation) and optical density of EVMS (ODEVMS) in indirect ELISA based on recombinant transmembrane and capsid proteins (TM/CA-iELISA). Green rectangle depicts the range of ODEVMS in which the model allows for estimation of EVMS%POS; B (right): Linearized regression function from the range presented in a green rectangle in the Fig. 2 A. Dark green broken line is a 95% confidence interval of the linear regression (green solid line). Light green broken line is a 95% prediction interval. Black broken line is a cut-off separating positive and negative EVMS
To illustrate the relationship between ODEVMS and EVMS%POS, EVMS%POS from the range of 1% to 49% was logarithmically transformed (natural logarithm). The EVMS model based on TM/CA-iELISA and applied to ODEVMS between 0.93 and 4.0 fit the data well (F1,47 = 610.4, p < 0.001) and was described by the following equation:
Its parameters are given in Table 1 and R2 was 0.927. Standardized residuals were normally distributed (p = 0.361) and homoscedasticity was retained (p = 0.425).
The relationship between ODEVMS measured using TM/CA-iELISA and EVMS%POS in the entire range of possible OD values was as follows:
SU-cELISA on positive EVMS
As in the case of the other ELISAs, the scatter plot showed a distinction in ODEVMS between samples with different EVMS%POS (Fig. 3). Then, ODEVMS showed a gradual decrease that was linearly linked to increasing EVMS%POS (r = -0.871, CI 95%: -0.973 – -0.491; p = 0.002), which, however, could be observed only up to the EVMS%POS of 15%. Then, the linear trend turned significantly weaker (r = -0.454, CI 95%: -0.687 – -0.137; p = 0.007). Such a trend was observed up to the EVMS%POS of 49%, and then it slightly changed but remained similarly weak (r = -0.584, CI 95%: -0.740 – -0.368; p < 0.001). A very narrow range of EVMS%POS (up to 15%) in which the relationship between ODEVMS and EVMS%POS followed a measurable trend indicated that SU-cELISA was unsuitable for estimation of EVMS%POS. As a result, it could not be used for estimation of the within-herd seroprevalence on the basis of EVMS.
Fig. 3.
The relationship between the proportion of seropositive individual milk samples in the equal-volume milk sample (EVMS%POS) and the optical density of EVMS (ODEVMS) in competitive ELISA based on surface glycoprotein antigen (SU-cELISA). Black broken line is a cut-off separating positive and negative EVMS
Discussion
Our study shows that a milk sample prepared by pooling equal volume of individual milk samples from all lactating animals in a herd, called by us an equal-volume milk sample (EVMS), is characterized by the presence of a very strong monotonic relationship between the proportion of individual milk samples from goats seropositive for SRLV infection used for preparation of this EVMS (EVMS%POS) and its OD value measured using a commercial indirect ELISA for CAE (ODEVMS). This relationship was observed despite the fact that milk samples were selected based on serological testing of respective serum samples not milk samples themselves and the agreement between OD values in these two materials is moderate, not only in CAE [8] but also in other infections [24, 42]. The strong monotonic relationship applies, however, only to a part of the range the two variables can take.
Our results offer a chance to use ODEVMS not only to classify a herd as seropositive or seronegative but also to estimate the proportion of seropositive lactating goats in a herd. This concept is very tempting as practical benefits from quantitative evaluation of a herd status based on testing only one sample are undeniable. Hope for this has encouraged many scientific teams to attain this goal by testing a BTM sample [34–39]. However, despite significant correlations and R2 ranging from 0.7 to 0.85 a considerable spread of OD values from the regression line could be observed in all these studies. Moreover, the higher within-herd prevalence was the further OD values tended to be located from the regression line. In our study we observed the same trend as well as differences in the character of the relationship in subsequent ranges of the dependent variable (i.e. EVMS%POS). In fact, the significant and strong correlation between the two variables was present in our study only for EVMS%POS < 50%. Within this range approximately 90% of variability could be explained by EVMS models. Above this value, the regression line became more or even completely horizontal in the case of sp-iELISA and TM/CA-ELISA, respectively. Therefore, the relationship between ODEVMS and EVMS%POS could not be described using a single function for the entire range of values of EVMS%POS (0% to 100%). Interestingly, a similar phenomenon could be noticed after careful inspection of scatter plots presented in many previous studies on BTM in bovine viral infections [34, 36–39]. In these studies, exponential, square root, and logarithmic functions were used to link the two variables in the whole range of their possible values. However, our observations suggest that OD values correlate significantly only with a limited range of values the analyzed variables may take. Therefore, we think it is pointless to try to construct a single model predicting the entire range of EVMS%POS. Above a particular value, which in our study seemed to be 50%, the EVMS%POS cannot be precisely estimated which is certainly a limitation of EVMS serological testing. In our opinion, the same limitation applies to most of so far published studies aiming to predict within-herd prevalence on the basis of OD value of BTM sample. Interestingly, this effect was absent in studies investigating this BTM testing in parasitic diseases [23, 24].
The regression equations we derived are only an illustration of the relationships observable in certain predefined and controlled laboratory conditions. It is unlikely that they can be directly used to estimate within-herd prevalences of CAE in field conditions. This is because EVMS%POS is only a very simplified proxy for the within-herd prevalence. First of all, it was made by mixing milk samples from seronegative and strongly seropositive goats while not all SRLV infected goats are strongly seropositive. In fact, the levels of antibodies against SRLV are known to fluctuate [3–5] which is likely to blur the differences between situations in which 1 strongly seropositive goats or several weakly seropositive goats are present in a herd. The study of Mazzei et al. [41] shows that when the variability coming from different antibody concentrations in individual milk samples is eliminated by using still the same positive and negative milk sample to prepare milk dilutions, the correlation between OD value and proportion of seropositive parts of milk in the pooled sample is virtually perfect. However, such a situation is purely theoretical and does not correspond to field conditions. Further studies should answer the question about the magnitude of influence this factor has on the estimations based on ODEVMS. Secondly, we artificially created EVMS based on 100 individual milk samples. The influence of a single goat’s seroreactivity on the seroreactivity of EVMS was probably relatively smaller than if EVMS was based on 10 milk samples. However, the magnitude of this effect remains unknown and cannot be reliably predicted by extrapolation of our results.
Another drawback to our study is the unknown analytical sensitivity of the method. Commercial indirect ELISAs used in our study allowed to detect the presence of 1 (TM/CA-iELISA) or 2 (sp-iELISA) seropositive individual milk samples out of 100. This could falsely suggest that EVMS serological testing might serve as a screening method. It has to be emphasized that the minimum amount of antibodies that can be detected by the ELISAs in EVMS (limit of detection) is unknown. Therefore, no conclusions can be drawn from the fact only 1 or 2 positive EVMS of the lowest EVMS%POS overlapped with negative EVMS. EVMS with e.g. 10% of positive individual milk samples could have also overlapped with negative EVMS if these positive milk samples had contained less antibodies. Testing EVMS may only indicate the seropositive status of the herd but not the seronegative status. The same applies to BTM testing. The freedom from disease can only be confirmed in properly-designed and properly-conducted disease surveys [16]. Our observation that SU-cELISA performed worse than the two indirect ELISAs is probably at least partly associated with lower analytical sensitivity of EVMS testing with this ELISA. SU-cELISA is based on a single SRLV antigen so it is able to capture only antibodies against SU, leaving anti-CA and anti-TM antibodies unattended. Therefore, is likely to detect fewer positive EVMS as it has also been shown in terms of individual milk samples [15] and serum [43].
The fact that a significant correlation between OD value of BTM samples and within-herd prevalence has been observed in many previous studies acts in favor of the concept of EVMS testing. EVMS overcomes one crucial weakness of BTM i.e. an inequality of milk sample volumes contributed by individual lactating females, which is likely to considerably lower the variability in predictive models. A concept close to EVMS was proposed and evaluated by McCarthy et al. [40]. They created pooled milk samples from 10 randomly selected heifers in a cattle herd. A diagnostic accuracy of serological test for lungworm infection carried out on this sample proved to be significantly higher than on BTM sample. Their study shows highly probable advantage of pooling equal volumes of milk over BTM in which contribution of individual animals to the total volume is unknown. Unfortunately, an attempt to estimate the within-herd seroprevalence was not made in this study. EVMS combines advantages of the pooled milk sample from the representative number of animals in a herd (equal representation of animals tested) and the BTM sample (representation of all lactating animals). Obviously, like BTM, EVMS does not include males, kids, and females before the first parturition, as well as goats in a dry off period. However, especially in goats, it is unlikely to considerably interfere with the assessment of the herd status as males constitute only a small part of a dairy herd (usually a few bucks are kept separately from females and can be tested individually) and goats usually are bred seasonally. As a result, most of lactating does are in a similar stage of lactation which eliminates a factor considered as a potential source of variability in BTM testing of dairy cattle herds [37, 44]. Moreover, the optimal moment for herd screening may easily be chosen. Modern milking parlors enable rapid and simple collection of an individual milk sample from the bucket (recording jar). As EVMS is prepared just by mixing such individual milk samples at equal volume, it adds very little work to farmer’s daily schedule. Compared to collecting individual blood or milk samples directly from individual animal’s vein or udder it not only saves money, time, and labor, but also spares animals additional stress.
Conclusions
Our study introduces the concept of serological testing of an equal-volume milk sample (EVMS) as a method of detecting SRLV-infected herds and estimating the proportion of strongly seropositive goats. Analogically to BTM, EVMS ensures the representation of all lactating animals in the herd, meanwhile eliminating the variability associated with different volumes of milk yielded by each animal. Our study demonstrates that in terms of SRLV infection a significant monotonic relationship between ODEVMS and EVMS%POS is observed only for a part of the range of values these variables may take and only when indirect ELISAs are used. Further field studies are warranted to assess practical benefits of EVMS serological testing not only in terms of CAE but also other infectious and parasitic diseases in dairy animals.
Methods
Equal-volume milk samples
Numerical results (raw and corrected OD values) of 200 individual serum and milk paired samples were purposively selected from the database used in the previous study regarding performance of 3 commercial immunoenzymatic assays on individual milk samples [14]. Serum and milk (lactoserum) samples from individual goats had been collected for this study between April and August 2019 (the first half of lactation) and screened using two indirect ELISAs – ID Screen MVV-CAEV Indirect Screening test (ID.vet Innovative Diagnostics, Grabels, France) containing the panel of peptides from SRLV structural proteins – surface glycoprotein (gp135, SU), transmembrane glycoprotein (gp46, TM), and capsid protein (p25/p28, CA) (henceforth referred to as sp-iELISA), and IDEXX MVV/CAEV p28 Ab Screening (IDEXX Laboratories, Westbrook, ME, USA) based on recombinant TM and CA antigen (TM/CA-iELISA), and one competitive ELISA – Small Ruminant Lentivirus Antibody Test Kit, cELISA (VMRD, Pullman, WA, USA) coated with SU (SU-cELISA). The following criteria of sample selection were applied: One hundred paired serum and milk samples came from 100 goats that tested positive for the presence of antibodies to SRLV (seropositive goats) in the 3 ELISAs, and their corrected OD was at least twofold higher than the manufacturers’ cut-off values which were as follows: serum-to-positive control ratio (S/P%) = 50% in sp-iELISA, S/P% = 110% in TM/CA-iELISA, and percentage of inhibition (PI) = 35% in SU-cELISA. Another 100 paired serum and milk samples came from 100 goats that tested negative for the presence of antibodies to SRLV (seronegative goats) in the 3 ELISAs, and their corrected OD was at least twofold lower than the manufacturers’ cut-off values. Raw and corrected OD of paired 200 serum and milk samples are summarized in Table 2 and individual results can be found in Table S2.
Table 2.
Numerical results of 3 ELISAs (optical density, OD) used in the study presented as the arithmetic mean ± standard deviation and range in parentheses
| ELISAa | Seronegative goats (n = 100) | Seropositive goats (n = 100) | ||||||
|---|---|---|---|---|---|---|---|---|
| serum | milk | serum | milk | |||||
| Raw OD | Corrected ODb [%] | Raw OD | Corrected ODb [%] | Raw OD | Corrected ODb [%] | Raw OD | Corrected ODb [%] | |
| sp-iELISA | 0.073 ± 0.038 (0.048 – 0.297) | 2.3 ± 3.6 (-0.3 – 20) | 0.065 ± 0.028 (0.045 – 0.240) | 1.5 ± 2.5 (-0.5 – 15) | 4.019 ± 0.248 (3.061 – 4.2) | 353 ± 34 (286 – 429) | 3.625 ± 0.690 (1.159 – 4.2) | 316 ± 59 (111 – 403) |
| TM/CA-iELISA | 0.160 ± 0.045 (0.100 – 0.330) | 8.2 ± 4.8 (1.8 – 20) | 0.169 ± 0.122 (0.051 – 0.933) | 9.2 ± 12 (-2.7 – 78) | 3.934 ± 0.297 (2.924 – 4.2) | 376 ± 48 (302 – 474) | 3.887 ± 0.443 (1.926 – 4.2) | 372 ± 63 (179 – 474) |
| SU-cELISA | 0.810 ± 0.156 (0.517 – 1.385) | -1.3 ± 12 (-45 – 9.9) | 0.737 ± 0.179 (0.275 – 1.596) | 7.6 ± 18 (-56 – 67) | 0.189 ± 0.085 (0.062 – 0.383) | 74 ± 12 (52 – 93) | 0.286 ± 0.120 (0.059 – 0.585) | 61 ± 15 (28 – 93) |
a ELISA used: sp-iELISA – indirect ELISA based on a mixture of synthetic viral peptides, TM/CA-iELISA – indirect ELISA based on recombinant transmembrane and capsid proteins, and SU-cELISA – competitive ELISA based on surface glycoprotein antigen
b corrected optical density: sp-iELISA and TM/CA-iELISA – serum-to-positive control ratio (S/P%), SU-cELISA – percentage of inhibition (PI)
By mixing randomly selected milk samples from seronegative goats with randomly selected milk samples from seropositive goats, 193 equal-volume milk samples (EVMS) were created. Simple random method of selection with returning was performed using the RAND.BETWEEN() function in Microsoft Excel. For each EVMS, 10 μl of 100 individual milk samples were mixed to amount to the volume of 1 ml. Ninety three EVMS were made of individual milk samples from seronegative goats (negative EVMS). One hundred EVMS were made of individual milk samples from seropositive and seronegative goats combined at a ratio from 1:99 to 100:0 (positive EVMS). The proportion of seropositive individual milk samples out of 100 individual milk samples (EVMS%POS) increased by 1% from 1% to 100%.
EVMS serological testing
Then, OD of EVMS (ODEVMS) of the 193 EVMS was measured using the 3 ELISAs. The ELISAs were performed according to manufacturers’ protocols. EVMS were diluted 1/2 in sp-iELISA and TM/CA-iELISA, and remained undiluted in SU-cELISA. These dilutions were chosen based on our previous study [15]. All other steps of the protocol remained unchanged compared to regular serum testing. ODEVMS was measured at a wavelength of 450 nm (sp-iELISA and TM/CA-iELISA) or 620 nm (SU-cELISA) using the scanner (Epoch Microplate Spectrophotometer, BioTek, USA). ODEVMS values exceeding the upper measuring limit of the scanner (OD > 4.0, OVERFLOW) were replaced with the value of 4.2.
Statistical analysis
OD values were presented as the arithmetic mean, standard deviation (± SD), and range. Normality of distribution was assessed using the Quantile–Quantile scatter plots and the Shapiro–Wilk test, and the skewness of distribution was expressed using coefficient of skewness (CoS) with CI 95%. The cut-off value separating negative and positive EVMS was set at the maximum (in the case of indirect ELISAs) or minimum (in the case of SU-cELISA) ODEVMS observed in negative EVMS rounded up or down, respectively, to the closest second decimal digit. Diagnostic specificity of EVMS testing was calculated as the number of negative EVMS below the cut-off value divided by 93 negative EVMS and the CI 95% was calculated using Wilson score method [45].
Linear correlation between ODEVMS above the cut-off value and EVMS%POS was determined using the Pearson product-moment correlation coefficient (r) and the CI 95% was calculated with the method of Altman et al. [45]. Strength of correlation was classified as follows: r = 0.00 to 0.19 – very weak; 0.20 to 0.49 – weak; 0.50 to 0.69 – moderate, 0.70 to 0.89 – strong, and 0.90 to 1.00 – very strong [46].
The relationship between EVMS%POS (explanatory, independent variable, x) and ODEVMS above the cut-off value (outcome, dependent variable, y) was investigated using the scatter plot and simple linear regression according to a general formula:
where β0 was an intercept (an y value at which x = 0), β1 was a slope of the linear regression line, and was a residual (error).
EVMS%POS was considered an independent variable in this model because its values were fixed by the study design, and they had an influence on ODEVMS which were, therefore, considered a dependent variable. Hence, the equation was as follows:
Depending on the shape of relationship, x variable was either entered in raw or logarithmically transformed (natural logarithm with e basis) form. To predict EVMS%POS based on ODEVMS the equation was inversed as follows:
Assumptions of the linear regression were evaluated as follows [47]: 1) shape of the relationship between x and y was assessed by visual inspection of the scatter plot and it was linearized by x transformation if necessary; 2) normality of residual distribution was assessed by visual inspection of the Quantile–Quantile scatter plots and using the Shapiro–Wilk test; 3) homoscedasticity was assessed by visual inspection of the standardized residual vs. predicted y variable plot and using Brown-Forsythe test on standardized residuals compared across 5 categories of EVMS%POS. The proportion of explained variability was evaluated by the coefficient of determination (R2). All statistical tests were two-tailed and a significance level (α) was set at 0.05. Statistical analysis was performed using TIBCO Statistica 13.3 (TIBCO Software Inc, Palo Alto, CA, USA).
Supplementary Information
Additional file 1: Table S1. Raw and corrected optical densities (OD) of 193 equal-volume milk samples (EVMS) measured using 3 ELISAs along with the proportion of seropositive individual milk samples in the equal-volume milk sample (EVMS%POS).
Additional file 2: Table S2. Raw and corrected optical densities (OD) of 200 serum and milk samples used in the experiment measured using 3 ELISAs – indirect ELISA based on a mix of synthetic viral peptides (sp-iELISA), indirect ELISA based on recombinant transmembrane and capsid proteins (TM/CA-iELISA), and competitive ELISA based on surface glycoprotein antigen (SU-cELISA).
Acknowledgements
Not applicable.
Abbreviations
- α
Significance level
- β0
Intercept of the linear regression line
- β1
Slope of the linear regression line
- BTM
Bulk-tank milk
- CAE
Caprine arthritis-encephalitis
- CI
Confidence interval
- CoS
Coefficient of skewness
- EVMS
Equal-volume milk sample
- EVMS%POS
The proportion of seropositive individual milk samples in the equal-volume milk sample
- OD
Optical density
- ODEVMS
Optical density of the equal-volume milk sample
- PI
Percentage of inhibition
- r
Pearson product-moment correlation coefficient
- R2
Coefficient of determination
- SD
Standard deviation
- S/P%
Sample-to-positive control ratio percentage
- SRLV
Small ruminant lentivirus
- SU-cELISA
Competitive ELISA based on surface glycoprotein of SRLV
- TM/CA-iELISA
Indirect ELISA based on recombinant transmembrane and capsid protein of SRLV
- sp-iELISA
Indirect ELISA based on a mixture of synthetic viral proteins
Authors’ contributions
AVP, MC, MS, and JK designed research; AVP, OS-J, AM-F, LW, MM, KB, DO, ET, and MC performed research; AVP, MC, and JK performed statistical and epidemiological analysis. AVP, MC, and JK wrote the draft manuscript. MS, CC, EB, IM-D, LW, and JK edited and reviewed the manuscript. All authors read and approved the final manuscript.
Funding
This work was financially supported by the National Science Center (grant number 2013/09/B/NZ6/03514) and by the Polish National Agency for Academic Exchange (NAWA) PROM (contract number POWR.03.03.00–00-PN13/18). The study was also financially supported by the grant from the Ministry of Science and Higher Education of the Republic of Poland, decision no. 9506/E-385/R/2018. The funding body provided financial support for modernizing and equipping the serological laboratory in which tests were performed. Sponsors did not have any role in study design, in the collection, analysis and interpretation of data, in the writing of the report, or in the decision to submit the article for publication.
Availability of data and materials
The database is available from the authors (Adrian-Valentin Potârniche and Michał Czopowicz) on request.
Declarations
Ethics approval and consent to participate
No ethics commission approval for this study was required according to Polish legal regulations (the Act on the Protection of Animals Used for Scientific or Educational Purposes of 15 January 2015) as collection of milk samples is not associated with any harm or pain for animals.
All the methods were performed in accordance with relevant guidelines and regulations.
The study is reported in accordance with ARRIVE guidelines.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Adrian-Valentin Potârniche, Email: adrian.potarniche@usamvcluj.ro.
Michał Czopowicz, Email: mczopowicz@gmail.com.
Olga Szaluś-Jordanow, Email: olga_szalus@sggw.edu.pl.
Agata Moroz-Fik, Email: agata_moroz@sggw.edu.pl.
Marcin Mickiewicz, Email: marcin_mickiewicz@sggw.edu.pl.
Kinga Biernacka, Email: kinga_biernacka@sggw.edu.pl.
Lucjan Witkowski, Email: lucjan_witkowski@sggw.edu.pl.
Iwona Markowska-Daniel, Email: iwona_markowska_daniel@sggw.edu.pl.
Emilia Bagnicka, Email: e.bagnicka@igbzpan.pl.
Constantin Cerbu, Email: constantin.cerbu@usamvcluj.ro.
Diana Olah, Email: diana_olah01@yahoo.com.
Emilia Trif, Email: emilia.ungureanu@usamvcluj.ro.
Marina Spinu, Email: marina.spinu@usamvcluj.ro.
Jarosław Kaba, Email: jaroslaw_kaba@sggw.edu.pl.
References
- 1.Adams DS, Crawford TB, Banks KL, McGuire TC, Perryman LE. Immune responses of goats persistently infected with caprine arthritis-encephalitis virus. Infect Immun. 1980;28:421–227. doi: 10.1128/iai.28.2.421-427.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rimstad E, East NE, Torten M, Higgins J, DeRock E, Pedersen NC. Delayed seroconversion following naturally acquired caprine arthritis-encephalitis virus infection in goats. Am J Vet Res. 1993;54:1858–1862. [PubMed] [Google Scholar]
- 3.Hanson J, Hydbring E, Olsson K. A long term study of goats naturally infected with caprine arthritis-encephalitis virus. Acta Vet Scand. 1996;37:31–39. doi: 10.1186/BF03548117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Czopowicz M, Szaluś-Jordanow O, Mickiewicz M, Witkowski L, Moroz A, Markowska-Daniel I, Reczyńska D, Bagnicka E, Kaba J. Fall in antibody titer to small ruminant lentivirus in the periparturient period in goats. Small Rumin Res. 2017;147:37–40. [Google Scholar]
- 5.Kaba J, Czopowicz M, Witkowski L, Szaluś-Jordanow O, Mickiewicz M, Markowska-Daniel I, Puchała R, Bagnicka E. Longitudinal study on seroreactivity of goats exposed to colostrum and milk of small ruminant lentivirus–infected dams. J Vet Res. 2022;66:511–521. doi: 10.2478/jvetres-2022-0071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.de Andrés D, Klein D, Watt NJ, Berriatua E, Torsteinsdottir S, Blacklaws BA, Harkiss GD. Diagnostic tests for small ruminant lentiviruses. Vet Microbiol. 2005;107:49–62. doi: 10.1016/j.vetmic.2005.01.012. [DOI] [PubMed] [Google Scholar]
- 7.Herrmann-Hoesing LM. Diagnostic assays used to control small ruminant lentiviruses. J Vet Diagn Invest. 2010;22:843–855. doi: 10.1177/104063871002200602. [DOI] [PubMed] [Google Scholar]
- 8.Adjadj NR, Vicca J, Michiels R, De Regge N. (Non-)Sense of Milk Testing in Small Ruminant Lentivirus Control Programs in Goats. Comparative Analysis of Antibody Detection and Molecular Diagnosis in Blood and Milk. Viruses. 2019;12:3. doi: 10.3390/v12010003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Plaza M, Sanchez A, Corrales JC, De La Fe C, Contreras A. Caprine arthritis encephalitis virus diagnosed by ELISA in lactating goats using milk samples. Small Rumin Res. 2009;81:189–192. [Google Scholar]
- 10.Brinkhof JM, Houwers DJ, Moll L, Dercksen D, van Maanen C. Diagnostic performance of ELISA and PCR in identifying SRLV-infected sheep and goats using serum, plasma and milk samples and in early detection of infection in dairy flocks through bulk milk testing. Vet Microbiol. 2010;142:193–198. doi: 10.1016/j.vetmic.2009.09.060. [DOI] [PubMed] [Google Scholar]
- 11.Barquero N, Arjona A, Domenech A, Toural C, de las Heras A, Fernández-Garayzabal JF, Ruiz-Santa Quiteria JA, Gomez-Lucia E. Diagnostic performance of PCR and ELISA on blood and milk samples and serological survey for small ruminant lentiviruses in central Spain. Vet Rec. 2011;168:20. doi: 10.1136/vr.c4951. [DOI] [PubMed] [Google Scholar]
- 12.Barquero N, Domenech A, Arjona A, Fernández-Garayzabal JF, Ruiz-Santa-Quiteria JA, Gomez-Lucia E. Comparison of two PCR and one ELISA techniques for the detection of small ruminant lentiviruses (SRLVs) in milk of sheep and goats. Res Vet Sci. 2013;94:817–819. doi: 10.1016/j.rvsc.2013.01.004. [DOI] [PubMed] [Google Scholar]
- 13.Barquero N, Gomez-Lucia E, Arjona A, Toural C, Heras Al, Fernández-Garayzabal JF, Domenech A. Evolution of specific antibodies and proviral DNA in milk of small ruminants infected by small ruminant lentivirus. Viruses. 2013;5:2614–2623. doi: 10.3390/v5102614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bernier Gosselin V, Dufour S, Zhang MZ, Middleton JR. Sensitivity and specificity of a competitive ELISA using frozen-thawed milk or serum for the diagnosis of small ruminant lentivirus infection in goats using a Bayesian latent class model. Small Rumin Res. 2018;167:29–31. [Google Scholar]
- 15.Potârniche AV, Czopowicz M, Szaluś-Jordanow O, Moroz A, Mickiewicz M, Witkowski L, Markowska-Daniel I, Bagnicka E, Cerbu C, Olah D, Spinu M, Kaba J. Diagnostic accuracy of three commercial immunoenzymatic assays for small ruminant lentivirus infection in goats performed on individual milk samples. Prev Vet Med. 2021;191:105347. doi: 10.1016/j.prevetmed.2021.105347. [DOI] [PubMed] [Google Scholar]
- 16.Martin SW, Shoukri M, Thorburn MA. Evaluation the health status of herds based on tests applied to individuals. Prev Vet Med. 1992;14:33–43. [Google Scholar]
- 17.Eiras C, Arnaiz I, Sanjuán ML, Yus E, Diéguez FJ. Bovine viral diarrhea virus: correlation between herd seroprevalence and bulk tank milk antibody levels using 4 commercial immunoassays. J Vet Diagn Invest. 2012;24:549–553. doi: 10.1177/1040638712440984. [DOI] [PubMed] [Google Scholar]
- 18.Klem TB, Tollersrud T, Osterås O, Stokstad M. Association between the level of antibodies in bulk tank milk and bovine respiratory syncytial virus exposure in the herd. Vet Rec. 2014;175:47. doi: 10.1136/vr.102403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.John EE, Nekouei O, McClure JT, Cameron M, Keefe G, Stryhn H. Investigation of within- and between-herd variability of bovine leukaemia virus bulk tank milk antibody levels over different sampling intervals in the Canadian Maritimes. Prev Vet Med. 2018;154:90–94. doi: 10.1016/j.prevetmed.2018.03.015. [DOI] [PubMed] [Google Scholar]
- 20.Isoda N, Asano A, Ichijo M, Ohno H, Sato K, Okamoto H, Nakao S, Kato H, Saito K, Ito N, Usui A, Takayama H, Sakoda Y. Assessment of the cost effectiveness of compulsory testing of introduced animals and bulk tank milk testing for bovine viral diarrhea in Japan. J Vet Med Sci. 2019;81:577–585. doi: 10.1292/jvms.18-0671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nielsen TD, Nielsen LR, Toft N, Houe H. Association between bulk-tank milk Salmonella antibody level and high calf mortality in Danish dairy herds. J Dairy Sci. 2010;93:304–310. doi: 10.3168/jds.2009-2528. [DOI] [PubMed] [Google Scholar]
- 22.Salgadu A, Firestone SM, Watt A, Thilakarathne DS, Condello AK, Siu D, Masukagami Y, Tivendale KA, Stevenson MA, Mansell PD, Browning GF, Wawegama NK. Evaluation of the MilA ELISA for the diagnosis of herd infection with Mycoplasma bovis using bulk tank milk and estimation of the prevalence of M. bovis in Australia. Vet Microbiol. 2022;270:109454. doi: 10.1016/j.vetmic.2022.109454. [DOI] [PubMed] [Google Scholar]
- 23.Salimi-Bejestani MR, Daniel RG, Felstead SM, Cripps PJ, Mahmoody H, Williams DJ. Prevalence of Fasciola hepatica in dairy herds in England and Wales measured with an ELISA applied to bulk-tank milk. Vet Rec. 2005;156:729–731. doi: 10.1136/vr.156.23.729. [DOI] [PubMed] [Google Scholar]
- 24.Mezo M, González-Warleta M, Castro-Hermida JA, Muiño L, Ubeira FM. Field evaluation of the MM3-SERO ELISA for detection of anti-Fasciola IgG antibodies in milk samples from individual cows and bulk milk tanks. Parasitol Int. 2010;59:610–615. doi: 10.1016/j.parint.2010.09.001. [DOI] [PubMed] [Google Scholar]
- 25.Ortega-Mora LM, Ferre I. Presence of Ostertagia ostertagi antibodies in bulk tank milk from cattle herds in northern Spain. Vet Parasitol. 2013;197:388–392. doi: 10.1016/j.vetpar.2013.05.021. [DOI] [PubMed] [Google Scholar]
- 26.Ploeger HW, Holzhauer M, Uiterwijk M, Van Engelen E. Comparison of two serum and bulk-tank milk ELISAs for diagnosing natural (sub)clinical Dictyocaulus viviparus infection in dairy cows. Vet Parasitol. 2014;199:50–58. doi: 10.1016/j.vetpar.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 27.Kowalczyk SJ, Czopowicz M, Weber CN, Müller E, Nalbert T, Bereznowski A, Kaba J. Herd-level seroprevalence of Fasciola hepatica and Ostertagia ostertagi infection in dairy cattle population in the central and northeastern Poland. BMC Vet Res. 2018;14:131. doi: 10.1186/s12917-018-1455-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Takeuchi-Storm N, Thamsborg SM, Enemark HL, Boes J, Williams D, Denwood MJ. Association between milk yield and milk anti-Fasciola hepatica antibody levels, and the utility of bulk tank milk samples for assessing within-herd prevalence on organic dairy farms. Vet Parasitol. 2021;291:109374. doi: 10.1016/j.vetpar.2021.109374. [DOI] [PubMed] [Google Scholar]
- 29.Nagel-Alne GE, Valle PS, Krontveit R, Sølverød LS. Caprine arthritis encephalitis and caseous lymphadenitis in goats: use of bulk tank milk ELISAs for herd-level surveillance. Vet Rec. 2015;176:173. doi: 10.1136/vr.102605. [DOI] [PubMed] [Google Scholar]
- 30.Gazzonis AL, Zanzani SA, Stradiotto K, Olivieri E, Villa L, Manfredi MT. Toxoplasma gondii Antibodies in Bulk Tank Milk Samples of Caprine Dairy Herds. J Parasitol. 2018;104:560–565. doi: 10.1645/17-44. [DOI] [PubMed] [Google Scholar]
- 31.Bauman CA, Jones-Bitton A, Jansen J, Kelton D, Menzies P. Evaluation of bulk tank milk PCR and bulk tank milk modified ELISA tests for the detection of paratuberculosis at the herd level in goat and sheep dairies in Ontario. Canada J Dairy Sci. 2019;102:511–520. doi: 10.3168/jds.2018-15020. [DOI] [PubMed] [Google Scholar]
- 32.Anastácio S, Carolino N, Sidi-Boumedine K, da Silva GJ. Q Fever Dairy Herd Status Determination Based on Serological and Molecular Analysis of Bulk Tank Milk. Transbound Emerg Dis. 2016;63:e293–300. doi: 10.1111/tbed.12275. [DOI] [PubMed] [Google Scholar]
- 33.Jansen W, Cargnel M, Boarbi S, Mertens I, Van Esbroeck M, Fretin D, Mori M. Belgian bulk tank milk surveillance program reveals the impact of a continuous vaccination protocol for small ruminants against Coxiella burnetii. Transbound Emerg Dis. 2022;69:e141–e152. doi: 10.1111/tbed.14273. [DOI] [PubMed] [Google Scholar]
- 34.Niskanen R. Relationship between the levels of antibodies to bovine viral diarrhoea virus in bulk tank milk and the prevalence of cows exposed to the virus. Vet Rec. 1993;133:341–344. doi: 10.1136/vr.133.14.341. [DOI] [PubMed] [Google Scholar]
- 35.Beaudeau F, Assié S, Seegers H, Belloc C, Sellal E, Joly A. Assessing the within-herd prevalence of cows antibody-positive to bovine viral diarrhoea virus with a blocking ELISA on bulk tank milk. Vet Rec. 2001;149:236–240. doi: 10.1136/vr.149.8.236. [DOI] [PubMed] [Google Scholar]
- 36.Hartman A, van Wuijckhuise L, Frankena K, Franken P, Wever P, de Wit J, Kramps J. Within-herd BHV-1 prevalence prediction from an ELISA on bulk milk. Vet Rec. 1997;140:484–485. doi: 10.1136/vr.140.18.484. [DOI] [PubMed] [Google Scholar]
- 37.Nylin B, Strøger U, Rønsholt L. A retrospective evaluation of a Bovine Herpesvirus-1 (BHV-1) antibody ELISA on bulk-tank milk samples for classification of the BHV-1 status of Danish dairy herds. Prev Vet Med. 2000;47:91–105. doi: 10.1016/s0167-5877(00)00163-x. [DOI] [PubMed] [Google Scholar]
- 38.Martínez S, Yus E, Sanjuán ML, Camino F, Eiras MC, Arnaiz I, Diéguez FJ. Bovine herpesvirus 1: within-herd seroprevalence and antibody levels in bulk-tank milk. Rev Sci Tech. 2016;35:899–904. doi: 10.20506/rst.35.3.2577. [DOI] [PubMed] [Google Scholar]
- 39.Nekouei O, Stryhn H, Van Leeuwen J, Kelton D, Hanna P, Keefe G. Predicting within-herd prevalence of infection with bovine leukemia virus using bulk-tank milk antibody levels. Prev Vet Med. 2015;122:53–60. doi: 10.1016/j.prevetmed.2015.10.009. [DOI] [PubMed] [Google Scholar]
- 40.McCarthy C, Höglund J, Christley R, Juremalm M, Kozlova I, Smith R, van Dijk J. A novel pooled milk test strategy for the herd level diagnosis of Dictyocaulus viviparus. Vet Parasitol X. 2019;1:100008. doi: 10.1016/j.vpoa.2019.100008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mazzei M, Carrozza ML, Bandecchi P, Mazzanti G, Mannelli A, Tolari F. Evaluation of an ELISA to detect antibodies to maedi-visna virus in individual and pooled samples of milk from sheep. Vet Rec. 2005;157:552–555. doi: 10.1136/vr.157.18.552. [DOI] [PubMed] [Google Scholar]
- 42.Ohlson A, Blanco-Penedo I, Fall N. Comparison of Bovine coronavirus-specific and Bovine respiratory syncytial virus-specific antibodies in serum versus milk samples detected by enzyme-linked immunosorbent assay. J Vet Diagn Invest. 2014;26:113–116. doi: 10.1177/1040638713509377. [DOI] [PubMed] [Google Scholar]
- 43.Czopowicz M, Szaluś-Jordanow O, Mickiewicz M, Moroz A, Witkowski L, Markowska-Daniel I, Reczyńska D, Bagnicka E, Kaba J. Decline of maternal antibodies to small ruminant lentivirus in goat kids. Anim Sci J. 2018;89:1364–1370. doi: 10.1111/asj.13038. [DOI] [PubMed] [Google Scholar]
- 44.Guidry J, Butler JE, Pearson RE, Weinland BT. IgA, igG1, IgG2, IgM, and BSA in serum and mammary secretion throughout lactation. Vet Immunol Immunopathol. 1980;1:329–341. doi: 10.1016/0165-2427(80)90012-4. [DOI] [PubMed] [Google Scholar]
- 45.Altman D, Machin D, Bryant T, Gardner M. Statistics with Confidence: Confidence Intervals and Statistical Guidelines, 2nd ed. Bristol: BMJ Books; 2000. p. 46–48;89–90.
- 46.Akoglu H. User's guide to correlation coefficients. Turk J Emerg Med. 2018;18:91–93. doi: 10.1016/j.tjem.2018.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zar JH. Biostatistical analysis, 5th ed. Upper Saddle River: Pearson Prentice Hall; 2010. p. 330–7.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Raw and corrected optical densities (OD) of 193 equal-volume milk samples (EVMS) measured using 3 ELISAs along with the proportion of seropositive individual milk samples in the equal-volume milk sample (EVMS%POS).
Additional file 2: Table S2. Raw and corrected optical densities (OD) of 200 serum and milk samples used in the experiment measured using 3 ELISAs – indirect ELISA based on a mix of synthetic viral peptides (sp-iELISA), indirect ELISA based on recombinant transmembrane and capsid proteins (TM/CA-iELISA), and competitive ELISA based on surface glycoprotein antigen (SU-cELISA).
Data Availability Statement
The database is available from the authors (Adrian-Valentin Potârniche and Michał Czopowicz) on request.