Abstract
Objectives:
Abdominal aortic aneurysm ultrasound surveillance varies between hospitals in the United Kingdom. University Hospitals Bristol and Weston adopt a 6-monthly surveillance interval for 4.5–4.9 cm abdominal aortic aneurysm, which is a deviation from nationally recommended 3-monthly intervals. Assessment of abdominal aortic aneurysm growth rate, and the concurrent impact of abdominal aortic aneurysm risk factors and medications prescribed for risk factors, may inform whether this change in surveillance intervals is safe and appropriate.
Methods:
This analysis was conducted retrospectively. A total of 1312 abdominal aortic aneurysm ultrasound scans from 315 patients between January 2015 and March 2020 were split into 0.5 cm groups, ranging from 3.0 to 5.5 cm. Abdominal aortic aneurysm growth rate was assessed with one-way analysis of variance. The impact of risk factors and risk factor medication on abdominal aortic aneurysm growth rate was analysed using multivariate and univariate linear regression and Kruskal–Wallis tests. Patient cause of death among surveillance patients was recorded.
Results:
Abdominal aortic aneurysm growth rate was significantly associated with increased abdominal aortic aneurysm diameter (p < 0.001). There was a significant whole-group reduction in growth rate from 0.29 to 0.19 cm/year in diabetics compared to non-diabetics (p = 0.02), supported by univariate linear regression (p = 0.04). In addition, gliclazide patients had lower growth rate compared to patients not on the medication (p = 0.04). One abdominal aortic aneurysm rupture occurred <5.5 cm resulting in death.
Conclusion:
Abdominal aortic aneurysm measuring 4.5–4.9 cm had a mean growth rate of 0.3 cm/year (± 0.18 cm/year). Therefore, mean growth rate and variability suggest patients are unlikely to surpass surgical threshold of 5.5 cm between the 6-monthly surveillance scans, supported by low rupture rates. This suggests the surveillance interval for 4.5–4.9 cm abdominal aortic aneurysm is a safe and appropriate deviation from national guidance. In addition, it may be pertinent to consider diabetic status when designing surveillance intervals.
Keywords: AAA, national guidance, surveillance interval, risk factors
Introduction
Abdominal aortic aneurysms (AAAs) carry a substantial burden on healthcare systems in developed countries. 1 Prior to ultrasound screening, AAAs were often identified as incidental findings, secondary to alternative imaging. 2 Consequently, there were large numbers of undiagnosed AAAs rupturing. 3 However, since the birth of ultrasound screening and surveillance, rupture-related mortality has significantly declined from 10.2 to 7.5 deaths per 100,000 in 2009 and 2017, respectively. 4
Despite reductions in AAA ruptures, the mechanisms that drive aneurysmal growth remain unclear. Thompson et al. 5 reported linear increases in growth rate (GR) with increased AAA diameter. They also reported that GR was influenced by cardiovascular, environmental and genetic risk factors (RFs). These include age, male sex, hypercholesterolaemia, hypertension and smoking. Smoking is suspected to have the greatest impact on AAA growth. 6 Smoking and hypertension are also linked to direct injury to the endothelium through toxins and high shear stress, respectively. Paradoxically, diabetic patients have lower prevalence of AAA and reduced GR. 7
Medications to treat RFs, for example metformin and statins, are associated with significantly reduced aneurysmal expansion.8,9 This relationship between RFs/medications and AAA GR has supported calls for their joint consideration when designing AAA surveillance intervals. β-blockers, such as propranolol, have shown to impede mechanisms for AAA growth, yet do not appear to impact GR. 10 This lack of relationship between propranolol and GR may justify no comparison of other β-blockers. However, bisoprolol, which is the most commonly prescribed β-blocker for surveillance patients at this Trust (University Hospitals Bristol and Weston (UHBW)), implements alternate mechanisms of action to treat hypertension. These cardio-selective β-1 blocking actions have been associated with reduced GR previously; 11 therefore, it is pertinent to evaluate this hypertensive medication.
Some hospitals in the United Kingdom have tailored surveillance intervals to suit their own evaluated GRs.12,13 UHBW have different surveillance intervals, with 4.5–4.9 cm AAA moving to 6-monthly scans rather than the recommended 3-month interval (Table 1). Aberrations from national guidance seen in the United Kingdom may be due to the lack of consensus surrounding published domestic14–16 and international criteria (Table 1). UHBW made this non-evidence-based deviation from national guidance, but its safety concerns have never been formally assessed, warranting detailed assessment of GRs. In addition, there have been no local considerations as to whether RFs/medications may be a risk/benefit to AAA progression, despite the abundance of literature surrounding these relationships. Their consideration may provide insight for appropriately set surveillance intervals and improve patient management.
Table 1.
University Hospitals Bristol and Weston and nationally recommended AAA surveillance intervals compared to other countries.
| Country | Diameter bands, cm | Surveillance interval, months |
|---|---|---|
| United Kingdom (UHBW) | 3.0–4.4 | 12 |
| 4.5–4.9 | 6 | |
| 5.0–5.4 | 3 | |
| United Kingdom (current) | 3.0–4.4 | 12 |
| 4.5–5.4 | 3 | |
| United Kingdom (proposed NICE changes) | 3.0–4.4 | 24 |
| 4.5–5.4 | 3 | |
| Denmark | 3.0–5.5 | 12 |
| Italy | 3.0–3.9 | 12 |
| 4.0–4.9 | 6 | |
| New Zealand | 3.0–5.5 | 12 |
| Norway | 3.0–3.9 | 24 |
| 4.0–4.5 | 12 | |
| 4.5–5.5 | 3–6 | |
| Sweden | 2.5–2.9 | 60 |
| 3.0–3.9 | 24 | |
| 4.0–4.4 | 12 | |
| 4.5–5.0 | 6 | |
| 5.1–5.4 | 3 | |
| United States | 2.5–2.9 | 60 |
| 3.0–3.4 | 36 | |
| 3.5–4.4 | 12 | |
| 4.5–5.4 | 6 |
AAA: abdominal aortic aneurysm; NICE: The National Institute of Health and Care Excellence; UHBW: University Hospitals Bristol and Weston NHS Foundation Trust.
The aims of this study therefore are (1) to assess GRs of all AAAs enrolled on the UHBW surveillance programme to inform whether the current changes made to the surveillance intervals at UHBW are safe and appropriate. UHBW AAA-related mortality will also be assessed for supporting information. (2) To evaluate the impact of RFs and their prescribed medications to determine their significance on GR.
Methods
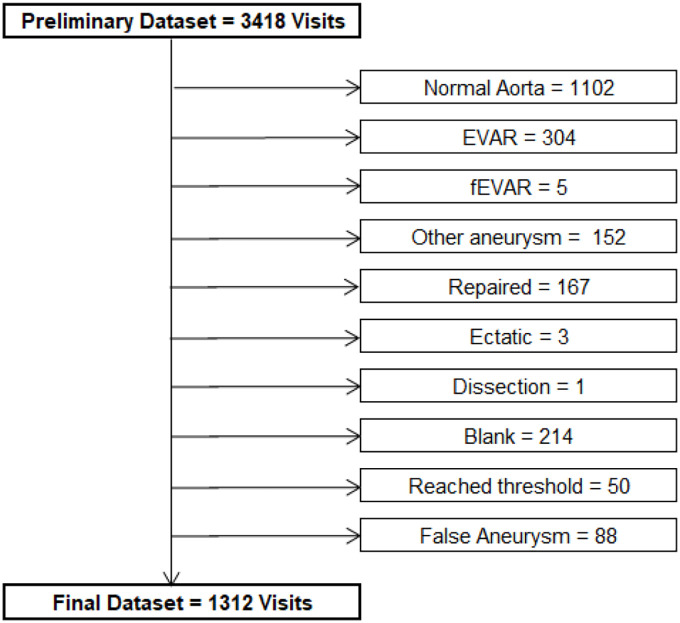
Data included in this analysis were examined in a retrospective manner. All patients who received a diagnostic ultrasound scan for the presence of AAA at UHBW, and were enrolled into the surveillance programme between January 2015 and March 2020, were included in the preliminary dataset. Data were extracted from a local department database in which all patient visits to UHBW were recorded. The preliminary dataset contained 3418 separate AAA scans in 710 patients, which reduced to 1312 AAA scans in 315 patients after appropriate exclusions were made. As any referral to UHBW querying aneurysm/hematoma is stored in the department database, referrals not specific to AAA were included in the preliminary data extraction. A strict exclusion criterion was developed to identify ‘true AAA’ patients between 3.0 and 5.5 cm (Figure 1). Referrals which did not query AAA, and those which did but the aortas were considered to be of normal calibre or >5.5 cm, were immediately excluded. AAAs >5.5 cm were excluded due to the very low sample size, unless the AAA had increased from a diameter <5.5 cm to >5.5 cm in the time which elapsed between two surveillance scans.
Figure 1.
Flow diagram illustrating exclusions made from the preliminary dataset.
EVAR: endovascular aneurysm repair; fEVAR: fenestrated endovascular aneurysm repair; Blank: aneurysm/hematoma scans recorded without any aneurysmal measurement.
Formal sample size calculations were not utilised as the entire eligible population were included in the analysis. No ethical approvals or participant consent were required; however, this analysis was registered with the UHBW research and development department and approval was granted.
AAA diameter acquisition was based on standard operating procedures followed during each ultrasound scan. All AAAs were assessed in transverse and longitudinal planes for anteroposterior diameter using a commercially available high-resolution ultrasound machine (Logiq E9, GE Medical). Ultrasound callipers were placed on the anteroposterior intima-lumen interfaces at the largest section of the aneurysm producing an accurate inner-wall to inner-wall diameter measurement. To increase reliability and confidence of measurements, multiple images were acquired in each plane. All scans were performed by trained vascular scientists and adjudicated by the vascular department to ensure accuracy.
The 1312 AAA measurements recorded were separated into groups. The groups were selected as: 3.0–3.4 cm; 3.5–3.9 cm; 4.0–4.4 cm; 4.5–4.9 cm; 5.0–5.5 cm. 7 By breaking down AAAs into smaller groups, calculated GRs could be related to a more precise AAA diameter, rather than generalising one GR to all AAAs. Importantly, with regard to the group breakdown analysis, some AAAs fall into multiple groups. For example, a patient’s first baseline AAA scan measures 3.4 cm which would be placed into the 3.0–3.4 cm group. During the next scan, the AAA has increased in diameter to 3.6 cm, therefore establishing a GR of 0.2 cm/year for the 3.0–3.4 cm group. However, as the AAA has increased in size to the next group, the baseline diameter for that group will be reset to 3.6 cm, as it is the first AAA measurement acquired when in that group. This method allows very specific and relative GR calculations for aneurysms within the five group sizes, which is important when establishing the risks of the 6-monthly interval for 4.5–4.9 cm.
In order to calculate GR, each patient must have attended UHBW for a AAA scan at least twice, otherwise there would be no record of change in diameter over time. GR was derived by dividing the change in diameters between the first and final scans by the time elapsed between them.
Secondary analysis was conducted to assess influence of RFs and medication on AAA growth. The RFs considered were diabetes, hypertension, hypercholesterolaemia, smoking status, gender and age. Medications assessed were bisoprolol, ramipril, amlodipine, atorvastatin, simvastatin, gliclazide and metformin. Patient RFs and prescribed medications were determined on a local digital care record system. RFs and medications could then be associated with the respective AAA to determine any potential effect on GR.
Cause of death was assessed to illustrate how many of the 315 patients enrolled on surveillance between January 2015 and March 2020 suffered AAA-related mortality. AAA diameter at the time of rupture was also recorded to help establish safety of the 6-monthly surveillance interval. Cause of death of all deceased patients was determined from digital care records.
Normal distributions of data were assessed employing Shapiro–Wilk tests. Patient demographics were assessed across all five AAA size groups using chi-square tests, unless the sample was <5, in which case a Fisher’s exact test was adopted. Annual GR between groups were compared using a one-way analysis of variance (ANOVA) with the Bonferroni correction method. Backward stepwise multiple linear regression models were designed to establish any influence of RFs or medications on AAA GR; univariate linear regression models were designed to directly assess each RF and medication on GR. Post hoc Kruskal–Wallis tests were carried out to compare patients confirmed with and without the presence of RFs, and patients prescribed and not prescribed each medication. Data were analysed using the statistical package SPSS (Ver.28 for Windows, Chicago, SPSS Inc) and presented as means (95% confidence intervals (CIs)). Statistical significance was set at p < 0.05.
Results
Demographics
Patient demographics are listed in Table 2. Combined, the patients analysed were of a mean age 80 years (95% CI; (79, 81); range: 55–100) at the time of their final scan recorded for this analysis. Mean AAA diameter for all five groups was 4.2 cm (95% CI (4.1, 4.3); range 3–5.8). When comparing all patients irrespective of AAA diameter, there were no differences in the number of hypertensive (52%) and normotensive (48%) patients. There were a higher proportion of non-diabetics (74%), hypercholesterolaemia patients (72%) and patients with a smoking history (78%) compared to diabetics (26%), normal cholesterol patients (27%) and patients with no smoking history (22%). Only age of >90 years was significantly different between the five AAA groups (p = 0.01), and there was no difference in the remaining genetic and environmental RFs (p > 0.05).
Table 2.
Demographics of AAAs on surveillance between January 2015 and March 2020, where AAAs can fall into multiple group sizes as they progress in diameter over time.
| Risk factors | Sample size, n (%) | p value | |||||
|---|---|---|---|---|---|---|---|
| 3.0–3.4 cm | 3.5–3.9 cm | 4.0–4.4 cm | 4.5–4.9 cm | 5.0–5.5 cm | 3.0–5.5 cm | ||
| Gender | |||||||
| Male | 82 (23) | 56 (16) | 60 (17) | 79 (22) | 76 (22) | 353 | >0.05 |
| Female | 8 (10) | 16 (20) | 16 (20) | 21 (26) | 20 (25) | 81 | >0.05 |
| Age | |||||||
| <70 years | 11 (21) | 15 (28) | 10 (19) | 8 (15) | 9 (17) | 53 | >0.05 |
| 70–79 years | 30 (20) | 19 (13) | 26 (18) | 37 (25) | 36 (24) | 148 | >0.05 |
| 80–89 years | 46 (23) | 34 (17) | 37 (19) | 44 (22) | 36 (18) | 197 | >0.05 |
| >90 years | 4 (11)* | 4 (11)* | 3 (8)* | 11 (30)* | 15 (41)* | 37 | 0.01 |
| Past medical history | |||||||
| Diabetes | 21 (23) | 17 (19) | 13 (14) | 20 (22) | 20 (22) | 91 | >0.05 |
| Hypertension | 47 (22) | 35 (17) | 37 (18) | 40 (19) | 50 (24) | 209 | >0.05 |
| Hypercholesterolaemia | 62 (24) | 45 (18) | 50 (20) | 55 (22) | 43 (17) | 255 | >0.05 |
| Current smoker | 23 (24) | 12 (13) | 15 (16) | 23 (24) | 23 (24) | 96 | >0.05 |
| Ever smoked | 42 (20) | 39 (18) | 43 (20) | 44 (21) | 43 (20) | 211 | >0.05 |
*Significant difference within groups p < 0.05.
Growth rate
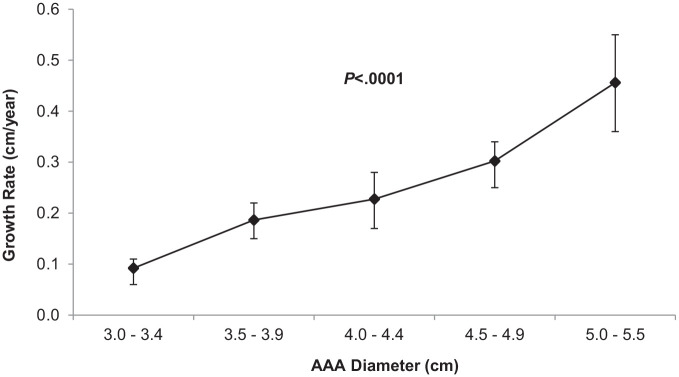
The mean GR for all patients was 0.25 cm/year. There was a significant increase in GR with an increase in AAA diameter (3.0–3.4 cm: 0.09 cm/year; 3.5–3.9 cm: 0.19 cm/year; 4.0–4.4 cm: 0.23 cm/year; 4.5–4.9 cm: 0.3 cm/year; 5.0–5.5 cm: 0.46 cm/year; p < 0.001) (Figure 2) when separated into 0.5 cm groups.
Figure 2.
Mean annual AAA growth rates (cm/year) with error bars indicating top and bottom end 95% confidence intervals.
Regression analysis
Univariate linear regression illustrated a significant effect of diabetes on AAA GR (p = 0.04); however, when performing backward stepwise multiple regression analysis in order to consider multiple RFs, this significance was not replicated (p > 0.05). No other RFs demonstrated an effect on GR. With regard to RF medication, gliclazide was the only medication to establish a significant effect on GR (p = 0.04), similarly when comparing multiple medications, the results became insignificant (p > 0.05).
Post hoc analysis
Post hoc Kruskal–Wallis tests supported the effects demonstrated by linear regression, with significantly higher AAA GR in non-diabetics (0.29 cm/year) and patients not prescribed gliclazide (0.26 cm/year) compared to diabetics (0.19 cm/year; p = 0.02) and patients prescribed gliclazide (0.1 cm/year; p = 0.004; Table 3), respectively. No differences were found for any remaining RFs or medications. One-way ANOVAs reinforced no effect of smoking and age on GR (p > 0.05).
Table 3.
Comparison of growth rate for patients with and without the presence of risk factors and risk factor medication.
| Risk factors | Growth rate (cm/year) | p Value a | p Value b |
|---|---|---|---|
| Age | |||
| <70 years | 0.30 [0.25, 0.34] | ||
| 70–79 years | 0.27 [0.24, 0.29] | ||
| 80–89 years | 0.24 [0.22, 0.25] | ||
| >90 years | 0.18 [0.14, 0.21] | >0.05 | N/A |
| Smoking status c | |||
| Current | 0.23 [0.18, 0.27] | ||
| Prior | 0.26 [0.21, 0.31] | ||
| Never | 0.28 [0.21, 0.35] | >0.05 | >0.05 |
| Gender | |||
| Male | 0.25 [0.21, 0.28] | ||
| Female | 0.29 [0.23, 0.34] | >0.05 | >0.05 |
| Hypercholesterolaemia | |||
| Yes | 0.24 [0.16, 0.29] | ||
| No | 0.22 [0.17, 0.26] | >0.05 | >0.05 |
| Diabetes | |||
| Yes | 0.19 [0.14, 0.23] | ||
| No | 0.29 [0.24, 0.32] | 0.02 | 0.04 |
| Hypertension | |||
| Yes | 0.25 [0.20, 0.29] | ||
| No | 0.25 [0.21, 0.28] | >0.05 | >0.05 |
| Medication | |||
| Hypercholesterolaemia | |||
| Atorvastatin | 0.24 [0.18, 0.29] | >0.05 | >0.05 |
| Simvastatin | 0.19 [0.13, 0.24] | >0.05 | >0.05 |
| Diabetes | |||
| Metformin | 0.20 [0.08, 0.33] | >0.05 | >0.05 |
| Gliclazide | 0.10 [0.04, 0.16] | 0.004 | 0.04 |
| Hypertension | |||
| Bisoprolol | 0.20 [0.14, 0.25] | >0.05 | >0.05 |
| Ramipril | 0.19 [0.13, 0.24] | >0.05 | >0.05 |
| Amlodipine | 0.24 [0.16, 0.31] | >0.05 | >0.05 |
CI: confidence interval; ANOVA: analysis of variance.
Data presented as means [95% CI].
Kruskal–Wallis or ANOVA performed.
Univariate linear regression performed.
ANOVA conducted.
Cause of death
There were a total of 93 (29.8%) deaths (Figure 3), of which 67 were male (mean, 95% CI; 83 years, 95% CI (81, 85)) and 26 were female (86 years, 95% CI (83, 89)). Of these, nine (10.3%) were associated with AAA which was the second most common known cause of death in these patients. AAA mortality encumbered seven AAA ruptures and two aortoduodenal fistula following elective endovascular aneurysm repair (EVAR). Six of the ruptures occurred in AAA >5.5 cm unsuitable for aneurysm repair, while one ruptured before reaching threshold, at 5.3 cm. There were no records showing post-operative in-hospital death following aneurysm repair.
Figure 3.
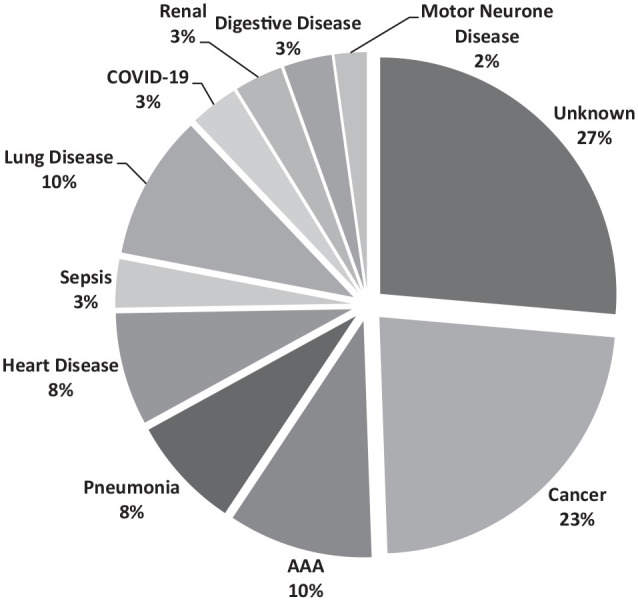
Cause of death among AAA surveillance patients.
Discussion
The aims of this analysis were to assess AAA GR and inform whether the current UHBW surveillance intervals were safe and appropriate. In addition, analysis of RFs/medication and their impact on GR may provide information as to how best to adapt surveillance intervals to patients.12,13 There were no differences in the demographics across each group, excluding patients aged >90 years, showing that the groups are well-matched and that findings are more comparable, reducing the risk of confounding variables.
The first key finding of this analysis is that AAA GR is positively associated with AAA diameter and is supported by an abundance of literature. Thompson et al. 5 compiled the AAA GRs of 18 studies with pooled estimates increasing by approximately 0.05 cm/year for each 0.5 cm increase in baseline diameter. Yet, there is substantial heterogeneity between GRs of these 18 studies, much of which was unexplained. Some of the studies are unavailable for direct comparison as Thompson et al. 5 received GR information by contacting the vascular laboratories directly. Nevertheless, AAA GRs in this analysis comply with multiple sources. Key information to note is that mean GR for 4.5–4.9 cm AAA were 0.3 cm/year [95% CI, 0.25, 0.34] within UHBW surveillance patients. Therefore, based on mean annual GR and variation, a 6-month surveillance interval would not increase the risk of patients reaching threshold between scans, compared to the nationally recommended 3 months. This provides validity upon the safety and appropriateness of altering surveillance intervals. Additional benefits to this safe 6-month interval is that there will be less frequent patient scans which can reduce demand on the department and improve cost-effectiveness of the surveillance programme.
Another point to consider is the number of AAA ruptures within UHBW surveillance patients. With only one AAA rupture <5.5 cm (ruptured at 5.3 cm) during the surveillance period analysed, UHBW rupture rates for 4.5–5.5 cm AAA are 0.31% over 5 years. National Abdominal Aortic Aneurysm Screening Programme (NAAASP) reported the same rupture rate (0.31%) in patients with the same size AAA between 2010 and 2017. 17 As NAAASP adopt a 3-monthly interval for this size AAA, there is no evidence of noticeable differences in rupture risks between the two surveillance intervals. Therefore, when considering AAA rupture rate alone, it is suitable to conclude that the deviation away from 3-monthly national recommendations is safe and appropriate. Moreover, when encompassing the GRs for AAA 4.5–4.9 cm, there is strong evidence to support this change in AAA surveillance intervals. However, there is controversy surrounding the optimum method for measuring AAA. NAAASP and National Institute of Health and Care Excellence (NICE) recommend internal aortic diameters, as conducted in the Multicentre Aneurysm Screening Study, 18 yet the UK surgical threshold of 5.5 cm is based on research assessing external aortic diameter.19,20 Internal and external diameters have shown to differ as much as 4 mm, 21 which could be a crucial difference between a ruptured aneurysm or intervention. It is important to note that UHBW rupture rates still remain valid when comparing to that of NAAASP, as both UHBW and NAAASP measure internal diameter. Nevertheless, based on this established variation between aortic diameters, the patient who suffered 5.3 cm AAA rupture and death may have undergone aneurysmal repair if external diameters were considered. Further insight into the differing rupture risks between measurement methods may help establish firm evidence-based surgical thresholds for internal aortic diameters.
An interesting side note with regard to AAA mortality encompasses two patients who died of aortoduodenal fistula formation following EVAR. This is a very rare complication of EVAR with an occurrence rate of 0.35%. 22 However, with 35 UHBW patients undergoing EVAR, aortoduodenal fistula occurred in 5.7% of this patient group which is a noticeably higher occurrence rate, 22 illustrating poorer outcomes for aortoduodenal fistula in AAA repair at UHBW. Detailed assessment of outcomes of AAA repair at UHBW could be a useful future direction for the study.
With regard to RFs, hypertension had no impact on GR, contradicting multiple sources.6,23 Hypertension has been shown to play a role in the formation of a AAA due to its relationship with shear stress and blood flow. Mechanical flow properties of a diseased aortic segment are disrupted with aneurysmal change, leading to an unequal distribution of shear forces during systole, also observed with turbulent flow patterns on colour and spectral Doppler ultrasound. 24 Shear stress on the aortic wall is directly related to the radius of the AAA and inversely to the thickness of the vessel wall, described by the Law of Laplace. 25 This reinforces that larger AAAs are at increased risk of rupture. 6 However, as no increase in GR was established, this UHBW sample may simply illustrate well-controlled hypertension, as some research points towards poorly controlled hypertension as the source of increased growth. Consequently, it is reasonable to believe that blood pressure control slows AAA progression and reduces the risk of rupture, potentially supported further by adherence to specific hypertensive medication. This analysis did demonstrate that bisoprolol and ramipril slightly reduced annual GR; however, this was insignificant. These results were very similar to that of Thompson et al. 26 who showed insignificant reductions in GR in patients taking these medications. It is worthwhile noting that most research assessing β-blockers and AAA centre around non-selective β-receptor propranolol rather than cardio-selective β-1 blocking bisoprolol. Unlike propranolol, a primary focus of bisoprolol is to inhibit renin secretion. 11 This prevents the eventual formation of angiotensin II which has been strongly linked to AAA expansion in animal models 27 and is a suspect for AAA progression in humans. Angiotensin II receptor blockers (ARBs) have been shown to reduce GR in human AAAs. 26 Therefore, it is possible to surmise that if inhibition of renin secretion occurs and no interaction with angiotensinogen from the liver takes place, this will prevent the cascade of reactions to angiotensin I and angiotensin II, consequently reducing AAA expansion. More recent studies which have included bisoprolol in their analysis have had too small a sample to make definitive conclusions on their impact; 26 repeating similar analyses such as this in a larger cohort may be of potential future benefit.
Angiotensin-converting enzyme inhibitors (ACEi) have had increasingly varied responses to AAA growth, demonstrating increases, decreases and no changes in GR. The results of this analysis support no effect of ACEi. Studies noting lower GRs have linked ACEi to reduced aortic inflammation processes. However, Kortekaas et al. 28 highlighted similar responses directly caused by the ACEi ramipril; yet they did not report any changes in GR after isolating this group of patients from patients with other RFs. It is possible that patients within this UHBW cohort prescribed ramipril are exhibiting similar aortic inflammation responses, but with no significant change to the aneurysmal growth.
An inverse relationship between diabetes and AAA development was initially established in the 2000s and supported by systematic reviews and meta-analyses. 29 This analysis has also shown the same relationship. Low prevalence of diabetes and AAA is also reflected within this sample, with only 22% diabetic, compared to the other cardiovascular RFs, hypertension (52%) and hypercholesterolemia (56%). The suspected reason is that diabetes increases aortic wall stiffness and decreases aortic wall degradation through an excess of vascular matrix collagen and decreased extracellular matrix proteolysis. 7 Whether these mechanisms may be a consequence of hyperglycaemia alone or hyperglycaemia and a combination of specific diabetic drug-use is difficult to determine. Yet, this analysis did highlight a significant association between gliclazide and further reduced GR, supported in other reports. 30 It is important to note, however, that the sample of gliclazide patients was only 18, thus hindering its statistical power. Contrary to many published reports, metformin showed no impact on GR. Metformin has repeatedly demonstrated a relationship with reduced AAA GR.8,30,31 A potential reason for this lack of effect of metformin is the low sample of diabetics within this analysis. Another reason may be that, as most studies report their diabetic cohorts to be taking metformin, reports often lack in-depth comparisons to include unmedicated diabetic patients. It could be that diabetes alone is the stronger protective mechanism against AAA rather than the actions of its therapeutic agents. Studies assessing diabetes and AAA which included all diabetic patients within their analyses, rather than focussing solely on medicated diabetics, still reported significant effects of diabetes.7,32 Univariate linear regression within this analysis highlighted an association between diabetes and GR, yet many of these diabetic patients are currently controlling their diabetes through improved diet, which suggests that diabetes is the more defensive factor.
There were notable limitations of this analysis. Data used for this study were extracted from the UHBW vascular department database, input by department staff at the time of the scan. Aortic diameters were not recorded in the department database for a total of 214 AAA ultrasound sans, which led to loss of data (Figure 1). In addition, as RF data were sourced retrospectively from digital care records, it was not possible to accurately account for when patients’ RFs were diagnosed. This also applied to RF medications prescribed to each patient. There is also no way to determine drug compliance rates. Therefore, patients with an RF may have been diagnosed very late into their surveillance and only recently prescribed medication, potentially skewing their impact on AAA GR. There were numerous patients who were also taking more than one of the RF medications analysed, making it difficult to pinpoint their influence and confounding factors. Finally, sample sizes were quite small for certain RFs and medications. Future studies should aim to include a larger sample with detailed information on time diagnosed with RFs and time spent complying with prescribed medication.
With a significant impact of diabetes and gliclazide on GR, it supports previous suggestions that RFs and medications should be considered when setting patient surveillance intervals at UHBW. Diabetic patients at UHBW illustrate protection against AAA growth and therefore may permit less frequent surveillance intervals. Finally, a mean GR and variation of growth for 4.5–4.9 cm AAA were 0.3 cm/year [95% CI, 0.25, 0.34], respectively, which is not enough to surpass threshold while waiting for the next surveillance scan. This is supported by low numbers of AAA ruptures <5.5 cm. Therefore, it is suitable to conclude that the current UHBW deviation away from national guidance for AAA surveillance intervals is safe and appropriate.
Acknowledgments
I would like to thank Thomas Berni for advising statistical analysis and interpretation. I would also thank Liza Hornby-Foster for her help with manuscript structuring.
Footnotes
Contributors: IH-F facilitated conceptualization, study design and analysis and interpretation of data. IH-F also drafted the manuscript and gave final approval for publication.
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding: The author received no financial support for the research, authorship and/or publication of this article.
Ethical approval: Not required. Data used for this study were gathered retrospectively from clinically required examinations and had been held since. All identifiable patient information was removed for the purpose of this study, therefore no ethical approval was required, as per The Declaration of Helsinki. Ethical approval was waived by University Hospitals Bristol and Weston Diagnostics and Therapies Audit Facilitator.
Guarantor: IH-F
ORCID iD: Ian Hornby-Foster  https://orcid.org/0000-0001-5500-3215
https://orcid.org/0000-0001-5500-3215
References
- 1. Sakalihasan N, Limet R, Defawe OD. Abdominal aortic aneurysm. Lancet 2005; 365: 1577–1589. [DOI] [PubMed] [Google Scholar]
- 2. Akkersdijk GJ, de Vries AC, Puylaert JB. Abdominal aortic aneurysm as an incidental finding in abdominal ultrasonography. Brit J Surg 1991; 78: 1261–1263. [DOI] [PubMed] [Google Scholar]
- 3. Anjum A, von Allmen R, Greenhalgh R. Explaining the decrease in mortality from abdominal aortic aneurysm rupture. J Vasc Surg 2012; 56: 876. [DOI] [PubMed] [Google Scholar]
- 4. Al-Balah A, Goodall R, Salciccioli J, et al. Trends in abdominal aortic aneurysm mortality in European Union 15+ countries from 1990-2017. J Brit Surg 2020; 107: 1459–1467. [DOI] [PubMed] [Google Scholar]
- 5. Thompson S, Brown L, Sweeting M, et al. Systematic review and meta-analysis of the growth and rupture rates of small abdominal aortic aneurysms: implications for surveillance-intervals and their cost-effectiveness. Health Tech Asses 2013; 17: 1–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Anagnostakos J, Lal BK. Abdominal aortic aneurysms. Prog Cardiovasc Dis 2021; 65: 34–43. [DOI] [PubMed] [Google Scholar]
- 7. De Rango P, Farchioni L, Fiorucci B, et al. Diabetes and abdominal aortic aneurysms. Eur J Vasc Endovasc Surg 2014; 47: 243–261. [DOI] [PubMed] [Google Scholar]
- 8. Golledge J, Moxon J, Pinchbeck J, et al. Association between metformin prescription and growth rates of abdominal aortic aneurysms. J Brit Surg 2017; 104: 1486–1493. [DOI] [PubMed] [Google Scholar]
- 9. Salata K, Syed M, Hussain MA, et al. Statins reduce abdominal aortic aneurysm growth, rupture, and perioperative mortality: a systematic review and meta-analysis. J Am Heart Assoc 2018; 7: e008657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Guessous I, Periard D, Lorenzetti D, et al. The efficacy of pharmacotherapy for decreasing the expansion rate of abdominal aortic aneurysms: a systematic review and meta-analysis. PLoS ONE 2008; 3: e1895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bennett MR, Chang CL, Tuffery C, et al. The impact of regular bisoprolol on the response to salbutamol in asthma: a double-blind randomized placebo-controlled crossover trial. Respirology 2021; 26: 225–232. [DOI] [PubMed] [Google Scholar]
- 12. Badger SA, Jones C, McClements J, et al. Surveillance strategies according to the rate of growth of small abdominal aortic aneurysms. Vasc Med 2011; 16: 415–421. [DOI] [PubMed] [Google Scholar]
- 13. McCarthy R, Shaw E, Whyman M, et al. Recommendations for screening intervals for small aortic aneurysms. J Vasc Surg 2003; 51: 1081–1087. [DOI] [PubMed] [Google Scholar]
- 14. Powell JT, Thompson SG. Should the frequency of surveillance for small abdominal aortic aneurysms be reduced? Eur J Vasc Endovasc Surg 2013; 46: 171–172. [DOI] [PubMed] [Google Scholar]
- 15. Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 practice guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic) a collaborative report from the American Association for Vascular Surgery/society for vascular surgery,* society for cardiovascular angiography and interventions, society for vascular medicine and biology, society of interventional radiology, and the ACC/AHA task force on practice guidelines. Circulation 2006; 113: 463–654. [DOI] [PubMed] [Google Scholar]
- 16. The RESCAN Collaborators RE. Surveillance-intervals for small abdominal aortic aneurysms. JAMA 2013; 309: 806–813. [DOI] [PubMed] [Google Scholar]
- 17. Oliver-Williams C, Sweeting MJ, Jacomelli J, et al. Safety of men with small and medium abdominal aortic aneurysms under surveillance in the NAAASP. Circulation 2019; 139: 1371–1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ashton HA, Buxton MJ, Day NE, et al. The Multicentre Aneurysm Screening Study (MASS) into the effect of abdominal aortic aneurysm screening on mortality in men: a randomised controlled trial. Lancet 2002; 360: 1531–1539. [DOI] [PubMed] [Google Scholar]
- 19. Powell JT, Brown LC, Forbes JF. Final 12-year follow-up of surgery versus surveillance in the UK small aneurysm trial. J Brit Surg 2007; 94: 702–708. [DOI] [PubMed] [Google Scholar]
- 20. Lederle FA, Wilson SE, Johnson GR, et al. Design of the abdominal aortic aneurysm detection and management study. J Vasc Surg 1994; 20: 296–303. [DOI] [PubMed] [Google Scholar]
- 21. Meecham L, Evans R, Buxton P, et al. Abdominal aortic aneurysm diameters: a study on the discrepancy between inner to inner and outer to outer measurements. Eur J Vasc Endovasc Surg 2015; 49: 28–32. [DOI] [PubMed] [Google Scholar]
- 22. Hosaka A, Nemoto M, Motoki M, et al. Aortoduodenal fistula after endovascular abdominal aortic aneurysm repair. Vasc Endovasc Surg 2020; 54: 445–448. [DOI] [PubMed] [Google Scholar]
- 23. Takagi H, Umemoto T and ALICE (All-Literature Investigation of Cardiovascular Evidence) Group. Association of hypertension with abdominal aortic aneurysm expansion. Ann Vasc Surg 2017; 39: 74–89. [DOI] [PubMed] [Google Scholar]
- 24. Khanafer KM, Bull JL, Upchurch GR, Jr, et al. Turbulence significantly increases pressure and fluid shear stress in an aortic aneurysm model under resting and exercise flow conditions. Ann Vasc Surg 2007; 21: 67–74. [DOI] [PubMed] [Google Scholar]
- 25. Rodel SGJ, Meerwaldt R, Geelkerken RH, et al. Risk factors for AAA growth and rupture; more than diameter alone. Latest Insight Abdom Aortic Aneurys Endovasc Repair 2012; 2012: 11–23. [Google Scholar]
- 26. Thompson A, Cooper JA, Fabricius M, et al. An analysis of drug modulation of abdominal aortic aneurysm growth through 25 years of surveillance. J Vasc Surg 2010; 52: 55–61. [DOI] [PubMed] [Google Scholar]
- 27. Kim CW, Kumar S, Son DJ, et al. Prevention of abdominal aortic aneurysm by Anti–MicroRNA-712 or Anti–MicroRNA-205 in angiotensin II–infused mice. Arterioscler Thromb Vasc Biol 2014; 34: 1412–1421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Kortekaas KE, Meijer CA, Hinnen JW, et al. ACE inhibitors potently reduce vascular inflammation, results of an open proof-of-concept study in the abdominal aortic aneurysm. PLoS ONE 2014; 9: e111952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Glover MJ, Kim LG, Sweeting MJ, et al. Cost-effectiveness of the National Health Service abdominal aortic aneurysm screening programme in England. Brit J Surg 2014; 101: 976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Itoga NK, Rothenberg KA, Suarez P, et al. Metformin prescription status and abdominal aortic aneurysm disease progression in the US veteran population. J Vasc Surg 2019; 69: 710–716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Thanigaimani S, Singh TP, Unosson J, et al. Association between metformin prescription and abdominal aortic aneurysm growth and clinical events: a systematic review and meta-analysis. Eur J Vasc Endovasc Surg 2021; 62: 747–756. [DOI] [PubMed] [Google Scholar]
- 32. Shantikumar S, Ajjan R, Porter KE, et al. Diabetes and the abdominal aortic aneurysm. Eur J Vasc Endovasc Surg 2010; 39: 200–207. [DOI] [PubMed] [Google Scholar]