Abstract
Background
The treatment of chronic type B aortic dissection by thoracic endovascular aortic repair has some challenges, and its long‐term outcomes remain unclear. This study aimed to analyze the 5‐year clinical outcomes of thoracic endovascular aortic repair of chronic type B aortic dissection, compare the differences between patients with and without adverse aortic events (AAEs), and identify risk factors for AAEs.
Methods and Results
Patients who underwent thoracic endovascular aortic repair of chronic type B aortic dissection from January 2009 to June 2017 were retrospectively enrolled. The primary end points were AAEs, including aorta‐related death, procedural complications, and disease progression requiring reintervention. Clinical outcomes were described at the 5‐year follow‐up visit. The secondary end point was the comparison of the results between patients with and without AAEs. Univariable and multivariable logistic analyses were used to identify potential risk factors for AAEs. A total of 214 patients were enrolled. AAEs occurred in 46 (21.5%) patients. Compared with patients without AAEs, those with AAEs had higher rates of residual type A aortic dissection (26.1% versus 4.2%, P<0.001) and aortic diameter ≥5.5 cm (69.6% versus 11.3%, P<0.001), and a lower rate of complete false lumen thrombosis (23.9% versus 89.9%, P<0.001). Meanwhile, the median interval from symptom onset to intervention was longer in patients with AAEs (26 months versus 12 months, P=0.004). Partial or no false lumen thrombosis (adjusted odds ratio [AOR], 14.71 [95% CI, 5.67–38.14; P<0.001]) and aortic diameter ≥5.5 cm (AOR, 10.16 [95% CI, 3.86–26.73; P<0.001]) were identified as independent risk factors for AAEs.
Conclusions
While thoracic endovascular aortic repair of chronic type B aortic dissection might be challenging in some cases, its long‐term outcomes appeared promising as this treatment was effective in preventing catastrophic aortic events. Patients with AAEs showed higher rates of residual type A aortic dissection and aortic diameter ≥5.5 cm, a lower rate of complete false lumen thrombosis, and a longer median interval from symptom onset to intervention. Failure of complete false lumen thrombosis and an aortic diameter ≥5.5 cm were predictors of AAEs.
Keywords: aortic remodeling, chronic type B aortic dissection, long‐term outcomes, thoracic endovascular aortic repair
Subject Categories: Cardiovascular Surgery, Stent
Nonstandard Abbreviations and Acronyms
- AAE
adverse aortic event
- AD
aortic dissection
- cTBAD
chronic type B aortic dissection
- FL
false lumen
- LCCA
left common carotid artery
- LSA
left subclavian artery
- RCCA
right common carotid artery
- TBAD
type B aortic dissection
- TEVAR
thoracic endovascular aortic repair
- TL
true lumen
Clinical Perspective
What Is New?
Patients with adverse aortic events showed higher rates of residual type A aortic dissection and aortic diameter ≥5.5 cm, a lower rate of complete false lumen thrombosis, and a longer median interval from symptom onset to intervention.
Failure of complete false lumen thrombosis and aortic diameter ≥5.5 cm are predictors of adverse aortic events.
What Are the Clinical Implications?
The long‐term outcome is promising and effective for thoracic endovascular repair of chronic type B aortic dissection.
More aggressive thoracic endovascular repair seems reasonable for chronic type B aortic dissection in suitable patients.
Aortic dissection (AD) is the most common life‐threatening disorder of the aorta, and its incidence has been reported to be ≈3/100 000 per year. 1 , 2 AD is categorized as acute phase (<14 days), subacute (15–90 days), and chronic (>90 days). 3 Chronic type B AD (cTBAD) also includes cases previously operated for type A AD, with persisting dissection of the descending aorta. 3 The optimal treatment for cTBAD remains unclear. Notably, aorta‐related complications might occur in 20% to 50% of patients with cTBAD. 4 Overall, ≈20% to 40% of patients with cTBAD developed false lumen (FL) enlargement requiring treatment. 5 Meanwhile, the estimated rupture rate was 30% once aortic expansion reached 60 mm. 6 Although great progress has been made during the past decades, the mortality and morbidity, including spinal cord ischemia, cerebrovascular events, and renal failure of open surgery, remains ≈30%. Thoracic endovascular aortic repair (TEVAR), a minimally invasive procedure, has become the dominant treatment for complicated acute type B AD (TBAD). 7 , 8 Despite the clear superiority of TEVAR over conventional open surgery for complicated TBAD, considerable debate exists on its application for cTBAD. Several large single‐center retrospective studies have reported the outcomes of TEVAR for cTBAD. In chronic cases, TEVAR appeared safe, with stroke and spinal cord ischemia rates of <3% in most series. Early mortality was also <5% in most series. 9 , 10 However, most previous studies were confined to small sample sizes with limited follow‐up. The consensus for TEVAR in the treatment of cTBAD has not been achieved, particularly because of poor aortic reverse remodeling, defined as gradual thrombosis of the FL and enlargement of the true lumen (TL) without enlargement of the total aortic diameter. 11 Herein, this study aimed to explore the long‐term outcomes of TEVAR for cTBAD and the challenges during this procedure. Additionally, we also attempted to identify the potential risk factors for adverse aortic events (AAEs).
METHODS
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Study Design
Patients diagnosed with cTBAD who underwent TEVAR at our center from January 2009 to June 2017 were enrolled. Their baseline information, surgical procedures, morbidity, and mortality were collected and analyzed. This study was approved by the Ethics Committee of Zhongshan Hospital, Fudan University (No. B2019‐231R). The need for informed consent was waived because of the retrospective nature of this study.
Data Collection
Baseline information, including age, sex, history of cardiovascular surgery, and comorbidities, including diabetes, hypertension, coronary artery disease, and chronic kidney dysfunction, was collected. Morphological information included the diameters of the FL and TL at 2 levels (level A, the maximal aortic diameter; level B, the distal end of the stent graft) (Figure 1), and the extent of the FL thrombosis. TEVAR‐related information, including the interval from symptom onset to TEVAR, the number and types of stent grafts, and the location of the proximal landing zone, was also collected. During the follow‐up period, aortic remodeling at 2 levels, aorta‐related death, complications, including stent‐induced new entry, retrograde type A AD, 12 and endoleaks, were recorded and analyzed. Stent‐induced new entry was defined as a new tear caused by the stent graft, excluding those arising from natural disease progression or iatrogenic injury from endovascular manipulation. 13 The annual change in diameters of TL and FL was defined as the diameter difference of 2 adjacent years and measured at level A and level B.
Figure 1. Illustration of 2 levels of measurement.

Level A, the maximal aortic diameter; Level B, the distal end of the stent graft.
Surgical Procedures
The indications for TEVAR included: (1) the patient complained of recurrent or persistent chest or back pain as well as compression symptoms; (2) progressive thoracic aortic enlargement (>1 cm/year); (3) development of FL aneurysms (with total aortic diameter ≥5.5 cm); (4) malperfusion syndrome; and (5) aortic rupture or impending rupture. 3 , 14 , 15 The procedure of TEVAR was previously described. 16 The embolization of left subclavian artery (LSA) was not routinely performed in our center, and they were just covered partially or totally if the left vertebral artery was not dominant. Otherwise, in situ fenestration and branched stent grafts were performed. However, if the angle between the aortic arch and the branches was unsuitable for the above procedures, a carotid‐subclavian artery bypass was performed. 17 Carotid‐carotid bypass was performed if the proximal landing zone was expected to involve the ostia of the left carotid artery. If the proximal landing zone was expected to involve the left carotid artery and the left vertebral artery was dominant, then a right common carotid artery (RCCA)‐left common carotid artery (LCCA)‐LSA bypass was performed. The chimney technique was performed only in emergency cases. If the endoleak was identified during the follow‐up and the origin of the endoleak was confirmed to be the subclavian artery, then we would embolize it.
For patients with extensively compressed or totally occluded TL, stent grafts could be advanced with the assistance of balloon dilation or pulled forward with the assistance of a snare from the brachial artery approach. If the attempt failed, a transabdominal aortic implantation was considered. For patients with residual type A AD after open aortic repair, 18 advance of the stent grafts could be achieved using a step‐by‐step technique assisted by a balloon. During this process, the balloon was first dilated proximal to the stent graft, which was then advanced as soon as the balloon was deflated. This procedure was repeated until the stent graft reached the proximal landing zone (Figure S1). For patients with complication of visceral ischemia, a bare stent was implanted if ischemia was sustained after TEVAR. The detailed strategy was previously described. 19
We sealed all intimal entry tears above the celiac artery in one stage. A staged intervention was performed for the residual lesions to prevent paraplegia. Cerebrospinal fluid drainage was not routinely performed and was applied as soon as the patient presented with paraplegia.
Follow‐Up Strategy
Technical success was defined as closure of the primary entry tear without a type I endoleak and conversion to correct type Ia endoleak. All patients were followed up at 3, 6, and 12 months and yearly thereafter. Computed tomography angiography was performed to observe aortic remodeling at 2 levels. AAEs, including aorta‐related death, procedural complications (endoleak, stent‐induced new entry, retrograde type A AD, aortic intimal intussusception, dilation of the residual FL), and disease progression requiring reintervention, were recorded. Other end points included all‐cause mortality, aortic remodeling, and intervention. Clinical outcomes were described at the 5‐year follow‐up visit.
Statistical Analysis
Data were analyzed using GraphPad Prism (GraphPad Software Inc., La Jolla, CA). Continuous variables are presented as means and SDs or medians with interquartile ranges depending on the distribution of the data. Categorical variables are presented as frequencies and percentages. For univariate analysis, the Student t‐test and Pearson Chi‐squared test were applied to analyze the differences between the 2 groups (with and without AAEs) for continuous variables and categorical variables, respectively. In the multivariable logistic model, all variables with significant differences at the level of P<0.05 in the univariate analysis were included. For analysis purposes, FL thrombosis was dichotomized into total versus partial and no thrombosis, because the number of cases with no thrombosis was small. Kaplan–Meier analysis was used to calculate the cumulative survival rate and freedom from reintervention. The survival time was censored when the patient was lost to follow‐up. All P values are 2‐tailed, and P<0.05 is defined as statistically significant.
RESULTS
Baseline Information and Procedural Details
A total of 214 patients diagnosed with cTBAD were enrolled and analyzed. All patients were followed for at least 5 years. The mean age was 57.1±3.2 (range, 32–86 years). The indications for TEVAR included recurrent pain in 79 patients (36.9%), aortic diameter >5.5 cm in 52 (24.3%), aggressive progression (>1 cm/year) in 49 (22.9%), and acute symptom onset in 34 (5.9%). The median interval from onset to intervention was 12 months (interquartile range, 9–33.5). A hybrid procedure was performed in 22 patients (10.3%). LCCA‐LSA bypass was the predominant procedure in 5 (2.3%), followed by LSA fenestration and RCCA‐LCCA bypass in 4 (1.9%) patients. Residual type A AD was observed in 19 (8.9%) patients, 3 of whom underwent TEVAR using the balloon‐assisted technique. The details of the baseline information and procedures were shown in Table 1. The technical success rate was 97.7%. Two patients had multiple entries in the aorta, and the TL could not be identified. One patient had severe calcification in the entire descending aorta, the stent graft could not be delivered to the proximal landing zone, and the procedure failed. The other 2 patients died of aortic rupture after iliac artery‐superior mesenteric artery bypass and cervical hematoma after RCCA‐LCCA and left vertebral artery transposition.
Table 1.
Baseline Information and Procedural Details
| No. (%) or −X±SD | |
|---|---|
| Men | 147 (68.7%) |
| Age, y | 57.1±3.2 (32–86) |
| Comorbidities | |
| Hypertension | 150 (70.1%) |
| Diabetes | 27 (12.6%) |
| CAD | 17 (7.9%) |
| Cerebral infraction | 14 (6.5%) |
| CKD | 14 (6.5%) |
| TAAD | 19 (8.9%) |
| Indications for intervention | |
| Recurrent pain | 79 (36.9%) |
| Aortic diameter >5.5 cm | 52 (24.3%) |
| Rapid growth | 49 (22.9%) |
| Acute onset | 34 (5.9%) |
| Interval from onset to intervention (mo) | 12 (9, 33.5) |
| Coverage of LSA | |
| No | 156 (72.9%) |
| Partially | 25 (11.7%) |
| Totally | 33 (15.4%) |
| Mean length of stent graft, mm | 184.6±32.8 (80–300) |
| Proximal technique | |
| LCCA‐LSA bypass | 5 (2.3%) |
| LSA fenestration | 4 (1.9%) |
| RCCA‐LCCA bypass | 4 (1.9%) |
| RCCA and LCCA chimney | 1 (0.5%) |
| LCCA and LSA chimney | 1 (0.5%) |
| RCCA‐LCCA‐LSA bypass | 1 (0.5%) |
| LCCA chimney | 1 (0.5%) |
| LSA chimney | 1 (0.5%) |
| LSA branched stent graft | 1 (0.5%) |
| Axillary‐axillary bypass | 1 (0.5%) |
| RCCA‐LCCA bypass and LVA transposition | 1 (0.5%) |
| Hybrid procedure | 1 (0.5%) |
CAD indicates coronary artery disease; CKD, chronic kidney disease; LCCA, left common carotid artery; LSA, left subclavian artery; LVA, left vertebral artery; RCCA, right common carotid artery; and TAAD, type A aortic dissection.
Aortic Remodeling
The TL diameter was significantly improved from 16.3±8.4 mm at baseline to 30.1±12.3 mm at the 5‐year follow‐up (P<0.01), and the FL diameter was significantly decreased from 25.1±10.3 mm at baseline to 13.1±8.5 mm at the 5‐year follow‐up (P<0.01) at level A. The TL diameter was significantly improved from 14.6±8.0 mm at baseline to 26.2±10.2 mm at the 5‐year follow‐up (P<0.01), and the FL diameter was significantly decreased from 20.3±10.1 mm at baseline to 14.0±8.7 mm at the 5‐year follow‐up (P<0.01) at level B. The annual diameter changes of TL and FL were shown in Figure 2. Complete FL thrombosis along the stent graft was observed in 162 (75.7%) patients, and no FL thrombosis was observed in 5 (2.3%) patients.
Figure 2. The annual diameter changes of the true lumen and false lumen at 2 levels.
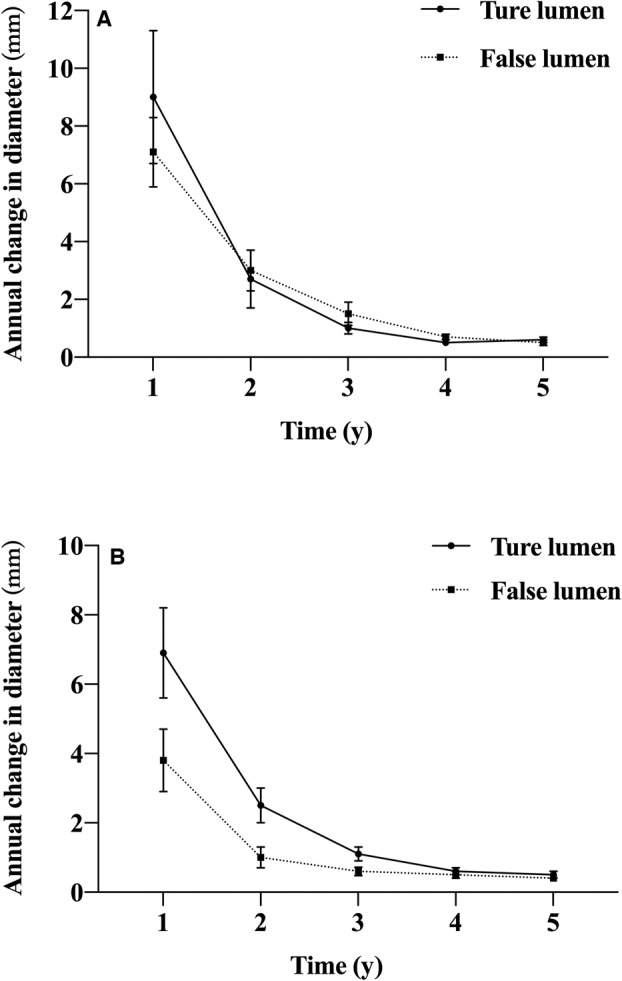
A, At level A, the diameter of true lumen increased by 9.0±2.3, 2.7±1.0, 1.0±0.2, 0.5±0.1, and 0.6±0.1 mm, and the diameter of false lumen decreased by 7.1±1.1, 3.0±0.7, 1.5±0.4, 0.7±0.1, and 0.5±0.1 mm at 1, 2, 3, 4, 5 years after endovascular repair. B, At level B, the diameter of true lumen increased by 6.9±1.3, 2.5±0.5, 1.1±0.2, 0.6±0.1, and 0.5±0.1 mm, and the diameter of false lumen decreased by 3.8±0.9, 1.0±0.3, 0.6±0.1, 0.5±0.1, and 0.4±0.1 mm at 1, 2, 3, 4, and 5 years after endovascular repair.
Mortality and Morbidity
The 30‐day morbidity rate was 5.1%. Femoral access occlusion occurred in 3 (1.4%) patients, and all of them underwent open repair. Two (0.9%) patients experienced atelectasis attributable to massive pleural effusion after TEVAR, and both recovered after ventilator therapy. Two patients (0.9%) died. One patient presented with malperfusion of the bowels and died of aortic rupture after iliac artery‐superior mesenteric artery bypass, and the other died of cervical hematoma after RCCA‐LCCA and left vertebral artery transposition. One patient developed spinal cord ischemia and recovered after cerebrospinal fluid drainage for 5 days. Stent‐graft–induced aortic intimal intussusception occurred in 1 patient (0.5%) and was detailed in our previous studies. 20
Reintervention was performed in 40 (18.7%) patients. Distal aneurysmal dilation was the most common indication in 20 (9.3%) patients. Other indications included type Ib endoleak in 6 patients (2.8%), distal stent‐induced new entry in 6 (2.8%) (Figure 3), femoral artery occlusion in 3 (1.4%), type II endoleak in 2 (0.9%), retrograde type A AD in 2 (0.9%), and type Ia in 1 (0.5%) patient (Figure 4). Two patients presented with endoleak type II from LSA and required embolization. One of them presented with endoleak type II 10 months after TEVAR, and he was complicated with endoleak type Ib, originating from the distal entry tear. A cuff stent was applied to seal the distal entry tear, and the LSA was embolized, and the patient had complete remodeling along the stent graft. The other patient presented with endoleak type II and type Ia at 18 months after TEVAR. He underwent re‐TEVAR and LSA embolization, but the patient died of aortic rupture at 22 months. The freedom from reintervention rate was 86.6% (95% CI, 81.2%–90.5%) at 5 years and 79.0% (95% CI, 71.7%–84.5%) at 10 years (Figure 5).
Figure 3. The occurrence of stent‐induced new entry.

The chronic type B aortic dissection was confirmed in an 82‐year‐old man (A), and he underwent endovascular repair by a stent graft (38*200 mm, Zenith). The false lumen was completely thrombosed at 5 months (B), as well as 12 months (C). However, the distal stent‐induced new entry was observed at 24 months (D), and a stent graft (38*200 mm. Zenith) was deployed to seal the distal stent‐induced new entry. The aortic remodeling was satisfactory at 6 months (E) and 24 months (F) after reintervention.
Figure 4. The occurrence of retrograde type A aortic dissection. A 74‐year‐old man complained of chest pain for 2 years.

The computed tomography show aortic dissection with entry tear in the distal to the left subclavian artery (A) and the false lumen was patent (B). A stent graft (36*200 mm, Valiant Captivia) was deployed. However, he suffered acute refractory chest pain at 26 days, and the computed tomography showed retrograde type A aortic dissection (C) and the false lumen was partially thrombosed (D). Hence, he underwent ascending aorta and aortic arch replacement and survived.
Figure 5. The cumulative survival and freedom from reintervention rates calculated by Kaplan–Meier analysis.

A, The 5‐year and 10‐year cumulative survival rates were 86.6% (95% CI, 81.2%–90.5%) and 79.0% (95% CI, 71.7%–84.5%). B, The 5‐year and 10‐year freedom from reintervention rates were 95.3% (95% CI, 91.5%–97.5%) and 89.9% (95% CI, 84.6%–93.5%).
During follow‐up, all‐cause mortality occurred in 20 (9.3%) patients. In 15 (7.0%) patients, the cause of mortality was aorta related. Among them, aortic rupture was the predominant cause in 10 (4.7%) patients, and 4 (1.9%) suffered from multi‐organ dysfunction after debranching procedures. The last patient (0.5%) suffered sudden death 1 day after TEVAR, and the exact cause of death remained unclear because of the lack of autopsy (Table S1). The cumulative 5‐year and 10‐year survival rates were 95.3% (95% CI, 91.5%–97.5%) and 89.9% (95% CI, 84.6%–93.5%) (Figure 5).
Univariable and Multivariable Analyses
The AAE rate was 21.5%. Compared with patients without AAEs, those with AAEs had higher rates of residual type A AD (26.1% versus 4.2%, P<0.001) and aortic diameter ≥5.5 cm (69.6% versus 11.3%, P<0.001), and a lower rate of complete FL thrombosis (23.9% versus 89.9%, P<0.001). The median interval from symptom onset to intervention was longer in patients with AAEs (26 months versus 12 months, P=0.004) (Table 2). In the multivariable analysis, partial or no FL thrombosis (adjusted odds ratio [AOR], 14.71 [95% CI, 5.67–38.14; P<0.001]) and aortic diameter ≥5.5 cm (AOR, 10.16 [95% CI, 3.86–26.73; P<0.001]) were identified as independent risk factors for AAEs (Table 3).
Table 2.
Comparison Between Patients With and Without AAEs
| AAEs | No AAEs | P value | |
|---|---|---|---|
| No. (%) or −X±SD | |||
| Sex | 0.615* | ||
| Men | 33 (71.7) | 114 (67.9) | |
| Women | 13 (28.3) | 54 (32.1) | |
| Age, y | 55.4±10.2 | 57.5±10.6 | 0.223‡ |
| Hypertension | 0.930* | ||
| Yes | 12 (26.1) | 7 (4.2) | |
| No | 34 (73.9) | 161 (95.8) | |
| Diabetes | 0.366* | ||
| Yes | 4 (8.7) | 23 (13.7) | |
| No | 42 (91.3) | 145 (86.3) | |
| Cerebral infraction | 0.999† | ||
| Yes | 3 (6.5) | 11 (6.5) | |
| No | 43 (93.5) | 157 (93.5) | |
| Coronary artery disease | 0.537† | ||
| Yes | 2 (4.3) | 15 (8.9) | |
| No | 44 (95.7) | 153 (91.1) | |
| Chronic kidney disease | 0.999† | ||
| Yes | 3 (6.5) | 11 (6.6) | |
| No | 43 (93.5) | 155 (93.4) | |
| TAAD | <0.001* | ||
| Yes | 12 (26.1) | 7 (4.2) | |
| No | 34 (73.9) | 161 (95.8) | |
| Symptom onset to intervention, mo | 26 (12, 51.8) | 12 (8, 30) | 0.004* |
| LSA sacrifice | 0.160* | ||
| No | 38 (82.6) | 118 (70.2) | |
| Partially | 2 (4.3) | 23 (13.7) | |
| Totally | 6 (13.0) | 27 (16.1) | |
| Mean length of stent grafts, mm | 187.2±35.8 | 183.9±32.0 | 0.545‡ |
| No. of stent grafts | 1.2±0.4 | 1.2±0.3 | 0.459* |
| Aortic diameter ≥5.5 cm | <0.001* | ||
| Yes | 32 (69.6) | 19 (11.3) | |
| No | 14 (30.4) | 149 (88.7) | |
| FL thrombosis along the stent graft | <0.001* | ||
| Totally | 11 (23.9) | 151 (89.9) | |
| Partially or patent | 35 (67.3) | 17 (10.1) | |
AAEs indicates adverse aortic events; FL, false lumen; LSA, left subclavian artery; and TAAD, type A aortic dissection.
Pearson Chi‐squared test.
Fisher exact test.
Student t‐test.
Table 3.
Multivariable Analysis of the Risk Factors for AAEs
| AOR | 95% CI | P value | |
|---|---|---|---|
| TAAD | 3.008 | 0.779–11.609 | 0.110 |
| Symptom onset to intervention | 1.006 | 0.977–1.007 | 0.309 |
| Partial or no FL thrombosis | 14.705 | 5.670–38.136 | <0.001 |
| Aortic diameter ≥5.5 cm | 10.159 | 3.861–26.733 | <0.001 |
AAEs indicates adverse aortic events; AOR, adjusted odds ratios; FL, false lumen; and TAAD, type A aortic dissection.
DISCUSSION
It is well understood that TEVAR can be performed with minimal morbidity for the acute setting of TBAD. However, little is known about its safety for the chronic setting of TBAD, as most patients have traditionally been treated with medical therapy. Although medical treatment is recommended for cTBAD, FL thrombosis after medical therapy was reported to be <4%. However, aneurysmal dilation, de novo dissection, and aortic rupture occurred in 20% to 50% of these patients. 21 Therefore, there has been an increasing interest in using TEVAR over medical treatment in patients with cTBAD, despite a lack of strong evidence demonstrating the superiority of TEVAR. A meta‐analysis of 567 patients treated with TEVAR showed a technical success rate of 89.9%, with an aneurysmal dilatation rate of 7.8%. 15 Similarly, early data also suggested that TEVAR was associated with acceptable short‐term outcomes. However, compared with open surgery, more reinterventions were required for TEVAR. The most important determinant of cTBAD prognosis after TEVAR was negative aortic remodeling, leading to adverse clinical outcomes. 22 In the present study, we enrolled 214 patients treated with TEVAR with a technical success rate of 97.7%. The 5‐year all‐cause mortality was 10.3%, and aorta‐related mortality rate was 7.0%, which were comparable with a previous study. 23
In terms of endovascular treatment, cTBAD has 2 distinguishing characteristics compared with acute AD that have a negative impact on clinical outcomes. First, cTBAD often has several mature fenestrations between the TL and FL. Second, the septum separating the TL from the FL in cTBAD is usually fibrotic and stiff. Because of both of these factors, promoting FL thrombosis in patients with cTBAD using a stent graft is difficult, because the radial force of the prosthesis cannot completely obliterate the FL of the dissection. Some investigators have noted that an inability to treat chronic dissections can be argued for early intervention. In this study, we found that the median interval of symptom onset was longer in patients with AAEs than in those without AAEs (P=0.004). Meanwhile, the septum became stiffer and thicker in patients with a longer history of AD. Hence, early endovascular intervention might be necessary to promote aortic reverse remodeling and avoid devastating events.
Occluded TL was identified as another challenge during the TEVAR. First, advancement of the stent graft was difficult in most patients with occluded TL, and auxiliary procedures, including 2.8% brachial artery access, 0.5% transabdominal aortic implantation, and 1.4% balloon dilatation assistance, were applied in the present study. Meanwhile, TL could not be distinguished in 2 (0.9%) patients, and the procedure failed. Of note, visceral arteries originating from the FL also made the procedure more challenging. There was a high risk of end‐organ ischemia when FL thrombosis occurred after extensive sealing of intimal entries. However, a patent FL was also responsible for the continuous dilation of the FL. Hence, initial TEVAR was performed to seal the primary entry tear, and close computed tomography angiography follow‐up was used to evaluate the remodeling of the distal aorta. We performed staged hybrid open endovascular repair to reconstruct the visceral arteries when the indications were met. 24 Three (1.4%) patients underwent this procedure when aneurysmal dilation was observed at 24, 60, and 68 months after TEVAR.
Because of the relatively long course of the disease, progression of the dissecting aortic aneurysm was observed in 52 (24.3%) patients. Because of this feature, additional procedures were necessary to acquire sufficient proximal landing zones. In our center, partial or total sacrifice of the LSA was sufficient for most patients with acute TBAD. However, we usually performed carotid‐carotid or carotid‐LSA bypass to avoid type Ia endoleak because of aneurysmal dilation in patients with cTBAD. Meanwhile, we identified aneurysmal dilation ≥5.5 cm as an independent predictor of AAE in this study. The results were consistent with those from Lee et al. 25 Lee et al demonstrated that good outcomes could be achieved in most patients with cTBAD treated by TEVAR and provided further insight into the complexity of this type of disorder. These authors concluded that there was no significant difference in outcomes between patients with long and short grafts, except that the patients having aneurysms with larger diameters required more frequent reinterventions. 26 , 27
Aortic reverse remodeling, including FL thrombosis, stabilization, and preferential TL flow, and reduction in the overall aortic diameter, varied among different studies. The investigation of stent‐grafts in aortic dissection trial demonstrated positive rates of aortic reverse remodeling with TEVAR in cTBAD. Leshnower et al also concluded that TEVAR in cTBAD stabilized dissecting aneurysm size and positively remodeled the descending thoracic aorta in 87% of patients. 27 A similar conclusion was also reported in another study. 28 However, outcomes were further stratified by the extent of the dissection. In this regard, patients with DeBakey IIIa achieved 100% FL thrombosis, while only 68% FL thrombosis was observed in those with DeBakey IIIb. 27 Meanwhile, it remains controversial whether implantation of the stent grafts alters the natural history of cTBAD, with specific regard to aneurysm progression in the visceral segment and the ultimate need for open aortic replacement or branched endograft within the perivisceral aorta. In this study, complete thrombosis along the stent grafts was observed in 162 patients (75.7%) (Figure S2). Partial or no FL thrombosis, identified as an independent predictor of AAE, was observed in 52 patients (24.3%). Thrombosis failure increased the risk of continuous dilation of the FL, leading to aortic rupture without aggressive reintervention. These results indicated that additional techniques to promote FL thrombosis might prevent devastating aortic events.
The presence of multiple entry tears was an outcome determinant. The optimal coverage of stent grafts remains controversial. To promote aortic remodeling, one study proposed implantation of longer stent graft. However, this approach increased the risk of paraplegia. 29 By contrast, Lee et al concluded that there was no significant difference in outcomes between patients with long and short grafts. 25 Additionally, Nienabe et al proposed the provisional extension to induce complete attachment (PETTICOAT) technique, which was characterized by the preservation of the spinal cord blood supply and the expansion of the distal FL. 30 However, whether this technique could promote aortic remodeling was controversial. 31 In our center, the initial TEVAR was performed to seal the primary entry tear, and the entry tears between the stent graft and the visceral artery were then dominant. A short‐cuff stent graft rather than one with a coverage of the whole thoracic descending aorta was implanted to seal these tears in ≥1 stages. The mean number of stent grafts used in the study was 1.2±0.3, and the mean length was 184.6±32.3 mm. Under this strategy, spinal cord ischemia occurred in only 1 (0.5%) patient, who fully recovered after cerebrospinal fluid drainage for 5 days.
This study has some limitations. First, this was a retrospective study that complied with its own nature. Second, we enrolled patients from January 2009 to June 2017; however, the devices used in endovascular therapy, the surgeon's experience, and perioperative care have improved dramatically in recent years, all of which might cause chronological bias. If the clinical outcomes of cTBAD treated with TEVAR are also improved, further exploration is needed. Meanwhile, some novel techniques, including “PETTICOAT,” simultaneous FL embolization, constrained bare stent, and some new devices, including taper stent graft and branched stent graft, have emerged. The long‐term effectiveness and safety of these innovations must also be demonstrated. Finally, while the chimney technique was applied in 4 (1.9%) patients in this cohort, it was only used for emergency cases. The sample size in this study was still too small, and the conclusions should be confirmed by large scale clinical trials. Despite these limitations, the present study enrolled 214 patients with a median follow‐up of 101.9±34.6 months, and we believe that our results might support clinical decision‐making and provide some insights for future research directions.
CONCLUSIONS
TEVAR treatment for cTBAD might have some challenges, including the difficulty of the stent graft passage because of the extremely compressed TL, the failure of complete aortic remodeling, and the controversy about the benefit of TEVAR in cTBAD. However, its long‐term outcomes might be promising, as this procedure was effective in preventing catastrophic aortic events with encouraging aortic remodeling and freedom from AAEs. Patients with AAEs showed higher rates of residual type A aortic dissection and aortic diameter ≥5.5 cm, a lower rate of complete FL thrombosis, and a longer median interval from symptom onset to intervention. Additional surveillance might be advisable in patients with failure of complete FL thrombosis and aortic diameter ≥5.5 cm, which were identified as predictors of AAEs.
Sources of Funding
This work was supported by the Project of Outstanding Academic Leaders of Shanghai Science and Technology Commission (19XD1401200), the National Nature Science Funds (81770508), the Clinical Excellent Doctor Training Project (DGF828008/001/002), and the Training Project for “Future Star” Doctor of Fudan University (2019).
Disclosures
None.
Supporting information
Table S1
Figures S1–S2
X. Jiang and Y. Liu contributed equally.
For Sources of Funding and Disclosures, see page 10.
Contributor Information
Weiguo Fu, Email: fu.weiguo@zs-hospital.sh.cn.
Zhihui Dong, Email: dzh926@126.com.
References
- 1. Hagan PG, Nienaber CA, Isselbacher EM, Bruckman D, Karavite DJ, Russman PL, Evangelista A, Fattori R, Suzuki T, Oh JK, et al. The international registry of acute aortic dissection (IRAD): new insights into an old disease. JAMA. 2000;283:897–903. doi: 10.1001/jama.283.7.897 [DOI] [PubMed] [Google Scholar]
- 2. Scott AJ, Bicknell CD. Contemporary management of acute type B dissection. Eur J Vasc Endovasc Surg. 2016;51:452–459. doi: 10.1016/j.ejvs.2015.10.026 [DOI] [PubMed] [Google Scholar]
- 3. Erbel R, Aboyans V, Boileau C, Bossone E, Bartolomeo RD, Eggebrecht H, Evangelista A, Falk V, Frank H, Gaemperli O, et al. 2014 ESC guidelines on the diagnosis and treatment of aortic diseases: document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The task force for the diagnosis and treatment of aortic diseases of the European Society of Cardiology (ESC). Eur Heart J. 2014;35:2873–2926. [DOI] [PubMed] [Google Scholar]
- 4. Winnerkvist A, Lockowandt U, Rasmussen E, Radegran K. A prospective study of medically treated acute type B aortic dissection. Eur J Vasc Endovasc Surg. 2006;32:349–355. [DOI] [PubMed] [Google Scholar]
- 5. Marui A, Mochizuki T, Mitsui N, Koyama T, Kimura F, Horibe M. Toward the best treatment for uncomplicated patients with type B acute aortic dissection: a consideration for sound surgical indication. Circulation. 1999;100:II275–II280. [DOI] [PubMed] [Google Scholar]
- 6. Davies RR, Goldstein LJ, Coady MA, Tittle SL, Rizzo JA, Kopf GS, Elefteriades JA. Yearly rupture or dissection rates for thoracic aortic aneurysms: simple prediction based on size. Ann Thorac Surg. 2002;73:17–27. doi: 10.1016/S0003-4975(01)03236-2 [DOI] [PubMed] [Google Scholar]
- 7. Fattori R, Cao P, De Rango P, Czerny M, Evangelista A, Nienaber C, Rousseau H, Schepens M. Interdisciplinary expert consensus document on management of type B aortic dissection. J Am Coll Cardiol. 2013;61:1661–1678. doi: 10.1016/j.jacc.2012.11.072 [DOI] [PubMed] [Google Scholar]
- 8. Appoo JJ, Bozinovski J, Chu MW, El‐Hamamsy I, Forbes TL, Moon M, Ouzounian M, Peterson MD, Tittley J, Boodhwani M, et al. Canadian cardiovascular society/Canadian Society of Cardiac Surgeons/Canadian Society for Vascular Surgery joint position statement on open and endovascular surgery for thoracic aortic disease. Can J Cardiol. 2016;32:703–713. doi: 10.1016/j.cjca.2015.12.037 [DOI] [PubMed] [Google Scholar]
- 9. Kang WC, Greenberg RK, Mastracci TM, Eagleton MJ, Hernandez AV, Pujara AC, Roselli EE. Endovascular repair of complicated chronic distal aortic dissections: intermediate outcomes and complications. J Thorac Cardiovasc Surg. 2011;142:1074–1083. doi: 10.1016/j.jtcvs.2011.03.008 [DOI] [PubMed] [Google Scholar]
- 10. Parsa CJ, Williams JB, Bhattacharya SD, Wolfe WG, Daneshmand MA, McCann RL, Hughes GC. Midterm results with thoracic endovascular aortic repair for chronic type B aortic dissection with associated aneurysm. J Thorac Cardiovasc Surg. 2011;141:322–327. doi: 10.1016/j.jtcvs.2010.10.043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Fanelli F, Cannavale A, O'Sullivan GJ, Gazzetti M, Cirelli C, Lucatelli P, Santoni M, Catalano C. Endovascular repair of acute and chronic aortic type B dissections: main factors affecting aortic remodeling and clinical outcome. JACC Cardiovasc Interv. 2016;9:183–191. doi: 10.1016/j.jcin.2015.10.027 [DOI] [PubMed] [Google Scholar]
- 12. Dong ZH, Fu WG, Wang YQ, Guo DQ, Xu X, Ji Y, Chen B, Jiang JH, Yang J, Shi ZY, et al. Retrograde type a aortic dissection after endovascular stent graft placement for treatment of type B dissection. Circulation. 2009;119:735–741. doi: 10.1161/CIRCULATIONAHA.107.759076 [DOI] [PubMed] [Google Scholar]
- 13. Dong Z, Fu W, Wang Y, Wang C, Yan Z, Guo D, Xu X, Chen B. Stent graft‐induced new entry after endovascular repair for Stanford type B aortic dissection. J Vasc Surg. 2010;52:1450–1457. doi: 10.1016/j.jvs.2010.05.121 [DOI] [PubMed] [Google Scholar]
- 14. Svensson LG, Kouchoukos NT, Miller DC, Bavaria JE, Coselli JS, Curi MA, Eggebrecht H, Elefteriades JA, Erbel R, Gleason TG, et al. Expert consensus document on the treatment of descending thoracic aortic disease using endovascular stent‐grafts. Ann Thorac Surg. 2008;85:S1–S41. [DOI] [PubMed] [Google Scholar]
- 15. Thrumurthy SG, Karthikesalingam A, Patterson BO, Holt PJ, Hinchliffe RJ, Loftus IM, Thompson MM. A systematic review of mid‐term outcomes of thoracic endovascular repair (TEVAR) of chronic type B aortic dissection. Eur J Vasc Endovasc Surg. 2011;42:632–647. doi: 10.1016/j.ejvs.2011.08.009 [DOI] [PubMed] [Google Scholar]
- 16. Fu WG, Shi Y, Wang YQ, Guo DQ, Xu X, Chen B, Jiang JH, Yang J, Shi ZY. Endovascular therapy for Stanford type B aortic dissection in 102 cases. Asian J Surg. 2005;28:271–276. doi: 10.1016/S1015-9584(09)60359-6 [DOI] [PubMed] [Google Scholar]
- 17. Shu X, Xu H, Wang E, Wang L, Guo D, Chen B, Fu W. Midterm outcomes of an adjustable puncture device for In situ fenestration during thoracic endovascular aortic repair. Eur J Vasc Endovasc Surg. 2022;63:43–51. doi: 10.1016/j.ejvs.2021.09.028 [DOI] [PubMed] [Google Scholar]
- 18. Sun L, Qi R, Zhu J, Liu Y, Zheng J. Total arch replacement combined with stented elephant trunk implantation: a new "standard" therapy for type a dissection involving repair of the aortic arch? Circulation. 2011;123:971–978. doi: 10.1161/CIRCULATIONAHA.110.015081 [DOI] [PubMed] [Google Scholar]
- 19. Liu Y, Jiang X, Chen B, Jiang J, Ma T, Dong Z, Fu W. Risk factors and treatment outcomes for type B aortic dissection with malperfusion requiring adjunctive procedures after thoracic endovascular aortic repair. J Vasc Surg. 2022;75:1192‐1200e2. DOI: 10.1016/j.jvs.2021.09.044. [DOI] [PubMed] [Google Scholar]
- 20. Ma T, Liu F, Chen B, Jiang JH, Shi Y, Guo DQ, Xu X, Dong ZH, Fu WG. Intraoperative stent‐graft‐induced aortic intimal intussusception during TEVAR for type B aortic dissection. J Endovasc Ther. 2021;28:860–870. doi: 10.1177/15266028211028213 [DOI] [PubMed] [Google Scholar]
- 21. Neuhauser B, Greiner A, Jaschke W, Chemelli A, Fraedrich G. Serious complications following endovascular thoracic aortic stent‐graft repair for type B dissection. Eur J Cardiothorac Surg. 2008;33:58–63. doi: 10.1016/j.ejcts.2007.10.010 [DOI] [PubMed] [Google Scholar]
- 22. Mani K, Clough RE, Lyons OT, Bell RE, Carrell TW, Zayed HA, Waltham M, Taylor PR. Predictors of outcome after endovascular repair for chronic type B dissection. Eur J Vasc Endovasc Surg. 2012;43:386–391. [DOI] [PubMed] [Google Scholar]
- 23. Nienaber CA, Kische S, Rousseau H, Eggebrecht H, Rehders TC, Kundt G, Glass A, Scheinert D, Czerny M, Kleinfeldt T, et al. Endovascular repair of type B aortic dissection: long‐term results of the randomized investigation of stent grafts in aortic dissection trial. Circ Cardiovasc Interv. 2013;6:407–416. doi: 10.1161/CIRCINTERVENTIONS.113.000463 [DOI] [PubMed] [Google Scholar]
- 24. Fang Y, Si Y, Yang J, Yue J, Chen B, Zhu T, Fu W. Viabahn open revascularization technique for renal artery revascularization reduces renal ischemia in thoracoabdominal aortic aneurysm hybrid open‐endovascular repair. Ann Vasc Surg. 2019;61:261–269. doi: 10.1016/j.avsg.2019.05.031 [DOI] [PubMed] [Google Scholar]
- 25. Lee M, Lee DY, Kim MD, Lee MS, Won JY, Park SI, Yoon YN, Lee S, Choi D, Ko YG. Outcomes of endovascular management for complicated chronic type B aortic dissection: effect of the extent of stent graft coverage and anatomic properties of aortic dissection. J Vasc Interv Radiol. 2013;24:1451–1460. doi: 10.1016/j.jvir.2013.06.007 [DOI] [PubMed] [Google Scholar]
- 26. Funaki B. Chronic complicated aortic dissection. J Vasc Interv Radiol. 2013;24:1460–1461. doi: 10.1016/j.jvir.2013.06.016 [DOI] [PubMed] [Google Scholar]
- 27. Leshnower BG, Szeto WY, Pochettino A, Desai ND, Moeller PJ, Nathan DP, ackson BM, Woo EY, Fairman RM, Bavaria JE. Thoracic endografting reduces morbidity and remodels the thoracic aorta in DeBakey III aneurysms. Ann Thorac Surg. 2013;95:914–921. doi: 10.1016/j.athoracsur.2012.09.053 [DOI] [PubMed] [Google Scholar]
- 28. Andacheh ID, Donayre C, Othman F, Walot I, Kopchok G, White R. Patient outcomes and thoracic aortic volume and morphologic changes following thoracic endovascular aortic repair in patients with complicated chronic type B aortic dissection. J Vasc Surg. 2012;56:644–650. doi: 10.1016/j.jvs.2012.02.050 [DOI] [PubMed] [Google Scholar]
- 29. Guangqi C, Xiaoxi L, Wei C, Songqi L, Chen Y, Zilun L, Shenming W. Endovascular repair of Stanford type B aortic dissection: early and mid‐term outcomes of 121 cases. Eur J Vasc Endovasc Surg. 2009;38:422–426. doi: 10.1016/j.ejvs.2009.04.015 [DOI] [PubMed] [Google Scholar]
- 30. Nienaber CA, Kische S, Zeller T, Rehders TC, Schneider H, Lorenzen B, Bünger C, Ince H. Provisional extension to induce complete attachment after stent‐graft placement in type B aortic dissection: the PETTICOAT concept. J Endovasc Ther. 2006;13:738–746. doi: 10.1583/06-1923.1 [DOI] [PubMed] [Google Scholar]
- 31. Bertoglio L, Rinaldi E, Melissano G, Chiesa R. The PETTICOAT concept for endovascular treatment of type B aortic dissection. J Cardiovasc Surg. 2019;60:91–99. doi: 10.23736/S0021-9509.17.09744-0 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1
Figures S1–S2


