This case report describes the successful treatment of a severe carbamazepine overdose using a combination of therapies and presents data suggesting mitochondrial dysfunction in carbamazepine intoxication.
Abstract
A 15-year-old female subject presented comatose, in respiratory failure and shock, after the intentional ingestion of ∼280 extended-release 200-mg carbamazepine tablets with a peak serum concentration of 138 µg/mL (583.74 µmol/L). The patient developed clinical seizures and an EEG pattern of stimulus-induced rhythmic, periodic, or ictal discharges, suggestive of significant cortical dysfunction. Due to the extremely high drug serum concentration and clinical instability, a combination of therapies was used, including lipid emulsion therapy, plasmapheresis, hemodialysis, continuous venovenous hemodiafiltration, and endoscopic intestinal decontamination. The patient’s elevated serum lactate level with a high mixed venous saturation suggested possible mitochondrial dysfunction, prompting treatment with barbiturate coma to reduce cerebral metabolic demand. The serum carbamazepine concentration declined steadily, with resolution of lactic acidosis, no long-term end-organ damage, and return to baseline neurologic function. The patient was eventually discharged in her usual state of health. In the laboratory, we demonstrated in vitro that the active metabolite of carbamazepine hyperpolarized the mitochondrial membrane potential, supporting the hypothesis that the drug caused mitochondrial dysfunction. We thus successfully treated a life-threatening carbamazepine overdose with a combination of modalities. Future studies are required to validate this aggressive approach. The occurrence of mitochondrial dysfunction must be confirmed in patients with carbamazepine toxicity and the need to treat it validated.
Carbamazepine is a lipophilic anticonvulsant with peak absorption at ∼12 to 24 hours after ingestion.1 It is a sodium channel blocker with anticholinergic properties.2 Carbamazepine poisoning can present with altered mental status leading to coma, respiratory failure, cardiac arrhythmias, and seizures.3 Serum concentrations >39 µg/mL (165 µmol/L) are associated with fatalities.3–5 We report a life-threatening intentional overdose of carbamazepine treated with a combination of therapies to enhance drug clearance. Barbiturate coma was induced for cerebral protection despite the patient being comatose; preliminary data are provided to support the hypothesis of functional tissue hypoxia in carbamazepine overdose.
Case Presentation
A confused 15-year-old female was found by her parents with an empty carbamazepine bottle (≤285 tablets, 200-mg extended-release formulation). She was brought to the emergency department, where she was unresponsive (Glasgow Coma Scale score, 5). She was intubated for airway protection, and charcoal was delivered via an orogastric tube. The initial carbamazepine serum concentration exceeded the assay upper limit (>20 µg/mL). The patient was transferred to the PICU of a tertiary referral hospital 9 hours after the ingestion.
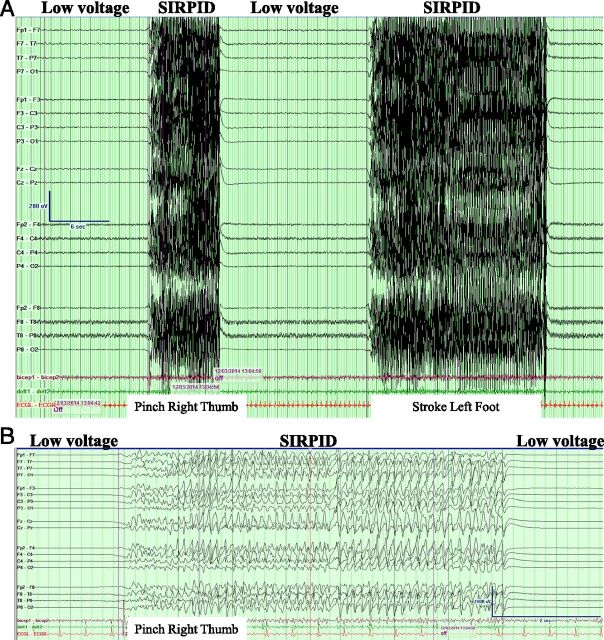
On arrival at the PICU, the patient was hypotensive, requiring volume resuscitation and a norepinephrine infusion. Her electrocardiogram showed a normal QRS interval (90 milliseconds) and a prolonged QTc (504 milliseconds). Initially, the patient demonstrated motor response to painful stimuli and had dilated but responsive pupils; she then quickly developed decerebrate posturing with no brainstem reflexes and pupils that were fixed and 9 mm dilated bilaterally. Her initial head computed tomography scan was normal. She developed generalized tonic-clonic seizures treated with lorazepam, propofol boluses, and a midazolam infusion. Continuous EEG monitoring showed low voltages with bursts of stimulus-induced rhythmic, periodic, or ictal discharges (SIRPIDs) (Fig 1).
FIGURE 1.
EEG of the study patient demonstrating low voltages with bursts of SIRPIDs during stimulation (pinch right thumb and stroke left foot). A, Compressed EEG. B, Uncompressed EEG.
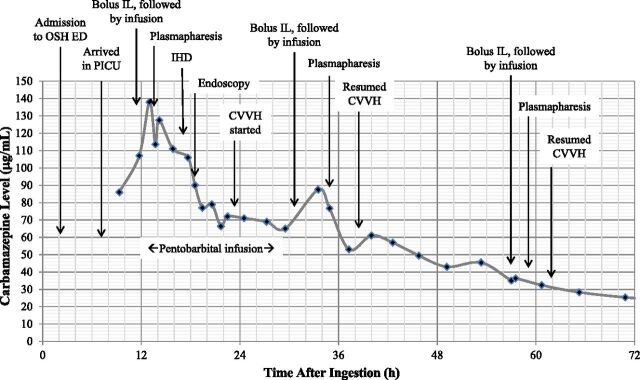
The initial venous blood gas analysis showed the following: pH, 7.33; Pco2, 42.7 mm Hg; bicarbonate, 22 mmol/L; and oxygen saturation, 98% (from a femoral catheter tip at L2). Her lactate level was 7.3 mmol/L. The initial carbamazepine serum concentration was 86 µg/mL (364 µmol/L). Extended urine and blood drug screens were otherwise positive only for midazolam and lorazepam, which had been administered to the patient. The time course of the patient’s carbamazepine serum concentration and interventions are shown in Fig 2.
FIGURE 2.
Total carbamazepine serum concentration over time. OSH, outside hospital; ED, emergency department; IL, lipid emulsion therapy; IHD, intermittent hemodialysis; CVVHD, continuous venovenous hemodialysis.
The patient deteriorated clinically with worsening lactic acidemia (lactate level, 10.6 mmol/L) and a peak carbamazepine concentration of 138 µg/mL (584 µmol/L) ∼13 hours after the suspected ingestion. Because of her worsening condition and rapidly increasing carbamazepine serum concentration, the patient was given lipid emulsion therapy (1.5 mL/kg bolus of a 20% fat emulsion, then infusion of 0.25 mL/kg/min for 60 minutes), followed by plasmapheresis of 1.5 plasma volume with 5% albumin replacement, and intermittent high-flux hemodialysis followed by continuous venovenous hemodiafiltration (CVVHDF). The pheresate was clear except after lipid treatment, when there was a separate lipemic phase. The patient had decreased bowel sounds and an ileus, preventing intestinal decontamination with motility agents or further use of activated charcoal. To prevent ongoing carbamazepine absorption from a potentially large number of ingested extended-release capsules, an endoscopic gastroduodenoscopy was performed, and 22 capsules were removed; more were visualized in the distal duodenum. She eventually passed >150 capsules in her stool.
The patient’s rising serum lactate level, together with a high venous oxygen saturation, increased the possibility that her tissues were not appropriately utilizing oxygen. To reduce cerebral metabolic demand, a pentobarbital infusion was started, which led to a low-voltage EEG and suppression of the SIRPIDs. Pentobarbital was discontinued when the lactate level was 3.7 mmol/L and venous oxygen saturation was 83%.
The patient’s clinical status steadily improved after these interventions. Brainstem reflexes returned on hospital day 4, when the serum carbamazepine concentration decreased to 25 µg/mL. At a carbamazepine concentration of 16 µg/mL on day 6, the patient began having purposeful movements and followed simple commands. Results of a brain and spine MRI on day 7 were normal. She was extubated that day but exhibited self-injurious behaviors necessitating reintubation. She was successfully extubated on day 10 in her baseline condition. On day 14, the patient was discharged to a psychiatric facility, where she reportedly did well.
Discussion
The patient presented with many characteristic symptoms of carbamazepine overdose. Fixed, dilated pupils, tachycardia, and decreased gastric motility with ileus were likely the result of carbamazepine’s anticholinergic effects.2 Other reported cardiovascular manifestations include hypotension, myocardial depression,6 and QRS and QTc prolongation.7 Carbamazepine frequently causes neurologic dysfunction such as dystonia, apnea, and coma.8 Seizures are likely caused by a combination of carbamazepine’s anticholinergic effect and antagonism of adenosine A1 receptors.9
The normal therapeutic serum concentration of carbamazepine is 4 to 12 µg/mL, with concentrations >39 µg/mL associated with severe toxicity.3 Our patient’s extremely high serum carbamazepine concentration and clinical instability prompted us to use a combination of treatment modalities to accelerate drug removal.
Lipid emulsion therapy was developed for local anesthetic toxicity but has been used in overdoses with other lipophilic compounds.10 We used it in the present case because carbamazepine is lipophilic and structurally similar to tricyclic antidepressants and local anesthetics.
We reasoned that plasmapheresis would remove lipid-bound carbamazepine and the 70% to 95% of drug that is protein bound.11 Plasmapheresis is an extracorporeal procedure that allows removal and replacement of selected blood components, including highly protein bound substances. Although the American Society for Apheresis does not offer recommendations regarding plasmapheresis in carbamazepine overdose,12 the use and variable effectiveness of plasmapheresis have been described in case reports.13–15 Significant rebound in carbamazepine serum concentrations have been reported after plasmapheresis.13 This scenario was not observed in our patient, possibly because we used CVVHDF after plasmapheresis. Lipid emulsion therapy can increase blood clot formation and fat deposition in extracorporeal circuits,16,17 which was not observed in this case.
Plasmapheresis was followed by 1 cycle of hemodialysis, then by CVVHDF, to remove free carbamazepine from the serum. Hemodialysis, hemofiltration, and continuous renal replacement therapy have been used in carbamazepine intoxication.18–21 The EXTRIP (Extracorporeal Treatments in Poisoning) Workgroup recommends extracorporeal interventions for severe intoxication because these interventions can achieve rapid and substantial removal of carbamazepine.22 Among treatment options, intermittent hemodialysis is preferred because it has efficacy similar to hemoperfusion, greater availability, and lower cost. In centers where hemodialysis is not available, hemoperfusion or continuous renal replacement therapies may be used. In our patient, intermittent hemodialysis was followed by CVVHDF for continuous drug elimination in the setting of suspected ongoing absorption from extended-release tablets.
In the present case, the potentially large dose ingested, delayed gastric motility, and known enterohepatic circulation of carbamazepine raised concern for ongoing drug absorption. Because repeated administration of activated charcoal and gastric decontamination with laxatives were prevented by the ileus, we attempted endoscopic removal of capsules, with elimination of 22 capsules (a quantity that, although a fraction of the total ingested, could have caused significant toxicity). Removal of more capsules was not feasible.
This combination of interventions was effective, as the peak carbamazepine serum concentration occurred ∼13 hours after suspected ingestion. This peak is much earlier than would otherwise have been expected; peak serum concentrations have been reported up to 96 hours after overdose of extended-release formulations.23 Elimination of carbamazepine would also have been delayed without intervention; in the setting of elevated serum concentrations, as seen in overdose, zero-order kinetics are observed (a fixed amount of drug is eliminated per unit of time).24 To definitively demonstrate which modality (plasmapheresis or continuous venovenous hemodialysis) was most effective in eliminating carbamazepine from the patient would require measurement of carbamazepine concentration in the dialysate and pheresate. Our institution did not previously have laboratory protocols for this process; we have subsequently developed procedures for any future cases.
The patient was comatose on presentation and developed generalized tonic-clonic seizures. SIRPIDs, identified by correlating patient stimulation with high-voltage discharges on video-EEG, are common in critically ill adults25 and reported in pediatric patients.26,27 Decreasing the serum carbamazepine concentration and treatment with pentobarbital may have contributed to the resolution of the SIRPIDs. As the patient recovered clinically, her EEG background normalized.
Our patient initially had a rising serum lactate level with high venous oxygen saturation. This combination suggested mitochondrial dysfunction (low oxygen extraction/consumption leading to elevated venous saturation) producing functional tissue hypoxia (anaerobic metabolism causing elevated lactate levels). To protect the brain from this possibility, we reduced cerebral metabolic demand with a pentobarbital coma. We were mindful that barbiturates induce cytochrome P450 3A4 enzymes, which facilitate carbamazepine clearance31,32 and have been used to accelerate metabolism of other drugs in overdose.33
Antiepileptic medications, including carbamazepine, may cause mitochondrial toxicity34,35 and oxidative stress36 after long-term use. The effects of acute carbamazepine toxicity on mitochondrial function are unknown. We tested in vitro our hypothesis that high serum carbamazepine levels may cause mitochondrial dysfunction. Mitochondrial membrane potential (ΔΨm) is a key indicator of mitochondrial function, including synthesis of adenosine triphosphate. Using the fluorescent ΔΨm indicator tetramethylrhodamine, ethyl ester29 in cultured live rat cortical neurons, we found that the main active metabolite of carbamazepine, carbamazepine 10,11-epoxide,30 hyperpolarized the ΔΨm at a concentration similar to that found in our patient (Fig 3). Mitochondrial hyperpolarization has been reported with prolonged complex I inhibition37 and exposure to neurotoxic substances,38,39 and it may be a response to toxic stimuli that precede mitochondrial membrane permeabilization and apoptosis.40 Our data suggest that decreasing metabolic demand in carbamazepine toxicity may be beneficial.
FIGURE 3.
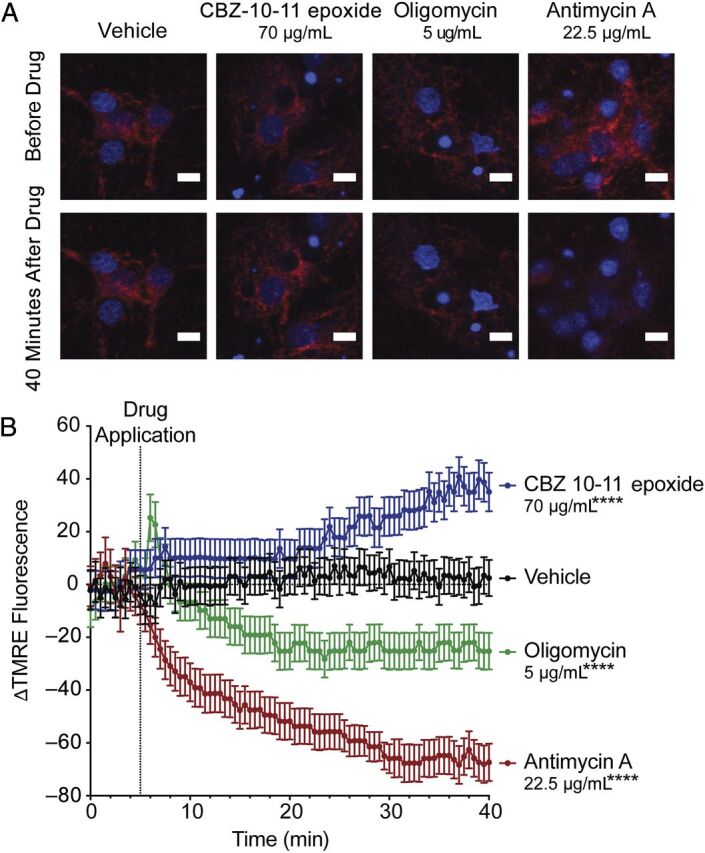
High concentrations of carbamazepine 10,11-epoxide (CBZ-10-11 epoxide) lead to acute mitochondrial hyperpolarization in cultured neurons in vitro. On day in vitro 18 to 21, cultured rat cortical neurons28 were loaded with the potentiometric cell–permeable fluorescent probe TMRE (tetramethylrhodamine, ethyl ester); its fluorescence on live cell confocal microscopy is known to correlate with mitochondrial membrane potential (ΔΨm).29 A, After 5 minutes to acquire a set of baseline images (top panels), drugs were applied at the indicated concentrations (lower panels). CBZ-10-11 epoxide was used because it is the main active metabolite of carbamazepine.30 B, TMRE fluorescence intensity was measured and is expressed as ΔF (percent change in fluorescence from baseline). This change in fluorescence intensity reflects the changes in ΔΨm.29 CBZ-10-11 epoxide significantly increased the TMRE signal, indicating hyperpolarized ΔΨm. Standard assay calibration was performed by using oligomycin (an adenosine triphosphate synthase inhibitor), which led to a transient hyperpolarization followed by depolarization, and antimycin A (a mitochondrial electron transport inhibitor), which strongly depolarized the membrane. ****P < .0001 (analysis of variance with Bonferroni's multiple comparison test; compared with vehicle). Data are presented as mean ± SD.
Conclusions
We present a case of carbamazepine overdose in which aggressive multimodal therapy resulted in an excellent outcome despite an extremely high peak serum concentration. Further study is required to separate the contributions of the individual interventions and to determine whether this approach is best practice in a massive overdose.
The use of barbiturate coma in a functionally anesthetized patient was driven by our hypothesis that cerebral oxygen utilization was impaired as a result of carbamazepine toxicity. Our preliminary laboratory data support this hypothesis; further laboratory and clinical research is necessary to verify that impaired oxygen utilization occurs and merits treatment.
Glossary
- CVVHDF
continuous venovenous hemodiafiltration
- SIRPID
stimulus-induced rhythmic, periodic, or ictal discharge
Footnotes
Dr Agulnik reviewed the clinical data for this case as well as the available literature, drafted the initial manuscript, and critically reviewed all versions of the manuscript; Dr Kelly reviewed the pheresis and hemodialysis clinical data for this case as well as the available literature, and critically reviewed the manuscript; Dr Bruccoleri reviewed the toxicology and lipid emulsion therapy clinical data for this case as well as the available literature, and critically reviewed the manuscript; Dr Yuskaitis reviewed the neurologic clinical data for this case as well as the available literature, and critically reviewed the manuscript; Dr Ebrahimi-Fakhari designed and conducted the in vitro study presented in the manuscript and critically reviewed the manuscript; Dr Sahin oversaw the in vitro study presented in the manuscript and critically reviewed the manuscript; Dr Bruccoleri reviewed the toxicology and lipid emulsion therapy clinical data for this case as well as the available literature, and critically reviewed the manuscript; and Dr Kohane reviewed the clinical data for this case and critically reviewed all versions of the manuscript; and all authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
FUNDING: No external funding.
References
- 1.Nelson L, Lewin N, Howland M, Hoffman R, Goldfrank L, Flomenbaum N. Goldfrank’s Toxicologic Emergencies. 9th ed. New York, NY: McGraw-Hill; 2011 [Google Scholar]
- 2.Spiller HA. Management of carbamazepine overdose. Pediatr Emerg Care. 2001;17(6):452–456 [DOI] [PubMed] [Google Scholar]
- 3.Hojer J, Malmlund HO, Berg A. Clinical features in 28 consecutive cases of laboratory confirmed massive poisoning with carbamazepine alone. J Toxicol Clin Toxicol. 1993;31(3):449–458 [DOI] [PubMed] [Google Scholar]
- 4.Fisher RS, Cysyk B. A fatal overdose of carbamazepine: case report and review of literature. J Toxicol Clin Toxicol. 1988;26(7):477–486 [DOI] [PubMed] [Google Scholar]
- 5.Tibballs J. Acute toxic reaction to carbamazepine: clinical effects and serum concentrations. J Pediatr. 1992;121(2):295–299 [DOI] [PubMed] [Google Scholar]
- 6.Gary NE, Byra WM, Eisinger RP. Carbamazepine poisoning: treatment by hemoperfusion. Nephron. 1981;27(4–5):202–203 [DOI] [PubMed] [Google Scholar]
- 7.Apfelbaum JD, Caravati EM, Kerns WP II, Bossart PJ, Larsen G. Cardiovascular effects of carbamazepine toxicity. Ann Emerg Med. 1995;25(5):631–635 [DOI] [PubMed] [Google Scholar]
- 8.Stremski ES, Brady WB, Prasad K, Hennes HA. Pediatric carbamazepine intoxication. Ann Emerg Med. 1995;25(5):624–630 [DOI] [PubMed] [Google Scholar]
- 9.Van Calker D, Steber R, Klotz KN, Greil W. Carbamazepine distinguishes between adenosine receptors that mediate different second messenger responses. Eur J Pharmacol. 1991;206(4):285–290 [DOI] [PubMed] [Google Scholar]
- 10.Jamaty C, Bailey B, Larocque A, Notebaert E, Sanogo K, Chauny JM. Lipid emulsions in the treatment of acute poisoning: a systematic review of human and animal studies. Clin Toxicol (Phila). 2010;48(1):1–27 [DOI] [PubMed] [Google Scholar]
- 11.Vree TB, Janssen TJ, Hekster YA, Termond EF, van de Dries AC, Wijnands WJ. Clinical pharmacokinetics of carbamazepine and its epoxy and hydroxy metabolites in humans after an overdose. Ther Drug Monit. 1986;8(3):297–304 [DOI] [PubMed] [Google Scholar]
- 12.Schwartz J, Winters JL, Padmanabhan A, et al. Guidelines on the use of therapeutic apheresis in clinical practice-evidence-based approach from the Writing Committee of the American Society for Apheresis: the sixth special issue. J Clin Apher. 2013;28(3):145–284 [DOI] [PubMed] [Google Scholar]
- 13.Kale PB, Thomson PA, Provenzano R, Higgins MJ. Evaluation of plasmapheresis in the treatment of an acute overdose of carbamazepine. Ann Pharmacother. 1993;27(7–8):866–870 [DOI] [PubMed] [Google Scholar]
- 14.Kozanoglu I, Kahveci S, Asma S, et al. Plasma-exchange treatment for severe carbamazepine intoxication: a case study. J Clin Apher. 2014;29(3):178–180 [DOI] [PubMed] [Google Scholar]
- 15.Duzova A, Baskin E, Usta Y, Ozen S. Carbamazepine poisoning: treatment with plasma exchange. Hum Exp Toxicol. 2001;20(4):175–177 [DOI] [PubMed] [Google Scholar]
- 16.Rodríguez B, Wilhelm A, Kokko KE. Lipid emulsion use precluding renal replacement therapy. J Emerg Med. 2014;47(6):635–637 [DOI] [PubMed] [Google Scholar]
- 17.Lee HM, Archer JR, Dargan PI, Wood DM. What are the adverse effects associated with the combined use of intravenous lipid emulsion and extracorporeal membrane oxygenation in the poisoned patient? Clin Toxicol (Phila). 2015;53(3):145–150 [DOI] [PubMed] [Google Scholar]
- 18.Kielstein JT, Schwarz A, Arnavaz A, Sehlberg O, Emrich HM, Fliser D. High-flux hemodialysis—an effective alternative to hemoperfusion in the treatment of carbamazepine intoxication. Clin Nephrol. 2002;57(6):484–486 [DOI] [PubMed] [Google Scholar]
- 19.Askenazi DJ, Goldstein SL, Chang IF, Elenberg E, Feig DI. Management of a severe carbamazepine overdose using albumin-enhanced continuous venovenous hemodialysis. Pediatrics. 2004;113(2):406–409 [DOI] [PubMed] [Google Scholar]
- 20.Azak A, Koçak G, Huddam B, Duranay M. Is conventional hemodialysis enough to manage carbamazepine intoxication? Blood Purif. 2012;33(4):225–226 [DOI] [PubMed] [Google Scholar]
- 21.Goktas U, Kati I, Yuce HH. Management of a severe carbamazepine overdose with continuous venovenous hemodiafiltration. Am J Emerg Med. 2010;28(2):260.e1–260.e2 [DOI] [PubMed] [Google Scholar]
- 22.Ghannoum M, Yates C, Galvao TF, et al. ; EXTRIP Workgroup . Extracorporeal treatment for carbamazepine poisoning: systematic review and recommendations from the EXTRIP workgroup. Clin Toxicol (Phila). 2014;52(10):993–1004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Graudins A, Peden G, Dowsett RP. Massive overdose with controlled-release carbamazepine resulting in delayed peak serum concentrations and life-threatening toxicity. Emerg Med (Fremantle). 2002;14(1):89–94 [DOI] [PubMed] [Google Scholar]
- 24.Winnicka RI, Topaciński B, Szymczak WM, Szymańska B. Carbamazepine poisoning: elimination kinetics and quantitative relationship with carbamazepine 10,11-epoxide. J Toxicol Clin Toxicol. 2002;40(6):759–765 [DOI] [PubMed] [Google Scholar]
- 25.Hirsch LJ, Claassen J, Mayer SA, Emerson RG. Stimulus-induced rhythmic, periodic, or ictal discharges (SIRPIDs): a common EEG phenomenon in the critically ill. Epilepsia. 2004;45(2):109–123 [DOI] [PubMed] [Google Scholar]
- 26.Skjei KL, Kessler SK, Abend NS. Stimulus-induced rhythmic, periodic, or ictal discharges in a 13-year-old girl after an overdose and respiratory arrest. Pediatr Neurol. 2011;45(5):350–351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Takenouchi T, Yap VL, Engel M, Perlman JM. Stimulus-induced seizure in sick neonates--novel observations with potential clinical implications. Epilepsia. 2010;51(2):308–311 [DOI] [PubMed] [Google Scholar]
- 28.Nie D, Sahin M. A genetic model to dissect the role of Tsc-mTORC1 in neuronal cultures. Methods Mol Biol. 2012;821:393–405 [DOI] [PubMed] [Google Scholar]
- 29.Joshi DC, Bakowska JC. Determination of mitochondrial membrane potential and reactive oxygen species in live rat cortical neurons. J Vis Exp. 2011;(51).pii: 2704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bertilsson L, Tomson T. Clinical pharmacokinetics and pharmacological effects of carbamazepine and carbamazepine-10,11-epoxide. An update. Clin Pharmacokinet. 1986;11(3):177–198 [DOI] [PubMed] [Google Scholar]
- 31.Kerr BM, Thummel KE, Wurden CJ, et al. Human liver carbamazepine metabolism. Role of CYP3A4 and CYP2C8 in 10,11-epoxide formation. Biochem Pharmacol. 1994;47(11):1969–1979 [DOI] [PubMed] [Google Scholar]
- 32.Hundt HK, Aucamp AK, Müller FO. Pharmacokinetic aspects of carbamazepine and its two major metabolites in plasma during overdosage. Hum Toxicol. 1983;2(4):607–614 [DOI] [PubMed] [Google Scholar]
- 33.George M, Kitzmiller JP, Ewald MB, O’Donell KA, Becter ML, Salhanick S. Methadone toxicity and possible induction and enhanced elimination in a premature neonate. J Med Toxicol. 2012;8(4):432–435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Santos NA, Medina WS, Martins NM, Mingatto FE, Curti C, Santos AC. Aromatic antiepileptic drugs and mitochondrial toxicity: effects on mitochondria isolated from rat liver. Toxicol In Vitro. 2008;22(5):1143–1152 [DOI] [PubMed] [Google Scholar]
- 35.Berger I, Segal I, Shmueli D, Saada A. The effect of antiepileptic drugs on mitochondrial activity: a pilot study. J Child Neurol. 2010;25(5):541–545 [DOI] [PubMed] [Google Scholar]
- 36.Li ZH, Li P, Randak T. Effect of a human pharmaceutical carbamazepine on antioxidant responses in brain of a model teleost in vitro: an efficient approach to biomonitoring. J Appl Toxicol. 2010;30(7):644–648 [DOI] [PubMed] [Google Scholar]
- 37.Forkink M, Manjeri GR, Liemburg-Apers DC, et al. Mitochondrial hyperpolarization during chronic complex I inhibition is sustained by low activity of complex II, III, IV and V. Biochim Biophys Acta. 2014;1837(8):1247–1256 [DOI] [PubMed] [Google Scholar]
- 38.Norman JP, Perry SW, Kasischke KA, Volsky DJ, Gelbard HA. HIV-1 trans activator of transcription protein elicits mitochondrial hyperpolarization and respiratory deficit, with dysregulation of complex IV and nicotinamide adenine dinucleotide homeostasis in cortical neurons. J Immunol. 2007;178(2):869–876 [DOI] [PubMed] [Google Scholar]
- 39.Perry SW, Norman JP, Litzburg A, Zhang D, Dewhurst S, Gelbard HA. HIV-1 transactivator of transcription protein induces mitochondrial hyperpolarization and synaptic stress leading to apoptosis. J Immunol. 2005;174(7):4333–4344 [DOI] [PubMed] [Google Scholar]
- 40.Ward MW, Huber HJ, Weisová P, Düssmann H, Nicholls DG, Prehn JH. Mitochondrial and plasma membrane potential of cultured cerebellar neurons during glutamate-induced necrosis, apoptosis, and tolerance. J Neurosci. 2007;27(31):8238–8249 [DOI] [PMC free article] [PubMed] [Google Scholar]