Abstract
Gut microbes and their metabolites are actively involved in the development and regulation of host immunity, which can influence disease susceptibility. Herein, we review the most recent research advancements in the gut microbiota–immune axis. We discuss in detail how the gut microbiota is a tipping point for neonatal immune development as indicated by newly uncovered phenomenon, such as maternal imprinting, in utero intestinal metabolome, and weaning reaction. We describe how the gut microbiota shapes both innate and adaptive immunity with emphasis on the metabolites short-chain fatty acids and secondary bile acids. We also comprehensively delineate how disruption in the microbiota–immune axis results in immune-mediated diseases, such as gastrointestinal infections, inflammatory bowel diseases, cardiometabolic disorders (e.g., cardiovascular diseases, diabetes, and hypertension), autoimmunity (e.g., rheumatoid arthritis), hypersensitivity (e.g., asthma and allergies), psychological disorders (e.g., anxiety), and cancer (e.g., colorectal and hepatic). We further encompass the role of fecal microbiota transplantation, probiotics, prebiotics, and dietary polyphenols in reshaping the gut microbiota and their therapeutic potential. Continuing, we examine how the gut microbiota modulates immune therapies, including immune checkpoint inhibitors, JAK inhibitors, and anti-TNF therapies. We lastly mention the current challenges in metagenomics, germ-free models, and microbiota recapitulation to a achieve fundamental understanding for how gut microbiota regulates immunity. Altogether, this review proposes improving immunotherapy efficacy from the perspective of microbiome-targeted interventions.
Keywords: gut microbiota dysbiosis, innate immune system, adaptive immune system, infection, cancer, inflammatory bowel diseases, fecal microbiota transplantation
1. Introduction
‘No man is an island’, said John Donne, to describe relations between a human being and society [1]. However, this is also true when describing human metabolism. From birth, humans, like all other animals, are colonized by microbes, especially on the skin and mucosal surfaces, such as the gastrointestinal tract (GIT). The GIT harbors a substantial collection of microorganisms known as the gut microbiota. It is a balanced composition of over 5000 species encompassed under bacteria (e.g., 99% of the intestinal microbiota is composed of Firmicutes, Bacteroidetes, Proteobacteria, and Actinobacteria), fungi (e.g., Candida), viruses (e.g., bacteriophages), and parasites (e.g., flagellates) [2,3,4,5,6,7,8]. The gut microbiota acts like a ‘superorganism’ inside the human host and aids in the assimilation of food, produces metabolites that nourish the host, protects the host from infection, maintains function and morphology of intestinal epithelial cells, and regulates host immunity [4,8,9,10,11,12]. Under healthy conditions, the gut microbiota is in a balanced state of ‘eubiosis’. However, during diseased conditions, the gut microbiota enters an imbalanced state of dysbiosis in which there is either a bloom of opportunistic pathogens or a reduction in beneficial commensals, or both. The beauty of the host–microbiota relationship lies in the fact that microbes shape every aspect of human metabolism. As such, in addition to digestive and skin disorders, the gut microbiota has the potential to influence the pathogenesis of diseases, such as obesity and asthma, and psychological disorders, such as Parkinson’s disease [13,14].
Gut microbiota crosstalk with host immunity is one of the major features for physiological stability and a mechanism for disease etiology. There are two branches of the immune system, i.e., innate and adaptive, that work together to protect the body from external and internal threats. The innate immune system is the ‘first line of defense’ and provides fast non-specific responses upon an immunological stimulus. Innate immunity involves granulocytes, natural killer cells, dendritic cells, and macrophages that engulf the pathogen and secrete cytokines and chemokines. In addition to recruitment of more innate immune cells, cytokines attract lymphocytes, i.e., B cells, which produce antibodies unique to the specific pathogenic insult, and T cells (generally categorized into helper T cells, cytotoxic T cells, and regulatory T cells (Treg cells)), both of which form the basis of adaptive immunity [15,16]. Both arms of the immune system are tightly regulated to avoid extremes of over-activation or exhaustion, for which the gut microbiota is an essential factor (summarized in Graphical Abstract). In this review, we provide an in-depth outline and discussion about how the gut microbiota as a whole, in addition to specific bacterial species and microbial-derived metabolites, regulates immune responses. We further discuss how the gut microbiota–immune axis is aberrant in prevalent chronic inflammatory diseases and how modulation of the gut microbiota could be a therapy or possible adjuvant for other current treatments.
2. Role of Gut Microbiota and Their Metabolites in Neonatal Immune System Development
The first microbial colonization in a neonate depends on the mode of delivery (C-section vs. vaginal delivery) and feeding (formula vs. maternal milk) [17,18,19]. For instance, formula feeding was found to lower the diversity of the gut microbiota and expand pathogenic bacteria, such as Enterobacteriaceae and Enterococcaceae; this gut microbiota dysbiosis contributed to greater mucosa inflammatory activity and worsened pathology in a necrotizing enterocolitis model [20]. Moreover, a possibility for C-section to disrupt mother-to-neonate transmission of specific microbial strains (e.g., LPS-expressing bacteria) was reported [21]. However, the neonatal immune system may be primed during intra-uterine development since microbes generally present in maternal gut and mouth, such as Firmicutes, Actinobacteria, and Proteobacteria, are found in the placenta, umbilical cord, and amniotic fluid [22,23]. While an in utero microbiome is still under investigation, a 2020 article by Rackaityte et al. proposes that bacterial colonization would be limited in the human intestine in utero [24]. Moreover, recent evidence for an in utero intestinal metabolome was delineated and found to be enriched with amino acids (e.g., tryptophan), vitamins (e.g., riboflavin), and, more interestingly, gut-microbiota-derived bile acids [25].
The hygiene hypothesis proposes that exposure to a plethora of microbes early in life is essential to develop a robust immune system [26]. During intra-uterine development, the fetal innate immune system is suppressed by Foxp3+ CD4+ Treg cells to prevent immune development against maternal antigens [12]. At and after birth, antigens from commensals are recognized by several pattern recognition receptors (PRRs), such as Toll-like receptors (TLRs), on intestinal epithelia, resulting in less production of antimicrobial peptides and establishment of immune tolerance [27]. Alongside these, Paneth cells produce antimicrobial peptides, such as phospholipase-2, lysozyme, and defensins, but these molecules do not act against commensals and rather protect the neonatal gut from opportunistic pathogens [22,28]. Bifidobacteria spp. is one of the major commensals that impact infantile immunity, such as T cell maturation [29]. The absence of Bifidobacteria resulted in the depletion of human milk oligosaccharide production and was associated with greater Th2/Th17 immune activation [30]. It is noteworthy that formula feeding is associated with less Bifidobacteria abundance, but the effect is transient [31]. After lactation, pups undergo a newly defined process called ‘weaning reaction’, which is a shift in the gut microbiota that occurs when the offspring transitions from breast milk to solid food [32]. Weaning reaction was found to increase bacterial and dietary metabolites, such as short-chain fatty acids (SCFA) and retinoic acid [32]. Inhibition of weaning causes a pathological imprinting for increased risk to allergic inflammation and colitis [32]. This matches other reports that the absence of early exposure to microbiota can induce immunoglobin E (IgE) over-production and hypersensitivity to a wide array of antigens, which leads to conditions such as asthma and inflammatory bowel diseases [33,34,35]. Overall, early immune system development is regulated by the gut microbiota and can have a long-lasting impact on disease susceptibility.
3. Interaction between Gut Microbiota and Host Innate Immune System
The interaction between the gut microbiota and host mucosal immune system is critical in maintaining host health because it is the first line of defense against encroaching gut microbes (summarized in Graphical Abstract). The mucosal surfaces are compartmentalized with immune responses, including a dense mucus layer, tight junction proteins, and antimicrobial proteins. Intestinal innate immune cells develop tolerance to commensal bacteria by identifying invasive pathogens and preventing their passage from the intestinal lumen to circulation [36]. After trespassing through the epithelial barrier, invasive bacteria and pathogen-associated molecular patterns (PAMPs, i.e., lipopolysaccharides/LPS) can stimulate the release of mucin by goblet cells and induce rapid reconstitution of the inner mucous layer [37]. PAMPs can also induce innate immune responses via activation of TLRs on neutrophils and macrophages [38].
Commensal bacteria can also prime dendritic cells (DCs) via their antigen presentation, which, in turn, can activate TLRs to train the innate immune system for recognition of pathogenic vs. commensal microbes [39]. Moreover, invading microbes are phagocytosed and eradicated by mucosal innate immune cells, such as DCs and macrophages in healthy conditions [40]. Of note, specific DC subsets can engulf selective bacterial species in the lamina propria at steady state [41]. It was also recently uncovered that the maturation of precursors of type 1 conventional DCs is mediated by gut-microbiota-induced tumor necrosis factor (TNF) secretion by monocytes and macrophages [42]. In addition to macrophages, neutrophils, and DCs, there are additional specialized epithelial cells, i.e., goblet cells and Paneth cells, that release various antimicrobials, such as mucins, defensins, lysozyme, secretory phospholipase A2, and cathelicidins; they serve as accessory immune cells to sustain gut innate immunity [43,44]. Innate lymphoid cells (ILCs) are another branch of the innate immune system that are mostly non-cytotoxic and secrete several effector cytokines [45]. Collectively, many innate immune cell populations maintain gut microbiota homeostasis.
In clinical illness, alterations of the enteric microenvironment promote opportunistic pathogen growth and reduce the abundance of commensal bacteria, i.e., gut microbiota dysbiosis [46], which causes imbalanced immune responses (summarized in Graphical Abstract). In a pathologic environment, neutrophils are excessively engaged into the site of inflammation or infection and can induce collateral mucosal damage via increasing pro-inflammatory cytokine secretion, matrix metalloprotease production, and pathologic immune cell activation [43,47]. Neutrophils are normally kept in a quiescent state to prevent perturbation of gut microbial ecology, which is mediated by the adapter protein downstream of kinase 3 [48]. Interestingly, induction of neutrophil extracellular traps (NETs) led to pathogen clearance and lowered inflammation [49]. Antibiotic-induced gut microbiota dysbiosis was also found to induce NETs formation, but this was associated with worsened inflammation [50], emphasizing that more investigation is needed to determine the role of intestinal NETs. Overall, an appropriate threshold or balance between the innate immune system and gut microbiota is essential to sustain homeostasis and prevent pathophysiologic outcomes.
4. Interaction between Gut Microbiota and Adaptive Immune System
The adaptive immune system in the gut mucosa comprises mainly intraepithelial lymphocytes (IELs) and lamina propria lymphocytes (LPLs) [51]. Among the IELs, γδ T cells are a distinct subset of T cells because they express the Helios transcription factor [52]. γδ T lymphocytes inhibit the mucosal dissemination of bacteria by secreting pro-inflammatory cytokines and antimicrobial proteins [53,54]. For example, γδ T cells stimulate CD4+ T cell responses, such as mucosal release of IL-22 and calprotectin [55]. Several gut bacteria species and their metabolites are noted to promote the expansion of γδ T cells, including Desulfovibrio-derived phosphatidylethanolamine and phosphatidylcholine [56]. Studies have shown that when intraepithelial γδ T cells are deficient, there is more bacterial translocation and expansion of invasive pathogens [57]. This is supported by diminished circulating γδ T cells in acutely septic patients [58,59] and reduced colonic γδ T cells in inflammatory bowel disease patients [60].
Interaction between the gut microbiota and adaptive immune system prevents bacterial translocation and infection (summarized in Graphical Abstract). This is supported by the finding that the gut adaptive immune system is suppressed in germ-free mice, and introduction of commensal bacteria can stimulate development of mucosal lymphocytes, e.g., CD4+ T cell and cytotoxic CD8+ T cells [61]. Both primary and secondary phases of cytotoxic CD8+ T cell immunity depend on CD4+ T cells, which require priming by professional antigen-presenting cells and are amplified by CD4+ T cell signaling [62]. CD8+ T cells eliminate intracellular pathogens (e.g., Salmonella), usually assisted by DC-mediated antigen presentation [63]. Salmonella enterica serovar Typhi can promote CD8+ T cells via epigenetic modification, i.e., histone methylation and acetylation [64]. Tissue resident memory CD8+ T cells are essential to protect against re-infection cases, and this can be studied through the Transient Microbiota Depletion-boosted Immunization model, which temporarily restrains microbiota-mediated colonization resistance [65]. Of note, B cells can also phagocytose pathogens, such as Salmonella and reactivate memory CD8+ T cells, via cross-presentation [66].
T helper 17 cells (Th17) also display distinct roles in both host protection and inflammatory responses. It appears that most Th17 responses are pathological, where one novel finding is that stem-like intestinal Th17 cells promote pathogenic effector T cells in extra-intestinal diseases [67]. Interestingly, Th17 cells stimulated by segmented filamentous bacteria (SFB) are non-inflammatory, whereas Th17 cells induced by Citrobacter spp. are pro-inflammatory [68]. Studies have shown that Th17 cells are absent in germ-free mice and are induced by specific microbes, such as SFB [69] and other commensal bacteria [70]. SFB-mediated IL-17 stimulation was found to be guided by cytokine (e.g., IL-6) signals [71]. The gut microbiota can also mediate Th17 responses. A study found that microbiome-dependent Th17 inflammation is regulated by α2,6-sialyl ligands, where α2,6-sialyltransferase deficiency induced mucosal Th17 responses [72]. Pathological Th17 cells can also be promoted by the Actinobacterium Eggerthella lenta through help by the cardiac glycoside reductase 2 enzyme [73] and Fusobacterium nucleatum via the short-chain fatty acid, butyrate [74].
Regulatory T cells (Treg) are another adaptive immune cell that provides immune tolerance in the GIT. Early in life, natural Treg cells are generated in the thymus via autoimmune regulator for self-tolerance [75,76], and then exposure to diet and microbiota sets in motion peripheral or inducible Treg production [32,77,78,79]. Gut microbiota can induce Treg cells by multiple mechanisms. For instance, ILCs can select for microbiota-specific RORγt+ Treg cells and prevent the expansion of Th17 cells to maintain immune tolerance in the intestine [80]. Helicobacter spp. [81] and Akkermansia muciniphila (A. muciniphila) [82] can also induce RORγt+ Treg cell-mediated immune responses. Comparatively, lowered levels of the gut-microbiota-derived metabolite propionate (a short chain fatty acid) can contribute to a pathological imbalance in the Th17/Treg cell differentiation [83,84].
The gut microbiota also performs a crucial role in regulating the production of secretory immunoglobulin A (IgA), which is primarily aimed against enteric commensals and bacterial antigens [85,86]. Secretory IgA can be produced either via T cell-dependent or T cell-independent pathways; T cell-dependent IgA production is more important in shaping gut microbiota homeostasis [87]. Early in life, IgA plasma cells have reactivity to commensal microbiota, which contributes to a balanced microbiome [88]. Additional evidence highlights antigenic imprinting that is essential for antibody response later in life [88,89]. This includes IgA secretion into breastmilk, where maternal transfer of IgA is imperative for immune development in the offspring [90]. When IgA is deficient, as shown in mice, gut commensals can easily cross the lamina propria, leading to enteric bacterial translocation [91].
5. Crosstalk between Microbial Metabolites and Immune Regulation
5.1. Short-Chain Fatty Acids
The gut microbiota has a huge metabolic capacity to convert host-derived and dietary components (lipids, carbohydrates, and proteins) into different metabolites that may be either favorable or dangerous for the host. Bacterial metabolites, such as short-chain fatty acids (SCFAs), secondary bile acids, lactic acid, and bacteriocins, have antimicrobial activities that protect against pathogenic bacteria [92,93]. SCFAs are produced by fermentation of indigestible carbohydrates by some commensals, including Faecalibacterium prausnitzii, Roseburia intestinalis, and Anaerostipes butyraticus [94]. SCFAs maintain intestinal homeostasis in normal colon by participating in intestinal repair through cellular proliferation and differentiation (Figure 1A). Acetate, mostly produced by Bifidobacteria spp., maintains gut–epithelial barrier function and regulates intestinal inflammation by activating the G-protein receptor (GPR) 43 [95]. Through GPR43 signaling, acetate promotes microbiome-reactive IgA production [96]. This relates to acetate being one of the major gut microbial metabolites to increase colonic IgA production and IgA coating on bacteria including Enterobacterales [97]. Acetate induction of IgA is essential to sustain gut microbiota in homeostasis. In pathophysiologic conditions, acetate and propionate, either alone or in combination, can effectively reduce inflammation by reducing Th1/Th17 and elevating Treg levels [98]. Likewise, acetate supplementation to dams with preeclampsia can restore fetal thymic Treg cell output [99], and acetate feeding to non-obese diabetic mice can reduce autoreactive T cells [100]. Acetate was also found to promote T cell differentiation into both effector and Treg cells, which minimized Citrobacter infection [101].
Figure 1.
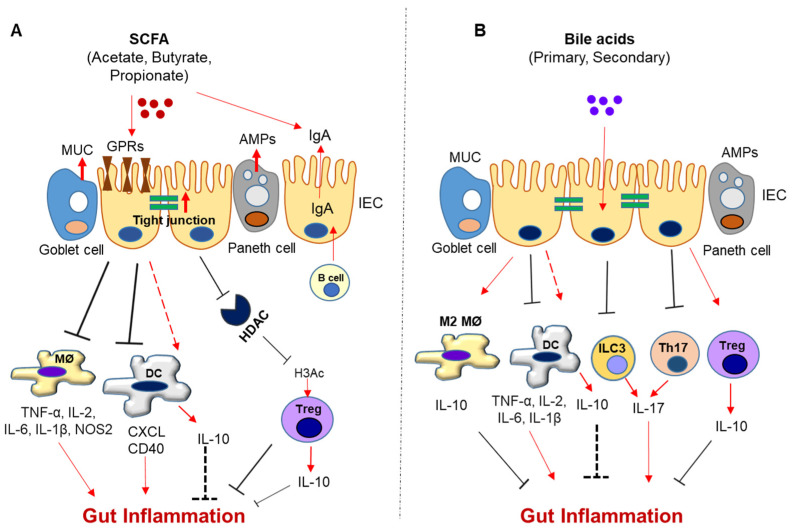
Possible mechanisms of short-chain fatty acids and bile acids positive effects on immune system in IBD. (A) Short-chain fatty acids (SCFAs) are fermented byproducts of dietary fiber. SCFAs are the ligands for G-protein receptors (GPRs) in which GPR activation upregulates mucin levels in goblet cells, antimicrobial peptides in Paneth cells, and tight junction proteins in intestinal epithelial cells. Moreover, SCFAs inhibit the secretion of pro-inflammatory cytokines (TNF-α, IL-2, IL-6, etc.) by macrophages, inhibit the expression of dendritic cell-migrated proteins (CXCL, CD40), and inhibit HDAC activity. HDAC inhibition allows for acetylation of histone 3 (H3Ac), which induces Treg differentiation and their secretion of anti-inflammatory cytokines, such as IL-10. Similarly, SCFA can promote DC-dependent anti-inflammatory IL-10 secretion. Finally, SCFAs induce IgA production from B cells. (B) Primary bile acids produced in the liver are metabolized by intestinal microbiota into secondary bile acids. Bile acids induce the polarization of macrophages and helper T cells into M2 macrophages and Treg, respectively, promoting anti-inflammatory IL-10 secretion. In addition, bile acids inhibit the secretion of pro-inflammatory cytokines (TNF-α, IL-2, IL-6, etc.) by DCs. Moreover, bile acids inhibit IL-17 secretion from ILC3 and Th17. Likewise, bile acids can promote DC-dependent IL-10 secretion. Overall, SCFA and bile acids reduce gut inflammation. SCFAs: Short-chain fatty acids, AMPs: Antimicrobial peptides, Mφ: Macrophages, DC: Dendritic cells, Tregs: T-Regulatory cells, Th1: T-helper 1, Th17: T helper 17, ILC3: Innate lymphoid cells type 3, IL: Interleukin, HDAC: Histone deacetylase, H3Ac: Acetylation of histone 3, TNF: Tumor necrosis factor, NOS2: Nitric oxide synthase 2, IgA: Immunoglobulin A, and CXCL: Chemokine (C-X-C motif) ligand. Red arrows denote activation, and black arrows denote inhibition.
Butyrate acts predominantly in intestinal homeostasis as an important energy source for colonocytes [95] and promotes release of mucin to maintain gut barrier homeostasis (Figure 1A) [102]. In addition to mucin, butyrate can promote the epithelial barrier through IL-10Rα-dependent repression of claudin-2 [103]. In regulating immune responses, butyrate can promote monocyte-to-macrophage differentiation by inhibiting histone deacetylase 3 (HDAC3) [104] and increasing the expression of IFN-γ and granzyme B in CD8+ T cells [105]. Moreover, butyrate can induce IL-22 secretion from T cells via promoting aryl hydrocarbon receptor (AhR) and hypoxia-inducible factor 1α expressions [106]. Similar to acetate, butyrate can modulate immune responses by activating GPR43 and inducing differentiation of Foxp3+ CD4+ Treg cells [100,107]. Butyrate can also promote inducible Treg production by accelerating fatty acid oxidation [108] and inhibiting HDAC [109,110]. Comparatively, the HDAC inhibitory effects of butyrate and propionate at high doses decreased class-switch DNA recombination in B cells, resulting in impairment of intestinal and systemic T-dependent and T-independent antibody responses [111]. This could explain findings from another report regarding an inverse correlation between high IgA levels and low SCFA levels that was associated with better immune tolerance [112]. Of note, in contrast to butyrate, propionate reduced IL-17 and IL-22 production by intestinal γδ T cells [113]. Overall, the main mechanisms that SCFAs maintain immune homeostasis in the intestine include HDAC inhibition, GPR signaling, inhibiting pro-inflammatory cytokine secretion, and promoting IgA production (Figure 1A).
5.2. Secondary Bile Acids
Bile acids are cholesterol-derived surfactants that primarily function to assimilate dietary lipids and fat-soluble vitamins. Primary bile acids (cholate and chenodeoxycholate) are produced in the liver and are secreted into the gallbladder conjugated to either taurine or glycine [114]. After traveling in the small intestine, 95% of bile acids are reabsorbed in the ileum, and the other 5% enter the colon. Conjugated cholate and chenodeoxycholate are then susceptible by the gut microbiota to a two-step bile salt hydrolase and dehydroxylation process that metabolizes them into the secondary bile acids deoxycholate (DCA) and lithocholate (LCA), respectively [115]. Secondary bile acids are regulated by intestinal clock-controlled bacteria, where a disruption in rhythmicity of the microbiota suppressed immune cell recruitment [116]. Moreover, gut microbiota regulation of secondary bile acids is apparent with the evidence that self-reinoculation (i.e., coprophagy) favored conjugated bile acids possibly due to the reduced total microbial load and low abundance of anaerobic microbiota [117]. Of note, oral administration of conjugated bile acids to newborn mice accelerated postnatal microbiota maturation [118].
Bile acids can drive metabolic and inflammatory mechanisms through activation of the nuclear receptor, Farnesoid X Receptor (FXR), or the G-protein coupled receptor, Takeda G protein-coupled receptor 5 (TGR5) [119]. Recent evidence implicates the role of secondary bile acids in both innate and adaptive immune responses. In terms of innate immunity, secondary bile acids, such as DCA, can activate TGR5, where this signaling inhibits monocyte-derived DC activation and NF-κB signaling [120] and promotes IL-10-dependent M2 macrophage polarization [121]. In relation to adaptive immunity, secondary bile acids were first noted to modulate gut RORγ+ Treg homeostasis, where genetic ablation of bile acid synthesis significantly depleted RORγ+ Treg cell counts [122]. Likewise, 3β-hydroxydeoxycholic acid (isoDCA) and isoDCA-producing Bacteroides consortia enhanced the peripheral generation of RORγt+ Treg cells by antagonizing FXR on DCs [123]. In addition to Treg cells, secondary bile acids, such as 3-oxoLCA and isoLCA, suppress Th17 cell function by inhibiting RORγT, a key Th17 cell-promoting transcription factor [124,125]. Unconjugated LCA also impeded Th1 activation by inhibiting ERK-1/2 phosphorylation via activation of the vitamin D receptor [126]. Furthermore, bile acid metabolism is also regulated by humoral immune responses, but dysfunction in the latter results in bile-acid-dependent small intestine enteropathy [127]. Overall, secondary bile acids modulate the gut microbiota–immune axis by lowering Th17/Treg cell differentiation, limiting pro-inflammatory cytokine secretion, and promoting M2 macrophage polarization (Figure 1B).
6. Influence of Environmental Microbiome Perturbation on the immune System
6.1. Antibiotic-Induced Microbiome Disturbances
Antibiotics have greatly improved humanity’s ability to fight infections. However, the impact of antibiotics on the microbiome was not considered until more recently. The neonatal gut microbiota and immune system can be susceptible to maternal programming when the dam microbiota is exposed to antibiotic treatment; as a result, the offspring has increased risk for developing disorders, including inflammatory bowel diseases and autoimmune diseases, and hypersensitivity, such as asthma [128,129,130,131,132,133,134]. Similarly, direct antibiotic treatment to infants, especially preterm infants, alters their microbial composition and increases susceptibility to various infections, such as necrotizing enterocolitis (NEC) [135,136,137]. It is notable that fecal microbiota transfer from NEC patients to germ-free mice demonstrated a significant reduction in butyrate and Treg levels [138]. Transient antibiotic exposure to infants can also cause microbiota-dependent suppression of type 3 ILCs, resulting in late-onset sepsis [139].
Antibiotics can have several direct and indirect negative impacts on adult human health, such as the development of antibiotic resistance for select microbial species and the loss of beneficial taxa [140]. For instance, a combined administration of meropenem, gentamicin, and vancomycin increased the abundance of pathobionts, such as Enterobacteriaceae, and diminished butyrate-producing commensals, such as Bifidobacterium [141]. Similar observations were seen when oral antibiotics lowered probiotic bacteria in the microbiota [142]. It has also been reported that ciprofloxacin rapidly decreased the richness and diversity of gut microbiota accompanied by shifts in Bacteroidetes, Lachnospiraceae, and Ruminococcaceae [143].
Exposure to antibiotics affects host immune responses, and this is linked to microbiota changes. For example, a study in mice demonstrated that antibiotic-induced alterations in the microbiota shifted the Th1/Th2 balance toward Th2-dominant immunity, which reduced lymphocytes [144]. Similar findings were found in newborn macaques after early-life antibiotic exposure that rendered the animals more susceptible to bacterial pneumonia, concurrent with neutrophil senescence, hyperinflammation, and macrophage dysfunction [145]. While changes in microbial populations after antibiotic treatment vary widely [141,146], a persistent theme appears to be the short-term (and in some cases, long-term) loss of certain keystone taxa and SCFA-producing bacteria [141,147]. As emphasized in Section 5.1, SCFAs stimulate CD4+ T cells and ILCs to produce anti-inflammatory IL-22 by several mechanisms [80], including inhibition of HDAC and stimulation of GPR41/43 [106]. SCFAs also maintain epithelial barrier function [148]. Consistent reports demonstrate that antibiotic exposure decreases SCFA levels [149,150,151]. Overall, increase of antibiotics use in both infants and adults suggests that these complications are likely to develop more acutely or more dominant in the future. Cautious use of antibiotics and continued research into the structure and function of the gut microbiota is a prerequisite to address these challenges.
6.2. Fecal Microbiota Transplantation
Fecal microbiota transplantation (FMT) is a procedure in which feces are transferred from one individual to another. The goal is to restore eubiosis by introducing beneficial commensals for reversing gut microbiota dysbiosis and restoring immune function. FMT has established itself as a widely used treatment for recurrent C. difficile infection [152]. Recent data suggest FMT may also be effective in the treatment of type I diabetes mellitus and IBD [153,154,155,156]. Ongoing research is investigating the potential of FMT in a multitude of other disorders with established links to gut microbiota dysbiosis, including cardiometabolic syndrome, autoimmune diseases, sleep apnea, depression, and schizophrenia [157,158,159,160,161]. Several mechanisms have been suggested regarding the benefits of FMT. One example involves the Gram-negative anaerobic bacterium Bacteroides fragilis (B. fragilis). B. fragilis contains an extraordinary part of the genomic DNA that has been used to produce capsular polysaccharides, which are known to be central virulence factors. Among the eight capsular polysaccharides loci of B. fragilis, there are two capsular polysaccharides that possess a zwitterionic charge motif [162]. A recent study demonstrated that B. fragilis and its metabolite polysaccharide A (one of the zwitterionic polymers) have the ability to restore dysfunctional Th1/Th2 balance in germ-free mice via TLR2-mediated activation of NF-κB [163]. It is the polysaccharide’s dual-charge structural motif that confers this ability [164,165]. Another mechanistic example for FMT includes rebalancing Th17 and Treg populations as seen in colitis patients [166]. Furthermore, restoration of SCFA levels is one other mechanism of the benefits of FMT, as shown with stroke recovery [167]. As can be expected, enteral broad-spectrum antibiotics can negate the positive effects of FMT, as seen in pre-term piglets with NEC [168]. While several beneficial effects of FMT have been mentioned, it is important to acknowledge that FMT could result in the possible transfer of pathogenic microbes present in the donor feces to the transplant patient, which can cause sepsis and other diseases [8,169].
6.3. Diet-, Probiotic-, and Prebiotic-Induced Microbiome Alterations
The gut microbiome has a wide range of metabolic activities, including metabolizing lipids, carbohydrates, and proteins. Many recent studies have focused specifically on the link between the microbiome and diet. Dietary food additives, such as emulsifying agents, ubiquitous in highly processed foods, increase host inflammation by altering the gut microbiome [170]. On the other hand, Mediterranean style diets increase the levels of SCFA-producing bacteria and minimize inflammation [171]. In addition, low-fat vegan diets improve insulin sensitivity and body composition in obese adults by changing the prevalence of Bacteroides and other gut microbes [172]. Other diets, such as a high protein diet, have limited effects on microbiota composition [173]. Below, we highlight in detail other dietary sources that can have either a negative or positive impact on the gut microbiota-immune axis.
6.3.1. High-Salt Diet
A high-salt diet (HSD) is associated with metabolic disorders, such as hypertension and obesity. Salt consumption greater than 20% of daily allowance is considered to be high salt intake. Salt, especially sodium, plays a crucial role in maintaining homeostasis. Sodium content in the blood regulates blood volume; higher salt increases blood volume, and, therefore, raises the blood pressure [174]. Apart from its direct effects on hemodynamics, high salt consumption can also alter the gut microbiota, which, in turn, aggravates metabolic disorders. The effect of HSD on gut microbial composition has been reported in several mouse models of various diseases [175,176,177,178]. A study by Hu et al. showed that chronic high salt intake led to enteric dysbiosis; particularly, the percentages of Actinobacteria, Firmicutes, and Bacteroidetes were markedly altered, and HSD caused gut leakiness, renal injury, and systolic blood pressure elevation [178]. Another recent study showed that administering HSD to mice for 3 weeks caused a significant increase in the Firmicutes/Bacteroidetes (F/B) ratio and Proteobacteria [179], both of which are classic markers of gut microbiota dysbiosis and are associated with metabolic disorders. Similarly, another study showed that HSD increased the F/B ratio and abundances of Lachnospiraceae and Ruminococcus but decreased the abundance of Lactobacillus [177]. The report by Miranda et al. further demonstrated that HSD decreases Lactobacillus spp. and butyrate production in a colitis mouse model [175]. In addition to microbiota changes, salt can affect immune responses. The main component of salt, i.e., sodium chloride (NaCl), induces pathogenic Th17 cells (IL-17-producing T helper cells) in both human and mouse naïve CD4+ T cell culture in vitro [180]. Similarly, HSD enhanced TNF-α and IL-17A in a p38-dependent manner from human lamina propria mononuclear cells [181] and stimulated intestinal Th17 responses but inhibited the function of Treg cells [182], all of which exacerbated the severity of colitis in mice. Furthermore, increased dietary salt intake upregulates Th17 cells and pro-inflammatory cytokines GM-CSF, TNF-α, and IL-2, which has made HSD an environmental risk factor for the development of autoimmune diseases [183]. Altogether, high salt intake is considered detrimental by causing negative effects on the gut microbiota and promoting pro-inflammatory mediators.
6.3.2. Dietary Polyphenols
Dietary polyphenols have also been increasingly recognized for their effects on gut microbiota. These micronutrients, including, but not limited to, flavonoids, anthocyanins, catechins and tannins, can be found in a variety of foods and beverages, such as vegetables, fruits, coffee, and tea. Though only a fraction of polyphenols is absorbed in the intestines [184], a larger unabsorbed portion remains in the gut and supports the growth of select bacterial groups [185]. For example, epigallocatechin-3-gallate (EGCG; a major catechin in green tea) promotes the growth of beneficial Bacteroides and Bifidobacterium and suppresses the bloom of pathogenic Fusobacterium, Bilophila, and Enterobacteriaceae [186]. Such microbiota-modulating effects of EGCG are noted to protect against colitis [187], high-fat diet-induced obesity [188,189,190], radiation-induced mucositis [191], and Clostridium difficile infection (CDI) [192] in mice. Though how EGCG impacts the microbiota is not well understood, several studies propose that it could be due to the bactericidal effects of EGCG, i.e., (i) generating H2O2 that damages the bacterial cell wall [193,194], (ii) inhibiting bacterial fatty acid and folate biosynthesis [195,196], and (iii) inducing oxidative stress and reactive oxidative species (ROS) formation in susceptible bacteria [197]. Beneficial effects of polyphenols, aside from EGCG, on gut microbiota were also noted and could be referred to in a review by Plamada and Vodnar [198]. Taken together, advances in this research area help to portray tea and other polyphenol-rich foods as a new subset of prebiotics.
6.3.3. Probiotics, Prebiotics, and Dietary Fiber
There is an abundance of research regarding the use of probiotics and prebiotics and studying their effects on microbiome composition. Probiotics, which often include organisms such as Lactobacillus, Bifidobacteria, and yeast, maintain integrity of the intestinal epithelial barrier by decreasing levels of LPS, protecting tight junctions, and decreasing levels of pro-inflammatory cytokines [199,200]. For a specific example, Lactobacillus johnsonii probiotic supplementation to dams stabilized both the maternal and offspring gut microbiota and protected pups from retroviral infection due to fewer Th2 immune responses [201]. Of note, it was recently shown that Peyer’s patches enhance and transmit probiotic (e.g., L. reuteri) signals to CCR6-expressing pre-germinal center-like B cells, promoting their differentiation and autocrine TGFβ-1 activation; this resulted in induction of PD-1-expressing Th1-dependent IgA, alleviation of gut microbiota dysbiosis, and protection from intestinal inflammation [202].
Prebiotics, including dietary fibers such as inulin, fructo-oligosaccharides, and galacto-oligosaccharides, selectively increase several probiotic populations, primarily Lactobacillus and Bifidobacteria. Increasing intake of dietary fiber, particularly fructans and galacto-oligosaccharides, elevated the abundance of Bifidobacterium and Lactobacillus spp. without changing the α-diversity [203]. A study has shown that when mice fed a chow diet were switched to a plant-based diet, there was a significant increase in Bacteroides and Alloprevotella and a decrease in Porphyromonadaceae and Erysipelotrichaceae [204]. Similarly, humans on a plant-based diet tend to have a higher population of Prevotella and are correlated with less susceptibility to gut disorders, such as IBD [2,205,206].
Both pro- and prebiotics increase SCFA levels, benefitting host immunity in various ways, including the inhibition of pro-inflammatory NF-κB pathways and induction of Treg cells [107,207]. The collective benefits of pro- and prebiotics explain their success in attenuating certain metabolic, allergic, and autoimmune diseases linked to gut microbiota dysbiosis [200,208,209,210,211]. However, it is important to acknowledge that probiotics only work when actively administered and have no proven long-term benefits. This relates to the limited knowledge about how long probiotic prophylaxis could stabilize the gut microbiota in preterm infants who are at greater risk for inflammatory diseases [212]. Albeit rarely, probiotic microbes themselves can cause bacterial infections and endotoxemia (Lactobacillus spp.), or negative side effects could come from a possible contamination (Mucormycetes) [8]. Similar thoughts and concerns should be applied to prebiotics as well.
7. Dysregulation of Microbiome–Immunity Interaction in Various Diseases
7.1. Gut Microbiota Dysbiosis and Immune Dysregulation
Gut epithelial cells and the mucosa serve as physical barriers against infection and endotoxemia. Gut microbiota metabolites, such as SCFA and secondary bile acids, also regulate gut permeability via immunomodulation. Of note, another gut-microbiota-derived metabolite inosine, produced by Bifidobacterium and A. muciniphila, heightens Th1 differentiation and effector function of naïve T cells [213]. Gut-microbiota-mediated immune responses are essential for preventing intestinal permeability. It is hypothesized that gut microbiota dysbiosis increases intestinal permeability from a ‘leaky gut,’ which allows opportunistic pathogens and their microbial products/toxins to invade the bloodstream and ultimately mount an inflammatory response [214,215,216]. Support for this idea comes from a number of known metabolites, such as phenolic and sulfur-containing compounds, that can harm the intestinal epithelia [217], disrupt intercellular tight junctions [218], and promote bacterial translocation [219]. These consequences, which also include immune cell dysfunction and inability to eliminate the invading pathogens, lead to inflammatory diseases [220,221]. This section of the review will discuss the microbiota-immune axis in prevalent intra- and extraintestinal diseases (Figure 2 and Table 1).
Figure 2.
Gut microbiota dysbiosis gives rise to several pathophysiological conditions. Gut microbiota dysbiosis can be induced by diet, antibiotics, and genetic factors. Gut microbiota dysbiosis can cause and sustain cancers, such as colorectal cancer and hepatocellular carcinoma, along with inflammatory diseases, autoimmune conditions, and cardiometabolic disorders. Gut microbiota dysbiosis-induced immune dysregulation is another etiological factor for disease among the many others listed, including age, sex, and medication.
Table 1.
Summary of gut microbiota–immune axis in various diseases.
| Diseases | Reference | Findings |
|---|---|---|
| Gastrointestinal Infections | Singer et al., 2019 [222] | Provide resistance against colonization and invasion by pathobiont. |
| Tovaglieri et al., 2019 [223] | Human gut microbiome metabolites induce expression of flagellin (a bacterial protein) increases EHEC motility and epithelial injury. | |
| IBD | Lee and chang, 2021 [224] | Gut microbiota dysbiosis of IBD patients is consistently marked by an overgrowth in Proteobacteria. |
| Furusawa et al., 2013 [110] | SCFA confers protection against IBD by maintaining gut barrier integrity, promoting Treg cell differentiation, and inhibiting histone deacetylases. | |
| Colorectal carcinoma |
Sepich-Poore et al., 2021 [225] | Generation of genotoxin such as Bacteroides fragilis toxin (Bft), cytolethal distending toxin (CDT), and colibactin. |
| Hale et al., 2017 [226] | Bacterial-derived secondary bile acids and hydrogen sulfide promote proinflammatory milieu that increases CRC risk. | |
| Yeoh et al., 2020 [227] | Bacteria such as F. nucleatum can adhere to colon tumors and aggravate tumorigenesis. | |
| Hepatocellular carcinoma | Lin et al., 1995 [228] | Systemic translocation of LPS promotes chronic liver injury and predisposes to HCC. |
| Singh et al., 2018 [229] | Excess butyrate production promotes HCC progression. | |
| Yoshimoto et al., 2013 [230] | Secondary bile acids promote carcinogenesis and impede anti-tumor immunosurveillance in the liver. | |
| Cardiometabolic disease | Cani et al., 2007 [231] Guasch-Ferré et al., 2017 [232] Millard et al., 2018 [233] |
LPS and other microbial ligands drive low-grade chronic inflammation and predispose to CVD. |
| Bacterial trimethylamine and its conversion to trimethylamine-N-oxide in the liver increases the risk of coronary artery disease, metabolic syndrome, stroke, and vascular inflammation. | ||
| Rheumatoid Arthritis |
Scher et al., 2013 [234] | Prevotella spp. Abundance is positively associated with new-onset rheumatoid arthritis. |
| Allergic Diseases |
Fazlollahi et al., 2018 [235] Bunyavanich et al., 2016 [236] |
Gut microbiota dysbiosis increases risk for allergic disease, e.g., food allergy and asthma. |
7.2. Gastrointestinal Infections
Depending on the context, the gut microbiota can either protect the host or increase risk of infection from exogenous pathogens. The role of the microbiome as a protective force is supported by research indicating that immature microbiomes of neonates are more susceptible to invasion by pathobionts [222]. There are several different mechanisms in which commensals can prevent colonization by pathogens and protect against infections, including competing for resources, releasing bacteriophages, and producing antimicrobial metabolites [237,238,239,240,241]. In contrast, microbiome metabolites, such as 4-methyl benzoic acid, 3,4-dimethylbenzoic acid, hexanoic acid, and heptanoic acid, have been shown to increase colonic epithelial damage, as seen by enterohemorrhagic E. coli in an organ-on-a-chip model [223]. Moreover, supernatant taken from commensal Escherichia albertii can also increase virulence of diarrheagenic E. coli species, resulting in a TLR5-mediated increase in IL-8 and an overall increased pro-inflammatory response by host intestinal cells [242].
Presence of certain commensals and changes in microbiome composition are linked to infection susceptibility by organisms such as Clostridium difficile, Salmonella typhimurium, Escherichia coli, vancomycin-resistant Enterococcus spp., and Citrobacter rodentium [238,239,241,243,244,245]. One of the best examples involves CDI, where innate immune cells are stimulated by C. difficile-toxins through the inflammasome and the TLR4, TLR5, and nucleotide-binding oligomerization domain-containing protein 1 (NOD1) signaling pathways [246,247]. Numerous pro-inflammatory cytokines (such as interleukin (IL)-12, IL-1β, IL-18, interferon gamma (IFN-γ), and tumor necrosis factor α (TNFα)) and chemokines (MIP-1a, MIP-2, and IL-8) are subsequently produced, resulting in increased mucosal permeability, mast cell degranulation, epithelial cell death, and neutrophilic infiltration [248]. Importantly, CDI is usually a result of antibiotic-mediated disruption of the gut microbiota [249]. Eradication of beneficial bacteria in the gut by certain antibiotics, particularly clindamycin, enables C. difficile to flourish [250], resulting in colitis and subsequent diarrhea [251,252]. Besides gut microbiota dysbiosis, immune cell populations, such as Th17- and IL-17-expressing cells, can promote recurrent CDI [253]. Comparatively, IL-33-activated ILCs can prevent CDI [254]. As gut microbiota depletion is a main cause for CDI, interventions that restore microbes could be of therapeutic value.
Prebiotics, such as dietary fiber and their fermented byproducts, i.e., SCFA, are possible treatments for CDI. For instance, dietary fibers, such as pectin, were able to restore gut microbiota eubiosis (denoted by increased Lachnospiraceae and decreased Enterobacteriaceae) and alleviate inflammation following C. difficile-induced colitis [255]. The butyrate producing bacterium Clostridium butyricum was similarly found to protect against CDI by increasing neutrophils, Th1, and Th17 cells in the early phase of infection; this was independent of GPR43 and GPR109a signaling [256]. As mentioned in Section 6.2, CDI can be effectively treated by FMT [152]. FMT is further supported in a prior study that showed that a Microbial Ecosystem Therapeutic, consisting of 33 bacterial strains isolated from human stool, could treat antibiotic-resistant C. difficile colitis [257]. Of note, similar observations were seen when the Microbial Ecosystem Therapeutic was applied to Salmonella typhimurium infection [258]. These findings emphasize that appropriate modulation of the gut microbiota and immune responses are imperative for preventing and fighting against infection.
7.3. Inflammatory Bowel Diseases
Inflammatory bowel diseases (IBD) develop due to defects in various factors, such as environment, gut microbes, immune system, and genetic factors. IBD involves chronic inflammation of the GIT. Crohn’s disease (CD) and ulcerative colitis (UC) are two distinct clinical conditions of IBD based on histopathological features, location of disease in the GIT, and symptoms [259]. In IBD, mucolytic bacteria and pathogenic bacteria degrade the mucosal barrier and increase the invasion of pathogens into deep intestinal tissues [224,260,261,262]. Alterations in the gut microbiota composition have been highly linked to the development and progression of IBD. IBD patients show reduced populations of Firmicutes and an expansion of Proteobacteria, Bacteroidetes, Enterobacteriaceae, and Bilophila [263,264,265]. In addition, many pro-inflammatory bacterial species are coated with IgA, as seen in IBD patients and colitis mouse models [266,267]. Gut microbes appear to play a direct role in IBD development on the basis of the evidence that germ-free mice are protected against colitis [268]. This is reinforced by the discovery that implantation of gut microbes from IBD mice to germ-free mice resulted in IBD for the latter group [268]. Likewise, dams with IBD can essentially transfer an ‘IBD microbiota’ to the offspring, for which the pups have reduced microbial diversity and fewer class-switched memory B cells and Treg cells in the colon [269]. The strong link between microbiota and IBD has moved forward metagenomic approaches to help better identify diagnostic and therapeutic targets [270].
FMT is proposed as a potential treatment, where treated UC patients were found to have an increased abundance of Faecalibaterium that corresponded with less RORγt+ Th17 cells and more Foxp3+ CD4+ Treg cells [166]. Administration of SCFAs is also thought to be a potential therapeutic for IBD patients [271]. Supporting evidence includes butyrate-mediated inhibition of pro-inflammatory neutrophil responses, i.e., NETs in colitic mice [272]. There are conflicting reports as to whether dietary fiber, the precursor for SCFA, could be a beneficial intervention for IBD patients. On one side, a specific multi-fiber mix was found to counteract intestinal inflammation via increasing IL-10 and Treg cells [273]. Opposingly, our research findings indicate a dichotomy in prebiotic fiber reactions for colitic mice, where pectin could alleviate inflammation compared with inulin, which aggravated the disease pathology [274]. Moreover, our study suggested that butyrate could be a detrimental microbial metabolite by increasing NLRP3 inflammatory signaling [274]. A probiotic cocktail, comparatively, alleviated inflammation by shifting the gut microbiota to an anti-inflammatory profile which included Akkermansia and Bifidobacterium [275]. These findings collectively indicate that more investigation is required to understand prebiotic fibers and SCFAs in IBD before implementing it in the clinics.
In addition to SCFA, secondary bile acids are implicated in IBD. DCA has been well-established to induce intestinal inflammation [276,277]. This could be due, in part, to bile-acid-mediated inhibition of Paneth cell function [278]. Yet, cholecystectomy-associated secondary bile acids, including DCA, ameliorated colitis in mice by inhibiting monocytes/macrophages recruitment [279]. Moreover, UDCA can also lower colitis severity by preventing the loss of Clostridium cluster XIVa and increasing the abundance of A. muciniphila [280]. The varying effects of bile acids could be related to their chemical structure and potential conjugated moieties. For instance, sulphated secondary bile acids may exert more pro-inflammatory effects compared with their unconjugated counterparts, as seen in IBD patients [281]. Certainly, more metabolomic profiling is necessary to understand the bile acid profile in IBD patients and determine the pro- or anti-inflammatory effects for each type of bile acid. In general, it appears that both SCFA and secondary bile acids have anti-inflammatory effects in the intestine (Figure 1A,B).
Several susceptibility genes that increase risk for IBD have been identified in recent years. Current research is focused on the idea that genetic predisposition, dysbiosis, and environmental factors, such as antibiotics, work in concert toward IBD. Nucleotide-binding oligomerization domain-containing protein 2 (NOD2, an immunological intracellular recognition protein) identifies intracellular muramyl dipeptide (MDP), an integral component of bacterial cell walls [282]. Loss of NOD2 function impairs inhibition of TLR2-mediated activation of NF-κB, resulting in an overactive Th1 response and weakened immunological tolerance to microbes [282]. Moreover, several other genes that increase susceptibility to IBD, including autophagy-related 16-like 1 (ATG16L1), caspase recruitment domain-containing protein 9 (Card9), and C-type lectin domain family 7 member A (CLEC7A), dysregulate T cell responses and create gut microbiota dysbiosis, also contributing to IBD [283,284,285]. Future studies should explore whether there are single nucleotide polymorphisms in genes related to microbial metabolite production for IBD patients.
7.4. Colorectal Carcinoma (CRC)
A growing body of literature suggests a role for microbiota in the development and progression of cancer. In scenarios where the immune system has maladaptive development, gut microbiota dysbiosis becomes a high risk, and the expansion of certain microbes can result in the production of mutagenic toxins [286]. These genotoxins include Bacteroides fragilis toxin (Bft), cytolethal distending toxin (CDT), and colibactin [225]. However, these highlight only a small number of bacterial-related toxins where more research is needed to identify and understand the carcinogenic potential with the full breadth of gut microbes [225].
Adenomatous and serrated polyps are two precancerous lesions that often progress to colorectal cancer (CRC). In patients with adenomas, several species, including Bilophila, Desulfovibrio, Mogibacterium, and the phylum Bacteroidetes, are increased in the feces, while patients with serrated polyps showed increases in the taxa Fusobacteria and class Erysipelotrichia [226]. Fusobacterium nucleatum (F. nucleatum) is characterized as an important microbe in CRC progression [287,288]. F. nucleatum promotes TLR4 signaling and E-cadherin/β-catenin signaling, ultimately leading to activation of NF-κB and reduced miR-1322 expression [289]. Regulatory micro-RNAs, such as miR-1322, can directly regulate the expression of CCL20, a cytokine that promotes CRC metastasis [287]. Other literature points to F. nucleatum adhesin A (FadA) as a key virulence factor that allows F. nucleatum to adhere, invade, and erode the colonic epithelia [227]. More recently, one study found that F. nucleatum can promote CRC by suppressing anti-tumor immunity through activation of the inhibitory receptors CEACAM1 and TIGIT1, which downregulate NK cells and T cells [290]. The F. nucleatum strain Fn7-1 was also demonstrated to aggravate CRC development by elevating Th17 responses [74]. These findings on F. nucleatum are alarming because this is a SCFA-producing bacterium [291], and SCFA have been, in general, highlighted as a potential therapeutic avenue for many inflammatory diseases. F. nucleatum predominantly produce acetate and butyrate, where it was recently suggested that F. nucleatum induces Th17 via free fatty acid receptor 2 (FFAR2), a SCFA receptor [74]. Yet, loss of FFAR2 in mice aggravated tumor bacterial load and over activated DCs, eventually promoting T cell exhaustion [292]. Moreover, butyrate from dietary fiber was found to be less metabolized in CRC cells because of the Warburg effect, allowing it to act as an HDAC inhibitor and promote acetylation of genes related to apoptosis [293]. These findings emphasize that the pathologic effects of F. nucleatum could be SCFA-independent, but further studies are needed to determine this possibility.
Another proposed mechanism in the development of CRC suggests that excessive dietary intake of sugars, proteins, and lipids could promote the growth of bile-tolerant microbes, which increase production of secondary bile acids, such as DCA and LCA, and by-products, such as hydrogen sulfide. Excessive secondary bile acids are genotoxic and may produce a pro-inflammatory environment that could promote the development of CRC [226]. In particular, DCA can stimulate intestinal carcinogenesis by activating epidermal growth factor receptor-dependent release of the metalloprotease ADAM-17 [294]. DCA also activates β-catenin signaling [295] and drives malignant transformations in Lgr5-expressing (Lgr5+) cancer stem cells [296] for CRC growth and invasiveness. However, bacteria associated with secondary bile acid production, i.e., Clostridium cluster XlVa, were significantly decreased in IBD patients, which was accompanied by reduced transformation of primary to secondary bile acids [297]. In addition to bile acids, the gut microbial metabolite folate can worsen CRC pathogenesis by triggering AhR signaling and expanding Th17 levels [298]. Similar to SCFA, more investigation is needed to discern the potential pro-tumorigenic effects of gut-microbiota-derived bile acids.
There are distinct microbiota-dependent immunological responses in CRC. In terms of innate immune responses, A. muciniphila enrichment facilitated M1 macrophage polarization in an NLRP3-dependent manner that suppressed colon tumorigenesis [299]. Likewise, intestinal adherent E. coli can increase IL-10-producing macrophages, which limits intestinal inflammation and restricts tumor formation [300]. In terms of adaptive immunity, microbial dysbiosis hyperstimulates CD8+ T cells to promote chronic inflammation and early T cell exhaustion, which contributes to colon tumor susceptibility [301]. Intestinal cancer cells can also respond to the microbiota by inducing calcineurin-dependent IL-6 secretion, which promotes tumor expression of the co-inhibitory molecules B7H3/B7H4 that diminish anti-tumor CD8+ T cells [302]. Comparatively, introduction of Helicobacter hepaticus induced T follicular helper cells that restored anti-tumor immunity in a mouse CRC model [303]. Compared with macrophages and Th17 cells, γδ T cells and resident memory T cells were found at lower frequencies in the colonic tissue of CRC patients [60]. It would be interesting to investigate whether an immune cell panel could be developed for early diagnosis of CRC.
7.5. Hepatocellular Carcinoma (HCC)
Hepatocellular carcinoma (HCC), the most common primary liver cancer, is the fourth leading cause of cancer-related mortality worldwide [304]. The main etiology for HCC pathogenesis comes from pre-existing liver diseases, such as nonalcoholic fatty liver disease (NAFLD) and steatohepatitis, that lead to cirrhosis [305]. This is further complicated by other concomitants in NAFLD patients, including insulin resistance, obesity, and metabolic disorders that further promote hepatic inflammation and tumorigenesis through IL-6 and TNF-α [306]. The liver is the ‘first stop’ for venous blood coming from the intestines, making it vulnerable to the gut microbiota via microbial translocation across the intestinal–epithelial barrier or contact with absorbed microbial metabolites [307]. The aforementioned well-known effects of gut microbiota dysbiosis, including disruption of gut barrier, translocation of microbes into the bloodstream, and subsequent inflammatory immune responses via induction of PRRs by PAMPs, such as LPS, are strongly correlated to the pathogenesis of NAFLD, liver cirrhosis, and HCC [228,307]. While it has long been thought that gut microbiota dysbiosis precedes the development of HCC, this causal relationship has not been explored in depth until more recently. Behary, Raposo et al. recently found, before HCC progression, that gut microbiota dysbiosis is in tandem with early onset liver injury that is followed by an LPS-dependent Th1- and Th17-mediated cytokine response [308]. Further investigation should determine whether gut microbiota dysbiosis is a cause or consequence in the liver injury preceding HCC.
Increased Enterobacteriaceae and Streptococcus and reduction in Akkermansia, alongside elevated levels of inflammatory mediators, such as CCL3, CCL4, CCL5, IL-8, and IL-13, have been noted in patients with NAFLD-associated HCC [309]. A more recent study found decreased abundance of SCFA-producing bacteria and increased LPS-producing bacteria in patients with cirrhosis-induced HCC but no significant evidence of gut microbiota dysbiosis in other liver diseases, such as hepatitis C, hepatitis B, or alcoholic liver disease [310]. Broadly speaking, however, it should be noted that altered microbial populations observed among multiple studies are not consistent with each other [309,311,312,313]. Furthermore, while it is generally thought that SCFAs produced by gut microbes have several benefits for humans, it was recently discovered that inulin, a precursor of the SCFA butyrate, can promote the progression to HCC in genetically altered dysbiotic mice [229]. Other studies have focused on the impact of microbial metabolites on HCC. For instance, a high-fat diet led to gut overgrowth of Gram-positive organisms that generate secondary bile acids, i.e., DCA [230]. DCA can work in concert with lipoteichoic acid to activate TLR2 and subsequently downregulate anti-tumor immunity, creating a microenvironment favorable for the development of HCC [314,315]. Overall, it appears that gut microbiota metabolites are potentially pro-tumorigenic for the liver.
7.6. Cardiovascular Disease
Cardiovascular disease (CVD) is heavily linked to metabolic syndrome, a condition which involves a set of interrelated diseases—mainly atherosclerosis, NAFLD, hypertension, and type II diabetes mellitus (TIIDM)—that arise from chronic, low-grade inflammation [316]. Many cells with high metabolic activity, such as parenchymal cells in the liver and pancreas, adipocytes, and skeletal myocytes, participate in extensive crosstalk with immune cells. Any perturbation of the microbiome has the potential to alter host immune function and, by extension, may have the ability to cause or alter disease processes in metabolically active tissues. The recognition of LPS and other microbial PAMPs by PRRs are thought to be a key driver in this low-grade inflammatory state [231]. Trimethylamine-N-oxide (TMAO), a microbial co-metabolite, is also noted to cause low-grade inflammation through NF-κB signaling, inflammasome activation, and increased production of free radicals [317,318]. Furthermore, TMAO leads to atherosclerosis and, thus, heart disease by impairing cholesterol metabolism in macrophages and contributing to the formation of foam cells [319]. Indeed, higher serum TMAO is correlated with increased risk of atherosclerosis, coronary artery disease, stroke, and vascular inflammation [232,233], and TMAO is currently being considered as a biomarker for adverse cardiovascular events [320]. More recent research has discovered phenylacetylglutamine (PAGln) as a microbial metabolite related to CVD via adrenergic receptor activation and pro-thrombotic effects [321,322]. There are multiple potential emerging roles for PAGln in cardiovascular medicine, such as being used as a diagnostic marker or even as a predictor for responsiveness to β-blocker therapy for CVD patients [322].
7.7. Diabetes
Diabetes mellitus is a disease separated into two classes: type I diabetes mellitus (TIDM) involves autoimmune destruction of pancreatic islet cells, while type II diabetes mellitus (TIIDM) involves acquired insulin insensitivity. Though much research involving microbiota and diabetes revolves around TIIDM and obesity, it has been shown that increasing dietary SCFA consumption can lead to altered microbiota and distinct immune profiles in TIDM patients [323]. Increasing dietary SCFAs, such as butyrate and acetate, were also shown to work synergistically to confer protection against autoreactive T cell populations and TIDM in mice [100]. Comparatively, administration of Parabacteroides distasonis accelerated the development of T1DM in a mouse model, and this was because of aberrant immune responses, including elevated CD8+ T cells and decreased Foxp3+ CD4+ Treg cells [324]. Of note, dysregulated bile acid metabolism was found to be a potential predisposing factor for islet autoimmunity and type 1 diabetes [325].
The microbiome and immune systems are both heavily involved in the pathogenesis of TIIDM. Branched-chain amino acids are produced by Prevotella copri (P. copri) and Bacteroides vulgatus spp., and P. copri directly induces insulin resistance in mouse models [326,327]. Depletion of commensal A. muciniphila compromises the intestinal barrier, resulting in translocation of endotoxin into the bloodstream and subsequent activation of CCR2+ monocytes. This results in conversion of pancreatic B1a cells into 4BL cells, which release inflammatory mediators and cause reversible or irreversible insulin resistance [328]. On the other hand, microbial metabolites, such as linoleic acid and docosahexaenoic acid, have protective effects against insulin resistance and TIIDM through anti-inflammatory effects and prevention of lipotoxicity [329]. FMT has also been shown to reduce fasting blood glucose levels and decrease insulin resistance in mice with TIIDM [330]. Furthermore, some of the therapeutic effects of several anti-diabetic drugs can be due, in part, to their ability to alter the microbiota [331,332,333].
7.8. Hypertension
Several studies have observed significantly altered microbiome compositions between normotensive and hypertensive mice, though specific microbial profiles in hypertensive mice are dependent on the hypertension model used [334,335,336,337]. In the angiotensin II model of hypertension, lack of microbiota in germ-free mice protected against hypertension partly by decreasing inflammatory cell populations in the blood [338]. Yet, germ-free mice were more prone to kidney injury following an angiotensin II and high-salt diet combination regimen [339]. Furthermore, reintroduction of microbiota to hypotensive germ-free mice re-established vascular contractility [340]. Generally, the microbiota composition differs between hypertensive and normotensive animals and, interestingly, cross-fostering hypertensive pups with normotensive dams can reduce blood pressure in the former group [341]. Similar to CVD, the gut metabolite TMAO also has relevance to hypertension. A recent study discovered TMAO exacerbated vasoconstriction via ROS in angiotensin II-induced hypertensive mice [342]. In a similar manner, high-salt-induced DC activation is associated with microbial dysbiosis-mediated hypertension [343]. Comparatively, the ketone body β-hydroxybutyrate has been shown to be decreased in high-salt-fed hypertensive rats; rescuing with the β-hydroxybutyrate precursor 1,3 butanediol decreased blood pressure and kidney inflammation through prevention of the NLRP3-mediated inflammasome [344]. While HSD has been shown elsewhere to decrease Lactobacillus spp. and induce Th17 cell populations, this appears to be through a distinctly different mechanism [176].
7.9. Rheumatoid Arthritis
The pathogenesis of rheumatoid arthritis (RA), a systemic autoimmune disease characterized primarily by inflammation of joints, is becoming more understood. RA is a multifactorial disease with multiple identified alleles and environmental factors conferring increased susceptibility to the disease. A potentially important microbial genus in the development of RA is Prevotella. This was first identified in 2013 by Scher et al., which found that patients with new onset RA had significantly increased abundance of Prevotella spp., particularly Prevotella copri, compared with healthy controls [234]. However, the Prevotella population did not increase in patients with chronic RA [234]. Since then, multiple studies have found further correlations between various Prevotella species and RA [345,346,347]. However, it is unclear whether Prevotella spp. itself contributes to the pathogenesis of RA, or the immunological environment created by RA increases abundance of Prevotella in the gut.
Other notable bacterial shifts in the gut microbiota for RA patients include a bloom in Proteobacteria, Clostridium cluster XlVa, and Ruminococcus, which were correlated with less CD4+ T cells and Treg cells [348]. Using the K/BxN autoimmune arthritis model, it was found that SFB-mediated cytotoxic T lymphocyte antigen-4 (CTLA-4) reduction caused autoreactive T follicular helper cells [349,350]. The accumulation of T follicular helper cells and Th17 cells in arthritis appears to be age-dependent [351], which helps to explain why RA is found mostly in the older population. Interestingly, though, the gut microbiota seems to predominantly affect T follicular helper cells, not Th17 cells, as confirmed by antibiotic treatment of the K/BxN autoimmune arthritis model [352]. Of note, it was recently reported that collagen-induced RA in mice causes an aberrancy in circadian rhythmic patterns in the gut microbiome, resulting in reduced barrier integrity due to an alteration in circulating microbial-derived factors, such as tryptophan metabolites [353].
SCFAs, specifically butyrate, have been proposed as a therapeutic option for RA. Butyrate supplementation was found to promote Treg cells by inhibiting HDAC expression, and it downregulated pro-inflammatory cytokine genes in RA [354]. Moreover, butyrate alleviated arthritis by directly inducing the differentiation of functional follicular Treg cells in vitro by enhancing histone acetylation via HDAC inhibition [355]. Furthermore, butyrate reduced arthritis severity by increasing the levels of AhR ligands, i.e., serotonin-derived metabolite 5-hydroxyindole-3-acetic acid, where AhR activation supported regulatory B cell function [356]. In addition to SCFA, the gut-microbiota-derived metabolites LCA, DCA, isoLCA, and 3-oxoLCA were also very recently found to exhibit anti-arthritis effects. Specifically, isoLCA and 3-oxoLCA inhibited Th17 differentiation and promoted M2 macrophage polarization [357]. These effects of secondary bile acids could be synergized with Parabacteroides distasonis probiotic supplementation [357]. The newfound findings of secondary bile acids are monumental and need additional investigation.
7.10. Allergic Diseases
Allergies occur when the immune system becomes hypersensitized to nonpathogenic foreign antigens. Common hypersensitivities include allergic rhinitis, food allergy, eczema, atopic dermatitis, and asthma. Several factors responsible for the development of allergies, such as reduced microbial exposure, cesarean delivery, diet, and antibiotic use are strongly linked to changes in gut microbiome composition [358,359,360,361]. Gut microbiota dysbiosis, in turn, increases risk for allergies, particularly food allergies [235,236]. Dysbiosis induced by antibiotic use is sufficient to increase allergic symptoms, elevate intestinal inflammation, and disrupt gut mucosal tight junction in sensitized mice [362]. A high-fat diet generally has effects similar to antibiotics, causing gut microbiota dysbiosis and subsequently increasing risk for food allergies [363]. Changes in gut microbiota composition immediately after birth, when the microbiome is still establishing, appears to have a particularly large impact on the development of allergic diseases later in life [364]. Of note, the vaginal microbiota can also reflect allergy risk, where Lactobacillus-dominated vaginal microbiota clusters were related to infant serum IgE status at 1 year of age [365].
Several studies reinforce the concept that dysbiosis is heavily linked to allergic disease, especially asthma. Individuals with atopic asthma have significantly higher fecal levels of Lactobacillus and E. coli compared with healthy individuals [366]. In terms of microbiota metabolites, 12,13-diHOME (a relatively uncharacterized linoleic acid) is commonly found in neonates at high risk for asthma [367]. It was recently found that the bacterial epoxide hydrolase, which produces 12,13-diHOME, is also higher in concentration during pulmonary inflammation, and 12,13-diHOME reduced Treg cells in the lung [368,369]. Comparatively, the AhR ligand tetrachlorodibenzo-p-dioxin was able to attenuate delayed-type hypersensitivity by inducing Treg cells, suppressing Th17 cells, and reversing gut microbiota dysbiosis [370]. Likewise, individuals with higher fecal SCFAs, such as butyrate and propionate, early in life had markedly decreased risk for development of asthma and atopy [371]. Of potential therapeutic value, SCFA supplementation could modulate T cells and DCs to alleviate asthma [372]. Similarly, maternal supplementation with dietary fiber or acetate was shown to protect neonates from asthma by promoting acetylation of the Foxp3 gene [373]. Dietary fiber feeding also gave protection from food allergens via retinal dehydrogenase activity in CD103+ DCs [374]. Of note, the dietary fiber inulin was recently found to promote allergen- and helminth-induced type 2 inflammation, and this was bile-acid dependent [375]. Overall, it appears that the influence of gut microbiota on allergies is highly regulated by metabolites, but each microbial product has independent effects that can either promote or demote hypersensitivity.
7.11. Psychiatric Disorders: The Gut–Brain Axis
The aforementioned information describes the gut microbiota to influence both intra- and extraintestinal diseases. One other organ that the gut microbiota can impact is the brain where a ‘stressed gut’ is becoming more recognized as a pathologic entity in several neurological disorders. For pre-term infants with an immature gut microbiota, Klebsiella overgrowth has been found to be highly predictive for brain damage and is associated with a pro-inflammatory immunological tone [376]. Parkinson’s disease is marked by an accumulation of alpha-synuclein in the gut, and patients often suffer from a leaky gut due to microbiota dysbiosis with higher populations of Prevotellaceae [13]. These symptoms can be reversed by administering probiotics [377,378]. Recently, the idea that microbiota shapes mental health has started gaining traction. Taxonomic and metabolic signatures have been proposed as a biomarker for stratifying major depressive disorder into mild, moderate, and severe symptom categories [379]. Several studies studying differences in microbiota between those who are mentally healthy and those with mental health disorders, such as anxiety and/or depression, have suggested that microbial colonization before and after birth plays a major role later in life. For instance, maternal stress can induce abnormal neurodevelopment in the offspring, which has been marked with a significant reduction of Bifidobacterium spp. [380]. Moreover, neonates delivered by C-section, as opposed to vaginal birth, have a greater risk of developing psychosis later in life [377,381]. Impressively, early-life oxytocin treatment can minimize behavior deficits seen in C-section delivered pups [382].
A cocktail of broad-spectrum, gut-microbiota-depleting antibiotics, specifically at the postnatal and weaning stages, can cause long-lasting effects of anxiety-related behavioral outcomes into adolescence and adulthood [383]. A recent elegant study by Li et al. delineated that infant exposure to antibiotics resulted in anxiety- and depression-like behaviors and memory impairments that were concurrent with an increase inflammatory milieu; similar findings were seen following long-term antibiotic treatment at the adolescent and adult stages in mice [384]. Early-life disruption of the gut microbiota could also cause sex-specific anxiety-like behavior, where LPS treatment to Wistar rats resulted in less social interaction in males compared with the females, who had an increase in social behavior [385]. It is noteworthy that FMT from an ‘aged microbiome’ to germ-free mice decreased SCFAs, and this was associated with cognitive decline [386]. The gut microbiota–immunity–brain axis is still in its nascency and requires investigation to establish mechanisms involved in immune regulation responsible for behavioral abnormalities and neurological disorders. However, it must be emphasized to look at other microorganisms besides bacteria because mucosal fungi were found to promote social behavior through complementary Th17 immune mechanisms [387].
8. Relationship between the Gut Microbiota and Their Metabolites in Immunotherapy
Presently, frontline immunotherapy treatments include T cells (checkpoint inhibitors, costimulatory receptor agonists), T cell modification, adoptive T cell transfer, autologous cytokine-induced killer cells, chimeric antigen receptor therapy, cytokines, oncolytic viruses, and vaccines [388,389]. In recent years, immunotherapy based on the application of immune checkpoint inhibitors (ICIs), including antibodies against CTLA-4, programmed cell death protein 1 (PD-1), and programmed death ligand 1 (PD-L1), has been approved as first- or second-line treatments in a variety of tumors [390]. In particular, ICIs that target PD-1 and its ligand PD-L1 have been approved by the U.S. Food and Drug Administration (FDA) for the treatment of 10 different cancer types [391]. Recent studies suggest the gut microbiota could be a significant determinant of the response to cancer immunotherapy in some preclinical and clinical studies [392,393,394]. Matson et al. showed that Bifidobacterium longum, Collinsella aerofaciens, and Enterococcus faecium have higher abundance in patients responding to PD-1 inhibitors [395]. Several studies have found significant differences in the microbiomes of responders vs. non-responders to PD-1 inhibitors, including increases in Faecalibacterium, Ruminococcus, and Akkermansia in responders and increases in Bacteroides in non-responders [392,396,397]. In addition, anti-PD-1 treatment for liver cancer patients resulted in elevated Faecalibacterium abundance and better progression-free survival [398].
Additional studies have further shown that the composition of gut bacteria can influence the metabolism of certain immunotherapeutic drugs. The fecal transfer from PD-1-treated responding patients to germ-free mice enhanced T cell responses and improved the effectiveness of PD-1 inhibitor therapy [395]. Inosine, which is produced by Bifidobacterium pseudolongum and Akkermansia muciniphila, also promoted anti-CTLA-4 and anti-PD-L1 therapy by activating T cells [213]. Comparatively, a recent study by Coutzac et al. showed that butyrate and propionate limited the efficacy of CTLA-4 inhibitors, which was associated with a higher Treg population and lower survival [399]. Of note, a newly isolated probiotic Lactobacillus strain (L. paracasei sh2020) promoted anti-PD-1 effects in CRC tumor-bearing mice by upregulating the expression of CXCL10 in the tumors and subsequently enhancing CD8+ T cell recruitment [400]. Remarkably, these anti-tumor effects occurred even in the presence of gut microbiota dysbiosis. These preclinical and clinical pieces of evidence support continued investigation to determine the requirement for gut microbiota to provide the maximum efficacy of immunotherapies (Figure 3). This includes possibly utilizing the gut microbiota to limit negative side effects from immunotherapies, such as ICI-related cardiotoxicity. Chen et al. elegantly described PD-1/PD-L1 inhibitor to deplete the Prevotellaceae and Rikenellaceae microbiota populations, reduce butyrate levels, and promote pro-inflammatory macrophage M1 polarization via downregulation of the PPARα-CYP4 × 1 axis [401]. Of therapeutic relevance, Prevotella loescheii recolonization and butyrate supplementation alleviated PD-1/PD-L1 inhibitor-related cardiotoxicity [401]. As immune checkpoints are often heterogeneous and not persistent, which can result in lower treatment response rate, drug resistance, and adverse reactions [402,403,404], gut-microbiota-targeted therapies could be essential adjuvants (Figure 3).
Figure 3.
Modifying the abundance of gut microbiota population may influence the outcomes of immunotherapy. A healthy gut microbiome can increase the bioavailability and efficacy of drugs in the host. Dysbiosis, caused by several depicted factors, may decrease the efficacy of the therapeutic drugs, leading to poor therapeutic outcomes. Modifying gut microbiota could increase the effectiveness of certain immunotherapeutic drugs, such as anti-PD-1 antibody, anti-PD-L1 antibody, and anti-CTL4 antibody treatments. Gut microbiota can be changed by supplementation with either antibiotics, probiotics, prebiotics, secondary bile acids, short-chain fatty acids (e.g., butyrate), inosine, or fecal matter transplantation.
The currently approved and available IBD therapies are anti-TNF agents, anti-integrin agents, anti-β7 monoclonal antibody, and Janus kinase (JAK) inhibitors. JAK inhibitors (e.g., baricitinib) were successful in restoring insulin signaling and improving myosteatosis following high-fat–high-sugar feeding, but it did not reverse diet-induced alterations to the gut microbiota in mice [405]. Anti-TNF inhibitors have improved clinical outcomes in both CD and UC, but they still require more randomized clinical trials [402]. However, it is notable that FMT was recently found to be a potential alternative therapy for CD patients with prior loss of response or intolerance to anti-TNF therapy (i.e., infliximab) [406]. Impressively, the probiotic Bifidobacterium longum (B. longum) CECT 7894 promoted infliximab efficacy in a mouse colitis model by reducing the abundance of opportunistic pathogens, i.e., Enterococcus and Pseudomonas, and increasing secondary bile acids [407]. Another recent study similarly found that both anti-TNF and anti-IL-12/23 therapies altered the gut microbiota to favor microbial species capable of secondary bile acid production [408]. The elevation in secondary bile acids may be due to anti-TNF treatment promoting the bloom of Clostridia spp. as part of the restoration of intestinal microbiota [409]. Bile acids are considered to be a potential metabolic biomarker for anti-TNF therapy response [410], but more research is needed to determine whether bile acids improve immunotherapy efficacy (Figure 3). There is a hint that secondary bile acids could be beneficial when considering the evidence for UDCA treatment to prevent CRC reoccurrence by inhibiting NF-κB signaling [411,412]. Moreover, UDCA was found to synergize with anti-PD1 effects to inhibit cancer progression in tumor-bearing mice [413]. Overall, it appears that the gut microbiota could be exploited as both a biomarker and therapeutic target to improve immunotherapy response.
9. Promises, Challenges, and Risks in Immune–Microbiome Research
The interplay between the microbiota and immune systems and their impact on diseases, including IBD, autoimmune arthritis, and cancer, is incredibly complex. One layer of complexity includes the challenge of showing the exact implication of a certain single or group of bacteria in the onset of disease or general host physiology. Colonization of microbes to germ-free models is a relevant strategy to better understand the potential effects of gut microorganisms in host health and disease [414]. However, the gut microbiota is much more than just a select few species. There is a strong dynamic in the microbiome environment, where species are either mutually exclusive or competitive for resources, and many microbes depend on one another for growth [415]. Another layer of complexity is including other interacting genetic and environmental factors, such as diet, smoking, drugs, and medications (Figure 2). This includes differences in the microbiota (and potentially immune responses) between urban vs. rural areas for individuals [416]. Notwithstanding, observations seen in rodent models is not always translatable to humans. It can be generally stated that humans and other mammals live in a ‘dirtier’ environment when compared with research rodents living in specific pathogen-free environments. Therefore, the cleanliness of the environment, reflecting the hygiene hypothesis, could impact the microbiota composition and disease susceptibility. This notion is supported with the recent finding that feralized mice (animals continuously exposed to a livestock farmyard-type environment) had a more stable gut microbiota and remained resistant to mutagen- and colitis-induced neoplasia when compared with hygienically born mice [417].
Several studies focusing on microbiome–immunity research have employed 16S rRNA sequencing to characterize the microbiome, but this method has limitations in that it can successfully identify genera but cannot provide distinctions at the species level [418]. Therefore, to achieve a more inclusive study of microbiomes, it is advisable that metagenomics must be combined with other -omics approaches [419]. Most recently, metatranscriptomics and metabolomics are rapidly becoming important to microbiome studies. Metagenomics generates the taxonomical profile of the sample, metatranscriptomics obtains a functional profile, and metabolomics finalizes the depiction by determining which byproducts are released by the microbiota in the environment [419]. Though each of these -omics approaches provide valuable information by themselves, it is suggested that a more complete picture come from combined -omics. One important benefit with these -omics approaches is that the raw files can be deposited into databases and then later mined for analysis by other research groups. One limitation that can arise when applying machine learning to compare multiple databases is the unevenness in sample size [420]. Moreover, -omics results could be considered study-specific, as it can be difficult to find overlapping patterns of gut microbiota changes between research and/or clinical studies. This is because the gut microbiota (plus their metabolites) and disease susceptibility can vary in humans depending on their geographic origin [421], and even the bacterial composition in common laboratory rodents can be different among research facilities and vendors [422]. Overall, -omics are surely advancing the biomedical field to identify potential diagnostic and therapeutic targets, but there are still some limitations to overcome.
10. Conclusions
In summary, the host immune system and the gut microbiome are heavily dependent upon each other for normal function and well-being of the host (summarized in Graphical Abstract). This review covered novel findings, including how fetal immune fitness is environmentally dependent on the maternal microbiota (healthy vs. dysbiosis or stressed). New mechanistic pathways have been discussed, such as SCFA and secondary bile acids modulating gut homeostasis by inducing Treg cells and IL-10 secretion (Figure 1A,B). Throughout the review, butyrate and its precursor dietary fiber were repeatedly mentioned to influence immune responses and act as potential therapeutics for many diseases, but some evidence suggest that their clinical practice may need to be disease contextualized. Comparatively, probiotics and FMT look to be more promising to restore gut microbiota eubiosis and alleviate inflammatory diseases. Moreover, gut microbiota appears to be a relevant target to improve current immunotherapies and abate their negative side effects (Figure 3). We also discussed the current challenges in microbiome research, which is essentially rooted in genetic and environmental factors (Figure 2), that makes each individual microbiota unique among humans and when comparing species models. We posit that recent developments in multi-omics methods, including epigenomics, meta-genomics, meta-proteomics, metabolomics, culturomics, and single-cell transcriptomics, will elucidate interactions between the gut microbiome and the immune system in health and disease [423]. As such, it will be exciting to predict the ‘specific’ host immune responses on the basis of gut microbiome profiles, which will support the development of ‘personalized microbiome-targeted’ therapy for immunologic diseases.
Abbreviations
| GIT | Gastrointestinal tract |
| TLRs | Toll-like receptors |
| Treg | Foxp3+ regulatory T cells |
| Th17 | T helper 17 cells |
| PRRs | Pattern recognition receptors |
| PAMPs | Pathogen-associated molecular patterns |
| DCs | Dendritic cells |
| APCs | Antigen-presenting cells |
| SCFAs | Short-chain fatty acids |
| ILCs | Innate lymphoid cells |
| FMT | Fecal microbiota transplantation |
| IBD | Inflammatory bowel disease |
| NOD2 | Nucleotide-binding oligomerization domain-containing protein 2 |
| MDP | Muramyl dipeptide |
| TMAO | Trimethylamine-N-oxide |
| CRC | Colorectal carcinoma |
| HCC | Hepatocellular carcinoma |
| ICIs | Checkpoint inhibitors |
| CTLA-4 | Cytotoxic T lymphocyte antigen-4 |
| PD-1 | Programmed cell death protein 1 |
| PD-L1 | Programmed death ligand 1 |
| RA | Rheumatoid arthritis |
| LPS | Lipopolysaccharide |
| SFB | Segmented filamentous bacteria |
| CD | Crohn’s disease |
| UC | Ulcerative colitis |
| IEB | Intestinal–epithelial barrier |
| NAFLD | Non-alcoholic fatty liver disease |
| GPR | G-protein receptor |
| ROS | Reactive oxygen species |
| TNF | Tumor necrosis factor |
| NET | Neutrophil extracellular trap |
| NLR | NOD-like receptor |
| IEL | Intraepithelial lymphocytes |
| IL | Interleukin |
| RORγ | RAR-related orphan receptor gamma |
| Ig | Immunoglobulin |
| GPR | G-protein receptor |
| HDAC | Histone deacetylase |
| AhR | Aryl hydrocarbon receptor |
| HSD | High-salt diet |
| F/B ratio | Firmicutes/Bacteroidetes ratio |
| NaCl | Sodium chloride |
| CDI | Clostridium difficile infection |
| TGFβ | Tumor growth factor beta |
| CA | Cholic acid |
| DCA | Deoxycholic acid |
| UDCA | Ursodeoxycholic acid |
| LCA | Lithocholic acid |
| CVD | Cardiovascular diseases |
| TIDM | Type I diabetes mellitus |
| TIIDM | Type II diabetes mellitus |
Author Contributions
P.S., C.C. and M.R.K., original draft preparation; P.S. and C.C., designed the figures; R.M.G., B.S.Y. and M.V.-K., reviewed, edited, and contributed scientific prospects to the review; P.S. and C.C., visualization; P.S., supervision; P.S., funding acquisition. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
P.S. is funded by Crohn’s and Colitis Foundation (CCF) and American Heart Association (AHA) Career Development Awards, grant numbers 854385 and 855256.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Donne J. No Man Is an Island. Taurus Press; Willow Dene, UK: 1975. Illustrated by Paul Peter Piech. [Google Scholar]
- 2.Beam A., Clinger E., Hao L. Effect of Diet and Dietary Components on the Composition of the Gut Microbiota. Nutrients. 2021;13:2795. doi: 10.3390/nu13082795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shanahan F., Ghosh T.S., O′Toole P.W. The Healthy Microbiome-What Is the Definition of a Healthy Gut Microbiome? Gastroenterology. 2021;160:483–494. doi: 10.1053/j.gastro.2020.09.057. [DOI] [PubMed] [Google Scholar]
- 4.Valdes A.M., Walter J., Segal E., Spector T.D. Role of the gut microbiota in nutrition and health. BMJ. 2018;361:k2179. doi: 10.1136/bmj.k2179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bull M.J., Plummer N.T. Part 1: The Human Gut Microbiome in Health and Disease. Integr. Med. 2014;13:17–22. [PMC free article] [PubMed] [Google Scholar]
- 6.Sacks D., Baxter B., Campbell B.C., Carpenter J.S., Cognard C., Dippel D., Eesa M., Fischer U., Hausegger K., Hirsch J.A. Multisociety Consensus Quality Improvement Revised Consensus Statement for Endovascular Therapy of Acute Ischemic Stroke. Int. J. Stroke. 2018;13:612–632. doi: 10.1016/j.jvir.2017.11.026. [DOI] [PubMed] [Google Scholar]
- 7.Matijasic M., Mestrovic T., Paljetak H.C., Peric M., Baresic A., Verbanac D. Gut Microbiota beyond Bacteria-Mycobiome, Virome, Archaeome, and Eukaryotic Parasites in IBD. Int J. Mol. Sci. 2020;21:2668. doi: 10.3390/ijms21082668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Giuffre M., Campigotto M., Campisciano G., Comar M., Croce L.S. A story of liver and gut microbes: How does the intestinal flora affect liver disease? A review of the literature. Am. J. Physiol. Gastrointest. Liver Physiol. 2020;318:G889–G906. doi: 10.1152/ajpgi.00161.2019. [DOI] [PubMed] [Google Scholar]
- 9.Gomaa E.Z. Human gut microbiota/microbiome in health and diseases: A review. Antonie Van Leeuwenhoek. 2020;113:2019–2040. doi: 10.1007/s10482-020-01474-7. [DOI] [PubMed] [Google Scholar]
- 10.Mills S., Stanton C., Lane J.A., Smith G.J., Ross R.P. Precision Nutrition and the Microbiome, Part I: Current State of the Science. Nutrients. 2019;11:923. doi: 10.3390/nu11040923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Adak A., Khan M.R. An insight into gut microbiota and its functionalities. Cell. Mol. Life Sci. 2019;76:473–493. doi: 10.1007/s00018-018-2943-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gensollen T., Iyer S.S., Kasper D.L., Blumberg R.S. How colonization by microbiota in early life shapes the immune system. Science. 2016;352:539–544. doi: 10.1126/science.aad9378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Giuffre M., Moretti R., Campisciano G., da Silveira A.B.M., Monda V.M., Comar M., Di Bella S., Antonello R.M., Luzzati R., Croce L.S. You Talking to Me? Says the Enteric Nervous System (ENS) to the Microbe. How Intestinal Microbes Interact with the ENS. J. Clin. Med. 2020;9:3705. doi: 10.3390/jcm9113705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cho Y., Shore S.A. Obesity, Asthma, and the Microbiome. Physiology. 2016;31:108–116. doi: 10.1152/physiol.00045.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.McComb S., Thiriot A., Akache B., Krishnan L., Stark F. Introduction to the Immune System. Methods Mol. Biol. 2019;2024:1–24. doi: 10.1007/978-1-4939-9597-4_1. [DOI] [PubMed] [Google Scholar]
- 16.Chan L., Karimi N., Morovati S., Alizadeh K., Kakish J.E., Vanderkamp S., Fazel F., Napoleoni C., Alizadeh K., Mehrani Y., et al. The Roles of Neutrophils in Cytokine Storms. Viruses. 2021;13:2318. doi: 10.3390/v13112318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dominguez-Bello M.G., Costello E.K., Contreras M., Magris M., Hidalgo G., Fierer N., Knight R. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA. 2010;107:11971–11975. doi: 10.1073/pnas.1002601107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mitchell C.M., Mazzoni C., Hogstrom L., Bryant A., Bergerat A., Cher A., Pochan S., Herman P., Carrigan M., Sharp K., et al. Delivery Mode Affects Stability of Early Infant Gut Microbiota. Cell Rep. Med. 2020;1:100156. doi: 10.1016/j.xcrm.2020.100156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Selma-Royo M., Calatayud Arroyo M., Garcia-Mantrana I., Parra-Llorca A., Escuriet R., Martinez-Costa C., Collado M.C. Perinatal environment shapes microbiota colonization and infant growth: Impact on host response and intestinal function. Microbiome. 2020;8:167. doi: 10.1186/s40168-020-00940-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Subramanian S., Geng H., Du C., Chou P.M., Bu H.F., Wang X., Swaminathan S., Tan S.C., Ridlon J.M., De Plaen I.G., et al. Feeding mode influences dynamic gut microbiota signatures and affects susceptibility to anti-CD3 mAb-induced intestinal injury in neonatal mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2022;323:G205–G218. doi: 10.1152/ajpgi.00337.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wampach L., Heintz-Buschart A., Fritz J.V., Ramiro-Garcia J., Habier J., Herold M., Narayanasamy S., Kaysen A., Hogan A.H., Bindl L., et al. Birth mode is associated with earliest strain-conferred gut microbiome functions and immunostimulatory potential. Nat. Commun. 2018;9:5091. doi: 10.1038/s41467-018-07631-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Negi S., Hashimoto-Hill S., Alenghat T. Neonatal microbiota-epithelial interactions that impact infection. Front. Microbiol. 2022;13:955051. doi: 10.3389/fmicb.2022.955051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Senn V., Bassler D., Choudhury R., Scholkmann F., Righini-Grunder F., Vuille-Dit-Bile R.N., Restin T. Microbial Colonization from the Fetus to Early Childhood-A Comprehensive Review. Front. Cell. Infect. Microbiol. 2020;10:573735. doi: 10.3389/fcimb.2020.573735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rackaityte E., Halkias J., Fukui E.M., Mendoza V.F., Hayzelden C., Crawford E.D., Fujimura K.E., Burt T.D., Lynch S.V. Viable bacterial colonization is highly limited in the human intestine in utero. Nat. Med. 2020;26:599–607. doi: 10.1038/s41591-020-0761-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li Y., Toothaker J.M., Ben-Simon S., Ozeri L., Schweitzer R., McCourt B.T., McCourt C.C., Werner L., Snapper S.B., Shouval D.S., et al. In utero human intestine harbors unique metabolome, including bacterial metabolites. JCI Insight. 2020;5:e138751. doi: 10.1172/jci.insight.138751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jain N. The early life education of the immune system: Moms, microbes and (missed) opportunities. Gut Microbes. 2020;12:1824564. doi: 10.1080/19490976.2020.1824564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gierynska M., Szulc-Dabrowska L., Struzik J., Mielcarska M.B., Gregorczyk-Zboroch K.P. Integrity of the Intestinal Barrier: The Involvement of Epithelial Cells and Microbiota-A Mutual Relationship. Animals. 2022;12:145. doi: 10.3390/ani12020145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Westrom B., Arevalo Sureda E., Pierzynowska K., Pierzynowski S.G., Perez-Cano F.J. The Immature Gut Barrier and Its Importance in Establishing Immunity in Newborn Mammals. Front. Immunol. 2020;11:1153. doi: 10.3389/fimmu.2020.01153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rabe H., Lundell A.C., Sjoberg F., Ljung A., Strombeck A., Gio-Batta M., Maglio C., Nordstrom I., Andersson K., Nookaew I., et al. Neonatal gut colonization by Bifidobacterium is associated with higher childhood cytokine responses. Gut Microbes. 2020;12:1847628. doi: 10.1080/19490976.2020.1847628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Henrick B.M., Rodriguez L., Lakshmikanth T., Pou C., Henckel E., Arzoomand A., Olin A., Wang J., Mikes J., Tan Z., et al. Bifidobacteria-mediated immune system imprinting early in life. Cell. 2021;184:3884–3898. doi: 10.1016/j.cell.2021.05.030. [DOI] [PubMed] [Google Scholar]
- 31.Chin N., Mendez-Lagares G., Taft D.H., Laleau V., Kieu H., Narayan N.R., Roberts S.B., Mills D.A., Hartigan-O′Connor D.J., Flaherman V.J. Transient Effect of Infant Formula Supplementation on the Intestinal Microbiota. Nutrients. 2021;13:807. doi: 10.3390/nu13030807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Al Nabhani Z., Dulauroy S., Marques R., Cousu C., Al Bounny S., Dejardin F., Sparwasser T., Berard M., Cerf-Bensussan N., Eberl G. A Weaning Reaction to Microbiota Is Required for Resistance to Immunopathologies in the Adult. Immunity. 2019;50:1276–1288. doi: 10.1016/j.immuni.2019.02.014. [DOI] [PubMed] [Google Scholar]
- 33.Roubaud-Baudron C., Ruiz V.E., Swan A.M., Jr., Vallance B.A., Ozkul C., Pei Z., Li J., Battaglia T.W., Perez-Perez G.I., Blaser M.J. Long-Term Effects of Early-Life Antibiotic Exposure on Resistance to Subsequent Bacterial Infection. mBio. 2019;10:e02820-19. doi: 10.1128/mBio.02820-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cahenzli J., Koller Y., Wyss M., Geuking M.B., McCoy K.D. Intestinal microbial diversity during early-life colonization shapes long-term IgE levels. Cell Host Microbe. 2013;14:559–570. doi: 10.1016/j.chom.2013.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.El Aidy S., Hooiveld G., Tremaroli V., Backhed F., Kleerebezem M. The gut microbiota and mucosal homeostasis: Colonized at birth or at adulthood, does it matter? Gut Microbes. 2013;4:118–124. doi: 10.4161/gmic.23362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang C., Li Q., Ren J. Microbiota-Immune Interaction in the Pathogenesis of Gut-Derived Infection. Front. Immunol. 2019;10:1873. doi: 10.3389/fimmu.2019.01873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.McGuckin M.A., Linden S.K., Sutton P., Florin T.H. Mucin dynamics and enteric pathogens. Nat. Rev. Microbiol. 2011;9:265–278. doi: 10.1038/nrmicro2538. [DOI] [PubMed] [Google Scholar]
- 38.Mogensen T.H. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin. Microbiol. Rev. 2009;22:240–273. doi: 10.1128/CMR.00046-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Minarrieta L., Ghorbani P., Sparwasser T., Berod L. Metabolites: Deciphering the molecular language between DCs and their environment. Semin. Immunopathol. 2017;39:177–198. doi: 10.1007/s00281-016-0609-6. [DOI] [PubMed] [Google Scholar]
- 40.Levy M., Kolodziejczyk A.A., Thaiss C.A., Elinav E. Dysbiosis and the immune system. Nat. Rev. Immunol. 2017;17:219–232. doi: 10.1038/nri.2017.7. [DOI] [PubMed] [Google Scholar]
- 41.Morikawa M., Tsujibe S., Kiyoshima-Shibata J., Watanabe Y., Kato-Nagaoka N., Shida K., Matsumoto S. Microbiota of the Small Intestine Is Selectively Engulfed by Phagocytes of the Lamina Propria and Peyer′s Patches. PLoS ONE. 2016;11:e0163607. doi: 10.1371/journal.pone.0163607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kohler A., Delbauve S., Smout J., Torres D., Flamand V. Very early-life exposure to microbiota-induced TNF drives the maturation of neonatal pre-cDC1. Gut. 2021;70:511–521. doi: 10.1136/gutjnl-2019-319700. [DOI] [PubMed] [Google Scholar]
- 43.Johansson M.E., Hansson G.C. Immunological aspects of intestinal mucus and mucins. Nat. Rev. Immunol. 2016;16:639–649. doi: 10.1038/nri.2016.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Humayun M., Ayuso J.M., Park K.Y., Martorelli Di Genova B., Skala M.C., Kerr S.C., Knoll L.J., Beebe D.J. Innate immune cell response to host-parasite interaction in a human intestinal tissue microphysiological system. Sci. Adv. 2022;8:eabm8012. doi: 10.1126/sciadv.abm8012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Artis D., Spits H. The biology of innate lymphoid cells. Nature. 2015;517:293–301. doi: 10.1038/nature14189. [DOI] [PubMed] [Google Scholar]
- 46.Lankelma J.M., van Vught L.A., Belzer C., Schultz M.J., van der Poll T., de Vos W.M., Wiersinga W.J. Critically ill patients demonstrate large interpersonal variation in intestinal microbiota dysregulation: A pilot study. Intensive Care Med. 2017;43:59–68. doi: 10.1007/s00134-016-4613-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fournier B.M., Parkos C.A. The role of neutrophils during intestinal inflammation. Mucosal Immunol. 2012;5:354–366. doi: 10.1038/mi.2012.24. [DOI] [PubMed] [Google Scholar]
- 48.Loh J.T., Lee K.G., Lee A.P., Teo J.K.H., Lim H.L., Kim S.S., Tan A.H., Lam K.P. DOK3 maintains intestinal homeostasis by suppressing JAK2/STAT3 signaling and S100a8/9 production in neutrophils. Cell Death Dis. 2021;12:1054. doi: 10.1038/s41419-021-04357-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Seo D.H., Che X., Kim S., Kim D.H., Ma H.W., Kim J.H., Kim T.I., Kim W.H., Kim S.W., Cheon J.H. Triggering Receptor Expressed on Myeloid Cells-1 Agonist Regulates Intestinal Inflammation via Cd177(+) Neutrophils. Front. Immunol. 2021;12:650864. doi: 10.3389/fimmu.2021.650864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Vong L., Yeung C.W., Pinnell L.J., Sherman P.M. Adherent-invasive Escherichia coli Exacerbates Antibiotic-associated Intestinal Dysbiosis and Neutrophil Extracellular Trap Activation. Inflamm. Bowel Dis. 2016;22:42–54. doi: 10.1097/MIB.0000000000000591. [DOI] [PubMed] [Google Scholar]
- 51.Delano M.J., Ward P.A. The immune system′s role in sepsis progression, resolution, and long-term outcome. Immunol. Rev. 2016;274:330–353. doi: 10.1111/imr.12499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Apostolov A.K., Hamani M., Hernandez-Vargas H., Igalouzene R., Guyennon A., Fesneau O., Marie J.C., Soudja S.M. Common and Exclusive Features of Intestinal Intraepithelial gammadelta T Cells and Other gammadelta T Cell Subsets. Immunohorizons. 2022;6:515–527. doi: 10.4049/immunohorizons.2200046. [DOI] [PubMed] [Google Scholar]
- 53.Ismail A.S., Severson K.M., Vaishnava S., Behrendt C.L., Yu X., Benjamin J.L., Ruhn K.A., Hou B., DeFranco A.L., Yarovinsky F., et al. Gammadelta intraepithelial lymphocytes are essential mediators of host-microbial homeostasis at the intestinal mucosal surface. Proc. Natl. Acad. Sci. USA. 2011;108:8743–8748. doi: 10.1073/pnas.1019574108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Holtmeier W., Kabelitz D. Gammadelta T cells link innate and adaptive immune responses. Chem. Immunol. Allergy. 2005;86:151–183. doi: 10.1159/000086659. [DOI] [PubMed] [Google Scholar]
- 55.Tyler C.J., McCarthy N.E., Lindsay J.O., Stagg A.J., Moser B., Eberl M. Antigen-Presenting Human gammadelta T Cells Promote Intestinal CD4(+) T Cell Expression of IL-22 and Mucosal Release of Calprotectin. J. Immunol. 2017;198:3417–3425. doi: 10.4049/jimmunol.1700003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Li Y., Wang Y., Shi F., Zhang X., Zhang Y., Bi K., Chen X., Li L., Diao H. Phospholipid metabolites of the gut microbiota promote hypoxia-induced intestinal injury via CD1d-dependent gammadelta T cells. Gut Microbes. 2022;14:2096994. doi: 10.1080/19490976.2022.2096994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tomasello E., Bedoui S. Intestinal innate immune cells in gut homeostasis and immunosurveillance. Immunol. Cell Biol. 2013;91:201–203. doi: 10.1038/icb.2012.85. [DOI] [PubMed] [Google Scholar]
- 58.Grimaldi D., Le Bourhis L., Sauneuf B., Dechartres A., Rousseau C., Ouaaz F., Milder M., Louis D., Chiche J.D., Mira J.P., et al. Specific MAIT cell behaviour among innate-like T lymphocytes in critically ill patients with severe infections. Intensive Care Med. 2014;40:192–201. doi: 10.1007/s00134-013-3163-x. [DOI] [PubMed] [Google Scholar]
- 59.Andreu-Ballester J.C., Tormo-Calandin C., Garcia-Ballesteros C., Perez-Griera J., Amigo V., Almela-Quilis A., Ruiz del Castillo J., Penarroja-Otero C., Ballester F. Association of gammadelta T cells with disease severity and mortality in septic patients. Clin. Vaccine Immunol. 2013;20:738–746. doi: 10.1128/CVI.00752-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Noble A., Pring E.T., Durant L., Man R., Dilke S.M., Hoyles L., James S.A., Carding S.R., Jenkins J.T., Knight S.C. Altered immunity to microbiota, B cell activation and depleted gammadelta/resident memory T cells in colorectal cancer. Cancer Immunol. Immunother. 2022;71:2619–2629. doi: 10.1007/s00262-021-03135-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Suzuki H., Jeong K.I., Itoh K., Doi K. Regional variations in the distributions of small intestinal intraepithelial lymphocytes in germ-free and specific pathogen-free mice. Exp. Mol. Pathol. 2002;72:230–235. doi: 10.1006/exmp.2002.2433. [DOI] [PubMed] [Google Scholar]
- 62.Bedoui S., Heath W.R., Mueller S.N. CD4(+) T-cell help amplifies innate signals for primary CD8(+) T-cell immunity. Immunol. Rev. 2016;272:52–64. doi: 10.1111/imr.12426. [DOI] [PubMed] [Google Scholar]
- 63.Belz G.T., Shortman K., Bevan M.J., Heath W.R. CD8alpha+ dendritic cells selectively present MHC class I-restricted noncytolytic viral and intracellular bacterial antigens in vivo. J. Immunol. 2005;175:196–200. doi: 10.4049/jimmunol.175.1.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sztein M.B., Bafford A.C., Salerno-Goncalves R. Salmonella enterica serovar Typhi exposure elicits ex vivo cell-type-specific epigenetic changes in human gut cells. Sci. Rep. 2020;10:13581. doi: 10.1038/s41598-020-70492-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Becattini S., Littmann E.R., Seok R., Amoretti L., Fontana E., Wright R., Gjonbalaj M., Leiner I.M., Plitas G., Hohl T.M., et al. Enhancing mucosal immunity by transient microbiota depletion. Nat. Commun. 2020;11:4475. doi: 10.1038/s41467-020-18248-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.De Wit J., Souwer Y., Jorritsma T., Klaasse Bos H., ten Brinke A., Neefjes J., van Ham S.M. Antigen-specific B cells reactivate an effective cytotoxic T cell response against phagocytosed Salmonella through cross-presentation. PLoS ONE. 2010;5:e13016. doi: 10.1371/journal.pone.0013016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Schnell A., Huang L., Singer M., Singaraju A., Barilla R.M., Regan B.M.L., Bollhagen A., Thakore P.I., Dionne D., Delorey T.M., et al. Stem-like intestinal Th17 cells give rise to pathogenic effector T cells during autoimmunity. Cell. 2021;184:6281–6298.e23. doi: 10.1016/j.cell.2021.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Omenetti S., Bussi C., Metidji A., Iseppon A., Lee S., Tolaini M., Li Y., Kelly G., Chakravarty P., Shoaie S., et al. The Intestine Harbors Functionally Distinct Homeostatic Tissue-Resident and Inflammatory Th17 Cells. Immunity. 2019;51:77–89. doi: 10.1016/j.immuni.2019.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ivanov I.I., Atarashi K., Manel N., Brodie E.L., Shima T., Karaoz U., Wei D., Goldfarb K.C., Santee C.A., Lynch S.V., et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell. 2009;139:485–498. doi: 10.1016/j.cell.2009.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tan T.G., Sefik E., Geva-Zatorsky N., Kua L., Naskar D., Teng F., Pasman L., Ortiz-Lopez A., Jupp R., Wu H.J., et al. Identifying species of symbiont bacteria from the human gut that, alone, can induce intestinal Th17 cells in mice. Proc. Natl. Acad. Sci. USA. 2016;113:E8141–E8150. doi: 10.1073/pnas.1617460113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sano T., Kageyama T., Fang V., Kedmi R., Martinez C.S., Talbot J., Chen A., Cabrera I., Gorshko O., Kurakake R., et al. Redundant cytokine requirement for intestinal microbiota-induced Th17 cell differentiation in draining lymph nodes. Cell Rep. 2021;36:109608. doi: 10.1016/j.celrep.2021.109608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Irons E.E., Cortes Gomez E., Andersen V.L., Lau J.T.Y. Bacterial colonization and TH17 immunity are shaped by intestinal sialylation in neonatal mice. Glycobiology. 2022;32:414–428. doi: 10.1093/glycob/cwac005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Alexander M., Ang Q.Y., Nayak R.R., Bustion A.E., Sandy M., Zhang B., Upadhyay V., Pollard K.S., Lynch S.V., Turnbaugh P.J. Human gut bacterial metabolism drives Th17 activation and colitis. Cell Host Microbe. 2022;30:17–30. doi: 10.1016/j.chom.2021.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Brennan C.A., Clay S.L., Lavoie S.L., Bae S., Lang J.K., Fonseca-Pereira D., Rosinski K.G., Ou N., Glickman J.N., Garrett W.S. Fusobacterium nucleatum drives a pro-inflammatory intestinal microenvironment through metabolite receptor-dependent modulation of IL-17 expression. Gut Microbes. 2021;13:1987780. doi: 10.1080/19490976.2021.1987780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Malchow S., Leventhal D.S., Lee V., Nishi S., Socci N.D., Savage P.A. Aire Enforces Immune Tolerance by Directing Autoreactive T Cells into the Regulatory T Cell Lineage. Immunity. 2016;44:1102–1113. doi: 10.1016/j.immuni.2016.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yang S., Fujikado N., Kolodin D., Benoist C., Mathis D. Immune tolerance. Regulatory T cells generated early in life play a distinct role in maintaining self-tolerance. Science. 2015;348:589–594. doi: 10.1126/science.aaa7017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ohnmacht C., Park J.H., Cording S., Wing J.B., Atarashi K., Obata Y., Gaboriau-Routhiau V., Marques R., Dulauroy S., Fedoseeva M., et al. Mucosal Immunology. The microbiota regulates type 2 immunity through RORgammat(+) T cells. Science. 2015;349:989–993. doi: 10.1126/science.aac4263. [DOI] [PubMed] [Google Scholar]
- 78.Ramanan D., Sefik E., Galvan-Pena S., Wu M., Yang L., Yang Z., Kostic A., Golovkina T.V., Kasper D.L., Mathis D., et al. An Immunologic Mode of Multigenerational Transmission Governs a Gut Treg Setpoint. Cell. 2020;181:1276–1290. doi: 10.1016/j.cell.2020.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sefik E., Geva-Zatorsky N., Oh S., Konnikova L., Zemmour D., McGuire A.M., Burzyn D., Ortiz-Lopez A., Lobera M., Yang J., et al. Mucosal Immunology. Individual intestinal symbionts induce a distinct population of RORgamma(+) regulatory T cells. Science. 2015;349:993–997. doi: 10.1126/science.aaa9420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lyu M., Suzuki H., Kang L., Gaspal F., Zhou W., Goc J., Zhou L., Zhou J., Zhang W., Bank J.R.I.L.C., et al. ILC3s select microbiota-specific regulatory T cells to establish tolerance in the gut. Nature. 2022;610:744–751. doi: 10.1038/s41586-022-05141-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chai J.N., Peng Y., Rengarajan S., Solomon B.D., Ai T.L., Shen Z., Perry J.S.A., Knoop K.A., Tanoue T., Narushima S., et al. Helicobacter species are potent drivers of colonic T cell responses in homeostasis and inflammation. Sci. Immunol. 2017;2:eaal5068. doi: 10.1126/sciimmunol.aal5068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Liu Y., Yang M., Tang L., Wang F., Huang S., Liu S., Lei Y., Wang S., Xie Z., Wang W., et al. TLR4 regulates RORgammat(+) regulatory T-cell responses and susceptibility to colon inflammation through interaction with Akkermansia muciniphila. Microbiome. 2022;10:98. doi: 10.1186/s40168-022-01296-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Du H.X., Yue S.Y., Niu D., Liu C., Zhang L.G., Chen J., Chen Y., Guan Y., Hua X.L., Li C., et al. Gut Microflora Modulates Th17/Treg Cell Differentiation in Experimental Autoimmune Prostatitis via the Short-Chain Fatty Acid Propionate. Front. Immunol. 2022;13:915218. doi: 10.3389/fimmu.2022.915218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Su X., Yin X., Liu Y., Yan X., Zhang S., Wang X., Lin Z., Zhou X., Gao J., Wang Z., et al. Gut Dysbiosis Contributes to the Imbalance of Treg and Th17 Cells in Graves′ Disease Patients by Propionic Acid. J. Clin. Endocrinol. Metab. 2020;105:3526–3547. doi: 10.1210/clinem/dgaa511. [DOI] [PubMed] [Google Scholar]
- 85.Abokor A.A., McDaniel G.H., Golonka R.M., Campbell C., Brahmandam S., Yeoh B.S., Joe B., Vijay-Kumar M., Saha P. Immunoglobulin A, an Active Liaison for Host-Microbiota Homeostasis. Microorganisms. 2021;9:2117. doi: 10.3390/microorganisms9102117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pabst O., Cerovic V., Hornef M. Secretory IgA in the Coordination of Establishment and Maintenance of the Microbiota. Trends Immunol. 2016;37:287–296. doi: 10.1016/j.it.2016.03.002. [DOI] [PubMed] [Google Scholar]
- 87.Sutherland D.B., Suzuki K., Fagarasan S. Fostering of advanced mutualism with gut microbiota by Immunoglobulin A. Immunol. Rev. 2016;270:20–31. doi: 10.1111/imr.12384. [DOI] [PubMed] [Google Scholar]
- 88.Vergani S., Muleta K.G., Da Silva C., Doyle A., Kristiansen T.A., Sodini S., Krausse N., Montano G., Kotarsky K., Nakawesi J., et al. A self-sustaining layer of early-life-origin B cells drives steady-state IgA responses in the adult gut. Immunity. 2022;55:1829–1842. doi: 10.1016/j.immuni.2022.08.018. [DOI] [PubMed] [Google Scholar]
- 89.New J.S., Dizon B.L.P., Fucile C.F., Rosenberg A.F., Kearney J.F., King R.G. Neonatal Exposure to Commensal-Bacteria-Derived Antigens Directs Polysaccharide-Specific B-1 B Cell Repertoire Development. Immunity. 2020;53:172–186. doi: 10.1016/j.immuni.2020.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Usami K., Niimi K., Matsuo A., Suyama Y., Sakai Y., Sato S., Fujihashi K., Kiyono H., Uchino S., Furukawa M., et al. The gut microbiota induces Peyer′s-patch-dependent secretion of maternal IgA into milk. Cell Rep. 2021;36:109655. doi: 10.1016/j.celrep.2021.109655. [DOI] [PubMed] [Google Scholar]
- 91.Mantis N.J., Rol N., Corthesy B. Secretory IgA′s complex roles in immunity and mucosal homeostasis in the gut. Mucosal Immunol. 2011;4:603–611. doi: 10.1038/mi.2011.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.De Sablet T., Chassard C., Bernalier-Donadille A., Vareille M., Gobert A.P., Martin C. Human microbiota-secreted factors inhibit shiga toxin synthesis by enterohemorrhagic Escherichia coli O157:H7. Infect. Immun. 2009;77:783–790. doi: 10.1128/IAI.01048-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Rea M.C., Clayton E., O′Connor P.M., Shanahan F., Kiely B., Ross R.P., Hill C. Antimicrobial activity of lacticin 3,147 against clinical Clostridium difficile strains. J. Med. Microbiol. 2007;56:940–946. doi: 10.1099/jmm.0.47085-0. [DOI] [PubMed] [Google Scholar]
- 94.Parada Venegas D., De la Fuente M.K., Landskron G., Gonzalez M.J., Quera R., Dijkstra G., Harmsen H.J.M., Faber K.N., Hermoso M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019;10:277. doi: 10.3389/fimmu.2019.00277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Yoo J.Y., Groer M., Dutra S.V.O., Sarkar A., McSkimming D.I. Gut Microbiota and Immune System Interactions. Microorganisms. 2020;8:1587. doi: 10.3390/microorganisms8101587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wu W., Sun M., Chen F., Cao A.T., Liu H., Zhao Y., Huang X., Xiao Y., Yao S., Zhao Q., et al. Microbiota metabolite short-chain fatty acid acetate promotes intestinal IgA response to microbiota which is mediated by GPR43. Mucosal Immunol. 2017;10:946–956. doi: 10.1038/mi.2016.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Takeuchi T., Miyauchi E., Kanaya T., Kato T., Nakanishi Y., Watanabe T., Kitami T., Taida T., Sasaki T., Negishi H., et al. Acetate differentially regulates IgA reactivity to commensal bacteria. Nature. 2021;595:560–564. doi: 10.1038/s41586-021-03727-5. [DOI] [PubMed] [Google Scholar]
- 98.Mandaliya D.K., Patel S., Seshadri S. The Combinatorial Effect of Acetate and Propionate on High-Fat Diet Induced Diabetic Inflammation or Metaflammation and T Cell Polarization. Inflammation. 2021;44:68–79. doi: 10.1007/s10753-020-01309-7. [DOI] [PubMed] [Google Scholar]
- 99.Hu M., Eviston D., Hsu P., Marino E., Chidgey A., Santner-Nanan B., Wong K., Richards J.L., Yap Y.A., Collier F., et al. Decreased maternal serum acetate and impaired fetal thymic and regulatory T cell development in preeclampsia. Nat. Commun. 2019;10:3031. doi: 10.1038/s41467-019-10703-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Marino E., Richards J.L., McLeod K.H., Stanley D., Yap Y.A., Knight J., McKenzie C., Kranich J., Oliveira A.C., Rossello F.J., et al. Gut microbial metabolites limit the frequency of autoimmune T cells and protect against type 1 diabetes. Nat. Immunol. 2017;18:552–562. doi: 10.1038/ni.3713. [DOI] [PubMed] [Google Scholar]
- 101.Park J., Kim M., Kang S.G., Jannasch A.H., Cooper B., Patterson J., Kim C.H. Short-chain fatty acids induce both effector and regulatory T cells by suppression of histone deacetylases and regulation of the mTOR-S6K pathway. Mucosal Immunol. 2015;8:80–93. doi: 10.1038/mi.2014.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Canani R.B., Costanzo M.D., Leone L., Pedata M., Meli R., Calignano A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 2011;17:1519–1528. doi: 10.3748/wjg.v17.i12.1519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Zheng L., Kelly C.J., Battista K.D., Schaefer R., Lanis J.M., Alexeev E.E., Wang R.X., Onyiah J.C., Kominsky D.J., Colgan S.P. Microbial-Derived Butyrate Promotes Epithelial Barrier Function through IL-10 Receptor-Dependent Repression of Claudin-2. J. Immunol. 2017;199:2976–2984. doi: 10.4049/jimmunol.1700105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Schulthess J., Pandey S., Capitani M., Rue-Albrecht K.C., Arnold I., Franchini F., Chomka A., Ilott N.E., Johnston D.G.W., Pires E., et al. The Short Chain Fatty Acid Butyrate Imprints an Antimicrobial Program in Macrophages. Immunity. 2019;50:432–445. doi: 10.1016/j.immuni.2018.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Luu M., Weigand K., Wedi F., Breidenbend C., Leister H., Pautz S., Adhikary T., Visekruna A. Regulation of the effector function of CD8(+) T cells by gut microbiota-derived metabolite butyrate. Sci. Rep. 2018;8:14430. doi: 10.1038/s41598-018-32860-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Yang W., Yu T., Huang X., Bilotta A.J., Xu L., Lu Y., Sun J., Pan F., Zhou J., Zhang W., et al. Intestinal microbiota-derived short-chain fatty acids regulation of immune cell IL-22 production and gut immunity. Nat. Commun. 2020;11:4457. doi: 10.1038/s41467-020-18262-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Smith P.M., Howitt M.R., Panikov N., Michaud M., Gallini C.A., Bohlooly Y.M., Glickman J.N., Garrett W.S. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science. 2013;341:569–573. doi: 10.1126/science.1241165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hao F., Tian M., Zhang X., Jin X., Jiang Y., Sun X., Wang Y., Peng P., Liu J., Xia C., et al. Butyrate enhances CPT1A activity to promote fatty acid oxidation and iTreg differentiation. Proc. Natl. Acad. Sci. USA. 2021;118:e2014681118. doi: 10.1073/pnas.2014681118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Arpaia N., Campbell C., Fan X., Dikiy S., van der Veeken J., deRoos P., Liu H., Cross J.R., Pfeffer K., Coffer P.J., et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature. 2013;504:451–455. doi: 10.1038/nature12726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Furusawa Y., Obata Y., Fukuda S., Endo T.A., Nakato G., Takahashi D., Nakanishi Y., Uetake C., Kato K., Kato T., et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature. 2013;504:446–450. doi: 10.1038/nature12721. [DOI] [PubMed] [Google Scholar]
- 111.Sanchez H.N., Moroney J.B., Gan H., Shen T., Im J.L., Li T., Taylor J.R., Zan H., Casali P. B cell-intrinsic epigenetic modulation of antibody responses by dietary fiber-derived short-chain fatty acids. Nat. Commun. 2020;11:60. doi: 10.1038/s41467-019-13603-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Rajput M., Momin T., Singh A., Banerjee S., Villasenor A., Sheldon J., Paudel P., Rajput R. Determining the association between gut microbiota and its metabolites with higher intestinal Immunoglobulin A response. Vet. Anim. Sci. 2023;19:100279. doi: 10.1016/j.vas.2022.100279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Dupraz L., Magniez A., Rolhion N., Richard M.L., Da Costa G., Touch S., Mayeur C., Planchais J., Agus A., Danne C., et al. Gut microbiota-derived short-chain fatty acids regulate IL-17 production by mouse and human intestinal gammadelta T cells. Cell Rep. 2021;36:109332. doi: 10.1016/j.celrep.2021.109332. [DOI] [PubMed] [Google Scholar]
- 114.Chiang J.Y. Bile acids: Regulation of synthesis. J. Lipid Res. 2009;50:1955–1966. doi: 10.1194/jlr.R900010-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ramirez-Perez O., Cruz-Ramon V., Chinchilla-Lopez P., Mendez-Sanchez N. The Role of the Gut Microbiota in Bile Acid Metabolism. Ann. Hepatol. 2017;16((Suppl. 1)):S21–S26. doi: 10.5604/01.3001.0010.5672. [DOI] [PubMed] [Google Scholar]
- 116.Heddes M., Altaha B., Niu Y., Reitmeier S., Kleigrewe K., Haller D., Kiessling S. The intestinal clock drives the microbiome to maintain gastrointestinal homeostasis. Nat. Commun. 2022;13:6068. doi: 10.1038/s41467-022-33609-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Bogatyrev S.R., Rolando J.C., Ismagilov R.F. Self-reinoculation with fecal flora changes microbiota density and composition leading to an altered bile-acid profile in the mouse small intestine. Microbiome. 2020;8:19. doi: 10.1186/s40168-020-0785-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Van Best N., Rolle-Kampczyk U., Schaap F.G., Basic M., Olde Damink S.W.M., Bleich A., Savelkoul P.H.M., von Bergen M., Penders J., Hornef M.W. Bile acids drive the newborn′s gut microbiota maturation. Nat. Commun. 2020;11:3692. doi: 10.1038/s41467-020-17183-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Chen M.L., Takeda K., Sundrud M.S. Emerging roles of bile acids in mucosal immunity and inflammation. Mucosal Immunol. 2019;12:851–861. doi: 10.1038/s41385-019-0162-4. [DOI] [PubMed] [Google Scholar]
- 120.Hu J., Wang C., Huang X., Yi S., Pan S., Zhang Y., Yuan G., Cao Q., Ye X., Li H. Gut microbiota-mediated secondary bile acids regulate dendritic cells to attenuate autoimmune uveitis through TGR5 signaling. Cell Rep. 2021;36:109726. doi: 10.1016/j.celrep.2021.109726. [DOI] [PubMed] [Google Scholar]
- 121.Biagioli M., Carino A., Cipriani S., Francisci D., Marchiano S., Scarpelli P., Sorcini D., Zampella A., Fiorucci S. The Bile Acid Receptor GPBAR1 Regulates the M1/M2 Phenotype of Intestinal Macrophages and Activation of GPBAR1 Rescues Mice from Murine Colitis. J. Immunol. 2017;199:718–733. doi: 10.4049/jimmunol.1700183. [DOI] [PubMed] [Google Scholar]
- 122.Song X., Sun X., Oh S.F., Wu M., Zhang Y., Zheng W., Geva-Zatorsky N., Jupp R., Mathis D., Benoist C., et al. Microbial bile acid metabolites modulate gut RORgamma(+) regulatory T cell homeostasis. Nature. 2020;577:410–415. doi: 10.1038/s41586-019-1865-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Campbell C., McKenney P.T., Konstantinovsky D., Isaeva O.I., Schizas M., Verter J., Mai C., Jin W.B., Guo C.J., Violante S., et al. Bacterial metabolism of bile acids promotes generation of peripheral regulatory T cells. Nature. 2020;581:475–479. doi: 10.1038/s41586-020-2193-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Hang S., Paik D., Yao L., Kim E., Trinath J., Lu J., Ha S., Nelson B.N., Kelly S.P., Wu L., et al. Bile acid metabolites control T(H)17 and T(reg) cell differentiation. Nature. 2019;576:143–148. doi: 10.1038/s41586-019-1785-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Paik D., Yao L., Zhang Y., Bae S., D′Agostino G.D., Zhang M., Kim E., Franzosa E.A., Avila-Pacheco J., Bisanz J.E., et al. Human gut bacteria produce Tau(Eta)17-modulating bile acid metabolites. Nature. 2022;603:907–912. doi: 10.1038/s41586-022-04480-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Pols T.W.H., Puchner T., Korkmaz H.I., Vos M., Soeters M.R., de Vries C.J.M. Lithocholic acid controls adaptive immune responses by inhibition of Th1 activation through the Vitamin D receptor. PLoS ONE. 2017;12:e0176715. doi: 10.1371/journal.pone.0176715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Mohammed A.D., Mohammed Z., Roland M.M., Chatzistamou I., Jolly A., Schoettmer L.M., Arroyo M., Kakar K., Tian Y., Patterson A., et al. Defective humoral immunity disrupts bile acid homeostasis which promotes inflammatory disease of the small bowel. Nat. Commun. 2022;13:525. doi: 10.1038/s41467-022-28126-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Yoshida S., Ide K., Takeuchi M., Kawakami K. Prenatal and early-life antibiotic use and risk of childhood asthma: A retrospective cohort study. Pediatr. Allergy Immunol. 2018;29:490–495. doi: 10.1111/pai.12902. [DOI] [PubMed] [Google Scholar]
- 129.Ahmadizar F., Vijverberg S.J.H., Arets H.G.M., de Boer A., Lang J.E., Garssen J., Kraneveld A., Maitland-van der Zee A.H. Early-life antibiotic exposure increases the risk of developing allergic symptoms later in life: A meta-analysis. Allergy. 2018;73:971–986. doi: 10.1111/all.13332. [DOI] [PubMed] [Google Scholar]
- 130.Kronman M.P., Zaoutis T.E., Haynes K., Feng R., Coffin S.E. Antibiotic exposure and IBD development among children: A population-based cohort study. Pediatrics. 2012;130:e794–e803. doi: 10.1542/peds.2011-3886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Tapiainen T., Koivusaari P., Brinkac L., Lorenzi H.A., Salo J., Renko M., Pruikkonen H., Pokka T., Li W., Nelson K., et al. Impact of intrapartum and postnatal antibiotics on the gut microbiome and emergence of antimicrobial resistance in infants. Sci. Rep. 2019;9:10635. doi: 10.1038/s41598-019-46964-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Nyangahu D.D., Lennard K.S., Brown B.P., Darby M.G., Wendoh J.M., Havyarimana E., Smith P., Butcher J., Stintzi A., Mulder N., et al. Disruption of maternal gut microbiota during gestation alters offspring microbiota and immunity. Microbiome. 2018;6:124. doi: 10.1186/s40168-018-0511-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Azad M.B., Konya T., Persaud R.R., Guttman D.S., Chari R.S., Field C.J., Sears M.R., Mandhane P.J., Turvey S.E., Subbarao P., et al. Impact of maternal intrapartum antibiotics, method of birth and breastfeeding on gut microbiota during the first year of life: A prospective cohort study. BJOG. 2016;123:983–993. doi: 10.1111/1471-0528.13601. [DOI] [PubMed] [Google Scholar]
- 134.Tormo-Badia N., Hakansson A., Vasudevan K., Molin G., Ahrne S., Cilio C.M. Antibiotic treatment of pregnant non-obese diabetic mice leads to altered gut microbiota and intestinal immunological changes in the offspring. Scand. J. Immunol. 2014;80:250–260. doi: 10.1111/sji.12205. [DOI] [PubMed] [Google Scholar]
- 135.Zou Z.H., Liu D., Li H.D., Zhu D.P., He Y., Hou T., Yu J.L. Prenatal and postnatal antibiotic exposure influences the gut microbiota of preterm infants in neonatal intensive care units. Ann. Clin. Microbiol. Antimicrob. 2018;17:9. doi: 10.1186/s12941-018-0264-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Gibson M.K., Wang B., Ahmadi S., Burnham C.A., Tarr P.I., Warner B.B., Dantas G. Developmental dynamics of the preterm infant gut microbiota and antibiotic resistome. Nat. Microbiol. 2016;1:16024. doi: 10.1038/nmicrobiol.2016.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Clark R.H., Bloom B.T., Spitzer A.R., Gerstmann D.R. Reported medication use in the neonatal intensive care unit: Data from a large national data set. Pediatrics. 2006;117:1979–1987. doi: 10.1542/peds.2005-1707. [DOI] [PubMed] [Google Scholar]
- 138.He Y., Du W., Xiao S., Zeng B., She X., Liu D., Du H., Li L., Li F., Ai Q., et al. Colonization of fecal microbiota from patients with neonatal necrotizing enterocolitis exacerbates intestinal injury in germfree mice subjected to necrotizing enterocolitis-induction protocol via alterations in butyrate and regulatory T cells. J. Transl. Med. 2021;19:510. doi: 10.1186/s12967-021-03109-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Niu X., Daniel S., Kumar D., Ding E.Y., Savani R.C., Koh A.Y., Mirpuri J. Transient neonatal antibiotic exposure increases susceptibility to late-onset sepsis driven by microbiota-dependent suppression of type 3 innate lymphoid cells. Sci. Rep. 2020;10:12974. doi: 10.1038/s41598-020-69797-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Lange K., Buerger M., Stallmach A., Bruns T. Effects of Antibiotics on Gut Microbiota. Dig. Dis. 2016;34:260–268. doi: 10.1159/000443360. [DOI] [PubMed] [Google Scholar]
- 141.Palleja A., Mikkelsen K.H., Forslund S.K., Kashani A., Allin K.H., Nielsen T., Hansen T.H., Liang S., Feng Q., Zhang C., et al. Recovery of gut microbiota of healthy adults following antibiotic exposure. Nat. Microbiol. 2018;3:1255–1265. doi: 10.1038/s41564-018-0257-9. [DOI] [PubMed] [Google Scholar]
- 142.Wei W., Li J., Liu F., Wu M., Xiong K., He Q., Zhang B., Deng Y., Li Y. Alteration of intestinal microecology by oral antibiotics promotes oral squamous cell carcinoma development. Mol. Immunol. 2022;149:94–106. doi: 10.1016/j.molimm.2022.06.013. [DOI] [PubMed] [Google Scholar]
- 143.Dethlefsen L., Relman D.A. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl. Acad. Sci. USA. 2011;108((Suppl. 1)):4554–4561. doi: 10.1073/pnas.1000087107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Oyama N., Sudo N., Sogawa H., Kubo C. Antibiotic use during infancy promotes a shift in the T(H)1/T(H)2 balance toward T(H)2-dominant immunity in mice. J. Allergy Clin. Immunol. 2001;107:153–159. doi: 10.1067/mai.2001.111142. [DOI] [PubMed] [Google Scholar]
- 145.Stevens J., Steinmeyer S., Bonfield M., Peterson L., Wang T., Gray J., Lewkowich I., Xu Y., Du Y., Guo M., et al. The balance between protective and pathogenic immune responses to pneumonia in the neonatal lung is enforced by gut microbiota. Sci. Transl. Med. 2022;14:eabl3981. doi: 10.1126/scitranslmed.abl3981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Vatanen T., Franzosa E.A., Schwager R., Tripathi S., Arthur T.D., Vehik K., Lernmark A., Hagopian W.A., Rewers M.J., She J.X., et al. The human gut microbiome in early-onset type 1 diabetes from the TEDDY study. Nature. 2018;562:589–594. doi: 10.1038/s41586-018-0620-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Reijnders D., Goossens G.H., Hermes G.D., Neis E.P., van der Beek C.M., Most J., Holst J.J., Lenaerts K., Kootte R.S., Nieuwdorp M., et al. Effects of Gut Microbiota Manipulation by Antibiotics on Host Metabolism in Obese Humans: A Randomized Double-Blind Placebo-Controlled Trial. Cell Metab. 2016;24:63–74. doi: 10.1016/j.cmet.2016.06.016. [DOI] [PubMed] [Google Scholar]
- 148.Perez-Reytor D., Puebla C., Karahanian E., Garcia K. Use of Short-Chain Fatty Acids for the Recovery of the Intestinal Epithelial Barrier Affected by Bacterial Toxins. Front. Physiol. 2021;12:650313. doi: 10.3389/fphys.2021.650313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zarrinpar A., Chaix A., Xu Z.Z., Chang M.W., Marotz C.A., Saghatelian A., Knight R., Panda S. Antibiotic-induced microbiome depletion alters metabolic homeostasis by affecting gut signaling and colonic metabolism. Nat. Commun. 2018;9:2872. doi: 10.1038/s41467-018-05336-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Vrieze A., Out C., Fuentes S., Jonker L., Reuling I., Kootte R.S., van Nood E., Holleman F., Knaapen M., Romijn J.A., et al. Impact of oral vancomycin on gut microbiota, bile acid metabolism, and insulin sensitivity. J. Hepatol. 2014;60:824–831. doi: 10.1016/j.jhep.2013.11.034. [DOI] [PubMed] [Google Scholar]
- 151.Zhao Y., Wu J., Li J.V., Zhou N.Y., Tang H., Wang Y. Gut microbiota composition modifies fecal metabolic profiles in mice. J. Proteome Res. 2013;12:2987–2999. doi: 10.1021/pr400263n. [DOI] [PubMed] [Google Scholar]
- 152.Green J.E., Davis J.A., Berk M., Hair C., Loughman A., Castle D., Athan E., Nierenberg A.A., Cryan J.F., Jacka F., et al. Efficacy and safety of fecal microbiota transplantation for the treatment of diseases other than Clostridium difficile infection: A systematic review and meta-analysis. Gut Microbes. 2020;12:1854640. doi: 10.1080/19490976.2020.1854640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.He L., Chen R., Zhang B., Zhang S., Khan B.A., Zhu D., Wu Z., Xiao C., Chen B., Chen F., et al. Fecal microbiota transplantation treatment of autoimmune-mediated type 1 diabetes mellitus. Front. Immunol. 2022;13:930872. doi: 10.3389/fimmu.2022.930872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Zhang W., Zou G., Li B., Du X., Sun Z., Sun Y., Jiang X. Fecal Microbiota Transplantation (FMT) Alleviates Experimental Colitis in Mice by Gut Microbiota Regulation. J. Microbiol. Biotechnol. 2020;30:1132–1141. doi: 10.4014/jmb.2002.02044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Costello S.P., Hughes P.A., Waters O., Bryant R.V., Vincent A.D., Blatchford P., Katsikeros R., Makanyanga J., Campaniello M.A., Mavrangelos C., et al. Effect of Fecal Microbiota Transplantation on 8-Week Remission in Patients with Ulcerative Colitis: A Randomized Clinical Trial. JAMA. 2019;321:156–164. doi: 10.1001/jama.2018.20046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Paramsothy S., Kamm M.A., Kaakoush N.O., Walsh A.J., van den Bogaerde J., Samuel D., Leong R.W.L., Connor S., Ng W., Paramsothy R., et al. Multidonor intensive faecal microbiota transplantation for active ulcerative colitis: A randomised placebo-controlled trial. Lancet. 2017;389:1218–1228. doi: 10.1016/S0140-6736(17)30182-4. [DOI] [PubMed] [Google Scholar]
- 157.Leong K.S.W., Jayasinghe T.N., Wilson B.C., Derraik J.G.B., Albert B.B., Chiavaroli V., Svirskis D.M., Beck K.L., Conlon C.A., Jiang Y., et al. Effects of Fecal Microbiome Transfer in Adolescents with Obesity: The Gut Bugs Randomized Controlled Trial. JAMA Netw. Open. 2020;3:e2030415. doi: 10.1001/jamanetworkopen.2020.30415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Badran M., Mashaqi S., Gozal D. The gut microbiome as a target for adjuvant therapy in obstructive sleep apnea. Expert Opin. Ther. Targets. 2020;24:1263–1282. doi: 10.1080/14728222.2020.1841749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Li N., Wang Q., Wang Y., Sun A., Lin Y., Jin Y., Li X. Fecal microbiota transplantation from chronic unpredictable mild stress mice donors affects anxiety-like and depression-like behavior in recipient mice via the gut microbiota-inflammation-brain axis. Stress. 2019;22:592–602. doi: 10.1080/10253890.2019.1617267. [DOI] [PubMed] [Google Scholar]
- 160.Zheng P., Zeng B., Liu M., Chen J., Pan J., Han Y., Liu Y., Cheng K., Zhou C., Wang H., et al. The gut microbiome from patients with schizophrenia modulates the glutamate-glutamine-GABA cycle and schizophrenia-relevant behaviors in mice. Sci. Adv. 2019;5:eaau8317. doi: 10.1126/sciadv.aau8317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Wang J.W., Kuo C.H., Kuo F.C., Wang Y.K., Hsu W.H., Yu F.J., Hu H.M., Hsu P.I., Wang J.Y., Wu D.C. Fecal microbiota transplantation: Review and update. J. Formos Med. Assoc. 2019;118((Suppl. 1)):S23–S31. doi: 10.1016/j.jfma.2018.08.011. [DOI] [PubMed] [Google Scholar]
- 162.Tzianabos A.O., Onderdonk A.B., Rosner B., Cisneros R.L., Kasper D.L. Structural features of polysaccharides that induce intra-abdominal abscesses. Science. 1993;262:416–419. doi: 10.1126/science.8211161. [DOI] [PubMed] [Google Scholar]
- 163.Wang Q., McLoughlin R.M., Cobb B.A., Charrel-Dennis M., Zaleski K.J., Golenbock D., Tzianabos A.O., Kasper D.L. A bacterial carbohydrate links innate and adaptive responses through Toll-like receptor 2. J. Exp. Med. 2006;203:2853–2863. doi: 10.1084/jem.20062008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Tzianabos A., Wang J.Y., Kasper D.L. Biological chemistry of immunomodulation by zwitterionic polysaccharides. Carbohydr. Res. 2003;338:2531–2538. doi: 10.1016/j.carres.2003.06.005. [DOI] [PubMed] [Google Scholar]
- 165.Tzianabos A.O., Finberg R.W., Wang Y., Chan M., Onderdonk A.B., Jennings H.J., Kasper D.L. T cells activated by zwitterionic molecules prevent abscesses induced by pathogenic bacteria. J. Biol. Chem. 2000;275:6733–6740. doi: 10.1074/jbc.275.10.6733. [DOI] [PubMed] [Google Scholar]
- 166.Huang C., Mei Q., Lou L., Huang Z., Fu Y., Fan J., Wang J., Yin N., Zheng Y., Lu Y., et al. Ulcerative Colitis in Response to Fecal Microbiota Transplantation via Modulation of Gut Microbiota and Th17/Treg Cell Balance. Cells. 2022;11:1851. doi: 10.3390/cells11111851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Lee J., d’Aigle J., Atadja L., Quaicoe V., Honarpisheh P., Ganesh B.P., Hassan A., Graf J., Petrosino J., Putluri N., et al. Gut Microbiota-Derived Short-Chain Fatty Acids Promote Poststroke Recovery in Aged Mice. Circ. Res. 2020;127:453–465. doi: 10.1161/CIRCRESAHA.119.316448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Brunse A., Offersen S.M., Mosegaard J.J., Deng L., Damborg P., Nielsen D.S., Sangild P.T., Thymann T., Nguyen D.N. Enteral broad-spectrum antibiotics antagonize the effect of fecal microbiota transplantation in preterm pigs. Gut Microbes. 2021;13:1849997. doi: 10.1080/19490976.2020.1849997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.De Leon L.M., Watson J.B., Kelly C.R. Transient flare of ulcerative colitis after fecal microbiota transplantation for recurrent Clostridium difficile infection. Clin. Gastroenterol. Hepatol. 2013;11:1036–1038. doi: 10.1016/j.cgh.2013.04.045. [DOI] [PubMed] [Google Scholar]
- 170.Viennois E., Merlin D., Gewirtz A.T., Chassaing B. Dietary Emulsifier-Induced Low-Grade Inflammation Promotes Colon Carcinogenesis. Cancer Res. 2017;77:27–40. doi: 10.1158/0008-5472.CAN-16-1359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Garcia-Mantrana I., Selma-Royo M., Alcantara C., Collado M.C. Shifts on Gut Microbiota Associated to Mediterranean Diet Adherence and Specific Dietary Intakes on General Adult Population. Front. Microbiol. 2018;9:890. doi: 10.3389/fmicb.2018.00890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Kahleova H., Rembert E., Alwarith J., Yonas W.N., Tura A., Holubkov R., Agnello M., Chutkan R., Barnard N.D. Effects of a Low-Fat Vegan Diet on Gut Microbiota in Overweight Individuals and Relationships with Body Weight, Body Composition, and Insulin Sensitivity. A Randomized Clinical Trial. Nutrients. 2020;12:2917. doi: 10.3390/nu12102917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Ford A.L., Nagulesapillai V., Piano A., Auger J., Girard S.A., Christman M., Tompkins T.A., Dahl W.J. Microbiota Stability and Gastrointestinal Tolerance in Response to a High-Protein Diet with and without a Prebiotic, Probiotic, and Synbiotic: A Randomized, Double-Blind, Placebo-Controlled Trial in Older Women. J. Acad. Nutr. Diet. 2020;120:500–516. doi: 10.1016/j.jand.2019.12.009. [DOI] [PubMed] [Google Scholar]
- 174.Agocs R., Sugar D., Szabo A.J. Is too much salt harmful? Yes. Pediatr. Nephrol. 2020;35:1777–1785. doi: 10.1007/s00467-019-04387-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Miranda P.M., De Palma G., Serkis V., Lu J., Louis-Auguste M.P., McCarville J.L., Verdu E.F., Collins S.M., Bercik P. High salt diet exacerbates colitis in mice by decreasing Lactobacillus levels and butyrate production. Microbiome. 2018;6:57. doi: 10.1186/s40168-018-0433-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Wilck N., Matus M.G., Kearney S.M., Olesen S.W., Forslund K., Bartolomaeus H., Haase S., Mahler A., Balogh A., Marko L., et al. Salt-responsive gut commensal modulates T(H)17 axis and disease. Nature. 2017;551:585–589. doi: 10.1038/nature24628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Wang C., Huang Z., Yu K., Ding R., Ye K., Dai C., Xu X., Zhou G., Li C. High-Salt Diet Has a Certain Impact on Protein Digestion and Gut Microbiota: A Sequencing and Proteome Combined Study. Front. Microbiol. 2017;8:1838. doi: 10.3389/fmicb.2017.01838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Hu J., Luo H., Wang J., Tang W., Lu J., Wu S., Xiong Z., Yang G., Chen Z., Lan T., et al. Enteric dysbiosis-linked gut barrier disruption triggers early renal injury induced by chronic high salt feeding in mice. Exp. Mol. Med. 2017;49:e370. doi: 10.1038/emm.2017.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Kumar S., Perumal N., Yadav P.K., Pandey R.P., Chang C.M., Raj V.S. Amoxicillin impact on pathophysiology induced by short term high salt diet in mice. Sci. Rep. 2022;12:19351. doi: 10.1038/s41598-022-21270-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Matthias J., Heink S., Picard F., Zeitrag J., Kolz A., Chao Y.Y., Soll D., de Almeida G.P., Glasmacher E., Jacobsen I.D., et al. Salt generates antiinflammatory Th17 cells but amplifies pathogenicity in proinflammatory cytokine microenvironments. J. Clin. Investig. 2020;130:4587–4600. doi: 10.1172/JCI137786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Monteleone I., Marafini I., Dinallo V., Di Fusco D., Troncone E., Zorzi F., Laudisi F., Monteleone G. Sodium chloride-enriched Diet Enhanced Inflammatory Cytokine Production and Exacerbated Experimental Colitis in Mice. J. Crohn’s Colitis. 2017;11:237–245. doi: 10.1093/ecco-jcc/jjw139. [DOI] [PubMed] [Google Scholar]
- 182.Wei Y., Lu C., Chen J., Cui G., Wang L., Yu T., Yang Y., Wu W., Ding Y., Li L., et al. High salt diet stimulates gut Th17 response and exacerbates TNBS-induced colitis in mice. Oncotarget. 2017;8:70–82. doi: 10.18632/oncotarget.13783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Kleinewietfeld M., Manzel A., Titze J., Kvakan H., Yosef N., Linker R.A., Muller D.N., Hafler D.A. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature. 2013;496:518–522. doi: 10.1038/nature11868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Manach C., Williamson G., Morand C., Scalbert A., Remesy C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005;81:230S–242S. doi: 10.1093/ajcn/81.1.230S. [DOI] [PubMed] [Google Scholar]
- 185.Tzounis X., Vulevic J., Kuhnle G.G., George T., Leonczak J., Gibson G.R., Kwik-Uribe C., Spencer J.P. Flavanol monomer-induced changes to the human faecal microflora. Br. J. Nutr. 2008;99:782–792. doi: 10.1017/S0007114507853384. [DOI] [PubMed] [Google Scholar]
- 186.Liu Z., de Bruijn W.J.C., Bruins M.E., Vincken J.P. Reciprocal Interactions between Epigallocatechin-3-gallate (EGCG) and Human Gut Microbiota In Vitro. J. Agric. Food Chem. 2020;68:9804–9815. doi: 10.1021/acs.jafc.0c03587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Wu Z., Huang S., Li T., Li N., Han D., Zhang B., Xu Z.Z., Zhang S., Pang J., Wang S., et al. Gut microbiota from green tea polyphenol-dosed mice improves intestinal epithelial homeostasis and ameliorates experimental colitis. Microbiome. 2021;9:184. doi: 10.1186/s40168-021-01115-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Sun X., Dey P., Bruno R.S., Zhu J. EGCG and catechin relative to green tea extract differentially modulate the gut microbial metabolome and liver metabolome to prevent obesity in mice fed a high-fat diet. J. Nutr. Biochem. 2022;109:109094. doi: 10.1016/j.jnutbio.2022.109094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Dey P., Olmstead B.D., Sasaki G.Y., Vodovotz Y., Yu Z., Bruno R.S. Epigallocatechin gallate but not catechin prevents nonalcoholic steatohepatitis in mice similar to green tea extract while differentially affecting the gut microbiota. J. Nutr. Biochem. 2020;84:108455. doi: 10.1016/j.jnutbio.2020.108455. [DOI] [PubMed] [Google Scholar]
- 190.Remely M., Ferk F., Sterneder S., Setayesh T., Roth S., Kepcija T., Noorizadeh R., Rebhan I., Greunz M., Beckmann J., et al. EGCG Prevents High Fat Diet-Induced Changes in Gut Microbiota, Decreases of DNA Strand Breaks, and Changes in Expression and DNA Methylation of Dnmt1 and MLH1 in C57BL/6J Male Mice. Oxid. Med. Cell. Longev. 2017;2017:3079148. doi: 10.1155/2017/3079148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Cai S., Xie L.W., Xu J.Y., Zhou H., Yang C., Tang L.F., Tian Y., Li M. (-)-Epigallocatechin-3-Gallate (EGCG) Modulates the Composition of the Gut Microbiota to Protect Against Radiation-Induced Intestinal Injury in Mice. Front. Oncol. 2022;12:848107. doi: 10.3389/fonc.2022.848107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Wu Z., Shen J., Xu Q., Xiang Q., Chen Y., Lv L., Zheng B., Wang Q., Wang S., Li L. Epigallocatechin-3-Gallate Improves Intestinal Gut Microbiota Homeostasis and Ameliorates Clostridioides difficile Infection. Nutrients. 2022;14:3756. doi: 10.3390/nu14183756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Liu X., Li J., Wang Y., Li T., Zhao J., Zhang C. Green tea polyphenols function as prooxidants to inhibit Pseudomonas aeruginosa and induce the expression of oxidative stress-related genes. Folia Microbiol. 2013;58:211–217. doi: 10.1007/s12223-012-0198-2. [DOI] [PubMed] [Google Scholar]
- 194.Cui Y., Oh Y.J., Lim J., Youn M., Lee I., Pak H.K., Park W., Jo W., Park S. AFM study of the differential inhibitory effects of the green tea polyphenol (-)-epigallocatechin-3-gallate (EGCG) against Gram-positive and Gram-negative bacteria. Food Microbiol. 2012;29:80–87. doi: 10.1016/j.fm.2011.08.019. [DOI] [PubMed] [Google Scholar]
- 195.Navarro-Martinez M.D., Navarro-Peran E., Cabezas-Herrera J., Ruiz-Gomez J., Garcia-Canovas F., Rodriguez-Lopez J.N. Antifolate activity of epigallocatechin gallate against Stenotrophomonas maltophilia. Antimicrob. Agents Chemother. 2005;49:2914–2920. doi: 10.1128/AAC.49.7.2914-2920.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Zhang Y.M., Rock C.O. Evaluation of epigallocatechin gallate and related plant polyphenols as inhibitors of the FabG and FabI reductases of bacterial type II fatty-acid synthase. J. Biol. Chem. 2004;279:30994–31001. doi: 10.1074/jbc.M403697200. [DOI] [PubMed] [Google Scholar]
- 197.Xiong L.G., Chen Y.J., Tong J.W., Huang J.A., Li J., Gong Y.S., Liu Z.H. Tea polyphenol epigallocatechin gallate inhibits Escherichia coli by increasing endogenous oxidative stress. Food Chem. 2017;217:196–204. doi: 10.1016/j.foodchem.2016.08.098. [DOI] [PubMed] [Google Scholar]
- 198.Plamada D., Vodnar D.C. Polyphenols-Gut Microbiota Interrelationship: A Transition to a New Generation of Prebiotics. Nutrients. 2021;14:137. doi: 10.3390/nu14010137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Cristofori F., Dargenio V.N., Dargenio C., Miniello V.L., Barone M., Francavilla R. Anti-Inflammatory and Immunomodulatory Effects of Probiotics in Gut Inflammation: A Door to the Body. Front. Immunol. 2021;12:578386. doi: 10.3389/fimmu.2021.578386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Compare D., Rocco A., Coccoli P., Angrisani D., Sgamato C., Iovine B., Salvatore U., Nardone G. Lactobacillus casei DG and its postbiotic reduce the inflammatory mucosal response: An ex-vivo organ culture model of post-infectious irritable bowel syndrome. BMC Gastroenterol. 2017;17:53. doi: 10.1186/s12876-017-0605-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Fonseca W., Malinczak C.A., Fujimura K., Li D., McCauley K., Li J., Best S.K.K., Zhu D., Rasky A.J., Johnson C.C., et al. Maternal gut microbiome regulates immunity to RSV infection in offspring. J. Exp. Med. 2021;218:e20210235. doi: 10.1084/jem.20210235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Liu H.Y., Giraud A., Seignez C., Ahl D., Guo F., Sedin J., Walden T., Oh J.H., van Pijkeren J.P., Holm L., et al. Distinct B cell subsets in Peyer′s patches convey probiotic effects by Limosilactobacillus reuteri. Microbiome. 2021;9:198. doi: 10.1186/s40168-021-01128-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.So D., Whelan K., Rossi M., Morrison M., Holtmann G., Kelly J.T., Shanahan E.R., Staudacher H.M., Campbell K.L. Dietary fiber intervention on gut microbiota composition in healthy adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2018;107:965–983. doi: 10.1093/ajcn/nqy041. [DOI] [PubMed] [Google Scholar]
- 204.Spinler J.K., Oezguen N., Runge J.K., Luna R.A., Karri V., Yang J., Hirschi K.D. Dietary impact of a plant-derived microRNA on the gut microbiome. ExRNA. 2020;2:11. doi: 10.1186/s41544-020-00053-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Yan J., Wang L., Gu Y., Hou H., Liu T., Ding Y., Cao H. Dietary Patterns and Gut Microbiota Changes in Inflammatory Bowel Disease: Current Insights and Future Challenges. Nutrients. 2022;14:4003. doi: 10.3390/nu14194003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Wu G.D., Chen J., Hoffmann C., Bittinger K., Chen Y.Y., Keilbaugh S.A., Bewtra M., Knights D., Walters W.A., Knight R., et al. Linking long-term dietary patterns with gut microbial enterotypes. Science. 2011;334:105–108. doi: 10.1126/science.1208344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Quigley E.M.M. Prebiotics and Probiotics in Digestive Health. Clin. Gastroenterol. Hepatol. 2019;17:333–344. doi: 10.1016/j.cgh.2018.09.028. [DOI] [PubMed] [Google Scholar]
- 208.Cukrowska B., Ceregra A., Maciorkowska E., Surowska B., Zegadlo-Mylik M.A., Konopka E., Trojanowska I., Zakrzewska M., Bierla J.B., Zakrzewski M., et al. The Effectiveness of Probiotic Lactobacillus rhamnosus and Lactobacillus casei Strains in Children with Atopic Dermatitis and Cow′s Milk Protein Allergy: A Multicenter, Randomized, Double Blind, Placebo Controlled Study. Nutrients. 2021;13:1169. doi: 10.3390/nu13041169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Cukrowska B., Bierla J.B., Zakrzewska M., Klukowski M., Maciorkowska E. The Relationship between the Infant Gut Microbiota and Allergy. The Role of Bifidobacterium breve and Prebiotic Oligosaccharides in the Activation of Anti-Allergic Mechanisms in Early Life. Nutrients. 2020;12:946. doi: 10.3390/nu12040946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Ho J., Nicolucci A.C., Virtanen H., Schick A., Meddings J., Reimer R.A., Huang C. Effect of Prebiotic on Microbiota, Intestinal Permeability, and Glycemic Control in Children with Type 1 Diabetes. J. Clin. Endocrinol. Metab. 2019;104:4427–4440. doi: 10.1210/jc.2019-00481. [DOI] [PubMed] [Google Scholar]
- 211.Kijmanawat A., Panburana P., Reutrakul S., Tangshewinsirikul C. Effects of probiotic supplements on insulin resistance in gestational diabetes mellitus: A double-blind randomized controlled trial. J. Diabetes Investig. 2019;10:163–170. doi: 10.1111/jdi.12863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Westaway J.A.F., Huerlimann R., Kandasamy Y., Miller C.M., Norton R., Watson D., Infante-Vilamil S., Rudd D. Exploring the long-term colonisation and persistence of probiotic-prophylaxis species on the gut microbiome of preterm infants: A pilot study. Eur. J. Pediatr. 2022;181:3389–3400. doi: 10.1007/s00431-022-04548-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Mager L.F., Burkhard R., Pett N., Cooke N.C.A., Brown K., Ramay H., Paik S., Stagg J., Groves R.A., Gallo M., et al. Microbiome-derived inosine modulates response to checkpoint inhibitor immunotherapy. Science. 2020;369:1481–1489. doi: 10.1126/science.abc3421. [DOI] [PubMed] [Google Scholar]
- 214.Ahmadi S., Razazan A., Nagpal R., Jain S., Wang B., Mishra S.P., Wang S., Justice J., Ding J., McClain D.A., et al. Metformin Reduces Aging-Related Leaky Gut and Improves Cognitive Function by Beneficially Modulating Gut Microbiome/Goblet Cell/Mucin Axis. J. Gerontol. A Biol. Sci. Med. Sci. 2020;75:e9–e21. doi: 10.1093/gerona/glaa056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Stevens B.R., Goel R., Seungbum K., Richards E.M., Holbert R.C., Pepine C.J., Raizada M.K. Increased human intestinal barrier permeability plasma biomarkers zonulin and FABP2 correlated with plasma LPS and altered gut microbiome in anxiety or depression. Gut. 2018;67:1555–1557. doi: 10.1136/gutjnl-2017-314759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Thevaranjan N., Puchta A., Schulz C., Naidoo A., Szamosi J.C., Verschoor C.P., Loukov D., Schenck L.P., Jury J., Foley K.P., et al. Age-Associated Microbial Dysbiosis Promotes Intestinal Permeability, Systemic Inflammation, and Macrophage Dysfunction. Cell Host Microbe. 2017;21:455–466. doi: 10.1016/j.chom.2017.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Hamer H.M., De Preter V., Windey K., Verbeke K. Functional analysis of colonic bacterial metabolism: Relevant to health? Am. J. Physiol. Gastrointest. Liver Physiol. 2012;302:G1–G9. doi: 10.1152/ajpgi.00048.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.McCall I.C., Betanzos A., Weber D.A., Nava P., Miller G.W., Parkos C.A. Effects of phenol on barrier function of a human intestinal epithelial cell line correlate with altered tight junction protein localization. Toxicol. Appl. Pharmacol. 2009;241:61–70. doi: 10.1016/j.taap.2009.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Cox L.M., Yamanishi S., Sohn J., Alekseyenko A.V., Leung J.M., Cho I., Kim S.G., Li H., Gao Z., Mahana D., et al. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell. 2014;158:705–721. doi: 10.1016/j.cell.2014.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Jacobs M.C., Haak B.W., Hugenholtz F., Wiersinga W.J. Gut microbiota and host defense in critical illness. Curr. Opin. Crit. Care. 2017;23:257–263. doi: 10.1097/MCC.0000000000000424. [DOI] [PubMed] [Google Scholar]
- 221.Van der Poll T., van de Veerdonk F.L., Scicluna B.P., Netea M.G. The immunopathology of sepsis and potential therapeutic targets. Nat. Rev. Immunol. 2017;17:407–420. doi: 10.1038/nri.2017.36. [DOI] [PubMed] [Google Scholar]
- 222.Singer J.R., Blosser E.G., Zindl C.L., Silberger D.J., Conlan S., Laufer V.A., DiToro D., Deming C., Kumar R., Morrow C.D., et al. Preventing dysbiosis of the neonatal mouse intestinal microbiome protects against late-onset sepsis. Nat. Med. 2019;25:1772–1782. doi: 10.1038/s41591-019-0640-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Tovaglieri A., Sontheimer-Phelps A., Geirnaert A., Prantil-Baun R., Camacho D.M., Chou D.B., Jalili-Firoozinezhad S., de Wouters T., Kasendra M., Super M., et al. Species-specific enhancement of enterohemorrhagic E. coli pathogenesis mediated by microbiome metabolites. Microbiome. 2019;7:43. doi: 10.1186/s40168-019-0650-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Lee M., Chang E.B. Inflammatory Bowel Diseases (IBD) and the Microbiome-Searching the Crime Scene for Clues. Gastroenterology. 2021;160:524–537. doi: 10.1053/j.gastro.2020.09.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Sepich-Poore G.D., Zitvogel L., Straussman R., Hasty J., Wargo J.A., Knight R. The microbiome and human cancer. Science. 2021;371:eabc4552. doi: 10.1126/science.abc4552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Hale V.L., Chen J., Johnson S., Harrington S.C., Yab T.C., Smyrk T.C., Nelson H., Boardman L.A., Druliner B.R., Levin T.R., et al. Shifts in the Fecal Microbiota Associated with Adenomatous Polyps. Cancer Epidemiol. Biomark. Prev. 2017;26:85–94. doi: 10.1158/1055-9965.EPI-16-0337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Yeoh Y.K., Chen Z., Wong M.C.S., Hui M., Yu J., Ng S.C., Sung J.J.Y., Chan F.K.L., Chan P.K.S. Southern Chinese populations harbour non-nucleatum Fusobacteria possessing homologues of the colorectal cancer-associated FadA virulence factor. Gut. 2020;69:1998–2007. doi: 10.1136/gutjnl-2019-319635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Lin R.S., Lee F.Y., Lee S.D., Tsai Y.T., Lin H.C., Lu R.H., Hsu W.C., Huang C.C., Wang S.S., Lo K.J. Endotoxemia in patients with chronic liver diseases: Relationship to severity of liver diseases, presence of esophageal varices, and hyperdynamic circulation. J. Hepatol. 1995;22:165–172. doi: 10.1016/0168-8278(95)80424-2. [DOI] [PubMed] [Google Scholar]
- 229.Singh V., Yeoh B.S., Chassaing B., Xiao X., Saha P., Aguilera Olvera R., Lapek J.D., Jr., Zhang L., Wang W.B., Hao S., et al. Dysregulated Microbial Fermentation of Soluble Fiber Induces Cholestatic Liver Cancer. Cell. 2018;175:679–694. doi: 10.1016/j.cell.2018.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Yoshimoto S., Loo T.M., Atarashi K., Kanda H., Sato S., Oyadomari S., Iwakura Y., Oshima K., Morita H., Hattori M., et al. Obesity-induced gut microbial metabolite promotes liver cancer through senescence secretome. Nature. 2013;499:97–101. doi: 10.1038/nature12347. [DOI] [PubMed] [Google Scholar]
- 231.Cani P.D., Amar J., Iglesias M.A., Poggi M., Knauf C., Bastelica D., Neyrinck A.M., Fava F., Tuohy K.M., Chabo C., et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56:1761–1772. doi: 10.2337/db06-1491. [DOI] [PubMed] [Google Scholar]
- 232.Guasch-Ferre M., Hu F.B., Ruiz-Canela M., Bullo M., Toledo E., Wang D.D., Corella D., Gomez-Gracia E., Fiol M., Estruch R., et al. Plasma Metabolites from Choline Pathway and Risk of Cardiovascular Disease in the PREDIMED (Prevention with Mediterranean Diet) Study. J. Am. Heart Assoc. 2017;6:e006524. doi: 10.1161/JAHA.117.006524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Millard H.R., Musani S.K., Dibaba D.T., Talegawkar S.A., Taylor H.A., Tucker K.L., Bidulescu A. Dietary choline and betaine; associations with subclinical markers of cardiovascular disease risk and incidence of CVD, coronary heart disease and stroke: The Jackson Heart Study. Eur. J. Nutr. 2018;57:51–60. doi: 10.1007/s00394-016-1296-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Scher J.U., Sczesnak A., Longman R.S., Segata N., Ubeda C., Bielski C., Rostron T., Cerundolo V., Pamer E.G., Abramson S.B., et al. Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. Elife. 2013;2:e01202. doi: 10.7554/eLife.01202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Fazlollahi M., Chun Y., Grishin A., Wood R.A., Burks A.W., Dawson P., Jones S.M., Leung D.Y.M., Sampson H.A., Sicherer S.H., et al. Early-life gut microbiome and egg allergy. Allergy. 2018;73:1515–1524. doi: 10.1111/all.13389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Bunyavanich S., Shen N., Grishin A., Wood R., Burks W., Dawson P., Jones S.M., Leung D.Y.M., Sampson H., Sicherer S., et al. Early-life gut microbiome composition and milk allergy resolution. J. Allergy Clin. Immunol. 2016;138:1122–1130. doi: 10.1016/j.jaci.2016.03.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Osbelt L., Wende M., Almasi E., Derksen E., Muthukumarasamy U., Lesker T.R., Galvez E.J.C., Pils M.C., Schalk E., Chhatwal P., et al. Klebsiella oxytoca causes colonization resistance against multidrug-resistant K. pneumoniae in the gut via cooperative carbohydrate competition. Cell Host Microbe. 2021;29:1663–1679. doi: 10.1016/j.chom.2021.09.003. [DOI] [PubMed] [Google Scholar]
- 238.Osbelt L., Thiemann S., Smit N., Lesker T.R., Schroter M., Galvez E.J.C., Schmidt-Hohagen K., Pils M.C., Muhlen S., Dersch P., et al. Variations in microbiota composition of laboratory mice influence Citrobacter rodentium infection via variable short-chain fatty acid production. PLoS Pathog. 2020;16:e1008448. doi: 10.1371/journal.ppat.1008448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Oliveira R.A., Ng K.M., Correia M.B., Cabral V., Shi H., Sonnenburg J.L., Huang K.C., Xavier K.B. Klebsiella michiganensis transmission enhances resistance to Enterobacteriaceae gut invasion by nutrition competition. Nat. Microbiol. 2020;5:630–641. doi: 10.1038/s41564-019-0658-4. [DOI] [PubMed] [Google Scholar]
- 240.Ducarmon Q.R., Zwittink R.D., Hornung B.V.H., van Schaik W., Young V.B., Kuijper E.J. Gut Microbiota and Colonization Resistance against Bacterial Enteric Infection. Microbiol. Mol. Biol. Rev. 2019;83:e00007-19. doi: 10.1128/MMBR.00007-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Jacobson A., Lam L., Rajendram M., Tamburini F., Honeycutt J., Pham T., Van Treuren W., Pruss K., Stabler S.R., Lugo K., et al. A Gut Commensal-Produced Metabolite Mediates Colonization Resistance to Salmonella Infection. Cell Host Microbe. 2018;24:296–307. doi: 10.1016/j.chom.2018.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Izquierdo M., Lopez J., Gallardo P., Vidal R.M., Ossa J.C., Farfan M.J. Bacteria from gut microbiota associated with diarrheal infections in children promote virulence of Shiga toxin-producing and enteroaggregative Escherichia coli pathotypes. Front. Cell. Infect. Microbiol. 2022;12:867205. doi: 10.3389/fcimb.2022.867205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Lesniak N.A., Schubert A.M., Flynn K.J., Leslie J.L., Sinani H., Bergin I.L., Young V.B., Schloss P.D. The Gut Bacterial Community Potentiates Clostridioides difficile Infection Severity. mBio. 2022;13:e0118322. doi: 10.1128/mbio.01183-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Thiemann S., Smit N., Roy U., Lesker T.R., Galvez E.J.C., Helmecke J., Basic M., Bleich A., Goodman A.L., Kalinke U., et al. Enhancement of IFNgamma Production by Distinct Commensals Ameliorates Salmonella-Induced Disease. Cell Host Microbe. 2017;21:682–694.e685. doi: 10.1016/j.chom.2017.05.005. [DOI] [PubMed] [Google Scholar]
- 245.Caballero S., Kim S., Carter R.A., Leiner I.M., Susac B., Miller L., Kim G.J., Ling L., Pamer E.G. Cooperating Commensals Restore Colonization Resistance to Vancomycin-Resistant Enterococcus faecium. Cell Host Microbe. 2017;21:592–602.e594. doi: 10.1016/j.chom.2017.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Bibbo S., Lopetuso L.R., Ianiro G., Di Rienzo T., Gasbarrini A., Cammarota G. Role of microbiota and innate immunity in recurrent Clostridium difficile infection. J. Immunol. Res. 2014;2014:462740. doi: 10.1155/2014/462740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Warny M., Keates A.C., Keates S., Castagliuolo I., Zacks J.K., Aboudola S., Qamar A., Pothoulakis C., LaMont J.T., Kelly C.P. p38 MAP kinase activation by Clostridium difficile toxin A mediates monocyte necrosis, IL-8 production, and enteritis. J. Clin. Investig. 2000;105:1147–1156. doi: 10.1172/JCI7545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Ishida Y., Maegawa T., Kondo T., Kimura A., Iwakura Y., Nakamura S., Mukaida N. Essential involvement of IFN-gamma in Clostridium difficile toxin A-induced enteritis. J. Immunol. 2004;172:3018–3025. doi: 10.4049/jimmunol.172.5.3018. [DOI] [PubMed] [Google Scholar]
- 249.Theriot C.M., Koenigsknecht M.J., Carlson P.E., Jr., Hatton G.E., Nelson A.M., Li B., Huffnagle G.B., Li J.Z., Young V.B. Antibiotic-induced shifts in the mouse gut microbiome and metabolome increase susceptibility to Clostridium difficile infection. Nat. Commun. 2014;5:3114. doi: 10.1038/ncomms4114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Rea M.C., Dobson A., O′Sullivan O., Crispie F., Fouhy F., Cotter P.D., Shanahan F., Kiely B., Hill C., Ross R.P. Effect of broad- and narrow-spectrum antimicrobials on Clostridium difficile and microbial diversity in a model of the distal colon. Proc. Natl. Acad. Sci. USA. 2011;108((Suppl. 1)):4639–4644. doi: 10.1073/pnas.1001224107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Buffie C.G., Jarchum I., Equinda M., Lipuma L., Gobourne A., Viale A., Ubeda C., Xavier J., Pamer E.G. Profound alterations of intestinal microbiota following a single dose of clindamycin results in sustained susceptibility to Clostridium difficile-induced colitis. Infect. Immun. 2012;80:62–73. doi: 10.1128/IAI.05496-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.McDonald L.C. Effects of short- and long-course antibiotics on the lower intestinal microbiome as they relate to traveller′s diarrhea. J. Travel Med. 2017;24:S35–S38. doi: 10.1093/jtm/taw084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Wang S., Deng W., Li F., Chen Y.E., Wang P.U. Blockade of T helper 17 cell function ameliorates recurrent Clostridioides difficile infection in mice. Acta Biochim. Biophys. Sin. 2021;53:1290–1299. doi: 10.1093/abbs/gmab107. [DOI] [PubMed] [Google Scholar]
- 254.Frisbee A.L., Saleh M.M., Young M.K., Leslie J.L., Simpson M.E., Abhyankar M.M., Cowardin C.A., Ma J.Z., Pramoonjago P., Turner S.D., et al. IL-33 drives group 2 innate lymphoid cell-mediated protection during Clostridium difficile infection. Nat. Commun. 2019;10:2712. doi: 10.1038/s41467-019-10733-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Wu Z., Xu Q., Wang Q., Chen Y., Lv L., Zheng B., Yan R., Jiang H., Shen J., Wang S., et al. The impact of dietary fibers on Clostridioides difficile infection in a mouse model. Front. Cell. Infect. Microbiol. 2022;12:1028267. doi: 10.3389/fcimb.2022.1028267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Hayashi A., Nagao-Kitamoto H., Kitamoto S., Kim C.H., Kamada N. The Butyrate-Producing Bacterium Clostridium butyricum Suppresses Clostridioides difficile Infection via Neutrophil- and Antimicrobial Cytokine-Dependent but GPR43/109a-Independent Mechanisms. J. Immunol. 2021;206:1576–1585. doi: 10.4049/jimmunol.2000353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Petrof E.O., Gloor G.B., Vanner S.J., Weese S.J., Carter D., Daigneault M.C., Brown E.M., Schroeter K., Allen-Vercoe E. Stool substitute transplant therapy for the eradication of Clostridium difficile infection: ′RePOOPulating′ the gut. Microbiome. 2013;1:3. doi: 10.1186/2049-2618-1-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Martz S.L., McDonald J.A., Sun J., Zhang Y.G., Gloor G.B., Noordhof C., He S.M., Gerbaba T.K., Blennerhassett M., Hurlbut D.J., et al. Administration of defined microbiota is protective in a murine Salmonella infection model. Sci. Rep. 2015;5:16094. doi: 10.1038/srep16094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Barbara G., Barbaro M.R., Fuschi D., Palombo M., Falangone F., Cremon C., Marasco G., Stanghellini V. Inflammatory and Microbiota-Related Regulation of the Intestinal Epithelial Barrier. Front. Nutr. 2021;8:718356. doi: 10.3389/fnut.2021.718356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Chassaing B., Darfeuille-Michaud A. The commensal microbiota and enteropathogens in the pathogenesis of inflammatory bowel diseases. Gastroenterology. 2011;140:1720–1728. doi: 10.1053/j.gastro.2011.01.054. [DOI] [PubMed] [Google Scholar]
- 261.Swidsinski A., Ladhoff A., Pernthaler A., Swidsinski S., Loening-Baucke V., Ortner M., Weber J., Hoffmann U., Schreiber S., Dietel M., et al. Mucosal flora in inflammatory bowel disease. Gastroenterology. 2002;122:44–54. doi: 10.1053/gast.2002.30294. [DOI] [PubMed] [Google Scholar]
- 262.Schwartz S.M., Kemnitz J.W. Age- and gender-related changes in body size, adiposity, and endocrine and metabolic parameters in free-ranging rhesus macaques. Am. J. Phys. Anthropol. 1992;89:109–121. doi: 10.1002/ajpa.1330890110. [DOI] [PubMed] [Google Scholar]
- 263.Gevers D., Kugathasan S., Denson L.A., Vazquez-Baeza Y., Van Treuren W., Ren B., Schwager E., Knights D., Song S.J., Yassour M., et al. The treatment-naive microbiome in new-onset Crohn′s disease. Cell Host Microbe. 2014;15:382–392. doi: 10.1016/j.chom.2014.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Machiels K., Joossens M., Sabino J., De Preter V., Arijs I., Eeckhaut V., Ballet V., Claes K., Van Immerseel F., Verbeke K., et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut. 2014;63:1275–1283. doi: 10.1136/gutjnl-2013-304833. [DOI] [PubMed] [Google Scholar]
- 265.Devkota S., Wang Y., Musch M.W., Leone V., Fehlner-Peach H., Nadimpalli A., Antonopoulos D.A., Jabri B., Chang E.B. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10−/− mice. Nature. 2012;487:104–108. doi: 10.1038/nature11225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Shapiro J.M., de Zoete M.R., Palm N.W., Laenen Y., Bright R., Mallette M., Bu K., Bielecka A.A., Xu F., Hurtado-Lorenzo A., et al. Immunoglobulin A Targets a Unique Subset of the Microbiota in Inflammatory Bowel Disease. Cell Host Microbe. 2021;29:83–93. doi: 10.1016/j.chom.2020.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Palm N.W., de Zoete M.R., Cullen T.W., Barry N.A., Stefanowski J., Hao L., Degnan P.H., Hu J., Peter I., Zhang W., et al. Immunoglobulin A coating identifies colitogenic bacteria in inflammatory bowel disease. Cell. 2014;158:1000–1010. doi: 10.1016/j.cell.2014.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Schaubeck M., Clavel T., Calasan J., Lagkouvardos I., Haange S.B., Jehmlich N., Basic M., Dupont A., Hornef M., von Bergen M., et al. Dysbiotic gut microbiota causes transmissible Crohn′s disease-like ileitis independent of failure in antimicrobial defence. Gut. 2016;65:225–237. doi: 10.1136/gutjnl-2015-309333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Torres J., Hu J., Seki A., Eisele C., Nair N., Huang R., Tarassishin L., Jharap B., Cote-Daigneault J., Mao Q., et al. Infants born to mothers with IBD present with altered gut microbiome that transfers abnormalities of the adaptive immune system to germ-free mice. Gut. 2020;69:42–51. doi: 10.1136/gutjnl-2018-317855. [DOI] [PubMed] [Google Scholar]
- 270.Franzosa E.A., Sirota-Madi A., Avila-Pacheco J., Fornelos N., Haiser H.J., Reinker S., Vatanen T., Hall A.B., Mallick H., McIver L.J., et al. Gut microbiome structure and metabolic activity in inflammatory bowel disease. Nat. Microbiol. 2019;4:293–305. doi: 10.1038/s41564-018-0306-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Facchin S., Vitulo N., Calgaro M., Buda A., Romualdi C., Pohl D., Perini B., Lorenzon G., Marinelli C., D′Inca R., et al. Microbiota changes induced by microencapsulated sodium butyrate in patients with inflammatory bowel disease. Neurogastroenterol. Motil. 2020;32:e13914. doi: 10.1111/nmo.13914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Li G., Lin J., Zhang C., Gao H., Lu H., Gao X., Zhu R., Li Z., Li M., Liu Z. Microbiota metabolite butyrate constrains neutrophil functions and ameliorates mucosal inflammation in inflammatory bowel disease. Gut Microbes. 2021;13:1968257. doi: 10.1080/19490976.2021.1968257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Hartog A., Belle F.N., Bastiaans J., de Graaff P., Garssen J., Harthoorn L.F., Vos A.P. A potential role for regulatory T-cells in the amelioration of DSS induced colitis by dietary non-digestible polysaccharides. J. Nutr. Biochem. 2015;26:227–233. doi: 10.1016/j.jnutbio.2014.10.011. [DOI] [PubMed] [Google Scholar]
- 274.Singh V., Yeoh B.S., Walker R.E., Xiao X., Saha P., Golonka R.M., Cai J., Bretin A.C.A., Cheng X., Liu Q., et al. Microbiota fermentation-NLRP3 axis shapes the impact of dietary fibres on intestinal inflammation. Gut. 2019;68:1801–1812. doi: 10.1136/gutjnl-2018-316250. [DOI] [PubMed] [Google Scholar]
- 275.Zhu Y., Xu Y., Wang X., Rao L., Yan X., Gao R., Shen T., Zhou Y., Kong C., Zhou L. Probiotic Cocktail Alleviates Intestinal Inflammation Through Improving Gut Microbiota and Metabolites in Colitis Mice. Front. Cell. Infect. Microbiol. 2022;12:886061. doi: 10.3389/fcimb.2022.886061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Xu M., Shen Y., Cen M., Zhu Y., Cheng F., Tang L., Zheng X., Kim J.J., Dai N., Hu W. Modulation of the Gut Microbiota-farnesoid X Receptor Axis Improves Deoxycholic Acid-induced Intestinal Inflammation in Mice. J. Crohn’s Colitis. 2021;15:1197–1210. doi: 10.1093/ecco-jcc/jjab003. [DOI] [PubMed] [Google Scholar]
- 277.Xu M., Cen M., Shen Y., Zhu Y., Cheng F., Tang L., Hu W., Dai N. Deoxycholic Acid-Induced Gut Dysbiosis Disrupts Bile Acid Enterohepatic Circulation and Promotes Intestinal Inflammation. Dig. Dis. Sci. 2021;66:568–576. doi: 10.1007/s10620-020-06208-3. [DOI] [PubMed] [Google Scholar]
- 278.Liu T.C., Kern J.T., Jain U., Sonnek N.M., Xiong S., Simpson K.F., VanDussen K.L., Winkler E.S., Haritunians T., Malique A., et al. Western diet induces Paneth cell defects through microbiome alterations and farnesoid X receptor and type I interferon activation. Cell Host Microbe. 2021;29:988–1001. doi: 10.1016/j.chom.2021.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Liu Y., Xu J., Ren X., Zhang Y., Ke Z., Zhou J., Wang Y., Zhang Y., Liu Y. Cholecystectomy-induced secondary bile acids accumulation ameliorates colitis through inhibiting monocyte/macrophage recruitment. Gut Microbes. 2022;14:2107387. doi: 10.1080/19490976.2022.2107387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Van den Bossche L., Hindryckx P., Devisscher L., Devriese S., Van Welden S., Holvoet T., Vilchez-Vargas R., Vital M., Pieper D.H., Vanden Bussche J., et al. Ursodeoxycholic Acid and Its Taurine- or Glycine-Conjugated Species Reduce Colitogenic Dysbiosis and Equally Suppress Experimental Colitis in Mice. Appl. Environ. Microbiol. 2017;83:e02766-16. doi: 10.1128/AEM.02766-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Duboc H., Rajca S., Rainteau D., Benarous D., Maubert M.A., Quervain E., Thomas G., Barbu V., Humbert L., Despras G., et al. Connecting dysbiosis, bile-acid dysmetabolism and gut inflammation in inflammatory bowel diseases. Gut. 2013;62:531–539. doi: 10.1136/gutjnl-2012-302578. [DOI] [PubMed] [Google Scholar]
- 282.Moltzau Anderson J., Lipinski S., Sommer F., Pan W.H., Boulard O., Rehman A., Falk-Paulsen M., Stengel S.T., Aden K., Hasler R., et al. NOD2 Influences Trajectories of Intestinal Microbiota Recovery After Antibiotic Perturbation. Cell. Mol. Gastroenterol. Hepatol. 2020;10:365–389. doi: 10.1016/j.jcmgh.2020.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Danne C., Michaudel C., Skerniskyte J., Planchais J., Magniez A., Agus A., Michel M.L., Lamas B., Da Costa G., Spatz M., et al. CARD9 in neutrophils protects from colitis and controls mitochondrial metabolism and cell survival. Gut. 2022 doi: 10.1136/gutjnl-2022-326917. [DOI] [PubMed] [Google Scholar]
- 284.Lavoie S., Conway K.L., Lassen K.G., Jijon H.B., Pan H., Chun E., Michaud M., Lang J.K., Gallini Comeau C.A., Dreyfuss J.M., et al. The Crohn′s disease polymorphism, ATG16L1 T300A, alters the gut microbiota and enhances the local Th1/Th17 response. Elife. 2019;8:e39982. doi: 10.7554/eLife.39982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Aden K., Tran F., Ito G., Sheibani-Tezerji R., Lipinski S., Kuiper J.W., Tschurtschenthaler M., Saveljeva S., Bhattacharyya J., Hasler R., et al. ATG16L1 orchestrates interleukin-22 signaling in the intestinal epithelium via cGAS-STING. J. Exp. Med. 2018;215:2868–2886. doi: 10.1084/jem.20171029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Zhou C.B., Zhou Y.L., Fang J.Y. Gut Microbiota in Cancer Immune Response and Immunotherapy. Trends Cancer. 2021;7:647–660. doi: 10.1016/j.trecan.2021.01.010. [DOI] [PubMed] [Google Scholar]
- 287.Xu C., Fan L., Lin Y., Shen W., Qi Y., Zhang Y., Chen Z., Wang L., Long Y., Hou T., et al. Fusobacterium nucleatum promotes colorectal cancer metastasis through miR-1322/CCL20 axis and M2 polarization. Gut Microbes. 2021;13:1980347. doi: 10.1080/19490976.2021.1980347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Hussan H., Clinton S.K., Roberts K., Bailey M.T. Fusobacterium′s link to colorectal neoplasia sequenced: A systematic review and future insights. World J. Gastroenterol. 2017;23:8626–8650. doi: 10.3748/wjg.v23.i48.8626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Yang Y., Weng W., Peng J., Hong L., Yang L., Toiyama Y., Gao R., Liu M., Yin M., Pan C., et al. Fusobacterium nucleatum Increases Proliferation of Colorectal Cancer Cells and Tumor Development in Mice by Activating Toll-Like Receptor 4 Signaling to Nuclear Factor-kappaB, and Up-regulating Expression of MicroRNA-21. Gastroenterology. 2017;152:851–866. doi: 10.1053/j.gastro.2016.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Gur C., Maalouf N., Shhadeh A., Berhani O., Singer B.B., Bachrach G., Mandelboim O. Fusobacterium nucleatum supresses anti-tumor immunity by activating CEACAM1. Oncoimmunology. 2019;8:e1581531. doi: 10.1080/2162402X.2019.1581531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Loesche W.J., Gibbons R.J. Amino acid fermentation by Fusobacterium nucleatum. Arch. Oral Biol. 1968;13:191–202. doi: 10.1016/0003-9969(68)90051-4. [DOI] [PubMed] [Google Scholar]
- 292.Lavoie S., Chun E., Bae S., Brennan C.A., Gallini Comeau C.A., Lang J.K., Michaud M., Hoveyda H.R., Fraser G.L., Fuller M.H., et al. Expression of Free Fatty Acid Receptor 2 by Dendritic Cells Prevents Their Expression of Interleukin 27 and Is Required for Maintenance of Mucosal Barrier and Immune Response Against Colorectal Tumors in Mice. Gastroenterology. 2020;158:1359–1372. doi: 10.1053/j.gastro.2019.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Donohoe D.R., Holley D., Collins L.B., Montgomery S.A., Whitmore A.C., Hillhouse A., Curry K.P., Renner S.W., Greenwalt A., Ryan E.P., et al. A gnotobiotic mouse model demonstrates that dietary fiber protects against colorectal tumorigenesis in a microbiota- and butyrate-dependent manner. Cancer Discov. 2014;4:1387–1397. doi: 10.1158/2159-8290.CD-14-0501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Dong W., Liu L., Dou Y., Xu M., Liu T., Wang S., Zhang Y., Deng B., Wang B., Cao H. Deoxycholic acid activates epidermal growth factor receptor and promotes intestinal carcinogenesis by ADAM17-dependent ligand release. J. Cell. Mol. Med. 2018;22:4263–4273. doi: 10.1111/jcmm.13709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Pai R., Tarnawski A.S., Tran T. Deoxycholic acid activates beta-catenin signaling pathway and increases colon cell cancer growth and invasiveness. Mol. Biol. Cell. 2004;15:2156–2163. doi: 10.1091/mbc.e03-12-0894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Fu T., Coulter S., Yoshihara E., Oh T.G., Fang S., Cayabyab F., Zhu Q., Zhang T., Leblanc M., Liu S., et al. FXR Regulates Intestinal Cancer Stem Cell Proliferation. Cell. 2019;176:1098–1112. doi: 10.1016/j.cell.2019.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Liu L., Yang M., Dong W., Liu T., Song X., Gu Y., Wang S., Liu Y., Abla Z., Qiao X., et al. Gut Dysbiosis and Abnormal Bile Acid Metabolism in Colitis-Associated Cancer. Gastroenterol. Res. Pract. 2021;2021:6645970. doi: 10.1155/2021/6645970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Ternes D., Tsenkova M., Pozdeev V.I., Meyers M., Koncina E., Atatri S., Schmitz M., Karta J., Schmoetten M., Heinken A., et al. The gut microbial metabolite formate exacerbates colorectal cancer progression. Nat. Metab. 2022;4:458–475. doi: 10.1038/s42255-022-00558-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Fan L., Xu C., Ge Q., Lin Y., Wong C.C., Qi Y., Ye B., Lian Q., Zhuo W., Si J., et al. A. muciniphila Suppresses Colorectal Tumorigenesis by Inducing TLR2/NLRP3-Mediated M1-Like TAMs. Cancer Immunol. Res. 2021;9:1111–1124. doi: 10.1158/2326-6066.CIR-20-1019. [DOI] [PubMed] [Google Scholar]
- 300.Zegarra Ruiz D.F., Kim D.V., Norwood K., Saldana-Morales F.B., Kim M., Ng C., Callaghan R., Uddin M., Chang L.C., Longman R.S., et al. Microbiota manipulation to increase macrophage IL-10 improves colitis and limits colitis-associated colorectal cancer. Gut Microbes. 2022;14:2119054. doi: 10.1080/19490976.2022.2119054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Yu A.I., Zhao L., Eaton K.A., Ho S., Chen J., Poe S., Becker J., Gonzalez A., McKinstry D., Hasso M., et al. Gut Microbiota Modulate CD8 T Cell Responses to Influence Colitis-Associated Tumorigenesis. Cell Rep. 2020;31:107471. doi: 10.1016/j.celrep.2020.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Peuker K., Strigli A., Tauriello D.V.F., Hendricks A., von Schonfels W., Burmeister G., Brosch M., Herrmann A., Kruger S., Nitsche J., et al. Microbiota-dependent activation of the myeloid calcineurin-NFAT pathway inhibits B7H3- and B7H4-dependent anti-tumor immunity in colorectal cancer. Immunity. 2022;55:701–717. doi: 10.1016/j.immuni.2022.03.008. [DOI] [PubMed] [Google Scholar]
- 303.Overacre-Delgoffe A.E., Bumgarner H.J., Cillo A.R., Burr A.H.P., Tometich J.T., Bhattacharjee A., Bruno T.C., Vignali D.A.A., Hand T.W. Microbiota-specific T follicular helper cells drive tertiary lymphoid structures and anti-tumor immunity against colorectal cancer. Immunity. 2021;54:2812–2824.e2814. doi: 10.1016/j.immuni.2021.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Yang J.D., Hainaut P., Gores G.J., Amadou A., Plymoth A., Roberts L.R. A global view of hepatocellular carcinoma: Trends, risk, prevention and management. Nat. Rev. Gastroenterol. Hepatol. 2019;16:589–604. doi: 10.1038/s41575-019-0186-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Huang D.Q., El-Serag H.B., Loomba R. Global epidemiology of NAFLD-related HCC: Trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 2021;18:223–238. doi: 10.1038/s41575-020-00381-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Park E.J., Lee J.H., Yu G.Y., He G., Ali S.R., Holzer R.G., Osterreicher C.H., Takahashi H., Karin M. Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell. 2010;140:197–208. doi: 10.1016/j.cell.2009.12.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Said I., Ahad H., Said A. Gut microbiome in non-alcoholic fatty liver disease associated hepatocellular carcinoma: Current knowledge and potential for therapeutics. World J. Gastrointest. Oncol. 2022;14:947–958. doi: 10.4251/wjgo.v14.i5.947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Behary J., Raposo A.E., Amorim N.M.L., Zheng H., Gong L., McGovern E., Chen J., Liu K., Beretov J., Theocharous C., et al. Defining the temporal evolution of gut dysbiosis and inflammatory responses leading to hepatocellular carcinoma in Mdr2 −/− mouse model. BMC Microbiol. 2021;21:113. doi: 10.1186/s12866-021-02171-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Ponziani F.R., Bhoori S., Castelli C., Putignani L., Rivoltini L., Del Chierico F., Sanguinetti M., Morelli D., Paroni Sterbini F., Petito V., et al. Hepatocellular Carcinoma Is Associated with Gut Microbiota Profile and Inflammation in Nonalcoholic Fatty Liver Disease. Hepatology. 2019;69:107–120. doi: 10.1002/hep.30036. [DOI] [PubMed] [Google Scholar]
- 310.Zheng R., Wang G., Pang Z., Ran N., Gu Y., Guan X., Yuan Y., Zuo X., Pan H., Zheng J., et al. Liver cirrhosis contributes to the disorder of gut microbiota in patients with hepatocellular carcinoma. Cancer Med. 2020;9:4232–4250. doi: 10.1002/cam4.3045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Liu Q., Li F., Zhuang Y., Xu J., Wang J., Mao X., Zhang Y., Liu X. Alteration in gut microbiota associated with hepatitis B and non-hepatitis virus related hepatocellular carcinoma. Gut Pathog. 2019;11:1. doi: 10.1186/s13099-018-0281-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Ren Z., Li A., Jiang J., Zhou L., Yu Z., Lu H., Xie H., Chen X., Shao L., Zhang R., et al. Gut microbiome analysis as a tool towards targeted non-invasive biomarkers for early hepatocellular carcinoma. Gut. 2019;68:1014–1023. doi: 10.1136/gutjnl-2017-315084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Grat M., Wronka K.M., Krasnodebski M., Masior L., Lewandowski Z., Kosinska I., Grat K., Stypulkowski J., Rejowski S., Wasilewicz M., et al. Profile of Gut Microbiota Associated with the Presence of Hepatocellular Cancer in Patients with Liver Cirrhosis. Transplant. Proc. 2016;48:1687–1691. doi: 10.1016/j.transproceed.2016.01.077. [DOI] [PubMed] [Google Scholar]
- 314.Ohtani N., Hara E. Gut-liver axis-mediated mechanism of liver cancer: A special focus on the role of gut microbiota. Cancer Sci. 2021;112:4433–4443. doi: 10.1111/cas.15142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Loo T.M., Kamachi F., Watanabe Y., Yoshimoto S., Kanda H., Arai Y., Nakajima-Takagi Y., Iwama A., Koga T., Sugimoto Y., et al. Gut Microbiota Promotes Obesity-Associated Liver Cancer through PGE(2)-Mediated Suppression of Antitumor Immunity. Cancer Discov. 2017;7:522–538. doi: 10.1158/2159-8290.CD-16-0932. [DOI] [PubMed] [Google Scholar]
- 316.Golonka R.M., Xiao X., Abokor A.A., Joe B., Vijay-Kumar M. Altered nutrient status reprograms host inflammation and metabolic health via gut microbiota. J. Nutr. Biochem. 2020;80:108360. doi: 10.1016/j.jnutbio.2020.108360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Hosseinkhani F., Heinken A., Thiele I., Lindenburg P.W., Harms A.C., Hankemeier T. The contribution of gut bacterial metabolites in the human immune signaling pathway of non-communicable diseases. Gut Microbes. 2021;13:1882927. doi: 10.1080/19490976.2021.1882927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Yue C., Yang X., Li J., Chen X., Zhao X., Chen Y., Wen Y. Trimethylamine N-oxide prime NLRP3 inflammasome via inhibiting ATG16L1-induced autophagy in colonic epithelial cells. Biochem. Biophys. Res. Commun. 2017;490:541–551. doi: 10.1016/j.bbrc.2017.06.075. [DOI] [PubMed] [Google Scholar]
- 319.Wang Z., Klipfell E., Bennett B.J., Koeth R., Levison B.S., Dugar B., Feldstein A.E., Britt E.B., Fu X., Chung Y.M., et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature. 2011;472:57–63. doi: 10.1038/nature09922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.Guasti L., Galliazzo S., Molaro M., Visconti E., Pennella B., Gaudio G.V., Lupi A., Grandi A.M., Squizzato A. TMAO as a biomarker of cardiovascular events: A systematic review and meta-analysis. Intern. Emerg. Med. 2021;16:201–207. doi: 10.1007/s11739-020-02470-5. [DOI] [PubMed] [Google Scholar]
- 321.Fang C., Zuo K., Jiao K., Zhu X., Fu Y., Zhong J., Xu L., Yang X. PAGln, an Atrial Fibrillation-Linked Gut Microbial Metabolite, Acts as a Promoter of Atrial Myocyte Injury. Biomolecules. 2022;12:1120. doi: 10.3390/biom12081120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Nemet I., Saha P.P., Gupta N., Zhu W., Romano K.A., Skye S.M., Cajka T., Mohan M.L., Li L., Wu Y., et al. A Cardiovascular Disease-Linked Gut Microbial Metabolite Acts via Adrenergic Receptors. Cell. 2020;180:862–877. doi: 10.1016/j.cell.2020.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Bell K.J., Saad S., Tillett B.J., McGuire H.M., Bordbar S., Yap Y.A., Nguyen L.T., Wilkins M.R., Corley S., Brodie S., et al. Metabolite-based dietary supplementation in human type 1 diabetes is associated with microbiota and immune modulation. Microbiome. 2022;10:9. doi: 10.1186/s40168-021-01193-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.Girdhar K., Huang Q., Chow I.T., Vatanen T., Brady C., Raisingani A., Autissier P., Atkinson M.A., Kwok W.W., Kahn C.R., et al. A gut microbial peptide and molecular mimicry in the pathogenesis of type 1 diabetes. Proc. Natl. Acad. Sci. USA. 2022;119:e2120028119. doi: 10.1073/pnas.2120028119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Lamichhane S., Sen P., Dickens A.M., Alves M.A., Harkonen T., Honkanen J., Vatanen T., Xavier R.J., Hyotylainen T., Knip M., et al. Dysregulation of secondary bile acid metabolism precedes islet autoimmunity and type 1 diabetes. Cell Rep. Med. 2022;3:100762. doi: 10.1016/j.xcrm.2022.100762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Pedersen H.K., Gudmundsdottir V., Nielsen H.B., Hyotylainen T., Nielsen T., Jensen B.A., Forslund K., Hildebrand F., Prifti E., Falony G., et al. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature. 2016;535:376–381. doi: 10.1038/nature18646. [DOI] [PubMed] [Google Scholar]
- 327.Zhou M., Shao J., Wu C.Y., Shu L., Dong W., Liu Y., Chen M., Wynn R.M., Wang J., Wang J., et al. Targeting BCAA Catabolism to Treat Obesity-Associated Insulin Resistance. Diabetes. 2019;68:1730–1746. doi: 10.2337/db18-0927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Bodogai M., O′Connell J., Kim K., Kim Y., Moritoh K., Chen C., Gusev F., Vaughan K., Shulzhenko N., Mattison J.A., et al. Commensal bacteria contribute to insulin resistance in aging by activating innate B1a cells. Sci. Transl. Med. 2018;10:eaat4271. doi: 10.1126/scitranslmed.aat4271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Huang J.P., Cheng M.L., Hung C.Y., Wang C.H., Hsieh P.S., Shiao M.S., Chen J.K., Li D.E., Hung L.M. Docosapentaenoic acid and docosahexaenoic acid are positively associated with insulin sensitivity in rats fed high-fat and high-fructose diets. J. Diabetes. 2017;9:936–946. doi: 10.1111/1753-0407.12505. [DOI] [PubMed] [Google Scholar]
- 330.Wang H., Lu Y., Yan Y., Tian S., Zheng D., Leng D., Wang C., Jiao J., Wang Z., Bai Y. Promising Treatment for Type 2 Diabetes: Fecal Microbiota Transplantation Reverses Insulin Resistance and Impaired Islets. Front. Cell. Infect. Microbiol. 2019;9:455. doi: 10.3389/fcimb.2019.00455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Deng X., Zhang C., Wang P., Wei W., Shi X., Wang P., Yang J., Wang L., Tang S., Fang Y., et al. Cardiovascular Benefits of Empagliflozin Are Associated with Gut Microbiota and Plasma Metabolites in Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2022;107:1888–1896. doi: 10.1210/clinem/dgac210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Montandon S.A., Jornayvaz F.R. Effects of Antidiabetic Drugs on Gut Microbiota Composition. Genes. 2017;8:250. doi: 10.3390/genes8100250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Wu H., Esteve E., Tremaroli V., Khan M.T., Caesar R., Manneras-Holm L., Stahlman M., Olsson L.M., Serino M., Planas-Felix M., et al. Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug. Nat. Med. 2017;23:850–858. doi: 10.1038/nm.4345. [DOI] [PubMed] [Google Scholar]
- 334.Li J., Zhao F., Wang Y., Chen J., Tao J., Tian G., Wu S., Liu W., Cui Q., Geng B., et al. Gut microbiota dysbiosis contributes to the development of hypertension. Microbiome. 2017;5:14. doi: 10.1186/s40168-016-0222-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Adnan S., Nelson J.W., Ajami N.J., Venna V.R., Petrosino J.F., Bryan R.M., Jr., Durgan D.J. Alterations in the gut microbiota can elicit hypertension in rats. Physiol. Genom. 2017;49:96–104. doi: 10.1152/physiolgenomics.00081.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 336.Marques F.Z., Nelson E., Chu P.Y., Horlock D., Fiedler A., Ziemann M., Tan J.K., Kuruppu S., Rajapakse N.W., El-Osta A., et al. High-Fiber Diet and Acetate Supplementation Change the Gut Microbiota and Prevent the Development of Hypertension and Heart Failure in Hypertensive Mice. Circulation. 2017;135:964–977. doi: 10.1161/CIRCULATIONAHA.116.024545. [DOI] [PubMed] [Google Scholar]
- 337.Santisteban M.M., Qi Y., Zubcevic J., Kim S., Yang T., Shenoy V., Cole-Jeffrey C.T., Lobaton G.O., Stewart D.C., Rubiano A., et al. Hypertension-Linked Pathophysiological Alterations in the Gut. Circ. Res. 2017;120:312–323. doi: 10.1161/CIRCRESAHA.116.309006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338.Karbach S.H., Schonfelder T., Brandao I., Wilms E., Hormann N., Jackel S., Schuler R., Finger S., Knorr M., Lagrange J., et al. Gut Microbiota Promote Angiotensin II-Induced Arterial Hypertension and Vascular Dysfunction. J. Am. Heart Assoc. 2016;5:e003698. doi: 10.1161/JAHA.116.003698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Avery E.G., Bartolomaeus H., Rauch A., Chen C.Y., N′Diaye G., Lober U., Bartolomaeus T.U.P., Fritsche-Guenther R., Rodrigues A.F., Yarritu A., et al. Quantifying the impact of gut microbiota on inflammation and hypertensive organ damage. Cardiovasc. Res. 2022 doi: 10.1093/cvr/cvac121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Joe B., McCarthy C.G., Edwards J.M., Cheng X., Chakraborty S., Yang T., Golonka R.M., Mell B., Yeo J.Y., Bearss N.R., et al. Microbiota Introduced to Germ-Free Rats Restores Vascular Contractility and Blood Pressure. Hypertension. 2020;76:1847–1855. doi: 10.1161/HYPERTENSIONAHA.120.15939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Abboud F.M., Cicha M.Z., Ericsson A., Chapleau M.W., Singh M.V. Altering Early Life Gut Microbiota Has Long-Term Effect on Immune System and Hypertension in Spontaneously Hypertensive Rats. Front. Physiol. 2021;12:752924. doi: 10.3389/fphys.2021.752924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Jiang S., Shui Y., Cui Y., Tang C., Wang X., Qiu X., Hu W., Fei L., Li Y., Zhang S., et al. Gut microbiota dependent trimethylamine N-oxide aggravates angiotensin II-induced hypertension. Redox Biol. 2021;46:102115. doi: 10.1016/j.redox.2021.102115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Ferguson J.F., Aden L.A., Barbaro N.R., Van Beusecum J.P., Xiao L., Simmons A.J., Warden C., Pasic L., Himmel L.E., Washington M.K., et al. High dietary salt-induced dendritic cell activation underlies microbial dysbiosis-associated hypertension. JCI Insight. 2019;5:e126241. doi: 10.1172/jci.insight.126241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Chakraborty S., Galla S., Cheng X., Yeo J.Y., Mell B., Singh V., Yeoh B., Saha P., Mathew A.V., Vijay-Kumar M., et al. Salt-Responsive Metabolite, beta-Hydroxybutyrate, Attenuates Hypertension. Cell Rep. 2018;25:677–689. doi: 10.1016/j.celrep.2018.09.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Lee Y.H. Causal association of gut microbiome on the risk of rheumatoid arthritis: A Mendelian randomisation study. Ann. Rheum. Dis. 2022;81:e3. doi: 10.1136/annrheumdis-2019-216747. [DOI] [PubMed] [Google Scholar]
- 346.Kishikawa T., Maeda Y., Nii T., Motooka D., Matsumoto Y., Matsushita M., Matsuoka H., Yoshimura M., Kawada S., Teshigawara S., et al. Metagenome-wide association study of gut microbiome revealed novel aetiology of rheumatoid arthritis in the Japanese population. Ann. Rheum. Dis. 2020;79:103–111. doi: 10.1136/annrheumdis-2019-215743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Pianta A., Arvikar S., Strle K., Drouin E.E., Wang Q., Costello C.E., Steere A.C. Evidence of the Immune Relevance of Prevotella copri, a Gut Microbe, in Patients with Rheumatoid Arthritis. Arthritis Rheumatol. 2017;69:964–975. doi: 10.1002/art.40003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Li Y., Zhang S.X., Yin X.F., Zhang M.X., Qiao J., Xin X.H., Chang M.J., Gao C., Li Y.F., Li X.F. The Gut Microbiota and Its Relevance to Peripheral Lymphocyte Subpopulations and Cytokines in Patients with Rheumatoid Arthritis. J. Immunol. Res. 2021;2021:6665563. doi: 10.1155/2021/6665563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 349.Teng F., Klinger C.N., Felix K.M., Bradley C.P., Wu E., Tran N.L., Umesaki Y., Wu H.J. Gut Microbiota Drive Autoimmune Arthritis by Promoting Differentiation and Migration of Peyer′s Patch T Follicular Helper Cells. Immunity. 2016;44:875–888. doi: 10.1016/j.immuni.2016.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 350.Bates N.A., Li A., Fan T., Cutcliffe M.P., Dagenet C.B., Sleiman K.C., Ma H., Tahsin S., Garrett C.S., Altemus J., et al. Gut Commensal Segmented Filamentous Bacteria Fine-Tune T Follicular Regulatory Cells to Modify the Severity of Systemic Autoimmune Arthritis. J. Immunol. 2021;206:941–952. doi: 10.4049/jimmunol.2000663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 351.Teng F., Felix K.M., Bradley C.P., Naskar D., Ma H., Raslan W.A., Wu H.J. The impact of age and gut microbiota on Th17 and Tfh cells in K/BxN autoimmune arthritis. Arthritis Res. Ther. 2017;19:188. doi: 10.1186/s13075-017-1398-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Block K.E., Zheng Z., Dent A.L., Kee B.L., Huang H. Gut Microbiota Regulates K/BxN Autoimmune Arthritis through Follicular Helper T but Not Th17 Cells. J. Immunol. 2016;196:1550–1557. doi: 10.4049/jimmunol.1501904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Simpkins D.A., Downton P., Gray K.J., Dickson S.H., Maidstone R.J., Konkel J.E., Hepworth M.R., Ray D.W., Bechtold D.A., Gibbs J.E. Consequences of collagen induced inflammatory arthritis on circadian regulation of the gut microbiome. FASEB J. 2023;37:e22704. doi: 10.1096/fj.202201728R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 354.He J., Chu Y., Li J., Meng Q., Liu Y., Jin J., Wang Y., Wang J., Huang B., Shi L., et al. Intestinal butyrate-metabolizing species contribute to autoantibody production and bone erosion in rheumatoid arthritis. Sci. Adv. 2022;8:eabm1511. doi: 10.1126/sciadv.abm1511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.Takahashi D., Hoshina N., Kabumoto Y., Maeda Y., Suzuki A., Tanabe H., Isobe J., Yamada T., Muroi K., Yanagisawa Y., et al. Microbiota-derived butyrate limits the autoimmune response by promoting the differentiation of follicular regulatory T cells. EBioMedicine. 2020;58:102913. doi: 10.1016/j.ebiom.2020.102913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 356.Rosser E.C., Piper C.J.M., Matei D.E., Blair P.A., Rendeiro A.F., Orford M., Alber D.G., Krausgruber T., Catalan D., Klein N., et al. Microbiota-Derived Metabolites Suppress Arthritis by Amplifying Aryl-Hydrocarbon Receptor Activation in Regulatory B Cells. Cell Metab. 2020;31:837–851. doi: 10.1016/j.cmet.2020.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 357.Sun H., Guo Y., Wang H., Yin A., Hu J., Yuan T., Zhou S., Xu W., Wei P., Yin S., et al. Gut commensal Parabacteroides distasonis alleviates inflammatory arthritis. Gut. 2023 doi: 10.1136/gutjnl-2022-327756. [DOI] [PubMed] [Google Scholar]
- 358.Adeyeye T.E., Yeung E.H., McLain A.C., Lin S., Lawrence D.A., Bell E.M. Wheeze and Food Allergies in Children Born via Cesarean Delivery: The Upstate KIDS Study. Am. J. Epidemiol. 2019;188:355–362. doi: 10.1093/aje/kwy257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Loh W., Tang M.L.K. The Epidemiology of Food Allergy in the Global Context. Int J. Environ. Res. Public Health. 2018;15:2043. doi: 10.3390/ijerph15092043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Leung A.S.Y., Wong G.W.K., Tang M.L.K. Food allergy in the developing world. J. Allergy Clin. Immunol. 2018;141:76–78. doi: 10.1016/j.jaci.2017.11.008. [DOI] [PubMed] [Google Scholar]
- 361.Borbet T.C., Pawline M.B., Zhang X., Wipperman M.F., Reuter S., Maher T., Li J., Iizumi T., Gao Z., Daniele M., et al. Influence of the early-life gut microbiota on the immune responses to an inhaled allergen. Mucosal Immunol. 2022;15:1000–1011. doi: 10.1038/s41385-022-00544-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 362.Zhang Q., Cheng L., Wang J., Hao M., Che H. Antibiotic-Induced Gut Microbiota Dysbiosis Damages the Intestinal Barrier, Increasing Food Allergy in Adult Mice. Nutrients. 2021;13:3315. doi: 10.3390/nu13103315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Hussain M., Bonilla-Rosso G., Kwong Chung C.K.C., Bariswyl L., Rodriguez M.P., Kim B.S., Engel P., Noti M. High dietary fat intake induces a microbiota signature that promotes food allergy. J. Allergy Clin. Immunol. 2019;144:157–170. doi: 10.1016/j.jaci.2019.01.043. [DOI] [PubMed] [Google Scholar]
- 364.Zimmermann P., Messina N., Mohn W.W., Finlay B.B., Curtis N. Association between the intestinal microbiota and allergic sensitization, eczema, and asthma: A systematic review. J. Allergy Clin. Immunol. 2019;143:467–485. doi: 10.1016/j.jaci.2018.09.025. [DOI] [PubMed] [Google Scholar]
- 365.McCauley K.E., Rackaityte E., LaMere B., Fadrosh D.W., Fujimura K.E., Panzer A.R., Lin D.L., Lynch K.V., Halkias J., Mendoza V.F., et al. Heritable vaginal bacteria influence immune tolerance and relate to early-life markers of allergic sensitization in infancy. Cell Rep. Med. 2022;3:100713. doi: 10.1016/j.xcrm.2022.100713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Okba A.M., Saber S.M., Abdel-Rehim A.S., Amin M.M., Mohamed D.A. Fecal microbiota profile in atopic asthmatic adult patients. Eur. Ann. Allergy Clin. Immunol. 2018;50:117–124. doi: 10.23822/EurAnnACI.1764-1489.48. [DOI] [PubMed] [Google Scholar]
- 367.Lundstrom S.L., Yang J., Kallberg H.J., Thunberg S., Gafvelin G., Haeggstrom J.Z., Gronneberg R., Grunewald J., van Hage M., Hammock B.D., et al. Allergic asthmatics show divergent lipid mediator profiles from healthy controls both at baseline and following birch pollen provocation. PLoS ONE. 2012;7:e33780. doi: 10.1371/journal.pone.0033780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Fujimura K.E., Sitarik A.R., Havstad S., Lin D.L., Levan S., Fadrosh D., Panzer A.R., LaMere B., Rackaityte E., Lukacs N.W., et al. Neonatal gut microbiota associates with childhood multisensitized atopy and T cell differentiation. Nat. Med. 2016;22:1187–1191. doi: 10.1038/nm.4176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Levan S.R., Stamnes K.A., Lin D.L., Panzer A.R., Fukui E., McCauley K., Fujimura K.E., McKean M., Ownby D.R., Zoratti E.M., et al. Elevated faecal 12,13-diHOME concentration in neonates at high risk for asthma is produced by gut bacteria and impedes immune tolerance. Nat. Microbiol. 2019;4:1851–1861. doi: 10.1038/s41564-019-0498-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 370.Abdulla O.A., Neamah W., Sultan M., Alghetaa H.K., Singh N., Busbee P.B., Nagarkatti M., Nagarkatti P. The Ability of AhR Ligands to Attenuate Delayed Type Hypersensitivity Reaction Is Associated with Alterations in the Gut Microbiota. Front. Immunol. 2021;12:684727. doi: 10.3389/fimmu.2021.684727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 371.Roduit C., Frei R., Ferstl R., Loeliger S., Westermann P., Rhyner C., Schiavi E., Barcik W., Rodriguez-Perez N., Wawrzyniak M., et al. High levels of butyrate and propionate in early life are associated with protection against atopy. Allergy. 2019;74:799–809. doi: 10.1111/all.13660. [DOI] [PubMed] [Google Scholar]
- 372.Cait A., Hughes M.R., Antignano F., Cait J., Dimitriu P.A., Maas K.R., Reynolds L.A., Hacker L., Mohr J., Finlay B.B., et al. Microbiome-driven allergic lung inflammation is ameliorated by short-chain fatty acids. Mucosal Immunol. 2018;11:785–795. doi: 10.1038/mi.2017.75. [DOI] [PubMed] [Google Scholar]
- 373.Thorburn A.N., McKenzie C.I., Shen S., Stanley D., Macia L., Mason L.J., Roberts L.K., Wong C.H., Shim R., Robert R., et al. Evidence that asthma is a developmental origin disease influenced by maternal diet and bacterial metabolites. Nat. Commun. 2015;6:7320. doi: 10.1038/ncomms8320. [DOI] [PubMed] [Google Scholar]
- 374.Tan J., McKenzie C., Vuillermin P.J., Goverse G., Vinuesa C.G., Mebius R.E., Macia L., Mackay C.R. Dietary Fiber and Bacterial SCFA Enhance Oral Tolerance and Protect against Food Allergy through Diverse Cellular Pathways. Cell Rep. 2016;15:2809–2824. doi: 10.1016/j.celrep.2016.05.047. [DOI] [PubMed] [Google Scholar]
- 375.Arifuzzaman M., Won T.H., Li T.T., Yano H., Digumarthi S., Heras A.F., Zhang W., Parkhurst C.N., Kashyap S., Jin W.B., et al. Inulin fibre promotes microbiota-derived bile acids and type 2 inflammation. Nature. 2022;611:578–584. doi: 10.1038/s41586-022-05380-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 376.Seki D., Mayer M., Hausmann B., Pjevac P., Giordano V., Goeral K., Unterasinger L., Klebermass-Schrehof K., De Paepe K., Van de Wiele T., et al. Aberrant gut-microbiota-immune-brain axis development in premature neonates with brain damage. Cell Host Microbe. 2021;29:1558–1572. doi: 10.1016/j.chom.2021.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 377.Ouabbou S., He Y., Butler K., Tsuang M. Inflammation in Mental Disorders: Is the Microbiota the Missing Link? Neurosci. Bull. 2020;36:1071–1084. doi: 10.1007/s12264-020-00535-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 378.Slyepchenko A., Carvalho A.F., Cha D.S., Kasper S., McIntyre R.S. Gut emotions—Mechanisms of action of probiotics as novel therapeutic targets for depression and anxiety disorders. CNS Neurol. Disord. Drug Targets. 2014;13:1770–1786. doi: 10.2174/1871527313666141130205242. [DOI] [PubMed] [Google Scholar]
- 379.Zhang X., Hou Y., Li Y., Wei W., Cai X., Shao H., Yuan Y., Zheng X. Taxonomic and Metabolic Signatures of Gut Microbiota for Assessing the Severity of Depression and Anxiety in Major Depressive Disorder Patients. Neuroscience. 2022;496:179–189. doi: 10.1016/j.neuroscience.2022.06.024. [DOI] [PubMed] [Google Scholar]
- 380.Galley J.D., Mashburn-Warren L., Blalock L.C., Lauber C.L., Carroll J.E., Ross K.M., Hobel C., Coussons-Read M., Dunkel Schetter C., Gur T.L. Maternal anxiety, depression and stress affects offspring gut microbiome diversity and bifidobacterial abundances. Brain Behav. Immun. 2023;107:253–264. doi: 10.1016/j.bbi.2022.10.005. [DOI] [PubMed] [Google Scholar]
- 381.O′Neill S.M., Curran E.A., Dalman C., Kenny L.C., Kearney P.M., Clarke G., Cryan J.F., Dinan T.G., Khashan A.S. Birth by Caesarean Section and the Risk of Adult Psychosis: A Population-Based Cohort Study. Schizophr. Bull. 2016;42:633–641. doi: 10.1093/schbul/sbv152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 382.Morais L.H., Golubeva A.V., Casey S., Scott K.A., Ramos Costa A.P., Moloney G.M., Dinan T.G., Cryan J.F. Early-life oxytocin attenuates the social deficits induced by caesarean-section delivery in the mouse. Neuropsychopharmacology. 2021;46:1958–1968. doi: 10.1038/s41386-021-01040-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Lynch C.M.K., Cowan C.S.M., Bastiaanssen T.F.S., Moloney G.M., Theune N., van de Wouw M., Florensa Zanuy E., Ventura-Silva A.P., Codagnone M.G., Villalobos-Manriquez F., et al. Critical windows of early-life microbiota disruption on behaviour, neuroimmune function, and neurodevelopment. Brain Behav. Immun. 2022;108:309–327. doi: 10.1016/j.bbi.2022.12.008. [DOI] [PubMed] [Google Scholar]
- 384.Li J., Pu F., Peng C., Wang Y., Zhang Y., Wu S., Wang S., Shen X., Li Y., Cheng R., et al. Antibiotic cocktail-induced gut microbiota depletion in different stages could cause host cognitive impairment and emotional disorders in adulthood in different manners. Neurobiol. Dis. 2022;170:105757. doi: 10.1016/j.nbd.2022.105757. [DOI] [PubMed] [Google Scholar]
- 385.Cuskelly A., Hoedt E.C., Harms L., Talley N.J., Tadros M.A., Keely S., Hodgson D.M. Neonatal immune challenge influences the microbiota and behaviour in a sexually dimorphic manner. Brain Behav. Immun. 2022;103:232–242. doi: 10.1016/j.bbi.2022.04.023. [DOI] [PubMed] [Google Scholar]
- 386.Lee J., Venna V.R., Durgan D.J., Shi H., Hudobenko J., Putluri N., Petrosino J., McCullough L.D., Bryan R.M. Young versus aged microbiota transplants to germ-free mice: Increased short-chain fatty acids and improved cognitive performance. Gut Microbes. 2020;12:1814107. doi: 10.1080/19490976.2020.1814107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Leonardi I., Gao I.H., Lin W.Y., Allen M., Li X.V., Fiers W.D., De Celie M.B., Putzel G.G., Yantiss R.K., Johncilla M., et al. Mucosal fungi promote gut barrier function and social behavior via Type 17 immunity. Cell. 2022;185:831–846. doi: 10.1016/j.cell.2022.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Allahverdiyev A., Tari G., Bagirova M., Abamor E.S. Current Approaches in Development of Immunotherapeutic Vaccines for Breast Cancer. J. Breast Cancer. 2018;21:343–353. doi: 10.4048/jbc.2018.21.e47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 389.Johnson L.A., June C.H. Driving gene-engineered T cell immunotherapy of cancer. Cell Res. 2017;27:38–58. doi: 10.1038/cr.2016.154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Ribas A., Wolchok J.D. Cancer immunotherapy using checkpoint blockade. Science. 2018;359:1350–1355. doi: 10.1126/science.aar4060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Sharma P., Hu-Lieskovan S., Wargo J.A., Ribas A. Primary, Adaptive, and Acquired Resistance to Cancer Immunotherapy. Cell. 2017;168:707–723. doi: 10.1016/j.cell.2017.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Routy B., Le Chatelier E., Derosa L., Duong C.P.M., Alou M.T., Daillere R., Fluckiger A., Messaoudene M., Rauber C., Roberti M.P., et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science. 2018;359:91–97. doi: 10.1126/science.aan3706. [DOI] [PubMed] [Google Scholar]
- 393.Chaput N., Lepage P., Coutzac C., Soularue E., Le Roux K., Monot C., Boselli L., Routier E., Cassard L., Collins M., et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann. Oncol. 2017;28:1368–1379. doi: 10.1093/annonc/mdx108. [DOI] [PubMed] [Google Scholar]
- 394.Sivan A., Corrales L., Hubert N., Williams J.B., Aquino-Michaels K., Earley Z.M., Benyamin F.W., Lei Y.M., Jabri B., Alegre M.L., et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science. 2015;350:1084–1089. doi: 10.1126/science.aac4255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.Matson V., Fessler J., Bao R., Chongsuwat T., Zha Y., Alegre M.L., Luke J.J., Gajewski T.F. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science. 2018;359:104–108. doi: 10.1126/science.aao3290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 396.Zheng Y., Wang T., Tu X., Huang Y., Zhang H., Tan D., Jiang W., Cai S., Zhao P., Song R., et al. Gut microbiome affects the response to anti-PD-1 immunotherapy in patients with hepatocellular carcinoma. J. Immunother. Cancer. 2019;7:193. doi: 10.1186/s40425-019-0650-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 397.Jin Y., Dong H., Xia L., Yang Y., Zhu Y., Shen Y., Zheng H., Yao C., Wang Y., Lu S. The Diversity of Gut Microbiome is Associated with Favorable Responses to Anti-Programmed Death 1 Immunotherapy in Chinese Patients With NSCLC. J. Thorac. Oncol. 2019;14:1378–1389. doi: 10.1016/j.jtho.2019.04.007. [DOI] [PubMed] [Google Scholar]
- 398.Li L., Ye J. Characterization of gut microbiota in patients with primary hepatocellular carcinoma received immune checkpoint inhibitors: A Chinese population-based study. Medicine. 2020;99:e21788. doi: 10.1097/MD.0000000000021788. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 399.Coutzac C., Jouniaux J.M., Paci A., Schmidt J., Mallardo D., Seck A., Asvatourian V., Cassard L., Saulnier P., Lacroix L., et al. Systemic short chain fatty acids limit antitumor effect of CTLA-4 blockade in hosts with cancer. Nat. Commun. 2020;11:2168. doi: 10.1038/s41467-020-16079-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Zhang S.L., Han B., Mao Y.Q., Zhang Z.Y., Li Z.M., Kong C.Y., Wu Y., Chen G.Q., Wang L.S. Lacticaseibacillus paracasei sh2020 induced antitumor immunity and synergized with anti-programmed cell death 1 to reduce tumor burden in mice. Gut Microbes. 2022;14:2046246. doi: 10.1080/19490976.2022.2046246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 401.Chen Y., Liu Y., Wang Y., Chen X., Wang C., Chen X., Yuan X., Liu L., Yang J., Zhou X. Prevotellaceae produces butyrate to alleviate PD-1/PD-L1 inhibitor-related cardiotoxicity via PPARalpha-CYP4X1 axis in colonic macrophages. J. Exp. Clin. Cancer Res. 2022;41:1. doi: 10.1186/s13046-021-02201-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Raad M.A., Chams N.H., Sharara A.I. New and Evolving Immunotherapy in Inflammatory Bowel Disease. Inflamm. Intest. Dis. 2016;1:85–95. doi: 10.1159/000445986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 403.Kaper J.B., Nataro J.P., Mobley H.L. Pathogenic Escherichia coli. Nat. Rev. Microbiol. 2004;2:123–140. doi: 10.1038/nrmicro818. [DOI] [PubMed] [Google Scholar]
- 404.Russo T.A., Johnson J.R. Medical and economic impact of extraintestinal infections due to Escherichia coli: Focus on an increasingly important endemic problem. Microbes Infect. 2003;5:449–456. doi: 10.1016/S1286-4579(03)00049-2. [DOI] [PubMed] [Google Scholar]
- 405.Collotta D., Hull W., Mastrocola R., Chiazza F., Cento A.S., Murphy C., Verta R., Alves G.F., Gaudioso G., Fava F., et al. Baricitinib counteracts metaflammation, thus protecting against diet-induced metabolic abnormalities in mice. Mol. Metab. 2020;39:101009. doi: 10.1016/j.molmet.2020.101009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Li Q., Ding X., Liu Y., Marcella C., Dai M., Zhang T., Bai J., Xiang L., Wen Q., Cui B., et al. Fecal Microbiota Transplantation is a Promising Switch Therapy for Patients with Prior Failure of Infliximab in Crohn′s Disease. Front. Pharmacol. 2021;12:658087. doi: 10.3389/fphar.2021.658087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Xiao F., Dong F., Li X., Li Y., Yu G., Liu Z., Wang Y., Zhang T. Bifidobacterium longum CECT 7894 Improves the Efficacy of Infliximab for DSS-Induced Colitis via Regulating the Gut Microbiota and Bile Acid Metabolism. Front. Pharmacol. 2022;13:902337. doi: 10.3389/fphar.2022.902337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Lee J.W.J., Plichta D., Hogstrom L., Borren N.Z., Lau H., Gregory S.M., Tan W., Khalili H., Clish C., Vlamakis H., et al. Multi-omics reveal microbial determinants impacting responses to biologic therapies in inflammatory bowel disease. Cell Host Microbe. 2021;29:1294–1304. doi: 10.1016/j.chom.2021.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Sanchis-Artero L., Martinez-Blanch J.F., Manresa-Vera S., Cortes-Castell E., Valls-Gandia M., Iborra M., Paredes-Arquiola J.M., Bosca-Watts M., Huguet J.M., Gil-Borras R., et al. Evaluation of changes in intestinal microbiota in Crohn′s disease patients after anti-TNF alpha treatment. Sci. Rep. 2021;11:10016. doi: 10.1038/s41598-021-88823-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.Ding N.S., McDonald J.A.K., Perdones-Montero A., Rees D.N., Adegbola S.O., Misra R., Hendy P., Penez L., Marchesi J.R., Holmes E., et al. Metabonomics and the Gut Microbiome Associated with Primary Response to Anti-TNF Therapy in Crohn′s Disease. J. Crohn’s Colitis. 2020;14:1090–1102. doi: 10.1093/ecco-jcc/jjaa039. [DOI] [PubMed] [Google Scholar]
- 411.Serfaty L., De Leusse A., Rosmorduc O., Desaint B., Flejou J.F., Chazouilleres O., Poupon R.E., Poupon R. Ursodeoxycholic acid therapy and the risk of colorectal adenoma in patients with primary biliary cirrhosis: An observational study. Hepatology. 2003;38:203–209. doi: 10.1053/jhep.2003.50311. [DOI] [PubMed] [Google Scholar]
- 412.Shah S.A., Volkov Y., Arfin Q., Abdel-Latif M.M., Kelleher D. Ursodeoxycholic acid inhibits interleukin 1 beta [corrected] and deoxycholic acid-induced activation of NF-kappaB and AP-1 in human colon cancer cells. Int. J. Cancer. 2006;118:532–539. doi: 10.1002/ijc.21365. [DOI] [PubMed] [Google Scholar]
- 413.Shen Y., Lu C., Song Z., Qiao C., Wang J., Chen J., Zhang C., Zeng X., Ma Z., Chen T., et al. Ursodeoxycholic acid reduces antitumor immunosuppression by inducing CHIP-mediated TGF-beta degradation. Nat. Commun. 2022;13:3419. doi: 10.1038/s41467-022-31141-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Grover M., Kashyap P.C. Germ-free mice as a model to study effect of gut microbiota on host physiology. Neurogastroenterol. Motil. 2014;26:745–748. doi: 10.1111/nmo.12366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Coyte K.Z., Rakoff-Nahoum S. Understanding Competition and Cooperation within the Mammalian Gut Microbiome. Curr. Biol. 2019;29:R538–R544. doi: 10.1016/j.cub.2019.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Lehtimaki J., Thorsen J., Rasmussen M.A., Hjelmso M., Shah S., Mortensen M.S., Trivedi U., Vestergaard G., Bonnelykke K., Chawes B.L., et al. Urbanized microbiota in infants, immune constitution, and later risk of atopic diseases. J. Allergy Clin. Immunol. 2021;148:234–243. doi: 10.1016/j.jaci.2020.12.621. [DOI] [PubMed] [Google Scholar]
- 417.Arnesen H., Hitch T.C.A., Steppeler C., Muller M.H.B., Knutsen L.E., Gunnes G., Angell I.L., Ormaasen I., Rudi K., Paulsen J.E., et al. Naturalizing laboratory mice by housing in a farmyard-type habitat confers protection against colorectal carcinogenesis. Gut Microbes. 2021;13:1993581. doi: 10.1080/19490976.2021.1993581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 418.Johnson J.S., Spakowicz D.J., Hong B.Y., Petersen L.M., Demkowicz P., Chen L., Leopold S.R., Hanson B.M., Agresta H.O., Gerstein M., et al. Evaluation of 16S rRNA gene sequencing for species and strain-level microbiome analysis. Nat. Commun. 2019;10:5029. doi: 10.1038/s41467-019-13036-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 419.Aguiar-Pulido V., Huang W., Suarez-Ulloa V., Cickovski T., Mathee K., Narasimhan G. Metagenomics, Metatranscriptomics, and Metabolomics Approaches for Microbiome Analysis. Evol. Bioinform. Online. 2016;12:5–16. doi: 10.4137/EBO.S36436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 420.Dias C.K., Starke R., Pylro V.S., Morais D.K. Database limitations for studying the human gut microbiome. PeerJ Comput. Sci. 2020;6:e289. doi: 10.7717/peerj-cs.289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 421.Porras A.M., Shi Q., Zhou H., Callahan R., Montenegro-Bethancourt G., Solomons N., Brito I.L. Geographic differences in gut microbiota composition impact susceptibility to enteric infection. Cell Rep. 2021;36:109457. doi: 10.1016/j.celrep.2021.109457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Long L.L., Svenson K.L., Mourino A.J., Michaud M., Fahey J.R., Waterman L., Vandegrift K.L., Adams M.D. Shared and distinctive features of the gut microbiome of C57BL/6 mice from different vendors and production sites, and in response to a new vivarium. Lab. Anim. 2021;50:185–195. doi: 10.1038/s41684-021-00777-0. [DOI] [PubMed] [Google Scholar]
- 423.Zheng D., Liwinski T., Elinav E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020;30:492–506. doi: 10.1038/s41422-020-0332-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.