Abstract
Introduction
Cardiovascular disease (CVD) is the leading cause of mortality in type 1 diabetes (T1D). However, there is a need for daily practice tools for identifying those more prone to suffer from these events. We aimed to assess the relationships between nuclear magnetic resonance (1H NMR)-based lipidomic analysis and several CVD risk variables (including preclinical carotid atherosclerosis) in individuals with T1D at high risk.
Methods
We included patients with T1D without CVD, with at least one of the following: age ≥ 40 years, diabetic kidney disease, or ≥ 10 years of evolution with another risk factor. The presence of plaque (intima-media thickness > 1.5 mm) was determined by standardized ultrasonography protocol. Lipidomic analysis was performed by 1H NMR. Bivariate and multivariate-adjusted differences in 1H NMR lipidomics were evaluated.
Results
We included n = 131 participants (49.6% female, age 46.4 ± 10.3 years, diabetes duration 27.0 ± 9.5 years, 47.3% on statins). Carotid plaques were present in 28.2% of the individuals (n = 12, with ≥ 3 plaques). Glucose (HbA1c), anthropometric (body mass index and waist circumference), and insulin resistance-related (fatty liver index and estimated glucose disposal rate) variables were those most associated with 1H NMR-derived lipidomic analysis (p < 0.01 for all). Regarding preclinical atherosclerosis, sphingomyelin was independently associated with carotid plaque presence (for 0.1 mmol/L increase, OR 0.50 [0.28–0.86]; p = 0.013), even after adjusting for age, sex, hypertension, statin use, mean 5-year HbA1c and diabetes duration. Furthermore, linoleic acid and ω-6 fatty acids remained independently associated with higher plaque burden (≥ 3 plaques) in multivariate models (0.17 [0.03–0.93] and 0.27 [0.07–0.97], respectively; p < 0.05 for both).
Conclusion
In our preliminary study of individuals with T1D at high risk, several 1H NMR-derived lipidomic parameters were independently associated with preclinical atherosclerosis. Specifically, ω-6 fatty acids and linoleic acid seem promising for identifying those with higher plaque burden.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13300-023-01372-x.
Keywords: Type 1 diabetes, Preclinical atherosclerosis, Vascular ultrasonography, Nuclear magnetic resonance, Lipidomic analysis
Key Summary Points
| Why carry out this study? |
| Cardiovascular disease is the leading cause of death among the type 1 diabetes population. However, the tools available in daily practice for identifying those more prone to suffer from these events are suboptimal. |
| This exploratory study aimed to assess the relationships between nuclear magnetic resonance-derived lipidomic variables and several cardiometabolic traits (including preclinical carotid atherosclerosis) among these individuals. |
| What was learned from the study? |
| Several lipidomic variables were independently associated with preclinical carotid atherosclerosis, even after adjusting for some confounders such as age, sex, hypertension, statin use, mean 5-year HbA1c and diabetes duration. Specifically, linoleic acid and ω-6 fatty acids seem promising in identifying those with higher plaque burden (≥ 3 carotid plaques). |
| Since the identification of individuals with T1D at higher risk for developing future CVD is challenging in daily practice, the lipidomic analysis seems promising in this field. Further studies with larger sample size and/or longitudinal designs are needed to confirm our preliminary results. |
Introduction
Cardiovascular disease (CVD) is still the leading cause of death among the type 1 diabetes population (T1D) [1, 2]. Although glycaemic control has been strongly associated with this increased risk, even in those with glucose levels meeting goals, cardiovascular death is threefold higher [3]. Thus, extra-glycaemic parameters are probably involved in this accelerated atherosclerosis. In addition to several conventional [4] and non-conventional [5] cardiovascular risk factors, circulating lipids are thought to play an important role in CVD pathogenesis [6, 7]. In fact, in a large cohort study, an increase of 1 mmol/L of low density lipoprotein-cholesterol (LDL-cholesterol) was associated with a 35–50% greater risk of overall CVD [8]. Further, our study group has recently shown that individuals with T1D had some subtle lipoprotein metabolism derangements that could be associated with atherosclerosis progression [9].
Lipidomic analysis is a new technique that identifies a broad range of lipid species, which could uncover some variables potentially associated with chronic complications in this population [10]. Preliminary data from our group also showed that some dietary-related lipid parameters could identify individuals with T1D more prone to suffer from CVD [11, 12]. However, the information regarding lipidomic-related variables with preclinical atherosclerosis, especially in Mediterranean countries, is scarce. Against this background, in this exploratory analysis, we aimed to assess the relationships between nuclear magnetic resonance (1H NMR) lipidomics and several CVD risk variables (including preclinical carotid atherosclerosis), in primary prevention individuals with T1D at high risk.
Methods
Study Participants
We performed a cross-sectional study in individuals with T1D followed at the diabetes unit of a tertiary hospital. All subjects were recruited from January 2016 to November 2018. We included participants with no previous history of CVD (coronary artery disease, ischemic stroke, peripheral artery disease and/or heart failure) and high CVD risk according to the main CVD prevention guidelines, as previously stated [13–15]: (a) age ≥ 40 years; (b) presence of any stage of diabetic kidney disease; (c) or ≥ 10 years of diabetes duration and at least one additional CVD risk factor. The following CVD risk factors were considered: family history of premature CVD (defined as < 55 years in men and < 65 years in women [16]), active smoking habit, hypertension, low high density lipoprotein-cholesterol (HDL-cholesterol) levels (< 40 mg/dL in men, < 45 mg/dL in women), high triglycerides levels (> 150 mg/dL), being already on statins, a former episode of preeclampsia/eclampsia, the presence of diabetic retinopathy, impaired hypoglycaemia awareness or a previous episode of severe hypoglycaemia in the last 2 years. Patients with clinical suspicion of latent autoimmune diabetes in adults (LADA), monogenic diabetes or prior pancreatic diseases (pancreatitis, surgery) were excluded.
The study protocol was conducted according to the principles of the Declaration of Helsinki and approved by the hospital Research Ethics Committees (HCB/2017/0977). Patients were included after giving their written informed consent.
Clinical and Laboratory Measures
Clinical data such as age, gender, family history of premature CVD in first-degree relatives, smoking habit (never, former or active), the presence of hypertension, as well as the use of certain medications (antihypertensive, lipid-lowering or antiplatelet) were registered. T1D-specific clinical information was also recorded: diabetes duration (years), current insulin therapy (continuous subcutaneous insulin infusion [CSII] or multiple daily insulin injection therapy [MDI]), presence of retinopathy and/or nephropathy, and history of severe hypoglycaemia or impaired hypoglycaemia awareness (Clarke test score > 3 points [17, 18]).
Anthropometric measures including height, weight, waist and hip circumference were also recorded and obtained with standard methods. Body mass index (BMI) was calculated as weight in kilograms divided by square height in metres. Blood pressure was registered using a blood pressure monitor after a few minutes of rest.
Laboratory data were obtained in the local laboratory using standardized assays to measure serum creatinine, urinary albumin-to-creatinine ratio (ACR), glycated haemoglobin (HbA1c; high-performance liquid chromatography and expressed in National Glycohemoglobin Standardization Program/Diabetes Control and Complications Trial units), fasting glucose and lipid profile (total cholesterol, triglycerides and HDL-cholesterol by direct methods; LDL-cholesterol by the Friedewald formula). The estimated glomerular filtration rate (eGFR) was obtained by the Chronic Kidney Disease-Epidemiology Collaboration equation (CKD-EPI) [19]. The HbA1c values available for each patient were used to calculate the mean HbA1c in the last 5 years. Insulin sensitivity was evaluated with the estimated glucose disposal rate (eGDR) formula, a well-validated score in T1D expressed in mg/kg/min [16, 20] and calculated as follows: 24.31 − 12.22 × waist-to-hip ratio − 3.29 × hypertension (0 = no, 1 = yes) − 0.57 × HbA1c (in %). Lastly, the fatty liver index (FLI) was calculated as described previously [21].
Diabetic kidney disease was considered if ACR was ≥ 30 mg/g or eGFR was < 60 ml/min/1.73 m2; or the use of angiotensin-converting enzyme inhibitors (ACEi) or angiotensin II receptor blockers (ARB), with no history of hypertension or CVD. The diagnosis of diabetic retinopathy was obtained from medical records and was always verified by an ophthalmologist. Hypertension was defined as consecutive determinations of systolic blood pressure ≥ 140 mmHg and diastolic blood pressure ≥ 90 mmHg or being treated with antihypertensive drugs (except those on ACEi/ARB for diabetic nephropathy).
1H NMR Lipidomic Analysis
1H NMR lipidomic analysis was performed as previously stated [12]. Lipophilic extracts were obtained from two 100-μL aliquots of freshly thawed plasma using the BUME method [22] with slight modifications. BUME was optimized for batch extractions with diisopropyl ether (DIPE) replacing heptane as the organic solvent. This procedure was performed with a BRAVO liquid-handling robot which can extract 96 samples at once. The upper lipophilic phase was completely dried in Speedvac until evaporation of organic solvents and frozen at − 80 °C until 1H NMR analysis. Lipid extracts were reconstituted in a solution of CDCl3/CD3OD/D2O (16:7:1, v/v/v) containing tetramethylsilane (TMS) at 1.18 mM as a chemical shift reference and transferred into 5-mm NMR glass tubes. 1H NMR spectra were measured at 600.20 MHz using an Avance III-600 Bruker spectrometer. A 90° pulse with water presaturation sequence (zgpr) was used. Quantification of lipid signals in 1H NMR spectra was carried out with LipSpin [23], in-house software based on Matlab (MATLAB. version 7.10.0 (R2010a); Natick, Massachusetts: The MathWorks Inc.; 2010.). Resonance assignments were based on values in the literature [24].
Carotid B-mode Ultrasound Imaging
Bilateral carotid artery B-mode ultrasound imaging was performed following a standardized protocol, as described previously [14, 15]. Acuson X300 and Acuson X700 (Siemens) ultrasound systems equipped with a linear transducer (frequency range 5–10 MHz) were used. Carotid intima-media thickness (IMT) was measured offline by semiautomatic software by the same experienced researcher (A.J.A.). The plaque was visualized using B-mode and colour Doppler examinations in both longitudinal and transverse planes to evaluate the presence of circumferential asymmetry. Carotid (bulb, internal or common carotid) plaque was defined as focal wall thickening encroaching into the arterial lumen by at least 50% of the surrounding IMT value or with a thickness of at least 1.5 mm measured from the media adventitia interphase to the intima-lumen surface [25]. The mean and the mean-maximum of all the carotid territories were recorded, as well as the maximum height of the carotid plaque. When plaque was present, the maximum IMT equalled the highest plaque height.
Statistical Analysis
Sample size could not be calculated before the analyses because of the absence of previous data on the distribution of lipidomic parameters. As a post hoc analysis, we calculated the sample size to achieve an absolute difference of 10% between groups for the lipidomic parameters. Assuming a two-tailed alpha error of 0.025, we obtained a statistical potency of 91%.
Data are presented as mean ± standard deviation, median (interquartile range) or number (percentage). The normal distribution of continuous variables was assessed by the Kolmogorov–Smirnov test. Differences in clinical and laboratory variables according to preclinical carotid atherosclerosis status were assessed with appropriate parametric and non-parametric tests. Relationships between 1H NMR lipidomic variables and several dichotomous cardiometabolic traits, as well as carotid atherosclerosis, were assessed with the Mann–Whitney U test. Spearman correlation analyses were further used to assess relationships between 1H NMR-related variables and continuous cardiometabolic parameters.
As an exploratory analysis, we assessed the independent relationships between 1H NMR lipidomics and carotid atherosclerosis (presence of at least one plaque; presence of ≥ 3 plaques; dependent variables). To this end, logistic binary regression models were constructed (and odds ratios [OR] and 95% confidence interval were reported, accordingly). Different models were assessed: (a) model 1: age- and sex-adjusted; (b) model 2: model 1 + presence of hypertension and statin use; and (c) model 3: model 2 + mean HbA1c in the last 5 years and T1D duration. The level of significance was set at p < 0.05. All the analyses were performed with the SPSS 26.0 statistical package (Chicago, IL).
Results
Subject Characteristics
A total of 131 participants were included in the study. Overall, the proportion of men and women was similar, the mean age was 46.4 years and the mean diabetes duration was 27.0 years. Regarding CVD risk factors, 27.5% were active smokers, 29% had hypertension and 47.3% and 33.6% were on statins and ACEi/ARB, respectively. Microvascular complications were present in 43.5% of the participants (37.4% had retinopathy and 13.7% had diabetic kidney disease). The characteristics are summarised in Table 1.
Table 1.
Characteristics of the study participants (n = 131)
| Clinical characteristics | |
| Female | 65 (49.6) |
| Age (years) | 46.4 ± 10.3 |
| Premature CVD in first-degree relatives* | 25 (19.1) |
| Active smoking habit | 36 (27.5) |
| Cumulative smoking (pack-years) | 0 (0–12.5) |
| Hypertension | 38 (29.0) |
| SBP (mmHg) | 126 ± 16 |
| DBP (mmHg) | 80 ± 8 |
| BMI (kg/m2) | 25.9 ± 4.1 |
| Waist circumference (cm) | 90 ± 12 |
| Women | 85 ± 11 |
| Men | 95 ± 11 |
| T1D duration (years) | 27.0 ± 9.5 |
| Diabetic kidney disease | 18 (13.7) |
| Diabetic retinopathy | 49 (37.4) |
| CSII therapy | 55 (42.0) |
| Laboratory characteristics | |
| Fasting plasma glucose (mg/dL) | 151 ± 66 |
| Haemoglobin A1c (%) | 7.5 ± 0.9 |
| Mean haemoglobin A1c in the last 5 years (%) | 7.7 ± 0.9 |
| Serum creatinine (mg/dL) | 0.82 ± 0.17 |
| eGFR (CKD-EPI; ml/min/1.73 m2) | 97.8 ± 15.9 |
| ALT (UI/L) | 22 ± 11 |
| Total cholesterol (mg/dL) | 188 ± 32 |
| HDL-cholesterol (mg/dL) | 60 ± 16 |
| LDL-cholesterol (mg/dL) | 111 ± 24 |
| Triglycerides (mg/dL) | 70 (54–90) |
| Non-HDL cholesterol (mg/dL) | 128 ± 28 |
| Remnant cholesterol (mg/dL) | 14 (11–18) |
| Total cholesterol/HDL-cholesterol ratio | 3.30 ± 0.89 |
| Fatty liver index | 24.8 (11.5–54.7) |
| eGDR (mg/kg/min) | 8.92 (6.65–10.21) |
| Pharmacological treatment | |
| Statins | 62 (47.3) |
| ACEi/ARB | 44 (33.6) |
| Antiplatelet drugs | 8 (6.1) |
Data are shown as n (percentage), mean ± SD or median (Q1–Q3)
ACEi angiotensin-converting enzyme inhibitor, ALT alanine aminotransferase, ARB angiotensin receptor blocker, BMI body mass index, CVD cardiovascular disease, CSII continuous subcutaneous insulin infusion, DBP diastolic blood pressure, eGDR estimated glucose disposal rate, eGFR estimated glomerular filtration rate, SBP systolic blood pressure, T1D type 1 diabetes
*Defined as < 55 years in men and < 65 years in women
On vascular ultrasonography, 37 (28.2%) had at least one carotid plaque (n = 12, with three or more plaques). Those with plaques were older (52.4 ± 9.7 vs. 44.1 ± 9.6 years; p < 0.001), had an increased prevalence of hypertension (43.2 vs. 23.4%; p = 0.024), a higher clinical systolic blood pressure (132 ± 15 mmHg vs. 124 ± 15 mmHg; p = 0.009) and a longer diabetes duration (31.1 ± 11.5 vs. 25.4 ± 8.2 years; p = 0.008; Table S1 in the supplementary material). The same variables were also directly associated with the number of carotid plaques (none, 1–2 plaques, ≥ 3 plaques; p for trend < 0.05 for all comparisons), plus the use of ACEi/ARB (28.7 vs. 66.7%), and inversely associated with eGFR (99 ± 16 vs. 88 ± 14 ml/min/1.73 m2) and LDL-cholesterol (113 ± 21 vs. 87 ± 14 mg/dL; for none vs. ≥ 3 plaques; p for trend < 0.05 for all comparisons; Table S2 in the supplementary material).
1H NMR-Based Lipidomic Analysis in Relation to Cardiometabolic Traits
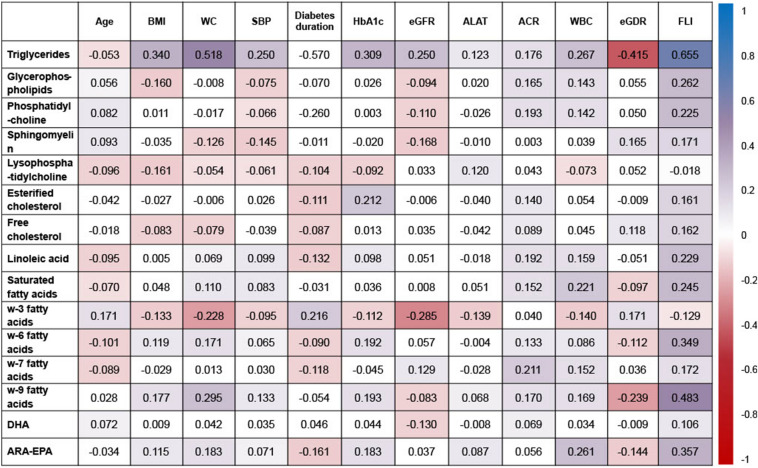
Whereas no differences were observed concerning age (Fig. 1), several between-gender differences were found: higher levels of 1H NMR triglycerides and ω-9 fatty acids, and lower levels of glycerophospholipids, phosphatidylcholine, sphingomyelin and ω-3 fatty acids were observed among male participants (p < 0.05; Table S3 in the supplementary material). Only minor differences were found regarding hypertension and smoking habit, whereas those on statins showed lower levels of linoleic and ω-6 fatty acids, and higher levels of sphingomyelin and arachidonic acid–eicosapentaenoic acid (ARA-EPA) (p < 0.05, Table S3). Among other adipose-related variables, FLI showed the strongest correlations with most of the 1H NMR-related lipidomic parameters, especially with 1H NMR triglycerides and ω-9 fatty acids (rs > 0.4 for both; Fig. 1). Finally, white blood cell count (as a marker of an inflammation-related variable) was directly associated with 1H NMR triglycerides, saturated fatty acids and ARA-EPA (rs = 0.2–0.3).
Fig. 1.
Associations between NMR lipidomics and clinical and laboratory parameters. Numbers in cells indicate Spearman’s R correlation index and the colour of each cell indicates the strength of association of that index according to the colour code shown in the right column. ACR albumin-to-creatinine ratio, ALAT alanine aminotransferase, BMI body mass index, eGDR estimated glucose disposal rate, eGFR estimated glomerular filtration rate, FLI fatty liver index, SBP systolic blood pressure, WBC white blood cells, WC waist circumference
Regarding T1D-specific risk factors, there were minor differences regarding diabetes duration and retinopathy status. However, HbA1c showed direct relationships with 1H NMR triglycerides, esterified cholesterol, ω-6 and ω-9 fatty acids, and ARA-EPA (p < 0.05 for all comparisons, Fig. 1). Further, only weak direct correlations were found with ACR (as a marker of diabetic kidney disease; rs = 0.1–0.2); and eGDR, as a marker of insulin sensitivity, was strongly and inversely associated with 1H NMR triglycerides (rs = − 0.415).
1H NMR-Based Lipidomic Analysis and Preclinical Carotid Atherosclerosis
Only levels of sphingomyelin were inversely associated with the presence of at least one carotid plaque (p < 0.05, Table 2), which maintained the statistical significance after adjusting for confounders such as age, sex, presence of hypertension, statin use, mean HbA1c in the last 5 years and diabetes duration (for 0.1 mmol/L increase, OR 0.50 [0.28–0.86]; p = 0.013; Table 3). When other variables associated with a higher plaque burden were assessed (presence of ≥ 3 plaques), inverse associations were found with esterified and free cholesterol, linoleic acid and ω-6 fatty acids (p < 0.05 for all comparisons; Table 2), which remained statistically associated in fully adjusted models (model 3; OR 0.055 (0.006–0.51), 0.009 (0.0–0.60), 0.17 (0.03–0.93) and 0.27 (0.07–0.97), for esterified cholesterol, free cholesterol, linoleic acid and ω-6 fatty acids, respectively; p < 0.05 for all; Table 3).
Table 2.
NMR lipidomics according to preclinical carotid atherosclerosis status
| Whole cohort (n = 131) | Presence of carotid plaque | Presence of ≥ 3 plaques | |||
|---|---|---|---|---|---|
| No (n = 94) | Yes (n = 37) | No (n = 119) | Yes (n = 12) | ||
| Triglycerides | 0.77 (0.51–1.08) | 0.76 (0.51–1.07) | 0.78 (0.50–1.43) | 0.77 (0.52–1.10) | 0.58 (0.40–1.01) |
| Glycerophospholipids | 2.49 (2.30–2.72) | 2.51 (2.29–2.69) | 2.39 (2.15–2.85) | 2.51 (2.27–2.74) | 2.21 (2.12–2.57) |
| Phosphatidylcholine | 2.40 (2.17–2.61) | 2.41 (2.18–2.59) | 2.35 (2.12–2.70) | 2.41 (2.18–2.62) | 2.26 (2.10–2.50) |
| Sphingomyelin | 0.61 (0.55–0.67) | 0.62 (0.56–0.69) | 0.57 (0.54–0.66)* | 0.62 (0.55–0.68) | 0.58 (0.55–0.61) |
| Lysophosphatidylcholine | 0.47 (0.41–0.53) | 0.48 (0.41–0.55) | 0.45 (0.41–0.51) | 0.48 (0.42–0.55) | 0.43 (0.40–0.46) |
| Esterified cholesterol | 4.65 (4.38–5.11) | 4.65 (4.43–5.09) | 4.67 (3.98–5.28) | 4.67 (4.43–5.19) | 4.38 (3.94–4.70)* |
| Free cholesterol | 2.08 (1.89–2.35) | 2.09 (1.94–2.35) | 2.08 (1.73–2.28) | 2.11 (1.91–2.36) | 1.90 (1.67–2.08)** |
| Linoleic acid | 4.14 (3.74–4.59) | 4.20 (3.80–4.58) | 4.06 (3.59–4.67) | 4.20 (3.77–4.61) | 3.67 (3.46–4.20)* |
| Saturated fatty acids | 4.77 (4.18–5.51) | 4.80 (4.20–5.44) | 4.63 (4.12–5.76) | 4.80 (4.17–5.51) | 4.65 (4.48–5.78) |
| ω-3 fatty acids | 0.24 (0.19–0.30) | 0.24 (0.19–0.30) | 0.25 (0.19–0.32) | 0.25 (0.19–0.30) | 0.22 (0.19–0.40) |
| ω-6 fatty acids | 2.73 (2.43–3.34) | 2.83 (2.49–3.37) | 2.60 (2.28–3.27) | 2.82 (2.47–3.35) | 2.30 (1.57–2.82)* |
| ω-7 fatty acids | 2.62 (2.27–3.05) | 2.63 (2.25–2.98) | 2.54 (2.34–3.28) | 2.63 (2.25–3.10) | 2.53 (2.41–2.80) |
| ω-9 fatty acids | 3.86 (3.09–4.79) | 3.79 (3.21–4.55) | 4.38 (2.73–5.57) | 3.86 (3.17–4.77) | 3.40 (2.15–4.83) |
| DHA | 0.088 (0.064–0.118) | 0.085 (0.065–0.114) | 0.097 (0.060–0.139) | 0.088 (0.065–0.114) | 0.115 (0.048–0.151) |
| ARA-EPA | 0.61 (0.51–0.82) | 0.61 (0.50–0.82) | 0.61 (0.51–0.81) | 0.61 (0.50–0.82) | 0.62 (0.57–0.75) |
Data are shown as median (Q1–Q3). Data are presented as mmol/L
ARA arachidonic acid, DHA docosahexaenoic acid, EPA eicosapentaenoic acid
*p < 0.05; **p < 0.01 between groups
Table 3.
Associations between NMR lipidomics and carotid atherosclerosis: multiple regression analysis
| Model 1* | Model 2* | Model 3* | ||||
|---|---|---|---|---|---|---|
| OR (95% CI) | p value | OR (95% CI) | p value | OR (95% CI) | p value | |
| Presence of carotid plaque | ||||||
| Triglycerides | 1.09 (0.73–1.63) | 0.674 | 1.09 (0.72–1.65) | 0.693 | 1.09 (0.72–1.64) | 0.697 |
| Glycerophospholipids | 0.65 (0.25–1.69) | 0.373 | 0.68 (0.26–1.80) | 0.435 | 0.67 (0.25–1.79) | 0.429 |
| Phosphatidylcholine | 0.64 (0.23–1.76) | 0.386 | 0.67 (0.24–1.86) | 0.444 | 0.64 (0.23–1.79) | 0.397 |
| Sphingomyelin | 0.52 (0.31–0.88) | 0.015 | 0.51 (0.30–0.89) | 0.018 | 0.50 (0.28–0.86) | 0.013 |
| Lysophosphatidylcholine | 0.78 (0.02–40.42) | 0.903 | 0.97 (0.02–53.45) | 0.989 | 1.29 (0.02–79.02) | 0.904 |
| Esterified cholesterol | 0.89 (0.45–1.74) | 0.734 | 0.91 (0.46–1.79) | 0.787 | 0.92 (0.45–1.88) | 0.824 |
| Free cholesterol | 0.50 (0.15–1.74) | 0.277 | 0.55 (0.16–1.94) | 0.352 | 0.54 (0.15–1.92) | 0.338 |
| Linoleic acid | 0.91 (0.53–1.55) | 0.721 | 0.92 (0.54–1.58) | 0.767 | 0.94 (0.55–1.61) | 0.814 |
| Saturated fatty acids | 0.91 (0.65–1.28) | 0.584 | 0.91 (0.65–1.28) | 0.580 | 0.90 (0.64–1.27) | 0.552 |
| ω-3 fatty acids | 1.83 (0.03–96.4) | 0.765 | 2.18 (0.04–120.0) | 0.704 | 1.48 (0.02–97.4) | 0.855 |
| ω-6 fatty acids | 0.82 (0.50–1.35) | 0.435 | 0.81 (0.49–1.35) | 0.421 | 0.81 (0.49–1.35) | 0.416 |
| ω-7 fatty acids | 1.40 (0.88–2.23) | 0.153 | 1.44 (0.90–2.31) | 0.125 | 1.47 (0.92–2.35) | 0.109 |
| ω-9 fatty acids | 1.05 (0.87–1.28) | 0.622 | 1.05 (0.87–1.28) | 0.596 | 1.06 (0.87–1.28) | 0.570 |
| DHA | 1.04 (0.94–1.14) | 0.443 | 1.05 (0.95–1.15) | 0.372 | 1.05 (0.95–1.16) | 0.369 |
| ARA-EPA | 0.98 (0.25–3.89) | 0.981 | 1.06 (0.26–4.38) | 0.932 | 1.11 (0.26–4.70) | 0.889 |
| Presence of ≥ 3 carotid plaques | ||||||
| Triglycerides | 0.84 (0.27–2.66) | 0.772 | 0.76 (0.21–2.75) | 0.677 | 0.72 (0.15–3.40) | 0.676 |
| Glycerophospholipids | 0.23 (0.03–1.62) | 0.140 | 0.27 (0.04–1.93) | 0.192 | 0.17 (0.01–2.32) | 0.185 |
| Phosphatidylcholine | 0.16 (0.02–1.367) | 0.094 | 0.17 (0.02–1.54) | 0.114 | 0.14 (0.01–1.80) | 0.130 |
| Sphingomyelin | 0.48 (0.21–1.10) | 0.082 | 0.53 (0.21–1.32) | 0.172 | 0.57 (0.20–1.59) | 0.282 |
| Lysophosphatidylcholine | 0.96 (0.0–962.3) | 0.991 | 3.05 (0.0–4726) | 0.766 | 0.73 (0.0–1380) | 0.934 |
| Esterified cholesterol | 0.12 (0.02–0.59) | 0.009 | 0.10 (0.02–0.58) | 0.010 | 0.055 (0.006–0.51) | 0.011 |
| Free cholesterol | 0.019 (0.001–0.39) | 0.010 | 0.025 (0.001–0.56) | 0.020 | 0.009 (0.0–0.60) | 0.027 |
| Linoleic acid | 0.36 (0.11–1.18) | 0.091 | 0.33 (0.93–1.20) | 0.092 | 0.17 (0.03–0.93) | 0.041 |
| Saturated fatty acids | 1.06 (0.63–1.80) | 0.827 | 1.05 (0.60–1.81) | 0.875 | 1.23 (0.66–2.28) | 0.512 |
| ω-3 fatty acids | 0.11 (0.0–71.7) | 0.500 | 0.18 (0.0–189.8) | 0.626 | 0.60 (0.0–724.8) | 0.888 |
| ω-6 fatty acids | 0.38 (0.13–1.06) | 0.063 | 0.38 (0.14–1.07) | 0.068 | 0.27 (0.07–0.97) | 0.045 |
| ω-7 fatty acids | 1.61 (0.73–3.51) | 0.236 | 1.77 (0.79–3.95) | 0.165 | 1.89 (0.74–4.79) | 0.181 |
| ω-9 fatty acids | 0.68 (0.38–1.021) | 0.192 | 0.69 (0.39–1.21) | 0.194 | 0.63 (0.34–1.15) | 0.132 |
| DHA | 0.99 (0.83–1.17) | 0.881 | 0.98 (0.84–1.20) | 0.981 | 1.01 (0.83–1.23) | 0.911 |
| ARA-EPA | 1.13 (0.08–15.91) | 0.930 | 1.49 (0.09–23.83) | 0.776 | 2.42 (0.12–49.46) | 0.565 |
Logistic regression model (OR and 95% confidence interval) is presented. All the data were expressed for increments of 1 mmol/L, except for sphingomyelin (0.1 mmol/L increase) and DHA (0.01 mmol/L increase)
ARA arachidonic acid, DHA docosahexaenoic acid, EPA eicosapentaenoic acid
*Model 1: age- and sex-adjusted; model 2: model 1 adjusted for age, sex, presence of hypertension and statin treatment; model 3: model 2 + mean HbA1c in the last 5 years and diabetes duration
Discussion
In our non-targeted and exploratory 1H NMR lipidomic analysis, we found several lipid species that could be associated with adverse cardiometabolic traits in individuals with T1D. Besides between-gender differences, glycaemic control (evaluated by HbA1c), anthropometric (BMI and waist circumference) and insulin resistance-related (i.e. FLI and eGDR) variables were those most associated with NMR-assessed lipidomic analysis. Furthermore, some of these parameters (especially those related to ω-6 fatty acids) were also independently associated with preclinical carotid atherosclerosis burden, a surrogate of future CVD events [26, 27]. To the best of our knowledge, this preliminary data represents one of the first contributions of the usefulness of 1H NMR-based lipidomics in the assessment of CVD risk in Mediterranean individuals with T1D.
Identifying the individuals with T1D more prone to suffering from CVD is challenging. Although there are several clinical scores validated for estimating such cardiovascular risk in this population [28, 29], some of them often fail when applied in daily practice [15]. To overcome these limitations, the use of biomarkers has gained attention in the last few years. Thus, either imaging [30] or circulating biomarkers [31–33] seem promising, especially in those populations in which current tools perform rather poorly (such as T1D). In this regard, high-throughput lipidomic analysis could have the potential to unravel new pathways and biomarkers in the field of CVD in T1D.
The role of various lipids in the pathogenesis of T1D, their mediation in inflammation and the complications of the entity are controversial. Some, such as ω-3 fatty acids and their derivatives, have been shown to reduce inflammation and autoimmunity by different mechanisms while others, such as ω-9 fatty acids, show otherwise [34]. Sphingomyelin is the most abundant sphingolipid in tissues and blood and has an eminently structural function in cell membranes. It has been associated with an increase in proinflammatory cytokines and it is even suggested that it could be pathophysiologically related to the formation and progression of atheromatosis [35]. In this sense, the inverse and independent association between sphingomyelin and the presence of carotid plaque in our sample could seem contradictory. Several reports have pointed out sphingomyelin as a crucial factor for glomerular and endothelial function [36]. In this sense, some previous studies from the Scandinavian population showed that this lipid variable was directly associated with worse kidney [37–39] and coronary heart disease outcomes [39]. Nevertheless, other studies using a more powerful lipidomic analysis (which could identify up to 106 different molecular lipid species) showed that several sphingomyelin subclasses were associated with worse kidney outcomes and cardiovascular mortality while other subclasses were associated with better outcomes [10, 38]. Furthermore, no significant relationships were found between any of the sphingomyelin subclasses and stroke [10]. Also, a prospection of the Multi-Ethnic Study of Atherosclerosis cohort [40], in the general population and primary prevention, found no association between sphingomyelin levels and incident CVD, and the association was even inverse in some models. In this regard, further studies should assess the relationships between the in-depth lipidomic analysis with the different manifestations of atherosclerosis.
Although there has been a lot of controversy regarding the role of ω-6 fatty acids, especially linoleic acid, in CVD prevention [41, 42], our findings showing inverse associations of these lipid species with atherosclerosis burden (≥ 3 carotid plaques) are reassuring. Our results are aligned with three recent meta-analyses in the general population; these include several biomarkers of dietary linoleic acid intake (adipose tissue and blood) and report a strong association of this fatty acid with CVD protection [43–45]. In this regard, our study group has also shown that this lipid species (assessed through means of gas chromatography) was inversely associated with the prevalence of symptomatic carotid artery disease in the general population [46], as well as with carotid atherosclerosis burden among the T1D population [11]. These previous and our actual findings concur with the recommendations released by the American Heart Association (AHA) to replace foods rich in saturated fatty acids with those rich in ω-6 fatty acids to decrease CVD risk [47], not only for the general population but also for high-risk populations such as patients with T1D.
Several strengths and limitations should be acknowledged. One of the main strengths is the novelty in the population studied, as all the previous reports came from Scandinavian countries. Since Mediterranean individuals have very different genetic and behavioural backgrounds, the extrapolation of the existing results could be difficult. Furthermore, instead of intima-media thickness, we assessed the presence of carotid plaque, a better predictor of future CVD [26, 27]. Several strengths and limitations should be acknowledged. One of the main strengths is the novelty in the population studied, as all the previous reports came from Scandinavian countries. Since Mediterranean individuals have very different genetic and behavioural backgrounds, the extrapolation of the existing results could be difficult. Furthermore, instead of intima-media thickness, we assessed the presence of carotid plaque, a better predictor of future CVD [26, 27]. Additionally, in our research group, we are exploring the clinical utility of multiple parameters determined with 1H NMR to identify those at higher cardiovascular risk to then be able to individualize the prevention strategy in each case. The great advantage of this technology is that several of these parameters can be simultaneously determined with a single sample (lipidomics, lipoprotein subclasses, inflammatory markers, metabolomics etc.), being able to have a broad perspective of various factors involved in the pathophysiology of cardiovascular disease and lowering costs. However, the study also has limitations. First, causality cannot be established because of the cross-sectional nature of the study. Second, as a result of the limited sample and the high number of comparisons, the study must be considered preliminary data. All the associations found should be confirmed in future studies with a bigger sample and/or with longitudinal follow-up. Third, given the paucity of previous data about lipidomic analyses in T1D, the sample size could not be calculated beforehand. Fourth, the ankle-brachial index, as a surrogate marker of cardiovascular disease, would have been interesting to analyse together with the other clinical variables and their correlation with lipidomic parameters but, unfortunately, the data was not available. Fifth, some other studies have assessed various parameters by 1H NMR such as lipoprotein subclass disturbances [48], but investigations analysing the use of this technique in lipidomic variables are scarce. Sixth, since the recruitment was mainly in 2017, the use of continuous glucose monitoring (CGM) or hybrid closed loop systems (HCL) was residual (< 5% for MCG and 0% for HCL). The use of CSII (started in our hospital in 2007–2008) improved glycaemic control in our sample (HbA1c 7.47% vs. 7.8% in multiple insulin dose group; t test p value 0.042 between groups), so on the basis of glycaemic control (mean HbA1c over the last 5 years), an overall improvement in the cardiovascular profile is to be expected in our study and therefore our findings should be interpreted with caution as they cannot be extrapolated to populations with lower use of this technology. Seventh, the finding of several significant associations has also been controversial for several of the parameters, so their applicability in real clinical practice should be analysed beforehand in studies with a larger sample. Finally, because of the aforementioned limitations, some of the relationships should be interpreted with caution. In this regard, the inverse associations between the free and esterified cholesterol with high atherosclerosis burden (≥ 3 plaques) could be misleading. On one hand, these two species were highly correlated with total and LDL-cholesterol in our sample (rs > 0.3–0.4, data not shown), which in turn were lower in those harbouring more carotid plaques (Table S2). Although these associations persisted (but were blunted) when including statin treatment in multivariate models, selection bias cannot be ruled out. On the other hand, this finding could reflect the need for better biomarkers of CVD than classical ones. Previous studies have already shown that LDL-cholesterol is not the best predictor of future CVD events in the setting of high-risk patients with a high background cardioprotective treatment use [49].
Conclusion
In our exploratory study including individuals with T1D at high CVD risk, some variables obtained from the 1H NMR-derived lipidomic analysis could be associated with preclinical atherosclerosis. Specifically, sphingomyelin levels were inversely associated with carotid plaques whereas the parameters related to ω-6 fatty acids, especially linoleic acid, could identify those individuals with a more advanced atherosclerosis burden. While the causal effect of these biomolecules in atherosclerosis should be confirmed in future studies, lipidomic analysis has the potential to unravel new pathways and biomarkers for CVD prevention among the T1D population.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
We thank the participants of the study.
Funding
No funding or sponsorship was received for this study or publication of this article. The Rapid Service Fee was funded by the authors. Antonio J. Amor received a research grant from the Associació Catalana de Diabetis (ACD), “Ajut per a la recerca en diabetis modalitat clínica 2018”. The award covered the costs of the analysis of lipidomic parameters using 1H NMR.
Author Contributions
All authors have discussed the results and commented on the final version of the manuscript. Tonet Serés-Noriega, Emilio Ortega, Marga Gimenez, Verónica Perea, Laura Boswell, Karla Mariaca, Carla Font, Alex Mesa, Clara Viñals, Jesús Blanco, Irene Vinagre, Adriana Pané, Laura Boswell, Enric Esmatjes, Ignacio Conget and Antonio J. Amor acquired and processed all clinical data; Antonio J. Amor performed the US measurements; Tonet Serés-Noriega and Antonio J. Amor contributed to data analysis and interpretation, wrote, reviewed and edited the manuscript. Antonio J. Amor and Verónica Perea contributed to the study concept and design. Marga Gimenez, Verónica Perea, Clara Viñals, Jesús Blanco, Irene Vinagre, Ignacio Conget and Antonio J. Amor supervised the study and participated in data analysis and interpretation. Antonio J. Amor designed the figures and had final responsibility for the decision to submit them for publication. Antonio J. Amor is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Disclosures
None of the funding sources played a role in the design, collection, analysis or interpretation of the data or in the decision to submit the manuscript for publication.
Compliance with Ethics Guidelines
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the hospital Clínic de Barcelona (HCB/2017/0977). All subjects were included after giving their written informed consent. All authors give consent for publication.
Prior Publication
The results of this work have not been published, partially or totally, in any other medium, either written or oral.
Data Availability
The data sets analyzed during the current study are available from the corresponding author upon reasonable request.
Contributor Information
Tonet Serés-Noriega, Email: tseresnoriega@gmail.com.
Antonio J. Amor, Email: ajamor@clinic.cat
References
- 1.Mayer-Davis EJ, Lawrence JM, Dabelea D, et al. Incidence trends of type 1 and type 2 diabetes among youths, 2002–2012. N Engl J Med. 2017;376(15):1419–1429. doi: 10.1056/NEJMoa1610187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Harjutsalo V, Pongrac Barlovic D, Groop P-H. Long-term population-based trends in the incidence of cardiovascular disease in individuals with type 1 diabetes from Finland: a retrospective, nationwide, cohort study. Lancet Diabetes Endocrinol. 2021;9(9):575–585. doi: 10.1016/S2213-8587(21)00172-8. [DOI] [PubMed] [Google Scholar]
- 3.Lind M, Svensson A-M, Kosiborod M, et al. Glycemic control and excess mortality in type 1 diabetes. N Engl J Med. 2014;371(21):1972–1982. doi: 10.1056/NEJMoa1408214. [DOI] [PubMed] [Google Scholar]
- 4.Nathan DM, Bebu I, Braffett BH, et al. Risk factors for cardiovascular disease in type 1 diabetes. Diabetes. 2016;65(5):1370–1379. doi: 10.2337/db15-1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Amor AJ, Vinagre I, Valverde M, et al. Preeclampsia is associated with increased preclinical carotid atherosclerosis in women with type 1 diabetes. J Clin Endocrinol Metab. 2020;105(1):85–95. doi: 10.1210/clinem/dgz031. [DOI] [PubMed] [Google Scholar]
- 6.Vergès B. Dyslipidemia in type 1 diabetes: a masked danger. Trends Endocrinol Metab. 2020;31(6):422–434. doi: 10.1016/j.tem.2020.01.015. [DOI] [PubMed] [Google Scholar]
- 7.O’Brien ST, Neylon OM, O’Brien T. Dyslipidaemia in type 1 diabetes: molecular mechanisms and therapeutic opportunities. Biomedicines. 2021;9(7):826. doi: 10.3390/biomedicines9070826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rawshani A, Rawshani A, Sattar N, et al. Relative prognostic importance and optimal levels of risk factors for mortality and cardiovascular outcomes in type 1 diabetes mellitus. Circulation. 2019;139(16):1900–1912. doi: 10.1161/CIRCULATIONAHA.118.037454. [DOI] [PubMed] [Google Scholar]
- 9.Amor AJ, Castelblanco E, Hernández M, et al. Advanced lipoprotein profile disturbances in type 1 diabetes mellitus: a focus on LDL particles. Cardiovasc Diabetol. 2020;19(1):126. doi: 10.1186/s12933-020-01099-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ferreira-Divino LF, Suvitaival T, Rotbain Curovic V, et al. Circulating metabolites and molecular lipid species are associated with future cardiovascular morbidity and mortality in type 1 diabetes. Cardiovasc Diabetol. 2022;21(1):135. doi: 10.1186/s12933-022-01568-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mesa A, Cofán M, Esmatjes E, et al. Biomarkers of fatty acid intake are independently associated with preclinical atherosclerosis in individuals with type 1 diabetes. Eur J Nutr. 2021;60(8):4595–4605. doi: 10.1007/s00394-021-02611-2. [DOI] [PubMed] [Google Scholar]
- 12.Amor AJ, Vinagre I, Valverde M, et al. Nuclear magnetic resonance-based metabolomic analysis in the assessment of preclinical atherosclerosis in type 1 diabetes and preeclampsia. Diabetes Res Clin Pract. 2021;1(171):108548. doi: 10.1016/j.diabres.2020.108548. [DOI] [PubMed] [Google Scholar]
- 13.Amor AJ, Ortega E, Giménez M, et al. Prevalence and factors associated with statin use in high-risk patients with type 1 diabetes from a specialized diabetes unit. Endocrinol Diabetes Nutr. 2019;66(8):512–519. doi: 10.1016/j.endinu.2019.02.004. [DOI] [PubMed] [Google Scholar]
- 14.Mesa A, Giménez M, Pueyo I, et al. Hyperglycemia and hypoglycemia exposure are differentially associated with micro- and macrovascular complications in adults with type 1 diabetes. Diabetes Res Clin Pract. 2022;1(189):109938. doi: 10.1016/j.diabres.2022.109938. [DOI] [PubMed] [Google Scholar]
- 15.Serés-Noriega T, Giménez M, Perea V, et al. Use of the steno T1 risk engine identifies preclinical atherosclerosis better than use of ESC/EASD-2019 in adult subjects with type 1 diabetes at high risk. Diabetes Care. 2022;45(10):2412–2421. doi: 10.2337/dc22-0118. [DOI] [PubMed] [Google Scholar]
- 16.Chillarón JJ, Goday A, Flores-Le-Roux JA, et al. Estimated glucose disposal rate in assessment of the metabolic syndrome and microvascular complications in patients with type 1 diabetes. J Clin Endocrinol Metab. 2009;94(9):3530–3534. doi: 10.1210/jc.2009-0960. [DOI] [PubMed] [Google Scholar]
- 17.Jansa M, Quirós C, Giménez M, Vidal M, Galindo M, Conget I. Psychometric analysis of the Spanish and Catalan versions of a questionnaire for hypoglycemia awareness. Med Clin (Barc) 2015;144(10):440–444. doi: 10.1016/j.medcli.2013.11.036. [DOI] [PubMed] [Google Scholar]
- 18.Clarke WL, Cox DJ, Gonder-Frederick LA, Julian D, Schlundt D, Polonsky W. Reduced awareness of hypoglycemia in adults with IDDM. A prospective study of hypoglycemic frequency and associated symptoms. Diabetes Care. 1995;18(4):517–522. doi: 10.2337/diacare.18.4.517. [DOI] [PubMed] [Google Scholar]
- 19.Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Williams KV, Erbey JR, Becker D, Arslanian S, Orchard TJ. Can clinical factors estimate insulin resistance in type 1 diabetes? Diabetes. 2000;49(4):626–632. doi: 10.2337/diabetes.49.4.626. [DOI] [PubMed] [Google Scholar]
- 21.Bedogni G, Bellentani S, Miglioli L, et al. The fatty liver index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006;6(1):33. doi: 10.1186/1471-230X-6-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Löfgren L, Ståhlman M, Forsberg GB, Saarinen S, Nilsson R, Hansson GI. The BUME method: a novel automated chloroform-free 96-well total lipid extraction method for blood plasma. J Lipid Res. 2012;53(8):1690–1700. doi: 10.1194/jlr.D023036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Barrilero R, Gil M, Amigó N, et al. LipSpin: a new bioinformatics tool for quantitative 1 H NMR lipid profiling. Anal Chem. 2018;90(3):2031–2040. doi: 10.1021/acs.analchem.7b04148. [DOI] [PubMed] [Google Scholar]
- 24.Vinaixa M, Ángel Rodríguez M, Rull A, et al. Metabolomic assessment of the effect of dietary cholesterol in the progressive development of fatty liver disease. J Proteome Res. 2010;9(5):2527–2538. doi: 10.1021/pr901203w. [DOI] [PubMed] [Google Scholar]
- 25.Touboul PJ, Hennerici MG, Meairs S, et al. Mannheim carotid intima-media thickness and plaque consensus (2004–2006–2011) Cerebrovasc Dis. 2012;34:290–296. doi: 10.1159/000343145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Brook RD, Bard RL, Patel S, et al. A negative carotid plaque area test is superior to other noninvasive atherosclerosis studies for reducing the likelihood of having underlying significant coronary artery disease. Arterioscler Thromb Vasc Biol. 2006;26(3):656–662. doi: 10.1161/01.ATV.0000200079.18690.60. [DOI] [PubMed] [Google Scholar]
- 27.Baber U, Mehran R, Sartori S, et al. Prevalence, impact, and predictive value of detecting subclinical coronary and carotid atherosclerosis in asymptomatic adults. J Am Coll Cardiol. 2015;65(11):1065–1074. doi: 10.1016/j.jacc.2015.01.017. [DOI] [PubMed] [Google Scholar]
- 28.Vistisen D, Andersen GS, Hansen CS, et al. Prediction of first cardiovascular disease event in type 1 diabetes mellitus the steno type 1 risk engine. Circulation. 2016;133(11):1058–1066. doi: 10.1161/CIRCULATIONAHA.115.018844. [DOI] [PubMed] [Google Scholar]
- 29.McGurnaghan SJ, McKeigue PM, Read SH, et al. Development and validation of a cardiovascular risk prediction model in type 1 diabetes. Diabetologia. 2021;64(9):2001–2011. doi: 10.1007/s00125-021-05478-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alexandratou M, Papachristodoulou A, Li X, et al. Advances in noninvasive carotid wall imaging with ultrasound: a narrative review. J Clin Med. 2022;11(20):6196. doi: 10.3390/jcm11206196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu L, Yan X, Tang Z, Feng B. Association between circulating oxidized OxLDL/LDL-C ratio and the severity of coronary atherosclerosis, along with other emerging biomarkers of cardiovascular disease in patients with type 2 diabetes. Diabetes Res Clin Pract. 2022;191:110040. doi: 10.1016/j.diabres.2022.110040. [DOI] [PubMed] [Google Scholar]
- 32.Serés-Noriega T, Giménez M, Perea V, et al. Quantification of glycoproteins by nuclear magnetic resonance associated with preclinical carotid atherosclerosis in patients with type 1 diabetes. Nutr Metab Cardiovasc Dis. 2021;31(7):2099–2108. doi: 10.1016/j.numecd.2021.03.021. [DOI] [PubMed] [Google Scholar]
- 33.Amor AJ, Vinagre I, Valverde M, et al. Nuclear magnetic resonance lipoproteins are associated with carotid atherosclerosis in type 1 diabetes and pre-eclampsia. Diabetes Metab Res Rev. 2021;37(1):e3362. [DOI] [PubMed]
- 34.Zhang J, Xiao Y, Hu J, Liu S, Zhou Z, Xie L. Lipid metabolism in type 1 diabetes mellitus: pathogenetic and therapeutic implications. Front Immunol. 2022;6(13):5942. doi: 10.3389/fimmu.2022.999108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Peters L, Kuebler WM, Simmons S. Sphingolipids in atherosclerosis: chimeras in structure and function. Int J Mol Sci. 2022;23(19):11948. doi: 10.3390/ijms231911948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Slotte JP. Biological functions of sphingomyelins. Prog Lipid Res. 2013;52(4):424–437. doi: 10.1016/j.plipres.2013.05.001. [DOI] [PubMed] [Google Scholar]
- 37.Mäkinen VP, Tynkkynen T, Soininen P, et al. Sphingomyelin is associated with kidney disease in type 1 diabetes (The FinnDiane Study) Metabolomics. 2012;8(3):369–375. doi: 10.1007/s11306-011-0343-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tofte N, Suvitaival T, Ahonen L, et al. Lipidomic analysis reveals sphingomyelin and phosphatidylcholine species associated with renal impairment and all-cause mortality in type 1 diabetes. Sci Rep. 2019;9(1):16398. doi: 10.1038/s41598-019-52916-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pongrac Barlovic D, Harjutsalo V, Sandholm N, Forsblom C, Groop PH. Sphingomyelin and progression of renal and coronary heart disease in individuals with type 1 diabetes. Diabetologia. 2020;63(9):1847–1856. doi: 10.1007/s00125-020-05201-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yeboah J, McNamara C, Jiang X-C, et al. Association of plasma sphingomyelin levels and incident coronary heart disease events in an adult population. Arterioscler Thromb Vasc Biol. 2010;30(3):628–633. doi: 10.1161/ATVBAHA.109.199281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ros E. Linoleic acid intake and reduction in mortality: the icing on the cake of health benefits from n–6 PUFAs? Am J Clin Nutr. 2020;112(1):3–4. doi: 10.1093/ajcn/nqaa062. [DOI] [PubMed] [Google Scholar]
- 42.Kim HK, Kang EY, Woong GG. Recent insights into dietary ω-6 fatty acid health implications using a systematic review. Food Sci Biotechnol. 2022;31(11):1365–1376. doi: 10.1007/s10068-022-01152-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Marklund M, Wu JHY, Imamura F, et al. Biomarkers of dietary omega-6 fatty acids and incident cardiovascular disease and mortality. Circulation. 2019;139(21):2422–2436. doi: 10.1161/CIRCULATIONAHA.118.038908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Borges MC, Schmidt AF, Jefferis B, et al. Circulating fatty acids and risk of coronary heart disease and stroke: individual participant data meta-analysis in up to 16 126 participants. J Am Heart Assoc. 2020;9(5):e013131. doi: 10.1161/JAHA.119.013131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li J, Guasch-Ferré M, Li Y, Hu FB. Dietary intake and biomarkers of linoleic acid and mortality: systematic review and meta-analysis of prospective cohort studies. Am J Clin Nutr. 2020;112(1):150–167. doi: 10.1093/ajcn/nqz349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lázaro I, Cofán M, Amor AJ, et al. Linoleic acid status in cell membranes inversely relates to the prevalence of symptomatic carotid artery disease. Stroke. 2021;52(2):703–706. doi: 10.1161/STROKEAHA.120.030477. [DOI] [PubMed] [Google Scholar]
- 47.Sacks FM, Lichtenstein AH, Wu JHY, et al. Dietary fats and cardiovascular disease: a presidential advisory from the American Heart Association. Circulation. 2017;136(3):e1–23. doi: 10.1161/CIR.0000000000000510. [DOI] [PubMed] [Google Scholar]
- 48.Llauradó G, Amigó N, Cano A, et al. Specific nuclear magnetic resonance lipoprotein subclass profiles and central arterial stiffness in type 1 diabetes mellitus: a case control study. J Clin Med. 2019;8(11):1875. doi: 10.3390/jcm8111875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Castañer O, Pintó X, Subirana I, et al. Remnant cholesterol, not LDL cholesterol, is associated with incident cardiovascular disease. J Am Coll Cardiol. 2020;76(23):2712–2724. doi: 10.1016/j.jacc.2020.10.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data sets analyzed during the current study are available from the corresponding author upon reasonable request.