Abstract
Background
Recently, PET/CT imaging with radiolabelled FAP inhibitors (FAPIs) has been widely evaluated in diverse diseases. However, rare report has been published using SPECT/CT, a more available imaging method, with [99mTc]Tc-labelled FAPI. In this study, we evaluated the potential effect of [99mTc]Tc-HFAPi in clinical analysis for digestive system tumours.
Methods
This is a single-centre prospective diagnostic efficiency study (Ethic approved No.: XJTU1AF2021LSK-021 of the First Affiliated Hospital of Xi’an Jiaotong University and ChiCTR2100048093 of the Chinese Clinical Trial Register). Forty patients with suspected or confirmed digestive system tumours underwent [99mTc]Tc-HFAPi SPECT/CT between January and June 2021. For dynamic biodistribution and dosimetry estimation, whole-body planar scintigraphy was performed at 10, 30, 90, 150, and 240 min post-injection in four representative patients. Optimal acquisition time was considered in all the patients at 60–90 min post-injection, then quantified or semi-quantified using SUVmax and T/B ratio was done. The diagnostic performance of [99mTc]Tc-HFAPi was calculated and compared with those of contrast-enhanced CT (ceCT) using McNemar test, and the changes of tumour stage and oncologic management were recorded.
Results
Physiological distribution of [99mTc]Tc-HFAPi was observed in the liver, pancreas, gallbladder, and to a lesser extent in the kidneys, spleen and thyroid. Totally, 40 patients with 115 lesions were analysed. The diagnostic sensitivity of [99mTc]Tc-HFAPi for non-operative primary lesions was similar to that of ceCT (94.29% [33/35] vs 100% [35/35], respectively; P = 0.5); in local relapse detection, [99mTc]Tc-HFAPi was successfully detected in 100% (n = 3) of patients. In the diagnosis of suspected metastatic lesions, [99mTc]Tc-HFAPi exhibited higher sensitivity (89.66% [26/29] vs 68.97% [20/29], respectively, P = 0.03) and specificity (97.9% [47/48] vs 85.4% [41/48], respectively, P = 0.03) than ceCT, especially with 100% (24/24) specificity in the diagnosis of liver metastases, resulting in 20.0% (8/40) changes in TNM stage and 15.0% (6/40) changes in oncologic management.
Conclusion
[99mTc]Tc-HFAPi demonstrates a greater diagnostic efficiency than ceCT in the detection of distant metastasis, especially in identifying liver metastases.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00259-022-06068-1.
Keywords: [99mTc]Tc-HFAPi, SPECT/CT, Digestive system cancer, Diagnostic efficiency
Introduction
An in-depth understanding of the tumour microenvironment has revealed a new player: cancer-associated fibroblasts (CAFs) [1]. The majority of epithelial tumours recruit fibroblasts and other non-malignant cells, stimulating them to become CAFs. This often leads to overexpression of membrane serine protease fibroblast activating protein alpha (FAP-α, also known as prolyl endopeptidase FAP), which is estimated to be overexpressed in approximately 90% of human cancers [2–4]. As FAP is mostly absent in healthy tissue, inhibitors of FAP (FAPIs) can be used in nuclear medicine for imaging [5]. Indeed, a large number of FAPI-based radiopharmaceuticals have been developed for PET/CT imaging, and a promising role for [68Ga]Ga-FAPI PET/CT in the diagnosis, staging, and radiotherapy planning of digestive tract cancers has been demonstrated [6–8].
[68Ga]Ga-FAPI PET/CT showed a higher sensitivity than [18F]FDG PET/CT in the detection of primary and metastatic lesions of various types of cancers [6, 9]. Recently, Koerber et al. reported the first clinical use of [68Ga]Ga-FAPI PET/CT for tumours in the lower intestinal tract [7]. Their results revealed that both primary and metastatic malignancies in the lower gastrointestinal tract can be reliably detected using [68Ga]Ga-FAPI PET/CT, leading to relevant changes in TNM status and oncologic management.
Due to its lower cost, SPECT/CT with technetium-99m (99mTc) is a more widely available, and 99mTc-labelled FAPIs are generally applicable tracers that are attractive options for imaging in clinical management when PET imaging is inaccessible or limited [10]. Lindner et al. reported 99mTc-labelled FAPIs and evaluated their biodistribution in tumour‐bearing mice [10], and found 99mTc-FAPI-34 showing strong and constant tumour accumulation. The preclinical application has indicated that it is a good candidate for scintigraphic imaging owing to the high contrast obtained via rapid tumour uptake and clearance from the rest of the body. Nevertheless, reliable clinical data are lacking, which only applied in two patients with ovarian and pancreatic cancer. Here, we report our first clinical experience with [99mTc]Tc-HYNIC-FAPI-04 ([99mTc]Tc-HFAPi) SPECT/CT applied in a cohort of patients with digestive system tumours. After quantifying tracer uptake in primary tumours and metastases, we compared the diagnostic efficiency of the [99mTc]Tc-HFAPi with the conventional imaging ceCT, which is routinely recommended in digestive system tumours [11–13].
Materials and methods
Radiopharmaceutical preparation
For 99mTc radiolabelling, 1 mL of 925–1295 MBq (25–35 mCi) of [99mTc]TcO4− saline solution was added to 25 µg of hydrazinonicotinamide-FAPI-04 (HYNIC-FAPI-04, abbreviated to HFAPi, Fig. S1), 3.0 mg of Trisodium triphenylphosphine-3,3′,3″-trisulfonate, and 2.0 mg of tricine, then incubated at 100 °C for 15 min. The radiochemical purity (RCP) was analysed by radio-HPLC and ITLC-SG, and the specific operation method is detailed in the supplementary information. For clinical use, the RCP was always greater than 95%. The reaction mixture was then filtered through a 0.20-mm Millex-LG filter (EMD Millipore) before agent injection.
Patients
This is a single-centre prospective diagnostic efficiency study of [99mTc]Tc-HFAPi SPECT/CT in digestive system tumours, with ceCT serving as the reference method, approved by the Clinical Research Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University (Ethic approved No.: XJTU1AF2021LSK-021) and Chinese Clinical Trial Register (Registration No.: ChiCTR2100048093). From January to June 2021, patients with suspected digestive system tumours who needed preoperative initial staging or posttreatment restaging were consecutively recruited at the First Affiliated Hospital of Xi’an Jiaotong University with written informed consent. Detailed Eligibility criteria is provided in the supplementary information. After a standard work-up including but not limited to ceCT, additional [99mTc]Tc-HFAPi SPECT/CT was performed (generally within 7 days).
Scintigraphy and SPECT/CT
Based on the previous reference dose [10, 14, 15], [99mTc]Tc-HFAPi was administered intravenously in amounts ranging from 790.4 to 930.2 MBq (21.36 to 25.14 mCi). For dynamic biodistribution, whole-body planar scintigraphy was performed at 10, 30, 90, 150 and 240 min in four representative patients. Base on biodistribution results, [99mTc]Tc-HFAPi imaging was performed 60–90 min following tracer injection in all the patients to get whole-body planar scintigraphy as well as SPECT/CT tomography fusion images. Whole-body scans were performed via GE Discovery 670 pro scanner system (GE Healthcare) equipped with low-energy high-resolution (LEHR) collimators in 18 cm/min velocity. Low-dose CT was performed for attenuation correction and anatomic localization. The patients were asked to self-report any abnormalities at 30 min after the examination was completed.
Biodistribution and dosimetry estimation
Visual analysis was applied to determine the integral biodistribution of the tracer as well as the transient and intersubject stability. For each subject, regions of interest (ROIs) were delineated over the identified organs: the heart, liver, lungs, kidneys, pancreas, spleen, brain, thyroid and salivary glands. The geometric mean count was determined for every organ from the background-corrected anterior and posterior counts. The results are expressed as a percentage of the initial injected activity after decay correction (%ID/organ). For dosimetry estimation, absorbance dose of different organs and effective dose were calculated using OLINDA/EXM 1.0 software (Vanderbilt, University, Nashville, TN, USA) as previously described [15].
SPECT/CT imaging review
[99mTc]Tc-HFAPi SPECT/CT scans were evaluated by 1 certified radiologist and 2 certified nuclear medicine physicians. They reach consensus when there is disagreement. Conventional imaging was interpreted by 2 certified radiologists with consensus but blind to the [99mTc]Tc-HFAPi SPECT/CT results. Fused SPECT/CT images were viewed on the Xeleris Workstation (version AW 4.7, GE Healthcare). For quantitative and semi-quantitative analyses, ROIs were drawn on transaxial images over the tumour with focally increased uptake. Quantitative calculating the SUVmax was based on an algorithm, which has been patented (Patent number: US11189374B2) [16]. The tumour-to-background (T/B) ratio was determined by dividing the maximum tumour uptake by the maximum contralateral muscle uptake. For skull lesions, T/B ratio was calculated as dividing the maximum tumour uptake by the maximum normal skull uptake.
Diagnosis and follow-up
Histopathology of biopsy/resected surgical specimens served as the gold standard for the final diagnosis. In cases in which the diagnosis of malignancy was not applicable, follow-up data after the SPECT/CT scans were requested. Referring to a similar study [9], the disease was defined as malignancy when (a) typical malignant features were confirmed by multi-modality imaging, (b) significant progression on follow-up imaging (significant increase in size), or (c) a significant decrease in size after anticancer treatment. All suspected lesions were followed up for no less than 6 months.
Immunohistochemistry (IHC) of FAP expression
FAP expression in 4 representative patients was analysed by immunohistochemistry, heat-mediated antigen retrieval was performed with Tris/EDTA buffer pH 9.0. The sections were incubated with 1:250 humanized anti-fibroblast activation antibody (Abcam, ab207178) at 4 °C overnight. After incubation with the labelled streptavidin–biotin (LSAB) complex, the slides were stained and visualized using the iView DAB detection system (ZSGB-BIO, Beijing, China). Typical lesions in high-power fields were photographed for visual comparison.
Statistical analyses
All statistical analyses were conducted using SPSS 25.0 statistical analysis software (IBM, Armonk, NY, USA). For organ biodistribution, the percentage of initial injected activity after decay correction (%ID) was used. To determine lesion uptake, the T/B ratio and SUVmax were used with the median ± interquartile range (IQR) because of non-normal distribution. McNemar test and chi-square test were employed to compare the diagnostic values between [99mTc]Tc-HFAPi SPECT/CT and ceCT. A receiver operating characteristic (ROC) curve was constructed to quantify the diagnostic performance of the T/B ratio and SUVmax by assessing the respective areas under the curve (AUCs). Two-tailed P values < 0.05 were considered significant.
Results
Radiopharmaceutical preparation
The structure of [99mTc]Tc-HFAPi was shown in (Fig. S1). The average radiochemical purity of [99mTc]Tc-HFAPi prepared from lyophilized kits, determined by radio-HPLC (Fig. S2) and ITLC-SG (Fig. S3), was over 95% with < 1% of free [99mTc]TcO4− as well as < 0.5% of [99mTc]Tc-colloid. [99mTc]Tc-HFAPi could be readily prepared in high specific activity (> 2.275 × 105 MBq/μmoL), and it was stable in the kit matrix as well as in the saline for > 6 h. More data on [99mTc]Tc-HFAPi preparation and preclinical studies will be reported in detail in a separate research paper.
Patient characteristics
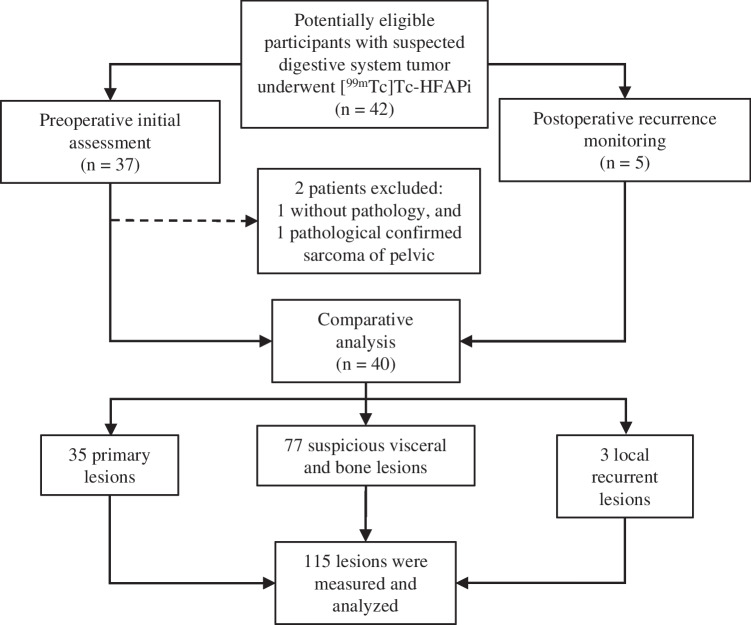
For 40 patients (25 male) enrolled, thirty-five patients had yet to undergo cancer-related surgery (30 treatment naïve and 5 with chemo/radio-therapy). Another 5 patients had already undergone surgery with/without chemotherapy and/or radiotherapy. The characteristics of the patients and primary lesions information are summarized in Table 1. Ultimately, 39 patients were confirmed to have malignant disease, whereas 1 was confirmed to have benign tumour (spindle cell tumour of intestine). The scheme of the study design is presented in Fig. 1.
Table 1.
Characteristics of patients that underwent [99mTc]Tc-HFAPi SPECT/CT
| Characteristic | Value |
|---|---|
| No. of patients | 40 |
| Age (years) | |
| Median | 64.5 |
| Interquartile range | 54.3–71.8 |
| Sex | |
| Male | 25 |
| Female | 15 |
| Site and pathology of primary disease | |
| Rectum Adenocarcinoma | 15 |
| Gastric Adenocarcinoma | 13 |
| Colonic Adenocarcinoma | 8 |
| Esophageal Squamous carcinoma | 1 |
| Intestinal Spindle cell tumour | 1 |
| Pancreatic carcinoma | 1 |
| Anal malignant melanoma | 1 |
| Clinical status before imaging | |
| Treatment-naive | 30 |
| Neoadjuvant chemo/radio-therapy | 5 |
| Resection surgery | 3 |
| Chemo/radio-therapy after surgery | 2 |
| Other imaging | |
| Contrast enhanced CT | 40 |
| Gastrointestinal endoscope | 38 |
| DWI | 11 |
| B ultrasound | 7 |
| 18F-FDG | 1 |
| Clinical questions for 99mTc-HFAPi | |
| Staging of cancer before surgery | 35 |
| Identification of disease recurrence and restaging | 5 |
Fig. 1.
The scheme of the study design
No drug-related side effects occurred during or after [99mTc]Tc-HFAPi injection, and SPECT/CT imaging was tolerated well by all patients. Vital parameters remained stable, and no patient reported any new symptoms during the observation period.
Dynamic biodistribution in organs and dosimetry estimation
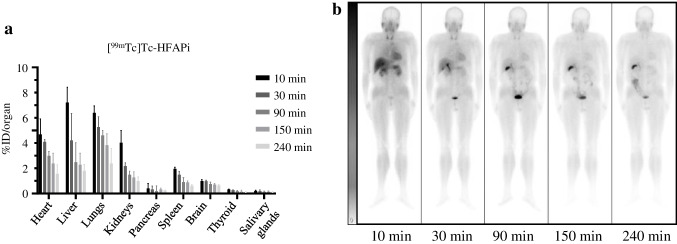
The dynamic physiological biodistribution of [99mTc]Tc-HFAPi in vital organs at 10, 30, 90, 150, and 240 min was measured in 4 patients and summarized using %ID/organ (patients’ information Table S1). Physiological distribution of [99mTc]Tc-HFAPi was observed in the liver, pancreas, gallbladder, and to a lesser extent in the kidneys, lungs, spleen, salivary glands, and thyroid glands, with rapid clearance of the radiotracer from these organs (Fig. 2a). Representations of a coronal section from whole-body SPECT are shown in Fig. 2b.
Fig. 2.
Biodistribution of [99mTc]Tc-HFAPi in different vital organs over time. a % Injection Dose (ID)/organ of [99mTc]Tc-HFAPi in heart, liver, lungs, kidneys, pancreas, spleen, brain, thyroid and salivary glands in different times. Data represent median ± interquartile range. b Example of background ROIs in patient #002 on planar scintigraphy images: whole body [99mTc]Tc-HFAPi scintigraphy was performed at 10, 30, 90, 150 and 240 min post-injection
A summary of dosimetric parameters for various organs is given in Table S2, and the mean effective dose equivalent of the whole body was 1.26 × 10−3 mSv/MBq, which is consistent with those for other molecules labelled with 99mTc [15, 17].
[99mTc]Tc-HFAPi for diagnosing primary lesions
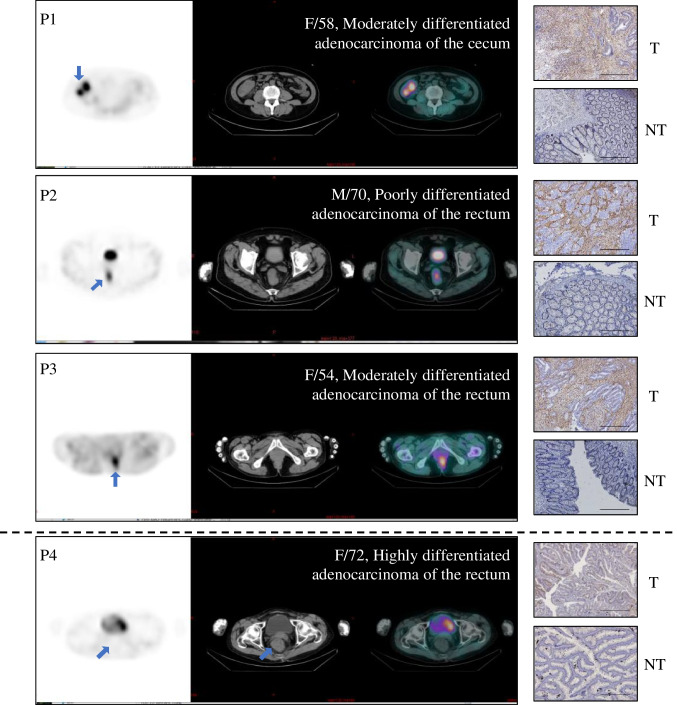
Thirty-five patients with unresected primary digestive system lesions, which were all pathologically confirmed as malignant lesions, were detected by [99mTc]Tc-HFAPi SPECT/CT with a nearly identical sensitivity of 94.29% (33/35) as ceCT (35/35, 100%, P = 0.5). The two false-negative of [99mTc]Tc-HFAPi were found highly differentiated rectal adenocarcinoma by pathology. Representative 3 true-positive and 1 false-negative lesions were stained with the anti-FAP antibody by IHC. As illustrated in Fig. 3, patient (P4) with false-negative lesions on [99mTc]Tc-HFAPi SPECT/CT showed the lowest expression of FAP compared with true-positive patients (P1-3), indicating that the uptake of [99mTc]Tc-HFAPi was associated with the expression of FAP.
Fig. 3.
SPECT/CT (left) and immunohistochemistry staining (right) of representative primary lesions and tumour-adjacent tissue. The graphs above the dotted line were representations of true positive cases (P1–P3), while below the dotted line is a representation of false negative case (P4) by [99mTc]Tc-HFAPi. Scale bar, 200 μm; T: primary tumour; NT: Tumour-adjacent tissue
Local recurrence was found in 60% (3/5) of patients by pathology or follow-up imaging, all of which were positively detected by ceCT and [99mTc]Tc-HFAPi SPECT/CT (Table S3).
[99mTc]Tc-HFAPi in the diagnosis of suspected metastatic lesions
After [99mTc]Tc-HFAPi examination, 77 suspected lesions were detected. Among them, 29 lesions in 9 patents were confirmed metastatic lesions by pathology (n = 4), multi-modality imaging (n = 11), or follow-up (n = 14). In total, [99mTc]Tc-HFAPi-positive metastases were observed in 8 patients with 26 lesions, including liver (n = 15), bone (n = 8), abdomen (n = 1), pelvic (n = 1) and mediastinum tissue (n = 1). Compared to ceCT, [99mTc]Tc-HFAPi exhibited higher sensitivity in the diagnosis of suspected metastatic lesions (89.66% [26/29] vs 68.97% [20/29], respectively, P = 0.03). For 48 benign lesions, [99mTc]Tc-HFAPi showed higher specificity than ceCT (97.9% [47/48] vs. 85.4% [41/48], respectively; P = 0.03). The only one false positive case turned out to be tuberculosis that has been reported previously [9, 18]. A comparison of diagnostic efficiency on benign or metastatic lesions between ceCT and [99mTc]Tc-HFAPi SPECT/CT is shown in Table 2. It is worth noting that for liver metastasis determination, 100% (24/24) specificity was achieved using [99mTc]Tc-HFAPi SPECT/CT, with 83.3% (20/24) by ceCT (Table 3). Although the non-statistical difference (P = 0.13) might be related to the relatively small sample size, subsequent studies with larger sample size are still worthwhile.
Table 2.
Diagnostic efficiency of [99mTc]Tc-HFAPi in suspected lesions compared with ceCT
| Basis of analysis and modality | Sensitivity TPRa (%) |
Specificity TNRb (%) |
Negative predict value NPV (%) |
Positive predict value PPV (%) |
Accuracy ACC (%) |
|---|---|---|---|---|---|
|
HFAPi-meta 95% CI |
89.7 (26/29) | 97.9 (47/48) | 94.0 (47/50) | 96.3 (26/27) | 94.8 (73/77) |
| 71.5–97.3 | 87.5–99.9 | 82.5–98.4 | 79.1–99.8 | 87.0–98.4 | |
|
ceCT-meta 95% CI |
69.0 (20/29) | 85.4 (41/48) | 82.0 (41/50) | 74.1 (20/27) | 79.2 (61/77) |
| 49.0–84.0 | 71.6–93.5 | 68.1–90.9 | 53.4–88.1 | 68.8–86.9 |
aTPR, true positive rate
bTNR, true negative rate
Table 3.
Diagnostic efficiency of [99mTc]Tc-HFAPi in liver metastasis compared with ceCT
| Basis of analysis and modality | Sensitivity TPR (%) |
Specificity TNR (%) |
Negative predict value NPV (%) |
Positive predict value PPV (%) |
Accuracy ACC (%) |
|---|---|---|---|---|---|
|
HFAPi-liver 95% CI |
88.2 (15/17) | 100.0 (24/24) | 92.3 (24/26) | 100.0 (15/15) | 95.1 (39/41) |
| 62.2–97.9 | 82.8–100.0 | 73.4–98.7 | 74.7–100.0 | 83.0–99.5 | |
|
ceCT-liver 95% CI |
100.0 (17/17) | 83.3 (20/24) | 100.0 (20/20) | 81.0 (17/21) | 90.2 (37/41) |
| 77.1–100.0 | 61.8–94.5 | 79.9–100.0 | 57.4–98.7 | 76.9–96.7 |
Clinical values of [99mTc]Tc-HFAPi
In suspected metastatic lesions which were not diagnosed coincidently by ceCT and [99mTc]Tc-HFAPi SPECT/CT, follow-up data were requested as described in methods (Diagnosis and follow-up section). As a result, [99mTc]Tc-HFAPi SPECT/CT suggested the metastasis (M) classification restaging in 8/40 (20.0%) patients (1 patient staged up and 7 staged down). Among the restaging, 6 of 8 patients changed the oncologic regimen, including 1 with new findings for bone metastasis who changed to systemic therapy and 5 for whom curative surgery was performed instead of systemic therapy, consistently with those of [99mTc]Tc-HFAPi imaging results. Changes in the clinical oncologic regimen are given in Table 4. The results indicate that [99mTc]Tc-HFAPi SPECT/CT can provide a strong basis for clinical decision-making.
Table 4.
Changes in metastatic staging and oncologic management according to [99mTc]Tc-HFAPi
| No | Sex | Age | State | Primary tumour | Metastasis stage and site from ceCT | Metastasis stage and site from [99mTc]Tc-HFAPi | Ways to confirm | Changing in stage | Clinical decision |
|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 69 | Treatment-naive | Adenocarcinoma of rectum | Mx | M1 with occipital bone | CT with bony change | Staging up | Palliative operation |
| 2 | F | 58 | Treatment-naive | Adenocarcinoma of the cecum | M1 with liver | M0 without liver | DWI: hepatic cyst | Staging down | Tend to operation |
| 3 | M | 81 | Treatment-naive | Adenocarcinoma of rectum | M1 with liver | M0 without liver | DWI: Hepatic spongy hemangioma | Staging down | Tend to operation |
| 4 | M | 55 | Treatment-naive | Gastric adenocarcinoma | M1 with liver | M0 without liver | DWI: no obvious abnormality | Staging down | Tend to operation |
| 5 | F | 22 | Treatment-naive | Gastric adenocarcinoma | M0 | M1 with skull, posterior bulbar tissue, peritoneum | Pathology of peritoneum, CT with bony change of the skull | Staging up | Palliative surgery and intraperitoneal chemotherapy |
| 6 | M | 58 | Treatment-naive | Gastric adenocarcinoma | Mx | M0 without liver | Follow up | Staging down | Tend to operation |
| 7 | F | 51 | Neoadjuvant chemotherapy | Adenocarcinoma of rectum | Mx | M0 without spleen | DWI: Splenic hemangioma | Staging down | Tend to operation |
SUVmax and T/B ratio in the diagnosis of benign and malignant disease
Our team previously designed an algorithm, which has been patented (Patent number: US11189374B2) [16], to calculate the SUVmax based on SPECT/CT and linearized it against the standard T/B ratio. The result indicating an obvious linear correlation with R2 of 0.735 (Fig. 4, P < 0.001) between SUVmax and T/B ratio.
Fig. 4.
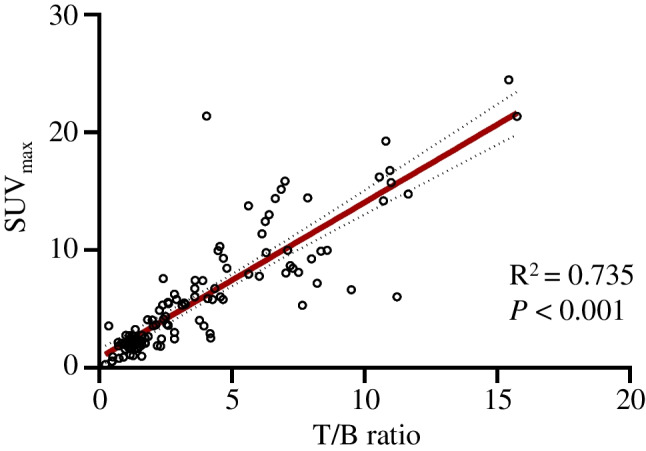
Linear relationship between SUVmax and T/B ratio. R: linear correlation coefficient
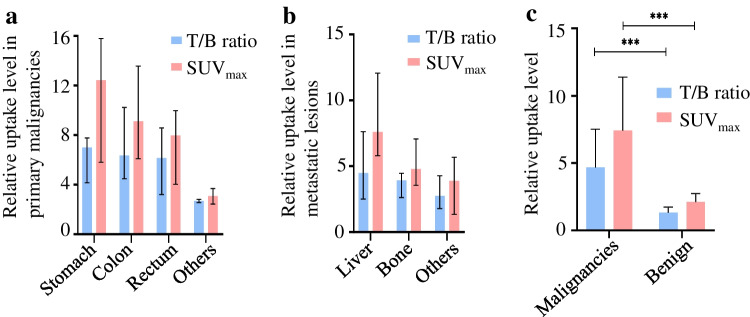
For primary malignant lesions, the median T/B ratio and SUVmax were 6.35 (IQR: 3.64 to 8.10) and 9.52 (IQR: 5.73 to 14.52), respectively; for all metastases, they were 3.65 (IQR:2.47 to 4.79) and 6.05 (IQR: 4.43 to 9.09), respectively. The SUVmax and T/B ratio for different malignant lesions by [99mTc]Tc-HFAPi are shown in Fig. 5a, b and Table S3, S4. High SUVmax and T/B ratios were found in gastric cancer and liver metastasis. For non-malignant lesions, few showed intense uptake of [99mTc]Tc-HFAPi, with a median T/B ratio and SUVmax of 1.32 (IQR: 1.01–1.74) and 2.11 (IQR: 1.68–2.75), respectively, both were significantly lower (P < 0.001) than malignancies (Fig. 5c).
Fig. 5.
Uptake of [99mTc]Tc-HFAPi in different lesions. T/B ratio (blue bar) and SUVmax (red bar) are shown in primary malignancies (a) and metastatic lesions (b). c The comparison of [99mTc]Tc-HFAPi uptake between benign and malignant lesions. Data represent median ± interquartile range. ***, P < 0.001
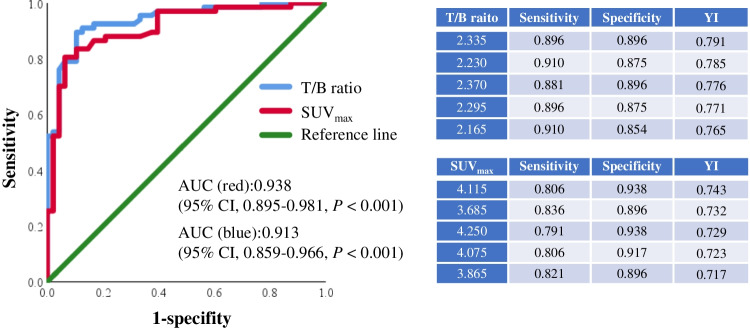
The ROC curve built with 67 malignant lesions (38 primary malignancies and 29 metastatic lesions) and 48 benign lesions yielded an AUC of 0.938 (95% CI, 0.895–0.981, P < 0.001) for the T/B ratio and 0.913 (95% CI, 0.859–0.966, P < 0.001) for SUVmax (Fig. 6, left). Youden’s index analysis revealed several optimal cut-off values for discriminating malignant from non-malignant lesions (Fig. 6, right).
Fig. 6.
The performance of T/B ratio (red line) and SUVmax (blue line) in the diagnosis of digestive system cancer by receiver operating characteristic (ROC) curves analysis (left). The charts (right) show several optimal cut-off values for discriminating malignant from non-malignant lesions. T/B ratio, tumour/background ratio; AUC, area under ROC curves; YI, Youden’s index
Discussion
Recent PET/CT studies with FAP inhibitors have been well developed, revealing strong PET signals across dozens of major cancers, especially digestive system cancers [6, 7, 19, 20]. With FAPI-specific PET imaging, patients do not require dietary preparation, and high-quality images can be obtained soon after tracer injection (10 min to 1 h, mainly 1 h) [21]. The superior value of [68Ga]Ga-FAPI PET/CT over [18F]FDG in detecting primary and metastatic lesions in digestive system cancers has been confirmed, most lesions showing higher tracer uptake with FAPI imaging, and it exhibits a promising role in the diagnosis, (re)staging, management, and treatment planning of digestive system cancers [22]. However, limited data about 99mTc-labelled FAPIs in clinical have been reported, with only application in two patients (ovarian and pancreatic cancer) [10]. Here, we evaluated the biodistribution of a newly developed [99mTc]Tc-HFAPi and the uptake in different digestive system cancers. Biodistribution studies have shown that [99mTc]Tc-HFAPi has good targeting of malignancies and fast renal clearance, with low uptake in normal organs, leading to a higher tumour-to-background ratio and have a relatively wide imaging time window, indicating it is a suitable clinical imaging agent for digestive system tumours. Comparisons of diagnostic efficiency in digestive system cancers between [99mTc]Tc-HFAPi and ceCT were performed in this study, since CT with contrast is routinely used for preoperational imaging in digestive system cancers based on NCCN guidelines [11–13].
Our study demonstrated that [99mTc]Tc-HFAPi and ceCT were comparable in detecting the primary tumours of digestive system. Almost all primary malignancies (n = 33) showed marked uptake of [99mTc]Tc-HFAPi, especially gastric cancers and colon cancers, with median T/B ratios of 7.01 and 6.35, and median SUVmax values of 12.43 and 9.13, respectively. This was in line with the previous PET imaging with 68Ga-labelled FAPI-04 [20], which showed the highest uptake in colon cancers and gastric cancers. Our study also covered small samples of oesophageal squamous carcinoma, pancreatic carcinoma, and anal malignant melanoma, which all showed good detection performance. In the detection of tumour recurrence in patients who received surgery (n = 5), 60% (3/5) of them showed recurrence and all had successful detection by [99mTc]Tc-HFAPi, consistent with the ceCT results. Although the samples were too few, they were particularly useful for their presence of elevated tumour markers but no clinical or morphological evidence, as previously indicated [9].
The liver is the main site of metastasis and a major cause of death in digestive system malignancies, leading to short PFS and extremely poor prognosis [23]. Several imaging methods have been utilized in the detection of liver metastasis, including ceCT, MRI, and [18F]FDG PET/CT, but all have limitations. Although ceCT is commonly used for diagnosing liver metastasis, its accuracy does not always meet clinical requirements [24, 25]. ceMRI is suggested to have advantage over ceCT in detecting small liver metastases (< 10 mm); however, criticism has been directed towards it because the cost does not match the clinical benefit [24, 26, 27]. [18F]FDG PET/CT is not routinely indicated for initial staging of digestive tract tumours [11–13] because of its low sensitivity for liver metastasis, particularly in patients who have received preoperative chemotherapy [25, 28, 29]. The low background of the normal liver leads to the potential application of FAPI tracers in patients with suspected liver metastases [7, 30]. Previous studies with PET FAPI agents have indicated an outstanding role in the diagnosis of liver metastasis [7, 20]. Our findings likewise support the implementation of [99mTc]Tc-HFAPi SPECT/CT in the identification of liver metastasis. In metastatic lesions, the highest uptake of [99mTc]Tc-HFAPi were achieved in liver metastasis, with a median T/B ratio and SUVmax of 4.48 and 7.59, respectively. Basically, [99mTc]Tc-HFAPi demonstrated satisfactory sensitivity (88.2%) for detecting liver metastasis. Moreover, due to the low expression of FAP in benign liver lesions [31], an extremely high specificity (100%) of [99mTc]Tc-HFAPi was achieved. Four cases of suspected liver metastasis in ceCT were negatively detected by [99mTc]Tc-HFAPi with minimal uptake, and the lesions were proven to be benign by biopsy or multi-modality imaging, thus excluding from metastasis and restaging from M1 to M0 and allowing the chance for radical surgery. Overall, [99mTc]Tc-HFAPi provided an accurate diagnosis of suspected liver metastases, which may avoid unnecessary misdiagnosis, correct tumour staging and promote clinical oncological decisions.
In addition to liver metastasis, [68Ga]Ga-FAPI outperformed traditional imaging in detecting bone metastases [9]. In the present study, 2 patients with multiple skeletal metastases showed visible uptake of [99mTc]Tc-HFAPi, which was often missed by ceCT. These results further demonstrate the diagnostic advantage of [99mTc]Tc-HFAPi for various distant metastasis, thus improving the staging of cancer and treatment modification.
The expression of FAP is also widely reported to be avid in tissue modelling, wound healing, and inflammation-induced fibrosis [32]. Our study with 99mTc-labelled FAPI also demonstrated pulmonary tuberculosis with moderate uptake (T/B ratio of 4.36). Moreover, uterine fibroids demonstrated diffuse uptake of [99mTc]Tc-HFAPi, which might be attributed to the activated fibroblasts, as shown in previous PET imaging studies [33, 34].
Altogether 2 primary tumour lesions demonstrated false-negative uptake of [99mTc]Tc-HFAPi. One might be attributed to obviously low expression of FAP, which was revealed by following IHC staining [35], while the other one still had moderate expression on IHC. The mechanisms underlying the discordance between FAP expression and [99mTc]Tc-HFAPi uptake might be due to the liganding of molecules and certain physical influences, which might alter the conformation of some membrane proteins and their functional state (activation or inactivation) [36]. When it is in a nonactivated conformation, it is inaccessible to its targeted inhibitors. However, this still needs to be further confirmed.
We know that sensitivity and specificity are equally important when Youden’s index is used to obtain the best cut-off value. Previous study adopted cut-off value based on the highest Youden’s index [37]. However, a fixed cut-off value often does not meet the needs of clinical decision-making. Different cut-off values are needed according to different disease states and clinical purposes, just as previous study indicated [38]. Here, we listed a series of cut-off values for reference, because larger samples and data from other tumour histotypes and different clinical statuses with [99mTc]Tc-HFAPi are needed to optimize this criterion.
There are several limitations to this study. First, a small patient cohort limited the statistical significance for some kinds of cancers, such as oesophageal, pancreatic, and gallbladder cancer. Second, although being the ideal reference standard, histopathological examination was not available in all lesions because of ethical and technical reasons. Third, further prospective studies with larger populations in head-to-head comparisons of [99mTc]Tc-HFAPi SPECT/CT and [68Ga]Ga-FAPI PET/CT are warranted to best comment on the superiority of the tracers to clarify the role of SPECT/CT.
Despite these limitations, to the best of our knowledge, this article might be the first application of a new 99mTc-labelled FAPI for digestive system tumours from a clinical perspective, and we confirmed its diagnostic efficacy in tumour staging and restaging, providing an important basis for clinical application and subsequent studies. Furthermore, our 99mTc-labelled FAPI might provide some future directions for drug labelling with 188Re, such as integration in FAP targeted diagnosis and targeted radionuclide therapy [10].
Conclusion
In this work, we have developed a new 99mTc-labelled molecular probe and transformed it for the first time for digestive system tumours study. The findings indicate selective uptake of [99mTc]Tc-HFAPi SPECT/CT and demonstrate a high target-to-background ratio for various types of digestive system cancers as well as related metastasis, especially liver metastasis, which contributes to the current literature on FAP inhibitor molecular imaging. Further studies with large populations and other cancer types should be done to draw firmer conclusions on the superiority of the tracers.
Supplementary Information
Below is the link to the electronic supplementary material.
Author contribution
Rui Gao conceived of the study and participated in its design and is responsible for the development of clinical research. Xi Jia and Xinru Li is primarily involved in registration of clinical trials, results analysis, drafting the manuscript and substantively revising. Bing Jia is responsible for the designing and guidance of preclinical drug related experiments. Ye Yang, Ting Ji and Xin Xie are responsible for medical history collection and data processing. Yuanbo Wang and Yan Liu prepared the [99mTc]Tc-HFAPi and collected SPECT/CT images. Yu Yao, Guanglin Qiu, Huixing Deng and Aimin Yang are responsible for providing clinical cases and MDT discussion. Zhaohui Zhu is responsible for manuscript review and proofreading. Si Chen provides algorithm of SUVmax calculating. All authors read and approved the final manuscript.
Data availability
The datasets used or analyzed during the current study are available from the corresponding authors on reasonable request.
Declarations
Ethics approval and consent to participate
This study was approved by the Clinical Research Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University (Ethic approved No.: XJTU1AF2021LSK-021) and Chinese Clinical Trial Register (Registration No.: ChiCTR2100048093), and all the patients gave written, informed consent before the study.
Consent for publication
All authors of the current manuscript meet the specified criteria for authorship and agreed to publish this manuscript.
Conflict of interest
The authors declare no competing interests.
Footnotes
This article is part of the Topical Collection on Oncology - Digestive tract
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xi Jia, Xinru Li and Bing Jia contributed equally to this work.
References
- 1.Chen X, Song E. Turning foes to friends: targeting cancer-associated fibroblasts. Nat Rev Drug Discov. 2019;18(2):99–115. doi: 10.1038/s41573-018-0004-1. [DOI] [PubMed] [Google Scholar]
- 2.Kobayashi H, Enomoto A, Woods SL, Burt AD, Takahashi M, Worthley DL. Cancer-associated fibroblasts in gastrointestinal cancer. Nat Rev Gastro Hepat. 2019;16(5):282–295. doi: 10.1038/s41575-019-0115-0. [DOI] [PubMed] [Google Scholar]
- 3.Lo A, Li C, Buza EL, Blomberg R, Govindaraju P, Avery D, et al. Fibroblast activation protein augments progression and metastasis of pancreatic ductal adenocarcinoma. JCI Insight. 2017;2(19). 10.1172/jci.insight.92232. [DOI] [PMC free article] [PubMed]
- 4.Lo A, Wang LS, Scholler J, Monslow J, Avery D, Newick K, et al. Tumor-promoting desmoplasia is disrupted by depleting FAP-expressing stromal cells. Cancer Res. 2015;75(14):2800–2810. doi: 10.1158/0008-5472.CAN-14-3041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Altmann A, Haberkorn U, Siveke J. The latest developments in imaging of fibroblast activation protein. J Nucl Med. 2021;62(2):160–167. doi: 10.2967/jnumed.120.244806. [DOI] [PubMed] [Google Scholar]
- 6.Chen H, Pang Y, Wu J, Zhao L, Hao B, Wu J, et al. Comparison of [68Ga]Ga-DOTA-FAPI-04 and [18F] FDG PET/CT for the diagnosis of primary and metastatic lesions in patients with various types of cancer. Eur J Nucl Med Mol I. 2020;47(8):1820–1832. doi: 10.1007/s00259-020-04769-z. [DOI] [PubMed] [Google Scholar]
- 7.Koerber SA, Staudinger F, Kratochwil C, Adeberg S, Haefner MF, Ungerechts G, et al. The role of 68Ga-FAPI PET/CT for patients with malignancies of the lower gastrointestinal tract: first clinical experience. J Nucl Med. 2020;61(9):1331–1336. doi: 10.2967/jnumed.119.237016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Loktev A, Lindner T, Mier W, Debus J, Altmann A, Jäger D, et al. A tumor-imaging method targeting cancer-associated fibroblasts. J Nucl Med. 2018;59(9):1423–1429. doi: 10.2967/jnumed.118.210435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen H, Zhao L, Ruan D, Pang Y, Hao B, Dai Y, et al. Usefulness of [68Ga]Ga-DOTA-FAPI-04 PET/CT in patients presenting with inconclusive [18F]FDG PET/CT findings. Eur J Nucl Med Mol I. 2021;48(1):73–86. doi: 10.1007/s00259-020-04940-6. [DOI] [PubMed] [Google Scholar]
- 10.Lindner T, Altmann A, Krämer S, Kleist C, Loktev A, Kratochwil C, et al. Design and development of 99mTc-labeled FAPI tracers for SPECT imaging and 188Re therapy. J Nucl Med. 2020;61(10):1507–1513. doi: 10.2967/jnumed.119.239731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Network NCCN, Clinical Practice Guidelines in Oncology. Colon Cancer, Version 3.2021. https://www.nccn.org/professionals/physician_gls/pdf/colon.pdf. Accessed 10 Sept 2021.
- 12.Network NCCN, Clinical Practice Guidelines in Oncology. Gastric Cancer, Version 1.2022. https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf. Accessed 20 Dec 2021.
- 13.Network NCCN, Clinical Practice Guidelines in Oncology. Rectal Cancer, Version 2.2021. https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf. Accessed 10 Sept 2021.
- 14.Jin X, Liang N, Wang M, Meng Y, Jia B, Shi X, et al. Integrin imaging with 99mTc-3PRGD2 SPECT/CT shows high specificity in the diagnosis of lymph node metastasis from non–small cell lung cancer. Radiology. 2016;281(3):958–966. doi: 10.1148/radiol.2016150813. [DOI] [PubMed] [Google Scholar]
- 15.Guanghui C, Shi G, Tiefeng JI, Qingjie MA, Bing J, Zuowei C, et al. Pharmacokinetics and radiation dosimetry of 99mTc-3PRGD2 in healthy individuals: a pilot study. Nucl Sci Tech. 2012;23(6):349–354. [Google Scholar]
- 16.Chen S. Method and system for calculating SUV normalization coefficient in a SPECT quantitative tomographic image. In. U.S. ed, 2020. https://pubchem.ncbi.nlm.nih.gov/patent/US-11189374-B2.
- 17.Urbán S, Meyer C, Dahlbom M, Farkas I, Sipka G, Besenyi Z, et al. Radiation dosimetry of 99mTc-PSMA I&S: a single-center prospective study. J Nucl Med. 2021;62(8):1075–1081. doi: 10.2967/jnumed.120.253476. [DOI] [PubMed] [Google Scholar]
- 18.Giesel FL, Heussel CP, Lindner T, Röhrich M, Rathke H, Kauczor H, et al. FAPI-PET/CT improves staging in a lung cancer patient with cerebral metastasis. Eur J Nucl Med Mol I. 2019;46(8):1754–1755. doi: 10.1007/s00259-019-04346-z. [DOI] [PubMed] [Google Scholar]
- 19.Giesel FL, Kratochwil C, Lindner T, Marschalek MM, Loktev A, Lehnert W, et al. 68Ga-FAPI PET/CT: biodistribution and preliminary dosimetry estimate of 2 DOTA-containing FAP-targeting agents in patients with various cancers. J Nucl Med. 2019;60(3):386–392. doi: 10.2967/jnumed.118.215913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pang Y, Zhao L, Luo Z, Hao B, Wu H, Lin Q, et al. Comparison of 68Ga-FAPI and 18F-FDG uptake in gastric, duodenal, and colorectal cancers. Radiology. 2021;298(2):393–402. doi: 10.1148/radiol.2020203275. [DOI] [PubMed] [Google Scholar]
- 21.Gilardi L, Airo FL, Demirci E, Clerici I, Omodeo SE, Ceci F. Imaging cancer-associated fibroblasts (CAFs) with FAPi PET. Biomedicines. 2022;10(3). 10.3390/biomedicines10030523. [DOI] [PMC free article] [PubMed]
- 22.Li M, Younis MH, Zhang Y, Cai W, Lan X. Clinical summary of fibroblast activation protein inhibitor-based radiopharmaceuticals: cancer and beyond. Eur J Nucl Med Mol Imaging. 2022;49(8):2844–2868. doi: 10.1007/s00259-022-05706-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hess KR, Varadhachary GR, Taylor SH, Wei W, Raber MN, Lenzi R, et al. Metastatic patterns in adenocarcinoma. Cancer-Am Cancer Soc. 2006;106(7):1624–1633. doi: 10.1002/cncr.21778. [DOI] [PubMed] [Google Scholar]
- 24.Sivesgaard K, Larsen LP, Sørensen M, Kramer S, Schlander S, Amanavicius N, et al. Diagnostic accuracy of CE-CT, MRI and FDG PET/CT for detecting colorectal cancer liver metastases in patients considered eligible for hepatic resection and/or local ablation. Eur Radiol. 2018;28(11):4735–4747. doi: 10.1007/s00330-018-5469-0. [DOI] [PubMed] [Google Scholar]
- 25.Chan K, Welch S, Walker-Dilks C, Raifu A. Evidence-based guideline recommendations on the use of positron emission tomography imaging in colorectal cancer. Clin Oncol-Uk. 2012;24(4):232–249. doi: 10.1016/j.clon.2011.11.008. [DOI] [PubMed] [Google Scholar]
- 26.Granata V, Catalano O, Fusco R, Tatangelo F, Rega D, Nasti G, et al. The target sign in colorectal liver metastases: an atypical Gd-EOB-DTPA “uptake” on the hepatobiliary phase of MR imaging. Abdom Imaging. 2015;40(7):2364–2371. doi: 10.1007/s00261-015-0488-7. [DOI] [PubMed] [Google Scholar]
- 27.Granata V, Fusco R, de Lutio Di Castelguidone E, Avallone A, Palaia R, Delrio P, et al. Diagnostic performance of gadoxetic acid–enhanced liver MRI versus multidetector CT in the assessment of colorectal liver metastases compared to hepatic resection. BMC Gastroenterol. 2019;19(1):129. 10.1186/s12876-019-1036-7. [DOI] [PMC free article] [PubMed]
- 28.Monteil J, Le Brun-Ly V, Cachin F, Zasadny X, Seitz J, Mundler O, et al. Comparison of 18FDG-PET/CT and conventional follow-up methods in colorectal cancer: a randomised prospective study. Digest Liver Dis. 2021;53(2):231–237. doi: 10.1016/j.dld.2020.10.012. [DOI] [PubMed] [Google Scholar]
- 29.Maffione AM, Lopci E, Bluemel C, Giammarile F, Herrmann K, Rubello D. Diagnostic accuracy and impact on management of 18F-FDG PET and PET/CT in colorectal liver metastasis: a meta-analysis and systematic review. Eur J Nucl Med Mol I. 2015;42(1):152–163. doi: 10.1007/s00259-014-2930-4. [DOI] [PubMed] [Google Scholar]
- 30.Giesel FL, Kratochwil C, Schlittenhardt J, Dendl K, Eiber M, Staudinger F, et al. Head-to-head intra-individual comparison of biodistribution and tumor uptake of 68Ga-FAPI and 18F-FDG PET/CT in cancer patients. Eur J Nucl Med Mol I. 2021;48(13):4377–4385. doi: 10.1007/s00259-021-05307-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shi X, Xing H, Yang X, Li F, Yao S, Congwei J, et al. Comparison of PET imaging of activated fibroblasts and 18F-FDG for diagnosis of primary hepatic tumours: a prospective pilot study. Eur J Nucl Med Mol I. 2021;48(5):1593–1603. doi: 10.1007/s00259-020-05070-9. [DOI] [PubMed] [Google Scholar]
- 32.Windisch P, Zwahlen DR, Giesel FL, Scholz E, Lugenbiel P, Debus J, et al. Clinical results of fibroblast activation protein (FAP) specific PET for non-malignant indications: systematic review. EJNMMI Res. 2021;11(1):18. doi: 10.1186/s13550-021-00761-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mona CE, Benz MR, Hikmat F, Grogan TR, Lueckerath K, Razmaria A, et al. Correlation of 68Ga-FAPi-46 PET biodistribution with FAP expression by immunohistochemistry in patients with solid cancers: interim analysis of a prospective translational exploratory study. J Nucl Med. 2022;63(7):1021–1026. doi: 10.2967/jnumed.121.262426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kessler L, Ferdinandus J, Hirmas N, Zarrad F, Nader M, Kersting D, et al. Pitfalls and common findings in 68Ga-FAPI PET: a pictorial analysis. J Nucl Med. 2022;63(6):890–896. doi: 10.2967/jnumed.121.262808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lin R, Lin Z, Chen Z, Zheng S, Zhang J, Zang J, et al. [(68)Ga]Ga-DOTA-FAPI-04 PET/CT in the evaluation of gastric cancer: comparison with [(18)F]FDG PET/CT. Eur J Nucl Med Mol Imaging. 2022;49(8):2960–2971. doi: 10.1007/s00259-022-05799-5. [DOI] [PubMed] [Google Scholar]
- 36.Chen Y, Lee H, Tong H, Schwartz M, Zhu C. Force regulated conformational change of integrin αVβ3. Matrix Biol. 2017;60–61:70–85. doi: 10.1016/j.matbio.2016.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang Y, Tang Y, Gao X, Gan Y, Hu S, Chen MF, et al. Optimization of prostate cancer patient lymph node staging via the integration of neutrophil-lymphocyte ratios, platelet-lymphocyte ratios, and (68)Ga-PSMA-PET-derived SUVmax values. Prostate. 2022;82(15):1415–1421. doi: 10.1002/pros.24415. [DOI] [PubMed] [Google Scholar]
- 38.Hoffmann MA, Muller-Hubenthal J, Rosar F, Fischer N, von Eyben FE, Buchholz HG, et al. Primary staging of prostate cancer patients with [(18)F]PSMA-1007 PET/CT compared with [(68)Ga]Ga-PSMA-11 PET/CT. J Clin Med. 2022;11(17). 10.3390/jcm11175064. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used or analyzed during the current study are available from the corresponding authors on reasonable request.