ACUTE RESPIRATORY DISTRESS SYNDROME (ARDS) after systemic infection is a known complication after congenital heart surgery. This can result in life-threatening hypoxemia in some critically ill infants. This complication assumes special clinical relevance after cardiac surgery because it leads to an imbalance among the available oxygen, impaired myocardial function, and increased oxygen requirements. Traditional ventilation techniques have been implicated in worsening lung injury. Prone ventilation is emerging as a promising adjuvant to lung-protective ventilatory strategies in ARDS in both the adult and pediatric populations.1 There is a paucity of literature on the use of prone ventilation in pediatric cardiac surgery. By presenting this case series, the authors want to convey the efficacy of prone ventilation in infants with severe refractory hypoxemia after systemic infection and ARDS after complex congenital heart surgery.
Case Reports
Case 1
A 1-month old, 3.8-kg child with an unobstructed coronary sinus type of total anomalous pulmonary venous connection with moderate pulmonary artery hypertension underwent corrective surgery. The child was weaned from cardiopulmonary bypass (CPB) with minimal inotropic support and half systemic pulmonary artery (PA) pressure. The sternum was kept open and stented because of poor lung compliance with elevated airway pressures (peak pressure during volume control ventilation of 30 cmH2O) and hypercarbia (PaCO2 = 55 mmHg). In the postoperative unit, the child was ventilated in the pressure-regulated volume control (PRVC) mode. The maintenance fluid was restricted to 500 to 1,500 mL/m2/d. PA pressures always remained subsystemic in the postoperative period (through percutaneous PA catheters placed at the time of surgery). The sternum was closed on the 1st postoperative day (POD). However, an attempt at weaning on POD 2 failed as the child became tachypneic on pressure-support ventilation. The development of lung infiltrates and falling white cell counts on POD 3 suggested the onset of systemic infection. Carbepenem (meropenem) was started based on the culture and sensitivity reports from the tracheal aspirates, which grew Pseudomonas aeruginosa. The inotropes were weaned off on POD 3 although the child continued to be on a ventilator. On POD 6, there was significant clinical deterioration with extensive bilateral lung infiltrates and hypoxemia suggestive of ARDS (Fig 1). Bedside echocardiography (ECHO) ruled out pulmonary venous obstruction. In view of the increasing stiffness of the lungs, a pressure-controlled mode of ventilation was instituted. Lung recruitment maneuvers (a sustained positive end-expiratory pressure of 20 cmH2O for 30 seconds) were performed intermittently to minimize atelectasis. Methylprednisolone was added to the treatment on POD 7.
Fig 1.
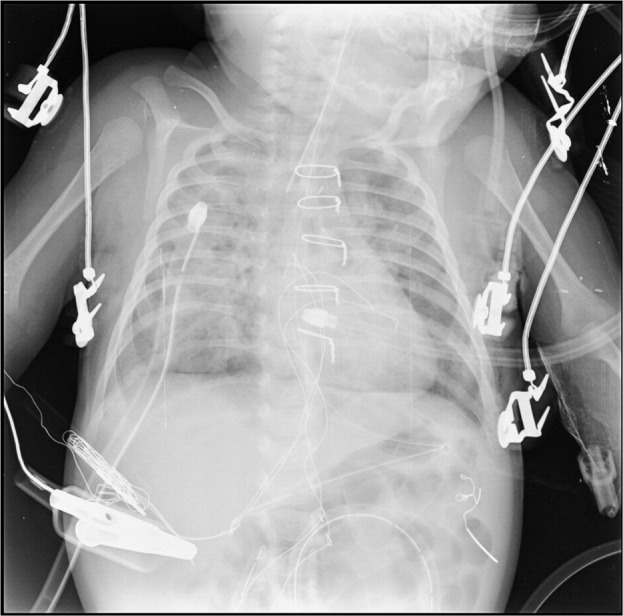
Bilateral lung infiltrates of ARDS on a chest x-ray in case 1.
Over the next few days, the oxygenation status of the child progressively deteriorated (pH = 7.41, PaO2 = 64.6 mmHg, PaCO2 = 44.6 mmHg, FIO2 = 1.0, and PaO2/FIO2 <100 on POD 12). The lack of clinical response to conventional ventilator strategies and worsening lung infiltrates with refractory hypoxemia prompted the use of prone ventilation. The pressure-controlled mode of ventilation was used during this period. The prone position was maintained for an average of 12 to 16 hours every day. Meticulous attention was paid to avoid pressure sores and eye injury during proning. Arterial blood gases were obtained within an hour of proning and every 4 hours thereafter. Analgesia and sedation were provided by continuous infusions of fentanyl and midazolam. Muscle relaxant was used only when the child was excessively agitated despite adequate sedation and analgesia. The patient was returned to the supine position during the night. Only minimal disconnections from the ventilator were permitted for intermittent tracheal suctioning. A lung recruitment maneuver was performed whenever there was a disconnection from the ventilator. Continuous enteral feeding was started to meet caloric requirements, but the volume of food was reduced by half in the prone position to minimize the risk of aspiration.
With the institution of the prone position, there was an immediate and sustained improvement in oxygenation and ventilation (Fig 2, Fig 3). This improvement was sustained even while the patient was in the supine position. The FIO2 levels could be reduced within 12 hours to an acceptable range. There was a slow but steady improvement in the chest x-ray over a period of 4 days. On the 18th POD, with sustained improvements in the oxygenation parameters and resolution of lung infiltrates, the child was shifted back to the PRVC mode in the supine position (Fig 4). Weaning was performed by progressive reduction of the ventilator rate and a spontaneous breathing trial on pressure-support ventilation. The child could be extubated successfully to nasal continuous positive airway pressure (CPAP) on POD 19. The child was further weaned from nasal CPAP to room air on POD 21. The child was discharged from the hospital on POD 28.
Fig 2.
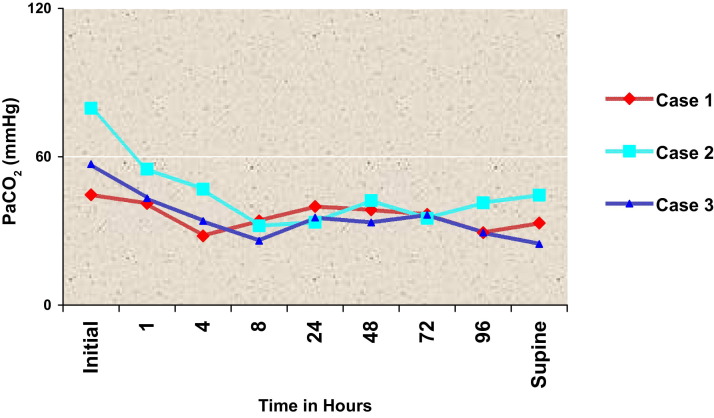
The trend in changes of PaO2/FIO2 before and during prone ventilation. Supine indicates PaO2/FIO2 ratio 6 hours after the termination of prone ventilation. (Color version of figure is available online.)
Fig 3.
The trend in changes of PaCO2 before and during prone ventilation. Supine indicates PaCO2 values 6 hours after the termination of prone ventilation. (Color version of figure is available online.)
Fig 4.

Clearance of lung infiltrates after extubation in case 1.
Case 2
A 1-month-old male child weighing 3.1 kg, with a preoperative diagnosis of coronary sinus type of total anomalous pulmonary venous connection and moderate pulmonary artery hypertension, underwent corrective cardiac surgery at the authors' center. The intraoperative period was uneventful, and the child was weaned from bypass with less than half systemic PA pressures and minimal inotropic support. The child was extubated on POD 2. However, the infant developed a pulmonary artery hypertensive crisis in the postextubation period with tachypnea and desaturation requiring reintubation. Pulmonary artery pressures stabilized with mechanical ventilation and pulmonary vasodilators (phenoxybenzamine and sildenafil). The child was extubated again on POD 5. Antibiotics (piperacillin/tazobactam) were initiated for the elevated white cell counts. On POD 10, the infant developed clinical and laboratory evidence of systemic infection and was reintubated. A chest x-ray revealed bilateral patchy opacities in the right lower lobe and the left upper lobe. The antibiotic was changed to meropenem based on the bronchoalveolar lavage and the blood culture, which grew Escherichia coli and Klebsiella pneumoniae, respectively. Bedside ECHO assessment ruled out pulmonary venous obstruction. Pressure-controlled ventilation with an intermittent lung recruitment maneuver did not bring about any satisfactory clinical improvement. Steroid therapy was initiated as an adjuvant therapy (POD 12). On POD 16, arterial blood gases deteriorated with hypercarbia and hypoxemia (pH = 7.24, PaO2 = 73.9 mmHg, PaCO2 = 71.8 mmHg, FIO2 = 1, and PaO2/FIO2 < 100). Prone ventilation was instituted on POD 16. Arterial blood gases showed consistent improvements in PaO2 and PaCO2 after placement in the prone position (Fig 2, Fig 3). The resolution of lung opacities occurred after ventilation for 6 days in the prone position. The child could be extubated successfully to nasal CPAP on the 25th POD and transitioned to room air on the 28th POD. He was discharged on the 37th POD.
Case 3
A 2-month-old, 4.75-kg male child diagnosed with severe postsubclavian coarctation of the aorta, large patent ductus arteriosus, and severe branch pulmonary artery stenosis presented for elective surgery. The child underwent coarctation repair, pulmonary artery patch plasty, and division of the ductus on CPB. He was separated from CPB with inotropic support of dobutamine at 5 μg/kg/min. Postoperatively, the child was ventilated in the PRVC mode. Weaning was not successful on POD 3 because of respiratory distress. Although the child was extubated to nasal CPAP on POD 5, he developed tachypnea with labored breathing and was reintubated immediately. ECHO revealed normal biventricular function and no residual gradient across the anastomotic sites. Bronchoscopy revealed normal airway anatomy. A chest x-ray was normal except for an elevated left hemidiaphragm. Repeated attempts at weaning failed. Bedside ECHO on the spontaneous breathing mode (without pressure support) revealed an akinetic left diaphragm. Diaphragmatic plication was performed to facilitate extubation. On POD 14, the child was extubated to nasal CPAP. Attempts to remove the noninvasive ventilatory support after 48 hours resulted in respiratory distress (POD 16). A chest radiograph showed patchy opacity in the left lower lobe. An empiric antibiotic (piperacillin/tazobactam) was started as the white cell counts dropped. Repeated attempts at weaning over the next few days continued to fail. Labored breathing and desaturation necessitated reintubation on POD 22. A chest radiograph showed bilateral hazy lung fields. There was severe hypoxemia requiring institution of high FIO2 levels. Prone ventilation was initiated as in the previous 2 cases. Prone ventilation was continued for 5 days until the resolution of lung opacities and oxygenation parameters were satisfactory (Fig 2, Fig 3). On the 35th POD, the child was extubated to nasal CPAP. However, on the 37th POD, the child developed respiratory distress with hemodynamic instability and was again reintubated. Inotropes and vasopressors were started to maintain hemodynamic stability. Fresh blood cultures grew Pseudomonas aeruginosa, which necessitated antibiotic therapy with meropenem. Despite the antibiotic therapy and inotropic support, the child developed severe septic shock and renal failure requiring peritoneal dialysis. Subsequent resuscitation measures failed, and the child died on the 40th POD.
Discussion
Acute lung injury/ARDS is a potential complication after congenital heart surgery with CPB.2 Causes of lung injury after cardiac surgery include the effects of sternotomy, systemic inflammatory response syndrome, increased extravascular lung water, collapse and atelectasis of the lung during CPB, and poor clearance of secretions in the postoperative period. Systemic infection is a well-recognized trigger for ARDS in children.3
Conventional mechanical ventilation with high tidal volumes and high inflation pressures may precipitate ventilator-induced lung injury by inducing shear stress between compliant and noncompliant regions of the lung in patients with ARDS. The ARDS network study has shown that a lung-protective ventilation strategy using low tidal volumes and limitation of the plateau pressures is most effective in patients with ARDS.4 Prone ventilation increasingly is being used as an alternative technique in critically ill patients with ARDS who do not respond to lung protective ventilation.5
Prone ventilation offers several physiologic benefits in patients with ARDS. The prone position causes a more uniform distribution of gravitational pleural pressure gradients.6 Dorsal regions of the lung, normally compressed by the heart and diaphragm, are allowed to expand more freely in prone position. The gravitational distribution of perfusion also is more homogenous in the prone position. This will minimize the ventilation–perfusion mismatching that typically occurs in ARDS and improve gas exchange. The reduction in gravitational pleural pressure gradients also prevents overdistention of alveolar units, thus protecting the lungs from ventilator-induced lung injury.
Prone ventilation can facilitate a consistent increase in oxygenation in 70% to 80% of adult patients when instituted early in the course of ARDS.6 In a meta-analysis of 10 studies of adult patients, Sud et al7 showed that prone ventilation improved oxygenation by 27% to 39% in the first 3 days of therapy. Studies in the pediatric population showed prone ventilation to be associated with improved oxygenation when instituted in the early phase of ARDS.8, 9 The PaCO2 response has not been projected in most of the studies. However, Gattinoni et al10 in a multicenter trial of 225 adult patients with ARDS showed a significant decrease of PaCO2 after prone positioning along with a concomitant survival benefit. This PaCO2 response has been attributed to an improved efficiency of alveolar ventilation with a decrease in physiologic deadspace ventilation. Although the optimal duration of proning to achieve the maximal benefits in gas exchange has not been clearly defined, current evidence suggests a longer duration of prone ventilation (20 hours/d) for a more sustained improvement in oxygenation.11, 12
Early studies failed to show a significant survival benefit for prone ventilation despite consistent improvement in gas exchange.11, 12, 13, 14 However, 2 randomized controlled trials in critically ill adult patients showed an improvement in mortality in a subgroup of patients with higher acute physiology scores.13, 15 A recent meta-analysis in adults showed a significant reduction in all-cause mortality among patients with very severe hypoxemia (PaO2/FIO2 < 100 mmHg).7 Thus, it may be more relevant to use prone ventilation in patients with a high risk of death from profound hypoxemia.
Prone positioning has not been accepted as a routine maneuver in several centers because it has been linked with potentially dangerous complications including endotracheal tube obstruction, pressure ulcers, and the dislodgement of intercostal drains and vascular access devices.7 Mancebo et al15 reported a low rate of complications after prone ventilation and concluded that prone positioning is both feasible and safe in adult patients. Proning is an attractive option in children because of the relatively small size of the patient. Fineman et al,16 in the secondary analysis of a multicenter randomized controlled trial, strongly recommended its safe use in critically ill infants and children. Their study highlighted that a comprehensive, evidence-based, and protocolized approach to prone positioning can minimize the incidence of adverse events. The fact that nutrition and comfort management goals also can be met in the prone position has further promoted the acceptance of proning as a therapeutic maneuver among pediatric critical care physicians.
Prone positioning is used less frequently in adult cardiac surgical patients. A major cause of reluctance has been the presence of sternotomy and issues related to wound infection. In the study by Maillet et al17 in adult cardiac surgical patients, there was a low incidence of sternal wound infections that did not influence the outcomes. The 2nd major issue related to proning may be the risk of hemodynamic instability after cardiac surgery. Brussel et al18 have shown that prone positioning can be accomplished without significant alteration in the hemodynamic state of adult cardiac surgical patients who develop acute respiratory failure in the postoperative period.
To the authors' knowledge, there are no data on prone ventilation in children after congenital heart surgery. The probable areas of concern in the pediatric population could be the presence of complex repairs, postoperative low-cardiac-output state, higher inotropic requirements, and the presence of multiple invasive catheters including intracardiac catheters. Cardiopulmonary interactions are likely to be more profound, and the likelihood of myocardial depression is high in this vulnerable group. ARDS with severe hypoxemia may necessitate the implementation of nonconventional strategies like high-frequency ventilation, inhaled nitric oxide, or the institution of extracorporeal membrane oxygenation. This may not be economically viable in a limited resources environment. The authors have resorted to prone ventilation as a rescue alternative in a similar scenario in a postoperative pediatric cardiac intensive care unit.
In the present case series, prone ventilation was initiated when pulmonary infiltrates were present in all 4 lung zones along with severe hypoxemia unresponsive to lung protective ventilation or recruitment strategies. A protocol-based prone positioning approach was used (Appendix 1). Nurses who were trained in the care of neonates and familiar with the protocol primarily were involved in the intensive care. The authors used the pressure-controlled mode of ventilation during this period and prone position was maintained for an average of 16 hours every day. The increase in PaO2 and the reduction in PaCO2 signified an improvement in ventilation-perfusion in the lungs (Fig 2, Fig 3). The authors were able to reduce the FIO2 to clinically acceptable levels. Moreover, the improvement in oxygenation and ventilation was extended to the period of supine ventilation. Invasive monitoring and a negative fluid balance were strictly followed during this period. There were no instances of hemodynamic instability in the 3 patients. The decision to stop prone ventilation was based on the disappearance of pulmonary infiltrates with a consistent improvement in oxygenation indices for at least 24 hours (PaO2 > 90 mmHg with FIO2 < 0.5 and PaO2/FIO2 > 200). Two patients developed facial edema, which resolved spontaneously after resuming the supine position. One patient developed an ulcer at the angle of the mouth because of pressure from the endotracheal tube. There were no episodes of endotracheal tube obstruction, dislodgement of intercostal drains, or intravascular catheters. There was no incidence of sternal wound infection. Most importantly, adopting a prone ventilation strategy might have averted probable mortality in 2 of the patients with life-threatening hypoxemia.
The use of steroids in ARDS is associated with reductionx in ventilation time, intensive care unit stay, lung injury score, and multiorgan dysfunction scores.19, 20 Low-dose steroids are not associated with increased infection rates.20 However, the authors cannot comment on the fact that the 2nd bout of sepsis in 1 of the 3 children was triggered by the steroid therapy.
The authors conclude that with ARDS complicating the postoperative course in neonates undergoing complex congenital heart repair, prone ventilation can be an attractive alternative when severe hypoxemia does not respond to conventional ventilatory management. Prone ventilation need not be adopted as a routine strategy for patients with ARDS. However, in a sicker subset of patients with refractory hypoxemia, this should be considered as a viable option. The use of a protocolized approach and trained personnel definitely can lower the risk of complications. Cardiac intensive care physicians should take it as a challenge to use prone ventilation in appropriate patients to obtain better postoperative outcomes.
Appendix 1. Prone Ventilation Protocol
Preparing the patient:
-
•
Ensure proper fixation and stability of the endotracheal tube, nasogastric tube, and monitoring catheters.
-
•
Proper eye care with pressure padding should be used to prevent injury.
-
•
Appropriate-sized bolsters (gel pillow) should be kept ready for support of the chest and pelvis and water cushions used to guard pressure points.
-
•
The intercostal drains, if present, should be clamped before proning to prevent air entering into the pleural cavity.
-
•
All clinicians should be aware of individual responsibilities and sequence of proning.
Placing the patient into the prone position:
-
•
A turning coordinator (pediatric cardiac intensivist) should coordinate the sequence of proning.
-
•
Infants and toddlers should be lifted off above the bed, turned 45°, then turned prone, and placed on the appropriate bolsters and cushions after the turn. The head should be kept in alignment with the body to avoid hyperextension. Head pillows should be positioned to prevent extreme lateral head rotation.
-
•
After positioning, the abdomen should be unrestrained.
-
•
Pressure points over forehead, knees, elbows, scrotum, and so on should be supported on water cushions.
-
•
The endotracheal tube need not be disconnected during the turn. If disconnected, perform a recruitment maneuver. After proning, reassess the security and patency of the endotracheal tube.
-
•
Check the security of monitoring catheters and intercostal drains.
Management in the prone position:
-
•
Maintain prone position for 12 to 16 hours/d.
-
•
Obtain an arterial blood gas 1 hour after proning and thereafter every 4 hours.
-
•
Pain and sedation management should be guided by age-appropriate pain and sedation assessment tools. The continuous infusion of narcotics is mandatory.
-
•
A restricted fluid protocol should be used.
-
•
Enteral feeding can be continued in the prone position. The volume of feeds should be restricted to half the volume to minimize the risk of aspiration.
-
•
If the patient deteriorates clinically in the prone position, consider restoring the supine position.
Placing the patient in supine position:
-
•
Follow the same sequence for turning to prone position.
-
•
Recheck stability and patency of endotracheal tube and vascular catheters.
-
•
Thorough skin assessment should be performed to detect pressure sores or ulcers.
-
•
Obtain a chest x-ray to confirm endotracheal tube position and to assess progress of lung pathology.
-
•
Obtain blood gas 1 hour after turning supine and thereafter every 4 hours.
-
•
Feed volumes can be restored to full volume.
References
- 1.Fessler H.E., Talmor D.S. Should prone positioning be routinely used for lung protection during mechanical ventilation? Respir Care. 2010;55:88–99. [PubMed] [Google Scholar]
- 2.Cui Q., Zhou H., Zhao R., et al. The effects of open lung ventilation on respiratory mechanics and hemodynamics in atelectatic infants after cardiopulmonary bypass. J Int Med Res. 2009;37:113–120. doi: 10.1177/147323000903700113. [DOI] [PubMed] [Google Scholar]
- 3.Randolph A.G. Management of acute lung injury and acute respiratory distress syndrome in children. Crit Care Med. 2009;37:2447–2454. doi: 10.1097/CCM.0b013e3181aee5dd. [DOI] [PubMed] [Google Scholar]
- 4.The Acute Respiratory Distress Syndrome Network Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301–1308. doi: 10.1056/NEJM200005043421801. [DOI] [PubMed] [Google Scholar]
- 5.Dellinger R.P., Levy M.M., Carlet J.M., et al. Surviving sepsis campaign: International guidelines for management of severe sepsis and septic shock; 2008. Crit Care Med. 2008;36:296–327. doi: 10.1097/01.CCM.0000298158.12101.41. [DOI] [PubMed] [Google Scholar]
- 6.Pelosi P., Brazzi L., Gattinoni L. Prone position in acute respiratory distress syndrome. Eur Respir J. 2002;20:1017–1028. doi: 10.1183/09031936.02.00401702. [DOI] [PubMed] [Google Scholar]
- 7.Sud S., Friedrich J.O., Taccone P., et al. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: Systematic review and meta-analysis. Intensive Care Med. 2010;36:585–599. doi: 10.1007/s00134-009-1748-1. [DOI] [PubMed] [Google Scholar]
- 8.Curley M.A., Thompson J.E., Arnold J.H. The effects of early and repeated prone positioning in pediatric patients with acute lung injury. Chest. 2000;118:156–163. doi: 10.1378/chest.118.1.156. [DOI] [PubMed] [Google Scholar]
- 9.Kornecki A., Frndova H., Coates A.L., et al. A randomized trial of prolonged prone positioning in children with acute respiratory failure. Chest. 2001;119:211–218. doi: 10.1378/chest.119.1.211. [DOI] [PubMed] [Google Scholar]
- 10.Gattinoni L., Vagginelli F., Carlesso E., et al. Decrease in PaCO2 with prone position is predictive of improved outcome in acute respiratory distress syndrome. Crit Care Med. 2003;31:2727–2733. doi: 10.1097/01.CCM.0000098032.34052.F9. [DOI] [PubMed] [Google Scholar]
- 11.Curley M.A., Hibberd P.L., Fineman L.D., et al. Effect of prone positioning on clinical outcomes in children with acute lung injury: A randomized controlled trial. JAMA. 2005;294:229–237. doi: 10.1001/jama.294.2.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Taccone P., Pesenti A., Latini R., et al. Prone positioning in patients with moderate and severe acute respiratory distress syndrome: A randomized controlled trial. JAMA. 2009;302:1977–1984. doi: 10.1001/jama.2009.1614. [DOI] [PubMed] [Google Scholar]
- 13.Gattinoni L., Tognoni G., Pesenti A., et al. Effect of prone positioning on the survival of patients with acute respiratory failure. N Engl J Med. 2001;345:568–573. doi: 10.1056/NEJMoa010043. [DOI] [PubMed] [Google Scholar]
- 14.Guerin C., Gaillard S., Lemasson S., et al. Effects of systematic prone positioning in hypoxemic acute respiratory failure: A randomized controlled trial. JAMA. 2004;292:2379–2387. doi: 10.1001/jama.292.19.2379. [DOI] [PubMed] [Google Scholar]
- 15.Mancebo J., Fernandez R., Blanch L., et al. A multicenter trial of prolonged prone ventilation in severe acute respiratory distress syndrome. Am J Respir Crit Care Med. 2006;173:1233–1239. doi: 10.1164/rccm.200503-353OC. [DOI] [PubMed] [Google Scholar]
- 16.Fineman L.D., LaBrecque M.A., Shih M.C., et al. Prone positioning can be safely performed in critically ill infants and children. Pediatr Crit Care Med. 2006;7:413–422. doi: 10.1097/01.PCC.0000235263.86365.B3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Maillet J.M., Thierry S., Brodaty D. Prone positioning and acute respiratory distress syndrome after cardiac surgery: A feasibility study. J Cardiothorac Vasc Anesth. 2008;22:414–417. doi: 10.1053/j.jvca.2007.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brussel T., Hachenberg T., Roos N., et al. Mechanical ventilation in the prone position for acute respiratory failure after cardiac surgery. J Cardiothorac Vasc Anesth. 1993;7:541–546. doi: 10.1016/1053-0770(93)90311-8. [DOI] [PubMed] [Google Scholar]
- 19.Foster J.R. Steroids for early acute respiratory distress syndrome: Critical appraisal of Meduri GU, Golden E, Freire AX, et al: Methylprednisolone infusion in early severe ARDS: Results of a randomized controlled trial. Chest. 2010;131:954–963. doi: 10.1097/PCC.0b013e3181c014eb. Pediatr Crit Care Med 11:404-407. [DOI] [PubMed] [Google Scholar]
- 20.Tang B.M., Craig J.C., Eslick G.D., et al. Use of corticosteroids in acute lung injury and acute respiratory distress syndrome: A systematic review and meta-analysis. Crit Care Med. 2009;37:1594–1603. doi: 10.1097/CCM.0b013e31819fb507. [DOI] [PubMed] [Google Scholar]