TO THE EDITOR:
Chimeric antigen receptor (CAR) T cells are associated with early (eg, cytokine release syndrome and immune effector cell–associated neurotoxicity syndrome) and late toxicities. Delayed adverse effects include prolonged cytopenia and hypogammaglobulinemia. Although hypogammaglobulinemia was well anticipated because of the expected on-target toxicity of the CD19-directed CAR T cells, prolonged cytopenia was initially underestimated.1,2 Their pathophysiology remains poorly understood, and their optimal management is not yet known. Prolonged and profound thrombocytopenias occurring after CAR T-cell infusion have been frequently reported in clinical trials and real-world studies (supplemental Tables 1 and 2). In ZUMA-1, 24% and 7% of patients treated with axicabtagene-ciloleucel (axi-cel) for diffuse large B-cell lymphoma (DLBCL) experienced grade 3 or 4 thrombocytopenia at days 30 and 90 after CAR T-cell infusion, respectively.3 In real-world experience, Logue et al4 reported the outcome of 70 patients with B-cell lymphoma receiving axi-cel in a single-center retrospective study. Grade 3 or 4 thrombocytopenia was found in 26% of patients at day 30 and in 5% at day 90. In a multicenter retrospective study of patients with DLBCL treated with axi-cel, Rejesky et al2 described the biphasic temporal course of CAR T-cell–associated cytopenia. After hematologic recovery postchemotherapy, neutrophil and platelet counts decreased after 3 weeks, reaching a nadir at ∼6 weeks after CAR T-cell infusion. Thrombopoietin receptor agonists (TPO-RAs) have been approved for the treatment of immune thrombocytopenic purpura (ITP) and severe aplastic anemia (AA). They have also been used for the treatment of thrombocytopenia after chemotherapy or allogeneic stem cell transplantation.5 However, the role of TPO-RAs in the management of prolonged thrombocytopenia after CAR T-cell therapy remains unknown.
We retrospectively analyzed the outcomes of patients with lymphoma treated with CD19 CAR T cells who developed prolonged and/or profound thrombocytopenia and who received TPO-RAs at our center (University Hospital of Rennes). Patients were included if they had received at least 1 platelet transfusion after CAR T-cell infusion for a platelet count <20 × 109/L and were treated with TPO-RAs. Patients whose disease relapsed after receiving CAR T cells were censored at the time of salvage therapy. Platelet recovery was defined as 2 consecutive platelet counts >50 × 109/L. The study was approved by the Ethics Committee of the University Hospital of Rennes (study MR-004) and performed according to the Declaration of Helsinki.
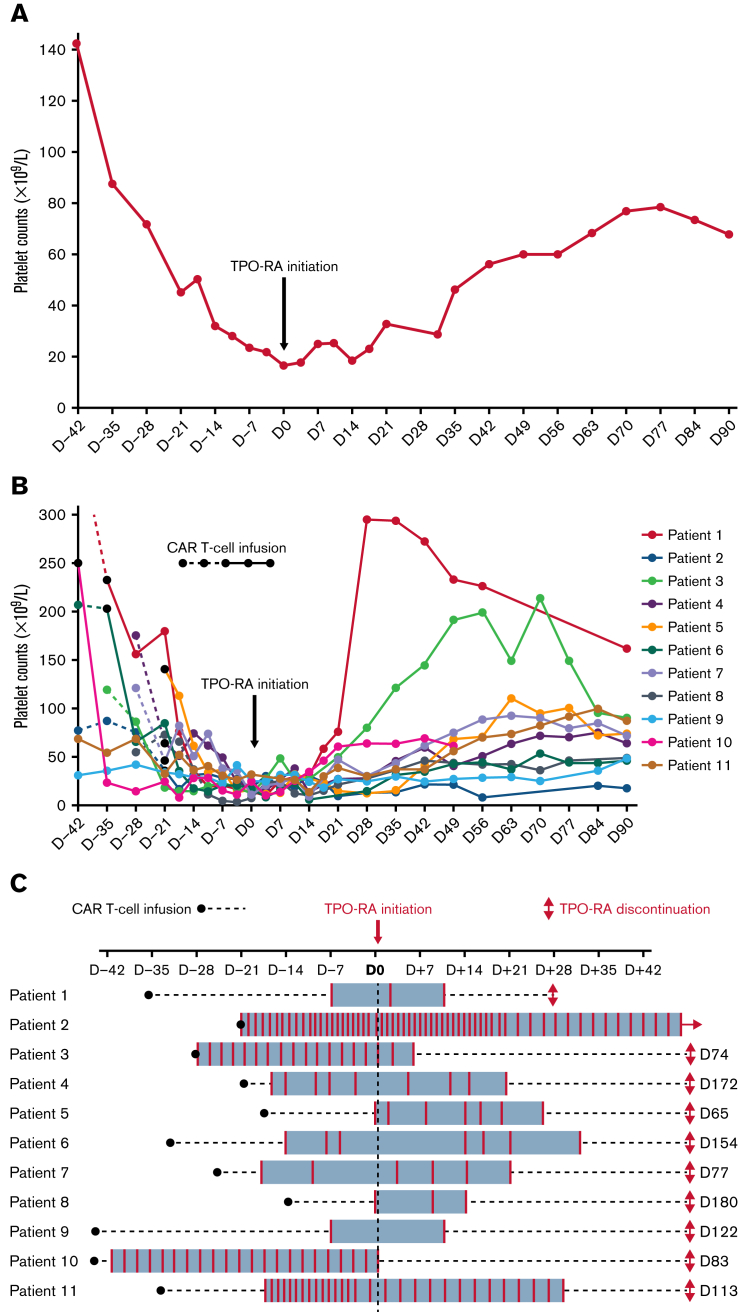
Between September 2019 and October 2021, 71 patients were treated with axi-cel, tisagenlecleucel, or brexucabtagene-autoleucel for DLBCL or mantle cell lymphoma and followed at our center. Grade 3 or 4 thrombocytopenia occurred in 46% of patients at 1 month, 25% at 2 months, and 15% at 3 months after CAR T-cell infusion. The median CAR-HEMATOTOX score was 2. Overall, 62% of the patients presenting with grade 3 or 4 thrombocytopenia after 1 month had a high CAR-HEMATOTOX score (≥2). Of the 71 patients, 11 (15.5%) were treated with TPO-RAs (patient characteristics are presented in Table 1). All patients had been treated with axi-cel for DLBCL, except 2 patients, who were treated with brexucabtagene-autoleucel for mantle cell lymphoma (patients 5 and 8). No patient had undergone transplantation before CAR T-cell therapy. The median CAR-HEMATOTOX score was 3. Ten patients were treated with eltrombopag at 75 mg per day, and 1 patient received romiplostim at 10 μg/kg per week (patient 8). TPO-RAs were started at a median of 27 days after CAR T-cell infusion, and the median duration of treatment with TPO-RAs was 97 days. Four patients were treated with anticoagulant therapy (patients 2, 4, 10, and 11); as a result, they had a higher transfusion threshold (platelets <50 × 109/L) and higher platelet transfusion rate. Platelet transfusion independence was achieved after a median of 17 days from TPO-RA initiation and 46 days from CAR T-cell infusion. Platelet recovery was achieved after a median of 46 and 76 days from TPO-RA initiation and CAR T-cell infusion, respectively. At the time of analysis, only 1 patient (patient 2) had not achieved transfusion independence, 6 months after CAR T-cell infusion (Figure 1). In our cohort, bone marrow aspiration was performed in 5 patients, showing hypoplastic bone marrow with rare megakaryocytes in 3 patients and normal bone marrow and megakaryocyte density in 2 patients. One patient experienced relapse at 3 months after CAR T-cell therapy (patient 10) and was censored at initiation of oral chemotherapy. All others were in remission after a median follow-up of 274 days. We did not observe any clinically significant adverse events that might have been related to TPO-RAs or, notably, any thromboembolic events.
Table 1.
Patient characteristics
| Characteristic | Value |
|---|---|
| Number of patients | 11 |
| Median age, y (range) | 64 (41-75) |
| Disease | |
| DLBCL | 9/11 |
| MCL | 2/11 |
| CAR T cells | |
| Axi-cel | 9/11 |
| Brexu-cel | 2/11 |
| Median CAR-HEMATOTOX score (range) | 3 (0-5) |
| Type of TPO-RA | |
| Eltrombopag | 10/11 |
| Romiplostim | 1/11 |
| Median follow-up from CAR T-cell infusion, d (range) | 274 (90-828) |
| Median platelets count at 1 mo of TPO-RA initiation, ×109/L (range) | 28 (13-295) |
| Median platelets count at 2 mo of TPO-RA initiation, ×109/L (range) | 60 (8-226) |
| Median platelets count at 3 mo of TPO-RA initiation, ×109/L (range) | 68 (17-162) |
| Median time between CAR T-cell infusion and start of TPO-RA, d (range) | 27 (14-45) |
| Median duration of TPO-RA, d (range) | 97 (27-180) |
| Number of patients free of platelet transfusion after TPO-RA | 10/11∗ |
| Median duration between start of TPO-RA and last transfusion, d (range)† | 17 (1-65) |
| Median duration between start of TPO-RA and platelet recovery, d (range)† | 46 (18-100) |
| Median duration between CAR T-cell infusion and last transfusion, d (range)† | 46 (21-104) |
| Median duration between CAR T-cell infusion and platelet recovery, d (range)† | 76 (53-130) |
Brexu-cel, brexucabtagene-autoleucel; MCL, mantle cell lymphoma.
Patient 2 was still undergoing transfusion at 6 mo after CAR T-cell infusion.
Patient 2 excluded, because still undergoing transfusion at 6 mo after CAR T-cell infusion. Platelet recovery was defined as 2 consecutive platelet counts >50 × 109/L.
Figure 1.
Platelet counts and platelet transfusion rates before and after TPO-RA initiation. (A) Median platelet counts before and after TPO-RA initiation for the entire cohort. Median platelet counts of the 11 patients are represented from day 42 before TPO-RA initiation (D-42) until day 90 after TPO-RA initiation (D90). (B) Individual platelet counts before and after TPO-RA initiation. Platelet counts of the 11 patients treated with TPO-RAs are shown from D-42 until D90. CAR T-cell infusion is represented with a black dot. Platelet counts before CAR T-cell infusion are represented with dashed lines, and those after infusion are represented with continuous lines. (C) Platelet transfusion rates before and after TPO-RA initiation. Black bullet point represents CAR T-cell infusion. Each red bar represents a platelet transfusion.
TPO-RAs demonstrated efficacy in ITP and severe AA. We evaluated the efficacy of TPO-RAs in patients treated with CAR T cells. In our cohort, patients treated with TPO-RAs achieved platelet recovery after a median time of 46 days and transfusion independence after a median of 17 days from initiation of TPO-RAs. In patients with chronic ITP treated with TPO-RAs (eltrombopag at 50-75 mg per day), the median platelet count increased to >53×109/L at day 15.6,7 Those with severe AA8 received eltrombopag at a higher dose of 150 mg per day, along with immunosuppressive therapy. Platelet transfusion independence was achieved after a mean time of 40 days from randomization and 26 days from TPO-RA initiation (TPO-RAs were started 14 days after randomization). Prolonged cytopenia after CAR T-cell therapy usually recovered spontaneously after a few weeks or months. Therefore, it is difficult to formally demonstrate in a retrospective study that TPO-RAs contribute to reducing the duration of platelet recovery and/or need for transfusion. However, the rapid improvement of thrombocytopenia in most patients, despite a high CAR-HEMATOTOX score, suggests some benefit of TPO-RAs. Interestingly, TPO-RAs were found to improve trilineage hematopoiesis in refractory patients with aplastic anemia, despite high levels of endogenous thrombopoietin.9 Furthermore, a recent study showed that TPO-RAs suppressed the inhibitory signal mediated by interferon-γ in hematopoietic stem cells in vitro.10 The impact of TPO-RAs on the 3 hematopoietic lineages through an antiinflammatory effect may be of importance in treating prolonged cytopenia associated with CAR T-cell therapy.
Overall, our study is the first to report the effect of TPO-RAs on prolonged CAR T-cell–associated cytopenia. Our results suggest a possible benefit of TPO-RAs in such patients, reducing the duration of platelet recovery and need for transfusion. However, prospective randomized studies are warranted to formally evaluate the benefit of TPO-RAs in this situation.
Conflict-of-interest disclosure: G.D. has received honoraria from Janssen. F.L. has received honoraria from Gilead Sciences, Kite, Roche, and Janssen. S.D.G. received honoraria from Gilead Sciences, AbbVie, and Janssen. G.M. has received honoraria from Bristol Myers Squibb. R.H. has received honoraria from Bristol Myers Squibb, Merck Sharp & Dohme, Gilead, Kite, Roche, Novartis, Janssen, and Celgene.
Acknowledgments
Contribution: All authors reviewed and approved the final manuscript.
Footnotes
Qualified researchers may request access to individual patient level data through the clinical study data request platform www.clinicalstudydatarequest.com.
The full-text version of this article contains a data supplement.
Supplementary Material
References
- 1.Fried S, Avigdor A, Bielorai B, et al. Early and late hematologic toxicity following CD19 CAR-T cells. Bone Marrow Transplant. 2019;54(10):1643–1650. doi: 10.1038/s41409-019-0487-3. [DOI] [PubMed] [Google Scholar]
- 2.Rejeski K, Perez A, Sesques P, et al. CAR-HEMATOTOX: a model for CAR T-cell-related hematologic toxicity in relapsed/refractory large B-cell lymphoma. Blood. 2021;138(24):2499–2513. doi: 10.1182/blood.2020010543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Locke FL, Ghobadi A, Jacobson CA, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1-2 trial. Lancet Oncol. 2019;20(1):31–42. doi: 10.1016/S1470-2045(18)30864-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Logue JM, Zucchetti E, Bachmeier CA, et al. Immune reconstitution and associated infections following axicabtagene ciloleucel in relapsed or refractory large B-cell lymphoma. Haematologica. 2021;106(4):978–986. doi: 10.3324/haematol.2019.238634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ghanima W, Cooper N, Rodeghiero F, Godeau B, Bussel JB. Thrombopoietin receptor agonists: ten years later. Haematologica. 2019;104(6):1112–1123. doi: 10.3324/haematol.2018.212845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bussel JB, Provan D, Shamsi T, et al. Effect of eltrombopag on platelet counts and bleeding during treatment of chronic idiopathic thrombocytopenic purpura: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;373(9664):641–648. doi: 10.1016/S0140-6736(09)60402-5. [DOI] [PubMed] [Google Scholar]
- 7.Cheng G, Saleh MN, Marcher C, et al. Eltrombopag for management of chronic immune thrombocytopenia (RAISE): a 6-month, randomised, phase 3 study. Lancet. 2011;377(9763):393–402. doi: 10.1016/S0140-6736(10)60959-2. [DOI] [PubMed] [Google Scholar]
- 8.Peffault de Latour R, Kulasekararaj A, Iacobelli S, et al. Severe Aplastic Anemia Working Party of the European Society for Blood and Marrow Transplantation Eltrombopag added to immunosuppression in severe aplastic anemia. N Engl J Med. 2022;386(1):11–23. doi: 10.1056/NEJMoa2109965. [DOI] [PubMed] [Google Scholar]
- 9.Zhao X, Feng X, Wu Z, et al. Persistent elevation of plasma thrombopoietin levels after treatment in severe aplastic anemia. Exp Hematol. 2018;58:39–43. doi: 10.1016/j.exphem.2017.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Alvarado LJ, Huntsman HD, Cheng H, et al. Eltrombopag maintains human hematopoietic stem and progenitor cells under inflammatory conditions mediated by IFN-γ. Blood. 2019;133(19):2043–2055. doi: 10.1182/blood-2018-11-884486. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.