Abstract
Purpose:
This article describes a case of didanosine (DDI)-associated retinal toxicity in a patient with a heterozygous pathogenic variant in the CRB1 gene.
Methods:
Case report.
Results:
A middle-aged patient with HIV controlled on HAART therapy, and a remote 10-year year history of treatment with DDI and tenofivir, presented with external ophthalmoplegia and well-circumscribed, midperipheral patterns of bilateral pigmentary retinopathy and chorioretinal atrophy in both eyes. Genetic testing revealed a heterozygous pathogenic variant in the CRB1 gene that encodes a protein (Crumbs homolog 1) involved in regulation of cell polarity and junctions and is localized adjacent to mitochondria in the ellipsoid and myoid area.
Conclusions:
This case highlights a potential role for genetic susceptibility to retinal toxicity in DDI-associated retinal toxicity. Large, prospective pharmacogenomics studies may be informative to further elucidate the role of genetic risk factors in drug-induced retinal toxicity.
Keywords: CRB1, didanosine, toxicity
Introduction
Characteristic features of didanosine (DDI)-associated retinal toxicity include a well-circumscribed midperipheral pattern of bilateral pigmentary retinopathy and chorioretinal atrophy. 1 -3 To our knowledge, no report of genetic risk factors in DDI-associated retinal toxicity has been previously reported. We describe a case of DDI-associated toxicity with ophthalmoplegia and an accompanying pathogenic mutation in the CRB1 gene, potentially causing genetic susceptibility in this patient.
Methods
Case Report
A middle-aged patient with a 35-year history of HIV infection and type 2 diabetes, both well-controlled on medication, was referred for management of cystoid macular edema (CME) after cataract surgery in the left eye. The patient had been treated with DDI of 250 mg daily for 10 years, and the therapy was discontinued 5 years before presentation. During the same period, the patient was also treated with tenofovir of 300 mg daily. HIV viral loads were most recently undetectable. Ocular history was significant for progressive nyctalopia over 10 years and esotropia with bilateral external ophthalmoplegia managed with prism glasses. Extraocular motility examination was significant for esotropia with restricted infraduction, supraduction, and abduction in both eyes. Best-corrected visual acuity was 20/20 OD and 20/30 OS and tensions were normal. The anterior segment was significant for cataract in the right eye and pseudophakia in the left.
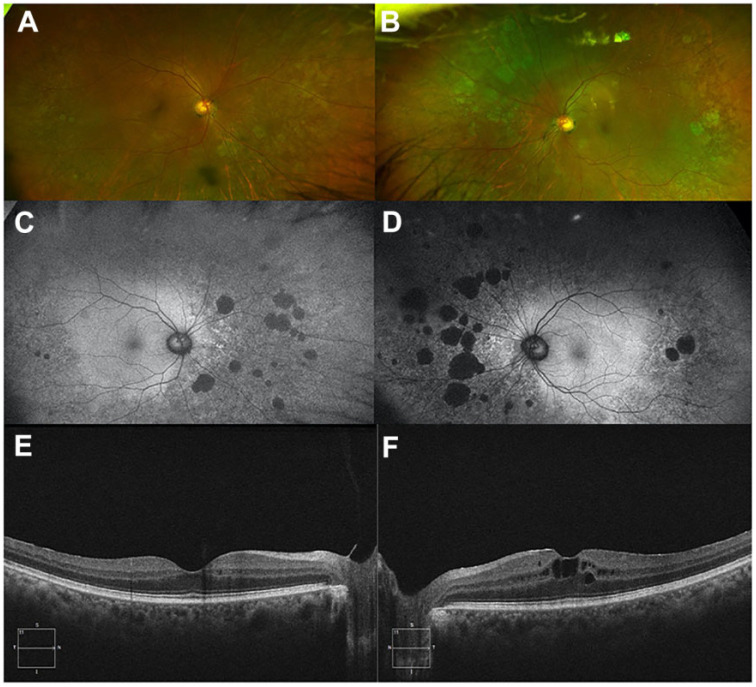
Dilated fundus examination was remarkable for symmetric pigmentary changes at the level of the retinal pigment epithelium and areas of concentric chorioretinal atrophy in the midperiphery with macula sparing in each eye. These findings were confirmed with widefield color fundus photography and fundus autofluorescence. Optical coherence tomography displayed grade 1 epiretinal membranes in both eyes and CME in the left eye (Figure 1).
Figure 1.
Widefield color fundus photography of the (A) right and (B) left eye shows characteristic midperipheral and peripheral chorioretinal degeneration in a well-circumscribed pattern. Widefield fundus autofluorescence of the (C) right and (D) left eye shows corresponding patches of hypoautofluorescent atrophy interspersed in a diffuse pattern of mottled autofluorescence. Optical coherence tomography images of the (E) right and (F) left eye shows grade 1 epiretinal membranes in both eyes and (F) cystoid macular edema in the left eye.
Results
To exclude an inherited retinal degeneration, genetic testing was performed. Mutational analysis revealed a heterozygous pathogenic variant in the CRB1 gene (c.1438T > C, p.Cyst480Arg).
Conclusions
Patients with DDI-associated retinal toxicity present with findings similar to mitochondrial disorders such as chronic progressive external ophthalmoplegia, Kearns-Sayre syndrome, maternally inherited diabetes and deafness, and mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes. DDI is a nucleoside reverse transcriptase inhibitor that inhibits mitochondrial DNA polymerase-γ.
There are several potential mechanisms that may be contributing factors in the development of our patient’s pigmentary retinopathy. First and most important, this patient presented with a long-standing history of DDI exposure, and the retinal features were classic for toxicity. 1 -3 Further evidence of mitochondrial impairment due to DDI toxicity was the presence of chronic progressive external ophthalmoplegia. Second, it has been postulated that tenofovir, the patient’s other medication, may compound retinal toxicity from DDI owing to further depletion of mitochondrial function. 4
Interestingly, a third pathogenic mechanism for this patient may be the detected pathogenic mutation in CRB1, which encodes the CRB1 protein involved in regulating cell polarity and cell junctions in Müller glial cells and photoreceptor inner segments (IS). 5 Mutations in CRB1 have been implicated in retinitis pigmentosa and Leber congenital amaurosis. More specifically, CRB1 is expressed in Müller glial cells as well as the IS of rods and cones. Electron microscopy and immunohistochemistry studies of photoreceptor IS have shown CRB1 to be localized in abundance adjacent to mitochondria in the ellipsoid and myoid areas. 5
It is plausible that certain genetic risk factors can make an individual susceptible to DDI-associated retinal toxicity. Given the limitations of genetic testing, it is possible that this patient may harbor a second, yet unidentified mutation. Despite the recent pseudophakic status of the left eye, the slow response of the patient’s CME over 1 year to topical prednisolone, nepafenac, and dorzolamide may suggest the CME is associated with a retinal degeneration in this patient with a heterozygous CRB1 pathogenic mutation. None of the DNA from patients previously reported to have DDI retina toxicity was genotyped, and thus it remains unknown whether DDI causes retinal toxicity in only genetically susceptible individuals. A large, prospective pharmacogenomics study may be informative to further elucidate the role of genetic risk factors in DDI-associated retinal toxicity.
Footnotes
Ethical Approval: This study adhered to the tenets of the Declaration of Helsinki. No institutional review board is required for case reports. No identifiable information is disclosed in this case report.
Statement of Informed Consent: Informed consent was obtained from the patient for publication of this case report and accompanying images.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
- 1. Gabrielian A, MacCumber MM, Kukuyev A, Mitsuyasu R, Holland GN, Sarraf D. Didanosine-associated retinal toxicity in adults infected with human immunodeficiency virus. JAMA Ophthalmol. 2013;131(2):255–259. doi:10.1001/jamaophthalmol.2013.579 [DOI] [PubMed] [Google Scholar]
- 2. Whitcup SM, Butler KM, Caruso R, et al. Retinal toxicity in human immunodeficiency virus-infected children treated with 2′,3′-dideoxyinosine. Am J Ophthalmol. 1992;113(1):1–7. doi:10.1016/s0002-9394(14)75744-7 [DOI] [PubMed] [Google Scholar]
- 3. Cobo J, Ruiz MF, Figueroa MS, et al. Retinal toxicity associated with didanosine in HIV-infected adults. AIDS. 1996;10(11):1297–1300. doi:10.1097/00002030-199609000-00022 [DOI] [PubMed] [Google Scholar]
- 4. Kearney BP, Sayre JR, Flaherty JF, Chen SS, Kaul S, Cheng AK. Drug-drug and drug-food interactions between tenofovir disoproxil fumarate and didanosine. J Clin Pharmacol. 2005;45(12):1360–1367. doi:10.1177/0091270005281351 [DOI] [PubMed] [Google Scholar]
- 5. Quinn PM, Pellissier LP, Wijnhold J. The CRB1 complex: following the trail of crumbs to a feasible gene therapy strategy. Front Neurosci. 2017;11:175. doi:10.3389/fnins.2017.00175 [DOI] [PMC free article] [PubMed] [Google Scholar]