Abstract
Simple Summary
Antimicrobial drugs, in addition to exerting antibiotic, antifungal, antiparasitic, or antiviral effects, may also affect the central nervous system and gut microbiota, thereby modulating brain and behavior. Zebrafish models can be used for studying the effects of antimicrobial drugs on the central nervous system. Here, we discuss recent findings on using zebrafish for assessing the effects of a wide range of antimicrobial drugs on brain and behavior in vivo.
Abstract
Antimicrobial drugs represent a diverse group of widely utilized antibiotic, antifungal, antiparasitic and antiviral agents. Their growing use and clinical importance necessitate our improved understanding of physiological effects of antimicrobial drugs, including their potential effects on the central nervous system (CNS), at molecular, cellular, and behavioral levels. In addition, antimicrobial drugs can alter the composition of gut microbiota, and hence affect the gut–microbiota–brain axis, further modulating brain and behavioral processes. Complementing rodent studies, the zebrafish (Danio rerio) emerges as a powerful model system for screening various antimicrobial drugs, including probing their putative CNS effects. Here, we critically discuss recent evidence on the effects of antimicrobial drugs on brain and behavior in zebrafish, and outline future related lines of research using this aquatic model organism.
Keywords: antimicrobial drugs, microbiota, antibiotic, zebrafish, brain, behavior
1. Introduction
Animal models are indispensable tools for translational biomedical research, including studying the systemic effects of various drugs in complex living systems [1]. Alongside rodent models, a small freshwater teleost fish, the zebrafish (Danio rerio), has become a powerful in vivo vertebrate system widely used in biomedicine [2]. Characterized by high genetic (~70%) and physiological homology to humans [3], zebrafish are also increasingly utilized in the central nervous system (CNS) research, including modeling neurodegeneration (e.g., Alzheimer’s, Parkinson’s, and Huntington’s diseases, amyotrophic lateral sclerosis) [4], epilepsy [5], affective disorders [6], addiction and various other drug-induced conditions [7]. In addition to offering multiple genetic models of CNS pathogenesis [8], zebrafish can also serve as sensitive pharmacological screens for major classes of neuroactive drugs [9], including antidepressants, anxiolytics, antipsychotics, antiepileptics, and anesthetics [10,11]. Zebrafish are also commonly used to assess central nervous action of various other chemicals, including CNS side effects of clinically used drugs [12] and neural deficits caused by toxins, environmental pollutants [13,14], and endocrine disruptors [15].
In general, antimicrobials represent a large diverse group of drugs used to prevent and treat infection, and include antibacterial (antibiotic), antiviral, antifungal, and antiparasitic agents [16]. Common antibiotics, classified based on their chemical structures and multiple modes of antimicrobial action, include beta-lactams, sulfonamides, aminoglycosides, tetracyclines, chloramphenicol, macrolides, glycopeptides, oxazolidinones, ansamycins, quinolones, streptogramins, and lipopeptides [17]. Typical classes of antifungal agents include polyenes, azoles, allylamines, echinocandins, and triterpenoids, that alter membrane permeability and/or inhibit the synthesis of the fungal wall [18]. Antiviral drugs have different mechanisms of action, inhibiting virus attachment, entry, uncoating, polymerase, nucleoside and nucleotide reverse transcriptase, integrase, and protease activity [19]. Antiparasitic drugs mainly include antiprotozoal agents [20]. A diverse array of other antimicrobial agents, acting via multiple biological mechanisms, includes chlorhexidine, triclosan, alcohols, hydrogen peroxide [21], non-steroidal anti-inflammatory drugs (NSAIDs) [22], and essential oils (e.g., basil, oregano, thyme, tea tree, coriander, and clove oils) [23].
The growing use and clinical importance of antimicrobial drugs necessitate our improved understanding of the complete spectrum of their physiological effects, including their potential effects on CNS at molecular, cellular, and behavioral levels. In addition to conventional antimicrobial properties that have been extensively tested in vivo and in vitro, these agents may impact CNS and behavior, both clinically and in animal models [24,25,26,27]. Like rodents, zebrafish represent a useful sensitive organism for screening the CNS effects of various antimicrobial drugs in vivo. Furthermore, antimicrobials can alter the composition of gut microbiota, and hence affect the gut–microbiota–brain axis, again modulating brain and behavioral processes. Recognizing the growing potential of zebrafish-based drug bioscreening, here we critically discuss recent evidence on central nervous effects of antimicrobial agents in zebrafish, summarize recent successes and challenges in this field, and outline future lines of research using this aquatic model organism.
2. Reported CNS Effects of Antimicrobial Drugs in Animal Models
2.1. Antibacterial Antibiotic Drugs
Mounting animal evidence demonstrates frequent CNS effects of commonly used antimicrobial drugs (also see Table 1). For example, in mice, various antibiotics, such as ampicillin, bacitracin, meropenem, neomycin, and vancomycin, evoke overt cognitive deficits, increase exploratory behavior, and alter brain expression of signaling molecules [24] and the permeability of the blood–brain barrier (BBB) [28]. Some antibiotics (e.g., ciprofloxacin, minocycline, ampicillin, neomycin, and vancomycin) lower systemic antioxidant activity [26], reduce apoptosis in rat brain [29], and increase rodent anxiety-like and impulsive behavior [30,31]. At least some of these effects may be indirect, and are probably mediated by gut microbiota status, since germ-free mice display motor hyperactivity, anxiety-like behavior [32], social deficit (e.g., social avoidance and diminished preference for social novelty) [33], as well as working memory deficits [24]. Microbiota can also affect behavioral characteristics in zebrafish, since axenic larvae exhibit hyperlocomotion corrected by microbiota colonization [34].
Table 1.
Selected examples of CNS effects of antimicrobials on CNS.
| Classes of Drugs | Representative Drugs | Effects in Zebrafish | References |
|---|---|---|---|
| Antibacterial | |||
| Phenols | triclosan | Inhibited acetylcholinesterase and dopaminergic activity, neuroapoptosis, reduced synaptic density, axonal length, higher expression of mir-137. | [35,36] |
| β-diketones | oxytetracycline | Decreased deiodinase 2/3, T3 levels, thyroid receptors, cortisol levels and serotonin synthesis. Increased exploratory behavior, motor activity, expression of parkin, pink1 and cd-11b, proliferation of glial cells, ventriculomegaly, altered (decreased at low, increased at high doses) anxiety-like behavior, cognitive deficits, aggression. | [37,38,39,40,41,42] |
| enrofloxatine | Increased corticotropin-releasing hormone (CRH), brain-derived neurotrophic factor (BDNF), neuropeptide Y, reduced adrenocorticotropic hormone (ACTH) and cortisol, proliferation of glial cells, ventriculomegaly, altered (decreased at low, increased at high doses) anxiety-like behavior, cognitive deficits, aggression. | [39,40,41,42,43,44] | |
| Aminoglycosides | neomycin, gentamicin |
Damaged lateral line hair cells. | [45] |
| Cephalosporins | ceftazidime | Increased locomotor activity, aggression and cognitive deficits. | [42] |
| ceftriaxone | Corrected exploratory behavior (disrupted by ethanol withdrawal), increased glutamate uptake. | [46] | |
| Sulfonamides | sulfamethoxazole | Cerebral ischemia, oxidative stress. | [47] |
| Lincosamides | lincomycin | Reduced ventricular volume, neuronal loss, locomotor activity, systemic oxidative stress and apoptosis, increased whole-body acetylcholinesterase and ATPase activity. | [48] |
| Penicillins | amoxicillin | Decreased locomotor activity, social behavior and oxidative stress. | [49] |
| Secoiridoids | sweroside | Reduced anxiety, improved cognitive performance, reduced brain acetylcholinesterase activity and oxidative stress. | [50] |
| Others | |||
| Essential oils | extract from
Thymus vulgaris |
Decreased anxiety-like behavior, improved cognitive function and acetylcholine neurotransmission. | [51] |
| Cationic surfactants | cetylpyridinium chloride |
Reduced locomotor and social activity, with lower serotonin, dopamine, and acetylcholine in brains of adult fish, but higher in juveniles. | [52] |
| Non-steroidal anti-inflammatory drugs (NSAIDS) | aspirin | Decreased anxiety, exploratory behavior and mobility. | [53,54] |
| mTOR inhibitors | rapamycin | Reduced seizures in epilepsy models. | [55,56] |
Triclosan, a widely used synthetic antimicrobial agent with poly-target (antibiotic and antifungal) action, also inhibits dopamine and increases acetylcholine neurotransmission, promotes neuronal apoptosis, and reduces synaptic density and axonal length in zebrafish [35]. Furthermore, triclosan downregulates the expression of brain genes that are important during neurodevelopment, including glial fibrillary acidic protein (GFAP) and myelin basic protein (MBP) that control myelination and axonal maintenance [57]. In contrast, mir-137, a short non-coding RNA associated with the mitogen-activated protein kinase (MAPK) pathway, is upregulated in zebrafish by triclosan, eventually impairing their auditory and visual sensitivity [36]. The neurotranscriptomic effects of triclosan can be mediated both by the regulation of DNA methylation [58] and by activation of other regulatory pathways, such as MAPK/ERK (extracellular signal-regulated kinases) [59].
CNS effects of β-diketone antibiotics, including fluoroquinolones and tetracyclines, have also been tested in zebrafish (Table 1). For example, a typical tetracycline antibiotic, oxytetracycline, affects the neuroendocrine system of juvenile zebrafish, such as the thyroid and adrenocorticotropic axes, as it reduces deiodinase 2 and 3, triiodothyronine T3, receptors of thyroid hormone, and whole-body cortisol levels [37]. In addition, the drug affects serotonin CNS signaling in the juvenile zebrafish, lowering brain expression of tryptophan hydroxylase, an enzyme involved in serotonin synthesis [37]. At the behavioral level, this antibiotic increases exploration and hyperactivity in zebrafish [38], whereas minocycline, another tetracycline, increased larval expression of parkin, pink1, and cd-11b genes, whose human orthologs are strongly implicated in Parkinson’s pathogenesis [39]. In line with this, minocycline evoked neuroprotective effects in a zebrafish larval model of Parkinson’s disease, preventing locomotor deficits and the loss of dopaminergic neurons [60].
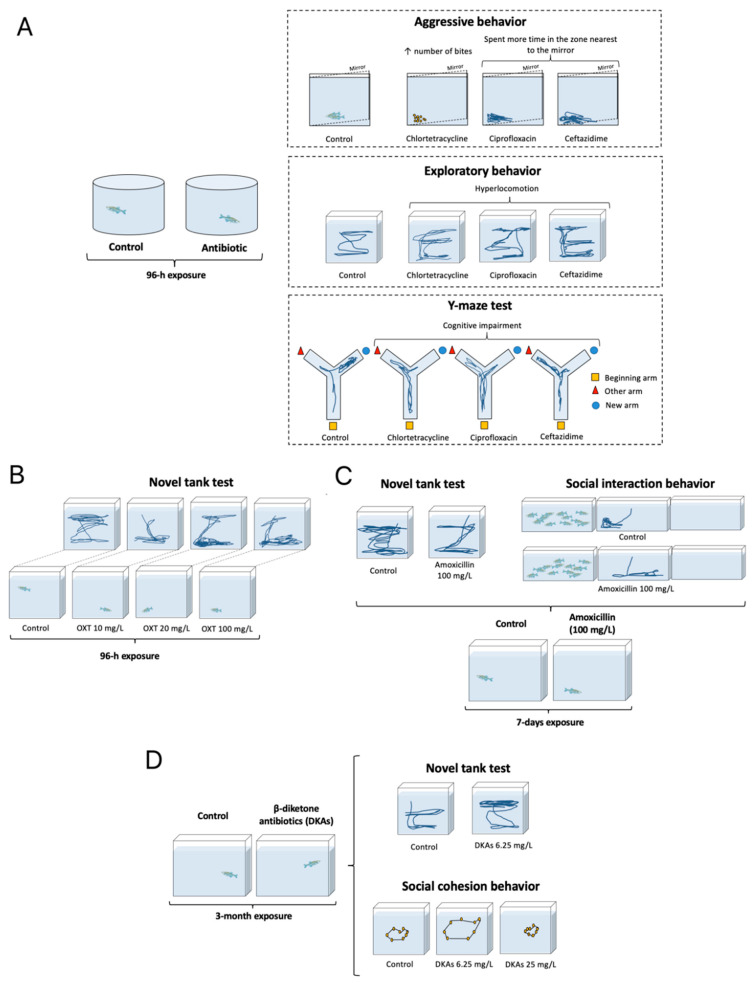
Fluoroquinols exert overt neurotoxic effects in zebrafish, impairing the development of embryos by hyperactivating the glutamate N-methyl-D-aspartate (NMDA) receptors [43]. In adult fish, exposure to these drugs increases whole-body corticotropin-releasing hormone (CRH) along with CNS levels of brain-derived neurotrophic factor (BDNF) and neuropeptide Y, but lowers plasma adrenocorticotropic hormone (ACTH) and cortisol [44]. In general, fluoroquinolones and tetracyclines are rather neuroactive in zebrafish (Table 1), and may cause ventriculomegaly, proliferation of glial cells, and neuronal apoptosis (e.g., see [40]), as well as dose-dependently increasing motor activity and altering (at low doses decreasing, and at high doses increasing) anxiety-like behavior in zebrafish [41]. Likewise, β-diketones impair zebrafish cognition (e.g., working memory) and promote aggressive behavior (Figure 1) [42].
Figure 1.
Selected examples of antimicrobial drugs’ effects on zebrafish behavior. (A) Exposure to chlortetracycline, ciprofloxacin, or ceftazidime for 96 h increases (↑) exploratory activity (more distance travelled), aggression (more bites in the mirror test), and cognitive deficits (more errors in the Y-maze) [42]. (B) Exposure to oxytetracycline (10–100 mg/L) for 96 h evokes anxiety-like behavior in the novel tank test [61]. (C) Exposure to amoxicillin (100 mg/L) for 7 days reduces distance travelled and social interaction [49]. (D) Chronic exposure to β-diketones (at 6.25 mg/L) increases time spent at the top of the test tank (an anxiolytic-like behavior) and alters (increases at 6.25, and decreases at 25 mg/L) shoaling behavior [41].
Aminoglycosides neomycin and gentamicin damage the lateral line hair cells in zebrafish larvae, impairing locomotion and the startle response [45]. Other antibiotics have also been studied in zebrafish, including screening the CNS effects of avermectin, sulfamethoxazole, lincomycin, and amoxicillin. Interestingly, in addition to overt neurotoxicity, avermectin also increases brain expression of gamma aminobutyric acid (GABA)-A receptor in another fish species, Carassius auratus [62]. During zebrafish embryogenesis, sulfamethoxazole causes cerebral ischemia and brain oxidative stress, activating CNS angiogenesis, probably mediated by vascular endothelial growth factor (VEGF) signaling, since its inhibition corrects the deficits [47]. Lincomycin also has neurotoxic effects, reducing ventricular volume and neuronal numbers, but increasing systemic oxidative stress and apoptosis in zebrafish larvae, activating their acetylcholinesterase and ATPase, and decreasing locomotor activity [48]. In adult zebrafish, amoxicillin reduces locomotor and social behavior, and promotes oxidative stress in the brain, strikingly paralleling some clinical symptoms observed in autistic patients (Figure 1) [49].
Ceftazidime, a cephalosporin antibiotic, increases locomotor activity in zebrafish, impairs their learning, and promotes aggression (Figure 1) [42]. Another cephalosporin, ceftriaxone, restores normal patterns of zebrafish exploratory behavior (disrupted by ethanol withdrawal), accompanied by increased brain glutamate transport [46]. Sweroside, a secoiridoid glycoside, reduces anxiety-like behavior and improves cognitive performance in zebrafish Y-maze and novel object-recognition tests, probably due to reduced brain acetylcholinesterase activity [50], as muscarinic acetylcholine receptors are involved in both learning and memory. Furthermore, impaired cognitive status can also be explained by increased oxidative stress [63].
2.2. Selected Other Antimicrobial Agents
In addition to its antimicrobial effects per se, thyme (Thymus vulgaris) essential oil decreases anxiety-like behavior, improves cognitive function and increases acetylcholine neurotransmission in zebrafish [51]. In contrast, an antimicrobial cationic surfactant cetylpyridinium chloride negatively impacts CNS, reducing fish locomotor and social activity, also age-dependently altering neuromediators (e.g., reducing serotonin, dopamine, and acetylcholine in adults, but increasing in juvenile fish) [52].
Antimicrobial properties have also been shown for NSAIDs [22], which also exert some CNS effects in zebrafish. For example, aspirin, a typical NSAID, evokes an anxiogenic-like action in adult zebrafish, likely mediated via the serotonergic system, given its similar serotonin-modulating effect in rodents (see [53] for discussion). Exposure to high doses of aspirin markedly inhibits exploratory behavior and mobility in zebrafish, which may suggest sedative and/or toxic side-effects of this drug [54].
Another atypical antimicrobial antifungal drug, rapamycin, is an important cellular inhibitor of the mammalian target of rapamycin (mTOR) signaling [64]. Rapamycin is remarkably neurotropic in both rodents and zebrafish, reducing seizures in various spontaneous (genetic) and chemically-induced epilepsy models [55]. In addition to inactivating mTOR in zebrafish larvae with experimental epilepsy, the impairment of fine branching of GABA-ergic neurons during neurodevelopment in this model is corrected by rapamycin, suggesting some putative additional mechanisms of its CNS action beyond directly affecting the mTOR signaling [56].
Furthermore, albeit not the main scope here, mounting evidence suggests that gut microbiota may impact some CNS functions in zebrafish models, for example, during morphine addiction. Altered microbiota composition is associated with affected behavior and brain and gut gene expression in morphine-treated fish, which also show conditioned place preference (CPP) to the drug. Interestingly, these alterations are corrected by an alkaloid synenin, whereas antibiotic treatment inhibits this process, hence implicating antibiotics and gut microbiota in morphine-related behaviors in zebrafish [65]. Again, while such CNS effects are probably mediated by indirect effects of antibiotics on gut microflora, rather than by direct action on neuronal processes in vivo, this aspect clearly merits further scrutiny in future zebrafish studies.
3. Discussion
Antimicrobial drugs, especially antibiotics, are among the most widely prescribed and used medications, with up to almost 80% of the global population having taken them in the last 6 months [66]. Such prevalent drug usage represents a serious biomedical problem, which is further complicated by antimicrobial drug resistance and risks of multiple systemic side effects. Thus, it becomes important to better understand a fuller spectrum of physiological effects of antimicrobial agents in vivo–the task that also involves testing their CNS effects in various experimental animals (including zebrafish).
There are several research aspects to consider in regard to studying CNS effects of antimicrobials in aquatic models, such as zebrafish. For example, recent evidence of potential neuroprotective effects of some antimicrobial drugs (see above) in animal models suggests an opportunity for their use for drug repurposing. Indeed, amoxicillin reduces ischemia in mice with cranio-cerebral trauma, a neuroprotective effect associated with lower migration of T cells into the meninges [67]. Minocycline, a tetracycline antibiotic, displays its putative neuroprotective properties by reducing cerebral edema during hemorrhage, neuroinflammation, neuronal degeneration, systemic inflammation [68], and neuronal apoptosis in rodents [29]. Azithromycin, another putative neuroprotective antibiotic, reduces ischemic brain damage and restores sensorimotor function in rat pups [69], probably due to the activation of anti-inflammatory M2-type microglia [70]. Doxycycline is also neuroprotective, apparently lowering neuroinflammation by activating antioxidant enzymes in rat brains [71]. Taken together, this evidence suggests that similar effects can also be expected in zebrafish models (Table 1), and indicates that a better focus is needed on assessing potential beneficial CNS effects of antimicrobial drugs in zebrafish screenings, in addition to traditional studies assaying their unwanted side effects on the central nervous system, both in experimental models and clinically.
Can zebrafish be in principle a valid, suitable aquatic model object for assessing a wider spectrum of CNS effects of antimicrobial drugs? It seems indeed likely, since zebrafish CNS is characterized by generally conserved neuroanatomy and neurotransmission, including well-developed glutamate-, GABA-, monoamine-, acetylcholine-, histamine-, glycine- [72], purine-ergic and endocannabinoid systems [73]. Zebrafish have a complex well-developed brain, and despite the lack of cerebral cortex and a clearly defined hippocampus, show otherwise high functionality of other structures that are neurally equivalent to those of mammals [74]. Zebrafish also exhibit a wide range of well-described behaviors, allowing the study of drug effects on locomotor, anxiety- and depression-like, and social phenotypes [75,76,77]. Collectively, this enables the use of zebrafish in translational modeling of neurodegenerative, affective, psychotic, neurodevelopmental, and addictive disorders [4,78,79].
Another advantage of zebrafish models for CNS drug screening is the economic benefit of such research (relative to that in rodents), given the simplicity of fish husbandry, handling, and experimental manipulation (e.g., compare adding drugs to fish water vs. using laborious systemic injections in rodents). Furthermore, these fish are characterized by external fertilization, allowing the eggs and embryos to be easily manipulated, which, in addition to optical clarity (including some adult strains, such as casper zebrafish), facilitates successful application of such aquatic models in assessing drug toxicity [80].
Overall, zebrafish hold a remarkable potential for drug development. For example, Prohema, a stabilized prostaglandin E2 (PGE2) product that increases regeneration of bone marrow, has now reached clinical trials [81]. Another relevant example is the ability of cyclooxygenase inhibitors to suppress the leukemia-like phenotype, first demonstrated in zebrafish and further validated in other animal models [82,83]. It is likely that such effects can therefore also be extended to CNS processes. For instance, the beneficial action of ramipril and quinapril, angiotensin-converting enzyme inhibitors, has been demonstrated in larval zebrafish, showing therapeutic effects on intracerebral hemorrhage [84]. Neuroprotection in zebrafish is included by PROTO-1, a benzothiophenacarboxamide that counteracts ototoxic effects of neomycin on fish hair cells [85]. Screening of nearly 400 PROTO-1-like drugs has identified a stable neuroprotective compound that has reached the clinical trial phase [86]. Thus, zebrafish continue to emerge as a suitable system for evaluating potential beneficial CNS properties of antimicrobial drugs.
Studying CNS effects in zebrafish has linked specific physiological profile of antimicrobials to altered neurotransmission (e.g., the acetylcholine, dopamine, and serotonin systems), neuroendocrine signaling (e.g., modulating thyroid and corticotropic axes), and other cellular processes (e.g., decreased or increased CNS oxidative stress, altered BDNF levels, see Table 1 for details). On the one hand, such studies are important since they are mechanistic in nature, and can show which genes are up- or down-regulated by the drug in question. For example, a new pathway of regulation of the mir-125b gene by triclosan in zebrafish, showing increased expression of mir-125b (that can be neurotoxic) via a novel, previously unrecognized signaling pathway [59], reflects a fundamental value of this type of research in zebrafish. Furthermore, antibiotics can impact DNA methylation, and hence exert epigenetic effects in the brain [58]. Thus, analyses of the entire spectrum of effects of antimicrobial agents in zebrafish may help explore the fundamental mechanisms of regulation of CNS development and functioning.
Nevertheless, multiple questions remain open in regard to assessing CNS effects of antimicrobial drugs by experimental models in general, and by using zebrafish screens in particular (see Table 2 for a summary of selected open questions in this field). For example, albeit poorly studied, the noradrenergic system in many ways has similar pathways and targets to the serotonergic system. As the latter is often affected by various antimicrobials (Table 1), it can be interesting to assess putative drug-induced central noradrenergic effects as well. Likewise, the reported impact of antimicrobial drugs on the thyroid and corticotropic axes (Table 1) raises the possibility of indirectly affecting other components of the endocrine system, given extensive horizontal connections within this system.
Table 2.
Selected open questions related to screening CNS effects of antimicrobial drugs in zebrafish.
| Open Questions |
|---|
|
Recent data show that antimicrobials can impact both neurons and glial cells. In zebrafish, while some antibiotics induce neuronal apoptosis accompanied by glia proliferation [40], the exact effects of these drugs on glial cells remain unexplored. Rodent studies demonstrate that exposure to particular antibiotics may impact gene expression in excitatory neurons, microglia, and astrocytes, with reduced efficiency of synaptic neurotransmission and cognitive deficits [88,89]. However, such effects on neurons may also be mediated by microglia. For example, antibiotics can lead to immature microglia unable to remodel dendritic spines, thereby resulting in cognitive deficits [88]. Some antibiotics, such as minocycline, inhibit microglial activation in rodents, especially the pro-inflammatory M1 microglia type [90,91]. Microglia can also induce transformation of astrocytes into the A1 phenotype, thus providing a neuroprotective effect [92]. Overall, there is likely a complicated interaction between neurons, microglia, and astrocytes, whose exact interplay in mediating CNS responses to antimicrobials is not fully understood in either rodents or fish.
Furthermore, various antibiotics modify the BBB permeability [93]. Clearly, this aspect warrants to be studied in zebrafish in a greater depth, as this would be less resource-consuming than in mammals, and may be fundamentally important, since altered BBB permeability relates to various brain diseases [94]. The link between antibiotics and neuroinflammation is another critical relevant topic for translational research, as modification of the microbiome composition can reduce neuroinflammation and alleviate Alzheimer’s symptoms [95]. However, the availability and mechanisms of this putative relationship in fish, as well as the possibility of using antibiotics to treat CNS diseases associated with neuroinflammation, merit further scrutiny.
Furthermore, antimicrobial drugs are also known to affect the epigenetic regulation of DNA expression. Indeed, some antibiotics (e.g., triclosan and minocycline) can alter DNA methylation both in rats and in zebrafish [58,90,96]. While such effects on some transcription factors (e.g., Nrf2) exist in zebrafish [59], the impact of antimicrobials on various other transcription factors, as well as at the level of acetylase and deacetylase activity, is yet to be studied in rodent or zebrafish models. An additional challenge is to explore the impact of other major experimental variables (e.g., sex, age, strain) on the brain–gut microbiota axis. For example, sex not only impacts brain pathogenesis, including autism, schizophrenia, and depression, but also affects the composition of gut microbiota [97]. Sex-dependent effects of antibiotics have been demonstrated in a mouse model of Alzheimer’s disease, affecting only males [98]. However, it remains unclear whether sex-specific CNS effects of antimicrobials can be found and replicated in fish. Likewise, the potential role of age, strain, and individual differences in CNS responses to antimicrobial drugs in zebrafish necessitates further translational studies (Table 2).
4. Concluding Remarks
As already noted, the aquatic zebrafish model is exceptionally well-positioned to serve as a tool for efficient and high-throughput drug screening. The latter may be highly relevant to assaying CNS effects of antimicrobial drugs. For example, unlike rodents, zebrafish larval assays allow a wide range of CNS drugs to be analyzed within minimally required time [84,99]. Importantly, zebrafish are vertebrate animals, and have a much higher genetic homology with humans [3] compared with other popular model organisms (e.g., Drosophila or C. elegans) that are commonly used for rapid drug screening. This enables the evaluation in zebrafish of a wide spectrum of physiological effects of a single drug, including assaying multiple CNS [100], cardiovascular [101], digestive [102], immune [103], and endocrine phenotypes [104].
Furthermore, the ability to perform rapid pharmacological studies enables efficient evaluation of drug–drug interactions in zebrafish, including testing combinations of a) several antimicrobial agents and b) an antimicrobial agent with another drug, that may synergistically or differentially modulate CNS functions. Moreover, it is possible to predict that studies of drug-induced CNS effects in zebrafish can be empowered by 3D modeling of their behavior, coupled by the application of artificial intelligence (AI) tools, in order to detect, recognize and decode neurophenotypic signatures for various antimicrobial drugs. The AI-based methods are becoming widely used in biomedicine, and their developing application to zebrafish drug screens clearly warrants further efforts.
Finally, the environmental impact of antimicrobial chemicals must also be considered in the context of utilizing zebrafish models as sensitive bioscreens for potential CNS effects of such drugs. Indeed, various antimicrobial drugs are widely used by humans, contaminating wastewater and leading to their release into the environment (e.g., see data on environmental pollution by triclosan and triclocarban [105]). From this standpoint, zebrafish are particularly well-suited for assessing the environmental impact of antimicrobial agents, including their acute neurotoxicity and their long-term, delayed, and/or early developmental effects on these aquatic organisms. For example, zebrafish have already been used to evaluate the toxicity of antiviral drugs, such as lopinavir and ritonavir [106], and their utility can be extended to include screening the CNS effects of multiple other antimicrobials. The fact that fish can typically be chronically exposed to chemicals (e.g., pollutants) via water immersion also more closely recapitulates the continuous aspect of human exposure to environmental hazards (e.g., compared to a more intermittent nature of chronic systemic injections of such drugs in rodent models).
In conclusion, mounting evidence summarized here demonstrates overt CNS effects of multiple antimicrobial drugs in mammals and zebrafish, emphasizing the latter as a particularly suitable organism for evaluating potential neurotoxic side effects, and rapid screening of CNS activity, of antimicrobial drugs. As such, a rigorous unbiased search for negative and positive CNS effects of antimicrobials in zebrafish pharmacological, genetic and pharmacogenetic models will not only improve the efficiency of preclinical profiling of these drugs in vivo, but can also facilitate a better translation of these findings into rodent studies and, eventually, into clinical settings.
Acknowledgments
A.V.K. is the Chair of the International Zebrafish Neuroscience Research Consortium (ZNRC) that coordinated this collaborative multi-laboratory project jointly with COBRAIN Center (headed by K.B.Y.) supported by the Republic of Armenia State Committee of Science (20TTCG-3A012 and N10-14/I-1) and the European Union-funded H2020 COBRAIN project (857600). The consortium provided a collaborative idea exchange platform for this study, and did not fund the study. The study partially used the facilities and equipment of the Resource Fund of Applied Genetics, MIPT support grant 075-15-2021-684.
Author Contributions
Conceptualization, M.M.K. and A.V.K.; methodology, investigation, M.M.K., D.S.G., M.S.d.A., T.O.K. and T.S.; data curation, M.M.K., E.V.P. and T.O.K.; writing—original draft preparation, M.M.K., T.G.A., K.A.D., D.S.G. and T.O.K.; writing—review and editing, K.A.D., M.S.d.A., E.V.P., K.B.Y. and A.V.K.; visualization, M.S.d.A.; supervision, K.B.Y., E.V.P. and A.V.K.; project administration, A.V.K.; funding acquisition, A.V.K. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This work was supported by St. Petersburg State University (project ID 93020614). T.O.K. is supported by Sirius University of Science and Technology (project NRB-RND-2116). The funders had no role in the design, analyses, and interpretation of the submitted study, or decision to publish.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Singh V.K., Seed T.M. How necessary are animal models for modern drug discovery? Expert Opin. Drug Discov. 2021;16:1391–1397. doi: 10.1080/17460441.2021.1972255. [DOI] [PubMed] [Google Scholar]
- 2.Beliaeva N.F., Kashirtseva V.N., Medvedeva N.V., Khudoklinova I., Ipatova O.M., Archakov A.I. Zebrafish as a model organism for biomedical studies. Biomeditsinskaia Khimiia. 2010;56:120–131. doi: 10.18097/pbmc20105601120. [DOI] [PubMed] [Google Scholar]
- 3.Howe K., Clark M.D., Torroja C.F., Torrance J., Berthelot C., Muffato M., Collins J.E., Humphray S., McLaren K., Matthews L., et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature. 2013;496:498–503. doi: 10.1038/nature12111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang J., Cao H. Zebrafish and Medaka: Important Animal Models for Human Neurodegenerative Diseases. Int. J. Mol. Sci. 2021;22:10766. doi: 10.3390/ijms221910766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stewart A.M., Desmond D., Kyzar E., Gaikwad S., Roth A., Riehl R., Collins C., Monnig L., Green J., Kalueff A.V. Perspectives of zebrafish models of epilepsy: What, how and where next? Brain Res. Bull. 2012;87:135–143. doi: 10.1016/j.brainresbull.2011.11.020. [DOI] [PubMed] [Google Scholar]
- 6.de Abreu M.S., Maximino C., Banha F., Anastácio P.M., Demin K.A., Kalueff A.V., Soares M.C. Emotional behavior in aquatic organisms? Lessons from crayfish and zebrafish. J. Neurosci. Res. 2020;98:764–779. doi: 10.1002/jnr.24550. [DOI] [PubMed] [Google Scholar]
- 7.Bao W., Volgin A.D., Alpyshov E.T., Friend A.J., Strekalova T.V., de Abreu M.S., Collins C., Amstislavskaya T.G., Demin K.A., Kalueff A.V. Opioid Neurobiology, Neurogenetics and Neuropharmacology in Zebrafish. Neuroscience. 2019;404:218–232. doi: 10.1016/j.neuroscience.2019.01.045. [DOI] [PubMed] [Google Scholar]
- 8.de Abreu M.S., Genario R., Giacomini A., Demin K.A., Lakstygal A.M., Amstislavskaya T.G., Fontana B.D., Parker M.O., Kalueff A.V. Zebrafish as a model of neurodevelopmental disorders. Neuroscience. 2020;445:3–11. doi: 10.1016/j.neuroscience.2019.08.034. [DOI] [PubMed] [Google Scholar]
- 9.Bruni G., Lakhani P., Kokel D. Discovering novel neuroactive drugs through high-throughput behavior-based chemical screening in the zebrafish. Front. Pharmacol. 2014;5:153. doi: 10.3389/fphar.2014.00153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zindler F., Stoll S., Baumann L., Knoll S., Huhn C., Braunbeck T. Do environmentally relevant concentrations of fluoxetine and citalopram impair stress-related behavior in zebrafish (Danio rerio) embryos? Chemosphere. 2020;261:127753. doi: 10.1016/j.chemosphere.2020.127753. [DOI] [PubMed] [Google Scholar]
- 11.Huang I.J., Sirotkin H.I., McElroy A.E. Varying the exposure period and duration of neuroactive pharmaceuticals and their metabolites modulates effects on the visual motor response in zebrafish (Danio rerio) larvae. Neurotoxicol. Teratol. 2019;72:39–48. doi: 10.1016/j.ntt.2019.01.006. [DOI] [PubMed] [Google Scholar]
- 12.Bittner L., Teixidó E., Keddi I., Escher B.I., Klüver N. pH-Dependent Uptake and Sublethal Effects of Antihistamines in Zebrafish (Danio rerio) Embryos. Environ. Toxicol. Chem. 2019;38:1012–1022. doi: 10.1002/etc.4395. [DOI] [PubMed] [Google Scholar]
- 13.Carlsson G., Patring J., Kreuger J., Norrgren L., Oskarsson A. Toxicity of 15 veterinary pharmaceuticals in zebrafish (Danio rerio) embryos. Aquat. Toxicol. (Amst. Neth.) 2013;126:30–41. doi: 10.1016/j.aquatox.2012.10.008. [DOI] [PubMed] [Google Scholar]
- 14.Galus M., Jeyaranjaan J., Smith E., Li H., Metcalfe C., Wilson J.Y. Chronic effects of exposure to a pharmaceutical mixture and municipal wastewater in zebrafish. Aquat. Toxicol. 2013;132–133:212–222. doi: 10.1016/j.aquatox.2012.12.016. [DOI] [PubMed] [Google Scholar]
- 15.Spaan K., Haigis A.C., Weiss J., Legradi J. Effects of 25 thyroid hormone disruptors on zebrafish embryos: A literature review of potential biomarkers. Sci. Total Environ. 2019;656:1238–1249. doi: 10.1016/j.scitotenv.2018.11.071. [DOI] [PubMed] [Google Scholar]
- 16.Antimicrobial Resistance. [(accessed on 2 December 2022)]. Available online: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance.
- 17.A Brief Overview of Classes of Antibiotics. [(accessed on 12 December 2022)]. Available online: https://www.compoundchem.com/2014/09/08/antibiotics/
- 18.Nivoix Y., Ledoux M.P., Herbrecht R. Antifungal Therapy: New and Evolving Therapies. Semin. Respir. Crit. Care Med. 2020;41:158–174. doi: 10.1055/s-0039-3400291. [DOI] [PubMed] [Google Scholar]
- 19.Kausar S., Said Khan F., Ishaq Mujeeb Ur Rehman M., Akram M., Riaz M., Rasool G., Hamid Khan A., Saleem I., Shamim S., Malik A. A review: Mechanism of action of antiviral drugs. Int. J. Immunopathol. Pharmacol. 2021;35:20587384211002621. doi: 10.1177/20587384211002621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Antiparasitic Drugs. [(accessed on 12 December 2022)]. Available online: https://my.clevelandclinic.org/health/drugs/22945-antiparasitic-drugs.
- 21.Williamson D.A., Carter G.P., Howden B.P. Current and Emerging Topical Antibacterials and Antiseptics: Agents, Action, and Resistance Patterns. Clin. Microbiol. Rev. 2017;30:827–860. doi: 10.1128/CMR.00112-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Al-Bakri A.G., Othman G., Bustanji Y. The assessment of the antibacterial and antifungal activities of aspirin, EDTA and aspirin-EDTA combination and their effectiveness as antibiofilm agents. J. Appl. Microbiol. 2009;107:280–286. doi: 10.1111/j.1365-2672.2009.04205.x. [DOI] [PubMed] [Google Scholar]
- 23.Sakkas H., Papadopoulou C. Antimicrobial Activity of Basil, Oregano, and Thyme Essential Oils. J. Microbiol. Biotechnol. 2017;27:429–438. doi: 10.4014/jmb.1608.08024. [DOI] [PubMed] [Google Scholar]
- 24.Fröhlich E.E., Farzi A., Mayerhofer R., Reichmann F., Jačan A., Wagner B., Zinser E., Bordag N., Magnes C., Fröhlich E., et al. Cognitive impairment by antibiotic-induced gut dysbiosis: Analysis of gut microbiota-brain communication. Brain Behav. Immun. 2016;56:140–155. doi: 10.1016/j.bbi.2016.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bercik P., Denou E., Collins J., Jackson W., Lu J., Jury J., Deng Y., Blennerhassett P., Macri J., McCoy K.D., et al. The intestinal microbiota affect central levels of brain-derived neurotropic factor and behavior in mice. Gastroenterology. 2011;141:599–609. doi: 10.1053/j.gastro.2011.04.052. [DOI] [PubMed] [Google Scholar]
- 26.Al-Naely A.J., Al-Hamadawi H.A., Alumeri J.K. An Examination of the Effect of Rutin against Neurotoxicity Induced by Ciprofloxacin Antibiotic in Wistar Rats. Arch. Razi Inst. 2022;77:835–841. doi: 10.22092/ari.2022.357094.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Czapińska-Ciepiela E. The risk of epileptic seizures during antibiotic therapy. Wiad. Lek. 2017;70:820–826. [PubMed] [Google Scholar]
- 28.Sun N., Hu H., Wang F., Li L., Zhu W., Shen Y., Xiu J., Xu Q. Antibiotic-induced microbiome depletion in adult mice disrupts blood-brain barrier and facilitates brain infiltration of monocytes after bone-marrow transplantation. Brain Behav. Immun. 2021;92:102–114. doi: 10.1016/j.bbi.2020.11.032. [DOI] [PubMed] [Google Scholar]
- 29.He J., Mao J., Hou L., Jin S., Wang X., Ding Z., Jin Z., Guo H., Dai R. Minocycline attenuates neuronal apoptosis and improves motor function after traumatic brain injury in rats. Exp. Anim. 2021;70:563–569. doi: 10.1538/expanim.21-0028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Glover M.E., Cohen J.L., Singer J.R., Sabbagh M.N., Rainville J.R., Hyland M.T., Morrow C.D., Weaver C.T., Hodes G.E., Kerman I.A., et al. Examining the Role of Microbiota in Emotional Behavior: Antibiotic Treatment Exacerbates Anxiety in High Anxiety-Prone Male Rats. Neuroscience. 2021;459:179–197. doi: 10.1016/j.neuroscience.2021.01.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mora S., Martín-González E., Prados-Pardo Á., Moreno J., López M.J., Pilar-Cuellar F., Castro E., Díaz Á., Flores P., Moreno M. Increased vulnerability to impulsive behavior after streptococcal antigen exposure and antibiotic treatment in rats. Brain Behav. Immun. 2020;89:675–688. doi: 10.1016/j.bbi.2020.08.010. [DOI] [PubMed] [Google Scholar]
- 32.Diaz Heijtz R., Wang S., Anuar F., Qian Y., Björkholm B., Samuelsson A., Hibberd M.L., Forssberg H., Pettersson S. Normal gut microbiota modulates brain development and behavior. Proc. Natl. Acad. Sci. USA. 2011;108:3047–3052. doi: 10.1073/pnas.1010529108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Desbonnet L., Clarke G., Shanahan F., Dinan T.G., Cryan J.F. Microbiota is essential for social development in the mouse. Mol. Psychiatry. 2014;19:146–148. doi: 10.1038/mp.2013.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Phelps D., Brinkman N.E., Keely S.P., Anneken E.M., Catron T.R., Betancourt D., Wood C.E., Espenschied S.T., Rawls J.F., Tal T. Microbial colonization is required for normal neurobehavioral development in zebrafish. Sci. Rep. 2017;7:11244. doi: 10.1038/s41598-017-10517-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang Y., Song J., Wang X., Qian Q., Wang H. Study on the toxic-mechanism of triclosan chronic exposure to zebrafish (Danio rerio) based on gut-brain axis. Sci. Total Environ. 2022;844:156936. doi: 10.1016/j.scitotenv.2022.156936. [DOI] [PubMed] [Google Scholar]
- 36.Liu J., Xiang C., Huang W., Mei J., Sun L., Ling Y., Wang C., Wang X., Dahlgren R.A., Wang H. Neurotoxicological effects induced by up-regulation of miR-137 following triclosan exposure to zebrafish (Danio rerio) Aquat. Toxicol. 2019;206:176–185. doi: 10.1016/j.aquatox.2018.11.017. [DOI] [PubMed] [Google Scholar]
- 37.Li J., Dong T., Keerthisinghe T.P., Chen H., Li M., Chu W., Yang J., Hu Z., Snyder S.A., Dong W., et al. Long-term oxytetracycline exposure potentially alters brain thyroid hormone and serotonin homeostasis in zebrafish. J. Hazard. Mater. 2020;399:123061. doi: 10.1016/j.jhazmat.2020.123061. [DOI] [PubMed] [Google Scholar]
- 38.Almeida A.R., Tacão M., Machado A.L., Golovko O., Zlabek V., Domingues I., Henriques I. Long-term effects of oxytetracycline exposure in zebrafish: A multi-level perspective. Chemosphere. 2019;222:333–344. doi: 10.1016/j.chemosphere.2019.01.147. [DOI] [PubMed] [Google Scholar]
- 39.Feng C.W., Wen Z.H., Huang S.Y., Hung H.C., Chen C.H., Yang S.N., Chen N.F., Wang H.M., Hsiao C.D., Chen W.F. Effects of 6-hydroxydopamine exposure on motor activity and biochemical expression in zebrafish (Danio rerio) larvae. Zebrafish. 2014;11:227–239. doi: 10.1089/zeb.2013.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Li J., Liu J., Zhang Y., Wang X., Li W., Zhang H., Wang H. Screening on the differentially expressed miRNAs in zebrafish (Danio rerio) exposed to trace β-diketone antibiotics and their related functions. Aquat. Toxicol. 2016;178:27–38. doi: 10.1016/j.aquatox.2016.07.009. [DOI] [PubMed] [Google Scholar]
- 41.Wang X., Zheng Y., Zhang Y., Li J., Zhang H., Wang H. Effects of β-diketone antibiotic mixtures on behavior of zebrafish (Danio rerio) Chemosphere. 2016;144:2195–2205. doi: 10.1016/j.chemosphere.2015.10.120. [DOI] [PubMed] [Google Scholar]
- 42.Petersen B.D., Pereira T.C.B., Altenhofen S., Nabinger D.D., Ferreira P.M.A., Bogo M.R., Bonan C.D. Antibiotic drugs alter zebrafish behavior. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2021;242:108936. doi: 10.1016/j.cbpc.2020.108936. [DOI] [PubMed] [Google Scholar]
- 43.Xi J., Liu J., He S., Shen W., Wei C., Li K., Zhang Y., Yue J., Yang Z. Effects of norfloxacin exposure on neurodevelopment of zebrafish (Danio rerio) embryos. Neurotoxicology. 2019;72:85–94. doi: 10.1016/j.neuro.2019.02.007. [DOI] [PubMed] [Google Scholar]
- 44.Tian D., Shi W., Yu Y., Zhou W., Tang Y., Zhang W., Huang L., Han Y., Liu G. Enrofloxacin exposure induces anxiety-like behavioral responses in zebrafish by affecting the microbiota-gut-brain axis. Sci. Total Environ. 2023;858:160094. doi: 10.1016/j.scitotenv.2022.160094. [DOI] [PubMed] [Google Scholar]
- 45.Han E., Ho Oh K., Park S., Chan Rah Y., Park H.C., Koun S., Choi J. Analysis of behavioral changes in zebrafish (Danio rerio) larvae caused by aminoglycoside-induced damage to the lateral line and muscles. Neurotoxicology. 2020;78:134–142. doi: 10.1016/j.neuro.2020.03.005. [DOI] [PubMed] [Google Scholar]
- 46.Agostini J.F., Costa N.L.F., Bernardo H.T., Baldin S.L., Mendes N.V., de Pieri Pickler K., Manenti M.C., Rico E.P. Ceftriaxone Attenuated Anxiety-Like Behavior and Enhanced Brain Glutamate Transport in Zebrafish Subjected to Alcohol Withdrawal. Neurochem. Res. 2020;45:1526–1535. doi: 10.1007/s11064-020-03008-z. [DOI] [PubMed] [Google Scholar]
- 47.Xu Y., Luo L., Chen J. Sulfamethoxazole induces brain capillaries toxicity in zebrafish by up-regulation of VEGF and chemokine signalling. Ecotoxicol. Environ. Saf. 2022;238:113620. doi: 10.1016/j.ecoenv.2022.113620. [DOI] [PubMed] [Google Scholar]
- 48.Cheng B., Jiang F., Su M., Zhou L., Zhang H., Cao Z., Liao X., Xiong G., Xiao J., Liu F., et al. Effects of lincomycin hydrochloride on the neurotoxicity of zebrafish. Ecotoxicol. Environ. Saf. 2020;201:110725. doi: 10.1016/j.ecoenv.2020.110725. [DOI] [PubMed] [Google Scholar]
- 49.Gonçalves C.L., Vasconcelos F.F.P., Wessler L.B., Lemos I.S., Candiotto G., Lin J., Matias M.B.D., Rico E.P., Streck E.L. Exposure to a high dose of amoxicillin causes behavioral changes and oxidative stress in young zebrafish. Metab. Brain Dis. 2020;35:1407–1416. doi: 10.1007/s11011-020-00610-6. [DOI] [PubMed] [Google Scholar]
- 50.Brinza I., Raey M.A.E., El-Kashak W., Eldahshan O.A., Hritcu L. Sweroside Ameliorated Memory Deficits in Scopolamine-Induced Zebrafish (Danio rerio) Model: Involvement of Cholinergic System and Brain Oxidative Stress. Molecules. 2022;27:5901. doi: 10.3390/molecules27185901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Capatina L., Todirascu-Ciornea E., Napoli E.M., Ruberto G., Hritcu L., Dumitru G. Thymus vulgaris Essential Oil Protects Zebrafish against Cognitive Dysfunction by Regulating Cholinergic and Antioxidants Systems. Antioxidants. 2020;9:1083. doi: 10.3390/antiox9111083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dong K., Li L., Chen C., Tengbe M.S., Chen K., Shi Y., Wu X., Qiu X. Impacts of cetylpyridinium chloride on the behavior and brain neurotransmitter levels of juvenile and adult zebrafish (Danio rerio) Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2022;259:109393. doi: 10.1016/j.cbpc.2022.109393. [DOI] [PubMed] [Google Scholar]
- 53.de Abreu M.S., Giacomini A., Demin K.A., Galstyan D.S., Zabegalov K.N., Kolesnikova T.O., Amstislavskaya T.G., Strekalova T., Petersen E.V., Kalueff A.V. Unconventional anxiety pharmacology in zebrafish: Drugs beyond traditional anxiogenic and anxiolytic spectra. Pharmacol. Biochem. Behav. 2021;207:173205. doi: 10.1016/j.pbb.2021.173205. [DOI] [PubMed] [Google Scholar]
- 54.Galstyan D.S., Demin K.A., Kolesnikova T.O., Kalueff A.V. Acute Behavioral Effects of Aspirin (Acetylsalicylic Acid) in Adult Zebrafish; Proceedings of the 27th Multidisciplinary International Neuroscience and Biological Psychiatry Conference “Stress and Behavior”; St-Petersburg, Russia. 16–18 September 2020. [Google Scholar]
- 55.Siebel A.M., Menezes F.P., da Costa Schaefer I., Petersen B.D., Bonan C.D. Rapamycin suppresses PTZ-induced seizures at different developmental stages of zebrafish. Pt BPharmacol. Biochem. Behav. 2015;139:163–168. doi: 10.1016/j.pbb.2015.05.022. [DOI] [PubMed] [Google Scholar]
- 56.Swaminathan A., Hassan-Abdi R., Renault S., Siekierska A., Riché R., Liao M., de Witte P.A.M., Yanicostas C., Soussi-Yanicostas N., Drapeau P., et al. Non-canonical mTOR-Independent Role of DEPDC5 in Regulating GABAergic Network Development. Curr. Biol. CB. 2018;28:1924–1937.e1925. doi: 10.1016/j.cub.2018.04.061. [DOI] [PubMed] [Google Scholar]
- 57.Gusso D., Altenhofen S., Fritsch P.M., Rübensam G., Bonan C.D. Oxytetracycline induces anxiety-like behavior in adult zebrafish. Toxicol. Appl. Pharmacol. 2021;426:115616. doi: 10.1016/j.taap.2021.115616. [DOI] [PubMed] [Google Scholar]
- 58.Kim J., Oh H., Ryu B., Kim U., Lee J.M., Jung C.R., Kim C.Y., Park J.H. Triclosan affects axon formation in the neural development stages of zebrafish embryos (Danio rerio) Environ. Pollut. 2018;236:304–312. doi: 10.1016/j.envpol.2017.12.110. [DOI] [PubMed] [Google Scholar]
- 59.Falisse E., Ducos B., Stockwell P.A., Morison I.M., Chatterjee A., Silvestre F. DNA methylation and gene expression alterations in zebrafish early-life stages exposed to the antibacterial agent triclosan. Environ. Pollut. 2018;243:1867–1877. doi: 10.1016/j.envpol.2018.10.004. [DOI] [PubMed] [Google Scholar]
- 60.Wang C., Huang W., Lin J., Fang F., Wang X., Wang H. Triclosan-induced liver and brain injury in zebrafish (Danio rerio) via abnormal expression of miR-125 regulated by PKCα/Nrf2/p53 signaling pathways. Chemosphere. 2020;241:125086. doi: 10.1016/j.chemosphere.2019.125086. [DOI] [PubMed] [Google Scholar]
- 61.Cronin A., Grealy M. Neuroprotective and Neuro-restorative Effects of Minocycline and Rasagiline in a Zebrafish 6-Hydroxydopamine Model of Parkinson’s Disease. Neuroscience. 2017;367:34–46. doi: 10.1016/j.neuroscience.2017.10.018. [DOI] [PubMed] [Google Scholar]
- 62.Zhao Y., Sun Q., Hu K., Ruan J., Yang X. Isolation, characterization, and tissue-specific expression of GABA A receptor α1 subunit gene of Carassius auratus gibelio after avermectin treatment. Fish Physiol. Biochem. 2016;42:83–92. doi: 10.1007/s10695-015-0119-9. [DOI] [PubMed] [Google Scholar]
- 63.Fukui K., Onodera K., Shinkai T., Suzuki S., Urano S. Impairment of Learning and Memory in Rats Caused by Oxidative Stress and Aging, and Changes in Antioxidative Defense Systems. Ann. N. Y. Acad. Sci. 2001;928:168–175. doi: 10.1111/j.1749-6632.2001.tb05646.x. [DOI] [PubMed] [Google Scholar]
- 64.Zullo A.J., Jurcic Smith K.L., Lee S. Mammalian target of Rapamycin inhibition and mycobacterial survival are uncoupled in murine macrophages. BMC Biochem. 2014;15:4. doi: 10.1186/1471-2091-15-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chen Z., Zhijie C., Yuting Z., Shilin X., Qichun Z., Jinying O., Chaohua L., Jing L., Zhixian M. Antibiotic-Driven Gut Microbiome Disorder Alters the Effects of Sinomenine on Morphine-Dependent Zebrafish. Front. Microbiol. 2020;11:946. doi: 10.3389/fmicb.2020.00946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.World Health Organization WHO Multi-Country Survey Reveals Widespread Public Misunderstanding about Antibiotic Resistance. [(accessed on 26 November 2022)]. Available online: https://www.who.int/about/policies/publishing/copyright/linking.
- 67.Benakis C., Brea D., Caballero S., Faraco G., Moore J., Murphy M., Sita G., Racchumi G., Ling L., Pamer E.G., et al. Commensal microbiota affects ischemic stroke outcome by regulating intestinal γδ T cells. Nat. Med. 2016;22:516–523. doi: 10.1038/nm.4068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Dai S., Hua Y., Keep R.F., Novakovic N., Fei Z., Xi G. Minocycline attenuates brain injury and iron overload after intracerebral hemorrhage in aged female rats. Neurobiol. Dis. 2019;126:76–84. doi: 10.1016/j.nbd.2018.06.001. [DOI] [PubMed] [Google Scholar]
- 69.Barks J.D.E., Liu Y., Wang L., Pai M.P., Silverstein F.S. Repurposing azithromycin for neonatal neuroprotection. Pediatr. Res. 2019;86:444–451. doi: 10.1038/s41390-019-0408-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Amantea D., Certo M., Petrelli F., Tassorelli C., Micieli G., Corasaniti M.T., Puccetti P., Fallarino F., Bagetta G. Azithromycin protects mice against ischemic stroke injury by promoting macrophage transition towards M2 phenotype. Pt 1Exp. Neurol. 2016;275:116–125. doi: 10.1016/j.expneurol.2015.10.012. [DOI] [PubMed] [Google Scholar]
- 71.Dik B., Coskun D., Bahcivan E., Er A. Doxycycline and meloxicam can treat neuroinflammation by increasing activity of antioxidant enzymes in rat brain. Pak. J. Pharm. Sci. 2019;32:391–396. [PubMed] [Google Scholar]
- 72.Horzmann K.A., Freeman J.L. Zebrafish Get Connected: Investigating Neurotransmission Targets and Alterations in Chemical Toxicity. Toxics. 2016;4:19. doi: 10.3390/toxics4030019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Joca S.R., Moreira F.A., Wegener G. Atypical Neurotransmitters and the Neurobiology of Depression. CNS Neurol. Disord. Drug Targets. 2015;14:1001–1011. doi: 10.2174/1871527314666150909114804. [DOI] [PubMed] [Google Scholar]
- 74.Calvo R., Schluessel V. Neural substrates involved in the cognitive information processing in teleost fish. Anim. Cogn. 2021;24:923–946. doi: 10.1007/s10071-021-01514-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Egan R.J., Bergner C.L., Hart P.C., Cachat J.M., Canavello P.R., Elegante M.F., Elkhayat S.I., Bartels B.K., Tien A.K., Tien D.H., et al. Understanding behavioral and physiological phenotypes of stress and anxiety in zebrafish. Behav. Brain Res. 2009;205:38–44. doi: 10.1016/j.bbr.2009.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kalueff A.V., Stewart A.M., Gerlai R. Zebrafish as an emerging model for studying complex brain disorders. Trends Pharmacol. Sci. 2014;35:63–75. doi: 10.1016/j.tips.2013.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Geng Y., Peterson R.T. The zebrafish subcortical social brain as a model for studying social behavior disorders. Dis. Model. Mech. 2019;12:dmm039446. doi: 10.1242/dmm.039446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Klee E.W., Schneider H., Clark K.J., Cousin M.A., Ebbert J.O., Hooten W.M., Karpyak V.M., Warner D.O., Ekker S.C. Zebrafish: A model for the study of addiction genetics. Hum. Genet. 2012;131:977–1008. doi: 10.1007/s00439-011-1128-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Tayanloo-Beik A., Hamidpour S.K., Abedi M., Shojaei H., Tavirani M.R., Namazi N., Larijani B., Arjmand B. Zebrafish Modeling of Autism Spectrum Disorders, Current Status and Future Prospective. Front. Psychiatry. 2022;13:911770. doi: 10.3389/fpsyt.2022.911770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.McGrath P., Li C.Q. Zebrafish: A predictive model for assessing drug-induced toxicity. Drug Discov. Today. 2008;13:394–401. doi: 10.1016/j.drudis.2008.03.002. [DOI] [PubMed] [Google Scholar]
- 81.North T.E., Goessling W., Walkley C.R., Lengerke C., Kopani K.R., Lord A.M., Weber G.J., Bowman T.V., Jang I.H., Grosser T., et al. Prostaglandin E2 regulates vertebrate haematopoietic stem cell homeostasis. Nature. 2007;447:1007–1011. doi: 10.1038/nature05883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yeh J.R., Munson K.M., Elagib K.E., Goldfarb A.N., Sweetser D.A., Peterson R.T. Discovering chemical modifiers of oncogene-regulated hematopoietic differentiation. Nat. Chem. Biol. 2009;5:236–243. doi: 10.1038/nchembio.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhang Y., Wang J., Wheat J., Chen X., Jin S., Sadrzadeh H., Fathi A.T., Peterson R.T., Kung A.L., Sweetser D.A., et al. AML1-ETO mediates hematopoietic self-renewal and leukemogenesis through a COX/β-catenin signaling pathway. Blood. 2013;121:4906–4916. doi: 10.1182/blood-2012-08-447763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Crilly S., Parry-Jones A., Wang X., Selley J.N., Cook J., Tapia V.S., Anderson C.S., Allan S.M., Kasher P.R. Zebrafish drug screening identifies candidate therapies for neuroprotection after spontaneous intracerebral haemorrhage. Dis. Model. Mech. 2022;15:dmm049227. doi: 10.1242/dmm.049227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Owens K.N., Santos F., Roberts B., Linbo T., Coffin A.B., Knisely A.J., Simon J.A., Rubel E.W., Raible D.W. Identification of genetic and chemical modulators of zebrafish mechanosensory hair cell death. PLoS Genet. 2008;4:e1000020. doi: 10.1371/journal.pgen.1000020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Smuga-Otto K. Zebrafish assay forges new approach to drug discovery. Proc. Natl. Acad. Sci. USA. 2018;115:5306–5308. doi: 10.1073/pnas.1806440115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.de Abreu M.S., Costa F., Giacomini A., Demin K.A., Petersen E.V., Rosemberg D.B., Kalueff A.V. Exploring CNS Effects of American Traditional Medicines using Zebrafish Models. Curr. Neuropharmacol. 2022;20:550–559. doi: 10.2174/1570159x19666210712153329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Chu C., Murdock M.H., Jing D., Won T.H., Chung H., Kressel A.M., Tsaava T., Addorisio M.E., Putzel G.G., Zhou L., et al. The microbiota regulate neuronal function and fear extinction learning. Nature. 2019;574:543–548. doi: 10.1038/s41586-019-1644-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Cordella F., Sanchini C., Rosito M., Ferrucci L., Pediconi N., Cortese B., Guerrieri F., Pascucci G.R., Antonangeli F., Peruzzi G., et al. Antibiotics Treatment Modulates Microglia-Synapses Interaction. Cells. 2021;10:2648. doi: 10.3390/cells10102648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wang H.T., Huang F.L., Hu Z.L., Zhang W.J., Qiao X.Q., Huang Y.Q., Dai R.P., Li F., Li C.Q. Early-Life Social Isolation-Induced Depressive-Like Behavior in Rats Results in Microglial Activation and Neuronal Histone Methylation that Are Mitigated by Minocycline. Neurotox. Res. 2017;31:505–520. doi: 10.1007/s12640-016-9696-3. [DOI] [PubMed] [Google Scholar]
- 91.Kobayashi K., Imagama S., Ohgomori T., Hirano K., Uchimura K., Sakamoto K., Hirakawa A., Takeuchi H., Suzumura A., Ishiguro N., et al. Minocycline selectively inhibits M1 polarization of microglia. Cell Death Dis. 2013;4:e525. doi: 10.1038/cddis.2013.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Li T., Liu T., Chen X., Li L., Feng M., Zhang Y., Wan L., Zhang C., Yao W. Microglia induce the transformation of A1/A2 reactive astrocytes via the CXCR7/PI3K/Akt pathway in chronic post-surgical pain. J. Neuroinflamm. 2020;17:211. doi: 10.1186/s12974-020-01891-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Wu Q., Zhang Y., Zhang Y., Xia C., Lai Q., Dong Z., Kuang W., Yang C., Su D., Li H., et al. Potential effects of antibiotic-induced gut microbiome alteration on blood-brain barrier permeability compromise in rhesus monkeys. Ann. N. Y. Acad. Sci. 2020;1470:14–24. doi: 10.1111/nyas.14312. [DOI] [PubMed] [Google Scholar]
- 94.Rosenberg G.A. Neurological diseases in relation to the blood-brain barrier. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2012;32:1139–1151. doi: 10.1038/jcbfm.2011.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Angelucci F., Cechova K., Amlerova J., Hort J. Antibiotics, gut microbiota, and Alzheimer’s disease. J. Neuroinflamm. 2019;16:108. doi: 10.1186/s12974-019-1494-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wang W., Sidoli S., Zhang W., Wang Q., Wang L., Jensen O.N., Guo L., Zhao X., Zheng L. Abnormal levels of histone methylation in the retinas of diabetic rats are reversed by minocycline treatment. Sci. Rep. 2017;7:45103. doi: 10.1038/srep45103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Jaggar M., Rea K., Spichak S., Dinan T.G., Cryan J.F. You’ve got male: Sex and the microbiota-gut-brain axis across the lifespan. Front. Neuroendocrinol. 2020;56:100815. doi: 10.1016/j.yfrne.2019.100815. [DOI] [PubMed] [Google Scholar]
- 98.Minter M.R., Zhang C., Leone V., Ringus D.L., Zhang X., Oyler-Castrillo P., Musch M.W., Liao F., Ward J.F., Holtzman D.M., et al. Antibiotic-induced perturbations in gut microbial diversity influences neuro-inflammation and amyloidosis in a murine model of Alzheimer’s disease. Sci. Rep. 2016;6:30028. doi: 10.1038/srep30028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Winter M.J., Pinion J., Tochwin A., Takesono A., Ball J.S., Grabowski P., Metz J., Trznadel M., Tse K., Redfern W.S., et al. Functional brain imaging in larval zebrafish for characterising the effects of seizurogenic compounds acting via a range of pharmacological mechanisms. Br. J. Pharmacol. 2021;178:2671–2689. doi: 10.1111/bph.15458. [DOI] [PubMed] [Google Scholar]
- 100.Rosa J.G.S., Lima C., Lopes-Ferreira M. Zebrafish Larvae Behavior Models as a Tool for Drug Screenings and Pre-Clinical Trials: A Review. Int. J. Mol. Sci. 2022;23:6647. doi: 10.3390/ijms23126647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yalcin H.C., Amindari A., Butcher J.T., Althani A., Yacoub M. Heart function and hemodynamic analysis for zebrafish embryos. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2017;246:868–880. doi: 10.1002/dvdy.24497. [DOI] [PubMed] [Google Scholar]
- 102.Zhao X., Pack M. Modeling intestinal disorders using zebrafish. Methods Cell Biol. 2017;138:241–270. doi: 10.1016/bs.mcb.2016.11.006. [DOI] [PubMed] [Google Scholar]
- 103.Novoa B., Figueras A. Zebrafish: Model for the study of inflammation and the innate immune response to infectious diseases. Adv. Exp. Med. Biol. 2012;946:253–275. doi: 10.1007/978-1-4614-0106-3_15. [DOI] [PubMed] [Google Scholar]
- 104.Lin S.W., Ge W. Differential regulation of gonadotropins (FSH and LH) and growth hormone (GH) by neuroendocrine, endocrine, and paracrine factors in the zebrafish—An in vitro approach. Gen. Comp. Endocrinol. 2009;160:183–193. doi: 10.1016/j.ygcen.2008.11.020. [DOI] [PubMed] [Google Scholar]
- 105.Abbott T., Kor-Bicakci G., Islam M.S., Eskicioglu C. A Review on the Fate of Legacy and Alternative Antimicrobials and Their Metabolites during Wastewater and Sludge Treatment. Int. J. Mol. Sci. 2020;21:9241. doi: 10.3390/ijms21239241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Czech B., Krzyszczak A., Boguszewska-Czubara A., Opielak G., Jośko I., Hojamberdiev M. Revealing the toxicity of lopinavir- and ritonavir-containing water and wastewater treated by photo-induced processes to Danio rerio and Allivibrio fischeri. Sci. Total Environ. 2022;824:153967. doi: 10.1016/j.scitotenv.2022.153967. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.