Abstract
Repellents are effective personal protective means against outdoor biting mosquitoes. Repellent formulations composed of EOs are finding increased popularity among consumers. In this study, after an initial screening of 11 essential oils (EOs) at the concentration of 33 μg/cm2, five of the most repellent EOs, Perovskia atriplicifolia, Citrus reticulata (fruit peels), C. reticulata (leaves), Mentha longifolia, and Dysphania ambrosioides were further investigated for repellent activity against Aedes aegypti mosquitoes in time span bioassays. When tested at the concentrations of 33 μg/cm2, 165 μg/cm2 and 330 μg/cm2, the EO of P. atriplicifolia showed the longest repellent effect up to 75, 90 and 135 min, respectively, which was followed by C. reticulata (peels) for 60, 90 and 120 min, M. longifolia for 45, 60 and 90 min, and C. reticulata (leaves) for 30, 45 and 75 min. Notably, the EO of P. atriplicifolia tested at the dose of 330 μg/cm2 showed complete protection for 60 min which was similar to the commercial mosquito repellent DEET. Gas chromatographic-mass spectrometric analyses of the EOs revealed camphor (19.7%), limonene (92.7%), sabinene (24.9%), carvone (82.6%), and trans-ascaridole (38.8%) as the major constituents of P. atriplicifolia, C. reticulata (peels), C. reticulata (leaves), M. longifolia, and D. ambrosioides, respectively. The results of the present study could help develop plant-based commercial repellents to protect humans from dengue mosquitoes.
Keywords: mosquito repellent, gas chromatography-mass spectrometry, bioactive compounds, Perovskia atriplicifolia, Citrus reticulata
1. Introduction
The yellow fever mosquito, Aedes aegypti L. (Diptera: Culicidae) is a vector of 54 different viruses and 2 species of Plasmodium causing dengue fever, chikungunya, zika, mayaro, yellow fever, and many other diseases [1]. The geographic distribution of Ae. aegypti is increasing rapidly and according to one estimate, half of the world’s population lives in areas where the environment has become suitable for yellow fever mosquitoes due to the abrupt climatic changes [2].
N,N-diethyl-meta-toluamide (DEET) is considered a gold-standard mosquito repellent for the protection of people against mosquito bites [3,4]. However, several studies have reported its adverse effects such as skin reactions, encephalopathies etc. upon extended usage [5,6,7]. Considering the toxic impact of DEET to humans, several plant species have been explored with the aim of finding natural compounds that could be used as an alternative to synthetic repellents. For example, Magnolia grandiflora [8], Mentha spicata [9], Citrus aurantifolia [10], Cymbopogon citratus [11], C. nardus [12], Dianthus caryophyllum [13] M. piperita [14], Nepeta cataria [15], Ferronia elephantum [16] and Carpesium abrotanoides [17] proved to be effective as repellents against Ae. aegypti. Moreover, the chemical constituents of a few EOs, such as thymol, carvacrol and α-terpinene [18], β-caryophyllene oxide [19], 1,8-cineole [20] and trans-nerolidol [17] have also been reported for their repellent effects against different mosquito species.
To find new plant-based sources of repellents for yellow fever mosquitoes, we have searched for plant species which showed biological activities against other insect pests but were not tested as repellents against Ae. aegypti. For example, Perovskia atriplicifolia has been reported as an antifeedant against Tribolium castaneum (Coleoptera: Tenebrionidae) [21] while Eucalyptus camaldulensis acted against Helicoverpa armigera (Lepidoptera: Noctuidae) [22]. Moreover, the fumigant toxicity of E. camaldulensis was also documented against Callosobruchus maculatus (Coleoptera: Chrysomelidae), Sitophilus oryzae (Coleoptera: Curculionidae) and T. castaneum [23]. The insecticidal and repellent activities of M. longifolia, C. reticulata and Dysphania ambrosioides were observed against Tribolium confusum (Coleoptera: Tenebrionidae), C. maculatus and Sitophilus zeamais (Coleoptera: Curculionidae) [24,25,26]. Therefore, we aimed to examine the repellent properties of EOs from P. atriplicifolia, C. reticulata, D. ambrosioides, E. camaldulensis, C. citratus, M. longifolia, Salvia moorcroftiana and Azadirachta indica plants against Ae. aegypti. Moreover, we identified the chemical composition of EOs that showed the highest repellent properties.
2. Results
2.1. Yield of Essential Oils
The aerial parts of M. longifolia and P. atriplicifolia were the richest in EO and yielded 1.33% and 1.21%, respectively, while the lowest amount (0.04%) of EO was produced from the bark of A. indica plants (Table 1).
Table 1.
Description of plants used and percent yield of the essential oils extracted by steam distillation process.
| Voucher | Plant Name | Abbreviation | Family | Growth Stage | Plant Part Used | Plant Collection Site | Yield % | |
|---|---|---|---|---|---|---|---|---|
| Coordinates | Elevation (m) | |||||||
| CUHA-225 | Dysphania ambrosioides | D. ambrosioides (I) | Amaranthaceae | Pre-flowering | Aerial parts | 34°12′11.0″ N 73°15′07.8″ E | 1200 | 0.09 |
| D. ambrosioides (II) | Fruiting | Aerial parts | 34°07′16.6″ N 73°19′54.73″ E | 1300 | 0.23 | |||
| CUHA-223 | Perovskia atriplicifolia | P. atriplicifolia | Lamiaceae | Pre-flowering | Aerial parts | 32°30′18.8″ N 69°45′00.3″ E | 1950 | 1.21 |
| CUHA-048 | Mentha longifolia | M. longifolia | Pre-flowering | Aerial parts | 34°07′20.5″ N 73°19′58.3″ E | 1300 | 1.33 | |
| CUHA-176 | Salvia moorcroftiana | S. moorcroftiana | Flowering | Flowers | 34°12′39.1″ N 73°18′38.4″ E | 1680 | 0.06 | |
| CUHA-227 | Azadirachta indica | A. indica | Meliaceae | Flowering | Bark | 30°16′06.6″ N 71°30′05.8″ E | 120 | 0.04 |
| CUHA-226 | Eucalyptus camaldulensis | E. camaldulensis (I) | Myrtaceae | Flowering | Leaves | 34°11′56.7″ N 73°14′37.1″ E | 1200 | 0.51 |
| E. camaldulensis (II) | Flower buds | 34°11′56.7″ N 73°14′37.1″ E | 1200 | 0.32 | ||||
| CUHA-228 | Cymbopogon citratus | C. citratus | Poaceae | Pre-flowering | Aerial parts | 31°28′19.0″ N 73°12′49.1″ E | 180 | 0.31 |
| CUHA-224 | Citrus reticulata | C. reticulata (I) | Rutaceae | Mature fruit stage | Leaves | 30°59′20.24″ N 72°53′21.84″ E | 170 | 0.15 |
| C. reticulata (II) | Fruit peels | 30°59′20.24″ N 72°53′21.84″ E | 170 | 0.29 | ||||
2.2. Chemical Composition of Essential Oils
EOs of P. atriplicifolia (PA-EO), C. reticulata (II) fruit peels (CR-EO (II)), M. longifolia (ML-EO), C. reticulata (I) leaves (CR-EO (I)), and D. ambrosioides aerial parts at a fruiting stage (II) (DA-EO (II)) showed the highest repellent activity, therefore the chemical composition was investigated by GC–MS (Table 2). The most abundant compounds in the PA-EO were camphor (19.7%), eucalyptol (12.1%) and limonene (10.9%). The ML-EO was rich in carvone which constituted 82.6% of the EO. The major compounds of CR-EO (I) EO were sabinene (24.9%), limonene (23.1%) and linalool (15.6%) whereas CR-EO (II) was dominated by limonene that composes 92.7% of the EO. The main components in the EO extracted from DA-EO (II) contained 28.2% α-terpinene, 15.8% p-cymene and 38.8% trans-ascaridole (Table 2).
Table 2.
Chemical composition of plant essential oils based on total ion chromatogram of GC-MS.
| Compound Name | RI ‡ | PA | ML | CR (I) | CR (II) | DA (II) | IM * |
|---|---|---|---|---|---|---|---|
| α-Thujene | 924 | 0.5 | 0.4 | MS, RI | |||
| α-Pinene | 928 | 3.8 | 0.1 | 1.5 | 0.5 | 0.3 | Std |
| Camphene | 943 | 3.8 | 0.1 | Std | |||
| Sabinene | 969 | 0.3 | 24.9 | 1.6 | MS, RI | ||
| β-Pinene | 972 | 2.3 | 0.1 | 1.8 | 0.2 | 0.6 | Std |
| 6-Methyl-5-heptene-2-one | 984 | 0.6 | MS, RI | ||||
| β-Myrcene | 988 | 0.7 | 0.1 | 2.9 | 2.5 | MS, RI | |
| 3-Octanol | 993 | 0.2 | MS, RI | ||||
| α-Phellandrene | 1002 | 0.1 | 0.1 | MS, RI | |||
| 3-Carene | 1008 | 0.4 | MS, RI | ||||
| α-Terpinene | 1015 | 0.3 | 1.1 | 0.1 | 28.2 | MS, RI | |
| p-Cymene | 1023 | 2.0 | 0.6 | 15.8 | MS, RI | ||
| Limonene | 1028 | 10.9 | 3.9 | 23.1 | 92.7 | 0.2 | Std |
| Eucalyptol | 1031 | 12.1 | 1.9 | 0.8 | Std | ||
| cis-β-Ocimene | 1036 | 0.2 | 0.1 | 1.1 | 0.3 | Std | |
| trans-β-Ocimene | 1047 | 0.1 | 4.9 | Std | |||
| γ-Terpinene | 1058 | 2.3 | 2.1 | 0.2 | 0.5 | MS, RI | |
| cis-Sabinene hydrate | 1066 | 0.3 | 0.1 | 0.2 | MS, RI | ||
| Terpinolene | 1088 | 0.3 | 0.6 | 0.1 | MS, RI | ||
| Dehydro-p-cymene | 1091 | 0.3 | MS, RI | ||||
| trans-Sabinene hydrate | 1098 | 0.2 | MS, RI | ||||
| Linalool | 1100 | 0.3 | 0.2 | 15.6 | 0.2 | Std | |
| cis-p-Menth-2-ene-1-ol | 1121 | 0.2 | 0.2 | MS, RI | |||
| Phenylacetonitrile | 1141 | 1.3 | MS, RI | ||||
| trans-p-Menth-2-ene-1-ol | 1142 | 0.2 | 1.6 | MS, RI | |||
| Camphor | 1147 | 19.7 | Std | ||||
| β-Citronellal | 1152 | 1.5 | 0.1 | MS, RI | |||
| Borneol | 1167 | 2.5 | 3.2 | Std | |||
| 4-Terpineol | 1179 | 0.3 | 0.2 | 5.1 | 0.2 | Std | |
| α-Terpineol | 1192 | 0.4 | 0.2 | 1.6 | 0.1 | Std | |
| 1,6-Dihydrocarveol | 1195 | 0.6 | MS, RI | ||||
| cis-Dihydrocarvone | 1196 | 1.4 | MS, RI | ||||
| trans-Carveol | 1219 | 0.3 | MS, RI | ||||
| cis-Geraniol | 1228 | 0.4 | MS, RI | ||||
| cis-Carveol | 1232 | 0.2 | MS, RI | ||||
| β-Citral | 1241 | 1.7 | MS, RI | ||||
| Carvone | 1247 | 82.6 | Std | ||||
| cis-Ascaridole | 1247 | 6.3 | MS, RI | ||||
| trans-Geraniol | 1254 | 0.2 | MS, RI | ||||
| Piperitone | 1256 | 0.2 | 1.5 | MS, RI | |||
| cis-Carvenone oxide | 1264 | 0.1 | 3.6 | MS, RI | |||
| α-Citral | 1270 | 2.2 | MS, RI | ||||
| trans-Carvone oxide | 1278 | 0.2 | MS, RI | ||||
| Bornyl acetate | 1288 | 4.2 | Std | ||||
| trans-Ascaridole | 1312 | 38.8 | MS, RI | ||||
| α-Terpinyl acetate | 1352 | 1.6 | MS, RI | ||||
| α-Copaene | 1381 | 0.4 | Std | ||||
| α-Gurjunene | 1416 | 0.2 | MS, RI | ||||
| trans-β-Caryophyllene | 1427 | 5.5 | 1.0 | 1.4 | 0.2 | MS, RI | |
| trans-α-Bergamotene | 1440 | 0.3 | MS, RI | ||||
| α-Humulene | 1461 | 6.3 | 0.2 | MS, RI | |||
| trans-β-Farnesene | 1458 | 0.2 | 0.2 | MS, RI | |||
| allo-Aromadendrene | 1468 | 1.1 | MS, RI | ||||
| Germacrene D | 1487 | 0.3 | MS, RI | ||||
| Viridiflorene | 1502 | 2.2 | MS, RI | ||||
| trans-α-Farnesene | 1509 | 0.2 | MS, RI | ||||
| β-Bisabolene | 1513 | 0.2 | MS, RI | ||||
| cis-Lachnophyllum ester | 1514 | 0.5 | MS, RI | ||||
| γ-Cadinene | 1520 | 0.6 | MS, RI | ||||
| δ-Cadinene | 1529 | 1.4 | MS, RI | ||||
| Caryophyllene oxide | 1588 | 1.6 | 1.0 | 0.1 | MS, RI | ||
| Ledol | 1600 | 8.1 | MS, RI | ||||
| δ-Cadinol | 1647 | 0.8 | MS, RI | ||||
| Monoterpenes | 28.0 | 4.3 | 65.0 | 98.4 | 45.8 | ||
| Oxygenated monoterpenes | 41.6 | 91.3 | 29.0 | 0.6 | 52.7 | ||
| Sesquiterpenes | 17.7 | 1.6 | 1.9 | 0.6 | 0.0 | ||
| Oxygenated sesquiterpenes | 10.5 | 1.0 | 0.1 | ||||
| Others | 0.2 | 2.4 | |||||
| Total Identified | 97.8 | 98.4 | 98.2 | 99.5 | 98.6 |
PA = Perovskia atriplicifolia; ML = Mentha longifolia; CR(I) = Citrus reticulata (fruit peels); CR(II) = C. reticulata (leaves); DA(II) = Dysphania ambrosioides (fruiting aerial parts). * In the identification method IM; MS = identification based on mass spectrum comparison with NIST-2008 library, RI = identification based on comparison of retention index with published data and Std = identification of compounds was made by comparing mass spectrum, retention index with published data as well as through the injection of standard compounds. ‡ Compounds listed are in order of elution from a DB-5 GC column. The retention index (RI) of a separated compound was calculated relative to C9–C26 retention time on the same parameter used for EOs analysis. The data shown in the table is the percentage compositions of different EOs where the values < 0.5% are approximate.
2.3. Repellency of Plant Essential Oils
In the screening bioassay, the numbers of female mosquito landings on the hand treated with different EOs or DEET were significantly lower (p < 0.001) than the negative control (solvent-treated hand). There were no mosquito landings on the hand treated with DEET, PA-EO and CR-EO (II) (Table 3).
Table 3.
Mosquito landings on human hand treated with different essential oils, DEET and negative control.
| Tested Substances | Average Number of Landings on Control (Negative) | Average Number of Landings on the Test Substance | p Value | df, t |
|---|---|---|---|---|
| DEET | 44.5 ± 1.76 | 0.00 ± 0.00 | <0.001 | 4, 25.21 |
| P. atriplicifolia | 35.33 ± 2.10 | 0.00 ± 0.00 | <0.001 | 4, 16.76 |
| C. reticulata (II) | 30.16 ± 1.13 | 0.00 ± 0.00 | <0.001 | 4, 26.51 |
| M. longifolia | 34.0 ± 0.93 | 3.16 ± 0.30 | <0.001 | 4, 29.47 |
| C. reticulata (I) | 35.00 ± 1.3 | 4.50 ± 0.22 | <0.001 | 4, 23.28 |
| D. ambrosioides (II) | 28.66 ± 0.84 | 9.66 ± 0.33 | <0.001 | 4, 19.67 |
| S. moorcroftiana | 37.16 ± 1.92 | 19.16 ± 0.60 | <0.001 | 4, 10.63 |
| D. ambrosioides (I) | 38.00 ± 1.90 | 22.00 ± 1.18 | <0.001 | 4, 12.92 |
| E. camaldulensis (II) | 37.00 ± 1.00 | 22.66 ± 0.84 | <0.001 | 4, 67.99 |
| E. camaldulensis (I) | 40.83 ± 1.30 | 30.33 ± 1.08 | <0.001 | 4, 8.53 |
| A. indica | 39.66 ± 2.52 | 34.16 ± 2.65 | <0.001 | 4, 9.77 |
| C. citratus | 46.00 ± 1.00 | 42.5 ± 1.17 | <0.001 | 4, 8.17 |
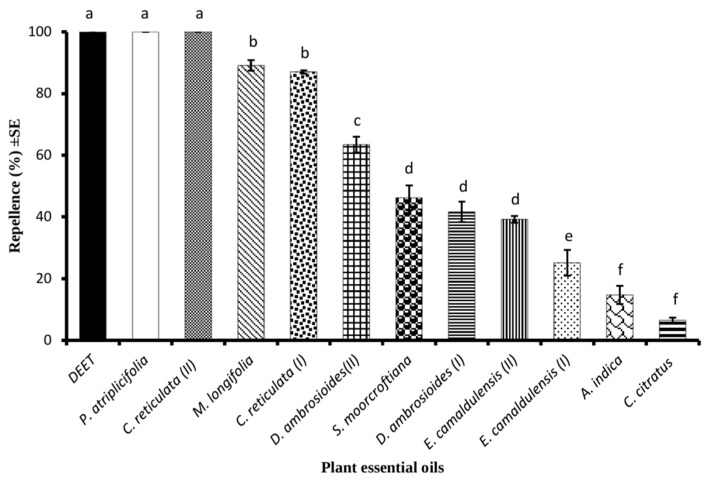
Out of eleven tested EOs, PA-EO and CA-EO (II) exhibited 100% of repellent effect which was similar (p > 0.05) to that of the positive control (DEET). The ML-EO and CR-EO (I) showed more than 85% of repellent effect whereas EO of C. citratus exhibited the lowest repellent effect of about 10% against Ae. aegypti females (Figure 1).
Figure 1.
Screening of eleven plant essential oils as repellents against Ae. aegypti females at the tested dose of 33 μg/cm2. Bars with different letters are significantly different (p < 0.05) from each other. Error bars on each column present standard error where n = 5.
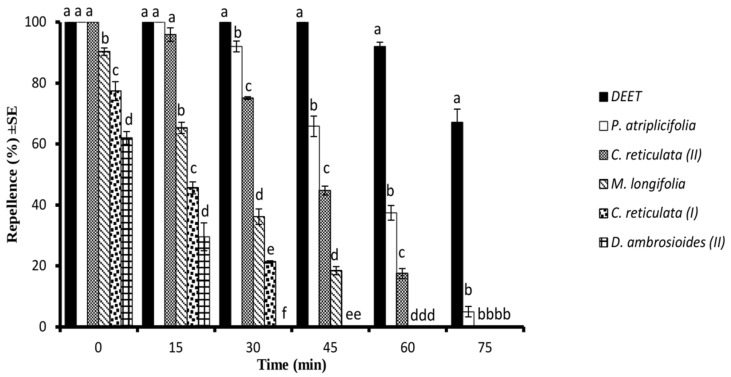
In the time span bioassay, the PA-EO and CR-EO (II) at a concentration of 33 µg/cm2 exhibited 100% of repellent effect against Ae. aegypti until 15 min after the treatment which was similar (p > 0.05) to the repellent effect displayed by DEET (positive control). Both of these EOs were active for 60 min. The DA-EO (II) showed the shortest repellent effect and was active only up to 15 min of post-treatment (Figure 2).
Figure 2.
Time span mosquito repellency of different essential oils and DEET at the tested concentration of 33 μg/cm2 against Ae. aegypti females. Different letters on the bars show a significant difference (p < 0.05) among different test substances at a specific period. Error bars on each column represent standard error where n = 5.
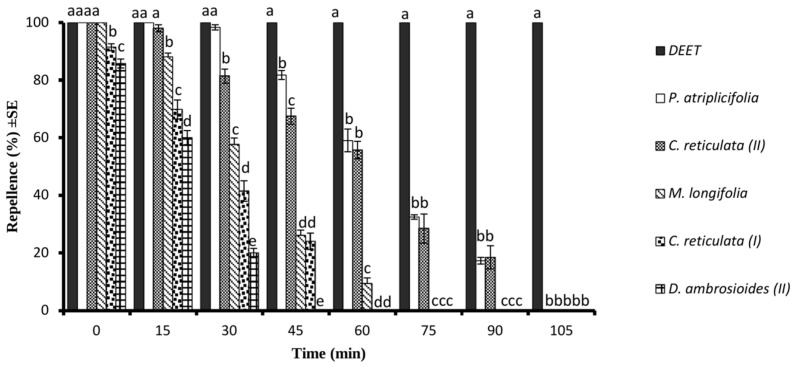
At the tested concentration of 165 μg/cm2, PA-EO showed repellent effect similar to that of DEET (p > 0.05) for 30 min whereas C. reticulata CR-EO (II) showed repellent effect of 100% for 15 min. However, both PA-EO and CR-EO (II) exhibited an active time span of 90 min. The ML-EO exhibited repellent effect of 100% only for a few minutes of application; after that it decreased with time and showed repellent effect of about 12% after 60 min post-treatment. The DA-EO (II) possessed the lowest repellent effect which lasted up to 30 min (Figure 3).
Figure 3.
Time span of mosquito repellent effect of different essential oils and DEET at the tested dose of 165 μg/cm2 against Ae. aegypti females. Different letters on the bars show a significant difference (p < 0.05) among different test substances at a specific period. Error bars on each column represent standard error where n = 5.
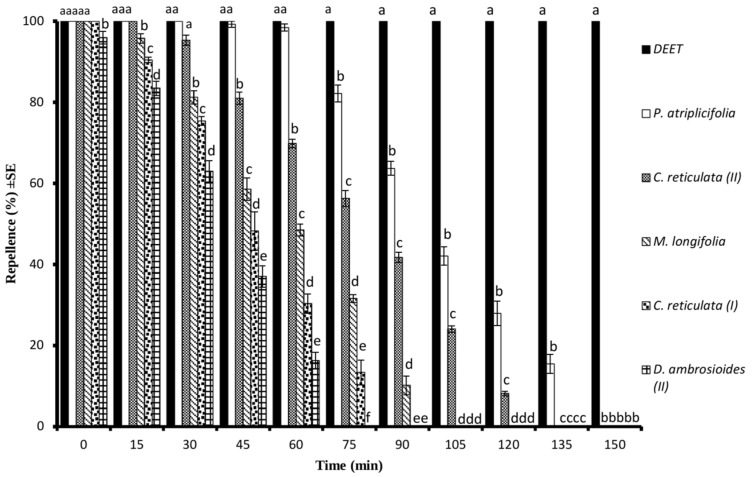
At the tested concentration of 330 μg/cm2, PA-EO showed complete protection similar to DEET (p > 0.05) for 60 min while CR-EO (II) exhibited complete protection for 30 min (Figure 4). The ML-EO showed complete protection immediately after application and the repellent effect lasted for 90 min. The highest time span of repellent effect, 135 min, was observed for PA-EO followed by the 120 min lasting repellent effect of CR-EO (II). The shortest repellence period of 60 min was observed for DA-EO (II) when tested at the highest dose of 330 μg/cm2 (Figure 4).
Figure 4.
Time span mosquito repellency of different essential oils and DEET at the tested dose of 330 μg/cm2 against Ae. aegypti females. Different letters on the bars show a significant difference (p < 0.05) among different test substances at a specific period. Error bars on each column represent standard error where n = 5.
3. Discussion
Repellent formulations composed of EOs are finding increased popularity among consumers and are generally considered environmentally safe compared to synthetic repellents. Five out of 11 EOs, tested in the current study at 1% concentration, exhibited a repellent effect of more than 60% against females of Ae. aegypti. The most active five EOs were further studied to check their repelling longevity. In the time span bioassay, all five EOs showed repellent effect of varying degrees and different active time spans when tested at different concentrations. The PA-EO proved the most effective repellent among all plant EOs at all tested concentrations. Moreover, the repelling effect of PA-EO was comparable to DEET for an extended period of time.
To the best of our knowledge, the PA-EO has not been previously tested against any mosquito species, however, a few studies reported the bioactivity of P. atriplicifolia EO against other insect pests. For example, P. atriplicifolia along with gamma radiation showed antifeedant [21] as well as insecticidal activity against the adults of T. castaneum [27]. There are some studies reporting bioactivities of other plant species of the genus Perovskia towards insects. For example, P. artemisioides showed repellent properties against Phthorimaea operculella (Lepidoptera: Gelechiidae) [28] and acted as a toxic fumigant against S. oryzae and T. castaneum [29].
In the current study, chemical analysis revealed that camphor, eucalyptol, limonene and α-humulene were the most abundant compounds in the PA-EO. The higher proportion of these compounds in PA-EO could be the reason for its prolonged repellent effect towards Ae. aegypti. Previously, camphor and camphor containing a fraction of Artemisia vulgaris were reported to repel Ae. aegypti females at 140 µg/cm2 concentration [30]. A study from Sweden presented results similar to our findings, that camphor-rich Tanacetum vulgare (Asterales: Asteraceae) EO showed 90% repellent activity against ticks [31]. Another study reported camphor as a repellent and toxic fumigant against Solenopsis invicta [32]. Likewise, eucalyptol, the second most abundant compound of P. atriplicifolia, was reported as a moderate antifeedant and oviposition deterrent against Ae. aegypti [20]. Moreover, eucalyptol was also found to be a toxic fumigant compound against An. sinensis [33]. In another study, the EO of Nepeta parnassica (Lamiales: Lamiaceae), having eucalyptol as the main constituent, displayed a good repellency against Ae. cretinus and Culex pipiens (Diptera: Culicidae) for 3 h and 2 h, respectively [34].
The chemical profiles of EOs are influenced by many factors such as the time of sample collection, the soil type and the climate. The chemical composition of PA-EO reported in the current study is qualitatively different to that from a previous study from Pakistan [35] whereas qualitatively similar but quantitatively different in a study reported from Iran [36]. The chemical composition of CR-EO II was different to that from a study conducted in India that presented EO comprised of 50% limonene [37]. The composition of ML-EO was quite different in a recent study from Pakistan [38] but similar to a study from Greece [39]. The composition of C. reticulata leaves CR-EO (I) was different to CR-EO (II) studied here as well as in a previous study that reported the chemical composition of C. reticulata leaves [40]. The harvesting stage of P. atriplicifolia reported in the current study and that of Dabiri and Sefidkon [36] were similar, however, the climate, soil type, and other factors were different, so a small difference in the chemical composition was found in both studies. The differences between the EOs reported by Erdemgil et al. [35] and the current study could be due to different climatic conditions and harvesting times. The variations found in the chemical profiles of EOs might be due to the different geographical regions from where the plant samples were collected as well as the physical condition of the samples.
We showed that CR-EO (II) was the second most effective repellent after P. atriplicifolia against Ae. aegypti. At the highest tested concentration, CR-EO (II) was active for 120 min, however, complete protection similar to DEET was observed only for a period of 30 min. Previously, the EO extracted from the fruit peel of C. reticulata was reported as a repellent against C. maculatus [24] and S. zeamais [41] while ethanol extracted from C. reticulata showed repellent activity against S. oryzae [42]. Moreover, the larvicidal activity of C. reticulata peels against Ae. aegypti is also documented [43]. In the current study, the most abundant compound identified from CR-EO (II) was limonene (92.7%). A number of previous studies showed the bioactivities of limonene against a number of organisms. For example, Türkoğlu et al. reported that limonene applied to cotton fabric has been used to avoid mosquito bites [44]. Moreover, limonene was documented as a repellent against Ae. aegypti females [45]. In another study, the larvicidal activity of R-(+)-limonene was reported against larvae of Ae. albopictus [46]. Therefore, the presence of limonene in the CR-EO (II) could strongly contribute to the repellent activity against Ae. aegypti. However, the shorter protection span exhibited by CR-EO could be due to the high volatility of limonene.
ML-EO showed repellent activity for periods ranging from 45 to 90 min and the most abundant compound of this EO was carvone. The chemical composition and repellent effect of ML-EO determined in current experiments are different to those reported in previous studies carried out in Pakistan and Saudi Arabia. According to a study from Pakistan, M. longifolia EO showed repellent effect for 90 min against Ae. aegypti at the concentration of 330 μg/cm2 while its major identified compound was piperitone oxide [47]. The study from Saudi Arabia revealed that the M. longifolia EO was active against C. pipiens and showed repellent effect for more than 43 min but at higher concentration of 1000 μg/cm2 [48] which was about three times higher than that used in current study.
The CR-EO (I) showed moderate repellent activity that lasted for a shorter period of time. Although limonene was present as the main compound in CR-EO (I), the repellent activity of the sample was far lower than that of the same plant as CR-EO (II). This might be due to the synergetic effect of other compounds as well as the lower proportion of limonene in CR-EO (I). The DA-EO (II) exhibited the shortest protection period compared to the other four most active EOs investigated in the current study. This oil did not show complete protection immediately after application even at the highest tested concentration. Our results are similar to the repellent activity of D. ambrosioides EO against Ae. aegypti which was reported by a study from Argentina [49]. Interestingly, the main compounds in DA-EO (II) harvested at the fruiting stage were trans-ascaridole, α-terpinene and p-cymene whereas α-terpinene (41.4%), germacrene D (16.2%) and p-cymene (14.7%) were identified as the major compounds of DA-EO (I) extracted from same plant species harvested at vegetative stage [50]. Thus, the difference in the bioactivity of DA-EOs (I) and (II) could be explained by their major constituents.
In the current study, no skin irritation/allergic reaction was observed on the treated area of any volunteer’s hand. Moreover, the EOs did not have an unpleasant smell. In our previous study, skin irritation and an irritating odor of EOs were reported [51]. Therefore, on the basis of the previous observation, skin sensitivity tests need to be performed before recommending EOs for commercial use. Our study revealed that EOs extracted from the aerial parts of P. atriplicifolia, M. longifolia and fruit peel of C. reticulata showed potential to be used to formulate plant-based mosquito repellent against females of Ae. aegypti.
4. Materials and Methods
4.1. Insect Rearing
Larvae of Ae. aegypti were obtained from the Dengue Control Unit, Railway Hospital, Multan and established in the laboratory under controlled conditions (25 ± 2 °C, R.H 65 ± 5% and photoperiod 12L:12D) at the Department of Entomology, Faculty of Agricultural Sciences & Technology, Bahauddin Zakariya University, Multan, Pakistan. Larvae were placed in plastic containers filled with 1 L of tap water. A fish diet (crude protein, 28%, crude fat 3%, crude fiber 4% with 10% moisture) was used to feed the larvae. Pupae were collected daily in plastic cups containing tap water and transferred to PLEXIGLAS® cages (30 × 30 × 30 cm) for adult emergence. Cotton soaked with 10% sucrose solution was placed in cages to feed the adults [19]. After 4–5 days, females were fed with blood using the immobilized pigeon method. A butter paper was placed on the inner side of the plastic jar filled with water and was kept in the adult cage as a substrate for oviposition. After oviposition, the butter paper with eggs was placed in the larval container filled with 1 L of tap water for mosquito hatching [9].
4.2. Collection of Plants
Different parts of plant species Perovskia atriplicifolia, Citrus reticulata, Dysphania ambrosioides, Eucalyptus camaldulensis, Cymbopogon citratus, Mentha longifolia, Salvia moorcroftiana and Azadirachta indica were collected from different areas of Pakistan. The parts of plants collected and their harvesting stage along with location coordinates and elevation are presented in Table 1. The identification of plant species was carried out by the plant taxonomist and the voucher specimens were submitted to the herbarium of the Department of Environmental Sciences, COMSATS University Islamabad, Abbottabad Campus, Abbottabad, Pakistan. The fresh plant material was either subjected to EO extraction on the same day of collection or stored in a freezer at −20 °C for 24–48 h until EO extraction.
4.3. Extraction of Essential Oils
The steam distillation method was used to extract the EO from the collected plant material as described in our previous publications [9,52]. A stainless-steel vessel (Liaqat Engineering Works, Faisalabad, Pakistan) was loaded with 2 kg of plant material and 2 L of distilled water. Water accumulated at the bottom of the steel vessel and had no direct contact with the plant material. The vessel was heated by an electric hot plate (Corning, NY, USA). Volatile compounds released from the plant materials along with steam were cooled down by using a condenser fitted on the head of the vessel and the distillate was collected in a separating funnel for 3 h. The collected distillate was extracted through liquid-liquid extraction using 210 mL (70 mL × 3) HPLC grade n-hexane (Daejung chemicals, Siheung, South Korea). The pooled hexane layers were dried by adding the anhydrous MgSO4 (Daejung chemicals, Siheung-si, South Korea)and filtered. The rotary evaporator (Buchi Labortechnik AG, Flawil, Switzerland) was used to evaporate the solvent at 25 °C under a vacuum. The obtained EO was weighed and the percentage yield of extracted EO was determined as described in our previous publication [53].
4.4. Chemical Analysis
Analysis of volatile compounds was carried out by using a HP 6890N gas chromatograph (GC) coupled with a HP 5973 mass spectrometer (MS) (Agilent Technologies Inc., Santa Clara, CA, USA). The GC was equipped with a DB-5 column (30 m length, 0.25 mm internal diameter, and 0.25 µm film coating comprised of 5% diphenyl and 95% dimethylpolysiloxane (Agilent, Santa Clara, CA, USA). The GC injector temperature was set at 235 °C while the GC oven temperature was maintained isothermally at 40 °C for 2 min, then increased at the rate of 4 °C/min up to 240 °C, and afterwards was kept isothermal for 8 min. Helium was used as a mobile phase with a constant flow of 1 mL/min. A diluted EO sample in n-hexane (Daejung chemicals, Siheung, South Korea) (500 ng/1 μL) was injected in a splitless mode set for 30 s. Electron ionization was performed at 70 eV where the ion source temperature was set constant at 180 °C. The mass spectra scan range was 30–400 amu. The total ion chromatogram peak area was used to find the percentage composition of each compound in an EO. EO components were identified by comparing their mass spectra with those present in the NIST-2008 MS library. Retention indices of separated compounds were determined relative to the retention times of a series of n-alkanes (C9–C24) (Merck, Darmstadt, Germany) analyzed at the same GC–MS parameters used for the EOs. Finally, the identified compounds were verified by injecting available standard compounds at the same parameters which were used for EOs analysis [9,53].
4.5. Repellency Bioassay
A human bait technique was used during the scotophase to test the repellence potential of EOs against Ae. aegypti females. Based on our previous experience, the 1%, 5%, and 10% solutions (10 mg/mL, 50 mg/mL, and 100 mg/mL,) of each EO and DEET were prepared using ethanol (Daejung chemicals, Siheung, South Korea) as a solvent. DEET (St. Louis, MO, USA) was used as a positive control. Twenty mated and blood-starved 4–5 days old female mosquitoes were released from the laboratory-reared colony in the experimental cage (30 × 30 × 30 cm). The hands of each subject were washed with scent-free liquid soap and allowed to dry for about 10 min before starting each test. Plastic gloves were used to cover each subject’s hand except for the 30 cm2 circular area on the dorsal side of the hand. An aliquot of 100 μL solution of test substance or pure solvent as a negative control was evenly applied on the exposed area of the hand and dried in air for three minutes before exposing the hand to mosquitoes. The hands of the subjects were exposed to the Ae. aegypti females in the experimental cage and mosquito landings, i.e., contact of the legs with the hand surface, were counted during the period of 5 min. The experiment was repeated randomly five times for both the test sample and the negative control. The human subjects (volunteers) were informed about the test procedure and consent was obtained before conducting repellency bioassays. The repellency percentage has been calculated by the formula presented by Azeem et al. [9]. Percentage repellency = [(Mc − Mt)/Mc] × 100 where Mc is the number of mosquito landings on the negative control (solvent) treated hand and Mt is the number of mosquito landings on the test substance treated hand.
4.6. Time Span Repellency Bioassay
Plant EOs that showed more than 60% repellency were further investigated to determine the maximum period of repellent activity. Time-span repellent bioassays were performed by following the same protocol as mentioned above in the repellency bioassay, except for the exposure of the same treated hand to the females of Ae. aegypti for 5 min after each 15 min time interval until the number of landings on control and treatment were the same. Time span bioassays were conducted by using test samples at the dosages of 33 μg/cm2, 165 μg/cm2 and 330 μg/cm2. The experiments were repeated five times and different females were employed for each replicate [17].
4.7. Statistical Analysis
Paired sample t-test was used to compare the number of landings of Ae. aegypti females on negative control and sample-treated hands. Statistical difference between the repellence of different EOs and DEET was analyzed by one-way ANOVA with post-hoc Bonferroni test. All the statistical tests were performed on Statistics 8 software (version 8.1, Tallahassee, FL, USA).
4.8. Ethical Approval
The Chairman of the Research Ethics and Biosafety Committee, Bahauddin Zakariya University, Multan, Pakistan provided the ethical approval.
5. Conclusions
The EO of P. atriplicifolia showed the best repellent activity of the EOs tested in our study. The complete protection by this EO from biting by females of Ae. aegypti lasted for more than 60 min and did not significantly differ from that of the gold-standard DEET. The EO of P. atriplicifolia could be further optimized, with the aim of developing an environmentally friendly and sustainable mosquito repellent formulation as an alternative to DEET.
Acknowledgments
This research was funded by International Foundation for Science (IFS), Sweden under grant number I-1-F-6041-1 to M.A. and by and by the Lithuanian state grant through Nature Research Centre, program 2 Climate and Eco-systems, Vilnius, Lithuania available to R.M. Tariq Zaman and Abdul Nazir are acknowledged for their help in collecting and identification of plants, respectively.
Author Contributions
M.A. and M.B. designed experiments, A.H., M.G.A., R.M. and M.M. performed experiments and collected data, M.A., R.M. and A.H. did data analysis and wrote the manuscript, and M.A. acquired funding. All authors contributed to finalizing the manuscript. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethical and Biosafety Committee of Bahauddin Zakariya University, Multan (protocol code No. 04 lURECl2022, approval date 21 September 2022).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of the compounds are available from the authors.
Funding Statement
This research work was financially supported by International Foundation for Science (IFS), Sweden under grant No. I-1-F-6041-1 to M.A. and by the Lithuanian state grant through Nature Research Centre, program 2 Climate and Ecosystems, Vilnius, Lithuania available to R.M.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.WRBU Walter Reed Biosystematics Unit (WRBU) Database. The USA. [(accessed on 1 November 2022)]. Available online: www.wrbu.si.edu.
- 2.Iwamura T., Guzman-Holst A., Murray K.A. Accelerating invasion potential of disease vector Aedes aegypti under climate change. Nat. Commun. 2020;11:1–10. doi: 10.1038/s41467-020-16010-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Debboun M., Frances S.P., Strickman D. Insect Repellents: Principles, Methods, and Uses. 1st ed. CRC Press; Boca Raton, FL, USA: 2006. [Google Scholar]
- 4.Mbuba E., Odufuwa O., Tenywa F., Philipo R., Tambwe M., Swai J.K., Moore J., Moore S. Single blinded semi-field evaluation of MAÏA® topical repellent ointment compared to unformulated 20% DEET against Anopheles gambiae, Anopheles arabiensis and Aedes aegypti in Tanzania. Malar. J. 2021;20:12. doi: 10.1186/s12936-020-03461-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gryboski J., Weinstein D., Ordway N.K. Toxic encephalopathy is apparently related to the use of an insect repellent. N. Engl. J. Med. 1961;264:289–291. doi: 10.1056/NEJM196102092640608. [DOI] [PubMed] [Google Scholar]
- 6.Lamberg S.I., Mulrennan J.A. Bullous Reaction to Diethyl Toluamide (DEET): Resembling a Blistering Insect Eruption. Arch. Dermatol. 1969;100:582–586. doi: 10.1001/archderm.1969.01610290066013. [DOI] [PubMed] [Google Scholar]
- 7.Windheuser J.J., Haslamx J.L., Caldwell L., Shaffer R.D. The use of N, N-diethyl-m-toluamide to enhance dermal and transdermal delivery of drugs. J. Pharm. Sci. 1982;71:1211–1213. doi: 10.1002/jps.2600711107. [DOI] [PubMed] [Google Scholar]
- 8.Ali A., Tabanca N., Demirci B., Raman V., Budel J.M., Baser K., Khan I.A. Insecticidal and biting deterrent activities of Magnolia grandiflora essential oils and selected pure compounds against Aedes aegypti. Molecules. 2020;25:1359. doi: 10.3390/molecules25061359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Azeem M., Zaman T., Tahir M., Haris A., Iqbal Z., Binyameen M., Nazir A., Shad S.A., Majeed S., Mozūraitis R. Chemical composition and repellent activity of native plants essential oils against dengue mosquito, Aedes aegypti. Ind. Crops Prod. 2019;140:111609. doi: 10.1016/j.indcrop.2019.111609. [DOI] [Google Scholar]
- 10.Soonwera M. Efficacy of essential oils from Citrus plants against mosquito vectors Aedes aegypti (Linn.) and Culex quinquefasciatus (Say) J. Agric. Technol. 2015;11:669–681. [Google Scholar]
- 11.Sritabutra D., Soonwera M., Waltanachanobon S., Poungjai S. Evaluation of herbal essential oil as repellents against Aedes aegypti (L.) and Anopheles dirus Peyton & Harrison. Asian Pac. J. Trop. Biomed. 2011;1:124–128. [Google Scholar]
- 12.Phasomkusolsil S., Soonwera M. Insect repellent activity of medicinal plant oils against Aedes aegypti (Linn.), Anopheles minimus (Theobald) and Culex quinquefasciatus Say based on protection time and biting rate. Southeast Asian J. Trop. Med. Public Health. 2010;41:831–840. [PubMed] [Google Scholar]
- 13.Tunón H., Thorsell W., Mikiver A., Malander I. Arthropod repellency, especially tick (Ixodes ricinus), exerted by extract from Artemisia abrotanum and essential oil from flowers of Dianthus caryophyllum. Fitoterapia. 2006;77:257–261. doi: 10.1016/j.fitote.2006.02.009. [DOI] [PubMed] [Google Scholar]
- 14.Kumar S., Wahab N., Warikoo R. Bioefficacy of Mentha piperita essential oil against dengue fever mosquito Aedes aegypti L. Asian Pac. J. Trop. Biomed. 2011;1:85–88. doi: 10.1016/S2221-1691(11)60001-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Reichert W., Ejercito J., Guda T., Dong X., Wu Q., Ray A., Simon J.E. Repellency assessment of Nepeta cataria essential oils and isolated nepetalactones on Aedes aegypti. Sci. Rep. 2019;9:1524. doi: 10.1038/s41598-018-36814-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Venkatachalam M., Jebanesan A. Repellent activity of Ferronia elephantum Corr. (Rutaceae) leaf extract against Aedes aegypti (L.) Bioresour. Technol. 2001;76:287–288. doi: 10.1016/S0960-8524(00)00096-1. [DOI] [PubMed] [Google Scholar]
- 17.Haris A., Azeem M., Binyameen M. Mosquito Repellent Potential of Carpesium abrotanoides Essential Oil and Its Main Components Against a Dengue Vector, Aedes aegypti (Diptera: Culicidae) J. Med. Entomol. 2022;59:801–809. doi: 10.1093/jme/tjac009. [DOI] [PubMed] [Google Scholar]
- 18.Choi W.-S., Park B.-S., Ku S.-K., Lee S.-E. Repellent activities of essential oils and monoterpenes against Culex pipiens pallens. J. Am. Mosq. Control Assoc. 2002;18:348–351. [PubMed] [Google Scholar]
- 19.Nararak J., Sathantriphop S., Kongmee M., Mahiou-Leddet V., Ollivier E., Manguin S., Chareonviriyaphap T. Excito-repellent activity of β-caryophyllene oxide against Aedes aegypti and Anopheles minimus. Acta Trop. 2019;197:1–8. doi: 10.1016/j.actatropica.2019.05.021. [DOI] [PubMed] [Google Scholar]
- 20.Klocke J.A., Darlington M.V., Balandrin M.F. 1, 8-Cineole (Eucalyptol), a mosquito feeding and ovipositional repellent from volatile oil of Hemizonia fitchii (Asteraceae) J. Chem. Ecol. 1987;13:2131–2141. doi: 10.1007/BF01012562. [DOI] [PubMed] [Google Scholar]
- 21.Ahmadi M., Moharramipour S., Abd Alla A. Antifeedant effect of gamma radiation and Perovskia atriplicifolia essential oil combination against Tribolium castaneum (Coleoptera: Tenebrionidae) J. Crop Prot. 2015;4:463–475. [Google Scholar]
- 22.Kathuria V., Kaushik N. Feeding inhibition of Helicoverpa armigera (Hübner) by Eucalyptus camaldulensis and Tylophora indica extracts. Insect Sci. 2005;12:249–254. doi: 10.1111/j.1005-295X.2005.00031.x. [DOI] [Google Scholar]
- 23.Negahban M., Moharramipour S. Fumigant toxicity of Eucalyptus intertexta, Eucalyptus sargentii and Eucalyptus camaldulensis against stored-product beetles. J. Appl. Entomol. 2007;131:256–261. doi: 10.1111/j.1439-0418.2007.01152.x. [DOI] [Google Scholar]
- 24.Saeidi M., Moharramipour S., Sefidkon F., Aghajanzadeh S. Insecticidal and repellent activities of Citrus reticulata, Citrus limon and Citrus aurantium essential oils on Callosobruchus maculatus. Integr. Prot. Stored Prod. IOBC/WPRS Bull. 2011;69:289–293. [Google Scholar]
- 25.Saeidi M., Moharramipour S. Insecticidal and repellent activities of Artemisia khorassanica, Rosmarinus officinalis and Mentha longifolia essential oils on Tribolium confusum. J. Crop Prot. 2013;2:23–31. [Google Scholar]
- 26.Langsi D., Nukenine E., Fokunang C., Suh C., Goudoungou W. Potentials of essential oils of Chenopodium ambrosioides L. and Cupressus sempervirens L. against stored maize pest, Sitophilus zeamais Motschulsky. J. Entomol. Zool. Stud. 2017;5:309–313. [Google Scholar]
- 27.Ahmadi M., Abdalla A.M.M., Moharramipour S. Combination of gamma radiation and essential oils from medicinal plants in managing Tribolium castaneum contamination of stored products. Appl. Radiat. Isot. 2013;78:16–20. doi: 10.1016/j.apradiso.2013.03.012. [DOI] [PubMed] [Google Scholar]
- 28.Sadeghi Z., Alizadeh Z., Khorrami F., Norouzi S., Moridi Farimani M. Insecticidal activity of the essential oil of Perovskia artemisioides Boiss. Nat. Prod. Res. 2021;35:5929–5933. doi: 10.1080/14786419.2020.1803311. [DOI] [PubMed] [Google Scholar]
- 29.Arabi F., Moharramipour S., Sefidkon F. Chemical composition and insecticidal activity of essential oil from Perovskia abrotanoides (Lamiaceae) against Sitophilus oryzae (Coleoptera: Curculionidae) and Tribolium castaneum (Coleoptera: Tenebrionidae) Int. J. Trop. Insect Sci. 2008;28:144–150. doi: 10.1017/S1742758408079861. [DOI] [Google Scholar]
- 30.Hwang Y.-S., Wu K.-H., Kumamoto J., Axelrod H., Mulla M.S. Isolation and identification of mosquito repellents in Artemisia vulgaris. J. Chem. Ecol. 1985;11:1297–1306. doi: 10.1007/BF01024117. [DOI] [PubMed] [Google Scholar]
- 31.Pålsson K., Jaenson T.G., Bæckström P., Borg-Karlson A.-K. Tick repellent substances in the essential oil of Tanacetum vulgare. J. Med. Entomol. 2008;45:88–93. doi: 10.1093/jmedent/45.1.88. [DOI] [PubMed] [Google Scholar]
- 32.Fu J., Tang L., Li W., Wang K., Cheng D., Zhang Z. Fumigant toxicity and repellence activity of camphor essential oil from Cinnamonum camphora Siebold against Solenopsis invicta workers (Hymenoptera: Formicidae) J. Insect Sci. 2015;15:129–134. doi: 10.1093/jisesa/iev112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Luo D.-Y., Yan Z.-T., Che L.-R., Zhu J.J., Chen B. Repellency and insecticidal activity of seven Mugwort (Artemisia argyi) essential oils against the malaria vector Anopheles sinensis. Sci. Rep. 2022;12:5337. doi: 10.1038/s41598-022-09190-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gkinis G., Michaelakis A., Koliopoulos G., Ioannou E., Tzakou O., Roussis V. Evaluation of the repellent effects of Nepeta parnassica extract, essential oil, and its major nepetalactone metabolite against mosquitoes. Parasitol. Res. 2014;113:1127–1134. doi: 10.1007/s00436-013-3750-3. [DOI] [PubMed] [Google Scholar]
- 35.Erdemgil F.Z., Ilhan S., Korkmaz F., Kaplan C., Mercangöz A., Arfan M., Ahmad S. Chemical composition and biological activity of the essential oil of Perovskia atriplicifolia. from Pakistan. Pharm. Biol. 2007;45:324–331. doi: 10.1080/13880200701212890. [DOI] [Google Scholar]
- 36.Dabiri M., Sefidkon F. Analysis of the essential oil from aerial parts of Perovskia atriplicifolia Benth. at different stages of plant growth. Flavour Fragr. J. 2001;16:435–438. doi: 10.1002/ffj.988. [DOI] [Google Scholar]
- 37.Goyal L., Kaushal S. Evaluation of chemical composition and antioxidant potential of essential oil from Citrus reticulata fruit peels. Adv. Res. 2018;15:1–12. doi: 10.9734/AIR/2018/41981. [DOI] [Google Scholar]
- 38.Abbas M.G., Haris A., Binyameen M., Nazir A., Mozūratis R., Azeem M. Chemical composition, larvicidal and repellent activities of wild plant essential oils against Aedes aegypti. Biology. 2022;12:8. doi: 10.3390/biology12010008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Koliopoulos G., Pitarokili D., Kioulos E., Michaelakis A., Tzakou O. Chemical composition and larvicidal evaluation of Mentha, Salvia, and Melissa essential oils against the West Nile virus mosquito Culex pipiens. Parasitol. Res. 2010;107:327–335. doi: 10.1007/s00436-010-1865-3. [DOI] [PubMed] [Google Scholar]
- 40.Lawal O.A., Ogunwande I.A., Owolabi M.S., Giwa-Ajeniya A.O., Kasali A.A., Abudu F.A., Sanni A.A., Opoku A.R. Comparative analysis of essential oils of Citrus aurantifolia Swingle and Citrus reticulata Blanco, from two different localities of Lagos State, Nigeria. Am. J. Essent. Oils Nat. Prod. 2014;2:8–12. [Google Scholar]
- 41.Fouad H.A., da Camara C.A. Chemical composition and bioactivity of peel oils from Citrus aurantiifolia and Citrus reticulata and enantiomers of their major constituent against Sitophilus zeamais (Coleoptera: Curculionidae) J. Stored Prod. Res. 2017;73:30–36. doi: 10.1016/j.jspr.2017.06.001. [DOI] [Google Scholar]
- 42.Akhtar M., Arshad M., Raza A.B.M., Chaudhary M.I., Iram N., Akhtar N., Mahmood T. Repellent effects of certain plant extracts against rice weevil, Sitophilus oryzae L.(Coleoptera: Curculionidae) Int. J. Agric. Appl. Sci. 2013;5:69–73. [Google Scholar]
- 43.Sutthanont N., Choochote W., Tuetun B., Junkum A., Jitpakdi A., Chaithong U., Riyong D., Pitasawat B. Chemical composition and larvicidal activity of edible plant-derived essential oils against the pyrethroid-susceptible and -resistant strains of Aedes aegypti (Diptera: Culicidae) J. Vector Ecol. 2010;35:106–115. doi: 10.1111/j.1948-7134.2010.00066.x. [DOI] [PubMed] [Google Scholar]
- 44.Türkoğlu G.C., Sarıışık A.M., Erkan G., Yıkılmaz M.S., Kontart O. Micro-and nano-encapsulation of limonene and permethrin for mosquito repellent finishing of cotton textiles. Iran. Polym. J. 2020;29:321–329. doi: 10.1007/s13726-020-00799-4. [DOI] [Google Scholar]
- 45.Nematollahi N., Ross P.A., Hoffmann A.A., Kolev S.D., Steinemann A. Limonene Emissions: Do Different Types Have Different Biological Effects? Int. J. Environ. Res. Public Health. 2021;18:10505. doi: 10.3390/ijerph181910505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Giatropoulos A., Papachristos D.P., Kimbaris A., Koliopoulos G., Polissiou M.G., Emmanouel N., Michaelakis A. Evaluation of bioefficacy of three Citrus essential oils against the dengue vector Aedes albopictus (Diptera: Culicidae) in correlation to their components enantiomeric distribution. Parasitol. Res. 2012;111:2253–2263. doi: 10.1007/s00436-012-3074-8. [DOI] [PubMed] [Google Scholar]
- 47.Iqbal S., Khan F.A., Haris A., Mozuratis R., Binyameen M., Azeem M. Essential oils of four wild plants inhibit the blood seeking behaviour of female Aedes aegypti. Exp. Parasitol. 2023;244:108424. doi: 10.1016/j.exppara.2022.108424. [DOI] [PubMed] [Google Scholar]
- 48.Al-Sarar A. Chemical Composition, Adulticidal and Repellent Activity of Essential Oils From Mentha longifolia L. and Lavandula dentata L. against Culex pipiens L. J. Plant Prot. Pathol. 2014;5:817–826. doi: 10.21608/jppp.2014.87995. [DOI] [Google Scholar]
- 49.Gillij Y., Gleiser R., Zygadlo J. Mosquito repellent activity of essential oils of aromatic plants growing in Argentina. Bioresour. Technol. 2008;99:2507–2515. doi: 10.1016/j.biortech.2007.04.066. [DOI] [PubMed] [Google Scholar]
- 50.Azeem M., Zaman T., Abbasi A.M., Abid M., Mozūratis R., Alwahibi M.S., Elshikh M.S. Pesticidal potential of some wild plant essential oils against grain pests Tribolium castaneum (Herbst, 1797) and Aspergillus flavus (Link, 1809) Arab. J. Chem. 2022;15:103482. doi: 10.1016/j.arabjc.2021.103482. [DOI] [Google Scholar]
- 51.Barnard D.R. Repellency of essential oils to mosquitoes (Diptera: Culicidae) J. Med. Entomol. 1999;36:625–629. doi: 10.1093/jmedent/36.5.625. [DOI] [PubMed] [Google Scholar]
- 52.Kumar S., Ahmad R., Saeed S., Azeem M., Mozūraitis R., Borg-Karlson A.-K., Zhu G. Chemical composition of fresh leaves headspace aroma and essential oils of four Coriander cultivars. Front. Plant Sci. 2022;13:1–9. doi: 10.3389/fpls.2022.820644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Azeem M., Iqbal Z., Emami S.N., Nordlander G., Nordenhem H., Mozūratis R., El-Seedi H.R., Borg-Karlson A.K. Chemical composition and antifeedant activity of some aromatic plants against pine weevil (Hylobius abietis) Ann. Appl. Biol. 2020;177:121–131. doi: 10.1111/aab.12586. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.