Abstract
Background
Bariatric surgery is superior to usual care for diabetes remission. Previous meta-analyses were limited by pooling observational and randomized trials, using various definitions of diabetes remission, and not controlling for various diabetes medications. The current meta-analysis aimed to compare bariatric surgery and usual care regarding the same.
Methods
We searched PubMed MEDLINE, Web of Science, SCOPUS, and Cochrane Library for relevant articles from the date of the first inception up to February 2023. The keywords diabetes remission, Bariatric surgery, metabolic surgery, lifestyles, usual care, GLIP-1 agonists, insulin use, gastric banding, biliopancreatic diversion, sleeve gastrectomy, and Roux-en-Y gastric bypass, were used. A datasheet was used to extract the relevant data.
Results
Diabetes remission (complete and prolonged) was higher among bariatric surgeries compared to usual care, odd ratio, 0.06, 95 CI, 0.02–0.25 and 0.12, 95 CI, 0.02–0.72, respectively. bariatric surgery patients were younger, had higher HbA1c, odd ratio, − 3.13, 95 CI, − 3.71 to 2.54, and 0.25, 95 CI, 0.02–0.48, respectively, insulin use was higher, and glucagon-like peptide agonists use was lower among bariatric surgery patients, odd ratio, 0.49, 95% CI, 0.24–0.97, and 3.06, 95% CI, 1.44–6.53, respectively.
Conclusion
Bariatric surgery was better than usual care in diabetes remission. Bariatric surgery patients were younger, had higher HbA1c, and received more insulin and lower GLP-1 agonists. No differences were evident regarding body mass index and the duration of diabetes. Further trials comparing the new anti-diabetic medications and different forms of bariatric surgery and controlling for the level of exercise and diet are recommended.
Keywords: Bariatric surgery, Metabolic surgery, Diabetes remission, Usual care, Lifestyles
Background
Diabetes mellitus is a chronic progressive disorder with a great burden on the patients, the healthcare system, and the community, according to International Diabetes Federation, 463 million adults were suffering from diabetes in the year 2019 with an annual death of 4.2 million globally. The global economic burden of diabetes is USD 760.3 billion in 2019 [1]. Diabetes mellitus is a progressive disease with high microvascular and macrovascular complications. In addition, the disease is associated with high mortality [2]. Diabetes mellitus and obesity are growing health burdens globally; they are usually associated (with diabesity) and put a high strain on the healthcare system.
Type 2 diabetes remission and reversal of metabolic abnormalities have been gaining momentum recently [3, 4]. Diabetes remission is defined as the glycated hemoglobin < 6.5% (48 mmol/mol) measured at least 3 months after stopping diabetes medications and is associated with reduced microvascular complications in particular among young age groups and those with lower comorbidities [5].
Diabetes mellitus remission can be induced with lifestyles (various dietary approaches including low calorie or low carbohydrate diet and various grades of physical activity), surgical intervention (bariatric surgery), and novel antidiabetic medications [6–8].
American College of Lifestyle Medicine's position statement is to target diabetes remission as the primary goal for patients with type 2 diabetes and use a therapeutic diet and exercise. Bariatric surgery showed similar results. However, lifestyles bear fewer side effects [9]. A recent review of meta-analysis stated that low-energy diets and formula meal replacement appear the most effective approaches, generally providing less energy than self-administered food-based diets with no support for any particular macronutrient profile or style [10].
Diabetes prevention through lifestyle modification backdated to the 1980s, evidence from a meta-analysis of randomized controlled trials showed the feasibility of sustained prevention of diabetes by reducing weight, physical activity, and Mediterranean diet [11]. World Health Organization (WHO) acknowledged diabetes reversal by caloric restriction [12], in addition, studies showed the effectiveness of bariatric surgery, carbohydrate restriction, or low-calorie diets [13].
It is stated that a 10% weight reduction is needed for meaningful health improvement. However, weight loss is difficult to achieve through lifestyle alone [14]. A large cohort published in the United Kingdom found that 5% weight loss is achievable in 14.3% and 12.5% of women and men, respectively [15]. Initial weight loss and dysglycemia can be obtained by different dietary approaches at the expense of constipation, alopecia, and cholelithiasis [16]. While cost might be a limiting factor for some diets [17]. Low carbohydrate diets for diabetes management is in use for more than two centuries and were found to be effective as an alternative to low-fat, low-calorie diets [18]. Previous studies showed weight reduction and diabetes remission through intensive lifestyle intervention [19, 20]. Other approaches for remission include novel hypoglycemic medications and bariatric surgery [3]. Gastrointestinal interventions intended for long-term weight loss have evolved since the fifties of the previous century. The new term metabolic surgery was developed to replace bariatric surgery given the concept of morbidity-related obesity surgeries. The term indicates improvement or even remission of metabolic disorders including type 2 diabetes [21]. The improvement in obesity-related metabolic disorders is mainly through sustainable weight loss and neuroendocrine mechanisms. Nearly a half million bariatric surgeries are currently performed annually worldwide [22]. There are many bariatric surgeries including sleeve gastrectomy, Roux-en-Y gastric bypass, and laparoscopic adjustable gastric banding. With the development of laparoscopic techniques, now laparoscopic sleeve gastrectomy (LSG) is gaining favor over other procedures [23].
Bariatric surgery was shown to induce sustainable diabetes remission between one to two-thirds depending on the surgical procedure [24].
Exercise plays a pivotal role in preventing and controlling DM since its effects include most vascular risk factors, with special effects on diabetes. Exercise is the best non-pharmacological therapy for the population in question [25–28].
A previous meta-analysis showed the beneficial effects of moderate-intensity exercise on type 2 diabetes risk (30% risk reduction). The same was observed with regular walking. In addition, the results remained robot after controlling for body mass index [29]. Literature on diabetes remission with bariatric surgery and usual diabetes care lacks. The available meta-analyses on comparisons are limited by pooling observational studies, using various definitions of diabetes remission, and including small studies. Therefore, we conducted this meta-analysis to compare usual care and bariatric surgery effects on diabetes remission and include randomized controlled trials with the most recent definition of diabetes remission.
Materials and methods
Eligibility criteria according to PICOS
Studies were included if they were randomized controlled trials on humans and compared bariatric surgery and usual diabetes care regarding diabetes remission. Retrospective studies, prospective cohorts, cross-sectional studies, case control, and case series were excluded.
Outcome measures
The outcome measures were:
Diabetes remission following bariatric surgery and usual care.
Diabetes remission
Diabetes remission is defined as the achievement of HbA1c of < 6.5 without diabetes medication for three months or longer. Prolonged remission is the maintenance of the same for one year, and permanent remission is HbA1c < 6.5 for five years or more. A glycated hemoglobin of < 6.5 estimated from the mean blood glucose using continuous glucose measurement and fasting blood glucose of < 126 mg/dl are acceptable [4].
Literature search and data extraction
A systematic literature search was conducted in PubMed MEDLINE, Web of Science, SCOPUS, and Cochrane Library from the date of the first inception up to February 2023. The reviewer searched the databases for relevant articles. The keywords diabetes remission, prolonged remission, Bariatric surgery, metabolic surgery, lifestyles, usual care, GLIP-1 agonists, insulin use, gastric banding, bypass surgery, biliopancreatic diversion, gastric bypass, sleeve gastrectomy, Roux-en-Y gastric bypass were used. In addition, the titles, abstracts, and references of the included studies were screened. We identified 432 studies and 318 stands after the removal of duplication, from them, 58 full texts were screened and only 12 studies were included in the final meta-analysis. A datasheet was used to extract the author's name year and country of publication, diabetes remission, HbA1c, age, body mass index, duration of diabetes, type of bariatric surgery, insulin, and GLP-1 agonist's use at baseline (Tables 1, 2, 3, 4, 5; Figs. 1, 2, 3, 4, 5, 6, 7, 8, 9, 10).
Table 1.
The revised risk of bias of the included randomized controlled trials
| Study | Randomization process bias | Deviation from the intended intervention | Missing outcome bias | Measurement of the outcome bias | Selective reporting results bias | Overall bias |
|---|---|---|---|---|---|---|
| Chong et al. 2017 [31] | High risk | Low | Unclear | Low | Low | Low |
| Courcoulas et al. 2020 [32] | High risk | Low | Unclear | Low | Low | Low |
| Ding et al. 2015 [33] | High risk | Low | Unclear | Low | Low | Low |
| Foschi et al. 2019 [34] | High risk | Low | Unclear | Low | Low | low |
| Halperin et al. 2014 [35] | High risk | Low | Unclear | Low | Low | Low |
| Kashyap et al. 2013 [36] | High risk | Unclear | Unclear | Unclear | Unclear | Low |
| Kirwan et al. 2022 [37] | High risk | Low | Unclear | Low | Low | Low |
| Liang et al. 2013 [38] | High risk | Low | Unclear | Low | Low | Low |
| Mingrone et al. 2021 [39] | High risk | Low | Unclear | Low | Low | Low |
| Parikh et al. 2014 [40] | High risk | Low | Unclear | Low | Low | low |
| Simonson et al. 2018 [41] | High risk | Low | Unclear | Low | Low | low |
| Sjöholm et al. 2022 [42] | High risk | Low | Unclear | Low | Low | Unclear |
Table 2.
Randomized controlled trials comparing different types of bariatric surgeries and medical treatments
| Author | Country | Diabetes remission (HbA1c criteria) | Type of surgery | Patients with remission (bariatric) | Patients with remission (usual care) |
|---|---|---|---|---|---|
| Chong et al. 2017 [31] | Taiwan, USA | < 6.5 | RYGB | 7/36 | 0/35 |
| Courcoulas et al. 2020 [32] | USA | < 6.5 | RYGB, AGB | 5/41 | 0/24 |
| Ding et al. 2015 [33] | USA | < 6.5 | AGB | 5/18 | 2/22 |
| Foschi et al. 2019 [34] | Italy | < 6 for complete, < 6.5 for partial | II, SG | 26/30 | 2/25 |
| Halperin et al. 2014 [35] | USA | < 6.5 | RYGB | 11/19 | 3/19 |
| Kashyap et al. 2013 [36] | USA | < 6 | RYGB, SG | 8/40 | 1/20 |
| Kirwan et al. 2022 [37] | USA | < 6.5 | RYGB, SG, and AGB | 60/160 | 2/76 |
| Liang et al. 2013 [38] | USA | < 6.5 | 28/30 | 0/70 | |
| Mingrone et al. 2021 [39] | Italy | < 6.5 | RYGB, BPD | 15/40 | 1/20 |
| Parikh et al. 2014 [40] | USA | < 6.5 | 13/20 | 0/24 | |
| Simonson et al. 2018 [41] | USA | < 6.5 | RYGB | 7/19 | 0/19 |
| Sjöholm et al. 2022 [42] | Sweden | < 6.5 | AGB and GB |
229/393 91/393 |
39/308 11/308 |
RYGB Roux-en-Y gastric bypass, AGB adjustable gastric banding, SG Sleeve gastrectomy, BPD Biliopancreatic diversion, II Ileal interposition with duodenal diversion sleeve gastrectomy
Table 3.
Basic characteristics of different types of bariatric surgeries and medical treatments
| Author | Age (mean ± SD) | Sex (F/M) | BMI (kg/m2) | Duration diabetes | Follow-up/years |
|---|---|---|---|---|---|
| Chong et al. 2017 [31] | 48.2 ± 8.4 | 46/71 | 32.4 ± 1.6 | 8.25 ± 5.05 | Two |
| Courcoulas et al. 2020 [32] | 46.55 ± 7.25 vs. 48.9 ± 4.7 | 33/41 vs. 17/20 | 35.56 ± 3.05 vs. 35.7 ± 3.3 | 6.85 ± 4.45 vs. 5.7 ± 5.6 | Five |
| Ding et al. 2015 [33] | 50.6 ± 12.6 vs. 51.4 ± 7.5 | 9/18 vs. 9/22 | 36.4 ± 3.0 vs. 36.7 ± 4.2 | Not mentioned | One |
| Foschi et al. 2019 [34] | 50.6 ± 1.9 vs. 55.0 ± 1.5 | 22/30 vs. 22/30 | 43.0 ± 1.5 vs. 41.9 ± 1.2 | 4.4 ± 0.6 vs. 4.4 ± 0.7 | Five |
| Halperin et al. 2014 [35] | 50.7 ± 7.6 vs. 52.6 ± 4.3 | 13/19 vs. 10/19 | 36.0 ± 3.5 vs. 36.5 ± 3.4 | 10.6 ± 6.6 vs. 10.2 ± 6.1 | One |
| Kashyap et al. 2013 [36] | 47.9 ± 9.85 vs. 50 ± 8.4 | 24/37 vs. 8/17 | 36.25 ± 2.9 vs. 35.8 ± 3.0 | 7.5 ± 4.75 vs. 10.5 ± 5.0 | Two |
| Kirwan et al. 2022 [37] | 49 ± 9 vs. 52 ± 7 | 137/195 vs. 75/121 | 37 ± 4 vs. 37 ± 3 |
9 ± 6 vs. 9 ± 6 STAMPEDE, TRIABETES, SLIMM-T2D, and CROSSROADS trials |
Three |
| Mingrone et al. 2021 [39] | 20/34 relapse | ≥ 35 | > 5 years | 10 years | |
| Parikh et al. 2014 [40] | 46.8 ± 8.1 vs. 53.9 ± 8.4 | 23/29 vs. 22/28 | 32.8 ± 1.7 vs. 32.4 ± 1.8 | NA | 6 months |
| Simonson et al. 2018 [41] | 50.7 ± 7.6 vs. 52.6 ± 4.3 | 13/19 vs. 10/19 | 36.3 ± 3.4. 6/19 vs. 7/19 < 35 | 10.6 ± 6.6 vs. 10.2 ± 6.1 | Three |
| Sjöholm et al. 2022 [42] | 48.6 ± 6.0 vs. 50.5 ± 6.3 | 236/393 vs. 182/308 | 42.4 ± 4.9 vs. 40.1 ± 4.7 | NA | 10 years |
Table 4.
Diabetter scoring for the included trials
| Study | Diabetter score out of 9 |
|---|---|
| Chong et al. 2017 [31] | 8 |
| Courcoulas et al. 2020 [32] | 6 |
| Ding et al. 2015 [33] | 7 |
| Foschi et al. 2019 [34] | 6 |
| Halperin et al. 2014 [35] | 7 |
| Kashyap et al. 2013 [36] | 8 |
| Kirwan et al. 2022 [37] | 8 |
| Liang et al. 2013 [38] | 7 |
| Mingrone et al. 2021 [39] | 7 |
| Parikh et al. 2014 [40] | 7 |
| Simonson et al. 2018 [41] | 8 |
| Sjöholm et al. 2022 [42] | 6 |
Table 5.
Randomized controlled trials comparing RYGB and different types of bariatric surgeries
| Author | Country | Diabetes remission (HbA1c criteria) | Type of surgery | Patients with remission RYGB | Patients with remission (other surgeries) |
|---|---|---|---|---|---|
| Courcoulas et al. 2020 [32] | USA | < 6.5 | RYGB, AGB | 6/20 | 4/21 |
| Kashyap et al. 2013 [36] | USA | < 6 | RYGB, SG | 6/18 | 2/19 |
| Mingrone et al. 2021 [39] | Italy | < 6.5 | RYGB, BPD | 5/20 | 10/20 |
Fig. 1.
Randomized controlled trials comparing different types of bariatric surgeries and medical treatments (the PRISMA chart)
Fig. 2.
Diabetes remission among different types of bariatric surgeries and medical treatments
Fig. 3.
Complete and prolonged diabetes remission among different types of bariatric surgeries and medical treatments
Fig. 4.
The age among different types of bariatric surgeries and medical treatments
Fig. 5.
HbA1c among patients with different types of bariatric surgeries and lifestyles
Fig. 6.
Duration of diabetes among patients with different types of bariatric surgeries and lifestyles
Fig. 7.
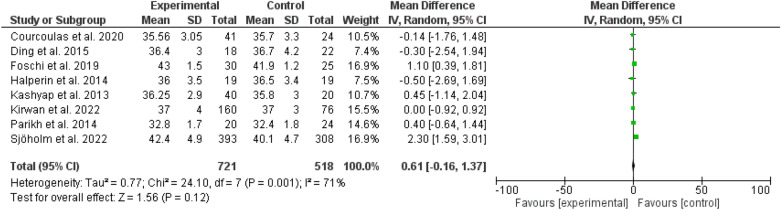
Body mass index among patients with different types of bariatric surgeries and lifestyles
Fig. 8.
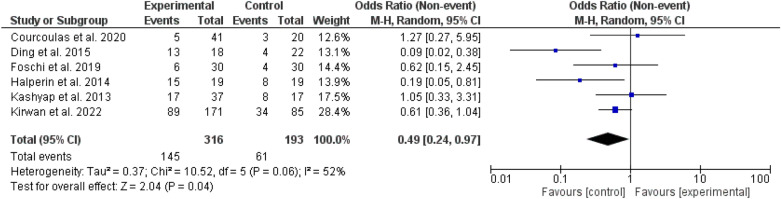
Insulin use among patients with different types of bariatric surgeries and lifestyles
Fig. 9.
Glucagon-like peptide agonists are used among patients with different types of bariatric surgeries and lifestyles
Fig. 10.
A comparison between RYGB and different types of bariatric surgeries regarding diabetes remission
Risk of bias assessment
The revised Cochrane risk of bias assessed tool (Rob.2) was used to assess the quality of the included studies [30].
Statistical analysis
The data were analyzed by the most recent version of the RevMan system (The method used for meta-analysis was DerSimonian and Laird). We pooled 12 randomized controlled trials to assess diabetes remission, of them six studies assessed prolonged remission. The dichotomous and continuous date data were entered manually and the fixed or random effects were applied depending on heterogeneity. In addition, 8 cohorts assessed the age, body mass index, and HbA1c, between bariatric surgery and the usual care groups. In the present meta-analysis, six trials compared insulin use and diabetes duration, while three trials compared GLP-1 agonist use. A P-value of < 0.05 was considered significant.
Results
We included 12 randomized controlled trials [31–42] and 1508 patients (846 bariatric surgery and 662 usual care including lifestyle and diabetes medications). The trials scored from 6–8 on Diabetter scoring system [43]. Diabetes remission was observed in 421 bariatric surgery patients and 193 in the usual care arm. The follow-up period ranged from 6 months to 10 years. Diabetes remission was higher among bariatric surgeries compared to usual care, odd ratio, 0.06, 95 CI, 0.02–0.25, Chi-square = 90.07, P-value for overall effect < 0.0001 the heterogeneity was substantial, I2, 88%, and P-value for heterogeneity < 0.0001 (Fig. 2).
Diabetes remission either complete or prolonged was higher among patients who underwent bariatric surgery, odd ratio, 0.12, 95 CI, 0.02–0.72, Chi-square = 33.63, P-value for overall effect, 0.02. The heterogeneity was significant, I2 for heterogeneity, 85%, and the P-value < 0.001 (Fig. 3).
Patients with bariatric surgery were younger (odd ratio, − 3.13, 95 CI, − 3.71 to 2.54, Chi-square = 18.39, P-value for overall effect, < 0.001, the heterogeneity was significant, I2, 62%, and P-value for heterogeneity, 0.01) (Fig. 4).
The glycated hemoglobin was higher among the bariatric surgery arm (odd ratio, 0.25, 95 CI, 0.02–0.48, Chi-square = 18.21, P-value for overall effect, 0.03 the heterogeneity was significant, I2, 62%, and P-value for heterogeneity, 0.01) (Fig. 5).
No differences were evident between bariatric surgery and usual care regarding the duration of diabetes (odd ratio, 0.02, 95 CI, − 0.54 to 0.58, P-value, 0.93), and body mass index (odd ratio, 0.61, 95 CI, − 0.16 to 1.37, P-value, 0.12) (Figs. 6, 7).
Insulin use was higher among the bariatric surgery arm, odd ratio, 0.49, 95% CI, 0.24–0.97, Chi-square, 10.52, and P-value for overall effect, 0.04, while glucagon-like agonists use was lower, odd ratio, 3.06, 95% CI, 1.44–6.53, Chi-square, 11.83, and P-value for overall effect, 0.004. The heterogeneity was 52% and 83% respectively (Figs 8, 9).
In the present meta-analysis, only three studies compared RYGB and other surgeries with no significant statistical difference (odd ratio, 1.27, 95% CI, 0.29–5.58, P-value for overall effect, 0.75, and Chi-square, 5.78. A significant heterogeneity was found, I2 for heterogeneity, 65%, P-value, 0.06) (Fig. 10).
Discussion
In the present meta-analysis, bariatric surgery achieved higher diabetes remission and prolonged remission rates, odd ratio, 0.06, 95 CI, 0.02–0.25, and odd ratio, 0.12, 95 CI, 0.02–0.72); the current findings supported Schauer et al. [21]. However, Schauer and colleagues included studies with a different cut-off of diabetes remission. Cresci et al. [44] conducted a meta-analysis and found more diabetes remission among the surgery arm; their work was limited by fewer events (only six among the lifestyle group). In addition, the authors included studies with short follow-ups and studies published by the same authors. Similarly, Yu et al. [45] and Kim et al. [46] results were limited by pooling both trials and observational studies conducted among certain ethnicities. Khorgami et al. [47] included seven randomized controlled trials with short follow-up duration and found similar results. This is the largest meta-analysis updating and supporting the previous findings. We excluded studies published by the same authors and retained the most recent [32]). In addition to a sub-analysis of four randomized controlled trials [37], and a 10-year update of Mingrone et al. [39]. Furthermore, the current meta-analysis updated the previous findings and included studies with up to 10 years of follow-up. Importantly, we calculated the relapse rate (297–146 = 151 in the metabolic surgery and 186–19 = 167 in the usual care group). The results showed that 50.8% of bariatric surgery and 89.7% of the usual care relapsed with long follow-ups. The above findings imply that the majority relapsed. It is interesting to note that patients with bariatric surgery received more insulin and lower glucagon-like peptide agonists at baseline, (GLP-1 studies were not included in the complete and prolonged remission sub-analysis). Intensive Insulin therapy was found to induce diabetes remission in nearly half of the patients at one year irrespective of body weight. The effect is through the enhancement of β-cell function [48]. Anti-diabetes medications in particular sodium-glucose co-transporters-2 inhibitors and glucagon-like peptide agonists were shown to induce diabetes remission in particular when combined with other therapies including insulin [49, 50]. Metformin combination with other oral hypoglycemic medication was proven to induce diabetes remission [51, 52]. In the present study, the patient who underwent bariatric surgery were younger and had higher HbA1c (odd ratio, − 3.13, 95 CI, − 3.71 to 2.54 and 0.25, 95 CI, 0.02–0.48, respectively). No differences were evident regarding body mass index and duration of diabetes. Previous studies reported that baseline HbA1c and short duration of diabetes as predictors of diabetes remission irrespective of body mass index [53, 54]. The current findings shined the light on the complexity of diabetes mellitus and its complexity. Diabetes mellitus is a vascular and multi-system disease; it is associated with various metabolic disorders including diabetes, hypertension, dyslipidemia, and metabolic-associated fatty liver disease (MAFLD). In this view, the choice of the best intervention is based on its effects on various diabetes-associated comorbidities [55]. Weight management is crucial in holistic diabetes care [56]. However, which strategy to achieve is still to be determined. The current findings supported bariatric surgery as first-line and compared to usual care and lifestyle intervention both in complete remission and prolonged remission. However, the heterogeneity observed limited our findings (not observed in complete and prolonged remission). Bariatric surgery induces a quick surge in GLP-1 earlier in the first weeks before weight loss [57]. The novel anti-diabetic medications including GLP-1-like agonists were recently approved for both diabetes and weight management. In addition, diabetes remission was observed in 66% to 81% [57]. Moreover, GLP-1 agonists reduced MAFLD, cardiac remodeling, and reduced pancreatic fat [50, 58, 59]. The most important question is who benefits the most? What is the role of novel antidiabetic medications? In addition, which intervention is for a particular patient with particular comorbidities? The strength of the current meta-analysis was the use of the recent definition of diabetes remission, the assessment of prolonged remission and relapse rate, and the assessment of insulin and GLIP-1 agonists use. In addition, we excluded studies published by the same authors and included new updates with longer follow-ups.
A comparison of different bariatric surgeries regarding diabetes remission
All types of bariatric surgery were superior to usual care in diabetes remission. The current study pooled various types of bariatric surgery, which is a major limitation. However, Uhe et al. [60] compared Roux-en-Y gastric bypass, sleeve gastrectomy, or one-anastomosis gastric bypass and found no difference regarding diabetes remission in agreement with the current findings in which there was no significant difference between RYGB and other types of bariatric surgeries. The small number of the included studies limited Uhe and colleagues' meta-analysis (three, six, and three at 3 months, one year, and 5 years, respectively).
Ding et al. [61] compared usual care and six bariatric surgeries and found that all were superior to usual care with Mini-gastric bypass the better, followed by biliopancreatic diversion, laparoscopic sleeve gastrectomy, and Roux-en-Y gastric bypass. However, at three years BPD, and mini-GBP were better. When considering all obesity comorbidities, Roux-en-Y gastric bypass was the best choice. In this meta-analysis, it is not possible to compare all six types of bariatric procedures.
Castellana et al. [62] included ten randomized controlled trials and compared the most two commonly used bariatric surgeries and found the superiority of Roux-en-Y gastric bypass over laparoscopic sleeve gastrectomy in the short-term. A recent interesting meta-analysis conducted by Fehervari et al. [63] found good weight loss and diabetes remission after sleeve gastrectomy conversion to Roux-en-Y gastric bypass. In this meta-analysis we compared RYGB arm with gastric banding, sleeve gastrectomy, and biliopancreatic diversion and no difference was found. A plausible explanation is the small number of the studies included. In addition, we compared malabsorption procedure in one arm with restrictive and malabsorption in the other arm.
Kwon et al. [64] assessed the length of the biliopancreatic and Roux limb in Roux-en-Y gastric bypass and found a higher rate of diabetes remission in the longer biliopancreatic but not the Roux limb. A more recent meta-analysis published in France [65] found that all bariatric surgeries were superior to medical treatment with one anastomosis gastric bypass and biliopancreatic diversion being the most effective followed by Roux-en-Y gastric bypass. Importantly, the authors observed a progressive decrease in diabetes remission over time regardless of the intervention. Li et al. [66] compared metabolic surgeries, restrictive procedures, and lifestyles and found that bariatric surgery was more effective in diabetes remission.
In conclusion, all types of bariatric surgeries were superior to lifestyle and usual care with mini-gastric bypass being the best followed by biliopancreatic diversion. Roux-en-Y gastric bypass was the best when considering all obesity comorbidities especially if the biliopancreatic limb is long. RGYB might be better in the real world because diabetes mellitus is usually accompanied by other comorbidities including hypertension dyslipidemia, and gastroesophageal reflux [67–69]. The high relapse needs to be observed in different interventions calls for longer randomized controlled trials.
The study limitations were the pooling of different bariatric surgeries, the high heterogeneity observed in some analyses, no information available regarding the level of exercise among the bariatric surgery patients, and the fact that we could not control for the different oral hypoglycemic medications.
Conclusion
Bariatric surgery was better than usual care in diabetes remission both in the short and long term. Bariatric surgery patients were younger, had higher HbA1c, and received more insulin and lower GLP-1-like agonists. No differences were evident regarding body mass index and the duration since diabetes diagnosis. No difference was found between RYGB and other bariatric surgeries regarding diabetes remission. Further trials comparing the new anti-diabetic medications and different forms of bariatric surgery and controlling for the level of exercise and diet are needed.
Abbreviations
- RYGB
Roux-en-Y gastric bypass
- AGB
Adjustable gastric banding
- SG
Sleeve gastrectomy
- BPD
Biliopancreatic diversion
- II
Ileal interposition with duodenal diversion sleeve gastrectomy
- GLP-1 agonists
Glucagon-like peptide-1 agonists
Author contributions
The two authors searched the literature. HM, the concept, and the design. HM. Analyzed the data, both authors drafted the manuscript and revised it critically before submission. Both authors read and approved the final manuscript.
Funding
This manuscript is self-funded and not supported by any institution or organization.
Availability of data and materials
The data used in this manuscript are available upon request.
Declarations
Ethical approval and consent to participate
We did not include articles published by us.
Consent for publication
Not applicable.
Competing interests
None.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.International Diabetes Federation. Diabetes Atlas 9th Edition. 2019. Available online: https://www.diabetesatlas.org/en/. Accessed 21 Apr 2022.
- 2.Galaviz KI, Narayan KMV, Lobelo F, Weber MB. Lifestyle and the prevention of type 2 diabetes: a status report. Am J Lifestyle Med. 2015;12:4–20. doi: 10.1177/1559827615619159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kalra S, Bantwal G, Kapoor N, Sahay R, Bhattacharya S, Anne B, et al. Quantifying remission probability in type 2 diabetes mellitus. Clin Pract. 2021;11(4):850–859. doi: 10.3390/clinpract11040100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Riddle MC, Cefalu WT, Evans PH, Gerstein HC, Nauck MA, Oh WK, et al. Consensus report: definition and interpretation of remission in type 2 diabetes. J Clin Endocrinol Metab. 2022;107(1):1–9. doi: 10.1210/clinem/dgab585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hounkpatin H, Stuart B, Farmer A, Dambha-Miller H. Association of type 2 diabetes remission and risk of cardiovascular disease in pre-defined subgroups. Endocrinol Diabetes Metab. 2021;4(3):e00280. doi: 10.1002/edm2.280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Courcoulas AP, Gallagher JW, Neiberg RH, Eagleton EB, DeLany JP, Lang W, Punchai S, Gourash W, Jakicic JM. Bariatric surgery vs lifestyle intervention for diabetes treatment: 5-year outcomes from a randomized trial. J Clin Endocrinol Metab. 2020;105(3):866–876. doi: 10.1210/clinem/dgaa006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brown A, McArdle P, Taplin J, Unwin D, Unwin J, Deakin T, et al. Dietary strategies for remission of type 2 diabetes: a narrative review. J Hum Nutr Diet. 2022;35(1):165–178. doi: 10.1111/jhn.12938. [DOI] [PubMed] [Google Scholar]
- 8.McInnes N, Hall S, Sultan F, Aronson R, Hramiak I, Harris S, et al. Remission of type 2 diabetes following a short-term intervention with insulin glargine, metformin, and dapagliflozin. J Clin Endocrinol Metab. 2020;105(8):248. doi: 10.1210/clinem/dgaa248. [DOI] [PubMed] [Google Scholar]
- 9.Kelly J, Karlsen M, Steinke G. Type 2 diabetes remission and lifestyle medicine: a position statement from the American College of lifestyle medicine. Am J Lifestyle Med. 2020;14(4):406–419. doi: 10.1177/1559827620930962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Churuangsuk C, Hall J, Reynolds A, Griffin SJ, Combet E, Lean MEJ. Diets for weight management in adults with type 2 diabetes: an umbrella review of published meta-analyses and systematic review of trials of diets for diabetes remission. Diabetologia. 2022;65(1):14–36. doi: 10.1007/s00125-021-05577-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Uusitupa M, Khan TA, Viguiliouk E, Kahleova H, Rivellese AA. Prevention of type 2 diabetes by lifestyle changes: a systematic review and meta-analysis. Nutrients. 2019;11:2611. doi: 10.3390/nu11112611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hallberg SJ, Gershuni VM, Hazbun TL, Athinarayanan SJ. Reversing type 2 diabetes: a narrative review of the evidence. Nutrients. 2019;11:766. doi: 10.3390/nu11040766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.World Health Organization. Global Report on Diabetes. 2016. Available online: https://www.who.int/publications/i/item/978924 1565257. Accessed 21 Apr 2021.
- 14.Kelly T, Unwin D, Finucane F. Low-carbohydrate diets in the management of obesity and type 2 diabetes: a review from clinicians using the approach in practice. Int J Environ Res Public Health. 2020;17(7):2557. doi: 10.3390/ijerph17072557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fildes A, Charlton J, Rudisill C, Littlejohns P, Prevost AT, Gulliford MC. Probability of an obese person attaining normal body weight: cohort study using electronic health records. Am J Public Health. 2015;105(9):e54–e59. doi: 10.2105/AJPH.2015.302773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Anderson JW, Conley SB, Nicholas AS. One hundred pound weight losses with an intensive behavioral program: changes in risk factors in 118 patients with long-term follow-up. Am J Clin Nutr. 2007;86(2):301–307. doi: 10.1093/ajcn/86.2.301. [DOI] [PubMed] [Google Scholar]
- 17.Finkelstein EA, Kruger E. Meta- and cost-effectiveness analysis of commercial weight loss strategies. Obesity (Silver Spring) 2014;22(9):1942–1951. doi: 10.1002/oby.20824. [DOI] [PubMed] [Google Scholar]
- 18.Rollo J. Account of two cases of diabetes mellitus, with remarks. Ann Med (Edinb). 1797;2:85–105. [PMC free article] [PubMed] [Google Scholar]
- 19.Taheri S, Zaghloul H, Chagoury O, Elhadad S, Ahmed SH, El Khatib N, et al. Effect of intensive lifestyle intervention on bodyweight and glycaemia in early type 2 diabetes (DIADEM-I): an open-label, parallel-group, randomised controlled trial. Lancet Diabetes Endocrinol. 2020;8(6):477–489. doi: 10.1016/S2213-8587(20)30117-0. [DOI] [PubMed] [Google Scholar]
- 20.de Hoogh IM, Oosterman JE, Otten W, Krijger AM, Berbée-Zadelaar S, Pasman WJ, et al. The effect of a lifestyle intervention on type 2 diabetes pathophysiology and remission: the stevenshof pilot study. Nutrients. 2021;13(7):2193. doi: 10.3390/nu13072193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schauer PR, Mingrone G, Ikramuddin S, Wolfe B. Clinical outcomes of metabolic surgery: efficacy of glycemic control, weight loss, and remission of diabetes. Diabetes Care. 2016;39(6):902–911. doi: 10.2337/dc16-0382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Angrisani L, Santonicola A, Iovino P, Formisano G, Buchwald H, Scopinaro N. Bariatric surgery worldwide 2013. Obes Surg. 2015;25:1822–1832. doi: 10.1007/s11695-015-1657-z. [DOI] [PubMed] [Google Scholar]
- 23.Khorgami Z, Andalib A, Corcelles R, Aminian A, Brethauer S, Schauer P. Recent national trends in the surgical treatment of obesity: sleeve gastrectomy dominates. Surg Obes Relat Dis. 2015;11:S6–S8. doi: 10.1016/j.soard.2015.10.012. [DOI] [Google Scholar]
- 24.Purnell JQ, Selzer F, Wahed AS, Pender J, Pories W, Pomp A, et al. Type 2 diabetes remission rates after laparoscopic gastric bypass and gastric banding: results of the longitudinal assessment of bariatric surgery study. Diabetes Care. 2016;39(7):1101–1107. doi: 10.2337/dc15-2138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Colberg SR, Sigal RJ, Fernhall B, Regensteiner JG, Blissmer BJ, Rubin RR, Chasan-Taber L, Albright AL, Braun B. Exercise and type 2 diabetes: the American College of Sports Medicine and the American Diabetes Association: joint position statement executive summary. Diabetes Care. 2010;33:2692–2696. doi: 10.2337/dc10-1548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bassuk SS, Manson JE. Epidemiological evidence for the role of physical activity in reducing the risk of type 2 diabetes and cardiovascular disease. J Appl Physiol. 1985;2005(99):1193–1204. doi: 10.1152/japplphysiol.00160.2005. [DOI] [PubMed] [Google Scholar]
- 27.Maiorana A, O’Driscoll G, Goodman C, Taylor R, Green D. Combined aerobic and resistance exercise improves glycemic control and fitness in type 2 diabetes. Diabetes Res Clin Pract. 2002;56:115–123. doi: 10.1016/S0168-8227(01)00368-0. [DOI] [PubMed] [Google Scholar]
- 28.Lima LC, Assis GV, Hiyane W, Almeida WS, Arsa G, Baldissera V, Campbell CS, Simões HG. Hypotensive effects of exercise performed around anaerobic threshold in type 2 diabetic patients. Diabetes Res Clin Pract. 2008;81:216–222. doi: 10.1016/j.diabres.2008.04.019. [DOI] [PubMed] [Google Scholar]
- 29.Physical Activity/Exercise and Diabetes American Diabetes Association. Diabetes Care. 2004;27 (Suppl 1): S58–S62. 10.2337/diacare.27.2007.S58. [DOI] [PubMed]
- 30.Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 31.Chong K, Ikramuddin S, Lee WJ, Billington CJ, Bantle JP, Wang Q, et al. National differences in remission of type 2 diabetes mellitus after Roux-en-Y gastric bypass surgery-subgroup analysis of 2-year results of the diabetes surgery study comparing Taiwanese with Americans with mild obesity (BMI 30–35 kg/m2) Obes Surg. 2017;27(5):1189–1195. doi: 10.1007/s11695-016-2433-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Courcoulas AP, Gallagher JW, Neiberg RH, Eagleton EB, DeLany JP, Lang W, et al. Bariatric surgery vs lifestyle intervention for diabetes treatment: 5-year outcomes from a randomized trial. J Clin Endocrinol Metab. 2020;105(3):866–876. doi: 10.1210/clinem/dgaa006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ding SA, Simonson DC, Wewalka M, Halperin F, Foster K, Goebel-Fabbri A, et al. Adjustable gastric band surgery or medical management in patients with type 2 diabetes: a randomized clinical trial. J Clin Endocrinol Metab. 2015;100(7):2546–2556. doi: 10.1210/jc.2015-1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Foschi D, Sorrentino L, Tubazio I, Vecchio C, Vago T, Bevilacqua M, et al. Ileal interposition coupled with duodenal diverted sleeve gastrectomy versus standard medical treatment in type 2 diabetes mellitus obese patients: long-term results of a case-control study. Surg Endosc. 2019;33(5):1553–1563. doi: 10.1007/s00464-018-6443-2. [DOI] [PubMed] [Google Scholar]
- 35.Halperin F, Ding SA, Simonson DC, Panosian J, Goebel-Fabbri A, Wewalka M, et al. Roux-en-Y gastric bypass surgery or lifestyle with intensive medical management in patients with type 2 diabetes: feasibility and 1-year results of a randomized clinical trial. JAMA Surg. 2014;149(7):716–726. doi: 10.1001/jamasurg.2014.514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kashyap SR, Bhatt DL, Wolski K, Watanabe RM, Abdul-Ghani M, Abood B, et al. Metabolic effects of bariatric surgery in patients with moderate obesity and type 2 diabetes: analysis of a randomized control trial comparing surgery with intensive medical treatment. Diabetes Care. 2013;36(8):2175–2182. doi: 10.2337/dc12-1596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kirwan JP, Courcoulas AP, Cummings DE, Goldfine AB, Kashyap SR, Simonson DC, et al. Diabetes remission in the alliance of randomized trials of medicine versus metabolic surgery in type 2 diabetes (ARMMS-T2D) Diabetes Care. 2022;45(7):1574–1583. doi: 10.2337/dc21-2441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Liang Z, Wu Q, Chen B, Yu P, Zhao H, Ouyang X. Effect of laparoscopic Roux-en-Y gastric bypass surgery on type 2 diabetes mellitus with hypertension: a randomized controlled trial. Diabetes Res Clin Pract. 2013;101(1):50–56. doi: 10.1016/j.diabres.2013.04.005. [DOI] [PubMed] [Google Scholar]
- 39.Mingrone G, Panunzi S, De Gaetano A, Guidone C, Iaconelli A, Capristo E, et al. Metabolic surgery versus conventional medical therapy in patients with type 2 diabetes: 10-year follow-up of an open-label, single-centre, randomised controlled trial. Lancet. 2021;397(10271):293–304. doi: 10.1016/S0140-6736(20)32649-0. [DOI] [PubMed] [Google Scholar]
- 40.Parikh M, Chung M, Sheth S, McMacken M, Zahra T, Saunders JK, et al. Randomized pilot trial of bariatric surgery versus intensive medical weight management on diabetes remission in type 2 diabetic patients who do NOT meet NIH criteria for surgery and the role of soluble RAGE as a novel biomarker of success. Ann Surg. 2014;260(4):617–622. doi: 10.1097/SLA.0000000000000919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Simonson DC, Halperin F, Foster K, Vernon A, Goldfine AB. Clinical and patient-centered outcomes in obese patients with type 2 diabetes 3 years after randomization to Roux-en-Y gastric bypass surgery versus intensive lifestyle management: the SLIMM-T2D study. Diabetes Care. 2018;41(4):670–679. doi: 10.2337/dc17-0487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sjöholm K, Carlsson LMS, Svensson PA, Andersson-Assarsson JC, Kristensson F, Jacobson P, et al. Association of bariatric surgery with cancer incidence in patients with obesity and diabetes: long-term results from the Swedish obese subjects study. Diabetes Care. 2022;45(2):444–450. doi: 10.2337/dc21-1335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.de Abreu SL, da Silva RBR, Galletti RP, Agareno GA, Colonno BB, de Sousa JHB, et al. Scores for predicting diabetes remission in bariatric surgery: a systematic review and meta-analysis. Obes Surg. 2023;33(2):600–610. doi: 10.1007/s11695-022-06382-5. [DOI] [PubMed] [Google Scholar]
- 44.Cresci B, Cosentino C, Monami M, Mannucci E. Metabolic surgery for the treatment of type 2 diabetes: a network meta-analysis of randomized controlled trials. Diabetes Obes Metab. 2020;22(8):1378–1387. doi: 10.1111/dom.14045. [DOI] [PubMed] [Google Scholar]
- 45.Yu J, Zhou X, Li L, Li S, Tan J, Li Y, et al. The long-term effects of bariatric surgery for type 2 diabetes: systematic review and meta-analysis of randomized and non-randomized evidence. Obes Surg. 2015;25(1):143–158. doi: 10.1007/s11695-014-1460-2. [DOI] [PubMed] [Google Scholar]
- 46.Kim JH, Pyo JS, Cho WJ, Kim SY. The effects of bariatric surgery on type 2 diabetes in Asian populations: a meta-analysis of randomized controlled trials. Obes Surg. 2020;30(3):910–923. doi: 10.1007/s11695-019-04257-w. [DOI] [PubMed] [Google Scholar]
- 47.Khorgami Z, Shoar S, Saber AA, Howard CA, Danaei G, Sclabas GM. Outcomes of bariatric surgery versus medical management for type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Obes Surg. 2019;29(3):964–974. doi: 10.1007/s11695-018-3552-x. [DOI] [PubMed] [Google Scholar]
- 48.Kramer CK, Zinman B, Choi H, Retnakaran R. Predictors of sustained drug-free diabetes remission over 48 weeks following short-term intensive insulin therapy in early type 2 diabetes. BMJ Open Diabetes Res Care. 2016;4:e000270. doi: 10.1136/bmjdrc-2016-000270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.McInnes N, Hall S, Sultan F, Aronson R, Hramiak I, Harris S, et al. Remission of type 2 diabetes following a short-term intervention with insulin glargine, metformin, and dapagliflozin. J Clin Endocrinol Metab. 2020;105:2532–2540. doi: 10.1210/clinem/dgaa248. [DOI] [PubMed] [Google Scholar]
- 50.Jastreboff AM, Aronne LJ, Ahmad NN, Wharton S, Connery L, Alves B, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205–216. doi: 10.1056/NEJMoa2206038. [DOI] [PubMed] [Google Scholar]
- 51.Jennings AS, Lovett AJ, George TM, Jennings JS. Getting to goal in newly diagnosed type 2 diabetes using combination drug “subtraction therapy”. Metabolism. 2015;64:1005–1012. doi: 10.1016/j.metabol.2015.04.008. [DOI] [PubMed] [Google Scholar]
- 52.Panikar V, Joshi SR, Bukkawar A, Nasikkar N, Santwana C. Induction of long-term glycemic control in type 2 diabetic patients using pioglitazone and metformin combination. J Assoc Physicians India. 2007;55:333–337. [PubMed] [Google Scholar]
- 53.Aung L, Lee WJ, Chen SC, Ser KH, Wu CC, Chong K, et al. Bariatric surgery for patients with early-onset vs late-onset type 2 diabetes. JAMA Surg. 2016;151(9):798–805. doi: 10.1001/jamasurg.2016.1130. [DOI] [PubMed] [Google Scholar]
- 54.Panunzi S, De Gaetano A, Carnicelli A, Mingrone G. Predictors of remission of diabetes mellitus in severely obese individuals undergoing bariatric surgery: do BMI or procedure choice matter? A meta-analysis. Ann Surg. 2015;261(3):459–467. doi: 10.1097/SLA.0000000000000863. [DOI] [PubMed] [Google Scholar]
- 55.Teck J. Diabetes-associated comorbidities. Prim Care. 2022;49(2):275–286. doi: 10.1016/j.pop.2021.11.004. [DOI] [PubMed] [Google Scholar]
- 56.Davies MJ, Aroda VR, Collins BS, Gabbay RA, Green J, Maruthur NM, et al. Management of hyperglycaemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) Diabetologia. 2022;65(12):1925–1966. doi: 10.1007/s00125-022-05787-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lok KH, Wareham NJ, Nair RS, How CW, Chuah LH. Revisiting the concept of incretin and enteroendocrine L-cells as type 2 diabetes mellitus treatment. Pharmacol Res. 2022;180:106237. doi: 10.1016/j.phrs.2022.106237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ghosal S, Datta D, Sinha B. A meta-analysis of the effects of glucagon-like-peptide 1 receptor agonist (GLP1-RA) in nonalcoholic fatty liver disease (NAFLD) with type 2 diabetes (T2D) Sci Rep. 2021;11:22063. doi: 10.1038/s41598-021-01663-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Al-Mrabeh A, Hollingsworth KG, Shaw JA, McConnachie A, Sattar N, Lean ME, Taylor R. 2-year remission of type 2 diabetes and pancreas morphology: a post-hoc analysis of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 2020;8:939–948. doi: 10.1016/S2213-8587(20)30303-X. [DOI] [PubMed] [Google Scholar]
- 60.Uhe I, Douissard J, Podetta M, Chevallay M, Toso C, Jung MK, et al. Roux-en-Y gastric bypass, sleeve gastrectomy, or one-anastomosis gastric bypass? A systematic review and meta-analysis of randomized-controlled trials. Obesity (Silver Spring) 2022;30(3):614–627. doi: 10.1002/oby.23338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ding L, Fan Y, Li H, Zhang Y, Qi D, Tang S, et al. Comparative effectiveness of bariatric surgeries in patients with obesity and type 2 diabetes mellitus: a network meta-analysis of randomized controlled trials. Obes Rev. 2020;21(8):e13030. doi: 10.1111/obr.13030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Castellana M, Procino F, Biacchi E, Zupo R, Lampignano L, Castellana F, et al. Roux-en-Y gastric bypass vs sleeve gastrectomy for remission of type 2 diabetes. J Clin Endocrinol Metab. 2021;106(3):922–933. doi: 10.1210/clinem/dgaa737. [DOI] [PubMed] [Google Scholar]
- 63.Fehervari M, Banh S, Varma P, Das B, Al-Yaqout K, Al-Sabah S, et al. Weight loss specific to indication, remission of diabetes, and short-term complications after sleeve gastrectomy conversion to Roux-en-Y gastric bypass: a systematic review and meta-analysis. Surg Obes Relat Dis. 2022;S1550–7289(22):00757–762. doi: 10.1016/j.soard.2022.11.004. [DOI] [PubMed] [Google Scholar]
- 64.Kwon Y, Lee S, Kim D, ALRomi A, Park SH, Lee CM, et al. Biliopancreatic limb length as a potential key factor in superior glycemic outcomes after Roux-en-Y gastric bypass in patients with type 2 diabetes: a meta-analysis. Diabetes Care. 2022;45(12):3091–3100. doi: 10.2337/dc22-0835. [DOI] [PubMed] [Google Scholar]
- 65.Solé T, Januel L, Denneval A, Williet N, Breton C, Blanc P, et al. Time impact on the antidiabetic effects of key bariatric surgeries: a network meta-analysis of randomized controlled trials with meta-regression. Surg Obes Relat Dis. 2022;18(6):832–845. doi: 10.1016/j.soard.2022.02.003. [DOI] [PubMed] [Google Scholar]
- 66.Lin C, Yeong TJJ, Lim WH, Ng CH, Yau CE, Chin YH, et al. Comparison of mechanistic pathways of bariatric surgery in patients with diabetes mellitus: a Bayesian network meta-analysis. Obesity (Silver Spring) 2022;30(7):1380–1390. doi: 10.1002/oby.23453. [DOI] [PubMed] [Google Scholar]
- 67.Han Y, Jia Y, Wang H, Cao L, Zhao Y. Comparative analysis of weight loss and resolution of comorbidities between laparoscopic sleeve gastrectomy and Roux-en-Y gastric bypass: a systematic review and meta-analysis based on 18 studies. Int J Surg. 2020;76:101–110. doi: 10.1016/j.ijsu.2020.02.035. [DOI] [PubMed] [Google Scholar]
- 68.Matar R, Monzer N, Jaruvongvanich V, Abusaleh R, Vargas EJ, Maselli DB, et al. Indications and outcomes of conversion of sleeve gastrectomy to Roux-en-Y gastric bypass: a systematic review and a meta-analysis. Obes Surg. 2021;31(9):3936–3946. doi: 10.1007/s11695-021-05463-1. [DOI] [PubMed] [Google Scholar]
- 69.Sha Y, Huang X, Ke P, Wang B, Yuan H, Yuan W, et al. Laparoscopic Roux-en-Y gastric bypass versus sleeve gastrectomy for type 2 diabetes mellitus in nonseverely obese patients: a systematic review and meta-analysis of randomized controlled trials. Obes Surg. 2020;30(5):1660–1670. doi: 10.1007/s11695-019-04378-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used in this manuscript are available upon request.