Abstract
The defense mechanism against harmful stimuli is inflammation. Indeed, neurodegenerative disorders can arise as a result of a persistent neuroinflammation. Beta-amyloid (Aβ1-42) is an early trigger in the origination of Alzheimer’s disease, leading to synaptic and cognitive impairments. Virgin olive oil (VOO) is correlated with a decreased risk of developing immune-inflammatory disorders, but the potential effects of the phenolic fraction (PF) from VOO in the modulation of neuroinflammatory processes in neutrophils remain unknown. In this study, we investigated the ability of the PF to modulate the activation of Aβ1-42-stimulated primary human neutrophils, focusing on the expression of gene and surface markers and the release of pro-inflammatory and chemoattractant mediators. Down-regulation of pro-inflammatory cytokine gene expression in Aβ1-42-treated neutrophils, among other changes, was reported. Furthermore, pretreatment with PF prevented neutrophil activation. The beneficial effects in the modulation of inflammatory responses show the relevance of VOO to achieve a healthier diet that can help prevent inflammatory diseases.
Keywords: hydroxytyrosol, immunonutrition, neuroinflammation, neutrophils, olive oil, phenols
1. Introduction
Virgin olive oil (VOO) is the major source of fatty acids in the traditional Mediterranean diet, and its ingestion is associated with a reduced risk of chronic degenerative diseases, including cardiovascular pathologies, immune-inflammatory disorders, and cancer [1,2,3]. The positive effects of VOO have been historically attributed to the extraordinary oleic acid content, but it is also largely recognized that the minor components like those present in the phenolic fraction (PF) may be of biological relevance [4,5]. In fact, the high concentration of phenolic compounds in the PF from VOO has been reported to be a relevant player in the beneficial effects associated with the Mediterranean diet [6]. Some of these phenolic compounds can exert one or more bioactivities, including anti-inflammatory, antioxidant, antimicrobial, antiproliferative, antiarrhythmic, and vasodilatory effects, as well as the capacity to regulate relevant cellular signaling pathways [7]. The Mediterranean diet, characterized by better cardiovascular health due to VOO, is a food model highly recognized by the scientific community and the World Health Organization.
Inflammation is a physiopathological phenomenon involved in the genesis of numerous diseases as a response of the immune system to injurious stimuli such as tissue injury, infection, or toxicants [8,9]. During physiological inflammation, with a primary role in the clearance of extracellular pathogens, neutrophils and monocytes are recruited from the circulation to sites of inflammation, infiltrated into the affected tissues, and then these cells produce oxidants, complement components, Fc receptors, prostaglandins, cytokines, and chemokines that cooperate to achieve healing and to restore homeostasis [10,11]. Upon identifying cellular injuries, immune cells like microglia, astrocytes, and activated T cells can cause neuroinflammation. Alzheimer’s disease (AD) and other neurodegenerative disorders can arise as a result of a persistent neuroinflammation. In this regard, beta-amyloid (Aβ1-42) is an early trigger in the development of AD, leading to synaptic and cognitive deficiency.
As the relationship between VOO and an anti-inflammatory response has been shown [12], it is relevant to unravel the contribution of the PF to these beneficial effects in human neutrophils. This information is key in order to clarify the possible positive impact of phenols during inflammation management and to better understand the complex mechanisms of neuroinflammation and how it is affected by the intake of VOO. The aim of this study was to evaluate whether the PF and its most abundant compound, hydroxytyrosol (HTyr), modulate the activation mediated by Aβ1-42 in human neutrophils, in order to unravel the mechanisms behind the beneficial effects of VOO. For this purpose, the Aβ1-42-induced expression of pro-inflammatory genes and surface markers and the release of pro-inflammatory cytokines in freshly isolated neutrophils from healthy volunteers exposed to the phenols were evaluated.
2. Materials and Methods
2.1. Materials
Hydroxytyrosol (HTyr) was purchased from Sigma-Aldrich. The phenolic fraction was extracted from a VOO (picual variety) that was obtained from Ofade Consulting (Seville, Spain). The extraction was carried out as described elsewhere [13] with some modifications [14], freeze dried, and stored at −80 °C until the analyses were carried out.
2.2. Chemical Characterization of PF
Quantitative and qualitative analysis of PF was carried out following the protocol COI/T20/29doc (International Olive Council). The analysis consists of a methanol extraction succeeded by quantification with high-performance liquid chromatography (HPLC) of the supernatant phase filtered. The HPLC system was equipped with a C18 reverse-phase column (4.6 mm × 25 cm), of type Spherisorb ODS-2 (5 mm), 100 Å, with a spectrophotometric UV detector at 280 nm and integrator. The amount of phenols was determined by measuring the areas of the related chromatographic peaks and expressed as mg of compound/kg of oil.
2.3. Blood Collection and Neutrophil Isolation
The rules of good clinical practice were followed when conducting this investigation. The research followed the guidelines set forth in the World Medical Association’s Helsinki Declaration. Buffy coats provided from the Regional Center for Blood Transfusions and the Tejidos Bank of Seville and Huelva were used to harvest neutrophils (S2200035). Human peripheral blood mononuclear cells were isolated by centrifugation on a gradient with Ficoll (Sigma, Madrid, Spain). Neutrophils were isolated by dextran sedimentation in a Ficoll Histopaque gradient (Sigma-Aldrich, St. Louis, MO, USA) and erythrocytes were removed with hypotonic lysis. RPMI 1640 medium supplemented with L-glutamine, penicillin, streptomycin, and 1% heat-inactivated fetal bovine serum was used to suspend the cells after isolation. Trypan blue exclusion was used to ensure that >97% of the neutrophil preparation was viable.
2.4. Cytotoxicity Assay
Neutrophils seeded in 96-well plates (1 × 105 cells/well) were incubated for 6 hours with or without different PF or HTyr concentrations (25 and 50 µg/mL). Then, the effect on cell viability was analyzed with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) colorimetric assay. Cell survival was measured as the percentage of absorbance compared with untreated control cells.
2.5. Cell Activation and Exposure to PF
Neutrophils were seeded at a density of 3 × 106 cells/mL and exposed to 500 nM of β-amyloid (Aβ1-42, Sigma-Aldrich) in the presence or absence of PF (25 and 50 µg/mL) or HTyr (41 µM) for 6 h.
2.6. Flow Cytometry
Membrane expression of CD16b (PE anti-human CD16b, BD Biosciences, San Jose, CA, USA), CD62L (FITC anti-human CD62L, Miltenyi Biotec, Bisley, UK), CD63 (PE-Cy7 anti-human CD63, BD Biosciences), and CXCR1 (APC anti-human CD181, BD Biosciences) on neutrophils was assessed with flow cytometry (FACS). According to the manufacturer’s instructions, neutrophils (106 cells) were treated with Aβ1-42 for 6 h and then incubated with the corresponding antibodies at room temperature, in the dark, for 15 min. Afterwards, the cells were fixed, and the erythrocytes were lysed with 20× volume of FACS lysing solution (BD Bioscience). At the Research, Technology, and Innovation Center (University of Seville), fluorescence intensity in the resulting suspension was measured with a FACS Canto II cytometer (BD Bioscience) with the support of FACS Canto II cell analyzer software (BD Bioscience). Each sample’s mean fluorescence intensity (MFI) was calculated using 104 counted cells. Forward scatterhigh (FSChigh), side scatterhigh (SSChigh), and CD16 high cells were used to gate neutrophils. Expression levels were shown as MFI after being adjusted for nonspecific binding of isotype control antibodies on donor neutrophils.
2.7. RNA Isolation and qRT-PCR Analysis
Total RNA was also obtained from cells using Trisure Reagent (Bioline GmbH, Berlin, Germany). RNA quality was evaluated with an A260/A280 ratio in a NanoDrop ND-1000 Spectrophotometer (Thermo Fisher Scientific, Wilmington, DE). RNA (1 µg) was subjected to reverse transcription (iScript, BioRad, Madrid, Spain) following the manufacturer’s indications. Ten ng of the resulting cDNA was employed as a template for real-time polymerase chain reaction (PCR) amplifications. The mRNA levels for individual genes were quantified using real-time PCR in a CFX96 system (BioRad). For each PCR reaction, cDNA template was added to Brilliant SYBR green QPCR Supermix (BioRad, CA, USA) containing the primer pairs for either gene or for glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and hypoxanthine phosphoribosyltransferase (HPRT) as housekeeping genes (Table 1). The average threshold cycle (Ct) values of all the triplicate amplification reactions were used to determine the relative mRNA expression of the candidate genes. Using the conventional 2-(ΔΔCt) technique, the magnitude of change in mRNA expression for candidate genes was determined. All data were reported as a percentage of controls after being adjusted to the content of the endogenous reference genes (GAPDH and HPRT).
Table 1.
Sequences of RT-PCR primers for gene expression analysis.
| Target | GenBank Accession Number | Direction | Sequence (5′ → 3′) |
|---|---|---|---|
| TLR4 | NM_138554.4 | Forward Reverse |
TGAGCAGTCGTGCTGGTATC CAGGGCTTTTCTGAGTCGTC |
| MPO | NM_000250 | Forward Reverse |
CAGCCCAGATATACCCCTCA GACAACACAGGCATCACCAC |
| NE | NM_001972.4 | Forward Reverse |
CATCGTGATTCTCCAGCTCA CTCACGAGAGTGCAGACGTT |
| COX-2 | NM_000963 | Forward Reverse |
TCCCATGGGTTGTGTGTTTA AGATCATCTCTGCCTGAGTATCTT |
| IFNγ | NM_000619.2 | Forward Reverse |
TCCCATGGGTTGTGTGTTTA AAGCACCAGGCATGAAATCT |
| IL-1β | NM_000576.2 | Forward Reverse |
CTGTCCTGCGTGTTGAAAGA TTCTGCTTGAGAGGTGCTGA |
| TNF-α | NM_000594.3 | Forward Reverse |
TCCTTCAGACACCCTCAACC AGGCCCCAGTTTGAATTCTT |
| IL-6 | NM_000600.4 | Forward Reverse |
TACCCCCAGGAGAAGATTCC TTTTCTGCCAGTGCCTCTTT |
| IL-8 | NM_000584 | Forward Reverse |
TAGCAAAATTGAGGCCAAGG AAACCAAGGCACAGTGGAAC |
| MMP-1 | NM_ 001145938 | Forward Reverse |
CTGCTTGACCCTCAGAGACC ATGCTGAAACCCTGAAGGTG |
| MMP-3 | NM_002422 | Forward Reverse |
GAGTGTCGGAGTCCAGCTTC GCAGTTTGCTCAGCCTATCC |
| MMP-9 | NM_004994 | Forward Reverse |
CAGGGATCTCCCCTCCTTAG GTCTTGTGGAGGCTTTGAGC |
| PPARγ | NM_005037 | Forward Reverse |
GCTGTGCAGGAGATCACAGA GGGCTCCATAAAGTCACCAA |
| GAPDH | NM_001289745.2 | Forward Reverse |
ACAGTCAGCCGCATCTTCTT ACGACCAAATCCGTTGACTC |
| HPRT | NM_002046.6 | Forward Reverse |
GAGTCAACGGATTTGGTCGT GACAAGCTTCCCGTTCTCAG |
2.8. Quantification of Cytokine Levels
The cytokine levels of IL-1β, IL-6, IL-8, TNF-α, and IFN-γ in cell supernatants were quantified with enzyme-linked immunosorbent assay (ELISA), according to the indications of the provider (Diaclone, Besançon, France).
2.9. Statistical Analysis
Data were assessed using Graph Pad Prism Version 6.1 software (San Diego, CA, USA). The arithmetic mean and standard error of the mean are used to express all values in the figures and text. There were three replicates of each experiment. One-way analysis of variance (ANOVA) was used to determine the statistical significance of any differences in each parameter between the groups, with the Dunnett test serving as the post hoc test. Statistics were considered significant at p values less than 0.05.
3. Results and Discussion
3.1. Characterization of the Phenolic Fraction
The composition of PF is detailed in Table 2. The main components found in the phenol fraction were the aldehyde form of oleuropein aglycone and the dialdehyde form of ligstroside aglycone, followed by HTyr and tyrosol, representing around 67% of the total PF. These results are in line with previous reports aiming to characterize the phenolic fraction of VOO [15]. Phenolic compounds are accumulated during fruit ripening, and both agronomic (i.e., cultivars or environmental factors) and processing factors (e.g., malaxation time) have an impact on their biosynthesis and biotransformation factors [15,16]. For instance, the content of HTyr has been reported to range from 0.28 to 7.57 mg/kg in olive oil in an evaluation of 80 different cultivars [17]. Thus, the content of HTyr in the test item is relatively high. The antioxidant and anti-inflammatory properties of olive phenols were recently reviewed, highlighting the relevance of HTyr [18].
Table 2.
Main composition of PF from VOO using COI/T20/29doc.
| Phenol Composition | µM Phenol (50 µg PF/mL) |
|---|---|
| Hydroxytyrosol | 41.07 |
| Tyrosol | 43.09 |
| Vanillic acid | 5.09 |
| p-Coumaric acid | 3.42 |
| Decarboxymethyl oleuropein aglycone (dialdehyde) = oleacein | 10.27 |
| Tyrosol acetate | 4.97 |
| Decarboxymethyl ligstroside aglycone (dialdehyde) = oleocanthal | 11.33 |
| Pinoresinol | 6.21 |
| Cinnamic acid | 6.89 |
| Acetoxy-pinoresinol | 6.22 |
| Oleuropein aglycone, oxidized aldehyde form | 39.18 |
| Ligstroside aglycone, dialdehyde form | 26.20 |
| Luteolin | 4.18 |
| Apigenin | 0.88 |
In fact, in terms of health properties, the European Food Safety Authority (EFSA) has declared that olive oil phenols contribute to the protection of blood lipids from oxidative stress, referring to 5 mg of HTyr and its derivatives per 20 g of olive oil. The oil evaluated contained 7 mg/20 g; thus, antioxidant activities at least are expected to occur. However, there is not a health claim concerning anti-inflammatory or other properties of VOO’s phenolic compounds because the research until now is still limited.
3.2. Effect of PF and HTyr on Neutrophil Viability
Cell survival was determined with MTT assay, and the viability at all doses assessed was >95% (data not shown). It was observed that PF up to 50 µg/mL and HTyr at 41 µM (equivalent concentration of HTyr found in PF at 50 µg/mL) for 6 h had no adverse effects on neutrophil viability. Considering these results, the rest of the assays were carried out at concentrations of 25 and 50 μg/mL.
3.3. Regulation of Myeloperoxidase, Neutrophil Elastase, and Cyclooxygenase-2 Gene Expression
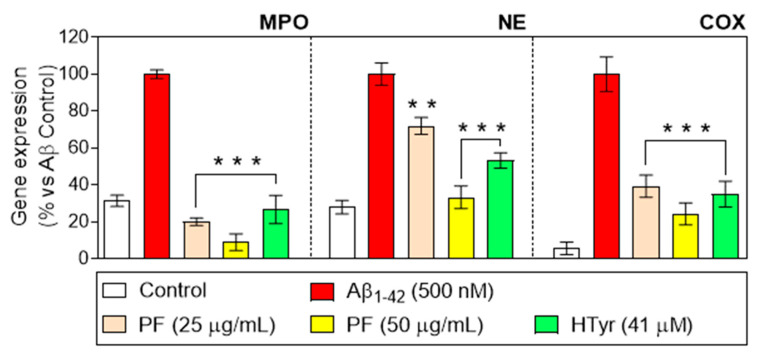
Gene expression following exposure to the PF and pure HTyr was evaluated in the Aβ1-42-activated neutrophils. Aβ1-42-treated neutrophils up-regulated myeloperoxidase (MPO), neutrophil elastase (NE), and cyclooxygenase-2 (COX-2) gene expression compared with untreated neutrophils (Figure 1). PF at both concentrations of 25 and 50 µg/mL, and HTyr at 41 µM, interfered with the enhanced transcriptional activity of MPO, NE, and COX-2 genes in Aβ1-42-treated neutrophils. In the case of PF, the response was observed in a dose-dependent manner. MPO is a local modulator of tissue injury and the subsequent inflammation related to many diseases, which is mostly expressed in neutrophils [19]. In the case of MPO gene expression, the PF led to a decrease lower than the basal levels (control), whereas the HTyr decreased to a lesser extent, indicating that the effects are due to the contribution of different compounds, rather than HTyr being the main contributor.
Figure 1.
Relative gene expression of myeloperoxidase (MPO), neutrophil elastase (NE), and cyclooxygenase-2 (COX-2) in primary human neutrophils stimulated with Aβ1-42 (500 nM), after treatment with the PF from VOO (25 and 50 μg/mL) or hydroxytyrosol (HTyr, 41 μM) for 6 h. Values are means of the three independent experiments in triplicate, with their standard errors represented by vertical bars (** p < 0.01; *** p < 0.001 vs. Aβ1-42-treated cells).
NE, which can be passively liberated or actively produced, has been correlated with the development of several inflammatory diseases [20], whereas cyclooxygenase-2 (COX-2) expression by the neutrophils results in PGE2 synthesis, which may account for alterations in tissue homeostasis. The regulation of NE seems to be more affected by other phenols present in the fraction rather than HTyr, compared with the other markers evaluated, since at 25 µg/mL the decrease, though significant, is not very relevant. The highest decrease is observed with the PF at 50 µg/mL where the levels are comparable to the control; thus, the increased concentration of phenols might be contributing to this effect. The pattern of gene expression found for COX-2 is similar to the other markers but the difference among samples is not very pronounced, suggesting in this case that HTyr is highly contributing to the regulation of COX-2.
Among the phenolic compounds present in the composition of olive oil, for instance, oleocanthal has shown in vitro inhibition of cyclooxygenase in ranges from 7–100 μM from the prostaglandin-biosynthesis pathway, indicating an anti-inflammatory potential similar to ibuprofen [21]. This study shows that the Aβ1-42-mediated increase in MPO expression was effectively prevented by PF and HTyr treatment. According to prior research, the heme enzyme MPO, whose expression is restricted to neutrophils and monocytes, is a key factor in determining how inflammation develops, catalyzing the formation of reactive oxygen intermediates [19]. In addition, MPO is accepted to be a local modulator of tissue injury and the resulting inflammation in many inflammatory diseases. Moreover, in neutrophils COX-2, which leads to pro-oxidant production and tissue damage, contributes to the neutrophils’ self-amplifying recruitment, further amplifying the inflammatory response [22]. In this study, the Aβ1-42-induced increase in the COX-2 gene and protein expression was markedly attenuated by the PF or HTyr treatment, suggesting that the phenolic components from VOO might reduce oxidative damage brought on by an inflammatory stimulation in human neutrophils.
Together with MPO, neutrophil azurophil granules contain high amounts of NE, which has been correlated with the development of several inflammatory diseases [20,23,24]. Under normal physiological conditions, this enzyme is controlled by endogenous serine proteinase, but in the presence of pro-oxidant species such as MPO-generated hypochlorous acid, this feedback mechanism is lost. This study suggests that PF and HTyr also have the capacity to inhibit the gene expression of NE in Aβ1-42-activated human neutrophils, contributing to the response modulation. These results are in line with Czerwińska et al. [25], who reported that oleacein (an isolated compound from the PF of VOO) was able to inhibit NE release in N-formyl-methionyl-leucyl-phenylalanine–stimulated human neutrophils. In addition, animal studies have shown the anti-inflammatory effects of phenols from olive oil, such as differences in the expression of COX-2, mPGES-1, and iNOS protein, and decreases in the levels of circulatory matrix metalloproteinase (MMP)-3 and pro-inflammatory cytokines in collagen-induced arthritis murine models [26]. The results obtained within this research provide a more in-depth insight into the mechanisms behind the immune response and how it is modulated by the VOO’s polyphenols from a commercial sample and specifically for HTyr.
3.4. Down-Regulation of Toll-like Receptor 4 and Pro-Inflammatory Cytokine Gene Expression and Secretion
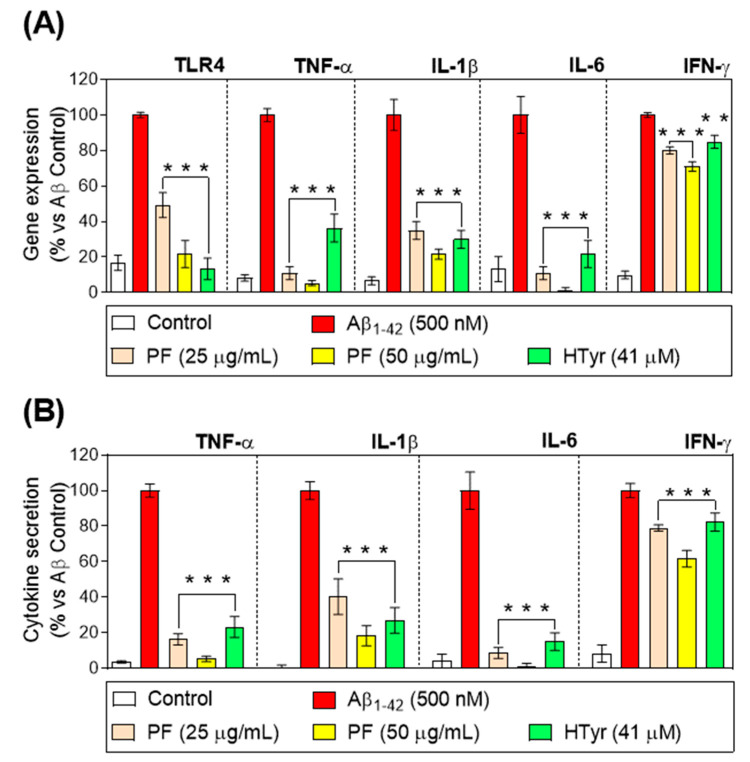
The stimulation of toll-like receptor (TLR) 4 by Aβ1-42 causes the release of vital immunoregulatory and pro-inflammatory cytokines, both of which are necessary to effectively activate the innate immune response. [27]. In this study, an up-regulation of the TLR4 gene expression in Aβ1-42-treated human neutrophils was reported, but this was counteracted by the treatment with PF and HTyr for 6 h (p < 0.001 vs. Aβ1-42-control, Figure 2A). Furthermore, the PF and HTyr significantly decreased the gene expression and production of the pro-inflammatory cytokines TNF-α, IL-1β, IL-6, and IFN-γ at all doses after 6 h (p < 0.001 vs. Aβ1-42-control, Figure 2B). The regulation of TLR4 is suggested to be due to the HTyr, as it was observed that the decrease was more pronounced when the exposure was performed with the isolated compound. This behavior was different for the rest of the markers in Figure 2. For instance, the gene expression of TFN-α and IL-6 is significantly different from the Aβ1-42-treated neutrophils and lower than the decrease effect caused by HTyr. This suggests that the other phenols (Table 2) in the test item are responsible for this counteracting effect. For the other parameters evaluated (IL-1β and IFN-γ) the pattern is similar, where the contribution seems to be balanced among the different phenols, since the HTyr is exerting the same effect as the PF.
Figure 2.
(A) Relative gene expression of TLR-4, tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-6, and interferon (IFN)-γ. (B) Cytokine expression of TNF-α, IL-1β, IL-6, and IFN-γ after the treatment of Aβ1-42-stimulated primary human neutrophils with the PF from VOO (25 and 50 μg/mL) or hydroxytyrosol (HTyr, 41 μM) for 6 h. Values are means of the three independent experiments in triplicate, with their standard errors represented by vertical bars (** p < 0.01; *** p < 0.001 vs. Aβ1-42-treated cells).
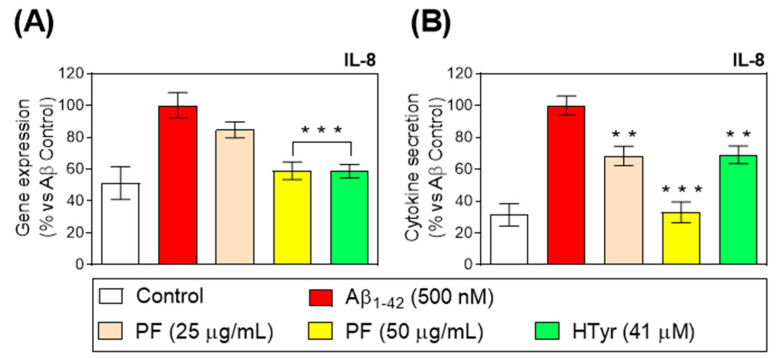
IL-8 was described to be one of the first chemokines triggering neutrophils after secretion by stimulated monocytes [28]. Aβ1-42-treated neutrophils up-regulated IL-8 gene expression and secretion compared with untreated neutrophils (Figure 3). In this study, the treatment of Aβ1-42-stimulated human neutrophils with the PF and HTyr for 6 h at concentrations up to 50 µg/mL (PF) and at 41 µM (HTyr) resulted in a decrease in the IL-8 gene expression and production by neutrophils. Furthermore, the PF at the highest concentration inhibited IL-8 release in a more significant manner than HTyr at 41 μM (p < 0.01; p < 0.001 vs. Aβ1-42-control, Figure 3).
Figure 3.
(A) Relative gene expression and (B) release of IL-8 in Aβ1-42-stimulated primary human neutrophils after treatment with the PF from VOO (25 and 50 μg/mL) or hydroxytyrosol (HTyr, 41 μM) for 6 h. Values are means of the three independent experiments in triplicate, with their standard errors represented by vertical bars (** p < 0.01; *** p < 0.001 vs. Aβ1-42-treated cells).
A great body of evidence has confirmed that human neutrophils are a target but also a source of several pro-inflammatory cytokines [29]. Interestingly, IL-8 is not only the most abundantly secreted cytokine by neutrophils, but also, neutrophils are its primary cellular target [28]. This cytokine activates neutrophils to induce chemotaxis, trigger burst, and degranulation and stimulates neutrophil adhesion to endothelial cells. In the present study, it was found that Aβ1-42-mediated IL-8 gene expression/secretion was markedly attenuated by the treatment with PF and HTyr in human neutrophils. This ability of modulating IL-8 secretion was also previously reported by Czerwińska et al. [25], who found that oleacein, the above-mentioned olive oil polyphenol, inhibited IL-8 secretion in stimulated human neutrophils. Moreover, PF and HTyr abrogated the gene expression of TLR in addition to decreasing the gene expression/secretion of key pro-inflammatory cytokines, including TNF-α, IL-1β, IL-6, and IFN-γ in Aβ1-42-activated human neutrophils and monocytes. In this regard, one of the relevant events during inflammation is leukocyte infiltration, which is regulated by several chemokines for neutrophils and monocytes, whose release is regulated by iNOS-derived NO [30]. In accordance with the aforementioned cytokines data, the treatment with PF (50 µg/mL) or HTyr (41µM) was able to prevent the induced protein expression of iNOS during the Aβ1-42 challenge.
It was recently reported that HTyr (up to 40 mg/kg) exerted antioxidative and anti-inflammatory activities in the splenic tissue after lipopolysaccharide (LPS)-mediated septic response in mice. In this study, animals ingested HTyr for 10 days, and results showed an enhancement of the survival rate and a decrease in the lactate dehydrogenase level in LPS-challenged mice. In addition, the oxidative damage was reduced as well the production levels of TNF-α, IL-1β, and IL-6. In this study, the mRNA expression of iNOS and NO production also increased [31]. Overall, the results reported are in accordance with our results, which explained the mechanisms behind the neutrophils’ role. In addition, a transcriptomic profile analysis reported the effects of HTyr on the whole-genome expression of endothelial cells under resting or pro-inflammatory conditions, resulting in 599 affected genes in IL-1β–stimulated conditions, highlighting the immunological, inflammatory, proliferative, and metabolic pathways affected [32]. These results help to explain how the body system, depending on the health of the subject, is affected at different levels following the ingestion of bioactive compounds.
3.5. Regulation of Metalloproteinases and Peroxisome Proliferator-Activated Receptor-γ Gene Expression
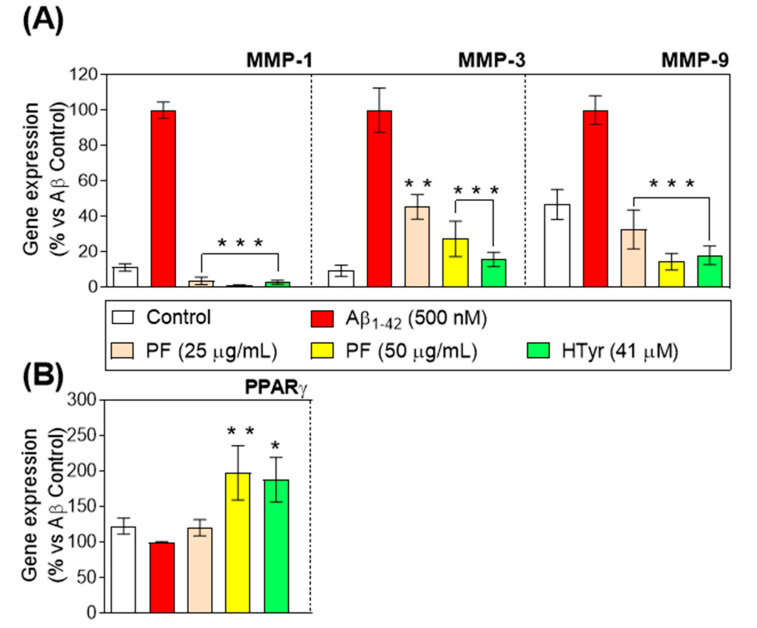
Metalloproteinases (MMPs) modulate the events of immune cell development, which are considered relevant for persistent inflammatory response [33]. Therefore, we investigated the influence of PF and HTyr on the gene expression of three major MMPs (MMP-1, MMP-3, and MMP-9). As peroxisome proliferator–activated receptor (PPAR)-γ agonists have been reported to inhibit MMPs and pro-inflammatory cytokines [34], whether the PF and HTyr can affect PPAR-γ gene expression in Aβ1-42-treated human neutrophils was also investigated. Aβ1-42-treated neutrophils up-regulated MMP gene expression compared with untreated neutrophils. As shown in Figure 4A, the incubation with PF for 6 h produced a significant down-regulation of all MMPs gene expression in Aβ1-42-treated human neutrophils. As can be observed, the effects of olive oil phenols on the expression of MMP could be considered highly biologically relevant, as it counteracts the effect, leading to even lower values of expression compared with the control for MMP-1, whereas the effect on MMP-3 and MMP-9 is a partial decrease. The effect of HTyr is very relevant for MMP-3, whereas in the case of MMP-9, results suggest that the effects are due to the different phenols contained as a whole, rather than because of HTyr exclusively. These findings were further endorsed by a significant up-regulation of PPAR-γ gene expression in Aβ1-42-treated human neutrophils following the incubation with 50 µg/mL of PF and 41 µM of HTyr (p < 0.05; p < 0.001 vs. Aβ1-42-control, respectively, Figure 4B).
Figure 4.
Relative gene expression of (A) MMP-1, MMP-3, and MMP-9 and (B) PPAR-γ in Aβ1-42-stimulated primary human neutrophils treated with the PF from VOO (25 and 50 μg/mL) or hydroxytyrosol (HTyr, 41 μM) for 6 h. Values are means of the three independent experiments in triplicate, with their standard errors represented by vertical bars (* p < 0.05; ** p < 0.01; *** p < 0.001 vs. Aβ1-42-treated cells).
MMPs, especially LPS-induced interstitial collagenase-1 (MMP-1), stromelysin-1 (MMP-3), and gelatinase B (MMP-9), have been described as involved in inflammation via destroying extracellular matrix elements and controlling cytokine signaling by interacting with COX pathways [35,36,37]. These results demonstrate that the PF and HTyr repressed the gene expression of MMP-1, MMP-3, and MMP-9 in human neutrophils, in agreement with other in vitro studies where HTyr, at nutritional concentrations, reduces MMP-9 and COX-2 induction in phorbol-12-myristate-13-acetate (PMA)-activated human monocytes [38]. It was also demonstrated that the PF and HTyr induced PPARγ up-regulation, which, by inhibiting MMP expression in inflamed tissues, plays a crucial part in the dynamic balance between overall matrix synthesis, deposition, and deterioration and affects the expression of pro-inflammatory cytokines [39].
Aβ1-42 is one of the key factors for the development of AD, as it can trigger neuronal inflammation. These results suggest that the modified expression of pro-inflammatory genes and surface markers, as well as the release of pro-inflammatory cytokines due to the phenols from VOO, are exerting a positive effect on the modulation of the physiological process leading to neuronal inflammation, and can be helpful in the management of these diseases.
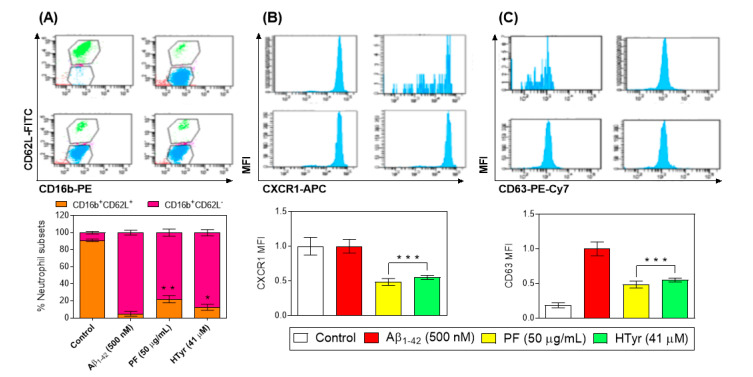
3.6. Olive Oil Phenols Prevent Activation of Human Neutrophils
The neutrophil population in peripheral blood can be discriminated with flow cytometry based on their forward- and side-scatter characteristics. After testing several combinations of antibodies, the best results were obtained by gating the CD16b and CD62L population. When cells were gated, we analyzed them according to their CD16b+ and CD62L- expression. The CD16b + CD62L- population was subjected to other activation markers analysis such as CXCR1 and CD63 (Figure 5). As expected, we found a higher content of activated neutrophils in Aβ1-42 -treated cells (p < 0.001 vs. Aβ1-42-control, Figure 5. Interestingly, we observed a significant decrease after the treatment with HTyr 41 μM compared with PF (50 μg/mL), which does not differ from the control, supporting the evidence that HTyr is one of the main contributors of the bioactivity described for phenols of olive oil.
Figure 5.
Membrane expression of (A) CD16b, (B) CXCR1, and (C) CD63 in Aβ1-42-stimulated primary human neutrophils treated with the PF from VOO (25 and 50 μg/mL) or hydroxytyrosol (HTyr, 41 μM) for 6 h. Values are means of the three independent experiments in triplicate, with their standard errors represented by vertical bars (* p < 0.05; ** p < 0.01; *** p < 0.001 vs. Aβ1-42-treated cells).
The innate immune system’s first line of defense is neutrophils, the most prevalent type of leukocyte in human blood [40]. Once activated, they are complex cells able to perform a substantial range of specific functions, and as effector cells of the innate immune response they are capable of regulating many pathological processes, including autoimmunity, cancer, and chronic inflammation. During inflammatory processes, neutrophils can interplay directly or through cytokines and chemokines with other immune cells to modify both the innate and adaptive immune responses [29]. The inflammatory response relies greatly on neutrophil activation, which entails a variety of different processes that lead to the synthesis of cytokines as well as neutrophil migration into tissues [29,41]. CD16b (FcγRIIIb) is exclusively expressed by human neutrophils and undergoes efficient ectodomain shedding upon neutrophil activation and apoptosis [42]. Moreover, neutrophils’ migration from the circulation into an area of inflammation involves L-selectin (CD62L) that is important in the initial attachment of leukocytes to the endothelium [43]. CXCR1 is a closely related receptor that recognizes CXC chemokines including CXCL8 (or IL-8) and CXCL6, and its down-regulation increases neutrophil adhesion and impairs its migration [44]. NE and other luminal proteins are stored in MPO-positive secretory lysosomes/primary granules of neutrophils, which contain an integral membrane protein, CD63, with an adaptor protein-3-dependent granule delivery system [45]. Once the activated population, based on CD16b++CD62L-CXCR1midCD63++ was established, it was found that the presence of PF and HTyr resulted in decreased frequencies of activated neutrophils when compared with Aβ1-42-activated neutrophils. Together, these results suggest that the anti-inflammatory properties of the PF and HTyr are not restricted to their antioxidant power, but also involve other properties, including the attenuation of neutrophil activation.
Beyond in vitro and animal studies, there are also some human studies reporting the potential anti-inflammatory effects of polyphenol-enriched olive oils. Patti et al. [46] reported differences in several parameters, including pro- and anti-inflammatory cytokines in patients with metabolic syndrome and hepatic steatosis, after the intake of 32 g of the test item during 60 days. The intake of 40 mL/day for 9 weeks of oils with different contents of minor polar compounds (706.36 and 485.01 mg/L) was also assessed in patients. Authors indicated an improvement in the biomarkers related to renal function as well as in inflammatory parameters and body composition, among other parameters, supporting the evidence that the phenolic compounds are contributing to the health benefits of this food [47]. However, de Santis et al. [48] recently stated that there is still a gap to solve concerning the correlation between the chemical characterization of VOO compounds and biological activity in humans, which is indispensable to clearly state health claims. The complex composition of olives, specifically regarding their polyphenol content and how it can vary depending on several factors, makes it hard to draw conclusions on the effects of olive oil phenols as such, since the synergy among the different components leads to different physiological responses.
4. Conclusions
VOO is known to be a significant bioactive food with a variety of advantageous qualities, and it may be useful in the treatment of various immune-inflammatory illnesses. On top of the fatty acid composition, where oleic acid as a major compound is considered an important contributor to these effects, VOO has other biological minor components that also account for its beneficial activities. The effect of the phenols from VOO on neutrophils, a key cell type in the immune response, was investigated in order to unravel the mechanisms behind the response. Results showed similar effects mediated by both total PF and isolated HTyr in Aβ1-42-activated human neutrophils, suggesting that HTyr plays a predominant role in the antioxidant and anti-inflammatory effects of the PF and strengthening the current knowledge concerning the main biological properties attributed to HTyr. Nevertheless, other bioactive substances found in the PF (e.g., tyrosol, pinoresinol, or oleocanthal, among others) may also contribute to PF’s beneficial effects. In fact, earlier studies have referred to these small polyphenols in olive oil as a type of natural substance with anti-inflammatory and antioxidant capabilities. Considering that Aβ1-42 is an early trigger in the pathogenesis of AD, leading to synaptic and cognitive impairments, these results show the beneficial effect of phenols in managing the inflammation associated with neuronal diseases, slowing down the development of the process.
Further studies are required to clarify the mechanisms underlying the beneficial effects of PF in the inflammatory process in order to design convenient therapeutic strategies and indications. Nevertheless, these results suggested that olive oil polyphenols have a significant potential to modulate inflammatory conditions distinguished by an over-activation of neutrophils, and thereby they could be an interesting alternative for the management of the inflammatory response.
Acknowledgments
Elena Grao-Cruces and Elvira Marquez-Paradas have the benefit of a doctoral fellowship (PREDOC_00749) from the Andalusian government and a departmental collaboration grant awarded by the Ministry of Education and Vocational Training from the Spanish government, respectively.
Author Contributions
Conceptualization, S.M.-d.l.P.; methodology, E.G.-C., E.M.-P. and C.M.C.-C.; formal analysis, S.L.-E., G.A. and C.S.-M.; investigation, F.R.-P.; resources, S.M.-d.l.P.; writing—original draft preparation, F.R.-P.; writing—review and editing, S.M.-d.l.P.; funding acquisition, S.M.-d.l.P. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This study was supported by the research grant TED2021-130521A-I00 (Ministry of Science and Innovation, Government of Spain) into the Recovery, Transformation, and Resilience Plan funding by NextGenerationEU.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Javier Basterra-Gortari F., Ruiz-Canela M., Martínez-González M.A., Babio N., Sorlí J.V., Fito M., Ros E., Gómez-Gracia E., Fiol M., Lapetra J., et al. Effects of a Mediterranean eating plan on the need for glucose-lowering medications in participants with type 2 diabetes: A subgroup analysis of the PREDIMED trial. Diabetes Care. 2019;42:1390–1397. doi: 10.2337/dc18-2475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Estruch R., Ros E., Salas-Salvadó J., Covas M.-I., Corella D., Arós F., Gómez-Gracia E., Ruiz-Gutiérrez V., Fiol M., Lapetra J., et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018;378:e34. doi: 10.1056/NEJMoa1800389. [DOI] [PubMed] [Google Scholar]
- 3.Schwingshackl L., Morze J., Hoffmann G. Mediterranean diet and health status: Active ingredients and pharmacological mechanisms. Br. J. Pharmacol. 2020;177:1241–1257. doi: 10.1111/bph.14778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Martin M.E., Millan-Linares M.C., Naranjo M.C., Toscano R., Abia R., Muriana F.J.G., Bermudez B., Montserrat-de la Paz S. Minor compounds from virgin olive oil attenuate LPS-induced inflammation via visfatin-related gene modulation on primary human monocytes. J. Food Biochem. 2019;43:e12941. doi: 10.1111/jfbc.12941. [DOI] [PubMed] [Google Scholar]
- 5.Cárdeno A., Aparicio-Soto M., Montserrat-de la Paz S., Bermudez B., Muriana F.J.G., Alarcón-de-la-Lastra C. Squalene targets pro- and anti-inflammatory mediators and pathways to modulate over-activation of neutrophils, monocytes and macrophages. J. Funct. Foods. 2015;14:779–790. doi: 10.1016/j.jff.2015.03.009. [DOI] [Google Scholar]
- 6.Reboredo-Rodríguez P., Varela-López A., Forbes-Hernández T.Y., Gasparrini M., Afrin S., Cianciosi D., Zhang J., Manna P.P., Bompadre S., Quiles J.L., et al. Phenolic compounds isolated from olive oil as nutraceutical tools for the prevention and management of cancer and cardiovascular diseases. Int. J. Mol. Sci. 2018;19:2305. doi: 10.3390/ijms19082305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Marković A.K., Torić J., Barbarić M., Brala C.J. Hydroxytyrosol, tyrosol and derivatives and their potential effects on human health. Molecules. 2019;24:2001. doi: 10.3390/molecules24102001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rivera-Jiménez J., Berraquero-García C., Pérez-Gálvez R., García-Moreno P.J., Espejo-Carpio F.J., Guadix A., Guadix E.M. Peptides and protein hydrolysates exhibiting anti- inflammatory activity: Sources, structural features and modulation mechanisms. Food Funct. 2022;13:12510–12540. doi: 10.1039/D2FO02223K. [DOI] [PubMed] [Google Scholar]
- 9.Qu X., Tang Y., Hua S. Immunological approaches towards cancer and inflammation: A cross talk. Front. Immunol. 2018;9:563. doi: 10.3389/fimmu.2018.00563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Montserrat-de la Paz S., Carrillo-Berdasco G., Rivero-Pino F., Villanueva-Lazo A., Millan-Linares M.C. Hemp Protein Hydrolysates Modulate Inflammasome-Related Genes in Microglial Cells. Biology. 2023;12:49. doi: 10.3390/biology12010049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schiller M., Ben-Shaanan T.L., Rolls A. Neuronal regulation of immunity: Why, how and where? Nat. Rev. Immunol. 2021;21:20–36. doi: 10.1038/s41577-020-0387-1. [DOI] [PubMed] [Google Scholar]
- 12.Aparicio-Soto M., Sánchez-Hidalgo M., Rosillo M.Á., Castejón M.L., Alarcón-De-La-Lastra C. Extra virgin olive oil: A key functional food for prevention of immune-inflammatory diseases. Food Funct. 2016;7:4492–4505. doi: 10.1039/C6FO01094F. [DOI] [PubMed] [Google Scholar]
- 13.Vazquez Roncero A., Janet del Valle M., Janet del Valle L. Componentes fenolicos de la aceituna. Grasas Aceites. 1976;27 [Google Scholar]
- 14.Cárdeno A., Sánchez-Hidalgo M., Aparicio-Soto M., Sánchez-Fidalgo S., Alarcón-De-La-Lastra C. Extra virgin olive oil polyphenolic extracts downregulate inflammatory responses in LPS-activated murine peritoneal macrophages suppressing NFκB and MAPK signalling pathways. Food Funct. 2014;5:1270–1277. doi: 10.1039/C4FO00014E. [DOI] [PubMed] [Google Scholar]
- 15.Miho H., Moral J., Barranco D., Ledesma-Escobar C.A., Priego-Capote F., Díez C.M. Influence of genetic and interannual factors on the phenolic profiles of virgin olive oils. Food Chem. 2021;342:128357. doi: 10.1016/j.foodchem.2020.128357. [DOI] [PubMed] [Google Scholar]
- 16.Miho H., Moral J., López-González M.A., Díez C.M., Priego-Capote F. The phenolic profile of virgin olive oil is influenced by malaxation conditions and determines the oxidative stability. Food Chem. 2020;314:126183. doi: 10.1016/j.foodchem.2020.126183. [DOI] [PubMed] [Google Scholar]
- 17.Miho H., Díez C.M., Mena-Bravo A., Sánchez de Medina V., Moral J., Melliou E., Magiatis P., Rallo L., Barranco D., Priego-Capote F. Cultivar influence on variability in olive oil phenolic profiles determined through an extensive germplasm survey. Food Chem. 2018;266:192–199. doi: 10.1016/j.foodchem.2018.06.002. [DOI] [PubMed] [Google Scholar]
- 18.Bucciantini M., Leri M., Nardiello P., Casamenti F., Stefani M. Olive Polyphenols: Antioxidant and Anti-Inflammatory Properties. Antioxidants. 2021;10:1044. doi: 10.3390/antiox10071044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Aratani Y. Myeloperoxidase: Its role for host defense, inflammation, and neutrophil function. Arch. Biochem. Biophys. 2018;640:47–52. doi: 10.1016/j.abb.2018.01.004. [DOI] [PubMed] [Google Scholar]
- 20.Papayannopoulos V., Metzler K.D., Hakkim A., Zychlinsky A. Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J. Cell Biol. 2010;191:677–691. doi: 10.1083/jcb.201006052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Beauchamp G.K., Keast R.S.J., Morel D., Lin J., Pika J., Han Q., Lee C.-H., Smith A.B., Breslin P.A.S. Phytochemistry: Ibuprofen-like activity in extra-virgin olive oil. Nature. 2005;437:45–46. doi: 10.1038/437045a. [DOI] [PubMed] [Google Scholar]
- 22.Yao C., Narumiya S. Prostaglandin-cytokine crosstalk in chronic inflammation. Br. J. Pharmacol. 2019;176:337–354. doi: 10.1111/bph.14530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Glennon-Alty L., Hackett A.P., Chapman E.A., Wright H.L. Neutrophils and redox stress in the pathogenesis of autoimmune disease. Free Radic. Biol. Med. 2018;125:25–35. doi: 10.1016/j.freeradbiomed.2018.03.049. [DOI] [PubMed] [Google Scholar]
- 24.Stock A.J., Kasus-Jacobi A., Pereira H.A. The role of neutrophil granule proteins in neuroinflammation and Alzheimer’s disease. J. Neuroinflamm. 2018;15:1–15. doi: 10.1186/s12974-018-1284-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Czerwińska M.E., Kiss A.K., Naruszewicz M. Inhibition of human neutrophils NEP activity, CD11b/CD18 expression and elastase release by 3,4-dihydroxyphenylethanol-elenolic acid dialdehyde, oleacein. Food Chem. 2014;153:1–8. doi: 10.1016/j.foodchem.2013.12.019. [DOI] [PubMed] [Google Scholar]
- 26.Montoya T., Sánchez-Hidalgo M., Castejón M.L., Rosillo M.Á., González-Benjumea A., Alarcón-De-la-lastra C. Dietary oleocanthal supplementation prevents inflammation and oxidative stress in collagen-induced arthritis in mice. Antioxidants. 2021;10:650. doi: 10.3390/antiox10050650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen L., Yu J. Modulation of Toll-Like Receptor Signaling in Innate Immunity by Natural Products. Int. Immunopharmacol. 2016;37:65–70. doi: 10.1016/j.intimp.2016.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zeilhofer H.U., Schorr W. Role of interleukin-8 in neutophil signaling. Curr. Opin. Hematol. 2000;7:178–182. doi: 10.1097/00062752-200005000-00009. [DOI] [PubMed] [Google Scholar]
- 29.Rosales C. Neutrophil: A cell with many roles in inflammation or several cell types? Front. Physiol. 2018;9:113. doi: 10.3389/fphys.2018.00113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kobayashi Y. The regulatory role of nitric oxide in proinflammatory cytokine expression during the induction and resolution of inflammation. J. Leukoc. Biol. 2010;88:1157–1162. doi: 10.1189/jlb.0310149. [DOI] [PubMed] [Google Scholar]
- 31.Alblihed M.A. Hydroxytyrosol ameliorates oxidative challenge and inflammatory response associated with lipopolysaccharide-mediated sepsis in mice. Hum. Exp. Toxicol. 2021;40:342–354. doi: 10.1177/0960327120949618. [DOI] [PubMed] [Google Scholar]
- 32.Carluccio M.A., Martinelli R., Massaro M., Calabriso N., Scoditti E., Maffia M., Verri T., Gatta V., Caterina R. De Nutrigenomic Effect of Hydroxytyrosol in Vascular Endothelial Cells: A Transcriptomic Profile Analysis. Nutrients. 2021;13:3990. doi: 10.3390/nu13113990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Khokha R., Murthy A., Weiss A. Metalloproteinases and their natural inhibitors in inflammation and immunity. Nat. Rev. Immunol. 2013;13:649–665. doi: 10.1038/nri3499. [DOI] [PubMed] [Google Scholar]
- 34.Youssef J., Badr M. Role of peroxisome proliferator-activated receptors in inflammation control. J. Biomed. Biotechnol. 2004;12:156–166. doi: 10.1155/S1110724304308065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lai W.-C., Zhou M., Shankavaram U., Peng G., Wahl L.M. Differential Regulation of Lipopolysaccharide-Induced Monocyte Matrix Metalloproteinase (MMP)-1 and MMP-9 by p38 and Extracellular Signal-Regulated Kinase 1/2 Mitogen-Activated Protein Kinases. J. Immunol. 2003;170:6244–6249. doi: 10.4049/jimmunol.170.12.6244. [DOI] [PubMed] [Google Scholar]
- 36.Löffek S., Schilling O., Franzke C.W. Series “matrix metalloproteinases in lung health and disease” edited by J. Müller-Quernheim and O. Eickelberg number 1 in this series: Biological role of matrix metalloproteinases: A critical balance. Eur. Respir. J. 2011;38:191–208. doi: 10.1183/09031936.00146510. [DOI] [PubMed] [Google Scholar]
- 37.Nissinen L., Kähäri V.M. Matrix metalloproteinases in inflammation. Biochim. Biophys. Acta Gen. Subj. 2014;1840:2571–2580. doi: 10.1016/j.bbagen.2014.03.007. [DOI] [PubMed] [Google Scholar]
- 38.Scoditti E., Nestola A., Massaro M., Calabriso N., Storelli C., De Caterina R., Carluccio M.A. Hydroxytyrosol suppresses MMP-9 and COX-2 activity and expression in activated human monocytes via PKCα and PKCβ1 inhibition. Atherosclerosis. 2014;232:17–24. doi: 10.1016/j.atherosclerosis.2013.10.017. [DOI] [PubMed] [Google Scholar]
- 39.Croasdell A., Duffney P.F., Kim N., Lacy S.H., Sime P.J., Phipps R.P. PPAR γ and the Innate Immune System Mediate the Resolution of Inflammation. PPAR Res. 2015;2015:e549691. doi: 10.1155/2015/549691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mantovani A., Cassatella M.A., Costantini C., Jaillon S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat. Rev. Immunol. 2011;11:519–531. doi: 10.1038/nri3024. [DOI] [PubMed] [Google Scholar]
- 41.Selders G.S., Fetz A.E., Radic M.Z., Bowlin G.L. An overview of the role of neutrophils in innate immunity, inflammation and host-biomaterial integration. Regen. Biomater. 2017;4:55–68. doi: 10.1093/rb/rbw041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang Y., Wu J., Newton R., Bahaie N.S., Long C., Walcheck B. ADAM17 cleaves CD16b (FcγRIIIb) in human neutrophils. Biochim. Biophys. Acta. 2013;1833:680–685. doi: 10.1016/j.bbamcr.2012.11.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ivetic A. A head-to-tail view of L-selectin and its impact on neutrophil behaviour. Cell Tissue Res. 2018;371:437–453. doi: 10.1007/s00441-017-2774-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hu N., Westra J., Rutgers A., Doornbos-Van der Meer B., Huitema M.G., Stegeman C.A., Abdulahad W.H., Satchell S.C., Mathieson P.W., Heeringa P., et al. Decreased CXCR1 and CXCR2 expression on neutrophils in anti-neutrophil cytoplasmic autoantibody-associated vasculitides potentially increases neutrophil adhesion and impairs migration. Arthritis Res. Ther. 2011;13:R201. doi: 10.1186/ar3534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Källquist L., Hansson M., Persson A.M., Janssen H., Calafat J., Tapper H., Olsson I. The tetraspanin CD63 is involved in granule targeting of neutrophil elastase. Blood. 2008;112:3444–3454. doi: 10.1182/blood-2007-10-116285. [DOI] [PubMed] [Google Scholar]
- 46.Patti A.M., Carruba G., Cicero A.F.G., Banach M., Nikolic D., Giglio R.V., Terranova A., Soresi M., Giannitrapani L., Montalto G., et al. Daily use of extra virgin olive oil with high oleocanthal concentration reduced body weight, waist circumference, alanine transaminase, inflammatory cytokines and hepatic steatosis in subjects with the metabolic syndrome: A 2-month intervention study. Metabolites. 2020;10:392. doi: 10.3390/metabo10100392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Noce A., Marrone G., Urciuoli S., Di Daniele F., Di Lauro M., Zaitseva A.P., Di Daniele N., Romani A. Usefulness of extra virgin olive oil minor polar compounds in the management of chronic kidney disease patients. Nutrients. 2021;13:581. doi: 10.3390/nu13020581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.De Santis S., Clodoveo M.L., Corbo F. Correlation between Chemical Characterization and Biological Activity: An Urgent Need for Human Studies Using Extra Virgin Olive Oil. Antioxidants. 2022;11:258. doi: 10.3390/antiox11020258. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.