Abstract
Background
Pristimantis is the most diverse genus of terrestrial frogs. Historically, it has been divided into several phenetic groups in order to facilitate species identification. However, in light of phylogenetic analysis, many of these groups have been shown to be non-monophyletic, denoting a high degree of morphological convergence and limited number of diagnostic traits. In this study, we focus on the Pristimantis myersi group, an assemblage of small rainfrogs distributed throughout the Andes of Ecuador and Colombia, whose external morphology is highly conserved, and its species diversity and evolutionary relationships largely unknown.
Methods
We inferred a new phylogenetic hypothesis for the frog genus Pristimantis, including all available sequences of the mtDNA 16S rRNA, as well as new DNA sequences from 175 specimens. Our sampling included 19 of the 24 species currently recognized as part of the Pristimantis myersi group.
Results
Our new evolutionary hypothesis recovered the P. myersi group as non-monophyletic and composed of 16 species. Therefore, we exclude P. albujai, P. bicantus, P. sambalan, and P. nelsongalloi in order to preserve the monophyly of the group. We discovered at least eight candidate species, most of them hidden under the names of P. leoni, P. hectus, P. festae, P. gladiator, and P. ocreatus.
Discussion
Our results reveal the occurrence of a high level of cryptic diversity to the species level within the P. myersi group and highlight the need to redefine some of its species and reassess their conservation status. We suggest that the conservation status of six species within the group need to be re-evaluated because they exhibit smaller distributions than previously thought; these species are: P. festae, P. gladiator, P. hectus, P. leoni, P. ocreatus, and P. pyrrhomerus. Finally, given that the Pristimantis myersi group, as defined in this work, is monophyletic and morphologically diagnosable, and that Trachyphrynus is an available name for the clade containing P. myersi, we implement Trachyphrynus as a formal subgenus name for the Pristimantis myersi group.
Keywords: Ecuador, Pristimantis, Andes, Evolutionary relationships, Cryptic diversity
Introduction
Pristimantis Jiménez de la Espada, 1870, is the most diverse genus of terrestrial frogs, with 595 species described (Frost, 2023). It is distributed from Honduras to southern Brazil, but concentrates a substantial part of its richness in the Andes of Colombia, Ecuador, and Peru (Lynch & Duellman, 1997; Heinicke, Duellman & Hedges, 2007; Hedges, Duellman & Heinicke, 2008; Duellman & Lehr, 2009; García et al., 2012; Frost, 2023). The taxonomy of Pristimantis is challenging because of its high diversity, relatively low intraspecific phenotypic variation, and few external diagnostic morphological characters (Lynch & Duellman, 1997; Duellman & Lehr, 2009; Guayasamin et al., 2015). Recent studies have shown that Pristimantis species richness is underestimated in part by the existence of morphologically cryptic species (Elmer & Cannatella, 2009; Padial & De la Riva, 2009; Yánez-Muñoz et al., 2010; Hutter & Guayasamin, 2015; Ortega-Andrade et al., 2015; Rivera-Correa & Daza, 2016; González-Durán et al., 2017; Páez & Ron, 2019).
Conceptual advances in the definition of species (de Queiroz, 2007; Padial & De la Riva, 2010; Camargo & Sites, 2013), and the implementation of integrative approaches including the use of DNA sequences, morphological and behavioral data, has allowed more robust and objective species delimitations. However, the inference of phylogenetic relationships based on molecular data is still pending for most Pristimantis species, despite several recent efforts (Bickford et al., 2007; Heinicke, Duellman & Hedges, 2007; Hedges, Duellman & Heinicke, 2008; Padial et al., 2010; Padial, Grant & Frost, 2014; Rivera-Correa & Daza, 2016; González-Durán et al., 2017; Guayasamin, Arteaga & Hutter, 2018; Páez & Ron, 2019), which limits the ability to identify and delimit independent evolutionary lineages hidden within complexes of morphologically cryptic species.
Our study focused on the Pristimantis myersi group. This assemblage has been phenetically defined by the combination of the following character states: small body size (SVL <28 mm), robust bodies, relative narrow heads and short snouts, cranial crest absents, tympanum differentiated, vocal slits and vomerine teeth present (vocal slits only absent in P. floridus), limbs short to moderate long, finger I shorter than finger II, toe V slightly longer than toe III and not extending to the proximal edge of the distal subarticular tubercle of the toe IV, and digital discs narrow (expanded in P. floridus) and rounded (lanceolate in P. hectus, P. lucidosignatus, and P. onorei) (Lynch & Duellman, 1997; Hedges, Duellman & Heinicke, 2008; Rödder & Schmitz, 2009).
The species belonging to this group inhabit rainforest, montane forests, and paramos of Colombia and Ecuador (Hedges, Duellman & Heinicke, 2008; Padial, Grant & Frost, 2014; Rojas-Runjaic, Delgado & Guayasamin, 2014; Rojas-Runjaic & Guayasamin, 2015; González-Durán et al., 2017; Guayasamin, Arteaga & Hutter, 2018). The group, as currently defined, is composed of 24 species: Pristimantis festae (Peracca, 1904); Pristimantis myersi (Goin & Cochran, 1963); Pristimantis celator (Lynch, 1976a); Pristimantis leoni (Lynch, 1976b); Pristimantis gladiator (Lynch, 1976b); Pristimantis pyrrhomerus (Lynch, 1976b); Pristimantis ocreatus (Lynch, 1981a); Pristimantis repens (Lynch, 1984); Pristimantis hectus (Lynch & Burrowes, 1990); Pristimantis verecundus (Lynch & Burrowes, 1990); Pristimantis scopaeus (Lynch, Carranza & Robayo, 1996); Pristimantis floridus (Lynch & Duellman, 1997); Pristimantis xeniolum (Lynch, 2001); Pristimantis jubatus (García & Lynch, 2006); Pristimantis bicantus Guayasamin & Funk, 2009; Pristimantis onorei Rödder & Schmitz, 2009; Pristimantis lucidosignatus Rödder & Schmitz, 2009; Pristimantis sirnigeli Yánez-Muñoz et al., 2010; Pristimantis munozi Rojas-Runjaic, Delgado & Guayasamin, 2014; Pristimantis mutabilis Guayasamin et al., 2015; Pristimantis sambalan Brito, Batallas & Yánez-Muñoz, 2017; Pristimantis albujai Brito, Batallas & Yánez-Muñoz, 2017; Pristimantis gralarias Guayasamin, Arteaga & Hutter, 2018; and Pristimantis nelsongalloi Valencia et al., 2019. Of these, 20 are found in Ecuador, between 900 and 4,150 masl (Frost, 2023).
As occur in other co-generic clusters, the Pristimantis myersi group is characterized by a remarkable paucity of discrete phenotypic characters useful to diagnosing the group. At the same time, some species in this group may exhibit striking intraspecific phenotypic variation in color pattern and skin texture (e.g., Arteaga et al., 2016; Rojas-Runjaic, Delgado & Guayasamin, 2014; Guayasamin et al., 2015). The combination of such phenomena represents a major source of taxonomic confusion; and the occurrence of instances of sympatry among some species of this group (e.g., Yánez-Muñoz et al., 2010; Rojas-Runjaic, Delgado & Guayasamin, 2014; Guayasamin et al., 2008) adds complexity to the system. In consequence, the occurrence of cryptic species (i.e., more than one morphologically similar species erroneously classified under the same name), taxonomic synonyms (i.e., two or more species names assigned to the same taxonomic species), and a very high percentage of species misidentifications in museum specimens is likely for the P. myersi group. These issues have been already highlighted by Yánez-Muñoz et al. (2010), who suggested that P. onorei and P. lucidosignatus may be junior synonyms of P. pyrrhomerus and P. floridus, respectively; and are also exemplified by the finding of the first record of P. myersi from museum specimens long misidentified as P. festae and P. ocreatus (Rojas-Runjaic & Guayasamin, 2015). Discovering the phylogenetic relationships of the Pristimantis myersi group and clarifying its taxonomic uncertainties is important, not only to improve our knowledge on its current species richness and to make adequate ecologic and biogeographic interpretations, but also for conservation purposes (Ortega-Andrade et al., 2021), such as re-evaluating the conservation status of already named species whose known extent of occurrence is altered as a consequence of their taxonomic redefinition, evaluating without further delay the status of those still undescribed, and to adequately guide conservation actions for all of them.
Our study aimed to assess the phylogeny and species diversity of the Pristimantis myersi group from a molecular perspective, and through a notably expanded taxon and geographic sampling, especially in Ecuador. This phylogenetic information allowed us to: (i) infer the evolutionary relationships between species of the group, (ii) redefine the species content of the group to render it monophyletic, and (iii) assess species diversity and species limits.
Materials & Methods
Ethics statement
We follow the guidelines for use of live amphibians and reptiles in field research from Beaupre et al. (2004).
Taxon sampling
We follow the taxonomy of Hedges, Duellman & Heinicke (2008) and Padial, Grant & Frost (2014) regarding family, genus, and arrangement of species groups. New mitochondrial DNA sequences from 175 specimens representing 31 species, were generated in the Laboratorio de Biología Evolutiva at Universidad San Francisco de Quito USFQ (LBE-USFQ), and the BioCamb’s molecular laboratory at Universidad Tecnológica Indoamérica (Appendix 1). Homologous sequences of some species in the P. myersi group available from previous studies as well as those of the outgroups, were downloaded from GenBank (http://www.ncbi.nlm.nih.gov/genbank/), 16 GenBank sequences required reidentification (Table 1). Voucher specimens for the new sequences are deposited at the Museo de la Universidad Tecnológica Indoamérica, Quito (MZUTI), Museo de Zoología de la Universidad San Francisco de Quito (ZSFQ), Museo de Zoología de la Pontificia Universidad Católica de Ecuador, Quito (QCAZ), and División de Herpetología (DHMECN) of the Instituto Nacional de Biodiversidad (INABIO). Other acronyms associated with terminals included in our phylogeny are JMG (Juan Manuel Guayasamin, field series, at USFQ), KU (University of Kansas, United States of America), and UVC (Universidad del Valle, Colombia).
Table 1. Species names of GenBank sequences re-identified in this study.
| Terminal name | Original name (GenBank code) |
|---|---|
| Pristimantis festae | Pristimantis trepidotus (EF493515) |
| Pristimantis ocreatus | Pristimantis thymelensis (JX564889) |
| Pristimantis sp. 1 | Pristimantis hectus (JN104680) |
| Pristimantis sp. 1 | Pristimantis hectus (JN371037) |
| Pristimantis sp. 2 | Pristimantis verecundus (EF493686) |
| Pristimantis sp. 10 | Pristimantis verecundus (KM675445) |
| Pristimantis sp. 10 | Pristimantis verecundus (KM675446) |
| Pristimantis sp. 10 | Pristimantis verecundus (KM675447) |
| Pristimantis sp. 10 | Pristimantis verecundus (KM675448) |
| Pristimantis sp. 10 | Pristimantis verecundus (KM675464) |
| Pristimantis sp. 10 | Pristimantis verecundus (KM675465) |
| Pristimantis sp. 10 | Pristimantis verecundus (KM675466) |
| Pristimantis sp. 10 | Pristimantis verecundus (KM675467) |
| Pristimantis sp. 13 | Pristimantis pyrrhomerus (EF493683) |
| Pristimantis sp. 18 | Pristimantis leoni (EF493684) |
| Pristimantis sp. 18 | Pristimantis librarius (MH516183) |
Our DNA sequence data includes 19 species currently assigned to the Pristimantis myersi group (P. albujai, P. bicantus, P. celator, P. festae, P. gladiator, P. gralarias, P. hectus, P. jubatus, P. leoni, P. lucidosignatus, P. munozi, P. mutabilis, P. nelsongalloi, P. ocreatus, P. onorei, P. pyrrhomerus, P. sambalan, P. sirnigeli, and P. verecundus), which represent coverage of 79% of the group (Appendix 1). Our first phylogenetic analysis included all available sequences of Pristimantis (760 terminals); the inferred tree allowed us to restrict the P. myersi group and, at the same time, to identify species that were erroneously placed in the group. In a second inference, we included only species of the monophyletic P. myersi group and closely related clades (219 terminals; Appendix 1). We included 10 species of several craugastorid genera as outgroups (Craugastor daryi, C. longirostris, Diasporus hylaeformis, D. vocator, Lynchius flavomaculatus, L. nebulanastes, Oreobates cruralis, O. saxatilis, Phrynopus auriculatus, and P. bracki) (Pinto-Sánchez et al., 2012), and used Agalychnis callidryas to root the trees. Due logistic limitations we were unable to obtain samples of three species of the Pristimantis myersi group whose geographic distributions are restricted to Colombia (i.e., P. repens, P. scopaeus, and P. xeniolum). We also failed to include samples of P. floridus in our study, as this species has not been found in recent years.
DNA extraction and PCR amplification for 16S rRNA gene
For DNA extraction, we followed the guanidine thiocyanate protocol designed by Peñafiel et al. (2019). For the Polymerase Chain Reaction (PCR) amplification of the 16S rRNA gene fragment, we followed the protocol designed by Guayasamin et al. (2017). For DNA amplification, we used the primers 16SC (GTRGGCCTAAAGCAGCCAC) and 16Sbr-H (5′-CCG GTC TGA ACT CAG ACG T-3′) designed by Darst & Cannatella (2004) and Palumbi et al. (1991), respectively. Each PCR reaction contained a final concentration of 1.5 mM MgCl2, 0.5 mM dNTP, 0.25 U/µL of Taq (Invitrogen) DNA polymerase, and 0.2 µM of each primer, in a total volume of 25 µL. The PCR protocol included an initial denaturation step of 4 min at 94 °C; followed by 1 min at 94 °C, 30 s at 57 °C, and 2 min at 72 °C, for 30 cycles, and a final extension of 8 min at 72 °C. The amplicons obtained were visualized by electrophoresis with a 2% agarose gel. The amplified samples were cleaned with ExoSAP, and cycle sequencing reactions were performed by Macrogen Sequencing Labs (Macrogen Inc., Korea). All fragments were sequenced in both forward and reverse directions.
The utility of mitochondrial genes, especially 16S, to uncover population structure and cryptic species has a long history in amphibian studies and has been the most widely used to delimit species (Fouquet et al., 2007; Funk, Caminer & Ron, 2012; Cryer et al., 2019; Páez & Ron, 2019; Guayasamin et al., 2020; Sánchez-Nivicela et al., 2021); moreover, because of the longer coalescence times of nuclear genes (approximately four times that of mtDNA), studies on closely related taxa heavily rely on fast-evolving genes (Avise, 2000; Vences et al., 2005; Zink & Barrowclough, 2008).
Sequence editing and phylogenetic analyses
All chromatogram sequences were fully inspected, assembled, compared against their reverse complements to detect errors, and manually edited using Geneious Pro 5.4.6 (Genematters Ltd.). We performed BLAST queries (https://blast.ncbi.nlm.nih.gov/Blast.cgi) for all the new sequences to verify their identity and to discard contaminations or mislabeling errors. GenBank accession codes of the generated sequences for the Pristimantis myersi group are in Appendix 1.
We performed the alignment with the online program MAFFT v7 (Katoh & Standley, 2013) under G-INS-I strategy (available at: https://mafft.cbrc.jp/alignment/server/index.html), and visualized it in Mesquite v3.6 (Maddison & Maddison, 2019). Uncorrected pairwise distances were calculated in MEGA7 (Kumar, Stecher & Tamura, 2016). Phylogenies were performed using Maximum Likelihood (ML) and Bayesian Inference (BI) methods. To obtain the best nucleotide substitution model, we used Model-Finder under the Bayesian information criterion (BIC) (Kalyaanamoorthy et al., 2017). Phylogenetic inference was made using ten replicates under the Maximun Likelihood (ML) criterium in IQ-Tree v2.1.3., which simultaneously finds the topology, and branch lengths that maximize the log-likelihood (Nguyen et al., 2015). Branch support was assessed from 100,000 ultrafast bootstraps (UFBoot2) replicates (Hoang et al., 2018; Minh, Nguyen & Haeseler, 2013), and SH-like approximate likelihood ratio test (Anisimova & Gascuel, 2006). To decrease the risk or overestimation branch support produced by UFBoot2, we implemented UFBoot optimization by nearest neighbor interchange search.
We produced two phylogenetic results using this methods. The first one using all available sequences of Pristimantis (742 terminals) and the second one using P. myersi group and closely related clades (215 terminals). For the first (742 terminals) we generated three phylogenetic trees using the ML described above and we made a topology test of them. The topology test was made using 1,000,000 replicates to analyze the Bootstrap Proportion using RELL approximation, Kishino-Hasegawa Test, Shimodaira-Hasegawa Test, Expected Likelihood Weights, and Approximately Unbiased (AU) Test (Kishino & Hasegawa, 1989; Kishino, Miyata & Hasegawa, 1990; Shimodaira & Hasegawa, 1999; Shimodaira, 2002; Strimmer & Rambaut, 2002), in IQ-Tree v2.1.3. All the topologies meet the 95% confidence set in all tests. For the second phylogeny, we generated 10 replicates using the ML method described above. Also, we produced two phylogenetic models of ML using regular bootstraps (Felsenstein, 1985); the branch support was obtained using 100 and 200 bootstraps, in IQ-Tree (Nguyen et al., 2015). The topology test was run as described in the previous analysis, and the selected topology meets the 95% confidence set in all tests. Bayesian inferences model were performed in MrBayes 3.2.7a software (Ronquist et al., 2012). We conducted four parallel runs of Monte Carlo Markov Chain (MCMC) for 10,000,000 generations, with sampling every 1,000 iterations and burning of 25% to estimate the Bayesian tree and Bayesian Posterior Probabilities (BPP). GTR+G+I was used as evolutionary model; this was selected using MrModelTest. Stationarity was assessed by examining the standard deviation of split frequencies and by plotting the –lnL per generation using Tracer 1.5.0 (Rambaut & Drummond, 2009). Finally, all trees generated were visualized in iTol v5 (Letunic & Bork, 2021) and edited in Adobe Illustrator 15.0.0 (Adobe Systems Inc.).
Species concept and candidate species
We follow the evolutionary species concept of Simpson (1961) as modified by Wiley (1978), which define a species as a lineage of ancestral descendant populations that maintains its identity from other such lineages and that has its own evolutionary tendencies and historical fate. These lineages can be operationally defined by contingent properties (e.g., fixed phenotypic traits, reproductive isolation, and reciprocal monophyly) that allow the discovery of their particular evolutionary trajectory (de Queiroz, 2007; Padial & De la Riva, 2010). Here, we delimited species and candidate species mainly based on molecular data and combining two approaches: monophyly verified by phylogenetic trees, and genetic distances (a non-tree based method).
Following Vieites et al. (2009) and Padial et al. (2010) we classified candidate species in three categories as follows: (1) Confirmed Candidate Species (CCS): deep genealogical lineages which exhibits fixed phenotypic characteristics that are consistent with its genetic divergence in differentiating them from other lineages, and that can be considered good species following standards of divergence for the group to which they belong, but that have not yet been formally described and named; (2) Unconfirmed Candidate Species (UCC): clades sister to nominal species, exhibiting relevant genetic divergence with respect to them (in this case we consider relevant distances ≥ 2%), but without further information on other lines of evidence (e.g., bioacoustics, morphology); and (3) Deep Conspecific Lineages (DCL): lineages exhibiting intraspecific divergence values above the typical threshold observed for conspecific populations of related species, but where other lines of evidence indicate that they do not differ at the species level. In these cases, genetic divergence might be correlated to geographic distance.
Genetic distances are a powerful tool for identifying and discovering species. When gene flow between populations is restricted, genetic distances should increase (Nei, 1972; Janzen, 2004). Thus, genetic distances between populations of the same species should be smaller than between different species. To identify candidate species, we set a 2% threshold of uncorrected distances for the gene 16S, because there is empirical evidence indicating that sister species in Pristimantis usually exhibit such genetic distance or greater (e.g., Padial & De la Riva, 2009; Ortega-Andrade et al., 2015) and other Neotropical frogs often differ by genetic distances <3% (e.g., Coloma et al., 2012; Funk, Caminer & Ron, 2012; Jungfer et al., 2013; Caminer & Ron, 2014; Blotto et al., 2021; Escalona et al., 2021).
Results
Taxon and geographic sampling
We were able to include samples of topotypic specimens (or from specimens collected near the type localities of their respective species) of all (but Pristimantis floridus and P. myersi) the already named Ecuadorian species of the Pristimantis myersi group, in our phylogenetic analyses. This represents a coverage of 79% of the species currently included in this species group. In addition, we included a number of specimens corresponding to undetermined (and presumably new) species belonging to the P. myersi group.
Phylogenetic relationships among species of the Pristimantis myersi group and its closely related clades
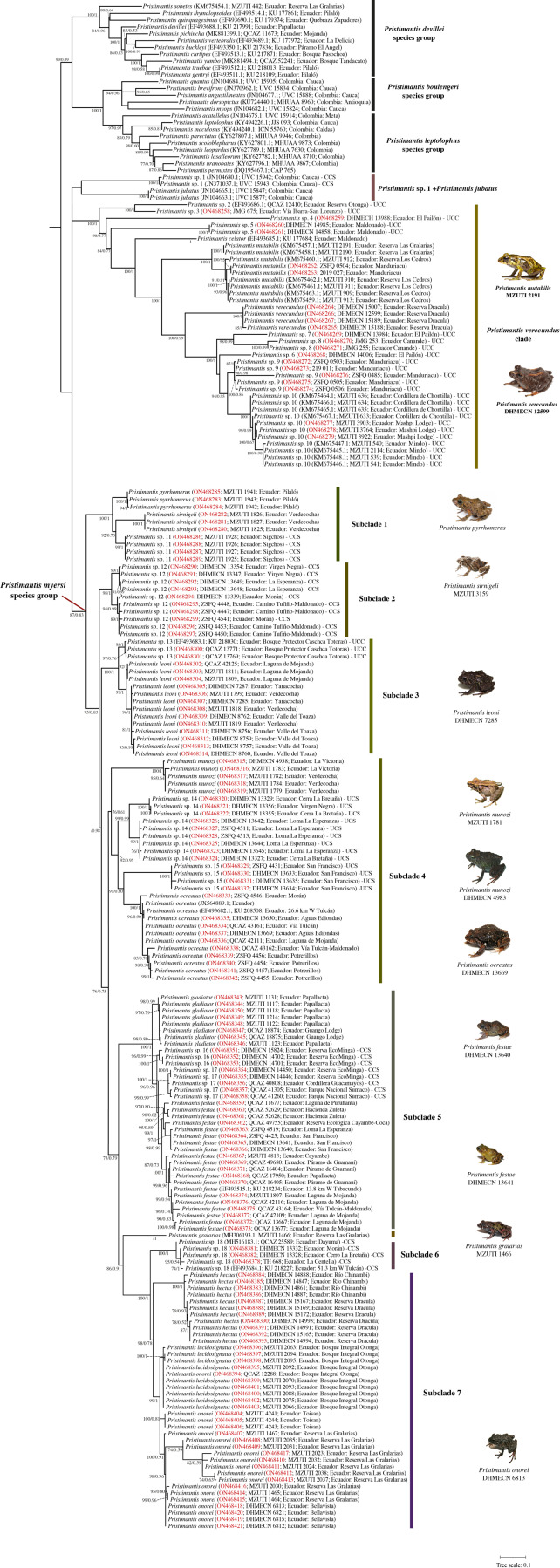
The optimal nucleotide substitution model for our first dataset (742 terminals) according to Model-Finder (lnL = 50343.3980; BIC = 112673.3001) was TlM2+F+R7. This first topology (Supplemental File 1) allowed us to exclude species previously assigned to the Pristimantis myersi group (see below). For the second dataset (215 terminals) the optimal nucleotide substitution model according to Model-Finder (lnL = −10135.4133; BIC = 23218.6410) was TlM2+F+R3, and our optimal ML topology is shown in Fig. 1 (lnL = −10091.1778). For Bayesian Inference analyses, MrModelTest selected GTR+I+G as the best substitution model (lnL = −53947.9102; AIC = 107915.8203). Maximum Likelihood and Bayesian topologies obtained from the second dataset are largely congruent. In general, Bayesian tree showed higher nodal support and lower number of collapsed nodes than the ML tree. Since no relevant incongruences were found, we present only the Maximun Likelihood tree of the second dataset, including support values for each node obtained from both ultrafast bootstraps of ML and Bayesian posterior probability (i.e., UFB/BPP) (Fig. 1). This tree is, in general, well-resolved, with most of the species previously assigned to the Pristimantis myersi group recovered as part of a well-supported clade (87%/0.83).
Figure 1. Maximum likelihood tree of the Pristimantis myersi species group and close relatives based on the mitochondrial gene 16S.
Node support is expressed in Bootstrap values (%), followed by Bayesian posterior probabilities; missing values indicate support below 70% (bootstrap) or 0.5 (posterior probability). Each terminal includes the following information: species name, GenBank code, voucher number, and locality. Outgroups are not shown. Codes in red indicate new sequences. Abbreviations: CCS = confirmed candidate species, and UCS = unconfirmed candidate species. Photographs of P. ocreatus and P. festae was taken by Diego Batallas-Revelo; photographs of P. verecundus, P. leoni, P. munozi and P. onorei was taken by Mario Yánez Muñoz. The photographs of P. mutabilis, P. pyrrhomerus, P. sirnigeli, and P. galarias are from Juan M. Guayasamin.
The clade of Pristimantis myersi, is grouped into a polytomy with other two strongly supported clades, one of them containing P. celator, P. mutabilis, P. verecundus, and several unnamed lineages (hereafter, P. verecundus clade; support = 100%/1; interspecific genetic distances: 2.6–16.3%), and the other (84%/1) composed of P. jubatus and a deeply divergent CCS (Pristimantis sp. 1); uncorrected p-distance between these two species was 8.2–8.3%. The successive sister clade (99%/0.99) to that described above, is composed of three monophyletic subclades corresponding to the P. devillei, P. boulengeri, and P. leptolophus species groups (Fig. 1).
From the 24 species previously assigned to the Pristimantis myersi group and sampled in our phylogeny, four (P. albujai, P. bicantus, P. nelsongalloi, and P. sambalan) were not recovered as part of that group. Contrary to expectations, P. albujai was inferred as sister to P. cf. mendax; the other three species were grouped in a clade (94%) that also includes P. caprifer and a sister clade that includes P. acuminatus and P. eriphus. Within this clade, the position of P. nelsongalloi renders P. bicantus paraphyletic (Fig. S1).
The Pristimantis verecundus clade (Fig. 1) is represented in our phylogeny by its nominal species (from Dracula Reserve, Carchi province, Ecuador), P. celator (Carchi province, Ecuador), P. mutabilis (Pichincha province, Ecuador), and nine Unconfirmed Candidate Species (UCC), namely: Pristimantis sp. 2 (Otonga, Cotopaxi province, Ecuador), Pristimantis sp. 3 (San Lorenzo road, Carchi province, Ecuador), Pristimantis sp. 4 (Dracula Reserve, Carchi province, Ecuador), Pristimantis sp. 5 (Maldonado-Chinambí road, Carchi province, Ecuador), Pristimantis sp. 6 (Dracula Reserve, Carchi province, Ecuador), Pristimantis sp. 7 (Dracula Reserve, Carchi province, Ecuador), Pristimantis sp. 8 (Canandé Reserve, Esmeraldas province, Ecuador) Pristimantis sp. 9 (Manduriacu Reserve, Pichincha province, Ecuador), and Pristimantis sp. 10 (Mindo, Mashpi, and Chontilla, Pichincha province, Ecuador). Interspecific uncorrected 16S p-distances within this group range between 2.6 and 16.3% (Table S2).
Within the Pristimantis myersi group we identified at least 18 independent and well-supported lineages corresponding to ten named species and at least eight candidate species (Fig. 2). These species-level lineages are arranged in seven major subclades. Below we describe the species composition, evolutionary relationships, and interspecific genetic divergence within each of them.
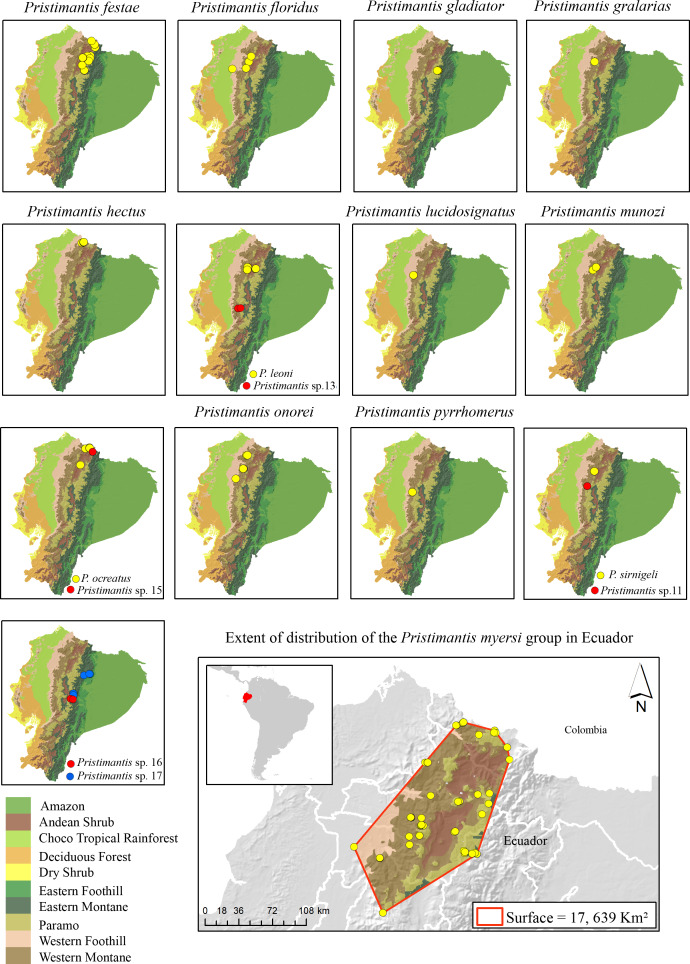
Figure 2. Distribution of the species in the Pristimantis myersi group (12 named species + five new candidate species) in the biogeographic regions of Ecuador.
Land cover: Modified from Ron, Guayasamin & Menéndez-Guerrero (2011).
Subclade 1 (Fig. 1): A strongly-supported clade (100%/1) composed of three lineages, namely: P. pyrrhomerus, P. sirnigeli, and a CCS (Pristimantis sp. 11). Pristimantis pyrrhomerus is represented by topotypes (from ca. 2 km E of Pilaló, Cotopaxi province, Ecuador) and it is sister to the other two lineages. In turn, P. sirnigeli it is represented by topotypes from Reserva Ecológica Verdecocha, Pichincha province, Ecuador and was recovered as sister to Pristimantis sp. 11 (from Sigchos, Cotopaxi province, Ecuador), a deeply divergent, geographically distant and morphologically distinguishable lineage. Genetic p-distances between P. sirnigeli and their two successive sisters, range between 2.7 and 3.7%, whereas p-distance between P. pyrrhomerus and Pristimantis sp. 11 was 2.2–2.3% (Table S3).
Subclade 2 (Fig. 1): A well-supported clade (98%/1) only composed of Pristimantis sp. 12, a CCS. All the specimens grouped in this subclade come from four different localities in Carchi province, Ecuador. The species exhibit genetic structure (although small) apparently related to geographic distance. Intraspecific genetic distances for this lineage do not surpass 0.8% (Table S4).
Subclade 3 (Fig. 1): A fully-supported clade (100%/1) composed of two lineages, namely: Pristimantis sp. 13 (UCC) from Bosque Protector Cashca Totoras (Bolívar province, Ecuador), and P. leoni. The latter is represented by specimens from six localities along its entire known distribution on northern Ecuador, including the type locality (Nudo de Mojanda). Pristimantis sp. 13 and P. leoni, are reciprocally monophyletic and their genetic divergence ranges between 1.3–2.2% (Table S5).
Subclade 4 (Fig. 1): A supported clade (76%/0.61) composed of four species: Pristimantis munozi, P. ocreatus, and two morphologically cryptic UCC. Pristimantis ocreatus is represented by specimens from at least six different locations along northern Ecuador, including its type locality (Tufiño, Carchi province, Ecuador) and exhibits genetic structure. It is sister to Pristimantis sp. 15, a deeply divergent UCC from Carchi province in northern Ecuador. Pristimantis sp. 14 is the successive sister of P. ocreatus and Pristimantis sp. 15, it is represented by specimens from three localities, also from Carchi province in northern Ecuador, and shows genetic structure. Finally, Pristimantis munozi, which is external to all other species in this subclade, is represented by type specimens from Verdecocha and an additional specimen from La Victoria, both localities are on the western slope of Pichincha volcano (Pichincha province, Ecuador). Uncorrected 16S p-distances among the species of this subclade ranges between 2.9 and 8.4% (Table S6). The sequence JX564889 (P. thymelensis) obtained from GenBank was recovered nested within P. ocreatus and shows none or minimal genetic divergent (0.0–0.8%) with other samples of this last species.
Subclade 5 (Fig. 1): A fully-supported clade (100%/1) formed by four lineages: Pristimantis gladiator, P. festae, and two CCS (Pristimantis sp. 16 and Pristimantis sp. 17). Pristimantis festae is represented by specimens of at least ten localities along northern Ecuador (Carchi, Imbabura, Pichincha, and Napo provinces), including its type locality (Papallacta, Napo province), shows genetic structure, with intraspecific p-distances between 0.0–1.9%. It is slightly divergent of Pristimantis sp. 17, its sister species. Pristimantis sp. 17 in turn is represented by specimens from three localities on the eastern slopes of the Ecuadorian Andes. Pristimantis sp. 16 is the successive sister species of these, is sympatric with Pristimantis sp. 17 at EcoMinga Reserve (Tungurahua province) and both are virtually indistinguishable from morphology. Finally, Pristimantis gladiator, which includes individuals from the type locality (Papallacta-Cuyuja, eastern versant of the Ecuadorian Andes) and surroundings, is the most external species and sister to all other lineages in the subclade. Interspecific genetic distances between all the lineages within this subclade ranges between 1.5–2.9% (Table S7).
Pristimantis gralarias (Fig. 1) is not treated as part of any subclade; it is sister to a large clade comprising the subclades 6 to 8. Uncorrected 16S p-distances between P. gralarias and all the other species in its sister clade are higher than 3.6%.
Subclade 6 (Fig. 1): A fully-supported clade (100%/1) only composed of a single CCS (Pristimantis sp. 18), from five localities in Orellana and Carchi provinces, Ecuador. Genetic distances between all the six terminals in this subclade range between 0.0 and 7.1% (Table S8).
Subclade 7 (Fig. 1): A well-supported clade (99%/1) composed of three species: Pristimantis hectus, P. lucidosignatus, and P. onorei. Pristimantis hectus is only represented by specimens from two different localities, distanced ca. 100 km from the type locality (Reserva La Planada, Colombia). It is sister to the group composed by the other two aforementioned species. Pristimantis onorei includes samples from four different localities at Pichincha (Las Gralarias and Bellavista reserves), Imbabura (Toisán), and Cotopaxi provinces (Otonga reserve, type locality), in Ecuador. Topotypic specimens morphologically attributable to P. lucidosignatus (Otonga reserve) were recovered intermixed and forming a polytomy with sympatric topotypic specimens of P. onorei, rendering the latter paraphyletic. Genetic distances between sympatric and topotypic specimens of P. lucidosignatus and P. onorei ranges between 0.0–0.3%; however, these distances increase to 0.9–2.9% between P. onorei from Toisán and other localities (Table S9). Finally, uncorrected p-distances between P. lucidosignatus + P. onorei and P. hectus were 2.9–6.4% (Table S9).
Discussion
Monophyly, phylogenetic relationships, and redefinition of the Pristimantis myersi group
Previous studies on the phylogenetic relationships of the genus Pristimantis inferred the P. myersi group—sensu Lynch & Duellman (1997); Hedges, Duellman & Heinicke (2008)—as monophyletic (Heinicke, Duellman & Hedges, 2007; Hedges, Duellman & Heinicke, 2008; Pyron & Wiens, 2011; Pinto-Sánchez et al., 2012; Guayasamin et al., 2015; González-Durán et al., 2017; Guayasamin, Arteaga & Hutter, 2018); however, in all of these studies the P. myersi species group was represented by a very limited taxon sampling, which included up to five species (P. festae, P. gralarias, P. ocreatus, and two unnamed species) which barely represent 13% of the named species currently assigned to the group. Sequences originally attributed to P. pyrrhomerus (KU 218030; GenBank accession number EF493683) and P. leoni (KU 218227; EF493684) by Heinicke, Duellman & Hedges (2007) correspond to two undescribed species (Pristimantis sp. 13 and P. sp. 18, respectively; see Fig. 1), whereas sequences referred as P. myersi by Guayasamin, Arteaga & Hutter (2018) (GenBank accession numbers: JX564889 and AY326009), originally referred as P. thymelensis by Darst & Cannatella (2004), were recovered as part of P. ocreatus in our phylogeny (see Fig. 1).
Pristimantis bicantus, P. albujai, P. sambalan, and P. nelsongalloi were all assigned to the P. myersi group in their original descriptions, solely on the basis of its overall similarity in external morphology (Guayasamin & Funk, 2009; Brito, Batallas & Yánez-Muñoz, 2017; Valencia et al., 2019); however, our phylogeny rejects these hypotheses, indicating that such morphological similarities may be better explained as evolutionary convergence of phenotypes among phylogenetically distant species. This phenomenon appears to be relatively common in Craugastoridae, where several phenetic groups have been shown to be non-monophyletic (Hedges, Duellman & Heinicke, 2008; Padial, Grant & Frost, 2014). Consequently, and in order to promote a supraspecific taxonomy based in natural groups (i.e., groups including a single common ancestral and all its descendants), we exclude P. albujai, P. bicantus, P. sambalan, and P. nelsongalloi from the P. myersi group. This action renders the P. myersi group monophyletic.
Pristimantis albujai appears in our topology closely related to P. cf. mendax, which also is part of the P. galdi group (Padial, Grant & Frost, 2014) according the redefinition proposed by Targino (2016) in its unpublished thesis. In contrast, Zumel, Buckley & Ron (2021), recovered P. albujai as part of the P. trachyblepharis species group, which is not closely related to the P. galdi group. Therefore, we cannot corroborate the allocation of P. albujai in the P. trachyblepharis group as proposed by Zumel, Buckley & Ron (2021). In turn, Pristimantis bicantus, P. nelsongalloi, and P. sambalan are all grouped in a well-supported clade in which is nested P. caprifer. The latter was relegated as unassigned to species group by Padial, Grant & Frost (2014) but later treated by Targino (2016) as part of her newly defined P. euphronides species group. Following Targino (2016), we suggest assigning P. bicantus, P. nelsongalloi, and P. sambalan as part of P. euphronides species group.
Based on an alleged congruence between morphological similarity and molecular-based topologies, Guayasamin, Arteaga & Hutter (2018) proposed a more inclusive redefinition of the P. myersi group, which encompassed its two successive sister clades, composed of P. verecundus, P. mutabilis, P. celator, P. jubatus, and several unnamed species closely related to these four species. Previously, Targino (2016) had also re-defined the P. myersi group in the same way, but only based in topology, because no unambiguously optimized phenotypic synapomorphy was recovered for that group. As far as we noted, these newly included species phenotypically differ from the species in the original P. myersi group at least in having digital discs expanded (weakly expanded to unexpanded in the P. myersi group; except in P. floridus, but its phylogenetic position has not been corroborated by molecular data), and by having discoidal fold (absent in the P. myersi group). Moreover, P. verecundus and its related species are arboreal (Lynch & Burrowes, 1990) whereas species in the P. myersi group are terrestrial or perch close to the ground (Hedges, Duellman & Heinicke, 2008; Padial, Grant & Frost, 2014). Pristimantis celator and P. jubatus further differ in a number of characters (i.e., absence of vocal slits) from all other species of the P. myersi group.
Taking into account that the expanded definition of the Pristimantis myersi group proposed by Targino (2016) and Guayasamin, Arteaga & Hutter (2018) lacks unambiguously optimized phenotypic synapomorphies, reduces its morphological diagnosability by making most characters in its definition polymorphic, and even is not supported in our study (Fig. 1), we conservatively propose returning to the previous definition of the P. myersi group (i.e., in our topology, the most inclusive clade containing P. pyrrhomerus and P. onorei, but not the clades of P. verecundus and P. jubatus; see Fig. 1), which is less inclusive but more phenotypically diagnosable.
Although herein excluded of the Pristimantis myersi group, we briefly discussed the species composition of P. verecundus and P. jubatus clades, as we discovered cryptic diversity to the species level within both. In the last clade, Pristimantis sp.1 (represented by the specimens UVC 15942 and 15043, from Cauca, Colombia) is sister and deeply divergent to P. jubatus (Fig. 1). It was originally sampled by Mendoza et al. (2015) and determined as Pristimantis hectus. We were able to examine a photo of one of these specimens in life and corroborate it is morphologically similar to P. hectus (including the characteristic lanceolate discs of that species). Nevertheless, this lineage is not phylogenetically related to our specimens of P. hectus which phenotypically matching with the species description and come from two localities about 45 km SW from La Planada (type locality). This lineage represents a typical case of cryptic species hidden by morphological convergence under another phylogenetically not related species (Patiño Ocampo, Duarte-Marín & Rivera-Correa, 2022).
Within the Pristimantis verecundus clade we discovered an astonishing cryptic diversity, with nine of the 12 independent evolutionary lineages that compose the clade (or 75%) corresponding to UCC. All of them were originally determined as P. verecundus based on their overall external morphology; however, our genetic evidence (i.e., topology and uncorrected p-distances) indicates that they presumably correspond to different species. All the species within this clade diverges in 6.3–14.6% from our samples of P. verecundus sensu stricto from Carchi province (Ecuador) about 45 km SW of La Planada, Colombia (type locality of P. verecundus). Further studies integrating morphology and bioacoustic data to our molecular evidence, will be required in order to corroborate the species limits within this clade.
In the subclade 1, the position of Pristimantis sirnigeli as part of the same clade of P. pyrrhomerus corroborates the close relationship between these two species already suggested by Yánez-Muñoz et al. (2010) solely on the basis of external morphology. On the other hand, based on a preliminary examination, Pristimantis sp. 11 (from Sigchos) is morphologically indistinguishable from topotypes of P. pyrrhomerus (from Pilaló) and their calls sound very similar (F.J.M. Rojas-Runjaic, pers. obs.), by which we originally determined the specimens from Sigchos as an additional population of P. pyrrhomerus. However, in our phylogenetic hypothesis these two are not sister lineages; Pristimantis sp. 11 is sister to P. sirnigeli, and P. pyrrhomerus is the successive sister of these two, so that P. sirnigeli renders P. pyrrhomerus paraphyletic (Fig. 1). Two different scenarios in which the monophyly of the group is preserved, can explain this situation: a) they are three deeply divergent conspecific lineages and P. sirnigeli is a junior synonym of P. pyrrhomerus, or b) Pristimantis sp. 11 is a morphologically cryptic species indistinguishable from P. pyrrhomerus but not directly related to that. Considering that, P. sirnigeli is readily distinguishable from their two successive sister lineages by morphology (e.g., P. sirnigeli has remarkably longer and thinner fingers, with digital discs more expanded than in P. pyrrhomerus, and dorsal skin much more tuberculate) and in their calls (F.J.M. Rojas-Runjaic, pers. obs.), and that genetic distances among these three lineages are greater than 2.2%, with P. sirnigeli being the most divergent (2.7–3.7%), we conclude that Pristimantis sp. 11 is in fact a morphologically cryptic CCS.
The subclade 3, included Pristimantis sp. 13, a CCS that has been sampled in all previous phylogenies of the P. myersi group. Despite this, its condition of as an undescribed species went unnoticed until now due it was erroneously determined as P. pyrrhomerus by Heinicke, Duellman & Hedges (2007), likely based on its similarity in external morphology. In the absence of sequences of P. pyrrhomerus of type locality (included by first time in this study), the error was perpetuated in subsequent phylogenies. Here we demonstrate that this lineage although seemingly similar to P. pyrrhomerus is not related to that. Pristimantis sp. 13 is the southern taxonomic replacement of its sister species P. leoni.
The subclade 4, includes Pristimantis sp. 14 and Pristimantis sp. 15. Although our genetic evidence (i.e., monophyly and genetic distances between 2.9 and 6.8%) strongly support these two lineages as independent species from P. ocreatus, we were unable to further analyze their morphology and bioacoustics; hence, we conservatively referred them as UCC. Lynch (1981) suggested that P. ocreatus is closely related to P. trepidotus (= P. festae), based on their similarity in size, proportions, hands and feet morphology, and color patterns. However, our phylogenetic analyses demonstrates that P. ocreatus is not closely related to P. festae.
Within subclade 5, we discovered two CCS (Pristimantis sp. 16 and P. sp. 17). Although they are sympatric at EcoMinga Reserve and are virtually indistinguishable in terms of morphology, they are not sister species. Pristimantis festae, to whom P. sp. 17 and P. sp. 16 are successive sister species, proved to be a widely distributed species in paramo environments of northeastern Ecuador. Its relatively wide geographic distribution is reflected in the genetic structured exhibited in our topology; however, besides it, the species shows a relatively low intraspecific genetic divergence (not higher than 1.9%).
The specimen QCAZ 11677 was determined as Pristimantis myersi and recovered as sister to P. festae in the unpublished phylogeny of Rojas-Runjaic (2012). In addition, that specimen was also determined as P. myersi (based on morphology) and illustrated in Rojas-Runjaic & Guayasamin (2015). Nevertheless, we treated this specimen herein as part of P. festae, mainly based on its incipient genetic divergence in relation to P. festae. Although alternatively, we could follow previous authors in treating QCAZ 11677 as P. myersi, and consequently also the more inclusive group containing it and the samples from San Francisco (which would render P. “myersi” and P. festae reciprocally monophyletic), the resulting intraspecific genetic divergence in P. “myersi” (up to 1.5%) would be similar to the divergence between it and P. festae (0.8–1.9%). We recognize the existence of sister species of amphibians exhibiting very low genetic distances, but they typically are further supported by the congruence of other lines of evidence, such as bioacoustics, and morphometry (e.g., Escalona et al., 2021). Given that we were unable to assess additional evidence on this issue, and faced with the impossibility of ruling out that the specimen QCAZ 11677 has been wrongly determined by Rojas-Runjaic & Guayasamin (2015), we conservatively referred it as part of P. festae.
Phylogenetic relationships among specimens of subclade 6, from five different localities (most of them in Carchi province, northern Ecuador) are not well resolved, and several of them exhibit remarkably long branches (likely due poor quality of the sequence TH 668). Despite this, the clade containing them is fully supported. We found high intra-specific genetic divergence reaching a variability up to 7%, even within the same population, similar studies that use mitochondrial DNA marker has been increasingly applied for evaluating the levels of genetic divergence, detecting barriers for gene flow that include Andean anurans (see Guarnizo, Amezquita & Bermingham, 2009; Rivera-Correa, Jimenez-Rivillas & Daza, 2017; Restrepo, Velasco & Daza, 2017; Rivera-Correa et al., 2022). We consider all of them conspecific and corresponding to Pristimantis sp. 18, an unnamed CCS. Within this clade is nested the specimen KU 218227, originally referred by Heinicke, Duellman & Hedges (2007) as P. leoni. Until now, this was the only available sequence for that species and consequently was included as such species in all subsequent phylogenies of the P. myersi group. Our topology, which is based in an exhaustive taxonomic and geographic sampling, reveals that the specimen KU 218227 is not related to P. leoni and actually correspond to Pristimantis sp. 18. Consequently, our study is also the first one to infer the phylogenetic position of P. leoni within the P. myersi group. We also highlight that the sequence of 16S available at GenBank of the specimen QCAZ 25589, first identified as P. librarius by Waddell et al. (2018) and presumably from an Amazonian locality, was recovered as part of the clade 6. It was inferred as sister to all the other five terminals of the clade 6, but barely diverges in 0.2% from three of them. Our evidence strongly suggest that this sample actually correspond to Pristimantis sp. 18 and likely comes from somewhere in the north of Carchi province. The Amazonian locality associated to this museum sample must be a mistake.
Subclade 7, groups Pristimantis hectus, P. lucidosignatus, and P. onorei. The type locality of Pristimantis hectus (La Planada) is located in the department of Nariño, southern Colombia (Lynch & Burrowes, 1990), ca. 45 km NE from the two localities sampled for this species in our study. There is not available molecular data from topotypes of P. hectus to compare with our specimens; however, we are confident that they are conspecific as their localities are nearby the type locality, and the specimens fully match with the morphological definition of P. hectus. However, based on the general pattern of geographic distribution exhibited by most of the species in the P. myersi group (i.e., species with small extent of occurrence), we consider unlikely that the geographic distribution of P. hectus extends much further south to Ecuador. There are some previous records of P. hectus for Pichincha, Cotopaxi and Imbabura (Appendix 1). These specimens morphologically resemble to that species but they likely correspond to different species.
Our phylogeny recovered P. onorei as paraphyletic due the position of topotypes morphologically attributable to P. lucidosignatus (i.e., bearing light markings on shanks) intermixed in a polytomy with topotypes of P. onorei. Thus, P. lucidosignatus might represent a junior synonym of P. onorei.
Definition of the Pristimantis myersi group and formalization of the subgenus Trachyphrynus
So far, no unambiguous phenotypic synapomorphies are known for the Pristimantis myersi group (Hedges, Duellman & Heinicke, 2008; Taboada et al., 2013). However, based on the newly inferred species composition of the group derived from our phylogeny, we phenotypically redefine it as follows (modified from Lynch & Duellman, 1997; Hedges, Duellman & Heinicke, 2008): (1) small body size (SVL in females 15.8–34.6 mm; in males 12.6–20.5 mm); (2) short snout; (3) robust body; (4) Toe V longer than Toe III, Finger I shorter than II; (5) digital discs narrow (expanded in P. floridus); and (6) cranial crests absent. In addition, all species in the group are found on low vegetation (<150 cm above the ground), at ground level, or underground. The Pristimantis myersi group as redefined herein, now contains 16 named species, namely: P. festae, P. floridus, P. gladiator, P. gralarias, P. hectus, P. leoni, P. lucidosignatus, P. munozi, P. myersi, P. ocreatus, P. onorei, P. pyrrhomerus, P. repens, P. scopaeus, P. sirnigeli, and P. xeniolum; and at least eight candidate species identified in our phylogeny. Despite P. repens, P. scopaeus, P. xeniolum, and P. floridus were not included in our phylogeny, we opted by maintain them as part of the group but highlighting that the assessment of their phylogenetic positions is pending.
Species groups have been widely and largely used in the taxonomy of Pristimantis to organize and to make more manageable this hiperdiverse genus (Lynch & Duellman, 1997; Hedges, Duellman & Heinicke, 2008); however, this taxonomic category is informal as it is not ruled by the ICZN (1999). To solve this issue, some authors have proposed the implementation of subgenera to formally name and define monophyletic and diagnosable clades within Pristimantis (e.g., Heinicke et al., 2018; Páez & Ron, 2019). Trachyphrynus (Goin & Cochran, 1963) was originally proposed as a genus name to allocate Trachyphrynus myersi (Goin & Cochran, 1963) but it was subsequently synonymized into Eleutherodactylus by Lynch (1968) and later into Pristimantis by Hedges, Duellman & Heinicke (2008). Considering that P. myersi is the nominal species of the homonymous species group, and that Trachyphrynus is an available name for the clade containing P. myersi, we implement Trachyphrynus as a formal subgenus name for the Pristimantis myersi group, as defined and delimited in this study.
Speciation
Lynch & Duellman (1997) described three general patterns of speciation in Ecuadorian Pristimantis: latitudinal, altitudinal, and trans-Andean replacement. Species in the P. myersi group mainly follow a pattern of latitudinal and altitudinal speciation rather than trans-Andean. For example, P. sirnigeli is sister to Pristimantis sp. 11 (Fig. 1); both species are on the Pacific versant of the Andes, but at different elevations and latitudes (Appendix 1, Fig. 2); the two species might be geographically isolated by the Toachi/Jacuntama/Sarapuyo/Pilatón river basin. A similar pattern is found between Pristimantis leoni and Pristimantis sp. 13 (Fig. 1). Pristimantis ocreatus is sister to Pristimantis sp. 15 (Fig. 1); the two species inhabit similar elevation and are geographically close, but isolated by a valley (Fig. 2). Canyons and dry valleys are the most likely geographic elements that disrupt gene flow (e.g., Lynch & Duellman, 1997; Coloma et al., 2012; Jungfer et al., 2013; Arteaga et al., 2016; Guayasamin et al., 2017; Guayasamin et al., 2020; Guayasamin et al., 2022; Yánez-Muñoz et al., 2018). For example, the Mira river valley restricts P. hectus to the north of Ecuador (Fig. 2); this basin is recognized as an important barrier for small vertebrates (Arteaga et al., 2016; Yánez-Muñoz et al., 2018). The canyon of the Pastaza River separates P. gladiator, Pristimantis sp. 16, and Pristimantis sp. 17 (Fig. 2). It seems that amphibian micro-endemism might mirror patterns of organisms already found in the same basin such as orchids and angiosperms (Jost, 2004; Jost & Shepard, 2017; Matsuda, 2018). There are also few examples of species that have reached new ecological zones (e.g., P. ocreatus found as high as higher 4,150 m).
This pattern agrees with the hypothesis that long mountain ranges fragmented by narrow transverse valleys promote allopatric divergence by limiting contact among contiguous populations, also explaining the limited distribution of numerous Andean frogs (Remsem, 1984; Graves, 1988; Wollenberg et al., 2008; Hutter & Guayasamin, 2015; Guayasamin et al., 2020; Guayasamin et al., 2022; Burrowes et al., 2020; De la Riva, 2020). A scenario of mostly allopatric speciation could serve to explain the presence of cryptic species; since selective pressures (biotic and abiotic) are similar for allopatric species that occupy analogous environmental niches; thus, ancestral morphologies and behaviors may be retained (Peterson, Soberón & Sánchez-Cordero, 1999; Graham et al., 2004; Wiens & Graham, 2005).
Wiens (2004) posited that lack of variability in populations, natural selection, pleiotropic effects, and gene flow from the centers of populations to their peripheries can act together to stabilize allopatric populations about their ancestral niche. If these processes are indeed at play in diverging lineages, we might then expect allopatric sister species in the P. myersi group to retain their ancestral morphologies and behaviors, resulting in cryptic species. However, while morphological traits are often informative of ecology, this is not necessarily the case (Losos, 2008). In order to make such a judgement in the case of cryptic species of Pristimantis, more detailed investigations into the relationships between ecology and the common morphology and behaviors in cryptic species pairs need to be conducted. Furthermore, it is generally unknown whether the traits shared by cryptic species in the P. myersi group are functional. In the absence of such information, sexual selection, or non-adaptive and non-directly selective explanations such as pleiotropy are equally plausible explanations for the retention of ancestral morphologies in allopatric cryptic species pairs as similar selective pressures. We expect such avenues of research to be particularly informative in developing a better understanding of cryptic speciation in Neotropical anurans.
Diversity of the Pristimantis myersi group in Ecuador
Numerous Pristimantis species remain undescribed because of the limited number of useful morphological traits for such a diverse genus. Thus, it should not surprise us that cryptic diversity is rampant when using alternative approaches, such as molecular phylogenies (Padial & De la Riva, 2009; Díaz, Hedges & Schmid, 2012; Padial et al., 2012; Fouquet et al., 2007; Kieswetter & Schneider, 2013; Hutter, Lambert & Wiens, 2017; Guayasamin et al., 2017; Guayasamin et al., 2022; Páez & Ron, 2019; Urgiles et al., 2019).
The higher species richness of the Pristimantis myersi group is concentrated in the montane forests of Ecuador. The group reaches its diversity peak in the western montane forest of the Andes, with nine named species (P. festae, P. floridus, P. gralarias, P. hectus, P. leoni, P. lucidosignatus, P. munozi, P. pyrrhomerus, P. onorei, and P. sirnigeli) in an altitudinal range from 1,589 to 3,487 m asl. Then, in the Paramo, with four named species (P. festae, P. gladiator, P. myersi, and P. ocreatus) in an altitudinal range from 3,853 to 4,068 m asl; and Andean shrub with two species (P. festae and P. leoni) in an altitudinal range from 2,834 to 2,901 m asl (Fig. 2 and Appendix 1).
Based on the results of this study, the number of species of the Pristimantis myersi group, as defined herein, would increase from 16 species to 23 (16 already recognized + 8 new candidate species), meaning that 34.8% of the known to date diversity of the P. myersi group is yet undescribed. Previous estimates of cryptic diversity on Neotropical amphibians’ range between 22–400%, but these studies are restricted to the Amazon region and are based on non-terraranan species (Fouquet et al., 2007; Funk, Caminer & Ron, 2012; Jungfer et al., 2013; Gehara et al., 2014; Rojas et al., 2018; Jaramillo et al., 2020). To date, no estimation of cryptic diversity for the Andean amphibians has been published. Our estimates, although restricted to a relatively small group, may apply to other Andean anurans with similar characteristics (e.g., Terrarana).
Impact on conservation
Clarifying taxonomic uncertainties is imperative not only to reveal true species richness within a group, but also for conservation purposes. Our study provides information that affects the conservation status of several species. Currently, the conservation status of five species of the Pristimantis myersi group have not been assessed under the criteria of the IUCN and Ortega-Andrade et al. (2021) (Table 2). Moreover, the extinction risk of several species requires a re-evaluation for the following reasons: (1) most species in the P. myersi group have geographic distributions that are actually smaller than currently recognized; (2) most species inhabit paramo and montane forest areas that are afflicted by severe anthropogenic factors (Menéndez-Guerrero & Graham, 2013); (3) several species (e.g., P. festae, P. leoni) actually represent species complexes. Specifically, we consider that the conservation status of six species within the group needs to be re-evaluated because they exhibit smaller distributions than previously thought; these species are: P. festae, P. gladiator, P. hectus, P. leoni, P. ocreatus, and P. pyrrhomerus (see Table 2). Finally, if the lineages identified as candidate species are described, this would require assessing their conservation status and, hopefully, designating priority areas for their conservation.
Table 2. Species in the Pristimantis myersi group, with information on the distribution and conservation status (IUCN and Ecuador’s Red List by Ortega-Andrade et al., 2021). Conservation status follows IUCN categories: Least concern (LC), Near Threatened (NT), Vulnerable (VU), Endangered (EN), No Evaluated (NE), and No information (–).
| Species | Distribution | IUCN | Ortega-Andrade et al. (2021) | Observations IUCN | Sources |
|---|---|---|---|---|---|
| P. festae | On the eastern slope of the Ecuadorian Andes, Sucumbíos, Napo and Tungurahua provinces and near southern Colombia. This study: Carchi, Pichincha, Imbabura, Napo province, Ecuador. | EN | VU | Requires re-evaluation | Coloma et al. (2004a), Frost (2023), Peracca (1904) and Ortega-Andrade et al. (2021) |
| P. floridus | Western flank of the Andes of Ecuador, Cotopaxi, Imbabura, and Pichincha provinces. This study: NA | VU | VU | − | Frost (2023), Lynch & Duellman (1997), Lynch, Coloma & Ron (2004) and Ortega-Andrade et al. (2021) |
| P. gladiator | On the Amazonian slopes of the Andes Carchi, Napo and Imbabura provinces; adjacent Putumayo, Colombia. This study: Napo province Ecuador. | VU | VU | Requires re-evaluation | Frost (2023), Almeida et al. (2019), Lynch (1976c) and Ortega-Andrade et al. (2021) |
| P. gralarias | Known only from the type locality, Ecuador (Pichincha province). This study: Type locality, Pichincha province, Ecuador. | − | VU | Requires evaluation | Frost (2023), Guayasamin, Arteaga & Hutter (2018) and Ortega-Andrade et al. (2021) |
| P. hectus | Western slope of the Cordillera Occidental in the Department of Nariño, Colombia; northwestern Ecuador (Esmeraldas and Imbabura provinces). This study: Carchi province, Ecuador. | VU | EN | Requires re-evaluation | Frost (2023), Cepeda-Quilindo et al. (2019), Lynch & Burrowes (1990) and Ortega-Andrade et al. (2021) |
| P. leoni | Southern Colombia and Amazonian slopes of the Andes in northern Ecuador (Carchi, Imbabura, Pichincha, Sucumbíos and Santo Domingo de los Tsáchilas provinces). This study: Only in the Imbabura and Pichincha provinces, Ecuador. | LC | EN | Requires re-evaluation | Castro et al. (2010), Frost (2023), Lynch (1976c), Lynch (1976b), Lynch (1976a) and Ortega-Andrade et al. (2021) |
| P. lucidosignatus | Known only from, Cotopaxi Province, and Tandapi, Pichincha Province, Ecuador.This Study: Cotopaxi province, Ecuador. | NE | DD | − | Frost (2023), Rödder & Schmitz (2009) and Ortega-Andrade et al. (2021) |
| P. munozi | Known only from the type locality (Pichincha province, Ecuador). This study: Type locality, Pichincha province, Ecuador. | − | VU | Requires evaluation | Frost (2023), Rojas-Runjaic, Delgado & Guayasamin (2014) and Ortega-Andrade et al. (2021) |
| P. myersi | On the southern, Cordillera Central in Colombia and in the North of Ecuador, Sucumbíos province. This study: We excluded Imbabura province. | LC | VU | − | Frost (2023), Castro, Herrera & Lymch (2004), Goin & Cochran (1963) and Ortega-Andrade et al. (2021) |
| P. ocreatus | Ecuador, Carchi, Imbabura, Napo and Cotopaxi provinces. This study: Carchi and Imbabura province, Ecuador. | EN | EN | Requires re-evaluation | Coloma et al. (2004b), Frost (2023), Lynch (1981a), Lynch (1981b) and Ortega-Andrade et al. (2021) |
| P. onorei | Ecuador, Santo Domingo de los Tsáchilas, Pichincha and Cotopaxi provinces. This study: Cotopaxi, Imbabura and Pichincha province, Ecuador. | − | DD | Requires evaluation | Frost (2023), Rödder & Schmitz (2009) and Ortega-Andrade et al. (2021) |
| P. pyrrhomerus | Ecuador, Pichincha, Imbabura, Carchi, Cotopaxi and Bolívar provinces. This study: Only Cotopaxi province, Ecuador. | EN | CR | Requires re-evaluation | Coloma et al. (2004c), Frost (2023), Lynch (1976b) and Ortega-Andrade et al. (2021) |
| P. repens | Colombia, Pasto and La Cruz, Department of Nariño. This study: NA | EN | − | − | Frost (2023), Rojas et al. (2019d) and Lynch (1984) |
| P. scopaeus | Known only from the type locality, Colombia (Municipal of Cajamarca). This study: NA | LC | − | − | Frost (2023), Castro, Gonzales-Duran & Herrera (2019) and Lynch, Carranza & Robayo (1996) |
| P. sirnigeli | Ecuador, Pichincha and Imbabura provinces. This study: Pichincha province, Ecuador. | − | EN | Requires evaluation | Frost (2023), Yánez-Muñoz et al. (2010) and Ortega-Andrade et al. (2021) |
| P. xeniolum | Known only from the type locality, Colombia (Department of Valle del Cauca). This study: NA | VU | − | − | Frost (2023), Castro et al. (2019) and Lynch (2001) |
Conclusions
Pristimantis myersi group (for which the name Trachyphrynus is applicable as subgenus), after the exclusion of P. albujai, P. bicantus, P. nelsongalloi, P. sambalan, P. jubatus, and P. verecundus clade, is monophyletic. Our study substantially contributes to a better understanding of the species richness and evolutionary relationships within the group. Species in the P. myersi group mainly follow mostly a pattern of latitudinal replacement rather than altitudinal or trans-Andean, promoting allopatric divergence. A scenario of allopatric speciation also explains the presence of cryptic species, but investigations into the relationships between ecology and the traits shared by particular cryptic species pairs are necessary to validate this hypothesis. Moving forward, obtaining calls would assist in solving species boundaries, particularly among sympatric lineages. We recommended an exhaustive taxonomic review of the Pristimantis myersi species group, as well as a re-evaluation of the conservation status of each species given the data provided herein.
Supplemental Information
Species names, group/clade, GenBank accession numbers, voucher numbers, coordinates and localities of Pristimantis myersi species group, P. verecundus clade, P. devillei, P. boulengeri and P. leptolophus species groups.
Node support is expressed in Bootstrap values; missing values indicate support below 70 (bootstrap). Each terminal includes the following information: GenBank code and species name. Highlighted branches indicated new sequences and excluded species of the Pristimantis myersi species group.
Node support is expressed in Bootstrap values; missing values indicate support below 70 (bootstrap). Each terminal includes the following information: GenBank code, species name and voucher number. Highlighted branches indicated sequences of Pristimantis myersi group + close relatives clade + excluded species.
Values are presented as percent distances calculated from uncorrected p values.
Acknowledgments
Special thanks to Diego Batallas-Revelo for his time and valuable information and field work in the province of Carchi. Instituto Nacional de Biodiversidad (INABIO), Museo de Zoología Universidad Tecnológica Indoamérica (MZUTI) and the Museo de Zoología de la Pontificia Universidad Católica de Ecuador, Quito (QCAZ-PUCE) provided tissues for phylogenetic analysis. FJMRR thanks Carlos Lasso, Paula Sánchez, Lina Mesa, Carlos Montaña, Claudia Múnera, and Claudia Medina from Instituto Alexander von Humboldt, Bogotá, Colombia (IAvH) who made possible the revision of the type series of Pristimantis hectus and P. verecundus. Also, thanks John D. Lynch, Jhon J. Ospina-Sarria, Teddy Angarita-Sierra, and Marvin Anganoy from the Instituto de Ciencias Naturales, Bogotá, Colombia (ICN) at the Universidad Nacional de Colombia, for allowing access to the type series of P. repens, P. scopaeus, and P. xeniolum; to Belisario Cepeda (Universidad de Nariño, Pasto, Colombia) who granted access to the specimens of P. myersi housed in the Colección Zoológica at the Universidad de Nariño (PSO-CZ); and to Ángela Mendoza-Henao (IAvH) for kindly sharing some information and photos of Colombian specimens. Technical support in laboratory was provided by Gabriela Gavilanes (LBE), and Diana Flores (BioCamb-UTI). Thanks to Mateo A. Vega-Yánez for his contribution in Map design. In addition, thanks to the people in the fieldwork and specimen collections (tissues) Juan Pablo Reyes and Javier Robayo (EcoMinga Reserve), Ítalo Tapia, Amanda Delgado, Jorge Brito, José Vieira, Alejandro Arteaga, Lucas Bustamente, Frank Pichardo, Jaime García, Rocío Vargas, and Kelsey Huisman. Fieldwork at Verdecocha Ecological Reserve was authorized by Mr. Jorge Enrique Maldonado. Finally, we are grateful to Nora Oleas and Tasman Rosenfeld for his accurate review comments and for the English review throughout the manuscript.
Funding Statement
This study was funded by the Universidad San Francisco de Quito (HUBI 5467, 5466; 11164, 16871). The Programa Inédita from Secretaría de Educación Superior, Ciencia, Tecnología e Innovación (Project PIC-20-INE-USFQ-001: Respuestas a la crisis de biodiversidad: la descripción de especies como herramienta de conservación) funded the molecular component of this work. Fernando J.M. Rojas-Runjaic is currently supported by National Council for Scientific and Technological Development (CNPq, PCI-MPEG/MCTIC, process # 300895/2022-4). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Contributor Information
Daniela Franco-Mena, Email: daniellafrancomena@gmail.com.
Fernando J.M. Rojas-Runjaic, Email: rojas_runjaic@yahoo.com.
Additional Information and Declarations
Competing Interests
The authors declare there are no competing interests.
Author Contributions
Daniela Franco-Mena conceived and designed the experiments, performed the experiments, analyzed the data, prepared figures and/or tables, authored or reviewed drafts of the article, and approved the final draft.
Juan M Guayasamin conceived and designed the experiments, performed the experiments, analyzed the data, prepared figures and/or tables, authored or reviewed drafts of the article, and approved the final draft.
Diego Andrade-Brito analyzed the data, authored or reviewed drafts of the article, and approved the final draft.
Mario H Yánez-Muñoz analyzed the data, authored or reviewed drafts of the article, and approved the final draft.
Fernando JM Rojas-Runjaic conceived and designed the experiments, performed the experiments, analyzed the data, prepared figures and/or tables, authored or reviewed drafts of the article, and approved the final draft.
Field Study Permissions
The following information was supplied relating to field study approvals (i.e., approving body and any reference numbers):
The Ministerio de Ambiente y Agua de Ecuador, provided permits for investigation, handling and collection of specimens: MAE-DNB-CM-2018-0105.
DNA Deposition
Data Availability
The following information was supplied regarding data availability:
Raw data, including Pristimantis myersi group genetic sequences, are available in the Supplemental Files.
References
- Almeida et al. (2019).Almeida D, Nogales F, Mueses-Cisneros JJ, Coloma LA, Ron SR. Pristimantis gladiator. The IUCN Red List of Threatened Species 2019: e.T56615A85863388. Downloaded on 08 November 2021. 2019. [DOI]
- Anisimova & Gascuel (2006).Anisimova M, Gascuel O. Approximate likelihood-ratio test for branches: a fast, accurate, and powerful alternative. Systematic Biology. 2006;55(4):539–552. doi: 10.1080/10635150600755453. [DOI] [PubMed] [Google Scholar]
- Arteaga et al. (2016).Arteaga A, Pyron RA, Peñafiel N, Romero-Barreto P, Culebras J, Bustamante L, Guayasamin JM. Comparative phylogeography reveals cryptic diversity and repeated patterns of cladogenesis for amphibians and reptiles in northwestern Ecuador. PLOS ONE. 2016;11(4):e0151746. doi: 10.1371/journal.pone.0151746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avise (2000).Avise JC. Phylogeography: the history and formation of species. Harvard University Press; Cambridge: 2000. p. 464 pp. [DOI] [Google Scholar]
- Beaupre et al. (2004).Beaupre SJ, Jacobson ER, Lillywhite HB, Zamudio K. Guidelines for use of live amphibians and reptiles in field and laboratory research. Herpetological Animal Care and Use Committee of the American Society of Ichthyologists and Herpetologists Lawrence: American Society of Ichthyologist and Herpetologist; 2004. [Google Scholar]
- Bickford et al. (2007).Bickford D, Lohman DJ, Sodhi N, Meier R, Winker K, Ingram KK, Das I. Cryptic species as a window on diversity and conservation. Ecology and Evolution. 2007;22(3):148–155. doi: 10.1016/j.tree.2006.11.004. [DOI] [PubMed] [Google Scholar]
- Blotto et al. (2021).Blotto BL, Lyra ML, Cardoso MCS, Rodriguez MT, Dias IR, Marciano-Jr E, Dal Vechio F, Orrico VGD, Brandão RA, De Assis CL, Lantyer-Silva ASF, Rutherford MG, Gagliardi-Urrutia G, Solé M, Baldo D, Nunes I, Cajade R, Torres A, Grant T, Jungfer K-H, da Silva HR, Haddad CFB, Faivovich J. The phylogeny of the Casque-headed Treefrogs (Hylidae: Hylinae: Lephyohylini) Cladistics. 2021;37(1):36–72. doi: 10.1111/cla.12409. [DOI] [PubMed] [Google Scholar]
- Brito, Batallas & Yánez-Muñoz (2017).Brito J, Batallas D, Yánez-Muñoz MH. Ranas terrestres Pristimantis (Anura: Craugastoridae) de los bosques montanos del río Upano, Ecuador: Lista anotada, patrones de diversidad y descripción de cuatro especies nuevas. Neotropical Biodiversity. 2017;3:125–126. doi: 10.1080/23766808.2017.1299529. [DOI] [Google Scholar]
- Burrowes et al. (2020).Burrowes PA, Navas CA, Jiménez-Robles O, Delgado P, De la Riva I. Climatic heterogeneity in the Bolivian Andes: are frogs trapped? South American Journal of Herpetology. 2020;18(1):1–12. doi: 10.2994/SAJH-D-18-00047.1. [DOI] [Google Scholar]
- Camargo & Sites (2013).Camargo A, Sites JJR. Species delimitation: a decade after the renaissance. In: Pavlinov IY, editor. The Species Problem. Ongoing Issues. London: IntechOpen; 2013. pp. 225–248. [DOI] [Google Scholar]
- Caminer & Ron (2014).Caminer MA, Ron SR. Systematics of treefrogs of the Hypsiboas calcaratus and Hypsiboas fasciatus species complex (Anura, Hylidae) with the description of four new species. ZooKeys. 2014;370:1–68. doi: 10.3897/zookeys.370.6291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castro, Gonzales-Duran & Herrera (2019).Castro F, Gonzales-Duran GA, Herrera I. Pristimantis scopaeus. The IUCN Red List of Threatened Species 2019: e.T56959A85882581. Downloaded on 08 November 2021. 2019 doi: 10.2305/IUCN.UK.2019-2.RLTS.T56959A85882581.en. [DOI]
- Castro et al. (2010).Castro F, Herrera M, Coloma LA, Ron SR, Lynch J, Almeida D, Nogales F, Yánez-Muñoz M. Pristimantis leoni (errata version published in 2016). The IUCN Red List of Threatened Species 2010: e.T56713A86448562. 08 November 2021. 2010. [DOI]
- Castro, Herrera & Lymch (2004).Castro F, Herrera MI, Lymch J. Pristimantis myersi. The IUCN Red List of Threatened Species 2004: e.T56781A11521544. Downloaded on 08 November 2021. 2004. [DOI]
- Castro et al. (2019).Castro F, Medina Rangel GF, Gonzales Duran GA, Herrera I, Lynch J. Pristimantis xeniolum. The IUCN Red List of Threatened Species 2019: e.T57058A85889291. Downloaded on 11 July 2020. 2019. [DOI]
- Cepeda-Quilindo et al. (2019).Cepeda-Quilindo B, Castro F, Cantillo G, Lymch J, Coloma LA, Herrera M, Anganoy M, Ron SR. Pristimantis hectus. The IUCN Red List of Threatened Species 2019: e.T56647A85862717. Downloaded on 08 November 2021. 2019. [DOI]
- Coloma et al. (2012).Coloma LA, Carvajal-Endara S, Dueñas JF, Paredes-Recalde A, Morales-Mite M, Almeida-Reinoso D, Tapia EE, Hutter CR, Toral E, Guayasamín JM. Molecular phylogenetics of stream frogs of the Hyloscirtus larinopygion group (Anura: Hylidae), and description of two new species from Ecuador. Zootaxa. 2012;3364:1–78. doi: 10.11646/zootaxa.3364.1.1. [DOI] [Google Scholar]
- Coloma et al. (2004a).Coloma LA, Ron S, Almeida D, Yánez-Muñoz M. Pristimantis festae (errata version published in 2017). The IUCN Red List of Threatened Species 2004: e.T57014A115203650. Downloaded on 08 November 2021. 2004a. [DOI]
- Coloma et al. (2004c).Coloma LA, Ron SR, Yánez-Muñoz M, Almeida D. Pristimantis pyrrhomerus. The IUCN Red List of Threatened Species 2004: e.T56892A11539576. Downloaded 08 November 2021. 2004c. [DOI]
- Coloma et al. (2004b).Coloma LA, Ron SR, Yánez-Muñoz M, Nogales F. Pristimantis ocreatus. The IUCN Red List of Threatened Species 2004:e.T56804A11534677. Downloaded on 08 November 2021. 2004b. [DOI]
- Cryer et al. (2019).Cryer J, Wynne F, Price SJ, Puschendorf R. Cryptic diversity in Lithobates warszewitschii (Amphibia, Anura, Ranidae) ZooKeys. 2019;838:49–69. doi: 10.3897/zookeys.838.29635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darst & Cannatella (2004).Darst CR, Cannatella DC. Novel relationships among hyloid frogs inferred from 12S and 16S mitochondrial DNA sequences. Molecular Phylogenetics and Evolution. 2004;31:462–475. doi: 10.1016/j.ympev.2003.09.003. [DOI] [PubMed] [Google Scholar]
- De la Riva (2020).De la Riva I. Unexpected beta-diversity radiations in highland clades of andean terraranae frogs. In: Rull V, Carnaval A, editors. Neotropical diversification: patterns and processes. Fascinating life sciences. Springer; Cham: 2020. pp. 741–764. [DOI] [Google Scholar]
- Díaz, Hedges & Schmid (2012).Díaz LM, Hedges SB, Schmid M. A new cryptic species of the genus Eleutherodactylus (Amphibia: Anura: Eleutherodactylidae) from Cuba. Zootaxa. 2012;3220:44–60. doi: 10.11646/zootaxa.3220.1.3. [DOI] [Google Scholar]
- Duellman & Lehr (2009).Duellman WE, Lehr E. Terrestrial breeding frogs (Strabomantidae) in Peru. Münster: Nature und Tiere Verlag, Naturwissenschaft; 2009. p. 382 pp. [Google Scholar]
- Elmer & Cannatella (2009).Elmer KR, Cannatella DC. Three new species of leaf litter frogs from the upper Amazon forests: cryptic diversity within Pristimantis ockendeni (Anura: Strabomantidae) in Ecuador. Zootaxa. 2009;1784:11–38. doi: 10.11646/zootaxa.1784.1.2. [DOI] [Google Scholar]
- Escalona et al. (2021).Escalona M, La Marca E, Castellanos M, Fouquet A, Crawford AJ, Rojas-Runjaic FJM, Giaretta AA, Señaris JC, Castroviejo-Fisher S. Integrative taxonomy reveals a new but common Neotropical treefrog, hidden under the name Boana xerophylla. Zootaxa. 2021;4981:401–448. doi: 10.11646/zootaxa.4981.3.1. [DOI] [PubMed] [Google Scholar]
- Felsenstein (1985).Felsenstein J. Confidence limits on Phylogenies: an approach using the bootstrap. Evolution. 1985;39(4):783–791. doi: 10.2307/2408678. [DOI] [PubMed] [Google Scholar]
- Jiménez de la Espada (1870).Jiménez de la Espada M. Fauna neotropicalis species quaedam nondum cognitae. Jornal de Sciências, Mathemáticas, Physicas e Naturaes. 1870;3:57–65. [Google Scholar]
- Fouquet et al. (2007).Fouquet A, Gilles A, Vences M, Marty C, Blanc M, Gemmell NJ. Underestimation of species richness in neotropical frogs revealed by mtDNA analyses. PLOS ONE. 2007;2:1–10. doi: 10.1371/journal.pone.0001109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frost (2023).Frost DR. Amphibian Species of the World: an Online Reference. Electronic Database accessible at American Museum of Natural History, New York, USA. Version 6.1https://amphibiansoftheworld.amnh.org/index.php. [8 February 2023];2023 doi: 10.5531/db.vz.0001. [DOI]
- Funk, Caminer & Ron (2012).Funk WC, Caminer M, Ron SR. High levels of cryptic species diversity uncovered in Amazonian frogs. Proceedings of the Royal Society, Biological Sciences. 2012;279:1806–1814. doi: 10.1098/rspb.2011.1653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García & Lynch (2006).García JC, Lynch JD. A new species of frog (genus Eleutherodactylus) from a cloud forest in Western Colombia. Zootaxa. 2006;1171:39–45. doi: 10.11646/zootaxa.1171.1.4. [DOI] [Google Scholar]
- García et al. (2012).García RJC, Crawford AJ, Mendoza ÁM, Ospina O, Cardenas H, Castro F. Comparative phylogeography of direct-developing frogs (Anura: Craugastoridae: Pristimantis) in the Southern Andes of Colombia. PLOS ONE. 2012;7:e46077. doi: 10.1371/journal.pone.0046077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gehara et al. (2014).Gehara M, Crawford AJ, Orrico VGD, Rodriguez A, Lötters S, Fouquet A, Baldo D, Barrientos LS, Brusquetti F, Castroviejo-Fisher S, De la Riva I, Ernst R, Faivovich J, Gagliardi-Urrutia G, Glaw F, Guayasamin J, Hölting M, Jansen M, Kok PJR, Kwet A, Lingnau R, Lyra M, Moravec J, Padial JM, Pombal J, Rojas-Runjaic FJM, Schulze A, Señaris JC, Solé M, Rodriguez MT, Twomey E, Haddad CFB, Vences M, Köhler J. High levels of diversity uncovered in a widespread nominal taxon: continental phylogeography of the Neotropical tree frog Dendropsophus minutus. PloS ONE. 2014;9:e103958. doi: 10.1371/journal.pone.0103958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goin & Cochran (1963).Goin CJ, Cochran DM. Two new genera of leptodactylid frogs from Colombia. Proceedings of the California Academy of Sciences. 1963;31(17):499–505. [Google Scholar]
- González-Durán et al. (2017).González-Durán GA, Targino M, Marco R, Grant T. Phylogenetic relationships and morphology of the Pristimantis leptolophus species group (Amphibia: Anura: Brachycephaloidea), with the recognition of a new species group in Pristimantis Jiménez de la Espada, 1870. Zootaxa. 2017;4243:42–74. doi: 10.11646/zootaxa.4243.1.2. [DOI] [PubMed] [Google Scholar]
- Graham et al. (2004).Graham CH, Ron SR, Santos JC, Schneider CJ, Moritz C. Integrating phylogenetics and environmental niche models to explore speciation mechanisms in dendrobatid frogs. Evolution. 2004;5:1781–1793. doi: 10.1111/j.0014-3820.2004.tb00461. [DOI] [PubMed] [Google Scholar]
- Graves (1988).Graves GR. Linearity of geographic range and its possible effect on the population structure of andean birds. The Auk. 1988;105:47–52. doi: 10.1093/auk/105.1.47. [DOI] [Google Scholar]
- Guarnizo, Amezquita & Bermingham (2009).Guarnizo CE, Amezquita A, Bermingham E. The relative roles of vicariance versus elevational gradients in the genetic differentiation of the high Andean tree frog, Dendropsophus labialis. Molecular Phylogenetics and Evolution. 2009;50:84–92. doi: 10.1016/j.ympev.2008.10.005. [DOI] [PubMed] [Google Scholar]
- Guayasamin, Arteaga & Hutter (2018).Guayasamin JM, Arteaga A, Hutter CR. A new (singleton) rainfrog of the Pristimantis myersi Group (Amphibia: Craugastoridae) from the northern Andes of Ecuador. Zootaxa. 2018;4527:323–334. doi: 10.11646/zootaxa.4527.3.2. [DOI] [PubMed] [Google Scholar]
- Guayasamin et al. (2022).Guayasamin JM, Brunner RM, Valencia-Aguilar A, Franco-Mena D, Ringler E, Medina Armijos A, Morochz C, Bustamante L, Maynard RJ, Culebras J. Two new glassfrogs (Centrolenidae: Hyalinobatrachium) from Ecuador, with comments on the endangered biodiversity of the Andes. PeerJ. 2022;10:e13109. doi: 10.7717/peerj.13109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guayasamin et al. (2008).Guayasamin JM, Castroviejo-Fisher S, Ayarzagüena J, Trueb L, Vilà C. Phylogenetic relationships of glassfrogs (Centrolenidae) based on mitochondrial and nuclear genes. Molecular Phylogenetics and Evolution. 2008;48:574–595. doi: 10.1016/j.ympev.2008.04.012. [DOI] [PubMed] [Google Scholar]
- Guayasamin et al. (2020).Guayasamin JM, Cisneros-Heredia DF, McDiarmid RW, Peña P, Hutter CR. Glassfrogs of ecuador: diversity, evolution, and conservation. Diversity. 2020;12:222. doi: 10.3390/d12060222. [DOI] [Google Scholar]
- Guayasamin & Funk (2009).Guayasamin JM, Funk WC. The amphibian community at Yanayacu Biological Station, Ecuador, with a comparison of vertical microhabitat use among Pristimantis species and the description of a new species of the Pristimantis myersi group. Zootaxa. 2009;2220:41–66. doi: 10.11646/zootaxa.2220.1.2. [DOI] [Google Scholar]
- Guayasamin et al. (2017).Guayasamin JM, Hutter CR, Tapia EE, Culebras J, Peñafiel N, Pyron RA, Morochz C, Funk WC, Arteaga A. Diversification of the rainfrog Pristimantis ornatissimus in the lowlands and Andean foothills of Ecuador. PLOS ONE. 2017;12:e0172615. doi: 10.1371/journal.pone.0172615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guayasamin et al. (2015).Guayasamin JM, Krynak T, Krynak K, Culebras J, Hutter CR. Phenotypic plasticity raises questions for taxonomically important traits: a remarkable new Andean rainfrog (Pristimantis) with the ability to change skin texture. Zoological Journal of the Linnean Society. 2015;173:913–928. doi: 10.1111/zoj.12222. [DOI] [Google Scholar]
- Hedges, Duellman & Heinicke (2008).Hedges SB, Duellman WE, Heinicke MP. New World direct-developing frogs (Anura: Terrarana): molecular phylogeny, classification, biogeography, and conservation. Zootaxa. 2008;1737:1–182. doi: 10.11646/ZOOTAXA.1737.1.1. [DOI] [Google Scholar]
- Heinicke, Duellman & Hedges (2007).Heinicke MP, Duellman WE, Hedges SB. Major Caribbean and Central American frog faunas originated by ancient oceanic dispersal. Proceedings of the National Academy of Sciences of the United States of America. 2007;104:10092–10097. doi: 10.1073/pnas.0611051104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinicke et al. (2018).Heinicke MP, Lemmon AR, Lemmon EM, McGrath K, Hedges SB. Phylogenomic support for evolutionary relationships of New World direct-developing frogs (Anura: Terraranae) Molecular Phylogenetics and Evolution. 2018;118:145–155. doi: 10.1016/j.ympev.2017.09.021. [DOI] [PubMed] [Google Scholar]
- Hoang et al. (2018).Hoang DT, Chernomor O, Haeseler A, Minh BQ, Vinh LS. UFBoot2: improving the ultrafast bootstrap approximation. Molecular Biology and Evolution. 2018;35(2):518–522. doi: 10.1093/molbev/msx281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutter & Guayasamin (2015).Hutter CR, Guayasamin JM. Cryptic diversity concealed in the Andean cloud forests: two new species of rainfrogs (Pristimantis) uncovered by molecular and bioacoustic data. Neotropical Biodiversity. 2015;1:36–59. doi: 10.1080/23766808.2015.1100376. [DOI] [Google Scholar]
- Hutter, Lambert & Wiens (2017).Hutter CR, Lambert SM, Wiens JJ. Rapid diversification and time explain amphibian richness at different scales in the Tropical Andes, Earth’s most biodiverse hotspot. The American Naturalist. 2017;190(6):828–843. doi: 10.5061/dryad.1555n. [DOI] [PubMed] [Google Scholar]
- ICZN (1999).ICZN The International Trust for Zoological Nomenclature. London, U.Khttps://www.iczn.org/ International Code of Zoological Nomenclature. Fourth Edition: adopted by the International Union of Biological Sciences. 1999 ISBN 0 85301 006 4.
- Janzen (2004).Janzen DH. Now is the time. Philosophical Transactions of the Royal Society B: Biological Sciences. 2004;359:731–732. doi: 10.1098/rstb.2003.1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaramillo et al. (2020).Jaramillo AF, De La Riva I, Guayasamin JM, Chaparro JC, Gagliardi-Urrutia G, Gutiérrez RC, Castroviejo-Fisher S. Vastly underestimated species richness of Amazonian salamanders (Plethodontidae: Bolitoglossa) and implications about plethodontid diversification. Molecular Phylogenetics and Evolution. 2020;149:106841. doi: 10.1016/j.ympev.2020.106841. [DOI] [PubMed] [Google Scholar]
- Jost (2004).Jost L. New Pleurothallid Orchids from the Cordillera del Condor of Ecuador. Selbyana. 2004;25:11–16. [Google Scholar]
- Jost & Shepard (2017).Jost L, Shepard A. Four new Teagueia (Orchidaceae: Pleurothallidinae) from the upper río Pastaza watershed of east-central Ecuador. Lankesteriana. 2017;17(2):261–278. doi: 10.15517/lank.v17i2.30159. [DOI] [Google Scholar]
- Jungfer et al. (2013).Jungfer K-H, Faivovich JA, Padial JM, Castroviejo-Fisher S, Lyra MM, Berneck BVM, Iglesias PP, Kok PJR, MacCulloch RD, Rodrigues MT, Verdade VK, Torres GCP, Chaparro JC, Valdujo PH, Reichle S, Moravec J, Gvoždík V, Gagliardi-Urrutia G, Ernst R, De la Riva I, Means DB, Lima AP, Señaris JC, Wheeler WC, Haddad CFB. Systematics of spiny-backed treefrogs (Hylidae: Osteocephalus): an Amazonian puzzle. Zoologica Scripta. 2013;42(4):351–380. doi: 10.1111/zsc.12015. [DOI] [Google Scholar]
- Kalyaanamoorthy et al. (2017).Kalyaanamoorthy S, Minh BQ, Wong TK, Haeseler Avon, Jermiin LS. ModelFinder: fast model selection for accurate phylogenetic estimates. Nature Methods. 2017;14(6):587–589. doi: 10.1038/nmeth.4285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katoh & Standley (2013).Katoh K, Standley DM. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Molecular Biology and Evolution. 2013;30:772–780. doi: 10.1093/molbev/mst010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kieswetter & Schneider (2013).Kieswetter CM, Schneider CJ. Phylogeography in the northern Andes: complex history and cryptic diversity in a cloud forest frog, Pristimantis w-nigrum (Craugastoridae) Molecular Phylogenetics and Evolution. 2013;69(3):417–429. doi: 10.1016/j.ympev.2013.08.007. [DOI] [PubMed] [Google Scholar]
- Kishino & Hasegawa (1989).Kishino H, Hasegawa M. Evaluation of the maximum likelihood estimate of the evolutionary tree topologies from DNA sequence data, and the branching order in hominoidea. Molecular Biology and Evolution. 1989;29:170–179. doi: 10.1007/BF02100115. [DOI] [PubMed] [Google Scholar]
- Kishino, Miyata & Hasegawa (1990).Kishino H, Miyata T, Hasegawa M. Maximum likelihood inference of protein phylogeny and the origin of chloroplasts. Molecular Biology and Evolution. 1990;31:151–160. doi: 10.1007/BF02109483. [DOI] [Google Scholar]
- Kumar, Stecher & Tamura (2016).Kumar S, Stecher G, Tamura K. MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Molecular Biology and Evolution. 2016;33(7):1870–1874. doi: 10.1093/molbev/msw054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letunic & Bork (2021).Letunic I, Bork P. Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Research. 2021;49(W1):W293–W296. doi: 10.1093/nar/gkab301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Losos (2008).Losos JB. Phylogenetic niche conservatism, phylogenetic signal and the relationship between phylogenetic relatedness and ecological similarity among species. Ecology Letters. 2008;11:995–1007. doi: 10.1111/j.1461-0248.2008.01229.x. [DOI] [PubMed] [Google Scholar]
- Lynch (1968).Lynch JD. Systematic Status of Some Andean leptodactylid frogs with a description of a new species of Eleutherodactylus. Herpetologica. 1968;24(4):289–300. [Google Scholar]
- Lynch (1976a).Lynch JD. New species of frogs (Leptodactylidae: Eleutherodactylus) from the Pacific versant of Ecuador. Occasional Papers of the Museum of Natural History, the University of Kansas. 1976a;55:1–33. [Google Scholar]
- Lynch (1976b).Lynch JD. Three new leptodactylid frogs (genus Eleutherodactylus) from the Andean slopes of Colombia and Ecuador. Herpetologica. 1976b;32:310–317. [Google Scholar]
- Lynch (1976c).Lynch JD. The species groups of the South American frogs of the genus Eleutherodactylus (Leptodactylidae) Occasional Papers of the Museum of Natural History, University of Kansas. 1976c;61:1–24. [Google Scholar]
- Lynch (1981a).Lynch JD. Leptodactylid frogs of the genus Eleutherodactylus in the Andes of northern Ecuador and adjacent Colombia. Miscellaneous Publication. The University of Kansas. Museum of Natural History. 1981a;72:1–46. doi: 10.5962/bhl.title.16289. [DOI] [Google Scholar]
- Lynch (1981b).Lynch JD. Two new species of Eleutherodactylus from western Colombia (Amphibia: Anura: Leptodactylidae) University of Michigan Museum of Zoology, Occasional Papers. 1981b;697:1–12. [Google Scholar]
- Lynch (1984).Lynch JD. A new species of Eleutherodactylus (Amphibia: Anura: Leptodactylidae) from southern Andean Colombia. Herpetologica. 1984;40(3):234–237. [Google Scholar]
- Lynch (2001).Lynch JD. A small amphibian fauna from a previously unexplored paramo of the Cordillera Occidental in western Colombia. Journal of Herpetology. 2001;35:226–231. doi: 10.2307/1566112. [DOI] [Google Scholar]
- Lynch & Burrowes (1990).Lynch JD, Burrowes PA. The frogs of the genus Eleutherodactylus (family Leptodactylidae) at the La Planada reserve in Southwestern Colombia with descriptions of eight new species. Occasional Papers of the Museum of Natural History, The University of Kansas. 1990;136:1–31. [Google Scholar]
- Lynch, Carranza & Robayo (1996).Lynch JD, Carranza PMR, Robayo MCA. Three new species of Eleutherodactylus (Amphibia: Leptodactylidae) from high elevations of the Cordillera Central of Colombia. Caldasia. 1996;18(3):329–342. [Google Scholar]
- Lynch, Coloma & Ron (2004).Lynch JD, Coloma LA, Ron S. Pristimantis floridus. The IUCN Red List of Threatened Species 2004: e.T56604A11501252. Downloaded on 08 November 2021. 2004. [DOI]
- Lynch & Duellman (1997).Lynch JD, Duellman WE. Frogs of the genus Eleutherodactylus (Leptodactylidae) in Western Ecuador: systematics, ecology, and biogeography. The University of Kansas Natural History Museum, Special Publications. 1997;23:1–236. doi: 10.5962/bhl.title.7951. [DOI] [Google Scholar]
- Maddison & Maddison (2019).Maddison W, Maddison WP. Mesquite: a modular system for evolutionary analysis. version 3.61http://mesquiteproject.org 2019
- Matsuda (2018).Matsuda E. Blooming mountains of biodiversity: an investigation of angiosperms on Cerro Mayordomo, Tungurahua, Ecuador. Independent Study Project (ISP) Collection 2783. 2018. https://digitalcollections.sit.edu/isp_collection/2783 https://digitalcollections.sit.edu/isp_collection/2783
- Mendoza et al. (2015).Mendoza ÁM, Ospina OE, Cárdenas-Henao H, García RJC. A likelihood inference of historical biogeography in the world’s most diverse terrestrial vertebrate genus: diversification of direct-developing frogs (Craugastoridae: Pristimantis) across the Neotropics. Molecular Phylogenetics and Evolution. 2015;85:50–58. doi: 10.1016/j.ympev.2015.02.001. [DOI] [PubMed] [Google Scholar]
- Menéndez-Guerrero & Graham (2013).Menéndez-Guerrero PA, Graham CH. Evaluating multiple causes of amphibian declines of Ecuador using geographical quantitative analyses. Ecography. 2013;36(7):756–769. doi: 10.1111/j.1600-0587.2012.07877.x. [DOI] [Google Scholar]
- Minh, Nguyen & von Haeseler (2013).Minh BQ, Nguyen MAT, von Haeseler A. Ultrafast approximation for phylogenetic bootstrap. Molecular Biology and Evolution. 2013;30(5):1188–1195. doi: 10.1093/molbev/mst024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nei (1972).Nei M. Genetic distance between between populations. American Naturalist. 1972;106:283–292. doi: 10.1086/282771. [DOI] [Google Scholar]
- Nguyen et al. (2015).Nguyen LT, Schmidt HA, von Haeseler A, Minh BQ. IQ-TREE: a fast and effective ftochastic flgorithm for estimating maximum-likelihood phylogenies. Molecular Biology and Evolution. 2015;32(1):268–274. doi: 10.1093/molbev/msu300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patiño Ocampo, Duarte-Marín & Rivera-Correa (2022).Patiño Ocampo E, Duarte-Marín S, Rivera-Correa M. Genética, bioacústica y morfología revelan una nueva especie oculta en Pristimantis dorsopictus (Anura: Strabomantidae) Revista Latinoamericana de Herpetología. 2022;5(1):60–90. doi: 10.22201/fc.25942158e.2022.1.305. [DOI] [Google Scholar]
- Ortega-Andrade et al. (2021).Ortega-Andrade HM, Rodes Blanco M, Cisneros-Heredia DF, Guerra Arévalo N, López De Vargas Machuca KG, Sánchez-Nivicela JC, Armijos-Ojeda D, Cáceres Andrade JF, Reyes-Puig C, Quezada Riera AB, Székely P, Rojas Soto OR, Székely D, Guayasamin JM, Siavichay Pesántez FR, Amador L, Betancourt R, Ramírez-Jaramillo SM, Timbe-Borja B, Gómez Laporta M, Webster Bernal JF, Oyagata Cachimuel LA, Chávez Jácome D, Posse V, Valle-Piñuela C, Padilla Jiménez D, Reyes-Puig JP, Terán-Valdez A, Coloma LA, Pérez Lara MB, Carvajal-Endara S, Urgilés M, Yánez Muñoz MH. Red List assessment of amphibian species of Ecuador: a multidimensional approach for their conservation. PLOS ONE. 2021;16(5):e0251027. doi: 10.1371/journal.pone.0251027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ortega-Andrade et al. (2015).Ortega-Andrade HM, Rojas-Soto OR, Valencia JH, de los Monteros AE, Morrone JJ, Ron SR, Cannatella DC. Insights from integrative systematics reveal cryptic diversity in Pristimantis frogs (Anura: Craugastoridae) from the upper Amazon basin. PLOS ONE. 2015;10:e0143392. doi: 10.1371/journal.pone.0143392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padial et al. (2012).Padial JM, Chaparro JC, Castroviejo-Fisher S, Guayasamin JM, Lehr E, Delgado AJ, Vaira M, Teixeira Jr M, Aguayo R, De La Riva I. A revision of species diversity in the Neotropical genus Oreobates (Anura: Strabomantidae), with the description of three new species from the Amazonian slopes of the Andes. American Museum Novitates. 2012;3752:1–55. doi: 10.1206/3752.2. [DOI] [Google Scholar]
- Padial, Grant & Frost (2014).Padial JM, Grant T, Frost DR. Molecular systematics of terraranas (Anura: Brachycephaloidea) with an assessment of the effects of alignment and optimality criteria. Zootaxa. 2014;3825:1–132. doi: 10.11646/zootaxa.3825.1.1. [DOI] [PubMed] [Google Scholar]
- Padial et al. (2010).Padial JM, Miralles A, De la Riva I, Vences M. Review: the integrative future of taxonomy. Frontiers in Zoology. 2010;7:1–14. doi: 10.1186/1742-9994-7-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padial & De la Riva (2009).Padial JM, De la Riva I. Integrative taxonomy reveals cryptic Amazonian species of Pristimantis (Anura: Strabomantidae) Zoological Journal of the Linnean Society. 2009;155:97–122. doi: 10.1111/j.1096-3642.2008.00424.x. [DOI] [Google Scholar]
- Padial & De la Riva (2010).Padial JM, De la Riva I. A response to recent proposals for integrative taxonomy. Biological Journal of the Linnean Society. 2010;101(3):747–756. doi: 10.1111/j.1095-8312.2010.01528.x. [DOI] [Google Scholar]
- Páez & Ron (2019).Páez NB, Ron SR. Systematics of Huicundomantis, a new subgenus of Pristimantis (Anura, Strabomantidae) with extraordinary cryptic diversity and eleven new species. ZooKeys. 2019;868:1–112. doi: 10.3897/zookeys.868.26766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palumbi et al. (1991).Palumbi SR, Martin A, Romano S, McMillan WO, Stice L, Grabowski G. Honolulu: University of Hawaii; 1991. p. 94 pp. [Google Scholar]
- Peñafiel et al. (2019).Peñafiel N, Flores DM, Rivero De Aguilar J, Guayasamin JM, Bonaccorso E. A cost-effective protocol for total DNA isolation from animal tissue. Neotropical Biodiversity. 2019;5(1):69–74. doi: 10.1080/23766808.2019.1706387. [DOI] [Google Scholar]
- Peracca (1904).Peracca MG. Viaggio del Dr. Enrico Festa nell’Ecuador e regioni vicine. Bollettino dei Musei di Zoologia ed Anatomia comparata, Università di Torino. 1904;19:1–41. doi: 10.5962/bhl.part.11596. [DOI] [Google Scholar]
- Peterson, Soberón & Sánchez-Cordero (1999).Peterson AT, Soberón J, Sánchez-Cordero V. Conservatism of ecological niches in evolutionary time. Science. 1999;285:1265–1267. doi: 10.1126/science.285.5431.1265. [DOI] [PubMed] [Google Scholar]
- Pinto-Sánchez et al. (2012).Pinto-Sánchez NR, Ibañez R, Madriñan S, Sanjur OI, Bermingham E, Crawford AJ. The great American biotic interchange in frogs: multiple and early colonization of Central America by the South American genus Pristimantis (Anura: Craugastoridae) Molecular Phylogenetics and Evolution. 2012;62:954–972. doi: 10.1016/j.ympev.2011.11.022. [DOI] [PubMed] [Google Scholar]
- Pyron & Wiens (2011).Pyron RA, Wiens JJ. A large-scale phylogeny of Amphibia including over 2800 species, and a revised classification of extant frogs, salamanders, and caecilians. Molecular Phylogenetics and Evolution. 2011;61:543–583. doi: 10.1016/j.ympev.2011.06.012. [DOI] [PubMed] [Google Scholar]
- Queiroz (2007).Queiroz KDe. Species concepts and species delimitation. Systematic Biology. 2007;56:879–886. doi: 10.1080/10635150701701083. [DOI] [PubMed] [Google Scholar]
- Rambaut & Drummond (2009).Rambaut A, Drummond AJ. Tracer version 1.5.0. www document. http://tree.bio.ed.ac.uk/software/tracer/ [17 October 2022];2009
- Remsem (1984).Remsem JV. High incidence of leapfrog pattern of geographic variation in Andean birds: implications for the speciation process. Science. 1984;224(4645):171–173. doi: 10.1126/science.224.4645.171. [DOI] [PubMed] [Google Scholar]
- Restrepo, Velasco & Daza (2017).Restrepo A, Velasco J, Daza JM. Extinction risk or lack of sampling in a threatened species: Genetic structure and environmental suitability of the Neotropical frog Pristimantis penelopus (Anura: Craugastoridae) Papeis Avulsos de Zoologia. 2017;57(1):1–15. doi: 10.11606/0031-1049.2017.57.01. [DOI] [Google Scholar]
- Rivera-Correa & Daza (2016).Rivera-Correa M, Daza J. Molecular phylogenetics of the Pristimantis lacrimosus species group (Anura: Craugastoridae) with the description of a new species from Colombia. Acta Herpetologica. 2016;11(1):31–45. [Google Scholar]
- Rivera-Correa et al. (2022).Rivera-Correa M, Correa-Medina H, Venegas-Valencia K, Daza JM. Genetic diversity, acoustic signal and geographic distribution of a colourful rain frog of the genus Pristimantis (Anura: Craugastoridae): Genetic diversity of the Pristimantis jaguensis. Herpetology Notes. 2022;15:215–227. [Google Scholar]
- Rivera-Correa, Jimenez-Rivillas & Daza (2017).Rivera-Correa M, Jimenez-Rivillas C, Daza JM. Phylogenetic analysis of the Neotropical Pristimantis leptolophus species group (Anura: Craugastoridae): molecular approach and description of a new polymorphic species. Zootaxa. 2017;4242(2):313–343. doi: 10.11646/zootaxa.4242.2.6. [DOI] [PubMed] [Google Scholar]
- Rödder & Schmitz (2009).Rödder D, Schmitz A. Two new Pristimantis (Anura, Strabomantidae) belonging to the myersi group from the Andean slopes of Ecuador. Revue Suisse de Zoologie. 2009;116(2):275–288. doi: 10.5962/bhl.part.79496. [DOI] [Google Scholar]
- Rojas et al. (2019d).Rojas RMA, Castro F, Guerrero JA, Lynch J, Herrera M, Gutiérrez-Cárdenas P. Pristimantis repens. The IUCN Red List of Threatened Species 2019: e.T56905A3052643. Downloaded on 08 November 2021. 2019d. [DOI]
- Rojas et al. (2018).Rojas RR, Fouquet A, Ron SR, Hernández-Ruz EJ, Melo-Sampaio PR, Chaparro JC, Vogt RC, De Carvalho VT, Cardoso-Pinheiro L, Avila RW, Pires-Farias I, Gordo M, Hrbek T. A Pan-Amazonian species delimitation: high species diversity within the genus Amazophrynella (Anura: Bufonidae) PeerJ. 2018;6:e4941. doi: 10.7717/peerj.4941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rojas-Runjaic (2012).Rojas-Runjaic FJM. Master’s Thesis. 2012. Species limits in direct-developing frogs of the Pristimantis myersi species group (Terrarana: Strabomantidae) pp. 1–48. [Google Scholar]
- Rojas-Runjaic, Delgado & Guayasamin (2014).Rojas-Runjaic FJM, Delgado AJ, Guayasamin JM. A new rainfrog of the Pristimantis myersi Group (Amphibia, Craugastoridae) from Volcán Pichincha, Ecuador. Zootaxa. 2014;3780(1):36–50. doi: 10.11646/zootaxa.3780.1.2. [DOI] [PubMed] [Google Scholar]
- Rojas-Runjaic & Guayasamin (2015).Rojas-Runjaic FJM, Guayasamin JM. First record of the rain frog Pristimantis myersi (Goin & Cochran, 1963) (Anura, Craugastoridae) for Ecuador. Check List. 2015;11(1):1542. doi: 10.15560/11.1.1542. [DOI] [Google Scholar]
- Ron, Guayasamin & Menéndez-Guerrero (2011).Ron SR, Guayasamin JM, Menéndez-Guerrero P. Biodiversity and conservation status of Ecuadorian amphibians. Amphibian Biology. 2011;9(Part 2):129–170. [Google Scholar]
- Ronquist et al. (2012).Ronquist F, Teslenko M, Van Der Mark P, Ayres DL, Darling A, Höhna S, Huelsenbeck JP. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Systematic Biology. 2012;61(3):539–542. doi: 10.1093/sysbio/sys029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sánchez-Nivicela et al. (2021).Sánchez-Nivicela JC, Urgiles VL, Cedeño Palacios J, AbadPeñafiel H, Guayasamin JM. Una fantástica nueva especie del grupo Pristimantis orcesi de los Andes sur De Ecuador. Neotropical Biodiversity. 2021;6(1):224–237. doi: 10.1080/23766808.2020.1869449. [DOI] [Google Scholar]
- Shimodaira (2002).Shimodaira H. An approximately unbiased test of phylogenetic tree selection. Systematic Biology. 2002;51(3):492–508. doi: 10.1080/10635150290069913. [DOI] [PubMed] [Google Scholar]
- Shimodaira & Hasegawa (1999).Shimodaira H, Hasegawa M. Multiple comparisons of log-likelihoods with applications to phylogenetic inference. Molecular Biology and Evolution. 1999;16(8):1114–1116. doi: 10.1093/oxfordjournals.molbev.a026201. [DOI] [Google Scholar]
- Simpson (1961).Simpson GG. Principles of animal taxonomy. Columbia University Press; New York: 1961. p. 247 pp. [DOI] [PubMed] [Google Scholar]
- Strimmer & Rambaut (2002).Strimmer K, Rambaut A. Inferring confidence sets of possibly misspecified gene trees. Proceedings of the Royal Society of London. Biological Sciences. 2002;269(1487):137–142. doi: 10.1098/rspb.2001.1862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taboada et al. (2013).Taboada C, Grant T, Lynch JD, Faivovich J. New morphological synapomorphies for the New World direct-developing frogs (Amphibia: Anura: Terrarana) Herpetologica. 2013;69(3):342–357. doi: 10.1655/HERPETOLOGICA-D-13-00019. [DOI] [Google Scholar]
- Targino (2016).Targino M. Doctoral Thesis. 2016. Phylogenetic analysis of Pristimantis Jiménez de la Espada, 1870, a megadiverse genus of amphibians (Anura, Brachycephaloidea, Craugastoridae) [DOI] [Google Scholar]
- Urgiles et al. (2019).Urgiles V, Székely P, Székely D, Christodoulides N, Sanchez-Nivicela J, Savage A. Genetic delimitation of Pristimantis orestes (Lynch, 1979) and P. saturninoi Brito others,2017 and description of two new terrestrial frogs from the Pristimantis orestes species group (Anura, Strabomantidae) ZooKeys. 2019;864:111–146. doi: 10.3897/zookeys.864.35102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valencia et al. (2019).Valencia J, Valladares-Suntasig F, Tipantiza-Tuguminago L, Dueñas M. A new species of terrestrial-breeding frog of the genus Pristimantis (Anura: Terrarana: Craugastoridae) from the eastern Andean slopes of the southern Ecuador. Zootaxa. 2019;4658:509–525. doi: 10.11646/zootaxa.4658.3. [DOI] [PubMed] [Google Scholar]
- Vences et al. (2005).Vences M, Thomas M, Van der Meijden A, Chiari Y, Vieites DR. Comparative performance of the 16S rRNA gene in DNA barcoding of amphibians. Frontiers in Zoology. 2005;2:1–12. doi: 10.1186/1742-9994-2-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vieites et al. (2009).Vieites DR, Wollenberg KC, Andreone F, Köhler J, Glaw F, Vences M. Vast underestimation of Madagascar’s biodiversity evidenced by an integrative amphibian inventory. Proceedings of the National Academy of Sciences of the United States of America. 2009;106(20):8267–8272. doi: 10.1073/pnas.0810821106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waddell et al. (2018).Waddell EH, Crotti M, Lougheed SC, Cannatella DC, Elmer KR. Hierarchies of evolutionary radiation in the world’s most species rich vertebrate group, the Neotropical Pristimantis leaf litter frogs. Systematics and Biodiversity. 2018;16(8):807–819. doi: 10.1080/14772000.2018.1503202. [DOI] [Google Scholar]
- Wiens (2004).Wiens JJ. Speciation and ecology revisited: phylogenetic niche conservatism and the origin of species. Evolution. 2004;58(1):193–197. doi: 10.1111/j.0014-3820.2004.tb01586.x. [DOI] [PubMed] [Google Scholar]
- Wiens & Graham (2005).Wiens JJ, Graham CH. Niche conservatism: integrating evolution, ecology, and conservation biology. Annual Review of Ecology, Evolution, and Systematics. 2005;36:519–539. doi: 10.1146/annurev.ecolsys.36.102803.095431. [DOI] [Google Scholar]
- Wiley (1978).Wiley EO. The evolutionary species concept reconsidered. Systematic Biology. 1978;27(1):17–26. doi: 10.2307/2412809. [DOI] [Google Scholar]
- Wollenberg et al. (2008).Wollenberg KC, Vieites DR, Van Der Meijden A, Glaw F, Cannatella DC, Vences M. Patterns of endemism and species richness in Malagasy cophyline frogs support a key role of mountainous areas for speciation. Evolution. 2008;62(8):1890–1907. doi: 10.1111/j.1558-5646.2008.00420.x. [DOI] [PubMed] [Google Scholar]
- Yánez-Muñoz et al. (2010).Yánez-Muñoz MH, Meza-Ramos P, Cisneros-Heredia DF, Reyes-Puig JP. Descripción de tres nuevas especies de ranas del género Pristimantis (Anura: Terrarana: Strabomantidae) de los bosques nublados del Distrito Metropolitano De Quito, Ecuador. Avances en Ciencias e Ingenierías. 2010;2:16–27. doi: 10.18272/aci.v2i3.40. [DOI] [Google Scholar]
- Yánez-Muñoz et al. (2018).Yánez-Muñoz MH, Reyes-Puig C, Reyes-Puig JP, Velasco JA, Ayala-Varela F, Torres-Carvajal O. A new cryptic species of Anolis lizard from northwestern South America (Iguanidae, Dactyloinae) ZooKeys. 2018;794:135–163. doi: 10.3897/zookeys.794.26936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zink & Barrowclough (2008).Zink RM, Barrowclough GF. Mitochondrial DNA under siege in avian phylogeography. Molecular Ecology. 2008;17(9):2107–2121. doi: 10.1111/j.1365-294X.2008.03737.x. [DOI] [PubMed] [Google Scholar]
- Zumel, Buckley & Ron (2021).Zumel D, Buckley D, Ron SR. The Pristimantis trachyblepharis species group, a clade of miniaturized frogs: description of four new species and insights into the evolution of body size in the genus. Zoological Journal of the Linnean Society. 2021;195(1):315–354. doi: 10.1093/zoolinnean/zlab044. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Species names, group/clade, GenBank accession numbers, voucher numbers, coordinates and localities of Pristimantis myersi species group, P. verecundus clade, P. devillei, P. boulengeri and P. leptolophus species groups.
Node support is expressed in Bootstrap values; missing values indicate support below 70 (bootstrap). Each terminal includes the following information: GenBank code and species name. Highlighted branches indicated new sequences and excluded species of the Pristimantis myersi species group.
Node support is expressed in Bootstrap values; missing values indicate support below 70 (bootstrap). Each terminal includes the following information: GenBank code, species name and voucher number. Highlighted branches indicated sequences of Pristimantis myersi group + close relatives clade + excluded species.
Values are presented as percent distances calculated from uncorrected p values.
Data Availability Statement
The following information was supplied regarding data availability:
Raw data, including Pristimantis myersi group genetic sequences, are available in the Supplemental Files.