Abstract
Aims
Left bundle branch area pacing (LBBAP) is a recent technique aiming at preservation of physiological ventricular electrical activation. Our goal was to assess mechanical synchrony parameters in relation to electrocardiographic features during LBBAP performed in routine practice.
Methods and results
From June 2020 to August 2021, all patients of our institution with permanent pacemaker implantation indication were eligible for LBBAP. A ‘qR’ pattern in V1 and a delay from pacing spike to the peak of the R-wave in V6 < 80 ms defined a successful LBBAP. Electrocardiogram and echocardiography were performed during spontaneous rhythm and LBBAP: left ventricular mechanical synchrony (LVMS) parameters using 2D Speckle tracking and interventricular mechanical delay (IVMD) were collected. LBBAP was attempted with success in 134/163 patients (82.2%). During LBBAP, the mean QRS width was 104 ± 12 ms. In patients with left bundle branch block (n = 47), LBBAP provided a significant decrease of QRS width from 139 ± 16 to 105 ± 12 ms (P < 0.001) with reduction of LVMS (53 ± 21 vs. 90 ± 46 ms, P = 0.009), and IVMD (14 ± 13 vs. 49 ± 18 ms, P < 0.001). In patients with right bundle branch block (n = 38), LBBAP led to a significant decrease of QRS width from 134 ± 14 to 106 ± 13 ms (P < 0.001) with no effect on LVMS and a reduction of IVMD (17 ± 14 vs. 50 ± 16 ms, P < 0.001).
Conclusion
LBBAP in routine practice preserved intra-ventricular mechanical synchrony in patients with narrow and RBBB QRS and improved asynchrony parameters in patients with LBBB.
Keywords: Left bundle branch area pacing, Left bundle branch block, Left ventricular synchrony parameters, Right bundle branch block 2D speckle tracking
What’s new?
Left bundle branch area pacing preserves mechanical synchrony in patients with narrow QRS and improves asynchrony parameters in patients with both right or left bundle branch block.
Patients with qR pattern in V1 and V6 RWPT > 80 ms have wider paced QRS and greater left ventricular mechanical dispersion time compared with patients with V6 RWPT ≤ 80 ms. Those data support the need for both qR pattern in V1 and RWPT ≤ 80 ms to define LBBAP success.
Whereas the presence of a left bundle branch potential at implant is a clear indication of a successful procedure, it is not associated with an additional benefit in terms of mechanical left ventricular synchrony.
Introduction
Left bundle branch area pacing (LBBAP) was first reported in 2017 by Huang et al.1 A prior study had already described cases of left ventricular septal pacing with paced QRS morphologies suggesting capture of the posterior fascicle of the left bundle.2 This conduction system pacing technique aimed at preserving left ventricular synchrony and avoiding the detrimental impact of conventional right ventricular pacing.3,4
Furthermore, in cases of proximal left bundle branch block (LBBB), LBBAP could potentially be an elegant technique to correct left ventricular asynchrony. However, data on electrocardiographic features of LBBAP and its echocardiography consequences are scarce to date. The aim of this study was to assess the feasibility of LBBAP in routine practice, and to evaluate mechanical synchrony parameters on echocardiography and their relationship with electrocardiographic features during LBBAP.
Methods
Study population
This was an observational, prospective, and single centre study. From June 2020 to August 2021, all patients with permanent pacemaker implantation indication5 were eligible for LBBAP. The decision of LBBAP rather than conventional pacing was left to operators’ discretion. The general rules were not to perform LBBAP in elderly, frail, and highly comorbid patients in whom the expected benefit from conduction system pacing was thought to be unlikely. The exclusion criteria were need for a right-sided approach, and age ≤ 18 years old. All patients with wide QRS and LVEF < 35% were excluded from this study and received conventional biventricular resynchronization whereas patients with narrow QRS and LVEF < 35% could be included. Of note, patients with non-specific intra-ventricular conduction delay were not excluded. All patients underwent a standard transthoracic echocardiography prior to implantation.
Pacemaker implantation
All patients provided informed consent for the implantation procedure and data collection. Pacemaker implantation was performed under local anaesthesia. Continuous 12-lead ECG was displayed throughout the entire procedure. The 3830 lead was advanced at the site of implantation using the C315 His sheath (Medtronic Inc, Minneapolis, MN). The lead was connected to an EP recording system and the pacing system analyser using a jumper cable. It was first directed to the His bundle area and then positioned 1 cm more distally on the virtual line between the His and the apex of the right ventricle in right anterior oblique view (30°). The presence of a ‘W’ QRS pattern in V1 (notch at the nadir of the QRS) during pacing identified an area of interest to initiate the fixation of the lead. In left anterior oblique view (30°), the lead was screwed perpendicular to the septum until obtaining a ‘qR’ pattern in lead V1.6 The potential presence of a left bundle branch (LBB) potential was noted. Electrical parameters were collected in both unipolar and bipolar modes.
LBBAP definition
LBBAP was considered successful using the standard criteria,7,8 i.e. qR pattern recorded in V1 associated with a delay from pacing spike to the peak of the R-wave in V6 (V6 RWPT) ≤ 80 ms regardless of the QRS axis. If after several screwing attempts a qR pattern in V1 was obtained with good electrical parameters but with a slightly longer V6 RWPT (>80 ms), the lead was left in place.
Electrocardiographic parameters
The following electrocardiographic parameters were collected: the QRS duration, the QRS axis, V6 RWPT, the R/Q ratio in V1. QRS duration was measured from the pacing stimulus or QRS onset (in case of isoelectric delay between spike and QRS onset) to the final QRS component in any of the 12 ECG leads. All those parameters were measured directly after the procedure using the EP recording system at a sweep speed of 100 or 200 mm/s. All measures were repeated in at least three QRS complexes and were averaged. Electrocardiographic patterns were also collected during high and low output in unipolar and bipolar configuration in a subgroup of patients. A 12-lead ECG was performed after the procedure, at hospital discharge and at each visit for pacemaker control.
Echocardiographic parameters
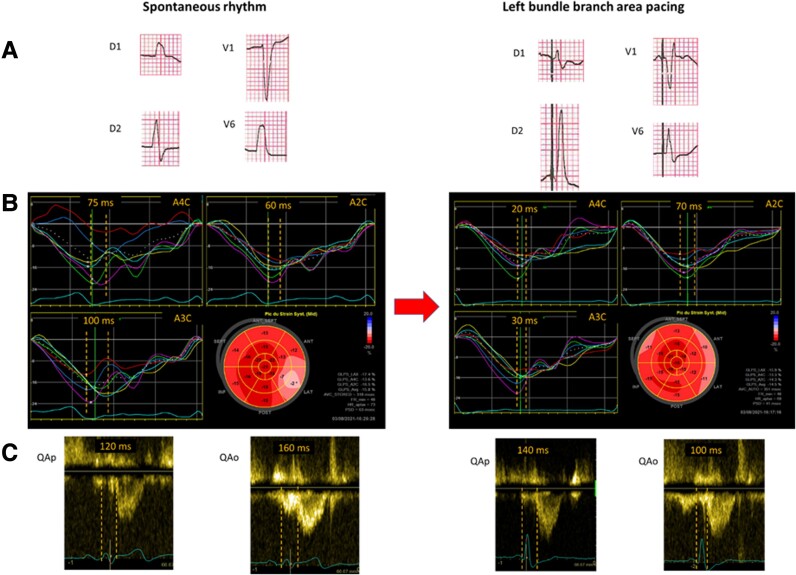
Echocardiography was performed within the first 3 months after pacemaker implantation using VIVID E95 ultrasound machines (General Electric, Boston, MA, USA). All the usual structural parameters were collected. Right heart dilatation was defined as either right atrial enlargement (right atrial area > 18 cm2) or right ventricular dilatation (right ventricular diastolic area > 28 cm2) in apical four-chamber view.9 The presence of a septal flash during LBBAP was assessed visually. It was defined as a premature contraction of the interventricular septum during pre-systolic time. The lead position into the interventricular septum was assessed in apical four-chamber view (A4C). The ratio between the distance from atrioventricular (AV) plane to the ventricular lead and the distance from AV plane to the apex of the left ventricle was calculated. This ratio was called ‘lead apex ratio’. The synchrony parameters collected were the following: the interventricular mechanical delay (IVMD), the mitral filling reported to the heart cycle in patient in sinus rhythm, the 2D speckle tracking in A4C, 2D speckle tracking in the apex three-chamber view (A3C), and 2D speckle tracking in the apex two-chamber view (A2C) view. The interventricular mechanical delay was defined as the time difference between the right ventricular pre-ejection time (QAp) and the left ventricular pre-ejection time (QAo). Pre-ejection times were measured from QRS start to the beginning of ejection blood flow (through the aortic valve and pulmonary valve). These synchrony parameters were assessed during pacing rhythm and spontaneous rhythm if available. Pacemaker were programmed in VVI mode at 30 bpm for a waiting period of at least 3 min to assess the presence of an intrinsic rhythm. The 2D speckle tracking was used to determine the systolic peak time of myocardial segments in the 3 views mentioned above. The left ventricular mechanical dispersion time (LVMDT) was defined as the time duration between the first and last systolic peak time recorded in each of the three views. The overall LVMDT was defined as the mean of the three dispersion times assessed in each of the three views (A4C, A3C, A2C) (Figure 2). All echocardiography data were acquired and analysed by the same operator who was blinded to the ECG parameters. Three cardiac revolutions were assessed and the values were averaged.
Figure 2.
Example of LBBAP effects in a 78 yo patient with no cardiomyopathy (LVEF 66%) and LBBB. Indication for pacing: syncope and intermittent complete AV block. (A) spontaneous QRS with LBBB of 120 ms width (left side) and LBB paced QRS of 110 ms width (right side). Note the presence of an R wave in V1 and a V6 RWPT of 50 ms. (B) mechanical dispersion measured with 2D speckle tracking during spontaneous rhythm (left side) and LBBAP (right side). Please note an improvement of LV synchrony during LBBAP (baseline and LBB paced overall mechanical dispersion of 78 vs. 40 ms, respectively). (C) Left and right pre-ejection times measurements during spontaneous rhythm (left side) and LBBAP (right). Please note a clear reduction in the left pre-ejection time (160 vs. 100 ms). QAo, left ventricular pre-ejection time; QAp, right ventricular pre-ejection time.
Follow-up
Hospital discharge was approved the day after the pacemaker implantation in absence of adverse events. Medical visits for pacemaker control and electrocardiographic recording were scheduled at 3 months and 1 year.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation. Normal distribution was assessed by a Shapiro–Wilk test. A student t test or a Mann–Whitney test was used when appropriate. A χ2 test or a Fisher exact test was used when appropriate. Correlation between two continuous variables was assessed by a Pearson test or a Spearman test when appropriate. A logistic regression analysis was performed to identify predictive factors of LBBAP failure. A multiple linear regression analysis was performed to identify predictive factors of QRS width and V6 RWPT. Variables included in the regression analysis were those with a P-value <0.05 in univariate analysis. Statistical significance was defined as a P-value <0.05. All tests were two-sided. The software used was SPSS statistic V23.
Results
Study population and characteristics of the LBBAP procedure
From June 2020 to August 2021, 163 patients underwent pacemaker implantation with an attempt of LBBAP. The baseline characteristics of the study population are described in Table 1. LBBAP was as successful in 134 patients (82.2%). In patients with failure of LBBAP (n = 29), 11 patients had a ‘qR’ pattern in V1 with an V6 RWPT >80 ms whereas no ‘qR’ pattern in V1 was observed in all others. Echocardiography was performed in 101/134 patients of the successful group (75.3%), 10/11 patients with ‘qR’ pattern only, at a mean follow-up of 2.5 ± 1.9 months post-implantation (Figure 1).
Table 1.
Characteristics of the study population
| Overall population (n = 163) | Successful procedures (n = 134) | Unsuccessful Procedure (n = 29) | P-value | |
|---|---|---|---|---|
| Age (years) | 76 ± 10 | 77 ± 10 | 75.5 ± 11.1 | 0.79 |
| BMI (Kg/m²) | 27.5 ± 4.8 | 27.2 ± 4.8 | 28.7 ± 4.5 | 0.10 |
| Hypertension | 111 (68.1%) | 89 (66.4%) | 22 (75.9%) | 0.39 |
| Dyslipidemia | 81 (49.7%) | 61 (45.5%) | 20 (60.0%) | 0.03 |
| Smoking | 51 (31.3%) | 41 (30.6%) | 10 (34.5%) | 0.66 |
| Diabetes | 46 (28.2%) | 35 (26.1%) | 11 (37.9%) | 0.26 |
| Creatinine blood level (µmoL/L) | 99 ± 40 | 95 ± 35 | 116 ± 56 | 0.09 |
| Atrial fibrillation | 66 (40.5%) | 50 (37.3%) | 16 (55.2%) | 0.10 |
| Cardiomyopathy | ||||
| ȃPrior valvular heart surgery | 28 (17.2%) | 19 (14.2%) | 9 (31.0%) | 0.06 |
| ȃTAVI | 27 (16.6%) | 24 (17.9%) | 3 (10.3%) | 0.42 |
| ȃCoronary disease | 22 (13.5%) | 15 (11.2%) | 7 (24.1%) | 0.08 |
| ȃDilated cardiomyopathy | 11 (6.7%) | 8 (6.0%) | 3 (10.3%) | 0.42 |
| ȃHypertrophic cardiomyopathy | 1 (0.6%) | 1 (0.7%) | 0 | |
| ȃAmyloidosis | 2 (1.2%) | 1 (0.7%) | 1 (3.4%) | 0.33 |
| NTproBNP (pg/mL) | 2909 ± 5127 | 2832 ± 5347 | 3276 ± 3998 | 0.18 |
| Electrocardiogram | ||||
| ȃMean QRS width (ms) | 119 ± 28 | 118 ± 28 | 127 ± 38 | 0.39 |
| ȃLeft bundle branch block | 54 (33.1%) | 47 (35.1%) | 7 (24.1%) | 0.29 |
| ȃRight bundle branch block | 42 (25.8%) | 38 (28.4%) | 4 (13.8%) | 0.16 |
| Echocardiography | ||||
| ȃLVEF (%) | 57 ± 12 | 58 ± 12 | 52 ± 14 | 0.03 |
| ȃLVEF 35–50% | 24 (14.7%) | 18 (13.4%) | 6 (20.7%) | 0.39 |
| ȃLVEF ≤ 35% | 14 (8.6%) | 8 (6.0%) | 6 (20.7%) | 0.02 |
| ȃLeft ventricular dilatation | 22 (13.5%) | 12 (9.0%) | 10 (34.5%) | 0.001 |
| ȃLeft ventricular hypertrophy | 42 (25.8%) | 30 (22.4%) | 12 (41.4%) | 0.06 |
| ȃLeft atrial dilatation | 108 (66.3%) | 86 (64.2%) | 22 (75.9%) | 0.29 |
| ȃRight heart dilatation | 46 (28.2%) | 31 (23.1%) | 15 (51.7%) | 0.003 |
| Pacemaker indication | ||||
| ȃHigh-degree AVB | 132 (81.0%) | 109 (81.3%) | 23 (79.3%) | 0.80 |
| ȃSinus node dysfunction | 6 (3.7%) | 5 (3.7%) | 1 (3.4%) | 1.0 |
| ȃUncontrolled atrial fibrillation | 21 (12.9%) | 19 (14.2%) | 2 (6.9%) | 0.37 |
| ȃOther | 4 (2.5%) | 1 (0.7%) | 3 (10.3%) | 0.02 |
AVB, atrioventricular block; BMI, body mass index; LBBB, left bundle branch block; LVEF, left ventricular ejection fraction; TAVI, trans-aortic valve implantation.
P < 0.05 are marked in bold.
Figure 1.
Flow chart. V6 RWPT, delay from pacing spike to the peak of the R wave in V6.
Characteristics of the study population are listed in Table 1. In multivariate analysis, the only predictive factor for unsuccessful procedures was right ventricular dilatation [Odds ratio (OR) = 3.9, 95% confidence interval (CI): 1.5–10.4, P = 0.006].
In the successful patient population, the mean procedure duration for single-chamber pacemaker and dual-chamber pacemaker were 41 ± 16 min (n = 29) and 47 ± 11 min (n = 102), respectively. The mean fluoroscopy duration was 4.3 ± 3.8 and 3.5 ± 2.9 min (Table 2). Single-chamber pacemakers were implanted for slowly conducted atrial fibrillation in eight patients and before AV nodal ablation for non-controlled atrial arrhythmia in 21.
Table 2.
Characteristics of the successful pacemaker implantation procedures
| Successful procedure n = 131a | |
|---|---|
| Single-chamber pacemaker | 29 (21.6%) |
| ȃTotal procedure duration (min) | 41 ± 16 [35; 30–52] |
| ȃTotal fluoroscopic duration (min) | 4.3 ± 3.8 [3.2; 2.1–5.4] |
| ȃTotal X-ray exposure (Gy.cm²) | 1.6 ± 1.9 [1.09; 0.53–2.1] |
| Dual-chamber pacemaker | 102 (76.1%) |
| ȃTotal procedure duration (min) | 47 ± 11 [45; 40–53] |
| ȃTotal fluoroscopic duration (min) | 3.5 ± 2.9 [2.5; 2–4.2] |
| ȃTotal X-Ray exposure (Gy.cm²) | 1.3 ± 1.1 [1.0; 0.52–1.66] |
| Unipolar pacing impedance (Ohms) | 559 ± 119 |
| Bipolar pacing impedance (Ohms) | 762 ± 113 |
| R-wave amplitude (mV) | 12.1 ± 5.3 |
| Unipolar pacing threshold (V @ 0.4 ms) | 0.56 ± 0.17 |
| Bipolar pacing threshold (V @ 0.4 ms) | 0.61 ± 0.23 |
[median, 1st quartile, 3rd quartile].
Three successful procedures were upgraded to cardiac resynchronization therapy and are not reported here.
Lead parameters and follow-up
Lead parameters and follow-up are presented in Table 2 (and Supplementary material online, Figure S1).
At implantation, the mean ventricular pacing threshold and ventricular R-wave amplitude were, respectively, 0.6 ± 0.24 V at 0.4 ms and 11.9 ± 5.3 mV. After a mean follow-up of 5.8 ± 3.5 months, these electrical parameters remained stable. However, compared with baseline, we observed a significant decrease in pacing impedance after 3 months (635 ± 165 Ohms vs. 450 ± 94; P < 0.001).
There was no pneumothorax or pericardial effusion related to the procedure. LBBAP lead dislodgement occurred in one patient the day after the implantation. Three patients died during follow-up, none of these deaths was related to a device dysfunction.
Electrocardiographic parameters
Electrocardiographic parameters are summarised in Table 3. In the successful population, the mean paced QRS duration was 104 ± 12 ms, QRS axis - 6 ± 46°, V6 RWPT 68 ± 10 ms, and R/Q ratio 0.6 ± 1.1. The R/Q ratio in V1 had no effect on QRS duration (or on echocardiography parameters). When measuring all paced QRS from pacing stimulus to the final QRS component, the mean paced QRS duration was 135 ± 15 ms.
Table 3.
ECG and echocardiography features during LBBAP
| Successful procedures | qR pattern in V1 with V6 RWPT > 80 ms | P-value | |
|---|---|---|---|
| Electrocardiographic parameters | n = 134 | n = 11 | |
| ȃQRS width (ms) | 104 ± 12 | 118 ± 11 | 0.001 |
| ȃQRS axis (°) | − 6 ± 46 | − 8 ± 45 | 0.99 |
| ȃV6 RWPT (ms) | 68 ± 10 | 97 ± 11 | <0.001 |
| ȃR/Q ratio in V1 | 0.6 ± 1.1 | 0.8 ± 1.1 | 0.71 |
| ȃS wave in V6 | 82 (61.2%) | 7 (63.6%) | 1.0 |
| Echocardiographic parameters | n = 101 | n = 10 | |
| ȃLead/Apex ratio | 0.38 ± 0.11 | 0.33 ± 0.1 | 0.25 |
| ȃ|QAp-QAo| (ms) | 11 ± 6 | 14 ± 13 | 0.55 |
| 2D speckle tracking parameters | n = 91 | n = 10 | |
| ȃA3C mechanical dispersion (ms) | 41 ± 26 | 68 ± 41 | 0.03 |
| ȃA4C mechanical dispersion (ms) | 55 ± 29 | 73 ± 35 | 0.20 |
| ȃA2C mechanical dispersion (ms) | 43 ± 28 | 64 ± 36 | 0.05 |
| Overall mechanical dispersion (ms) | 46 ± 20 | 69 ± 30 | 0.04 |
A2C, apex two-chamber view; A3C: apex three-chamber view; A4C: apex four-chamber view; V6 RWPT: delay from pacing spike to the peak of the R wave in V6; QAp: left ventricular pre-ejection time; QAo: right ventricular pre-ejection time.
P < 0.05 are marked in bold.
A left ventricular axis beyond—30° (n = 41 patients) was associated with a wider QRS (110 ± 12 vs. 102 ± 12 ms, P = 0.006) (see Supplementary material online, Table S1).
In narrow QRS population, a slight increase of QRS width from 89 ± 17 to 101 ± 10 ms (P < 0.001) was observed during LBBAP. Conversely, in patients with LBBB, or right bundle branch block (RBBB), LBBAP decreased the QRS width from 139 ± 16 to 105 ± 12 ms (P < 0.001) and from 134 ± 14 to 106 ± 13 ms (P < 0.001) respectively (Table 4).
Table 4.
ECG and echocardiographic data during spontaneous rhythm and successful left LBBAP
| Successful population n = 101 | P | Left bundle branch block n = 26 | P | Right bundle branch block n = 33 | P | Narrow QRS n = 42 | P | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LBBAP | Spontaneous rhythm | LBBAP | Spontaneous rhythm | LBBAP | Spontaneous rhythm | LBBAP | Spontaneous rhythm | |||||
| QRS width and electromechanical parameters assessment | ||||||||||||
| n = 101 | n = 62 | n = 26 | n = 18 | n = 33 | n = 19 | n = 42 | n = 25 | |||||
| ȃQRS width (ms) | 104 ± 12 | 119 ± 28 | <0.001 | 105 ± 12 | 139 ± 16 | <0.001 | 106 ± 13 | 134 ± 14 | <0.001 | 101 ± 10 | 89 ± 17 | <0.001 |
| ȃQAp (ms) | 123 ± 22 | 123 ± 30 | 0.67 | 124 ± 20 | 110 ± 21 | 0.01 | 122 ± 26 | 156 ± 21 | <0.001 | 125 ± 23 | 108 ± 20 | 0.003 |
| ȃQAo (ms) | 109 ± 22 | 119 ± 34 | 0.05 | 112 ± 19 | 159 ± 31 | <0.001 | 105 ± 22 | 106 ± 18 | 0.99 | 111 ± 23 | 102 ± 21 | 0.04 |
| ȃ|QAp-QAo|(ms) | 15 ± 13 | 34 ± 24 | <0.001 | 14 ± 13 | 49 ± 18 | <0.001 | 17 ± 14 | 50 ± 16 | <0.001 | 13 ± 11 | 12 ± 11 | 0.90 |
| 2D speckle tracking assesment | ||||||||||||
| n = 91 | n = 59 | n = 22 | n = 16 | n = 27 | n = 18 | n = 42 | n = 25 | |||||
| ȃA3C mechanical dispersion (ms) | 41 ± 27 | 60 ± 41 | <0.001 | 43 ± 26 | 100 ± 49 | 0.001 | 47 ± 25 | 42 ± 29 | 0.83 | 35 ± 27 | 48 ± 22 | 0.13 |
| ȃA4C mechanical dispersion (ms) | 56 ± 29 | 56 ± 44 | 0.79 | 62 ± 28 | 105 ± 51 | 0.002 | 56 ± 31 | 36 ± 19 | 0.09 | 51 ± 27 | 41 ± 28 | 0.25 |
| ȃA2C mechanical dispersion (ms) | 43 ± 28 | 46 ± 35 | 0.81 | 52 ± 33 | 66 ± 50 | 0.74 | 40 ± 23 | 38 ± 24 | 0.38 | 40 ± 29 | 39 ± 24 | 0.71 |
| ȃOverall mechanical dispersion (ms) | 47 ± 20 | 53 ± 36 | 0.27 | 53 ± 21 | 90 ± 46 | 0.009 | 48 ± 18 | 35 ± 19 | 0.09 | 42 ± 20 | 43 ± 18 | 0.71 |
A2C, apex two-chamber view; A3C, apex three-chamber view; A4C, apex four-chamber view; QAp, left ventricular pre-ejection time; QAo, right ventricular pre-ejection time.
P < 0.05 are marked in bold.
The presence of a LBB potential at implantation (25.3%, n = 34/134) was not associated with narrower QRS width (see Supplementary material online, Table S1).
In multiple linear regression analysis, the predictive factors of QRS width were left ventricular mass index, and baseline QRS width (Table 5). The predictive factors of V6 RWPT <80 ms were left ventricular mass index [OR = 0.13, 95% CI: 0.05–0.21, P = 0.001] and lead apex ratio [OR = −36.8, 95% CI (− 61.4–−12.1), P = 0.004] (see Supplementary material online, Table S2).
Table 5.
Predictive factors of QRS width during LBBAP. Univariate and multiple linear regressions
| Univariate analysis | Multivariate analysis | |||||
|---|---|---|---|---|---|---|
| Odds Ratio | 95% CI | P-value | Odds ratio | 95% CI | P-value | |
| V6 RWPT | 0.11 | −0.92–0.31 | 0.28 | |||
| Left ventricular mass index | 0.19 | 0.09–0.29 | <0.001 | 0.15 | 0.06–0.24 | 0.001 |
| QRS axis | − 0.01 | − 0.05–0.04 | 0.78 | |||
| R/Q ratio in V1 | − 0.5 | − 2.4–1.4 | 0.60 | |||
| Baseline QRS width | 0.14 | 0.07–0.21 | <0.001 | 0.18 | 0.10–0.26 | <0.001 |
| LVEF | − 0.14 | − 0.32–0.05 | 0.14 | |||
| |QAp-QAo| | − 0.04 | − 0.23–0.16 | 0.73 | |||
| Lead apex ratio | 28.9 | 7.1–50.6 | 0.01 | 17 | −3.8–38.2 | 0.11 |
V6 RWPT, delay from pacing spike to the peak of the R wave in V6; LVEF, left ventricular ejection fraction; QAp, left ventricular pre-ejection time; QAo, right ventricular pre-ejection time.
P < 0.05 are marked in bold.
In patients with a qR pattern in V1 and V6 RWPT > 80 ms, QRS width was wider (118 ± 11 vs. 104 ± 12 ms, P = 0.001) than in successful LBBAP population.
Echocardiographic parameters
Echocardiographic parameters of mechanical asynchrony are displayed in Table 4. In the overall successful patients, we observed during LBBAP a LVMDT of 41 ± 27 ms in A3C, 56 ± 29 ms in A4C, and 43 ± 28 ms in A2C views. The overall LVMDT was correlated with the width of the QRS (rs = 0.35, P < 0.001). Although this correlation was statistically significant, the rs of 0.35 indicated a rather weak correlation. In patients with V6 RWPT > 80 ms, there was a higher LVMDT (69 ± 30 vs. 46 ± 20 ms, P = 0.04) but no significant difference in terms of inter ventricular mechanical synchrony (|QAp-QAo| 11 ± 6 vs. 14 ± 13, P = 0.55) compared with patients with V6 RWPT < 80 ms (Table 3).
The interventricular septum was the first segment activated in all patients. Of note, a septal flash could be observed in 63/101 patients (62.3%). It was associated with a significant increase in the overall mechanical dispersion time when compared with patients without a septal flash (50 ± 19 vs. 40 ± 19 ms, P < 0.001) but it was not related to larger paced QRS (105 ± 13 ms vs. 103 ± 10 ms). Consistent with the RBBB pattern during LBBAP, the QAp was systematically longer than the QAo with a mean IVMD of 15 ± 13 ms.
In patients with LBBB, the LVMDT in A3C and A4C view decreased significantly, respectively from 100 ± 49 to 43 ± 26 ms (P = 0.001) and from 105 ± 51 to 62 ± 28 ms (P = 0.002) during LBBAP as compared with baseline, whereas the LVMDT in A2C did not significantly decrease (66 ± 50 vs. 52 ± 33 ms, P = 0.74). The overall mechanical dispersion significantly decreased in LBBAP when compared with baseline (90 ± 46 vs. 53 ± 21, P = 0.009).
The IVMD decreased from 49 ± 18 to 14 ± 13 ms (P < 0.001). QAo decreased significantly from 159 ± 31 to 112 ± 19 ms (P < 0.001) whereas QAp increased from 110 ± 21 to 124 ± 20 ms (P = 0.01; example in Figure 2).
In patients with RBBB, LBBAP decreased IVMD from 50 ± 16 to 17 ± 14 ms (P < 0.001) without significantly modifying LVMDT (example in Supplementary material online, Figure S2).
In narrow QRS population, no significant difference was reported regarding the interventricular and intra left ventricular synchrony during LBBAP as compared with baseline rhythm. Concordant with the RBBB pattern, the QAp was significantly prolonged during LBBAP from 108 ± 20 to 125 ± 23 ms (P = 0.003; example in Supplementary material online, Figure S3).
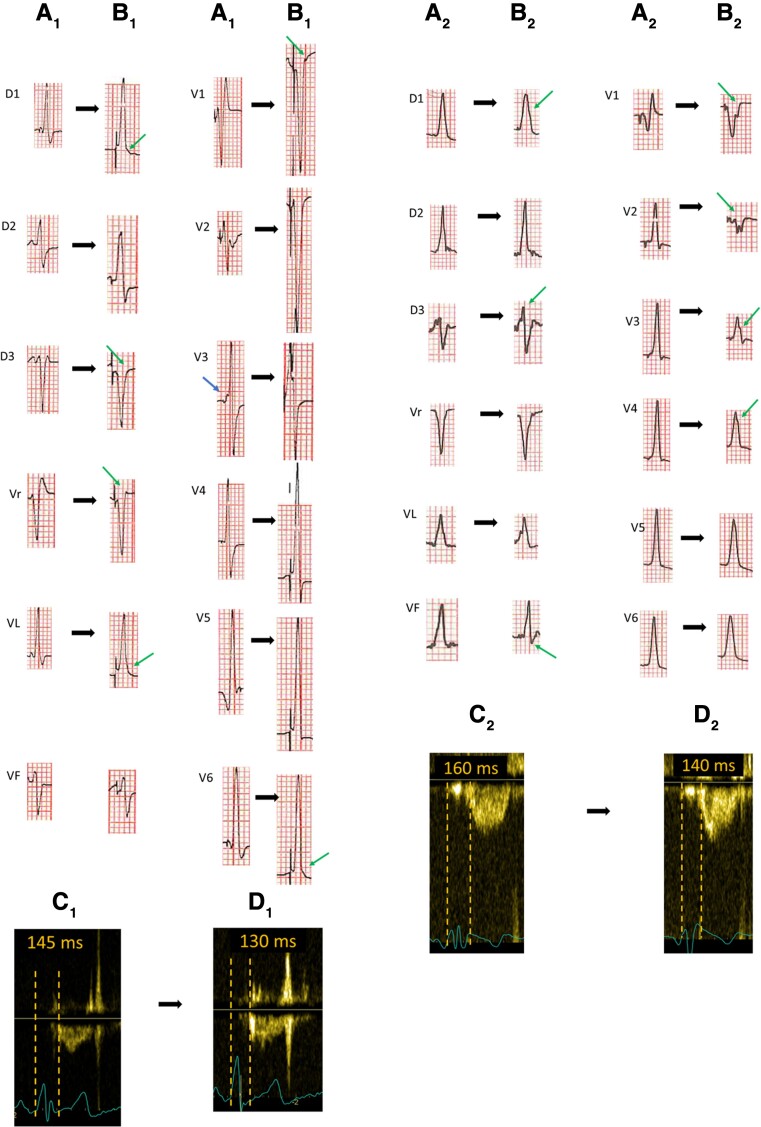
Capture of the surrounding septal myocardium
Capture of the surrounding septal myocardium is presented in Figure 3. Several observations were consistent with a direct interventricular septum capture during LBBAP.
Figure 3.
Capture of the surrounding myocardium. Example of two patients. (A) electrocardiogram with LBBAP at low bipolar output. (B) Electrocardiogram of the same patient with LBBAP at high-bipolar output resulting in anodal capture. (C) Right ventricular pre-ejection time during LBBAP at low bipolar output. (D) Right ventricular pre-ejection time of the same patient during LBBAP at high-bipolar output. Blue arrow, delta wave pattern; green arrows, indicates changes in QRS morphology secondary to anodal capture. LBBAP, left bundle branch area pacing.
In some patients, the recording of a ‘pseudo’ delta wave following the pacing spike and preceding the rapid QRS deflection in the precordial leads demonstrated septal capture. The septal flash observed in echocardiography (63/101 patients), could be attributed to a conflict between direct septal activation and left ventricular depolarisation by the LBB impulse.
In the RBBB population, LBBAP systematically provided a reduction of both QRS width and IVMD (Table 4).
R-wave disappearance in lead V1 without significant QRS widening was reported in 56.1% of the patients in whom high voltage bipolar LBBAP was delivered (46/82 patients). This observation was thought to be related to anodal capture. During anodal capture, no significant changes were observed in LVMDT during 2D speckle tracking assessment (n = 19 patients). Conversely, a decrease of IVMD from 14.8 ± 11.5 to 5.2 ± 12.6 ms was noticed thanks to a shortening of QAp (P = 0.001). Patients in whom anodal capture could be observed had a significantly thicker interventricular septum than the remaining population (11.5 ± 2.6 vs. 9.8 ± 2.0 mm, P = 0.02).
Discussion
In this study, LBBAP slightly lengthened QRS duration in patients with baseline narrow QRS, whereas it significantly shortened QRS duration in patients with baseline RBBB and LBBB. These electrical features were associated with a non-significant impact on ventricular mechanical assessment in patients with narrow QRS and RBBB and with an improvement/normalization of pre-existing asynchrony in patients with LBBB. Our data suggested that it was a safe and effective technique for use in routine practice but much larger multi-center studies are now necessary to assess definitive safety and efficacy of this conduction system pacing approach.
Definition of LBBAP, success rate, and standard electrical parameters
In our study, we have used the association of a ‘qR’ pattern in V1 and an V6 RWPT ≤ 80 ms to define successful LBBAP. In the first publications, the presence of a LBB potential was required to define success.10,11 A LBB potential was recorded in 25.3% of our patients. However, the presence of a LBB potential had no influence on QRS width and LVMDT. This would not support the requirement of LB potential recording to define successful LBBAP.
Other criteria have now been described to precisely differentiate selective (s LBBC) vs. non-selective capture of the LBB (ns LBBC). Prolongation of the V6 RWPT is a recognized marker of transition from ns LBBC to left ventricular septal capture, whereas V1 RWPT prolongation could indicate a transition from ns LBBC to selective capture when decreasing pacing output.8 The distinction between s LBBC and ns LBBC is therefore mainly based on V6 RWPT and/or V1 RWPT prolongations at low pacing output. At the time, our study was initiated, the precise criteria differentiating s LBBC and ns LBBC were not yet clearly established and the measurement of V6–V1 RWPT interval had not been described. We did not pay attention to those criteria during the vast of majority of the inclusion period (June 2020—August 2021). Of note, the QRS prolongation usually occurs at a pacing amplitude way below that of the final programming. As we selected patients with short V6 RWPT (< 80 ms, criteria nearly 100% specific of LBB capture),7 we ensured that the LBB was effectively captured at programmed output (even if non selectively captured). Although comparing dyssynchrony parameters between s LBBC and ns LBBC patients could have been elegant, there was little to no chance that these parameters would be different between these two groups at final programmed pacing output (during ns LBBC QRS pattern).
Compared to patients with V6 RWPT < 80 ms, we observed that patients with qR pattern in V1 and V6 RWPT > 80 ms, had wider paced QRS and greater LVMDT secondary to delayed left lateral wall activation. This feature has been considered as related to left ventricular septal pacing and therefore to the lack of effective LBB capture.12 Our data are in line with these previous studies and support the need for both qR pattern in lead V1 and V6 RWPT ≤ 80 ms criteria to define LBBAP success.
As others, we observed a decrease in pacing impedance values at 3 months while other parameters were stable.13 This could be secondary to the alteration of the surrounding tissue with fibrosis development around the helix.
Left ventricular mechanical synchrony during LBBAP
The mean paced QRS width reported here, was within the range of that of reported in the literature.10,13 Sun el al.14 compared LBBAP with conventional right ventricular pacing using 2D speckle tracking. The authors found that the mechanical dispersion ranges during LBBAP, were equivalent to those observed in our study. Although statistically non-significant, we report here a trend toward to a greater mechanical dispersion in A4C view in 2D speckle tracking during LBBAP as compared to native narrow QRS activation. This is consistent with the fact that QRS width were significantly wider during LBBAP (101 ± 10 ms) than during spontaneous narrow QRS activation (89 ± 17 ms).
In LBBB patients, the QRS width significantly decreased, and the echocardiographic assessment showed improvement in ventricular synchrony. QAo > 140 ms is a marker of intra-ventricular dyssynchrony. This parameter, as well as LVMD and IVMD was markedly reduced during LBBAP (from 159 ± 31 to 112 ± 19 ms). Our results are in line with those of Huang et al.15 study showing a significant reduction of QRS width during LBBAP in LBBB patients and a significant improvement of left ventricular ejection fraction and functional health status during follow-up. Li et al.16 found the same results with a significant benefit in left ventricular remodelling at 6 months compared with standard cardiac resynchronisation therapy (CRT).
In RBBB patients, LBBAP provided QRS narrowing and reduced IVMD. Our study is one of the very few highlighting this finding.17–19 This phenomenon could not be the result of retrograde activation in the left and His bundles followed by antegrade activation of the right bundle. This was necessarily due to rapid activation of the right ventricle, which could be explained by the following hypotheses potentially associated: the capture of the surrounding myocardium (either using high amplitude unipolar pacing or bipolar pacing; i.e. anodal capture) initiating the right ventricle activation or/and the presence of septal connections between right and LBBs (below the level of block in the right bundle), as demonstrated in an animal study by Lazzara et al.20
ECG characteristics during LBBAP
QRS morphology during LBBAP varied from one patient to another without clear understanding. As QRS width was correlated with left ventricular mechanical synchrony (i.e. LVMDT), predictive factors of paced QRS width during LBBAP are of importance. We found that the left ventricular mass index, and the native QRS width were independent predictive factors of paced QRS width. The relationship between QRS width and left ventricular mass index is consistent with physiopathology of myocardial depolarisation. Native narrow QRS complexes attest of healthy ventricular conduction system. Then, LBBAP benefits from this underlying setting and results in narrow paced QRS. We also observed that a left QRS axis deviation, although associated with a wider QRS, did not affect LV mechanical synchrony.
Anodal capture during high-bipolar output has already been described in the literature.21–23 It leads to disappearance or reduction of the R-wave in V1 secondary to direct capture of the right part of interventricular septum. Echocardiographic assessment in this setting confirms this underlying mechanism by reducing IVMD. In our study, the anodal capture was observed in patients with thicker interventricular septum probably due to the close relationship between the proximal lead electrode and the surface of the right interventricular septum.
Limitations
RVP has been clearly demonstrated as the cause of intra and interventricular dyssynchrony. Although our study demonstrated that LBBAP preserved intra-ventricular mechanical synchrony in patients with baseline narrow and RBBB QRS and improved asynchrony parameters in patients with native LBBB, there was no comparison with conventional right ventricular pacing.
Echocardiographic data for mechanical synchrony assessment were lacking in 34/145 patients (23.4%) of the study population due to follow-up performed in other centres. The dependence on good image quality and intra-observer variability, inherent limitations of any echocardiography exam, also have to be stressed. Moreover, after pacemaker implantation, some patients did not have ventricular escape rhythm, precluding echo assessment in spontaneous rhythm. As the echocardiography were performed at a mean of 2.5 ± 1.9 months post-implantation, we cannot exclude that a left ventricular remodelling could have occurred in LBBB patients. The difference of left ventricular synchrony during intrinsic and LBBAP could therefore have been minimized in case of LV remodelling.
The short follow-up duration (5.8 ± 3.5 months) should also be pointed out in the limitations of our study and did not allow us to confirm the stability of the pacing parameters over the long term.
Some of our patients had a ‘qR’ pattern in V1 but a V6 RWPT > 80 ms during pacing. Those patients who did not strictly fulfil our successful criteria for LBBAP have impaired our success rate. However, they underwent echocardiography analysis which confirmed that the mechanical results were not optimal. This underlined the need to assess this parameter during lead implantation to ensure the best results. Due to the small number of patients with qR pattern in V1 and V6 RWPT > 80 ms (n = 11), data reported from that patient group should be taken with cautious and confirmed in larger studies.
Of note, V6 RWPT criteria suffers from some limitations. It is not sensitive enough to diagnose LBB capture in some cases as V6 RWPT might be prolonged due to slower propagation via a diseased His-purkinje conduction system or substantial LV dilatation.24 In our study, 11 patients had qR pattern in V1 and V6 RWPT > 80 ms. To clarify our results, we have considered those 11 procedures as LBBAP failures although they could correspond to LBB capture with prolonged V6 RWPT due to latency in the His-purkinje conduction system. The use of new criteria (i.e. V6-V1 RWPT interval measurement during decreasing pacing output) would facilitate a better distinction between left ventricular septal pacing and LBBAP area pacing in those borderline cases.
Conclusion
LBBAP was found to preserve mechanical synchrony in patients with narrow QRS, whereas it was associated with an improvement of asynchrony parameters, in patients with both right and LBBB. We also found that paced QRS width was associated with left ventricular mass index, and preprocedural QRS width. Although the presence of a LBB potential at implant was a clear indication of a successful procedure, it was not associated with an additional benefit in terms of mechanical left ventricular synchrony.
Supplementary Material
Contributor Information
Adrian Mirolo, Department of cardiology, Rouen University Hospital, CHU—Hôpitaux de Rouen, 1 rue de Germont, 76031 Rouen, France.
Corentin Chaumont, Department of cardiology, Rouen University Hospital, CHU—Hôpitaux de Rouen, 1 rue de Germont, 76031 Rouen, France.
Nathanael Auquier, Department of cardiology, Jacques Monod Hospital, 29 Av. Pierre Mendès France, 76290 Montivilliers, France.
Arnaud Savoure, Department of cardiology, Rouen University Hospital, CHU—Hôpitaux de Rouen, 1 rue de Germont, 76031 Rouen, France.
Benedicte Godin, Department of cardiology, Rouen University Hospital, CHU—Hôpitaux de Rouen, 1 rue de Germont, 76031 Rouen, France.
Florence Vandevelde, Department of cardiology, Jacques Monod Hospital, 29 Av. Pierre Mendès France, 76290 Montivilliers, France.
Helene Eltchaninoff, Department of cardiology, Rouen University Hospital, CHU—Hôpitaux de Rouen, 1 rue de Germont, 76031 Rouen, France.
Frederic Anselme, Department of cardiology, Rouen University Hospital, CHU—Hôpitaux de Rouen, 1 rue de Germont, 76031 Rouen, France.
Supplementary material
Supplementary material is available at Europace online.
Funding
No funding declared.
Data availability
The authors of this study were unable to find a valid data repository for the data produced in this study. Supporting documents are available from Dr Adrian Mirolo (amirolo@free.fr).
References
- 1. Huang W, Su L, Wu S, Xu L, Xiao F, Zhou Xet al. A novel pacing strategy with low and stable output: pacing the left bundle branch immediately beyond the conduction block. Can J Cardiol 2017;33:1736.e1–.e3. [DOI] [PubMed] [Google Scholar]
- 2. Mafi-Rad M, Luermans JGLM, Blaauw Y, Janssen M, Crijns HJ, Prinzen FWet al. Feasibility and acute hemodynamic effect of left ventricular septal pacing by transvenous approach through the interventricular septum. Circ Arrhythm Electrophysiol 2016;9:e003344. [DOI] [PubMed] [Google Scholar]
- 3. Zhang XH, Chen H, Siu CW, Yiu KH, Chan WS, Lee KLet al. New-onset heart failure after permanent right ventricular apical pacing in patients with acquired high-grade atrioventricular block and Normal left ventricular function. J Cardiovasc Electrophysiol 2008;19:136–41. [DOI] [PubMed] [Google Scholar]
- 4. Sweeney MO, Hellkamp AS, Ellenbogen KA, Greenspon AJ, Freedman RA, Lee KLet al. Adverse effect of ventricular pacing on heart failure and atrial fibrillation among patients with Normal baseline QRS duration in a clinical trial of pacemaker therapy for sinus node dysfunction. Circulation 2003;107:2932–7. [DOI] [PubMed] [Google Scholar]
- 5. Brignole M, Auricchio A, Baron-Esquivias G, Bordachar P, Boriani G, Breithardt OAet al. 2013 ESC guidelines on cardiac pacing and cardiac resynchronization therapyThe task force on cardiac pacing and resynchronization therapy of the European society of cardiology (ESC). developed in collaboration with the European heart rhythm association (EHRA). Eur Heart J Oxford Academic 2013;34:2281–329. [DOI] [PubMed] [Google Scholar]
- 6. Huang W, Chen X, Su L, Wu S, Xia X, Vijayaraman P. A beginner’s guide to permanent left bundle branch pacing. Heart Rhythm 2019;16:1791–6. [DOI] [PubMed] [Google Scholar]
- 7. Jastrzębski M, Kiełbasa G, Curila K, Moskal P, Bednarek A, Rajzer Met al. Physiology-based electrocardiographic criteria for left bundle branch capture. Heart Rhythm 2021;18:935–43. [DOI] [PubMed] [Google Scholar]
- 8. Jastrzębski M, Burri H, Kiełbasa G, Curila K, Moskal P, Bednarek Aet al. The V6-V1 interpeak interval: a novel criterion for the diagnosis of left bundle branch capture. Europace 2022;24:40–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PAet al. Recommendations for chamber quantification. Eur J Echocardiogr 2006;7:79–108. [DOI] [PubMed] [Google Scholar]
- 10. Padala SK, Ellenbogen KA. Left bundle branch pacing is the best approach to physiological pacing. Heart Rhythm O2 2020;1:59–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Ponnusamy SS, Arora V, Namboodiri N, Kumar V, Kapoor A, Vijayaraman P. Left bundle branch pacing: a comprehensive review. J Cardiovasc Electrophysiol 2020;31:2462–73. [DOI] [PubMed] [Google Scholar]
- 12. Curila K, Jurak P, Jastrzebski M, Prinzen F, Waldauf P, Halamek Jet al. Left bundle branch pacing compared to left ventricular septal myocardial pacing increases interventricular dyssynchrony but accelerates left ventricular lateral wall depolarization. Heart Rhythm 2021;18:1281–9. [DOI] [PubMed] [Google Scholar]
- 13. Gao J, Zhang BH, Zhang N, Sun M, Wang R. The electrocardiogram characteristics and pacing parameters of permanent left bundle branch pacing: a systematic review and meta-analysis. J Interv Card Electrophysiol 2022;63:215–24. [DOI] [PubMed] [Google Scholar]
- 14. Sun Z, Di B, Gao H, Lan D, Peng H. Assessment of ventricular mechanical synchronization after left bundle branch pacing using 2-D speckle tracking echocardiography. Clin Cardiol 2020;43:1562–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Huang W, Wu S, Vijayaraman P, Su L, Chen X, Cai Bet al. Cardiac resynchronization therapy in patients with nonischemic cardiomyopathy using left bundle branch pacing. JACC Clin Electrophysiol 2020;6:849–58. [DOI] [PubMed] [Google Scholar]
- 16. Li X, Qiu C, Xie R, Ma W, Wang Z, Li Het al. Left bundle branch area pacing delivery of cardiac resynchronization therapy and comparison with biventricular pacing. ESC Heart Fail 2020;7:1711–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Li X, Li H, Ma W, Ning X, Liang E, Pang Ket al. Permanent left bundle branch area pacing for atrioventricular block: feasibility, safety, and acute effect. Heart Rhythm 2019;16:1766–73. [DOI] [PubMed] [Google Scholar]
- 18. Li X, Fan X, Li H, Ning X, Liang E, Ma Wet al. ECG Patterns of successful permanent left bundle branch area pacing in bradycardia patients with typical bundle branch block. Pacing Clin Electrophysiol 2020;43:781–90. [DOI] [PubMed] [Google Scholar]
- 19. Zhu K, Lin M, Li L, Chang D, Li Q. Left bundle branch pacing shortened the QRS duration of a right bundle branch block. J Electrocardiol 2021;68:153–6. [DOI] [PubMed] [Google Scholar]
- 20. Lazzara R, Yeh BK, Samet P. Functional transverse interconnections within the his bundle and the bundle branches. Circ Res 1973;32:509–15. [DOI] [PubMed] [Google Scholar]
- 21. Zhang S, Zhou X, Gold MR. Left bundle branch pacing: JACC review topic of the week. J Am Coll Cardiol 2019;74:3039–49. [DOI] [PubMed] [Google Scholar]
- 22. Vijayaraman P, Huang W. Atrioventricular block at the distal his bundle: electrophysiological insights from left bundle branch pacing. Hear Case Rep 2019;5:233–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Vijayaraman P, Subzposh FA, Naperkowski A, Panikkath R, John K, Mascarenhas Vet al. Prospective evaluation of feasibility and electrophysiologic and echocardiographic characteristics of left bundle branch area pacing. Heart Rhythm 2019;16:1774–82. [DOI] [PubMed] [Google Scholar]
- 24. Jastrzêbski M. ECG And pacing criteria for differentiating conduction system pacing from myocardial pacing. Arrhythmia Electrophysiol Rev 2021;10:172–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The authors of this study were unable to find a valid data repository for the data produced in this study. Supporting documents are available from Dr Adrian Mirolo (amirolo@free.fr).