Supplemental Digital Content is Available in the Text.
Spinal cord stimulation treatment in Sweden decreases disability days and consequently indirect costs. Effect of SCS seemed to have little association with patient characteristics.
Keywords: Neuromodulation, Spinal cord stimulation, Administrative register, Productivity loss, Work ability, Effectiveness.
Abstract
Current research indicates that spinal cord stimulation (SCS) has a positive short-term impact on outcomes, such as quality of life, pain, and productivity in patients with chronic neuropathic pain. However, there is a need for studies on larger population samples. This study used data from Swedish national registers to analyze change and predictors of sick leave and disability pension 2 years before and after SCS treatment. Patients with SCS implanted between 2006 and 2017, and a reference group consisting of 5 individuals matched to each SCS patient without replacement with respect to age, sex, and region of residence, were included. A difference-in-difference approach was used to compare the average change (2 years after treatment vs 2 years before treatment) in net disability days and indirect cost related to disability days for the SCS group, compared with the average change for the reference group. The results showed that SCS treatment in Sweden is associated with a decrease of 21 disability days and consequent decrease in indirect cost of €4127 in working age patients. Large work loss prior to index date was also demonstrated (average 214 days before 1 year), indicating a significant burden on the patient, employers, and the society at large. The number of disability days varied considerably depending on age, sex, socioeconomic variables, and comorbidities; however, the effect of SCS seemed to have little association with patient characteristics. This economic benefit needs to be considered, as well as the clinical outcome, when evaluating the full societal value of SCS.
1. Introduction
Chronic pain, defined as a pain condition lasting more than 3 months, affects one in 5 Europeans and can stem from a variety of causes and has substantial effect on the quality of life and costs.1,3,7,12,21,27 Neuropathic pain is defined as a lesion or disease affecting the somatosensory nervous system.14 The prevalence of chronic neuropathic pain is uncertain, but some studies indicate it to be between 7% and 10%.34 It is associated with a high burden for the individual, health care, and society.1,3,7,21 Neuropathic pain commonly affects the back, and back pain is one of the most common causes of sick leave.2,27 Indirect costs because of the absence from work constitutes the majority of total costs related to chronic pain conditions.11
Treatment of neuropathic pain commonly includes pharmacotherapy, but the efficacy on pain relief is often unsatisfactory.8,10 Multiple nonpharmacological treatments for chronic neuropathic pain are available, such as physical therapy, cognitive behavioral therapy, multimodal rehabilitation, and interventional pain treatments, including radiofrequency ablation. Spinal cord stimulation (SCS) is a treatment option that has been used for more than 50 years to reduce neuropathic pain.25 Multiple trials have shown SCS to be a comparably safe and often effective treatment for chronic conditions like failed back surgery syndrome (FBSS), complex regional pain syndrome, and painful diabetic polyneuropathy.4,5,17,18,20,23
Most investigations on SCS are randomized controlled trials involving special selected patients, with only a few trials evaluating a heterogenous population representative of clinical practice.12,24,30,32 The current evidence indicates that SCS may exert a positive impact on the overall quality of life, pain, and work ability,19,31 and an observational study revealed that after initiation of SCS treatment of patients who had undergone lumbar spine surgery, sick leave and disability pension decreased.15 However, the patient populations evaluated to date have been small, and information concerning long-term effects and the influence of patient characteristics (eg, clinical, and socioeconomic) is lacking.
Moreover, much of the information on predictors of the success of SCS treatment, which is crucial to designing effective therapy, is today weak or conflicting, and guidelines are often based exclusively on expert opinion. The uniquely complete information in Swedish national registries covering almost all healthcare visits, diagnoses, procedures, and disability payments for the entire population allowed us to conduct a retrospective observational cohort study designed to identify potential predictors of positive outcome (for example, socioeconomics, comorbidities), as well as the impact of SCS treatment on the ability to work.
The aim of the study was to analyze the impact of SCS on sick leave and long-term disability pension and what predictors are associated with the impact.
2. Methods
2.1. Study design and patient selection
This is a retrospective observational cohort study of individuals treated with SCS. The study used a research database consisting of patient-level data from several Swedish national and local registers. Population-based data from the National Patient Register, the Cause of Death Register, Longitudinal integrated database for health insurance and labor market studies (“LISA”), and the Swedish Social Insurance Agency were extracted for the study population. Data from the different data sources were linked on patient level through the personal national ID. The national registers in Sweden have a nation-wide coverage and contain detailed information on the provision of care on an individual level. Due to a closed data system (data have always been reported to the same registers, which have always been maintained by the same authorities), patients are not lost to follow-up, unless they emigrate from the country.
The study period was from January 1, 2000, to December 31, 2019, the maximal period during which relevant data could be extracted from the Swedish Social Security Agency. The individuals included were of working age (21-62 years) who began SCS treatment because of neuropathic pain during 2006 to 2017 (as identified from the Swedish National Patient Register) based on procedure coding (NOMESCO Classification of Surgical Procedures [NCSP]). The identification period started in 2006 to identify prior drug use in the Prescribed Drug Register, which started in July 2005. In line with a previous study and for comparability, only patients with “permanent” SCS treatment, defined as permanent insertion of the device within 100 days of the test insertion,15 were included. Test and permanent implants were identified from the same NCSP code ABD30 (“implantation of spinal stimulation device”). A reference group was included to rule out the effect of societal changes that may impact the use of disability benefits (eg, unemployment, changes to the social security system). The reference group consisted of 5 individuals matched to each patient without replacement with respect to age, sex, and region of residence.
All individuals who live or work in Sweden are automatically covered by the Swedish social insurance providing compensation for income losses. These compensations are registered in the register from the Social Insurance Agency. The insurance system comprises several different types of benefits, which may be granted depending on, for example, the length of time for which the benefit can be granted and age of the insured individual. For simplicity, these benefit types are referred to as sick leave benefits and disability pension. Sick leave benefits are compensations granted for a distinct number of days and cannot be permanent. Disability pension can be granted if the disorder is “chronic,” in this context meaning that sufficiently severe to prohibit the person from ever going back to work. During the study period, the first day of a sick leave period was an unpaid, qualifying day and the following 13 days are a “sick pay” period, covered by the employer. Only sick leaves exceeding 14 days are registered in the Social Insurance Agency's register, from the day which they are reimbursed by the public social insurance plan. However, for all sick episodes exceeding 14 days, the first 14 days are registered. Sick leave and disability pension were measured concerning net disability days. Net disability days were defined as the degree of compensation (the percentage of the patient's working time, which is covered by sick leave benefits and/or disability pension) multiplied with the gross number of days with granted sick leave or disability pension. Net disability days included both normal working days, weekends, and holidays. Because disability pension is only approved for long-term disability, it is less likely that disability pension use will change over a four-year period. Therefore, net days of sick leave and disability pension were analyzed separately in a sensitivity analysis.
The indirect cost of sick leave and disability pension was measured by assigning a monetary value to net disability days. The most commonly used approach to value the indirect cost of reduced work productivity is the human capital approach.33 This approach assumes that the relevant value of reduced productivity equals to the present value of all lost future earnings for the individual. Assuming that the value of production is equal to the total labor cost from the employer's view, gross wage was calculated from the average wage per day (including weekends and holidays) depending on sex and education level in Sweden based on publicly available statistics from Statistics Sweden, with an additional 31.42% of payroll taxes.28,29 Indirect costs were expressed in Euro (€) 2020 using mean yearly exchange rate of SEK/€10.4867.
2.2. Potential predictors
Index date was defined as the date of first SCS implantation. Untreated control individuals were assigned their matched counterpart's index date. Potential predictors were observed during a period of up to 5 years prior to index date depending on variable (maximum look-back period was 5 years). Calendar year of index date was included because minor reforms have been made to the Swedish social insurance system during the study period, which may have an impact on the use of such benefits.26 Calendar year was also intended to capture any effects from developments of the SCS treatment that occurred during the study period. Considered predictors, collected from the national registers, were as follows:
(1) Age in years at index date
(2) Sex (female, male)
(3) Country of birth (Sweden, Europe except Sweden, other)
(4) Calendar year of index date
(5) Previous spine surgeries identified by NCSP codes (code list available in Supplementary Table 1, available at http://links.lww.com/PAIN/B699)
(6) Elixhauser comorbidity index,9 measured by diagnoses (ICD-10) 2 years prior to index date
(7) Education level: primary, secondary, and postsecondary or postgraduate (combined because of few individuals had postgraduate education)
(8) Employment status (employed/not employed)
(9) Annual income in Euro (€) inflated to 2020 price level
(10) Any use of strong opioids 3 months preceding index date (identified using Anatomic Therapeutic Chemical classification system, ATC, code N02A)
(11) Any use of weak opioids 3 months preceding index date (identified using Anatomic Therapeutic Chemical classification system, ATC, code N02A)
(12) Any use of nonopioid pain medicine 3 months preceding index date (ATC code N02B)
(13) Any use of depression medicine 3 months preceding index (ATC code N06A–B).
2.3. Statistical analysis
A difference-in-difference (DiD) approach was used to create a model in which the change in net disability days in SCS patients before and after the start of treatment is compared with the change during the same calendar period in control individuals. The change in net disability days was measured as the difference in total days from month 12 to 24 after index date (“after period”) compared with month 24 to 12 before index date (“before period”). The DiD model subtracts the average change over time in the reference group from the average change over time in the exposed group.13 The period of 12 to 24 months before and after index date was chosen to wash out the initial increase in disability days in the months before and after SCS implantation that could be related to recovering from the procedure, rather than long-term effect on disability. As a sensitivity analysis, the change in net disability days was measured as the difference in total days from month 25 to 36 months after index date compared with month 36 to 24 before the index date. It was required for patients to have data in these periods. Double differencing handles the problem of time-variant trends in society (such as unemployment and changes in the social security system) and biases resulting from permanent differences between the treated and reference groups.
The estimator of DiD is defined as the average change in outcome from the before period to the after period in the exposed individuals subtracted with the average change in outcome during the same periods in control individuals (equation 1). Individual i belongs to group G (a dummy variable that takes the value 1 if a patient is an SCS patient and 0 if the patient is a control) and is observed at time T (a dummy variable that takes the value 1 if in the after period and 0 if in the before period).
| (1) |
The effect of SCS on the net disability days (, ) is identified by the interaction term estimated by the in the ordinary least squares regression model (equation 2):
| (2) |
where is the outcome net disability days. The estimated intercept captures systematical time-invariant differences in outcome between patients. For every calendar year of possible index dates, captures the time-variant component in outcome, which is common for all patients. are the potential predictors. captures unobservable characteristics, which are assumed to be independent of the group indicator and have the same distribution over time. Potential predictors were interacted with the group indicator G and the time indicator T to measure the association of the predictors and effect of SCS on net disability days (equation 3).
| (3) |
The indirect cost of sick leave and disability pension has a mass point at zero and is heavily left skewed; costs were analyzed using a two-part regression model, which is common when analyzing, for example, healthcare costs.6 The first part of the model uses a logistic regression to model the probability of having indirect cost larger than zero. The second part models indirect costs larger than zero using generalized linear model with gamma family and log link. Indirect costs per period and group, and the difference between them, are then predicted based on this regression model.
Data management and statistical analyses were conducted using MySQL and Stata16.
3. Results
3.1. Baseline characteristics
The final study population included 1082 SCS patients and 5410 control individuals. Seven hundred twenty-four patients were deemed to have test implantation (only one implantation code registered, or more than 100 days between subsequent implantation) and were therefore excluded from the study. Table 1 shows participant characteristics at index date. Due to matching, the 2 groups were similar in age (mean 47 years) and gender (56% were female). On average, income and education level were lower in the SCS group compared with the reference group. Use of opioids, non-opioid pain drugs, and anti-depression medicine were substantially higher in the SCS group.
Table 1.
Participant characteristics.
| SCS patients (n = 1082) | Control individuals (n = 5410) | ||||
|---|---|---|---|---|---|
| Mean or percent | SE or count | Mean or percent | SE or count | ||
| Age at index (y) | 47.1 | 0.3 | 47.1 | 0.1 | |
| Follow-up time (y) | 4.4 | 0.4 | 5.0 | 0.3 | |
| Sex | |||||
| Male | 44% | 480 | 44% | 2400 | |
| Female | 56% | 602 | 56% | 3010 | |
| Birth country | |||||
| Sweden | 86% | 931 | 82% | 4409 | |
| Europe, except Sweden | 9% | 99 | 9% | 489 | |
| Other | 5% | 52 | 9% | 512 | |
| Education level | |||||
| Primary education | 19% | 201 | 13% | 721 | |
| Secondary education | 58% | 629 | 48% | 2571 | |
| Postsecondary/postgraduate education | 22% | 242 | 37% | 2026 | |
| Employment status | |||||
| Employed | 47% | 508 | 69% | 3734 | |
| Not employed | 53% | 574 | 31% | 1663 | |
| Income (000, €) | 21.6 | 0.6 | 26.6 | 0.8 | |
| Elixhauser comorbidity index | 0.8 | 0 | 0.3 | 0 | |
| Prior spine surgery | 23% | 253 | <1% | 7 | |
| Nonopioid pain medication usage (at least 1 dispensation in the prior 3 mo) | 45% | 484 | 5% | 282 | |
| Depression medicine usage (at least 1 dispensation in the prior 3 mo) | 46% | 494 | 8% | 411 | |
| Any opioid usage (at least 1 dispensation in the prior 3 mo) | 55% | 594 | 4% | 201 | |
| Strong opioid usage (at least 1 dispensation in the prior 3 mo) | 27% | 288 | 1% | 34 | |
| Weak opioid usage (at least 1 dispensation in the prior 3 mo) | 36% | 394 | 3% | 173 | |
SCS, spinal cord stimulation.
3.2. Trends in net disability days
Figure 1 shows the trend in crude (unadjusted) net disability days by period (before and after) and by group (SCS and control). Mean number of net disability days in the SCS group was 214 (standard deviation [SD] 142) in the before period and 194 days (SD 153) in the after period. In the reference group, mean net disability days was 33 (SD 93) and 34 (SD 94) in the before and after period, respectively. Net disability days increased slightly in the reference group (mean crude difference 1.2 days). A larger numerical change in net disability days was seen in the SCS group (mean crude difference −19.9 days).
Figure 1.
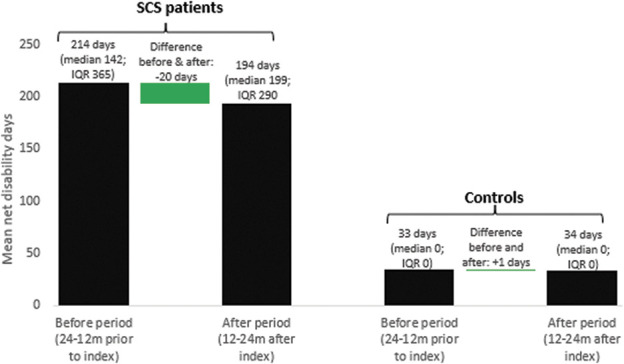
Mean crude number of net disability days in the before and after period by group. Green bars represent the difference in mean net disability days from before and after period. SCS, spinal cord stimulation.
As shown by the main regression findings in Table 2, on average, SCS reduced the net number of disability days by 21 days (P < 0.001). Without considering treatment status and period, higher age, more comorbidities, use of either strong or weak opioids, and more extensive usage of nonopioids and antidepressives were associated with a larger number of disability days (P < 0.001). Men and individuals with higher education level had fewer such days (P < 0.001).
Table 2.
Difference-in-difference (DiD) estimates of net disability days
| Variable | Coefficient (standard error) | Interaction effect of potential predictors and treatment effect (predictor* ) |
|---|---|---|
| SCS group effect, | 112.0 (5.8)*** | |
| Period effect, (1 if after index date, 0 if before index date) | 1.2 (0.7) | |
| Treatment effect, | −21.2 (4.1)*** | |
| Calendar year | −4.4 (0.4)*** | |
| Predictors | ||
| Age | 1.9 (0.1)*** | 0.6 (0.4) |
| Male | −11.0 (2.1)*** | −8.4 (8.2) |
| Comorbidities | 14.2 (1.7)*** | 5.0 (3.9) |
| Any opioid use 3 mo before index date | 44.9 (15.0)*** | 8.0 (11.1) |
| Use of strong opioid 3 mo before index date | −6.7 (13.2) | −15.1 (26.7) |
| Use of weak opioid 3 mo before index date | −23.9 (13.6)* | 12.8 (11.3) |
| Any nonopioid pain medicine use 3 mo before index date | 27.6 (5.4)*** | 6.0 (9.9) |
| Any antidepression medicine use 3 mo before index date | 40.8 (4.6)*** | 21.9 (9.4)*** |
| Previous spine surgery | −3.8 (8.1) | 16.0 (82.5) |
| Country of birth: Europe, not Sweden (ref: Sweden) | −5.2 (4.2) | −27.6 (15.6)* |
| Country of birth: other (ref: Sweden) | −37.0 (4.4)*** | −24.9 (20.7) |
| Education level: secondary school (ref: primary school) | −20.4 (4.0)*** | 11.8 (11.6) |
| Education level: postsecondary/postgraduate (ref: primary school) | −35.8 (4.0)*** | 7.6 (12.9) |
| Yearly income (€) | −1.4 (1.9) | 8.9 (20.0) |
| Unemployed | 73.5 (3.1)*** | 13.0 (8.2) |
Negative coefficient indicates decreased net disability days ***Significant on 1% level, **Significant on 5% level, *Significant on 10% level. Ref.=reference. SCS, spinal cord stimulation.
Interactions of potential predictors, group (SCS or control), and period (before or after index date) were used to identify the difference in treatment effect by predictor. Male patients and being born outside Europe were numerically associated with better treatment effect compared with other treated patients, although not statistically significant (P = 0.228). Higher age was numerically associated with poorer treatment effect (net disability days increased), although not statistically significant (P = 0.154). Similarly, the use of nonopioid pain medicine and any opioids was associated, but not statistically significantly, with a poorer treatment effect. Use of antidepression medicine was significantly associated with poorer treatment effect (P < 0.001). No statistically significant difference in treatment effect was seen whether patient had a prior spine surgery or not.
3.3. Indirect costs
The indirect cost associated with sick leave and disability pension were estimated by group and period using the two-part regression model (Table 3). Mean indirect cost decreased in the SCS groups from the before to the after period and slightly increased in the reference group. Mean saving of indirect costs on the treated SCS group was €4127.
Table 3.
Yearly indirect costs of spinal cord stimulation group and controls by period, predicted using two-part regression model.
| N | Before period (month 24-12 preceding index date), mean (standard error) | After period (month 24-12 after index date), mean (standard error) | Difference mean (standard error) | |
|---|---|---|---|---|
| SCS group | 1082 | 19,895 (652) | 16,146 (598) | −3749 (885) |
| Reference group | 5410 | 5352 (173) | 5729 (183) | 378 (252) |
| Difference | 14,543 (675) | 10,417 (625) | DiD: −4127/Patient |
SCS, spinal cord stimulation.
3.4. Sensitivity analyses
Sick leave and disability pension were modelled separately in a sensitivity analysis (Table 4). SCS was associated with a positive impact on sick leave (decreases net sick leave days, coef. −39.0, P < 0.01) but associated with a negative impact on disability pension (coef. 17.8, P < 0.01). Mean net disability days continuously increased in the SCS group from month 36 before index date until the month of the treatment start and thereafter continuously decreased until month 36 after index date (data not shown). In an additional sensitivity analysis, net disability days in month 25 to 36 after index date was compared with month 25 to 36 before index before in each group. This sensitivity analysis showed that SCS treatment had no impact on net disability days, indicating that in the third year after SCS, net disability days decreased to a similar level as 3 years before index date (coef. 1.2, P = 0.796, Table 4). This was also confirmed looking at trajectory of net disability days 36 months before and after index date (Supplementary Fig. 1, available at http://links.lww.com/PAIN/B699). Full regression model results for the sensitivity analyses are presented in Supplementary Table 2 to 4, available at http://links.lww.com/PAIN/B699.
Table 4.
Sensitivity analysis on difference-in-difference (DiD) estimates of net disability days
| Variable | Coefficient (standard error) |
|---|---|
| Sensitivity analysis: including only sick leave | |
| SCS group effect, | 73.8 (4.9)*** |
| Period effect, (1 if after index date, 0 if before index date) | 0.7 (0.7) |
| Treatment effect, | −39.0 (4.4)*** |
| Sensitivity analysis: including only disability pension | |
| SCS Group effect, | 38.2 (5.7)*** |
| Period effect, (1 if after index date, 0 if before index date) | 0.5 (0.5) |
| Treatment effect, | 17.8 (3.3)*** |
| Sensitivity analysis: total net disability days 3 y before index date compared with 3 y before index | |
| SCS Group effect, | 91.9 (5.9)*** |
| Period effect, (1 if after index date, 0 if before index date) | 0.5 (0.9) |
| Treatment effect, | 1.2 (4.6) |
Negative coefficient indicates decreased net disability days. SCS, spinal cord stimulation.
4. Discussion
4.1. Summary of results
This study aimed to analyse the association of potential predictors and the impact of SCS treatment on sick leave and disability pension in a representative Swedish cohort using nation-wide register data. Large loss of work ability prior to treatment was identified because patients had on average 214 net disability days 2 years prior to index date. This adds details to the existing literature about the impact that chronic pain has on the patient and the society at large in Sweden. SCS was associated with a statistically significantly reduction in net disability days. The estimated decrease in net disability days was 21 days per patient. The reduction in net disability days corresponded to a decrease in indirect costs of €4127 per patient. SCS could potentially lead to savings in societal costs related to disability days even when extrapolating the results to larger patient population. For 1000 patients treated during a year, this entails a potential reduction of this cost of around €4.1 million during only one year, although caution should be used when extrapolating results to years outside the study period. The total cost saving is expected to be higher because the effect on work ability is likely to persist over a longer period than what is studied here. However, to be able to estimate the cost-effectiveness of SCS treatment, the potential cost saving should be related to the additional treatment cost. In the future, the data presented in this study may be used together with other health and cost-related data, to fully estimate the cost-effectiveness of SCS treatment.
In this study, a reference group from the general population was included to rule out potential effects of societal changes that may impact the use of disability benefits. Given the retrospective register study design, patients in this study were not randomized to SCS treatment group or control group. Predictors that were deemed to have potential effect on disability days were included in the regression models to make the groups as comparable as possible, given the data availability. Nevertheless, there could be some unobservable factors that might explain the treatment effect. We consider it unlikely that the SCS patients would have improved (ie, disability days reduced) without the SCS treatment given that SCS is usually indicated for patients with intractable pain who have undergone several past treatments, such as spine surgery without improvement. Furthermore, a consistently high number of disability days was observed up to 3 years before treatment. This indicates that it is unlikely that the high number of disability days at baseline (treatment) is due to chance, and disability days at follow-up would regress to a lower value closer to a population average even without treatment (“regression to the mean”). Disregarding treatment and period in relation to index date, higher age, more comorbidities, more use of opioids, nonopioid pain medicines, and antidepression medicine were significantly associated with higher net disability days. Male sex was associated with less net disability days compared with female sex. This is consistent with previous findings of higher all-cause sick insurance use in women compared with men in Sweden but also in SCS-treated patients in Finland.16,22 Higher education level was also associated with less net disability days. No statistically significant difference in effect of treatment on disability days was detected for most of the potential predictors included in this study. However, the use of antidepression medicines was significantly associated with poorer treatment effect (P < 0.001). This relationship may be investigated in more detail in future studies. Previous studies using register data in Finland and Sweden, respectively, indicated that sick leave decreased after SCS in patients with prior spine surgery (ie, FBSS).15,16 Although this study indicate that net disability days decreased after SCS overall, no difference in treatment effect was seen in patients with prior spine surgery compared with those without prior spine surgery. This might be a surprising result given that SCS has been extensively evaluated for FBSS, which is the main indication for SCS. However, this result could potentially indicate that SCS for other indications may have a similar treatment effect as for FBSS, in this data material.
4.2. Study limitations
We acknowledge that our study has some limitations. Not all data on sick leave were available for analysis, which may underestimate sick leave days to some extent. Short-term episodes (≤14 days) are the responsibility of the employer in Sweden and are therefore not recorded in the Social Insurance Register. Thus, such episodes could not be included in the analyses. However, the first 14 days for all recorded sick leave episodes are recorded in the register. The findings of this study are based on Swedish data and are as such directly not transferable to other countries. In particular, differences in social insurance system between countries may entail different propensity to use such benefits, possibly entailing different impact of SCS on return to work. Another important limitation is the uncertainty in the estimated size of the SCS cohort. In this study, we included only patients based on having 2 consecutive codes for implantation registered within 100 days. Some clinics may perform the SCS procedure without a test simulation. Furthermore, some clinics may have a longer waiting time than 100 days. Therefore, the sample size might be underestimated. Explantations were not accounted for, which may to some extent overestimate the number of patients with permanent implants, to some extent.
4.3. Study strengths
Our study has some major strengths. This study relies to a large extent on Swedish register-based data, which are known to have a high degree of completeness ( 99%) Reporting of certain variables used in this study is not voluntary, so for healthcare visits and drug dispensations, all the necessary information can be expected to have been present. This entails that virtually all SCS procedures, all diagnoses, prescription drugs, and socioeconomic variables can be captured. The Swedish social security number allows following patients over time and allows data to be linked to other registers. Minimal exclusion criteria were applied; therefore, it could be argued that the study provides a more representative depiction of events in a real-world setting, compared with smaller, more selective studies.
5. Concluding remarks
In conclusion, this study demonstrates that SCS treatment in Sweden is associated with a decrease in disability days and consequently a decrease in indirect cost in working age patients. Large work loss prior to index date was also demonstrated, indicating a significant burden on the employers, the patient, and the society at large. The number of disability days varied considerably depending on the predictors age, sex, education, comorbidities, use of either strong or weak opioids, and use of nonopioids and antidepression medicines. The effect of SCS appeared to have little association with the chosen predictors, except for the use of antidepression medicines, which was the only predictor associated with a statistically significant treatment effect of SCS, indicating that the use of antidepressants may decrease the treatment effect of SCS. The economic benefit needs to be considered, as well as the clinical outcome, when evaluating the full societal value of SCS.
Conflict of interest statement
E. Söreskog, T. Jacobson, and F. Borgström are employees of Quantify Research providing consultancy services to medical device companies and other private and public organizations. T. Kirketeig has received lecturing fees and Uppsala University Hospital research grants from Abbott/St. Jude Medical.
Appendix A. Supplemental digital content
Supplemental digital content associated with this article can be found online at http://links.lww.com/PAIN/B699.
Supplementary Material
Acknowledgements
The study was financially supported by Abbott Inc., a company manufacturing and marketing SCS products. Some of the data in this study were summarized previously in a Licentiate thesis (“Spinal cord stimulation in chronic pain: A study of health outcomes and costs”, Karolinska Institutet, 2021). No data or analytical code will be made publicly available. The study was not preregistered.
Footnotes
Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article.
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal's Web site (www.painjournalonline.com).
Contributor Information
Trolle Jacobson, Email: Trolle.Jacobson@sironagroup.se.
Terje Kirketeig, Email: terje.kirketeig@surgsci.uu.se.
Peter Fritzell, Email: peterfritzell@gmail.com.
Rolf Karlsten, Email: Rolf.Karlsten@akademiska.se.
Niklas Zethraeus, Email: niklas.zethraeus@ki.se.
Fredrik Borgström, Email: fredrik.borgstrom@quantifyresearch.com.
References
- [1].Attal N, Lanteri-Minet M, Laurent B, Fermanian J, Bouhassira D. The specific disease burden of neuropathic pain: results of a French nationwide survey. PAIN 2011;152:2836–43. [DOI] [PubMed] [Google Scholar]
- [2].Bouhassira D, Lantéri-Minet M, Attal N, Laurent B, Touboul C. Prevalence of chronic pain with neuropathic characteristics in the general population. PAIN 2008;136:380–7. [DOI] [PubMed] [Google Scholar]
- [3].Breivik H, Collett B, Ventafridda V, Cohen R, Gallacher D. Survey of chronic pain in Europe: prevalence, impact on daily life, and treatment. Eur J Pain 2006;10:287. [DOI] [PubMed] [Google Scholar]
- [4].Chakravarthy K, Richter H, Christo PJ, Williams K, Guan Y. Spinal cord stimulation for treating chronic pain: reviewing preclinical and clinical data on paresthesia‐free high‐frequency therapy. Neuromodulation 2018;21:10–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Deer T, Pope J, Hayek S, Narouze S, Patil P, Foreman R, Sharan A, Levy R. Neurostimulation for the treatment of axial back pain: a review of mechanisms, techniques, outcomes, and future advances. Neuromodulation 2014;17:52–68. [DOI] [PubMed] [Google Scholar]
- [6].Diehr P, Yanez D, Ash A, Hornbrook M, Lin DY. Methods for analyzing health care utilization and costs. Annu Rev Public Health 1999;20:125–44. [DOI] [PubMed] [Google Scholar]
- [7].Doth AH, Hansson PT, Jensen MP, Taylor RS. The burden of neuropathic pain: a systematic review and meta-analysis of health utilities. PAIN 2010;149:338–44. [DOI] [PubMed] [Google Scholar]
- [8].Dworkin RH, O’connor AB, Backonja M, Farrar JT, Finnerup NB, Jensen TS, Kalso EA, Loeser JD, Miaskowski C, Nurmikko TJ. Pharmacologic management of neuropathic pain: evidence-based recommendations. PAIN 2007;132:237–51. [DOI] [PubMed] [Google Scholar]
- [9].Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care 1998;36:8–27. [DOI] [PubMed] [Google Scholar]
- [10].Finnerup NB, Attal N, Haroutounian S, McNicol E, Baron R, Dworkin RH, Gilron I, Haanpää M, Hansson P, Jensen TS. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol 2015;14:162–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Gustavsson A, Bjorkman J, Ljungcrantz C, Rhodin A, Rivano-Fischer M, Sjolund KF, Mannheimer C. Socio-economic burden of patients with a diagnosis related to chronic pain--register data of 840,000 Swedish patients. Eur J Pain 2012;16:289–99. [DOI] [PubMed] [Google Scholar]
- [12].Hayek SM, Veizi E, Hanes M. Treatment‐limiting complications of percutaneous spinal cord stimulator implants: a review of eight years of experience from an academic center database. Neuromodulation 2015;18:603–9. [DOI] [PubMed] [Google Scholar]
- [13].Imbens GW, Wooldridge JM. Recent developments in the econometrics of program evaluations. J Econ Lit 2009;47:5–86. [Google Scholar]
- [14].International Association for the Study of Pain (IASP). Terminology. Pain Terms: Neuropathic pain, 2017. Available at: https://www.iasp-pain.org/Education/Content.aspx?ItemNumber=1698. Accessed October 4, 2022. [Google Scholar]
- [15].Jonsson E, Hansson‐Hedblom A, Kirketeig T, Fritzell P, Hägg O, Borgström F. Cost and health outcomes patterns in patients treated with spinal cord stimulation following spine surgery—a register‐based study. Neuromodulation 2020;23:626–33. [DOI] [PubMed] [Google Scholar]
- [16].Kaijankoski H, Nissen M, Ikäheimo TM, von Und Zu Fraunberg M, Airaksinen E, Huttunen J. Effect of spinal cord stimulation on early disability pension in 198 failed back surgery syndrome patients: case-control study. Neurosurgery 2019;84:1225–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Kemler MA, de Vet HC, Barendse GA, van den Wildenberg FA, van Kleef M. Effect of spinal cord stimulation for chronic complex regional pain syndrome Type I: five-year final follow-up of patients in a randomized controlled trial. J Neurosurg 2008;108:292–8. [DOI] [PubMed] [Google Scholar]
- [18].Kemler MA, Reulen JP, Barendse GA, van Kleef M, de Vet HC, van den Wildenberg FA. Impact of spinal cord stimulation on sensory characteristics in complex regional pain syndrome type IA randomized trial. Anesthesiology 2001;95:72–80. [DOI] [PubMed] [Google Scholar]
- [19].Kumar K, Hunter G, Demeria D. Spinal cord stimulation in treatment of chronic benign pain: challenges in treatment planning and present status, a 22-year experience. Neurosurgery 2006;58:481–96. [DOI] [PubMed] [Google Scholar]
- [20].Kumar K, Taylor RS, Jacques L, Eldabe S, Meglio M, Molet J, Thomson S, O'Callaghan J, Eisenberg E, Milbouw G. Spinal cord stimulation versus conventional medical management for neuropathic pain: a multicentre randomised controlled trial in patients with failed back surgery syndrome. PAIN 2007;132:179–88. [DOI] [PubMed] [Google Scholar]
- [21].Langley PC, Van Litsenburg C, Cappelleri JC, Carroll D. The burden associated with neuropathic pain in Western Europe. J Med Econ 2013;16:85–95. [DOI] [PubMed] [Google Scholar]
- [22].Lidwall U, Bill S, Palmer E, Olsson Bohlin C. Mental disorder sick leave in Sweden: a population study. Work 2018;59:259–72. [DOI] [PubMed] [Google Scholar]
- [23].North RB, Kidd DH, Farrokhi F, Piantadosi SA. Spinal cord stimulation versus repeated lumbosacral spine surgery for chronic pain: a randomized, controlled trial. Neurosurgery 2005;56:98–106. discussion 106-7. [DOI] [PubMed] [Google Scholar]
- [24].Pope JE, Deer TR, Falowski S, Provenzano D, Hanes M, Hayek SM, Amrani J, Carlson J, Skaribas I, Parchuri. Multicenter retrospective study of neurostimulation with exit of therapy by explant. Neuromodulation 2017;20:543–52. [DOI] [PubMed] [Google Scholar]
- [25].Shealy CN, Mortimer JT, Reswick JB. Electrical inhibition of pain by stimulation of the dorsal columns: preliminary clinical report. Anesth Analg 1967;46:489–91. [PubMed] [Google Scholar]
- [26].Social Insurance Agency (Försäkringskassan). MiDAS, Sjukpenning och Rehabiliteringspenning (MiDAS, sick leave and rehabilitation compensations Report), 2011. Available at: https://www.forsakringskassan.se/wps/wcm/connect/f1e0dce5-e310-4d6d-8076-d4493534c10b/MiDAS_Sjukpenning_och_rehabiliteringspenning_Version_1_02.pdf?MOD=AJPERES. Accessed April 10, 2022. [Google Scholar]
- [27].Social Insurance Agency (Försäkringskassan). Vad kostar olika sjukdomar? Sjukpenningkostnader fördelade efter sjukskrivningsdiagnos [What is the cost of different diseases? Sick leave costs by diagnosis], 2002. Available at: https://www.forsakringskassan.se/wps/wcm/connect/43121e9e-6768-4fe8-a202-7a9d10b4e48c/redovisar_2002_02.pdf?MOD=AJPERES. Accessed April 10, 2022. [Google Scholar]
- [28].Statistics Sweden (Statistiska Centralbyrån). Genomsnittlig månadslön, kr efter Utbildningsnivå SUN 2000, kön och år [Average monthly wage in SEK by education level SUN 2000, sex and year], 2019. Available at: https://www.statistikdatabasen.scb.se/. Accessed April 10, 2022. [Google Scholar]
- [29].The Swedish Tax Agency (Skatteverket). Arbetsgivaravgifter [Payroll taxes], 2021. Available at: https://www.skatteverket.se/foretagochorganisationer/arbetsgivare/arbetsgivaravgifterochskatteavdrag/arbetsgivaravgifter.4.233f91f71260075abe8800020817.html. Accessed April 10, 2022. [Google Scholar]
- [30].Thomson SJ, Kruglov D, Duarte RV. A spinal cord stimulation service review from a single centre using a single manufacturer over a 7.5 year follow‐up period. Neuromodulation 2017;20:589–99. [DOI] [PubMed] [Google Scholar]
- [31].Tshomba Y, Psacharopulo D, Frezza S, Marone EM, Astore D, Chiesa R. Predictors of improved quality of life and claudication in patients undergoing spinal cord stimulation for critical lower limb ischemia. Ann Vasc Surg 2014;28:628–32. [DOI] [PubMed] [Google Scholar]
- [32].Van Buyten JP, Wille F, Smet I, Wensing C, Breel J, Karst E, Devos M, Pöggel‐Krämer K, Vesper J. Therapy‐related explants after spinal cord stimulation: results of an international retrospective chart review study. Neuromodulation 2017;20:642–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].van den Hout WB. The value of productivity: human-capital versus friction-cost method. Ann Rheum Dis 2010;69:i89–91. [DOI] [PubMed] [Google Scholar]
- [34].van Hecke O, Austin SK, Khan RA, Smith BH, Torrance N. Neuropathic pain in the general population: a systematic review of epidemiological studies. PAIN 2014;155:654–62. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental digital content associated with this article can be found online at http://links.lww.com/PAIN/B699.


