Abstract
Colonic metastasis from ovarian cancer is extremely rare, with only seven reported cases. A 77-year-old woman who had previously undergone surgery for ovarian cancer was admitted to a local hospital with anal bleeding. Histopathological analysis confirmed the presence of adenocarcinoma. Colonoscopy revealed a descending colon tumor. The patient was diagnosed with Union for International Cancer Control T3N0M0 descending colon cancer or colon metastasis of the ovarian cancer. Laparoscopic left colectomy was performed; intraoperative frozen section diagnosis confirmed metastasis from ovarian cancer, and the absence of invasion to the serosal surface suggested hematogenous metastasis. This is the first case of colonic metastasis from ovarian cancer that was diagnosed using an intraoperative frozen section and laparoscopically treated.
Keywords: Ovarian cancer, Metastatic colon cancer, Clear cell carcinoma
Introduction
Ovarian cancer is the leading cause of cancer-related deaths among women with gynecological cancers. Although distant metastasis is the most common cause of death, colonic metastasis by hematogenous dissemination is extremely rare; only 7 cases have been reported [1–7]. Ovarian cancer tends to spread through direct invasion, intraperitoneal seeding, and lymphatic or hematogenous dissemination [1, 8]. Most cases of colonic metastasis from ovarian cancer are caused by direct invasion or intraperitoneal seeding; however, hematogenous dissemination is rare. Furthermore, colonic metastases from ovarian and primary colorectal cancers have similar endoscopic findings. To date, there are no case reports in which the diagnosis of metastasis of ovarian cancer to the colon or primary colorectal cancer has been made preoperatively. We present a case of colonic metastasis from ovarian cancer that was diagnosed using an intraoperative frozen section. The patient underwent a laparoscopic left colectomy with regional lymph node dissection. To the best of our knowledge, this is the first report of such a case.
Case Report
A 77-year-old woman was admitted to a local hospital with a chief complaint of anal bleeding. She had previously been diagnosed with stage IC ovarian carcinoma and had undergone bilateral salpingo-oophorectomy, total hysterectomy, and omentectomy at the gynecology department of our hospital 3 years earlier. Intraoperative findings included no peritoneal dissemination or liver metastasis. The final histopathological examination showed right ovarian clear cell carcinoma. She received adjuvant chemotherapy with a total of six cycles of carboplatin and paclitaxel on a monthly basis. The patient initially presented to a local hospital, and a colonoscopy was performed. Colonoscopy revealed a raised tumor, approximately 40 mm in diameter, in the descending colon. Biopsies were performed at four sites under colonoscopy; however, the histopathology of the biopsy specimen revealed no malignancy, and the patient was referred to our hospital for further evaluation and treatment. Laboratory investigations revealed normal serum hemoglobin levels. The levels of tumor markers such as carcinoembryonic antigen, carbohydrate antigen 19-9, and carbohydrate antigen 125 (CA125) were within the normal ranges at 1.4 ng/mL (normal, 0–5.0 ng/mL), 9 U/mL (normal, 0–37.0 U/mL), and 7 U/mL (normal, 0–30.0 U/mL), respectively. However, the serum anti-p53 antibody level was slightly elevated at 3.06 U/mL (normal, 0–1.3 U/mL). Colonoscopy was re-performed at our hospital, and six more biopsies were performed. Additionally, the endoscopic image revealed atypical findings (two humps and elevation of the submucosa) with regard to typical colorectal cancer (shown in Fig. 1). Since the patient had a history of ovarian cancer, the possibility of metastasis of ovarian cancer could not be ruled out. This was supported by the gynecologist. However, at the time of diagnosis of ovarian cancer, the CA125 level was elevated at 54 U/mL, but it was within the normal range before colorectal tumor surgery. Histopathological analysis revealed the tumor to be adenocarcinoma; however, the biopsy was too small to evaluate whether it was primary colon cancer or metastasis from the ovary. Abdominal computed tomography (CT) revealed a bulky intraluminal mass in the descending colon, with no evidence of liver metastasis, enlarged lymph nodes, or peritoneal metastasis. Fluorodeoxyglucose positron emission tomography (FDG-PET)/CT scans showed focal uptake (maximum standardized uptake value, 8.5) in the descending colon. The patient was diagnosed with Union for International Cancer Control T3N0M0 descending colon cancer or colon metastasis of ovarian cancer, and a laparoscopic colectomy was planned. In addition, we decided to perform an intraoperative frozen section of the resected tumor. Subsequently, laparoscopic left colectomy with regional lymph node dissection and peritoneal lavage cytology were performed. No findings related to peritoneal dissemination were observed. Intraoperative frozen section diagnosis confirmed ovarian cancer metastasis. The surgery was completed after confirming that there was no dissemination of ovarian cancer by laparoscopy.
Fig. 1.
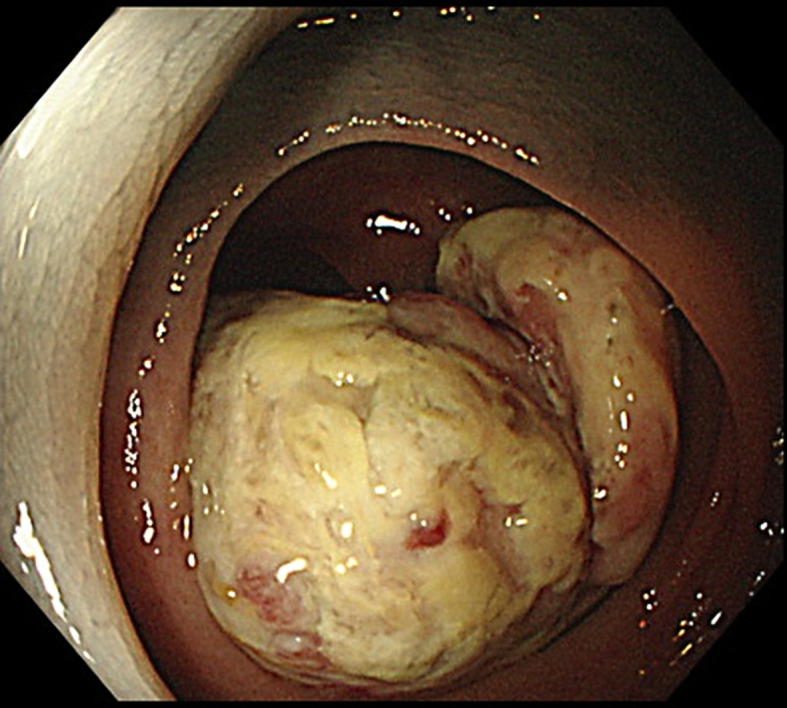
Colonoscopy shows an elevated tumorous lesion in the descending colon.
Microscopically, the tumor showed solid growth of clear cells admixed with glandular and cystic growth of cells with hobnail features that had invaded the entire colonic wall. The histopathological features were similar to those of previously resected ovarian cancer, and a diagnosis of metastasis of clear cell carcinoma to the descending colon was confirmed. Immunostaining was not performed because the diagnosis of ovarian cancer metastasis could be made with the hematoxylin and eosin staining findings. No lymph node metastasis was detected (shown in Fig. 2, 3). Peritoneal lavage cytology was class 1. The patient was discharged 10 days postoperatively and underwent six cycles of adjuvant chemotherapy on a monthly basis with carboplatin and paclitaxel for recurrent ovarian carcinoma with colorectal metastasis. There were no signs of recurrence at the 1-year follow-up.
Fig. 2.
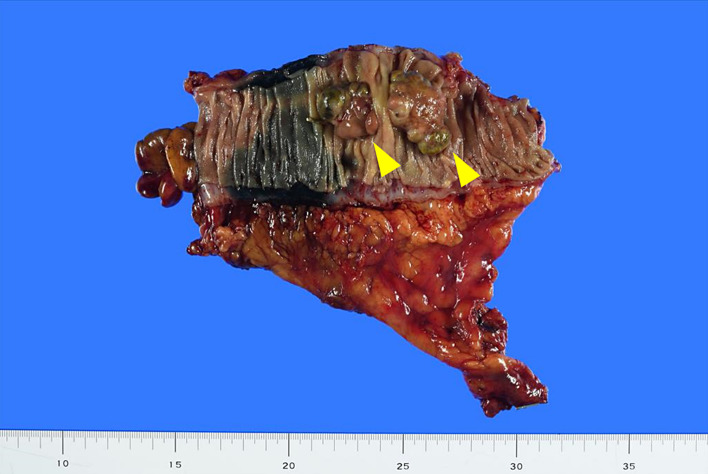
Macroscopic evaluation of the resected specimen. An elevated tumor measuring approximately 50 mm in diameter is observed in the descending colon (arrowhead).
Fig. 3.
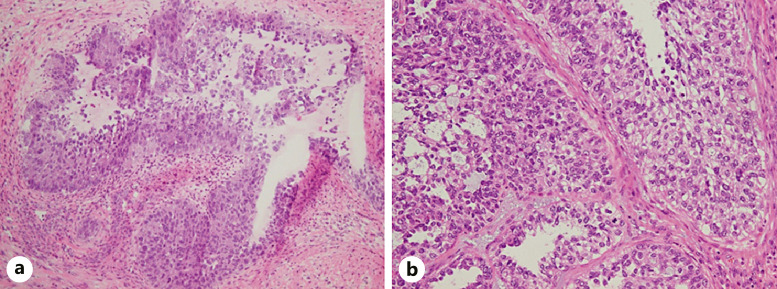
Histology of the tumor excised from the descending colon. a Intraoperative frozen section of the tumor showing solid growth of clear cells admixed with glandular and cystic growth of cells with hobnail features. b Histopathology of the paraffin-embedded specimen showing the cytoplasm of the tumor cells more clearly and brightly than the intraoperative frozen section (hematoxylin and eosin stain, magnification ×20).
Conclusion
Colonic metastasis is rare and accounts for 0.1–1.0% of all colorectal tumors [9]. Colorectal metastasis from ovarian cancer accounts for 6% of colorectal cancers in women [10]. We report the case of a patient who underwent a surgery for ovarian cancer and presented with a descending colon tumor 2 years later, which was confirmed to be colon metastasis, and she underwent colectomy. Ovarian cancer tends to spread through direct invasion and intraperitoneal seeding, but rarely hematogenously [1, 8]. In this case, a complete surgical resection for ovarian cancer was performed before colon metastasis, and peritoneal lavage cytology was negative; therefore, hematogenous metastasis may have occurred. Metastatic forms may be distinguished as dissemination or hematogenesis based on the presence or absence of serosal involvement, respectively. In this case, the two humps of the tumor consisted of a single mass that continued below the submucosa. The metastatic tumor primarily involved the colonic mucosa, with only focal invasion of the superficial colonic muscularis propria, and the tumor did not involve the attached pericolic adipose tissue. Therefore, these pathological findings indicated that the mode of metastasis was hematogenous dissemination. However, it was difficult to determine whether the tumor was infiltrating from the serous or mucosal surface based on preoperative CT findings; the pathological findings ultimately confirmed that it was a hematogenous metastasis to the colon.
We identified previously published case reports of hematogenic colorectal metastasis from ovarian cancer through PubMed search, using “colon metastasis” and “ovarian cancer” as keywords. Only 7 cases of colorectal metastasis from ovarian cancer that appeared to have metastasized hematogenously have been reported, and their clinicopathological findings are summarized in Table 1[1–7]. The metastasis site in all cases was in the left colon including the rectum. The site of origin of ovarian cancer was on the left side in 4 cases, on the right side in 2 cases, and unknown in 2 cases. The relationship between the site of ovarian cancer and the location of metastases is unclear. The median time to colon metastasis following primary surgery for ovarian cancer ranges from 8 months to 19 years. Since the histological type of ovarian cancer differs between reports, no unified view has been obtained, although clear cell carcinoma was the most reported histological type (37.5%). Surgical treatment was performed in all cases except for one in which chemotherapy was administered. As for the surgical technique, laparoscopic colectomy can be considered a safe and minimally invasive procedure that permits visual examination of the abdominal cavity. This is the first reported case of laparoscopic resection of colon metastases from ovarian cancer. In addition, although almost all previous reports indicated a poor prognosis for colonic metastasis of ovarian tumors, there was no distant metastasis or recurrence in the present case.
Table 1.
Colorectal metastasis of ovarian cancer with possible hematogenous
| Case | Year | Authors | Age | Side of ovarian cancer | Time to recurrence | Symptoms (yes/no) | Location | Stage (FIGO) | Metastatic route | Histology | Treatment | Postoperative course |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1999 | Sean et al. [1] | 44 | Left | 9 yr | Yes | S | IC | Hemat. | Clear cell carcinoma | Open surgery | No recurrence |
| 2 | 2004 | Zighelboim et al. [2] | 39 | N/A | 6 mo | Yes | S | N/A | N/A | High-grade serous carcinoma and endometrioid adenocarcinoma | Open surgery | 2 yr; no recurrence |
| 3 | 2004 | Cynthia et al. [3] | 35 | Left | 19 yr | Yes | R | III | Hemat. | Clear cell carcinoma | Open surgery | Hepatic metastasis |
| 4 | 2012 | Aqsa et al. [4] | 53 | Left | 2 yr | No | R | IC | Hemat. or Lymph. | N/A | Robot-assisted surgery | No recurrence |
| 5 | 2014 | Amzerin et al. [5] | 61 | N/A | 11 mo | Yes | R | IV | N/A | N/A | Chemotherapy | 1 yr; dead |
| 6 | 2015 | Kim et al. [6] | 70 | Right | 8 yr | Yes | R | IIIB | N/A | Papillary serous adenocarcinoma | Open surgery | 15 mo; no recurrence |
| 7 | 2019 | Bhange et al. [7] | 33 | Left | 13 yr | No | R | IV | N/A | Papillary serous adenocarcinoma | Open surgery | No recurrence |
| 8 | 2020 | Present case | 78 | Right | 2 yr | Yes | D | IC | Hemat. | Clear cell carcinoma | Laparoscopic surgery | 6 mo; no recurrence |
S, sigmoid colon; R, rectum; D, descending colon; Hemat., hematogenous metastasis; Lymph., lymphatic metastasis; yr, year; mo, month; N/A, not available.
In the present case, distinguishing ovarian cancer from colon cancer preoperatively was difficult owing to the biopsy specimen, which was only a small portion of the tumor that did not characterize the entire lesion. Previous studies have reported that immunohistochemical evaluation is useful for differentiating ovarian and colon cancers [1]. Ovarian metastasis has been reported to be positive for cytokeratin 7 and estrogen receptor and negative for cytokeratin 20 [11]. All previously reported cases were confirmed using postoperative specimens. Although endometrioid and mucinous types of ovarian cancer are indistinguishable from colon cancer by histology, ovarian clear cell carcinoma can be distinguished from adenocarcinoma. In this case, the preoperative diagnosis of the tumor was inconclusive, and frozen section diagnosis was important during the surgery. Although we consequently performed surgery as scheduled, it was possible to alter the other methods of operation depending on the diagnosis. To the best of our knowledge, this is the first case in which colon metastasis was diagnosed using an intraoperative frozen section. In addition, treatment by laparoscopic surgery allowed for the minimally invasive and extensive examination of the abdominal cavity. Few reports have compared endoscopic surgery with conventional laparotomy, and there is no consensus on the choice of the procedure, which is currently decided by the surgeon. However, there are reports that minimally invasive techniques, such as laparoscopy and robotic surgery, for locally recurrent cases, contribute to improved perioperative outcomes [12]. Minimally invasive surgery is reported to cause less intraoperative blood loss, shorter hospital stay, fewer postoperative complications, and no significant difference in the postoperative disease-free survival rate compared with open surgery [13–15]. On lymphadenectomy for colon metastasis of ovarian cancer, O’Hanlan et al. [8] hypothesized that ovarian metastasis to the gastrointestinal tract might mimic the colonic pattern of dissemination. Moreover, they concluded that the resection of a wedge mesentery to include the paracolic and intermediate nodes and removal of the intestine while including sufficient longitudinal margins should be considered to achieve a complete resection of the tumor [8].
In conclusion, we report a case of colon metastasis from ovarian cancer in which intraoperative frozen section diagnosis was performed and laparoscopic colectomy was the treatment of choice. It is important to note that ovarian cancer can metastasize to the colon via hematogenous dissemination. Although immunohistochemical evaluation is helpful for definitive diagnosis, hematoxylin and eosin staining may be sufficient to confirm the metastasis of ovarian cancer with certain histologic subtypes. Further accumulation of data and investigation of colon metastasis from ovarian cancer is necessary. The CARE Checklist has been completed by the authors for this case report, attached as online supplementary material (for all online suppl. material, see www.karger.com/doi/10.1159/000529299).
Acknowledgments
We would like to thank Editage [http://www.editage.com] for editing and reviewing this manuscript for English language.
Statement of Ethics
This study protocol was reviewed, and the need for approval was waived by the Research Ethics Committee of the Faculty of Medicine of the University of Tokyo. This retrospective review of patient data did not require ethical approval in accordance with local/national guidelines. Written informed consent was obtained from the patient for the publication of the details of their medical case and any accompanying images.
Conflict of Interest Statement
The authors have no conflicts of interest to declare.
Funding Sources
This study did not receive any funding.
Author Contributions
Kentaro Abe and Hiroyuki Anzai made substantial contributions to the study concept or the data analysis or interpretation; Satoko Eguchi, Masako Ikemura, Aya Shinozaki-Ushiku, Takahide Shinagawa, Hirofumi Sonoda, Yuichiro Yoshioka, Yuzo Nagai, Shinya Abe, Hiroyuki Matsuzaki, Yuichiro Yokoyama, Shigenobu Emoto, Koji Murono, Kazuhito Sasaki, Hiroaki Nozawa, Tetsuo Ushiku, and Soichiro Ishihara drafted the manuscript or revised it critically for important intellectual content; Kentaro Abe, Hiroyuki Anzai, Satoko Eguchi, Masako Ikemura, Aya Shinozaki-Ushiku, Takahide Shinagawa, Hirofumi Sonoda, Yuichiro Yoshioka, Yuzo Nagai, Shinya Abe, Hiroyuki Matsuzaki, Yuichiro Yokoyama, Shigenobu Emoto, Koji Murono, Kazuhito Sasaki, Hiroaki Nozawa, Tetsuo Ushiku, and Soichiro Ishihara approved the final version of the manuscript to be published and agreed to be accountable for all aspects of the work.
Funding Statement
This study did not receive any funding.
Data Availability Statement
All data generated or analyzed during this study are included in this article and its online supplementary material files. Further inquiries can be directed to the corresponding author.
References
- 1. Dowdy SC, Pfeifer EA, Longcope DC, Cliby WA. Unusual recurrence of ovarian carcinoma 9 years after initial diagnosis. Gynecol Oncol. 1999;74(3):495–8. 10.1006/gyno.1999.5465. [DOI] [PubMed] [Google Scholar]
- 2. Zighelboim I, Broaddus R, Ramirez PT. Atypical sigmoid metastasis from a high-grade mixed adenocarcinoma of the ovary. Gynecol Oncol. 2004;94:850–3. 10.1016/j.ygyno.2004.05.058. [DOI] [PubMed] [Google Scholar]
- 3. Trastour C, Rahili A, Schumacker C, Effi AB, Bernard JL. Hematogenous rectal metastasis 20 years after removal of epithelial ovarian cancer. Gynecol Oncol. 2004;94(2):584–8. 10.1016/j.ygyno.2004.05.022. [DOI] [PubMed] [Google Scholar]
- 4. Aqsa A, Droubi S, Amarnath S, Haddad F, Deeb L. Colorectal metastasis from ovarian neoplasm mimicking primary colon cancer. Case Rep Gastroenterol. 2021;15(1):41–6. 10.1159/000510934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Amzerin M, Garcia C, Stanciu C, Veys I, Awada A, Errihani H, et al. Case Report: mammary and rectal metastases from an ovarian cancer: report of two cases and review of literature. F1000Res. 2014;3:255. 10.12688/f1000research.2644.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Kim JR, Kim BM, Kim YM, Lee WA, Namgung H. Colonic metastasis presenting as an intraluminal fungating mass 8 years after surgery for ovarian cancer. Ann Coloproctol. 2015;31(5):198–201. 10.3393/ac.2015.31.5.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Bhange SA, Bhansali M, Shaikh T, Ajgaonkar U. Colonic metastases 13 years after the primary ovarian cancer: a case study with a brief review of literature. BMJ Case Rep. 2019;12(7):e230127. 10.1136/bcr-2019-230127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. O’Hanlan KA, Kargas S, Schreiber M, Burrs D, Mallipeddi P, Longacre T, et al. Ovarian carcinoma metastases to gastrointestinal tract appear to spread like colon carcinoma: implications for surgical resection. Gynecol Oncol. 1995;59(2):200–6. 10.1006/gyno.1995.0008. [DOI] [PubMed] [Google Scholar]
- 9. Balthazar EJ, Rosenberg HD, Davidian MM. Primary and metastatic scirrrhous carcinoma of the rectum. AJR Am J Roentgenol. 1979;132(5):711–5. 10.2214/ajr.132.5.711. [DOI] [PubMed] [Google Scholar]
- 10. Lindner V, Gasser B, Debbiche A, Tomb L, Vetter JM, Walter P. Ovarian metastasis of colorectal adenocarcinomas. A clinico-pathological study of 41 cases. Ann Pathol. 1999;19(6):492–8. [PubMed] [Google Scholar]
- 11. Loy TS, Calaluce RD, Keeney GL. Cytokeratin immunostaining in differentiating primary ovarian carcinoma from metastatic colonic adenocarcinoma. Mod Pathol. 1996;9(11):1040–4. [PubMed] [Google Scholar]
- 12. Uccella S, Franchi MP, Cianci S, Zorzato PC, Bertoli F, Alletti SG, et al. Laparotomy vs. minimally invasive surgery for ovarian cancer recurrence: a systematic review. Gland Surg. 2020;9(4):1130–9. 10.21037/gs-20-353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Fagotti A, Costantini B, Gallotta V, Cianci S, Ronsini C, Petrillo M, et al. Minimally invasive secondary cytoreduction plus HIPEC versus open surgery plus HIPEC in isolated relapse from ovarian cancer: a retrospective cohort study on perioperative outcomes. J Minim Invasive Gynecol. 2015;22(3):428–32. 10.1016/j.jmig.2014.11.008. [DOI] [PubMed] [Google Scholar]
- 14. Magrina JF, Cetta RL, Chang YH, Guevara G, Magtibay PM. Analysis of secondary cytoreduction for recurrent ovarian cancer by robotics, laparoscopy and laparotomy. Gynecol Oncol. 2013;129(2):336–40. 10.1016/j.ygyno.2013.01.015. [DOI] [PubMed] [Google Scholar]
- 15. Eriksson AGZ, Graul A, Yu MC, Halko A, Chi DS, Zivanovic O, et al. Minimal access surgery compared to laparotomy for secondary surgical cytoreduction in patients with recurrent ovarian carcinoma: perioperative and oncologic outcomes. Gynecol Oncol. 2017;146(2):263–7. 10.1016/j.ygyno.2017.05.022. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this article and its online supplementary material files. Further inquiries can be directed to the corresponding author.