Abstract
Small intestinal bacterial overgrowth is a small bowel disorder characterised by excessive amounts of bacteria populating the small intestine leading to symptoms of abdominal pain, bloating and change in bowel habit. This creates some degree of diagnostic uncertainty due to the overlap of these symptoms with numerous other gastrointestinal conditions. Quantitative culture of jejunal aspirates is the gold standard diagnostic test but has largely been replaced by glucose and lactulose breath tests due to their relative ease and accessibility. The approach to treatment centres around reducing bacterial numbers through antibiotic therapy and managing any predisposing factors. Further research is required in order to define the optimum antibiotic choice and duration of therapy as well as the potential diagnostic utility of home breath testing and capsule-based technology.
Keywords: NUTRITION, SMALL BOWEL, BACTERIAL OVERGROWTH, ANTIBIOTIC THERAPY
Introduction
The human gut microbiota is an intricate ecosystem of microbes that numerically out populate cells of the host. Bacterial numbers are maintained by several mechanisms including small intestinal motility and integrity, gastric acid and pancreaticobiliary secretions and local immunity. Small intestinal bacterial overgrowth (SIBO) is a state of dysbiosis caused by perturbation of this complex environment and is defined as an increase in bacterial burden in the small bowel. In healthy individuals, small intestinal levels are thought to be approximately <104 colony forming units per millilitre (CFU/mL) with a bacterial colony count exceeding 105 CFU/mL3 compatible with a diagnosis of SIBO.1 2
As the clinical presentation of SIBO is often non-specific with abdominal pain, bloating and diarrhoea, it can go largely underdiagnosed making the estimation of its current prevalence all the more difficult. This review aims to summarise current knowledge on the aetiology, clinical manifestations and risk factors for SIBO as well as strategies for its effective diagnosis and treatment.
Aetiology and risk factors
When compared with the colon, the small intestine is home to relatively few bacteria due to its hostile environment. Though little in number, these enteric bacteria carry out several important functions in the host including modulation and education of the human immune system and protection from pathogens.3
Host homoeostatic processes aim to prevent overgrowth and maintain stable levels of intestinal bacteria. The action of gastric acid and pancreaticobiliary secretions alongside local immune cells help destroy bacteria. The normal structure and antegrade peristaltic activity of the small bowel itself works by trapping and sweeping bacteria towards the colon.1 4 It is the disruption of these processes which can predispose patients to the development of SIBO and these are subdivided into four main mechanisms (table 1):
Table 1.
Aetiology and associated risk factors for development of SIBO
| Gut dysmotility | Altered GI secretions | Anatomical alterations | Impaired immunity |
| Diabetes mellitus | Medications—proton pump inhibitors | Roux-en-Y gastric bypass | Hypogammaglobulinaemia |
| Connective tissue disorders for example, systemic sclerosis, Ehlers Danlos | Chronic pancreatitis | Strictures, adhesions, fistulae (Crohn’s) | Combined variable immunodeficiency |
| Visceral neuropathies and myopathies | Chronic liver disease | Ileocecal valve impairment /resection Gastrectomy |
|
| Medications—opioids, anticholinergics | Small bowel diverticular disease | ||
| Parkinson’s disease Radiation enteropathy |
GI, gastrointestinal; SIBO, small intestinal bacterial overgrowth.
Gut dysmotility.
Anatomical changes.
Altered gastrointestinal (GI) secretions.
Impaired gut immunity.
Gut dysmotility
Under normal physiological action the GI system co-ordinates a series of complex smooth muscle contractions known as the migrating motor complex (MMC) that is responsible for propagating food and debris through the GI tract during periods of fasting. This electromechanical process additionally serves an important function by pushing bacteria further down the gut to prevent stagnation and thereby any opportunity of colonisation.5 Decreased MMC activity is associated with an increased prevalence of SIBO.6 Chander Roland et al found mean small bowel transit times in SIBO were almost double that of controls, 7.89 hours and 3.93 hours, respectively (p=0.02)7 suggesting that small bowel stasis may play a significant role.8
Small bowel motility may be reduced by intrinsic damage of enteric nerves and muscles or by the continued use of medications such as opioids and anticholinergics, which may diminish MMC activity.9
Visceral neuropathies are a long-term complication of poorly controlled diabetes and damaged enteric nerves can result in gastroparesis and impaired gastric motility predisposing to SIBO development. Delayed gastric emptying and prolonged transit time are also common findings in patients with Parkinson’s disease (PD) thereby favouring colonisation and proliferation of intestinal bacteria. In one study of 103 patients with PD, 25% were found to have SIBO.10 SIBO rates are as high as 40% in patients with systemic sclerosis, a multisystem connective tissue disorder which can present with GI dysmotility and dysphagia.11 12
Altered GI secretions
Normal physiological pH of the stomach ranges from 1 to 3 and is maintained by the release of acid from the gastric parietal cells. This acidic environment forms the fundamental backbone of the gastric barrier which kills approximately 99% of all bacteria preventing colonisation in the stomach and proximal duodenum so its failure may result in SIBO development.9 13
Hypochlorhydria or achlorhydria, typically from prolonged use of acid suppressants such as histamine-2 receptor antagonists and proton pump inhibitors (PPIs) may be a contributing factor.14 15 A meta-analysis of 19 observational studies conducted by Su et al in 2018 showed a statistically significant increased risk of SIBO and PPI use OR 1.71 (95% CI 1.2 to 2.4).16
The pancreas serves an integral role in digestion with the secretion of a key enzymes including proteases and lipases. These enzymes also help in maintaining low bacterial numbers in the upper GI tract through its antibacterial properties and indirect action of stimulating anterograde gut motility. The prevalence of SIBO is therefore increased in states of impaired pancreatic exocrine function such as chronic pancreatitis (CP).17 18 Patients with CP have numerous additional risk factors for SIBO including diabetes, medications such as opioids and PPIs, alcohol consumption as well as GI surgery, with the latter associated with the greatest increased risk. A meta-analysis showed a pooled prevalence of SIBO in non-surgical CP patients was 25.7% (95% CI 8.1% to 57.6%) compared with 54.1% (95% CI 23.2% to 82.1%) in CP patients with a GI surgical history.17
Bile also has antimicrobial properties, and a reduction in its production as seen in conditions such as chronic liver disease and cirrhosis may lead to increased susceptibility to SIBO.19 Gut motility may also be impaired in cirrhosis leading to greater intestinal transit time.20
Anatomical abnormalities
Both congenital and acquired distortions in the normal anatomical structure of the small intestine can predispose to SIBO. This can happen as a result of obstructed passage or damage of enteric neuronal pathways causing ineffective clearance of the small bowel with the net effect of promoting colonisation of bacteria. Diverticula in the small bowel though mostly asymptomatic and incidental can conceal and grow bacteria in their respective outpouchings.
SIBO is a recognised sequelae of Roux-en-Y gastric bypass (RYGB) surgery which creates a blind intestinal loop that precipitates stasis of bacteria and shielding from gastric acid.13 In one study, RYGB patients were more than twice as likely to have a positive glucose breath test (GBT) when compared with those with normal GI anatomy (73.4% vs 36%, respectively).21
Inflammatory bowel disease has a strong association with SIBO. Results from a recent meta-analysis showed the odds of SIBO prevalence determined by a positive hydrogen BT in those with Crohn’s and ulcerative colitis was 10.86 (95% CI 2.76 to 42.69) and 7.99 (95% CI 1.66 to 38.35), respectively.22 Numerous factors predispose this patient group to developing bacterial overgrowth including the formation of fistulae, adhesions and strictures as seen in Crohn’s disease.9 Furthermore, impaired ileocecal valve function is a well-documented risk factor for SIBO.8 23 Damage to the valve either by surgery or IBD itself directly allows retrograde translocation of colonic bacteria. A meta-analysis found that in 194 patients with Crohn’s disease that had undergone resection of the ileocecal valve disease, the prevalence of SIBO was 33% (95% CI 19.2% to 50.6%).22
Impaired immunity
At the local level, the gut immune system is a diverse array of immune cells, physical and chemical barriers whose sole function is to protect and prevent perturbation of the gut microbiome. Paneth cells found at the base of intestinal crypts secrete defensins, antimicrobial peptides which contribute to the innate mucosal barrier.24 In addition, secretory IgA antibodies play a central role in maintaining low bacterial numbers by blocking attachment and preventing proliferation in the small bowel.25 Such tight and regulated control of microbiota composition is therefore preordained by normal functioning of the immune system and disruption of this such as in common variable immunodeficiency, hypogammaglobulinaemia and IgA deficiency increases the risk of SIBO development.26 27
Clinical features
Commonly SIBO presents with a range of non-specific symptoms including weight loss, bloating, flatulence, diarrhoea, abdominal pain and distention mimicking other GI conditions. The frequency of each of these symptoms however varies among patients. Diagnosing SIBO based on clinical symptomology alone is therefore difficult but there should be a higher index of suspicion in patients with predisposing risk factors.3 13
Chronic diarrhoea and bloating are the most commonly reported symptoms with the diarrhoea seen in SIBO thought to be a consequence of increased deconjugation of bile acids by intestinal bacteria leading to fat malabsorption.28 Bacterial metabolism is another driver of diarrhoea with the production of several osmotically active substances including short chain fatty acids.29 The BSG guidelines on chronic diarrhoea suggest empirical treatment in patients with suggestive symptoms and clear aetiological risk factors for SIBO.30
With increased number of bacteria in the small bowel, fermentation also occurs at much greater levels and is the process behind post prandial bloating. What follows bloating is the false sense of satiety and may be accompanied by nausea and discomfort. Over time patients begin to avoid eating in order to prevent these symptoms as a form of learnt behaviour which can eventually result in poor nutritional intake and weight loss.18
Vitamin B12 deficiency can be a consequence of SIBO. This occurs as a result of utilisation of the vitamin by bacteria. It is partially metabolised to inactive analogues, which compete with normal vitamin B12 binding and absorption.18
Investigations
Small bowel aspirate
Small bowel aspirate via endoscopy from the duodenum or jejunum is often considered the ‘gold standard’ in establishing a diagnosis of SIBO. A bacterial concentration of >103 CFUs/mL in a small bowel aspirate culture is diagnostic of SIBO. The most common species identified include Bacteroides, Enterococcus and Lactobacillus.31 This method has several pitfalls and is not typically used due to the invasiveness of the procedure, time consumption and the cost implications.
Limitations include difficulty in aspirating a sufficient sample, risks of gastric or oropharyngeal cross-contamination of the sample and the low yield of bacterial cultures which requires careful microbiological technique, despite which only approximately 40% of the total gut flora can be identified using conventional culture methods.32 However, as strengthening of microbiological analysis of endoscopic sampling improves this is a promising area for further research.4 8
Hydrogen breath tests
Breath tests (BT) is considered the first line investigation as it is non-invasive, safe and easy to perform and more so as home testing kits are now available. This test works on the principle that the metabolism of a substrate (typically a carbohydrate) by the bacterial flora will lead to a production of hydrogen or methane which is absorbed and excreted in the breath and is then subsequently measured.
The most common substrate used is a readily metabolised carbohydrate such as lactulose, fructose and glucose in preference to complex carbohydrates which have been found to be less sensitive and specific.33 Lactulose is a non-absorbable substance that is normally metabolised by gut bacteria in the colon leading to the production of hydrogen and/or methane. In individuals without SIBO, the administration of lactulose results in a single peak in breath hydrogen/methane within 2–3 hours due to the metabolism of lactulose by colonic flora (figure 1). In patients with SIBO, administration of lactulose results in an early peak in breath hydrogen/methane levels due to metabolism by small bowel bacteria (figure 2).34 Under normal circumstances glucose is rapidly absorbed from the proximal small bowel but when used as a substrate in the presence of SIBO, it is metabolised to hydrogen in the small bowel lumen prior to absorption.
Figure 1.
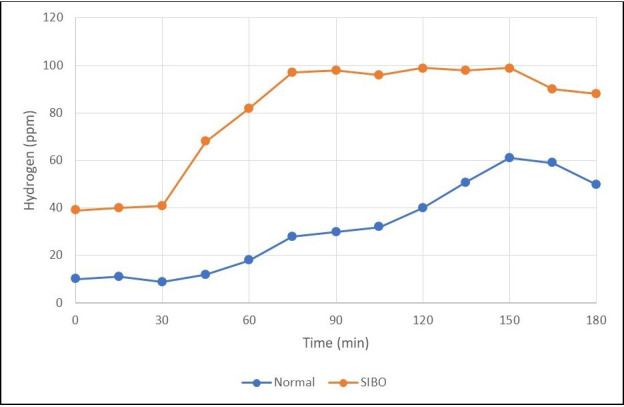
Breath test in a healthy patient versus a patient with small bowel intestinal overgrowth (SIBO) showing an earlier rise in hydrogen.
Figure 2.
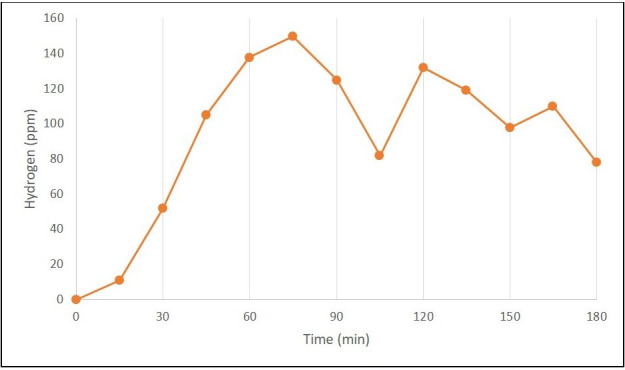
Breath test in a patient with Small Bowel Intestinal Overgrowth with double peak representing the early rise seen in the small intestine followed by a short duration fall in hydrogen before the expected rise in hydrogen in the large bowel.
Currently, there is no universally accepted diagnostic criteria for a positive hydrogen BT.
The North American consensus has proposed an interpretation for lactulose BT results suggesting that two peaks is not required for the diagnosis of SIBO which concurs with the UK Association of Gastrointestinal Physiologists. Their other recommendations commonly used for diagnosis are summarised in box 1: 9 35
Box 1. Interpretation of breath testing results—recommendations.
1. A rise of ≥20 ppm from baseline in hydrogen during the test should be considered positive for fructose and lactose breath testing.
2. A rise of ≥20 ppm from baseline in hydrogen by 90 min min should be considered a positive test for small intestinal bacterial overgrowth.
3. A level of ≥10 ppm be considered positive for methane on a breath test.
Breath test has a lower sensitivity (glucose 44%, lactulose 31%) but higher specificity (glucose 80%, lactulose 86%) when compared with small bowel aspirate via endoscopy.52
Several substrates are used with varying sensitivity and specificity and the best substrate is yet to be identified.
Capsule-based technology
A novel capsule-based technology; that can provide real time measurements for SIBO is currently under development. Intraluminal gases such as hydrogen, carbon dioxide and methane are measured as the capsule travels through the gut using sensors and measuring conductivity.36 Ultrasound imaging can be used as an accurate marker for the location of the capsule. A single-blinded trial in 12 healthy subjects evaluating capsule endoscopy vs BT concluded that the capsule had a high sensitivity in measuring luminal hydrogen concentrations, providing information on the site of intestinal gas production while demonstrating safety and reliability.37
Management
Diet and lifestyle
A key principle in the management of SIBO is to identify and treat any predisposing factors using a combination of dietary, lifestyle, medical and surgical therapies. Lifestyle measures include avoidance of sugar, reduction in NSAID use and smoking cessation.38 Fermentable oligosaccharides, disaccharides, and monosaccharides and polyols (FODMAPs) are short-chain carbohydrates that are poorly absorbed and are osmotically active in the intestinal lumen where they are rapidly fermented by small intestinal bacteria. A diet low in FODMAPs improves bloating and gas in patients with irritable bowel syndrome so may also help in symptomatic SIBO patients but should be delivered by a trained dietician.39 40
Antibiotics
The mainstay of therapy for SIBO is antibiotics to reduce rather than eradicate the burden of small intestinal bacteria and to reverse the mucosal inflammation associated with overgrowth and malabsorption.41
Overall antibiotics normalise SIBO in 51.1% of patients with abnormal BTs, which is significantly higher than placebo (9.8%).42 Antibiotic choice is influenced by local antibiotic resistance rates, cost, patient allergies and risk factors for drug-resistance (recent or repeated prior exposure). The BSG advocates an empirical trial for those with a high-test probability for SIBO.43
Rifaximin 550 mg two times per day for 7–14 days has the strongest evidence base for treatment of SIBO but other antibiotic treatments shown to have an effective response include ciprofloxacin 500 mg two times per day, metronidazole 250 mg three times a day, trimethoprim 160/800 mg two times per day and norfloxacin 400 mg four times a day.44
A meta-analysis of rifaximin efficacy in SIBO treatment which included 32 clinical trials (7 RCT, 24 cohort and 1 cross-over trial) comprising a total of 1331 patients45 found the response rate to Rifaximin therapy with follow-up BT to vary from 61% to 78%.31 Early studies with ciprofloxacin, metronidazole, tetracycline, amoxicillin-clavulanic acid, trimethoprim and norfloxacin suggested these antibiotics to be equally as effective, in particular metronidazole. They all reported symptom improvement with treatment compared with placebo and on repeat BT.46 Later studies reversed this opinion and concluded that clinical response rates may be higher or equal with rifaximin.47
A second course of antibiotics is offered to patients if they have a partial improvement in symptoms or an early recurrence (<3 months) and in these cases a different antibiotic therapy should be prescribed. The most commonly used antibiotics which have reported the best complete improvement of symptoms alongside reduction of recurrence of <3 months has been seen with rifaximin and vancomycin.31 48 Recurrence after one course of antibiotic therapy has been shown to be more likely among older patients, a surgical history of appendectomy and those with a history of long-term PPI use, for reasons which are unclear.49
Prophylactic antibiotics should be considered in those patients who have had repeated courses of antibiotic treatment for four or more distinct episodes within a year.45 The typical prophylactic regimen is two or three different low-dose long-term antibiotics that should be rotated 5–10 days out of every month or every other week and given for 10–14 days. There are unfortunately no controlled trials to guide the duration of treatment or management of recurrent SIBO and recommendations are commonly based on clinical experience.48 A recently published study from 2021 compared the effectiveness of rotating antibiotics vs a single course and found it to be more effective in maintaining remission (70.8% vs 50.8%), improving quality of life and reducing bloating. This benefit may be more beneficial in those with anatomical of physiological risk factors but further research is required to elucidate these effects.50 Adverse events from recurrent antibiotic use including diarrhoea, abdominal pain and C. diff infection are fairly uncommon and occur in less than 5% of patients.45 Patients with no improvement in symptoms after two courses of antibiotic therapy or progressive symptoms should be evaluated for alternative diagnoses such as coeliac disease and IBS.
The role of probiotics
Several studies have looked at the effect of probiotics on SIBO, but these studies lack consistency not only in the formulations used but also in the duration of treatment, populations assessed and diagnostic methods implemented. A meta-analysis of 18 studies showed no significant difference in the improvement of symptoms of patients with SIBO on probiotics as compared with the control group.51
Conclusion
Clinicians should consider a diagnosis of SIBO in patients with abdominal bloating, change in bowel habit and predisposing risk factors such as gut dysmotility, anatomical changes, altered GI secretions and impaired gut immunity.
BT are used as they are a cheaper and non-invasive diagnostic test compared with small bowel aspirate, but results should be interpreted with caution given their limited sensitivity, specificity and no universally accepted diagnostic criteria.
Furthermore, with no established regimen for treatment, an empirical approach is often adopted. Rifaximin is the most accepted first line antibiotic therapy for SIBO, but its use may be limited due to its high cost and availability. Treatments are commonly heterogeneous which also highlight the need for further research to establish an optimum antibiotic regimen and provide guidance on administering prophylactic treatment.
Key points.
Small intestinal bacterial overgrowth can arise as a consequence of disorders in small intestinal motility, gastric acid and pancreaticobiliary secretion, local immunity and alterations in the anatomy of the gastrointestinal tract.
Chronic diarrhoea and bloating are the most common reported symptoms, and the index of suspicion should be high in those with predisposing risk factors.
Breath testing is considered the first line investigation although the sensitivity and specificity of the test is low.
Antibiotics are the mainstay of therapy, but a clear universal protocol is yet to be established.
Footnotes
Contributors: JFA and PP co-wrote the article. AR conceptualised and provided oversight and overall supervision of this manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Not applicable.
References
- 1. Bures J, Cyrany J, Kohoutova D, et al. Small intestinal bacterial overgrowth syndrome. World J Gastroenterol 2010;16:2978–90. 10.3748/wjg.v16.i24.2978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Singh VV, Toskes PP. Small bowel bacterial overgrowth: presentation, diagnosis, and treatment. Curr Gastroenterol Rep 2003;5:365–72. 10.1007/s11894-003-0048-0 [DOI] [PubMed] [Google Scholar]
- 3. Jandhyala SM, Talukdar R, Subramanyam C, et al. Role of the normal gut microbiota. World J Gastroenterol 2015;21:8787–803. 10.3748/wjg.v21.i29.8787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Dukowicz AC, Lacy BE, Levine GM. Small intestinal bacterial overgrowth: a comprehensive review. Gastroenterol Hepatol 2007;3:112–22. [PMC free article] [PubMed] [Google Scholar]
- 5. Stotzer PO, Björnsson ES, Abrahamsson H. Interdigestive and postprandial motility in small-intestinal bacterial overgrowth. Scand J Gastroenterol 1996;31:875–80. 10.3109/00365529609051995 [DOI] [PubMed] [Google Scholar]
- 6. Pimentel M, Soffer EE, Chow EJ, et al. Lower frequency of MMC is found in IBS subjects with abnormal lactulose breath test, suggesting bacterial overgrowth. Dig Dis Sci 2002;47:2639–43. 10.1023/A:1021039032413 [DOI] [PubMed] [Google Scholar]
- 7. Roland BC, Ciarleglio MM, Clarke JO, et al. Small intestinal transit time is delayed in small intestinal bacterial overgrowth. J Clin Gastroenterol 2015;49:571–6. 10.1097/MCG.0000000000000257 [DOI] [PubMed] [Google Scholar]
- 8. Chander Roland B, Mullin GE, Passi M, et al. A prospective evaluation of ileocecal valve dysfunction and intestinal motility derangements in small intestinal bacterial overgrowth. Dig Dis Sci 2017;62:3525–35. 10.1007/s10620-017-4726-4 [DOI] [PubMed] [Google Scholar]
- 9. Reddymasu SC, Sostarich S, McCallum RW. Small intestinal bacterial overgrowth in irritable bowel syndrome: are there any predictors? BMC Gastroenterol 2010;10:23. 10.1186/1471-230X-10-23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Tan AH, Mahadeva S, Thalha AM, et al. Small intestinal bacterial overgrowth in Parkinson's disease. Parkinsonism Relat Disord 2014;20:535–40. 10.1016/j.parkreldis.2014.02.019 [DOI] [PubMed] [Google Scholar]
- 11. Morrisroe K, Baron M, Frech T, et al. Small intestinal bacterial overgrowth in systemic sclerosis. J Scleroderma Relat Disord 2020;5:33–9. 10.1177/2397198319863953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Polkowska-Pruszyńska B, Gerkowicz A, Szczepanik-Kułak P, et al. Small intestinal bacterial overgrowth in systemic sclerosis: a review of the literature. Arch Dermatol Res 2019;311:1–8. 10.1007/s00403-018-1874-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Bohm M, Siwiec RM, Wo JM,. Diagnosis and management of small intestinal bacterial overgrowth. Nutr Clin Pract 2013;28:289–99. 10.1177/0884533613485882 [DOI] [PubMed] [Google Scholar]
- 14. Ratuapli SK, Ellington TG, O'Neill M-T, et al. Proton pump inhibitor therapy use does not predispose to small intestinal bacterial overgrowth. Am J Gastroenterol 2012;107:730–5. 10.1038/ajg.2012.4 [DOI] [PubMed] [Google Scholar]
- 15. Revaiah PC, Kochhar R, Rana SV, et al. Risk of small intestinal bacterial overgrowth in patients receiving proton pump inhibitors versus proton pump inhibitors plus prokinetics. JGH Open 2018;2:47–53. 10.1002/jgh3.12045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Su T, Lai S, Lee A, et al. Meta-Analysis: proton pump inhibitors moderately increase the risk of small intestinal bacterial overgrowth. J Gastroenterol 2018;53:27–36. 10.1007/s00535-017-1371-9 [DOI] [PubMed] [Google Scholar]
- 17. Capurso G, Signoretti M, Archibugi L, et al. Systematic review and meta‐analysis: small intestinal bacterial overgrowth in chronic pancreatitis. United European Gastroenterol J 2016;4:697–705. 10.1177/2050640616630117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Zaidel O, Lin HC. Uninvited guests: the impact of small intestinal bacterial overgrowth on nutritional status. Pract Gastroenterol 2003;27:27–33. [Google Scholar]
- 19. Ghosh G, Jesudian AB. Small intestinal bacterial overgrowth in patients with cirrhosis. J Clin Exp Hepatol 2019;9:257–67. 10.1016/j.jceh.2018.08.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kalaitzakis E, Sadik R, Holst JJ, et al. Gut transit is associated with gastrointestinal symptoms and gut hormone profile in patients with cirrhosis. Clin Gastroenterol Hepatol 2009;7:346–52. 10.1016/j.cgh.2008.11.022 [DOI] [PubMed] [Google Scholar]
- 21. Dolan RD, Baker J, Harer K, et al. Small intestinal bacterial overgrowth: clinical presentation in patients with Roux-en-Y gastric bypass. Obes Surg 2021;31:564–9. 10.1007/s11695-020-05032-y [DOI] [PubMed] [Google Scholar]
- 22. Shah A, Morrison M, Burger D, et al. Systematic review with meta-analysis: the prevalence of small intestinal bacterial overgrowth in inflammatory bowel disease. Aliment Pharmacol Ther 2019;49:624–35. 10.1111/apt.15133 [DOI] [PubMed] [Google Scholar]
- 23. Roland BC, Ciarleglio MM, Clarke JO, et al. Low ileocecal valve pressure is significantly associated with small intestinal bacterial overgrowth (SIBO). Dig Dis Sci 2014;59:1269–77. 10.1007/s10620-014-3166-7 [DOI] [PubMed] [Google Scholar]
- 24. Salzman NH. Paneth cell defensins and the regulation of the microbiome: détente at mucosal surfaces. Gut Microbes 2010;1:401–6. 10.4161/gmic.1.6.14076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Mantis NJ, Forbes SJ. Secretory IgA: arresting microbial pathogens at epithelial borders. Immunol Invest 2010;39:383–406. 10.3109/08820131003622635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Jones RM, Neish AS. Recognition of bacterial pathogens and mucosal immunity. Cell Microbiol 2011;13:670–6. 10.1111/j.1462-5822.2011.01579.x [DOI] [PubMed] [Google Scholar]
- 27. Pignata C, Budillon G, Monaco G, et al. Jejunal bacterial overgrowth and intestinal permeability in children with immunodeficiency syndromes. Gut 1990;31:879–82. 10.1136/gut.31.8.879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Ghoshal UC, Ghoshal U. Small intestinal bacterial overgrowth and other intestinal disorders. Gastroenterol Clin North Am 2017;46:103–20. 10.1016/j.gtc.2016.09.008 [DOI] [PubMed] [Google Scholar]
- 29. Sachdev AH, Pimentel M. Gastrointestinal bacterial overgrowth: pathogenesis and clinical significance. Ther Adv Chronic Dis 2013;4:223–31. 10.1177/2040622313496126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Arasaradnam RP, Brown S, Forbes A, et al. Guidelines for the investigation of chronic diarrhoea in adults: British Society of gastroenterology, 3rd edition. Gut 2018;67:1380–99. 10.1136/gutjnl-2017-315909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Pimentel M, Saad RJ, Long MD, et al. Acg clinical guideline: small intestinal bacterial overgrowth. Am J Gastroenterol 2020;115:165–78. 10.14309/ajg.0000000000000501 [DOI] [PubMed] [Google Scholar]
- 32. Giamarellos-Bourboulis E, Tang J, Pyleris E, et al. Molecular assessment of differences in the duodenal microbiome in subjects with irritable bowel syndrome. Scand J Gastroenterol 2015;50:1076–87. 10.3109/00365521.2015.1027261 [DOI] [PubMed] [Google Scholar]
- 33. Riordan SM, McIver CJ, Duncombe VM, et al. Evaluation of the rice breath hydrogen test for small intestinal bacterial overgrowth. Am J Gastroenterol 2000;95:2858–64. 10.1111/j.1572-0241.2000.02317.x [DOI] [PubMed] [Google Scholar]
- 34. Rezaie A, Buresi M, Lembo A, et al. Hydrogen and Methane-Based breath testing in gastrointestinal disorders: the North American consensus. Am J Gastroenterol 2017;112:775–84. 10.1038/ajg.2017.46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. AGIP Best Practice Statement for HMBT , 2019. Available: https://www.bsg.org.uk/wp-content/uploads/2019/12/AGIP-Best-Practice-Statement-for-HMBT_24.01.19.pdf
- 36. Kalantar-Zadeh K, Berean KJ, Ha N, et al. A human pilot trial of ingestible electronic capsules capable of sensing different gases in the gut. Nature Electronics 2018;1:79–87. 10.1038/s41928-017-0004-x [DOI] [Google Scholar]
- 37. Erdogan A, Rao SSC, Gulley D, et al. Small intestinal bacterial overgrowth: duodenal aspiration vs glucose breath test. Neurogastroenterol Motil 2015;27:481–9. 10.1111/nmo.12516 [DOI] [PubMed] [Google Scholar]
- 38. Viome . Heal your heart through your gut, 2018. Available: https://www.viome.com/blog/heal-your-heart-through-your-gut-understanding-gut-heart-connection [Accessed 27 Feb 2022].
- 39. Ghoshal UC, Shukla R, Ghoshal U. Small intestinal bacterial overgrowth and irritable bowel syndrome: a bridge between functional organic dichotomy. Gut Liver 2017;11:196–208. 10.5009/gnl16126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Vasant DH, Paine PA, Black CJ, et al. British Society of gastroenterology guidelines on the management of irritable bowel syndrome. Gut 2021;70:1214–40. 10.1136/gutjnl-2021-324598 [DOI] [PubMed] [Google Scholar]
- 41. Haboubi NY, Lee GS, Montgomery RD. Duodenal mucosal morphometry of elderly patients with small intestinal bacterial overgrowth: response to antibiotic treatment. Age Ageing 1991;20:29–32. 10.1093/ageing/20.1.29 [DOI] [PubMed] [Google Scholar]
- 42. Shah SC, Day LW, Somsouk M, et al. Meta-Analysis: antibiotic therapy for small intestinal bacterial overgrowth. Aliment Pharmacol Ther 2013;38:925–34. 10.1111/apt.12479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Arasaradnam RP, et al. Guidelines for the investigation of chronic diarrhoea in adults. Gut 2018:1–20. 10.1136/gutjnl-2017-315909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Quigley EMM, Murray JA, Pimentel M. AGA clinical practice update on small intestinal bacterial overgrowth: expert review. Gastroenterology 2020;159:1526–32. 10.1053/j.gastro.2020.06.090 [DOI] [PubMed] [Google Scholar]
- 45. Gatta L, Scarpignato C. Systematic review with meta-analysis: rifaximin is effective and safe for the treatment of small intestine bacterial overgrowth. Aliment Pharmacol Ther 2017;45:604–16. 10.1111/apt.13928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Tahan S, Melli LCFL, Mello CS, et al. Effectiveness of trimethoprim-sulfamethoxazole and metronidazole in the treatment of small intestinal bacterial overgrowth in children living in a slum. J Pediatr Gastroenterol Nutr 2013;57:316–8. 10.1097/MPG.0b013e3182952e93 [DOI] [PubMed] [Google Scholar]
- 47. Di Stefano M, Miceli E, Missanelli A, et al. Absorbable vs. non-absorbable antibiotics in the treatment of small intestine bacterial overgrowth in patients with blind-loop syndrome. Aliment Pharmacol Ther 2005;21:985–92. 10.1111/j.1365-2036.2005.02397.x [DOI] [PubMed] [Google Scholar]
- 48. BSG . Management of difficult to treat SIBO, 2020. Available: https://www.bsg.org.uk/clinical-articles-list/management-of-difficult-to-treat-small-intestinal-bacterial-overgrowth-dr-andreyev-and-dr-poon-highlight-a-case-study-on-a-difficult-case-of-sibo/ [Accessed 27 Feb 2022].
- 49. Lauritano EC, Gabrielli M, Scarpellini E, et al. Small intestinal bacterial overgrowth recurrence after antibiotic therapy. Am J Gastroenterol 2008;103:2031–5. 10.1111/j.1572-0241.2008.02030.x [DOI] [PubMed] [Google Scholar]
- 50. Richard N, Desprez C, Wuestenberghs F, et al. The effectiveness of rotating versus single course antibiotics for small intestinal bacterial overgrowth. United European Gastroenterol J 2021;9:645–54. 10.1002/ueg2.12116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Zhong C, Qu C, Wang B, et al. Probiotics for preventing and treating small intestinal bacterial overgrowth: a meta-analysis and systematic review of current evidence. J Clin Gastroenterol 2017;51:300–11. 10.1097/MCG.0000000000000814 [DOI] [PubMed] [Google Scholar]
- 52. Gibson GR, Cummings JH, Macfarlane GT, et al. Alternative pathways for hydrogen disposal during fermentation in the human colon. Gut 1990;31:679–83. 10.1136/gut.31.6.679 [DOI] [PMC free article] [PubMed] [Google Scholar]


