Abstract
Almost regular hexagonal arrays of microscopic pyramids consisting of soot nanoparticles are formed on the surface of graphitized hollow filaments, which are resistively heated to ∼1800–2400 °C under an Ar atmosphere containing trace amounts of oxygen (∼300 ppm). At higher temperatures (T > 2300 °C, approximately) the soot particles are represented mainly by multishell carbon nano-onions. The height and width of the pyramids are strongly dependent on the temperature of the resistive heating, diminishing from 5 to 10 μm at T ≈ 1800 °C to ∼1 μm at 2300–2400 °C. Quasi-hexagonal arrays of the micropyramids are organized in the convex “craters” on the surface of the microtubes, which grow with the time of the thermal treatment. The pyramids always point normally to the surface of the craters, except at the boundaries between the craters, where the normal direction is not well-defined. The pyramids are soft and can be easily destroyed by touching them but can be hardened by heating them under an oxygen-free atmosphere. The pyramids are observed only on the exterior surface of the microtubes, not on their inner surface. This suggests that the thermophoretic force generated by a strong temperature gradient near the external surface of the tubes may be the cause of the micropyramid formation. Electrostatic charging of the soot nanoparticles due to thermionic emission may also be relevant to this phenomenon. The micropyramids can function as field emission point sources, as demonstrated with the use of a micronanoprobing station, mounted in a scanning electron microscope.
Keywords: self-assembly, carbon nano-onions, resistive heating, thermophoretic force, micropyramids, hexagonal array
Carbon is known for its capacity to form numerous architectures on the nanoscale as well as on the microscale. Apart of the most well-known carbon single and multishell tubes, fullerenes, and graphene, there have been discovered many interesting entities such as carbon nanohorns and nanocones,1−3 conical microcrystals,4 carbon whiskers,5,6 graphite micropyramids,7 graphene flowers, and vertical graphene.8−12 The electrical, optical, and morphological peculiarities of these architectures give them prospects for advanced applications such as field electron emission sources,9,10 supercapacitors,13 biosensors,11 ultrablack materials,14 and superhydrophobic15 and bactericidal16 coatings. The methods of production of these structures include argon plasma etching of graphite substrates,7,14 reactive sputtering using methane gas,9 thermal chemical vapor deposition on carbon fibers,10 reduction of graphene oxide nanosheets,11 combustion flame deposition,8 microwave plasma chemical vapor deposition followed by bias-assisted reactive ion etching,16 and a wood charcoal heat treatment above 2000 °C.6
Here we report soot micropyramids, which we have found on the surface of resistively heated amorphous carbon microtubes under an Ar atmosphere, to which a vanishingly small amount of air was admixed. The pyramids always point along the local normal direction to the tube’s surface, suggesting that the soot nanoparticles, formed under the oxygen-deficient atmosphere, organize into pyramids under the action of some force, directed outward from the surface. Moreover, the pyramids are assembled in almost hexagonal arrays, resembling the spontaneously formed hexagonal patterns, which appear at certain deformable interfaces under the action of normally applied forces and which have been extensively studied in the past. Examples include the Rosenweig instability of a ferromagnetic fluid under a magnetic field,17 gravity-driven Rayleigh–Taylor instability of suspended liquids,18 elastic solids,19 and electrostatically induced structure formations at the polymer–air interface.20 The patterns arise due to the competition of external fields (gravity, magnetic, or electrostatic), which amplify the interface deformation with surface tension and elastic energy, which have stabilizing effects. Linear stability analyses have established the dispersion relation between the amplitude growth rates of the normal modes of deformation and the wavenumbers of the modes. The modes with the highest growth rates dominate and determine the characteristic wavelength of the pattern. Hexagonal patterns arise due to the resonance of the dominant modes, whose wave vectors form an equilateral triangle in the reciprocal space, as shown by nonlinear stability analysis.18,21
A natural candidate for the role of such a normally directed force in our system of soot particles on the surface of a resistively heated microtube is the thermophoretic force, Fth, which results from the difference of the average momentum transferred to the particle by molecules arriving from the forward and the backward directions, with respect to the temperature gradient’s direction.22 More rapid molecules, which arrive from the region of higher temperature, transfer larger momentum on average than the molecules that arrive from the colder regions. This results in a drag force on the particle, which drives it from hotter regions to colder ones. Thermoelectric phenomena, such as electrostatic charging of the particles due to thermionic loss of electrons at high temperatures, may be also essential for the process of the micropyramid formation.
Results and Discussion
Figure 1 presents the most representative features of the microscale morphology observed on the surface of a tube heated to T = 2000–2100 °C and at an oxygen concentration of ∼300 ppm for 5 h. The surface of the tube, which was initially smooth, became covered by microcraters (Figure 1a) whose concave bottoms are roofed by the arrays of the features which we call micropyramids, because their shape resembles this geometric form (Figure 1b,c). No similar structure was found on the surface of the control tubes heated without air admixing (see Figure S1 in the Supporting Information). The pyramids have sharp apexes with sub-micrometer curvature radii, and the apexes of some pyramids are split. The surface of the pyramids is not smooth; rather, it has a porous aspect (see Figure 1c and Figure S2 in the Supporting Information for a high-resolution image). The pyramids are often prolonged by a thread whose thickness does not exceed 100 nm (Figure 1d). A fragment of an extremity of an intentionally broken tube is shown in Figure 1e. The image reveals the interface between the loose material of the pyramids and the monolithic structure of the tube wall, which was not yet etched by the oxygen-containing atmosphere. The pyramids appear only on the outer surface of the tube, but not on its interior wall, shown by the white arrow.
Figure 1.
The most characteristic morphology features of the carbon microtubes resistively heated under an oxygen-deficient atmosphere. (a) Craters on the surface of a microtube. (b, c) Array of pyramids at different magnifications. (d) Almost hexagonal array of the pyramids. (e) Interface between pyramid bottoms and the unchanged wall of a tube. The white arrow indicates the inner surface of the tube. (f) Pyramids with thin filaments on the top. (g) Magnified view of a crater. Note that the pyramids point normally to the walls of the craters. (h) Boundary between two craters. (i) Irregular multiapex pyramids at the boundary.
As seen from the top, the pyramid arrays have a nearly hexagonal coordination in the centers of the craters (Figure 1f). The pyramids are always oriented normally to the concave crater walls (Figure 1g). At the boundaries between the craters, where the normal direction is not defined, the pyramids have irregular shapes characterized by split apexes (Figure 1g,i).
To collect the material of the pyramids for a Raman spectroscopy characterization, a tube was touched by a narrow strip of Scotch tape. Some of the pyramids were partially destroyed by this operation (Figure 2a), which allowed the examination of their inner structure. A high-resolution SEM image reveals that the pyramids consist of nanoparticles whose diameters do not exceed 30 nm (Figure 2b). The fact that the pyramids can be easily disrupted by touching signifies that the particles are not connected by covalent bonds; rather, they are held together by van der Waals forces. A high-resolution TEM image reveals that particles are have a closed shell, similar to the particles produced by the carbon arc method.23 Some particles can be identified as multishell carbon nano-onions (CNOs) (Figure 2c). The graphitization degree and the fraction of CNOs increase at higher temperatures. For a tube heated to T ≈ 2380°C (Figure 2d) the quasi-totality of the particles, collected on the surface of the tubes, can be classified as CNOs. The Raman spectra of the particles (Figure 2e,f) have well-resolved G (graphitic) and D (disordered) bands,24 meaning that the particles contain a relatively low concentration of defects of the sp2 coordination of carbon atoms. The ratio of the intensities of the G and the D peaks, IG/ID, grows with temperature, implying the improving the sp2 coordination of the CNO particles.
Figure 2.
Inner structure of the pyramids. (a) Pyramids after the
application
of Scotch tape to the surface of a tube (T ≈
2000 °C, 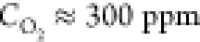 ). Some pyramids are partially destroyed,
and some are completely removed. Note that the soot pyramids cover
the pyramidal elevations of the unchanged material of the tubes. (b)
High-resolution SEM image of the interior of a partially destroyed
pyramid. (c, d) HRTEM images of the material of the miccropyramids,
formed at T ≈ 2000 °C and T ≈ 2380 °C, respectively. A CNO particle is shown in
(c) by the white arrow. (e, f) Raman spectra of the material of the
pyramids, formed at T ≈ 2000 °C and T ≈ 2380 °C, respectively.
). Some pyramids are partially destroyed,
and some are completely removed. Note that the soot pyramids cover
the pyramidal elevations of the unchanged material of the tubes. (b)
High-resolution SEM image of the interior of a partially destroyed
pyramid. (c, d) HRTEM images of the material of the miccropyramids,
formed at T ≈ 2000 °C and T ≈ 2380 °C, respectively. A CNO particle is shown in
(c) by the white arrow. (e, f) Raman spectra of the material of the
pyramids, formed at T ≈ 2000 °C and T ≈ 2380 °C, respectively.
The mechanism of formation of the soot particles on the surface of the resistively heated graphitized microtubes is not yet clear. The physicochemical conditions at the surface of the microtubes are different from the conditions of the well-understood processes of soot production by hydrocarbon flames,25,26 spark discharges,27 and laser ablation.28 In the flames, the process starts by gas-phase chemical reactions leading to the formation of polycyclic aromatic hydrocarbons (PAHs), followed by the nucleation of particles via the PAH dimerization, particle growth by the surface reactions, and particle coagulation. It is unlikely that the carbonized microtubes can be the source of PAH precursors, because at temperatures above ∼1000 K carbon films are almost completely dehydrogenated.29 In the cases of spark discharge and laser ablation, soot nuclei are formed due to evaporation of a small amount of the carbon electrode or carbon target material and condensation of the supersaturated vapor in the neutral gas flow. The soot nuclei coagulate, forming the soot particles, which aggregate eventually. This mechanism is also unlikely to be relevant to the formation of the soot particles in our system, because the temperature of the resistive heating of the microtubes is well below that of the graphite sublimation point (around 4000 K at ambient pressure).30
To the best of our knowledge, soot formation on the surface of graphitized materials exposed to temperatures close to 2000 °C and oxygen concentrations of a few hundred ppm has not reported to date, despite a great deal of literature on the oxidation of graphite materials, especially for nuclear applications.31 Typically, the temperature at which graphite oxidation was studied did not exceed 1000 K. At these temperatures, the basal graphite plane is inert to molecular oxygen. The graphite oxidation proceeds from the zigzag and armchair active sites of the graphite crystallites, attacked by molecular oxygen, with desorption of CO and CO2. Oxidation of multilayer graphene flakes suspended on an amorphous carbon grid in air and heated by laser to temperatures close to 2000 K resulted in the layer-by-layer thinning of the flakes, assuming that flakes are eroded in a highly anisotropic manner. However, no formation of carbon nanoparticles, such as soot, has been reported.32
In order to gain a deeper insight into the conditions of the soot particle formation in our system, we have carried out a control experiment, in which a 25 μm thick pyrolytic graphite film was resistively heated to 2000 °C under an Ar atmosphere at an oxygen concentration of 600 ppm. The sample was eroded over the course of 20 min of treatment (Figure S3), but no soot or pyramid formation was detected. In particular, the graphite stripe remained shiny, while the carbonized chitosan tubes became violet black upon etching. This means that the mechanism of the soot formation in our system requires a relatively high number of defects of the sp2 coordination of the carbon material.
The size of the pyramids and their number per unit surface depend strongly on the temperature of the resistive heating (Table 1). Within the temperature interval 1800–2200 °C, in which the micropyramids arrays can be unambiguously identified, the average distance between the pyramid centers decreases by ∼6 times as the temperature increases. The pyramids also became more “slim”, and their apexes are more often continued by a thin filament.
Table 1. Average Distance between the Pyramid
Centers as a Function of Resistive Heating Temperature, for 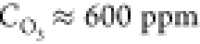 .
.
| 1800 °C | 1900 °C | 2000 °C | 2100 °C | 2200 °C | |
|---|---|---|---|---|---|
| λ (μm) | 6.2 ± 0.2 | 5.8 ± 0.3 | 4.3 ± 0.2 | 2.8 ± 0.2 | 1.0 ± 0.1 |
The craterlike features imply that the pyramids do not appear simultaneously on the surface of the tubes. Indeed, we have found that initially small, isolated craters, having an almost perfectly circular shape and containing a few pyramids, appear on the surface of the tubes a few dozen minutes after the beginning of the resistive heating (Figure S4a,b in the Supporting Information). During the experiment, the craters became wider and deeper, and the number of pyramids in them increased. New craters appeared and grew. Eventually, all the surfaces became covered by the craters, which touched each other (Figure S4c,d).
The characteristic time of the pyramid formation can be strongly
reduced by increasing the oxygen concentration in the vessel atmosphere.
Thus, almost regular arrays of the pyramids were observed on the tubes
heated at T = 2000 °C and  after 120 min of resistive heating. A few
craters were observed on the surface of the tubes, but the majority
of the pyramids were formed outside them (Figure S5 in the Supporting Information). Apparently, the rate of
the pyramid formation affects their mutual coordination. This fact
follows from an analysis of the Voronoi diagrams of the SEM images
of the pyramid arrays produced at at T = 2000 °C
and at the relative oxygen concentrations
after 120 min of resistive heating. A few
craters were observed on the surface of the tubes, but the majority
of the pyramids were formed outside them (Figure S5 in the Supporting Information). Apparently, the rate of
the pyramid formation affects their mutual coordination. This fact
follows from an analysis of the Voronoi diagrams of the SEM images
of the pyramid arrays produced at at T = 2000 °C
and at the relative oxygen concentrations 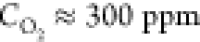 and
and 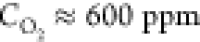 (Figure 3). By definition, the Voronoi cell of a vortex is defined
by the ensemble of the points that are closer to a given vortex than
to any other vortex of the system. The edges of the polygons correspond
roughly to the faces of the pyramids, and the vortices of the polygons
correspond to the edges of the pyramids. In our analysis, the vortices
were defined as the extremities of the micropyramids. At the lower
oxygen concentration, the absolute majority (∼80%) of the Voronoi
cells have 6 edges, while at the higher oxygen concentration, the
fraction of the 6-coordinated cells drops to around 50%. A further
increase of oxygen concentration to
(Figure 3). By definition, the Voronoi cell of a vortex is defined
by the ensemble of the points that are closer to a given vortex than
to any other vortex of the system. The edges of the polygons correspond
roughly to the faces of the pyramids, and the vortices of the polygons
correspond to the edges of the pyramids. In our analysis, the vortices
were defined as the extremities of the micropyramids. At the lower
oxygen concentration, the absolute majority (∼80%) of the Voronoi
cells have 6 edges, while at the higher oxygen concentration, the
fraction of the 6-coordinated cells drops to around 50%. A further
increase of oxygen concentration to  led to rapid consumption of the microtubes
(typically, within 10–20 min) and irregular shapes of the pyramids,
as well as a poor degree of their hexagonal arrangement.
led to rapid consumption of the microtubes
(typically, within 10–20 min) and irregular shapes of the pyramids,
as well as a poor degree of their hexagonal arrangement.
Figure 3.
Voronoi analysis
of the pyramid arrangement. Color coding of the
Voronoi cell coordination: brown, 4-edged cells; blue, 5-edged cells;
green, 6-edged cells; pink, 7-edged cells; orange, 8-edged cells.
(a) Superposition of the SEM image of a crater bottom for a tube heated
at T ≈ 2000 °C and 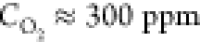 during 4 h and the Voronoi diagram, calculated
by the pyramid vortices. (b) Coloring of the Voronoi diagram cells
according to the number of cell edges. (c) Distribution of the cells
by the number of their edges. (d–f) The same as (a)–(c),
respectively, for a tube heated at T ≈ 2000
°C and
during 4 h and the Voronoi diagram, calculated
by the pyramid vortices. (b) Coloring of the Voronoi diagram cells
according to the number of cell edges. (c) Distribution of the cells
by the number of their edges. (d–f) The same as (a)–(c),
respectively, for a tube heated at T ≈ 2000
°C and 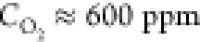 over 2 h (see Figure S5 in the Supporting Information).
over 2 h (see Figure S5 in the Supporting Information).
The hypothetical mechanism of the pyramids growing and the propagation of the pyramids arrays is shown in Figure 4. We suppose that a pyramid’s formation starts from a local defect on the surface of a tube, such as a local microscale elevation (Figure 4a). Soot particles, generated at the bottom of the defect, roll up along its walls in the vertex direction, increasing the defect’s height and transforming it into a pyramid. Simultaneously, local cavities are formed at the bottom of the pyramid, because of the particle excavation from the tube. The particles start to roll along the walls of the cavities, forming new pyramids at their borders (Figure 4b). This process is repeated, leading to the formation of pyramid arrays (Figure 4c). A similar process may start from a defect in the form of a microcavity on the surface of a tube. The growth of the pyramids seems to be limited by their sputtering from the pyramid’s vortices. The formation of the concave “craters” signifies that the pyramids somehow accelerate the transformation of the graphitized material in soot, because the older pyramids have propagated more deeply in the walls of the tubes. This is probably because the migration of the soot particles onto the pyramid’s walls unmasks the tube’s surface and makes it more accessible to the molecules of oxygen present in the gas mixture.
Figure 4.

Hypothetical mechanism of pyramid growth and pyramid arrays propagation.
The height of the pyramids in the largest craters is much smaller than the craters’ depth (see Figure 1g). This means that the soot nanoparticles, which constitute the pyramids, are constantly removed from the pyramids and replaced by new particles, which are produced by the interaction of oxygen with the tube’s walls. To clarify the fate of the eliminated particles, we placed a tungsten wire close to a microtube. In 1 h of resistive heating at T = 2300 °C, the so-called graphene flowers8,9 formed on the surface of the wire (Figure S6). Thus, the carbon material did not burn completely; rather, it sputtered into the space around the tubes, where it can be sedimented on a support.
The pyramids can be stabilized by sintering soot particles under an oxygen-free atmosphere. This operation makes them mechanically stable (Figure 5), so that the pyramids are wholly unrooted rather than smashed when they are touched. The sintering procedure also increases the height to base aspect ratio of the pyramids and cleans up the space between them from the soot particles, so that the pyramids are transformed into needlelike structures, well separated from each other. We suppose that the effect of the “cleaning up” of the space between the pyramids is due to the fact that, in the absence of oxygen, soot particles are no longer produced in the course of the erosion of the material of the tube. However, they are not sintered immediately and therefore migrate to the top of the pyramids, increasing their height to width aspect ratio. The particles that are inside the pyramids (and not on their surface) are less mobile, and for them the probability of sintering is higher; therefore these particles constitute the rigid “skeleton” of the pyramids, making them more robust mechanically.
Figure 5.
Normal view (a) and side view (b) of the micropyramids
produced
at T ≈ 2000 °C and 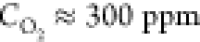 and stabilized by heating at T ≈ 2300 °C without air admixing during 30 min. The sintering
of soot particles rigidifies the pyramids, so that they are not smashed
or disrupted when touched (cf. Figure 2a); rather, they are wholly unrooted (c).
and stabilized by heating at T ≈ 2300 °C without air admixing during 30 min. The sintering
of soot particles rigidifies the pyramids, so that they are not smashed
or disrupted when touched (cf. Figure 2a); rather, they are wholly unrooted (c).
The ensemble of the experimental facts presented
above allows us
to suppose the existence of a force normally directed to the interface
between the soot layer and the material of the tube, which is not
yet transformed into soot by its interaction with oxygen. A natural
candidate for the role of such a force is thermophoretic force, Fth, which results from the difference of the
average momentum transferred to the particle by molecules arriving
from the forward and the backward directions, with respect to the
temperature gradient’s direction. More rapid molecules, which
arrive from the region of higher temperature, transfer larger momentum
on average than the molecules that arrive from the colder regions.
This results in a drag force on the particle, which drives it from
hotter regions to colder ones. The fact that the pyramids do not appear
inside the tubes, where the temperature is presumably uniform, seems
to support the hypothesis that the temperature gradient giving rise
to thermophoretic force is the factor that causes the formation of
pyramids. The intensity of Fth can be
estimated from the following considerations. At T = 2000 °C, the mean free path of argon atoms is 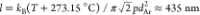 , where kB is
the Boltzmann constant, p is the pressure of the
gas, and dAr ≈ 0.4 nm is the diameter
of the argon atoms. From the HRTEM images (Figure 2c,d), one can evaluate the characteristic
diameter of the soot particles as a ≈ 10 nm.
Since the Knudsen number l/a ≈
43 is large, the thermophoretic force can be calculated in the free
molecular limit22 as
, where kB is
the Boltzmann constant, p is the pressure of the
gas, and dAr ≈ 0.4 nm is the diameter
of the argon atoms. From the HRTEM images (Figure 2c,d), one can evaluate the characteristic
diameter of the soot particles as a ≈ 10 nm.
Since the Knudsen number l/a ≈
43 is large, the thermophoretic force can be calculated in the free
molecular limit22 as 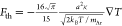 where κ ≈ 0.074 W m–1 K–1 is the argon gas thermal conductivity at the
given temperature33 and mAr is the mass of argon atoms. The temperature gradient
at the filament’s surface is usually evaluated with the assumption
that there exists a layer of almost immovable gas around the filament
(the so-called stagnant layer of Langmuir).34 Coherent anti-Stokes Raman scattering measurements35 and computer simulations36 estimate
this gradient to ∼105–106 K m–1 for tungsten filaments heated to ∼2000 K at
the normal pressure of the nitrogen surrounding the filament. Assuming
that, for argon, the temperature gradient is on the same order of
magnitude, one can estimate the thermophoretic force acting on a particle
of the characteristic size a ≈ 10 nm as Fth ≈ 10–15–10–14 N. It is several orders of magnitude smaller than
the typical adhesion force of soot particles measured by atomic force
microscopy to be in the range of a few nanonewtons.37 Therefore, it is hardly possible that the thermophoretic
force alone can detach the soot particles from each other. Conversely,
it is likely that, at elevated temperatures, the average kinetic energy
of the particles, E = 3kB(T + 273.15 °C)/2, takes over the van der Waals
adhesion energy, allowing the rearrangement of the mutual position
of the particles. The thermophoretic force field might then bias these
rearrangements so they have the effect of migrating the particles
outward from the tube’s surface. At the tips of the pyramids,
the particles have a relatively small number of neighbors and can
be “evaporated” into the hot gas surrounding the tube.
This scenario is in line with current theories about the formation
of nanoparticle aggregates, which assume that the coagulation efficiency
of the particles depends on both their size and temperature.38,39 At sufficiently high temperatures, smaller soot particles are likely
to bounce off rather than adhere to each other because of the relatively
high kinetic energy compared with the interaction energy of the particles
(the so-called thermal rebound effect). Hou et al.38 simulated the coagulation of soot particles in the framework
of the Hamaker model, in which the effective interaction of two spherical
atomic clusters is the sum of all pairwise Lennard–Jones interactions
of the atoms belonging to opposite clusters. As follows from their
model, the van der Waals interaction cannot hold together two spherical
soot particles of diameters ≤10 nm when the temperature exceeds T ≈ 2000 °C. These values are uncertain, since
the soot particles are mostly aspherical at this temperature. Moreover,
in close packing, each particle has more than one neighbor; therefore,
it needs more kinetic energy to escape from them. Also, the formation
of covalent bonds between some particles is not excluded. Nevertheless,
it seems plausible that, at high temperatures, which are reached in
the resistively heated tubes, the soot particles may move with respect
to each other, at least on the surface of the pyramids, where they
have a smaller average number of contacts. It may then be possible
to make an analogy between the ensemble of soot particles at high
temperatures and a layer of suspended viscous fluid whose interface
is deformed by gravity.18 This analogy
is supported by the existence of thin filaments by which the extremities
of some pyramids are prolonged. Such filaments are unlikely to be
formed by the random aggregation of soot particles. Rather, they seem
to be the result of the viscous flow in a force field. Another argument
for the liquidlike behavior of the soot layer on the surface of resistively
heated microtubes is the circular growth of pyramid clusters (Figure S4). The already formed pyramids seem
to induce the formation of subsequent generations of pyramids in their
neighborhood. Similar behavior was observed for arrays of pending
droplets in the layers of a suspended viscous fluid film,18 in which the concentric rings of hexagonally
coordinated droplet arrays grow around the initial perturbations.
where κ ≈ 0.074 W m–1 K–1 is the argon gas thermal conductivity at the
given temperature33 and mAr is the mass of argon atoms. The temperature gradient
at the filament’s surface is usually evaluated with the assumption
that there exists a layer of almost immovable gas around the filament
(the so-called stagnant layer of Langmuir).34 Coherent anti-Stokes Raman scattering measurements35 and computer simulations36 estimate
this gradient to ∼105–106 K m–1 for tungsten filaments heated to ∼2000 K at
the normal pressure of the nitrogen surrounding the filament. Assuming
that, for argon, the temperature gradient is on the same order of
magnitude, one can estimate the thermophoretic force acting on a particle
of the characteristic size a ≈ 10 nm as Fth ≈ 10–15–10–14 N. It is several orders of magnitude smaller than
the typical adhesion force of soot particles measured by atomic force
microscopy to be in the range of a few nanonewtons.37 Therefore, it is hardly possible that the thermophoretic
force alone can detach the soot particles from each other. Conversely,
it is likely that, at elevated temperatures, the average kinetic energy
of the particles, E = 3kB(T + 273.15 °C)/2, takes over the van der Waals
adhesion energy, allowing the rearrangement of the mutual position
of the particles. The thermophoretic force field might then bias these
rearrangements so they have the effect of migrating the particles
outward from the tube’s surface. At the tips of the pyramids,
the particles have a relatively small number of neighbors and can
be “evaporated” into the hot gas surrounding the tube.
This scenario is in line with current theories about the formation
of nanoparticle aggregates, which assume that the coagulation efficiency
of the particles depends on both their size and temperature.38,39 At sufficiently high temperatures, smaller soot particles are likely
to bounce off rather than adhere to each other because of the relatively
high kinetic energy compared with the interaction energy of the particles
(the so-called thermal rebound effect). Hou et al.38 simulated the coagulation of soot particles in the framework
of the Hamaker model, in which the effective interaction of two spherical
atomic clusters is the sum of all pairwise Lennard–Jones interactions
of the atoms belonging to opposite clusters. As follows from their
model, the van der Waals interaction cannot hold together two spherical
soot particles of diameters ≤10 nm when the temperature exceeds T ≈ 2000 °C. These values are uncertain, since
the soot particles are mostly aspherical at this temperature. Moreover,
in close packing, each particle has more than one neighbor; therefore,
it needs more kinetic energy to escape from them. Also, the formation
of covalent bonds between some particles is not excluded. Nevertheless,
it seems plausible that, at high temperatures, which are reached in
the resistively heated tubes, the soot particles may move with respect
to each other, at least on the surface of the pyramids, where they
have a smaller average number of contacts. It may then be possible
to make an analogy between the ensemble of soot particles at high
temperatures and a layer of suspended viscous fluid whose interface
is deformed by gravity.18 This analogy
is supported by the existence of thin filaments by which the extremities
of some pyramids are prolonged. Such filaments are unlikely to be
formed by the random aggregation of soot particles. Rather, they seem
to be the result of the viscous flow in a force field. Another argument
for the liquidlike behavior of the soot layer on the surface of resistively
heated microtubes is the circular growth of pyramid clusters (Figure S4). The already formed pyramids seem
to induce the formation of subsequent generations of pyramids in their
neighborhood. Similar behavior was observed for arrays of pending
droplets in the layers of a suspended viscous fluid film,18 in which the concentric rings of hexagonally
coordinated droplet arrays grow around the initial perturbations.
Thermoelectric phenomena may be also relevant to the pyramid formation.
A grounded carbon spherical nanoparticle of the diameter a ≈ 10 nm loses one electron every ∼110 μs at T = 2000 K (∼1727 °C) and every ∼0.7
μs at T = 2400 K (∼2127 °C), as
calculated by the Richardson–Dushman equation for the thermoelectric
current,  , where s = πa2 is the surface of the particle, A = 1.2 × 106 A m–2 K–2 is the Richardson constant, and Φ = 7.63 × 10–19 J is the work function for carbon.40,41 Filippov et
al. considered the possibility of an electrostatic breakdown (Coulomb
explosion) of soot agglomerates heated by laser pulses to high temperatures.40 If the analogy between the soot layer at high
temperature and a layer of viscous liquid having a finite surface
tension is correct, then charging particles on the surface of the
layer may lead to the development of electrohydrodynamic instability.42 However, unlike the particles in the isolated
soot clusters, the particles on the surface of the tubes constantly
restore their electroneutrality due to the electron flux from the
grounded tubes. Unfortunately, in the absence of literature and data
on the conductivity and effective permittivity of the soot layer at
high temperatures, it is impossible to make reliable estimates of
the surface charge.
, where s = πa2 is the surface of the particle, A = 1.2 × 106 A m–2 K–2 is the Richardson constant, and Φ = 7.63 × 10–19 J is the work function for carbon.40,41 Filippov et
al. considered the possibility of an electrostatic breakdown (Coulomb
explosion) of soot agglomerates heated by laser pulses to high temperatures.40 If the analogy between the soot layer at high
temperature and a layer of viscous liquid having a finite surface
tension is correct, then charging particles on the surface of the
layer may lead to the development of electrohydrodynamic instability.42 However, unlike the particles in the isolated
soot clusters, the particles on the surface of the tubes constantly
restore their electroneutrality due to the electron flux from the
grounded tubes. Unfortunately, in the absence of literature and data
on the conductivity and effective permittivity of the soot layer at
high temperatures, it is impossible to make reliable estimates of
the surface charge.
Theoretically, it might be possible to discriminate which of the two factors is more relevant to the pyramids formation, by heating the carbonized tubes in an oven, thus eliminating the temperature gradient at the tube surface. However, the maximum temperature at which the alumina ovens can operate in a continuous regime does not exceed 1600–1700 °C, which is essentially below of lowest temperature at which we succeeded to observe the pyramids. The graphitic ovens can reach much higher temperatures (up to 3000 °C), but they are incompatible with the oxygen-containing atmosphere.
The arrays of the carbon micropyramids may be explored as the field emitter arrays.9,10,43 As a first proof of concept, we present here preliminary results to test the capacity of field emission properties of individual pyramids. We used a micronanoprobing station mounted in a scanning electron microscope (see Figure 6a). The nanoprobes are standard tungsten tips without any coating (such as, for example, gold to improve the work function of the tip probe). The measurements are taken at 10–6 mbar at ambient temperature. An example of the current–voltage (I–V) characteristics of a pyramid, which is separated from the tip by a 200 nm wide gap, is shown in Figure 6b. The observed emission curves are very similar to those observed in a previous work devoted to the emission from carbon nanotube films and approximated by the classical Fowler–Nordheim (FN) model.43 The first few sweeps show noisy curves (blue, red, and green lines).
Figure 6.
Measurement of the tunneling current from the micropyramids. (a) The setup: four micronanomanipulator IMINAs mounted around a cryo stage (600–77 K). (b) Kammrath SEM image of the nanoprobe in front of a pyramid (d = 200 nm) and the emitted current vs applied potential. The current increases with a threshold bias of −75 V. (c) SEM image and anomalous (non-FN) emitted current versus bias curve for two pyramids. (d) I–V curve for a pyramid annealed at 2300 °C.
After several sweeps the I–V characteristics became regular (black lines). This is usually attributed to a “conditioning” of the emitter, such as oxygen desorption.43 The threshold of electron emission is −75 V, and the measured emitted current reaches 60 nA for a bias of −100 V. It should be said that the emissive characteristics of the pyramids are highly variable and may demonstrate completely different I–V curves. Figure 6c shows two type of I–V curves recorded for two other pyramids. The shape of the curve is very different from that expected by the FN model. The current increases slowly up to a sharp increase at different bias voltages with a saturation at 0.5 nA, which is a value 1 order of magnitude lower than for that the pyramid shown on the Figure 6b. Possibly, the morphology of the pyramids changes during the I–V measurement; however, a plateau of emissive current is reached. The FN-like emission curves were detected for the micropyramids annealed at 2300 °C (Figure 6d).
From these measurements it follows that the carbon micropyramids can be considered as the field emission sources, yet their emissive characteristics are very variable. This is not surprising since, according to the FN theory, the tunneling currents are highly dependent on the shape of the extremities of the emitters (mathematically, they are expressed by the local field enhancement factor), and the extremities of the pyramids are all different. We need many more studies to understand the behavior of individual pyramids, to probe the emissivity with lower tip radius curvature (these experiments were done with 500 nm but tips with 100 and 50 nm), and to see the effect of the pyramid shapes and morphology, as well as the coating on the tungsten tip.
Conclusion and Outlook
Hexagonal arrays of micropyramids composed of soot nanoparticles are observed on the surface of graphitized microtubes resistively heated to 1800–2400 °C under an Ar atmosphere containing a small (∼300–600 ppm) fraction of oxygen. The few micrometer tall and wide micropyramids are arranged in quasi-regular hexagonal arrays on the surface of the microtubes. The pyramids point normally to the walls of the craters, except for the borders between the craters, where the normal direction is not defined. The pyramids are formed only on the external walls of the microtubes. HRTEM reveals that the increasing fraction of soot particles is morphologically identical with carbon nano-onions, when the resistive heating temperature is increased. The pyramids are soft and can be easily destroyed by touching them, but they can be rigidified by a resistive heating without oxygen admixing under the Ar stream passing through the system.
The mechanism of the pyramid formation and their self-assembly in the quasi-hexagonal arrays is not yet clear. The fact that the pyramids are always directed parallel to the local normals to the tube surface indicates the existence of some factor that favors the drift of the nanoparticles in these directions. A thermophoretic force, arising as the consequence of a strong temperature gradient in the vicinity of the tube, is a natural candidate for the role of such a factor. Under this assumption, one can draw an analogy with the gravity-driven Rayleigh–Taylor instability of a thin suspended liquid layer. It is also possible that the electrostatic charging of the soot particle layer due to thermoelectric emission contributes to the pyramid formation and their assembly in the hexagonal arrays.
The regular mutual arrangement and the sharp extremities of the micropyramids allow us to consider them as prospective microstructures for advanced applications. In our study, we have demonstrated that the individual carbon micropyramids emit a tunneling current upon application of a local electrical field. This indicates the principal possibility to create field emitter arrays45 on the base of the micropyramid ensembles. To achieve this goal, a reliable approach should be found for the formation of the pyramid arrays on the flat substrates and on sufficiently large surfaces. The pyramid ensembles on the flat substrates may then also be investigated for the creation of superhydrophobic15 and bactericidal16 coatings, as well as ultrablack materials.14
Methods
The experimental setup for the generation of the soot micropyramid arrays is shown schematically in Figure 7. An electroconductive hollow-core microtube derived from a chitosan self-rolled microtube, made according to the procedure given in ref (44), was placed inside quartz vessel 1, where it was suspended on crocodile-like electrodes. An electrical current was generated in the tube by the DC power supply 2. The vessel was filled with an air/argon mixture at a normal pressure of p = 101.3 kPa with the use of valves 3 and 4, guided by the multichannel gas flow controller 5.
Figure 7.

Experimental setup to produce arrays of self-assembled soot micropyramids.
After passing through the system, the gas mixture
was rejected
in atmosphere 6. The relative concentration, 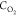 , of oxygen was varied between 300 and 3000
ppm. The temperature of the microtube was measured by Modline 5 Raytec
optical pyrometer 7 conjugated with a computer 8. Typically, the formation
of well-developed pyramid patterns requires several dozens of minutes
at
, of oxygen was varied between 300 and 3000
ppm. The temperature of the microtube was measured by Modline 5 Raytec
optical pyrometer 7 conjugated with a computer 8. Typically, the formation
of well-developed pyramid patterns requires several dozens of minutes
at 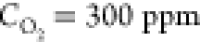 , but at a relatively high O2 concentration, this time period was shorter. After a Joule heating
experiment, the filament was gently dismounted and examined by scanning
electron microscopy (SEM) and transmission electron microscopy (TEM).
Soot particles transferred onto Scotch tape were examined by Raman
spectroscopy (Horiba Labram 300, λ = 632 nm). The field emission
was measured with the use of a nanoprobing station mounted in an FEG-MEB
XL30 scanning electron microscope. The station consisted of four IMINA
micronanomanipulators mounted around a Kammrath cryostage (600–77
K). Panasonic pyrolytic graphite sheets were purchased from Farnell
Electronics.
, but at a relatively high O2 concentration, this time period was shorter. After a Joule heating
experiment, the filament was gently dismounted and examined by scanning
electron microscopy (SEM) and transmission electron microscopy (TEM).
Soot particles transferred onto Scotch tape were examined by Raman
spectroscopy (Horiba Labram 300, λ = 632 nm). The field emission
was measured with the use of a nanoprobing station mounted in an FEG-MEB
XL30 scanning electron microscope. The station consisted of four IMINA
micronanomanipulators mounted around a Kammrath cryostage (600–77
K). Panasonic pyrolytic graphite sheets were purchased from Farnell
Electronics.
Acknowledgments
We thank L. Vidal, S. Knopf, B. Rety, and A. Beda (all-IS2M CNRS) for their assistance with electron microscopy imaging and the carbonization of chitosan microtubes. The measurements of the tunneling currents were done with the financial support by the CNRS, the Région Grand Est and FEDER (EU) through the NanoteraHertz project, and the French National Research Agency (ANR) through the MIXES project (Grant ANR-19-CE09-0028).
Supporting Information Available
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsnano.2c04395.
Control experiment on resistive heating of a tube without air admixing, high-resolution SEM images of the micropyramids, Control experiment withetching of a pyrolytic graphite film, growth of “craters” on the surface of the resistively heated microtubes, morphology of a microtube, resistively heated at T = 2000 °C and the oxygen relative concentration
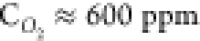 , and graphene flowers grown on the surface
of a tungsten wire suspended in the vicinity of a resistively heated
tube (PDF)
, and graphene flowers grown on the surface
of a tungsten wire suspended in the vicinity of a resistively heated
tube (PDF)
The authors declare no competing financial interest.
Supplementary Material
References
- Harris P. J. F.; Tsang S.; Claridge J. B.; Green M. High-Resolution Electron Microscopy Studies of a Microporous Carbon Produced By Arc-Evaporation. J. Chem. Soc. Faraday Trans. 1994, 90, 2799–2802. 10.1039/ft9949002799. [DOI] [Google Scholar]
- Iijima S.; Yudasaka M.; Yamada R.; Bandow S.; Suenaga K.; Kokai F.; Takahashi K. Nano-Aggregates of Single-Walled Graphitic Carbon Nano-Horns. Chem. Phys. Lett. 1999, 309, 165–170. 10.1016/S0009-2614(99)00642-9. [DOI] [Google Scholar]
- Naess S.; Elgsaeter A.; Helgesen G.; Knudsen K. Carbon Nanocones: Wall Structure and Morphology. Sci. Technol. Adv. Mater. 2009, 10 (6pp), 065002 10.1088/1468-6996/10/6/065002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gogotsi Y.; Dimovski S.; Libera J. Conical Crystals of Graphite. Carbon. 2002, 40, 2263–2284. 10.1016/S0008-6223(02)00067-2. [DOI] [Google Scholar]
- Gillot J.; Bollmann W.; Lux B. Cristaux de Graphite en Forme de Cigare et a Structure Conique. Carbon. 1968, 6, 381–387. 10.1016/0008-6223(68)90033-X. [DOI] [Google Scholar]
- Saito Y.; Arima T. Features of Vapor-Grown Cone-Shaped Graphitic Whiskers Deposited in the Cavities of Wood Cells. Carbon. 2007, 45, 248–255. 10.1016/j.carbon.2006.10.002. [DOI] [Google Scholar]
- Glad X.; Poucques L.; Bougdira J. Formation Mechanism of Graphite Hexagonal Pyramids by Argon Plasma Etching of Graphite Substrates. J. Phys. D Appl. Phys. 2015, 48 (8pp), 495304. 10.1088/0022-3727/48/49/495304. [DOI] [Google Scholar]
- Despres J. F.; Genseki A.; Odawara O. Characterization of an Unusual Uarbon Entity: ‘Gypsum Flower’-Like Carbon. J. Mater. Chem. 1997, 7, 1877–1879. 10.1039/a701597f. [DOI] [Google Scholar]
- Lee K.-Y.; Fujimoto K.; Ohkura S.; Honda S.; Katayama M.; Hirao T.; Oura K. Study of Electron Field Emission and Structural Properties of Nanostructured Carbon Thin Films Deposited by Hot-Filament Assisted Reactive Sputtering Using Methane Gas. Vacuum. 2002, 66, 239–243. 10.1016/S0042-207X(02)00148-3. [DOI] [Google Scholar]
- Iwai Y.; Muramatsu K.; Tsuboi S.; Jyouzuka A.; Nakamura T.; Onizuka Y.; Mimura H. X-ray Tube Using a Graphene Flower Cloth Field Emission Cathode. Appl. Phys. Express 2013, 6 (3pp), 105102. 10.7567/APEX.6.105102. [DOI] [Google Scholar]
- Du J.; Yue R.; Ren F.; Yao Z.; Jiang F.; Yang P.; Du Y. Novel Graphene Flowers Modified Carbon Fibers for Simultaneous Determination of Ascorbic Acid, Dopamine and Uric Acid. Biosens. Bioelectron. 2014, 53, 220–224. 10.1016/j.bios.2013.09.064. [DOI] [PubMed] [Google Scholar]
- Wang Z.; Li Y.; Liu J.; Tian G.; Liu G.; Wang M.; Ogata H.; Gong W.; Vipin K.; Melvin G.; Ortiz-Medina J.; Morimoto S.; Hashimoto Y.; Terrones M.; Endo M. Microwave Plasma-Induced Growth of Vertical Graphene From Fullerene Soot. Carbon. 2021, 172, 26–30. 10.1016/j.carbon.2020.09.086. [DOI] [Google Scholar]
- Huang Q.; Wang J.; Liu F.; Chang X.; Chen H.; Lin H.; Han S. Graphene-Constructed Flower-Like Porous Co(OH)2 with Tunable Hierarchical Morphologies for Supercapacitors. RSC Adv. 2016, 6, 16745–16750. 10.1039/C6RA00562D. [DOI] [Google Scholar]
- Sun Y.; Evans J.; Ding F.; Liu N.; Liu W.; Zhang Y.; He S. Bendable, Ultra-Black Absorber Based on a Graphite Nanocone Nanowire Composite Structure. Opt. Express. 2015, 23, 20115–20123. 10.1364/OE.23.020115. [DOI] [PubMed] [Google Scholar]
- Meng L.-Y.; Park S.-J. Superhydrophobic Carbon-Based Materials: a Review of Synthesis, Structure, and Applications. Carbon Lett. 2014, 15, 89–104. 10.5714/CL.2014.15.2.089. [DOI] [Google Scholar]
- Fisher L.; Yang Y.; Yuen M.-F.; Zhang W.; Nobbs A.; Su B. Bactericidal Activity of Biomimetic Diamond Nanocone Surfaces. Biointerfaces. 2016, 11 (6pp), 011014 10.1116/1.4944062. [DOI] [PubMed] [Google Scholar]
- Cowley M. D.; Rosenweig R. E. The Interfacial Stability of a Ferromagnetic Fluid. J. Fluid Mech. 1967, 30, 671–688. 10.1017/S0022112067001697. [DOI] [Google Scholar]
- Fermigier M.; Limat L.; Wesfried J. E.; Boudinet P.; Quiliet C. Two-Dimensional Patterns in Rayleigh-Taylor Instability of a Thin Layer. J. Fluid Mech. 1992, 236, 349–383. 10.1017/S0022112092001447. [DOI] [Google Scholar]
- Mora S.; Phou T.; Fromental J.-M.; Pomeau Y. Gravity Driven Instability in Elastic Solid Layers. Phys. Rev. Lett. 2014, 113 (5pp), 178301. 10.1103/PhysRevLett.113.178301. [DOI] [PubMed] [Google Scholar]
- Schäffer E.; Thurn-Albrecht T.; Russel T. P.; Steiner U. Electrically Induced Structure Formation and Pattern Transfer. Nature. 2000, 403, 874–877. 10.1038/35002540. [DOI] [PubMed] [Google Scholar]
- Chakrabarti A.; Mora S.; Richard F.; Phou T.; Fromental J.-M.; Pomeau Y.; Audoly B. Selection of Hexagonal Buckling Patterns by the Elastic Rayleigh-Taylor Instability. J. Mech. Phys. Solids. 2018, 121, 234–257. 10.1016/j.jmps.2018.07.024. [DOI] [Google Scholar]
- Waldmann L. Uber die Kraft Eines Inhomogenen Gases auf Kleine Suspendierte Kugeln. Z. Naturforsch. 1959, 14, 589–599. 10.1515/zna-1959-0701. [DOI] [Google Scholar]
- Bacsa W. S.; de Heer W. A.; Ugarte D.; Châtelain A. Raman Spectroscopy of Closed-Shell Carbon Particles. Chem. Phys. Lett. 1993, 211, 346–352. 10.1016/0009-2614(93)87071-A. [DOI] [Google Scholar]
- Codorniu Pujals D.; Arias de Fuentes O.; Desdin Garcia L. F.; Cazzanelli E.; Caputi L. S. Raman Spectroscopy of Polyhedral Carbon Nano-Onions. Appl. Phys. A: Mater. Sci. Process. 2015, 120, 1339–1345. 10.1007/s00339-015-9315-9. [DOI] [Google Scholar]
- Bockhorn H.Soot Formation in Combustion; Springer: 1995. [Google Scholar]
- Frenklach M. Reaction Mechanism of Soot Formation in Flames. Phys. Chem. chem. Phys. 2002, 4, 2028–2037. 10.1039/b110045a. [DOI] [Google Scholar]
- Hagen F. P.; Rinkenburger A.; Gunther J.; Bockhorn H.; Niessner R.; Suntz R.; Loukou A.; Trimis D.; Haisch C. Spark-Discharge Generated Soot: Varying Nanostructure and Reactivity Against Oxidation with Molecular Oxygen by Synthetic Conditions. J. Aerosol Sci. 2020, 143, 105530. 10.1016/j.jaerosci.2020.105530. [DOI] [Google Scholar]
- Ajtai T.; Utry N.; Pintér M.; Kiss-Albert G.; Puskas R.; Tapai C.; Kecskeméti G.; Smausz T.; Hopp B.; Bozoki Z.; Konya Z.; Szabo G. Microphysical Properties of Carbonaceous Aerosol Particles Generated by Laser Ablation of a Graphite Target. Atmos. Meas. Technol. 2015, 8, 1207–1215. 10.5194/amt-8-1207-2015. [DOI] [Google Scholar]
- Hopf C.; Angot T.; Aréou E.; Dürbeck T.; Jacob W.; Martin C.; Pardanaud C.; Roubin P.; Schwarz-Selinger T. Characterization of Temperature-Induced Changes in Amorphous Hydrogenated Carbon Thin Films. Diam. Relat. Mater. 2013, 37, 97–103. 10.1016/j.diamond.2013.05.004. [DOI] [Google Scholar]
- Abrahamson J. Graphite Sublimation Temperatures, Carbon Arcs and Crystallite Erosion. Carbon. 1974, 12, 111–141. 10.1016/0008-6223(74)90019-0. [DOI] [Google Scholar]
- Kane J. J.; Contescu C. I.; Smith R. E.; Strydom G.; Windes W. W. Understanding the Reaction of Nuclear Graphite with Molecular Oxygen: Kinetics, Transport, and Structural Evolution. J. Nucl. Mater. 2017, 493, 343–367. 10.1016/j.jnucmat.2017.06.001. [DOI] [Google Scholar]
- Ermakov V. A.; Alaferov A. V.; Vaz A.; Perim E.; Autrero P. A.; Paupitz R.; Galvao D. S.; Moshkalov S. A. Burning Graphene Layer-By-Layer. Sci. Rep. 2015, 5 (8pp), 11546. 10.1038/srep11546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bich E.; Millat J.; Vogel E. The Viscosity and Thermal Conductivity of Pure Monatomic Gases from Their Normal Boiling Point up to 5000 K in the Limit of Zero Density and at 0.101325 MPa. J. Phys. Chem. Ref. Data. 1990, 19, 1289. 10.1063/1.555846. [DOI] [Google Scholar]
- Langmuir I. Convection and Conduction of Heat in Gases. Phys. Rev. 1912, 34, 401–422. 10.1103/PhysRevSeriesI.34.401. [DOI] [Google Scholar]
- Devonshire R.; Dring I.; Hoey G.; Porter F. M.; Williams D. R.; Greenhalgh D. A. Accurate CARS Measurement and Fluid-Flow Modelling of the Temperature Distribution Around a Linear Incadescent Filament. Chem. Phys. Lett. 1986, 129, 191–196. 10.1016/0009-2614(86)80195-6. [DOI] [Google Scholar]
- Boutebba S.; Kaabar W. Simulation of Natural Convection Heat Transfer of Nitrogen in a Cylindrical Enclosure. Period. Polytech. Mech. Eng. 2016, 60, 256–261. 10.3311/PPme.9688. [DOI] [Google Scholar]
- Liu Y.; Song C.; Lv G.; Chen N.; Zhou H.; Jing X. Determination of the Attractive Force, Adhesive force, Adhesion Energy and Hamaker Constant of Soot Particles Generated From a Premixed Methane/Oxygen Flame by AFM. Appl. Surf. Sci. 2018, 433, 450–457. 10.1016/j.apsusc.2017.10.030. [DOI] [Google Scholar]
- Hou D.; Zong D.; Lindberg C.; Kraft M.; You X. On the Coagulation Efficiency of Carbonaceous Nanoparticles. J. Aerosol Sci. 2020, 140, 105478. 10.1016/j.jaerosci.2019.105478. [DOI] [Google Scholar]
- Wang H. C.; Kasper G. Filtration Efficiency of Nanometer-Size Aerosol Particles. J. Aerosol Sci. 1991, 22, 31–41. 10.1016/0021-8502(91)90091-U. [DOI] [Google Scholar]
- Filippov A. V.; Markus M. W.; Roth P. In-Situ Characterization of Ultrafine Particles by Laser-Induced Incadescence: Sizing and Particle Structure Determination. J. Aerosol. Sci. 1999, 30, 71–87. 10.1016/S0021-8502(98)00021-4. [DOI] [Google Scholar]
- Robers W.; Schröder H.; Kompa K. L.; Niessner R. Photo- und Thermo-ionization von Aerosolen durch Gepulstes Laserlicht. Zeit. Phys. Chem. 1988, 159, 129–148. 10.1524/zpch.1988.159.Part_2.129. [DOI] [Google Scholar]
- Firouznia M.; Saintillan D. Electrohydrodynamic Instabilities in Freely Suspended Viscous Films Under Normal Electric Fields. Phys. Rev. Fluids. 2021, 6 (17pp), 103703. 10.1103/PhysRevFluids.6.103703. [DOI] [Google Scholar]
- Di Bartolomeo A.; Scarfato A.; Giubileo F.; Bobba F.; Biasiucci M.; Cucolo A. M.; Santucci S.; Passacantando M. A Local Field Emission Study of Partially Aligned Carbon-Nanotubes by Atomic Force Microscope Probe. Carbon. 2007, 45, 2957–2971. 10.1016/j.carbon.2007.09.049. [DOI] [Google Scholar]
- Beda A.; Yamada H.; Egunov A.; Matei Ghimbeu C.; Malval J.-P.; Saito Y.; Luchnikov V. Carbon Microtubes Derived from Self-Rolled Chitosan Acetate Films and Graphitized by Joule Heating. J. Mater. Sci. 2019, 54, 11345–11356. 10.1007/s10853-019-03675-6. [DOI] [Google Scholar]
- Giubileo F.; Di Bartolomeo A.; Iemmo L.; Luongo G.; Urban F. Field Emission from Carbon Nanostructures. Appl. Sci. 2018, 8 (21pp), 526. 10.3390/app8040526. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.








