Abstract
Human papillomavirus (HPV) infection detected in oropharyngeal squamous cell carcinoma (OPSCC) is associated with a better survival outcome from previous literature. However, Thailand and several Asian countries have a low prevalence of HPV-associated OPSCC and, therefore, have a low positive rate of immunostaining with p16. Tumor microenvironments (TME), including tumor-infiltrating CD8+ lymphocytes (CD8+ TIL) and programmed death ligand 1 (PD-L1), are proposed as significant prognostic indicators in addition to p16. Objectives: Explore the expression p16, CD8+ TIL, and PD-L1 and its value as prognostic indicators for overall survival (OS) in patients with OPSCC. Materials and Methods: Data from patients with OPSCC diagnosed from 2012 to 2018 were recovered from medical records and national registry. All available glass slides and slides of immunohistochemistry (IHC) of p16, CD8, and PD-L1 were reviewed. The TME was classified into four types according to the expression pattern of PD-L1 and CD8+TIL. Overall survival (OS) was assessed using the Kaplan–Meier method and Cox regression model analysis. Results: In 160 OPSCC patients, p16 was positive in 27 (16.88%). The density of CD8+ TIL was higher in the p16+ and PD-L1+ groups (p = 0.005, 0.039); however, there was no association between p16 and the status of PD-L1. P16 and CD8+ TIL were significant prognostic factors for better OS (p = 0.007, 0.001), but not PD-L1 status (p = 0.317). Among the four types of TME, carcinoma showed mainly type IV TME (PD-L1−/TIL+), while OPSCCs with type I TME (PD-L1+/TIL+) had the best survival outcome. Conclusions: The positivity of p16 and the density of CD8+ TIL were associated with better OS in OPSCC, while the status of PD-L1 was not significantly related to OS. OPSCC with type I TME (PD-L1+/TIL+) showed the best prognosis of all types of TME.
Keywords: oropharyngeal squamous cell carcinoma (OPSCC), survival, human papillomavirus (HPV), p16, CD8, PD-L1
1. Introduction
Oropharyngeal squamous cell carcinoma (OPSCC) has been in the spotlight with a rapidly increasing incidence over the past decade. Evidence shows that high-risk human papillomavirus (HPV) infection is a causative pathogen of OPSCC and has a favorable prognostic value for patients with OPSCC [1]. The attributable fraction for HPV is greater than 60% in the United States and 40% in Europe [2,3]. However, in Thailand, the reported prevalence of high-risk HPV infection in OPSCC is only about 8.7–15.6% [4,5,6,7].
Although HPV-driven tumorigenesis is associated with the chronic inflammatory process of viral infection; the host immune responses are possibly different from smoking carcinogenesis. The immune system plays a vital role in tumor eradication, so cancer cells develop mechanisms to avoid detection or dysregulate the immune system. These mechanisms include the development of tolerance to T cells, the alteration of HLA class I, the inhibition of inflammatory cytokines, and the evasion of the immune checkpoint [8]. Thus, components of the tumor microenvironment (TME), such as lymphocytes, macrophages, or immune checkpoints, are believed to play an essential role in the inhibition and/or development of cancer cells [9,10].
CD8+ lymphocytes are cytotoxic T-lymphocytes that function as an antigen-specific immune response. These T-lymphocytes play a role in the tumor microenvironment by increasing antitumor immune responses. In HPV-associated cancers, many studies have observed a high density of tumor-infiltrating CD8+ lymphocytes (CD8+ TIL), and an increase in CD8+ TIL leads to better OS (overall survival) outcomes [11,12,13].
Programmed death protein 1 (PD-1) is a transmembrane receptor expressed by T cells, B cells, monocytes, and dendritic cells, which plays a role in the immune checkpoint cascade. Binding of PD-1 to its ligands, the programmed death ligand 1 (PD-L1) on tumor cells, helps them escape immune surveillance [8]. Tumor PD-L1 is associated with various prognoses [14,15,16]. The expression of PD-L1 on both cancer cells and immune cells is shown to be associated with survival outcomes and responses to immune checkpoint inhibitors [16,17]. In particular, the expression is controlled by many factors related to oncogenic pathways or inflammatory pathways such as IFN- γ [18,19]. The up-regulation of PD-L1 on tumor cells in the oropharynx is an adaptive immune response during chronic viral infection [15,20]. In some studies, high expression of PD-L1 is observed in HPV-associated cancers [16], with malignant transformation in deep tonsillar crypts where HPV infection often occurs [20].
The low prevalence of HPV-associated OPSCC results in a low positive rate of p16 expression and a higher discordant rate between p16 and PCR for HPV DNA when compared to western countries [7], so the role of p16 as the sole prognostic factor may be less significant compared to areas with a high prevalence of HPV. Ang et al. originally suggested a risk classification based on HPV and smoking statuses [21]. A low prevalence of HPV and a high rate of smoking in the Thai population may result in fewer patients in the low-risk group, so the classification using only these two factors may not fully reflect the risk of death in this setting. This study aims to demonstrate the role of CD8+TIL and PD-L1 as additional prognostic factors for survival outcomes in a setting with a low prevalence of HPV.
2. Materials and Methods
2.1. Study Population and Design
Patients with OPSCC aged 18 years or older treated at Siriraj Hospital between January 2012 and December 2018 were included. Patients with all stages of the disease were included, except for recurrent cases, regardless of the treatment received. Most of the patients received definitive chemoradiation, followed by radiation alone and surgery with adjuvant chemoradiation. Patient data was collected from electronic medical records. The study endpoint was the OS outcome, defined as the time from diagnosis to death from any cause or the end of the study period on 30 April 2020. The date of death was acquired from the Thai national registry. All available glass slides and the additional IHC staining of p16, CD8, and PD-L1 performed in formalin-fixed paraffin-embedded tumor blocks were reviewed and evaluated by a pathologist. This study was approved by the Siriraj Institutional Review Board, (EC2) 336/2562.
2.2. Immunohistochemical Method
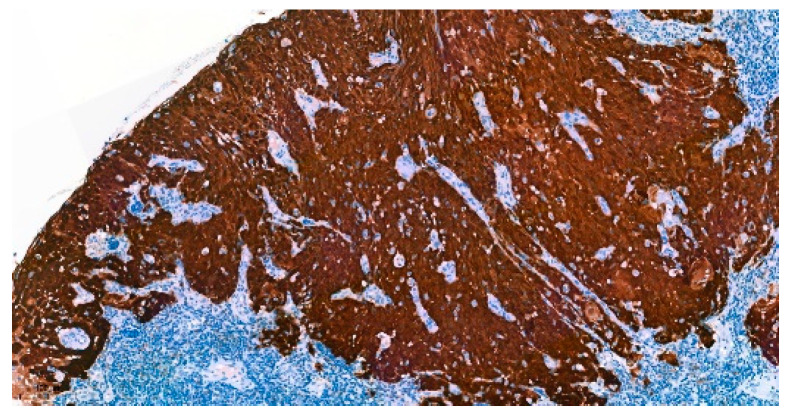
Formalin-fixed paraffin-embedded blocks of tumor specimens were analyzed for IHC of p16, CD8, and PD-L1. Paraffin-embedded tumor sections were deparaffinized and incubated with monoclonal antibodies. IHC of p16 was performed using a mouse monoclonal primary antibody against p16INK4a (CINtec® Histology, Ventana, AZ, USA) (Figure 1). The positive expression of p16 was interpreted with strong and diffuse nuclear and cytoplasmic staining (block staining) ≥ 70% of tumor cells [22].
Figure 1.
Immunohistochemistry of p16-positive OPSCC (100×).
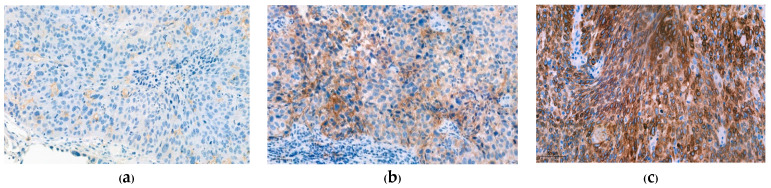
CD8 IHC was performed using CD8 (C8/144B, Cell Marque) mouse monoclonal primary antibody. Expression of CD8 was calculated as the percentage of CD8+ lymphocytes infiltrating the tumor divided by the total number of tumor cells and classified into four groups (Figure 2): CD8 ≥ 10%, CD8 ≥ 5% but < 10%, CD8 ≥ 1% but < 5%, and CD8 < 1% [11].
Figure 2.
Immunohistochemical staining for CD8 (100×). (a) CD8 ≥ 1% but < 5%; (b) CD8 ≥ 5% but <10%; (c) CD8 ≥ 10%.
PD-L1 expression in a tissue microarray by IHC used a mouse IgG antibody against PD-L1 (22C3, PD-L1 IHC 22C3 pharmDx). The expression of PD-L1 was evaluated using the Combined Positive Score (CPS) and classified into three groups: CPS < 1, CPS ≥ 1 but < 20, and CPS ≥ 20, according to the interpretation manual (Figure 3). Staining intensity was also visually assessed and manually scored as 1+ (weak intensity), 2+ (moderate intensity), and 3+ (strong intensity).
Figure 3.
Immunohistochemical staining for PD-L1 (200×). (a) CPS ≥ 1 but < 20 with weak intensity (1+); (b) CPS ≥ 20 with moderate intensity (2+); (c) CPS ≥ 20 with strong intensity (3+).
2.3. Tumor Microenvironment
Tumor microenvironments were classified into 4 types as suggested by Teng et al. by the status of PD-L1 and CD8+TIL which was type I (PD-L1+/TIL+, adaptive immune resistance), type II (PD-L1−/TIL−, immunological ignorance), type III (PD-L1+/TIL−, intrinsic induction), and type IV (PD-L1−/TIL+, tolerance) [23]. PD-L1+ was defined as CPS ≥ 1 and TIL+ was defined as CD8 ≥ 1% in this study.
2.4. Statistical Analysis
Baseline characteristics were presented as mean ± standard deviation (SD) for continuous data and number (percentage) for categorical data. Continuous data was compared using t tests. Chi-square and Fisher’s exact tests were applied to the categorical data. OS rates were assessed using the Kaplan–Meier method, with a comparison between the two groups using the log-rank test. Cox proportional-hazards models were used to estimate the hazard ratio (HR). Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) version 22.0 software. A p-value < 0.05 was considered statistically significant.
3. Results
3.1. Immunohistochemical Analysis
160 patients met the inclusion criteria and were enrolled in this study. Of these, immunohistochemical staining of p16 was positive in 27 patients (16.88%). The results of CD8+TIL density and the Combined Positive Score (CPS) of PD-L1 expression are shown in Table 1. The usual cut-off point for pembrolizumab treatment was CPS ≥ 1, which represented 29.1% of the study groups. However, none of the patients in this study were treated with immune checkpoint inhibitors. PD-L1 intensity was assessed in all cases with CPS ≥ 1.
Table 1.
IHC results.
| CD8+ TIL, n(%) | (n = 159) |
| <1% | 59 (37.1) |
| ≥1%, <5% | 54 (34.0) |
| ≥5%, <10% | 19 (11.9) |
| ≥10% | 27 (17.0) |
| PD-L1: CPS, n(%) | (n = 158) |
| CPS < 1 | 112 (70.9) |
| CPS ≥ 1, < 20 | 31 (19.6) |
| CPS ≥ 20 | 15 (9.5) |
| PD-L1: Intensity, n(%) | (n = 46) |
| 1+ (weak) | 24 (52.2) |
| 2+ (moderate) | 19 (41.3) |
| 3+ (strong) | 3 (6.5) |
The high density of CD8+ TIL was associated with positivity of p16 expression (p = 0.005) and PD-L1 expression (p = 0.039). However, the expression of PD-L1 was not statistically associated with the status of p16 (p = 0.596), as shown in Table 2.
Table 2.
Association between p16, CD8+ TIL, and PD-L1.
| p16− | p16+ | p-Value | PD-L1, CPS < 1 | PD-L1, CPS ≥ 1 | p-Value | |
|---|---|---|---|---|---|---|
| CD8+ TIL, n(%) | 0.005 * | 0.039 * | ||||
| <1% | 51 (38.6) | 8 (29.6) | 48 (42.9) | 11 (23.9) | ||
| ≥1%, <5% | 50 (37.9) | 4 (14.8) | 39 (34.8) | 15 (32.6) | ||
| ≥5%, <10% | 14 (10.6) | 5 (18.5) | 11 (9.8) | 8 (17.4) | ||
| ≥10% | 17 (12.9) | 10 (37.0) | 14 (12.5) | 12 (26.1) | ||
| PD-L1, n(%) | 0.596 | |||||
| CPS < 1 | 94 (71.8) | 18 (66.7) | ||||
| CPS ≥ 1 | 37 (28.2) | 9 (33.3) |
* p-Value < 0.05.
3.2. Patient Characteristics
Most of the patients (93.75%) were men and 91.08% were smokers. Of these, 7.01% had no history of smoking or alcohol consumption. Furthermore, patients with p16+ OPSCC were younger than those with p16− as expected (p = 0.045). High CD8+ TIL and positive PD-L1 were associated with a lower rate of metastasis (p = 0.040, 0.035). The baseline characteristics according to each IHC are shown in Table 3.
Table 3.
Relationships between patient characteristics and IHC.
| p16− (n = 133) |
p16+ (n = 27) |
p-Value | CD8+ TIL < 1% (n = 59) |
CD8+ TIL ≥ 1% (n = 100) | p-Value | PD-L1; CPS < 1 (n = 112) | PD-L1; CPS ≥ 1 (n = 46) | p-Value | |
|---|---|---|---|---|---|---|---|---|---|
| Age | 0.045 * | 0.754 | 0.445 | ||||||
| Mean ± SD | 61.12 ± 11.18 | 56.19 ± 13.42 | 60.58 ± 11.87 | 59.97 ± 11.61 | 59.73 ± 12.03 | 61.30 ± 10.94 | |||
| Sex, n (%) | 0.066 | 0.325 | 0.156 | ||||||
| Male | 127 (95.5) | 23 (85.2) | 57 (96.6) | 92 (92.0) | 107 (95.5) | 41 (89.1) | |||
| Female | 6 (4.5) | 4 (14.8) | 2 (3.4) | 8 (8.0) | 5 (4.5) | 5 (10.9) | |||
| Smoking status, n (%) | 0.058 | 0.015 * | 0.223 | ||||||
| Yes | 122 (93.1) | 21 (80.8) | 57 (98.3) | 85 (86.7) | 103 (92.8) | 38 (86.4) | |||
| No | 9 (6.9) | 5 (19.2) | 1 (1.7) | 13 (13.3) | 8 (7.2) | 6 (13.6) | |||
| Alcohol use, n (%) | 0.094 | 0.28 | 0.302 | ||||||
| Yes | 84 (66.7) | 11 (44.0) | 39 (70.9) | 55 (57.9) | 71 (67.0) | 23 (53.5) | |||
| Social drinking | 16 (12.7) | 6 (24.0) | 6 (10.9) | 16 (16.8) | 14 (13.2) | 8 (18.6) | |||
| Never | 26 (20.6) | 8 (32.0) | 10 (18.2) | 24 (25.3) | 21 (19.8) | 12 (27.9) | |||
| Subsite, n (%) | 0.081 | 0.139 | 0.132 | ||||||
| Base of tongue | 60 (45.1) | 11 (40.7) | 27 (45.8) | 43 (43.0) | 44 (39.3) | 26 (56.5) | |||
| Tonsil | 49 (36.8) | 15 (55.6) | 19 (32.2) | 45 (45.0) | 48 (42.9) | 15 (32.6) | |||
| Others | 24 (18.0) | 1 (3.7) | 13 (22.0) | 12 (12.0) | 20 (17.9) | 5 (10.9) | |||
| Pathological grading, n (%) | 0.698 | 0.815 | 0.069 | ||||||
| Well diff. | 18 (13.6) | 3 (13.6) | 9 (15.3) | 12 (12.8) | 16 (15.1) | 5 (10.9) | |||
| Moderately diff. | 88 (66.7) | 13 (59.1) | 39 (66.1) | 61 (64.9) | 73 (68.9) | 26 (56.5) | |||
| Poorly diff. | 26 (19.7) | 6 (27.3) | 11 (18.6) | 21 (22.3) | 17 (16.0) | 15 (32.6) | |||
| T stage, n (%) | 0.144 | 0.317 | 0.89 | ||||||
| T1 | 10 (7.5) | 6 (22.2) | 6 (10.2) | 10 (10.0) | 12 (10.7) | 4 (8.7) | |||
| T2 | 28 (21.1) | 5 (18.5) | 8 (13.6) | 25 (25.0) | 22 (19.6) | 10 (21.7) | |||
| T3 | 52 (39.1) | 9 (33.3) | 23 (39.0) | 37 (37.0) | 41 (36.6) | 19 (41.3) | |||
| T4 | 43 (32.3) | 7 (25.9) | 22 (37.3) | 28 (28.0) | 37 (33.0) | 13 (28.3) | |||
| N stage, n (%) | 0.007 * | 0.304 | 0.554 | ||||||
| N0 | 27 (20.3) | 4 (14.8) | 16 (27.1) | 15 (15.0) | 23 (20.5) | 8 (17.4) | |||
| N1 | 19 (14.3) | 11 (40.7) | 11 (18.6) | 19 (19.0) | 24 (21.4) | 6 (13.0) | |||
| N2 | 62 (46.6) | 6 (22.2) | 22 (37.3) | 45 (45.0) | 44 (39.3) | 22 (47.8) | |||
| N3 | 25 (18.8) | 6 (22.2) | 10 (16.9) | 21 (21.0) | 21 (18.8) | 10 (21.7) | |||
| M stage, n (%) | 0.213 | 0.040 * | 0.035 * | ||||||
| M0 | 122 (91.7) | 27 (100.0) | 52 (88.1) | 97 (97.0) | 102 (91.1) | 46 (100.0) | |||
| M1 | 11 (8.3) | 0 (0.0) | 7 (11.9) | 3 (3.0) | 10 (8.9) | 0 (0.0) | |||
| Stage (AJCC 8th), n (%) | <0.001 * | 0.954 | 0.578 | ||||||
| I | 3 (2.3) | 6 (22.2) | 4 (6.8) | 5 (5.0) | 8 (7.1) | 1 (2.2) | |||
| II | 9 (6.8) | 10 (37.0) | 7 (11.9) | 12 (12.0) | 12 (10.7) | 7 (15.2) | |||
| III | 24 (18.0) | 11 (40.7) | 12 (20.3) | 23 (23.0) | 25 (22.3) | 10 (21.7) | |||
| IV | 97 (72.9) | 0 (0.0) | 36 (61.0) | 60 (60.0) | 67 (59.8) | 28 (60.9) | |||
| Stage (AJCC 7th), n (%) | 0.368 | 0.461 | 0.62 | ||||||
| I | 3 (2.3) | 2 (7.4) | 3 (5.1) | 2 (2.0) | 5 (4.5) | 0 (0.0) | |||
| II | 9 (6.8) | 1 (3.7) | 5 (8.5) | 5 (5.0) | 7 (6.3) | 3 (6.5) | |||
| III | 24 (18.0) | 3 (11.1) | 8 (13.6) | 19 (19.0) | 20 (17.9) | 7 (15.2) | |||
| IV | 97 (72.9) | 21 (77.8) | 43 (72.9) | 74 (74.0) | 80 (71.4) | 36 (78.3) |
* p-Value < 0.05.
3.3. Survival Outcomes
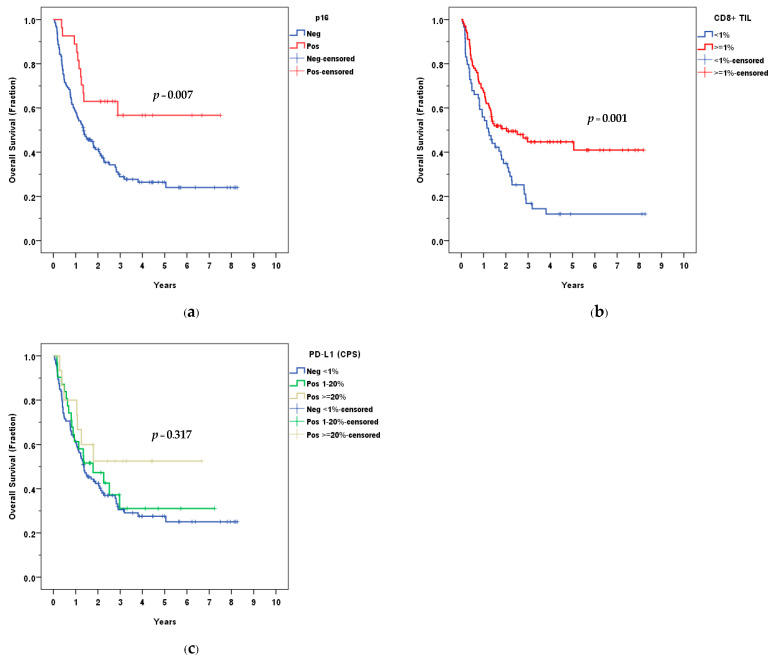
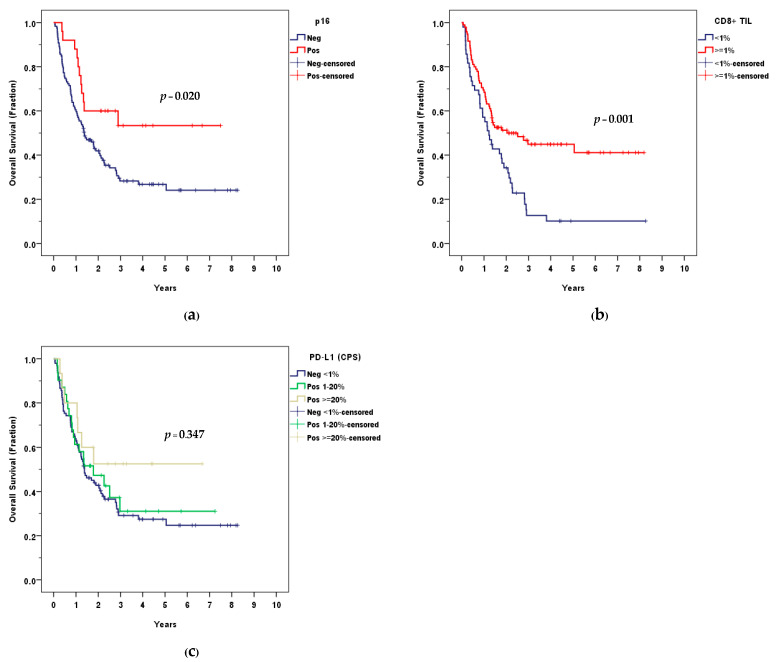
The median follow-up time was 1.38 years (range, 0.06–8.26), 1.34 years in p16− OPSCC, and 2.43 years in p16+ OPSCC. Kaplan–Meier analysis yielded a three-year OS of 28.9% and 56.7% for p16− and p16+ patients, respectively. The difference in OS between both groups was statistically significant (p = 0.007).
The median survival time was 1.41 years (95% CI, 0.88–1.95). The median survival time was 1.35 years (95% CI, 0.87–1.83) in the p16−group, while no median survival was reached in the p16+ group. The Kaplan–Meier survival curve is shown in Figure 4a.
Figure 4.
Overall survival (OS) according to: (a) p16; (b) CD8+ TIL; (c) PD-L1 (CPS).
OS was not significantly different according to the level of expression of PD-L1 (p = 0.317) (Figure 4c); while the high density of CD8+ TIL was associated with better OS, using a percentage of CD8+ TIL at ≥1%, ≥ 5%, and ≥10% as cut-off points (p = 0.001, 0.009, 0.006). The OS between CD8+ TIL < 1% and ≥1% was demonstrated in Figure 4b. When analyzing only the p16– group, a high density of CD8+ TIL was associated with better OS using ≥1% as the cut-off point (p = 0.015).
A subgroup analysis excluding patients in stage I (AJCC 7th) and patients with distant metastases was performed. Similar to the findings from the overall study population, the positivity of p16 and the high density of CD8+ TIL were associated with better OS (p = 0.020, 0.001). Again, OS was not significantly different according to the level of expression of PD-L1 (p = 0.347). The Kaplan–Meier survival curve is shown in Appendix A (Figure A1).
High stages, negative p16, and low CD8+ TIL density were predictors of poor OS outcomes in multivariate analysis (HR 2.43, 95% CI 1.11–5.32, p = 0.026; HR 1.98, 95% CI 1.05–3.71, p = 0.034; HR 1.77, 95% CI 1.18–2.67, p = 0.006) as shown in Table 4.
Table 4.
Cox regression model for overall survival (OS).
| Univariate HR (95% CI) | p-Value | Adjusted HR (95% CI) | p-Value | |
|---|---|---|---|---|
| Smoking status: yes (former/current) | 3.46 (1.27–9.41) | 0.015 * | 2.48 (0.90–6.82) | 0.079 |
| Stage: III-IV | 2.18 (1.01–4.70) | 0.047 * | 2.43 (1.11–5.32) | 0.026 * |
| p16: negative | 2.32 (1.24–4.33) | 0.009 * | 1.98 (1.05–3.71) | 0.034 * |
| CD8+ TIL: low density | 1.89 (1.28–2.78) | 0.001 * | 1.77 (1.18–2.67) | 0.006 * |
| PD-L1: negative | 1.33 (0.85–2.07) | 0.211 | 1.13 (0.71–1.80) | 0.603 |
* p-Value < 0.05.
3.4. Tumor Microenvironment
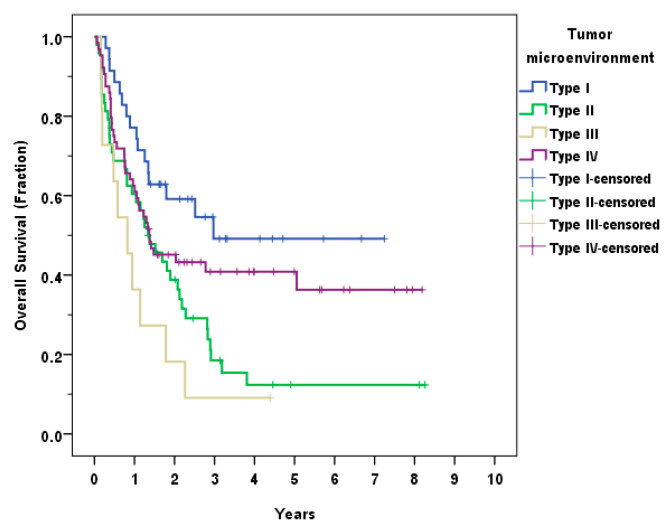
The patients were classified into four groups according to the TME as follows: type I (PD-L1+/TIL+, adaptive immune resistance) 35 (22.2%), type II (PD-L1−/TIL−, immunological ignorance) 48 (30.4%), type III (PD-L1+/TIL−, intrinsic induction) 11 (7.0%), and type IV (PD-L1−/TIL+, tolerance) 64 (40.5%). The OS differed significantly between the four types (p = 0.004). The group with the best survival outcome was type I, followed by type IV, II and III, respectively. The Kaplan–Meier analysis of the TME types is shown in Figure 5.
Figure 5.
Overall survival (OS) according to the type of tumor microenvironment (TME).
4. Discussion
The role of TME has been widely studied in recent years, especially for OPSCC, where HPV is known to be one of the causal factors. HPV-associated OPSCC demonstrated favorable survival and treatment outcomes in the previous literature [21,24,25]. The favorable survival outcome was also observed in recurrent or metastatic head and neck squamous cell carcinoma (HNSCC) [26]. The higher density of TILs was observed in HPV+ OPSCC; the TIL is a part of adaptive immunity and has a protective effect against tumor activity, as evidenced by a better outcome in the high TIL subgroup of patients with OPSCC [27,28]. However, similar to reports from many Asian countries, the prevalence of HPV+ OPSCC in Thailand was considerably lower compared to those in the United States and European countries. In our study, the rate of p16+ from tumor specimens was 16.88%, while OS was better in the p16+ group, similar to other previous studies.
Despite a low p16+ OPSCC in this study, the high density of CD8+ TIL was still associated with a favorable survival outcome and a lower rate of distant metastases. Additionally, there was a significant association between CD8+ TIL density and positive expression of p16. All OPSCCs with a higher CD8+ TIL density had a better OS than those with a lower CD8+ TIL, as well as in the p16− OPSCC subgroup. Previous studies found that the role of TIL in survival outcome and response to chemoradiation was demonstrated in HPV+ OPSCC, but there is still controversy about HPV- OPSCC [27,29,30]. Our results showing good OS in patients with high CD8+ TIL suggested that TIL could be a predictive prognostic factor, especially in the low-HPV-prevalence population. The role of TIL in cancer treatment has attracted research interests [31]. For example, the CheckRad-CD8 trial showed a promising result of induction chemoimmunotherapy and radioimmunotherapy (RIT) using a combination of durvalumab and tremelimumab in patients with an increase in CD8+ immune cells [32]. However, more studies are needed to assess the efficacy of immune checkpoint inhibitors based on the TIL.
Several immune checkpoints regulate T cell activity, including PD-1/PD-L1. Anti-PD-1, such as pembrolizumab and nivolumab, have been approved by the FDA to treat recurrent and metastatic HNSCC [9]. Tumor PD-L1 was associated with various prognoses in previous studies. For example, a meta-analysis by Yang et al. [33] found no significant differences in OS and progression-free survival (PFS) between PD-L1 positive and negative OPSCC; however, low CD8+ TIL HNSCC and worse OS was observed in the PD-L1 positive subgroup. However, another meta-analysis by Tang et al. [16] in the OPSCC subgroup did not show a significant correlation between PD-L1 expression and OS but better disease-free survival (DFS) in patients with higher expression of PD-L1. Our study showed 29.1% positive PD-L1 expression using a cut-off CPS ≥ 1 but found no statistical difference in OS between the positive and negative subgroups. However, there was a trend towards a better OS in the PD-L1 positive group.
The relationship of PD-L1 with TIL and HPV is also equivocal; some evidence demonstrated an association between PD-L1 and TIL or HPV [16,34,35]. Positive PD-L1 was associated with high CD8+ TIL in our study, but no association with p16 was observed. Despite the proven benefit of anti-PD-1 in those with recurrent and metastatic HNSCC [36], the prognostic value of PD-L1 IHC for survival was still controversial. Furthermore, since the rate of positive expression of PD-L1 in this study was remarkably low, further studies should be suggested to ratify the clinical outcomes of anti-PD-1 treatment in the Thai population.
Classification of TME into four types based on PD-L1 and TIL was suggested by Teng et al. [23] in 2015 and was applied to several cancer studies later. Our study showed the best survival outcome in type I TME (PD-L1+/TIL+) similar to the findings in melanoma, as well as in HNSCC, which showed the most favorable prognosis in type I (PD-L1+/TIL+) and type IV (PD-L1−/TIL+) [23,34,35,37]. The results suggested that type I and IV had better results according to TIL status, highlighting the importance of the immune response in tumor control.
We acknowledge some limitations in our study. The study is retrospective and has a short median follow-up time. The HPV-positivity status was not confirmed by PCR. However, the discordant rate of p16 and PCR for HPV DNA was less than 25% [7]. Moreover, due to the limitations of our medical database, we could not report other types of survival outcomes apart from OS.
Biomarkers that reflect tumor cell proliferation, such as p53 or Ki-67 (MIB-1), have been investigated in other studies with controversial results on survival and recurrent outcomes, although some reported Ki-67 as an unfavorable prognostic factor [38,39]. Some studies also showed an association between these biomarkers and cancer aggressiveness [40,41]. We did not examine the role of proliferative markers in this study. Nevertheless, it is another field to be explored.
Our results suggested a benefit of the integration of p16 and TIL in predicting survival outcomes and, possibly with further studies, treatment response. Therefore, we postulated that additional IHC staining of TILs would provide additional prognostic information on patient outcomes, especially in areas with low HPV and settings where HPV validation could not be performed. More studies are needed to confirm its value in the prediction of clinical response and to identify other potential prognostic biomarkers.
5. Conclusions
The prevalence of HPV-associated OPSCC in Thailand is low. Positive p16 and high CD8+ TIL density were associated with better OS in OPSCC patients, while PD-L1 status was not significantly related to OS. Of all types of TME, the adaptive immune resistance phenotype (type I, PD-L1+/TIL+) provided the best prognosis.
Acknowledgments
The authors acknowledge Chulaluk Komoltri for her assistance in statistical analysis and Jeerapa Kerdnoppakhun for her assistance in manuscript preparation.
Appendix A
Figure A1.
Overall survival (OS) of a subgroup analysis without patients in stage I and patients with distant metastases according to: (a) p16; (b) CD8+ TIL; (c) PD-L1 (CPS).
Author Contributions
Conceptualization, K.A., N.L., J.P., N.R. and W.P.; data curation, K.A., N.L., J.P., N.R. and W.P.; formal analysis, K.A., N.L., J.P., N.R. and W.P.; funding acquisition, W.P.; investigation, K.A., N.L., J.P., N.R. and W.P.; methodology, K.A., N.L., J.P., N.R. and W.P.; project administration, K.A., N.L., J.P., N.R. and W.P.; resources, N.L., J.P., N.R. and W.P.; supervision, N.L., J.P., N.R. and W.P.; validation, K.A., N.L., J.P., N.R. and W.P.; visualization, K.A., N.L. and W.P.; writing—original draft preparation, K.A., N.L. and N.R.; writing—review and editing, J.P. and W.P. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
This study was conducted according to the Declaration of Helsinki and was approved by the Siriraj Institutional Review Board, (EC2) 336/2562.
Informed Consent Statement
Patient consent was waived due to the retrospective nature of the study and the analysis used anonymous clinical data.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy and ethical issues.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This study was funded by the Health Systems Research Institute (grant number 63-140).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Wang M.B., Liu I.Y., Gornbein J.A., Nguyen C.T. HPV-Positive Oropharyngeal Carcinoma: A Systematic Review of Treatment and Prognosis. Otolaryngol.-Head Neck Surg. Off. J. Am. Acad. Otolaryngol.-Head Neck Surg. 2015;153:758–769. doi: 10.1177/0194599815592157. [DOI] [PubMed] [Google Scholar]
- 2.National Comprehensive Cancer Network Head and Neck Cancers (Version 2.2018) [(accessed on 22 June 2018)]. Available online: https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf.
- 3.Mehanna H., Beech T., Nicholson T., El-Hariry I., McConkey C., Paleri V., Roberts S. Prevalence of human papillomavirus in oropharyngeal and nonoropharyngeal head and neck cancer--systematic review and meta-analysis of trends by time and region. Head Neck. 2013;35:747–755. doi: 10.1002/hed.22015. [DOI] [PubMed] [Google Scholar]
- 4.Nopmaneepaisarn T., Tangjaturonrasme N., Rawangban W., Vinayanuwattikun C., Keelawat S., Bychkov A. Low preva-lence of p16-positive HPV-related head-neck cancers in Thailand: Tertiary referral center experience. BMC Cancer. 2019;19:1050. doi: 10.1186/s12885-019-6266-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chotipanich A., Siriarechakul S., Mungkung O.O. Role of high-risk human papillomavirus in the etiology of oral and oro-pharyngeal cancers in Thailand: A case-control study. SAGE Open Med. 2018;6:2050312118765604. doi: 10.1177/2050312118765604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pongsapich W., Jotikaprasardhna P., Lianbanchong C., Phumchan A., Siritantikorn S., Chongkolwatana C. Human Papil-lomavirus Infection in Oral Cavity and Oropharyngeal Cancers: Are They the Same Story? J. Med. Assoc. Thail. Chotmaihet Thangphaet. 2016;99:684–690. [PubMed] [Google Scholar]
- 7.Arsa L., Siripoon T., Trachu N., Foyhirun S., Pangpunyakulchai D., Sanpapant S., Jinawath N., Pattaranutaporn P., Jinawath A., Ngamphaiboon N. Discrepancy in p16 expression in patients with HPV-associated head and neck squamous cell carcinoma in Thailand: Clinical characteristics and survival outcomes. BMC Cancer. 2021;21:504. doi: 10.1186/s12885-021-08213-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ferris R.L. Immunology and Immunotherapy of Head and Neck Cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2015;33:3293–3304. doi: 10.1200/JCO.2015.61.1509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lechien J.R., Descamps G., Seminerio I., Furgiuele S., Dequanter D., Mouawad F., Badoual C., Journe F., Saussez S. HPV Involvement in the Tumor Microenvironment and Immune Treatment in Head and Neck Squamous Cell Carcinomas. Cancers. 2020;12:1060. doi: 10.3390/cancers12051060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Welters M.J.P., Santegoets S.J., van der Burg S.H. The Tumor Microenvironment and Immunotherapy of Oropharyngeal Squamous Cell Carcinoma. Front. Oncol. 2020;10:545385. doi: 10.3389/fonc.2020.545385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fukushima Y., Someya M., Nakata K., Hori M., Kitagawa M., Hasegawa T., Tsuchiya T., Gocho T., Ikeda H., Hirohashi Y., et al. Influence of PD-L1 expression in immune cells on the response to radiation therapy in patients with oropharyngeal squamous cell carcinoma. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 2018;129:409–414. doi: 10.1016/j.radonc.2018.08.023. [DOI] [PubMed] [Google Scholar]
- 12.Ou D., Adam J., Garberis I., Blanchard P., Nguyen F., Levy A., Casiraghi O., Gorphe P., Breuskin I., Janot F., et al. Clinical relevance of tumor infiltrating lymphocytes, PD-L1 expression and correlation with HPV/p16 in head and neck cancer treated with bio- or chemo-radiotherapy. Oncoimmunology. 2017;6:e1341030. doi: 10.1080/2162402X.2017.1341030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Russell S., Angell T., Lechner M., Liebertz D., Correa A., Sinha U., Kokot N., Epstein A. Immune cell infiltration patterns and survival in head and neck squamous cell carcinoma. Head Neck Oncol. 2013;5:24. [PMC free article] [PubMed] [Google Scholar]
- 14.Solomon B., Young R.J., Rischin D. Head and neck squamous cell carcinoma: Genomics and emerging biomarkers for im-munomodulatory cancer treatments. Pt 2Semin. Cancer Biol. 2018;52:228–240. doi: 10.1016/j.semcancer.2018.01.008. [DOI] [PubMed] [Google Scholar]
- 15.De Meulenaere A., Vermassen T., Aspeslagh S., Huvenne W., Van Dorpe J., Ferdinande L., Rottey S. Turning the tide: Clinical utility of PD-L1 expression in squamous cell carcinoma of the head and neck. Oral Oncol. 2017;70:34–42. doi: 10.1016/j.oraloncology.2017.05.002. [DOI] [PubMed] [Google Scholar]
- 16.Tang H., Zhou X., Ye Y., Zhou Y., Wu C., Xu Y. The different role of PD-L1 in head and neck squamous cell carcinomas: A meta-analysis. Pathol. Res. Pract. 2020;216:152768. doi: 10.1016/j.prp.2019.152768. [DOI] [PubMed] [Google Scholar]
- 17.Kim H.R., Ha S.J., Hong M.H., Heo S.J., Koh Y.W., Choi E.C., Kim E.K., Pyo K.H., Jung I., Seo D., et al. PD-L1 expression on immune cells, but not on tumor cells, is a favorable prognostic factor for head and neck cancer pa-tients. Sci. Rep. 2016;6:36956. doi: 10.1038/srep36956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yi M., Niu M., Xu L., Luo S., Wu K. Regulation of PD-L1 expression in the tumor microenvironment. J. Hematol. Oncol. 2021;14:10. doi: 10.1186/s13045-020-01027-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zerdes I., Matikas A., Bergh J., Rassidakis G.Z., Foukakis T. Genetic, transcriptional and post-translational regulation of the programmed death protein ligand 1 in cancer: Biology and clinical correlations. Oncogene. 2018;37:4639–4661. doi: 10.1038/s41388-018-0303-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lyford-Pike S., Peng S., Young G.D., Taube J.M., Westra W.H., Akpeng B., Bruno T.C., Richmon J.D., Wang H., Bishop J.A., et al. Evidence for a role of the PD-1:PD-L1 pathway in immune resistance of HPV-associated head and neck squamous cell carcinoma. Cancer Res. 2013;73:1733–1741. doi: 10.1158/0008-5472.CAN-12-2384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ang K.K., Harris J., Wheeler R., Weber R., Rosenthal D.I., Nguyen-Tân P.F., Westra W.H., Chung C.H., Jordan R.C., Lu C., et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N. Engl. J. Med. 2010;363:24–35. doi: 10.1056/NEJMoa0912217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lewis J.S., Jr., Beadle B., Bishop J.A., Chernock R.D., Colasacco C., Lacchetti C., Moncur J.T., Rocco J.W., Schwartz M.R., Seethala R.R., et al. Human Papillomavirus Testing in Head and Neck Carcinomas: Guideline From the College of American Pathologists. Arch. Pathol. Lab. Med. 2018;142:559–597. doi: 10.5858/arpa.2017-0286-CP. [DOI] [PubMed] [Google Scholar]
- 23.Teng M.W., Ngiow S.F., Ribas A., Smyth M.J. Classifying Cancers Based on T-cell Infiltration and PD-L1. Cancer Res. 2015;75:2139–2145. doi: 10.1158/0008-5472.CAN-15-0255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fakhry C., Westra W.H., Li S., Cmelak A., Ridge J.A., Pinto H., Forastiere A., Gillison M.L. Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J. Natl. Cancer Inst. 2008;100:261–269. doi: 10.1093/jnci/djn011. [DOI] [PubMed] [Google Scholar]
- 25.Ragin C.C., Taioli E. Survival of squamous cell carcinoma of the head and neck in relation to human papillomavirus infection: Review and meta-analysis. Int. J. Cancer. 2007;121:1813–1820. doi: 10.1002/ijc.22851. [DOI] [PubMed] [Google Scholar]
- 26.Argiris A., Li S., Ghebremichael M., Egloff A.M., Wang L., Forastiere A.A., Burtness B., Mehra R. Prognostic significance of human papillomavirus in recurrent or metastatic head and neck cancer: An analysis of Eastern Cooperative Oncology Group trials. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2014;25:1410–1416. doi: 10.1093/annonc/mdu167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Näsman A., Romanitan M., Nordfors C., Grün N., Johansson H., Hammarstedt L., Marklund L., Munck-Wikland E., Da-lianis T., Ramqvist T. Tumor infiltrating CD8+ and Foxp3+ lymphocytes correlate to clinical outcome and human papilloma-virus (HPV) status in tonsillar cancer. PLoS ONE. 2012;7:e38711. doi: 10.1371/journal.pone.0038711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Saber C.N., Grønhøj Larsen C., Dalianis T., von Buchwald C. Immune cells and prognosis in HPV-associated oropharyngeal squamous cell carcinomas: Review of the literature. Oral Oncol. 2016;58:8–13. doi: 10.1016/j.oraloncology.2016.04.004. [DOI] [PubMed] [Google Scholar]
- 29.Nordfors C., Grün N., Tertipis N., Ährlund-Richter A., Haeggblom L., Sivars L., Du J., Nyberg T., Marklund L., Munck-Wikland E., et al. CD8+ and CD4+ tumour infiltrating lymphocytes in relation to human papillomavirus status and clinical outcome in tonsillar and base of tongue squamous cell carcinoma. Eur. J. Cancer. 2013;49:2522–2530. doi: 10.1016/j.ejca.2013.03.019. [DOI] [PubMed] [Google Scholar]
- 30.Wansom D., Light E., Thomas D., Worden F., Prince M., Urba S., Chepeha D., Kumar B., Cordell K., Eisbruch A., et al. Infiltrating lymphocytes and human papilloma-virus-16--associated oropharyngeal cancer. Laryngoscope. 2012;122:121–127. doi: 10.1002/lary.22133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Raskov H., Orhan A., Christensen J.P., Gögenur I. Cytotoxic CD8(+) T cells in cancer and cancer immunotherapy. Br. J. Cancer. 2021;124:359–367. doi: 10.1038/s41416-020-01048-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hecht M., Eckstein M., Rutzner S., von der Grün J., Illmer T., Klautke G., Laban S., Hautmann M.G., Brunner T.B., Ta-maskovics B., et al. Induction chemoimmunotherapy followed by CD8+ immune cell-based patient selection for chemotherapy-free radioimmunotherapy in locally advanced head and neck cancer. J. Immunother. Cancer. 2022;10:e003747. doi: 10.1136/jitc-2021-003747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yang W.F., Wong M.C.M., Thomson P.J., Li K.Y., Su Y.X. The prognostic role of PD-L1 expression for survival in head and neck squamous cell carcinoma: A systematic review and meta-analysis. Oral Oncol. 2018;86:81–90. doi: 10.1016/j.oraloncology.2018.09.016. [DOI] [PubMed] [Google Scholar]
- 34.Balermpas P., Rödel F., Krause M., Linge A., Lohaus F., Baumann M., Tinhofer I., Budach V., Sak A., Stuschke M., et al. The PD-1/PD-L1 axis and human papilloma virus in patients with head and neck cancer after adjuvant chemoradiotherapy: A multicentre study of the German Cancer Consortium Radiation Oncology Group (DKTK-ROG) Int. J. Cancer. 2017;141:594–603. doi: 10.1002/ijc.30770. [DOI] [PubMed] [Google Scholar]
- 35.Sanchez-Canteli M., Granda-Díaz R., Del Rio-Ibisate N., Allonca E., López-Alvarez F., Agorreta J., Garmendia I., Montuenga L.M., García-Pedrero J.M., Rodrigo J.P. PD-L1 expression correlates with tumor-infiltrating lymphocytes and better prognosis in patients with HPV-negative head and neck squamous cell carcinomas. Cancer Immunol. Immunother. CII. 2020;69:2089–2100. doi: 10.1007/s00262-020-02604-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Huang Z., Zheng S., Ding S., Wei Y., Chen C., Liu X., Li H., Xia Y. Prognostic role of programmed cell death ligand-1 ex-pression in head and neck cancer treated with programmed cell death protein-1/programmed cell death ligand-1 inhibitors: A meta-analysis based on clinical trials. J. Cancer Res. Ther. 2021;17:676–687. doi: 10.4103/jcrt.JCRT_1606_20. [DOI] [PubMed] [Google Scholar]
- 37.De Meulenaere A., Vermassen T., Aspeslagh S., Deron P., Duprez F., Laukens D., Van Dorpe J., Ferdinande L., Rottey S. Tumor PD-L1 status and CD8(+) tumor-infiltrating T cells: Markers of improved prognosis in oropharyngeal cancer. Oncotarget. 2017;8:80443–80452. doi: 10.18632/oncotarget.19045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sittel C., Ruiz S., Volling P., Kvasnicka H.M., Jungehülsing M., Eckel H.E. Prognostic significance of Ki-67 (MIB1), PCNA and p53 in cancer of the oropharynx and oral cavity. Oral Oncol. 1999;35:583–589. doi: 10.1016/S1368-8375(99)00041-X. [DOI] [PubMed] [Google Scholar]
- 39.Liu J., Zhang M., Rose B., Veillard A.S., Jones D., Zhang X., Soon Lee C., Milross C., Hong A. Ki67 Expression has Prognostic Significance in Relation to Human Papillomavirus Status in Oropharyngeal Squamous Cell Carcinoma. Ann. Surg. Oncol. 2015;22:1893–1900. doi: 10.1245/s10434-014-4237-x. [DOI] [PubMed] [Google Scholar]
- 40.Macluskey M., Chandrachud L.M., Pazouki S., Green M., Chisholm D.M., Ogden G.R., Schor S.L., Schor A.M. Apoptosis, proliferation, and angiogenesis in oral tissues. Possible relevance to tumour progression. J. Pathol. 2000;191:368–375. doi: 10.1002/1096-9896(2000)9999:9999<::AID-PATH652>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 41.Mallick S., Breta M., Gupta S.D., Dinda A.K., Mohanty B.K., Singh M.K. Angiogenesis, Proliferative Activity and DNA Ploidy in Oral Verrucous Carcinoma: A Comparative Study Including Verrucous Hyperplasia and Squamous Cell Carcinoma. Pathol. Oncol. Res. POR. 2015;21:1249–1257. doi: 10.1007/s12253-014-9856-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy and ethical issues.