Abstract
Purpose
To investigate the association of the maximal severity of pneumonia on CT scans obtained within 6-week of diagnosis with the subsequent development of post-COVID-19 lung abnormalities (Co-LA).
Methods
COVID-19 patients diagnosed at our hospital between March 2020 and September 2021 were studied retrospectively. The patients were included if they had (1) at least one chest CT scan available within 6-week of diagnosis; and (2) at least one follow-up chest CT scan available ≥ 6 months after diagnosis, which were evaluated by two independent radiologists. Pneumonia Severity Categories were assigned on CT at diagnosis according to the CT patterns of pneumonia and extent as: 1) no pneumonia (Estimated Extent, 0%); 2) non-extensive pneumonia (GGO and OP, <40%); and 3) extensive pneumonia (extensive OP and DAD, >40%). Co-LA on follow-up CT scans, categorized using a 3-point Co-LA Score (0, No Co-LA; 1, Indeterminate Co-LA; and 2, Co-LA).
Results
Out of 132 patients, 42 patients (32%) developed Co-LA on their follow-up CT scans 6–24 months post diagnosis. The severity of COVID-19 pneumonia was associated with Co-LA: In 47 patients with extensive pneumonia, 33 patients (70%) developed Co-LA, of whom 18 (55%) developed fibrotic Co-LA. In 52 with non-extensive pneumonia, 9 (17%) developed Co-LA: In 33 with no pneumonia, none (0%) developed Co-LA.
Conclusions
Higher severity of pneumonia at diagnosis was associated with the increased risk of development of Co-LA after 6–24 months of SARS-CoV-2 infection.
Abbreviations: ARDS, acute respiratory distress syndrome; COVID-19, coronavirus disease 2019; OP, organizing pneumonia; Co-LA, post-COVID-19 lung abnormalities; DAD, diffuse alveolar damage; GGO, ground-glass opacity; ILA, interstitial lung abnormalities; ILD, interstitial lung disease; PE, pulmonary embolism; SARS-CoV2, severe acute respiratory syndrome coronavirus 2; 2019-nCoV, 2019 novel coronavirus
Keywords: COVID-19, COVID-19 pneumonia, COVID-19 related lung abnormalities, Lung, Abnormalities, Chest CT
Highlights
-
•
Post-COVID-19 lung abnormalities (Co-LA) was confirmed in 33% of COVID-19 patients.
-
•
Higher severity of COVID-19 pneumonia at diagnosis was associated with the likelihood of Co-LA.
-
•
The likelihood of Co-LA was 69% with extensive pneumonia and 18% with non-extensive pneumonia.
-
•
Fibrotic Co-LA was noted in 26 of 42 Co-LA patients (62%) with extensive pneumonia at diagnosis.
1. Introduction
In March 2020, the Society of Thoracic Radiology, the American College of Radiology and the Radiological Society of North America published the first guidelines for COVID-19 pneumonia on chest CT [1], [2], [3]. Bilateral, multifocal, peripheral, and often rounded ground-glass opacity (GGO), associated with consolidation or crazy-paving pattern as well as similar findings of organizing pneumonia, were considered typical CT findings of COVID-19 pneumonia [3]. COVID-19 affected over 500 million people worldwide, causing pneumonia, and CT is the key for diagnosis and monitoring.
Several recent studies have reported CT findings that are interpreted as probable sequalae of COVID-19 [4], [5], [6], [7], [8], [9], [10], [11]. More specifically, some residual cases of COVID-19 have been associated with interstitial changes, including reticulation or traction bronchiectasis, which resemble the CT findings for other diseases such as fibrotic interstitial lung abnormalities (ILA) or acute respiratory distress syndrome (ARDS) related pulmonary fibrosis [12], [13], [14], [15]. However, few studies investigated whether the initial COVID-19 pneumonia might lead to post-COVID-19 lung abnormalities (Co-LA).
To fill the gap, the present study was to investigate the correlation between the maximal severity of pneumonia on a CT scan obtained within a 6-week window around the time of diagnosis (−2 to +4 weeks), and the eventual development of Co-LA. We tested the hypothesis that the presence of severe pneumonia on a CT scan obtained around the time of the diagnosis is associated with subsequent development of fibrotic lung disease.
2. Materials and methods
2.1. Patient selection, inclusion, and exclusion
This study was approved by the institutional review board (IRB#2021P000981), and it was performed in accordance with principles of the declaration of Helsinki. COVID-19 patients diagnosed between March 1st 2020 and September 23rd 2021 were studied retrospectively. The inclusion criteria were: (1) at least one CT scan was available within a 6-week window around the time of the COVID-19 diagnosis (−2 to +4 weeks); and (2) at least one follow-up chest CT scan was available 24 weeks or more after the original diagnosis. Patients with preexisting interstitial lung disease (ILD)/ ILA were excluded due to the difficulty in distinguishing these pre-existing conditions from post-COVID-19 lung abnormalities (Co-LA).
2.2. Demographic and clinical data
Demographic data including age, sex, body mass index (BMI), smoking status, and vaccination status were obtained from the electronic medical record. The number of hospital stays, hospitalization, ICU admission, presence of ARDS, presence of pulmonary embolism (PE), platelet, D-dimer, and past medical history such as malignancy, hypertension, diabetes mellitus, coronary artery disease, chronic kidney disease, lung disease, and all-cause death were also collected as clinical data. Lung diseases in this study included asthma, chronic obstructive pulmonary disease, lung cancers, and lung surgery.
2.3. CT scans of chest
Chest CT scans were performed using standard chest CT protocols at our institute with or without administration of the intravenous contrast. Images were reconstructed with a slice thickness from 1 to 3 mm, and all the section CT images were reviewed in a lung window: window width of 1000–2000 Hounsfield units; window level of − 700 to − 500 Hounsfield units.
2.4. Image evaluation and classification
Initial CT scans were classified into 6-point-scale scores as Initial CT Scores, which is a modified method of the pattern categorization by Jin et al. [16]: 0: No Pneumonia (Estimated Extent, 0%), 1: GGO (<10%), 2: Transition from GGO to OP patten (10–20%), 3: OP patten (20–40%), 4: Extensive OP pattern (40–70%), and 5: diffuse alveolar damage (DAD) pattern (>70%) (Table 1) [17]. Two thoracic radiologists (HT, HH) independently reviewed and scored all available CT scans within the − 2 to + 4 weeks window of COVID-19 diagnosis. The maximal severity CT score (Pneumonia Score) was determined using the highest score of serial CT scans in each patient. Pneumonia Scores were further classified into three Pneumonia Severity Categories as follows: 1) no pneumonia (Pneumonia Score = 0); 2) non-extensive pneumonia (Pneumonia Score = 1, 2, or 3); and 3) extensive pneumonia (Pneumonia Score = 4 or 5). Interobserver agreement of Pneumonia Severity Categories was also calculated.
Table 1.
Pneumonia Score: CT severity score based on pattern and extent of COVID-19.
| Pneumonia Score | Pattern | Estimated Extent* |
|---|---|---|
| 0 | No pneumonia | 0% |
| 1 | GGO | < 10% |
| 2 | Transition from GGO to OP patten | 10–20% |
| 3 | OP pattern† | 20–40% |
| 4 | Extensive OP pattern | 40–70% |
| 5 | DAD pattern‡ | > 70% |
GGO, ground-glass opacity; OP, organizing pneumonia; DAD, diffuse alveolar damage;
AIP, acute interstitial pneumonia
*Extent was estimated based on the percentage of lung involvement in the entire bilateral lungs
†OP patten is characterized by multifocal bilateral parenchymal consolidation
‡DAD pattern is characterized by diffuse or multifocal GGOs/consolidation predominantly in dependent lung region, may be accompanied by lung volume loss and traction bronchiectasis
Follow-up CT images were categorized on a three-point-scale score, referred to here as the Co-LA Score: 0, No Co-LA, 1, Indeterminate Co-LA, and 2, Co-LA. Co-LA Score is defined by applying the framework of ILA [12], [13]. Briefly, ILA is defined as incidental CT findings of non-dependent abnormalities affecting more than 5% of any lung zone (upper, middle, and lower lung zones are demarcated by the levels of the inferior aortic arch and right inferior pulmonary vein) on chest CT, where interstitial disease was not previously suspected [12]. The findings include ground-glass or reticular abnormalities, lung distortion, traction bronchiectasis/bronchiolectasis, honeycombing, and non-emphysematous cysts [12], [13]. Co-LA was subcategorized according to the presence/absence of fibrotic change: fibrotic or non-fibrotic Co-LA by the same two experienced thoracic radiologists mentioned above (HT, HH).
In the event of discrepancy between the readings for the Initial CT Score or Co-LA Score, a third experienced thoracic radiologist (MN) reviewed the corresponding cases and determined the final score. The interobserver agreement of the Co-LA Score was calculated.
2.5. Statistical analyses
Numeric values were expressed as follows; mean ± standard deviation with normal distribution and median (interquartile range) with non-normal distribution. Categorical data including sex, smoking history, death, all-cause ICU admission, presence of PE, lung disease, ARDS, and past medical history among three groups by Co-LA Score were assessed using Fisher’s exact test. Age and BMI among three groups were examined with one-way ANOVA, while platelet, D-dimer, and hospital stays were examined with the Kruskal-Wallis test by ranks. The contingency table of Co-LA vs Pneumonia Score was described. The difference of distribution according to Co-LA Score was also examined with Fisher’s exact test. Co-LA Score was assessed with a multivariate linear regression analysis to confirm the presence or absence of confounder factors. Statistical analyses were conducted with R 4.0.4. Two-sided P values less than 0.050 were considered statistically significant.
3. Results
3.1. Inclusion criteria of the cohort
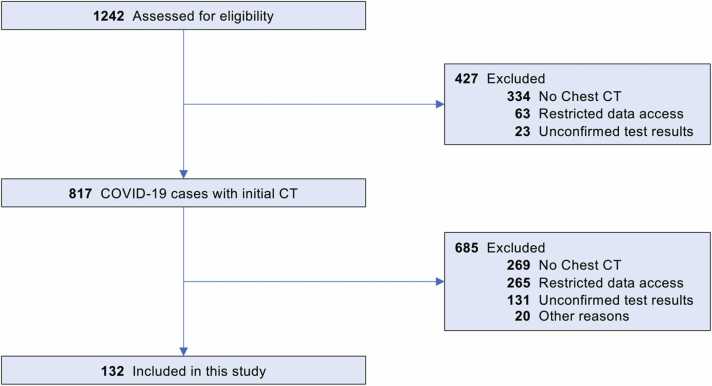
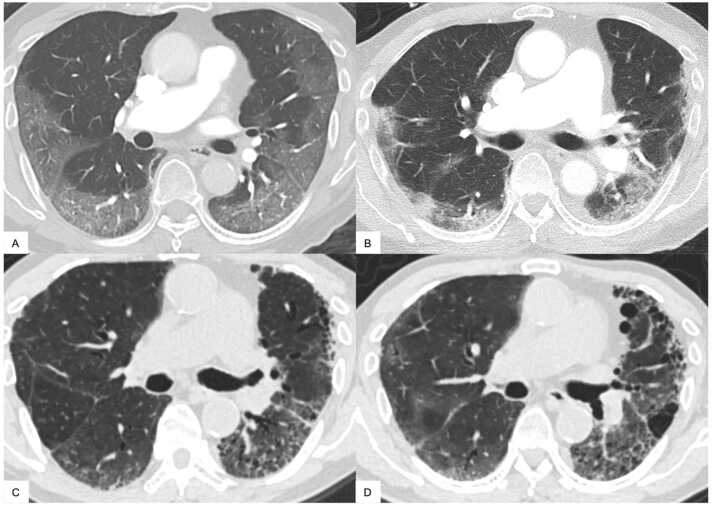
We first identified 1242 patients diagnosed with COVID-19 utilizing our institutional research patient data repository (RPDR). The flow chart for inclusion/exclusion criteria is shown in Fig. 1. Four hundred twenty-five cases were excluded because of the following reasons: no available chest CT scans (n = 334), restricted data access (n = 63), unconfirmed COVID-19 test results (n = 28). As such, 817 COVID-19 patients with at least one available CT scan were identified. An additional 269 cases were excluded because they had no CT scan in a six-week window (−2 to +4) around diagnosis, and another 20 due to preexisting interstitial lung disease (ILD) or ILA (n = 17), lung transplant after COVID-19 infection (n = 1), and post cardiac arrest (n = 2). As a result, 528 COVID-19 patients were identified who had at least one appropriate initial CT scan, but 396 of these did not have a CT scan 6 months or more after diagnosis and were thus excluded. Ultimately, a total of 132 COVID-19 patients (62.9 ± 14.2 years old; 57 males) were included in this study. Fig. 2 shows typical results from a patient with severe COVID-19 pneumonia who subsequently developed fibrotic Co-LA with serial CT scans at 1 month, 10 months and 16 months.
Fig. 1.
Inclusion and exclusion criteria.
Fig. 2.
An 82-year-old man without vaccination, lung disease, or smoking history. A. Initial CT scan was performed six days after diagnosis. Well-defined ground glass opacities (GGOs) were extended in subpleural region, while central area was spared. Initial CT score and Pneumonia Score was 4. B. Follow-up CT at one month after diagnosis. The previously identified GGOs became denser consolidation with architectural distortion indicating fibrosis with indication of traction bronchiolectasis. C. Follow-up CT at ten months after diagnosis. GGO and consolidation have decreased. However, asymmetrical moderate to severe traction bronchiectasis/bronchiolectasis has developed in subpleural areas. Microcystic honeycombing was also suspected. D. Follow-up CT at 16 months after diagnosis. Subpleural GGO with progression of asymmetrical severe traction bronchiectasis/bronchiolectasis, consistent with fibrotic Co-LA. Co-LA, post-COVID-19 lung abnormalities; GGO, ground-glass opacity.
3.2. Demographic data
Summarized demographic data for the whole cohort are provided in Table S1. The median interval from diagnosis to initial CT was 1.5 days, while the median interval to the last follow-up CT was 11.7 months. Demographic and clinical data as stratified by Co-LA Score are provided in Table 2. Co-LA patients had longer hospital stays (P < 0.001), and higher probability of hospitalization (P = 0.002), ARDS (P < 0.001), or ICU admission (P = 0.004). There was no difference in presence of PE, platelet, D-dimer, comorbidities, and all-cause death among the three groups of Co-LA, Indeterminate Co-LA, and No Co-LA.
Table 2.
Demographic data and clinical information stratified by Co-LA Score.
| Co-LA (n = 42) | Indeterminate Co-LA (n = 13) |
No Co-LA (n = 77) | P value | |
|---|---|---|---|---|
| Age, yrs | 62.2 ± 15.0 | 64.2 ± 16.7 | 63.9 ± 11.8 | 0.79 |
| Male | 23 (55%) | 4 (31%) | 30 (39%) | 0.17 |
| BMI, kg/m2 | 29.9 ± 8.1 | 28.4 ± 4.4 | 29.3 ± 6.6 | 0.76 |
| Smoking history Never Former/Current |
26 (62%) 16 (38%) |
8 (62%) 5 (38%) |
40 (52%) 37 (48%) |
0.51 |
| PE | 7 (17%) | 2 (15%) | 7 (9%) | 0.50 |
| Platelet, 103/μL | 190 [144–251] (n = 36) | 204 [164–296] (n = 10) | 239 [173–299] (n = 59) | 0.14 |
| D-dimer, ng/mL | 1105 [874–2020] (n = 33) | 1650 [580–4085] (n = 8) | 1590 [888–3644] (n = 47) | 0.51 |
| Vaccination | 2 (5%) | 0 (0%) | 8 (10%) | 0.54 |
| Hospital stays, days | 6 (0–14) | 5 (0–9) | 14 (8–34) | < 0.001 |
| Hospitalization | 40 (95%) | 8 (62%) | 57 (74%) | 0.002 |
| ICU admission | 15 (36%) | 1 (8%) | 9 (12%) | 0.004 |
| ARDS | 22 (52%) | 1 (8%) | 13 (17%) | < 0.001 |
| Lung disease | 16 (38%) | 8 (62%) | 37 (48%) | 0.51 |
| HT | 22 (52%) | 8 (62%) | 51 (66%) | 0.33 |
| CAD | 7 (17%) | 1 (8%) | 11 (14%) | 0.81 |
| DM | 10 (24%) | 2 (15%) | 15 (19%) | 0.77 |
| CKD | 7 (17%) | 1 (8%) | 10 (13%) | 0.75 |
| Malignancy | 25 (60%) | 8 (62%) | 46 (60%) | > 0.99 |
| All-cause death | 2 (5%) | 2 (15%) | 11 (14%) | 0.22 |
ARDS; acute respiratory distress syndrome; BMI, body mass index; CAD, coronary artery disease;
CKD, chronic kidney disease; Co-LA, post-COVID-19 lung abnormalities; DM, diabetes mellitus;
HT, hypertension; PE, pulmonary embolus
3.3. Difference between initial CT score and maximal severity CT score (Pneumonia Score)
Twenty-seven patients (20%) had more than one CT scan within the − 2 to + 4 weeks interval from diagnosis. Of these 27 patients, 13 (48%) had an increase in pneumonia severity over time, i.e., their maximal severity CT score (Pneumonia Score) was not established based on their initial CT scan but rather on a subsequent CT scan, which showed signs of more severe pneumonia. The relationship between Initial CT Score and Pneumonia Score is summarized in Table S2.
3.4. Pneumonia score and pneumonia severity categories
Pneumonia Scores were 0 in 33 (25%), 1 in 11 (8.3%), 2 in 27 (20%), 3 in 14 (11%), 4 in 36 (27%), and 5 in 11 (8.3%), respectively (Table 3). Pneumonia Severity Categories were no pneumonia in 33 (25%), non-extensive pneumonia in 52 (39%), and extensive pneumonia in 47 (36%). Interobserver agreement of the Pneumonia Severity Categories was substantial (weighted kappa score = 0.71).
Table 3.
Pneumonia Score vs Co-LA Score.
| Presence/Absence of Co-LA at follow-up CT, n (%) |
|||
|---|---|---|---|
| Pneumonia Score | Co-LA (n = 42) | Indeterminate Co-LA (n = 13) | No Co-LA (n = 77) |
| 0 | 0 (0%) | 3 (9%) | 30 (91%) |
| 1 | 0 (0%) | 2 (18%) | 9 (82%) |
| 2 | 4 (15%) | 5 (18%) | 18 (67%) |
| 3 | 5 (36%) | 2 (14%) | 7 (50%) |
| 4 | 24 (67%) | 1 (3%) | 11 (30%) |
| 5 | 9 (82%) | 0 (0%) | 2 (18%) |
Co-LA, post-COVID-19 lung abnormalities.
3.5. Co-LA score
Out of 132 patients, 42 patients (33%) developed Co-LA on the follow-up CT scan obtained within 6–24 months. Co-LA Scores were 0 in 77 (58%), 1 in 13 (10%), and 2 in 42 (32%). (Table 3) Out of 42 patients with Co-LA, 22 patients had fibrotic Co-LA and 20 had non-fibrotic Co-LA. Interobserver agreement of Co-LA was substantial (weight kappa score = 0.71).
3.6. Pneumonia score vs Co-LA score
The contingency table between Pneumonia Score and Co-LA score is shown in Table 3. Co-LA developed in 0 out of 33 patients (0%) with Pneumonia Score = 0, 0 out of 11 patients (0%) with Pneumonia Score = 1, 4 out of 27 patients (15%) with Pneumonia Score = 2, 5 out of 14 patients (36%) with Pneumonia Score = 3, 24 out of 36 patients (67%) with Pneumonia Score = 4, and 9 out of 11 patients (82%) with Pneumonia Score = 5. The higher the Pneumonia Score, the more patients developed Co-LA.
3.7. Pneumonia severity categories vs Co-LA
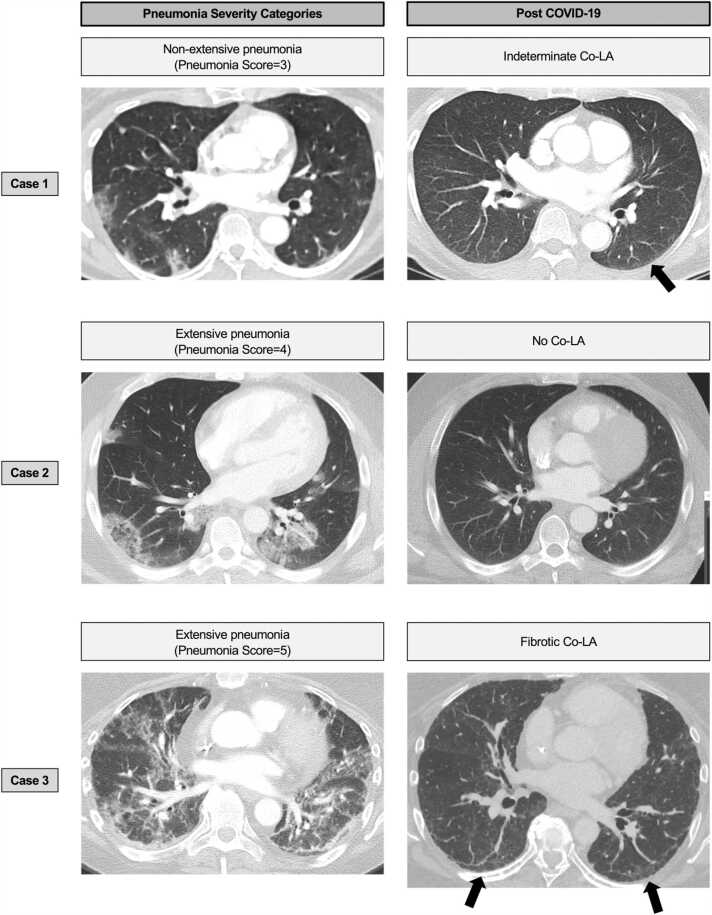
Pneumonia Severity Categories and the presence/absence of Co-LA and fibrosis is summarized in Table 4. Out of 47 patients with extensive pneumonia, 33 patients (70%) developed Co-LA, of which 18 (55%) out of 33 developed fibrotic Co-LA. Out of 52 patients with non-extensive pneumonia, 9 patients (17%) developed Co-LA, of which 7 (78%) out of 9 developed non-fibrotic Co-LA. Out of 33 patients with no pneumonia, 0 patients (0%) developed Co-LA. Pneumonia Severity Categories demonstrated the different distribution, which was confirmed in every two-group comparison (P < 0.001). (Table 4, Fig. 3).
Table 4.
Pneumonia Severity Categories vs Co-LA Scores with presence/absence of fibrosis at 6–24 months after diagnosis.
| Presence/absence of Co-LA at follow-up CT, n (%) |
||||
|---|---|---|---|---|
| Pneumonia Severity Categories | Fibrotic Co-LA (n = 20) | Non-fibrotic Co-LA (n = 22) | Indeterminate Co-LA (n = 13) | No Co-LA (n = 77) |
| No pneumonia | 0 (0%) | 0 (0%) | 3 (9%) | 30 (91%) |
| Non-extensive pneumonia | 2 (4%) | 7 (14%) | 9 (17%) | 34 (65%) |
| Extensive pneumonia | 18 (38%) | 15 (32%) | 1 (2%) | 13 (28%) |
Co-LA, post-COVID-19 lung abnormalities.
Fig. 3.
Pneumonia Severity Categories vs Co-LA. Case 1. 81-year-old woman without vaccination, lung disease, or smoking history. Initial CT was performed one day after diagnosis. Focal consolidation or ground-glass opacities (GGOs) were confirmed mainly in peripheral lesions, which is consistent with Pneumonia Score 3. On CT scan performed 22 months after diagnosis, minimal subpleural GGOs were seen in the subpleural region of left lower lobe, which is consistent with indeterminate Co-LA. Case 2. 42-year-old woman without lung disease or smoking history. Initial CT scan was performed three days after diagnosis. Multiple consolidation or ground-glass opacities (GGOs), including reversed halo pattern, were confirmed mainly in peripheral areas. This case is consistent with Pneumonia Score 4. On CT scans obtained nine months after diagnosis, all the opacities have resolved, which is consistent with no Co-LA. Case 3. 56-year-old woman with unknown smoking history, and without vaccination or lung disease. CT performed 22 days after diagnosis showed diffuse GGOs or consolidations accompanying focal spared areas, which is consistent with Pneumonia Score 5. On CT scan obtained 16.5 months after diagnosis, subpleural linear opacity, GGO, and interlobular septal thickening remained in subpleural areas. Mild traction bronchiolectasis was also identified, consistent with fibrotic Co-LA. Co-LA, post-COVID-19 lung abnormalities; GGO, ground-glass opacity.
Similar analyses were performed in the patients with CT scans in 12–24 months (Table S3, n = 63). The results were consistent among three analyses with different timeframes of follow-up CT scans.
3.8. Association of pneumonia score with Co-LA score
A multivariable linear regression analysis of the continuous Co-LA Score on Pneumonia Score (continuous), adjusted for age, sex, BMI, and smoking history, was performed; see the results in Table 5. Pneumonia Score was significantly associated with the Co-LA Score (P < 0.001) in the presence of these common confounders, confirming the results reported in Table 4.
Table 5.
Multiple linear regression analysis of Co-LA Score on Pneumonia Score, age, sex, BMI, and smoking history.
| Estimate | Standard error | t value | P value | |
|---|---|---|---|---|
| (Intercept) | -0.166 | 0.503 | -0.331 | 0.74 |
| Age | 0.005 | 0.005 | 1.031 | 0.31 |
| Sex | 0.030 | 0.134 | 0.225 | 0.82 |
| BMI | -0.006 | 0.009 | -0.670 | 0.50 |
| Smoking history | -0.074 | 0.135 | -0.552 | 0.58 |
| Pneumonia Score | 0.326 | 0.039 | 8.377 | < 0.001 |
Co-LA, post-COVID-19 lung abnormalities.
4. Discussion
In this study, we present information about the natural progression of Covid pneumonia, reporting data on 132 COVID-19 patients who had at least one available CT scan within a 6-week window (−2 to +4 weeks) of the diagnosis and at least one follow-up chest CT scan more than six months after diagnosis. Of these 132 patients, one-third eventually developed Co-LA: 70% of the patients who originally had extensive pneumonia, 17% of those with non-extensive pneumonia, and 0% of those with no pneumonia. Of the 47 patients with extensive pneumonia, 18 (38%) went on to develop fibrotic Co-LA. To our knowledge, previous reports from China and Europe studied fewer than 120 patients with up to one year following SARS-CoV-2 infection [4], [5], [6], [7], [8], [9], [10], [11], [15]. The present study represents the first report from a US academic medical center. In addition, this study is unique because the study population included a wider spectrum of patients who presented with a varying degrees of initial lung involvement, ranging from no pneumonia to severe pneumonia. In contrast, prior studies investigated only hospitalized patients, mostly with severe pneumonia. This retrospective and observational study may be valuable when SARS-CoV-2 is changing and evolving constantly, which makes prospective study difficult to plan.
Both the Pneumonia Severity Categories and the Co-LA Score had substantial (weight kappa score >0.7) interobserver agreements and were considered reproducible, which is one of the important characteristics for the ‘good imaging criteria’. The Pneumonia Score captured the severity of the pneumonia more accurately than the Initial CT Score, because CT findings of pneumonia sometimes occur with a delay from the diagnosis of SARS-CoV-2 infection. (Table S2) We avoided using ILA terminology in COVID-19 patients because ILA is defined as an incidental finding [12]. Instead, we coined the term ‘post-COVID-19 lung abnormalities’ (Co-LA) for the CT findings observed following SARS-CoV-2 infection, even though the image interpretation approach was similar to that employed with ILA [12]. Indeed, the CT findings after COVID-19 pneumonia in some patients were very similar to those seen in ILD [12]. To avoid confusion, the time frame described represents the period from date of the diagnosis of SARS-CoV2 infection throughout the manuscript.
Of interest, there were no statistically significant differences in age, sex or smoking status among the three groups of Co-LA, Intermediate Co-LA, and No Co-LA. As expected, there was highly significant difference in hospital stays, hospitalization, ICU admission, and ARDS. There were large numbers of patients with underlying lung disease, malignancy, and other comorbidities, as could be expected for a study at a tertiary academic center. Of note, there was a decrease in platelets in the Co-LA group, whereas no significant difference was observed in PE or D-dimer level among the three groups of Co-LA, Intermediate Co-LA, or No Co-LA. Furthermore, no significant difference was observed in the prevalence of lung disease, malignancy, and other comorbidities among these groups. There was no significant difference in all-cause mortality, presumably because only long-term survivors could be selected by the present study design.
When the relationship between Pneumonia Score and Co-LA at follow-up CT was investigated in Table 2, more than half of the patients with Pneumonia Score 4 and 5 developed Co-LA. Therefore, we categorized Pneumonia Score 4 and 5 as extensive pneumonia in the Pneumonia Severity Categories. The patients with Pneumonia Score 1–3 were further categorized as non-extensive pneumonia in the Pneumonia Severity Categories. Thus, we developed a contingency table between Pneumonia Severity Categories and Co-LA in Table 3. The subsequent results clearly demonstrated: (1) that 70% of patients with extensive pneumonia developed Co-LA, whereas none of the patients without pneumonia developed Co-LA: and (2) that 38% of patients with extensive pneumonia developed fibrotic Co-LA, whereas 4% of patients with non-extensive pneumonia developed fibrotic Co-LA and none of the patients without pneumonia developed fibrotic Co-LA.
In June 2020, autopsy cases of COVID-19 with pulmonary fibrosis were reported; acute DAD with fibrosis accompanying fibroblast and honeycombing-like remodeling was seen in histopathological specimen of lungs [18]. In COVID-19 cases, remodeling reactions of lungs to ARDS/DAD, cytokine storm, thromboembolism or mechanical ventilation can induce fibroblasts, followed by subsequent pulmonary fibrosis [19], [20], [21]. COVID-19 pulmonary fibrosis shares a variety of characteristics with idiopathic pulmonary fibrosis (IPF), which strengthens the association between post COVID-19 pulmonary fibrosis and IPF [22], [23].
CT images of the case leading to autopsy showed traction bronchiectasis, septal thickening, consolidation with parenchymal change, suggesting pulmonary fibrosis [18]. Notably, recent studies have shown that more than one third of severe COVID-19 patients had fibrotic-change progression in six-month follow-up chest CTs [4], [5]. Luger et al. have reported that reticulation did not diminish at one-year follow-up [11], [15]. Our similar analyses in patients with CT scans in 6–24 months (Table 4, n = 132) and in 12–24 months (Table S3, n = 63) reconfirmed the persistent nature of the Co-LA throughout analyses with two different timeframes.
The long-term prognosis of patients with ARDS is poor despite improving mechanical ventilation therapy [24]. COVID-19 infection and DAD with other causes share the indistinguishable pathological presentation of DAD [25]. It is well known that ARDS is one of the etiologies for the subsequent development of fibrotic lung disease. In our study, it was not possible to distinguish between the effect of severe COVID-19 pneumonia and the effect of ARDS and associated high-concentration oxygen administration mechanical ventilation, since the majority of the patients with COVID-19 extensive pneumonia had ICU admission and ARDS.
Several papers have reported that COVID-19 and IPF share genetic signals [26], [27]. However, the association of genetic variants of MUC5B, linked to IPF and ILA, seems to confer protection against COVID-19, although the combined effect of all other IPF risk loci seem to bring higher risks for COVID-19 severity [26], [27], [28]. It is important to investigate whether patients who developed fibrotic Co-LA might share common genetic variants with IPF patients.
This study had several limitations. First, it is a retrospective study from a single institution in the United States. Confirmation of the results by multi-center and international studies will be needed in the future. Furthermore, the underlying genetic predisposition of patients were not included in this study. We plan to investigate the relationship between potential genetic markers and the development of Co-LA as a next step. In this retrospective study, 106 (64%) out of 166 patients had pulmonary CT angiography for suspected PE inpatients with prolonged elevation of D-dimer value and often with symptoms of dyspnea. This helps explain why these patients had one or more further CT scans prescribed in the next 6–24 months. On the other hand, those who had died or discharged hopelessly never obtained the follow-up CT studies. Out of 528 patients with an initial CT scan, only 132 (25%) received the further CT scan(s) needed for inclusion in this study, which may have created a potential bias. It was not possible to differentiate between the consequence of severe COVID-19 pneumonia versus ventilator-related oxygen toxicity or lung injury, because many of the patients with severe COVID-19 pneumonia did have intubation with ventilator assistance at ICU. It could have been more desirable to have excluded ventilated patients focusing on moderate disease: it is conceivable that mild-moderate pneumonia will be the most frequent forms following the wide spread of vaccination. It is also necessary to conduct a future study with a longer observational period to distinguish fibrotic-like scars as sequelae of acute inflammatory damage from slowly progressive fibrosing tissue remodeling that may have a longer course and more subtle evolution.
In conclusion, higher severity of pneumonia at diagnosis was associated with increased likelihood of subsequent development of Co-LA at 6–24 months following SARS-CoV-2 infection. In patients with no pneumonia noted on initial CT scans, none developed Co-LA. Assessment of the presence and severity of pneumonia at diagnosis may help optimize monitoring and surveillance strategies for COVID-19 patients, especially with regard to the development of Co-LA.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest/Funding
MN is supported by R01CA203636, U01CA209414, R01HL111024, and R01CA240592. BM is supported by R01EB030470. BDL is supported by 1OT2HL162087. KMK is supported by AI150575, AI165382, and DE025208. HH is supported by NIH R01CA203636, 5U01CA209414, NIH/NHLBI, 2R01HL111024, NIH R01HL135142, and NIH/NHLBI 1R01HL130974. They receive the grant or personal fee outside the submitted work. The other co-authors have nothing to disclose.
Ethical statement
This study was approved by the National Bioethics Committee in Iceland (IRB#2021P000981) and the institutional review boards of Brigham and Women’s Hospital. Written informed consents were obtained from all the participants. This study does not include animal experiment. We use only demographic and image data. Thus, we state that this study is based on the declaration of Helsinki.
CRediT authorship contribution statement
Takuya Hino: Conceptualization, Methodology, Investigation, Formal analysis, Data curation, Formal analysis, Writing − original draft preparation, Visualization. Mizuki Nishino: Methodology, Investigation, Writing − review & editing. Vladimir I. Valtchinov: Resource, Data curation, Writing − review & editing. Staci Gagne: Methodology, Writing − review & editing. Elizabeth Gay: Writing − review & editing. Noriaki Wada: Writing − review & editing. Shu Chi Tseng: Writing − review & editing. Bruno Madore: Methodology, Writing − review & editing. Kousei Ishigami: Writing − review & editing. Yi Li: Methodology, Formal analysis, Writing − review & editing. David C. Christiani: Methodology, Resource, Formal analysis, Writing − review & editing. Charles R. G. Guttmann: Writing − review & editing. Gary M. Hunninghake: Methodology, Writing − review & editing. Bruce D. Levy: Writing − review & editing. Kenneth M. Kaye: Methodology, Writing − review & editing. Hiroto Hatabu: Conceptualization, Methodology, Resources, Investigation, Formal analysis, Writing − original draft preparation, Project administration, Supervision.
Footnotes
Institution from which the work originated: Brigham and Women’s Hospital and Harvard Medical School, 75 Francis Street, Boston, MA 02115, USA.
Supplementary data associated with this article can be found in the online version at doi:10.1016/j.ejro.2023.100483.
Appendix A. Supplementary material
Supplementary material
.
References
- 1.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R., Niu P., Zhan F., Ma X., Wang D., Xu W., Wu G., Gao G.F., Tan W. China Novel Coronavirus Investigating and Research Team, A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Naming the coronavirus disease (COVID-19) and the virus that causes it. World Health Organization (WHO) [Published online February 10, 2020]. 〈https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it〉. Accessed Oct 14th, 2022.
- 3.Simpson S., Kay F.U., Abbara S., Bhalla S., Chung J.H., Chung M., Henry T.S., Kanne J.P., Kligerman S., Ko J.P., Litt H. Radiological Society of North America Expert Consensus Statement on Reporting Chest CT Findings Related to COVID-19. Endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA - Secondary Publication. J. Thorac. Imaging. 2020;35:219–227. doi: 10.1097/RTI.0000000000000524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Han X., Fan Y., Alwalid O., Li N., Jia X., Yuan M., Li Y., Cao Y., Gu J., Wu H., Shi H. Six-month follow-up chest CT findings after severe COVID-19 pneumonia. Radiology. 2021;299:E177–E186. doi: 10.1148/radiol.2021203153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Caruso D., Guido G., Zerunian M., Polidori T., Lucertini E., Pucciarelli F., Polici M., Rucci C., Bracci B., Nicolai M., Cremona A., De Dominicis C., Laghi A. Post-Acute Sequelae of COVID-19 Pneumonia: Six-month Chest CT Follow-up. Radiology. 2021;301:E396–E405. doi: 10.1148/radiol.2021210834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Huang C., Huang L., Wang Y., Li X., Ren L., Gu X., Kang L., Guo L., Liu M., Zhou X., Luo J., Huang Z., Tu S., Zhao Y., Chen L., Xu D., Li Y., Li C., Peng L., Li Y., Xie W., Cui D., Shang L., Fan G., Xu J., Wang G., Wang Y., Zhong J., Wang C., Wang J., Zhang D., Cao B. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021;397:220–232. doi: 10.1016/S0140-6736(20)32656-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vijayakumar B., Tonkin J., Devaraj A., Philip K.E.J., Orton C.M., Desai S.R., Shah P.L. CT Lung Abnormalities after COVID-19 at 3 Months and 1 Year after Hospital Discharge. Radiology. 2022;303:444–454. doi: 10.1148/radiol.2021211746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pan F., Yang L., Liang B., Ye T., Li L., Li L., Liu D., Wang J., Hesketh R.L., Zheng C. Chest CT Patterns from Diagnosis to 1 Year of Follow-up in Patients with COVID-19. Radiology. 2022;302:709–719. doi: 10.1148/radiol.2021211199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen Y., Ding C., Yu L., Guo W., Feng X., Yu L., Su J., Xu T., Ren C., Shi D., Wu W., Yi P., Liu J., Tao J., Lang G., Li Y., Xu M., Sheng J., Li L., Xu K. One-year follow-up of chest CT findings in patients after SARS-CoV-2 infection. BMC Med. 2021;19:191. doi: 10.1186/s12916-021-02056-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yin X., Xi X., Min X., Feng Z., Li B., Cai W., Fan C., Wang L., Xia L. Long-term chest CT follow-up in COVID-19 Survivors: 102-361 days after onset. Ann. Transl. Med. 2021;9:1231. doi: 10.21037/atm-21-1438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Luger A.K., Sonnweber T., Gruber L., Schwabl C., Cima K., Tymoszuk P., Gerstner A.K., Pizzini A., Sahanic S., Boehm A., Coen M., Strolz C.J., Wöll E., Weiss G., Kirchmair R., Feuchtner G.M., Prosch H., Tancevski I., Löffler-Ragg J., Widmann G. Chest CT of Lung Injury 1 Year after COVID-19 Pneumonia: The CovILD Study. Radiology. 2022;304:462–470. doi: 10.1148/radiol.211670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hatabu H., Hunninghake G.M., Richeldi L., Brown K.K., Wells A.U., Remy-Jardin M., Verschakelen J., Nicholson A.G., Beasley M.B., Christiani D.C., San José Estépar R., Seo J.B., Johkoh T., Sverzellati N., Ryerson C.J., Graham Barr R., Goo J.M., Austin J.H.M., Powell C.A., Lee K.S., Inoue Y., Lynch D.A. Interstitial lung abnormalities detected incidentally on CT: a Position Paper from the Fleischner Society. Lancet Respir. Med. 2020;8:726–737. doi: 10.1016/S2213-2600(20)30168-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hata A., Schiebler M.L., Lynch D.A., Hatabu H. Interstitial lung abnormalities: state of the art. Radiology. 2021;301:19–34. doi: 10.1148/radiol.2021204367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Desai S.R., Wells A.U., Rubens M.B., Evans T.W., Hansell D.M. Acute respiratory distress syndrome: CT abnormalities at long-term follow-up. Radiology. 1999;210:29–35. doi: 10.1148/radiology.210.1.r99ja2629. [DOI] [PubMed] [Google Scholar]
- 15.Han X., Fan Y., Alwalid O., Zhang X., Jia X., Zheng Y., Shi H. Fibrotic interstitial lung abnormalities at 1-year follow-up CT after severe COVID-19. Radiology. 2021;301:E438–E440. doi: 10.1148/radiol.2021210972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jin C., Tian C., Wang Y., Wu C.C., Zhao H., Liang T., Liu Z., Jian Z., Li R., Wang Z., Li F., Zhou J., Cai S., Liu Y., Li H., Li Z., Liang Y., Zhou H., Wang X., Ren Z., Yang J. A Pattern Categorization of CT Findings to Predict Outcome of COVID-19 Pneumonia. Front Public Health. 2020;8 doi: 10.3389/fpubh.2020.567672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee K.S. Pneumonia associated with 2019 novel coronavirus: Can computed tomographic findings help predict the prognosis of the disease? Korean J. Radiol. 2020;21:257–258. doi: 10.3348/kjr.2020.0096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schwensen H.F., Borreschmidt L.K., Storgaard M., Redsted S., Christensen S., Madsen L.B. Fatal pulmonary fibrosis: a post-COVID-19 autopsy case. J. Clin. Pathol. 2021;74:400–402. doi: 10.1136/jclinpath-2020-206879. [DOI] [PubMed] [Google Scholar]
- 19.Yim J., Lim H.H., Kwon Y. COVID-19 and pulmonary fibrosis: therapeutics in clinical trials, repurposing, and potential development. Arch. Pharm. Res. 2021;44:499–513. doi: 10.1007/s12272-021-01331-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ambardar S.R., Hightower S.L., Huprikar N.A., Chung K.K., Singhal A., Collen J.F. Post-COVID-19 Pulmonary Fibrosis: Novel Sequelae of the Current Pandemic. J. Clin. Med. Res. 2021;10:2452. doi: 10.3390/jcm10112452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.A.E. John, C. Joseph, G. Jenkins, A.L. Tatler, COVID-19 and pulmonary fibrosis: A potential role for lung epithelial cells and fibroblasts, Immunol. Rev. 302, 2021: 228–240. [DOI] [PMC free article] [PubMed]
- 22.Rai D.K., Sharma P., Kumar R. Post covid 19 pulmonary fibrosis. Is it real threat? Indian J. Tube. 2021;68:330–333. doi: 10.1016/j.ijtb.2020.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Crisan-Dabija R., Pavel C.A., Popa I.V., Tarus A., Burlacu A. “A Chain Only as Strong as Its Weakest Link”: An Up-to-Date Literature Review on the Bidirectional Interaction of Pulmonary Fibrosis and COVID-19. J. Proteome Res. 2020;19:4327–4338. doi: 10.1021/acs.jproteome.0c00387. [DOI] [PubMed] [Google Scholar]
- 24.Chiumello D., Coppola S., Froio S., Gotti M. What’s next after ARDS: long-term outcomes. Respir. Care. 2016;61:689–699. doi: 10.4187/respcare.04644. [DOI] [PubMed] [Google Scholar]
- 25.Konopka K.E., Nguyen T., Jentzen J.M., Rayes O., Schmidt C.J., Wilson A.M., Farver C.F., Myers J.L. Diffuse alveolar damage (DAD) resulting from coronavirus disease 2019 Infection is Morphologically Indistinguishable from Other Causes of DAD. Histopathology. 2020;77:570–578. doi: 10.1111/his.14180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Allen R.J., Guillen-Guio B., Croot E., Kraven L.M., Moss S., Stewart I., Jenkins R.G., Wain L.V. Genetic overlap between idiopathic pulmonary fibrosis and COVID-19. Eur. Respir. J. 2022;60 doi: 10.1183/13993003.03132-2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fadista J., Kraven L.M., Karjalainen J., Andrews S.J., Geller F., COVID-19 Host Genetics Initiative. Baillie J.K., Wain L.V., Jenkins R.G., Feenstra B. Shared genetic etiology between idiopathic pulmonary fibrosis and COVID-19 severity. EBioMedicine. 2021;65 doi: 10.1016/j.ebiom.2021.103277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hunninghake G.M., Hatabu H., Okajima Y., Gao W., Dupuis J., Latourelle J.C., Nishino M., Araki T., Zazueta O.E., Kurugol S., Ross J.C., San José Estépar R., Murphy E., Steele M.P., Loyd J.E., Schwarz M.I., Fingerlin T.E., Rosas I.O., Washko G.R., O’Connor G.T., Schwartz D.A. MUC5B promoter polymorphism and interstitial lung abnormalities. N. Engl. J. Med. 2013;368:2192–2200. doi: 10.1056/NEJMoa1216076. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material