Abstract
Objective:
This study aimed to identify methodological aspects involved in determining anthropometric measurements among studies assessing the nutritional status of individuals with cystic fibrosis (CF).
Methods:
A search of the literature was performed on MEDLINE via Pubmed, Embase, and Web of Science databases. The population comprised children and adolescents with CF. Observational studies and clinical trials using anthropometric and body composition measures and indices determined by dual-energy X-ray absorptiometry (DXA) and bioelectrical impedance assessment (BIA) were included. Use of a standardized procedure for data collection was defined when details on the instruments and their calibration were given, the measuring procedures were described, and when it was clear measures had been determined by a trained team, or the use of an anthropometric reference manual was cited. Data extracted were expressed as absolute and relative frequencies.
Results:
A total of 32 articles were included, and a total of 233 measures or indices were observed. The most frequently used measures were body mass index (kg/m2; 35%), weight (kg; 33%), and height (cm; 33%). Among the 28 studies that used anthropometric measures, 21 (75%) provided a complete or partial description of the measurement instruments used, 3 (11%) reported information on equipment calibration, 10 (36%) indicated the measurement procedures employed by assessors, and 2 (7%) stated a trained team had carried out the measurements.
Conclusions:
The poor description of measuring procedures precluded a meaningful evaluation of data quality. Scientific debate on this theme can help raise awareness of the need to ensure quality in collecting and fully presenting data.
Keywords: Adolescent, Body composition, Body mass index, Cystic fibrosis, Child
Resumo
Objetivo:
Identificar os aspectos metodológicos relacionados à aferição de medidas antropométricas em pesquisas que avaliaram o estado nutricional de indivíduos com fibrose cística (FC).
Métodos:
Realizou-se busca nas bases Medical Literature Analysis and Retrieval System Online (MEDLINE) via United States National Library of Medicine (PubMed), Embase e Web of Science. A população foi composta de crianças e adolescentes com FC. Foram incluídos estudos observacionais e ensaios clínicos que utilizaram medidas e índices antropométricos ou dados de composição corporal obtidos pela dual-energy X-ray absorptiometry (DXA) e bioimpedância elétrica (BIA). Considerou-se o uso de procedimento padronizado na coleta dos dados quando o estudo apresentou informações sobre os instrumentos, a calibração dos equipamentos, os detalhes dos procedimentos de mensuração e a realização de treinamento. Foram apresentadas as frequências absolutas e relativas dos dados extraídos.
Resultados:
Trinta e dois artigos foram selecionados, sendo observadas 233 medidas ou índices. Desse total, os mais utilizados foram índice de massa corporal (kg/m2; 35%), peso corporal (kg; 33%) e altura (cm; 33%). De 28 estudos que utilizaram medidas antropométricas, 21 (75%) apresentaram detalhes totais ou parciais dos instrumentos utilizados nas aferições, três (11%) apresentaram informações sobre a calibração dos equipamentos, dez (36%) apresentaram a indicação dos procedimentos de mensuração utilizados pelos avaliadores e dois (7%) mencionaram a realização de treinamento da equipe responsável pelas aferições.
Conclusões:
Há pouco detalhamento dos procedimentos de mensuração nas publicações, dificultando a avaliação da qualidade dos dados. O debate científico sobre o tema pode contribuir para conscientizar sobre a necessidade de assegurar a qualidade na coleta dos dados e sua apresentação completa nos artigos.
Palavras-chave: Adolescente, Composição corporal, Índice de massa corporal, Fibrose cística, Criança
INTRODUCTION
Cystic fibrosis (CF) is a hereditary recessive genetic disease that affects several organs and systems because of dysfunction of the CFTR (cystic fibrosis transmembrane conductance regulator) protein. 1 In spite of therapeutic improvements in recent years, according to the Brazilian Cystic Fibrosis Registry (Registro Brasileiro de Fibrose Cística), the median age at death is 19.4 years. 2 Malnutrition and recurrent pulmonary exacerbations are recognized strong predictors of CF mortality. 3
Close monitoring of nutritional status is recommended for CF individuals, through periodical follow-up of anthropometry and body composition parameters. 4 The goal for pediatric CF individuals is to attain a body mass index for age (BMI/A) above the 50th percentile because this is associated with better pulmonary function. 5 Weight gain should thus take place through an increase in lean mass as opposed to body fat only. Besides BMI monitoring, body composition evaluation methods such as skinfolds measurement, dual-energy X-ray absorptiometry (DXA), and bioelectrical impedance assessment (BIA) are recommended. 4,6
The most used method for nutritional assessment, anthropometry, is frequently associated with measurement errors in patients, introduced by instruments, operators, or changes in the subjects’ body composition. 7 Since the validity of results of any survey depends on the absence of systematic methodological errors, 8 measurements such as weight and height must be obtained through accurate procedures to be suitably interpreted. 9
The objective of this study was to identify methodological aspects involved in determining anthropometric measurements among studies assessing the nutritional status of children and adolescents with CF. Preliminary searches of the International Prospective Register of Systematic Reviews (PROSPERO) and JBI Evidence Synthesis revealed no previous reviews on the topic, justifying the conducting of the present scoping review.
METHOD
The study was carried out based on the recommendations of the Joanna Briggs for scoping reviews (https://jbi.global), and the study protocol was registered with the Open Science Framework (https://osf.io/q2acf/). A systematic search of the literature was performed on MEDLINE via PubMed, Embase, and Web of Science databases. The following research questions were addressed:
Which anthropometric and body composition measures are used for evaluating the nutritional status of children and adolescents with CF in clinical practice and in research?
Are standardized procedures employed to obtain these anthropometric measurements? and
Which reference populations are used to classify the nutritional status of people with CF?
The search criteria were based on the PCC strategy (population, concept, and context). The population comprised children and adolescents with CF of both genders. Concept was defined as anthropometric data, including the use of simple anthropometric measurements: 9 body weight, body height, waist circumference, and skinfolds; the use of anthropometric indices: weight-for-age, height-for-age, BMI, and BMI/A; and the use of body composition-related measurements or indices obtained by BIA, DXA, or an equation: lean mass, body fat mass, and percentage of fat. Context was defined as CF.
Eligibility criteria were as follows: articles that included children and adolescents with CF, age 6–18 years, even if individuals of other ages were also assessed; involved evaluation of nutritional status or evolution of childhood growth, or the statistical relationship of anthropometric and body composition measurements with clinical outcomes, such as pulmonary function; and used at least one of the anthropo-metric measurements or indices cited in the main concept of the present study.
Exclusion criteria were as follows: studies that included hospitalized subjects, individuals on a lung, pancreas, or liver transplantation waiting list, those who had undergone any type of transplant surgery, pregnant women, and individuals with associated conditions such as celiac disease, Crohn’s disease, or cancer. Experimental studies assessing the pharmacodynamics and pharmacokinetics of drugs or studies with self-referenced body composition or anthropometric measurements were also excluded.
The search strategy was defined by two reviewers (FMDE and DPB) who conducted independent searches. The search included observational studies or clinical trials pursuant to the study objective. To make the scoping review possible, the studies included were restricted to those published between January 2014 and December 2021 and to those articles in English and Portuguese.
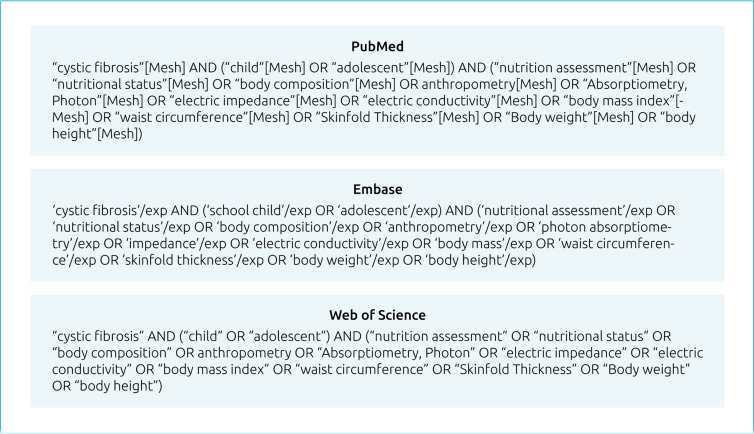
Specific descriptors for each database were used: MeSH terms (Medical Subject Head) in Pubmed and thesaurus Emtre® in Embase®. Initially, keywords associated with the PCC acronym were searched in Pubmed and then in the other databases; adaptations were made when no corresponding matches were found in the MeSH terms. The terms were linked by Boolean operators AND (restriction) and OR (addition) and constituted search phrases (Figure 1) used on the databases. The use of EndNote online (Clarivate Analytics, Boston, MA, USA) helped manage and organize the studies retrieved, removing duplicates.
Figure 1. Search terms applied in databases selected.
An initial selection of the studies was made based on titles and abstracts, independently by two authors (FMDE and DPB). When abstracts did not include age group or a description of anthropometric or body composition measurements, the methods section of the article was consulted. Differences were resolved by consensus.
As part of the selection process, two independent reviewers (FMDE and DPB) applied the Downs and Black checklist. 10 The original checklist contains 27 questions and was devised and validated to assess the methodological quality of observational studies and clinical trials on the domains of reporting, external validity, internal validity (bias), confounding/selection bias, and power. Because the present study included articles with different designs, only questions 1, 2, 3, 5, 6, 7, 10, 11, 12, 16, and 20 were applied. Differences were resolved by consensus. The maximum possible score for each article was 12 points. Articles scoring 9 or more points were considered eligible. For question 20 [“Were the main outcome measures used accurate (valid and reliable)?”], 10 only the methodological aspects pertaining to the use of anthropometric and body composition data to focus on the abovementioned concept were evaluated.
Data were extracted to characterize the studies and methodological aspects that allowed the use of standardized anthropometric data collection procedures, 9 namely,
Anthropometric and body composition measures and indices used;
Source of each measurement – either by direct measurement or extracted from medical record;
Details on instruments, such as manufacturer, type, and scale;
Calibration of instruments;
Details on measuring procedures, such as descriptions of measuring techniques, clothing worn, and presence of accessories;
Training given;
Use of anthropometric reference manual;
Reference curves; and
Criteria for grading nutritional status.
The data extracted were recorded by the two independent evaluators (FMDE and DPB) using a chart. Differences were resolved by consensus.
The data obtained were analyzed using the statistical software package Stata version 13 (Stata Corp LP, TX, USA) and expressed qualitatively (descriptive text) or quantitatively (in tables) in the form of absolute and relative frequencies.
RESULTS
The initial search resulted in the retrieval of 667 articles, excluding duplicates; 249 (37%) of these were subsequently selected. Of this initial total, 32 (13%) articles were considered eligible and included in this scoping review (Figure 2; Tables 1 and 2). 11-42
Figure 2. Flow diagram of process of article search, selection, and inclusion.
Table 1. Characteristics of studies included in scoping review (2014–2018).
| Author and year | Study type* | Country | Sample size |
|---|---|---|---|
| Bizzarri et al., 201 51 1 | 2 | Italy | CF and CFRD group: 17. Control group CF, normal GTT: 52 |
| Bruzzese et al., 2018 12 | 3 | Italy | Lactobacillus rhamnosus GG group: 41 Placebo group: 40 |
| Calella et al., 2018 13 | 1 | The United Kingdom | 69 |
| Dentini et al., 2017 14 | 1 | Brazil | Group A (BCC): 50. Group B (non-BCC): 134 |
| Doulgeraki et al., 2017 15 | 1 | Greece | HMI: 19. Without HMI: 82 |
| El Attar et al., 2017 16 | 1 | Egypt | 50 |
| Engelen and Deutz, 2014 17 | 1 | The United States | CF group: 15. Control group: 17 |
| González-Jiménez et al., 2017 18 | 1 | Spain | Pancreatic sufficiency: 371. Exocrine pancreatic insufficiency: 80 |
| Goss et al., 2015 19 | 1 | The United States and United Kingdom | 13,777; 3,968 |
| Goss et al., 2018 20 | 2 | Canada and the United States | 5,149; 37,772 |
| Groleau et al., 2014 21 | 3 | The United States | LXS group: 27. Placebo group: 36 |
| Haack and Garbi-Novaes, 2014 22 | 1 | Brazil | Without deficit: 34. With deficit: 13 |
| Hauschild et al., 2016 23 | 1 | Brazil | CF group: 46. Group without CF: 24 |
| Hauschild et al., 2018 24 | 2 | Brazil | 38 |
| Hortencio et al., 2015 25 | 2 | Brazil | 52 |
| Isa et al., 2016 26 | 1 | Bahrain | 47 |
| Kelly et al., 2016 27 | 1 | The United States | CF group: 97. Control group: 199 |
| Ledder et al., 2015 28 | 2 | Australia | 59 |
| Okuro et al., 2017 29 | 1 | Brazil | CF group: 55. Control group: 185 |
| Ong et al., 2017 30 | 2 | The United States | 1,375 |
| Papalexopoulou et al.,2018 31 | 1 | The United Kingdom | CF group in study 1: 18. Study 2: 29 |
| Sands et al., 2015 32 | 1 | Poland | 89 |
| Sheikh et al., 2014 33 | 1 | The United States | CF group: 208. Control group: 390 |
| Welsh et al., 2014 34 | 2 | Australia | 98 |
| Woestenenk et al., 2017 35 | 1 | Holland | 474 |
*Type of study: 1: cross-sectional; 2: longitudinal: 3: clinical trial. BCC: Burkholderia cepacia complex; CF: cystic fibrosis; CFRD: cystic fibrosis-related diabetes; HMI: history of meconium ileus; LXS: LYM-X-SORB™; GTT: glucose tolerance test.
Table 2. Characteristics of studies included in scoping review (2019–2021).
| Author and year of publication | Study type* | Country | Sample size |
|---|---|---|---|
| Barbosa et al., 2020 36 | 1 | Brazil | 31 |
| Campos et al., 2020 37 | 1 | Brazil | CF group: 30. Control group (healthy individuals): 25 |
| Hak et al., 2019 38 | 2 | Holland | 57 |
| Owen et al., 2021 39 | 1 | The United Kingdom | 37 |
| Phong et al., 2020 40 | 1 | The United States | 49 |
| Poulimeneas et al., 2020 41 | 1 | Greece | 76 |
| Sainath et al., 2019 42 | 2 | The United States, Canada, and Italy | 22 |
*Type of study: 1: cross-sectional; 2: longitudinal. CF: cystic fibrosis.
Regarding study type, 22 (69%) were cross-sectional, 8 (25%) were longitudinal, and 2 (6%) were clinical trials. Studies were mostly carried out in Europe (34%), South America (25%), and North America (22%). Sample size ranged from 15 to 42,921: 24 (75%) studies involved fewer than 100 subjects with CF, 5 (16%) had 101–500 subjects, and 3 (9%) included more than 500 subjects (Tables 1 and 2). 11-42
The articles employed, on average, seven measures or indices (median=6.5; quartile 1=4; quartile 3=10; values=1–19). Overall, a total of 233 measures or indices were used, predominantly derived from anthropometry (n=165; 71%) (Table 3). 11-42 For anthropometric data, a similar proportion of measures (52%) and indices (48%) were employed. The most frequently used anthropometric measures were body weight (n=28; 33%), height (n=28; 33%), and BMI (n=28; 35%). For body composition, DXA was the most frequently used measuring technique (n=55; 81%); lean mass was determined by DXA in 25% (n=14) of measurements.
Table 3. Distribution of anthropometric and body composition measures and indices, by measuring method cited in articles.
| Method | n (%) | n (%) |
|---|---|---|
| Anthropometry | 165 (71) | |
| Measurements | ||
| Weight | 28 (33) | |
| Height | 28 (33) | |
| Others | 29 (34) | |
| Total | 85 (100) | |
| Indices or equations | ||
| Body mass index | 28 (35) | |
| Height for age | 9 (11) | |
| Body mass index for age | 7 (9) | |
| Target height | 6 (8) | |
| Weight for age | 5 (6) | |
| Upper arm muscle area | 4 (5) | |
| Upper arm fat area | 3 (4) | |
| Others (chest index, height velocity, hip to shoulder width index, % ideal body weight, puberty status, ratio of leg or trunk length to height, the sum of the three skinfold thickness, weight for height) | 18 (22) | |
| Total | 80 (100) | |
| Body composition | 6,8 (29) | |
| Dual-energy X-ray absorptiometry | ||
| Lean mass measurements | 14 (25) | |
| Lean mass index | 10 (18) | |
| Fat mass measurements | 10 (18) | |
| Fat mass index | 10 (18) | |
| Bone mineral (density and content) | 11 (20) | |
| Total | 55 (100) | |
| Bioimpedance | ||
| Fat free mass; fat mass | 7 (54) | |
| Resistance/height; reactance/height; resistance index | 3 (23) | |
| Hydration status; phase angle | 3 (23) | |
| Total | 13 (100) | |
| TOTAL | 233 (100) | |
Of the total anthropometric measurements reported (n=85), 60 (71%) comprised primary data collected during study execution, 17 (20%) were secondary data obtained from medical records, and 6 (7%) involved data extracted from patient Registries, while for 2 (2%) studies, the first measurement was performed directly and the remainder was obtained from medical records.
Of the total studies (n=28) which used anthropometric measures, 21 (75%) reported full or partial details on the measuring instruments applied, 3 (11%) provided information on instrument calibration, 10 (36%) detailed the measuring procedures applied by evaluators, and 2 (7%) reported giving training to the team that took the measurements. The anthropometric measures employed by the studies, together with methods applied, are listed in Table 4.
Table 4. Description of anthropometric measures cited in articles.
| Measurements | Total | Details of instruments | Use of calibrated instruments | Details of measuring procedures | Training given | ||||
|---|---|---|---|---|---|---|---|---|---|
| n | n | % | n | % | n | % | n | % | |
| Abdominal skinfold thickness | 1 | 1 | 100 | 0 | - | 1 | 100 | 1 | 100 |
| Birth weight | 2 | 1 | 50 | 0 | - | 0 | - | 0 | - |
| Height | 28 | 22 | 79 | 0 | - | 10 | 36 | 2 | 7 |
| Hip width | 1 | 1 | 100 | 0 | - | 1 | 100 | 1 | 100 |
| Leg length | 1 | 0 | - | 0 | - | 1 | 100 | 1 | 100 |
| Mid-upper arm circumference | 6 | 4 | 67 | 0 | - | 3 | 50 | 1 | 17 |
| Parental height | 7 | 3 | 43 | 0 | - | 0 | - | 0 | - |
| Sagittal chest depth | 1 | 1 | 100 | 0 | - | 1 | 100 | 1 | 100 |
| Shoulder width | 1 | 1 | 100 | 0 | - | 1 | 100 | 1 | 100 |
| Subscapular skinfold thickness | 2 | 2 | 100 | 0 | - | 1 | 50 | 1 | 50 |
| Transverse chest width | 1 | 1 | 100 | 0 | - | 1 | 100 | 1 | 100 |
| Triceps skinfold thickness | 5 | 4 | 80 | 0 | - | 2 | 40 | 1 | 20 |
| Trunk length | 1 | 0 | - | 0 | - | 1 | 100 | 1 | 100 |
| Weight | 28 | 22 | 79 | 3 | 11 | 9 | 32 | 2 | 7 |
| Total | 85 | 63 | 3 | 32 | 14 | ||||
Three (4%) articles cited the use of reference manuals 7,9 to define the measuring procedures.
The most commonly cited reference populations were those of the Centers for Disease Control and Prevention (CDC) 14,19-21,27,29,31,33,40-42 and of the World Health Organization (WHO); 18,22-26,37 however, references were also made to specific populations such as Italian, 11 Greek, 15 Egyptian, 16 Dutch, 35,38 and British. 39 The cutoff points adopted by the authors 17,18,22,24,39,42 were mainly derived from consensuses or specific recommendations for CF subjects. 5,6,43,44
DISCUSSION
This review identified anthropometry as the most widely used tool for nutritional assessment of individuals with CF, where BMI (35%), body weight (33%), and height (33%) were the most commonly reported measures. Few articles described the methods used to obtain measurements, hampering the assessment of these aspects. Ascertaining the quality of the technical procedures adopted in the studies and in clinical practice is the key to minimize misclassifications of nutritional status, which may impact clinical and nutritional approaches in treatment planning.
Methodological studies with a focus on assessing the quality of anthropometric measures in CF are scarce. 45 This critical review of the leading articles concerning nutritional assessment of CF individuals revealed that, although the instruments used for most of the anthropometric measurements were reported, very few studies described calibration procedures. This finding implies a significant risk of systematic errors. In addition, few studies described measuring procedures in detail, compromising reliability and affecting the potential for comparisons between reference values and other values of interest. In addition, few measurements were taken by trained investigators, and the use of standardized procedures by others studies remained merely implicit.
The “Consensus Report on Nutrition for Pediatric Patients” with CF 43 states that each specialized center should maintain calibrated anthropometric instruments and measurement protocols and also perform team training, in order to ensure reliable measurements. Since many studies included in this review used data taken from medical records, these recommendations seem to be critical to avoid inaccurate conclusions. Studies carried out using medical record-based data may not be reliable due to failures in recording of the data, different approaches used to obtain measurements, or missing data from the medical records. 46 The same criticism may be applied to studies based on patient registries, since these may include the additional bias of methodological heterogeneity in measurements collected by different institutions. This possible source of error can be reduced by adopting standard criteria for datum measurement 47 and quality control during the collection of data. Professional capacity building and the standardization of techniques may also improve the precision and accuracy of measurements. 48,49
According to Fleiss, 50 the importance of error-free measurements lies in the fact that statistics are incapable of correcting an unreliable datum. Unreliable data may lessen the strength of correlation between variables, result in incorrect decisions for tests of hypotheses, and reduce the power of statistical tests, leading to the need to increase the sample size.
Since CF is a fairly rare disease, studies involving patients with CF include relatively small samples, generally fewer than 100 patients. The current review identified studies that included only 15 17 and 17 subjects with CF. 11 As the power of the statistical test is intrinsically related to sample size, a difference between groups may not be detected when the sample size is too small. The decision to include all patients may mitigate precision issues, but if the measurements are not standardized, other methodological errors may remain and jeopardize the internal validity of the results. 8 Measurement errors in the nutritional evaluation of individuals with CF may have significant implications for the patient’s overall status and impact treatment decisions.
The finding that anthropometry was the most frequent measure adopted in the nutritional studies was expected, given that they have validated psychometric properties, are readily collected, and are of low cost. The ability of BMI to evaluate body composition is still a matter of debate, 51,52 and DXA has emerged as the preferred approach to investigate body composition in the case of individuals with CF. 4 However, its high cost has been a constraint to broader application of the technique.
Since people with CF have the potential to grow normally, there is no specific growth curve for this group. The usual reference values for the general population, such as the CDC and WHO curves, were the most frequently adopted in the studies.
The studies describing body fat and lean mass used equations derived from anthropometric measurements, resistance and reactance measurements of BIA, or measurements provided by DXA. These data may have intrinsic limitations, since the equations may have been constructed for other populations or be specific for the manufacturers of the DXA apparatus.
Limitations of the present study included constraints on the search period and language of publication. The defining of the score for considering a study eligible was arbitrary and may have led to the exclusion of some important articles. However, all restrictions served to reduce the number of articles and make the review possible. No complementary searches of other sources, such as gray literature, were carried out, given the articles obtained included different anthropometric and body composition methods generally recommended.
The findings of this review highlight the key role played by hospitals and health services as a source of data in CF. Nutritional assessment is important to guide decisions on patient treatment. Scientific debate on this theme can help raise awareness of the need to ensure quality in collecting and presenting complete data, allowing readers to analyze the internal validity of the studies.
In studies evaluating nutritional status of children and adolescents with CF, few details were given on the measurement procedures used to obtain the commonly used anthropometric measures, hindering a more far-reaching analysis of the nutritional picture in CF and aggregation of data.
Footnotes
Funding This study did not receive any funding.
REFERENCES
- 1.Egan ME, Greene DM, Voynow JA. Kliegman RM, Stanton BF, St Geme JW, Schor NF, Behrman RE, et al. Nelson: tratado de pediatria. 20a ed. Rio de Janeiro:: Elsevier; 2018. Fibrose cística; pp. 2098–112. [Google Scholar]
- 2.Grupo Brasileiro de Estudos de Fibrose Cística . São Paulo (SP): GBEFC; 2019. [[cited 2022 Jan 07]]. Registro Brasileiro de Fibrose Cística. Relatório anual de 2019 [homepage on the Internet] Available from: http://portalgbefc.org.br/ckfinder/userfiles/files/REBRAFC_2019.pdf . [Google Scholar]
- 3.Stephenson AL, Tom M, Berthiaume Y, Singer LG, Aaron SD, Whitmore GA, et al. A contemporary survival analysis of individuals with cystic fibrosis: a cohort study. Eur Respir J. 2015;45:670–9. doi: 10.1183/09031936.00119714. [DOI] [PubMed] [Google Scholar]
- 4.Athanazio RA, Silva LV, Filho, Vergara AA, Ribeiro AF, Riedi CA, Procianoy ED, et al. Brazilian guidelines for the diagnosis and treatment of cystic fibrosis. J Bras Pneumol. 2017;43:219–45. doi: 10.1590/S1806-37562017000000065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stallings VA, Stark LJ, Robinson KA, Feranchak AP, Quinton H. Clinical Practice Guidelines on Growth and Nutrition Subcommittee, et al. Evidence-based practice recommendations for nutrition-related management of children and adults with cystic fibrosis and pancreatic insufficiency: results of a systematic review. J Am Diet Assoc. 2008;108:832–9. doi: 10.1016/j.jada.2008.02.020. [DOI] [PubMed] [Google Scholar]
- 6.Turck D, Braegger CP, Colombo C, Declercq D, Morton A, Pancheva R, et al. ESPEN-ESPGHAN-ECFS guidelines on nutrition care for infants, children, and adults with cystic fibrosis. Clin Nutr. 2016;35:557–77. doi: 10.1016/j.clnu.2016.03.004. [DOI] [PubMed] [Google Scholar]
- 7.Lohman TG, Roche AF, Martorell R. Anthropometric standardization reference manual. Champaign: Human Kinetics Books; 1988. [Google Scholar]
- 8.Medronho RA, Bloch KV, Luiz RR, Werneck GL. Epidemiologia. 2a ed. São Paulo: Editora Atheneu; 2009. [Google Scholar]
- 9.World Health Organization . Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee. Geneva: WHO; 1995. [PubMed] [Google Scholar]
- 10.Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52:377–84. doi: 10.1136/jech.52.6.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bizzarri C, Montemitro E, Pedicelli S, Ciccone S, Majo F, Cappa M, et al. Glucose tolerance affects pubertal growth and final height of children with cystic fibrosis. Pediatr Pulmonol. 2015;50:144–9. doi: 10.1002/ppul.23042. [DOI] [PubMed] [Google Scholar]
- 12.Bruzzese E, Raia V, Ruberto E, Scotto R, Giannattasio A, Bruzzese D, et al. Lack of efficacy of Lactobacillus GG in reducing pulmonary exacerbations and hospital admissions in children with cystic fibrosis: a randomised placebo controlled trial. J Cyst Fibros. 2018;17:375–82. doi: 10.1016/j.jcf.2017.10.014. [DOI] [PubMed] [Google Scholar]
- 13.Calella P, Valerio G, Thomas M, McCabe H, Taylor J, Brodlie M, et al. Association between body composition and pulmonary function in children and young people with cystic fibrosis. Nutrition. 2018;48:73–6. doi: 10.1016/j.nut.2017.10.026. [DOI] [PubMed] [Google Scholar]
- 14.Dentini P, Marson FA, Bonadia LC, Bertuzzo CS, Ribeiro AF, Levy CE, et al. Burkholderia cepacia complex in cystic fibrosis in a Brazilian reference center. Med Microbiol Immunol. 2017;206:447–61. doi: 10.1007/s00430-017-0521-2. [DOI] [PubMed] [Google Scholar]
- 15.Doulgeraki A, Petrocheilou A, Petrocheilou G, Chrousos G, Doudounakis SE, Kaditis AG. Body composition and lung function in children with cystic fibrosis and meconium ileus. Eur J Pediatr. 2017;176:737–43. doi: 10.1007/s00431-017-2906-z. [DOI] [PubMed] [Google Scholar]
- 16.El Attar MM, Azab NM, Hamed DH, Tawfik AS. Growth assessment in Egyptian children with cystic fibrosis: a single center study. Egyptian Paediatric Association Gazette. 2017;65:21–4. doi: 10.1016/j.epag.2017.01.001. [DOI] [Google Scholar]
- 17.Engelen MP, Com G, Deutz NE. Increased whole body hydroxyproline production as assessed by a new stable isotope technique is associated with hip and spine bone mineral loss in cystic fibrosis. Clin Nutr. 2014;33:1117–21. doi: 10.1016/j.clnu.2013.12.008. [DOI] [PubMed] [Google Scholar]
- 18.González-Jiménez D, Muñoz-Codoceo R, Garriga-García M, Molina-Arias M, Álvarez-Beltrán M, García-Romero R, et al. Excess weight in patients with cystic fibrosis: is it always beneficial? Nutr Hosp. 2017;34:578–83. doi: 10.20960/nh.620. [DOI] [PubMed] [Google Scholar]
- 19.Goss CH, MacNeill SJ, Quinton HB, Marshall BC, Elbert A, Knapp EA, et al. Children and young adults with CF in the USA have better lung function compared with the UK. Thorax. 2015;70:229–36. doi: 10.1136/thoraxjnl-2014-205718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Goss CH, Sykes J, Stanojevic S, Marshall B, Petren K, Ostrenga J, et al. Comparison of nutrition and lung function outcomes in patients with cystic fibrosis living in Canada and the United States. Am J Respir Crit Care Med. 2018;197:768–75. doi: 10.1164/rccm.201707-1541OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Groleau V, Schall JI, Dougherty KA, Latham NE, Maqbool A, Mascarenhas MR, et al. Effect of a dietary intervention on growth and energy expenditure in children with cystic fibrosis. J Cyst Fibros. 2014;13:572–8. doi: 10.1016/j.jcf.2014.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Haack A, Garbi-Novaes MR. Cystic fibrosis patients assisted by a program nutrition therapy: assessment of the use of supplements in patients colonized and non colonized by P. aeruginosa. Rev Invest Clin. 2014;66:136–43. [PubMed] [Google Scholar]
- 23.Hauschild DB, Barbosa E, Moreira EA, Ludwig N, Neto, Platt VB, Piacentini E, Filho, et al. Nutrition status parameters and hydration status by bioelectrical impedance vector analysis were associated with lung function impairment in children and adolescents with cystic fibrosis. Nutr Clin Pract. 2016;31:378–86. doi: 10.1177/0884533615627157. [DOI] [PubMed] [Google Scholar]
- 24.Hauschild DB, Rosa AF, Ventura JC, Barbosa E, Moreira EA, Ludwig N, Neto, et al. Association of nutritional status with lung function and morbidity in children and adolescents with cystic fibrosis: a 36-month cohort study. Rev Paul Pediatr. 2018;36:31–8. doi: 10.1590/1984-0462/;2018;36;1;00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hortencio TD, Nogueira RJ, Marson FA, Hessel G, Ribeiro JD, Ribeiro AF. Factors impacting the growth and nutritional status of cystic fibrosis patients younger than 10 years of age who did not undergo neonatal screening. Rev Paul Pediatr. 2015;33:3–11. doi: 10.1016/j.rpped.2014.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Isa HM, Al-Ali LF, Mohamed AM. Growth assessment and risk factors of malnutrition in children with cystic fibrosis. Saudi Med J. 2016;37:293–8. doi: 10.15537/smj.2016.3.13476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kelly A, Schall J, Stallings VA, Zemel BS. Trabecular and cortical bone deficits are present in children and adolescents with cystic fibrosis. Bone. 2016;90:7–14. doi: 10.1016/j.bone.2016.04.030. [DOI] [PubMed] [Google Scholar]
- 28.Ledder O, Oliver MR, Heine RG, Graham J, Volders E, Robinson PJ. Clinical audit results in earlier nutritional intervention in malnourished children with cystic fibrosis with improved outcome. J Paediatr Child Health. 2015;51:988–93. doi: 10.1111/jpc.12888. [DOI] [PubMed] [Google Scholar]
- 29.Okuro RT, Ribeiro MA, Ribeiro JD, Minsky RC, Schivinski CI. Alternative indexes to estimate the functional capacity from the 6-minute walk test in children and adolescents with cystic fibrosis. Respir Care. 2017;62:324–32. doi: 10.4187/respcare.04625. [DOI] [PubMed] [Google Scholar]
- 30.Ong T, Schechter M, Yang J, Peng L, Emerson J, Gibson RL, et al. Socioeconomic status, smoke exposure, and health outcomes in young children with cystic fibrosis. Pediatrics. 2017;139:e20162730. doi: 10.1542/peds.2016-2730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Papalexopoulou N, Dassios TG, Lunt A, Bartlett F, Perrin F, Bossley CJ, et al. Nutritional status and pulmonary outcome in children and young people with cystic fibrosis. Respir Med. 2018;142:60–5. doi: 10.1016/j.rmed.2018.07.016. [DOI] [PubMed] [Google Scholar]
- 32.Sands D, Umławska W, Zielińska A. A cross-sectional study of growth, nutritional status and body proportions in children and adolescents at a medical center specializing in the treatment of cystic fibrosis in Poland. Arch Med Sci. 2015;11:155–63. doi: 10.5114/aoms.2015.49207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sheikh S, Zemel BS, Stallings VA, Rubenstein RC, Kelly A. Body composition and pulmonary function in cystic fibrosis. Front Pediatr. 2014;15(2):33. doi: 10.3389/fped.2014.00033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Welsh L, Robertson CF, Ranganathan SC. Increased rate of lung function decline in Australian adolescents with cystic fibrosis. Pediatr Pulmonol. 2014;49:873–7. doi: 10.1002/ppul.22946. [DOI] [PubMed] [Google Scholar]
- 35.Woestenenk JW, Gulmans VA, van der Ent CK, Houwen RH. Height assessment in the Dutch-origin pediatric cystic fibrosis population. Nutr Clin Pract. 2017;32:130–2. doi: 10.1177/0884533616639109. [DOI] [PubMed] [Google Scholar]
- 36.Barbosa RR, Liberato FM, Coelho PF, Vidal PR, Carvalho RB, Donadio MV. Sleep-disordered breathing and markers of morbidity in children and adolescents with cystic fibrosis. Pediatr Pulmonol. 2020;55:1974–83. doi: 10.1002/ppul.24780. [DOI] [PubMed] [Google Scholar]
- 37.Campos NE, Heinzmann-Filho JP, Becker NA, Schiwe D, Gheller MF, Almeida IS, et al. Evaluation of the exercise intensity generated by active video gaming in patients with cystic fibrosis and healthy individuals. J Cyst Fibros. 2020;19:434–41. doi: 10.1016/j.jcf.2020.01.001. [DOI] [PubMed] [Google Scholar]
- 38.Hak SF, Arets HG, van der Ent CK, van der Kamp HJ. Rapid early increase in BMI is associated with impaired longitudinal growth in children with cystic fibrosis. Pediatr Pulmonol. 2019;54:1209–15. doi: 10.1002/ppul.24343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Owen E, Williams JE, Davies G, Wallis C, Grant RL, Fewtrell MS. Growth, body composition, and lung function in prepubertal children with cystic fibrosis diagnosed by newborn screening. Nutr Clin Pract. 2021;36:1240–46. doi: 10.1002/ncp.10604. [DOI] [PubMed] [Google Scholar]
- 40.Phong RY, Taylor SL, Robinson BA, Jhawar S, Nandalike K. Utility of mid-upper arm circumference in diagnosing malnutrition in children with cystic fibrosis. Nutr Clin Pract. 2020;35:1094–100. doi: 10.1002/ncp.10593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Poulimeneas D, Grammatikopoulou MG, Petrocheilou A, Kaditis AG, Vassilakou T. Triage for malnutrition risk among pediatric and adolescent outpatients with cystic fibrosis, using a disease-specific tool. Children (Basel). 2020;7:269. doi: 10.3390/children7120269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sainath NN, Schall J, Bertolaso C, McAnlis C, Stallings VA. Italian and North American dietary intake after ivacaftor treatment for Cystic Fibrosis Gating Mutations. J Cyst Fibros. 2019;18:135–43. doi: 10.1016/j.jcf.2018.06.004. [DOI] [PubMed] [Google Scholar]
- 43.Borowitz D, Baker RD, Stallings V. Consensus report on nutrition for pediatric patients with cystic fibrosis. J Pediatr Gastroenterol Nutr. 2002;35:246–59. doi: 10.1097/00005176-200209000-00004. [DOI] [PubMed] [Google Scholar]
- 44.Sinaasappel M, Stern M, Littlewood J, Wolfe S, Steinkamp G, Heijerman HG, et al. Nutrition in patients with cystic fibrosis: a European Consensus. J Cyst Fibros. 2002;1:51–75. doi: 10.1016/s1569-1993(02)00032-2. [DOI] [PubMed] [Google Scholar]
- 45.Coburn-Miller C, Casey S, Luong Q, Cameron N, Hocevar-Trnka J, Leung DH, et al. Standardization of research-quality anthropometric measurement of infants and implementation in a multicenter study. Clin Transl Sci. 2015;8:330–3. doi: 10.1111/cts.12283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pellen N, Guéganton L, Bertrand PD, Rault G. Lessons from the on-site quality audit of data transmitted to the French cystic fibrosis registry. Orphanet J Rare Dis. 2018;13(Suppl 1):9. doi: 10.1186/s13023-017-0750-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.World Health Organization [homepage on the Internet] Child growth standards. [[cited 2021 Jun 21]]. Available from: http://www.who.int/childgrowth/standards/en/
- 48.Grellety E, Golden MH. The effect of random error on diagnostic accuracy illustrated with the anthropometric diagnosis of malnutrition. PLoS One. 2016;11:e0168585. doi: 10.1371/journal.pone.0168585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Leidman E, Mwirigi LM, Maina-Gathigi L, Wamae A, Imbwaga AA, Bilukha OO. Assessment of anthropometric data following investments to ensure quality: Kenya Demographic Health Surveys Case Study, 2008 to 2009 and 2014. Food Nutr Bull. 2018;39:406–19. doi: 10.1177/0379572118783181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fleiss JL. Design and analysis of clinical experiments. London: John Wiley & Sons; 1986. [Google Scholar]
- 51.Jensen NS, Camargo TF, Bergamaschi DP. Índice de massa corpórea e perímetro da cintura são bons indicadores para classificação do estado nutricional de crianças. Ciênc Saúde Colet. 2016;21:1175–80. doi: 10.1590/1413-81232015214.138712015. [DOI] [PubMed] [Google Scholar]
- 52.Vanderwall C, Clark RR, Eickhoff J, Carrel AL. BMI is a poor predictor of adiposity in young overweight and obese children. BMC Pediatr. 2017;17:135. doi: 10.1186/s12887-017-0891-z. [DOI] [PMC free article] [PubMed] [Google Scholar]