Abstract
A series of physiological and pathological changes occur after radiotherapy and accidental exposure to ionizing radiation (IR). These changes cause serious damage to human tissues and can lead to death. Radioprotective countermeasures are radioprotective agents that prevent and reduce IR injury or have therapeutic effects. Based on a good understanding of radiobiology, a number of protective agents have achieved positive results in early clinical trials. The present review grouped known radioprotective agents according to biochemical categories and potential clinical use, and reviewed radiation countermeasures, i.e., radioprotectors, radiation mitigators and radiotherapeutic agents, with an emphasis on their current status and research progress. The aim of the present review is to facilitate the selection and application of suitable radioprotectors for clinicians and researchers, to prevent or reduce IR injury.
Keywords: radioprotectors, mitigators, radiation therapeutic agents, radiation injury
1. Introduction
Ionizing radiation (IR) refers to energy released by atoms in the form of particles (neutrons, α or β particles) or electromagnetic waves (X-or γ-rays), which is sufficient to ionize atoms or molecules (1). Exposure to IR from these sources, including natural and artificial radiation, can have lethal consequences (2,3). The effects of IR are divided into deterministic effects, such as acute radiation sickness and radiation cataracts, and stochastic effects, such as radiation-induced cancer and genetic diseases (4). Even low-dose IR can cause DNA damage, and produce free radicals and reactive oxygen, causing DNA and protein damage, which can result in cell death, teratogenesis or carcinogenesis (5,6).
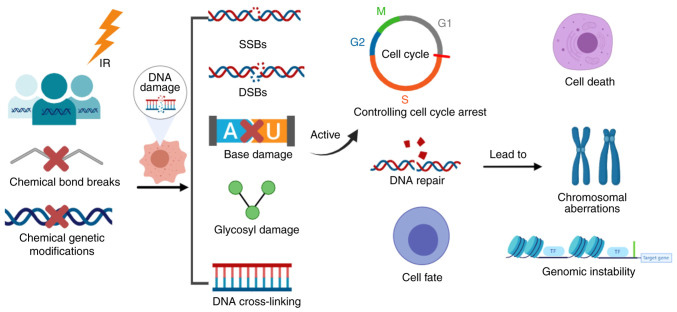
Exposure to IR can lead to chemical bond breaks or a variety of other chemical genetic modifications that can cause damage to biological macromolecules, particularly DNA, resulting in biochemical and biological cascade reactions. IR-induced DNA damage includes single-strand breaks and double-strand breaks (DSBs) (7–9), nucleotide base damage, glycosyl damage and DNA cross-linking, with DSBs being the most serious. DNA damage can activate a series of cellular DNA damage response signals, which control cell cycle arrest, DNA repair and cell fate. It can lead to serious consequences, such as cell death, chromosomal aberrations and genomic instability (Fig. 1). Furthermore, DNA damage followed by abnormal repair and genetic mutations is an important link in the development of tumors (8,10). Therefore, there is an urgent need to identify effective countermeasures against the harmful effects of IR.
Figure 1.
Pathway of radiation response. IR, ionizing radiation; SSBs, single-strand breaks; DSBs, double-strand breaks. The figure was generated using online drawing software tools (https://www.medpeer.cn).
2. Strategies to reduce radiation injury
IR countermeasures protect organisms against the harmful effects of IR and reduce tissue damage (11). Based on the time of administration relative to IR exposure, countermeasures against radiation injury are classified as radioprotectors, radiation mitigators or therapeutics (12–15). No radioprotective agent is both non-toxic at an effective dosage and capable of protecting normal cells from IR damage while maintaining the radiosensitivity of tumor cells.
IR-induced injury can be prevented using chemical compounds, biological agents, Chinese herbal extracts or cellular therapy (16,17). These measures reduce or improve radiation-induced tissue damage, and thus promote rehabilitation. Radioprotective countermeasures are radioprotective agents that prevent and reduce IR injury, or have therapeutic effects. The mechanism of action of radioprotective agents varies, yet the most common protective mechanism involves scavenging free radicals and enhancing DNA repair, thereby inhibiting oxidation and protecting cells.
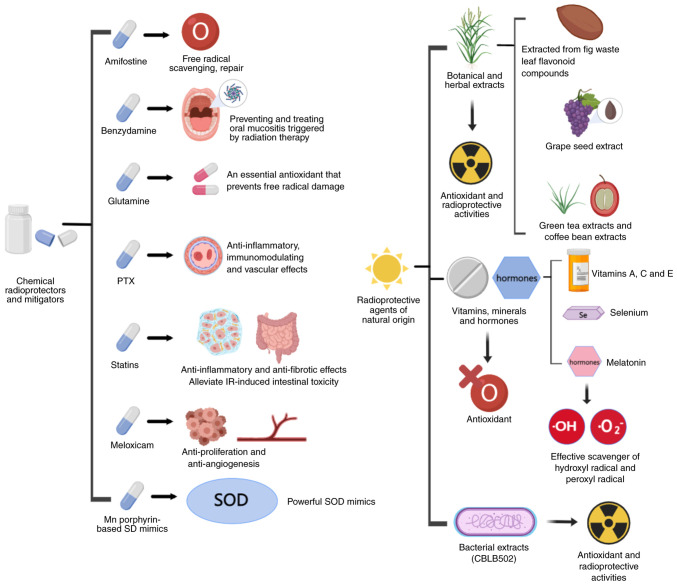
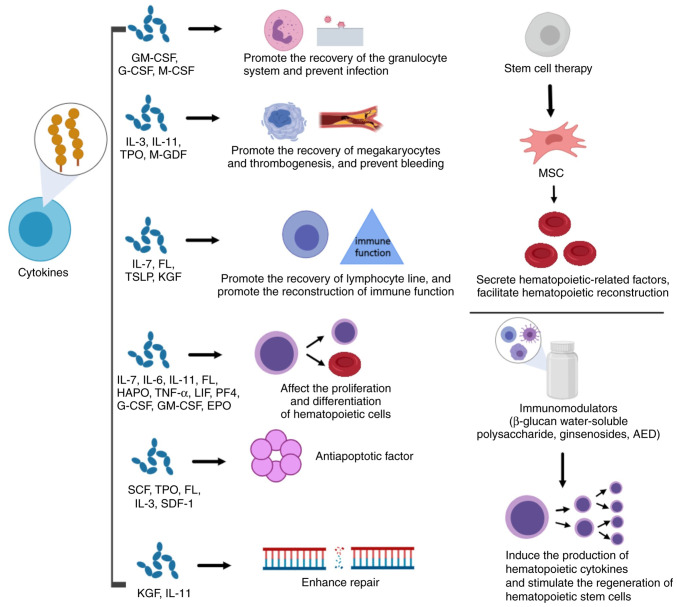
In the present review, the radioprotective mechanisms, and clinical and preclinical applications, of radioprotectors, radiation mitigators and radiation therapeutic agents are summarized and discussed. Known radioprotective agents were grouped according to biochemical categories and potential clinical use, and radiation countermeasures, i.e., radioprotectors, radiation mitigators and radiotherapeutic agents, were described as was their probable mechanism of action (Figs. 2 and 3). These measures, and the main emerging therapies, are listed in Tables I and II.
Figure 2.
Main radioprotectors or mitigators, and their probable mechanism of action. PTX, pentoxifylline; SOD, superoxide dismutase; Mn, manganese. The figure was generated using online drawing software tools (https://www.medpeer.cn).
Figure 3.
Main radiation treatment agents, along with their probable mechanism of action. GM-CSF, granulocyte-macrophage colony stimulating factor; G-CSF, granulocyte colony stimulating factor; M-CSF, macrophage colony stimulating factor; IL, interleukin; TPO, thrombopoietin; M-GDF, megakaryocyte growth development factor; FL, Flt-3 ligand; TSLP, thymic stromal lymphopoietin; KGF, keratinocyte growth factor; HAPO, hemangiopoietin; LIF, leukemia inhibitory factor; PF4, platelet factor 4; EPO, erythropoietin; SCF, stem cell factor; SDF-1, stromal cell-derived factor-1; MSC, mesenchymal stem cell; AED, 5-androstenone. The figure was generated using online drawing software tools (https://www.medpeer.cn).
Table I.
List of main radioprotectors or mitigators along with the probable mechanism of action.
| Type | Example | Mechanism of action | Limitations | (Refs.) |
|---|---|---|---|---|
| Chemical radioprotectors and mitigators | Most of them have low potency, short time of action, high toxicity and severe side effects (hypotension, vomiting, flushing) | |||
| Amifostine | Free radical scavenging, DNA repair | (18–31) | ||
| Benzydamine | Preventing and treating oral mucositis triggered by radiation therapy | (32–37) | ||
| Glutamine | An essential antioxidant that prevents free radical damage | (39–50) | ||
| Pentoxifylline (PTX) | Anti-inflammatory, immunomodulating and vascular effects | (51–56) | ||
| Statins | ||||
| Lovastatin | Anti-inflammatory and anti-fibrotic effects | (64) | ||
| Simvastatin and pravastatin | Alleviate IR-induced intestinal toxicity | (65–66) | ||
| Carbamazepine (CBZ) | Further research is needed | (70–73) | ||
| Meloxicam | Anti-proliferation and anti-angiogenesis | (74–78) | ||
| Metformin | Reducing tumor stem cells, and suppressing proliferation and hypoxia | (79–85) | ||
| Mn porphyrin-based SD mimics | Powerful SOD mimics | (86) | ||
| Redox nanoparticles | Free radical scavenging | (87–89) | ||
| Toll-like receptors | Inhibits apoptosis and improves cell survival | (90–102) | ||
| S1P | Promotes survival | (100,103–106) | ||
| Radioprotective agents of natural origin | Only in vitro and animal experiments have been performed, no clinical validation | |||
| Botanical and herbal extracts | Extracted from fig waste leaf flavonoid compounds | Strong antioxidant activity | (111,112) | |
| Grape seed extract | High antioxidant and free radical-scavenging capabilities | (107) | ||
| Green tea extracts and coffee bean extracts | Certain radioprotective effects | (108,113) | ||
| Vitamins, minerals and hormones | Vitamins A, C and E, and selenium | Antioxidant | (114–119) | |
| Melatonin | Effective scavenger of hydroxyl radical and peroxyl radical | (120,121) | ||
| Bacterial extracts | Recombinant polypeptide derived from Salmonella flagellin (CBLB502) | Antioxidant and radioprotective activities | (92,122–125) |
GM-CSF, granulocyte-macrophage colony stimulating factor; G-CSF, granulocyte colony stimulating factor; M-CSF, macrophage colony stimulating factor; IL, interleukin; TPO, thrombopoietin; M-GDF, megakaryocyte growth development factor; FL, Flt-3 ligand; TSLP, thymic stromal lymphopoietin; KGF, keratinocyte growth factor; HAPO, hemangiopoietin; LIF, leukemia inhibitory factor; PF4, platelet factor 4; EPO, erythropoietin; SCF, stem cell factor; SDF-1, stromal cell-derived factor-1; MSC, mesenchymal stem cell; AED, 5-androstenone.
Table II.
List of main radiation treatment agents along with the probable mechanism of action.
| Type | Example | Mechanism of action | Limitations | (Refs.) |
|---|---|---|---|---|
| Stem cell therapy | MSC | Secrete hematopoietic-related factors, facilitate hematopoietic reconstruction | Occurrence of severe GvHD | (127–152) |
| Cytokines | Regulate immune, nerve and endocrine function; participate in inflammation; promote wound healing; regulate hematopoiesis | Combination of cytokines needs further study | ||
| GM-CSF, G-CSF, M-CSF | Promote the recovery of the granulocyte system and preven infection | (129) | ||
| IL-3, IL-11, TPO, M-GDF | Promote the recovery of megakaryocytes and thrombogenesis, and prevent bleeding | (129) | ||
| IL-7, FL, TSLP, KGF | Promote the recovery of the lymphocyte line, and promote the reconstruction of immune function | (129) | ||
| IL-3, IL-6, IL-11, FL, HAPO, TNF-α, LIF, PF4, G-CSF, GM-CSF, EPO | Affect the proliferation and differentiation of hematopoietic cells | (153) | ||
| SCF, TPO, FL, IL-3, SDF-1 | Antiapoptotic factor | (153) | ||
| KGF, IL-11 | Enhance repair | (154) | ||
| Immunomodulators | β-glucan watersoluble polysaccharide, ginsenosides, AED | Induce the production of hematopoietic cytokines and stimulate the regeneration of hematopoietic stem cells | Proinflammatory response and immunogenicity; long-term use may cause allergies and organ damage | (159–161) |
GvHD, graft-vs. -host disease; GM-CSF, granulocyte-macrophage colony stimulating factor; G-CSF, granulocyte colony stimulating factor; M-CSF, macrophage colony stimulating factor; IL, interleukin; TPO, thrombopoietin; M-GDF, megakaryocyte growth development factor; FL, Flt-3 ligand; TSLP, thymic stromal lymphopoietin; KGF, keratinocyte growth factor; HAPO, hemangiopoietin; LIF, leukemia inhibitory factor; PF4, platelet factor 4; EPO, erythropoietin; SCF, stem cell factor; SDF-1, stromal cell-derived factor-1; MSC, mesenchymal stem cell; AED, 5-androstenone.
Radioprotectors in clinical use
Amifostine (AMF)
AMF is a United States Food and Drug Administration-approved selective normal tissue radioprotector that can be hydrolyzed and dephosphorylated to active N-(2-mercaptoethyl)-1,3-propanediamine by cell membrane-bound alkaline phosphatases, the sulfhydryl structure of which scavenges oxygen free radicals from tissue (18,19). Treatment with AMF during radiotherapy for lung cancer can reduce the incidence of severe radiation pneumonitis by 16%. AMF is mainly used to prevent radiotherapy-induced mucositis, dry mouth, dysphagia, pulmonary fibrosis and pneumonia (20–26). In previous studies, AMF selectively protected healthy cells against the harmful effects of radiotherapy, whereas cancer cells remained radiosensitive (27,28). However, AMF has a narrow therapeutic index, is administered intravenously and is toxic. AMF needs to be administered intravenously before radiotherapy; however, the drug is rapidly cleared from blood circulation, which cannot protect the gut. Furthermore, AMF is easily metabolized and sensitive to gastric acid, making direct oral administration an obstacle (19,29,30). Recently, it has been reported that a research team has produced orally available SP@AMF (31), which can prevent intestinal damage caused by radiation and prolong the survival period without affecting tumor regression in mice. Therefore, radioprotectors with oral administration, high efficacy, low toxicity and a long duration of action are needed.
Benzydamine
Benzydamine is a nonsteroidal anti-inflammatory drug that can inhibit the inflammatory factors TNF-α and IL-1β, and has antipyretic and analgesic effects (32–37). Radiotherapy-induced oral mucositis (RTOM) refers to inflammation of the oral mucosa caused by radiotherapy, which accounts for 80% of the complications from head and neck tumor radiotherapy. Benzydamine mouthwash has been reported to reduce RTOM pain in radioactive oral mucositis (38). In patients receiving radiotherapy, benzydamine oral rinse significantly reduced the rate of RTOM (32). Epstein et al (33) reported that benzydamine oral rinse reduced the rate of RTOM triggered by a high radiotherapy dose. Thus, benzydamine is recommended for the prevention and treatment of RTOM with simple radiotherapy for head and neck tumors in moderate doses.
Glutamine
Glutamine plays an important role in reducing radiotherapy side effects and improving body tolerance to radiotherapy (39–41). Radiotherapy kills cancer cells as well as normal cells in the body, especially intestinal mucosa cells, which can cause nausea, vomiting, diarrhea and other symptoms in patients receiving radiotherapy. A number of clinical studies have confirmed that glutamine is helpful in the repair of intestinal mucosa after radiotherapy and has significant effects on preventing and relieving adverse symptoms in patients with radioenteritis (41–44). An animal experiment showed that adding glutamine to feed could reduce acute radiation damage to intestinal mucosa structures and improve the antioxidant capacity of radiation-damaged animals (45). Similarly, the preventive effect of glutamine on acute radioesophagitis has been confirmed in several clinical trials (46–48). IR-induced enteritis was decreased by compound glutamine capsules. It has also been reported that the use of glutamine in patients receiving radiotherapy for breast cancer can significantly reduce the incidence of radiation injury (49). Therefore, taking compound glutamine capsules during radiotherapy can effectively prevent and improve the symptoms of radiation enteritis and esophagitis. Moreover, the combined supplementation of β-hydroxy-β-methylbutyrate, L-glutamine and L-arginine improved radiation-induced acute intestinal damage (50). Therefore, glutamine has an important protective effect for patients receiving radiotherapy and can improve the immune function of the body.
Pentoxifylline (PTX)
PTX is a methyl xanthine derivative with anti-inflammatory, immunomodulatory and vascular effects (51–53). PTX can be used for the treatment of delayed skin changes, such as skin fibrosis and necrosis caused by radiotherapy. It is usually combined with vitamin E after breast resection and reconstruction. It can also be used in the conservative treatment of radioactive bone necrosis (54). PTX can also decrease the risk of radiation-induced oral mucositis after oral administration. In an experimental pilot study, PTX suppressed TNF receptor upregulation and neuronal responsiveness in patients with RTOM (55). Similarly, radiotherapy combined with PTX and vitamin E reduced the incidence of severe oral mucositis and dysphagia after adjusting for age (56). Therefore, PTX treatment should be considered for radiodermatitis and radiation-induced mucositis.
Statins
Statins are used to lower lipid levels (57), and can ameliorate IR-induced inflammation and fibrotic remodeling (16,58). Statins have been found to reverse radiation-induced gene expression disorders such as p53. p53 is closely associated with radiosensitivity and radiation-induced cell death; notably, statins can target the p53-controlled mevalonate pathway, thereby reducing the risk of cancer (59,60). The IR-induced expression of proinflammatory and profibrotic marker genes was revealed to be attenuated by statins in vitro and in vivo (61–63). These effects were mediated by the attenuation of Rho signaling. Ostrau et al (64) reported that lovastatin showed tissue-specific anti-inflammatory and anti-fibrotic effects in vivo. Furthermore, simvastatin and pravastatin alleviated IR-induced intestinal toxicity by inhibiting Rho-ROCK signaling (65,66). Wedlake et al (67) demonstrated that statins reduced IR-induced acute gastrointestinal symptoms in a group of patients receiving pelvic radiotherapy, and showed long-term protective effects. However, while oral administration of simvastatin was shown to improve hematopoietic damage and gastrointestinal dysfunction caused by radiation in male mice, it worsened radiation-induced symptoms in female mice, which was related to sex-specific differences in gut flora (68). This suggests that using statins for treatment may be sex-specific. Based on these pleiotropic effects, especially on IR-induced intestinal toxicity, statins have potential as radioprotectors and mitigators for IR-induced injuries. In addition, statins can prevent radiation-induced heart disease. It was found that statins can inhibit activation of the transcription factor NF-κB during the acute response period after normal tissue exposure to radiation, and can significantly reduce TGF-β1, ROCK I and phosphorylated-Akt expression, thereby ameliorating radiation-induced cardiac fibrosis (69).
Carbamazepine (CBZ)
CBZ is an inducer of autophagy, which has been approved for the clinical treatment of bipolar disorder, trigeminal neuralgia and epilepsy (70). The safety of CBZ for patients with a variety of diseases has led to it being considered for radiation protection in humans. CBZ administration prior to radiation has been shown to increase hematopoietic cell survival and autophagy (71). However, despite its radioprotective and mitigative effects in normal murine tissue in vitro and in vivo, CBZ did not exert such effects in human cells in vitro. In addition, no reduction in side effects was observed in patients on CBZ for radiotherapy of trigeminal neuralgia, head-and-neck cancer or lung cancer (72,73).
Meloxicam
Meloxicam is a nonsteroidal anti-inflammatory drug (NSAID). It is a selective inhibitor of cyclooxygenase-2 (COX-2), with anti-proliferative and anti-angiogenetic effects, and is beneficial in alleviating gastrointestinal toxicity and improving radiation tolerability (74–76). Meloxicam can be used as a radioprotector in the mandible of irradiated rats. In addition, a single dose of meloxicam administered 1 h after a lethal radiation dose can achieve an increased survival rate of 30 days; however, it has been reported to be ineffective to administer meloxicam 24 h after lethal exposure (75). Intercellular adhesion molecule-1 (ICAM-1) and COX-2 are known factors involved in causing myocardial infarction after radiation exposure and were previously reported to be elevated after radiation exposure (77). According to Uehara et al (78), meloxicam somewhat upregulated radiation-induced expression of ICAM-1 and COX-2, i.e., the synergistic effect of NSAIDs with radiation was found, which would be a limitation of the clinical application of meloxicam.
Metformin
Metformin is a biguanide used for the treatment of type II diabetes. Previously, it has been report that metformin can exert radiosensitivity and radioprotective effects, and it can enhance the radiation response by reducing tumor stem cells, and suppressing proliferation and hypoxia (79–82). Additionally, metformin may prevent IR-induced esophageal carcinoma invasion and metastasis (83).
In vitro studies have shown, when administered 2 h before irradiation, metformin increased the expression of the BCL2 gene, and reduced the expression of BAX and CASP3 genes, thereby suppressing IR-induced apoptosis (84,85). Therefore, metformin has potential as a novel radioprotector against IR-induced apoptosis.
Emerging radioprotectors
Manganese (Mn) porphyrin-based superoxide dismutase (SOD) mimics
Mn porphyrins are powerful SOD mimics that have radioprotective effects in the lung, prostate and brain (86). Lead Mn porphyrins, namely MnTE-2-PyP5+ (BMX-010, AEOL10113), MnTnBuOE-2-PyP5+ (BMX-001) and MnTnHex-2-PyP5+, have entered clinical trials for glioma, head and neck cancer, anal cancer and multiple brain metastases, as well as for radioprotection of normal tissues during cancer radiotherapy (86).
Redox nanoparticles
Low-molecular-weight (LMW) nitroxide compounds have potential as radioprotectors or mitigators. Among the various nitroxide radicals, 4-amino-2,2,6,6-tetramethylpiperidine-1-oxyl (4-amino-TEMPO, also known as tempamine or NH2-TEMPO), had the highest radioprotective efficacy in vitro (87,88). Feliciano et al (89) reported that a novel nanoparticle-based radioprotective agent, NH2-TEMPO-containing redox nanoparticles, had good bioavailability and low toxicity. Most LMW compounds have extremely poor bioavailability, impairing their therapeutic efficacy and limiting their clinical use; therefore, their conversion into redox nanoparticles can effectively remove radiation-induced reactive oxygen species (ROS), with characteristic long-term bioavailability and extended tissue residence time.
Toll-like receptors (TLRs)
TLRs are pattern recognition receptors, which have been studied extensively in radiation protection in recent years (90–98). TLR2, TLR5 or TLR9 agonists have been shown to inhibit radiation-induced apoptosis and improve cell survival (91–93). The TLR4 agonist lipopolysaccharide has been reported to protect IR-induced bone marrow damage and reduce the mortality of mice after irradiation (94). The TLR5 ligand CBLB502 has been shown to serve a radioprotective role in mouse and rhesus monkey models of bone marrow, gut and reproductive damage (95,96). A novel TLR9 agonist containing synthetic immunomodulatory CpR (R=2′-deoxy-7-dezaguanosine) dinucleotide and 3′-3′-attached novel structures can also protect mice from radiation-induced gastrointestinal syndrome (97). There is growing clinical evidence that radiotherapy combined with new TLR agonists targeting TLR3, TLR7/8 or TLR9, in addition to protecting normal tissues from radiation and sensitizing cancer cells to ionizing radiation, may enhance antitumor immunity. Mechanistic studies have shown that TLR agonists can enhance dendritic cell-mediated T-cell initiation after radiotherapy, in some cases leading to the production of systemic antitumor immunity and immune memory (98–102). Additionally, it has been reported that one of the protective mechanisms of gut flora against radioactive intestinal injury is resistance through the TLR signaling pathway. Bacterial DNA binding TLR9 act as radiation protection by activating the transcription factor NF-κB. In addition to gut flora, this mechanism is also applicable to oral microorganisms (90), which supports the protection against radiotherapy-induced oral mucositis. Furthermore, activation of the TLR9 pathway protected against IR-induced intestinal injury (90,99). These findings suggested that activating the TLR signaling pathway may have significant radioprotective effects, indicating that TLRs have great advantages as a new target for radiation protection.
Sphingosine 1-phosphate (S1P)
S1P increases the likelihood of human cell survival by activating the phosphoinositide 3-kinase/AKT and mitochondrion-dependent pathways (100,103). Nitzsche et al (104) showed that S1P can maintain blood perfusion and microvascular patency in ischemic penumbra. Moreover, S1P and its analogs (FTY720/fingolimod) preserved testicular and ovarian function and fertility in IR-exposed animals and humans (105,106).
Radioprotective agents of natural origin
Some of the aforementioned chemical radioprotective agents are unsuitable for clinical use because of their toxicity and adverse side effects. Therefore, effective radioprotective agents with low toxicity have become a focus of research.
Botanical and herbal extracts
Some botanical and herbal plants, such as black tea extract, have radioprotective properties (107–110). Flavonoid compounds extracted from fig leaf exert strong antioxidant effects by removing superoxide anion and hydroxyl free radicals (111,112). Moreover, they significantly reduced the levels of ROS and malondialdehyde, and increased catalase, SOD and glutathione peroxidase activities in MC3T3-E1 mouse calvaria-derived preosteoblast cells. In another study (107), the blood samples of subjects who received 100, 300, 600 or 1,000 mg grape-seed extract were subjected to 1.5 Gy of X-ray radiation. The grape-seed extract reduced IR-induced DNA damage, exhibiting high antioxidant and free radical-scavenging effects. Furthermore, green tea and coffee bean extracts are reported to exert radioprotective effects (108,113).
Vitamins, minerals and hormones
Vitamins A, C and E, and selenium, are antioxidants that act as radioprotective agents (114). Dietary vitamin A in soybean oil can prevent the internal radiation-induced biological effects of radionuclides in mice (115). Vitamins C and E can reduce chromosome breakage in human lymphocytes by scavenging ROS, thus protecting against internal radiation damage caused by the radionuclide iodine-131 (116–118). Vitamin E, alone or in combination with WR-3689 [S-2((3-methylaminopropyl)amino)ethylphosphorothioic acid], increased the survival rate of IR-exposed mice (118). Selenium can also protect against free radical-induced damage. Selenium reversed the effects of IR on spermatogenesis in mice, thereby reducing damage to the testicles (119). Melatonin, a hormone produced by the pineal gland can scavenge hydroxyl radicals and peroxyl radicals to exert a radioprotective effect (120,121).
Bacterial extracts
Some bacterial species are resistant to radiation (122). A recombinant polypeptide from Salmonella flagellin (CBLB502) exhibited high radioprotective efficacy in mice and primates (92). In addition, an extracellular polysaccharide from the radiation-resistant bacterium Deinococcus showed antioxidant and radioprotective activities in vitro (123). Lactobacillus rhamnoides GG has been reported to protect the intestinal epithelium from radiation damage by releasing lipophosphate, promoting macrophage activation and the migration of mesenchymal stem cells (124). Furthermore, another study (125) reported that intestinal microorganisms are the primary regulators against radiation damage and protect the hematopoietic and gastrointestinal systems, among which Psilocytidae and Enterococcaceae and their downstream metabolites (propionate and tryptophan) have the greatest defense. The study suggests that microbial metabolites can protect against radiation damage; therefore, bacterial extracts have potential as radioprotective agents.
Radiation therapeutic agents
IR affects hematopoietic tissues, and significantly reduces the number of neutrophils and platelets. The resulting decrease in the number of peripheral blood cells can cause sepsis, bleeding, anemia and, in some cases, death (126). Therefore, radiation therapeutics aim to promote the recovery of hematopoietic function. At present, acute radiation sickness is treated clinically using biological agents, such as cytokines and mesenchymal stem cells (MSCs), modulate normal immune system function (127) and the secretion of hematopoietic growth factors, and promote reconstruction of the hematopoietic microenvironment (128). Biological agents have advantages such as low immunogenicity, and ease of transfection and expression of exogenous genes. Therefore, they are used to treat IR-induced injury (129). Table II lists the main radiation therapeutic agents along with the probable mechanism of action.
Stem cells
MSCs originate in the embryonic mesoderm and are the most important stem cells in the bone marrow matrix. MSCs are capable of multidirectional differentiation and immune regulation (127). MSCs secrete a variety of hematopoiesis-related factors, such as interleukin (IL)-6, Flt-3 ligand (FL), stem cell factor (SCF), granulocyte colony-stimulating factor (G-CSF) and granulocyte-macrophage colony stimulating factor (GM-CSF), making up for IR-induced deficiencies in cytokines. Therefore, MSCs can repair IR-induced damage to bone marrow stromal cells and the hematopoietic microenvironment. In addition, MSCs have hematopoietic support functions in bone marrow. The described characteristics of MSCs render them important therapeutics for IR-induced injury (130–132).
Liu et al (133) reported that MSCs promote recovery of hematopoietic function. Additionally, MSCs can inhibit T-cell activation and proliferation by secreting cytokines, growth factors and receptors. Co-transplantation of MSCs and hematopoietic stem cells into recipient bone marrow has been shown to prevent and reduce graft-versus-host disease (GvHD) (134). MSCs exhibit ectopic differentiation and block distal microvessels and other inherent defects (135). Further research will likely lead to the development of more effective and safer MSC-based therapeutics for IR-induced injuries.
Numerous studies and clinical trials have shown that MSCs have a significant effect on wound recovery from radiation burns (136–138). For the first time, stem cells were transplanted into a patient with deep skin burns and it was revealed that the patient exhibited faster healing. In addition, paracrine factors from stem cells used to treat local radiation burns in rats, can lead to increased skin regeneration and decreased leukocyte infiltration. Furthermore, Wharton's jelly-derived-MSC-derived conditioned medium (MSC-CM) (139) was revealed to be effective for the treatment of IR-induced skin wounds in rats, suggesting that MSC-CM could serve as the basis of novel cell-free treatments for radiation dermatitis.
For radiation-shock trauma, stem cells have been shown to enhance the response of the body to infection and improve chronic diseases, such as osteomyelitis and osteoarthritis that radiation-shock trauma can induce (140). In addition, compartment syndrome and segmental bone defects can also be treated with stem cell transplantation (141,142).
For treating acute radiation syndrome, hematopoietic stem cell transplantation can be used to rebuild hematopoietic functions, which attracted attention after the 1999 Tokaimura nuclear accident in Japan. Furthermore, MSCs can promote the implantation of hematopoietic stem cells and prevent the occurrence of GvHD (143), thus improving patient survival. In addition, a number of studies have shown that MSCs have a good tissue repair effect on the gastrointestinal tract, lung injury and burn-burn complex injury caused by radiation (144–146).
Radiation cataracts are also a serious complication caused by IR, and Maleki et al (147) showed that umbilical cord MSCs can be induced by the vitreous to differentiate into lens fibroblasts, thus providing the possibility for treatment of cataracts. In 2016, the Zhongshan University Eye Hospital used endogenous stem cells to regenerate new lenses in situ, successfully restoring complete vision to children with congenital cataracts (148).
Radiation exposure can also cause IR-induced xerostomia, which is a permanent side effect of radiotherapy for head and neck cancer that damages the large salivary glands (149–151). A clinical trial (152) of 33 patients with xerostomia caused by radiotherapy for oropharyngeal squamous cell carcinoma evaluated the long-term safety of injecting autologous adipose tissue-derived MSCs/stromal cells into the mandibular glands. The results showed that this therapy was safe and beneficial for IR-induced dry mouth-related symptoms.
In summary, MSC-based stem cell therapy has been used in a variety of radiation-induced diseases as an effective post-radiation disease treatment strategy and has played an important role in clinical practice.
Cytokines and immunomodulators
Cytokines regulate immune, nerve and endocrine function; participate in inflammation; promote wound healing; and regulate hematopoiesis. Some cytokines have marked effects on IR-induced injuries (129). Several cytokines promote recovery of granulocytes and prevent infection (129), such as GM-CSF, G-CSF and macrophage colony-stimulating factor, whereas others promote recovery of megakaryocytes and thrombogenesis, and prevent bleeding, such as IL-3, IL-11, thrombopoietin (TPO) and megakaryocyte growth development factor. Other cytokines promote lymphocyte-mediated recovery of immune function, such as IL-7, FL, thymic stromal lymphopoietin and keratinocyte growth factor (KGF). Some cytokines are involved in early differentiation, namely IL-3, IL-6, IL-11, FL, hemangiopoietin, TNF-α, leukemia inhibitory factor and platelet factor 4, whereas others act on the anaphase of differentiation (e.g., G-CSF, GM-CSF and erythropoietin). Cytokines with antiapoptotic effects include SCF, TPO, FL, IL-3 and stromal cell-derived factor-1 (153). Finally, some cytokines promote tissue repair and ameliorate multiple organ dysfunction syndrome, such as KGF and IL-11 (154). However, cytokines are pleiotropic, i.e., they exert varying biological effects on different target cells; for example, IL-11 can effectively promote the recovery of gastrointestinal mucosa (153). Certain cytokines interact with other cytokines, forming a complex cytokine network in vivo. Therefore, blocking or promoting one cytokine may affect the activities of other cytokines. Some cytokines increase sensitivity to IR when used alone, such as tumor growth factor-β, IL-6 and TNF-α (155–157). These cytokines must be used in combination with other cytokines. Therefore, further research is needed to identify the optimum combinations of cytokines for the treatment of IR injury.
Immunomodulators are not cytokines but induce the production of hematopoietic cytokines and stimulate the regeneration of hematopoietic stem cells. Cytokine release stimulates the growth, differentiation and proliferation of hematopoietic progenitor cells and stem cells. Therefore, immunomodulators are important therapeutics for IR-induced injury. Based on its immunopharmacological effect, β-glucan water-soluble polysaccharide has been used to modulate the host immune response (158). Glucan can increase the number of endogenous multifunctional hematopoietic stem cells in irradiated mice (159). Ginsenosides have also been shown to stimulate the production of IL-1 and IL-6. Administration of 100 mg/kg ginsenoside 24 h before γ-ray irradiation significantly increased the survival rate of mice, as well as the numbers of bone marrow cells, spleen cells, peripheral blood neutrophils, lymphocytes and platelets, and the GM-CFC level (160). The dehydroepiandrosterone derivative 5-androstenone has been reported to stimulate the synthesis of IL-1, IL-3 and IL-6 to promote recovery of the irradiated hematopoietic system in mice (161).
Gene therapy
There is much interest in gene therapy for radiation protection (162,163). At present, gene therapy for IR-induced injury typically involves growth factor and free radical inhibitor genes, to inhibit apoptosis and enhance damage repair (163). In addition, gene transfer vectors, multiple gene combinations and targeted specific gene therapy can be used to prevent harmful effects of IR. SOD, CAT, snail homolog 2, multidrug resistant gene 1, IL-3, KGF and erythropoietin have been evaluated in preclinical models regarding their radioprotective potential (164). Zhang et al (165) reported that IR-induced SOD2 overexpression increased the radiosensitivity of HT-29 colon cancer cells and prevented IR induced damage to normal colon (CCD841) cells.
3. Conclusion
Radiation exposure events occur occasionally, yet increasing numbers of patients are undergoing radiotherapy for tumors, which can have severe side effects. The present review discussed the clinical potential of radioprotectors, radiation mitigators and radiation therapeutic agents.
The ideal radioprotective agent would exert a protective effect when administered before or after IR exposure, prevent or repair IR-induced tissue damage, have a rapid onset of action and long half-life, be administered orally, and be resistant to the deleterious effects of IR and high temperatures. However, at present, most radioprotective agents with marketing approval or in clinical trials are administered intravenously. Accidental radiation exposure occur occasionally, yet increasing numbers of patients are undergoing radiotherapy for tumors, which can have severe side effects. The present review discussed the clinical potential of radioprotectors, radiation mitigators and radiation therapeutic agents. This review may help to provide improved ideas and application value for clinical radiological protection.
Acknowledgements
Not applicable.
Funding Statement
This work was supported by grants from the National Natural Science Foundation of China (grant nos. 81773363, 81872558 and 81972969).
Availability of data and materials
The data used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
LCL prepared and drafted the manuscript. LCL, ZZL and SMM searched the relevant literature and revised the manuscript. LL and XDL critically reviewed the manuscript. Data authentication is not applicable. All authors have read and approved the final version of the manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Obrador E, Salvador R, Villaescusa JI, Soriano JM, Estrela JM, Montoro A. Radioprotection and radiomitigation: From the bench to clinical practice. Biomedicines. 2020;8:461. doi: 10.3390/biomedicines8110461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mishra K, Alsbeih G. Appraisal of biochemical classes of radioprotectors: Evidence, current status and guidelines for future development. 3 Biotech. 2017;7:292. doi: 10.1007/s13205-017-0925-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Li X, Wang X, Miao L, Guo Y, Yuan R, Ren J, Huang Y, Tian H. Design, synthesis, and biological evaluation of a novel aminothiol compound as potential radioprotector. Oxid Med Cell Longev. 2021;2021:4714649. doi: 10.1155/2021/4714649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mishra KN, Moftah BA, Alsbeih GA. Appraisal of mechanisms of radioprotection and therapeutic approaches of radiation countermeasures. Biomed Pharmacother. 2018;106:610–617. doi: 10.1016/j.biopha.2018.06.150. [DOI] [PubMed] [Google Scholar]
- 5.Liu J, Bi K, Yang R, Li H, Nikitaki Z, Chang L. Role of DNA damage and repair in radiation cancer therapy: A current update and a look to the future. Int J Radiat Biol. 2020;96:1329–1338. doi: 10.1080/09553002.2020.1807641. [DOI] [PubMed] [Google Scholar]
- 6.Santivasi WL, Xia F. Ionizing radiation-induced DNA damage, response, and repair. Antioxid Redox Signal. 2014;21:251–259. doi: 10.1089/ars.2013.5668. [DOI] [PubMed] [Google Scholar]
- 7.Zhao L, Bao C, Shang Y, He X, Ma C, Lei X, Mi D, Sun Y. The determinant of DNA repair pathway choices in ionising radiation-induced DNA double-strand breaks. Biomed Res Int. 2020;2020:4834965. doi: 10.1155/2020/4834965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yao M, Rogers L, Suchowerska N, Choe D, Al-Dabbas MA, Narula RS, Lyons JG, Sved P, Li Z, Dong Q. Sensitization of prostate cancer to radiation therapy: Molecules and pathways to target. Radiother Oncol. 2018;128:283–300. doi: 10.1016/j.radonc.2018.05.021. [DOI] [PubMed] [Google Scholar]
- 9.Kavanagh JN, Redmond KM, Schettino G, Prise KM. DNA double strand break repair: A radiation perspective. Antioxid Redox Signal. 2013;18:2458–2472. doi: 10.1089/ars.2012.5151. [DOI] [PubMed] [Google Scholar]
- 10.Huang RX, Zhou PK. DNA damage response signaling pathways and targets for radiotherapy sensitization in cancer. Signal Transduct Target Ther. 2020;5:60. doi: 10.1038/s41392-020-0150-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Allegra AG, Mannino F, Innao V, Musolino C, Allegra A. Radioprotective agents and enhancers factors. Preventive and therapeutic strategies for oxidative induced radiotherapy damages in hematological malignancies. Antioxidants (Basel) 2020;9:1116. doi: 10.3390/antiox9111116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kalman NS, Zhao SS, Anscher MS, Urdaneta AI. Current status of targeted radioprotection and radiation injury mitigation and treatment agents: A critical review of the literature. Int J Radiat Oncol Biol Phys. 2017;98:662–682. doi: 10.1016/j.ijrobp.2017.02.211. [DOI] [PubMed] [Google Scholar]
- 13.Deas SD, Huprikar N, Skabelund A. Radiation exposure and lung disease in today's nuclear world. Curr Opin Pulm Med. 2017;23:167–172. doi: 10.1097/MCP.0000000000000349. [DOI] [PubMed] [Google Scholar]
- 14.Gudkov SV, Popova NR, Bruskov VI. Radioprotectors: History, trends and prospects. Biofizika. 2015;60:801–811. (In Russian) [PubMed] [Google Scholar]
- 15.Singh VK, Seed TM. Pharmacological management of ionizing radiation injuries: Current and prospective agents and targeted organ systems. Expert Opin Pharmacother. 2020;21:317–337. doi: 10.1080/14656566.2019.1702968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Patyar RR, Patyar S. Role of drugs in the prevention and amelioration of radiation induced toxic effects. Eur J Pharmacol. 2018;819:207–216. doi: 10.1016/j.ejphar.2017.12.011. [DOI] [PubMed] [Google Scholar]
- 17.Mun GI, Kim S, Choi E, Kim CS, Lee YS. Correction to: Pharmacology of natural radioprotectors. Arch Pharm Res. 2019;43:272–274. doi: 10.1007/s12272-019-01194-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cheema AK, Li Y, Girgis M, Jayatilake M, Fatanmi OO, Wise SY, Seed TM, Singh VK. Alterations in tissue metabolite profiles with amifostine-prophylaxed mice exposed to gamma radiation. Metabolites. 2020;10:211. doi: 10.3390/metabo10050211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.King M, Joseph S, Albert A, Thomas TV, Nittala MR, Woods WC, Vijayakumar S, Packianathan S. Use of amifostine for cytoprotection during radiation therapy: A review. Oncology. 2020;98:61–80. doi: 10.1159/000502979. [DOI] [PubMed] [Google Scholar]
- 20.Barbosa SCM, Pereira VBM, Wong DVT, Santana APM, Lucetti LT, Carvalho LL, Barbosa CRN, Callado RB, Silva CAA, Lopes CDH, et al. Amifostine reduces inflammation and protects against 5-fluorouracil-induced oral mucositis and hyposalivation. Braz J Med Biol Res. 2019;52:e8251. doi: 10.1590/1414-431x20188251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chang H, Yi W, Wang X, Tao Y, Yang X, Chen C, Zhang W, Zhou S, Liu S, Li X, et al. Effectiveness and safety of different amifostine regimens: Preliminary results of a phase II multicenter randomized controlled trial. Chin J Cancer Res. 2018;30:307–314. doi: 10.21147/j.issn.1000-9604.2018.03.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ferraiolo DM, Veitz-Keenan A. Insufficient evidence for interventions to prevent dry mouth and salivary gland dysfunction post head and neck radiotherapy. Evid Based Dent. 2018;19:30–31. doi: 10.1038/sj.ebd.6401295. [DOI] [PubMed] [Google Scholar]
- 23.Kim JM, Kim JW, Choi ME, Kim SK, Kim YM, Choi JS. Protective effects of curcumin on radioiodine-induced salivary gland dysfunction in mice. J Tissue Eng Regen Med. 2019;13:674–681. doi: 10.1002/term.2833. [DOI] [PubMed] [Google Scholar]
- 24.Ma SJ, Rivers CI, Serra LM, Singh AK. Long-term outcomes of interventions for radiation-induced xerostomia: A review. World J Clin Oncol. 2019;10:1–13. doi: 10.5306/wjco.v10.i1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Riley P, Glenny AM, Hua F, Worthington HV. Pharmacological interventions for preventing dry mouth and salivary gland dysfunction following radiotherapy. Cochrane Database Syst Rev. 2017;7:CD012744. doi: 10.1002/14651858.CD012744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Torun N, Muratli A, Serim BD, Ergulen A, Altun GD. Radioprotective effects of amifostine, L-carnitine and vitamin e in preventing early salivary gland injury due to radioactive iodine treatment. Curr Med Imaging Rev. 2019;15:395–404. doi: 10.2174/1573405614666180314150808. [DOI] [PubMed] [Google Scholar]
- 27.Luby AO, Subramanian C, Buchman LK, Lynn JV, Urlaub KM, Nelson NS, Donneys A, Cohen MS, Buchman SR. Amifostine prophylaxis in irradiated breast reconstruction: A study of oncologic safety in vitro. Ann Plast Surg. 2020;85:424–429. doi: 10.1097/SAP.0000000000002110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Molkentine JM, Fujimoto TN, Horvath TD, Grossberg AJ, Garcia CJG, Deorukhkar A, de la Cruz Bonilla M, Lin D, Samuel ELG, Chan WK, et al. Enteral activation of WR-2721 mediates radioprotection and improved survival from lethal fractionated radiation. Sci Rep. 2019;9:1949. doi: 10.1038/s41598-018-37147-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yu X, Li M, Zhu L, Li J, Zhang G, Fang R, Wu Z, Jin Y. Amifostine-loaded armored dissolving microneedles for long-term prevention of ionizing radiation-induced injury. Acta Biomater. 2020;112:87–100. doi: 10.1016/j.actbio.2020.05.025. [DOI] [PubMed] [Google Scholar]
- 30.Singh VK, Seed TM. The efficacy and safety of amifostine for the acute radiation syndrome. Expert Opin Drug Saf. 2019;18:1077–1090. doi: 10.1080/14740338.2019.1666104. [DOI] [PubMed] [Google Scholar]
- 31.Zhang D, Zhong D, Ouyang J, He J, Qi Y, Chen W, Zhang X, Tao W, Zhou M. Microalgae-based oral microcarriers for gut microbiota homeostasis and intestinal protection in cancer radiotherapy. Nat Commun. 2022;13:1413. doi: 10.1038/s41467-022-28744-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Daugėlaitė G, Užkuraitytė K, Jagelavičienė E, Filipauskas A. Prevention and treatment of chemotherapy and radiotherapy induced oral mucositis. Medicina (Kaunas) 2019;55:25. doi: 10.3390/medicina55020025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Epstein JB, Silverman S, Jr, Paggiarino DA, Crockett S, Schubert MM, Senzer NN, Lockhart PB, Gallagher MJ, Peterson DE, Leveque FG. Benzydamine HCl for prophylaxis of radiation-induced oral mucositis: Results from a multicenter, randomized, double-blind, placebo-controlled clinical trial. Cancer. 2001;92:875–885. doi: 10.1002/1097-0142(20010815)92:4<875::AID-CNCR1396>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 34.Nicolatou-Galitis O, Bossi P, Orlandi E, Bensadoun RJ. The role of benzydamine in prevention and treatment of chemoradiotherapy-induced mucositis. Support Care Cancer. 2021;29:5701–5709. doi: 10.1007/s00520-021-06048-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Manoharan V, Fareed N, Battur H, Khanagar S, Praveena J. Effectiveness of mouthrinses in prevention and treatment of radiation induced mucositis: A systematic review. J Cancer Res Ther. 2020;16((Suppl)):S1–S10. doi: 10.4103/jcrt.JCRT_176_18. [DOI] [PubMed] [Google Scholar]
- 36.Yu YT, Deng JL, Jin XR, Zhang ZZ, Zhang XH, Zhou X. Effects of 9 oral care solutions on the prevention of oral mucositis: A network meta-analysis of randomized controlled trials. Medicine (Baltimore) 2020;99:e19661. doi: 10.1097/MD.0000000000019661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chitapanarux I, Tungkasamit T, Petsuksiri J, Kannarunimit D, Katanyoo K, Chakkabat C, Setakornnukul J, Wongsrita S, Jirawatwarakul N, Lertbusayanukul C, et al. Randomized control trial of benzydamine HCl versus sodium bicarbonate for prophylaxis of concurrent chemoradiation-induced oral mucositis. Support Care Cancer. 2018;26:879–886. doi: 10.1007/s00520-017-3904-4. [DOI] [PubMed] [Google Scholar]
- 38.Kakoei S, Pardakhty A, Hashemipour MA, Larizadeh H, Kalantari B, Tahmasebi E. Comparison the pain relief of amitriptyline mouthwash with benzydamine in oral mucositis. J Dent (Shiraz) 2018;19:34–40. [PMC free article] [PubMed] [Google Scholar]
- 39.Papanikolopoulou A, Syrigos N, Vini L, Papasavva M, Lazopoulos G, Kteniadakis S, Spandidos DA, Charpidou A, Drakoulis N. Use of oral glutamine in radiation-induced adverse effects in patients with thoracic and upper aerodigestive malignancies: Results of a prospective observational study. Oncol Lett. 2022;23:19. doi: 10.3892/ol.2021.13137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chang SC, Lai YC, Hung JC, Chang CY. Oral glutamine supplements reduce concurrent chemoradiotherapy-induced esophagitis in patients with advanced non-small cell lung cancer. Medicine (Baltimore) 2019;98:e14463. doi: 10.1097/MD.0000000000014463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cao DD, Xu HL, Xu M, Qian XY, Yin ZC, Ge W. Therapeutic role of glutamine in management of radiation enteritis: A meta-analysis of 13 randomized controlled trials. Oncotarget. 2017;8:30595–30605. doi: 10.18632/oncotarget.15741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vidal-Casariego A, Calleja-Fernández A, de Urbina-González JJ, Cano-Rodríguez I, Cordido F, Ballesteros-Pomar MD. Efficacy of glutamine in the prevention of acute radiation enteritis: A randomized controlled trial. JPEN J Parenter Enteral Nutr. 2014;38:205–213. doi: 10.1177/0148607113478191. [DOI] [PubMed] [Google Scholar]
- 43.Vidal-Casariego A, Calleja-Fernández A, Cano-Rodríguez I, Cordido F, Ballesteros-Pomar MD. Effects of oral glutamine during abdominal radiotherapy on chronic radiation enteritis: A randomized controlled trial. Nutrition. 2015;31:200–204. doi: 10.1016/j.nut.2014.08.003. [DOI] [PubMed] [Google Scholar]
- 44.Erbil Y, Oztezcan S, Giriş M, Barbaros U, Olgaç V, Bilge H, Küçücük H, Toker G. The effect of glutamine on radiation-induced organ damage. Life Sci. 2005;78:376–382. doi: 10.1016/j.lfs.2005.04.068. [DOI] [PubMed] [Google Scholar]
- 45.Ersin S, Tuncyurek P, Esassolak M, Alkanat M, Buke C, Yilmaz M, Telefoncu A, Kose T. The prophylactic and therapeutic effects of glutamine- and arginine-enriched diets on radiation-induced enteritis in rats. J Surg Res. 2000;89:121–125. doi: 10.1006/jsre.1999.5808. [DOI] [PubMed] [Google Scholar]
- 46.Alshawa A, Cadena AP, Stephen B, Reddy A, Mendoza TR, McQuinn L, Lawhorn K, Zarifa A, Bernhardt AM, Fessaheye S, et al. Effects of glutamine for prevention of radiation-induced esophagitis: A double-blind placebo-controlled trial. Invest New Drugs. 2021;39:1113–1122. doi: 10.1007/s10637-021-01074-w. [DOI] [PubMed] [Google Scholar]
- 47.Topkan E, Yavuz MN, Onal C, Yavuz AA. Prevention of acute radiation-induced esophagitis with glutamine in non-small cell lung cancer patients treated with radiotherapy: Evaluation of clinical and dosimetric parameters. Lung Cancer. 2009;63:393–399. doi: 10.1016/j.lungcan.2008.06.015. [DOI] [PubMed] [Google Scholar]
- 48.Vidal-Casariego A, Calleja-Fernández A, Ballesteros-Pomar MD, Cano-Rodríguez I. Efficacy of glutamine in the prevention of oral mucositis and acute radiation-induced esophagitis: A retrospective study. Nutr Cancer. 2013;65:424–429. doi: 10.1080/01635581.2013.765017. [DOI] [PubMed] [Google Scholar]
- 49.Rubio I, Suva LJ, Todorova V, Bhattacharyya S, Kaufmann Y, Maners A, Smith M, Klimberg VS. Oral glutamine reduces radiation morbidity in breast conservation surgery. JPEN J Parenter Enteral Nutr. 2013;37:623–630. doi: 10.1177/0148607112474994. [DOI] [PubMed] [Google Scholar]
- 50.Yavas C, Yavas G, Celik E, Buyukyoruk A, Buyukyoruk C, Yuce D, Ata O. Beta-hydroxy-beta-methyl-butyrate, L-glutamine, and L-arginine supplementation improves radiation-induce acute intestinal toxicity. J Diet Suppl. 2019;16:576–591. doi: 10.1080/19390211.2018.1472709. [DOI] [PubMed] [Google Scholar]
- 51.Setiadi A, Korim WS, May CN, Yao ST. Systemic administration of pentoxifylline attenuates the development of hypertension in renovascular hypertensive rats. Hypertens Res. 2020;43:667–678. doi: 10.1038/s41440-020-0412-6. [DOI] [PubMed] [Google Scholar]
- 52.Hendawy N. Pentoxifylline attenuates cytokine stress and Fas system in syngeneic liver proteins induced experimental autoimmune hepatitis. Biomed Pharmacother. 2017;92:316–323. doi: 10.1016/j.biopha.2017.05.084. [DOI] [PubMed] [Google Scholar]
- 53.Seirafianpour F, Mozafarpoor S, Fattahi N, Sadeghzadeh-Bazargan A, Hanifiha M, Goodarzi A. Treatment of COVID-19 with pentoxifylline: Could it be a potential adjuvant therapy? Dermatol Ther. 2020;33:e13733. doi: 10.1111/dth.13733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wang K, Tepper JE. Radiation therapy-associated toxicity: Etiology, management, and prevention. CA Cancer J Clin. 2021;71:437–454. doi: 10.3322/caac.21689. [DOI] [PubMed] [Google Scholar]
- 55.Price ML, Lai YE, Marcus KL, Robertson JB, Lascelles BDX, Nolan MW. Early radiation-induced oral pain signaling responses are reduced with pentoxifylline treatment. Vet Radiol Ultrasound. 2021;62:255–263. doi: 10.1111/vru.12943. [DOI] [PubMed] [Google Scholar]
- 56.Andreyev HJN, Matthews J, Adams C, Gothard L, Lucy C, Tovey H, Boyle S, Anbalagan S, Musallam A, Yarnold J, et al. Randomised single centre double-blind placebo controlled phase II trial of Tocovid SupraBio in combination with pentoxifylline in patients suffering long-term gastrointestinal adverse effects of radiotherapy for pelvic cancer: The PPALM study. Radiother Oncol. 2022;168:130–137. doi: 10.1016/j.radonc.2022.01.024. [DOI] [PubMed] [Google Scholar]
- 57.Pedro-Botet J, Pintó X. LDL-cholesterol: The lower the better. Clin Investig Arterioscler. 2019;31((Suppl 2)):S16–S27. doi: 10.1016/j.arteri.2019.10.003. [DOI] [PubMed] [Google Scholar]
- 58.Lee MS, Liu DW, Hung SK, Yu CC, Chi CL, Chiou WY, Chen LC, Lin RI, Huang LW, Chew CH, et al. Emerging challenges of radiation-associated cardiovascular dysfunction (RACVD) in modern radiation oncology: Clinical practice, bench investigation, and multidisciplinary care. Front Cardiovasc Med. 2020;7:16. doi: 10.3389/fcvm.2020.00016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Moon SH, Huang CH, Houlihan SL, Regunath K, Freed-Pastor WA, Morris JP, IV, Tschaharganeh DF, Kastenhuber ER, Barsotti AM, Culp-Hill R, et al. p53 represses the mevalonate pathway to mediate tumor suppression. Cell. 2019;176:564–580.e19. doi: 10.1016/j.cell.2018.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Li P, Liu X, Zhao T, Li F, Wang Q, Zhang P, Hirayama R, Chen W, Jin X, Zheng X, et al. Comparable radiation sensitivity in p53 wild-type and p53 deficient tumor cells associated with different cell death modalities. Cell Death Discov. 2021;7:184. doi: 10.1038/s41420-021-00570-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kwak SY, Park S, Kim H, Lee SJ, Jang WS, Kim MJ, Lee S, Jang WI, Kim AR, Kim EH, et al. Atorvastatin inhibits endothelial PAI-1-mediated monocyte migration and alleviates radiation-induced enteropathy. Int J Mol Sci. 2021;22:1828. doi: 10.3390/ijms22041828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pathak R, Kumar VP, Hauer-Jensen M, Ghosh SP. Enhanced survival in mice exposed to ionizing radiation by combination of gamma-tocotrienol and simvastatin. Mil Med. 2019;184((Suppl 1)):S644–S651. doi: 10.1093/milmed/usy408. [DOI] [PubMed] [Google Scholar]
- 63.Anscher MS, Chang MG, Moghanaki D, Rosu M, Mikkelsen RB, Holdford D, Skinner V, Grob BM, Sanyal A, Wang A, Mukhopadhyay ND. A phase II study to prevent radiation-induced rectal injury with lovastatin. Am J Clin Oncol. 2018;41:544–548. doi: 10.1097/COC.0000000000000320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ostrau C, Hülsenbeck J, Herzog M, Schad A, Torzewski M, Lackner KJ, Fritz G. Lovastatin attenuates ionizing radiation-induced normal tissue damage in vivo. Radiother Oncol. 2009;92:492–499. doi: 10.1016/j.radonc.2009.06.020. [DOI] [PubMed] [Google Scholar]
- 65.Ziegler V, Henninger C, Simiantonakis I, Buchholzer M, Ahmadian MR, Budach W, Fritz G. Rho inhibition by lovastatin affects apoptosis and DSB repair of primary human lung cells in vitro and lung tissue in vivo following fractionated irradiation. Cell Death Dis. 2017;8:e2978. doi: 10.1038/cddis.2017.372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bachelet JT, Granzotto A, Ferlazzo M, Sonzogni L, Berthel E, Devic C, Foray N. First radiobiological characterization of the McCune-Albright syndrome: Influence of the ATM protein and effect of statins + bisphosphonates treatment. Int J Radiat Biol. 2021;97:317–328. doi: 10.1080/09553002.2021.1864045. [DOI] [PubMed] [Google Scholar]
- 67.Wedlake LJ, Silia F, Benton B, Lalji A, Thomas K, Dearnaley DP, Blake P, Tait D, Khoo VS, Andreyev HJ. Evaluating the efficacy of statins and ACE-inhibitors in reducing gastrointestinal toxicity in patients receiving radiotherapy for pelvic malignancies. Eur J Cancer. 2012;48:2117–2124. doi: 10.1016/j.ejca.2011.12.034. [DOI] [PubMed] [Google Scholar]
- 68.Cui M, Xiao H, Li Y, Zhang S, Dong J, Wang B, Zhu C, Jiang M, Zhu T, He J, et al. Sexual dimorphism of gut microbiota dictates therapeutics efficacy of radiation injuries. Adv Sci (Weinh) 2019;6:1901048. doi: 10.1002/advs.201901048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang K, He X, Zhou Y, Gao L, Qi Z, Chen J, Gao X. Atorvastatin ameliorates radiation-induced cardiac fibrosis in rats. Radiat Res. 2015;184:611–620. doi: 10.1667/RR14075.1. [DOI] [PubMed] [Google Scholar]
- 70.Do TM, Unis GD, Kattar N, Ananth A, McCoul ED. Neuromodulators for atypical facial pain and neuralgias: A systematic review and meta-analysis. Laryngoscope. 2021;131:1235–1253. doi: 10.1002/lary.29162. [DOI] [PubMed] [Google Scholar]
- 71.Guéguen Y, Bontemps A, Ebrahimian TG. Adaptive responses to low doses of radiation or chemicals: Their cellular and molecular mechanisms. Cell Mol Life Sci. 2019;76:1255–1273. doi: 10.1007/s00018-018-2987-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Di Stefano G, Maarbjerg S, Truini A. Trigeminal neuralgia secondary to multiple sclerosis: From the clinical picture to the treatment options. J Headache Pain. 2019;20:20. doi: 10.1186/s10194-019-0969-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Bal W, Łabuz-Roszak B, Tarnawski R, Lasek-Bal A. Effectiveness and safety of CyberKnife radiosurgery in treatment of trigeminalgia-experiences of Polish neurological and oncological centres. Neurol Neurochir Pol. 2020;54:28–32. doi: 10.5603/PJNNS.a2020.0009. [DOI] [PubMed] [Google Scholar]
- 74.Khalil NY, Aldosari KF. Meloxicam. Profiles Drug Subst Excip Relat Methodol. 2020;45:159–197. doi: 10.1016/bs.podrm.2019.10.006. [DOI] [PubMed] [Google Scholar]
- 75.Yamasaki MC, Roque-Torres GD, Peroni LV, Nascimento EHL, Salmon B, Oliveira ML, Freitas DQ, Correr-Sobrinho L. Does the administration of meloxicam before head and neck radiotherapy reduce the risk of mandibular osteoradionecrosis? An animal model study. Clin Oral Investig. 2021;25:3739–3745. doi: 10.1007/s00784-020-03701-x. [DOI] [PubMed] [Google Scholar]
- 76.Yamasaki MC, Nejaim Y, Roque-Torres GD, Freitas DQ. Meloxicam as a radiation-protective agent on mandibles of irradiated rats. Braz Dent J. 2017;28:249–255. doi: 10.1590/0103-6440201701271. [DOI] [PubMed] [Google Scholar]
- 77.Zhou Y, Dong X, Xiu P, Wang X, Yang J, Li L, Li Z, Sun P, Shi X, Zhong J. Meloxicam, a selective COX-2 inhibitor, mediates hypoxia-inducible factor-(HIF-) 1α signaling in hepatocellular carcinoma. Oxid Med Cell Longev. 2020;2020:7079308. doi: 10.1155/2020/7079308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Uehara Y, Murata Y, Shiga S, Hosoi Y. NSAIDs diclofenac, indomethacin, and meloxicam highly upregulate expression of ICAM-1 and COX-2 induced by X-irradiation in human endothelial cells. Biochem Biophys Res Commun. 2016;479:847–852. doi: 10.1016/j.bbrc.2016.09.120. [DOI] [PubMed] [Google Scholar]
- 79.Zhang KF, Wang J, Guo J, Huang YY, Huang TR. Metformin enhances radiosensitivity in hepatocellular carcinoma by inhibition of specificity protein 1 and epithelial-to-mesenchymal transition. J Cancer Res Ther. 2019;15:1603–1610. doi: 10.4103/jcrt.JCRT_297_19. [DOI] [PubMed] [Google Scholar]
- 80.Wang J, Wang Y, Han J, Mei H, Yu D, Ding Q, Zhang T, Wu G, Peng G, Lin Z. Metformin attenuates radiation-induced pulmonary fibrosis in a murine model. Radiat Res. 2017;188:105–113. doi: 10.1667/RR14708.1. [DOI] [PubMed] [Google Scholar]
- 81.Da F, Guo J, Yao L, Gao Q, Jiao S, Miao X, Liu J. Pretreatment with metformin protects mice from whole-body irradiation. J Radiat Res. 2021;62:618–625. doi: 10.1093/jrr/rrab012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Adeberg S, Bernhardt D, Harrabi SB, Nicolay NH, Hörner-Rieber J, König L, Repka M, Mohr A, Abdollahi A, Weber KJ, et al. Metformin enhanced in vitro radiosensitivity associates with G2/M cell cycle arrest and elevated adenosine-5′-monophosphate-activated protein kinase levels in glioblastoma. Radiol Oncol. 2017;51:431–437. doi: 10.1515/raon-2017-0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Nakayama A, Ninomiya I, Harada S, Tsukada T, Okamoto K, Nakanuma S, Sakai S, Makino I, Kinoshita J, Hayashi H, et al. Metformin inhibits the radiation-induced invasive phenotype of esophageal squamous cell carcinoma. Int J Oncol. 2016;49:1890–1898. doi: 10.3892/ijo.2016.3676. [DOI] [PubMed] [Google Scholar]
- 84.Rezaei N, Neshasteh-Riz A, Mazaheri Z, Koosha F, Hoormand M. The combination of metformin and disulfiram-cu for effective radiosensitization on glioblastoma cells. Cell J. 2020;22:263–272. doi: 10.22074/cellj.2020.6798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kolivand S, Motevaseli E, Cheki M, Mahmoudzadeh A, Shirazi A, Fait V. The anti-apoptotic mechanism of metformin against apoptosis induced by ionizing radiation in human peripheral blood mononuclear cells. Klin Onkol. 2017;30:372–379. doi: 10.14735/amko2017372. [DOI] [PubMed] [Google Scholar]
- 86.Batinic-Haberle I, Tovmasyan A, Huang Z, Duan W, Du L, Siamakpour-Reihani S, Cao Z, Sheng H, Spasojevic I, Alvarez Secord A. H2O2-driven anticancer activity of Mn porphyrins and the underlying molecular pathways. Oxid Med Cell Longev. 2021;2021:6653790. doi: 10.1155/2021/6653790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Feliciano CP, Nagasaki Y. Oral nanotherapeutics: Redox nanoparticles attenuate ultraviolet B radiation-induced skin inflammatory disorders in Kud: Hr-hairless mice. Biomaterials. 2017;142:162–170. doi: 10.1016/j.biomaterials.2017.07.015. [DOI] [PubMed] [Google Scholar]
- 88.Wang F, Gao P, Guo L, Meng P, Fan Y, Chen Y, Lin Y, Guo G, Ding G, Wang H. Radio-protective effect and mechanism of 4-Acetamido-2,2,6,6-tetramethylpiperidin-1-oxyl in HUVEC cells. Environ Health Prev Med. 2017;22:14. doi: 10.1186/s12199-017-0616-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Feliciano CP, Tsuboi K, Suzuki K, Kimura H, Nagasaki Y. Long-term bioavailability of redox nanoparticles effectively reduces organ dysfunctions and death in whole-body irradiated mice. Biomaterials. 2017;129:68–82. doi: 10.1016/j.biomaterials.2017.03.011. [DOI] [PubMed] [Google Scholar]
- 90.Liu Z, Lei X, Li X, Cai JM, Gao F, Yang YY. Toll-like receptors and radiation protection. Eur Rev Med Pharmacol Sci. 2018;22:31–39. doi: 10.26355/eurrev_201801_14097. [DOI] [PubMed] [Google Scholar]
- 91.Chen Y, Xu Y, Du J, Guo J, Lei X, Cui J, Liu C, Cheng Y, Li B, Gao F, et al. Radioprotective effects of heat-killed mycobacterium tuberculosis in cultured cells and radiosensitive tissues. Cell Physiol Biochem. 2016;40:716–726. doi: 10.1159/000452583. [DOI] [PubMed] [Google Scholar]
- 92.Shi T, Li L, Zhou G, Wang C, Chen X, Zhang R, Xu J, Lu X, Jiang H, Chen J. Toll-like receptor 5 agonist CBLB502 induces radioprotective effects in vitro. Acta Biochim Biophys Sin (Shanghai) 2017;49:487–495. doi: 10.1093/abbs/gmx034. [DOI] [PubMed] [Google Scholar]
- 93.Zheng L, Asprodites N, Keene AH, Rodriguez P, Brown KD, Davila E. TLR9 engagement on CD4 T lymphocytes represses gamma-radiation-induced apoptosis through activation of checkpoint kinase response elements. Blood. 2008;111:2704–2713. doi: 10.1182/blood-2007-07-104141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Liu C, Zhang C, Mitchel RE, Cui J, Lin J, Yang Y, Liu X, Cai J. A critical role of toll-like receptor 4 (TLR4) and its' in vivo ligands in basal radio-resistance. Cell Death Dis. 2013;4:e649. doi: 10.1038/cddis.2013.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Burdelya LG, Krivokrysenko VI, Tallant TC, Strom E, Gleiberman AS, Gupta D, Kurnasov OV, Fort FL, Osterman AL, Didonato JA, et al. An agonist of toll-like receptor 5 has radioprotective activity in mouse and primate models. Science. 2008;320:226–230. doi: 10.1126/science.1154986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Bai H, Sun F, Yang G, Wang L, Zhang Q, Zhang Q, Zhan Y, Chen J, Yu M, Li C, et al. CBLB502, a Toll-like receptor 5 agonist, offers protection against radiation-induced male reproductive system damage in mice. Biol Reprod. 2019;100:281–291. doi: 10.1093/biolre/ioy173. [DOI] [PubMed] [Google Scholar]
- 97.Saha S, Bhanja P, Liu L, Alfieri AA, Yu D, Kandimalla ER, Agrawal S, Guha C. TLR9 agonist protects mice from radiation-induced gastrointestinal syndrome. PLoS One. 2012;7:e29357. doi: 10.1371/journal.pone.0029357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Walshaw RC, Honeychurch J, Choudhury A, Illidge TM. Toll-like receptor agonists and radiation therapy combinations: An untapped opportunity to induce anticancer immunity and improve tumor control. Int J Radiat Oncol Biol Phys. 2020;108:27–37. doi: 10.1016/j.ijrobp.2020.04.020. [DOI] [PubMed] [Google Scholar]
- 99.Schmitt H, Ulmschneider J, Billmeier U, Vieth M, Scarozza P, Sonnewald S, Reid S, Atreya I, Rath T, Zundler S, et al. The TLR9 agonist cobitolimod induces IL10-producing wound healing macrophages and regulatory T cells in ulcerative colitis. J Crohns Colitis. 2020;14:508–524. doi: 10.1093/ecco-jcc/jjz170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Ogretmen B. Sphingolipid metabolism in cancer signalling and therapy. Nat Rev Cancer. 2018;18:33–50. doi: 10.1038/nrc.2017.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Li Y, Jiang X, Luo T, Xia J, Lee MJ, Weichselbaum RR, Lin W. TLR3 agonist nanoscale coordination polymer synergizes with immune checkpoint blockade for immunotherapy of cancer. Biomaterials. 2022;290:121831. doi: 10.1016/j.biomaterials.2022.121831. [DOI] [PubMed] [Google Scholar]
- 102.Zhang Y, Feng Z, Liu J, Li J, Su Q, Zhang J, Huang P, Wang W, Liu J. Polarization of tumor-associated macrophages by TLR7/8 conjugated radiosensitive peptide hydrogel for overcoming tumor radioresistance. Bioact Mater. 2022;16:359–371. doi: 10.1016/j.bioactmat.2021.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wang H, Huang H, Ding SF. Sphingosine-1-phosphate promotes the proliferation and attenuates apoptosis of Endothelial progenitor cells via S1PR1/S1PR3/PI3K/Akt pathway. Cell Biol Int. 2018;42:1492–1502. doi: 10.1002/cbin.10991. [DOI] [PubMed] [Google Scholar]
- 104.Nitzsche A, Poittevin M, Benarab A, Bonnin P, Faraco G, Uchida H, Favre J, Garcia-Bonilla L, Garcia MCL, Léger PL, et al. Endothelial S1P1 signaling counteracts infarct expansion in ischemic stroke. Circ Res. 2021;128:363–382. doi: 10.1161/CIRCRESAHA.120.316711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zelinski MB, Murphy MK, Lawson MS, Jurisicova A, Pau KY, Toscano NP, Jacob DS, Fanton JK, Casper RF, Dertinger SD, Tilly JL. In vivo delivery of FTY720 prevents radiation-induced ovarian failure and infertility in adult female nonhuman primates. Fertil Steril. 2011;95:1440–1445.e1-e7. doi: 10.1016/j.fertnstert.2011.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Guzel Y, Bildik G, Oktem O. Sphingosine-1-phosphate protects human ovarian follicles from apoptosis in vitro. Eur J Obstet Gynecol Reprod Biol. 2018;222:19–24. doi: 10.1016/j.ejogrb.2018.01.001. [DOI] [PubMed] [Google Scholar]
- 107.Ghasemnezhad Targhi R, Saba V. Grape seed extract alleviates radiation-induced damages in human blood lymphocytes. Avicenna J Phytomed. 2020;10:398–406. [PMC free article] [PubMed] [Google Scholar]
- 108.Ansari L, Banaei A, Dastranj L, Majdaeen M, Vafapour H, Zamani H, Ataei G, Abedi-Firouzjah R. Evaluating the radioprotective effect of single dose and daily oral consumption of green tea, grape seed, and coffee bean extracts against gamma irradiation. Appl Radiat Isot. 2021;174:109781. doi: 10.1016/j.apradiso.2021.109781. [DOI] [PubMed] [Google Scholar]
- 109.Ghosh D, Pal S, Saha C, Chakrabarti AK, Datta SC, Dey SK. Black tea extract: A supplementary antioxidant in radiation-induced damage to DNA and normal lymphocytes. J Environ Pathol Toxicol Oncol. 2012;31:155–166. doi: 10.1615/JEnvironPatholToxicolOncol.v31.i2.70. [DOI] [PubMed] [Google Scholar]
- 110.Ježovičová M, Koňariková K, Ďuračková Z, Keresteš J, Králik G, Žitňanová I. Protective effects of black tea extract against oxidative DNA damage in human lymphocytes. Mol Med Rep. 2016;13:1839–1844. doi: 10.3892/mmr.2015.4747. [DOI] [PubMed] [Google Scholar]
- 111.Shen N, Wang T, Gan Q, Liu S, Wang L, Jin B. Plant flavonoids: Classification, distribution, biosynthesis, and antioxidant activity. Food Chem. 2022;383:132531. doi: 10.1016/j.foodchem.2022.132531. [DOI] [PubMed] [Google Scholar]
- 112.Li C, Yu M, Li S, Yang X, Qiao B, Shi S, Zhao C, Fu Y. Valorization of Fig (Ficus carica L.) waste leaves: HPLC-QTOF-MS/MS-DPPH system for online screening and identification of antioxidant compounds. Plants (Basel) 2021;10:2532. doi: 10.3390/plants10112532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Kamran MZ, Ranjan A, Kaur N, Sur S, Tandon V. Radioprotective agents: Strategies and translational advances. Med Res Rev. 2016;36:461–493. doi: 10.1002/med.21386. [DOI] [PubMed] [Google Scholar]
- 114.Mortazavi SM, Rahimi S, Mosleh-Shirazi MA, Arjomandi M, Soleimani A, Koohi Hossein-Abadi O, Haghani M, Alavi M. A comparative study on the life-saving radioprotective effects of vitamins A, E, C and over-the-counter multivitamins. J Biomed Phys Eng. 2015;5:59–66. [PMC free article] [PubMed] [Google Scholar]
- 115.Harapanhalli RS, Narra VR, Yaghmai V, Azure MT, Goddu SM, Howell RW, Rao DV. Vitamins as radioprotectors in vivo. II. Protection by vitamin A and soybean oil against radiation damage caused by internal radionuclides. Radiat Res. 1994;139:115–122. doi: 10.2307/3578741. [DOI] [PubMed] [Google Scholar]
- 116.Safaei M, Jafarpour SM, Mohseni M, Salimian M, Akbari H, Karami F, Aliasgharzadeh A, Farhood B. Vitamins E and C prevent DNA double-strand breaks in peripheral lymphocytes exposed to radiations from iodine-131. Indian J Nucl Med. 2018;33:20–24. doi: 10.4103/ijnm.IJNM_89_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Narra VR, Howell RW, Sastry KS, Rao DV. Vitamin C as a radioprotector against iodine-131 in vivo. J Nucl Med. 1993;34:637–640. [PubMed] [Google Scholar]
- 118.Srinivasan V, Weiss JF. Radioprotection by vitamin E: Injectable vitamin E administered alone or with WR-3689 enhances survival of irradiated mice. Int J Radiat Oncol Biol Phys. 1992;23:841–845. doi: 10.1016/0360-3016(92)90657-4. [DOI] [PubMed] [Google Scholar]
- 119.Bagheri H, Salajegheh A, Javadi A, Amini P, Shekarchi B, Shabeeb D, Eleojo Musa A, Najafi M. Radioprotective effects of zinc and selenium on mice spermatogenesis. J Biomed Phys Eng. 2020;10:707–712. doi: 10.31661/jbpe.v0i0.957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Liang S, Jin YX, Yuan B, Zhang JB, Kim NH. Melatonin enhances the developmental competence of porcine somatic cell nuclear transfer embryos by preventing DNA damage induced by oxidative stress. Sci Rep. 2017;7:11114. doi: 10.1038/s41598-017-11161-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mao A, Guo H, Liu Y, Wang F, Tang J, Liao S, Zhang Y, Sun C, Xia X, Zhang H. Exogenous melatonin modulates carbon ion radiation-induced immune dysfunction in mice. Toxicology. 2019;417:35–41. doi: 10.1016/j.tox.2019.01.019. [DOI] [PubMed] [Google Scholar]
- 122.Shibai A, Satoh K, Kawada M, Kotani H, Narumi I, Furusawa C. Complete genome sequence of a radioresistant bacterial strain, Deinococcus grandis ATCC 43672. Microbiol Resour Announc. 2019;8:e01226–19. doi: 10.1128/MRA.01226-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lin SM, Baek CY, Jung JH, Kim WS, Song HY, Lee JH, Ji HJ, Zhi Y, Kang BS, Bahn YS, et al. Antioxidant activities of an exopolysaccharide (DeinoPol) produced by the extreme radiation-resistant bacterium Deinococcus radiodurans. Sci Rep. 2020;10:55. doi: 10.1038/s41598-019-56141-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Riehl TE, Alvarado D, Ee X, Zuckerman A, Foster L, Kapoor V, Thotala D, Ciorba MA, Stenson WF. Lactobacillus rhamnosus GG protects the intestinal epithelium from radiation injury through release of lipoteichoic acid, macrophage activation and the migration of mesenchymal stem cells. Gut. 2019;68:1003–1013. doi: 10.1136/gutjnl-2018-316226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Guo H, Chou WC, Lai Y, Liang K, Tam JW, Brickey WJ, Chen L, Montgomery ND, Li X, Bohannon LM, et al. Multi-omics analyses of radiation survivors identify radioprotective microbes and metabolites. Science. 2020;370:eaay9097. doi: 10.1126/science.aay9097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Akeem S, Lukman O, Eltahir K, Fatai O, Abiola B, Khadijat O. Bone marrow and peripheral blood cells toxicity of a single 2.0 Gy Cobalt60 ionizing radiation: An animal model. Ethiop J Health Sci. 2019;29:195–202. doi: 10.4314/ejhs.v29i2.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Chin SP, Mohd-Shahrizal MY, Liyana MZ, Then KY, Cheong SK. High dose of intravenous allogeneic umbilical cord-derived mesenchymal stem cells (CLV-100) infusion displays better immunomodulatory effect among healthy volunteers: A phase 1 clinical study. Stem Cells Int. 2020;2020:8877003. doi: 10.1155/2020/8877003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Lykov AP, Bondarenko NA, Poveshchenko OV, Kabakov AV, Surovtseva MA, Kim II, Kazakov OV, Poveshchenko AF, Iankaĭte EV. Therapeutic potential of a biomedical cellular product in rats with lower limb ischaemia. Angiol Sosud Khir. 2020;26:37–43. doi: 10.33529/ANGIO2020315. (In Russian) [DOI] [PubMed] [Google Scholar]
- 129.DiCarlo AL, Horta ZP, Aldrich JT, Jakubowski AA, Skinner WK, Case CM., Jr Use of growth factors and other cytokines for treatment of injuries during a radiation public health emergency. Radiat Res. 2019;192:99–120. doi: 10.1667/RR15363.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Pourgholaminejad A, Aghdami N, Baharvand H, Moazzeni SM. The effect of pro-inflammatory cytokines on immunophenotype, differentiation capacity and immunomodulatory functions of human mesenchymal stem cells. Cytokine. 2016;85:51–60. doi: 10.1016/j.cyto.2016.06.003. [DOI] [PubMed] [Google Scholar]
- 131.Qiao S, Ren H, Shi Y, Liu W. Allogeneic compact bone-derived mesenchymal stem cell transplantation increases survival of mice exposed to lethal total body irradiation: A potential immunological mechanism. Chin Med J (Engl) 2014;127:475–482. [PubMed] [Google Scholar]
- 132.Nevens F, van der Merwe S. Mesenchymal stem cell transplantation in liver diseases. Semin Liver Dis. 2022;42:283–292. doi: 10.1055/s-0042-1755328. [DOI] [PubMed] [Google Scholar]
- 133.Liu FD, Tam K, Pishesha N, Poon Z, Van Vliet KJ. Improving hematopoietic recovery through modeling and modulation of the mesenchymal stromal cell secretome. Stem Cell Res Ther. 2018;9:268. doi: 10.1186/s13287-018-0982-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Court AC, Le-Gatt A, Luz-Crawford P, Parra E, Aliaga-Tobar V, Bátiz LF, Contreras RA, Ortúzar MI, Kurte M, Elizondo-Vega R, et al. Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response. EMBO Rep. 2020;21:e48052. doi: 10.15252/embr.201948052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Haarer J, Johnson CL, Soeder Y, Dahlke MH. Caveats of mesenchymal stem cell therapy in solid organ transplantation. Transpl Int. 2015;28:1–9. doi: 10.1111/tri.12415. [DOI] [PubMed] [Google Scholar]
- 136.Jo H, Brito S, Kwak BM, Park S, Lee MG, Bin BH. Applications of mesenchymal stem cells in skin regeneration and rejuvenation. Int J Mol Sci. 2021;2:2410. doi: 10.3390/ijms22052410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Huang YZ, Gou M, Da LC, Zhang WQ, Xie HQ. Mesenchymal stem cells for chronic wound healing: Current status of preclinical and clinical studies. Tissue Eng Part B Rev. 2020;26:555–570. doi: 10.1089/ten.teb.2019.0351. [DOI] [PubMed] [Google Scholar]
- 138.Benderitter M, Herrera-Reyes E, Tamarat R. Mesenchymal stromal cells in the regeneration of radiation-induced organ sequelae: Will they make the difference? J Radiol Prot. 2022;42 doi: 10.1088/1361-6498/ac6dd8. [DOI] [PubMed] [Google Scholar]
- 139.Sun J, Zhang Y, Song X, Zhu J, Zhu Q. The healing effects of conditioned medium derived from mesenchymal stem cells on radiation-induced skin wounds in rats. Cell Transplant. 2019;28:105–115. doi: 10.1177/0963689718807410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Dao A, McDonald MM, Savage PB, Little DG, Schindeler A. Preventing osteolytic lesions and osteomyelitis in multiple myeloma. J Bone Oncol. 2022;37:100460. doi: 10.1016/j.jbo.2022.100460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Jiang X, Yang J, Liu F, Tao J, Xu J, Zhang M. Embryonic stem cell-derived mesenchymal stem cells alleviate skeletal muscle injury induced by acute compartment syndrome. Stem Cell Res Ther. 2022;13:313. doi: 10.1186/s13287-022-03000-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Rahyussalim AJ, Nasser MK, Al As'ady FM, Kurniawati T. Umbilical cord-derived mesenchymal stem cells implantation on Hemivertebra defect with three-year follow-up: Biological approach in congenital scoliosis treatment-a case report. Int J Surg Case Rep. 2022;99:107602. doi: 10.1016/j.ijscr.2022.107602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Fujii S, Miura Y. Immunomodulatory and regenerative effects of MSC-derived extracellular vesicles to treat acute GVHD. Stem Cells. 2022;40:977–990. doi: 10.1093/stmcls/sxac057. [DOI] [PubMed] [Google Scholar]
- 144.Gong W, Guo M, Han Z, Wang Y, Yang P, Xu C, Wang Q, Du L, Li Q, Zhao H, et al. Mesenchymal stem cells stimulate intestinal stem cells to repair radiation-induced intestinal injury. Cell Death Dis. 2016;7:e2387. doi: 10.1038/cddis.2016.276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Hou G, Li J, Liu W, Wei J, Xin Y, Jiang X. Mesenchymal stem cells in radiation-induced lung injury: From mechanisms to therapeutic potential. Front Cell Dev Biol. 2022;10:1100305. doi: 10.3389/fcell.2022.1100305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Leclerc T, Thepenier C, Jault P, Bey E, Peltzer J, Trouillas M, Duhamel P, Bargues L, Prat M, Bonderriter M, Lataillade JJ. Cell therapy of burns. Cell Prolif. 2011;44((Suppl 1)):S48–S54. doi: 10.1111/j.1365-2184.2010.00727.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Maleki M, Ghanbarvand F, Reza Behvarz M, Ejtemaei M, Ghadirkhomi E. Comparison of mesenchymal stem cell markers in multiple human adult stem cells. Int J Stem Cells. 2014;7:118–126. doi: 10.15283/ijsc.2014.7.2.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Lin H, Ouyang H, Zhu J, Huang S, Liu Z, Chen S, Cao G, Li G, Signer RA, Xu Y, et al. Lens regeneration using endogenous stem cells with gain of visual function. Nature. 2016;531:323–328. doi: 10.1038/nature17181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Xing Y, Li B, He J, Hua H. Labial gland mesenchymal stem cell derived exosomes-mediated miRNA-125b attenuates experimental Sjogren's syndrome by targeting PRDM1 and suppressing plasma cells. Front Immunol. 2022;13:871096. doi: 10.3389/fimmu.2022.871096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Sun T, Liu S, Yang G, Zhu R, Li Z, Yao G, Chen H, Sun L. Mesenchymal stem cell transplantation alleviates Sjögren's syndrome symptoms by modulating Tim-3 expression. Int Immunopharmacol. 2022;111:109152. doi: 10.1016/j.intimp.2022.109152. [DOI] [PubMed] [Google Scholar]
- 151.Jansson PM, Lynggaard CD, Carlander AF, Jensen SB, Follin B, Hoeeg C, Kousholt BS, Larsen RT, Grønhøj C, Jakobsen KK, et al. Mesenchymal stromal/stem cell therapy for radiation-induced salivary gland hypofunction in animal models: A protocol for a systematic review and meta-analysis. Syst Rev. 2022;11:72. doi: 10.1186/s13643-022-01943-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Lynggaard CD, Grønhøj C, Jensen SB, Christensen R, Specht L, Andersen E, Andersen TT, Ciochon UM, Rathje GS, Hansen AE, et al. Long-term safety of treatment with autologous mesenchymal stem cells in patients with radiation-induced xerostomia: Primary results of the MESRIX phase I/II randomized trial. Clin Cancer Res. 2022;28:2890–2897. doi: 10.1158/1078-0432.CCR-21-4520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Hérodin F, Drouet M. Cytokine-based treatment of accidentally irradiated victims and new approaches. Exp Hematol. 2005;33:1071–1080. doi: 10.1016/j.exphem.2005.04.007. [DOI] [PubMed] [Google Scholar]
- 154.Peng Y, Wu S, Tang Q, Li S, Peng C. KGF-1 accelerates wound contraction through the TGF-β1/Smad signaling pathway in a double-paracrine manner. J Biol Chem. 2019;294:8361–8370. doi: 10.1074/jbc.RA118.006189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Chen Y, Rubin P, Williams J, Hernady E, Smudzin T, Okunieff P. Circulating IL-6 as a predictor of radiation pneumonitis. Int J Radiat Oncol Biol Phys. 2001;49:641–648. doi: 10.1016/S0360-3016(00)01445-0. [DOI] [PubMed] [Google Scholar]
- 156.Damm R, Pech M, Haag F, Cavalli P, Gylstorff S, Omari J, Seidensticker R, Ricke J, Seidensticker M, Relja B. TNF-α indicates radiation-induced liver injury after interstitial high dose-rate brachytherapy. In Vivo. 2022;36:2265–2274. doi: 10.21873/invivo.12955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Wang J, Xu Z, Wang Z, Du G, Lun L. Corrigendum to ‘TGF-beta signaling in cancer radiotherapy’ [Cytokine 148 (2021) 155709] Cytokine. 2022;149:155753. doi: 10.1016/j.cyto.2021.155753. [DOI] [PubMed] [Google Scholar]
- 158.Lee S, Ki CS. Inflammatory responses of macrophage-like RAW264.7 cells in a 3D hydrogel matrix to ultrasonicated schizophyllan. Carbohydr Polym. 2020;229:115555. doi: 10.1016/j.carbpol.2019.115555. [DOI] [PubMed] [Google Scholar]
- 159.Moorlag SJCFM, Khan N, Novakovic B, Kaufmann E, Jansen T, van Crevel R, Divangahi M, Netea MG. β-Glucan induces protective trained immunity against mycobacterium tuberculosis infection: A key role for IL-1. Cell Rep. 2020;31:107634. doi: 10.1016/j.celrep.2020.107634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Song JY, Han SK, Bae KG, Lim DS, Son SJ, Jung IS, Yi SY, Yun YS. Radioprotective effects of ginsan, an immunomodulator. Radiat Res. 2003;159:768–774. doi: 10.1667/0033-7587(2003)159[0768:REOGAI]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 161.Wu T, Liu W, Fan T, Zhong H, Zhou H, Guo W, Zhu X. 5-Androstenediol prevents radiation injury in mice by promoting NF-κB signaling and inhibiting AIM2 inflammasome activation. Biomed Pharmacother. 2020;121:109597. doi: 10.1016/j.biopha.2019.109597. [DOI] [PubMed] [Google Scholar]
- 162.Everett WH, Curiel DT. Gene therapy for radioprotection. Cancer Gene Ther. 2015;22:172–180. doi: 10.1038/cgt.2015.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Khalifa J, François S, Rancoule C, Riccobono D, Magné N, Drouet M, Chargari C. Gene therapy and cell therapy for the management of radiation damages to healthy tissues: Rationale and early results. Cancer Radiother. 2019;23:449–465. doi: 10.1016/j.canrad.2019.07.002. [DOI] [PubMed] [Google Scholar]
- 164.Maier P, Veldwijk MR, Wenz F. Radioprotective gene therapy. Expert Opin Biol Ther. 2011;11:1135–1151. doi: 10.1517/14712598.2011.580271. [DOI] [PubMed] [Google Scholar]
- 165.Zhang Z, Lang J, Cao Z, Li R, Wang X, Wang W. Radiation-induced SOD2 overexpression sensitizes colorectal cancer to radiation while protecting normal tissue. Oncotarget. 2017;8:7791–7800. doi: 10.18632/oncotarget.13954. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used and/or analyzed during the current study are available from the corresponding author on reasonable request.