Abstract
Background
Lennox–Gastaut syndrome (LGS) is a severe developmental and epileptic encephalopathy characterized by drug-resistant epilepsy with multiple seizure types starting in childhood, a typical slow spike-wave pattern on electroencephalogram, and cognitive dysfunction.
Methods
We performed a systematic literature review according to the PRISMA guidelines to identify, synthesize and appraise the burden of illness in LGS (including “probable” LGS). Studies were identified by searching MEDLINE, Embase and APA PsychInfo, Cochrane’s database of systematic reviews, and Epistemonikos. The outcomes were epidemiology (incidence, prevalence or mortality), direct and indirect costs, healthcare resource utilization, and patient and caregiver health-related quality of life (HRQoL).
Results
The search identified 22 publications evaluating the epidemiology (n = 10), direct costs and resource (n = 10) and/or HRQoL (n = 5). No studies reporting on indirect costs were identified. With no specific ICD code for LGS in many regions, several studies had to rely upon indirect methods to identify their patient populations (e.g., algorithms to search insurance claims databases to identify “probable” LGS). There was heterogeneity between studies in how LGS was defined, the size of the populations, ages of the patients and length of the follow-up period. The prevalence varied from 4.2 to 60.8 per 100,000 people across studies for probable LGS and 2.9–28 per 100,000 for a confirmed/narrow definition of LGS. LGS was associated with high mortality rates compared to the general population and epilepsy population. Healthcare resource utilization and direct costs were substantial across all studies. Mean annual direct costs per person varied from $24,048 to $80,545 across studies, and home-based care and inpatient care were significant cost drivers. Studies showed that the HRQoL of patients and caregivers was adversely affected, although only a few studies were identified. In addition, studies suggested that seizure events were associated with higher costs and worse HRQoL. The risk of bias was low or moderate in most studies.
Conclusions
LGS is associated with a significant burden of illness featuring resistant seizures associated with higher costs and worse HRQoL. More research is needed, especially in evaluating indirect costs and caregiver burden, where there is a notable lack of studies.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13023-023-02626-4.
Keywords: Burden of illness, Direct costs, Indirect costs, Caregiver burden, Health-related quality of life, Developmental and epileptic encephalopathy, Epilepsy, Lennox–Gastaut syndrome, Seizures
Background
The recognition of a specific disease entity is vital for every aspect of patient care, from diagnosis, management and treatment to advancements in health outcomes through research and health policy (Fig 1). The definition of Lennox–Gastaut syndrome (LGS) has been evolving since Lennox and Davis (1950) [1] and Gastaut et al (1960) [2] described the electroclinical features of a severe, refractory, childhood epileptic encephalopathy with slow spike and wave electroencephalogram (EEG) formations (Fig 1A). Studies elucidating LGS over the years have established a triad of classical features that can be used in clinical practice and research [3–5] (Fig 1B): 1. Multiple types of drug-resistant seizures, including tonic, atonic, and atypical absences, with onset generally before the age of 8 years (peak age of onset between 3 and 5 years). Tonic seizures and atypical absences are mandatory for diagnosis. 2. Interictal EEG pattern of diffuse, slow spike-wave complexes; and 3. developmental delay. However, importantly, it has also become clear that there are caveats to these features which can make LGS challenging to define and diagnose [5]. For example, none of the 3 features alone is pathognomonic, the main seizure types are not always present, and the features and progression of LGS is variable [6–11]. In addition, LGS is associated with multiple etiologies, as opposed to the known monogenetic causes that can be diagnostic for other epilepsy syndromes such as Dravet syndrome (DS) and tuberous sclerosis complex (TSC). Indeed, LGS can often arise from another severe infantile epilepsy syndrome including infantile spasms syndrome, early infantile developmental and epileptic encephalopathy, epilepsy of infancy with migrating focal seizures and TSC [12].
Fig 1.
Timeline to show A the history of the characterization and treatment of LGS B the triad of symptoms characteristic of LGS and the clinical implications of the characterization of the syndrome. From: Lennox et al (1950) [1], Gastraut et al (1966) [2], Niedermeyer (1969) [90], Beaumanoir (1985) [91], ILAE 1989 [92], Engel (2001) [93], Arzimanoglou et al (2009) [3], Scheffer (2017) [94], World Health Organization (ICD-11) (2022) [64]. CLB, clobazam; CBD, cannabidiol; EEG, electroencephalogram; FFA, fenfluramine; FLB, felbamate; ILAE, International League Against Epilepsy; LGS, Lennox–Gastaut syndrome; LTG, lamotrigine; RUF, rufinamide; TPM, topiramate
Despite the challenges of defining LGS, there have been significant clinical improvements over the years [5, 13–16]. There are now seven anti-seizure medications (ASMs) specifically indicated in patients with LGS in the US [15]—clobazam, felbamate, lamotrigine, topiramate, rufinamide, cannabidiol and fenfluramine, with efficacy demonstrated in clinical trials for seizures associated with drops in LGS patients (Fig 1A) [17–23]. Other interventions include the ketogenic diet, vagus nerve stimulation, and corpus callosotomy [5, 13–16].
The term “burden of illness” encompasses outcomes including the epidemiology (prevalence/incidence and mortality), healthcare resource utilization, costs, and patient and caregiver health-related quality of life (HRQoL) [24, 25]. In addition to clinical research investigating therapies, evaluating the burden of illness is key to understanding the challenges and needs of patients and their caregivers, together with understanding the impact and challenges on healthcare systems and society. Systematic literature reviews (SLRs) evaluating the burden of illness in DS and TSC have recently been published [25, 26]. However, there are no SLRs on this topic for LGS.
We conducted an SLR with a narrative synthesis to comprehensively identify, synthesize and appraise the evidence associated with the burden of illness in individuals with LGS, as well as to identify gaps in the evidence. The focus of this SLR was on the overall burden as opposed to the impact of specific interventions.
Methods
We carried out an SLR following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [27]. The protocol was registered with Prospero (CRD42022333410).
Literature search
We searched MEDLINE, Embase and APA PsychInfo (all via OVID), Cochrane’s database of systematic reviews (CDSR, Wiley), and Epistemonikos. The search strategy took the following form: ((terms for Lennox–Gastaut syndrome) AND (modified search filters for epidemiology, resource use, and health-related quality of life)) and it was developed by an information specialist and checked by the authors [28–31]. Conference proceedings were searched via Embase and we checked the reference lists of eligible studies and relevant systematic reviews [32]. The search was not limited by language and it was reported in web-only material using a search narrative [33]. The search terms used to identify the relevant literature can be seen in the Additional file 1: (Supplementary information: Search terms). The databases were searched from inception to 10th September 2021.
Eligibility criteria
A summary of the eligibility criteria for this review according to population, intervention, comparator, outcomes and study design (PICOS) is presented in Table 1. Eligible studies were those that reported original data on the above outcomes without the impact of an intervention. In vitro and in vivo studies, preclinical studies, review articles, SLRs, editorials, and case studies were not eligible, however the reference lists of reviews and SLRs were searched to ensure all relevant studies were captured. In addition, economic evaluations (e.g cost-effectiveness, cost-utility and budget impact models) reporting on specific interventions without any original data on e.g HRQoL (e.g utilities) and healthcare resource utilization were excluded, however the reference lists were searched for publications containing original data. No limits were applied (e.g regarding publication dates or language), except for conference abstracts that were limited to the previous 3 years.
Table 1.
PICOS criteria for inclusion and exclusion of studies
| PICOS | Inclusion | Exclusion |
|---|---|---|
| Population | Patients with LGS, including clearly defined “probable” LGS | Other DEEs |
| Intervention | Studies assessing the overall burden of illness of LGS without the impact of an intervention | Studies assessing the impact of interventionsa |
| Comparator | N/A | – |
| Outcomes |
Prevalence/incidence Economic burden Direct and indirect costs Resource use Patient and caregiver HRQoL and broader patient-reported outcomes Mortality |
– |
| Study designs/publication type |
Publications reporting original data, including: Healthcare insurance claims data Chart reviews Registry studies Electronic healthcare databases studies Epidemiologic surveys Observational studies Economic evaluations if they contain original data on resource utilisation and HRQoL |
In vitro and in vivo studies Preclinical studies Case reports and case series Clinical studies reporting only efficacy and safety data Economic evaluations reporting on specific interventionsb Reviewsc, SLRsc, comments, letters, editorials and press releases |
| Limits | Searches of conferences in Embase were limited to the previous 3 years (conferences 2018-current)d | None |
HRQoL, health-related quality of life; LGS, Lennox–Gastaut syndrome; N/A, non-applicable; SLR, systematic literature review
aThe focus is on the overall burden as opposed to the impact of specific interventions
bEconomic evaluations reporting on specific interventions without any original data on e.g HRQoL (utilities) and healthcare resource utilisation will be excluded, however, the reference lists will be searched to ensure all relevant studies are captured in this SLR.
cReviews and SLRs will be excluded from final inclusion but the reference lists will be searched to ensure all relevant studies are captured in this SLR.
dThe rationale for excluding conference abstracts > 3 years is that this should be sufficient time for the data to be published in peer-review journals if they are of sufficient importance and quality. The rationale for including conference abstracts ≤3 years is to include the most up-to-date data.
Study selection
Publications identified via the bibliographic database searches were managed and screened in Covidence, which is an online tool that helps to facilitate the systematic review process [34]. To ensure the accuracy of the screening process, two independent reviewers assessed relevant articles. Initially, primary screening was performed by reviewing the title and abstract and articles were excluded if they were clearly not relevant according to the PICOS eligibility criteria (e.g if the article was about a different condition and clearly not about patients with LGS etc). For all potentially relevant articles, full articles were obtained, and secondary screening was performed by reviewing the full text against the PICOS criteria. The reasons for exclusion at secondary screening were clearly documented according to the PICOS criteria. Disagreements regarding the inclusion and/or exclusion of studies at both primary and secondary screening were generally resolved by discussion between the two reviewers, or if needed by a third reviewer.
Data extraction and synthesis
Data were extracted into tables in MS Word by one reviewer, which was checked for accuracy by a second independent reviewer. The prespecified variables were as follows: Study and population characteristics i.e study type, observation period, population definition and number, age (mean, standard deviation [SD], median, range), percentage of male patients, and outcomes reported; epidemiology outcomes i.e incidence, prevalence and mortality as reported by the authors; healthcare resource utilization (mean and SD per person per year [PPPY]) as reported by the authors including inpatient, outpatient, home nursing, equipment, physiotherapy, medications; annual hospitalisation rates; length of hospital stay; anti-seizure medication use; direct costs i.e total direct costs and a breakdown of costs for individual healthcare resources as reported by the authors (mean and SD PPPY); indirect costs (e.g costs from loss of productivity). In addition, quantitative HRQoL measures for patients or caregivers (measure used, patient or caregiver and value) and qualitative HRQoL studies were described descriptively. Data were extracted for patients with LGS, LGS subpopulations (patients with and without seizures and with and without rescue medication) and control populations used by the authors (e.g age matched non-LGS patients).
During the development of the protocol (Prospero: CRD42022333410), it was anticipated that the studies identified in this SLR would be diverse with regard to study design, definition of LGS (especially as there is no specific ICD-9 or 10 code for LGS in some regions and diagnosis can be challenging), size of the populations, the ages of the patients and the length of the follow-up period, and therefore the synthesis of the data was planned to be narrative in nature, without any statistical analyses/comparisons (e.g meta-analyses). Costs were extracted as given in the source. Summaries of the costs across the different studies were provided in the narrative synthesis in US dollars ($) using a simple conversion from Euros (€) to US dollars at an exchange rate of €1.00 Euro =$1.06 (xe.com on 21st June 2022).
Quality assessment/risk of bias
The quality assessment of the prevalence studies was performed using a tool developed by Hoy et al. [35]. The quality of the cost of illness studies was assessed using a tool from the British Medical Journal Checklist for economic submissions [36] that was adapted by Molinier et al. [37]. The quality assessment of the qualitative and quantitative HRQoL studies was conducted using the Critical Appraisal Skills Programme, CASP; https://casp-uk.net/ that was adapted by Gallop et al. [38].
Results
Search results
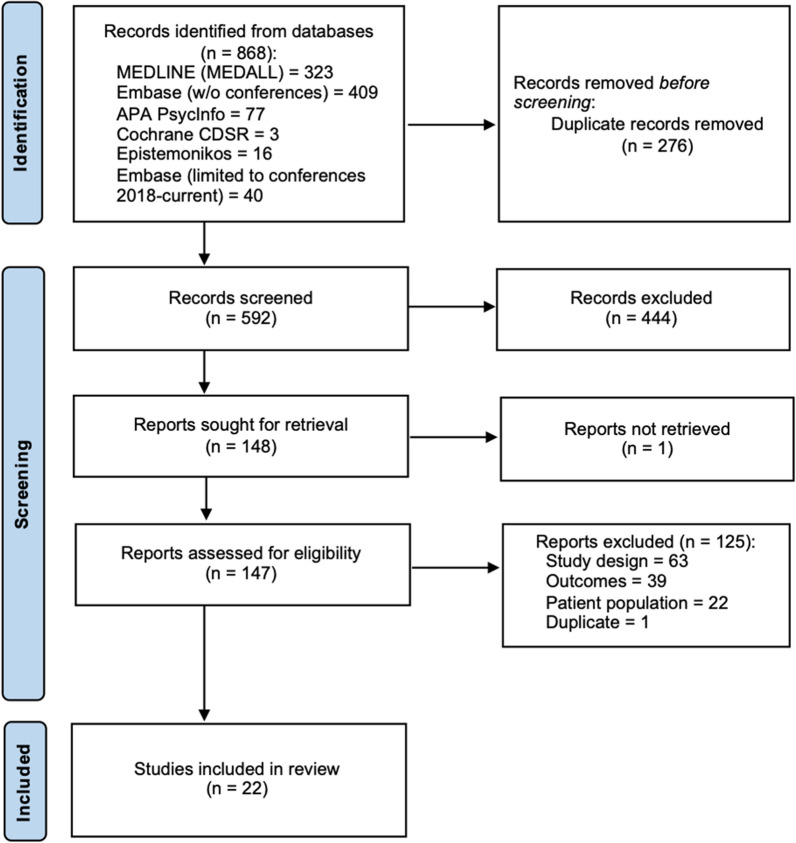
The electronic database searches initially identified 868 articles, of which 595 were screened after the removal of duplicates. After the primary screening, a total of 149 articles were retrieved for secondary screening (full-text assessment), and of these, 22 met the eligibility criteria (Fig 2). The study characteristics are summarized in Table 2, epidemiology data are summarized in Table 3 and the costs and resource data are presented in Additional file 2: Table S1. The results of the quality assessments/risk of bias are presented in Additional file 2: Tables S2–5.
Fig 2.
PRISMA flow diagram.
Adapted from Page et al (2021) [95]
Table 2.
Study characteristics
| Study, year (Publication type) Country Database | Study type and source of data | Time period | Population: Number | Definition of LGS | Characteristics: Age, mean (SD; median [range]), years; Male, % | Outcomes |
|---|---|---|---|---|---|---|
| Burden-of-illness/costs and resource | ||||||
|
Piña-Garza [46] (Full) USA MEDLINE and Embase |
Retrospective insurance claims analysis: Medicaid multi-state database of six states | Observation period: mean (SD): 11.1 (4.5) years (1997/98–2012/13) |
Probable LGS: N=14,712 |
≥2 medical claims ≥30 days apart for specified epilepsy or unspecified epilepsy (ICD-9-CM codes) Machine-learning technique based on a number of clinical variables (including use of rufinamide during the testing & training period) |
16.3 (15.7); 52.5% male |
Epidemiology Resource Direct costs |
|
Strzelczyk 2021 [47](Full) Germany MEDLINE, Embase and APA PsycInfo |
Retrospective insurance claims analysis: Vilua Healthcare research database, entries for >4 million people i.e ~5% of the German population | 10 years (2007–2016) |
Broad LGS: N=545 Narrow LGS: N=102 |
Broad definition: Algorithm considering: ≥1 ICD-10 diagnosis of G40 (epilepsy)/G41 (status epilepticus) and ≥1 claim of either rufinamide or felbamate (during the year of identification), OR ≥2 different ASMs in combination with ≥1 developmental delay diagnosis and no diagnosis code for competing etiologies Narrow definition: ≥1 documented G40/G41 claim before their 6th birthday |
Broad LGS: 31.4 (2–89); 53% male Narrow LGS: 7.4 (2–14); 52% male |
Epidemiology Mortality Resource Direct costs |
|
Chin 2021 [40] (Full) UK MEDLINE and Embase |
Retrospective analysis using electronic medical records from healthcare databases: Primary care data (CPRD) linked to secondary care data (HES), and general population mortality data (ONS) |
~31 years (1987–2018) Follow-up: mean (SD; median [range]): 11.7 (8.2; 9.6 [0.2; 32.2]) years |
Confirmed LGS: N=110 Probable LGS: N=146 Full cohort: N=256 |
Confirmed LGS: CPRD read code for LGS Probable LGS: ICD-10 code/ CPRD read code for epilepsy Rufinamide within a year of diagnosis |
Confirmed LGS: 7.0 (10.0; 3.5 [0–61]); 66% male Probable LGS: 8.9 (11.0; 4 [0–54]); 56% male Full cohort: 8.1 (10.6; 4 [0–61]); 60% male |
Epidemiology Mortality Resource |
|
Reaven 2018 [54] (Full) (LGS vs controls) USA MEDLINE, Embase and APA PsycInfo |
Retrospective insurance claims analysis: Truven Health Analytics MarketScan® Research Databases, entries for ~60 million people with commercial or Medicaid insurance coverage |
2010–2015 |
Probable LGS: Commercial: N=2270 Medicaid: N= 3749 |
A diagnosis code for refractory epilepsy and A diagnosis code for developmental delay/intellectual disability and A prescription for at least one selected ASM. Excluded patients with diagnosis codes for conditions suggestive of other etiologies Modified version of the algorithm developed by Piña-Garza et al [46] (above) |
Commercial: 13 (9.8 [0–62]); 53.0% male Medicaid: 13 (10.5 [0–60]); 52.9% male |
Resource Direct costs |
|
Reaven 2019 [53] (Full) (LGS vs other DEEs; seizure events vs no events) USA MEDLINE, Embase and APA PsycInfo |
Probable LGS: Commercial: N=2269 Medicaid: N= 3730 |
Commercial: 13 (9.8 [0–62]); 53.0% male Medicaid: 13 (10.5 [0–60]); 52.8% male |
||||
|
Stockl 2019 [56] (Abstract) (Hospitalizations) USA Embase |
Probable LGS: Commercial: N=2520 Medicaid: N=4613 |
As above, but also patients with an acute inpatient hospitalization (≥1 day LOS) | NR | Resource | ||
|
Stockl 2019 [55] (Abstract) (Commercial; Costs & Resource) USA Embase |
Retrospective insurance claims analysis: IBM® MarketScan® Commercial & Medicaid databases | 2015–2016 |
Probable LGS: Commercial: N=1296 |
≥1 ASM claim and medical claims with ≥1 diagnosis code for LGS or refractory epilepsy or ≥1 claim for clobazam or rufinamide. The LGS cohort required a diagnosis code for LGS or all of the following: oooooo≥1 diagnosis code for refractory epilepsy oooooo≥1 diagnosis code for intellectual disability/developmental delay, ooooooNo diagnosis codes that preclude LGS |
Commercial: 16.5; NR |
Epidemiology Resource (ASM use only) Direct costs |
|
Hollenack 2019 [52] (Abstract) (Medicaid; Costs & Resource USA Embase |
2014–2015 |
Probable LGS: Medicaid: N= 5186 |
Medicaid: 18.3; 58.2% male | |||
|
Hollenack 2019 [41] (Abstract) (prevalence) USA Embase |
Commercial: 2016– 2017 Medicaid: 2016 |
Probable LGS: Commercial: N=2273 Medicaid: N=4786 |
NR | |||
|
François 2017 [51] (Full) USA MEDLINE |
Retrospective insurance claims analysis: Truven Health MarketScan® claims databases, entries for >40 million people with commercial or Medicaid insurance coverage | 2010–2014 |
LGS pre-clobazam initiation Commercial/medicare: N=1384 Medicaid: N=1365 |
≥2 medical claims with a diagnosis of generalized convulsive or non-convulsive epilepsy ≥30 days apart, or ≥1 medical claim with a diagnosis of generalized convulsive epilepsy and ≥1 medical claim with a diagnosis of non-convulsive epilepsy (≥30 days apart (ICD-9-CM codes) ≥1 of the epilepsy diagnosis codes in the primary position ≥1 medical claim with a diagnosis for developmental disorder or cognitive impairment (ICD-9-CM codes) |
Commercial/medicare: 31.0 (23.2); 49.3% male Medicaid: 24.1 (17.9); 51.8% male |
Resource Direct costs |
|
Umeno 2019 [57] (Full) Japan Embase |
Retrospective study on the use of the welfare system in children with DEEs in Japan | 2015–2016 | LGS: N=30 | Children aged 0 to 16 years diagnosed at the Department of Child Neurology, Okayama University Hospital | NR | Resource (welfare system) |
| Epidemiology alone | ||||||
|
Trevathan 1997 [44] (Full) MADDS study USA MEDLINE and Embase |
Population-based, cross-sectional study: (MADDS): a multiple-source, EEG laboratory-based, case ascertainment system across Atlanta |
1975–1977 | Children with LGS: N=23 |
Epilepsy Two or more seizure types that included tonic, atonic, atypical absence, and/or myoclonic seizures with multiple falls Interictal EEG that demonstrated slow spike-wave discharges |
NR; 73.9% male | Prevalence |
|
Autry 2010 [48] (Full) MADDS study USA |
1975–2001 | LGS: N=34 | NR; NR | Mortality | ||
|
Sidenvall 1996 [43] (Full) Northern Sweden MEDLINE and Embase |
Observational study: Active epilepsy assessed in children aged 0–16 years in an area of northern Sweden with ~ 250,000 inhabitants (~50,000 children) | 1985–1986 | LGS: N=9 | Diagnosed using standard ILAE criteria | NR; NR | Prevalence |
|
Rantala 1999 [42] (Full) Finland MEDLINE and Embase |
Retrospective study: Children seen at the Department of Pediatrics University of Oulu | 1976–1993 | LGS: N=25 |
At least two types of the most common seizure types (tonic-axial, atonic, and absence seizures) Slow spike waves |
13.0 [6.5–17.9]; 56% male | Prevalence and incidence |
|
Heiskala 1997 [45] (Full) Finland MEDLINE and Embase |
Retrospective study: Health records from the Helsinki metropolitan area & the province of Uusimaa | 1975–1985 | LGS: N=75 |
A minimum of two types of typical though nonspecific epileptic seizures (e.g. astatic seizures, tonic seizures during sleep, and atypical absence seizures) Mental deficiency Bursts of diffuse slow spike-waves in the EEG |
NR; NR | Incidence |
|
Beilmann 1999 [39] (Full) Estonia MEDLINE |
Observational study: Patients with epilepsy seen at the Tartu University Hospital, sourced through multiple methods | 1995 –1997 | NR | Diagnosed using standard ILAE criteria | NR; NR | Prevalence |
| HRQoL | ||||||
|
Auvin 2021 [61] (Full) UK and France |
Observational study: On-line surveys | 2018–2019 | Patients and/or caregivers of individuals with DEEs estimated a patient’s health state from vignettes of hypothetical patients | NA | NA; NA | Patient HRQoL: quantitative (VAS) |
|
Radu 2019 [62] (Abstract) NR Embase |
Observational study: On-line surveys | 2018 | NA | NA; NA | Patient HRQoL: quantitative (VAS) | |
|
Gallop 2010 [58] (Full) US, UK & Italy MEDLINE, Embase and APA PsycInfo |
Observational study: Open-ended interviews and HRQoL instruments | NR |
Parents of patients with LGS recruited through support groups and websites (UK and US) and through clinicians (Italy): N=40 |
NR | 12 (4–43); 63% male |
Caregiver HRQoL: Quantative (SF-36v2, HADS) and qualitative Patient HRQoL: Qualitative |
|
Murray 1993 [60] (Full) Australia MEDLINE, Embase and APA PsycInfo |
Observational study: Questionnaires & in-depth interviews | NR | Parents of patients with LGS who attended a self-help group: N=41 | NR | 13 (3–34); NR | Caregiver HRQoL: Qualitative |
|
Gibson 2014 [59] (Full) USA MEDLINE, Embase and APA PsycInfo |
Observational study: General experience and parent survey | NR | Parents of patients with LGS who had contacted a support hotline: N=96 surveys returned | NR | [3.5–36 years]; NR | Caregiver HRQoL: Qualitative |
ASM anti-seizure medication; CPRD Clinical Practice Research Datalink; DEE, developmental and epileptic encephalopathy; DS, Dravet syndrome; HADS, Hospital Anxiety and Depression Scale; HES, Hospital Episode Statistics; HRQoL, health-related quality of life; ILAE, International League Against Epilepsy; LGS, Lennox–Gastaut syndrome; LOS, length of stay; NA, non-applicable; MADDS, Metropolitan Atlanta Developmental Disabilities Study; NR, not reported; ONS, Office for National Statistics; SD, standard deviation; VAS, visual analogue scale
Table 3.
Epidemiology: Incidence/prevalence and mortality in patients with LGS
| Study | Country | Study type | Year | Population (n) | Incidence/prevalence | Mortality | |
|---|---|---|---|---|---|---|---|
| Parameter | Value | ||||||
|
Piña-Garza 2017 [46] (Full) |
USA | Retrospective claims analysis | 2012/13 |
Probable LGS patients N=14,712 |
Proportion of epilepsy patients with probable LGS (10-year age groups) | 8.4% at age 10 decreasing to 2% in the 60-year-old cohort | NR |
|
Strzelczyk 2021 [47] (Full) |
Germany | Retrospective claims analysis | 2016 |
Broad LGS N=545 Narrow LGS N=102 |
Prevalence (standardized to German GKV population) |
Broad LGS: 39.2 per 100,000 people Narrow LGS: 6.5 per 100,000 people |
Broad LGS: 10.01% [157 events] Narrow LGS: 2.88% [six events] (p < 0.001 vs control) Control: 0.01% [one event] |
|
Chin 2021 [40] (Full) |
UK | Retrospective analysis of electronic medical records | 2017 |
Confirmed LGS: N=74 for prevalence Probable LGS: N=106 for prevalence Full cohort: N=180 for prevalence; N=122 for mortality |
Prevalence (standardized to UK CPRD population) |
Confirmed LGS: 2.89 per 100,000 people* Probable LGS: 4.20 per 100,000 people* Full cohort: 5.78 per 100,000 people* |
Crude mortality rate per 1000 person-years: Confirmed LGS: 6.12 [11 events] Probable LGS: 4.17 [7 events] Full cohort: 18 events |
|
Hollenack 2019 [41] (Abstract) |
USA | Retrospective claims analysis | 2016/ 2017 |
Probable LGS: Commercial: N=2273 Medicaid: N=4786 |
Prevalence |
Commercial: 13.0 per 100,000 people* Medicaid: 60.8 per 100,000 people* |
NR |
|
Trevathan 1997 [44] MADDS study (Full) |
USA | Population-based, cross-sectional study | 1975–1977 | LGS: N=23 |
Prevalence of LGS at age 10 years Proportion of epilepsy patients with LGS |
26 per 100,000 people* 4% of epilepsy patients |
NR |
|
Autry 2010 [48] MADDS study (Full) |
Original cohort: 1975–1977 Follow-up: 1975 to 2001 |
LGS: N=34 | NR | NR | Mortality ratio (95% CI): 13.92 (7.19-24.31) for LGS vs 3.11 (2.39-3.98) for all epilepsy | ||
|
Sidenvall 1996 [43] (Full) |
Northern Sweden | Observational | 1985 | LGS: N=9 |
Prevalence N (%) of epilepsy patients with LGS |
20 per 100,000 people* n=9 (5.8%) of all epilepsy cases |
NR |
|
Rantala 1999 [42] (Full) |
Finland | Retrospective | 1976–1993 | LGS: N=25 | Prevalence | 28 per 100,000 people (95% CI: 18–41)* | NR |
| Annual incidence |
1.93 per 100,000 children <15 years (95% CI: 1.25-2.85/100,000) |
||||||
|
Heiskala 1997 [45] (Full) |
Finland | Retrospective | 1975–1985 |
LGS (broad definition): N=75 LGS children 0–14 years: N=NR |
Annual incidence |
Broad definition: 2.1 per 100,000 people Children 0–14 years: 1.9 per 100,000 |
NR |
|
Beilmann 1999 [39] (Full) |
Estonia | Observational | 1997 | LGS: N=NR | Prevalence | 10 per 100,000 people* | NR |
CI confidence interval; CPRD UK Clinical Practice Research Datalink; GKV German population covered by statutory health insurance (Gesetzliche Krankenversicherung; GKV); NR not reported
*Converted to per 100,000 people
Epidemiology: incidence/prevalence and mortality
Overall, 9 studies (10 publications) reported on the epidemiology of LGS (Table 3). Prevalence, reported in 7 studies, varied from 4.2–60.8 per 100,000 people across studies for probable LGS and 2.9–28 per 100,000 people for a confirmed/narrow definition of LGS [15, 39–44]. Incidence, reported in 2 studies, was estimated at approximately 1.9 per 100,000 people in both studies [42, 45]. In addition, 3 studies reported on the proportion of LGS among epilepsy patients, varying from 4 to 8.4% during childhood [43, 44, 46] [42, 45]. The risk of bias in the studies reporting on prevalence/incidence was low in 3 studies [40, 41, 47] and moderate in 5 studies [39, 42–45] (Additional file 2: Table S2).
Trevathan et al (1997) reported the lifetime prevalence of LGS at the age 10 years to be 26 per 100,000 people (0.26 per 1000 people) [44]. The study used data from the Metropolitan Atlanta Developmental Disabilities Study (MADDS) that encompassed children aged 10 years who were born from January 1, 1975, through December 31, 1977; children with LGS were identified via multiple sources including public schools, special education programs, hospitals, selected physicians’ offices, and EEG laboratories [44]. More recent studies (2019–2021) have used databases covering large populations including claims databases in the USA [41] and Germany [47] and the Clinical Practice Research Datalink (CPRD) database of health records in the UK [40]. These studies have estimated the prevalence of probable LGS to be 13.0–60.8 per 100,000 people in the USA [41], 39.2 per 100,000 people in Germany [47], and 4.2 per 100,000 people in the UK [40]. Using a more stringent definition of LGS resulted in prevalence rates of 6.5 per 100,000 people in Germany [47] and 2.89 per 100,000 people in the UK [40]. Other older, smaller population-based studies have reported the prevalence of LGS to be 10 per 100,000 people in Estonia [39], 2.1–28 per 100,000 people in Finland [42, 45], and 20 per 100,000 people in Sweden [43]. While LGS is a rare syndrome, Piña-Garza et al. [46] reported that LGS accounts for 8.4% of epilepsy cases at age 10 decreasing to 2% in the 60-year-old cohort in the USA. In addition, Trevathan et al (1997) (MADDS study) [44] reported that it accounted for 4% of childhood epilepsy cases, while the study in Sweden [43] found that among the childhood epilepsy syndromes, 5.8% of patients had LGS (Table 2).
Mortality was reported in 3 studies, although with different methods (mortality ratio, mortality rate as a percentage of patients who died divided by the total number of patients in the study, and mortality rate per 1000 person-years) [40, 47, 48]. Overall, the studies showed that mortality was substantially higher in LGS patients than in children with epilepsy and controls (Table 3) [40, 47, 48]. For example, using the MADDS data source, Autry et al. [48] reported a mortality ratio (ratio of observed-to-expected deaths) of 13.92 (95% confidence interval [CI] 7.19–24.31) for children with LGS versus 3.11 (95% CI 2.39–3.98) for all children with epilepsy over a 26-year follow-up period. The retrospective claims analysis from Germany reported a mortality rate (the percentage of patients who died divided by the total number of patients in the study) over the 10-year study period of 10.01% for patients according to the broad definition of LGS and 2.88% for the narrow definition versus 0.01% for the age- and sex-matched control population (p < 0.001) [47]. Using data from the CPRD (electronic health records) linked to mortality data from the Office of National Statistics (ONS) in the UK, Chin et al. [40] estimated the mortality rates to be 6.12 deaths per 1000 person-years for confirmed LGS and 4.17 deaths per 1000 person-years for probable LGS over a follow-up period of 1796 and 1679 patient-years, substantially higher than the mortality rates for the general population and the epilepsy population (0.6 per 1000 person-years in England in 2018 [49], and 0.9 per 1000 person-years in the UK [50], respectively).
Healthcare resource utilization and costs
Overall, 9 studies (9 publications), all published from 2017 to 2021, were identified that reported on resource utilization and/or direct costs; 7 reported on both resource utilization and direct costs [46, 47, 51–55] (6 from the USA and 1 from Germany), and 2 reported on resource utilization only (1 from the USA using insurance claims databases and 1 from the UK using electronic health records) [40, 56] (Table 2 and Additional file 2: Table S1). By country, most of the studies were from the USA (7 out of 9 studies). All of the costs studies were retrospective analyses of insurance claims databases and the mean annual direct costs were €22,787 in Germany (n = 1 study), and varied from $28,461 to $80,545 in the USA across studies (n = 6 studies). The risk of bias in the studies reporting on costs and/or resources was assessed to be low in 6 studies [40, 46, 47, 51, 53, 54] and moderate in 3 studies, which were conference abstracts that provided limited information [52, 55, 56] (Additional file 2: Table S3). No study reporting the indirect costs of patients with LGS or their caregivers was identified, although one paper reported that a large proportion of caregivers of patients with LGS struggled to access the available medical expense subsidies in Japan [57] (Table 2).
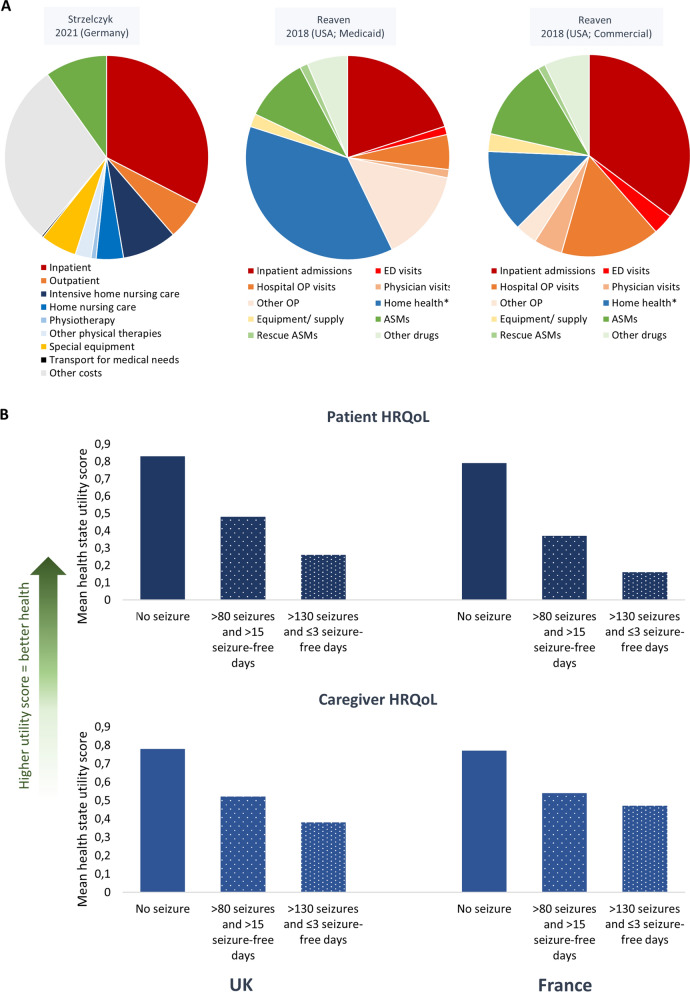
Total healthcare costs and healthcare resource utilization associated with LGS were substantially higher than matched controls [54], non-LGS epilepsy patients [46], and patients with other DEEs, including DS and TSC [52, 53, 55] (Fig 3A, Additional file 2: Table S1). For example, Reaven et al. [54] reported that, compared to matched controls, annual costs for services were 17 to 20 times higher for patients with possible LGS, and annual costs for drugs were 16 to 38 times higher (Fig 3B). In addition, among LGS patients, costs and healthcare resource utilization were reported to be higher in patients with a seizure event versus those without (Fig 3C) [53], and costs and hospitalizations/ length of stay (LOS) were higher in those prescribed rescue medication vs those not [47] (Additional file 3: Fig S1).
Fig 3.
A Total direct costs in LGS patients (blue) versus control patients (grey), patients with other epilepsies (lilac), and in LGS patients with seizure events vs without seizure events and LGS patients in years where patients were prescribed with rescue medication vs years where patients were not prescribed rescue medication (blue) B resource utilization and costs in LGS patients vs controls; C resource utilization and costs in LGS patients with seizure events vs without seizure events.
Adapted from: Reaven et al (2018) [54], Piña-Garza et al (2017) [46], Stockl et al (2019) [55], Hollenack et al (2019) [52], Reaven et al (2019) [53] and Strzelczyk et al (2021) [47] .* Costs were converted from Euros to US dollars on 21st June 2022 (exchange rate 1.00 Euro =1.06 US Dollars [xe.com]). ASM, anti-seizure medication; DS, Dravet syndrome; ED, emergency department; LGS, Lennox–Gastaut syndrome; PPPY, per person per year; OP, outpatient; TSC, tuberous sclerosis complex
The studies showed that patients with LGS require a multitude of healthcare resources including general/family practitioner visits, outpatient visits to the neurologist/epilepsy clinic, laboratory and radiology services, medication, physiotherapy/habilitation services and other physical therapies, inpatient and emergency department visits, as well as home-based nursing [40, 47, 51, 53, 54, 56] (Additional file 2: Table S1). Across the studies, inpatient care and home-based care were significant cost drivers, whereas pharmacy costs, particularly for ASMs, generally represented a smaller share of all healthcare costs (Fig 4A, Additional file 2: Table S1) [46, 47, 51–55]. In Germany the greatest contributors to healthcare costs were inpatient care (33%) especially hospital stays related to epilepsy, followed by home nursing care (including intensive home nursing) (13%), and medication (10%) (Fig 4A, Additional file 2: Table S1) [47]. ASMs represented only 14% of the overall medication costs despite their ubiquitous use. Similarly, Piña-Garza et al. [46] reported that the main contributor to healthcare costs was home-based care in children aged 1–18 years (mean costs of $12,396 to $18,360 PPPY across across different pediatric age groups) and long-term care in the 60-year-old cohort ($16,215 PPPY), whereas mean pharmacy costs encompassed a smaller proportion of the costs at $1592–$5630 PPPY across all age groups. In the study by Reaven et al. [54], the largest contributors to the healthcare costs in LGS patients were inpatient care in the commercial-insured cohort and home health services (home-based nursing, personal care, and residential habilitation services) in the medic aid-insured cohort (Fig 4A, Additional file 2: Table S1).
Fig 4.
A Cost drivers according to inpatient and emergency department admissions (red), outpatient visits (orange), home health (home nursing and physiotherapy; blue), equipment (yellow) and medication (green) costs (where reported) B Patient and caregiver HRQoL according to number of drop seizures per month and number of seizure-free days in the UK and France. Mean health state utility scores for a hypothetical patient with LGS and a hypothetical caregiver of a patient with LGS. A Adapted from: Strzelczyk et al (2021) [47] and Reaven et al (2018) [54]. Home health= home-based nursing, personal care, and residential habilitation services. B Adapted from Auvin et al (2021) [61]. ASM, anti-seizure medication; ED, emergency department; HRQoL, health-related quality of life; OP, outpatient
While the costs of ASMs may account for a relatively small proportion of the overall healthcare costs, data from several studies suggest that the burden of ASMs on patients and the overall pharmacy costs is high. For example, in a study based on IBM® MarketScan® medic aid database, the mean number of distinct ASMs during the 12-month study period averaged 2.4, 2.3, and 2.2, in probable LGS, probable DS, and other refractory epilepsies, respectively, accounting for 67.3%, 62.8%, 52.3% of the pharmacy costs [52]. Likewise, using the IBM® MarketScan® commercial database, the mean number of distinct ASMs was 3.4, 2.6, and 2.1, respectively, representing 72.6%, 70.8%, and 65.8% of the pharmacy costs [55]. In addition, Chin et al (2021) reported that in patients with confirmed LGS the mean number of ASMs was 6.7 (SD 3.4) during the 8–12 years follow-up period, with only 3.7% being prescribed only one ASM during this period, while the mean number of ASMs per year was 1.06 during 2010–13 and 1.12 during 2014–17 [40]; this suggests that patients had to switch to different ASMs many times over the course of the study period. Similarly, in the study in Germany, patients generally received 1–3 different ASM combinations each year (range: 1–9) and most patients received 2–4 different ASMs [range: 1–12] over the entire 10-year observable period [47]. Of note, the use of other drugs was also substantially higher in LGS patients than in control populations (Fig 2B) [54]. In both Germany [47] and the UK [40] the most commonly prescribed ASMs were valproate, lamotrigine, clobazam, oxcarbazepine (in Germany) and levetiracetam (in the UK); in the UK rufinamide was prescribed in 27% of patients with confirmed LGS. In the US, Pina-Garza et al. [46] reported LGS patients may be inadequately treated; the majority, but importantly not all probable LGS patients had ≥1 claim for an ASM (62.6–82.3% across age cohorts), while clobazam and rufinamide, both specifically licensed for LGS in the US, were infrequently used, especially in the older cohorts (17.5% for clobazam and 7.4% for rufinamide in patients ≤5 years, 5.5% for both ASMs in the 20-year cohort and <1.0% of patients in the 60-year cohort).
Regarding hospitalizations and LOS, in Germany there was a large variation between patients, with some being hospitalized many times (median hospitalization rate: 1 [range: 0–13 PPPY]) with a median LOS of 3 days (range: 0–804 days) [47]. In a study describing the nature of acute hospitalizations in LGS patients, 46–58% were considered epilepsy-related across health plans (Commercial and Medicaid) compared to 63–70% for DS, 26% for TSC, and 37–51% for other DEEs; 6–7% were pneumonia-related, and approximately 2% were injury-related [56]. Across the different DEEs, admission to the intensive care unit (ICU) accounted for 31% of all hospitalizations, and ICU use was associated with a longer LOS (mean 8.0 [SD 16.8] days vs. 4.0 [SD 7.9] days for non-ICU use). Furthermore, readmission rates were high, with approximately 9–10% of patients readmitted within the month and 42–45% within a year of discharge [56]. Concerning injuries, 67% of LGS patients reported at least one injury over the 10-year study period in Germany [47], while in the study by Reaven et al. [53], 22% (2329 of 10,618) of LGS patients with a medically-treated seizure event across insurance types had an injury.
Health-related quality of life
Five studies, published between 1993 and 2021, were identified that reported on the HRQoL of patients with LGS and/or their caregivers using qualitative and/or quantitative methods; one study reported on both caregiver and patient perspectives [58], 2 focused on caregivers [59, 60] and 2 on patients [61, 62] (Table 2). Three studies used qualitative methods (one for both patient and caregiver HRQoL, and two for caregiver HRQoL); the quality of the qualitative studies was assessed to be grade I (highest methodological and reporting quality) in one study and grade III (limitations in methodological and reporting quality) in 2 studies (Additional file 2: Table S4). Three studies used quantitative methods (one assessed caregiver HRQoL using the SF-36 and the HADS (Hospital Anxiety and Depression Scale), while two studies used a VAS (visual analog scale). The quality of the quantitative studies was assessed to be grade II (moderate-high methodological and reporting quality) in 2 studies and grade III in one study (Additional file 2: Table S5).
All three studies reporting on the HRQoL of caregivers found that LGS had substantial negative effects. Gallop et al. [58] investigated the HRQoL of parent caregivers (N = 40) both quantitively and qualitatively. Using the SF-36v2, a widely used generic instrument to measure health status, the parents’ mental health summary score (a composite of social functioning, vitality and mental health) was found to be below average (45 for LGS parents vs. 50 for US general population). In addition, 58% of parents experienced anxiety according to the HADS. Using semi-structured interviews with the parents, the authors determined that the parents were impacted physically, emotionally, socially and financially. Gibson [59] similarly reported that LGS had a significant physical, emotional, social and financial impact on parents, while siblings were also affected, frequently taking on a caregiver role themselves early in life. Furthermore, Murray reported in 1993 that parents had to deal with a lot of uncertainty around diagnosis, etiology, seizure activity, treatment and prognosis, which caused feelings of guilt and stress in some parents [60].
Gallop et al. [58] also investigated parents’ perceptions of how LGS impacted their children, describing the profound impact that the syndrome conferred on the daily lives of children with LGS; in particular, physical, social, cognitive and behavioral aspects of their lives were disrupted. Studies by Auvin et al. [61] and Radu et al. [62] used an alternative method of measuring patient HRQoL in a quantitative manner whereby patients and/or caregivers of patients with LGS, DS, or other epilepsies were asked to score the patient HRQoL based on hypothetical vignettes of patients with LGS (or DS) according to how many seizures and seizure-free days the hypothetical patient experienced. These studies have suggested that fewer seizures and additional seizure-free days are associated with better patient HRQoL, with seizure-free days having the greatest consequence on HRQoL (Fig 4B). Of note, this vignette-based method for estimating HRQoL focused specifically on the impact of seizure frequency/seizure-free days as opposed to a more holistic HRQoL assessment. The values were used in the NICE (National Institute for Health and Care Excellence) appraisal in the UK to inform the cost-effectiveness of cannabidiol for the adjuvant treatment of seizures associated with LGS [63].
Discussion
We conducted a comprehensive SLR following pre-specified inclusion/exclusion criteria to understand the burden of LGS, providing a descriptive summary of the literature while identifying gaps in the current knowledge. The epidemiology studies confirm that LGS is a rare syndrome, whereby the prevalence varied from 4.2 to 60.8 per 100,000 people across studies for probable LGS and 2.9 to 28 per 100,000 people for confirmed/narrow definition of LGS. Annual costs per patient and healthcare resource utilization were substantial across all studies, confirming the high economic burden associated with LGS. In addition, studies showed that the HRQoL of patients and caregivers was adversely affected, and seizure events were associated with higher costs and worse HRQoL. Furthermore, LGS was associated with high mortality rates compared to the general population and those with epilepsy in general.
The burden of illness studies relies heavily on how the disease is defined. However, for LGS there are inherent difficulties in using the currently available source data where there has been no specific code for LGS in the ICD coding in many regions. An extension code for LGS in the ICD-10-CM coding system (G40. 812) has only been available since 2015, and it is only used in some regions. The recent release of ICD-11 includes a specific code for LGS (8A62.1 Lennox–Gastaut syndrome) [64], however, it may be many years before the benefits are seen in real-world research, including the burden of illness studies, which require a sufficient number of patients and observation period. Without a precise definition of LGS and a lack of consistency in using recent and specific diagnostic codes for LGS, there was considerable heterogeneity between studies on how LGS was defined. The retrospective claims analysis studies generally used a “probable LGS” definition that included appropriate ICD-9 or ICD-10 codes for epilepsy and developmental delay and a prescription for a relevant ASM. Piña-Garza et al (2017) developed a machine-learning classification model to identify patients with LGS [46], which was subsequently used and modified for other studies [53, 54, 56]. The methods were latterly reported in more detail by Vekeman et al (2019), showing that an LGS classifier that used input variables including the number of distinct ASMs received, epilepsy-related outpatient/inpatient visits, electroencephalogram procedures and claims for delayed development, showed high sensitivity and specificity in identifying LGS patients [65]. On the other hand, Chin et al (2021) was able to circumvent the limitations of the ICD coding by using the electronic medical records from the CPRD in the UK that included a code for LGS (the confirmed LGS group) [40]. Other smaller retrospective, observational studies reporting on the epidemiology of LGS included patients who had been diagnosed based on standard diagnostic/ILAE criteria [39, 42–45, 48], while the HRQoL studies generally recruited caregivers and/or patients through support groups, patient associations, clinicians and/or websites [58–62]. Other areas of heterogeneity across the studies included the size of the populations, the ages of the patients and the length of the follow-up period. Another limitation is that LGS is likely to be underdiagnosed/ misdiagnosed, especially in adults [11, 14], and therefore many aspects of the burden of illness may have been underestimated.
Many of the studies identified are recently published, particularly those on costs and healthcare resources, which may reflect the development of new treatment options, whereby the data can be used to inform payers and health technology assessment agencies. We did not look at the impact of interventions on our outcomes. However, compared to standard of care, studies have estimated that rufinamide is a cost-effective treatment [66, 67], while cannabidiol was found to be cost-effective in a model that was based on absolute seizure frequency and seizure-free days and took account of the impact of caregivers’ burden [68], but in a different model, it was determined not to be cost-effective at a willingness-to-pay threshold of $150,000/ quality-adjusted life-year [69]. As with DS [70–73], studies have suggested that seizure events in LGS are associated with higher costs and worse HRQoL, and therefore it is hoped that current and future improvements in the treatment will help to alleviate some of the burdens on healthcare resources and costs and improve the HRQoL of patients and caregivers.
We found only a few studies evaluating the HRQoL in patients and caregivers. In a recent SLR describing the impact of a range of DEEs (including LGS) on the HRQoL of caregivers and the wider family unit Gallop et al. [58] identified only one study of sufficient quality regarding caregivers of patients with LGS (also identified hererin) [38]. However, across the DEEs, the authors found that most of the studies reported a negative impact on the HRQoL of caregivers, and they highlighted the challenges of evaluating caregiver HRQoL, especially using generic instruments, because parents of young children with complex chronic diseases adapt and become habituated to their decrease in quality of life [38]. The same can apply to patients, although measuring HRQoL in patients with LGS generally requires a parent proxy because of the patient’s young age and/or cognitive dysfunction, as well as not knowing a time before their condition with which to compare to.
We identified no studies reporting on indirect costs, and therefore it is likely that the overall economic burden has been underestimated due to studies only reporting on direct costs. Indeed, the financial burden among patients and caregivers has also been little studied across other DEEs including DS and TSC [25]. However, the few studies that are available on DS caregivers and TSC patients or caregivers have shown that indirect costs are relevant [72, 74–76], with a particular impact on mothers having to reduce their working hours or stop working completely [72, 74, 77]. Overall, evaluating the caregiver burden including indirect costs, productivity losses and HRQoL requires complex, preferably prospective, studies that may have been hindered by a lack of having large patient advocacy groups from which to recruit participants, as well as a lack of priority for health research funding, among other factors.
Strengths and limitations
As far as we are aware this is the first SLR on the burden of illness in LGS. The SLR has several strengths in that it was a comprehensive review performed according to the PRISMA guidelines, with no limitations on language or date (except for abstracts), whereby the search terms were developed by an experienced information scientist.
The main limitations have been discussed above, including the challenges around identifying the LGS population and the paucity of studies, especially on caregiver HRQoL and indirect costs. Furthermore, many of the studies, especially regarding costs and resources, were conducted in the USA, with limited evidence in Europe, and no evidence from other parts of the world despite not having any language restrictions. This country-bias may reflect the lack of research funding as described above, with perhaps some publication bias against non-native English speaking countries [78, 79]. The situation in low and middle income countries is especially complex and many aspects of the burden of illness, especially mortality, costs, and resources are likely to be very different from high income countries. For example, a study of the global burden of epilepsy (among other conditions) reported that 81% of epilepsy-deaths occurred in low and middle countries [80], reflecting both the larger number of people with epilepsy and the limited resources with which to effectively manage and treat patients [81]. Overall, there is an imperative need for innovative solutions to improve the outcomes of patients in these countries [82, 83].
We restricted the search specifically to LGS, excluding studies which encompassed refractory epilepsy in general, even if they had a few LGS cases within a more general population of refractory epilepsy patients, which may have limited our evidence base, especially as LGS can evolve from or be part of other epilepsy syndromes, e.g. TSC [84]. In addition, while the literature was searched in major electronic databases, we may still have missed some studies from other databases or from the grey literature. We did not identify any extra relevant studies when we searched the reference lists of other SLRs, reviews and economic evaluations suggesting that our methods, including the search terms and databases used, were robust.
Future research directions
In this SLR we have found that there is lack of studies reporting on indirect costs, and patient and caregiver HRQoL. These are extremely important and relevant outcomes for this complex syndrome that goes beyond seizures. In addition, there is a lack of studies on all aspects of the burden of illness in low and middle income countries, and more research is needed in these areas, possibly through enhanced collaboration with established research groups in high income countries and the pharmaceutical industry. Patient advocacy groups for LGS should be empowered across all countries, perhaps through the use of virtual events and an increase in on-line resources and communities.
Studies in this SLR have reported that seizure events are associated with higher resource and costs, and lower patient HRQoL. Cannabidiol and fenfluramine are the most recently approved therapies for the treatment of seizures associated with LGS (Fig 1). ASMs in clinical development include soticlestat and carisbamate. Soticlestat has recently successfully completed Phase 2 clinical deveopment as an adjunctive therapy in pediatric patients with DS or LGS [85], with Phase 3 trials underway (NCT04938427 and NCT05163314). A Phase 3 clinical trial assessing carisbamate in patients with LGS has also been initiated (NCT05219617). It will certainly be of interest in the future to determine the impact that these potential and new ASMs [89] have on the burden of illness in LGS, especially patient and caregiver HRQoL. Of concern though is the apparent lack of other therapies in development for patients with LGS at present.
Also of note is that LGS has multiple and varied etiologies, and with advances in neuroimaging and genetics the conditions within this syndrome are now largely defined and named by their etiology. These etiologies are a more precise guide to prognosis and management than the designation of LGS. William Lennox’s views on eugenics [87, 88] have resulted in his name being removed from named lectures in the USA. The International League Against Epilepsy (ILAE) decided to keep the term Lennox–Gastaut in the 2022 ILAE Syndromes Definitions arguing that this was because medications are licensed for LGS and that a better collective name had not been coined for DEE with slow spike and wave and tonic seizures in childhood [86]. However, the term Lennox–Gastaut may gradually be retired in the next decade.
Conclusions
LGS is associated with a substantial burden of illness, with seizure events associated with higher costs and worse HRQoL. However, with no ICD-10 coding for LGS available in many regions, several studies had to rely upon indirect methods to identify their patient populations. More research is needed, especially in evaluating the caregiver burden, including HRQoL, productivity losses and indirect costs, where there is a notable lack of studies.
Supplementary Information
Additional file 2. Supplementary Table S1: Healthcare resource utilization and costs in patients with LGS; Table S2: Quality assessment checklist for prevalence studies; Table S3: Quality assessment checklist of cost-of-illness studies; Table S4: Quality assessment checklist for qualitative HRQoL studies; Table S5: HRQoL: Quality assessment checklist for quantitative HRQoL studies.
Additional file 3. Supplementary Figure S1: Costs, annual hospitalization rate and length of stay (LOS) in LGS patients in years where patients were prescribed with rescue medication vs years where patients were not prescribed rescue medication.
Acknowledgements
Chris Cooper developed the search terms, and assistance with the literature screening and medical writing, under the direction of the authors, was provided by Amanda Prowse (Lochside Medical Communications Ltd) and Emma Melchor.
Abbreviations
- ASMs
Anti-seizure medications
- CI
Confidence interval
- CPRD
Clinical Practice Research Datalink
- DEE
Developmental and epileptic encephalopathy
- DS
Dravet syndrome
- EEG
Electroencephalogram
- HADS
Hospital Anxiety and Depression Scale
- HRQoL
Health-related quality of life
- ICU
Intensive care unit
- LGS
Lennox–Gastaut syndrome
- LOS
Length of stay
- MADDS
Metropolitan Atlanta Developmental Disabilities Study
- NICE
National Institute for Health and Care Excellence
- ONS
Office of National Statistics
- PICOS
population, intervention, comparator, outcomes and study design
- PRISMA
Preferred reporting items for systematic reviews and meta-analyses
- PPPY
Per person per year
- SD
Standard deviation
- SLR
Systematic literature review
- TSC
Tuberous sclerosis complex
Author contributions
AS and SSB developed the idea for this study and conceived the paper. AS, SZ, PS, FR and SSB wrote the paper, discussed the results, and contributed to the final manuscript. All authors read and approved the final manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. Medical writing assistance was funded by GW Pharma (Germany) GmbH.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
AS reports personal fees and grants from Angelini Pharma, Desitin Arzneimittel, Eisai, Jazz/GW Pharmaceuticals companies, Marinus Pharma, Precisis, Takeda, UCB, UNEEG medical, and Zogenix. SZ reports research support from Epilepsy Research UK, Scottish Government Digital Health & Care, UCB Pharma, Dravet Syndrome UK, Dravet Syndrome Foundation, Tenovus Foundation and Glasgow Children’s Hospital Charity. He or his institution have received honoraria for consultancy work, educational symposia and advisory boards from GW Pharma, Zogenix, UCB, Encoded Therapeutics, Takeda and Eisai. PS reports personal fees and grants from Angelini Pharma, Eisai, Jazz Pharmaceuticals Biomarin, UCB, Proveca, and Zogenix. FR reports personal fees from Angelini Pharma, Arvelle Therapeutics, Eisai GmbH, GW Pharmaceuticals companies, and UCB and grants from the Detlev-Wrobel-Fonds for Epilepsy Research, the Deutsche Forschungsgemeinschaft, the LOEWE Programme of the State of Hesse, and the European Union. SSB reports personal fees from Eisai, Desitin Pharma, GW Pharmaceuticals companies, Ethypharm, UCB, and Zogenix.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
11/4/2024
In table 2 age ranges were shown as references: Broad LGS: 31.4 [2–89]; 53% male Narrow LGS: 7.4 [2–14]; 52% male; 12 [4–43]; 63% male; 13 [3–34]; NR. It should be: Broad LGS: 31.4(2–89); 53% male; Narrow LGS: 7.4 (2–14); 52% male; 12 (4–43); 63% male; 13 (3–34); NR
References
- 1.Lennox WG, Davis JP. Clinical correlates of the fast and the slow spike-wave electroencephalogram. Pediatrics. 1950;5(4):626–44. [PubMed] [Google Scholar]
- 2.Gastaut H, Roger J, Soulayrol R, et al. Childhood epileptic encephalopathy with diffuse slow spike-waves (otherwise known as “petit mal variant”) or Lennox syndrome. Epilepsia. 1966;7(2):139–79. 10.1111/j.1528-1167.1966.tb06263.x. [DOI] [PubMed] [Google Scholar]
- 3.Arzimanoglou A, French J, Blume WT, et al. Lennox–Gastaut syndrome: a consensus approach on diagnosis, assessment, management, and trial methodology. Lancet Neurol. 2009;8(1):82–93. 10.1016/s1474-4422(08)70292-8. [DOI] [PubMed] [Google Scholar]
- 4.Camfield PR. Definition and natural history of Lennox–Gastaut syndrome. Epilepsia. 2011;52(s5):3–9. 10.1111/j.1528-1167.2011.03177.x. [DOI] [PubMed] [Google Scholar]
- 5.Cross JH, Auvin S, Falip M, et al. Expert opinion on the management of Lennox–Gastaut syndrome: treatment algorithms and practical considerations. Front Neurol. 2017;8:505. 10.3389/fneur.2017.00505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kerr M, Kluger G, Philip S. Evolution and management of Lennox–Gastaut syndrome through adolescence and into adulthood: Are seizures always the primary issue? Epileptic Disord. 2011;13(Suppl 1):S15-26. 10.1684/epd.2011.0409. [DOI] [PubMed] [Google Scholar]
- 7.Yagi K. Evolution of Lennox–Gastaut syndrome: a long-term longitudinal study. Epilepsia. 1996;37(Suppl 3):48–51. 10.1111/j.1528-1157.1996.tb01821.x. [DOI] [PubMed] [Google Scholar]
- 8.Asadi-Pooya AA, Bazrafshan M, Farazdaghi M. Long-term medical and social outcomes of patients with Lennox–Gastaut syndrome. Epilepsy Res. 2021;178:106813. 10.1016/j.eplepsyres.2021.106813. [DOI] [PubMed] [Google Scholar]
- 9.Asadi-Pooya AA, Bazrafshan M, Farazdaghi M. Cluster analysis of a large dataset of patients with Lennox–Gastaut syndrome. Seizure. 2021;92:36–9. 10.1016/j.seizure.2021.08.005. [DOI] [PubMed] [Google Scholar]
- 10.Asadi-Pooya AA, Farazdaghi M. Age at onset in patients with Lennox–Gastaut syndrome: correlations with clinical manifestations. J Clin Neurosci. 2022;100:138–42. 10.1016/j.jocn.2022.04.020. [DOI] [PubMed] [Google Scholar]
- 11.Piña-Garza JE, Chung S, Montouris GD, et al. Challenges in identifying Lennox–Gastaut syndrome in adults: a case series illustrating its changing nature. Epilepsy Behav Case Rep. 2016;5:38–43. 10.1016/j.ebcr.2016.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Riney K, Bogacz A, Somerville E, et al. International League Against Epilepsy classification and definition of epilepsy syndromes with onset at a variable age: position statement by the ILAE Task Force on Nosology and Definitions. Epilepsia. 2022;63(6):1443–74. 10.1111/epi.17240. [DOI] [PubMed] [Google Scholar]
- 13.Asadi-Pooya AA. Lennox–Gastaut syndrome: a comprehensive review. Neurol Sci. 2018;39(3):403–14. 10.1007/s10072-017-3188-y. [DOI] [PubMed] [Google Scholar]
- 14.Montouris G, Aboumatar S, Burdette D, et al. Expert opinion: proposed diagnostic and treatment algorithms for Lennox–Gastaut syndrome in adult patients. Epilepsy Behav. 2020;110:107146. 10.1016/j.yebeh.2020.107146. [DOI] [PubMed] [Google Scholar]
- 15.Strzelczyk A, Schubert-Bast S. Expanding the treatment landscape for Lennox–Gastaut syndrome: current and future strategies. CNS Drugs. 2021;35(1):61–83. 10.1007/s40263-020-00784-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Varadkar S. Cannabidiol for drop seizures in Lennox–Gastaut syndrome. Lancet. 2018;391(10125):1006–7. 10.1016/S0140-6736(18)30135-1. [DOI] [PubMed] [Google Scholar]
- 17.Glauser T, Kluger G, Sachdeo R, et al. Rufinamide for generalized seizures associated with Lennox–Gastaut syndrome. Neurology. 2008;70(21):1950–8. 10.1212/01.wnl.0000303813.95800.0d. [DOI] [PubMed] [Google Scholar]
- 18.Motte J, Trevathan E, Arvidsson JFV, et al. Lamotrigine for generalized seizures associated with the Lennox–Gastaut syndrome. New Engl J Med. 1997;337(25):1807–12. 10.1056/NEJM199712183372504. [DOI] [PubMed] [Google Scholar]
- 19.Ng YT, Conry JA, Drummond R, et al. Randomized, phase III study results of clobazam in Lennox–Gastaut syndrome. Neurology. 2011;77(15):1473–81. 10.1212/WNL.0b013e318232de76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sachdeo RC, Glauser TA, Ritter F, et al. A double-blind, randomized trial of topiramate in Lennox–Gastaut syndrome. Neurology. 1999;52(9):1882–7. 10.1212/wnl.52.9.1882. [DOI] [PubMed] [Google Scholar]
- 21.The Felbamate Study Group in Lennox–Gastaut S. Efficacy of felbamate in childhood epileptic encephalopathy (Lennox–Gastaut syndrome). New Engl J Med. 1993;328(1):29–33. 10.1056/NEJM199301073280105. [DOI] [PubMed] [Google Scholar]
- 22.Thiele EA, Marsh ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox–Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391(10125):1085–96. 10.1016/s0140-6736(18)30136-3. [DOI] [PubMed] [Google Scholar]
- 23.Knupp KG, Scheffer IE, Ceulemans B, et al. Efficacy and safety of fenfluramine for the treatment of seizures associated with Lennox–Gastaut syndrome: a randomized clinical trial. JAMA Neurol. 2022;79(6):554-64. 10.1001/jamaneurol.2022.0829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brown ML, Lipscomb J, Snyder C. The burden of illness of cancer: economic cost and quality of life. Annu Rev Public Health. 2001;22:91–113. 10.1146/annurev.publhealth.22.1.91. [DOI] [PubMed] [Google Scholar]
- 25.Zöllner JP, Franz DN, Hertzberg C, et al. A systematic review on the burden of illness in individuals with tuberous sclerosis complex (TSC). Orphanet J Rare Dis. 2020;15(1):23. 10.1186/s13023-019-1258-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sullivan J, Deighton AM, Vila MC, et al. The clinical, economic, and humanistic burden of Dravet syndrome–a systematic literature review. Epilepsy Behav. 2022;130:108661. 10.1016/j.yebeh.2022.108661. [DOI] [PubMed] [Google Scholar]
- 27.Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. 10.1371/journal.pmed.1000097s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Royle P, Bain L, Waugh N. Systematic reviews of epidemiology in diabetes: finding the evidence. BMC Med Res Methodol. 2005;5(1):2. 10.1186/1471-2288-5-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Centre for Reviews and Dissemination. NHS EED economics filter: MEDLINE (Ovid) York: CRD; 2010
- 30.Suzy Paisley, Andrew Booth, Shaila Mensinkai. Chapter 12: health-related quality of life studies. 2005 [cited Jun 10 2020]. In: Etext on Health Technology Assessment (HTA) Information Resources [Internet]. [cited Jun 10 2020]. Available from: https://www.nlm.nih.gov/archive/20060905/nichsr/ehta/chapter12.html.
- 31.CADTH. Strings Attached: CADTH's Database Search Filters 2021 [Available from: https://www.cadth.ca/strings-attached-cadths-database-search-filters.
- 32.Cooper C, Booth A, Britten N, et al. A comparison of results of empirical studies of supplementary search techniques and recommendations in review methodology handbooks: a methodological review. Syst Rev. 2017;6(1):234. 10.1186/s13643-017-0625-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cooper C, Dawson S, Peters J, et al. Revisiting the need for a literature search narrative: a brief methodological note. Res Synth Methods. 2018;9(3):361–5. 10.1002/jrsm.1315. [DOI] [PubMed] [Google Scholar]
- 34.Covidence. Systematic review management platform [Available from: https://www.covidence.org/]
- 35.Hoy D, Brooks P, Woolf A, et al. Assessing risk of bias in prevalence studies: modification of an existing tool and evidence of interrater agreement. J Clin Epidemiol. 2012;65(9):934–9. 10.1016/j.jclinepi.2011.11.014. [DOI] [PubMed] [Google Scholar]
- 36.Drummond MF, Sculpher MJ, Claxton K, et al. Methods for the economic evaluation of health care programmes. Oxford: Oxford University Press; 2015. [Google Scholar]
- 37.Molinier L, Bauvin E, Combescure C, et al. Methodological considerations in cost of prostate cancer studies: a systematic review. Value Health. 2008;11(5):878–85. 10.1111/j.1524-4733.2008.00327.x. [DOI] [PubMed] [Google Scholar]
- 38.Gallop K, Lloyd AJ, Olt J, et al. Impact of developmental and epileptic encephalopathies on caregivers: a literature review. Epilepsy Behav. 2021;124:108324. 10.1016/j.yebeh.2021.108324. [DOI] [PubMed] [Google Scholar]
- 39.Beilmann A, Napa A, Soot A, et al. Prevalence of childhood epilepsy in Estonia. Epilepsia. 1999;40(7):1011–9. [DOI] [PubMed] [Google Scholar]
- 40.Chin RFM, Pickrell WO, Guelfucci F, et al. Prevalence, healthcare resource utilization and mortality of Lennox–Gastaut syndrome: retrospective linkage cohort study. Seizure. 2021;91:159–66. [DOI] [PubMed] [Google Scholar]
- 41.Hollenack K, Story T, Acs A, et al. Prevalence of probable dravet syndrome, Lennox–Gastaut syndrome, and other refractory epilepsies in commercial and medicaid populations in the United States. J Manag Care Spec Pharm. 2019;25(3-A SUPPL):S58. [Google Scholar]
- 42.Rantala H, Putkonen T. Occurrence, outcome, and prognostic factors of infantile spasms and Lennox–Gastaut syndrome. Epilepsia. 1999;40(3):286–9. [DOI] [PubMed] [Google Scholar]
- 43.Sidenvall R, Forsgren L, Heijbel J. Prevalence and characteristics of epilepsy in children in northern Sweden. Seizure. 1996;5(2):139–46. [DOI] [PubMed] [Google Scholar]
- 44.Trevathan E, Murphy CC, Yeargin-Allsopp M. Prevalence and descriptive epidemiology of Lennox–Gastaut syndrome among Atlanta children. Epilepsia. 1997;38(12):1283–8. [DOI] [PubMed] [Google Scholar]
- 45.Heiskala H. Community-based study of Lennox–Gastaut syndrome. Epilepsia. 1997;38(5):526–31. [DOI] [PubMed] [Google Scholar]
- 46.Pina-Garza JE, Montouris GD, Vekeman F, et al. Assessment of treatment patterns and healthcare costs associated with probable Lennox–Gastaut syndrome. Epilepsy Behav. 2017;73:46–50. [DOI] [PubMed] [Google Scholar]
- 47.Strzelczyk A, Schubert-Bast S, Simon A, et al. Epidemiology, healthcare resource use, and mortality in patients with probable Lennox–Gastaut syndrome: a population-based study on German health insurance data. Epilepsy Behav. 2021;115: 107647. [DOI] [PubMed] [Google Scholar]
- 48.Autry AR, Trevathan E, Van Naarden Braun K, et al. Increased risk of death among children with Lennox–Gastaut syndrome and infantile spasms. J Child Neurol. 2010;25(4):441–7. [DOI] [PubMed] [Google Scholar]
- 49.Office for National Statistics (ONS). Death registrations, Populations and Age Standardised Rates, England 1981 to 2018 2020 [Available from: https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/deaths/adhocs/11168deathregistrationspopulationsandagestandardisedratesengland1981to2018.]
- 50.Myland M, Buysse B, Tsong W, et al. Seizure frequency, healthcare resource utilisation and mortality in childhood epilepsy: a retrospective cohort study using the THIN database. Arch Dis Child. 2019;104(11):1070. 10.1136/archdischild-2019-316910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Francois C, Stern JM, Ogbonnaya A, et al. Use and cost comparison of clobazam to other antiepileptic drugs for treatment of Lennox–Gastaut syndrome. J Mark Access Health Policy. 2017;5(1):1318691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hollenack KA, Story TJ, Acs A, et al. Pnd34 economic burden of probable Lennox–Gastaut syndrome, probable Dravet syndrome, and other refractory epilepsies for united states medicaid health plans. Value Health. 2019;22(Supplement 2):S276. [Google Scholar]
- 53.Reaven NL, Funk SE, Lyons PD, et al. The direct cost of seizure events in severe childhood-onset epilepsies: a retrospective claims-based analysis. Epilepsy Behav. 2019;93:65–72. [DOI] [PubMed] [Google Scholar]
- 54.Reaven NL, Funk SE, Montouris GD, et al. Burden of illness in patients with possible Lennox–Gastaut syndrome: a retrospective claims-based study. Epilepsy Behav. 2018;88:66–73. [DOI] [PubMed] [Google Scholar]
- 55.Stockl KM, Hollenack KA, Acs A, et al. Pnd27 economic burden of probable Lennox–Gastaut syndrome, probable Dravet syndrome, and other refractory epilepsies for united states commercial health plans. Value Health. 2019;22(Supplement 2):S274. [Google Scholar]
- 56.Stockl K, Funk S, Reaven N, et al. Inpatient hospitalizations and readmissions among patients with probable Lennox–Gastaut syndrome, dravet syndrome, tuberous sclerosis complex, and other refractory epilepsies. J Manag Care Spec Pharm. 2019;25(10-A SUPPL):S57–8. [Google Scholar]
- 57.Umeno J. A survey on the utilization status of social welfare services for pediatric patients with epilepsy. [Japanese]. No To Hattatsu. 2019;51(4):23–49.
- 58.Gallop K, Wild D, Verdian L, et al. Lennox–Gastaut Syndrome (LGS): development of conceptual models of health-related quality of life (HRQL) for caregivers and children. Seizure. 2010;19(1):23–30. [DOI] [PubMed] [Google Scholar]
- 59.Gibson PA. Lennox–Gastaut syndrome: impact on the caregivers and families of patients. J Multidiscip Healthc. 2014;7:441–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Murray J. Coping with the uncertainty of uncontrolled epilepsy. Seizure. 1993;2(3):167–78. [DOI] [PubMed] [Google Scholar]
- 61.Auvin S, Damera V, Martin M, et al. The impact of seizure frequency on quality of life in patients with Lennox–Gastaut syndrome or Dravet syndrome. Epilepsy Behav. 2021;123: 108239. [DOI] [PubMed] [Google Scholar]
- 62.Radu X, Damera V, Martin M, et al. Pro58 quality of life in patients with Dravet syndrome or Lennox Gastaut syndrome in the Uk: higher seizure frequency has a substantial negative impact on quality of life. Value Health. 2019;22(Supplement 2):S346. [Google Scholar]
- 63.National Institute for Health And Care Excellence (NICE). Cannabidiol with clobazam for treating seizures associated with Lennox–Gastaut syndrome 2019 [Available from: https://www.nice.org.uk/guidance/ta615/documents/final-appraisal-determination-document]
- 64.World Health Organization. International Classification of Diseases 11th Revision (ICD-11) 2022 [Available from: https://www.who.int/standards/classifications/classification-of-diseases]
- 65.Vekeman F, Piña-Garza JE, Cheng WY, et al. Development of a classifier to identify patients with probable Lennox–Gastaut syndrome in health insurance claims databases via random forest methodology. Curr Med Res Opin. 2019;35(8):1415–20. 10.1080/03007995.2019.1595552. [DOI] [PubMed] [Google Scholar]
- 66.Benedict A, Verdian L, Maclaine G. The cost effectiveness of rufinamide in the treatment of Lennox–Gastaut syndrome in the UK. Pharmacoeconomics. 2010;28(3):185–99. [DOI] [PubMed] [Google Scholar]
- 67.Verdian L, Yi Y. Cost-utility analysis of rufinamide versus topiramate and lamotrigine for the treatment of children with Lennox–Gastaut Syndrome in the United Kingdom. Seizure. 2010;19(1):1–11. [DOI] [PubMed] [Google Scholar]
- 68.Wijnen B, Armstrong N, Ramaekers B, et al. Cannabidiol for adjuvant treatment of seizures associated with Lennox–Gastaut syndrome and dravet syndrome: an evidence review group perspective of a NICE single technology appraisal. Pharmacoeconomics. 2020;38(10):1043–53. [DOI] [PubMed] [Google Scholar]
- 69.Neuberger EE, Carlson JJ, Veenstra DL. Cost-effectiveness of cannabidiol adjunct therapy versus usual care for the treatment of seizures in Lennox–Gastaut syndrome. Pharmacoeconomics. 2020;38(11):1237–45. [DOI] [PubMed] [Google Scholar]
- 70.Strzelczyk A, Kurlemann G, Bast T, et al. Exploring the relationships between composite scores of disease severity, seizure-freedom and quality of life in Dravet syndrome. Neurol Res Pract. 2022;4(1):22. 10.1186/s42466-022-00186-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lagae L, Brambilla I, Mingorance A, et al. Quality of life and comorbidities associated with Dravet syndrome severity: a multinational cohort survey. Dev Med Child Neurol. 2018;60(1):63–72. 10.1111/dmcn.13591. [DOI] [PubMed] [Google Scholar]
- 72.Strzelczyk A, Schubert-Bast S, Bast T, et al. A multicenter, matched case-control analysis comparing burden-of-illness in Dravet syndrome to refractory epilepsy and seizure remission in patients and caregivers in Germany. Epilepsia. 2019;60(8):1697–710. 10.1111/epi.16099. [DOI] [PubMed] [Google Scholar]
- 73.Strzelczyk A, Kalski M, Bast T, et al. Burden-of-illness and cost-driving factors in Dravet syndrome patients and carers: a prospective, multicenter study from Germany. Eur J Paediatr Neurol. 2019;23(3):392–403. 10.1016/j.ejpn.2019.02.014. [DOI] [PubMed] [Google Scholar]
- 74.Grau J, Zöllner JP, Schubert-Bast S, et al. Direct and indirect costs and cost-driving factors of Tuberous sclerosis complex in children, adolescents, and caregivers: a multicenter cohort study. Orphanet J Rare Dis. 2021;16(1):282. 10.1186/s13023-021-01899-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sancho J, Peña P, Rufo M, et al. Health and non-health care resources use in the management of adult outpatients with drug-resistant epilepsy in Spain: a cost-of-illness study (LINCE study). Epilepsy Res. 2008;81(2–3):176–87. 10.1016/j.eplepsyres.2008.05.008. [DOI] [PubMed] [Google Scholar]
- 76.Zöllner JP, Grau J, Rosenow F, et al. Direct and indirect costs and cost-driving factors in adults with tuberous sclerosis complex: a multicenter cohort study and a review of the literature. Orphanet J Rare Dis. 2021;16(1):250. 10.1186/s13023-021-01838-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Nabbout R, Dirani M, Teng T, et al. Impact of childhood Dravet syndrome on care givers of patients with DS, a major impact on mothers. Epilepsy Behav. 2020;108:107094. 10.1016/j.yebeh.2020.107094. [DOI] [PubMed] [Google Scholar]
- 78.Harris M, Macinko J, Jimenez G, et al. Measuring the bias against low-income country research: an Implicit Association Test. Globalization Health. 2017;13(1):80. 10.1186/s12992-017-0304-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Harris M, Marti J, Watt H, et al. Explicit bias toward high-income-country research: a randomized, blinded, crossover experiment of english clinicians. Health Aff. 2017;36(11):1997–2004. 10.1377/hlthaff.2017.0773. [DOI] [PubMed] [Google Scholar]
- 80.Feigin VL, Nichols E, Alam T, et al. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019;18(5):459–80. 10.1016/S1474-4422(18)30499-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Singh G, Sander JW. The global burden of epilepsy report: implications for low-and middle-income countries. Epilepsy Behav. 2020;105:106949. 10.1016/j.yebeh.2020.106949. [DOI] [PubMed] [Google Scholar]
- 82.Davies EH, Fieggen K, Wilmshurst J, et al. Demonstrating the feasibility of digital health to support pediatric patients in South Africa. Epilepsia Open. 2021;6(4):653–62. 10.1002/epi4.12527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Le VT, Le Thuy MA, Nguyen DH, et al. Epilepsy surgery program in a resource-limited setting in Vietnam: a multicentered collaborative model. Epilepsia Open. 2022;7(4):710–7. 10.1002/epi4.12650.10.1002/epi4.12650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Camfield P, Camfield C. Long-term prognosis for symptomatic (secondarily) generalized epilepsies: a population-based study. Epilepsia. 2007;48(6):1128–32. [DOI] [PubMed] [Google Scholar]
- 85.Hahn CD, Jiang Y, Villanueva V, et al. A phase 2, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of soticlestat as adjunctive therapy in pediatric patients with Dravet syndrome or Lennox–Gastaut syndrome (ELEKTRA). Epilepsia. 2022;63(10):2671–83. 10.1111/epi.17367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Specchio N, Wirrell EC, Scheffer IE, et al. International league against epilepsy classification and definition of epilepsy syndromes with onset in childhood: Position paper by the ILAE task force on nosology and definitions. Epilepsia. 2022;63(6):1398–442. 10.1111/epi.17241. [DOI] [PubMed]
- 87.Offen ML. Dealing with “defectives” Foster Kennedy and William Lennox on eugenics. Neurology. 2003;61(5):668. 10.1212/WNL.61.5.668. [DOI] [PubMed] [Google Scholar]
- 88.Rosso M, Chu D, Santoro JD. William Gordon Lennox (1884–1960). J Neurol. 2021;268(9):3512–3. 10.1007/s00415-020-10338-z. [DOI] [PubMed] [Google Scholar]
- 89.Strzelczyk A, Schubert-Bast S. Psychobehavioural and cognitive adverse events of anti-seizure medications for the treatment of developmental and epileptic encephalopathies. CNS Drugs. 2022:36(10):1079–111. 10.1007/s40263-022-00955-9. [DOI] [PMC free article] [PubMed]
- 90.Niedermeyer E. The Lennox–Gastaut syndrome: a severe type of childhood epilepsy. Dtsch Z Nervenheilkd. 1969;195(4):263–82. 10.1007/bf00242459. [DOI] [PubMed] [Google Scholar]
- 91.Beaumanoir A, et al. The Lennox–Gastaut syndrome. In: Roger J, Bureau M, Dravet C, et al., editors. Epileptic syndromes in infancy, childhood, and adolescence. London: John Libbey; 1985. p. 89–99. [Google Scholar]
- 92.International League Against Epilepsy. Proposal for revised classification of epilepsies and epileptic syndromes. Commission on classification and terminology of the international league against epilepsy. Epilepsia. 1989;30(4):389–99. 10.1111/j.1528-1157.1989.tb05316.x. [DOI] [PubMed] [Google Scholar]
- 93.Engel J Jr. A proposed diagnostic scheme for people with epileptic seizures and with epilepsy: report of the ILAE task force on classification and terminology. Epilepsia. 2001;42(6):796–803. 10.1046/j.1528-1157.2001.10401.x. [DOI] [PubMed] [Google Scholar]
- 94.Scheffer IE, Berkovic S, Capovilla G, et al. ILAE classification of the epilepsies: position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017;58(4):512–21. 10.1111/epi.13709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. 10.1136/bmj.n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 2. Supplementary Table S1: Healthcare resource utilization and costs in patients with LGS; Table S2: Quality assessment checklist for prevalence studies; Table S3: Quality assessment checklist of cost-of-illness studies; Table S4: Quality assessment checklist for qualitative HRQoL studies; Table S5: HRQoL: Quality assessment checklist for quantitative HRQoL studies.
Additional file 3. Supplementary Figure S1: Costs, annual hospitalization rate and length of stay (LOS) in LGS patients in years where patients were prescribed with rescue medication vs years where patients were not prescribed rescue medication.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.