Abstract
小鼠动物模型作为科学研究中最常用的实验工具,在两个世纪的发展中受到了研究人员的广泛青睐。小鼠白血病动物模型出现于 20 世纪 30 年代,在近 90 年的发展过程中,研究者们开发出了多种类型的小鼠白血病模型来模拟人类白血病的发生发展和药物治疗过程,这在一定程度上推动了白血病发生发展分子机制的阐明和靶向治疗白血病药物的研发进程。考虑到髓系白血病,特别是急性髓系白血病目前在临床上没有很好的治疗措施,急需阐明其新的分子机制和开发新的治疗靶点,本文重点综述了近几年最常用的髓系白血病小鼠模型,包括小鼠品系、髓系白血病细胞类型、成模方式等,以期为相关科研工作者在髓系白血病研究过程中选择动物模型时提供一定的参考。
Keywords: 髓系白血病, 小鼠品系, 异种移植模型, 患者来源的异种移植模型
Abstract
Mouse animal models are the most commonly used experimental tools in scientific research, which have been widely favored by researchers. The animal model of mouse leukemia appeared in the 1930s. During the past 90 years, researchers have developed various types of mouse leukemia models to simulate the development and treatment of human leukemia in order to promote effectively the elucidation of the molecular mechanism of leukemia' development and progression, as well as the development of targeted drugs for the treatment of leukemia. Considering that to myeloid leukemia, especially acute myeloid leukemia, there currently is no good clinical treatment, it is urgent to clarify its new molecular mechanism and develop new therapeutic targets. This review focuses on the various types of mouse models about myeloid leukemia used commonly in recent years, including mouse strains, myeloid leukemia cell types, and modeling methods, which are expected to provide a reference for relevant researchers to select animal models during myeloid leukemia research.
Keywords: myeloid leukemia, mouse strains, xenograft models, patient derived xenograft model
引言
白血病是一类造血干细胞恶性克隆性疾病,克隆性白血病细胞因为增殖失控、分化障碍、凋亡受阻等机制在骨髓和其他造血组织中大量增殖累积,并浸润其他非造血组织和器官,同时抑制正常造血功能。在 2018 年肿瘤年报中,新增 437 033 例白血病病例,占所有新增肿瘤病例的 2.4%[1]。常见白血病可分为髓系和淋巴系两大类,根据白血病的分化程度和自然病程的长短,髓系白血病又可分为急性髓系白血病(acute myeloid leukemia,AML)和慢性髓系白血病(chronic myeloid leukemia,CML)两大类。AML 是一种血液系统恶性增生性疾病,也是一种遗传异质性癌症,起源于造血干细胞,起因是骨髓中未成熟原始粒细胞快速且不受控制的生长,其大量积聚并渗入其他组织,导致 AML 患者正常造血功能的抑制。CML 是一种骨髓增生性疾病,以费城(Ph)染色体为特征,9 号染色体(ch9)和 22 号染色体(ch22)的长臂之间相互易位。该易位导致 9 号染色体上的 ABL 基因和 22 号染色体上的 BCR 基因融合,形成编码 BCR-ABL 癌蛋白的 DNA 序列,该癌蛋白是促进不受控制的骨髓细胞生成的组成型活性酪氨酸激酶。这种疾病最初由 Craigie、Bennett 和 Virchow 在 1845 年描述[2],占所有白血病的 15%。大多数 CML 患者处于慢性期,其症状可以通过格列卫等靶向药物进行控制,但是 CML 患者一旦进入急变期,将转变为 AML 并伴随一系列 AML 的临床特征,如发热、出血、贫血和浸润等。
小鼠是由小家鼠演变而来,作为模式生物的一种,被广泛应用于科学研究中。它广泛分布于世界各地,经长期人工饲养选择培育,已育成 1 000 多近交系和独立的远交群。早在 17 世纪就有人用小鼠做实验,有文献记载的可以追溯到 19 世纪[3],小鼠已成为使用量最大、研究最详尽的哺乳类实验动物。1930 年,人们开始使用小鼠白血病模型[4]。如今小鼠的品系有上千种,用于白血病动物模型的一般分为近交系和突变系两种,简要总结见表 1。
表 1. Mouse strains used in common mouse models of leukemia.
常见白血病模型所用小鼠品系
| 小鼠品系 | 特征 | |
| 近交系 | C57BL/6 | 作为产生自发突变和诱发突变的同基因型小鼠的背景品系 |
| FVB/N | 带有对 B 型 Friend 白血病病毒敏感的 Fv1b 因子 | |
| BALB/C | 具有白化、免疫缺陷的特征,对致癌物敏感 | |
| CB-17 | BALB/C 同源近交产生的小鼠品系 | |
| 突变系 | BALB/c-nude | 为 BALB/C 的突变品系,胸腺发育不良,免疫 T 细胞缺失 |
| SCID | CB-17 SCID 小鼠为 CB-17 的 SCID 突变系(常染色体隐性突变),丧失 B 淋巴细胞与 T 淋巴细胞的功能 | |
| NOD-SCID | 将 SCID 突变基因导入到非肥胖糖尿病背景小鼠(NOD 小鼠)获得,在 SCID 小鼠的基础上降低了 NK 细胞活性 | |
| NSG | NOD/SCID/IL2-Rγ-/-,NOD-SCID 小鼠模型上导入 IL-2 受体 γ 链缺失突变,具有重度免疫缺陷表型,无成熟 T 细胞、B 细胞和功能性 NK 细胞,是迄今为止免疫缺陷程度最高的工具小鼠 |
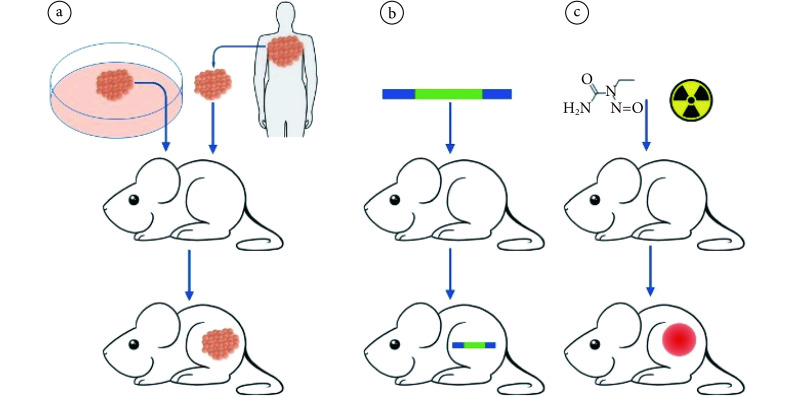
科学研究过程中会根据实验目的不同选择不同的小鼠品系和白血病小鼠模型。常用的白血病小鼠模型有三种:异种移植模型、基因修饰型白血病模型和诱发性白血病模型(见图 1)。髓系白血病小鼠模型已经在科学研究中得到广泛使用,但目前少见相关综述,为了科研工作者能更加方便快捷地选择符合实验目的的小鼠模型,现对近年相关文献进行总结。
图 1.
Common mouse models of leukemia
常用的白血病小鼠模型
a. xenograft model: leukemia cells derived from leukemia cell lines or patient-derived leukemia cells are transplanted into mice by various means to obtain leukemia mice; b. genetic modification model: a genetically modified mouse model was obtained by introducing a gene of interest into a mouse genome; c. induced model: the mouse is induced to produce artificial leukemia by chemical or physical means
a. 异种移植模型:白血病细胞系来源或患者来源的白血病细胞通过各种手段移植到小鼠体内,得到白血病小鼠;b. 基因修饰模型:通过将目的基因导入小鼠基因组,获得基因修饰小鼠模型;c. 诱发性模型:通过化学或物理手段诱导使小鼠产生人为的白血病
1. 异种移植模型
异种移植模型是最常用的动物模型,其具体内容就是将白血病细胞移植到免疫缺陷的小鼠体内,细胞来源可分为白血病细胞系细胞(见表 2)、患者来源的细胞以及转染的小鼠骨髓细胞。
表 2. Myeloid leukemia cell lines.
常用髓系白血病细胞株
| 髓系白血病分类 | 常见细胞株 | |
| 急性髓系白血病细胞系 | 急性髓性白血病微分化型(M0) | M0-91、MOLM-13、MOLM-14、OCI-AML、SKNO-1、CMK-11-5 |
| 粒细胞白血病未分化型(M1) | KG-1a | |
| 粒细胞白血病部分分化型(M2) | Kasumi-1 | |
| 早幼粒细胞型(M3) | HL-60、NB4 | |
| 粒-单核细胞型(M4) | ML-2 | |
| 单核细胞型(M5) | THP-1、MV-4-11、SHI-1 | |
| 红白血病(M6) | HEL、TF-1 | |
| 巨核细胞型(M7) | M-07e | |
| 慢性髓系白血病细胞 | K562、CML-T1、KU812、BV173 | |
| 小鼠白血病细胞 | C1498、EL9611 | |
根据实验目的选择不同的小鼠和细胞后,通常的接种方式有皮下接种、眼眶后接种、骨髓接种和尾静脉接种。在接种细胞之前,需要尽可能地破坏小鼠体内残存的免疫力,方法有射线辐射法和药物处理两种。射线辐射是指用半致死的 γ 射线辐射小鼠,该方法始于 1956 年 Congdon 等[5]发现射线可以很大程度上破坏小鼠的免疫力。药物处理是指用免疫抑制剂来破坏小鼠的残存免疫力,常用的有环磷酰胺等,在 1951 年 Burchenal 等[6]首次用药物来抑制小鼠的免疫力。
1.1. 皮下接种
皮下接种是较为常见的一种接种方式,其优点是操作简便,易于观察,成瘤效果好(见表 3[7-28])。从近年来的文献中我们可以看到,BALB/c-nude 小鼠在皮下接种受体小鼠中最受欢迎,这可能与它没有毛的特性有关,有利于实验者观察肿瘤的生长情况。皮下接种的验证成瘤方式为直接观察,并用游标卡尺测量肿瘤的长和宽,计算肿瘤体积公式为:V = (长度 × 宽度2)/2(其中宽度定义为两次测量中较小的一次,长度定义为两者中较大者)。在肿瘤达到 100~150 mm3时开始使用药物处理,当肿瘤过大或者注射细胞 30 天左右处死小鼠,取出肿瘤和需要的脏器。肿瘤和脏器可以切片染色,观察白血病的侵染情况;也可以通过将其制成蛋白样品来探究白血病发生发展的分子机制;还可以将组织块分散为单细胞悬液,通过流式细胞仪分选出目标细胞群,进一步实验。
表 3. Mouse models for leukemia by subcutaneous inoculation.
皮下注射接种小鼠白血病模型
| 接种细胞种类和数量 | 受体小鼠 | 参考文献 |
| 2 × 106个 M0-91(AML) | SCID | [7] |
| 3 × 106~2 × 107个 SKNO-1(AML) | BALB/c-nude | [8-9] |
| MOLM-14(AML) | BALB/c-nude | [10] |
| 1 × 106~5 × 106个 OCI-AML(AML) | SCID | [11-12] |
| 3 × 106~1 × 107个 HL-60(AML) | SCID | [12] |
| BALB/c-nude | [13-14] | |
| BALB/C | [15] | |
| 5 × 106~2 × 107个 Kasumi-1(AML) | BALB/c-nude | [9] |
| SCID | [16] | |
| 2 × 106个 HEL(AML) | BALB/c-nude | [17] |
| SCID | [18] | |
| 1.4 × 105~1 × 107个 MV-4-11(AML) | SCID | [19] |
| NOD-SCID | [20] | |
| SCID | [12, 21] | |
| BALB/c-nude | [22] | |
| 1 × 107个 SHI-1(AML) | BALB/c-nude | [23] |
| 2 × 106个 CMK-11-5(AML)
2 × 106个 TF-1(AML) |
SCID | [18] |
| 1 × 107个 NB4(AML) | BALB/c-nude | [9] |
| 2 × 106~2 × 107个 K562(CML) | BALB/c-nude | [9, 24] |
| NOD-SCID | [25] | |
| 6 × 106个 TEL-ABLT315I-BaF3 细胞(CML) | BALB/c-nude | [26] |
| 1 × 106个 FLAG-EWSR1/ELF5 转导的 NIH3T3 细胞 | NSG | [27] |
| 4 × 106个 AML 患者细胞 | NSG | [28] |
1.2. 眼眶后接种
眼眶后接种在异种移植模型中属于应用较少的方式。在近 5 年的文章中,McGill 等[29]通过眼眶后接种将 C1498 细胞移植到 C57BL/6 小鼠(1 × 106个细胞/小鼠)研究蓝莓提取物抗 AML 功效;Ferreira 等[30]将从 hCG-PML-RARα 转基因小鼠获得的白血病细胞(2 × 106个细胞/小鼠)注射到 10~12 周龄的 NOD-SCID 小鼠眼丛中,评估磷酸乙醇胺能否成为治疗白血病的药物;Zhang 等[31]将 MV-4-11 细胞(2.5 × 106个细胞/小鼠)通过眼眶后接种到 NOD-SCID 小鼠,证明了 CD244 可作为治疗 AML 的新型治疗靶标。从上述三例实验我们可以发现,NOD-SCID 小鼠是眼眶后接种的首选实验对象,而眼眶后接种的最大缺点可能在于实验难度过大,建模成功率不高。
1.3. 骨髓接种
骨髓接种是指通过钻骨针在小鼠股骨钻孔,然后使用微量进样器将白血病细胞直接注入骨髓腔的接种方式。这种方式通常都用来观察白血病细胞对骨的破坏性以及白细胞骨转移后的治疗方式。Nóbrega-Pereira 等[32]将 HEL 细胞注射到 NSG 小鼠(1 × 105个细胞/小鼠)的右胫骨中,研究靶向耐药性白血病细胞的线粒体代谢途径的分子机制;Yoshimi 等[33]将从慢性粒单核细胞白血病(chronic myelomonocytic leukemia,CMML)和青少年髓单核细胞白血病(juvenile myelomonocytic leukemia,JMML)患者的外周血中分离得到的 CD34+细胞注射到 NSG 小鼠(0.2 × 106~1.18 × 106个细胞/小鼠)的股骨中,研究在 NSG 小鼠中 CMML 和 JMML 疾病起始和成熟白血病细胞的发展;Mori 等[22]将 MV-4-11-luc 细胞(1 × 106细胞/小鼠)注射到雌性 NOD-SCID 小鼠的左胫骨骨髓,通过全身的生物发光成像监测肿瘤生长;Liyanage 等[11]将原始 AML 细胞(2 × 106~3 × 106个细胞/小鼠)注射到雌性 NOD-SCID 小鼠的右股骨中,一段时间后处死取左股骨,收集骨髓腔中的细胞,通过流式定量评估左股骨中的原代 AML 植入;Zhang 等[31]将 MV-4-11 细胞(2.5 × 106个细胞/小鼠)注射到 NOD-SCID 小鼠胫骨中,用作评估靶向 CD244 治疗白血病的疗效。骨髓接种白血病细胞之后,通过基于流式细胞术的血液(纵向)和外周器官(末期疾病阶段)中肿瘤细胞数量的计数来评估肿瘤进展。NSG 小鼠和 NOD-SCID 小鼠是骨髓接种的热门动物,这与它们的重度免疫缺陷有关;骨髓接种的最大优点在于其模拟发源于骨髓的白血病在小鼠体内的发展过程,这对研究白血病在人体的发展具有重要借鉴作用。
1.4. 尾静脉接种
尾静脉接种是指将细胞通过尾静脉直接接种到小鼠的血液系统中,主要优点是可形成全身性扩散的白血病模型,符合白血病临床进展规律,此模型以动物生存期作为评价药效的主要指标。尾静脉注射白血病细胞后,可通过白细胞(white blood cell,WBC)计数、活体成像(注射细胞带有荧光素酶基因)、流式细胞仪每周监测血液学计数等方法,并利用以下标准验证白血病模型是否构建成功:外周血中存在至少 1%的原始细胞和 3 × 104细胞/L 以上的白细胞增多,血红蛋白水平低于 10 g/dL,血小板低于 5 × 105细胞/L[34]。表 4 显示了尾静脉接种小鼠常用的细胞类型和受体小鼠[35-59],统计结果表明接种人源白血病细胞的受体小鼠多数为 NSG 小鼠;接种小鼠白血病细胞 C1498 细胞则多选用 C57BL/6 小鼠。患者来源的异种移植模型(patient derived xenograft model,PDX)是将患者来源的外周血单核细胞经过增殖接种到小鼠体内,构建白血病小鼠模型。将患者的血液样品经过密度梯度离心,得到外周血单核细胞;使用单克隆抗体 CD45-FITC、CD19-APC、CD10-PerCP-Cy5.5、CD34-PE-Cy7 和 NG2-PE 进行免疫表型分析,分选出目标细胞群,经过体外培养达到足够数量,将细胞注射到小鼠的尾静脉中[60]。
表 4. Mouse models for leukemia by tail vein inoculation.
尾静脉注射接种小鼠白血病模型
| 接种细胞种类和数量 | 受体小鼠 | 参考文献 |
| 3.6 × 105~5 × 106个 MOLM-13(AML) | NSG | [35-37] |
| NOG | [12] | |
| 6 × 105~2 × 106个 MOLM-14(AML) | NSG | [38-39] |
| 5 × 104~2 × 106个 HL-60(AML) | NSG | [39-40] |
| NOD-SCID | [29, 41] | |
| 1 × 106~2 × 106个 MV-4-11(AML) | NSG | [39, 42] |
| 2 × 106个 KG-1a(AML) | NSG | [39] |
| 2.5 × 106个 KG-1(AML) | NOD-SCID | [29] |
| 2.5 × 106个 HEL(AML) | NSG | [43] |
| 2 × 106个 K562(CML) | NOD-SCID | [44] |
| 1 × 106个 BV173(CML) | NOG | [45] |
| 5 × 106个 EL9611 | BALB/C | [23] |
| 1 × 105~5 × 105个 C1498 | C57BL/6 | [35, 46-47] |
| 1 × 105~1.7 × 107个 AML 患者 AML 细胞 | NSG | [35, 48-50] |
| NOG | [51] | |
| SCID | [19, 52] | |
| 1 × 106~2.5 × 106个 CML 患者 CD34+细胞 | NSG | [33, 53-54] |
| NOG | [55] | |
| 1 × 106个 PML/RARα 转基因小鼠获得的白血病细胞 | NOD-SCID | [34] |
| 4 × 105个白血病小鼠的骨髓细胞 | C57BL/6 | [56] |
| 5 × 104个用逆转录病毒 MLL-AF9-IRES-GFP 感染的骨髓细胞 | C57BL/6 | [57] |
| 原代 MLL-AF9 白血病小鼠收获骨髓 GFP+(MLL-AF9+)细胞 | C57BL/6 | [58] |
| 1 × 106个用 MigR1-MLL-AF9 旋转接种的 C57BL/6 小鼠骨髓中分离的 HSPC | C57BL/6 | [59] |
| 1 × 106个 MLL-Af4 或 MLL-AF9 转导的小鼠骨髓细胞 | NSG | [54] |
2. 基因修饰型白血病模型
目前从文献中得知骨髓肿瘤的遗传形式与至少 10 个基因的种系突变有关,包括 ANKRD26、CEBPA、DDX41、ETV6、GATA2、RUNX1、SRP72、TERC、TERT 和 TP53[61-63]。在最近的一项研究中,SRSF2、SF3B1、U2AF1、ZRSR2、ASXL1、EZH2、BCOR 或 STAG2 中突变的存在定义了 AML 的独特遗传亚型,其与临床证实的继发性 AML 具有相似的临床病理学特征[64]。Basova[65]通过将 PU.1 敲降模型(PU.1ure/ure)与 p53-/-突变小鼠杂交得到 PU.1ure/urep53-/-小鼠,在这种小鼠中产生高度侵袭性的 AML。Omidvar 等[66]将 MRP8BCL-2 小鼠与 MRP8NRASD12 小鼠杂交,产生 MRP8[BCL-2/NRASD12]小鼠,具有显著较差的存活率,根据血样结果可分为人类 AML 样或未成熟的小鼠骨髓性白血病。Tsai 等[67]对新生非 M3 的 391 名 AML 患者进行基因检测,发现 37 名患者(9.5%)具有 cohesin 基因突变,同时在这 37 例病例中检测上述白血病常见突变基因,结果显示,30 名(81.1%)患者至少还有一种其他基因突变,如 FLT3-ITD(21.6%)、NPM1(21.6%)、RUNX1 或 ASXL1。Mullenders 等[68]描述了 cohesin 在造血干细胞和祖细胞(hematopoietic stem and progenitor cell,HSPC)稳态中的作用。首先,他们用针对 Rad21、Smc3 和 Smc1a 基因的 shRNA 转染小鼠骨髓 HSPC,结果表明它们的重新植入能力和髓样分化缺陷迅速增加。然后,他们构建具有 cohesin 下调的小鼠模型,一系列检测结果显示该模型小鼠具有骨髓瘤形成的临床特征。Harada 等[69]在 C57BL/6 小鼠中构建 GATA2 亚型突变体(G2fGN/fGN),检查年轻(42~99 天龄)、中年(100~249 天龄)和老年(250 天或更多)小鼠的造血指数,发现 G2fGN/fGN小鼠在其一生中患有血小板减少症,并且在老年时期患有白细胞增多症,这些发现表明 G2fGN/fGN小鼠中的 GATA2 严重损害了 HSPC,这导致了造血系统肿瘤的发展,这些数据意味着 GATA2 可能参与或促成人类白血病的发生。Kaur 等[70]通过构建 NPM1c+ 慢病毒转染人 CD34+HSPC,转染效率为 5%~20%,通过心内注射将转染和未转染的 HSPC 的混合物植入 NSG 新生鼠中,在重建后 8 周开始监测小鼠的人白细胞重建和外周血中的绿色荧光蛋白(green fluorescent protein,GFP)表达。结果显示,注射转染 NPM1c+ 的 HSPC 的小鼠存活时间显著低于对照组小鼠,且病毒转染效率越高,存活时间越短;模型小鼠表现出贫血、血细胞生成障碍、骨髓和血液中白血病细胞的存在,以及白血病细胞向其他器官的浸润等现象,结果显示 NPM1c 可能参与白血病的发生发展。
3. 诱发性白血病模型
诱发性白血病模型是指用物理或化学的方式作用于小鼠,使其产生白血病。化学手段是通过一些致癌或致突变的药剂,如烷基化剂 N-乙基-亚硝基脲(N-Ethyl-N-nitrosourea,ENU)[71-72]。物理方法则是通过一些电离辐射(如 γ 射线)诱发小鼠形成白血病模型。诱发性白血病模型的优点是操作方便,缺点是成模的效率低,并且模型小鼠的个体差异过大。Bernard 等[73]在 8 周龄 WKAH/Hkm 大鼠上,每周 5 天口服给药 6 mg 的 N-丁基亚硝基脲(N-Butylnitrosourea,BNU)(先溶解在 0.3 mL 乙醇中,再用饮用水进一步稀释至 15 mL),连续给药 24 周,接着观察所有大鼠 12 个月,并通过流式细胞术检测外周血监测白血病进程。BNU 处理组总诱导白血病为 60% ± 6%,在白血病大鼠中,白细胞的数量急剧增加,还观察到贫血和血小板减少症。Sugiyama 等[74]使用 Long-Evans 大鼠,通过尾静脉注射二羟甲基丁酸(dimethylolbutanoic acid,DMBA)的脂质乳剂(含有 15% 棉籽油、0.5% DMBA、大豆卵磷脂和 Pluronic F68)构建大鼠白血病模型,使用 25~45 mg/kg 的 DMBA 剂量,在 27 天开始以 10~14 天的间隔进行 5~6 次,作为标准方案,通常使用降低剂量为 45、40、35、30 和 25 mg/kg,因为老年动物对 DMBA 的耐受性较差。在注射 DMBA 前 1 天和注射 7 天后进行血液检查,并进行肝脏活组织检查。行活组织检查时,在乙醚麻醉下通过腹部切口切除肝脏周边的楔形(约 50 mg)。通过每周一次的血液检查发现,外周血在晚期显示轻度或中度白细胞增多;在组织学检查中,大的胚细胞侵入肝窦,最终完全取代窦状隙;在脾脏中,白血病细胞侵入红髓,淋巴滤泡保存至晚期;在淋巴结、骨髓和肾上腺中层不断发现白血病浸润。另外,DMBA 诱导的白血病在 N-ras 基因的密码子 61 处始终具有 A > T 颠换二碱基突变。
4. 结论与展望
肿瘤小鼠模型可以用来研究肿瘤的发生发展机制,也可用来评价治疗肿瘤的临床前药物的效果,从而为药物研发提供临床前药效学的实验数据支持,以及预测该临床前药物可能的不良反应和毒副作用;尽管药物在小鼠上进行过临床前的动物实验研究,但是从动物模型到肿瘤临床试验的平均成功转换率低于 8%[75],甚至存在可能导致实验患者死亡的风险[76]。因此完善改进现有的临床前实验动物模型,提高临床前药物动物水平实验结果适用于人体水平的准确率,是亟待解决的科学问题。本文着重介绍了髓系白血病小鼠的异种移植模型,这也是目前髓系白血病研究中最常用的模型,从最初的皮下接种细胞发展到现在的静脉接种细胞,从原先最基础的近交系小鼠发展到现在的 NSG 突变系小鼠,建模成功率和有效性都有了极大改善。基因修饰模型是在基因编辑技术基础上发展起来的动物模型,目前,基因修饰小鼠白血病模型应用还不广泛,建模技术还有待完善和进一步提高,通过后续科学家们共同的努力,相信今后髓系白血病的基因修饰白血病模型可能为临床前白血病药物的筛选提供更加贴合临床实际的动物模型,从而助力临床髓系白血病药物研发的进程。
利益冲突声明:本文全体作者均声明不存在利益冲突。
Funding Statement
国家自然科学基金(81770176)
References
- 1.Bray F, Ferlay J, Soerjomataram I, et al Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Dameshek W Chronic lymphocytic leukemia—an accumulative disease of immunolgically incompetent lymphocytes. Blood. 1967;29(4 Suppl):566–584. [PubMed] [Google Scholar]
- 3.Nugent E Humblebees vs. field-mice. Science. 1883;2(35):470. doi: 10.1126/science.ns-2.35.470. [DOI] [PubMed] [Google Scholar]
- 4.Richter M N, Macdowell E C Studies on leukemia in mice: I. The experimental transmission of leukemia. J Exp Med. 1930;51(4):659–673. doi: 10.1084/jem.51.4.659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Congdon C C, Gengozian N, Makinodan T Agglutinin production in normal, sublethally irradiated, and lethally irradiated mice treated with mouse bone marrow. J Immunol. 1956;77(4):250–256. [PubMed] [Google Scholar]
- 6.Burchenal J H, Webber L F, Johnston S F Mechanisms of amethopterin resistance in leukemia. II. Effect of cortisone on sensitive and resistant mouse leukemias. Proc Soc Exp Biol Med. 1951;78(1):352–354. doi: 10.3181/00379727-78-19070. [DOI] [PubMed] [Google Scholar]
- 7.Smith K M, Fagan P C, Pomari E, et al Antitumor activity of entrectinib, a Pan-TRK, ROS1, and ALK inhibitor, in ETV6-NTRK3-positive acute myeloid leukemia . Mol Cancer Ther. 2018;17(2):455–463. doi: 10.1158/1535-7163.MCT-17-0419. [DOI] [PubMed] [Google Scholar]
- 8.Gao X N, Yan F, Lin J, et al AML1/ETO cooperates with HIF1alpha to promote leukemogenesis through DNMT3a transactivation. Leukemia. 2015;29(8):1730–1740. doi: 10.1038/leu.2015.56. [DOI] [PubMed] [Google Scholar]
- 9.Shan Zhiling, Zhu Xinyu, Ma Pengpeng, et al PML(NLS-) protein: A novel marker for the early diagnosis of acute promyelocytic leukemia. Mol Med Rep. 2017;16(4):5418–5424. doi: 10.3892/mmr.2017.7272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Green A S, Maciel T T, Hospital M A, et al Pim kinases modulate resistance to FLT3 tyrosine kinase inhibitors in FLT3-ITD acute myeloid leukemia. Sci Adv. 2015;1(8):e1500221–e1500233. doi: 10.1126/sciadv.1500221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liyanage S U, Hurren R, Voisin V, et al Leveraging increased cytoplasmic nucleoside kinase activity to target mtDNA and oxidative phosphorylation in AML. Blood. 2017;129(19):2657–2666. doi: 10.1182/blood-2016-10-741207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Floc'h N, Ashton S, Taylor P, et al Optimizing therapeutic effect of Aurora B inhibition in acute myeloid leukemia with AZD2811 nanoparticles. Mol Cancer Ther. 2017;16(6):1031–1040. doi: 10.1158/1535-7163.MCT-16-0580. [DOI] [PubMed] [Google Scholar]
- 13.Li Jingdong, Zi Youmei, Wang Wanling, et al Long noncoding RNA MEG3 inhibits cell proliferation and metastasis in chronic myeloid leukemia via targeting miR-184. Oncol Res. 2018;26(2):297–305. doi: 10.3727/096504017X14980882803151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gu Yueli, Si Jinchun, Xiao Xichun, et al miR-92a inhibits proliferation and induces apoptosis by regulating methylenetetrahydrofolate dehydrogenase 2(MTHFD2) expression in acute myeloid leukemia. Oncol Res. 2017;25(7):1069–1079. doi: 10.3727/096504016X14829256525028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhu Baomin, Zhang Huanying, Yu Lianling Novel transferrin modified and doxorubicin loaded Pluronic 85/lipid-polymeric nanoparticles for the treatment of leukemia: In vitro and in vivo therapeutic effect evaluation . Biomed Pharmacother. 2017;86:547–554. doi: 10.1016/j.biopha.2016.11.121. [DOI] [PubMed] [Google Scholar]
- 16.Chen L T, Chen C T, Jiaang W T, et al BPR1J373, an oral multiple tyrosine kinase inhibitor, targets c-KIT for the treatment of c-KIT-driven myeloid leukemia. Mol Cancer Ther. 2016;15(10):2323–2333. doi: 10.1158/1535-7163.MCT-15-1006. [DOI] [PubMed] [Google Scholar]
- 17.Heo S K, Noh E K, Kim J Y, et al Targeting c-KIT (CD117) by dasatinib and radotinib promotes acute myeloid leukemia cell death. Sci Rep. 2017;7(1):1–12. doi: 10.1038/s41598-016-0028-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ishikawa Y, Gamo K, Yabuki M, et al A novel LSD1 inhibitor T-3775440 disrupts GFI1B-containing complex leading to transdifferentiation and impaired growth of AML cells. Mol Cancer Ther. 2017;16(2):273–284. doi: 10.1158/1535-7163.MCT-16-0471. [DOI] [PubMed] [Google Scholar]
- 19.Morgado-Palacin I, Day A, Murga M, et al Targeting the kinase activities of ATR and ATM exhibits antitumoral activity in mouse models of MLL-rearranged AML. Sci Signal. 2016;9(445):ra91–ra107. doi: 10.1126/scisignal.aad8243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Maes T, Mascaro C, Tirapu I, et al ORY-1001, a potent and selective covalent KDM1A inhibitor, for the treatment of acute leukemia. Cancer Cell. 2018;33(3):495–511. doi: 10.1016/j.ccell.2018.02.002. [DOI] [PubMed] [Google Scholar]
- 21.Puente-Moncada N, Costales P, Antolin I, et al Inhibition of FLT3 and PIM kinases by EC-70124 exerts potent activity in preclinical models of acute myeloid leukemia. Mol Cancer Ther. 2018;17(3):614–624. doi: 10.1158/1535-7163.MCT-17-0530. [DOI] [PubMed] [Google Scholar]
- 22.Mori M, Kaneko N, Ueno Y, et al Gilteritinib, a FLT3/AXL inhibitor, shows antileukemic activity in mouse models of FLT3 mutated acute myeloid leukemia. Invest New Drugs. 2017;35(5):556–565. doi: 10.1007/s10637-017-0470-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen Meiyu, Xiong Fei, Ma Liang, et al Inhibitory effect of magnetic Fe3O4 nanoparticles coloaded with homoharringtonine on human leukemia cells in vivo and in vitro . Int J Nanomedicine. 2016;11:4413–4422. doi: 10.2147/IJN.S105543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Park G T, Heo J R, Kim S U, et al The growth of K562 human leukemia cells was inhibited by therapeutic neural stem cells in cellular and xenograft mouse models. Cytotherapy. 2018;20(9):1191–1201. doi: 10.1016/j.jcyt.2018.05.008. [DOI] [PubMed] [Google Scholar]
- 25.Bellavia D, Raimondo S, Calabrese G, et al Interleukin 3-receptor targeted exosomes inhibit in vitro and in vivo chronic myelogenous leukemia cell growth . Theranostics. 2017;7(5):1333–1345. doi: 10.7150/thno.17092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu Xuesong, Wang Beilei, Chen Cheng, et al Discovery of (E)-N-(4-((4-methylpiperazin-1-yl)methyl)-3-(trifluoromethyl)phenyl)-3-((3-(2-(pyridin-2-yl)vinyl)-1H-indazol-6-yl)thio)propanamide (CHMFL-ABL-121) as a highly potent ABL kinase inhibitor capable of overcoming a variety of ABL mutants including T315I for chronic myeloid leukemia. Eur J Med Chem. 2018;160:61–81. doi: 10.1016/j.ejmech.2018.10.007. [DOI] [PubMed] [Google Scholar]
- 27.Endo A, Tomizawa D, Aoki Y, et al EWSR1/ELF5 induces acute myeloid leukemia by inhibiting p53/p21 pathway. Cancer Sci. 2016;107(12):1745–1754. doi: 10.1111/cas.13080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Carretta M, De Boer B, Jaques J, et al Genetically engineered mesenchymal stromal cells produce IL-3 and TPO to further improve human scaffold-based xenograft models. Exp Hematol. 2017;51:36–46. doi: 10.1016/j.exphem.2017.04.008. [DOI] [PubMed] [Google Scholar]
- 29.McGill C M, Brown T J, Cheng Y Y, et al Therapeutic effect of blueberry extracts for acute myeloid leukemia. Int J Biopharm Sci. 2018;1(1):102–115. [PMC free article] [PubMed] [Google Scholar]
- 30.Ferreira A K, Santana-Lemos B, Rego E M, et al Synthetic phosphoethanolamine has in vitro and in vivo anti-leukemia effects . Br J Cancer. 2013;109(11):2819–2828. doi: 10.1038/bjc.2013.510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhang Feifei, Liu Xiaoye, Chen Chiqi, et al CD244 maintains the proliferation ability of leukemia initiating cells through SHP-2/p27(kip1) signaling. Haematologica. 2017;102(4):707–718. doi: 10.3324/haematol.2016.151555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nóbrega-Pereira S, Caiado F, Carvalho T A, et al VEGFR2-mediated reprogramming of mitochondrial metabolism regulates the sensitivity of acute myeloid leukemia to chemotherapy. Cancer Res. 2018;78(3):731–741. doi: 10.1158/0008-5472.CAN-17-1166. [DOI] [PubMed] [Google Scholar]
- 33.Yoshimi A, Balasis M E, Vedder A, et al Robust patient-derived xenografts of MDS/MPN overlap syndromes capture the unique characteristics of CMML and JMML. Blood. 2017;130(4):397–407. doi: 10.1182/blood-2017-01-763219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Torello C O, Shiraishi R N, Della Via F I, et al Reactive oxygen species production triggers green tea-induced anti-leukaemic effects on acute promyelocytic leukaemia model. Cancer Lett. 2018;414:116–126. doi: 10.1016/j.canlet.2017.11.006. [DOI] [PubMed] [Google Scholar]
- 35.Jin Yanxia, Yang Qian, Liang Li, et al Compound kushen injection suppresses human acute myeloid leukaemia by regulating the Prdxs/ROS/Trx1 signalling pathway. J Exp Clin Cancer Res. 2018;37(1):277–289. doi: 10.1186/s13046-018-0948-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Battula V L, Le P M, Sun J C, et al AML-induced osteogenic differentiation in mesenchymal stromal cells supports leukemia growth. JCI Insight. 2017;2(13):e90036–e90053. doi: 10.1172/jci.insight.90036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cartellieri M, Feldmann A, Koristka S, et al Switching CAR T cells on and off: a novel modular platform for retargeting of T cells to AML blasts. Blood Cancer J. 2016;6(8):e458–e475. doi: 10.1038/bcj.2016.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Carter B Z, Mak P Y, Wang X, et al Focal adhesion kinase as a potential target in AML and MDS. Mol Cancer Ther. 2017;16(6):1133–1144. doi: 10.1158/1535-7163.MCT-16-0719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Saland E, Boutzen H, Castellano R, et al A robust and rapid xenograft model to assess efficacy of chemotherapeutic agents for human acute myeloid leukemia. Blood Cancer J. 2015;5:e297–e304. doi: 10.1038/bcj.2015.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tashiro H, Sauer T, Shum T, et al Treatment of acute myeloid leukemia with T cells expressing chimeric antigen receptors directed to C-type lectin-like molecule 1. Molecular Therapy. 2017;25(9):2202–2213. doi: 10.1016/j.ymthe.2017.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chen Jiajie, Zhou Wei, Cai Nan, et al In vivo murine model of leukemia cell-induced spinal bone destruction . Biomed Res Int. 2017;2017:1–6. doi: 10.1155/2017/3521481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhang C C, Yan Z, Pascual B, et al Gemtuzumab ozogamicin (GO) inclusion to induction chemotherapy eliminates leukemic initiating cells and significantly improves survival in mouse models of acute myeloid leukemia. Neoplasia. 2018;20(1):1–11. doi: 10.1016/j.neo.2017.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Saenz D T, Fiskus W, Manshouri T, et al BET protein bromodomain inhibitor-based combinations are highly active against post-myeloproliferative neoplasm secondary AML cells. Leukemia. 2017;31(3):678–687. doi: 10.1038/leu.2016.260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xu Xuefen, Zhang Xiaobo, Zhang Yi, et al Wogonin reversed resistant human myelogenous leukemia cells via inhibiting Nrf2 signaling by Stat3/NF-kappa B inactivation. Sci Rep. 2017;7:1–15. doi: 10.1038/srep39950. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 45.Morita M, Nishinaka Y, Kato I, et al Dasatinib induces autophagy in mice with Bcr-Abl-positive leukemia. Int J Hematol. 2017;105(3):335–340. doi: 10.1007/s12185-016-2137-5. [DOI] [PubMed] [Google Scholar]
- 46.Shen Na, Yan Fei, Pang Jiuxia, et al Inactivation of receptor tyrosine kinases reverts aberrant DNA methylation in acute myeloid leukemia. Clin Cancer Res. 2017;23(20):6254–6266. doi: 10.1158/1078-0432.CCR-17-0235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Deng Rong, Shen Na, Yang Yang, et al Targeting epigenetic pathway with gold nanoparticles for acute myeloid leukemia therapy. Biomaterials. 2018;167:80–90. doi: 10.1016/j.biomaterials.2018.03.013. [DOI] [PubMed] [Google Scholar]
- 48.Her Z, Yong K S, Paramasivam K, et al An improved pre-clinical patient-derived liquid xenograft mouse model for acute myeloid leukemia. J Hematol Oncol. 2017;10(1):162–175. doi: 10.1186/s13045-017-0532-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Paczulla A M, Dirnhofer S, Konantz M A, et al Long-term observation reveals high-frequency engraftment of human acute myeloid leukemia in immunodeficient mice. Haematologica. 2017;102(5):854–864. doi: 10.3324/haematol.2016.153528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Venton G, Perez-Alea M, Baier C, et al Aldehyde dehydrogenases inhibition eradicates leukemia stem cells while sparing normal progenitors. Blood Cancer J. 2016;6(9):e469–e477. doi: 10.1038/bcj.2016.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fukushima N, Minami Y, Kakiuchi S, et al Small-molecule Hedgehog inhibitor attenuates the leukemia-initiation potential of acute myeloid leukemia cells. Cancer Sci. 2016;107(10):1422–1429. doi: 10.1111/cas.13019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pomeroy E J, Lee L A, Lee R D W, et al Ras oncogene-independent activation of RALB signaling is a targetable mechanism of escape from NRAS(V12) oncogene addiction in acute myeloid leukemia. Oncogene. 2017;36(23):3263–3273. doi: 10.1038/onc.2016.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Peterson L F, Lo M C, Liu Yihong, et al Induction of p53 suppresses chronic myeloid leukemia. Leuk Lymphoma. 2017;58(9):2165–2175. doi: 10.1080/10428194.2016.1272682. [DOI] [PubMed] [Google Scholar]
- 54.Lin H, Woolfson A, Jiang X New mouse models to investigate the efficacy of drug combinations in human chronic myeloid leukemia. Methods Mol Biol. 2016;1465:187–205. doi: 10.1007/978-1-4939-4011-0_16. [DOI] [PubMed] [Google Scholar]
- 55.Jin Yanli, Yao Yiwu, Chen Li, et al Depletion of gamma-catenin by histone deacetylase inhibition confers elimination of CML stem cells in combination with imatinib. Theranostics. 2016;6(11):1947–1962. doi: 10.7150/thno.16139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Laouedj M, Tardif M R, Gil L, et al S100A9 induces differentiation of acute myeloid leukemia cells through TLR4. Blood. 2017;129(14):1980–1990. doi: 10.1182/blood-2016-09-738005. [DOI] [PubMed] [Google Scholar]
- 57.Minzel W, Venkatachalam A, Fink A, et al Small molecules co-targeting CKI alpha and the transcriptional kinases CDK7/9 control AML in preclinical models. Cell. 2018;175(1):171–185. doi: 10.1016/j.cell.2018.07.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Long J, Fang W Y, Chang L, et al Targeting HDAC3, a new partner protein of AKT in the reversal of chemoresistance in acute myeloid leukemia via DNA damage response. Leukemia. 2017;31(12):2761–2770. doi: 10.1038/leu.2017.130. [DOI] [PubMed] [Google Scholar]
- 59.Liang K, Volk A G, Haug J S, et al Therapeutic targeting of MLL degradation pathways in MLL-rearranged leukemia. Cell. 2017;168(1/2):59–72. doi: 10.1016/j.cell.2016.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lopez-Millan B, Sanchez-Martinez D, Roca-Ho H, et al NG2 antigen is a therapeutic target for MLL-rearranged B-cell acute lymphoblastic leukemia. Leukemia. 2019;33:1557–1569. doi: 10.1038/s41375-018-0353-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Godley L A Inherited predisposition to acute myeloid leukemia. Semin Hematol. 2014;51(4):306–321. doi: 10.1053/j.seminhematol.2014.08.001. [DOI] [PubMed] [Google Scholar]
- 62.Polprasert C, Schulze I, Sekeres M A, et al Inherited and somatic defects in DDX41 in myeloid neoplasms. Cancer Cell. 2015;27(5):658–670. doi: 10.1016/j.ccell.2015.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhang M Y, Churpek J E, Keel S B, et al Germline ETV6 mutations in familial thrombocytopenia and hematologic malignancy. Nat Genet. 2015;47(2):180–185. doi: 10.1038/ng.3177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lindsley R C, Mar B G, Mazzola E, et al Acute myeloid leukemia ontogeny is defined by distinct somatic mutations. Blood. 2015;125(9):1367–1376. doi: 10.1182/blood-2014-11-610543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Basova P, Pospisil V, Savvulidi F, et al Aggressive acute myeloid leukemia in PU.1/p53 double-mutant mice. Oncogene. 2014;33(39):4735–4745. doi: 10.1038/onc.2013.414. [DOI] [PubMed] [Google Scholar]
- 66.Omidvar N, Kogan S, Beurlet S, et al BCL-2 and mutant NRAS interact physically and functionally in a mouse model of progressive myelodysplasia. Cancer Res. 2007;67(24):11657–11667. doi: 10.1158/0008-5472.CAN-07-0196. [DOI] [PubMed] [Google Scholar]
- 67.Tsai C H, Hou H A, Tang J L, et al Prognostic impacts and dynamic changes of cohesin complex gene mutations in De Novo acute myeloid leukemia. Blood. 2017;130(1):663–669. doi: 10.1038/s41408-017-0022-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Mullenders J, Aranda-Orgilles B, Lhoumaud P, et al Cohesin loss alters adult hematopoietic stem cell homeostasis, leading to myeloproliferative neoplasms. J Exp Med. 2015;212(11):1833–1850. doi: 10.1084/jem.20151323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Harada N, Hasegawa A, Hirano I, et al GATA2 hypomorphism induces chronic myelomonocytic leukemia in mice. Cancer Sci. 2019;110(4):1183–1193. doi: 10.1111/cas.13959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kaur M, Drake A C, Hu Guangan, et al Induction and therapeutic targeting of human NPM1c(+) myeloid leukemia in the presence of autologous immune system in mice. J Immunol. 2019;202(6):1885–1894. doi: 10.4049/jimmunol.1800366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Joslin J M, Fernald A A, Tennant T R, et al Haploinsufficiency of EGR1, a candidate gene in the del(5q), leads to the development of myeloid disorders. Blood. 2007;110(2):719–726. doi: 10.1182/blood-2007-01-068809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lu E P, Mclellan M, Ding Li, et al Caspase-9 is required for normal hematopoietic development and protection from alkylator-induced DNA damage in mice. Blood. 2014;124(26):3887–3895. doi: 10.1182/blood-2014-06-582551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Bernard N, Devevey L, Jacquemont C, et al A new model of pre-B acute lymphoblastic leukemia chemically induced in rats. Exp Hematol. 2005;33(10):1130–1139. doi: 10.1016/j.exphem.2005.06.018. [DOI] [PubMed] [Google Scholar]
- 74.Sugiyama T, Osaka M, Koami K, et al 7,12-DMBA-induced rat leukemia: a review with insights into future research. Leuk Res. 2002;26(12):1053–1068. doi: 10.1016/S0145-2126(02)00045-0. [DOI] [PubMed] [Google Scholar]
- 75.Mak I W, Evaniew N, Ghert M Lost in translation: animal models and clinical trials in cancer treatment. Am J Transl Res. 2014;6(2):114–118. [PMC free article] [PubMed] [Google Scholar]
- 76.Perrin S Preclinical research: Make mouse studies work. Nature. 2014;507(7493):423–425. doi: 10.1038/507423a. [DOI] [PubMed] [Google Scholar]