Abstract
Background
Malaria is a major public health issue with substantial risks among vulnerable populations. Currently, the World Health Organization (WHO) recommends SP-IPTp in the second and third trimesters. However, the efficacy of SP-IPTp is threatened by the emergence of sulfadoxine-pyrimethamine resistant malaria parasites due to single nucleotide polymorphisms in the Plasmodium falciparum dihydrofolate reductase and dihydropteroate synthetase genes. This study aimed to assess the current prevalence of Pfdhfr/Pfdhps mutations in P. falciparum isolates collected from individuals residing in Ile-Ife, Nigeria, and also present maps of the prevalence of Pfdhps 431V and 581G within Nigeria and surrounding countries.
Methods
Between October 2020 and April 2021, samples were collected as dried blood spots among 188 participants who showed malaria positivity with a histidine-rich-protein-based rapid diagnostic test (RDT). Nested PCR assays were used to confirm falciparum in the samples with RDT positivity, and to amplify fragments of the Pfdhfr/Pfdhps genes followed by targeted amplicon sequencing. Published data since 2007 on the prevalence of the Pfdhps genotypes in Nigeria and the neighbouring countries were used to produce maps to show the distribution of the mutant genotypes.
Results
Only 74 and 61 samples were successfully amplified for the Pfdhfr and Pfdhps genes, respectively. At codons resulting in N51I, C59R, and S108N, Pfdhfr carried mutant alleles of 97.3% (72/74), 97.3% (72/74) and 98.6% (73/74), respectively. The Pfdhps gene carried mutations at codons resulting in amino acid changes at 431–436-437–540-581–613; I431V [45.9%, (28/61)], A581G [31.1% (19/61)] and A613S [49.2% (30/61)]. Constructed haplotypes were mainly the triple Pfdhfr mutant 51I-59R-108N (95.9%), and the most common haplotypes observed for the Pfdhps gene were the ISGKAA (32.8%), ISGKGS (8.2%), VAGKAA (14.8%), VAGKAS (9.8%) and VAGKGS (14.8%). In the context of the previously published data, a high prevalence of 431V/581G mutations was found in the study population. It seems quite evident that the Pfdhps 431V, 581G and 613S often co-occur as Pfdhps-VAGKGS haplotype.
Conclusion
This study showed that the prevalence of VAGKGS haplotype seems to be increasing in prevalence. If this is similar in effect to the emergence of 581G in East Africa, the efficacy of SP-IPTp in the presence of these novel Pfdhps mutants should be re-assessed.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12936-023-04487-5.
Keywords: SP resistance, Molecular marker, Dihydropteroate, Dihydrofolate, Malaria, Pregnancy
Background
The threat of malaria continues to pose a major public health issue across the globe, specifically in sub-Saharan African countries, and especially among pregnant women and children under the age of five. In 2021 alone, the World Health Organization (WHO) reported that an estimated 247 million cases of malaria occurred worldwide, while the estimated number of deaths from malaria stood at 619,000 [1]. According to the report, approximately 27% of global malaria morbidity and 31.9% of mortality are attributed to Nigeria [1]. Currently, the WHO recommends chemopreventive strategies, including the use of sulfadoxine-pyrimethamine (SP) as intermittent preventive treatment during pregnancy (SP-IPTp), the use of SP (or other anti-malarials) as perennial malaria chemoprevention (PMC) and seasonal malaria chemoprevention (SMC) with amodiaquine and SP [2].
The SP-IPTp strategy, administered at fixed time points, at four-week intervals, during antenatal visits, is currently implemented in 33 sub-Saharan African countries [1]. It has been documented to ameliorate malarial illness and its associated risks both in maternal and fetal health outcomes in regions with medium to high malaria transmission [3]. The strategy is also considered one of the cost-effective malaria control tools available [4]. The effectiveness of SP-IPTp is measured by using metrics such as reduction in the incidence of maternal malaria infection, reduction in low birth weight (LBW), decrease in preterm delivery cases, and improved haematological parameters [4]. In Nigeria, the provision of SP-IPTp is recommended as part of antenatal care of all pregnant women [5]. The dosing schedule is typically set from the second trimester, and at least three doses are given before delivery, each at least one month apart.
The combination of sulfadoxine and pyrimethamine works synergistically, and preferentially, to target dihydropteroate synthetase (PfDHPS) and dihydrofolate reductase (PfDHFR), both enzymes involved in the folate biosynthesis pathway. Molecular mechanisms underpinning SP resistance are caused by single nucleotide polymorphisms (SNPs) in two distinct genes that codes for PfDHPS and PfDHFR, that is Pfdhfr and Pfdhps genes. Polymorphisms in codons of Pfdhps causing amino acid substitutions in PfDHPS at S/A436F, A437G, K540E, and A613S have been found to be associated with sulfadoxine resistance while SNPs in Pfdhfr causing amino acid changes at position 51, 59, 108 and 164 of PfDHFR confer pyrimethamine resistance [6, 7]. Pfdhfr triple mutant alleles resulting in changes N51I, C59R, S108N (IRN) and in Pfdhps gene, the A437G mutation is prevalent in West and Central African countries [8, 9]. In contrast, a quintuple mutant that combines the Pfdhfr triple (IRN) haplotype and the Pfdhps double mutant at A437G and K540E is highly prevalent in East Africa [6] while the Pfdhps K540E mutation is rarely found in West and Central Africa [10].
However, the narrative in East Africa has escalated in the last decade with the emergence of a SNP in Pfdhps causing an alanine to glycine mutation at position 581 of PfDHPS [4, 11]. This results in the ‘super resistant’ sextuple haplotype of Pfdhfr/Pfdhps (mainly the constructed haplotype: CIRNI-SGEGA). There is evidence that the CIRNI-SGEGA haplotype signals a loss of sensitivity of Plasmodium falciparum to SP and compromise the effectiveness of the SP-IPTp strategy including a reduction in its ability to prevent low birthweights [5, 6, 12, 13]. In contrast, in West Africa, the absence of 540 and 581 genotypes was reassuring for the continuation of SP-IPTp efficacy. However, recent reports have shown that the molecular repertoire of P. falciparum in West Africa may be changing. For instance, a SNP resulting in an isoleucine to valine change (Pfdhps I431V) has emerged in Nigeria and other parts of West Africa [14]. The mutant was first reported in the UK from a malaria infection imported from Nigeria [15] and it also appeared in other surveys conducted in Nigeria, and subsequently in Cameroon [16–18]. More recently, polymorphisms in Pfdhps resulting in amino acid substitutions at 431, 581 and 613 were reported in P. falciparum isolates from Ghana [19]. It has been suggested that the new mutant might disrupt sulfadoxine binding to its active site, thus reducing the susceptibility of P. falciparum to the drug [14]. Considering the huge burden of malaria in Nigeria, particularly among pregnant women, and the lack of recent data on the extent of the spread of the Pfdhps 431V/581G mutation in the country and the region as a whole, there is an urgent need to evaluate the current prevalence of the Pfdhps 431V, Pfdhps 431V/-581G and Pfdhps 581G/-613S bearing parasites and, subsequently, the impact of these haplotypes on the protective efficacy of IPTp.
The present study assesses the prevalence of SNPs in Pfdhps in southwest Nigeria where the prevalence Pfdhps 431V/581G mutations are suspected to be increasing.
Methods
Study design
This was a cross-sectional study for sampling possible P. falciparum infection in pregnant women above 18 years who had received SP for IPTp and additionally, in both asymptomatic and febrile healthcare-seeking adults at a general outpatient clinic. The study participants were all tested for malaria by rapid diagnostic test (RDT) and those who tested positive were treated in line with the national guidelines for the treatment of uncomplicated malaria.
Study area and site
The study was conducted at the Obafemi Awolowo University Teaching Hospitals Complex (OAUTHC) located in the Southwest region of Nigeria, at Ile-Ife in Osun State. Ile-Ife is situated in the Ife/Ijesa Senatorial district and the city has a population of approximately 400,763 people according to the last estimate of Nigeria’s population (https://populationstat.com/nigeria/ife). Ile-Ife stands between latitudes 7°28N and 7°45N, and longitudes 4°30E and 4°34E. The transmission of P. falciparum in this area is all-year-round [20]. The pregnant women were recruited at the Antenatal Clinic of OAUTHC, Ile-Ife. In addition, healthcare-seeking adults in the General Outpatient Department of the hospital also participated in the study.
Ethics approval
Ethics approval was obtained from the Health Research and Ethics Committee (HREC) of the Obafemi Awolowo University Teaching Hospitals Complex, Ile-Ife (Protocol Number-ERC/2020/02/24). The prospective participants were provided with adequate information about the study and written informed consent was obtained from all the study participants eligible for participation.
Sample collection
The samples were collected from the study participants between October 2020 and April 2021. Two millilitres of blood samples were collected in EDTA vacutainer tubes and 50 µl aliquots of each sample were spotted on 903 Whatman protein saver cards and stored as dried blood spot (DBS) samples. The DBS samples were stored with desiccant at room temperature prior to shipment to the Centre of Medical Parasitology, University of Copenhagen, for genomic analysis.
DNA extraction
DNA was extracted in 1 ml DNA 96-well plates using DNA extraction kits supplied by Omega Bio-tek (Norcross, GA, USA). Each DBS sample was cut into the 1-ml well and the DNA was extracted according to the manufacturer’s manual. DNA samples were stored at − 20 °C until further downstream applications.
Detection of P. falciparum infections
A nested PCR assay on the RDT positive samples for the detection of malaria parasite was performed as previously reported [21, 22]. The first PCR amplified the entire rPLU6 (TTAAAATTGTTGCAGTTAAAACG) and rPLU5 (CCTGTTGTTGCCTTAAACTTC). Primers for the nested PCR step were Plasm-all-n1fw (CCTTCAGTACCTTATGAGAAATC) and Plasm-all-n2rw (TCTGTCAATCCTACTCTTGTCTT). The cycle conditions for the primary PCR were: initial denaturation at 94 °C for 15 min followed by 30 cycles of annealing at 58 °C for 2 min, extension at 72 °C for 2 min, denaturation at 94 °C for 1 min with a final round of 58 °C for 2 min and 72 °C for 5 min. The conditions for the nested amplification were initial activation 94℃ for 15 min, denaturation at 94 °C for 1 min, annealing at 55 °C for 2 min, and extension at 72 °C for 2 min, with the last three steps repeated in 30 cycles. The amplification product from nested PCR was approximately 170 base pairs.
Sequencing of molecular Pfdhps and Pfdhfr resistance markers
All samples positive for P. falciparum malaria were analysed using an Illumina targeted amplicon sequencing technique as described elsewhere with minor modifications [23].
Data analysis
Sequencing data were analysed using Galaxy tools [24], trimmomatic [25], Python, and MS Excel (2016). Data were analysed with MS Excel and a plot was constructed with a plotting package in GraphPad® software. The distributions of the haplotypes containing Pfdhps 431V allele among the two populations were compared using Fisher’s exact test. The difference was considered statistically significant if the P-value was less than 0.05.
Mapping from previous reports on Pfdhps 431V, 437G, 540E, 581G and 613S resistant alleles
A survey of published reports since 2007 till date on the prevalence of Pfdhps 431V and 581G mutations in Nigeria [14, 16, 17, 26–35], and neighbouring countries [18, 36–47], was conducted, with no regards to age groups or specific population subgroups, to extract information on the total number of samples with mutations at the following positions 431, 437, 540, 581, 613, and the number of changes at each position. The mapping was performed in R version 4.0.3, and used shape files obtained from the Database of Global Administrative Areas Map (GADM) https://gadm.org/download_country.html.
Results
A total of 561 participants consisting of pregnant women (n = 175) and healthcare-seeking adults (n = 386) gave their consent to donate 2 ml of venous blood, with a total of 188 samples (pregnant women (n = 40) and healthcare-seeking adults (n = 148)) being positive for malaria by RDT. However, assay by PCR revealed P. falciparum DNA in 128 samples out of 188 RDT positive samples. Only 33 (out of 40) samples among the asymptomatic pregnant women revealed malaria positivity after the PCR assay; 30 from the samples collected at enrolment during the third trimester and 3 collected at delivery (these were pooled together due a limited sample size during delivery). On the other hand, 88 (out of 148) samples among individuals recruited at the Outpatient Clinic were confirmed to be positive for malaria by PCR.
Prevalence of Pfdhps and Pfdhfr haplotypes
All the PCR-based P. falciparum positive samples (n = 121) were genotyped for Pfdhfr and Pfdhps, out of which, 74 and 61 samples were successfully amplified, sequenced and fully profiled for mutations in Pfdhfr (50, 51, 59, 108 and 164) and Pfdhps (431, 436, 437, 540, 581 and 613), respectively. A high prevalence of Pfdhfr mutant alleles (omitting mixed genotype infections) was detected at N51I (97.3%, 72/74), C59R (97.3%; 72/74) and S108N (98.6%; 73/74). The most common SNP in Pfdhps were the A437G identified in 100% of the samples. The prevalence of mutations at I431V, A581G and A613S were found to be 40.9% (25/61), 24.6% (15/61) and 42.6% (26/61), respectively, while no mutants were found at codon resulting in the amino acid substitution at K540E (Table 1). In addition, 49.2% of the samples (30/61) carried the 436 A allele (Table 1).
Table 1.
The prevalence of dhfr and dhps SNPs in P. falciparum isolates from pregnant women and healthcare seeking adults in Ile-Ife, Southwest Nigeria
| Gene | Group | SNP | Wild type n (%) | Mutant type n (%) | Mixed type n (%) |
|---|---|---|---|---|---|
| Pfdhfr | Pregnant women | N51I | 0 (0) | 16 (100) | 0 (0) |
| C59R | 0 (0) | 16 (100) | 0 (0) | ||
| S108N | 0 (0) | 16 (100) | 0 (0) | ||
| I164L | 16 (100) | 0 (0) | 0 (0) | ||
| Community | N51I | 1 (1.7) | 56 (96.6) | 1 (1.7) | |
| C59R | 1 (1.7) | 56 (96.6) | 1 (1.7) | ||
| S108N | 0 (0) | 57 (98.3) | 1 (1.7) | ||
| I164L | 58 (100) | 0 (0) | 0 (0) | ||
| ALL | N51I | 1 (1.4) | 72 (97.2) | 1 (1.4) | |
| C59R | 1 (1.4) | 72 (97.2) | 1 (1.4) | ||
| S108N | 0 (0.0) | 73 (98.6) | 1 (1.4) | ||
| I164L | 74 (100.0) | 0 (0.0) | 0 (0.00) | ||
| Pfdhps | Pregnant women | I431V | 11 (61.1) | 6 (33.3) | 1 (5.6) |
| S436A | 8 (44.4) | 9 (50) | 1 (5.6) | ||
| A437G | 0 (0) | 18 (100) | 0 (0) | ||
| K540E | 18 (100) | 0 (0) | 0 (0) | ||
| A581G | 16 (88.8) | 1 (5.6) | 1 (5.6) | ||
| A613S | 13 (72.2) | 4 (22.2) | 1 (5.6) | ||
| Community | I431V | 22 (51.2) | 19 (44.2) | 2 (4.6) | |
| S436A | 17 (39.5) | 21 (48.8) | 5 (11.7) | ||
| A437G | 0 (0) | 43 (100) | 0 (0) | ||
| K540E | 43 (100) | 0 (0) | 0 (0) | ||
| A581G | 26 (60.5) | 14 (32.6) | 3 (6.9) | ||
| A613S | 18 (41.8) | 22 (51.1) | 3 (6.9) | ||
| All | I431V | 33 (54.1) | 25 (40.9) | 3 (4.9) | |
| S436A | 25 (41.0) | 30 (49.2) | 6 (9.8) | ||
| A437G | 0 (0.0) | 61 (100) | 0 (0.0) | ||
| K540E | 61 (100) | 0 (0.00) | 0 (0.0) | ||
| A581G | 42 (68.9) | 15 (24.6) | 4 (6.6) | ||
| A613S | 31 (50.8) | 26 (42.6) | 4 (6.6) |
Out of those successfully amplified and sequenced for Pfdhps and Pfdhfr genes, 61 and 74 of the Pfdhps and Pfdhfr sequences, respectively, were constructed into Pfdhfr/Pfdhps haplotypes, omitting infections with mixed genotypes at more than one codon (Table 2). For Pfdhfr, the triple mutant C50-51I-59R-108N-164I (CIRNI) haplotype was almost at fixation with a frequency of 95.8% (71/74). The other Pfdhfr haplotypes observed were the CNRNI (1.4%, n = 1) and ICNI (1.4%, n = 1). Eight distinct haplotypes of the Pfdhps gene were identified namely; ISGKAA, VAGKAA, VAGKGS, VAGKAS, ISGKGS, IAGKAS, IAGKAA and IAGKGS at a prevalence of 20 (32.8%), 9 (14.8%), 9 (14.8%), 6 (9.8%), 5 (8.2%), 4 (6.6%), 3 (4.9%) and 1 (1.6%), respectively. Combining Pfdhfr and Pfdhps haplotypes resulted in 10 discrete genotypes as presented in Table 2. The most abundant genotype was the quadruple mutants consisting of IRNI + ISGKAA (33.9%), IRNI + IAGKAA (5.4%) and NRNI + VAGKAA (1.8%). In the present data, three different sextuple mutant haplotypes existed, ranging from IRNI + ISGKGS, IRNI + IAGKGS to IRNI + VAGKAS, and all contributed 19.6% of the total haplotypic data. Another extensive mutant haplotype with 7 (septuple) mutant alleles was found in the isolates with genotypes IRNI + VAGKGS (16.1%).
Table 2.
Genetic diversity of Pfdhfr and Pfdhps and the combined Pfdhfr/dhps haplotypes in P. falciparum isolates from Ile-Ife, Southwest Nigeria
| Gene | Category of mutation | Haplotype | Community, n (%) | Pregnant, n (%) | Total, n (%) |
|---|---|---|---|---|---|
| Pfdhfr (n = 74) | Wild type | CNCSI | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Double | CNRNI | 1 (1.7) | 0 (0.0) | 1 (1.4) | |
| CICNI | 1 (1.7) | 0 (0.0) | 1 (1.4) | ||
| Triple | CIRNI | 55 (94.9) | 16 (100.0) | 71 (95.8) | |
| Pfdhps (n = 61) | Wild type | ISAKAA | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Single | ISGKAA | 12 (27.9) | 8 (44.4) | 20 (32.8) | |
| IAGKAA | 2 (4.7) | 1 (5.6) | 3 (5.8) | ||
| Double | IAGKAS | 2 (4.7) | 2 (11.1) | 4 (6.6) | |
| VAGKAA | 5 (11.6) | 4 (22.2) | 5 (9.62) | ||
| Triple | ISGKGS | 5 (11.6) | 0 (0.0) | 9 (14.8) | |
| IAGKGS | 1 (2.3) | 0 (0.0) | 1 (1.6) | ||
| VAGKAS | 5 (11.6) | 1 (5.6) | 6 (9.8) | ||
| Quardruple | VAGKGS | 8 (18.6) | 1 (5.6) | 9 (14.8) | |
| Pfdhfr/Pfdhps (n = 56) | Quadruple mutant | IRNI + ISGKAA | 11 (28.2) | 8 (47.1) | 19 (33.9) |
| IRNI + IAGKAA | 2 (5.1) | 1 (5.9) | 3 (5.4) | ||
| NRNI + VAGKAA | 1 (2.6) | 0 (0.0) | 1 (1.8) | ||
| Quintuple mutant | ICNI + VAGKAS | 1 (2.6) | 0 (0.0) | 1(1.8) | |
| IRNI + IAGKAS | 2 (5.1) | 2 (11.8) | 4 (7.2) | ||
| IRNI + VAGKAA | 4 (10.3) | 4 (23.5) | 8 (14.2) | ||
| Sextuple mutant | IRNI + ISGKGS | 5 (12.8) | 0 (0.0) | 5 (8.9) | |
| IRNI + IAGKGS | 1 (2.6) | 0 (0.0) | 1 (1.8) | ||
| IRNI + VAGKAS | 4 (10.3) | 1 (5.9) | 5 (8.9) | ||
| Septuple mutant | IRNI + VAGKGS | 8 (20.5) | 1 (5.9) | 9 (16.1) |
The frequency of the Pfdhfr IRNI haplotype was as expected, widespread in both pregnant women and in the community arm. The prevalence of the haplotypes that contained 431V mutation (VAGKAA, VAGKAS, VAGKGS) appeared not to be statistically significant between the two groups (Fisher’s exact test, P-value = 0.568) (Fig. 1).
Fig. 1.
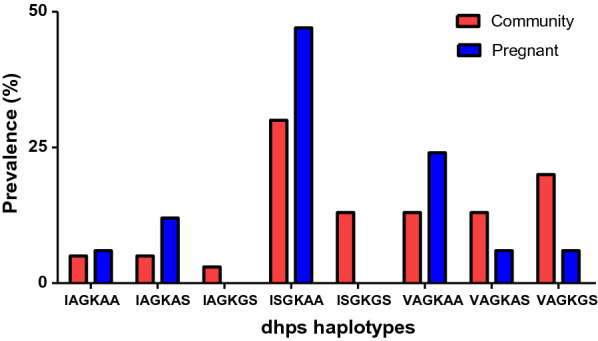
Bar Chart comparing the prevalence of dhps haplotypes among pregnant women and community setting (Npregnant women = 17, Ncommunity = 40)
Current knowledge on the distribution of Pfdhps genotypes in Nigeria and the neighbouring countries
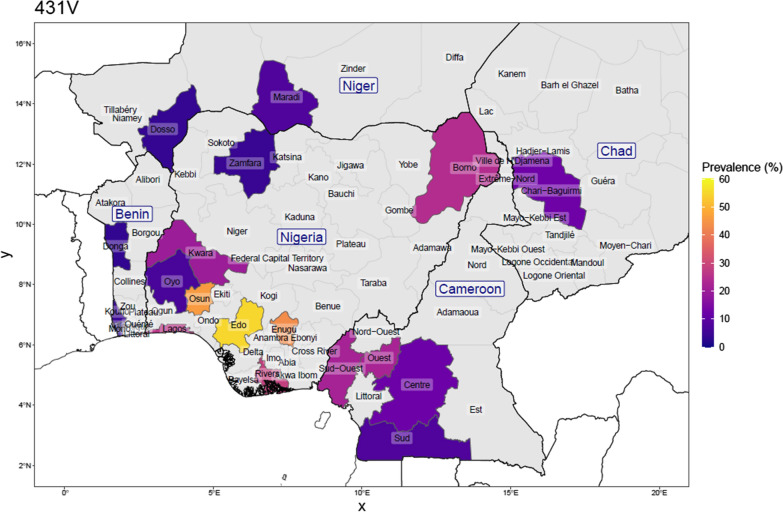
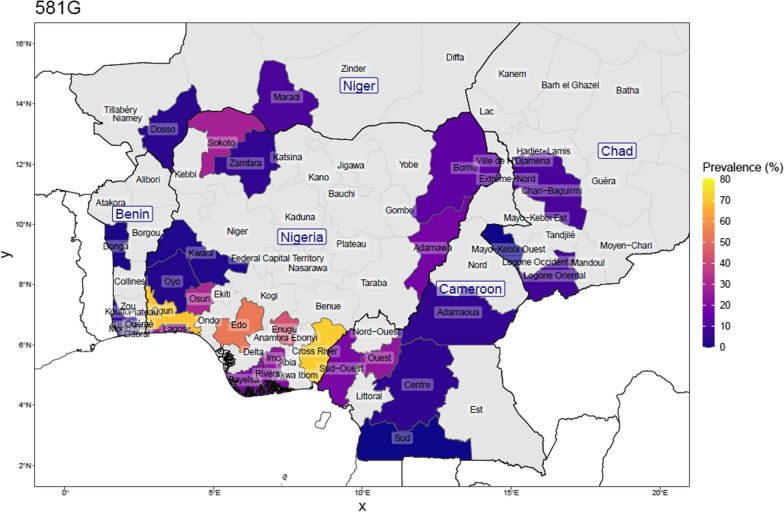
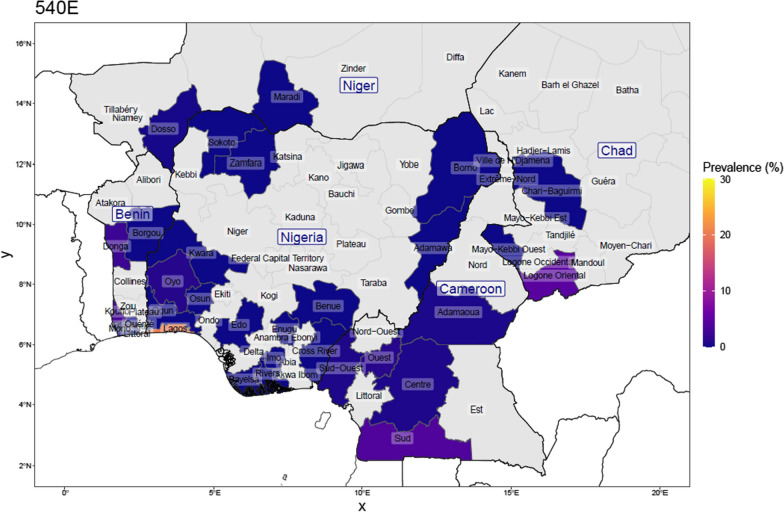
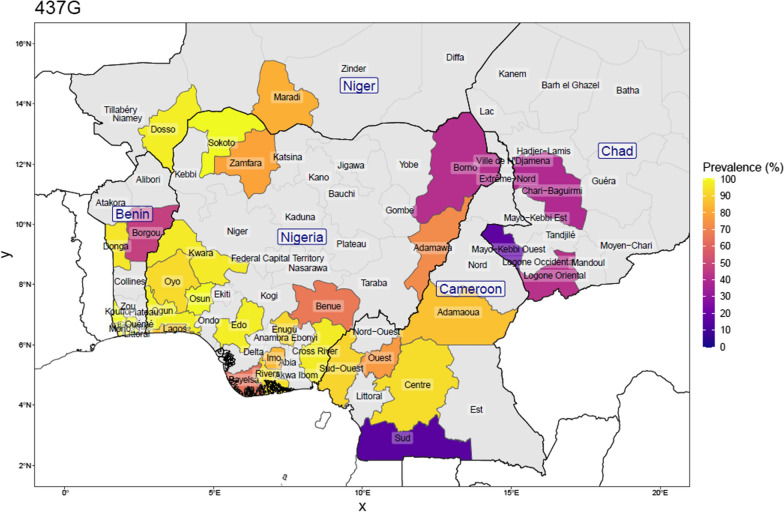
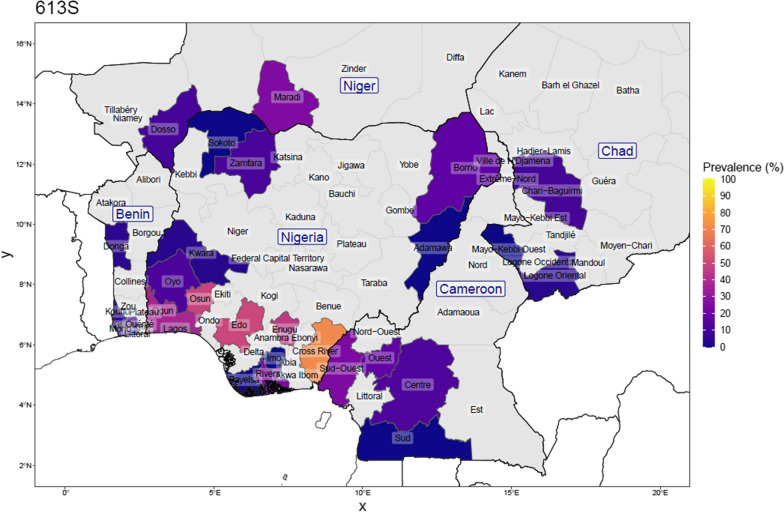
Genotype data from the literature spanning surveys done between 2007 and 2020, were combined with the genotype data from the current study to assess the current distribution of Pfdhps mutations, and to briefly describe the patterns observed (Figs. 2, 3, 4, 5, 6). Published surveys (Additional file 1: Review of surveys on dhps mutation in Nigeria and neighbouring countries) of Pfdhps 431V were limited to only 9 States in Nigeria whereas those of other Pfdhps SNPs had wider coverage (15 States). The Pfdhps 431V mutation reports, show that it has already reached a prevalence of 54.0% in Edo, closely followed by Enugu (43.3%) as well as Osun (45.9%) in the present study. The Pfdhps 431V allele has also been reported in the surveys conducted in Borno (24.5%), Kwara (20.0%), and Lagos (25.9%), Oyo (6.6%), and Rivers (28.4%) and Zamfara (4.0%) states. The data on Pfdhps 437G mutation gathered from published studies revealed that it is widespread throughout Nigeria (prevalence ranging from 41.4 to 100%). Borno state in the Northeast region being the only area where the prevalence of Pfdhps 437G was less than 50%. The mutation has already attained 100% in most places in Nigeria. By contrast, the surveys reporting on Pfdhps 540E (Fig. 4) showed that the mutation is very rare in Nigeria. Of the fifteen states where published surveys were conducted, Pfdhps 540E was detected only in Lagos state (20.1%) and Oyo state (1.0%), and it appears to be non-existent in other states. Variations in the prevalence of Pfdhps-581G and -613S were observed across the country and broadly there is a geographical division between a high prevalence of the Pfdhps 581G/613S (Figs. 5, 6) in the southern States compared to northern states. A much higher prevalence of Pfdhps 581G was observed in Ogun and Cross-River States reaching 69.8 and 71.4%. Some other States in the South-south and Southeast showed a prevalence between 20 and 50%.
Fig. 2.
Prevalence of the Pfdhps-431V mutation in Nigeria, and bordering countries by upper administrative unit, using published studies of surveys done between 2007 and 2021 (for Nigeria: 25 surveys from 16 studies; Ntotal samples = 2262, and for neighbouring countries: 32 surveys from 27 studies; Ntotal samples = 3248). This is calculated as the number with the mutation among the number of samples determined at that position. Proportions were weighed by each study’s sample size for regions where more than one study was conducted
Fig. 3.
Prevalence of the dhps-581G mutation in Nigeria, and bordering countries by upper administrative unit, using published studies of surveys done between 2007 and 2021 (for Nigeria: 25 surveys from 16 studies; Ntotal samples = 2609, and for neighbouring countries: 32 surveys from 27 studies; Ntotal samples = 5705). This is calculated as the number with the mutation among the number of samples determined at that position. Proportions were weighed by each study’s sample size for regions where more than one study was conducted
Fig. 4.
Prevalence of the dhps-540E mutation in Nigeria, and bordering countries by upper administrative unit, using published studies of surveys done between 2007 and 2021 (for Nigeria: 25 surveys from 16 studies; Ntotal samples = 2869, and for neighbouring countries: 32 surveys from 27 studies; Ntotal samples = 6519). This is calculated as the number with the mutation among the number of samples determined at that position. Proportions were weighed by each study’s sample size for regions where more than one study was conducted
Fig. 5.
Prevalence of the dhps-437G mutation in Nigeria, and bordering countries by upper administrative unit, using published studies of surveys done between 2007 and 2021 (for Nigeria: 25 surveys from 16 studies; Ntotal samples = 2794, and for neighbouring countries: 32 surveys from 27 studies; Ntotal samples = 6587). This is calculated as the number with the mutation among the number of samples determined at that position. Proportions were weighed by each study’s sample size for regions where more than one study was conducted
Fig. 6.
Prevalence of the dhps-613S mutation in Nigeria, and bordering countries by upper administrative unit, using published studies of surveys done between 2007 and 2021(for Nigeria: 25 surveys from 16 studies; Ntotal samples = 2606, and for neighbouring countries: 32 surveys from 27 studies; Ntotal samples = 3936). This is calculated as the number with the mutation among the number of samples determined at that position. Proportions were weighed by each study’s sample size for regions where more than one study was conducted
Considering the studies from four neighbouring countries, the prevalence of SP resistance markers was reported in 10 surveys in Benin, in 14 surveys in Cameroon, in 3 surveys in Chad and in 5 surveys in Niger. The samples collected in Cameroon pre-2011 had no 431V and 581G mutation. The Pfdhps 540E mutation was also rarely found in the four neighbouring countries (0.75%). Among the samples reported in all the surveys carried out in Cameroon, 16.6%, 8.5% and 17.7% carried the Pfdhps-431V, -581G and -613S mutants, respectively. In Benin, the data from the 10 surveys indicate 6.8%, 5.9% and 9.7% prevalence of the Pfdhps-431V, -581G and -613S mutations, respectively. Both Chad and Niger Republic survey data indicated a similar trend.
Discussion
The WHO encourages the use of SP-IPTp as part of antenatal care in malaria-endemic settings in Africa. In this study, asymptomatic falciparum malaria was found in 19% of pregnant women. Previous reports on asymptomatic malaria in pregnancy, have shown asymptomatic malaria in pregnancy is prevalent across sub-Saharan African (SSA) countries, and this carries an increased risk of maternal anaemia compared to non-infected pregnant women [48, 49]. It has been well established that asymptomatic malaria infection is associated with poor maternal or fetal health [48]. SP resistance is known to modify the outcome of SP-IPTp strategy. The occurrence of maternal and placental malaria among pregnant women who received SP-IPT may be linked to reduced drug susceptibility of the malaria parasites due to SP resistance as a result of emergence of SNPs in the Pfdhfr/Pfdhps genes of P. falciparum [4, 11, 50–52].
Findings from this study showed that the SNPs in Pfdhfr N51I, -C59R, -S108N (Pfdhfr haplotype CIRNI) and Pfdhps-A437G are widespread in the studied population, reaching a prevalence of beyond 95%. Similar findings have been reported in Nigeria and other countries across SSA [17, 53]. This suggests that the quadruple mutations are already fixed in these populations [18, 32, 54]. Indications from other studies however revealed that the protective effect of SP-IPTp remains intact, regardless of the high prevalence of this quadruple Pfdhfr-Pfdhps mutant [2, 55].
However, the emerging of certain Pfdhps mutant haplotypes in the West African region raise concerns about the continuing effectiveness of SP-IPTp. The present study has substantiated findings from previous studies that suggested the emergence of other Pfdhfr/Pfdhps mutant alleles in Nigeria and the West African region. The present study showed that some P. falciparum isolates were carrying additional mutations at codons I431V, A581G and A613S particularly in the South. However, the frequency of these new mutations is not fixed and furthermore it is not clear how far the new mutations will influence the efficacy of SP-IPTp (and possibly other preventive interventions using SP alone; e.g. the newly revised Perennial Malaria Chemoprevention of infants). Before the advent of the Pfdhps 431V mutation in Nigeria, East Africa witnessed a similar scenario with the emergence of the quintuple (IRN + 437G + 540E) and sextuple (IRN + 437G + 540E + 581G) mutant haplotypes. These were associated with a reduction in the mean birth weight and other metrics of SP-IPTp failure in Tanzania [13, 56]. Although no 540E mutation was observed in the present study, it is possible that the emerging Pfdhps 431V and its co-occurrence with 581G and 613S could lead to a higher order mutant haplotype, and this alternative sextuple mutant might compromise the chemopreventive impact of SP-IPTp in West Africa.
Previous studies indicated that the prevalence of Pfdhps 431V varies across regions in Nigeria in clinical isolates collected at various respective study periods [13–15]. For instance, a low prevalence of Pfdhps 431V mutation was reported in Southwest Nigeria, whereas the prevalence in the parasite population is widespread in the Southeast and Northeast Nigeria among the samples collected about a decade ago [13]. Figures 2 and 3 give a view of the spatial distribution of Pfdhps-431V and -581G in Nigeria and neighbouring countries, using data reported in surveys conducted between 2007 and 2020. This study was the first survey in Osun State and it revealed a high prevalence of the Pfdhps-431V (45.9%) and -581G (31.1%) mutations. The Pfdhps-431V mutation is found to be higher than compared to the neighbouring states, both Oyo and Kwara. Considering the upsurge of mutation in the present study, it would be necessary to bear in mind that the data for Oyo/Kwara are mostly based on older surveys. Oguike et al. [14] provided data in a study site located in Ibadan, approximately 100 km from the present study site, with a frequency of 0 and then,6.5% from a sequential sampling design between 2003 and 2008. There is a high possibility that the level of Pfdhps mutant alleles might have also increased in all the surrounding states. A more recent report by Oboh et al. [16] in Lagos and Edo States revealed a prevalence of 28.5% and 51.4%, respectively, in clinical samples collected between 2016 and 2017 [16]. This is in support that the prevalence of Pfdhps 431V mutation is increasing across the Nigerian population. This present data revealed a frequency of 31.1% for the dhps-581G bearing parasites. This also suggests an upsurge in the circulating 581G mutant allele in Southwest Nigeria similar to previous findings from Lagos and Ogun States [14, 16]. The SNPs A581G and I431V co-occur in 21.3% of the parasite population in this study. This was suggested to render the parasite less susceptible to sulfadoxine [17]. However, the paradigm is already changing to a higher order mutant allele combination consisting the Nigerian sextuple or septuple mutant haplotypes.
It is worth noting that eight distinct haplotypes were detected on the Pfdhps gene starting from ISGKAA, a single mutation, to VAGKGS with 4 mutant alleles. Other haplotypes were IAGKAA, IAGKAS, ISGKGS, VAGKAA, IAGKGS and VAGKAS. The wild haplotype ISAKAA was not found in this study in contrast to the findings reported by Oboh et al. [16]. Most haplotypes were present among the study populations except ISGKGS and IAGKGS which were only present in the group seeking healthcare at the Outpatient Unit. An important observation from this study is a relatively high prevalence of sextuple and septuple haplotypes when the SNPs from both Pfdhfr and Pfdhps genes were combined. Both haplotypes contributed approximately 25.0% of the P. falciparum isolates in the present study, and their presence might pose a high probability of the malaria parasite becoming extensively resistant to SP in Nigeria.
Despite the limitations of the study, such as the small sample size and the use of only one study site, the present data provides information about the current prevalence of molecular markers of SP resistance in Southwest Nigeria. The study revealed that a high frequency of the emerging mutations now exists within the region. In previous studies, other regions in Nigeria depicted a much higher frequency of Pfdhps 431V. Thus, the results of the present study, conducted in the Southwest region of the country, suggest a need to reassess the current prevalence of Pfdhps mutation in other regions as the current prevalence in those regions might have reached an alarming proportion. However, the exact implication of an increase in the frequency of mutation has not been established. Therefore, further work is suggested to examine in vivo susceptibility of the mutant malaria parasites to SP and evaluate the efficacy of SP-IPTp in the context of the emerging mutations.
Supplementary Information
Additional file 1. Review of surveys on dhps mutation in Nigeria and neighbouring countries.
Acknowledgements
This study was supported by an EDCTP2 Preparatory Fellowship (TMA2018PF-2537) to Adebanjo Adegbola.
Abbreviations
- SP-IPTp
Sulfadoxine-Pyrimethamine for Intermittent preventive treatment during pregnancy
- Pfdhps
Plasmodium falciparum Dihydropteroate synthetase
- Pfdhfr
Plasmodium falciparum Dihydrofolate reductase
- PCR
Polymerase chain reaction
- PMC
Perennial malaria chemoprevention
- SMC
Seasonal malaria chemoprevention
- SNPs
Single nucleotide polymorphisms
- RDT
Rapid diagnostic test
- OAUTHC
Obafemi Awolowo University Teaching Hospitals Complex
- HREC
Health Research and Ethics Committee
- DBS
Dried blood spot
- GADM
The Database of Global Administrative Areas Map (GADM)
- SSA
Sub-Saharan African
- WHO
World Health Organization
Author contributions
AJA, OAI, OOB, MA and CR designed the study. OAI, AEU, BAA, OB and AJA performed all clinical and point of care diagnostic testing. HH, EFH, MA and AJA performed molecular parasite quantifcation assays. HH, EFH, AJA and AM conducted analysis and pooling of data to produce the maps. AJA, OOB, OAI, AEU, HH, MA, AM, CR prepared and revised the manuscript. All authors read and approved the final manuscript.
Funding
AJA is supported by a EDCTP Preparatory fellowship (TMA2018PF-2537) for his training at the Centre for Medical Parasitology and Immunology, University of Copenhagen, Denmark.
Availability of data and materials
Data are available upon reasonable request to the corresponding author.
Declarations
Ethics approval and consent to participate
Ethics approval was obtained from the Health Research and Ethics Committee (HREC) of the Obafemi Awolowo University Teaching Hospitals Complex, Ile-Ife (Protocol Number-ERC/2020/02/24). The prospective participants were provided with adequate information about the study and written informed consent was obtained from all the study participants eligible for participation.
Consent for publication
Not applicable.
Competing interests
The authors have declared no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.WHO . World Malaria Report. Geneva: World Health Organization; 2021. [Google Scholar]
- 2.WHO . Guidelines for malaria. Geneva: World Health Organization; 2022. [Google Scholar]
- 3.Mbonye AK, Birungi J, Yanow SK, Shokoples S, Malamba S, Alifrangis M, et al. Prevalence of Plasmodium falciparum resistance markers to sulfadoxine-pyrimethamine among pregnant women receiving intermittent preventive treatment for malaria in Uganda. Antimicrob Agents Chemother. 2015;59:5475–5482. doi: 10.1128/AAC.00507-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van Eijk AM, Larsen DA, Kayentao K, Koshy G, Slaughter DEC, Roper C, et al. Effect of Plasmodium falciparum sulfadoxine-pyrimethamine resistance on the effectiveness of intermittent preventive therapy for malaria in pregnancy in Africa: a systematic review and meta-analysis. Lancet Infect Dis. 2019;19:546–556. doi: 10.1016/S1473-3099(18)30732-1. [DOI] [PubMed] [Google Scholar]
- 5.Maduka O. End malaria for good: a review of current strategies and future novelties for malaria elimination in Nigeria. MalariaWorld J. 2018;9:1. [PMC free article] [PubMed] [Google Scholar]
- 6.Spalding M, Eyase F, Akala H, Bedno S, Prigge S, Coldren R, et al. Increased prevalence of the pfdhfr/phdhps quintuple mutant and rapid emergence of pfdhps resistance mutations at codons 581 and 613 in Kisumu, Kenya. Malar J. 2010;9:338. doi: 10.1186/1475-2875-9-338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kuesap J, Suphakhonchuwong N, Kalawong L, Khumchum N. Molecular markers for sulfadoxine/pyrimethamine and chloroquine resistance in Plasmodium falciparum in Thailand. Korean J Parasitol. 2022;60:109–116. doi: 10.3347/kjp.2022.60.2.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jiang T, Chen J, Fu H, Wu K, Yao Y, Eyi JUM, et al. High prevalence of Pfdhfr-Pfdhps quadruple mutations associated with sulfadoxine-pyrimethamine resistance in Plasmodium falciparum isolates from Bioko Island, Equatorial Guinea. Malar J. 2019;18:101. doi: 10.1186/s12936-019-2734-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jiang T, Cheng W, Yao Y, Tan H, Wu K, Li J. Molecular surveillance of anti-malarial resistance Pfdhfr and Pfdhps polymorphisms in African and Southeast Asia Plasmodium falciparum imported parasites to Wuhan, China. Malar J. 2020;9:434. doi: 10.1186/s12936-020-03509-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pearce RJ, Pota H, Evehe MS, Bâel H, Mombo-Ngoma G, Malisa AL, et al. Multiple origins and regional dispersal of resistant dhps in African Plasmodium falciparum malaria. PLoS Med. 2009;6:e1000055. doi: 10.1371/journal.pmed.1000055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gutman J, Kalilani L, Taylor S, Zhou Z, Wiegand RE, Thwai KL, et al. The A581G mutation in the gene encoding Plasmodium falciparum dihydropteroate synthetase reduces the effectiveness of sulfadoxine-pyrimethamine preventive therapy in malawian pregnant women. J Infect Dis. 2015;211:1997–2005. doi: 10.1093/infdis/jiu836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Minja DT, Schmiegelow C, Mmbando B, Boström S, Oesterholt M, Magistrado P, et al. Plasmodium falciparum mutant haplotype infection during pregnancy associated with reduced birthweight, Tanzania. Emerg Infect Dis. 2013;19:1446–1454. doi: 10.3201/eid1909.130133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hansson H, Minja DTR, Moeller SL, Lusingu JPA, Bygbjerg IC, Yde AM, et al. Reduced birth weight caused by sextuple drug-resistant Plasmodium falciparum infection in early second trimester. J Infect Dis. 2021;224:1605–1613. doi: 10.1093/infdis/jiab117. [DOI] [PubMed] [Google Scholar]
- 14.Oguike MC, Falade CO, Shu E, Enato IG, Watila I, Baba ES, et al. Molecular determinants of sulfadoxine-pyrimethamine resistance in Plasmodium falciparum in Nigeria and the regional emergence of dhps 431V. Int J Parasitol Drugs Drug Resist. 2016;6:220–229. doi: 10.1016/j.ijpddr.2016.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sutherland CJ, Fifer H, Pearce RJ, bin Reza F, Nicholas M, Haustein T, et al. Novel pfdhps haplotypes among imported cases of Plasmodium falciparum malaria in the United Kingdom. Antimicrob Agents Chemother. 2009;53:3405–3410. doi: 10.1128/AAC.00024-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Oboh MA, Singh US, Antony HA, Ndiaye D, Badiane AS, Ali NA, et al. Molecular epidemiology and evolution of drug-resistant genes in the malaria parasite Plasmodium falciparum in southwestern Nigeria. Infect Genet Evol. 2018;66:222–228. doi: 10.1016/j.meegid.2018.10.007. [DOI] [PubMed] [Google Scholar]
- 17.Quan H, Igbasi U, Oyibo W, Omilabu S, Chen SB, Shen HM, et al. High multiple mutations of Plasmodium falciparum-resistant genotypes to sulphadoxine-pyrimethamine in Lagos, Nigeria. Infect Dis Poverty. 2020;9:91. doi: 10.1186/s40249-020-00712-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chauvin P, Menard S, Iriart X, Nsango SE, Tchioffo MT, Abate L, et al. Prevalence of Plasmodium falciparum parasites resistant to sulfadoxine/pyrimethamine in pregnant women in Yaoundé, Cameroon: emergence of highly resistant pfdhfr/pfdhps alleles. J Antimicrob Chemother. 2015;70:2566–2571. doi: 10.1093/jac/dkv160. [DOI] [PubMed] [Google Scholar]
- 19.Zhao L, Pi L, Qin Y, Lu Y, Zeng W, Xiang Z, et al. Widespread resistance mutations to sulfadoxine-pyrimethamine in malaria parasites imported to China from Central and Western Africa. Int J Parasitol Drugs Drug Resist. 2020;12:1–6. doi: 10.1016/j.ijpddr.2019.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ajala O, Olayiwola W. An assessment of the growth of Ile-Ife, Osun State Nigeria, using multi-temporal imageries. J Geogr Geol. 2013;5:43–54. [Google Scholar]
- 21.Touré M, Petersen PT, Bathily SN, Sanogo D, Wang CW, Schiøler KL, et al. Molecular evidence of malaria and zoonotic diseases among rapid diagnostic test-negative febrile patients in low-transmission season, Mali. Am J Trop Med Hyg. 2017;96:335–337. doi: 10.4269/ajtmh.16-0700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Snounou G, Pinheiro L, Gonçalves A, Fonseca L, Dias F, Brown KN, et al. The importance of sensitive detection of malaria parasites in the human and insect hosts in epidemiological studies, as shown by the analysis of field samples from Guinea Bissau. Trans R Soc Trop Med Hyg. 1993;87:649–653. doi: 10.1016/0035-9203(93)90274-T. [DOI] [PubMed] [Google Scholar]
- 23.Nag S, Ursing J, Rodrigues A, Crespo M, Krogsgaard C, Lund O, et al. Proof of concept: used malaria rapid diagnostic tests applied for parallel sequencing for surveillance of molecular markers of anti-malarial resistance in Bissau, Guinea-Bissau during 2014–2017. Malar J. 2019;18:252. doi: 10.1186/s12936-019-2894-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ewels P, Magnusson M, Lundin S, Käller M. MultiQC: summarize analysis results for multiple tools and samples in a single report. Bioinformatics. 2016;32:3047–3048. doi: 10.1093/bioinformatics/btw354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics. 2014;30:2114–2120. doi: 10.1093/bioinformatics/btu170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Beshir KB, Muwanguzi J, Nader J, Mansukhani R, Traore A, Gamougam K, et al. Prevalence of Plasmodium falciparum haplotypes associated with resistance to sulfadoxine-pyrimethamine and amodiaquine before and after upscaling of seasonal malaria chemoprevention in seven African countries: a genomic surveillance study. Lancet Infect Dis. 2022. Online ahead of print. [DOI] [PubMed]
- 27.Esu E, Tacoli C, Gai P, Berens-Riha N, Pritsch M, Loescher T, et al. Prevalence of the Pfdhfr and Pfdhps mutations among asymptomatic pregnant women in Southeast Nigeria. Parasitol Res. 2018;117:801–807. doi: 10.1007/s00436-018-5754-5. [DOI] [PubMed] [Google Scholar]
- 28.Baba E, Hamade P, Kivumbi H, Marasciulo M, Maxwell K, Moroso D, et al. Effectiveness of seasonal malaria chemoprevention at scale in west and central Africa: an observational study. Lancet. 2020;396:1829–1840. doi: 10.1016/S0140-6736(20)32227-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Balogun ST, Sandabe U, Sodipo O, Okon K, Akanmu A. Single nucleotide polymorphisms of Pfdhfr and Pfdhps genes: implications for malaria prophylactic strategies in Maiduguri, Northeast Nigeria. J Trop Med. 2021;2021:8840089. doi: 10.1155/2021/8840089. [DOI] [Google Scholar]
- 30.Ikpa T, Sha'a KK, Auta I. Molecular markers of sulfadoxine-pyrimethamine resistant malaria prior to intermittent preventive treatment among pregnancies in Makurdi, Nigeria. Int J Biol Chem Sci. 2014;8:1961–1968. doi: 10.4314/ijbcs.v8i5.1. [DOI] [Google Scholar]
- 31.Iwalokun BA, Iwalokun SO, Adebodun V, Balogun M. Carriage of mutant dihydrofolate reductase and dihydropteroate synthase genes among Plasmodium falciparum isolates recovered from pregnant women with asymptomatic infection in Lagos, Nigeria. Med Princ Pract. 2015;24:436–443. doi: 10.1159/000430987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fagbemi KA, Adebusuyi SA, Nderu D, Adedokun SA, Pallerla SR, Amoo AOJ, et al. Analysis of sulphadoxine-pyrimethamine resistance-associated mutations in Plasmodium falciparum isolates obtained from asymptomatic pregnant women in Ogun State, Southwest Nigeria. Infect Genet Evol. 2020;85:104503. doi: 10.1016/j.meegid.2020.104503. [DOI] [PubMed] [Google Scholar]
- 33.Kayode AT, Ajogbasile FV, Akano K, Uwanibe JN, Oluniyi PE, Eromon PJ, et al. Polymorphisms in Plasmodium falciparum dihydropteroate synthetase and dihydrofolate reductase genes in Nigerian children with uncomplicated malaria using high-resolution melting technique. Sci Rep. 2021;11:471. doi: 10.1038/s41598-020-80017-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chijioke-Nwauche I, Oguike MC, Nwauche CA, Beshir KB, Sutherland CJ. Antimalarial drug resistance markers in human immunodeficiency virus (HIV)-positive and HIV-negative adults with asymptomatic malaria infections in Port Harcourt, Nigeria. Trans R Soc Trop Med Hyg. 2021;115:531–537. doi: 10.1093/trstmh/trab061. [DOI] [PubMed] [Google Scholar]
- 35.Ahouidi A, Ali M, Almagro-Garcia J, Amambua-Ngwa A, Amaratunga C, Amato R, et al. An open dataset of Plasmodium falciparum genome variation in 7,000 worldwide samples. Wellcome Open Res. 2021;6:42. doi: 10.12688/wellcomeopenres.16168.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Huijben S, Macete E, Mombo-Ngoma G, Ramharter M, Kariuki S, Desai M, et al. Counter-selection of antimalarial resistance polymorphisms by intermittent preventive treatment in pregnancy. J Infect Dis. 2020;221:293–303. doi: 10.1093/infdis/jiz451. [DOI] [PubMed] [Google Scholar]
- 37.Svigel SS, Adeothy A, Kpemasse A, Houngbo E, Sianou A, Saliou R, et al. Low prevalence of highly sulfadoxine-resistant dihydropteroate synthase alleles in Plasmodium falciparum isolates in Benin. Malar J. 2021;20:72. doi: 10.1186/s12936-021-03605-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ogouyèmi-Hounto A, Ndam NT, Fadégnon G, Azagnandji C, Bello M, Moussiliou A, et al. Low prevalence of the molecular markers of Plasmodium falciparum resistance to chloroquine and sulphadoxine/pyrimethamine in asymptomatic children in Northern Benin. Malar J. 2013;12:413. doi: 10.1186/1475-2875-12-413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ogouyèmi-Hounto A, Ndam NT, Kinde Gazard D, d'Almeida S, Koussihoude L, Ollo E, et al. Prevalence of the molecular marker of Plasmodium falciparum resistance to chloroquine and sulphadoxine/pyrimethamine in Benin seven years after the change of malaria treatment policy. Malar J. 2013;12:147. doi: 10.1186/1475-2875-12-147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Moussiliou A, De Tove YS-S, Doritchamou J, Luty AJF, Massougbodji A, Alifrangis M, et al. High rates of parasite recrudescence following intermittent preventive treatment with sulphadoxine-pyrimethamine during pregnancy in Benin. Malar J. 2013;12:195. doi: 10.1186/1475-2875-12-195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Moyeh MN, Njimoh DL, Evehe MS, Ali IM, Nji AM, Nkafu DN, et al. Effects of drug policy changes on evolution of molecular markers of Plasmodium falciparum resistance to chloroquine, amodiaquine, and sulphadoxine-pyrimethamine in the South West Region of Cameroon. Malar Res Treat. 2018;2018:7071383. doi: 10.1155/2018/7071383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Apinjoh TO, Mugri RN, Miotto O, Chi HF, Tata RB, Anchang-Kimbi JK, et al. Molecular markers for artemisinin and partner drug resistance in natural Plasmodium falciparum populations following increased insecticide treated net coverage along the slope of mount Cameroon: cross-sectional study. Infect Dis Poverty. 2017;6:136. doi: 10.1186/s40249-017-0350-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.L'Episcopia M, Kelley J, Djeunang Dongho BG, Patel D, Schmedes S, Ravishankar S, et al. Targeted deep amplicon sequencing of antimalarial resistance markers in Plasmodium falciparum isolates from Cameroon. Int J Infect Dis. 2021;107:234–241. doi: 10.1016/j.ijid.2021.04.081. [DOI] [PubMed] [Google Scholar]
- 44.Tuedom AGB, Sarah-Matio EM, Moukoko CEE, Feufack-Donfack BL, Maffo CN, Bayibeki AN, et al. Antimalarial drug resistance in the Central and Adamawa regions of Cameroon: prevalence of mutations in P. falciparum crt, Pfmdr1, Pfdhfr and Pfdhps genes. PLoS ONE. 2021;16:e0256343. doi: 10.1371/journal.pone.0256343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sarah-Matio EM, Guillochon E, Nsango SE, Abate L, Ngou CM, Bouopda GA, et al. Genetic diversity of Plasmodium falciparum and distribution of antimalarial drug resistance mutations in symptomatic and asymptomatic infections. Antimicrob Agents Chemother. 2022;66:e0018822. doi: 10.1128/aac.00188-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Das S, Kérah-Hinzoumbé C, Kebféné M, Srisutham S, Nagorngar TY, Saralamba N, et al. Molecular surveillance for operationally relevant genetic polymorphisms in Plasmodium falciparum in Southern Chad, 2016–2017. Malar J. 2022;21:83. doi: 10.1186/s12936-022-04095-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Grais RF, Laminou IM, Woi-Messe L, Makarimi R, Bouriema SH, Langendorf C, et al. Molecular markers of resistance to amodiaquine plus sulfadoxine-pyrimethamine in an area with seasonal malaria chemoprevention in south central Niger. Malar J. 2018;17:98. doi: 10.1186/s12936-018-2242-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yimam Y, Nateghpour M, Mohebali M, Abbaszadeh Afshar MJ. A systematic review and meta-analysis of asymptomatic malaria infection in pregnant women in sub-Saharan Africa: a challenge for malaria elimination efforts. PLoS ONE. 2021;16:e0248245. doi: 10.1371/journal.pone.0248245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Obebe OO, Olajuyigbe OO, Falohun OO. Prevalence of asymptomatic Plasmodium falciparum infection in pregnant women in Nigeria: a systematic review and meta-analysis. Ann Parasitol. 2020;66:283–294. doi: 10.17420/ap6603.266. [DOI] [PubMed] [Google Scholar]
- 50.Cisse M, Awandare GA, Soulama A, Tinto H, Hayette M-P, Guiguemdé RT. Recent uptake of intermittent preventive treatment during pregnancy with sulfadoxine-pyrimethamine is associated with increased prevalence of Pfdhfr mutations in Bobo-Dioulasso, Burkina Faso. Malar J. 2017;16:38. doi: 10.1186/s12936-017-1695-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Conrad MD, Mota D, Foster M, Tukwasibwe S, Legac J, Tumwebaze P, et al. Impact of intermittent preventive treatment during pregnancy on Plasmodium falciparum drug resistance-mediating polymorphisms in Uganda. J Infect Dis. 2017;216:1008–1017. doi: 10.1093/infdis/jix421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Matondo SI, Temba GS, Kavishe AA, Kauki JS, Kalinga A, van Zwetselaar M, et al. High levels of sulphadoxine-pyrimethamine resistance Pfdhfr-Pfdhps quintuple mutations: a cross sectional survey of six regions in Tanzania. Malar J. 2014;13:152. doi: 10.1186/1475-2875-13-152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Pacheco MA, Schneider KA, Cheng Q, Munde EO, Ndege C, Onyango C, et al. Changes in the frequencies of Plasmodium falciparum dhps and dhfr drug-resistant mutations in children from Western Kenya from 2005 to 2018: the rise of Pfdhps S436H. Malar J. 2020;19:378. doi: 10.1186/s12936-020-03454-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ojurongbe O, Nguetse CN, Fayemiwo SA, Falade CO, Ojurongbe TA, Thomas BN, et al. High prevalence of dihydrofolate reductase gene mutations in Plasmodium falciparum parasites among pregnant women in Nigeria after reported use of sulfadoxine-pyrimethamine. Pathog Glob Health. 2018;112:86–92. doi: 10.1080/20477724.2017.1422615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Falade CO, Yusuf BO, Fadero FF, Mokuolu OA, Hamer DH, Salako LA. Intermittent preventive treatment with sulphadoxine-pyrimethamine is effective in preventing maternal and placental malaria in Ibadan, south-western Nigeria. Malar J. 2007;6:88. doi: 10.1186/1475-2875-6-88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Minja D, Schmiegelow C, Mmbando B, Boström S, Oesterholt M, Pehrson C, et al. Plasmodium falciparum mutant haplotype infection during pregnancy associated with reduced birthweight, Tanzania. Emerg Infect Dis. 2013;19:1446–1454. doi: 10.3201/eid1909.130133. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Review of surveys on dhps mutation in Nigeria and neighbouring countries.
Data Availability Statement
Data are available upon reasonable request to the corresponding author.