Abstract
Objectives:
Efforts to conceptualize risk factors for postoperative delirium in older adults have focused on the time proximate to the episode, but how early-life exposures influence delirium risk is poorly understood.
Methods:
An observational cohort of 547 patients aged 70+ undergoing major non-cardiac surgery at two academic medical centers in Boston. Demographic characteristics, cognition, parental education, health, and participation in cognitively stimulating activities were assessed prior to surgery. Delirium incidence and severity were measured daily during hospitalization.
Results:
Higher paternal education was associated with significantly lower incidence of delirium (X2(1, N=547)= 8.35, p<.001; odds ratio OR= .93, 95% CI, .87 to .98) and inversely associated with delirium severity (r(545)= −.13, p<.001). Higher maternal education was associated with lower delirium incidence but did not reach statistical significance. The effect of paternal education on delirium incidence was independent of the patient’s education, estimated premorbid intelligence, medical comorbidities, neighborhood disadvantage, and participation in cognitively stimulating activities (X2(2, N=547)= 31.22, p<.001).
Conclusions:
Examining early-life exposures may yield unique insights into the risks and pathogenesis of delirium.
Clinical Implications:
Evaluating long-term factors that increase vulnerability to delirium may improve our ability to calculate risk. It may guide clinical decision-making and inform pre- and post-operative recommendations.
Keywords: childhood socioeconomic status, delirium, social determinants of health
INTRODUCTION
Delirium is a common postoperative complication occurring in up to 65% of older adults undergoing selected surgeries (Marcantanio, 2017) and is associated with increased costs (Gou et al., 2021), adverse health outcomes, and increased mortality (Inouye et al., 2011; Jankowski et al., 2011). Delirium is a highly preventable condition (Hamrick & Meyer, 2013), and early detection and treatment may mitigate delirium-related adverse outcomes (McCusker et al., 2002). In efforts to modify risk and to mitigate negative sequelae, delirium research has predominantly incorporated immediate effect lifecourse models (Liu et al., 2013). Most studies have examined risk factors that are proximate to delirium incidence, including cognitive impairment, frailty, preexisting medical comorbidities, and sensory impairment (Inouye et al., 2014; McCusker et al., 2013).
Social and historical factors interact with psychological and biological predispositions, particularly during critical and sensitive periods in childhood, to influence general and cognitive health later in life (Keage et al., 2016; Rogers et al., 2009; Seeman et al., 2010). Low childhood socioeconomic status (low SES) has been linked to adverse childhood events, including maltreatment, malnourishment, as well as exposure to environmental hazards and family dysfunction (Felitti et al., 1998; Halfon et a., 2017; Rogers et al., 2015), which has, in turn, emerged as predictors of social mobility, health, and emotional well-being in adulthood. Parental education, a well-established marker of childhood SES, has been associated with signs of biological aging (Cuevas et al., 2021) and absolute cognitive functioning in adulthood (Luo & Waite, 2005; Everson-Rose et al., 2003).
Research exploring the association between early-life exposure and delirium is scarce, but efforts are underway to fill this knowledge gap (Arias et al., 2022; Wu et al., 2021). We have recently identified that residing in the most disadvantaged neighborhoods (area deprivation index, ADI > 44) is associated with a higher risk of incident delirium and severity (Arias et al., 2020). This may be because individual-level vulnerabilities to disease (e.g., genetic predisposition, age) act in synergy with individual-level factors to affect the central nervous system’s ability to withstand insult. In another United States (US) -based study, Davoudi and colleagues (2017) used preoperative electronic health records to predict postoperative delirium. They found that socioeconomic status, a well-established social determinant of health (SDOH), and surgeon characteristics, along with other known individual-level risk factors (e.g., age, diagnosed substance use, and complexity of the person’s medical history) were important predictors of delirium (Davoudi et al., 2017). Neuronal aging, neurotransmitter imbalance, systemic inflammation, and dysregulation in the endocrine system, pathophysiological mechanisms involved in delirium (Kazmierski et al., 2013; Mulkey et al., 2018; Westhoff et al., 2013), are also affected by SDOH factors, particularly in older adults (Northwood, Ploeg, Markle-Reid, & Sherifali, 2018), and may explain the association between SDOH factors and delirium incidence. The cognitive reserve hypothesis (Arenaza-Urquijo et al., 2020; Satz, 1993), which poses that exposure to risk and protective factors throughout the lifespan influence’s the central nervous system’s ability to cope with insult, provides a rationale for examining the association between delirium and early-life exposure (Jones et al., 2006). In fact, neuroimaging and biomarker studies on delirium (Cavallari et al., 2017; Fong et al., 2020) suggest that central nervous systems vulnerabilities are present in persons at risk for delirium even in the absence of frank cognitive impairment. Thus, characterizing the associations between SDOH on acute cognitive changes is an important step in advancing delirium science and elucidating how brain vulnerability to acute changes in cognition may alter long-term cognitive trajectories in older adults.
Examining the contribution of early-life exposures on cognitive health in older adults is also pertinent to the work with patients from underrepresented groups. Non-Hispanic Black and Hispanic/Latino(a) older adults are at greater risk of developing Alzheimer’s disease and related dementias (ADRD) (Mehta et al., 2017). Moreover, Non-Hispanic Black and Hispanic/Latino(a) older adults are more likely than their Non-Hispanic White counterparts to develop postoperative complications (Brooks et al., 2013; Azin et al., 2020). Unfortunately, rates of acute cognitive changes have not been systematically reported in the context of SDOH factors. A PubMed search expanding from 2000 to 2022 using the terms “delirium” and “social determinants of health” or “race” or “socioeconomic status” identified 54 peer-reviewed manuscripts in English. Of those, less than 25% were empirical studies examining the association between delirium and race, socioeconomic, and contextual factors in older adults in medical settings in the United States (Arias et al., 2020; Bilotta, Lauretta, Borozdina, Mizikov, & Rosa, 2013; Campbell et al., 2014; Haddad et al., 2020; Kagansky et al., 2004; Khan et al., 2016). This is a significant gap in this research given that ethnoracially and socioeconomically underrepresented groups experience high rates of poverty. Twenty-four and 28% of Non-Hispanic Black and Hispanic/Latino(a) families, respectively, experienced poverty in 2019 (Creamer, 2020). Finally, transgenerational trauma experiences and psychological distress are prevalent in persons from socially and culturally -marginalized groups (Matheson et al., 2019; Phipps & Degges-White, 2014).
We examined whether parental education was related to delirium incidence and severity. Additionally, we explored whether these associations were explained by cognitive performance, medical comorbidities, and participant’s education at baseline, characteristics known to increase delirium risk. We hypothesized that paternal and maternal education would predict delirium incidence independent of other cognitive reserve markers, medical comorbidities, and neighborhood-level characteristics. Overall, paternal education is an important predictor of educational attainment and epigenetic aging (Joyce et al., 2021; Meng & Groth, 2018; Keage et al., 2016) in adulthood. In addition, maternal education has been associated with global cognitive performance (Vable et al., 2018) and offspring mortality (Myrskylä, Elo, Kohler, & Martikainen, 2014) in older adults. In some studies, the association between maternal education and cognition in adulthood persists even after controlling for paternal education (Rogers et al., 2009). In light of the evidence suggesting that paternal and maternal education may influence health in older adulthood via independent pathways, our goal was to explore these associations independently. We believe that given gender roles and acquisition of power differences between men and women in the US, the relationship between paternal education and delirium incidence will be independent of the association between maternal education and delirium incidence.
METHODS
Design
This is a prospective observational cohort of patients enrolled between June 18, 2010 and August 8, 2013. Baseline interviews were conducted face-to-face in participants’ homes prior to surgery. Variables collected at baseline or baseline characteristics included the participants’ demographic characteristics, cognitive functioning, and daily functioning. Medical comorbidities and details about the surgery were collected via medical record review. During hospitalization, participants were evaluated for delirium daily. Study procedures were completed by research associates who received intensive initial training with semiannual inter-rater reliability checks (Schmitt et al., 2015).
Setting and Participants
This paper includes a subset of the patients enrolled in the Successful Aging after Elective Surgery (SAGES; Schmitt et al., 2015) study (N=560). Specifically, patients who were missing data on the self-report scale of lifetime cognitive activities (Cognitive Activities Scale; CAS; n=3; 1%) and parental education (n=10; 2%) were excluded from this analysis, and our overall sample size was 547 (Wilson et al., 2003). As described elsewhere (Schmitt et al., 2015), eligible participants were 70+ years of age, English speaking, scheduled to undergo major elective surgery at one of two Harvard-affiliated academic medical centers, and with an anticipated length of stay of at least three days. Exclusion criteria included evidence of dementia or baseline education-adjusted Modified Mini-Mental State (3MS) examination score ≤ 69 (Teng & Chui, 1987), a history of delirium, hospitalization within three months, terminal condition, legal blindness, severe deafness, and history of schizophrenia, psychosis, or alcohol abuse. Written informed consent was obtained following procedures approved by the institutional review boards of Beth Israel Deaconess Medical Center and Brigham and Women’s Hospital, the two study hospitals, and Hebrew SeniorLife, the study coordinating center.
Measurements
Paternal and maternal education levels were assessed via a face-to-face interview one month after surgery. Patients were asked to select the highest grade of formal education completed by his/her parents, from 0 to 20+ years. Parental education levels were considered as indicators of childhood SES (Baird et al., 2017; Galobardes et al., 2006).
The delirium assessment which was conducted daily until discharge, included a brief cognitive screen (i.e., orientation, short-term recall, attention tasks) and an abbreviated Delirium Symptom Interview [DSI] (Albert et al., 1992). Family members and hospital staff were interviewed to assess acute changes in alertness or cognitive function. In addition, the information collected via the DSI was used to score the Confusion Assessment Method [CAM] long-form (Inouye et al., 1990), a standardized approach to identify delirium with high sensitivity (94%–100%), high specificity (90%–95%), and high inter-rater reliability [kappa statistic 0.92 in 71 paired ratings] (Inouye et al., 1990; Inouye et al., 2016). Patients were classified as delirious if they met criteria based on the CAM or via a well-validated chart review process (Inouye et al., 2014; Saczynski et al., 2014). The CAM Severity score (CAM-S), scored 0 to 19 (higher scores indicate more severe delirium), was calculated daily using the CAM scores (Inouye et al., 2014). For this study, delirium severity was defined according to the highest CAM-S (CAM-S Peak) value during hospitalization. Research staff scoring the CAM long-form were trained and supervised by a team of experts, including a seasoned geriatrician and a delirium expert (ERM). The team conducting inpatient assessments was independent of the team collecting baseline and the follow-up assessments (Schmitt et al., 2005).
Individual variables included demographics, cognition, and comorbidity. Demographic variables included age, sex, race, educational level, and living situation. Cognitive functioning was assessed with a comprehensive neuropsychological protocol that included measures of premorbid functioning, attention, processing speed, mental flexibility, language fluency, confrontation naming, and verbal learning and memory (Fong et al., 2015) and yielded a weighted composite score, the General Cognitive Performance (GCP) score (Jones et al., 2010), calibrated to have a mean of 50 and a standard deviation of 10 (Gross et al., 2014). Estimated premorbid intelligence was assessed using the Wechsler Test of Adult Reading [WTAR] (Green et al., 2008). The total score was transformed to an age-adjusted standard score (mean of 100, standard deviation of 10). The cognitive activities scale (CAS), collected at a face-to-face interview one month after surgery, was used to assess participation in 25 cognitively stimulating activities at four time-points: 12, 18, and 40 years of age and at the time of the interview (Wilson et al., 2003; Wilson et al., 2002). Responses from 12 to 18 years of age were averaged to calculate early-life CAS scores; responses from 40 years were used for mid-life CAS scores, and current responses were used for late-life CAS scores. The CAS Total Score assessed patients’ participation in each cognitively stimulating activity. The scale ranged from 0 (never participated in the activity) to 5 (participated almost every day), with higher scores indicating increased participation. The presence of comorbid medical conditions was rated with the Charlson Comorbidity Index, scored 0 to 35 (35= highest) (Charlson et al., 1987). Neighborhood-level characteristics were evaluated using the Area Deprivation Index (ADI) (Kind et al., 2014; Singh et al., 2003) using the location of the patient’s reported primary residence reported at baseline. The ADI, which ranges from 1 to 100 (100= most disadvantaged), was developed using 17 US census indicators to measure the level of disadvantage of the local neighborhood.
Statistical Analyses
Potential risk factors associated with delirium incidence were examined in multivariate logistic regression models. We evaluated whether paternal education predicted delirium incidence and whether this association remained robust after other individual and neighborhood-level variables were incorporated into the model separately (series models) and then together (full model). Model fit and predictive power were assessed using Nagelkerke R2, (Nagelkerke et al., 1981) and Pearson’s chi-square test (Hosmer et al., 1997), respectively. We present standardized regression beta coefficients such that each beta coefficient expresses the difference in log odds of delirium corresponding to one standard deviation difference in the predictor. Spearman’s correlations were used to examine bivariate associations between parental education and markers of cognitive reserve. Linear regressions were used to assess the association between parental education with delirium severity (Figure 1 and Figure 2). All analyses were performed using R, version 3.6.1 (Team, 2013). The a priori two-tailed alpha was .05, and all variables were checked for skewness and normality.
Figure 1. Association between Paternal Education and Delirium Incidence in the Sample (N=547).

The graph illustrates results from a logistic regression model (black line) and 95% confidence intervals (grey area), which were calculated using a standard formula (mean ±1.96 * SE). The X-axis reflects paternal education in years, with black lines along the X-axis reflecting the number of cases on each year of education reported. The Y-axis reflects the probability of delirium risk (N=547). *
Figure 2. Association between Paternal Education and Delirium Severity in the Sample (N=547).
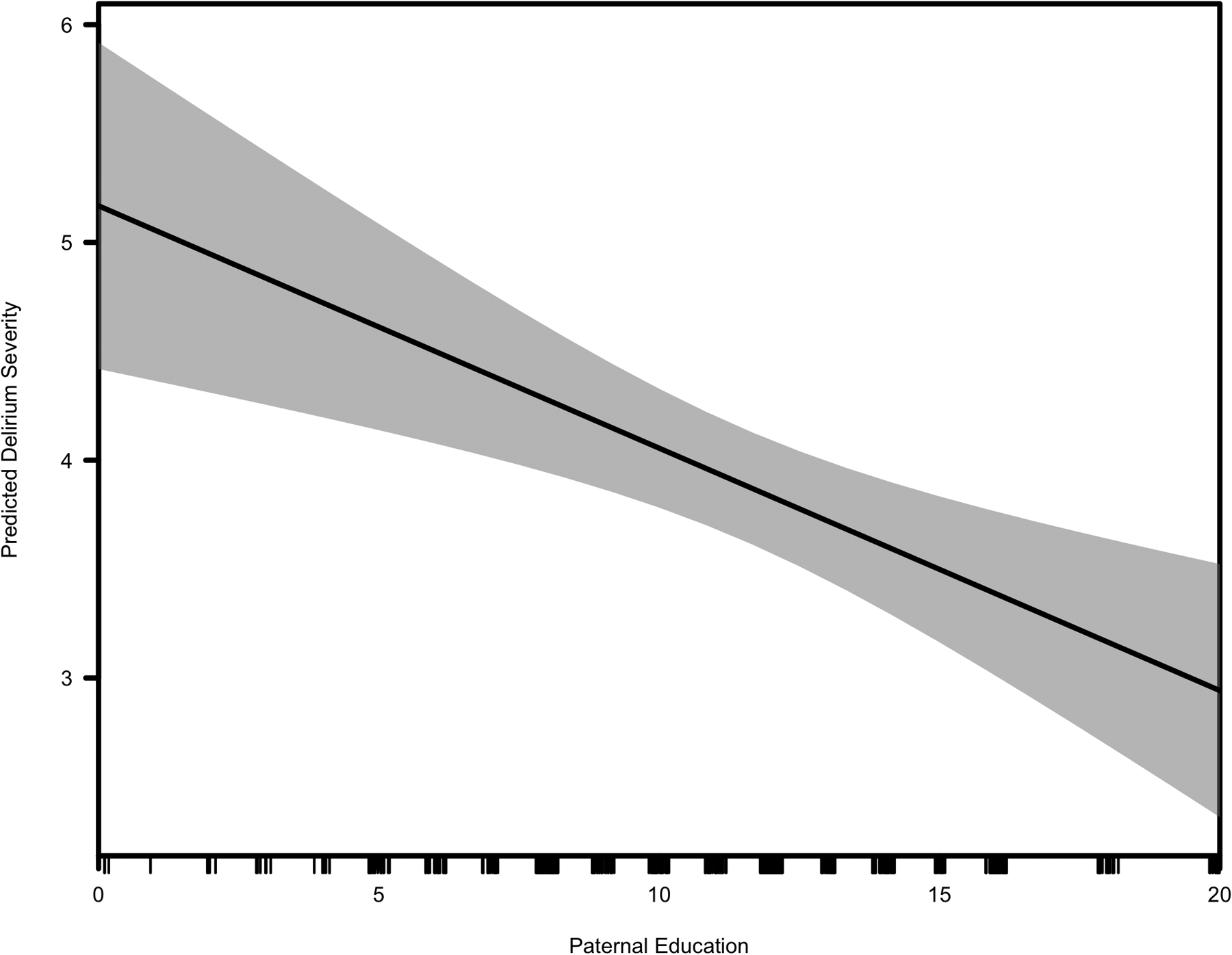
The graph illustrates results from a linear regression model (black line) and 95% confidence intervals (grey area), which were calculated using a standard formula (mean ±1.96 * SE). The X-axis reflects paternal education in years, with black lines along the X-axis reflecting the number of cases on each year of education reported. The Y-axis reflects delirium severity (N=547).
RESULTS
Cohort Characteristics
Table 1 presents the demographic and clinical characteristics of our sample (N=547). Participants were, on average, 76.6 (SD=5.1) years of age [mean (SD)], married (58%), and living with others (70%). At the onset of the study, the sample was cognitively unimpaired, 3MS mean score of 93.5 (SD=5.2). Incident postoperative delirium occurred in 23% of the sample, and the average peak CAM-S score was 8.1 (SD= 3.6) in those with delirium and 2.6 (SD=1.6) in those without delirium.
Table 1.
Baseline Characteristics of the Sample and Parental Education (N= 547)*
| Entire Sample | Delirium (N=128) | No-Delirium (N=419) | |
|---|---|---|---|
| Age in years, mean (SD) | 76.6 (5.1) | 77.3 (4.8) | 76.4 (5.2) |
| Female, N (%) | 318 (58) | 78 (61) | 240 (57) |
| Non-white, N (%) | 41 (7) | 13 (10) | 28 (7) |
| Participant education in years, mean (SD) | 14.9 (2.9) | 14.7 (2.9) | 15.1 (2.9) |
| WTAR, mean (SD) | 37.9 (9.9) | 35.7 (9.9) | 38.6 (9.8) |
| GCP, mean (SD) | 57.8 (7.2) | 54.9 (6.3) | 58.6 (7.2) |
| CAS, mean (SD) | |||
| Early-life CAS | 49.9 (8.9) | 48.8 (8.8) | 50.2 (9.0) |
| Mid-life CAS | 49.7 (10.1) | 50.9 (10.0) | 49.4 (10.2) |
| Late-life CAS | 49.9 (9.9) | 48.9 (9.2) | 50.2 (10.1) |
| CCI, mean (SD) | 1 (1.3) | 1.3 (1.4) | 1.0 (1.2) |
| ADI National Percentile, mean (SD) | 16 (14.6) | 18.8 (19.1) | 15.1 (12.9) |
| Father’s Education in years, mean (SD) | 11.5 (4.2) | 10.5 (4.3) | 11.8 (4.1) |
| Mother’s Education in years, mean (SD) | 11.2 (3.6) | 10.9 (3.8) | 11.2 (3.6) |
SD- Standard Deviation; WTAR- Wechsler Test of Adult Reading, scored 0–50, highest scores= better performance; GCP- General Cognitive Performance, scored 0–50, highest scores= better performance; CAS- Cognitive Activity Scale, scored 0–5, highest score= more frequent participation in an activity; Early-life CAS- Average of CAS at 12 years old and CAS at 18 years old; CCI- Charlson Comorbidity Index, scored 0–35, highest scores= increased comorbidity; ADI- Area Deprivation Index, scored 0–100, highest scores= increased neighborhood-level disadvantage.
Parental Education and Delirium
Paternal and maternal education (0 to 20 years) were normally distributed, with means of 11.5 (SD=4.2) and 11.2 (SD=3.6), respectively. Paternal and maternal education were strongly associated with r(545)=.53, p<.001. Both paternal and maternal education were associated with patient’s education r(545)=.38; r(545)=.36, p<.001. Paternal but not maternal education was a significant predictor of incident delirium (Figure 1), with the probability of delirium decreasing as paternal education increased.
Paternal education was also inversely and significantly associated with delirium severity r(545)=−.13, p<.001. That is, the delirium severity decreased as paternal education increased (Figure 2). While maternal education was also inversely associated with delirium severity, that difference did not reach statistical significance r(545)=−.06, p=.181.
Logistic regression models (Table 2) found that paternal education predicted incident delirium (X2(1, N=547)= 8.98, p<.001). Model fit improved when WTAR (X2(2)= 13.32, p<.001), GCP (X2(2, N=547)= 31.22, p<.001), CCI (X2(2, N=547)= 14.17, p<.001), and ADI (X2(2, N=547)= 13.2, p<.001) were added individually, but not with addition of CAS Total Score. A multivariate logistic regression model, which incorporated individual, neighborhood-level factors, paternal education, and GCP was statistically significant predicting delirium incidence (X2(7, N=547)= 38.91, p<.001), and explained 10% of the variance in delirium incidence (Nagelkerke R2).
Table 2.
Logistic Regression Models for Delirium Incidence using Standardized Beta Coefficients (N= 547)§
| Predictors | Bivariable Models | Series Models B | Full Model | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Intercept | NA | −0.39 | −1.21*** | −1.22*** | −1.23*** | −1.21*** | −1.27*** | −1.22*** | −1.29*** |
| Paternal Education | −0.07*** | −0.07*** | −0.29** | −0.24** | −0.28*** | −0.31*** | −0.22* | −0.29*** | −0.28** |
| Participant’s Education | −0.05 | -- | −0.02 | -- | -- | -- | -- | -- | 0.17 |
| WTAR | −0.03*** | -- | -- | −0.21** | -- | -- | -- | -- | −0.06 |
| ADI National Percentile | 0.02** | -- | -- | -- | 0.19** | -- | -- | -- | 0.15 |
| CAS Total Score | −0.07 | -- | -- | -- | -- | 0.02 | -- | -- | 0.12 |
| GCP | −0.07*** | -- | -- | -- | -- | -- | −0. 49*** | -- | −0.51*** |
| CCI | 0.19** | -- | -- | -- | -- | -- | -- | 0.22** | 0.16 |
| R2 | NA | 0.02** | 0.02* | 0.04** | 0.03** | 0.02* | 0.08*** | 0.04** | 0.10*** |
WTAR- Wechsler Test of Adult Reading; ADI- Area Deprivation Index; Total CAS- Cognitive Activity Scale; GCP- General Cognitive Performance; CCI- Charlson Comorbidity Index; Standardized Beta Coefficients- Beta coefficients in standard deviation units that are used to compare the effect of each independent variables on the dependent variable.
P < .001,
P < .01,
P < .05
Maternal education did not emerge as a predictor of incident delirium (X2(1, N=547)= 1.87, p=.392) and its predictive utility did not improve when WTAR (X2(2, N=547)= 8.12, p=.017), GCP (X2(2, N=547)= 26.59, p<.001), CCI (X2(2, N=547)= 6.51, p=.038), and ADI (X2(2, N=547)= 5.85, p=.053) were added individually.
Parental Education and Markers of Cognitive Reserve
Paternal education was associated with WTAR, (r(545)= 0.30, p<.001); CAS Total Score, (r(545)= 0.26, p<.001); and GCP (r(545)=0.14, p=.002), and inversely associated with CCI, (r(545)= −0.10, p=.016) and ADI national percentiles (r(545)= −0.16, p<.001). Maternal education was positively associated with WTAR (r(545)= 0.30, p<.001); CAS Total Score, (r(545)= 0.23, p<.001); and GCP (r(545)= =0.16, p<.001), and negatively associated with ADI national percentiles, (r(545)=−0.17, p<.001).
DISCUSSION
In our study, delirium occurred in 23% of the sample, which is consistent with recent evidence suggesting that the global prevalence of postoperative delirium ranges from 17 to 24% (Abate, Checkole, Mantedafro, Basu, & Aynalem, 2021). Related to our hypothesis, we found that paternal education emerged as a significant predictor of delirium incidence, and higher paternal education resulted in lower rates of delirium and the association persisted independent of other risk factors for delirium, including patient’s education, premorbid intelligence, participation in cognitive activities across the lifecourse, medical comorbidities, and neighborhood-level characteristics. Paternal education was also inversely associated with delirium severity and suggested an exposure-response relationship. These findings are consistent with the growing body of literature on the association between early-life exposure and long-term health outcomes. Childhood socioeconomic status reflect aspects of a family’s access to financial, medical, and social resources and may be relevant to acute changes in cognition in patients without cognitive impairment. While maternal education was a predictor of delirium incidence, the relationship did not reach statistical significance. Our study is a novel demonstration of the potential importance early-life variables have on delirium incidence and severity.
Cognitive outcomes in adulthood (Aizer & Currie, 2014; Wilson & Shuey, 2019) have been associated with maternal education. In our study maternal education did not emerge as a significant predictor of delirium incidence or severity. Our findings are in line with previous studies suggesting that paternal education is associated with adverse health outcomes in adulthood independent of maternal characteristics (Meng & Groth, 2018; Luo & Waite, 2005). Social, historical, and economic factors have a powerful effect on women living in the US. Historically, women have faced limited access to education and have been uniquely impacted by generational changes in family structure and gender roles (Buckley, 2018). Participants in the SAGES cohort were at least 70 years old at the time of recruitment (~2010) and thus experienced their early childhood and adolescent years in the 1930s and 1940s. According to data from the US Department of Labor, 20% to 28% of women were employed outside the home between 1920 to 1940 (United States Department of Labor, 2020). Given these historical trends, paternal rather than maternal, education may have been a stronger influence on the family’s socioeconomic status.
In this analysis, participants’ education did not emerge as a significant predictor of delirium incidence. This finding is incongruent with other studies identifying educational level as a risk factor for delirium (Jones et al., 2006). Participants in the SAGES cohort are highly educated and are predominantly women. It is possible that there is not enough range in years of education or adequate representation in the low education group to capture differences between participants with high and low educational levels. Alternatively, it is plausible that the role of education as a protective factor is different in men than in women. To date, there is at least some evidence suggesting that education’s role in cognitive reserve differs across racial and ethnic groups (Avila et al., 2020).
Previous studies have suggested that early-life exposures, such as parental education, can influence late-life health outcomes via cognitive functioning (Turrell et al., 2002) or adult SES (Haas, 2008). In our sample, the effect of paternal education on delirium incidence persisted independently of cognitive function at baseline and adult SES. Thus, it is possible that paternal education is associated with delirium risk via pathways not systematically assessed in our study (e.g., nutrition during formative years, childhood illnesses). Additionally, other paternal characteristics may contribute to the association between paternal education and delirium incidence. Paternal mental health, physical health, and age have been found to be associated with offspring health later in life (Manios et al., 2007; Vervoort, Delger, & Soubry, 2021). For example, advanced parental age has been associated with offspring mortality (Myrskylä, Elo, Kohler, & Martikainen, 2014), and that association is, at least in part, mediated by early parental loss. It is worth highlighting that the association between paternal education may be explained by factors that preceded both paternal education and delirium and not necessarily related to childhood SES (e.g., familial predisposition to a given condition, shared exposure to environmental factors). Thus, causal association cannot be derived from this analysis. Collection of more detailed information on family history in clinical practice and research may provide a more complete representation of the overall risk profile for each patient.
A major strength of this study includes our large, well-characterized cohort contributing a wealth of clinical and demographic data, including parental education. The well-validated measures of delirium and delirium severity are also strengths. The availability of parental education, proxies of childhood SES, allowed us to take an innovative approach to investigating delirium. Our work has the potential to advance delirium and perioperative research by prompting investigators to consider the role of early-life factors in influencing delirium risk. These findings indicate that characteristics present long before surgery may render some patients more vulnerable to delirium and, more generally, to poor postoperative outcomes.
Several limitations of this study must be considered. Our cohort is well-educated, largely non-Hispanic White, from a discrete geographical area, and therefore generalizability to other samples will need to be evaluated. Education level for patients in other US-based studies have hanged from 7.7 (4.4) to 14.7 (3.4) years (Avila et al., 2021), with non-Hispanic White being significantly more educated than their Non-white counterparts (Shaw &. Spokane, 2008). The relationship between paternal education and delirium incidence was weak, as paternal education explained a small portion of the variance in delirium risk. Moreover, we have little information about the quality of education parents received or whether they were schooled in English and in the US. Years of education have traditionally predicted earning potential (Turner, Tamura, Mulholland, and Baier, 2007), however, evidence also suggests that the quantity of education alone does not fully capture the schooling experience (Manly, 2006). Inasmuch, access to material resources, participation in programs that provide remedial support, and interruptions in one’s academic trajectory are not accounted for by years of education (Behrman & Birdsall, 1983). In our study, it is possible that higher parental years of education did not necessarily translate into increased income, but that it instead captured exposures within the home environment (e.g., access to books, opportunities to participate in cognitively stimulating activities) that promote cognitive health. Having additional markers of parental income, education, and home characteristics would have allowed us to test our assumptions. Future studies which include such additional markers related to parental education are needed in order to conduct more refined analyses and identify other aspects of the early-life environment that might influence vulnerability to acute cognitive changes in adulthood.
Other studies examining childhood SES calculate composite SES measures including parental occupation and the offspring’s place of birth (Glymour et al., 2008). Information to calculate composite measures of childhood SES was not available for all participants in our cohort. Future studies examining cognitive health trajectories in older adults should assess parental characteristics (e.g., parental health, occupation, involvement in a patient’s life, and patient’s access to social support in childhood and adolescence) that may facilitate our ability to quantify early life exposure. Conducting nuanced analyses of parental characteristics would allow us to better quantify the influence of these factors on cognitive health and help to elucidate the mechanisms by which childhood SES is associated with acute and long-term cognitive changes.
While our cohort is homogenous in terms of demographic characteristics, aspects of our findings that paternal education is a significant predictor of delirium incidence are likely still relevant to persons from socioeconomically and ethnoracially underrepresented groups because of its close relationship to childhood SES. In persons from historically marginalized communities, the influence of low childhood SES on long-term cognitive health likely interacts with other social, systemic, and environmental stressors. Recent evidence suggests that cumulative stress, operationalized using composite constructs that included exposure to discrimination, stressful life events, and low socioeconomic status in adulthood, explains some of the disparities in cognitive functioning and health that exist between Black and Hispanic/Latino(a) with Non-Hispanic White persons in the US (Chen et al., 2022; Hoffmann, 2016; McClendon et al., 2021). Moreover, long-term exposure to certain environmental toxins, which are disproportionately represented in low-income neighborhoods (Ogunsola & Mehtar, 2020), have been linked to neurodegeneration in adulthood (Cerza et al., 2019; Agin et al., 2020). As such, low childhood SES, which is already more prevalent in historically marginalized groups, may interact with other social and systemic vulnerabilities to affect cognition in underrepresented groups.
In summary, this study provides an exploratory examination of delirium incidence and severity and early-life exposures. This study represents a novel exploration in the field of delirium, and our results contribute to the growing body of literature highlighting the potential benefit of assessing early-life contributors to cognitive health in older age. These findings support incorporating markers of childhood socioeconomic status into delirium research. Examining early-life exposures will help yield unique insights into novel risk factors that enhance vulnerability to delirium and further elucidate the fundamental pathogenesis of delirium.
CLINICAL IMPLICATIONS.
Delirium prevention evaluations in older adults will benefit from examining immediate as well as long-term factors that may confer increased vulnerability. This is particularly relevant for older adults from socioeconomically and ethnoracially underrepresented groups given that they are more likely to experience individual as well as synergistic effects of social and systemic marginalization. Because early-life factors may not be or may no longer be modifiable risk factors in older adulthood, healthcare providers will have limited control over them in clinical settings. Nevertheless, a comprehensive understanding of a patient’s risk profile will assist with calculating risk/benefit ratios associated with certain interventions (Arias, Bursian, Sappenfield, & Price, 2018). Importantly, this information has the potential to inform clinical decision-making as it may assist clinical teams to determine whether a patient may benefit from additional support when navigating medical settings. In the context of our study, it is possible that patients with lower paternal education may benefit from a more comprehensive assessment of other factors that may increase vulnerability (e.g., current SES, access to support within the home, active substance use). For patients deemed to have a high-risk profile, prehabilitation and post-discharge support may be incorporated into standard care.
Acknowledgments
There are no changes in institutional affiliations to report. This work was funded in part by grants no. P01AG031720 (SKI) and R24AG054259 (SKI) from the National Institute on Aging. Dr. Marcantonio’s time in part by K24AG035075; Dr. Franchesca Arias’ time was supported in part by grant no. 2019-AARFD-644816 of the Alzheimer’s Association; Dr. Inouye holds the Milton and Shirley F. Levy Family Chair at Hebrew SeniorLife/Harvard Medical School. This work is dedicated to the memory of Joshua Bryan Inouye Helfand. The authors gratefully acknowledge the contributions of the patients, family members, nurses, physicians, research team, and members of the Executive Committee who participated in the Successful Aging after Elective Surgery (SAGES) study. Dataset supporting this project is available to researchers subject to a non-disclosure agreement. For details on how to access the data, please, visit: The Aging Brain Center at the Hinda and Arthur Marcus Institute for Aging Research, Hebrew SeniorLife, https://www.marcusinstituteforaging.org/research/aging-brain-center/team. The authors have no competing interests or conflicts to declare. The funding sources had no involvement in the design, analysis, or reporting of the results.
REFERENCES
- Abate SM, Checkole YA, Mantedafro B, Basu B, & Aynalem AE (2021). Global prevalence and predictors of postoperative delirium among non-cardiac surgical patients: a systematic review and meta-analysis. International Journal of Surgery Open, 32, 100334. [Google Scholar]
- Agin A, Blanc F, Bousiges O, Villette C, Philippi N, Demuynck C, … & Heintz D (2020). Environmental exposure to phthalates and dementia with Lewy bodies: contribution of metabolomics. Journal of Neurology, Neurosurgery & Psychiatry, 91(9), 968–974. [DOI] [PubMed] [Google Scholar]
- Aizer A, & Currie J (2014). The intergenerational transmission of inequality: maternal disadvantage and health at birth. science, 344(6186), 856–861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arenaza-Urquijo EM, & Vemuri P (2020). Improving the resistance and resilience framework for aging and dementia studies. Alzheimer’s research & therapy, 12(1), 1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arias F, Alegria M, Kind AJ, Jones RN, Travison TG, Marcantonio ER, … & Inouye SK (2022). A framework of social determinants of health for delirium tailored to older adults. Journal of the American Geriatrics Society, 70(1), 235–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arias F, Bursian AC, Sappenfield JW, & Price CC (2018). Delirium history and preoperative mild neurocognitive disorder: an opportunity for multidisciplinary patient-centered care. The American Journal of Case Reports, 19, 1324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arias F, Chen F, Fong TG, Shiff H, Alegria M, Marcantonio ER, … & Inouye SK (2020). Neighborhood-Level Social Disadvantage and Risk of Delirium Following Major Surgery. Journal of the American Geriatrics Society, 68(12), 2863–2871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avila JF, Rentería MA, Jones RN, Vonk JM, Turney I, Sol K, … & Manly JJ (2021). Education differentially contributes to cognitive reserve across racial/ethnic groups. Alzheimer’s & Dementia, 17(1), 70–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azin A, Hirpara DH, Doshi S, Chesney TR, Quereshy FA, & Chadi SA (2020). Racial Disparities in Surgery: A Cross-Specialty Matched Comparison Between Black and White Patients. Annals of Surgery Open, 1(2), e023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albert MS, Levkoff SE, Reilly C, Liptzin B, Pilgrim D, Cleary PD, … & Rowe JW (1992). The delirium symptom interview: an interview for the detection of delirium symptoms in hospitalized patients. Topics in geriatrics, 5(1), 14–21. [DOI] [PubMed] [Google Scholar]
- Baird J, Jacob C, Barker M, Fall CH, Hanson M, Harvey NC, … & Cooper C (2017, March). Developmental origins of health and disease: a lifecourse approach to the prevention of non-communicable diseases. In Healthcare (Vol. 5, No. 1, p. 14). Multidisciplinary Digital Publishing Institute. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behrman JR, & Birdsall N (1983). The quality of schooling: Quantity alone is misleading. The American Economic Review, 73(5), 928–946. [Google Scholar]
- Bilotta F, Lauretta MP, Borozdina A, Mizikov VM, & Rosa G (2013). Postoperative delirium: risk factors, diagnosis and perioperative care. Minerva Anestesiol, 79(9), 1066–1076. [PubMed] [Google Scholar]
- Brooks Carthon JM, Jarrín O, Sloane D, & Kutney-Lee A (2013). Variations in postoperative complications according to race, ethnicity, and sex in older adults. Journal of the American Geriatrics Society, 61(9), 1499–1507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckles KS (2017). Maternal socio-economic status and the well-being of the next generation (s). In The Oxford Handbook of Women and the Economy. New York: Oxford University Press. [Google Scholar]
- Campbell NL, Cantor BB, Hui SL, Perkins A, Khan BA, Farber MO, … & Boustani MA (2014). Race and documentation of cognitive impairment in hospitalized older adults. Journal of the American Geriatrics Society, 62(3), 506–511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavallari M, Dai W, Guttmann CR, Meier DS, Ngo LH, Hshieh TT, … & Alsop DC (2017). Longitudinal diffusion changes following postoperative delirium in older people without dementia. Neurology, 89(10), 1020–1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cerza F, Renzi M, Gariazzo C, Davoli M, Michelozzi P, Forastiere F, & Cesaroni G (2019). Long-term exposure to air pollution and hospitalization for dementia in the Rome longitudinal study. Environmental Health, 18(1), 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Charlson ME, Pompei P, Ales KL, & MacKenzie CR (1987). A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. Journal of chronic diseases, 40(5), 373–383. [DOI] [PubMed] [Google Scholar]
- Chen R, Weuve J, Misra S, Cuevas A, Kubzansky LD, & Williams DR (2022). Racial disparities in cognitive function among middle-aged and older adults: the roles of cumulative stress exposures across the life course. The Journals of Gerontology: Series A, 77(2), 357–364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Creamer J (2020). Inequalities persist despite decline in poverty for all major race and Hispanic origin groups. US Census Bureau, September, 15. [Google Scholar]
- Cuevas AG, Greatorex-Voith S, Abuelezam N, Eckert N, & Assari S (2020). Educational mobility and telomere length in middle-aged and older adults: testing three alternative hypotheses. Biodemography and Social Biology, 66(3–4), 220–235. [DOI] [PubMed] [Google Scholar]
- Davoudi A, Ozrazgat-Baslanti T, Ebadi A, Bursian AC, Bihorac A, & Rashidi P (2017, October). Delirium prediction using machine learning models on predictive electronic health records data. In 2017 IEEE 17th International Conference on Bioinformatics and Bioengineering (BIBE) (pp. 568–573). IEEE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Everson-Rose SA, Mendes de Leon CF, Bienias JL, Wilson RS, & Evans DA (2003). Early life conditions and cognitive functioning in later life. American journal of epidemiology, 158(11), 1083–1089. [DOI] [PubMed] [Google Scholar]
- Felitti VJ, Anda RF, Nordenberg D, Williamson DF, Spitz AM, Edwards V, & Marks JS (1998). Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults: The Adverse Childhood Experiences (ACE) Study. American journal of preventive medicine, 14(4), 245–258. [DOI] [PubMed] [Google Scholar]
- Fong TG, Hshieh TT, Wong B, Tommet D, Jones RN, Schmitt EM, … & Inouye SK (2015). Neuropsychological profiles of an elderly cohort undergoing elective surgery and the relationship between cognitive performance and delirium. Journal of the American Geriatrics Society, 63(5), 977–982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fong TG, Vasunilashorn SM, Ngo L, Libermann TA, Dillon ST, Schmitt EM, … & SAGES Study Group. (2020). Association of plasma neurofilament light with postoperative delirium. Annals of neurology, 88(5), 984–994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galobardes B, Smith GD, & Lynch JW (2006). Systematic review of the influence of childhood socioeconomic circumstances on risk for cardiovascular disease in adulthood. Annals of epidemiology, 16(2), 91–104. [DOI] [PubMed] [Google Scholar]
- Glymour MM, Avendaño M, Haas S, & Berkman LF (2008). Lifecourse social conditions and racial disparities in incidence of first stroke. Annals of epidemiology, 18(12), 904–912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gou RY, Hshieh TT, Marcantonio ER, Cooper Z, Jones RN, Travison TG, … & SAGES Study Group. (2021). One-year Medicare costs associated with delirium in older patients undergoing major elective surgery. JAMA surgery, 156(5), 430–442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green RE, Melo B, Christensen B, Ngo LA, Monette G, & Bradbury C (2008). Measuring premorbid IQ in traumatic brain injury: an examination of the validity of the Wechsler Test of Adult Reading (WTAR). Journal of clinical and experimental neuropsychology, 30(2), 163–172. [DOI] [PubMed] [Google Scholar]
- Gross AL, Jones RN, Fong TG, Tommet D, & Inouye SK (2014). Calibration and validation of an innovative approach for estimating general cognitive performance. Neuroepidemiology, 42(3), 144–153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haas S (2008). Trajectories of functional health: the ‘long arm’of childhood health and socioeconomic factors. Social science & medicine, 66(4), 849–861. [DOI] [PubMed] [Google Scholar]
- Haddad DN, Mart MF, Wang L, Lindsell CJ, Raman R, Nordness MF, … & Patel MB (2020). Socioeconomic factors and intensive care unit-related cognitive impairment. Annals of surgery, 272(4), 596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halfon N, Larson K, Son J, Lu M, & Bethell C (2017). Income inequality and the differential effect of adverse childhood experiences in US children. Academic Pediatrics, 17(7), S70–S78. [DOI] [PubMed] [Google Scholar]
- Hamrick I, & Meyer F (2013). Perioperative management of delirium and dementia in the geriatric surgical patient. Langenbeck’s archives of surgery, 398(7), 947–955. [DOI] [PubMed] [Google Scholar]
- Hoffmann JP (2016). Cumulative stress and substance use from early adolescence to emerging adulthood. Journal of Drug Issues, 46(3), 267–288. [Google Scholar]
- Hosmer DW, Hosmer T, Le Cessie S, & Lemeshow S (1997). A comparison of goodness-of-fit tests for the logistic regression model. Statistics in medicine, 16(9), 965–980. [DOI] [PubMed] [Google Scholar]
- Inouye SK, Kosar CM, Tommet D, Schmitt EM, Puelle MR, Saczynski JS, … & Jones RN (2014). The CAM-S: development and validation of a new scoring system for delirium severity in 2 cohorts. Annals of internal medicine, 160(8), 526–533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inouye SK, Leo-Summers L, Zhang Y, Bogardus ST Jr, Leslie DL, & Agostini JV (2005). A chart-based method for identification of delirium: validation compared with interviewer ratings using the confusion assessment method. Journal of the American Geriatrics Society, 53(2), 312–318. [DOI] [PubMed] [Google Scholar]
- Inouye SK, Marcantonio ER, Kosar CM, Tommet D, Schmitt EM, Travison TG, … & Jones RN (2016). The short-term and long-term relationship between delirium and cognitive trajectory in older surgical patients. Alzheimer’s & Dementia, 12(7), 766–775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inouye SK, van Dyck CH, Alessi CA, Balkin S, Siegal AP, & Horwitz RI (1990). Clarifying confusion: the confusion assessment method: a new method for detection of delirium. Annals of internal medicine, 113(12), 941–948. [DOI] [PubMed] [Google Scholar]
- Inouye SK, Westendorp RG, & Saczynski JS (2014). Delirium in elderly people. The Lancet, 383(9920), 911–922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jankowski CJ, Trenerry MR, Cook DJ, Buenvenida SL, Stevens SR, Schroeder DR, & Warner DO (2011). Cognitive and functional predictors and sequelae of postoperative delirium in elderly patients undergoing elective joint arthroplasty. Anesthesia & Analgesia, 112(5), 1186–1193. [DOI] [PubMed] [Google Scholar]
- Jones RN, Rudolph JL, Inouye SK, Yang FM, Fong TG, Milberg WP, … & Marcantonio ER (2010). Development of a unidimensional composite measure of neuropsychological functioning in older cardiac surgery patients with good measurement precision. Journal of clinical and experimental neuropsychology, 32(10), 1041–1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones RN, Yang FM, Zhang Y, Kiely DK, Marcantonio ER, & Inouye SK (2006). Does educational attainment contribute to risk for delirium? A potential role for cognitive reserve. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 61(12), 1307–1311. [DOI] [PubMed] [Google Scholar]
- Joyce BT, Gao T, Koss K, Zheng Y, Cardenas A, Heiss J, … & Hou L (2021). Impact of paternal education on epigenetic ageing in adolescence and mid-adulthood: a multi-cohort study in the USA and Mexico. International Journal of Epidemiology. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kagansky N, Rimon E, Naor S, Dvornikov E, Cojocaru L, & Levy S (2004). Low incidence of delirium in very old patients after surgery for hip fractures. The American Journal of Geriatric Psychiatry, 12(3), 306–314. [PubMed] [Google Scholar]
- Kazmierski J, Banys A, Latek J, Bourke J, & Jaszewski R (2013). Cortisol levels and neuropsychiatric diagnosis as markers of postoperative delirium: a prospective cohort study. Critical Care, 17(2), 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan BA, Perkins A, Hui SL, Gao S, Campbell NL, Farber MO, & Boustani MA (2016). Relationship between African-American race and delirium in the intensive care unit. Critical care medicine, 44(9), 1727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keage HA, Muniz G, Kurylowicz L, Van Hooff M, Clark L, Searle AK, … & McFarlane A (2016). Age 7 intelligence and paternal education appear best predictors of educational attainment: The Port Pirie Cohort Study. Australian Journal of Psychology, 68(1), 61–69. [Google Scholar]
- Kind AJ, Jencks S, Brock J, Yu M, Bartels C, Ehlenbach W, … & Smith M (2014). Neighborhood socioeconomic disadvantage and 30-day rehospitalization: a retrospective cohort study. Annals of internal medicine, 161(11), 765–774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S, Jones RN, & Glymour MM (2010). Implications of lifecourse epidemiology for research on determinants of adult disease. Public Health Reviews, 32(2), 489–511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo Y, & Waite LJ (2005). The impact of childhood and adult SES on physical, mental, and cognitive well-being in later life. The Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 60(2), S93–S101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manios Y, Costarelli V, Kolotourou M, Kondakis K, Tzavara C, & Moschonis G (2007). Prevalence of obesity in preschool Greek children, in relation to parental characteristics and region of residence. BMC Public Health, 7(1), 1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manly JJ (2006). Deconstructing race and ethnicity: implications for measurement of health outcomes. Medical care, S10–S16. [DOI] [PubMed] [Google Scholar]
- Marcantonio ER (2017). Delirium in hospitalized older adults. New England Journal of Medicine, 377(15), 1456–1466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matheson K, Foster MD, Bombay A, McQuaid RJ, & Anisman H (2019). Traumatic experiences, perceived discrimination, and psychological distress among members of various socially marginalized groups. Frontiers in psychology, 10, 416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClendon J, Chang K, Boudreaux MJ, Oltmanns TF, & Bogdan R (2021). Black-White racial health disparities in inflammation and physical health: Cumulative stress, social isolation, and health behaviors. Psychoneuroendocrinology, 131, 105251. [DOI] [PubMed] [Google Scholar]
- McCusker J, Cole M, Abrahamowicz M, Primeau F, & Belzile E (2002). Delirium predicts 12-month mortality. Archives of internal medicine, 162(4), 457–463. [DOI] [PubMed] [Google Scholar]
- Meng Y, & Groth SW (2018). Fathers count: the impact of paternal risk factors on birth outcomes. Maternal and child health journal, 22(3), 401–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta KM, & Yeo GW (2017). Systematic review of dementia prevalence and incidence in United States race/ethnic populations. Alzheimer’s & Dementia, 13(1), 72–83. [DOI] [PubMed] [Google Scholar]
- Mulkey MA, Hardin SR, Olson DM, & Munro CL (2018). Pathophysiology review: seven neurotransmitters associated with delirium. Clinical Nurse Specialist, 32(4), 195–211. [DOI] [PubMed] [Google Scholar]
- Myrskylä M, Elo IT, Kohler IV, & Martikainen P (2014). The association between advanced maternal and paternal ages and increased adult mortality is explained by early parental loss. Social Science & Medicine, 119, 215–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagelkerke NJ (1991). A note on a general definition of the coefficient of determination. Biometrika, 78(3), 691–692. [Google Scholar]
- Northwood M, Ploeg J, Markle-Reid M, & Sherifali D (2018). Integrative review of the social determinants of health in older adults with multimorbidity. Journal of Advanced Nursing, 74(1), 45–60. [DOI] [PubMed] [Google Scholar]
- Ogunsola FT, & Mehtar S (2020). Challenges regarding the control of environmental sources of contamination in healthcare settings in low-and middle-income countries-a narrative review. Antimicrobial Resistance & Infection Control, 9(1), 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phipps RM, & Degges-White S (2014). A new look at transgenerational trauma transmission: Second-generation Latino immigrant youth. Journal of Multicultural Counseling and Development, 42(3), 174–187. [Google Scholar]
- Rogers MA, Plassman BL, Kabeto M, Fisher GG, McArdle JJ, Llewellyn DJ, … & Langa KM (2009). Parental education and late-life dementia in the United States. Journal of geriatric psychiatry and neurology, 22(1), 71–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers R, Eagle TF, Sheetz A, Woodward A, Leibowitz R, Song M, … & Eagle KA (2015). The relationship between childhood obesity, low socioeconomic status, and race/ethnicity: lessons from Massachusetts. Childhood Obesity, 11(6), 691–695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saczynski JS, Kosar CM, Xu G, Puelle MR, Schmitt E, Jones RN, … & Inouye SK (2014). A tale of two methods: chart and interview methods for identifying delirium. Journal of the American Geriatrics Society, 62(3), 518–524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seeman T, Epel E, Gruenewald T, Karlamangla A, & McEwen BS (2010). Socio-economic differentials in peripheral biology: Cumulative allostatic load. Annals of the New York Academy of Sciences, 1186(1), 223–239. [DOI] [PubMed] [Google Scholar]
- Shaw BA, & Spokane LS (2008). Examining the association between education level and physical activity changes during early old age. Journal of aging and health, 20(7), 767–787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmitt EM, Saczynski JS, Kosar CM, Jones RN, Alsop DC, Fong TG, … & Sperling R (2015). The successful aging after elective surgery study: cohort description and data quality procedures. Journal of the American Geriatrics Society, 63(12), 2463–2471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh GK (2003). Area deprivation and widening inequalities in US mortality, 1969–1998. American journal of public health, 93(7), 1137–1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Team, R. C. (2013). R: A language and environment for statistical computing.
- Teng E, & Chui H (1987). The modified mini-mental state examination (3MS). Can J Psychiatry, 41(2), 114–21. [PubMed] [Google Scholar]
- Turner C, Tamura R, Mulholland SE, & Baier S (2007). Education and Income of the States of the United States: 1840–2000. Journal of Economic Growth, 12(2), 101–158. [Google Scholar]
- Turrell G, Lynch JW, Kaplan GA, Everson SA, Helkala EL, Kauhanen J, & Salonen JT (2002). Socioeconomic position across the lifecourse and cognitive function in late middle age. The Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 57(1), S43–S51. [DOI] [PubMed] [Google Scholar]
- United States Department of Labor, Womens Bureau (2020). 100 Years of Working Women. https://www.dol.gov/agencies/wb/data/occupations-decades-100. Accessed 10/17, 2020.
- Vable AM, Eng CW, Mayeda ER, Basu S, Marden JR, Hamad R, & Glymour MM (2018). Mother’s education and late-life disparities in memory and dementia risk among US military veterans and non-veterans. J Epidemiol Community Health, 72(12), 1162–1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vervoort I, Delger C, & Soubry A (2021). A multifactorial model for the etiology of neuropsychiatric disorders: the role of advanced paternal age. Pediatric Research, 1–14. [DOI] [PubMed] [Google Scholar]
- Westhoff D, Witlox J, Koenderman L, Kalisvaart KJ, de Jonghe JF, van Stijn MF, … & van Gool WA (2013). Preoperative cerebrospinal fluid cytokine levels and the risk of postoperative delirium in elderly hip fracture patients. Journal of Neuroinflammation, 10(1), 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson RS, Barnes LL, & Bennett DA (2003). Assessment of lifetime participation in cognitively stimulating activities. Journal of clinical and experimental neuropsychology, 25(5), 634–642. [DOI] [PubMed] [Google Scholar]
- Wilson RS, De Leon CFM, Barnes LL, Schneider JA, Bienias JL, Evans DA, & Bennett DA (2002). Participation in cognitively stimulating activities and risk of incident Alzheimer disease. Jama, 287(6), 742–748. [DOI] [PubMed] [Google Scholar]
- Willson AE, & Shuey KM (2019). A longitudinal analysis of the intergenerational transmission of health inequality. The Journals of Gerontology: Series B, 74(1), 181–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu TT, Zegers M, Kooken R, Griffith JL, Molnar BE, Devlin JW, & van den Boogaard M (2021). Social Determinants of Health and Delirium Occurrence and Duration in Critically Ill Adults. Critical Care Explorations, 3(9). [DOI] [PMC free article] [PubMed] [Google Scholar]


