Abstract
Liver fibrosis is caused by chronic liver damage and results in the aberrant accumulation of extracellular matrix during disease progression. Despite the identification of the HAT enzyme p300 as a major factor for liver fibrosis, the development of therapeutic agents targeting the regulation of p300 has not been reported. We validated a novel p300 inhibitor (A6) on the improvement of liver fibrosis using two mouse models, mice on a choline-deficient high-fat diet and thioacetamide-treated mice. We demonstrated that pathological hall-marks of liver fibrosis were significantly diminished by A6 treatment through Masson’s trichrome and Sirius red staining on liver tissue and found that A6 treatment reduced the expression of matricellular protein genes. We further showed that A6 treatment improved liver fibrosis by reducing the stability of p300 protein via disruption of p300 binding to AKT. Our findings suggest that targeting p300 through the specific inhibitor A6 has potential as a major therapeutic avenue for treating liver fibrosis.
Keywords: A6, Histone acetyltransferase, Inhibitor, Liver fibrosis, p300
INTRODUCTION
Fibrotic disease is an irreversible disease in which the excess deposition of extracellular matrix (ECM) leads to the replacement of normal tissue with fibrous tissue (1). Fibrotic diseases have varied etiologies and pathologies across multiple organs and tissues, including the liver, lungs, skin, and kidney (2), but are commonly found in the final stages of chronic inflammatory reactions within these tissues. The development and progression of fibrosis are regulated by signaling pathways associated with inflammation and tissue regeneration, including pathogen-associated molecular patterns (PAMP), cytokines (IL13, IL21, and TGF-β1), chemokines (MCP-1 and MIP 1b), angiogenic factors (VEGF), and growth factors (PDGF) (1-3).
Hepatic fibrosis is a leading cause of high morbidity and mortality worldwide (4). The fibrotic reaction is generated in response to many forms of chronic liver injury, such as alcohol consumption, viral hepatitis, non-alcoholic fatty liver disease, and cholesteric liver diseases (5). Recent studies on the pathogenesis and treatment of liver fibrosis have focused on the molecular mechanisms driving the pathogenesis of liver fibrosis (6, 7). However, there are still no approved pharmacotherapies for liver fibrosis.
Epigenetic changes regulate physiology, embryogenesis, and tissue development and arise from alterations in DNA methylation, histone modifications, and microRNAs (8, 9). Epigenetic dysregulation is a common factor in the progression of human diseases (10-13), including fibrosis of the liver, kidney and lungs (14-16). Histone acetyltransferases (HAT) and histone deacetylases (HDAC) are critical regulators of epigenetic changes in normal and disease states (17, 18). One HAT protein, p300, is an E1A binding protein that acetylates specific lysine residues on histones and modulates the transcription of genes involved in cell growth, proliferation, survival (19, 20), and organ development. p300 levels are commonly elevated across various fibrotic tissues (21-23), where this enhanced p300 activity promotes the transcription of maternal genes and results in increased collagen synthesis and fibrosis. Recent studies have demonstrated that loss of HDAC3 in the uterus increases p300 recruitment to COL1A1 and COL1A2 genes, leading to the accumulation of endometrial fibrosis and the development of endometriosis (23). Drugs that target epigenetic regulation are currently being tested in clinical trials to alleviate the epigenetic dysregulation driving a wide variety of diseases (24).
Recent studies have reported that p300 is also a crucial factor in the pathogenesis and progression of liver fibrosis (16, 25). Although p300 plays a critical role in the development of liver fibrosis, more research is needed to identify therapeutic agents that can efficiently alter p300 function and expression.
The present study revealed that treatment with a p300-specific inhibitor, A6, significantly reduced liver fibrosis and matricellular proteins in two mouse models. In addition, the expression levels of fibrosis marker genes were significantly reduced by A6 treatment compared to vehicle. In hepatic stellate cells, the binding of p300 to AKT and stabilization of p300 protein were diminished by A6 treatment. Together, our results demonstrate the potential of p300 as a major therapeutic target for liver fibrosis and propose the novel p300 inhibitor A6 as a promising therapeutic agent for liver fibrosis.
RESULTS
Protein levels of p300 are increased during liver fibrosis in mice on CD-HFD
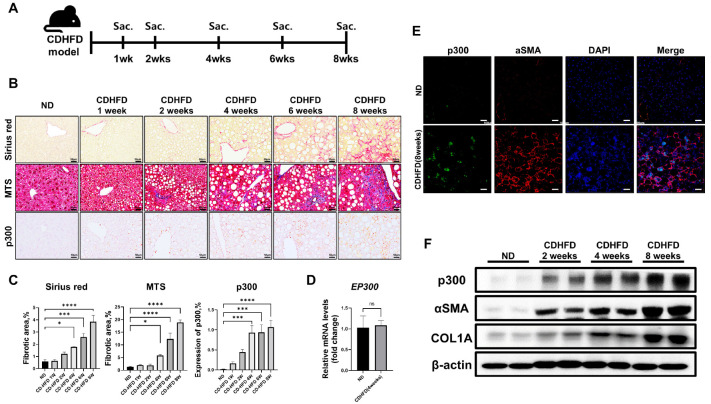
We investigated the expression levels of p300 in the liver during the progression of liver fibrosis in male mice on a choline-deficient, high-fat diet (CD-HFD). We generated liver fibrosis in C57BL/6 mice and sacrificed at 1, 2, 4, 6, and 8 weeks after starting CD-HFD (Fig. 1A). The CD-HFD-induced fibrous liver showed collagen accumulation over this timeframe, as visualized by Sirius red and Masson’s trichrome staining (MTS) (Fig. 1B, C). Within 4 weeks, the level of p300 protein in the liver of mice on CD-HFD was significantly increased compared to mice on a normal diet (ND). However, we detected no difference in p300 mRNA levels across these groups (Fig. 1D). After 8 weeks of CD-HFD, we detected increased levels of p300 localized to sites of increased fibrosis through immunofluorescence (IF) staining of p300 and αSMA, a marker of activated myofibroblasts (Fig. 1E). Similarly, western blot analysis verified that p300 protein levels increased simultaneously with αSMA and type I collagen (COL1A) during the progression of liver fibrosis due to CD-HFD (Fig. 1F). These results show that p300 expression increases during the progression of liver fibrosis.
Fig. 1.
The level of p300 during liver fibrosis. (A) Schematic showing the timeline of animal experiments for the CD-HFD model. (B) Representative photomicrographs of Sirius red staining, MTS, and immunohistochemistry (IHC) analysis of p300 in liver tissues from mice on ND and 1, 2, 4, 6, and 8 weeks after CD-HFD. (C) Quantification data of histological staining images (n = 5) by ImageJ software. Data are represented as mean ± SEM, *P < 0.05, ***P < 0.001, and ****P < 0.0001 by ordinary one-way ANOVA test. (D) The mRNA expression level of p300 in the liver of mice after 4 weeks of ND and CD-HFD (n = 5). ns = not significant. (E) Co-immunofluorescence of p300 and αSMA in mouse liver tissue at 8 weeks of ND and CD-HFD. (F) Western blot analysis of p300, αSMA, and Col1a in CD-HFD-induced mouse liver tissue. β-actin was used as the sample loading control.
CD-HFD-induced mouse liver fibrosis is improved by p300-specific inhibitors
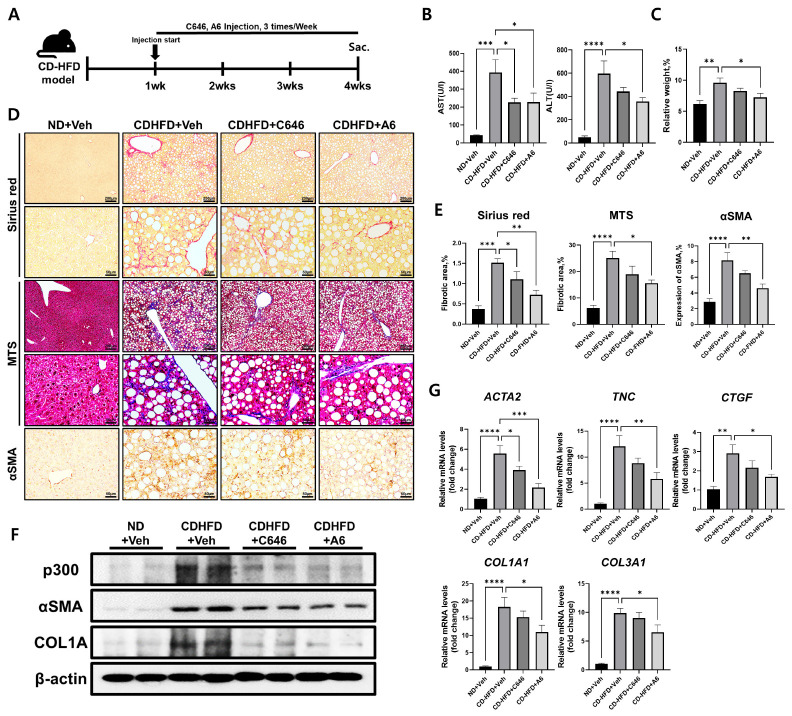
We previously reported that binding to acetyl-CoA, a p300 substrate, was comparable between A6 and a conventional p300 inhibitor, C646 (26). Furthermore, A6 demonstrated anti-fibrotic activity in TGF-β1-stimulated lung fibroblasts and in bleomycin-induced murine lung fibrosis in vivo. In this study, we examined whether the inhibition of p300 activity by A6 or C646 ameliorated the CD-HFD-induced liver fibrosis in mice. One week after starting the mice on CD-HFD, we intraperitoneally injected either A6 or C646 3 times a week for 4 weeks (Fig. 2A). We first assessed liver damage in the mice by measuring the serum levels of aspartate aminotransferase (AST) and alanine transaminase (ALT; n = 5 for each group). Serum AST and ALT levels were significantly reduced by both p300 inhibitors compared to vehicle (Fig. 2B). In addition, the increase in size and weight of the liver and body weight of the mice due to 4 weeks of CD-HFD were significantly restored by A6 treatment compared to vehicle (liver weight/body weight, P < 0.05; body weight, P < 0.05; n = 5 for each group; Fig. 2C and Supplementary Fig. 1). We next verified the anti-fibrotic effect of p300 inhibitors on CD-HFD-induced mouse liver fibrosis. The p300 inhibitor-treated mice exhibited a significant decrease in the intensity of Sirius red staining (C646, P < 0.05; A6, P < 0.01), MTS (A6, P < 0.05), and expression levels of αSMA (A6, P < 0.01) in the liver of mice fed a CD-HFD (Fig. 2D, E). Similarly, western blot analysis verified that p300, αSMA, and COL1A protein levels decreased in mice liver tissues with p300 inhibitors at 4 weeks after CD-HFD (Fig. 2F). The reduction of fibrosis after treatment with p300 inhibitors was further verified by quantification of mRNA levels of fibrosis markers, actin alpha 2, smooth muscle(αSMA) (ACTA2; C646, P < 0.05; A6, P < 0.001), tenascin C (TNC; A6; P < 0.01), collagen type 1, alpha 1 (Col1a1; A6, P < 0.05), collagen type 3, alpha 1 (Col3a1; A6, P < 0.05), and connective tissue growth factor (CTGF; A6, P < 0.05) (Fig. 2G). However, there was no detectable change in mRNA levels of p300, regardless of diet or the administration of p300 inhibitors (Supplementary Fig. 2). These results show that liver fibrosis induced by CD-HFD is ameliorated by the selective reduction of p300 expression and activity.
Fig. 2.
Amelioration of CD-HFD-induced liver fibrosis by p300 inhibitors. (A) Experimental design to investigate the effects of p300 inhibitors on CD-HFD-induced mouse liver fibrosis. Mice were fed CD-HFD for one week prior to treatment. Vehicle (0.01% DMSO 100 μl), C646 (5 mg/kg), or A6 (5 mg/kg) was administered intraperitoneally 3 times a week for an additional 3 weeks (n = 5). (B) Serum levels of AST and ALT after 4 weeks of CD-HFD, with or without p300 inhibitors (n = 5). (C) Average ratio of liver weight to body weight of mice on CD-HFD after treatment with vehicle, C646, or A6 (n = 5). (D, E) Representative photomicrographs (D) and percentages (E) of Sirius red staining, MTS, and IHC analysis of αSMA in mice liver tissues with or without p300 inhibitors at 4 weeks after CD-HFD (n = 5). (F) West-ern blot analysis of p300, αSMA, and COL1A in mice liver tissues with or without p300 inhibitors at 4 weeks after CD-HFD. β-actin was used as the sample loading control. (G) RT-qPCR expression analysis of fibrosis marker genes ACTA2 (αSMA), TNC, Col1a1, Col3a1, and CTGF in the mice liver with or with-out p300 inhibitors at 4 weeks after CD-HFD. Data are represented as mean ± SEM, *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001 by ordinary one-way ANOVA test.
TAA-induced mouse liver fibrosis is ameliorated by p300-specific inhibitors
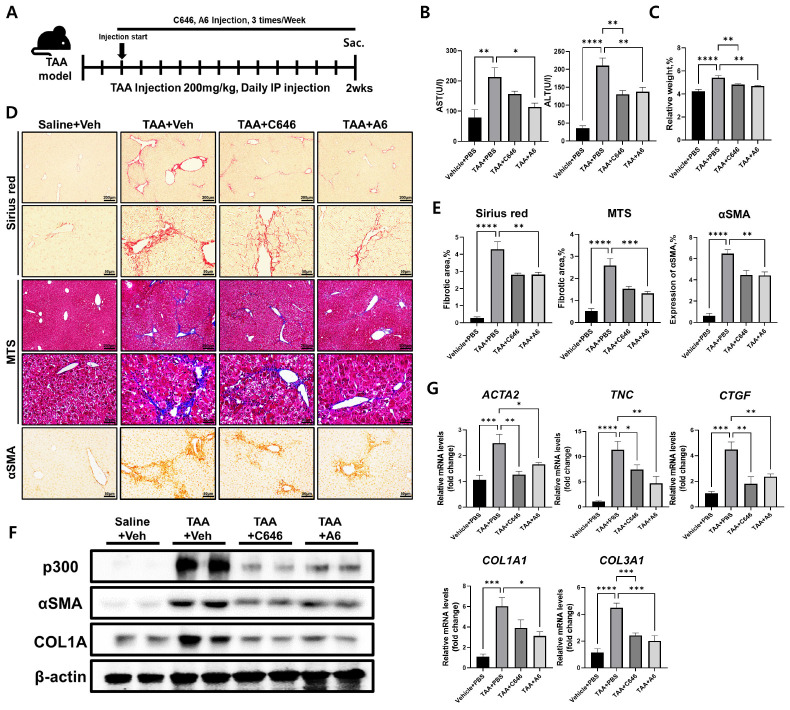
To further validate the broad anti-fibrotic effects of p300 inhibitors in liver fibrotic diseases of various etiologies, we utilized a second method for generating fibrosis across various animal tissues (27). Instead of feeding mice a CD-HFD, the thioacetamide (TAA) model induces fibrosis in murine livers through daily intraperitoneal injection of TAA for 2 weeks. We started intraperitoneal injections of A6 or C646 two days after the first TAA injection and continued p300 inhibitor administration 3 times a week for 2 weeks (Fig. 3A). Similar to the results of the CD-HFD-induced liver fibrosis mouse model, serum AST and ALT levels were significantly reduced by p300 inhibitors compared to vehicle (Fig. 3B). The increase in the ratio of liver weight to body weight of the mice due to the TAA-induced fibrosis was significantly restored by treatment with C646 (P < 0.01) or A6 (P < 0.01) compared to vehicle (Fig. 3C and Supplementary Fig. 3). In addition, p300 inhibitors significantly alleviated the progression of TAA-induced fibrosis and significantly reduced the elevated protein and mRNA levels of mouse liver fibrosis markers (Fig. 3D-G). However, there was no significant change in mRNA levels of p300, regardless of TAA injection or the administration of p300 inhibitors (Supplementary Fig. 4). Similarly, western blot analysis verified that p300, αSMA, and COL1A protein levels decreased in liver tissues from mice treated with TAA with p300 inhibitors (Fig. 3F). In both CD-HFD and TAA models, selective inhibition of p300 activity effectively suppresses the progression of liver fibrosis.
Fig. 3.
Improvement of TAA-induced liver fibrosis by p300 inhibitors. (A) Experimental design to investigate the anti-fibrotic effects of p300 inhibitors on TAA-induced mouse liver fibrosis. Vehicle (0.01% DMSO 100 μl), C646 (5 mg/kg), and A6 (5 mg/kg) were administered intraperitoneally 3 times a week for 2 weeks in TAA-injected mice (n = 5). Saline was used for control. (B) Serum AST and ALT levels of TAA-treated mice with or without p300 inhibitors (n = 5). (C) Average ratio of liver weight to body weight of TAA-injected mice with vehicle, C646, or A6 (n = 5). (D, E) Representative photomicrographs of liv-er tissues (D) and percentages (E) of Sirius red staining, MTS, and IHC analysis of αSMA from TAA-treated mice with or without p300 inhibitors (n = 5). (F) Western blot analysis of p300, αSMA, and COL1A in liver tissues from mice treated with TAA with or without p300 inhibitors. β-actin was used as the sample loading control. (G) RT-qPCR expression analysis of fibrosis marker genes ACTA2(αSMA), TNC, Col1a1, Col3a1, and CTGF in the liver of TAA-treated mice with or without p300 inhibitors at 2 weeks after TAA injection. Data are represented as mean ± SEM, *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001 by ordinary one-way ANOVA test.
p300 inhibitors suppress TGF-β1-induced pro-fibrotic response by interfering with interactions between AKT and p300
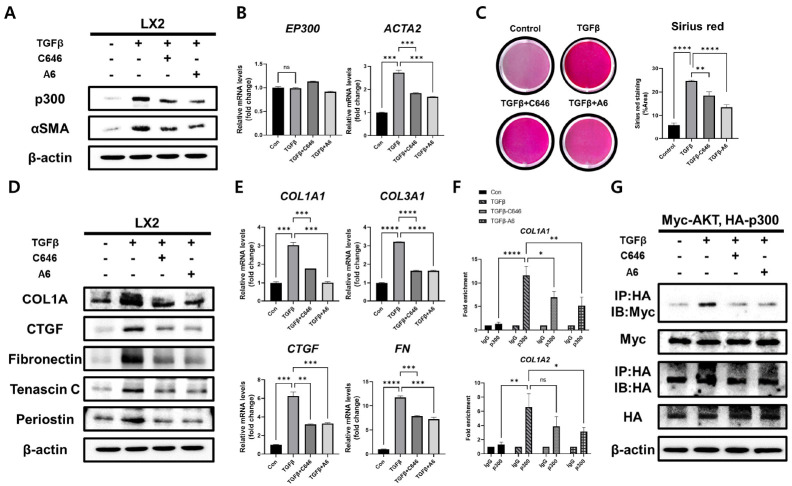
Hepatic stellate cells play a critical role in liver development and fibrosis (28). Previous works have implicated p300 function in stellate cell differentiation and secretion of ECM (25). To elucidate the molecular mechanism for the therapeutic effect of p300 inhibitors on liver fibrosis, the human hepatic stellate cell line (LX2) was treated with TGF-β to in-duce a fibrotic response. When LX2 cells were stimulated by TGF-β1, the protein and gene expression levels of αSMA increased, but this effect on αSMA levels was significantly reduced by the addition of p300 inhibitors (Fig. 4A, B). Treatment with the p300 inhibitors after TGF-β1-induced fibrosis also significantly decreased the elevated collagen levels within the cell matrix (Fig. 4C) and ameliorated the TGF-β1-induced increases in fibrosis marker proteins, COL1A, CTGF, fibronectin (FN), TNC, and periostin (Fig. 4D). As expected, the mRNA levels of fibrosis marker genes COL1A1, COL3A1, CTGF, and FN were significantly reduced in TGF-β1-treated LX2 cells by p300 inhibitor treatment compared with control (Fig. 4E). Since p300 is recruited to the COL1A1 and COL1A2 genes to increase collagen expression (23), we investigated whether the recruitment of p300 to the collagen gene is affected by p300 inhibitors via the chromatin immunoprecipitation (ChIP) assay. When p300 inhibitors were applied to TGF-β1-treated LX2 cells, the recruitment of p300 to the collagen gene was significantly reduced (C646, P < 0.05; A6, P < 0.01 on COL1A1 gene and A6, P < 0.05 on COL1A2 gene; Fig. 4F).
Fig. 4.
Suppression of pro-fibrotic responses in TGF-β1-stimulated LX2 cells via disruption of AKT interaction by p300 inhibitor. (A) Western blot analysis of p300 and αSMA in TGF-β1-stimulated LX2 cells with or without p300 inhibitors. (B) RT-qPCR analysis of expression of p300 and αSMA in TGF-β1-stimulated LX2 cells with or without p300 inhibitors. (C) Representative photomicrographs and percentages of Sirius red staining in TGF-β1-stimulated LX2 cells with or without p300 inhibitors (n = 5). (D) Western blot analysis of fibrosis marker proteins, COL1A, CTGF, FN, TNC, and Periostin, in TGF-β1-stimulated LX2 cells with or without p300 inhibitors. (E) RT-qPCR expression analysis of fibrosis marker genes, COL1A1, COL3A1, CTGF, and FN in TGF-β1-stimulated LX2 cells with or without p300 inhibitors. (F) ChIP assay using IgG or anti-p300 antibody in TGF-β1-stimulated LX2 cells with or without p300 inhibitors. Data are represented as mean ± SEM, *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001 by ordinary one-way ANOVA test. (G) Immunoprecipitation assay using anti-HA or anti-Myc antibodies in LX2 cells (co-transfected Myc-AKT and HA-AKT). β-actin was used as the sample loading control.
AKT is activated during fibrosis through upstream signal cascades that include TGF-β and PDGF, a platelet-derived growth factor (29, 30). Previous studies have shown that p300 interacts with activated AKT in fibrotic hepatic stellate cells (25). In addition, AKT phosphorylation of Ser1834 on p300 is important for regulating the stability and catalytic activity of p300 (25, 31). Therefore, we examined whether disrupting the interaction between p300 and AKT could be contributing to the reduction in p300 protein levels after selective p300 inhibition. Immunoprecipitation of p300 from TGF-β1-stimulated LX2 cells confirmed that the binding affinity of p300 with AKT was decreased by the addition of p300 inhibitors in TGF-β1 treated stellate cells (Fig. 4G). These results suggest that inhibiting the enzymatic activity of p300 induces the dissociation of p300 from AKT and leads to suppression of TGF-β1-induced pro-fibrotic responses.
DISCUSSION
Hepatic fibrotic disease has a high prevalence worldwide and generally progresses to cirrhosis with high mortality (4). Liver fibrosis pathogenesis can be initiated by many diseases, but current studies aim to elucidate the underlying molecular mechanisms. Recent efforts have focused on the regulation of histone acetylation by HATs and HDACs in the progression of fibrotic disease (32, 33). Normally, the HAT enzyme p300 activates gene transcription by inducing acetylation of histone tails (19). Under conditions that promote fibrosis, p300 induces the expression of fibrosis-related genes, such as collagen, CTGF, TNC, and FN (22, 34). In this study, we demonstrated that selective inhibition of p300 alleviates liver fibrosis pathology and pro-fibrotic gene expression in two different mouse models and a hepatic stellate cell line.
Recent studies have utilized p300 inhibitors, including artificial (C646) and natural compounds (Plumbagin, Curcumin, and Gacrcinol), for the treatment of fibrosis (15, 24). However, these compounds inhibit other HAT enzymes in addition to p300. Furthermore, the inhibitory activity of these compounds does not reach levels sufficient for therapeutic efficacy. Therefore, we introduced a novel p300 inhibitor, A6, as a treatment for hepatic fibrosis. Previous studies have shown that A6 has high anti-fibrotic activity in TGF-β-induced lung fibroblast cells and bleomycin-induced mouse lung fibrosis models (26). We demonstrate that both A6 and C646 significantly suppress mouse liver fibrosis induced by either CD-HFD or TAA. Intriguingly, we observed that A6 treatment was more effective than C646 in alleviating serum AST and ALT levels. Similarly, A6 was previously shown to be more effective than C646 in pulmonary fibrosis. A6 has many pharmacokinetic advantages over C646, including a longer half-life with a higher steady-state volume of distribution and a higher total plasma clearance rate than C646 (26). Additional studies will be needed to evaluate the relative efficacy of A6 compared to other p300-specific inhibitors.
To elucidate the molecular mechanism for the therapeutic effect of p300 inhibitors on liver fibrosis, we induced a fibrotic response in the human hepatic stellate cell line, LX2. In this study, the expression levels of fibrosis-related genes increased in TGF-β-induced LX2 cells and significantly decreased in A6-treated cells compared to TGF-β-treated cells alone. Compared to C646, treatment with A6 more significantly suppressed TGF-β-induced pro-fibrotic gene expression in LX2 cells. The superior effects of A6 in mouse liver fibrosis, for both in vivo and in vitro models, suggest that A6 has therapeutic potential across multiple fibrotic diseases where p300 modulates disease progression.
AKT-related signaling pathways are activated in multiple fibrotic diseases and phosphorylation by AKT has been shown to regulate p300 function and stability (25, 35). To determine whether p300 inhibitors reduce the binding affinity between AKT and p300, we treated TGF-β-stimulated LX2 cells with A6 or C646. Treatment with these p300 inhibitors greatly reduced both the protein binding between p300 and AKT and the protein levels of p300. Future studies will need to address whether p300 inhibition affects AKT activity and also how disrupting the interaction with AKT leads to destabilization of the p300 protein. Because we did not detect any changes in p300 mRNA levels across treatments, proteasomal degradation by USP7, which is known to interact with p300, may be responsible for regulating p300 protein levels (36). Additional studies will be necessary to clarify the relationship between p300 function and stabilization during the development of liver fibrosis.
In conclusion, we demonstrated that A6, a p300 selective inhibitor, suppresses the progression of murine liver fibrosis induced by CD-HFD or TAA treatment. We also show that inhibition of p300 activity disrupts the interaction between p300 and AKT and suppresses TGF-β1-induced pro-fibrotic responses. These results suggest that selective inhibition of p300 by A6 has therapeutic potential in treating fibrosis of the liver and other organs.
MATERIALS AND METHODS
Detailed information on Materials and methods is available in the supplementary section.
Funding Statement
ACKNOWLEDGEMENTS We thank Dr. Jae-woo Kim for providing the LX2 cell line. This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean Government (MIST) (No. 2018R1A5A2025079 and 2020R1A2C3003303 to H.-G.Y.).
Footnotes
CONFLICTS OF INTEREST
The authors have no conflicting interests.
REFERENCES
- 1.Wynn TA. Cellular and molecular mechanisms of fibrosis. J Pathol. 2008;214:199–210. doi: 10.1002/path.2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Weiskirchen R, Weiskirchen S, Tacke F. Organ and tissue fibrosis: molecular signals, cellular mechanisms and translational implications. Mol Aspects Med. 2019;65:2–15. doi: 10.1016/j.mam.2018.06.003. [DOI] [PubMed] [Google Scholar]
- 3.Parola M, Pinzani M. Pathophysiology of organ and tissue fibrosis. Mol Aspects Med. 2019;65:1. doi: 10.1016/j.mam.2019.02.001. [DOI] [PubMed] [Google Scholar]
- 4.Lim YS, Kim WR. The global impact of hepatic fibrosis and end-stage liver disease. Clin Liver Dis. 2008;12:733–746. vii. doi: 10.1016/j.cld.2008.07.007. [DOI] [PubMed] [Google Scholar]
- 5.Aydin MM, Akcali KC. Liver fibrosis. Turk J Gastroenterol. 2018;29:14–21. doi: 10.5152/tjg.2018.17330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kisseleva T, Brenner D. Molecular and cellular mechanisms of liver fibrosis and its regression. Nat Rev Gastroenterol Hepatol. 2021;18:151–166. doi: 10.1038/s41575-020-00372-7. [DOI] [PubMed] [Google Scholar]
- 7.Roehlen N, Crouchet E, Baumert TF. Liver fibrosis: mechanistic concepts and therapeutic perspectives. Cell. 2020;9:875. doi: 10.3390/cells9040875.a00dcb356af64fbbabd610298e7d96f7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dupont C, Armant DR, Brenner CA. Epigenetics: definition, mechanisms and clinical perspective. Semin Reprod Med. 2009;27:351–357. doi: 10.1055/s-0029-1237423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim SH, Park J, Park JW, et al. SET7-mediated TIP60 methylation is essential for DNA double-strand break repair. BMB Rep. 2022;55:541–546. doi: 10.5483/BMBRep.2022.55.11.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xue T, Qiu X, Liu H, et al. Epigenetic regulation in fibrosis progress. Pharmacol Res. 2021;173:105910. doi: 10.1016/j.phrs.2021.105910. [DOI] [PubMed] [Google Scholar]
- 11.Sarkar S, Horn G, Moulton K, et al. Cancer development, progression, and therapy: an epigenetic overview. Int J Mol Sci. 2013;14:21087–21113. doi: 10.3390/ijms141021087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Surace AEA, Hedrich CM. The role of epigenetics in autoimmune/inflammatory disease. Front Immunol. 2019;10:1525. doi: 10.3389/fimmu.2019.01525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rai R, Verma SK, Kim D, et al. A novel acetyltransferase p300 inhibitor ameliorates hypertension-associated cardio-renal fibrosis. Epigenetics. 2017;12:1004–1013. doi: 10.1080/15592294.2017.1370173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chung S, Kim S, Son M, et al. Inhibition of p300/CBP-associated factor attenuates renal tubulointerstitial fibrosis through modulation of NF-kB and Nrf2. Int J Mol Sci. 2019;20:1554. doi: 10.3390/ijms20071554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee SY, Kim MJ, Jang S, et al. Plumbagin suppresses pulmonary fibrosis via inhibition of p300 histone acetyltransferase activity. J Med Food. 2020;23:633–640. doi: 10.1089/jmf.2019.4670. [DOI] [PubMed] [Google Scholar]
- 16.Yao W, Wang T, Huang F. p300/CBP as a key nutritional sensor for hepatic energy homeostasis and liver fibrosis. Biomed Res Int. 2018;2018:8168791. doi: 10.1155/2018/8168791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yoon S, Eom GH. HDAC and HDAC inhibitor: from cancer to cardiovascular diseases. Chonnam Med J. 2016;52:1–11. doi: 10.4068/cmj.2016.52.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hillyar C, Rallis KS, Varghese J. Advances in epigenetic cancer therapeutics. Cureus. 2020;12:e11725. doi: 10.7759/cureus.11725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dancy BM, Cole PA. Protein lysine acetylation by p300/CBP. Chem Rev. 2015;115:2419–2452. doi: 10.1021/cr500452k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Giles RH, Peters DJ, Breuning MH. Conjunction dysfunction: CBP/p300 in human disease. Trends Genet. 1998;14:178–183. doi: 10.1016/S0168-9525(98)01438-3. [DOI] [PubMed] [Google Scholar]
- 21.Ghosh AK. FAT-free p300 is good for scar-free tissue repair. J Cell Biochem. 2014;115:1486–1489. doi: 10.1002/jcb.24820. [DOI] [PubMed] [Google Scholar]
- 22.Ghosh AK, Varga J. The transcriptional coactivator and acetyltransferase p300 in fibroblast biology and fibrosis. J Cell Physiol. 2007;213:663–671. doi: 10.1002/jcp.21162. [DOI] [PubMed] [Google Scholar]
- 23.Kim TH, Yoo JY, Choi KC, et al. Loss of HDAC3 results in nonreceptive endometrium and female infertility. Sci Transl Med. 2019;11:eaaf7533. doi: 10.1126/scitranslmed.aaf7533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dekker FJ, Haisma HJ. Histone acetyl transferases as emerging drug targets. Drug Discov Today. 2009;14:942–948. doi: 10.1016/j.drudis.2009.06.008. [DOI] [PubMed] [Google Scholar]
- 25.Dou C, Liu Z, Tu K, et al. P300 acetyltransferase mediates stiffness-induced activation of hepatic stellate cells into tumor-promoting myofibroblasts. Gastroenterology. 2018;154:2209–2221. doi: 10.1053/j.gastro.2018.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hwang SY, Park SY, Hong JY, et al. Field-based rational design of p300 histone acetyltransferase inhibitor and systematic evaluation as an anti-fibrotic agent. Chem Commun (Camb) 2020;56:9795–9798. doi: 10.1039/D0CC03553J. [DOI] [PubMed] [Google Scholar]
- 27.Ravichandra A, Schwabe RF. Mouse models of liver fibrosis. Methods Mol Biol. 2021;2299:339–356. doi: 10.1007/978-1-0716-1382-5_23. [DOI] [PubMed] [Google Scholar]
- 28.Puche JE, Saiman Y, Friedman SL. Hepatic stellate cells and liver fibrosis. Compr Physiol. 2013;3:1473–1492. doi: 10.1002/cphy.c120035. [DOI] [PubMed] [Google Scholar]
- 29.Yamaguchi T, Yoshimura T, Ohara T, Fujisawa M, Tong G, Matsukawa A. PolyI:C suppresses TGF-β1-induced Akt phosphorylation and reduces the motility of A549 lung carcinoma cells. Mol Biol Rep. 2021;48:6313–6321. doi: 10.1007/s11033-021-06625-1. [DOI] [PubMed] [Google Scholar]
- 30.Zhang X, Zhao F, Zhao JF, Fu HY, Huang XJ, Lv BD. PDGF-mediated PI3K/AKT/beta-catenin signaling regulates gap junctions in corpus cavernosum smooth muscle cells. Exp Cell Res. 2018;362:252–259. doi: 10.1016/j.yexcr.2017.11.025. [DOI] [PubMed] [Google Scholar]
- 31.Huang WC, Chen CC. Akt phosphorylation of p300 at Ser-1834 is essential for its histone acetyltransferase and transcriptional activity. Mol Cell Biol. 2005;25:6592–6602. doi: 10.1128/MCB.25.15.6592-6602.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shen F, Zhuang S. Histone acetylation and modifiers in renal fibrosis. Front Pharmacol. 2022;13:760308. doi: 10.3389/fphar.2022.760308.d6057582fad847759123626466b7680b [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li M, Zheng Y, Yuan H, Liu Y, Wen X. Effects of dynamic changes in histone acetylation and deacetylase activity on pulmonary fibrosis. Int Immunopharmacol. 2017;52:272–280. doi: 10.1016/j.intimp.2017.09.020. [DOI] [PubMed] [Google Scholar]
- 34.Mao L, Liu L, Zhang T, Wu X, Zhang T, Xu Y. MKL1 mediates TGF-β-induced CTGF transcription to promote renal fibrosis. J Cell Physiol. 2020;235:4790–4803. doi: 10.1002/jcp.29356. [DOI] [PubMed] [Google Scholar]
- 35.Hu X, Xu Q, Wan H, et al. PI3K-Akt-mTOR/PFKFB3 pathway mediated lung fibroblast aerobic glycolysis and collagen synthesis in lipopolysaccharide-induced pulmonary fibrosis. Lab Invest. 2020;100:801–811. doi: 10.1038/s41374-020-0404-9. [DOI] [PubMed] [Google Scholar]
- 36.Zeng M, Zhang X, Xing W, Wang Q, Liang G, He Z. Cigarette smoke extract mediates cell premature senescence in chronic obstructive pulmonary disease patients by up-regulating USP7 to activate p300-p53/p21 pathway. Toxicol Lett. 2022;359:31–45. doi: 10.1016/j.toxlet.2022.01.017. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.