Introduction
The T-helper-2 cytokines interleukin 4 (IL-4) and IL-13 play a key role in the pathogenesis of allergic disorders. Dupilumab is a fully human monoclonal antibody against the IL-4 alpha chain receptor, mitigating the T-helper-2 response.1 In June 2022 it became the first Food and Drug Administration approved biologic for the treatment of moderate-to-severe atopic dermatitis in children 6 months to 5 years old. Cutaneous adverse reactions associated with dupilumab are increasingly reported, with facial rash being the most common.2 Psoriasiform eruptions have also been reported in patients being treated with dupilumab.3 We present a case of a pediatric patient who developed a pityriasis rosea like-reaction after initiation of dupilumab therapy, which resolved with cessation of the medicine.
Case report
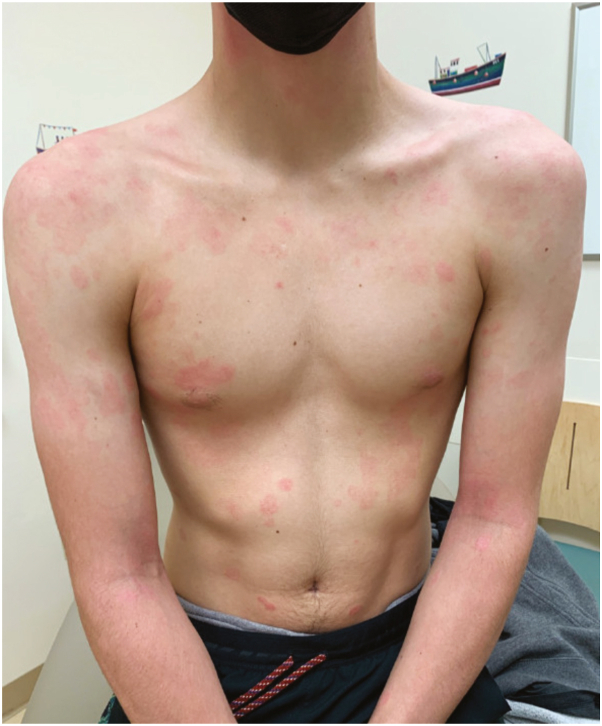
A 16-year-old boy with a history of atopic dermatitis presented to the dermatology clinic for evaluation of a pruritic rash located primarily on face, trunk, and extremities. Rash onset was 6 days prior to presentation, shortly after initiation of dupilumab 300 mg subcutaneously every 2 weeks by an outside dermatologist. On exam, there were erythematous, oval-to-round wrinkly plaques with peripheral collarette of scale and central clearance on the trunk and extremities (Fig 1) accompanied by macular erythema of the face. There was sparing of mucous membranes, palms, and soles. Although the KOH was negative, the decision was made to treat empirically for Malassezia associated dermatitis, as this can occur in the setting of dupilumab. Patient was prescribed a combination of itraconazole 200 mg once daily for 2 weeks, and ketoconazole 2% shampoo once daily. Dupilumab was continued.
Fig 1.
Clinical presentation. Multiple oval-to-round erythematous plaques with peripheral collarette of scale and central clearance.
Upon follow-up 3 weeks later, the rash had worsened despite anti-fungal therapy, and a biopsy was performed. Histopathologic examination of a punch biopsy specimen from the left flank revealed parakeratosis, mild acanthosis, spongiosis, and perivascular lymphohistiocytic inflammation with scattered eosinophils, consistent with a pityriasiform dermatitis (Fig 2). GMS stain was negative for fungal organisms. A diagnosis of pityriasis rosea-like drug eruption to dupilumab was made based on clinical and histopathologic findings. Unfortunately, the patient experienced no treatment response to triamcinolone 0.1%, tacrolimus 0.03%, or mometasone 0.1% creams. Phototherapy was not available near patient’s home. Ultimately, dupilumab was discontinued with resultant clearance of the rash within 2 weeks. The patient reported the rash was more bothersome than his baseline atopic dermatitis and was satisfied with the outcome after stopping dupilumab.
Fig 2.
Punch biopsy from the left flank. Mild acanthosis and spongiosis with mounded parakeratosis and perivascular lymphohistiocytic inflammation with scattered eosinophils (hematoxylin and eosin, 100×, inset highlighting eosinophils).
Discussion
Since 2017, dupilumab has been an effective and globally recognized Food and Drug Administration approved biologic drug that addresses the underlying T-helper-2 pathway in allergic diseases.4 Overall, dupilumab is a safe and well-tolerated drug. Adverse cutaneous reactions do not always necessitate discontinuation of the drug. Injection site reactions, facial erythema, and psoriasiform reactions are the most common cutaneous reactions to dupilumab. Some have theorized psoriasiform lesions as a marker for effective treatment with dupilumab, reflecting a potential shift toward cutaneous IL-23 and T-helper-17 pathway activation with suppression of type 2 immunity.5 In contrast to the treatment response in our case of a pityriasis rosea-like drug eruption, most cases of psoriasiform eruptions in the setting of dupilumab achieve clearance with use of topical medium-strength to potent steroids. In contrast drug induced or idiopathic pityriasis rosea tends to be refractory to treatment and as it is mediated by a different immunologic mechanism in response to viral triggers. Other rarer cutaneous reactions that have been described with dupilumab include erythema nodosum and urticaria.6,7
To our knowledge, this is the first case of pityriasis rosea-like drug eruption induced by dupilumab. Pityriasis rosea-like drug eruptions mimic pityriasis rosea, a common idiopathic and typically self-limited cutaneous eruption. Pityriasis rosea initially presents as a solitary scaly, pink, or flesh-colored plaque, the “herald patch”, followed by an eruption of scaling patches on the trunk and upper extremities. It is thought to be associated with systemic reaction to human herpesvirus 6 and 7. In comparison, a pityriasis rosea-like drug eruption may be more violaceous and redder in color and may lack a herald patch. Pityriasis rosea-like drug eruptions have been reported with non-steroidal anti-inflammatory drugs and angiotensin converting enzyme inhibitors, among many other drugs.8 In these cases, the mechanism underlying the eruption is unclear and resolution was achieved with drug withdrawal. Duration greater than 3 months and biopsy showing increased eosinophilic infiltrate can help distinguish a pityriasis rosea-like drug eruption from pityriasis rosea.
Other biologic agents reported to cause pityriasis rosea-like drug eruption include rituximab and imatinib.9,10 In both cases, the drug was either discontinued or the treatment course was already completed by the time of diagnosis, and the rash resolved within 2 weeks.
Although biologic agents have increased selectivity, new rare adverse reactions can occur. We share this case as an example of a cutaneous reaction to dupilumab that may be under-recognized due to lack of representation in the literature. A pityriasis rosea-like drug eruption should be considered in a patient on dupilumab with erythematous scaling plaques on the trunk and extremities.
Conflicts of interest
None disclosed.
Footnotes
Funding sources: None.
IRB approval status: Not applicable.
Patient consent: Consent for the publication of all patient photographs and medical information was provided by the authors at the time of article submission to the journal stating that all patients gave consent for their photographs and medical information to be published in print and online and with the understanding that this information may be publicly available.
References
- 1.Harb H., Chatila T.A. Mechanisms of dupilumab. Clin Exp Allergy. 2020;50(1):5–14. doi: 10.1111/cea.13491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Muzumdar S., Skudalski L., Sharp K., Waldman R.A. Dupilumab facial redness/dupilumab facial dermatitis: a guide for clinicians. Am J Clin Dermatol. 2022;23(1):61–67. doi: 10.1007/s40257-021-00646-z. [DOI] [PubMed] [Google Scholar]
- 3.Napolitano M., Scalvenzi M., Fabbrocini G., Cinelli E., Patruno C. Occurrence of psoriasiform eruption during dupilumab therapy for adult atopic dermatitis: a case series. Dermatol Ther. 2019;32 doi: 10.1111/dth.13142. [DOI] [PubMed] [Google Scholar]
- 4.Shirley M. Dupilumab: first global approval. Drugs. 2017;77(10):1115–1121. doi: 10.1007/s40265-017-0768-3. [DOI] [PubMed] [Google Scholar]
- 5.Parker J.J., Sugarman J.L., Silverberg N.B., et al. Psoriasiform dermatitis during dupilumab treatment for moderate-to-severe atopic dermatitis in children. Pediatr Dermatol. 2021;38(6):1500–1505. doi: 10.1111/pde.14820. [DOI] [PubMed] [Google Scholar]
- 6.Mustin D.E., Cole E.F., Blalock T.W., Kalangara M.E., Stoff B.K., Feldman R.J. Dupilumab-induced erythema nodosum. JAAD Case Rep. 2021;19:41–43. doi: 10.1016/j.jdcr.2021.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mastorino L., Ortoncelli M., Virginia B., et al. Dupilumab-induced urticaria. Dermatol Ther. 2021;34(6) doi: 10.1111/dth.15117. [DOI] [PubMed] [Google Scholar]
- 8.Atzori L., Pinna A.L., Ferreli C., Aste N. Pityriasis rosea-like adverse reaction: review of the literature and experience of an Italian drug-surveillance center. Dermatol Online J. 2006;12(1):1. [PubMed] [Google Scholar]
- 9.Sezer E., Erkek E., Cetin E., Sahin S. Pityriasis rosea-like drug eruption related to rituximab treatment. J Dermatol. 2013;40(6):495–496. doi: 10.1111/1346-8138.12103. [DOI] [PubMed] [Google Scholar]
- 10.Brazzelli V., Prestinari F., Roveda E., et al. Pityriasis rosea-like eruption during treatment with imatinib mesylate: description of 3 cases. J Am Acad Dermatol. 2005;53(5 suppl 1):S240–S243. doi: 10.1016/j.jaad.2004.10.888. [DOI] [PubMed] [Google Scholar]