Abstract
Patient: Male, 68-year-old
Final Diagnosis: CML
Symptoms: Leukocytosis • splenomegaly
Clinical Procedure: Bone marrow biopsy
Specialty: Hematology
Objective:
Rare disease
Background:
Breakpoint cluster region (BCR)-Abelson murine leukemia (ABL1) and Janus Kinase-2 (JAK2) mutations have been thought to be mutually exclusive in myeloproliferative neoplasms (MNPs), but recent data suggest that they can occur together.
Case report:
A 68-year-old man was referred to the hematology clinic because of an elevated white blood cell count. His medical history included type II diabetes mellitus, hypertension, and retinal hemorrhage. Fluorescence in situ hybridization analysis of the bone marrow was positive for BCR-ABL1 in 66/100 cells. Conventional cytogenetics was positive for the Philadelphia chromosome in 16/20 counted cells. The percentage of BCR-ABL1 was 12%. Considering the patient’s age and medical comorbidities, he was started on imatinib 400 mg once daily. Further tests showed JAK2 V617F mutation positivity and absence of acquired von Willebrand disease. He was then started on aspirin 81 mg and hydroxyurea 500 mg once daily, which was later increased to 1000 mg daily. The patient achieved a major molecular response after 6 months of treatment, with undetectable BCR-ABL1 levels.
Conclusions:
BCR-ABL1 and JAK2 mutations can co-existence in MNPs. Physicians must suspect the presence of one of the MPNs in chronic myeloid leukemia (CML) patients with persistent or increased thrombocytosis, an atypical course of the disease, or hematological abnormalities despite evidence of response or remission of CML. Therefore, testing for JAK2 should be performed accordingly. Combining cytoreductive therapy with tyrosine kinase inhibitors (TKIs) is a therapeutic option when both mutations are present, and TKI alone is not sufficient to control peripheral blood cell counts.
Keywords: Janus Kinase 2; EXOSC5 Protein, Human; Thrombocytosis
Background
Chronic myeloid leukemia (CML) is a clonal hematopoietic stem cell disorder that causes bone marrow hyperplasia. It is a myeloproliferative neoplasm (MPN) with an estimated incidence of 1.9 cases per 100,000 adults and a death rate of 0.3 cases per 100,000 adults recorded in 2019 in the United States [1]. It is characterized by the presence of fusion between the Abelson murine leukemia (ABL1) gene on chromosome 9 and the breakpoint cluster region (BCR) gene on chromosome 22, leading to the expression of the oncoprotein BCR-ABL1. The derived chromosome is termed Philadelphia chromosome. BCR-ABL1 is a constitutively active tyrosine kinase that promotes growth and replication through downstream signaling pathways involving RAF, RAS, JAN kinase, STAT, and MYC [2]. Other MPNs include polycythemia vera (PV), essential thrombocythemia (ET), and primary myelofibrosis (PMF), which are collectively known as Philadelphia-negative MPNs. Diagnostic criteria for all MPNs have been developed by the World Health Organization [3]. The presence of recurrent genetic mutations is central to diagnosis. JAK2 mutation is the most common mutation detected in Philadelphia-negative MPNs, which also causes cell proliferation through the JAK–RAS–STAT pathway [4]. JAK2 mutations are present in up to 95% of PV cases and 65% of ET and PMF cases [5]. BCR-ABL1 and JAK2 mutations are thought to be mutually exclusive, but recent data and case reports have described cases in which these mutations occur concomitantly [6–13]. In this report, we describe a unique coincidence presentation of CML and JAK2-positive thrombocytosis one month after initiating tyrosine kinase inhibitor (TKI) therapy.
Case Report
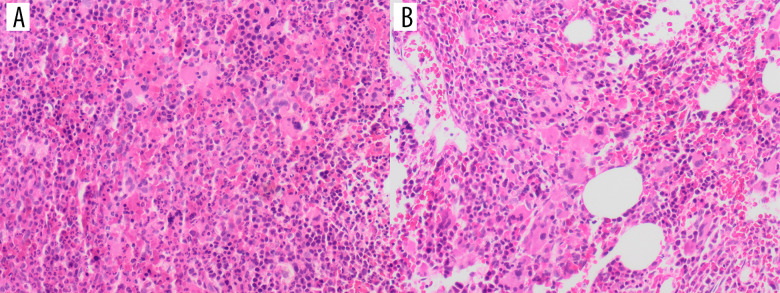
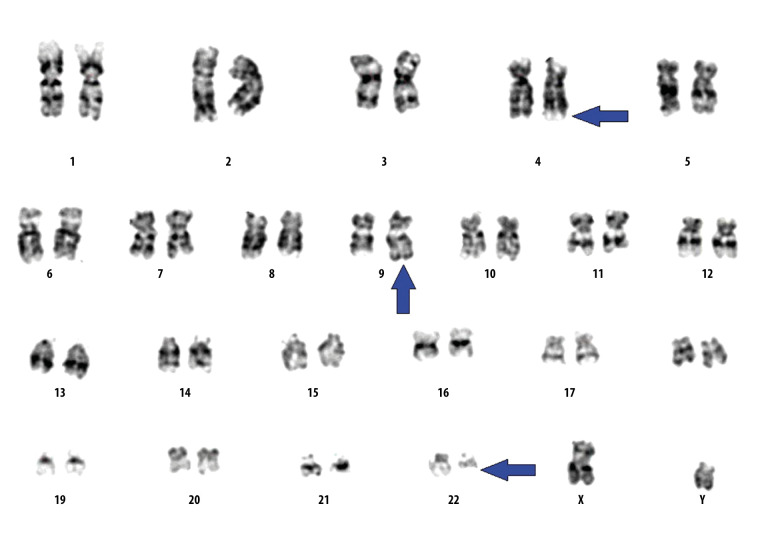
A 68-year-old man was referred to the hematology clinic because of an elevated white blood cell (WBC) count. He was diagnosed with type II diabetes mellitus and hypertension, after which he developed retinal hemorrhage. Prior to retinal laser photocoagulation, he was found to have a high white blood cell count. He was started empirically on hydroxyurea (500 mg once daily) and was referred to the clinic for further evaluation. Physical examination revealed splenomegaly (spleen size: 13 cm). His initial complete blood count (CBC) revealed the following: hemoglobin (Hb), 14.1 g/dL; WBCs, 34.6×109/L; platelets, 539×109/L; basophils, 0.34×109/L; eosinophils, 0.85×109/L; and neutrophils, 27.47×109/L. Bone marrow biopsy showed hypercellular marrow (cellularity estimated at 100%) with increased tri-lineage hematopoiesis, prominent megakaryopoiesis, and increased reticulin staining. The percentage of blasts was <1% as shown in Figure 1. Fluorescence in situ hybridization (FISH) analysis of the bone marrow was positive for BCRABL1 in 66/100 cells, with Table 1 showing only 1 fusion signal, 1 signal for bcr, and 2 signals for abl, which indicates BCR/ ABL rearrangement, representing a Philadelphia translocation with loss of 1 BCR copy and Figure 2 showing 2 Green (BCR), 3 Red (ABL) and 1 Fusion (BCR/ABL). Conventional cytogenetics was positive for the Philadelphia chromosome in 16/20 counted cells, showing 3-way translocation 46,XY,t(4;9;22) (q31;q34;q11.2)[17]/46,XY [4] as described in Figure 3. Real-time quantitative polymerase chain reaction (RT-qPCR) showed that the percentage of BCR-ABL1 was 12%. Considering the patient’s age and medical comorbidities, he was thus started on imatinib 400 mg once daily.
Figure 1.
(A) Bone marrow biopsy showed hypercellularity ~100% (H&E stain, x50) with myeloid hyperplasia. Megakaryocytes are seen with variable sizes, and morphology includes dwarf forms and multiple erythroid islands. (B) Megakaryocytes are increased and show small loose clusters.
Table 1.
Signals per probe of an analysis of 100 interphase fluorescence in situ hybridization (FISH) nuclei with probe for BCR/ABL.
| BCR | ABL | Fusion signals | Number of cells (%) |
|---|---|---|---|
| 2 | 2 | 0 | 34 (34) |
| 2 | 3 | 1 | 66 (66) |
Figure 2.
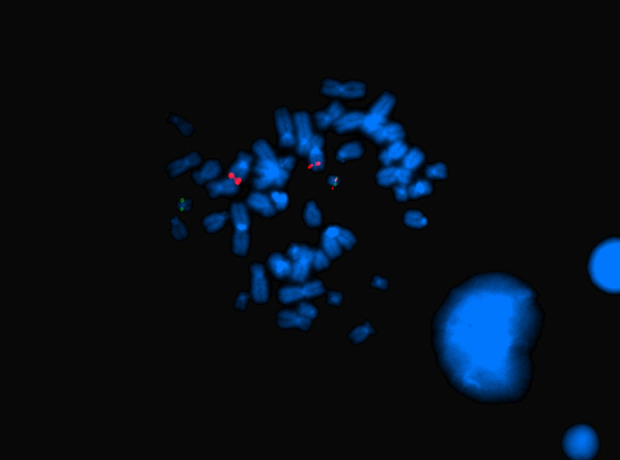
FISH analysis of a metaphase cell with LSI BCR/ABL revealed the following signal distribution: 2 signals for ABL on both chromosomes 9 (red), 1 signal for BCR on 1 chromosome 22 (green), and 1 fusion signal on the derivative chromosome 22.
Figure 3.
Chromosomal analysis revealed the presence of variant Philadelphia chromosome as a result of 3-way translocation between the long arms of chromosome 4,9 and 22 at bands 4q31,9q34, and 22q11.2 (arrows).
After 1 month of therapy, the patient’s WBC count normalized and BCR-ABL1 percentage by RT-qPCR decreased to 9.5%, but his platelet count increased to 1543×109/L. Repeat bone marrow biopsy was negative for accelerated phase/blast phase CML. Further tests showed that JAK2 V617F mutation was positive, and the genomic DNA from the material obtained was screened with the Mutascreen Kit from IPSOGEN for presence of the mutation c. 1849G<T (p.V617F). Exon 14 of the Janus Kinase 2 gene (JAK2) was analyzed using real-time PCR. The measured results were compared with a reference sample, but the allele burden was not assessed. Tests for acquired von Willebrand disease yielded negative results. He was then started on aspirin 81 mg and hydroxyurea 500 mg once daily, which was later increased to 1000 mg daily. Throughout his treatment, the patient achieved all the CML treatment milestones, and he achieved a major molecular response after 6 months of treatment. His platelet count decreased to 400–500×109/L, but his spleen remained enlarged at 13 cm. During follow-up, the patient remained in major molecular response with undetectable levels of BCR-ABL1.
Discussion
The JAK2 V617F and BCR-ABL1 oncogenes are thought to be mutually exclusive; however, rare cases have been reported in which the occurrence of one may precede the occurrence of the other. Several attempts have been made to identify the prevalence of concomitant BCR-ABL1 and JAK2 mutations in patients with MPNs. Pieri et al tested 314 patients with BCRABL1-positive CML and found that 8 of them (2.5%) had the JAK2 V617F mutation [14]. Martin-Cabrera et al reported that among patients diagnosed with a myeloproliferative neoplasm by the presence of either JAK2 V617F, BCR-ABL1, or MPL mutations, 0.2% were found to be positive for both JAK2 V617F and BCR-ABL1 [15]. More recently, Soderquist et al reported a 0.4% prevalence of concurrent JAK2 V617F and BCR-ABL1 mutations when concomitant testing was performed on all patients suspected to have underlying MPN [10].
In our literature review of case reports, the diagnosis varied among CML preceding an MPN, an MPN preceding CML, or the co-existence of both disorders simultaneously [10,12]. The majority of the reviewed cases were initially diagnosed with MPN, ET, PV, or PMF, and had a JAK2 V617F mutation and later developed CML. The time from diagnosis of CML to MPN varied, ranging from 2 to 18 years after the initial diagnosis. The average duration from the diagnosis of MPN to CML later is longer than that from the diagnosis of CML to MPN later (7 vs 2 years) [12,16,17]. Although JAK2 and BCR-ABL were considered mutually exclusive drivers of mutation, it was repeatedly reported in the literature that they can co-exist. A case series by Soderquist et al indicated that both mutations can present in various manners [18–20].
Several theories attempt to explain the occurrence of both mutations simultaneously. One theory suggests the existence of 2 separate clones, in which one clone is suppressed by the proliferation of the other clone. Another theory suggests that there is a single clone that includes a relatively smaller sub-clone with both mutations. Other theories point to the possibility of a single clone harboring both mutations [21–25]. Ping et al proposed that the JAK clone will usually achieve dominance once the BCR-ABL clone is suppressed, although their patient had a recent diagnosis of PV [26].
In our case, thrombocytosis became more pronounced after starting TKI (imatinib), with a decrease in BCR-ABL1 RT-qPCR levels. It is likely that these 2 mutations exist in different clones, and once the CML clone is suppressed, the JAK2 V617F clone becomes more active. The patient responded well to TKI and cytoreductive (hydroxyurea) therapy. The other options would include combining TKI and JAK inhibitors in such cases.
The limitations in our case report include not measuring the JAK2 V617F allele burden and inability to perform more complex testing for clonality, as we believe it is important to differentiate between clonal evolution vs selection.
Conclusions
Although BCR-ABL1 and JAK2 mutations are thought to be mutually exclusive, cases with co-existence of both mutations and acquiring new mutations have been reported. Physicians must suspect the presence of one of the MPNs in CML patients with persistent or increased thrombocytosis, an atypical course of the disease, or hematological abnormalities despite evidence of response or remission of CML. Therefore, testing for JAK2 should be performed. Combining cytoreductive therapy with TKI is a therapeutic option when both mutations are present, and TKI alone is not sufficient to control peripheral blood cell counts.
Acknowledgments
We thank Ms. Heba Hashem, Department of Pathology and Laboratory Medicine, Ministry of the National Guard – Health Affairs, Jeddah, Saudi Arabia for her kind help and review in this case. Also, we appreciate the help of Dr. Abdullah Bukhari, PGY5 hematopathology, for his valuable input in reviewing the slides of this patient.
Footnotes
Publisher’s note: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher
Declaration of Figures’ Authenticity
All figures submitted have been created by the authors who confirm that the images are original with no duplication and have not been previously published in whole or in part.
References:
- 1.Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127(20):2391–405. doi: 10.1182/blood-2016-03-643544. [DOI] [PubMed] [Google Scholar]
- 2.National Cancer Institute https//seer.cancer.gov/statfacts/html/cmyl.html, data accessed 09/03/2020.
- 3.Cilloni D, Saglio G. Molecular pathways: BCR-ABL. Clin Cancer Res. 2012;18(4):930–37. doi: 10.1158/1078-0432.CCR-10-1613. [DOI] [PubMed] [Google Scholar]
- 4.Baxter EJ, Scott LM, Campbell PJ, et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet. 2005;365(9464):1054–61. doi: 10.1016/S0140-6736(05)71142-9. [published correction appears in Lancet. 2005;366(9480):122] [DOI] [PubMed] [Google Scholar]
- 5.Cazzola M, Kralovics R. From Janus kinase 2 to calreticulin: The clinically relevant genomic landscape of myeloproliferative neoplasms. Blood. 2014;123(24):3714–19. doi: 10.1182/blood-2014-03-530865. [DOI] [PubMed] [Google Scholar]
- 6.Pahore ZA, Shamsi TS, Taj M, et al. JAK2V617F mutation in chronic myeloid leukemia predicts early disease progression. J Coll Physicians Surg Pak. 2011;21(8):472–75. [PubMed] [Google Scholar]
- 7.Tabassum N, Saboor M, Ghani R, Moinuddin M. Frequency of JAK2 V617F mutation in patients with Philadelphia positive Chronic Myeloid Leukemia in Pakistan. Pak J Med Sci. 2014;30(1):185–88. doi: 10.12669/pjms.301.3906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hassan A, Dogara LG, Babadoko AA, et al. Coexistence of JAK2 and BCRABL mutation in patient with myeloproliferative neoplasm. Niger Med J. 2015;56(1):74–76. doi: 10.4103/0300-1652.149177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhou A, Knoche EM, Engle EK, et al. Concomitant JAK2 V617F-positive polycythemia vera and BCR-ABL-positive chronic myelogenous leukemia treated with ruxolitinib and dasatinib. Blood Cancer J. 2015;5(10):e351. doi: 10.1038/bcj.2015.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Soderquist CR, Ewalt MD, Czuchlewski DR, et al. Myeloproliferative neoplasms with concurrent BCR-ABL1 translocation and JAK2 V617F mutation: A multi-institutional study from the bone marrow pathology group. Mod Pathol. 2018;31(5):690–704. doi: 10.1038/modpathol.2017.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mousinho F, Azevedo AP, Mendes T, et al. Concomitant presence of JAK2V617F mutation and BCR-ABL translocation in two patients: A new entity or a variant of myeloproliferative neoplasms (Case report) Mol Med Rep. 2018;18(1):1001–6. doi: 10.3892/mmr.2018.9032. [DOI] [PubMed] [Google Scholar]
- 12.Bader G, Dreiling B. Concurrent JAK2-positive myeloproliferative disorder and chronic myelogenous leukemia: A novel entity? A case report with review of the literature. J Investig Med High Impact Case Rep. 2019;7:2324709619832322. doi: 10.1177/2324709619832322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sağlam B, Albayrak M, Yıldız A, et al. A rare entity in chronic myelocytic leukemia: Coexistence of BCR ABL1 translocation and JAK2 V617F mutation. Case report. SN Compr Clin Med. 2020;2:478–80. [Google Scholar]
- 14.Pieri L, Spolverini A, Scappini B, et al. Concomitant occurrence of BCR-ABL and JAK2V617F mutation. Blood. 2011;118(12):3445–46. doi: 10.1182/blood-2011-07-365007. [DOI] [PubMed] [Google Scholar]
- 15.Martin-Cabrera P, Haferlach C, Kern W, et al. BCR-ABL1-positive and JAK2 V617F-positive clones in 23 patients with both aberrations reveal biologic and clinical importance. Br J Haematol. 2017;176(1):135–39. doi: 10.1111/bjh.13932. [DOI] [PubMed] [Google Scholar]
- 16.Pingali SR, Mathiason MA, Lovrich SD, Go RS. Emergence of chronic myelogenous leukemia from a background of myeloproliferative disorder: JAK2V617F as a potential risk factor for BCR-ABL translocation. Clin Lymphoma Myeloma. 2009;9(5):E25–29. doi: 10.3816/CLM.2009.n.080. [DOI] [PubMed] [Google Scholar]
- 17.Zhou A, Knoche EM, Engle EK, et al. Concomitant JAK2 V617F-positive polycythemia vera and BCR-ABL-positive chronic myelogenous leukemia treated with ruxolitinib and dasatinib. Blood Cancer J. 2015;5(10):e351. doi: 10.1038/bcj.2015.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lorenzo M, Grille S, Stevenazzi M. Emergence of BCR-ABL1 chronic myeloid leukemia in a JAK2-V617F polycythemia vera. J Hematol. 2020;9(1–2):23–29. doi: 10.14740/jh591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Soderquist CR, Ewalt MD, Czuchlewski DR, et al. Myeloproliferative neoplasms with concurrent BCR-ABL1 translocation and JAK2 V617F mutation: A multi-institutional study from the bone marrow pathology group. Mod Pathol. 2018;31(5):690–704. doi: 10.1038/modpathol.2017.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Park SH, Chi HS, Cho YU, et al. Two cases of myeloproliferative neoplasm with a concurrent JAK2 (V617F) mutation and BCR/ABL translocation without chronic myelogenous leukemia phenotype acquisition during hydroxyurea treatment. Ann Lab Med. 2013;33(3):229–32. doi: 10.3343/alm.2013.33.3.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Laibe S, Tadrist Z, Arnoulet C, Sainty D, Mozziconacci MJ. A myeloproliferative disorder may hide another one. Leuk Res. 2009;33(8):1133–36. doi: 10.1016/j.leukres.2009.01.034. [DOI] [PubMed] [Google Scholar]
- 22.Hassankrishnamurthy S, Mody MD, Kota VK. A case of chronic myelogenous leukemia occurring in a patient treated for essential thrombocythemia. Am J Case Rep. 2019;20:10–14. doi: 10.12659/AJCR.911854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bee PC, Gan GG, Nadarajan VS, et al. A man with concomitant polycythaemia vera and chronic myeloid leukemia: The dynamics of the two disorders. Int J Hematol. 2010;91(1):136–39. doi: 10.1007/s12185-009-0471-6. [DOI] [PubMed] [Google Scholar]
- 24.Wang X, Tripodi J, Kremyanskaya M, et al. BCR-ABL1 is a secondary event after JAK2V617F in patients with polycythemia vera who develop chronic myeloid leukemia. Blood. 2013;121(7):1238–39. doi: 10.1182/blood-2012-11-467787. [DOI] [PubMed] [Google Scholar]
- 25.Krämer A, Reiter A, Kruth J, et al. JAK2-V617F mutation in a patient with Philadelphia-chromosome-positive chronic myeloid leukaemia. Lancet Oncol. 2007;8(7):658–60. doi: 10.1016/S1470-2045(07)70206-1. [DOI] [PubMed] [Google Scholar]
- 26.Bee PC, Gan GG, Nadarajan VS, et al. A man with concomitant polycythaemia vera and chronic myeloid leukemia: The dynamics of the two disorders. Int J Hematol. 2010;91(1):136–39. doi: 10.1007/s12185-009-0471-6. [DOI] [PubMed] [Google Scholar]