Abstract
Context:
Knee muscle atrophy and weakness are common impairments after anterior cruciate ligament (ACL) reconstruction. Blood flow restriction (BFR) training represents a new approach to treat such impairments. However, limited evidence currently exists to support this intervention in related patients.
Objective:
To appraise literature comparing the effects of BFR training with conventional therapy on knee muscle morphological and strength properties in ACL-reconstructed patients.
Data Sources:
PubMed, SPORTDiscus, CINAHL, and Cochrane Central Register databases were searched for relevant articles from January 1991 through April 2021.
Study Selection:
Articles were minimum Level 3 evidence focusing on knee muscle morphologic as well as extensor and flexor strength outcomes in ACL-reconstructed patients of all graft types.
Study Design:
Systematic review.
Level of Evidence:
Level 2.
Data Extraction:
Critical appraisal instruments (Downs and Black checklist, Cochrane Collaboration tool, ROBINS-1 tool) were used to evaluate study quality. We independently calculated effect sizes (ESs) (Cohen d) between groups in each study. The Strength of Recommendation Taxonomy grading scale was used for clinical recommendations.
Results:
Six articles (4 randomized control studies, 1 nonrandomized study, and 1 case-control study) met inclusion criteria. Exercises paired with BFR training included open kinetic chain, closed kinetic chain, and passive applications. Diverse assessments and time of intervention were observed across studies. ESs ranged from trivial to large in favor of BFR training for muscle morphological (d = 0.06 to 0.81) and strength assessments (d = −0.12 to 1.24) with CIs spanning zero.
Conclusion:
At this time, grade B or inconsistent and limited-quality patient-oriented evidence exists to support using BFR training to improve or maintain thigh muscle size as well as knee extensor and flexor strength in ACL-reconstructed patients. ESs indicated no consistent clinically meaningful differences when compared with conventional therapy. Subsequent analyses should be repeated as new evidence emerges to update practice guidelines.
Keywords: ACL reconstruction, blood flow restriction training, rehabilitation
Rehabilitation protocols after ACL reconstruction focus on restoring patient function using the limb symmetry index.1,34 Thigh muscle atrophy22,31,41 as well as residual quadriceps and hamstring weakness22,23,40,42 are commonly observed after ACL reconstruction. These specific deficits may persist greater than 2 years postreconstruction7,22,24 and negatively influence recurrent ACL injury risk, 9 kinesiophobia, 32 and return to physical activity,2,3 which affect long-term health.
Current clinical recommendations indicate moderate-to-high resistance training (~60%-80% 1 repetition maximum [1RM]) be prescribed to address muscular strength asymmetries. 16 However, loads of this intensity are initially contraindicated after surgery and may be too strenuous at the end of the rehabilitation protocol because of joint pain or kinesiophobia.5,15 An alternative mode that addresses these limitations is blood flow restriction (BFR) training. BFR training is a rehabilitative technique where vascular occlusion of the limb is externally applied as the patient completes prescribed exercise.10,18 Contemporary BFR training utilizes approximately 20% to 40% of the patient’s 1RM to produce relatively equal benefits as traditional high-intensity training. 36
Arve et al 4 examined BFR training effects on quadriceps size and strength after knee surgery, including ACL reconstruction. Their appraisal determined that inconsistent evidence exists to support BFR training for treating associated patient populations. 4 Similarly, Barber-Westin and Noyes 6 analyzed literature investigating the effects of BFR training on muscle atrophy as well as knee extensor and flexor weakness across a multitude of patients, including those having undergone ACL reconstruction. They noted that many articles reported positive outcomes. However, some studies included varying knee pathologies (eg, osteoarthritis), which clouded the specific effect on ACL-reconstructed patients. Since these publications, new related research has been disseminated. Thus, the purpose of this systematic review was to compare the effectiveness of BFR training with conventional treatment to improve quadriceps and hamstring muscle size as well as strength in patients undergoing rehabilitation after ACL reconstruction. An updated analysis is warranted to produce contemporary evidence that guides utility of this therapeutic intervention and identifies areas for future study.
Methods
Inclusion and Exclusion Criteria
Articles representing level 3 or higher evidence published in English over the past 30 years were selected if they measured thigh muscle morphological properties (ie, volume, thickness, or anatomic cross-sectional area [ACSA]) and knee extensor or flexor strength before and after BFR training in ACL-reconstructed patients. Patients of all ages and those with a history of concomitant injury at the time of ACL reconstruction were included.
Articles were excluded if they only reported preoperative use of BFR training and applied the intervention solely to healthy participants. Other instances of exclusion entailed a lack of analyzing thigh muscle size or strength and articles classified as case studies. Duplicate studies, prior systematic reviews, and meta-analysis as well as works published in languages other than English were also excluded.
Search Strategy
We followed the 2009 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 30 A literature search was conducted with the date range of January 1991 through April 2021 using PubMed, SPORTDiscus, CINAHL, and Cochrane Central Register of Controlled Trials databases. Search terms included the following key phrases: (anterior cruciate ligament injury OR ACL injury OR anterior cruciate ligament tear OR ACL tear OR ACL reconstruction) AND (blood flow restriction training OR blood flow restriction exercise OR blood flow restriction therapy OR BFR therapy OR KAATSU training OR occlusion resistance training OR vascular occlusion).
Study Selection and Data Extraction
Studies identified from the eligibility criteria were independently analyzed by 2 authors (MC, BP). Articles were initially screened by title and abstract to determine eligibility. A thorough evaluation of each work followed to definitively determine inclusion. Any disagreements regarding inclusion of an article study were arbitrated by a third author (GV).
The following items were extracted from each article: study design, patient demographics, occlusion and exercise protocols, muscle size and/or strength assessment methods, and study dropouts. In applicable instances, primary authors were contacted to obtain unpublished information.
Study Quality Assessment
The Downs and Black checklist was implemented to score study quality. 13 Risk of bias was assessed using the Cochrane Collaboration 17 and ROBINS-1 38 tools for randomized and nonrandomized control studies, respectively. Clinical recommendations were rated per the Strength of Recommendation Taxonomy (SORT) grading scale. 14 Two authors (MC and BP) completed each appraisal tool separately. Disagreements in scoring between the authors were resolved by a third author (GV).
Post hoc analysis of data presented in surveyed articles was completed by calculating Cohen d effect sizes (ESs) and corresponding 95% CIs. ESs were interpreted as ≥0.80 = large, 0.50 to 0.79 = moderate, 0.20 to 0.49 = small, and <0.20 = trivial. 11 Positive ESs signaled a favorable benefit of BFR training and negative ESs signaled no benefit of BFR training.
Results
Figure 1 illustrates our search results.
Figure 1.
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram of literature search and appraisal. BFR, blood flow restriction.
Table 1 outlines characteristics of the articles included in this systematic review. Articles spanned those analyzing the effects of BFR training immediately after to 5 years after surgery. Five12,19,20,33,39 of the 6 studies used BFR training within 6 months of ACL reconstruction. One study 21 used it 2 years after. The majority of reconstructions utilized a hamstring tendon autograft (n = 117) followed by a bone–patellar tendon–bone autograft (n = 31) and quadriceps tendon autograft (n = 3). Table 2 details study quality. Bias assessments are reported in Appendices A and B (available in the online version of this article). Table 3 describes occlusion and exercise interventions.
Table 1.
Study characteristics, design, and group demographics for each included study
| Study | Study Design | Group Demographics a | Time Postreconstruction to Intervention a | Graft Type |
|---|---|---|---|---|
| Curran et al 12 | Randomized control trial | BFR groups: n = 18 (10 m, 8 f); age, 15.7 ± 1.3
y (Concentric with BFR and eccentric with BFR groups combined) Control groups: n = 16 (5 m, 11 f); age, 17.4 ± 3.5 y (Concentric and eccentric groups combined) |
All groups: 10 weeks |
BFR groups: BPTB graft (n = 13), STG graft (n =
3), QT graft (n = 2) Control groups: BPTB graft (n = 12), STG graft (n = 3), QT graft (n = 1) |
| Hughes et al 19 | Randomized control trial | BFR group: n = 12 (7 m, 5 f); age, 29 ± 7 y Control group: n = 12 (10 m, 2 f); age, 29 ± 7 y |
BFR group: 23 ± 2 days to postsurgery
testing/intervention Control group: 24 ± 1 days to postsurgery testing/intervention |
Hamstring tendon autograft (n = 24) |
| Iversen et al 20 | Randomized control trial | BFR group: n = 12 (7 m, 5 f); age, 24.9 ± 7.4 y Control group: n = 12 (7 m, 5 f); age, 29.8 ± 9.3 y |
Both groups: 2 days | Hamstring tendon autograft (n = 24) |
| Ohta et al 33 | Randomized control trial | BFR group: n = 22 (13 m, 9 f); age, 28 ± 9.7 y Control group: n = 22 (12 m, 10 f); age, 30 ± 9.7 y |
Both groups: 0 weeks | Semitendinosus tendon autograft (n = 44) |
| Takarada et al 39 | Nonrandomized control study | BFR group: n = 8 (4 m, 4 f); age, 22.8 ± 0.9 y (m) and 22.0 ±
0.9 y (f) Control group: n = 8 (4 m, 4 f); age, 23.3 ± 1.3 y (m), 22.8 ± 1.0 y (f) |
Both groups: 3 days | Hamstring STG autograft (n = 16) |
| Kilgas et al 21 | Case-control study (nonequivalent pre-post test control) | ACL/BFR group: n = 9 (3 m, 6 f); age, 26 ± 8 y Healthy control group: n = 9 (3 m, 6 f); age, 26 ± 6 y |
ACL/BFR group: 5 ± 2 y Healthy control group: N/A |
Patellar tendon autograft (n = 6) Hamstring tendon autograft (n = 3) |
ACL, anterior cruciate ligament; BFR, blood flow restriction; BPTB, bone–patellar tendon–bone; f, female; m, male; N/A, not applicable; QT, quadriceps tendon; STG, semitendinosus-gracilis tendon.
Values are expressed as mean ± standard deviation.
Table 2.
Downs and Black checklist for quality assessment scores a
| Study Reporting | External Validity | Internal Validity—Bias | Internal Validity—Confounding | Power | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Study | 1 | 2 | 3 | 4 | 5 b | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | 26 | 27 c | Total |
| Curran et al 12 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 23 |
| Hughes et al 19 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 23 |
| Iversen et al 20 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 18 |
| Ohta et al 33 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 16 |
| Takarada et al 39 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 12 |
| Kilgas et al 21 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 16 |
Study reporting corresponds to questions 1 to 10, external validity questions 11 to 13, internal validity—bias questions 14 to 20, internal validity—confounding questions 21 to 26, and power question 27.
Possible score of 2.
Possible score of up to 5.
Table 3.
Occlusion and exercise protocols for each included study a
| Study | Tourniquet System and Occlusion Pressure/Cuff Width | Occlusion Protocol and Duration | Exercises | Frequency, Duration, and Total treatment Sessions | Dropouts in Study |
|---|---|---|---|---|---|
| Curran et al 12 | PTS personalized tourniquet system 80% LOP 11.5 cm width |
Unilateral occlusion. Intermittent occlusion (occlusion during exercise, no occlusion during rest. 2 min between sets). Total occlusion: not reported |
BLAST! leg press system (60°-20° knee flexion arc). 5× 10
repetitions (first set 20% 1RM all groups [no BFR for first
set]) Concentric groups: 70% 1RM concentric knee extension, 20% 1RM eccentric knee extension Eccentric groups: 20% 1RM concentric knee extension, 70% 1RM eccentric knee extension 1RM assessed every week |
8 weeks Total treatment sessions: 16 Supervised rehabilitation program |
Allocation and enrollment time point: contralateral ACL tear (n
= 1) Prior to RTA testing: Did not return for follow-up (n = 2), contralateral ACL tear (n = 1) Total: n = 4 |
| Hughes et al 19 | PTS personalized tourniquet system 80% LOP 11.5 cm cuff width |
Bilateral occlusion. Applied to injured side, then noninjured limb | Unilateral leg press training. Both legs trained (injured,
followed by noninjured limb) BFR group: 4 sets (30, 15, 15, 15 repetitions). 30 s of interset rest Load: 30% predicted 1RM b (readjusted at mid-training; increased by 10% if completing all repetitions on two subsequent sessions) Control group: 3× 10. 30 s of interset rest Load: 70% predicted 1RM b Both groups: take-home rehabilitation |
2× per week for 8 weeks. Total treatment sessions: 16 Take-home rehabilitation program: 3× per week |
4 participants (unplanned additional surgery [n = 1], reasons unrelated to study [n = 3]) |
| Iversen et al 20 | Pneumatic occlusion cuff Day 1: 130 mm Hg. Increased by 10 mm Hg daily. Maximum pressure 180 mm Hg. 14 cm cuff width |
Unilateral occlusion. 5× (5 min occlusion, 3 min
rest). Total occlusion 25 min per session |
Isometric quadriceps contractions, leg extensions over knee
roll, and straight leg raises. (20 repetitions each
exercise) Upper body inclined to 45° |
Completed twice daily from 2 to 14 days. Supervised rehabilitation exercises: 16 days Total treatment sessions: 24 |
None |
| Ohta et al 33 | Pneumatic cuff 180 mm Hg Cuff width not stated |
Unilateral occlusion Week 1: no occlusion Weeks 2 to 16: with occlusion Total occlusion: not reported |
Straight leg raises and hip joint abduction (20 × 5 s)
c
Hip joint adduction with ball (weeks 1 to 12 [2 sets 20 × 5]) Half-squat exercise (20 × 6 s) c Step-up exercise (1 × 20: 25 cm box) |
Total 16 weeks Straight leg raises and hip joint abduction: weeks 1 to 8, 2× per day Hip joint adduction with ball: twice daily, 6× per week |
2 participants in BFR group (discomfort or dull pain in lower limb) |
| OKC knee flexion exercise with tubing (weeks 13 to 16 [1 × 20],
45° to 100°) Half-squat walking (60 steps) |
Half-squat exercise: weeks 5 to 16, 2× per day Step-up exercise: weeks 5 to 16, 3× per day, 6× per week OKC knee flexion exercise: weeks 9 to 16, 1 set daily/6× per week weeks 9 through 12, 2 sets daily: 6× per week Half-squat walking: weeks 13 to 16, 3× per day, 6× per week Total treatment sessions: ~96 |
||||
| Takarada et al 39 | Pneumatic cuff 180 to 260 mm Hg (average: 238 ± 8 mm Hg) 9 cm cuff width |
Unilateral occlusion. BFR group: Intermittent occlusion (5 sets of 5 min on / 3 min off) Total occlusion: 25 min total Control group: Sham cuff application for 37 min (no occlusion) |
Intermittent occlusion with no movement (upper body inclined to 45°) | Days 3 to 14 postreconstruction Daily application 2× per day Total treatment sessions: 22 Supervised rehabilitation program |
None |
| Kilgas et al 21 | Pneumatic cuff 50% participant’s LOP 18 cm cuff width |
Unilateral occlusion. Continuous occlusion during exercise and
rest (1 min), deflated between exercises (2 min) Total occlusion: 18 to 25 min per session |
3 exercises: single-leg knee extensions and bodyweight half squats (3 × 30 each) and 3 × 2 min walking | 5× per week, 4 weeks
d
Total treatment sessions: 20 |
None |
1RM, 1 repetition maximum; BFR, blood flow restriction; LOP, limb occlusion pressure; OKC, open kinetic chain; PTS, personalized tourniquet system; RTA, return to activity.
Table includes frequency and duration of the intervention and dropouts for each included study.
1RM calculated from 10RM strength testing.
Progressive increase in load.
Take-home program with 5 supervised sessions.
Morphological Measures
Table 4 details outcome measures assessed and comparisons within the articles. Each study analyzed at least 1 muscle morphologic measure.12,19-21,33,39 Knee extensors were assessed in all studies. Results varied with regard to knee extensor volume, cross-sectional area (CSA) or thickness when compared between groups. Three studies demonstrated positive BFR training effects. These include significantly reduced atrophy during the early phase of healing, 39 maintenance of limb symmetry ratios 16 weeks after surgery, 33 and improved muscle thickness symmetry after 4 weeks of training 2 or more years after ACL reconstruction. 21 In contrast, three studies found no differences in maintaining knee extensor CSA, 20 vastus lateralis muscle thickness, 19 and rectus femoris muscle volume between groups. 12 Within-group improvements in knee extensor morphologic properties were noted in 2 studies.19,21 The sole study that analyzed single muscle fiber diameter by fiber type of the vastus lateralis noted atrophy in type II fibers after a 16-week BFR training intervention in both groups. However, differences were insignificant within and between groups. 33 It should be noted that only 8 cases within each group were selected for muscle fiber analysis. 33 Knee flexor muscle morphological changes were evaluated in 2 studies via magnetic resonance imaging.33,39 No differences were identified between groups when BFR training was implemented within the first 2 to 16 weeks after surgery.33,39 Patients in both studies had their ACL reconstructed with a hamstring tendon autograft.
Table 4.
Outcome measures and statistical analysis within each included study
| Study | Morphological Properties | Knee Strength | ||
|---|---|---|---|---|
| Results a | Comparisons | Results a | Comparisons | |
| Curran et al 12 |
Preintervention to
postintervention: Rectus femoris volume via MRI (cm3) BFR group: Δ 1.13 ± 3.05 Control group: Δ 2.26 ± 3.11 Preoperation to postintervention Rectus femoris volume via MRI (cm3) BFR group: Δ −1.7 ± 2.5 Control group: Δ −2.7 ± 3.8 |
Between groups: P = 0.31 Between groups: P = 0.40 |
Preoperation to
postintervention: Isokinetic knee extension strength (N·m) BFR group: Δ −12.4 ± 19.2 Control group: Δ −15.0 ± 34.4 MVIC (N·m) BFR group: Δ −16.3 ± 31.1 Control group: Δ −11.8 ± 38.3 |
Between groups: Isokinetic knee extension strength (N·m) P = 0.49 MVIC P = 0.88 |
| Hughes et al 19 |
Muscle thickness via ultrasound (postsurgery to
postintervention) Vastus lateralis (cm) BFR group: Δ 0.11 ± 0.10 Control group: Δ 0.13 ± 0.06 |
Main effects for timeP <
0.01 Between-group differences: P = 0.33 |
Presurgery to posttraining
Unilateral leg press 10RM strength BFR group: 104% ± 18% injured limb, 33% ± 12% noninjured limb Control group: 106% ± 21% injured limb, 40% ± 16% noninjured limb Scaled isokinetic strength testing Extension (nm/kg): 60 deg/s: BFR group: Δ −0.13 ± 0.44 injured limb, Δ 0.26 ± 0.31 noninjured limb Control group: Δ −0.31 ± 0.52 injured limb, Δ 0.07 ± 0.54 noninjured limb 150 deg/s: BFR group: Δ 0.07 ± 0.27 injured limb, Δ 0.19 ± 0.24 noninjured limb Control group: Δ −0.15 ± 0.38 injured limb, Δ 0.14 ± 0.43 noninjured limb 300 deg/s: BFR group: Δ 0.03 ± 0.20 injured limb, Δ 0.11 ± 0.18 noninjured limb Control group: Δ −0.13 ± 0.23 injured limb, Δ 0.09 ± 0.30 noninjured limb Flexion (nm/kg): 60 deg/s: BFR group: Δ −0.06 ± 0.22 injured limb, Δ 0.05 ± 0.17 noninjured limb Control group: Δ −0.22 ± 0.15 injured limb, Δ 0.05 ± 0.20 noninjured limb 150 deg/s: BFR group: Δ −0.07 ± 0.14 injured limb, Δ 0.09 ± 0.16 noninjured limb Control group: Δ −0.17 ± 0.14 injured limb, Δ 0.04 ± 0.20 noninjured limb 300 deg/s: BFR group: Δ −0.03 ± 0.10 injured limb, Δ 0.09 ± 0.09 noninjured limb Control group: Δ −0.12 ± 0.18 injured limb, Δ 0.05 ± 0.16 noninjured limb |
10RM strength: Main effect for time Injured limb: P < 0.01 Noninjured limb: P < 0.01 Between-group differences: Injured limb: P = 0.22 Noninjured limb: P = 0.39 Isokinetic strength: Knee extensor 60 deg/s Between groups Injured limb: P = 0.20 Within group: P < 0.01 Knee extensor 150 deg/s and 300 deg/s: Significant decrease in control group (P < 0.01), no change in BFR group Noninjured limb: increase for all speeds for both groups: P < 0.05. Knee flexor 60 deg/s, 150 deg/s, and 300 deg/s: Between groups: significant decrease in injured limb for control group P < 0.01 Noninjured limb: increase at all speeds for both groups P < 0.05 |
| Iversen et al 20 |
ACSA of Quadriceps: 40% ACSA BFR group: 77 ± 2.5 pre, 67.7 ± 2.7 post (Δ −9.7 ± 1.0) Control group: 75.4 ± 3.2 pre, 66.1 ± 3.3 post (Δ −9.2 ± 0.8) 50% ACSA BFR group: 87 ± 3.6 pre, 73.9 ± 3.5 post (Δ −13.7 ± 0.9) Control group: 82.8 ± 3.4 pre, 71.3 ± 3.2 post (Δ −11.5 ± 0.7) |
P = 0.63 | N/A | N/A |
| Ohta et al 33 |
CSA ratios via MRI: Knee extensors BFR group: 91 ± 7 pre, 101 ± 11 post Control group: 92 ± 11 pre, 92 ± 12 post Knee flexor + adductor muscle groups BFR group: 99 ± 3 pre, 105 ± 19 post Control group: 97 ± 11 pre, 102 ± 23 post Muscle fiber diameter by fiber type ratios (preop/postop): Vastus lateralis: BFR group: Type I 103 ± 10, type II 102 ± 8 Control group: Type I 95 ± 11, type II 97 ± 7 |
Between groups
Knee extensors P = 0.04 Knee flexor + adductor muscle groups No difference (P value not expressed) Type I muscle fiber No difference (P value not expressed) Type II muscle fiber No difference (P value not expressed) |
Isokinetic torque limb ratios: Knee extensors Concentric 60° BFR group: 84 ± 13 pre, 76 ± 16 post Control group: 86 ± 14 pre, 55 ± 17 post Concentric 180° BFR group: 84 ± 14 pre, 77 ± 13 post Control group: 90 ± 9 pre, 65 ± 13 post Isometric 60° BFR group: 92 ± 19 pre, 84 ± 19 post Control group: 94 ± 21 pre, 65 ± 19 post Knee flexors Concentric 60° BFR group: 96 ± 21 pre, 81 ± 14 post Control group: 90 ± 16 pre, 72 ± 15 post Concentric 180° BFR group: 96 ± 19 pre, 84 ± 18 post Control group: 99 ± 16 pre, 74 ± 12 post Isometric 60° BFR group: 91 ± 18 pre, 72 ± 11 post Control group: 94 ± 17 pre, 62 ± 14 post |
Between groups: Knee extensors Concentric 60° P < 0.01 Concentric 180° P < 0.01 Isometric 60° P < 0.01 Knee flexors Concentric 60° P = 0.05 Concentric 180° P = 0.04 Isometric 60° P = 0.02 |
| Takarada et al 39 |
CSA via MRI: BFR group Total thigh CSA: 167.5 ± 5.1 pre; 156.3 ± 6.5 post Extensors: 54.0 ± 3.0 pre; 48.5 ± 2.6 post (9.4% ± 1.6%) Flexors: 46.2 ± 2.6 pre; 41.7 ± 2.2 post (9.2% ± 2.6%) Control group Total thigh CSA: 161.0 ± 4.7 pre; 137.5 ± 6.4 post Extensors: 52.8 ± 3.3 pre; 42.1 ± 3.4 post (20.7% ± 2.2%) Flexors: 49.1 ± 4.7 pre; 44.8 ± 5.4 post (11.3% ± 2.6%) |
Within-group comparison: Total CSA, extensors, flexors: P < 0.05 Between-group comparison Knee extensors: P < 0.05 (smaller decrease in favor of BFR group) Knee flexors: P = 0.69 |
N/A | N/A |
| Kilgas et al 21 |
Muscle thickness via ultrasound: ACL/BFR Training group Rectus femoris: Involved limb: 1.83 ± 0.33 pre, 2.02 ± 0.32 post Uninvolved limb: 2.10 ± 0.31 pre, 2.09 ± 0.31 post LSI 87% ± 5% pre, 96% ± 4% post Thickness (↑11% ± 5%) Vastus lateralis: Involved limb: 2.25 ± 0.39 pre, 2.47 ± 0.40 post Uninvolved limb: 2.10 ± 0.31 pre, 2.09 ± 0.31 post LSI 90% ± 6% pre, 97% ± 3% post. Thickness (↑10% ± 6%) Healthy control group Rectus femoris: LSI 99% ± 5% Vastus lateralis: LSI 101% ± 3% |
Within-group comparison
Rectus femoris: P < 0.01, LSI P = 0.03 Uninvolved limb P = 0.76 Vastus lateralis: P < 0.01, LSI P = 0.02. Uninvolved limb P = 0.47 Between-group comparison: Rectus femoris: LSI P = 0.28 Vastus lateralis: LSI P = 0.03 |
Estimated 1RM testing (Cybex knee extension)
ACL/BFR training group Involved limb: 34.9 ± 13.8 pre, 41.6 ± 16.8 post Uninvolved limb: 39.7 ± 15.4 pre, 41.8 ± 17.1 post LSI: 88% ± 4% pre; 99% ± 5% post. Knee extensor strength: ↑20% ± 14% Healthy control group LSI: 99% ± 5% |
Within-group comparison
ACL group: Involved limb strength: P < 0.01 Between-group comparison LSI post, P = 0.95 |
ACL, anterior cruciate ligament; ACSA, anatomic cross-sectional area; BFR, blood flow restriction; CSA, cross-sectional area; LSI, limb symmetry index; MRI, magnetic resonance imaging; MVIC, maximal voluntary isometric contraction; N/A, not applicable; RM, repetition maximum.
Values are expressed as mean ± standard deviation.
Figure 2 presents the results from our post hoc analysis of morphological measures. Overall, the magnitude of quadriceps morphologic changes ranged from trivial to large (d = 0.06 to 0.81) with wide CIs suggesting inconclusive favorable benefit among studies. ESs for knee flexor morphological outcome measures ranged from small in favor of conventional treatment to trivial in favor of BFR training (d = −0.20 to 0.13) with wide CIs, corresponding to a nonclinically meaningful difference between treatments.
Figure 2.
Post hoc analysis of included muscle morphological outcomes via Cohen d effect size calculations. Comparisons are between BFR training groups and control groups. Calculated effect sizes (95% CIs in parentheses) are above each outcome measure. Colors reference to included articles: red, Curran et al 10 ; blue, Hughes et al 18 ; orange, Iversen et al 19 ; gray, Ohta et al 32 ; green, Takarada et al 39 ; black, Kilgas et al. 20 ACSA, anatomic cross-sectional area; BFR, blood flow restriction; CSA, cross-sectional area.
Strength Measures
Knee strength was evaluated in 4 studies.12,19,21,33 Assessment techniques included estimated 1RM 21 or 10RM, 19 isokinetic,12,19,33 and maximal voluntary isometric contraction (MVIC) testing.12,33 Knee extensors were assessed in 4 studies12,19,21,33 and knee flexors in 2 studies.19,33 For studies that paired BFR training with a leg press intervention, varying results were observed. Curran et al 12 identified no differences in knee extensor isokinetic strength and MVIC regardless of BFR training assignment and exercise mode (concentric or eccentric training) at 70% of 1RM. Similarly, there were no differences between groups in Hughes et al, 19 where knee extension peak moment at 60 deg/s decreased when comparing presurgery with postintervention measures. Both groups showed a similar increase for scaled 10RM from pre- to postintervention for both limbs. In contrast, participants in the BFR training group maintained knee extension peak moment at 150 and 300 deg/s, while significant decreases were observed in the conventional therapy group completing repetitions at a predicted 70% of 1RM. 19 Knee flexion peak moment decreased in both groups for all isokinetic testing velocities after the intervention period when compared with presurgery levels, but the magnitude for loss was greater in the control group. 19 Both groups had an increase in noninjured knee extension and flexion peak moment at all velocities from presurgery to postintervention. 19 In Ohta et al, 33 knee extensor and flexor muscle isokinetic and isometric strength loss was reduced in the BFR training group when compared with the control at 16 weeks post ACL reconstruction using both open kinetic chain (OKC) and closed kinetic chain (CKC) knee extensor and flexor exercises.
In the sole study that applied BFR training to participants greater than 2 years post ACL reconstruction, knee extensor strength measured via estimated 1RM increased in the involved limb after 4 weeks. 21 Accordingly, knee extensor limb symmetry index increased and approached 100% within participants. 21
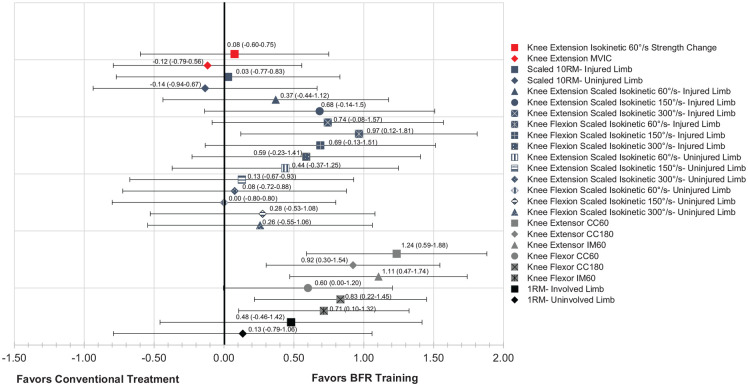
Figure 3 presents the findings of our post hoc analysis for strength measures. ESs ranged from trivial (d = −0.14) in favor of conventional treatment to large (d = 1.24) in favor of BFR training. Only Ohta et al 33 and Hughes et al 19 had strength measures with large, clinically meaningful ESs in favor of BFR training. The range in ESs and predominantly nonclinically meaningful findings signal a lack of consensus on the benefit of BFR training over conventional treatment among studies.
Figure 3.
Post hoc analysis of included muscle strength outcomes via Cohen d effect size calculations. Comparisons are between BFR training groups and control groups. Calculated effect sizes (95% CIs in parentheses) are above each outcome measure. Colors reference to included articles: red, Curran et al 10 ; blue, Hughes et al 18 ; gray, Ohta et al 32 ; black, Kilgas et al. 20 1RM, 1 repetition maximum; 10RM, 10 repetition maximum; BFR, blood flow restriction; CC60, concentric contraction 60°; CC180, concentric contraction 180°; IM60, isometric 60°; MVIC, maximal voluntary isometric contraction.
Discussion
The benefit of BFR training to maintain or improve quadriceps and hamstring muscle size, and knee extension and flexion strength in ACL-reconstructed patients is inconclusive at this time. Although we report predominantly positive ESs suggesting favorable benefit of BFR training, accompanying CIs indicate the results were not clinically meaningful compared with conventional treatment. Coincidentally, 1 study 33 noting clinically meaningful findings in favor of BFR training scored poorly in methodological quality, which hinders inference of results. Postsurgical impairments may have contributed to the lack of precision and large variability in CIs from our post hoc analysis. Based on standards of the SORT scale, grade B inconsistent evidence exists to support BFR training to maintain or improve muscle size and knee extensor and flexor strength in patients post ACL reconstruction.
Morphological Measures
Of the 12 muscle morphological measures reported, only knee extensor CSA 33 and total thigh CSA 41 were clinically meaningful. In contrast, Lixandrão et al 25 reported mean muscle mass to increase by 7.22% after high-load intensity training and BFR training combined with no difference reported between the 2 interventions (mean ES difference = 0.10 ± 0.10 [−0.10 to 0.30]) in a healthy population. 25 Of the clinically meaningful measures in our review, Takarada et al 39 implemented BFR training passively and Ohta et al 33 had the lowest methodological quality score. Postsurgical impairments (eg, postsurgical pain, disuse atrophy, arthrogenic muscle inhibition, and neural drive deficiencies) in a clinical population may explain the lack of positive findings observed in these studies compared with Lixandrão et al. 25 Further research is warranted to understand the clinical utility of BFR training on knee muscle morphological measures post ACL reconstruction.
Muscle Strength
From the 24 muscle strength measures reported, only knee extensor isokinetic strength at 60 deg/s,19,33 150 deg/s, 33 and 300 deg/s 33 and knee flexor isokinetic strength at 60 deg/s, 150 deg/s, and 300 deg/s in 1 study 33 were clinically meaningful. In contrast, Lixandrão et al 25 reported mean strength gain to be 14.36% for both high-load intensity training and BFR training combined, but high-load resistance training had a clinically meaningful greater increase in mean strength by an additional 7.36% (mean ES = 0.63 ± 0.09 [0.43-0.80]). 25 Given the conflicting results we discovered, the influence of postsurgical impairments should be considered. The presence of pain and effusion 35 is known to hinder muscle activation and strength output, which may have affected the results within each study. In Hughes et al, 19 the BFR training group noted a significantly greater decrease in pain and effusion postintervention when compared with the control group completing exercises at a higher load. In contrast, Curran et al 12 reported no group differences in change of subjective knee outcome scores (measured via International Knee Documentation Committee [IKDC]) and strength measures between groups. Tolerance of lower loads and a potential analgesic effect of BFR training on clinical populations 8 may explain the improvements observed in isokinetic knee extension in Hughes et al. 19 A positive effect linked to isokinetic knee extension and flexion in Ohta et al 33 should be interpreted with caution given the poor appraisal of their experimental methods.
As previously stated, variability was present among a multitude of factors between the included studies. Heavy focus on quadriceps dominant exercises was common, where exercises spanned across CKC,12,19,21,33 OKC,20,33 or mixture of both.21,33 Furthermore, mode and intensity of exercises ranged from a passive application 39 to low-load under occlusion.19-21,33 Occlusion protocols differed between set pressures20,33,39 and personalized pressures specific to each participant.12,19,21 Current practice recommendations support the practice of personalized pressures that account for factors (eg, limb circumference 26 and cuff width 28 ) to reduce the risk for adverse events related to extreme pressures.29,36 Additionally, 1 study 12 used an intermittent mode, where occlusion was released during rest periods between sets. Theories for BFR training include supplementation of low-intensity exercises with venous occlusion to stimulate higher threshold, fast twitch muscle fibers through increased metabolite build-up.27,37 Releasing occlusion during rest periods between sets may affect the level of metabolic stress in the muscles being trained. It is unknown if the varying occlusion parameters influenced study results among the included studies. Interestingly, Lixandrão et al 25 noted that main effects reported in their systematic review were not influenced by the heterogenous occlusion parameters, intervention protocols, and outcome measures observed in healthy populations. It is unknown how the variability between study methods in our systematic review influenced the effect of BFR training on muscle size as well as knee extensor and flexor strength in an ACL-reconstructed population. Consistent intervention protocols and refinement in occlusion parameters with similar subjective characteristics (eg, ACL graft and presence of concomitant injuries) are warranted in future research to provide higher quality data for future analyses.
Overall, study quality assessment scores were satisfactory among the articles in our review. The Downs and Black tool identified a wide range of scores with an average score of 18. For the studies21,33,39 scoring below average, selection bias and lack of controlling for bias was identified. The only study 33 with clinically meaningful results had the poorest quality rating. High risk of bias was linked to a lack of allocation concealment in 2 randomized controlled trials,20,33 a lack of blinding of participants in all randomized controlled trials,12,19,20,33 and confounding variables in the nonrandomized studies.21,39
Limitations
The influence of pain and inhibition, postoperative deficiencies, patient characteristics, and variable study methods across studies in our review hindered the ability to determine an overall effect. Another limitation was the scarcity of data on BFR training to improve knee flexor strength post ACL reconstruction. Knee flexor strength was only assessed in 2 studies.19,33 Another limitation was strength assessments only depicted 1RM/10RM or isokinetic concentric strength. Ultimately, this constrained the findings related to other forms of assessment, such as eccentric strength and functional testing assessments. Research on the impact for BFR training to improve subjective knee function in patients long after ACL reconstruction is scant. Only 2 studies12,19 implemented patient reported outcomes pre- and postintervention.
Conclusion
Few studies have examined the effect of BFR training on knee muscle size and knee extensor and flexor strength in patients post ACL reconstruction. From the current literature, inconsistent evidence is present on the effect of BFR training to maintain or improve knee muscle size and knee extensor and flexor strength in patients post ACL reconstruction. There was no indication of consistent clinically meaningful differences between BFR training and conventional therapy. Variability resulting from postsurgical impairments, intervention, and occlusion protocols cloud the overall effect. A need remains for high-quality research studies to understand the effect that BFR training has on ACL-reconstructed patients. Future research should explore residual deficiencies in knee strength, muscle size, and subjective knee outcomes after the first year post ACL reconstruction.
Supplemental Material
Supplemental material, sj-docx-1-sph-10.1177_19417381211070834 for Effects of Blood Flow Restriction Training on Clinical Outcomes for Patients With ACL Reconstruction: A Systematic Review by Mark Colapietro, Brandon Portnoff, Sayers John Miller, Wayne Sebastianelli and Giampietro L. Vairo in Sports Health: A Multidisciplinary Approach
Footnotes
The authors report no potential conflicts of interest in the development and publication of this article.
ORCID iD: Giampietro L. Vairo  https://orcid.org/0000-0002-7575-5016
https://orcid.org/0000-0002-7575-5016
References
- 1. Adams D, Logerstedt D, Hunter-Giordano A, Axe MJ, Snyder-Mackler L. Current concepts for anterior cruciate ligament reconstruction: a criterion-based rehabilitation progression. J Orthop Sports Phys Ther. 2012;42:601-614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ardern CL, Taylor NF, Feller JA, Webster KE. Return-to-sport outcomes at 2 to 7 years after anterior cruciate ligament reconstruction surgery. Am J Sports Med. 2012;40:41-48. [DOI] [PubMed] [Google Scholar]
- 3. Ardern CL, Webster KE, Taylor NF, Feller JA. Return to sport following anterior cruciate ligament reconstruction surgery: a systematic review and meta-analysis of the state of play. Br J Sports Med. 2011;45:596-606. [DOI] [PubMed] [Google Scholar]
- 4. Arve EH, Madrak E, Warren AJ. Does blood flow restriction training improve quadriceps measures after arthroscopic knee surgery? A critically appraised topic. Int J Athl Ther Train. 2020;25:221-226. [Google Scholar]
- 5. Baez SE, Hoch MC, Hoch JM. Psychological factors are associated with return to pre-injury levels of sport and physical activity after ACL reconstruction. Knee Surg Sports Traumatol Arthrosc. 2020;28:495-501. [DOI] [PubMed] [Google Scholar]
- 6. Barber-Westin S, Noyes FR. Blood flow–restricted training for lower extremity muscle weakness due to knee pathology: a systematic review. Sports Health. 2019;11:69-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Bourne MN, Bruder AM, Mentiplay BF, Carey DL, Patterson BE, Crossley KM. Eccentric knee flexor weakness in elite female footballers 1-10 years following anterior cruciate ligament reconstruction. Phys Ther Sport. 2019;37:144-149. [DOI] [PubMed] [Google Scholar]
- 8. Bryk FF, dos Reis AC, Fingerhut D, et al. Exercises with partial vascular occlusion in patients with knee osteoarthritis: a randomized clinical trial. Knee Surg Sports Traumatol Arthrosc. 2016;24:1580-1586. [DOI] [PubMed] [Google Scholar]
- 9. Carofino B, Fulkerson J. Medial hamstring tendon regeneration following harvest for anterior cruciate ligament reconstruction: fact, myth, and clinical implication. Arthroscopy. 2005;21:1257-1265. [DOI] [PubMed] [Google Scholar]
- 10. Centner C, Wiegel P, Gollhofer A, Koenig D. Effects of blood flow restriction training on muscular strength and hypertrophy in older individuals: a systematic review and meta-analysis. Sports Med. 2019;49:95-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Cohen J. Statistical Power Analysis for the Behavioral Sciences. Routledge; 2013. [Google Scholar]
- 12. Curran MT, Bedi A, Mendias CL, Wojtys EM, Kujawa MV, Palmieri-Smith RM. Blood flow restriction training applied with high-intensity exercise does not improve quadriceps muscle function after anterior cruciate ligament reconstruction: a randomized controlled trial. Am J Sports Med. 2020;48:825-837. [DOI] [PubMed] [Google Scholar]
- 13. Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52:377-384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ebell MH, Siwek J, Weiss BD, et al. Strength of recommendation taxonomy (SORT): a patient-centered approach to grading evidence in the medical literature. Am Fam Physician. 2004;69:548-556. [PubMed] [Google Scholar]
- 15. Flanigan DC, Everhart JS, Pedroza A, Smith T, Kaeding CC. Fear of reinjury (kinesiophobia) and persistent knee symptoms are common factors for lack of return to sport after anterior cruciate ligament reconstruction. Arthroscopy. 2013;29:1322-1329. [DOI] [PubMed] [Google Scholar]
- 16. Garber CE, Blissmer B, Deschenes MR, et al. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43:1334-1359. [DOI] [PubMed] [Google Scholar]
- 17. Higgins JPT, Altman DG, Gotzsche PC, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Hughes L, Paton B, Rosenblatt B, Gissane C, Patterson SD. Blood flow restriction training in clinical musculoskeletal rehabilitation: a systematic review and meta-analysis. Br J Sports Med. 2017;51:1003-1011. [DOI] [PubMed] [Google Scholar]
- 19. Hughes L, Rosenblatt B, Haddad F, et al. Comparing the effectiveness of blood flow restriction and traditional heavy load resistance training in the post-surgery rehabilitation of anterior cruciate ligament reconstruction patients: a UK national health service randomised controlled trial. Sports Med. 2019;49:1787-1805. [DOI] [PubMed] [Google Scholar]
- 20. Iversen E, Røstad V, Larmo A. Intermittent blood flow restriction does not reduce atrophy following anterior cruciate ligament reconstruction. J Sport Health Sci. 2016;5:115-118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Kilgas MA, Lytle LLM, Drum SN, Elmer SJ. Exercise with blood flow restriction to improve quadriceps function long after ACL reconstruction. Int J Sports Med. 2019;40:650-656. [DOI] [PubMed] [Google Scholar]
- 22. Konrath JM, Vertullo CJ, Kennedy BA, Bush HS, Barrett RS, Lloyd DG. Morphologic characteristics and strength of the hamstring muscles remain altered at 2 years after use of a hamstring tendon graft in anterior cruciate ligament reconstruction. Am J Sports Med. 2016;44:2589-2598. [DOI] [PubMed] [Google Scholar]
- 23. Kuenze CM, Foot N, Saliba SA, Hart JM. Drop-landing performance and knee-extension strength after anterior cruciate ligament reconstruction. J Athl Train. 2015;50:596-602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Lepley AS, Grooms DR, Burland JP, Davi SM, Kinsella-Shaw JM, Lepley LK. Quadriceps muscle function following anterior cruciate ligament reconstruction: systemic differences in neural and morphological characteristics. Exp Brain Res. 2019;237:1267-1278. [DOI] [PubMed] [Google Scholar]
- 25. Lixandrão ME, Ugrinowitsch C, Berton R, et al. Magnitude of muscle strength and mass adaptations between high-load resistance training versus low-load resistance training associated with blood-flow restriction: a systematic review and meta-analysis. Sports Med. 2018;48:361-378. [DOI] [PubMed] [Google Scholar]
- 26. Loenneke JP, Allen KM, Mouser JG, et al. Blood flow restriction in the upper and lower limbs is predicted by limb circumference and systolic blood pressure. Eur J Appl Physiol. 2015;115:397-405. [DOI] [PubMed] [Google Scholar]
- 27. Loenneke JP, Fahs CA, Wilson JM, Bemben MG. Blood flow restriction: the metabolite/volume threshold theory. Med Hypotheses. 2011;77:748-752. [DOI] [PubMed] [Google Scholar]
- 28. Loenneke JP, Thiebaud RS, Fahs CA, Rossow LM, Abe T, Bemben MG. Effect of cuff type on arterial occlusion. Clin Physiol Funct Imaging. 2013;33:325-327. [DOI] [PubMed] [Google Scholar]
- 29. McEwen JA, Owens JG, Jeyasurya J. Why is it crucial to use personalized occlusion pressures in blood flow restriction (BFR) rehabilitation? J Med Biol Eng. 2019;39:173-177. [Google Scholar]
- 30. Moher D, Liberati A, Tetzlaff J, Altman DG. for the PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535-b2535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Norte GE, Knaus KR, Kuenze C, et al. MRI-based assessment of lower-extremity muscle volumes in patients before and after ACL reconstruction. J Sport Rehabil. 2018;27:201-212. [DOI] [PubMed] [Google Scholar]
- 32. Norte GE, Solaas H, Saliba SA, Goetschius J, Slater LV, Hart JM. The relationships between kinesiophobia and clinical outcomes after ACL reconstruction differ by self-reported physical activity engagement. Phys Ther Sport. 2019;40:1-9. [DOI] [PubMed] [Google Scholar]
- 33. Ohta H, Kurosawa H, Ikeda H, Iwase Y, Satou N, Nakamura S. Low-load resistance muscular training with moderate restriction of blood flow after anterior cruciate ligament reconstruction. Acta Orthop Scand. 2003;74:62-68. [DOI] [PubMed] [Google Scholar]
- 34. Palmieri-Smith R, Lepley L. Quadriceps strength asymmetry following ACL reconstruction alters knee joint biomechanics and functional performance at time of return to activity. Am J Sports Med. 2015;43:1662-1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Palmieri-Smith RM, Villwock M, Downie B, Hecht G, Zernicke R. Pain and effusion and quadriceps activation and strength. J Athl Train. 2013;48:186-191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Patterson SD, Hughes L, Warmington S, et al. Blood flow restriction exercise position stand: considerations of methodology, application, and safety. Front Physiol. 2019;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Scott BR, Loenneke JP, Slattery KM, Dascombe BJ. Exercise with blood flow restriction: an updated evidence-based approach for enhanced muscular development. Sports Med. 2015;45:313-325. [DOI] [PubMed] [Google Scholar]
- 38. Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355.i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Takarada Y, Takazawa H, Ishii N. Applications of vascular occlusion diminish disuse atrophy of knee extensor muscles. Med Sci Sports Exerc. 2000;32:2035-2039. [DOI] [PubMed] [Google Scholar]
- 40. Thomas AC, Villwock M, Wojtys EM, Palmieri-Smith RM. Lower extremity muscle strength after anterior cruciate ligament injury and reconstruction. J Athl Train. 2013;48:610-620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Thomas AC, Wojtys EM, Brandon C, Palmieri-Smith RM. Muscle atrophy contributes to quadriceps weakness after ACL reconstruction. J Sci Med Sport. 2016;19:7-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Vairo GL. Knee flexor strength and endurance profiles after ipsilateral hamstring tendons anterior cruciate ligament reconstruction. Arch Phys Med Rehabil. 2014;95:552-561. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-docx-1-sph-10.1177_19417381211070834 for Effects of Blood Flow Restriction Training on Clinical Outcomes for Patients With ACL Reconstruction: A Systematic Review by Mark Colapietro, Brandon Portnoff, Sayers John Miller, Wayne Sebastianelli and Giampietro L. Vairo in Sports Health: A Multidisciplinary Approach