Abstract
Alzheimer's disease (AD) which is associated with cognitive dysfunction and memory lapse has become a health concern. Various targets and pathways have been involved in AD's progress, such as deficit of acetylcholine (ACh), oxidative stress, inflammation, β-amyloid (Aβ) deposits, and biometal dyshomeostasis. Multiple pieces of evidence indicate that stress oxidative participation in an early stage of AD and the generated ROS could enable neurodegenerative disease leading to neuronal cell death. Hence, antioxidant therapies are applied in treating AD as a beneficial strategy. This review refers to the development and use of antioxidant compounds based on natural products, hybrid designs, and synthetic compounds. The results of using these antioxidant compounds were discussed with the given examples, and future directions for the development of antioxidants were evaluated.
1. Introduction
Alzheimer's disease (AD), a neurodegenerative disorder, is associated with progressive neural loss, dementia, and other symptoms. Recently, due to population aging, the prevalence of AD for people over 65 had an adverse socioeconomic impact [1]. In spite of efforts, the exact etiologic of AD is not yet characterized [2]. There is evidence showing that some hidden players such as deficit of acetylcholine (ACh) [3], tau-protein aggregation [4], oxidative stress inflammation [5, 6], β-amyloid (Aβ) deposits [7], and dyshomeostasis of biometals [8] are involved in the progress of AD [9]. Among these, the amyloid β (Aβ) peptide fibrillization and senile plaques play a role in AD's pathological hallmarks [10]. Abnormal processing of amyloid precursor protein (APP) via β- and γ-secretases results in Aβ peptides which undergo conformational changes to generate organized β-sheet amyloid structures [11, 12]. Other studies have shown that increased biological markers related to oxidative stress, such as oxidized biological macromolecules (lipids, proteins, DNA, and RNA) [13] and 8-hydroxyguanosine (8-OHG) [14], as well as decreased antioxidant enzyme activity [15], were observed in the brain. In age-related disorders, oxidative stress produces peroxides and free radicals that induce cell death.
Reactive oxygen species (ROS) such as superoxide anion, hydroxyl radical (OH•), and hydrogen peroxide radicals (H2O2•) are produced in redox reactions [16–19]. Overproduction of ROS caused by higher consumption of O2 in the brain is responsible for the cognitive dysfunction observed in AD. The lack of balance between the prooxidant and the antioxidant elements is associated with protein oxidation and accumulation of Aβ. Increased ROS production would be able to impact the function of synapses. To reduce the rate of progression, antioxidant therapy has been suggested because of the prominent role of oxidative stress in AD [7, 20].
Various studies have indicated that antioxidants like vitamins E and C can be prolonged the progression of dementia [19]. Also, the nature of AD inspired the researcher to the development of multitargeted-directed ligands (MTDLs) which provide a more effective treatment than existing drugs [20, 21]. In a study conducted by Rossi et al., the MTDLs were selected based on hepatic, neuronal, and microglial cell toxicity [22]. In this study, the MTDLs were selected based on known pharmacophores in the natural products, hybrid designs, and synthetic compounds. Furthermore, we will review the role of antioxidant agents and their outcomes achieved in the treatment of AD.
1.1. Natural Product-Based Antioxidant Agents in AD
Natural products have been used to treat and reduce the progression of AD [21]. Extensive use of natural products due to less toxicity and fewer side effects makes them a very popular treatment [23]. The studies have confirmed the advantages of natural products such as vitamins C, E, luteolin, melatonin, curcumin [24], quercetin, resveratrol [25], huperzine A, and rosmarinic acid in the treatment of AD [26, 27].
1.2. Phenolic-Based Natural Compounds in AD
Ferreres and coworkers have investigated the effects of ellagic acid and its derivatives against oxidative stress, AD, and depression. Since the phenolic compounds could be antioxidants and neuroprotective agents, the phenolic composition of aqueous and hydromethanolic extracts was determined by HPLC-DAD-ESI/MSn. The results illustrated that both extracts and ellagic acid had radical scavenging capacity compared to those of ascorbic acid. This indicated antidepressant activity while their anticholine esterase activities were weak [28] (Figure 1).
Figure 1.
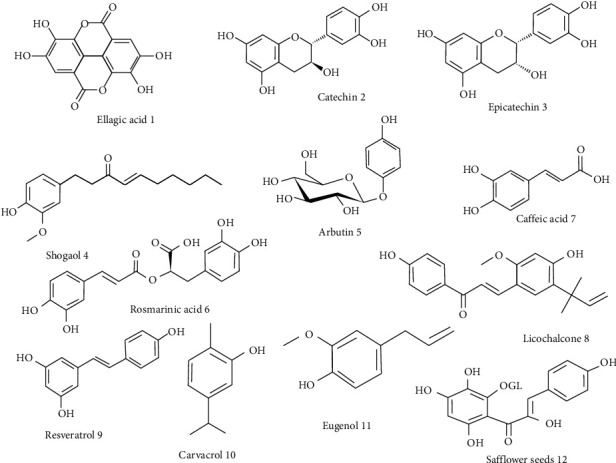
Natural compounds with phenolic structures.
Cimini et al. evaluated the impact of the polyphenolic extract of cocoa against human AD. Cocoa polyphenols exerted their activity via triggering neuroprotection by activating the BDNF survival pathway, which led to the counteraction of neurite dystrophy, both on Aß plaque and Aß oligomers treated cells. Based on these results, cocoa powder could be used as a preventive agent for neurodegeneration [29]. For the development of polyphenolic compounds, Oligopin containing flavan-3-L units such as catechin (C) and epicatechin (EC) in pine bark extract have been identified (Figure 1). This polyphenol compound exerts its activity via blocking oligomer formation of Aβ1–40 and Aβ1–42, as well as tau in vitro [30].
In a similar study, Na et al. assessed the effect of 6-shogaol, an active constituent of ginger, with antioxidant and anti-inflammatory activities for the treatment of AD (Figure 1). It is suggested that Aβ (1–42) upregulated the cysteinyl leukotriene 1 receptor (CysLT1R) and cathepsin B. Results showed that 6-shogaol inhibited the CysLT1R and downregulated cathepsin B in both in vitro and in vivo models. Moreover, it decreases Aβ deposition in the brain of APPSw/PS1-dE9 Tg mice. For better results, one year later, this team evaluated the effect of 6-shogaol on sortilin-related receptor 1 (SORL1). Both in vitro and in vivo results indicated that the activation of SORL1 by 6-shogaol downregulates BACE, sAPPb, and Aβ as the preventive agent for the treatment of AD [31, 32]. The protective effects of the phenolic compound arbutin (from Pyrus biossieriana) with anti-inflammatory and antioxidant effects of streptozotocin (STZ)-induced neurotoxicity in rats have been compared. It is indicated that arbutin improves spatial memory and decreases oxidative and nitrosative stress. Overall, the neuroprotective effect of arbutin is associated with its antioxidants and free radical scavenging effects [33].
The inhibition of cholinesterase and antioxidant properties of various extracts of Sideritis albiflora and Sideritis leptoclada, which included rosmarinic acid and caffeic acid, were analyzed (Figure 1). The high antioxidant activity for the acetone extract of S. leptoclada in different assays such as β‐carotene‐linoleic acid (IC50: 17.23 ± 0.11 μg/ml), DPPH• (IC50: 28.14 ± 0.05 μg/ml), and ABTS (IC50: 15.18 ± 0.02 μg/ml) assays, and the hexane extracts of sideritis species indicated moderate inhibitory activity on AChE and BChE [34]. Another multifunctional anti-Alzheimer agent is licochalcone B (LCB), as the main constituent of the root of Glycyrrhiza inflates (Figure 1). Cao et al. have evaluated the anti-AD activity of LCB through various tests. Based on the results, LCB could block amyloid-beta (Aβ42) self-aggregation (IC50 = 2.16 ± 0.24 μM), disaggregate preformed Aβ42 fibrils, and decrease metal-induced Aβ42 aggregation via chelating metal ions. Also, upon exposure to LCB, the ROS generation was inhibited dose-dependently in SH-SY5Y cells, and the observed antioxidant activity of LCB was more potent than Cur [35]. The evaluation of antioxidant and neuronal cell protective effects of different fractions of Erigeron annuus leaf, including caffeic acid 7, showed that the highest antioxidant activity was related to butanol fraction because of the highest total phenolic contents (396.49 mg of GAE/g) and neuroprotective effects on neuronal cells [36].
Resveratrol (RSV) was isolated from P. suffruticosa, a member of a family of polyphenolic compounds called stilbenes (Figure 1). RSV reduces oxidative stress and has DPPH-free radical scavenging, as well as β-secretase inhibitory activity. The neuroprotective effect of this compound has been confirmed by regulating cholinergic, antioxidant, and anti-inflammatory pathways [37]. To develop functional foods for regulating glucose homeostasis and neuroprotection, Videira and coworkers conducted a study. They observed that a polyphenol-rich is extracted from polyvinylpolypyrrolidone (PVPP) winery residue with strong antioxidant activity blocks α-glucosidase (Ki = 166.9 μg/mL) and aldose reductase (Ki = 127.5 μg/mL). PVPP-white wine extract is capable of blocking rat brain AChE, decreasing ROS generation, and preserving the cell redox stat [38].
Kumar and coworkers have been investigating the neuroprotective mechanisms of clove oil in intracerebroventricular (icv)-colchicine-induced cognitive dysfunction in rats. The main constituents of clove oil include the phenolic compound carvacrol and eugenol (Figure 1). After the administration of clove oil, an impaired cognitive performance in the Morris water maze (MWM) was observed that results in oxidative stress increased AChE level, and neuroinflammation and mitochondrial dysfunction. The main neuroprotective effect of clove oil may be partly because of its mitochondrial restoring and antioxidant properties along with a microglial inhibitory mechanism [39].
Cholinergic dysfunction and oxidative stress are involved in AD. Safflower seeds with phenolic structure have a reputation as an antioxidant agent that could improve cholinergic compounds like serotonin (Figure 1). Cho and coworkers investigated the effects of safflower seed extract on scopolamine-induced memory impairment in a mouse model. The results showed that safflower seed extract could improve memory function via inhibition of the AChE activity. In comparison, the safflower seed decreases ROS production and increases antioxidant enzyme levels, indicating the protective role of the safflower seed extract against oxidative stress [40].
1.3. Salvianolic Acid and Flavonoid Natural Compounds in AD
There is evidence that metal ions generate oxidative damage to neuronal cells. Cao et al. studied the effects of salvianolic acid A (Sal A) on the treatment of AD (Figure 2(a)). Obtained results showed that Sal A blocks amyloid-beta (Aβ), self-aggregation and disaggregates preformed Aβ fibrils, decreases metal-induced Aβ aggregation, and prevents the formation of ROS in SH-SY5Y cells [41]. Many Chinese herbs with antioxidant properties have been regarded as neuroprotective agents. Gazova and coworkers selected salvianolic acid B (Sal B), which is extracted from Salvia miltiorrhiza and anemarrhenae asphodeloides to evaluate their dissociation potential towards Aβ42 peptide fibrils and neuroprotective effect (Figure 2(b)). Most compounds could be able to dissociate Aβ42 fibrils. One of the sarsasapogenin derivatives showed a decreased level of nitride oxide production [42].
Figure 2.
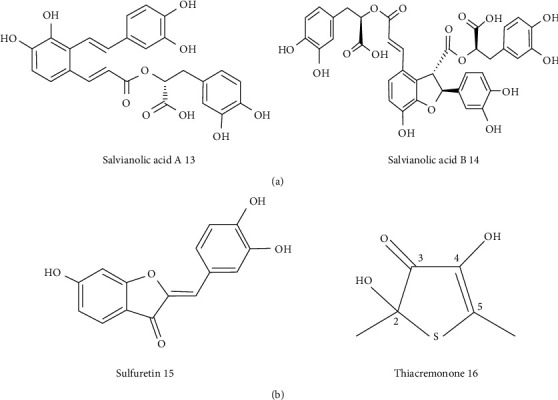
(a) Salvianolic acid in the treatment of AD. (b) Flavonoid compounds in AD.
The neuroprotective effects of sulfuretin 15 (Figure 2(b)), a member of flavonoid glycosides with antioxidant properties which are isolated from the stem bark of Albizzia julibrissin and heartwood of Rhus verniciflua, have been investigated by Kwon et al. They found that sulfuretin decreases the release of lactate dehydrogenase and accumulation of ROS associated with Aβ25–35-induced neurotoxicity in neuronal cells. It upregulates the expression of heme oxygenase-1 (HO-1) and protects neuronal cells from Aβ25–35-induced neurotoxicity via activation of Nrf/HO-1 and PI3K/Akt signaling pathways [43]. Various studies show that the accumulation of amyloid-beta (Aβ) peptide in AD is related to oxidative stress and inflammatory responses. Hence, Yun et al. investigated the neuroprotective effects of thiacremonone isolated from garlic, which has antioxidant and anti-inflammatory properties (Figure 2(b)). Thiacremonone could be able to improve memory by the stimulating antioxidant system when it upregulated the expression of peroxiredoxin 6 (PRDX6) for reducing oxidative stress of macromolecules such as protein and lipids. Exposure by thiacremonone, H2O2, and Aβ1–42 in embryonic neuronal cells inhibited the activation of NF-κB and ERK pathways [44].
1.4. Other Structures in Natural Compounds in AD
Hritcu and coworkers have assessed possible anxiolytic, antidepressant, and antioxidant properties of F. angulata essential oil, which is used for treating digestive pains, haemorrhoids, snake bites, and ulcers. In the scopolamine-induced rat model of Alzheimer's disease, the results showed anxiolytic, antioxidant, and antidepressant-like effects. It is proposed that F. angulate essential oil inhalation relieves scopolamine-induced anxiety and depression by the decrease of oxidative stress in the rat amygdala [45].
Today, using a multitarget-directed ligand (MTDL) strategy, the design of a new indole drug, synthesis of novel chromone+ donepezil hybrids, and development of carbazole scaffold based as potential anti-Alzheimer agents were conducted [46–49]. To develop a therapeutic strategy against AD by using an MTDL approach, the antiamyloidogenic, antioxidant, and neuroprotective properties were found in the Asparagus racemosus aqueous extract and its secondary metabolite sarsasapogenin (SRS) 17 (Figure 3). SRS dose-dependently inhibits AChE, BuChE, BACE1, and MAO-B as well as blocks Aβ42 fibrillization up to 68%. In addition, the SRS indicated a neuroprotective effect on PC12 cells against Aβ42 and H2O2-mediated cytotoxicity. Thus, SRS could be served as an MTDL compound against AD [50]. To relieve the inflammatory progression of AD, Lee and coworkers found tetramethylpyrazine (TMP) 18 (Figure 3).
Figure 3.
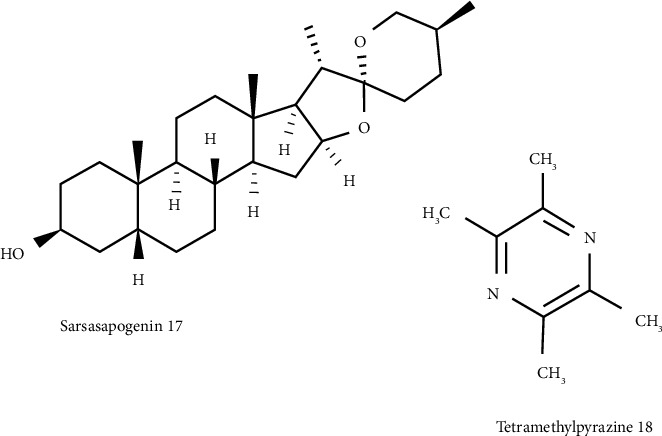
Other structures in natural compounds.
TMP is able to block the Aβ25–35 fibrillization and production of nitric oxide. This compound was an example of controlling Aβ-related neuropathology [51]. For stress-related disease prevention, herbs such as Ipomoea aquatica Forsk (HAEIA) were used to diminish oxidative stress in the brain. The results of Sivaraman and coworkers indicated that HAEIA significantly upregulates the expression of the enzymes, including superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPX), and glutathione reductase (GPR). HAEIA prevents the progression of DNA damage and as a potential antistress compound could be applied in AD [52].
1.5. Hybrid-Based Antioxidant Agents in AD
Since the nature of AD includes a complex cascade of molecular events in the brain, multifunctional drugs could be beneficial. Conjugation of the AChE inhibition and antioxidant properties in a molecular hybrid is known as an effective strategy.
2. Phenolic Acids-Based Hybrids
Šebestík et al. hybrid the naturally occurring phenolic acid compounds (derivatives of caffeic acid, rosmarinic acid, and trolox), with choline to account for the recognition by AChE. Most of the hybrids indicated AChE inhibitory activity and good antioxidant properties [53]. Curcumin which is extracted from plant rhizomes “Curcuma longa Linn” has anti-inflammatory and antioxidant activities and scavenges free radicals while protecting the brain from lipid peroxidation.
2.1. Curcumin-Based Hybrids
For the design of hybrid drugs, Elmegeed et al. combined the promising heterocyclic nucleus with the essential pharmacophoric features of the curcumin moiety. This hybrid compound decreases AchE activity, urinary 8-OHG level, serum caspase-3 level, and brain P53 level relative to the control group [54].
2.2. Donepezil-Based Hybrid
Using a hybrid strategy for the design of multifunctional anti-AD agents, Tripathi et al. introduced biphenyl-3-oxo-1,2,4-triazine-linked piperazine derivatives (see Figure 4(a)). They connected biaryl-1,2,4-triazines with intrinsic antioxidant property with the piperazine moiety as a bioisostere of the piperidine ring in donepezil, an FDA-approved drug against dementia, via the different lengths of alkyl linkers. The most active compounds in this series were able to inhibit AChE (IC50: 0.2 ± 0.01 μM) more effectively than standard donepezil (IC50: 0.1 ± 0.002 μM). Also based on ex vivo studies, AChE inhibition and reversal of the scopolamine-induced oxidative stress were observed [55].
Figure 4.
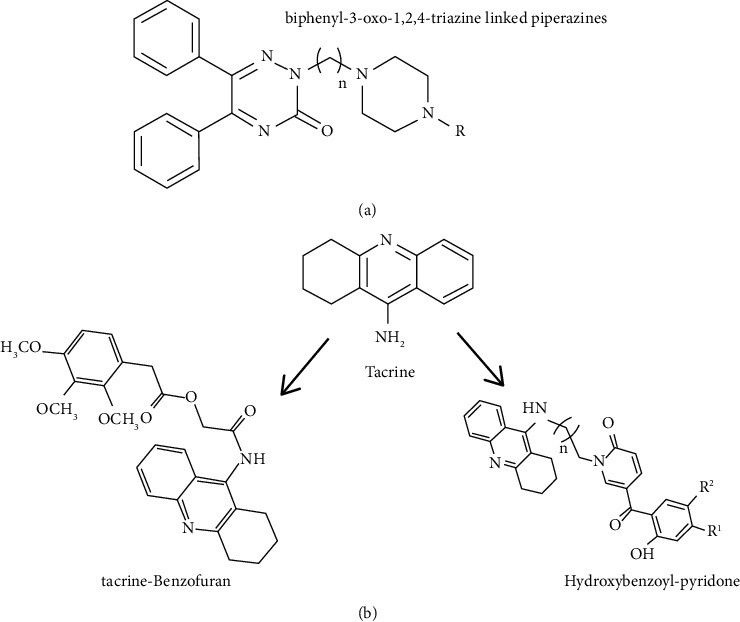
(a) Structure of biphenyl-3-oxo-1,2,4-triazine linked piperazine derivatives 19. (b) Structures of tacrine-based hybrids 20 and 21.
The novel tacrine-(hydroxybenzoyl-pyridone) (TAC-HBP) hybrids have been designed and synthesized by Santos and coworkers. All the hybrid compounds inhibit AChE in the submicromolar range (IC50 = 0.57–0.78 μM) compared to the parent tacrine, the well-known AChE inhibitor, and showed good DPPH radical scavenging capacity (EC50 = 204–249 μM) conferred by the hydroxybenzoyl-pyridone (HBP) moiety. The most active compound chelates of various biometals pFe = 13.9, pCu = 6.0, and pZn = 6.0, at pH 6.0, CL/CM = 10, and CM = 10−6 M [56]. Also, this group conjugated tacrine with benzofuran (BF) to develop a single agent which inhibits AChE and amyloid-beta peptide aggregation as well as chelate metals (Fe, Cu) and associated extra antioxidant activity. TAC-BF hybrids showed the submicromolar range of AChE activity while could able to inhibit self- and Cu-mediated Aβ aggregation (Figure 4(b)) [57].
Using tacrine, trolox, and β-carboline derivatives with versatile biological activities as promising leads for developing AD, the tacrine-trolox and tacrine-tryptoline hybrids with different linker chain lengths were designed and synthesized. These hybrids demonstrated moderate to high AChE inhibitory activity (IC50: 17.37–2200 nM) and BuChE inhibitory activity (IC50: 3.16–128.82 nM) as well as free radical scavenging activities (IC50: 11.48–49.23 μM). Docking experiments confirmed the binding of compounds to both the CAS and PAS of enzymes [58].
2.3. Synthetic Antioxidant Compounds in AD
Carbazole is naturally occurring and possesses an extensive range of biological activities used to develop anti-Alzheimer agents. The most potent multifunctional compound demonstrated effective and selective AChE inhibition, Aβ disaggregation, and metal chelation action. The radical scavenging assay of DPPH results showed 69.16 and 81.64% DPPH inhibition for the most active compounds [59]. Taj and coworkers also have been synthesized the benzamide derivatives which showed antioxidant and anti-Alzheimer activities. The most active compound in this series indicated the high inhibition of ACEs, as well as the highest scavenging percentage by DPPH assay [60].
Isoalloxazine derivatives are another class of compounds that have been evaluated for their neuroprotective potential by Machhi and coworkers. Based on AChE inhibitory activity, the most active compounds with good CNS penetration could improve cognitive ability in both scopolamine and Aβ1–42-induced Alzheimer's-like conditions in rodents. The antioxidant potential is confirmed by in vivo results. Additionally, these compounds could activate the Wnt/b-catenin pathway as evidenced by improved p-GSK-3, b-catenin, and neuroD1 levels in Aβ1–42-induced Alzheimer's rat brain [61]. The discovery of compounds with the ability to prevent the formation of free radicals in the brain is a beneficial strategy in the treatment of AD. Thus, synthesis and biological evaluation of 5-oxo-5H-furo[3,2-g]chromene-6-carbaldehydes derivatives against AChE and BChE has been reported by Mphahlele and coworkers. Various enzyme targets, such as β-secretase (BACE-1) and lipoxygenase-15 (LOX-15), as well as potential antioxidant are involved in AD. Evaluating the AChE and β-secretase inhibitory activity resulted in the most active compound in this series showing inhibitory effects, and it revealed modest antioxidant activity (IC50 = 7.4–23.9 μM) relative to the ascorbic acid (IC50 = 4.8 μM) [62]. Indole alkaloids are used as a vital pharmacological agent due to their biological effects. Yar and coworkers have assessed a series of selected synthetic indole derivatives to find new antioxidants via suppressing oxidative stress. DPPH scavenging assay for most of the compounds indicated significant to moderate antioxidant activity (IC50 value: 399.07–140.0 ± 50 μM) [63].
As another example of the application of indole alkaloids in the design of cholinesterase inhibitors, Kanhed et al. made an effort to develop a series of indoloquinoxaline derivatives as multitarget-directed ligands. The indoloquinoxaline scaffold has been used as a promising entity that has stable binding affinities with both the AChE and BuChE enzymes [2, 3]. The synthesized compounds exhibited modest to good AChE inhibitory activity. The most active compound in this series inhibits the self-induced Aβ1–42 aggregation and possesses antioxidant activity [64].
Accumulation of the amyloid-beta peptide (Aβ) in neuritic plaques is connected to inflammatory responses due to the production of nitrosative stress that stimulates the expression of inducible nitric oxide synthase (iNOS). Increased nitric oxide leads to neuronal death and memory impairment. Therefore, treatment of AD using anti-inflammatory and antioxidant agents reduces the risk of developing AD. Guevara and coworkers assessed the effects of systemic administration of aminoguanidine (AG) (100 mg/kg/day for 4 days), as an iNOS inhibitor with anti-inflammatory and antioxidant properties, on spatial memory when inflammatory responses induced by an injection of Aβ25–35 [100 μM] into the temporal cortex (TCx) of rats. AG treatment of the Aβ25–35-treated group improved spatial memory, which was linked with reductions in reactive gliosis, IL-1β, TNF-α, and nitrite levels, as well as in neurodegeneration in the TCx and hippocampus (Hp) [65].
The effect of pure DDN (2,3-dichloro-5,8-dihydroxy-1,4-naphthoquinone) on the inhibition of AChE and Aβ42 aggregation was assessed, and the inhibition of Aβ42 aggregation (more than 90% at 25 μM) and on AChE (IC50 = 14.5 μM) was determined [66]. Inspired by the widespread use of triazine derivatives, Sinha et al. screened the novel triazine derivatives for their cholinesterase inhibition, and the most active compounds were selected for biological evaluation. The results confirmed the anticholinesterase and antioxidant activities of choosing compounds and indicated that they exerted their activities through decreasing in Aβ1–42 burden and cytochrome c as well as cleaved caspase-3 levels in the brain. Immunoblot and immunofluorescence data showed that these compounds could be able to improve the ratio of pGSK3/GSK3 and β-catenin which suggested positive involvement of the novel triazine derivatives in the Wnt/β-catenin pathway [67]. To develop potential multitargeted therapeutics, one may refer to the synthesis of seleno-dihydropyrimidinones compounds. As illustrated in Figure 5, for the design of novel AChE inhibitors, Braga and coworkers combined biologically active dihydropyrimidinones (DHPMs) and organoselenium compounds which have a vital role in modulating oxidative stress in the brain. All of the synthesized compounds demonstrated AChE inhibitory activity compared to the standard drugs while displaying good antioxidant activity via various mechanisms of action [68].
Figure 5.
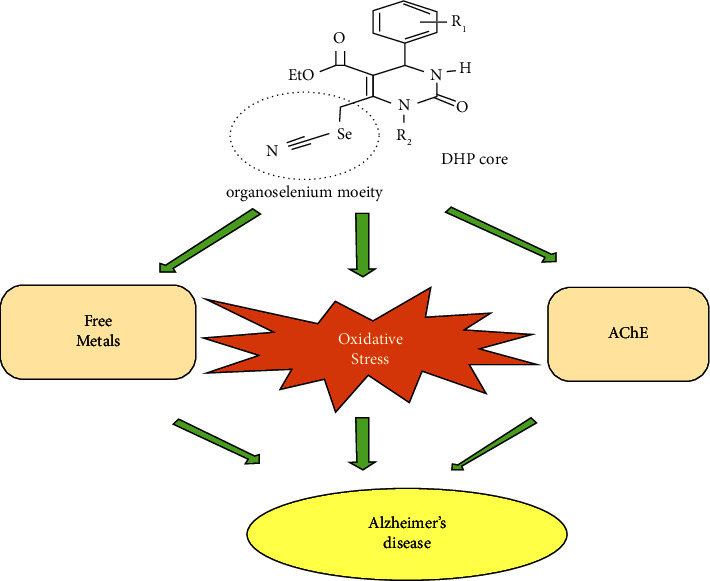
Structure of seleno-dihydropyrimidinones derivatives.
3. Analogues of Bioactive Compounds in AD
A potent antioxidant ferulic acid agent with antifibrillogenic and fibril-destabilizing effects against Aß was dimerized in order to increase its pharmacological efficiency against pathogenic Aβ oligomers. Results showed that one of the dimeric derivatives of ferulic acid, KMS4001, could decrease the Aβ1–40 and Aβ1–42 levels in the frontal cortex of APP/PS1 mutant transgenic mice [69]. Inspired by the important role of a bioactive compound Gx-50 for the treatment of AD, which is extracted from Sichuan pepper (Zanthoxylum bungeanum), various analogs like N-phenethyl cinnamide derivatives were designed and synthesized by Chai et al. Using a DPPH radical scavenging assay, antioxidant activities of synthesized compounds were evaluated. The most active compound could be able to inhibit the disaggregation of Cu2+-induced Aβ1–42 aggregation and indicate the potent antioxidative activity [70].
The irreversible fibril formation of various proteins, the main mechanism responsible for AD, has been introduced as an important target for developing anti-AD agents. Maity et al. reported a modified analog of curcumin, which possesses an antioxidant and antiamyloid activity aimed at improved stability. A pyrazole derivative of curcumin indicated notable potency in arresting the fibrillization of β-lactoglobulin (β-lg). Molecular docking revealed that such a result may be due to its interaction with aggregation-prone regions of the protein and prevention interactions between monomers, leading to suppression of the overall aggregation process [71].
Various studies have revealed that endogenous cannabinoid is implicated in the central nervous system and related diseases like AD. Valles and coworkers assessed the effect of WIN 55,212-2, a synthetic cannabinoid, on astrocytes function via the evaluation of different targets related to AD. This compound could be able to upregulate the expression of antioxidant enzyme Cu/Zn SOD and prevent inflammation induced by Aβ1-42 in cultured astrocytes through the increase of TNF-α, IL-1β, p-65, COX-2, and iNOS levels protein expression while reducing the expression of PPAR-γ. Hence, WIN 55,212-2 is an example of the beneficial effects of cannabinoids in the treatment of AD [72].
4. Other Synthetic Antioxidant Compounds in AD
Cholinesterase inhibitory, antioxidant, neuroprotective, and modulation of Aβ aggregation activities of flavone-8-acrylamide derivatives were evaluated by Amooru and coworkers. Modest antioxidant activity, strong neuroprotective capacities, accelerated Aβ aggregation, and selective inhibition of the AChE over BuChE was indicated by these novel multifunctional anti-Alzheimer agents [73]. Metal ions such as Cu, Fe, and Zn interacted with soluble and aggregated forms of Aβ peptide leading to the development of AD. Singh and coworkers synthesized the compound 2,6-bis[2-[(4-carboxyphenyl) methylene] hydrazide] as a novel Cu chelator and assessed its chelation ability in Cu-mediated neurotoxicity in the eye tissues of transgenic Drosophila expressing human amyloid β. Determination of antioxidant markers such as SOD and MDA showed a significant rescue [74].
Bukhari et al. synthesized a series of novel carbonyl compounds and evaluated their antioxidant activity and in vitro cytotoxicity. Most of the compounds were protective against Aβ-induced neuronal cell death in PC12 cells, and among them, the most active compound showed free radical scavenging activity (18.39 mM) and higher AChE inhibitory activity than donepezil [75]. Amyloid beta (Aβ) misfolding and aggregation are strongly implicated in AD. In pursuit of the development of aggregation modulators as effective therapeutic agents for AD, Ramesh et al. designed and synthesized a series of natural amino acids, L-dopa, and dopamine-appended derivatives of naphthalene diimide (NDI). Based on the results, L-dopa (NLD) and dopamine (NDP) conjugated NDIs could be able to modulate Aβ42-aggregation effectively and quench ROS [76].
Although a large number of compounds with antioxidant molecules have been synthesized, few have been successfully tested in clinical trials. This requires a better understanding of the mechanisms by which antioxidants work and where and when they are most effective. This understanding provides a rational approach that could lead to greater drug success in the future. However, the therapeutic use of synthetic molecules has largely been disappointing, largely due to inappropriate assumptions about how antioxidants work [77]. For example, scavenging the hydroxyl radical (•OH) is impractical, but preventing its formation by reducing the production of hydrogen peroxide (H2O2) can be effective in preventing damage. One of the misconceptions in this field is related to the removal of superoxide (O2•−) or H2O2 by small molecules [78]. This is because antioxidant enzymes react with those oxidants millions of times faster than small molecules, providing a dominant antioxidant defense [79]. The Mn porphyrin compounds have been synthesized, and some of them showed very high SOD activity [80]. Although whether the underlying mechanism is via SOD-like activity or another action remains elusive in some cases, the protective and therapeutic effects of many Mn porphyrins have been demonstrated in nonhuman animal models of diseases [81]. These preclinical results suggest the potential of Mn porphyrins in the clinical therapy of diseases [78]. Currently, a phase I clinical trial of MnTDE-2-ImP5+ in patients with amyotrophic lateral sclerosis showed no toxicity at therapeutic doses [82].
Although currently available treatments help manage the disease, they do not cure the disease [83, 84]. An important feature of Alzheimer's disease is oxidative stress. Despite evidence of the role of oxidative stress in the pathogenesis of Alzheimer's disease, none of the available treatment options have investigated oxidative stress [83]. Recently, important results of the effect of antioxidants in reducing the effects of oxidative stress on the central nervous system have been reported. Some antioxidants have proven positive effects on AD, but some still need attention and work [84]. Numerous studies and evidence show that oxidative damage is involved in the pathogenesis of AD through different pathways. Therefore, new therapeutic strategies for preventing oxidative damage may offer therapeutic efficacy against AD. Combining natural bioactivity with approved drugs could become an approach to prevent the onset of AD. An integrated system of antioxidants with several drugs may be more effective [84]. In addition, there is little information about the role of antioxidants in AD and the difference between the results of animal models and clinical trials. For this reason, there are doubts about the success of antioxidant therapy for AD. Investigating a more detailed approach to the connection between inflammation and AD and the integrated role of antioxidants in reducing inflammation should be considered [84]. Recently, the relationship between oxidative stress and the effect of natural antioxidants from natural sources and AD pathology has been discussed. This can lead to providing new therapeutic strategies.
It should be noted that the use of natural antioxidants has some limitations. Compared to synthetic antioxidants, natural antioxidants are mostly much more polar. Therefore, natural antioxidants (phenolic) are commonly not sufficiently soluble in the lipid phase [85]. This low solubility limits their efficiency in bulk lipids. Furthermore, in comparison to synthetic antioxidants with high purity (100%), natural antioxidants have a low degree of purity so a small part of the added composition is an antioxidant, and adding higher concentrations of natural antioxidants is necessary [85]. On the other hand, natural antioxidants are mainly less active and substrate specific. The antioxidant activity of natural antioxidants depends on synergists present both in the stabilized food and in the antioxidant preparation [86]. Other restrictions on the use of natural antioxidants are that these antioxidants possess certain toxicity so that too high concentrations of antioxidants are undesirable [87].
5. Conclusion
Alzheimer's disease (AD) is featured with cognitive dysfunction and memory lapse. Although the disease mechanism is yet to be properly characterized by various players such as deficit of acetylcholine (ACh), oxidative stress inflammation, β-amyloid (Aβ) deposits, and dyshomeostasis of biometals are involved in the progress of AD. Multiple pieces of evidence indicated that intracellular ROS with an important role in the pathogenesis of AD increased in brains' AD. ROS could be able to damage a wide range of critical biomolecules related to neurodegenerative disease and lead to neuronal cell death. The antioxidant compounds have been considered an effective therapeutic strategy. In this review, compounds with antioxidant activity that have played a role in AD are classified into three classes. Most of the investigated antioxidant reagents were obtained from natural products as one of the main classes of antioxidants. The majority of natural product-based antioxidants were phenolic compounds that could act as hydrogen donors or chelate metal ions and inhibit the oxidation of low-density lipoproteins (LDL) leading to a decrease in risks of neurodegenerative diseases. Hybrid compounds, like other classes of compounds, were designed in order to have antioxidant and anti-AD activities in a single entity. The last classes of antioxidant compounds related to AD were synthetic reagents with different structures. Considering these three classes of compounds, information is provided for further direction of anti-AD compound design. Most of the investigated compounds in the current paper target a variety of pathways and proteins involved in AD. This multitargeted feature provides useful benefits to tackle the intertwined pathogenesis of AD. Collectively, using antioxidant compounds in the treatment of AD due to their significant outcomes serves as a promising strategy.
Acknowledgments
The authors gratefully acknowledge the technical support provided for this project by the Graduate University of Advanced Technology, Kerman, Iran.
Data Availability
All data generated or analyzed during this study are included in this published article.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Sippl W., Contreras J.-M., Parrot I., Rival Y. M., Wermuth C. G. Structure-based 3D QSAR and design of novel acetylcholinesterase inhibitors. Journal of Computer-Aided Molecular Design . 2001;15(5):395–410. doi: 10.1023/a:1011150215288. [DOI] [PubMed] [Google Scholar]
- 2.Knopman D. S. Current treatment of mild cognitive impairment and Alzheimer’s disease. Current Neurology and Neuroscience Reports . 2006;6(5):365–371. doi: 10.1007/s11910-996-0016-9. [DOI] [PubMed] [Google Scholar]
- 3.Mesulam M.-M., Guillozet A., Shaw P., Levey A., Duysen E., Lockridge O. Acetylcholinesterase knockouts establish central cholinergic pathways and can use butyrylcholinesterase to hydrolyze acetylcholine. Neuroscience . 2002;110(4):627–639. doi: 10.1016/s0306-4522(01)00613-3. [DOI] [PubMed] [Google Scholar]
- 4.Castellani R. J., Plascencia-Villa G., Perry G. The amyloid cascade and Alzheimer’s disease therapeutics: theory versus observation. Laboratory Investigation . 2019;99(7):958–970. doi: 10.1038/s41374-019-0231-z. [DOI] [PubMed] [Google Scholar]
- 5.Gaggelli E., Kozlowski H., Valensin D., Valensin G. Copper homeostasis and neurodegenerative disorders (Alzheimer’s, prion, and Parkinson’s diseases and amyotrophic lateral sclerosis) Chemical Reviews . 2006;106(6):1995–2044. doi: 10.1021/cr040410w. [DOI] [PubMed] [Google Scholar]
- 6.Cheignon C., Tomas M., Bonnefont-Rousselot D., Faller P., Hureau C., Collin F. Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Biology . 2018;14:450–464. doi: 10.1016/j.redox.2017.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rajasekhar K., Chakrabarti M., Govindaraju T. Function and toxicity of amyloid beta and recent therapeutic interventions targeting amyloid beta in Alzheimer’s disease. Chemical Communications . 2015;51(70):13434–13450. doi: 10.1039/c5cc05264e. [DOI] [PubMed] [Google Scholar]
- 8.Peterson L. K., Fujinami R. S. Inflammation, demyelination, neurodegeneration and neuroprotection in the pathogenesis of multiple sclerosis. Journal of Neuroimmunology . 2007;184(1-2):37–44. doi: 10.1016/j.jneuroim.2006.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sanabria-Castro A., Alvarado-Echeverría I., Monge-Bonilla C. Molecular pathogenesis of Alzheimer’s disease: an update. Annals of Neurosciences . 2017;24(1):46–54. doi: 10.1159/000464422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.dos Santos Picanco L. C., Ozela P. F., de Fatima de Brito Brito M., et al. Alzheimer’s disease: a review from the pathophysiology to diagnosis, new perspectives for pharmacological treatment. Current Medicinal Chemistry . 2018;25(26):3141–3159. doi: 10.2174/0929867323666161213101126. [DOI] [PubMed] [Google Scholar]
- 11.Vassar R., Kovacs D. M., Yan R., Wong P. C. The β-secretase enzyme BACE in health and Alzheimer’s disease: regulation, cell biology, function, and therapeutic potential. Journal of Neuroscience . 2009;29(41):12787–12794. doi: 10.1523/jneurosci.3657-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Andrade S., Ramalho M. J., Loureiro J. A., Pereira M. C. Interaction of natural compounds with biomembrane models: a biophysical approach for the Alzheimer’s disease therapy. Colloids and Surfaces B: Biointerfaces . 2019;180:83–92. doi: 10.1016/j.colsurfb.2019.04.019. [DOI] [PubMed] [Google Scholar]
- 13.Huang X., Moir R. D., Tanzi R. E., Bush A. I., Rogers J. T. Redox‐active metals, oxidative stress, and Alzheimer’s disease pathology. Annals of the New York Academy of Sciences . 2004;1012(1):153–163. doi: 10.1196/annals.1306.012. [DOI] [PubMed] [Google Scholar]
- 14.Nunomura A., Perry G., Aliev G., et al. Oxidative damage is the earliest event in Alzheimer disease. Journal of Neuropathology & Experimental Neurology . 2001;60(8):759–767. doi: 10.1093/jnen/60.8.759. [DOI] [PubMed] [Google Scholar]
- 15.Marcus D. L., Thomas C., Rodriguez C., et al. Increased peroxidation and reduced antioxidant enzyme activity in Alzheimer’s disease. Experimental Neurology . 1998;150(1):40–44. doi: 10.1006/exnr.1997.6750. [DOI] [PubMed] [Google Scholar]
- 16.Bursal E., Köksal E., Gülçin İ., Bilsel G., Gören A. C. Antioxidant activity and polyphenol content of cherry stem (Cerasus avium L.) determined by LC–MS/MS. Food Research International . 2013;51(1):66–74. doi: 10.1016/j.foodres.2012.11.022. [DOI] [Google Scholar]
- 17.Gülcin I. Antioxidant activity of food constituents: an overview. Archives of Toxicology . 2012;86(3):345–391. doi: 10.1007/s00204-011-0774-2. [DOI] [PubMed] [Google Scholar]
- 18.LeBel C. P., Ischiropoulos H., Bondy S. C. Evaluation of the probe 2’, 7’-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chemical Research in Toxicology . 1992;5(2):227–231. doi: 10.1021/tx00026a012. [DOI] [PubMed] [Google Scholar]
- 19.Kulshreshtha A., Piplani P. Ameliorative effects of amide derivatives of 1, 3, 4-thiadiazoles on scopolamine induced cognitive dysfunction. European Journal of Medicinal Chemistry . 2016;122:557–573. doi: 10.1016/j.ejmech.2016.06.046. [DOI] [PubMed] [Google Scholar]
- 20.Bajda M., Guzior N., Ignasik M., Malawska B. Multi-target-directed ligands in Alzheimer’s disease treatment. Current Medicinal Chemistry . 2011;18(32):4949–4975. doi: 10.2174/092986711797535245. [DOI] [PubMed] [Google Scholar]
- 21.González J. F., Alcántara A. R., Doadrio A. L., Sánchez-Montero J. M. Developments with multi-target drugs for Alzheimer’s disease: an overview of the current discovery approaches. Expert Opinion on Drug Discovery . 2019;14(9):879–891. doi: 10.1080/17460441.2019.1623201. [DOI] [PubMed] [Google Scholar]
- 22.Rossi M., Freschi M., de Camargo Nascente L., et al. Sustainable drug discovery of multi-target-directed ligands for Alzheimer’s disease. Journal of Medicinal Chemistry . 2021;64(8):4972–4990. doi: 10.1021/acs.jmedchem.1c00048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kim Thu D., Vui D. T., Ngoc Huyen N. T., Duyen D. K., Thanh Tung B. The use of Huperzia species for the treatment of Alzheimer’s disease. Journal of Basic and Clinical Physiology and Pharmacology . 2019;31(3) doi: 10.1515/jbcpp-2019-0159. [DOI] [PubMed] [Google Scholar]
- 24.Lee W.-H., Loo C.-Y., Bebawy M., Luk F., Mason R. S., Rohanizadeh R. Curcumin and its derivatives: their application in neuropharmacology and neuroscience in the 21st century. Current Neuropharmacology . 2013;11(4):338–378. doi: 10.2174/1570159x11311040002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ahmed T., Javed S., Javed S., et al. Resveratrol and Alzheimer’s disease: mechanistic insights. Molecular Neurobiology . 2017;54(4):2622–2635. doi: 10.1007/s12035-016-9839-9. [DOI] [PubMed] [Google Scholar]
- 26.Laurent C., Eddarkaoui S., Derisbourg M., et al. Beneficial effects of caffeine in a transgenic model of Alzheimer’s disease-like tau pathology. Neurobiology of Aging . 2014;35(9):2079–2090. doi: 10.1016/j.neurobiolaging.2014.03.027. [DOI] [PubMed] [Google Scholar]
- 27.Bui T. T., Nguyen T. H. Natural product for the treatment of Alzheimer’s disease. Journal of Basic and Clinical Physiology and Pharmacology . 2017;28(5):413–423. doi: 10.1515/jbcpp-2016-0147. [DOI] [PubMed] [Google Scholar]
- 28.Ferreres F., Grosso C., Gil-Izquierdo A., Valentão P., Andrade P. B. Ellagic acid and derivatives fromCochlospermum angolensiswqAnti-depressant, anti-cholinesterase and anti-oxidant activities. Phytochemical Analysis . 2013;24(6):534–540. doi: 10.1002/pca.2429. [DOI] [PubMed] [Google Scholar]
- 29.Cimini A., Gentile R., D’Angelo B., et al. Cocoa powder triggers neuroprotective and preventive effects in a human Alzheimer’s disease model by modulating BDNF signaling pathway. Journal of Cellular Biochemistry . 2013;114(10):2209–2220. doi: 10.1002/jcb.24548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ono K., Zhao D., Wu Q., et al. Pine bark polyphenolic extract attenuates amyloid-β and tau misfolding in a model system of Alzheimer’s disease neuropathology. Journal of Alzheimer’s Disease . 2020;15:1–10. doi: 10.3233/JAD-209007. [DOI] [PubMed] [Google Scholar]
- 31.Na J. Y., Song K., Lee J. W., Kim S., Kwon J. 6-Shogaol has anti-amyloidogenic activity and ameliorates Alzheimer’s disease via CysLT1R-mediated inhibition of cathepsin B. Biochemical and Biophysical Research Communications . 2016;477(1):96–102. doi: 10.1016/j.bbrc.2016.06.026. [DOI] [PubMed] [Google Scholar]
- 32.Na J.-Y., Song K., Lee J.-W., Kim S., Kwon J. Sortilin-related receptor 1 interacts with amyloid precursor protein and is activated by 6-shogaol, leading to inhibition of the amyloidogenic pathway. Biochemical and Biophysical Research Communications . 2017;484(4):890–895. doi: 10.1016/j.bbrc.2017.02.029. [DOI] [PubMed] [Google Scholar]
- 33.Dastan Z., Pouramir M., Ghasemi-Kasman M., et al. Arbutin reduces cognitive deficit and oxidative stress in animal model of Alzheimer’s disease. International Journal of Neuroscience . 2019;129(11):1145–1153. doi: 10.1080/00207454.2019.1638376. [DOI] [PubMed] [Google Scholar]
- 34.Deveci E., Tel‐Çayan G., Duru M. E., Öztürk M. Phytochemical contents, antioxidant effects, and inhibitory activities of key enzymes associated with Alzheimer’s disease, ulcer, and skin disorders of Sideritis albiflora and Sideritis leptoclada. Journal of Food Biochemistry . 2019;43(12) doi: 10.1111/jfbc.13078.e13078 [DOI] [PubMed] [Google Scholar]
- 35.Cao Y., Xu W., Huang Y., Zeng X. Licochalcone B, a chalcone derivative from Glycyrrhiza inflata, as a multifunctional agent for the treatment of Alzheimer’s disease. Natural Product Research . 2020;34(5):736–739. doi: 10.1080/14786419.2018.1496429. [DOI] [PubMed] [Google Scholar]
- 36.Jeong C.-H., Jeong H. R., Choi G. N., Kim D.-O., Lee U., Heo H. J. Neuroprotective and anti-oxidant effects of caffeic acid isolated from Erigeron annuus leaf. Chinese Medicine . 2011;6(1):25–29. doi: 10.1186/1749-8546-6-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Liu S., Li Y., Yi F., et al. Resveratrol oligomers from Paeonia suffruticosa protect mice against cognitive dysfunction by regulating cholinergic, antioxidant and anti-inflammatory pathways. Journal of Ethnopharmacology . 2020;260 doi: 10.1016/j.jep.2020.112983.112983 [DOI] [PubMed] [Google Scholar]
- 38.Rocha L., Neves D., Valentão P., Andrade P. B., Videira R. A. Adding value to polyvinylpolypyrrolidone winery residue: a resource of polyphenols with neuroprotective effects and ability to modulate type 2 diabetes-relevant enzymes. Food Chemistry . 2020;329 doi: 10.1016/j.foodchem.2020.127168.127168 [DOI] [PubMed] [Google Scholar]
- 39.Kumar A., Aggrawal A., Pottabathini R., Singh A. Possible neuroprotective mechanisms of clove oil against icv-colchicine induced cognitive dysfunction. Pharmacological Reports . 2016;68(4):764–772. doi: 10.1016/j.pharep.2016.03.005. [DOI] [PubMed] [Google Scholar]
- 40.Kim J. H., He M. T., Kim M. J., et al. Safflower (Carthamus tinctorius L.) seed attenuates memory impairment induced by scopolamine in mice via regulation of cholinergic dysfunction and oxidative stress. Food & Function . 2019;10(6):3650–3659. doi: 10.1039/c9fo00615j. [DOI] [PubMed] [Google Scholar]
- 41.Cao Y. Y., Wang L., Ge H., et al. Salvianolic acid A, a polyphenolic derivative from Salvia miltiorrhiza bunge, as a multifunctional agent for the treatment of Alzheimer’s disease. Molecular Diversity . 2013;17(3):515–524. doi: 10.1007/s11030-013-9452-z. [DOI] [PubMed] [Google Scholar]
- 42.Bednarikova Z., Gancar M., Wang R., et al. Extracts from Chinese herbs with anti-amyloid and neuroprotective activities. International Journal of Biological Macromolecules . 2021;179:475–484. doi: 10.1016/j.ijbiomac.2021.03.013. [DOI] [PubMed] [Google Scholar]
- 43.Kwon S. H., Ma S. X., Hwang J. Y., Lee S. Y., Jang C. G. Involvement of the Nrf2/HO-1 signaling pathway in sulfuretin-induced protection against amyloid beta25-35 neurotoxicity. Neuroscience . 2015;304:14–28. doi: 10.1016/j.neuroscience.2015.07.030. [DOI] [PubMed] [Google Scholar]
- 44.Yun H.-M., Jin P., Park K.-R., et al. Thiacremonone potentiates anti-oxidant effects to improve memory dysfunction in an APP/PS1 transgenic mice model. Molecular Neurobiology . 2016;53(4):2409–2420. doi: 10.1007/s12035-015-9208-0. [DOI] [PubMed] [Google Scholar]
- 45.Bagci E., Aydin E., Mihasan M., Maniu C., Hritcu L. Anxiolytic and antidepressant‐like effects of Ferulago angulata essential oil in the scopolamine rat model of Alzheimer’s disease. Flavour and Fragrance Journal . 2016;31(1):70–80. doi: 10.1002/ffj.3289. [DOI] [Google Scholar]
- 46.Lamie P. F., Abdel-Fattah M. M., Philoppes J. N. J. J. o.E. I., Chemistry M. Design and synthesis of new indole drug candidates to treat Alzheimer’s disease and targeting neuro-inflammation using a multi-target-directed ligand (MTDL) strategy. Journal of Enzyme Inhibition and Medicinal Chemistry . 2022;37(1):2660–2678. doi: 10.1080/14756366.2022.2126464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kumar N., Gupta P., Bansal S. Progress and development of carbazole scaffold based as potential anti-alzheimer agents using MTDL approach. Letters in Drug Design and Discovery . 2022;19(12):1049–1067. doi: 10.2174/1570180819666220314144219. [DOI] [Google Scholar]
- 48.Malek R., Refouvelet B., Benchekroun M., et al. Synthesis and biological evaluation of novel chromone+ donepezil hybrids for Alzheimer’s disease therapy. Current Alzheimer Research . 2019;16(9):815–820. doi: 10.2174/1567205016666191011112624. [DOI] [PubMed] [Google Scholar]
- 49.Mathew B. J. C. C., Screening H. T. Privileged pharmacophore of FDA approved drugs in combination with chalcone framework: a new hope for Alzheimer’s treatment. Combinatorial Chemistry & High Throughput Screening . 2020;23(9):842–846. doi: 10.2174/1386207323999200728122627. [DOI] [PubMed] [Google Scholar]
- 50.Kashyap P., Muthusamy K., Niranjan M., Trikha S., Kumar S. Sarsasapogenin: a steroidal saponin from Asparagus racemosus as multi target directed ligand in Alzheimer’s disease. Steroids . 2020;153 doi: 10.1016/j.steroids.2019.108529.108529 [DOI] [PubMed] [Google Scholar]
- 51.Kim M., Kim S. O., Lee M., et al. Tetramethylpyrazine, a natural alkaloid, attenuates pro-inflammatory mediators induced by amyloid β and interferon-γ in rat brain microglia. European Journal of Pharmacology . 2014;740:504–511. doi: 10.1016/j.ejphar.2014.06.037. [DOI] [PubMed] [Google Scholar]
- 52.Sivaraman D., Panneerselvam P., Muralidharan P. Brain free radical quenching capacity of Ipomoea aquatica by rejuvenating vital anti-oxidant enzymes on beta-amyloid induced stress in swiss albino mice. Der Pharmacia Lettre . 2015;7(4):01–12. [Google Scholar]
- 53.Sebestík J., Marques S. M., Falé P. L., et al. Bifunctional phenolic-choline conjugates as anti-oxidants and acetylcholinesterase inhibitors. Journal of Enzyme Inhibition and Medicinal Chemistry . 2011;26(4):485–497. doi: 10.3109/14756366.2010.529806. [DOI] [PubMed] [Google Scholar]
- 54.Elmegeed G. A., Ahmed H. H., Hashash M. A., Abd-Elhalim M. M., El-kady D. S. Synthesis of novel steroidal curcumin derivatives as anti-Alzheimer’s disease candidates: evidences-based on in vivo study. Steroids . 2015;101:78–89. doi: 10.1016/j.steroids.2015.06.003. [DOI] [PubMed] [Google Scholar]
- 55.Tripathi P. N., Srivastava P., Sharma P., et al. Biphenyl-3-oxo-1,2,4-triazine linked piperazine derivatives as potential cholinesterase inhibitors with anti-oxidant property to improve the learning and memory. Bioorganic Chemistry . 2019;85:82–96. doi: 10.1016/j.bioorg.2018.12.017. [DOI] [PubMed] [Google Scholar]
- 56.Chand K., Alsoghier H. M., Chaves S., Santos M. A. Tacrine-(hydroxybenzoyl-pyridone) hybrids as potential multifunctional anti-Alzheimer’s agents: AChE inhibition, antioxidant activity and metal chelating capacity. Journal of Inorganic Biochemistry . 2016;163:266–277. doi: 10.1016/j.jinorgbio.2016.05.005. [DOI] [PubMed] [Google Scholar]
- 57.Fancellu G., Chand K., Tomás D., et al. Novel tacrine-benzofuran hybrids as potential multi-target drug candidates for the treatment of Alzheimer’s Disease. Journal of Enzyme Inhibition and Medicinal Chemistry . 2020;35(1):211–226. doi: 10.1080/14756366.2019.1689237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Teponnou G. A. K., Joubert J., Malan S. F. Tacrine, trolox and tryptoline as lead compounds for the design and synthesis of multi-target agents for Alzheimer’s disease therapy. The Open Medicinal Chemistry Journal . 2017;11(1):24–37. doi: 10.2174/1874104501711010024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Mishra C. B., Gusain S., Shalini S., et al. Development of novel carbazole derivatives with effective multifunctional action against Alzheimer’s diseases: design, synthesis, in silico, in vitro and in vivo investigation. Bioorganic Chemistry . 2020;95 doi: 10.1016/j.bioorg.2019.103524.103524 [DOI] [PubMed] [Google Scholar]
- 60.Taj M. B., Tirmizi S. A., Raheel A., Ali H. B. M., Qureshi S., Alshatir H. Benzamide derivatives as potential candidates for anti-alzheimer, anti-fatigue, anti-urease and anti-oxidant activity. Journal of the Chilean Chemical Society . 2017;62(1):3342–3344. doi: 10.4067/s0717-97072017000100007. [DOI] [Google Scholar]
- 61.Machhi J., Sinha A., Patel P., et al. Neuroprotective potential of novel multi-targeted isoalloxazine derivatives in rodent models of Alzheimer’s disease through activation of canonical wnt/β-catenin signalling pathway. Neurotoxicity Research . 2016;29(4):495–513. doi: 10.1007/s12640-016-9598-4. [DOI] [PubMed] [Google Scholar]
- 62.Mphahlele M. J., Gildenhuys S., Agbo E. N. In vitro evaluation and docking studies of 5-oxo-5H-furo[3,2-g]chromene-6-carbaldehyde derivatives as potential anti-alzheimer’s agents. International Journal of Molecular Sciences . 2019;20(21):p. 5451. doi: 10.3390/ijms20215451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yar M., Arshad M., Farooq A., et al. Synthesis and DPPH scavenging assay of reserpine analogues, computational studies and in silico docking studies in AChE and BChE responsible for Alzheimer’s disease. Brazilian Journal of Pharmaceutical Sciences . 2015;51(1):53–61. doi: 10.1590/s1984-82502015000100006. [DOI] [Google Scholar]
- 64.Kanhed A. M., Patel D. V., Patel N. R., et al. Indoloquinoxaline derivatives as promising multi-functional anti-Alzheimer agents. Journal of Biomolecular Structure and Dynamics . 2020;23:1–18. doi: 10.1080/07391102.2020.1840441. [DOI] [PubMed] [Google Scholar]
- 65.Díaz A., Rojas K., Espinosa B., et al. Aminoguanidine treatment ameliorates inflammatory responses and memory impairment induced by amyloid-beta 25-35 injection in rats. Neuropeptides . 2014;48(3):153–159. doi: 10.1016/j.npep.2014.03.002. [DOI] [PubMed] [Google Scholar]
- 66.Khelifi I., Tourrette A., Dhouafli Z., et al. The antioxidant 2, 3‐dichloro, 5, 8‐dihydroxy, 1, 4‐naphthoquinone inhibits acetyl‐cholinesterase activity and amyloid β42 aggregation: a dual target therapeutic candidate compound for the treatment of Alzheimer’s disease. Biotechnology and Applied Biochemistry . 2020;67 doi: 10.1002/bab.1870. [DOI] [PubMed] [Google Scholar]
- 67.Sinha A., Tamboli R. S., Seth B., et al. Neuroprotective role of novel triazine derivatives by activating Wnt/β catenin signaling pathway in rodent models of Alzheimer’s disease. Molecular Neurobiology . 2015;52(1):638–652. doi: 10.1007/s12035-014-8899-y. [DOI] [PubMed] [Google Scholar]
- 68.Canto R. F. S., Barbosa F. A. R., Nascimento V., de Oliveira A. S., Brighente I. M. C., Braga A. L. Design, synthesis and evaluation of seleno-dihydropyrimidinones as potential multi-targeted therapeutics for Alzheimer’s disease. Organic and Biomolecular Chemistry . 2014;12(21):3470–3477. doi: 10.1039/c4ob00598h. [DOI] [PubMed] [Google Scholar]
- 69.Jung J.-S., Yan J.-J., Li H.-M., et al. Protective effects of a dimeric derivative of ferulic acid in animal models of Alzheimer’s disease. European Journal of Pharmacology . 2016;782:30–34. doi: 10.1016/j.ejphar.2016.04.047. [DOI] [PubMed] [Google Scholar]
- 70.Chai T., Zhao X. B., Wang W. F., Qiang Y., Zhang X. Y., Yang J. L. Design, synthesis of N-phenethyl Cinnamide derivatives and their biological activities for the treatment of Alzheimer’s disease: antioxidant, beta-amyloid disaggregating and rescue effects on memory loss. Molecules . 2018;23(10):p. 2663. doi: 10.3390/molecules23102663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Maity S., Pal S., Sardar S., et al. Inhibition of amyloid fibril formation of β-lactoglobulin by natural and synthetic curcuminoids. New Journal of Chemistry . 2018;42(23):19260–19271. doi: 10.1039/c8nj03194k. [DOI] [Google Scholar]
- 72.Aguirre-Rueda D., Guerra-Ojeda S., Aldasoro M., et al. WIN 55,212-2, agonist of cannabinoid receptors, prevents amyloid β1-42 effects on astrocytes in primary culture. PLoS One . 2015;10(4) doi: 10.1371/journal.pone.0122843.e0122843 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Shaik J. B., Yeggoni D. P., Kandrakonda Y. R., et al. Synthesis and biological evaluation of flavone-8-acrylamide derivatives as potential multi-target-directed anti Alzheimer agents and investigation of binding mechanism with acetylcholinesterase. Bioorganic Chemistry . 2019;88 doi: 10.1016/j.bioorg.2019.102960.102960 [DOI] [PubMed] [Google Scholar]
- 74.Singh S. K., Sinha P., Mishra L., Srikrishna S. Neuroprotective role of a novel copper chelator against induced neurotoxicity. International Journal of Alzheimer’s Disease . 2013;2013:9. doi: 10.1155/2013/567128.567128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bukhari S. N. A., Jantan I., Masand V. H., et al. Synthesis of α, β-unsaturated carbonyl based compounds as acetylcholinesterase and butyrylcholinesterase inhibitors: characterization, molecular modeling, QSAR studies and effect against amyloid β-induced cytotoxicity. European Journal of Medicinal Chemistry . 2014;83:355–365. doi: 10.1016/j.ejmech.2014.06.034. [DOI] [PubMed] [Google Scholar]
- 76.Ramesh M., Makam P., Voshavar C., et al. l-Dopa and dopamine conjugated naphthalenediimides modulate amyloid β toxicity. Organic and Biomolecular Chemistry . 2018;16(41):7682–7692. doi: 10.1039/c8ob01691g. [DOI] [PubMed] [Google Scholar]
- 77.Forman H. J., Davies K. J., Ursini F. J. F. R. B., Medicine How do nutritional antioxidants really work: nucleophilic tone and para-hormesis versus free radical scavenging in vivo. Free Radical Biology and Medicine . 2014;66:24–35. doi: 10.1016/j.freeradbiomed.2013.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Forman H. J., Zhang H. J. N. R. D. D. Targeting oxidative stress in disease: promise and limitations of antioxidant therapy. Nature Reviews Drug Discovery . 2021;20(9):689–709. doi: 10.1038/s41573-021-00233-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sies H. Strategies of antioxidant defense. European Journal of Biochemistry . 1993;215(2):213–219. doi: 10.1111/j.1432-1033.1993.tb18025.x. [DOI] [PubMed] [Google Scholar]
- 80.Batinic-Haberle I., Tovmasyan A., Spasojevic I. J. R. b. An educational overview of the chemistry, biochemistry and therapeutic aspects of Mn porphyrins–from superoxide dismutation to H2O2-driven pathways. Redox Biology . 2015;5:43–65. doi: 10.1016/j.redox.2015.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rabbani Z. N., Spasojevic I., Zhang X., et al. Antiangiogenic action of redox-modulating Mn (III) meso-tetrakis (N-ethylpyridinium-2-yl) porphyrin, MnTE-2-PyP5+, via suppression of oxidative stress in a mouse model of breast tumor. Free Radical Biology and Medicine . 2009;47(7):992–1004. doi: 10.1016/j.freeradbiomed.2009.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Benatar M. J. N. Lost in translation: treatment trials in the SOD1 mouse and in human ALS. Neurobiology of Disease . 2007;26(1):1–13. doi: 10.1016/j.nbd.2006.12.015. [DOI] [PubMed] [Google Scholar]
- 83.Pritam P., Deka R., Bhardwaj A., et al. Antioxidants in Alzheimer’s disease: current therapeutic significance and future prospects. Biology . 2022;11(2):p. 212. doi: 10.3390/biology11020212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Collins A. E., Saleh T. M., Kalisch B. E. J. A. Naturally occurring antioxidant therapy in Alzheimer’s disease. Antioxidants . 2022;11(2):p. 213. doi: 10.3390/antiox11020213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Pokorný J. J. E. Are natural antioxidants better–and safer–than synthetic antioxidants? European Journal of Lipid Science and Technology . 2007;109(6):629–642. doi: 10.1002/ejlt.200700064. [DOI] [Google Scholar]
- 86.Evans R. J. N. Health effects and applications Optimizing lipid stability with natural inhibitors . Vol. 45. Urbana, Illinois: AOCS Press; 1997. [Google Scholar]
- 87.Butler G. P. Protein-polyphenol interactions: nutritional aspects proanthocyanidin, tannin . France: Groupe Polyphenols; 1992. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.


