Abstract
In the revised diagnostic classification systems ICD-11 and DSM-5, secondary, organic forms of obsessive-compulsive disorder (OCD) are implemented as specific nosological entities. Therefore, the aim of this study was to clarify whether a comprehensive screening approach, such as the Freiburg-Diagnostic-Protocol for patients with OCD (FDP-OCD), is beneficial for detecting organic OCD forms. The FDP-OCD includes advanced laboratory tests, an expanded magnetic resonance imaging (MRI) protocol, and electroencephalography (EEG) investigations as well as automated MRI and EEG analyses. Cerebrospinal fluid (CSF), [18F]fluorodeoxyglucose positron emission tomography, and genetic analysis were added for patients with suspected organic OCD. The diagnostic findings of the first 61 consecutive OCD inpatients (32 female and 29 male; mean age: 32.7 ± 12.05 years) analyzed using our protocol were investigated. A probable organic cause was assumed in five patients (8%), which included three patients with autoimmune OCD (one patient with neurolupus and two with specific novel neuronal antibodies in CSF) and two patients with newly diagnosed genetic syndromes (both with matching MRI alterations). In another five patients (8%), possible organic OCD was detected (three autoimmune cases and two genetic cases). Immunological serum abnormalities were identified in the entire patient group, particularly with high rates of decreased “neurovitamin” levels (suboptimal vitamin D in 75% and folic acid in 21%) and increased streptococcal (in 46%) and antinuclear antibodies (ANAs; in 36%). In summary, the FDP-OCD screening led to the detection of probable or possible organic OCD forms in 16% of the patients with mostly autoimmune forms of OCD. The frequent presence of systemic autoantibodies such as ANAs further support the possible influence of autoimmune processes in subgroups of patients with OCD. Further research is needed to identify the prevalence of organic OCD forms and its treatment options.
Subject terms: Diagnostic markers, Molecular neuroscience
Introduction
Obsessive-compulsive disorder (OCD) is a common and severely disabling mental disorder with a lifetime prevalence of 1 to 3.5% [1–3] and is associated with a significant loss of quality of life [4]. It is assumed that OCD has a multi-factorial pathogenesis with biological, psychological, and social causes, as well as additional external stressors like critical life events or infections [5–7]. Biologically, dysbalance of the cortico–striato–thalamo–cortical circuits, a dysregulation of serotonergic, glutamatergic, or dopaminergic neurotransmissions, as well as (epi-)genetic alterations, are assumed [8–11]. In contrast to these primary forms of OCD, the DSM-5 and the upcoming ICD-11 distinguish obsessive-compulsive symptoms (OCS) in the context of organic causes—i.e., “organic OCD” forms [12, 13] from classical OCD. Organic OCD forms are based on clear-cut organic findings to which OCS can be attributed, like neurological lesions or tumors [14, 15]. In recent years, autoimmune mechanisms have been increasingly discussed in the context of organic OCD [16]. The most prominent example is Pediatric Autoimmune Neuropsychiatric Disorder Associated with Streptococcal Infections (PANDAS) [17–19]. There is also increasing evidence for organic OCD independent of infection due to different neuronal antibodies or in the context of systemic autoimmune disorders such as lupus erythematosus [16, 20–22]. Non-inflammatory causes could include neurodegenerative diseases such as Huntington’s disease or genetic syndromes such as velocardiofacial syndrome (cf. [11]). To date, little is known about the prevalence of organic OCD [9].
Study rationale
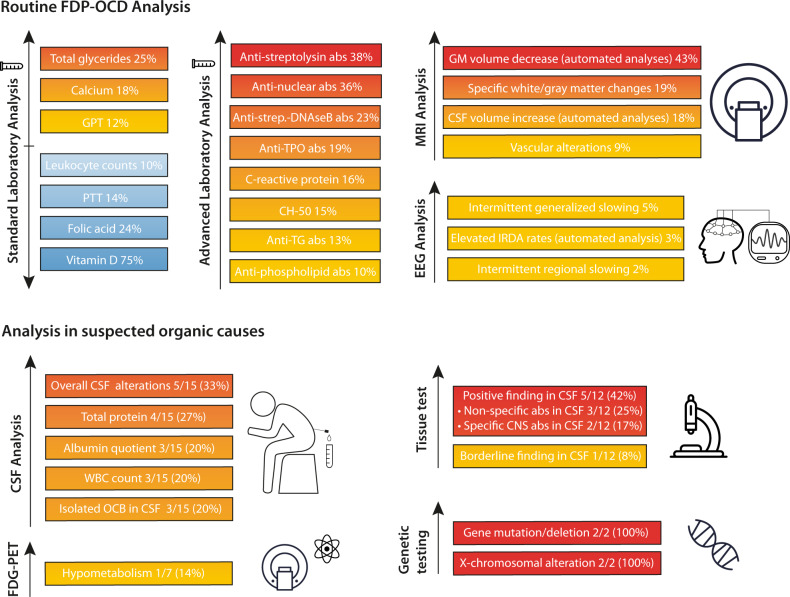
Organic OCD is gaining clinical importance and has also been included as a diagnosis in the DSM-5 and ICD-11 criteria. However, it is largely unclear how detailed the diagnostic work-up should be for the individual patient. At the Department of Psychiatry and Psychotherapy of the Medical Center, University of Freiburg, Germany, the Freiburg Diagnostic Protocol for patients with OCD (FDP-OCD) was developed (see Fig. 1). This article presents the array of diagnostic abnormalities and number of organic OCD cases when applying the FDP-OCD screening approach in the entire collective of patients with OCD.
Fig. 1. Freiburg diagnostic protocol for patients with OCD (FDP-OCD).
AMPA α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid, ANA antinuclear antibody, ANCA antineutrophil cytoplasmic antibody, AP alkaline phosphatase, C3/C4/C3d complement component 3/4/3d, CASPR2 Contactin-associated protein-like 2, CGI Clinical Global Impression, CK Creatine kinase, CH50 complement hemolytic activity, CSF Cerebrospinal fluid, cMRI cranial magnetic resonance imaging, CRP C-reactive protein, CRMP5 collapsing response-mediator protein-5, DNAse Deoxyribonuclease, DPPX dipeptidyl-peptidase–like protein 6, DTI Diffusion tensor imaging, DWI Diffusion-weighted imaging, ECG electrocardiogram, EEG Electroencephalogram, FDG-PET fluorodeoxyglucose - positron emission tomography, FLAIR Fluid-attenuated inversion recovery, γ-GT Gamma-glutamyl transferase, GABA Gamma-aminobutyric acid, GAD glutamic acid decarboxylase, GOT Glutamic-Oxaloacetic transaminase, GPT Glutamic-Pyruvic transaminase, HbA1c Hemoglobin A1c, HDL high-density lipoprotein, IgA Immunoglobulin A, IgG Immunoglobulin G, IgM Immunoglobulin M, INR International normalized ratio, LG1 leucine-rich glioma-inactivated 1, LDL low-density lipoprotein, MPRAGE Magnetization Prepared Rapid Acquisition Gradient Echo, NMDA N-methyl-D-aspartate, PCASL pseudo-continuous arterial spin labeling, PTT Partial thromboplastin time, R Receptor, SCID-1/2 Structured Clinical Interview for DSM-IV, T3 Triiodothyronine, T4 Thyroxine, TG Thyroglobulin, TPO Thyroid peroxidase, TRAKs Thyroid-stimulating hormone-receptor-antibodies, TSH Thyroid-stimulating hormone, Y-BOCS Yale-Brown Obsessive Compulsive Scale, Zic4 zinc-finger protein 4.
Patients and methods
The FDP-OCD screening approach has been developed over the past years based on the Freiburg Diagnostic Protocol in Psychosis (FDPP) for patients with psychosis [23] and is presented here for the first time together with first results. The retrospective analysis was performed according to the Declaration of Helsinki and after ethical approvement from the Ethics Committee of the University of Freiburg (No. 21 1290).
Patient group
The first 61 consecutive inpatients (32 females and 29 males with a mean age of 32.7 ± 12.05 years) treated in 2020 and 2021 on our ward specialized for OCD psychotherapy (https://www.uniklinik-freiburg.de/psych/stationen/station-6.html), who agreed to the offered diagnostic work-up, were included in the present study. Patients with the following admission codes according to the criteria of the ICD-10 [24] were analyzed: F42.0 (predominantly obsessional thoughts or ruminations), F42.1 (predominantly compulsive acts), and F42.2 (mixed obsessional thoughts and acts). All diagnoses were assigned based on patient history and clinical examination by experienced senior psychiatrists. Patients under 18, patients with obsessive-compulsive (anankastic) personality disorder (ICD-10: F60.5), and patients with mild cognitive impairment or dementia were excluded. Of the 61 included patients, five suffered from predominantly obsessional thoughts or ruminations (F42.0), six from predominantly compulsive acts (F42.1), and 50 from mixed obsessional thoughts and acts (F42.2).
FDP-OCD screening
The FDP-OCD screening includes advanced laboratory analysis with screening for pathogens and serum immunological markers, as well as instrumental diagnostics including an extended 3 Tesla magnetic resonance imaging (MRI) protocol, and electrocardiography (ECG). Extended diagnostic investigations in patients with suspected organic cases included cerebrospinal fluid (CSF) analysis, [18F]fluorodeoxyglucose positron emission tomography (FDG-PET), tissue-based assays on murine brain tissue for the detection of novel anti-central nervous system (CNS) antibodies, genetic and neuropsychological testing (Fig. 1).
Laboratory analyses
Blood analyses were offered to all patients as a routine procedure upon admission. The methodology of the tests has been described in previous publications [23, 25–27]. In addition to these routine diagnostic tests, 15 selected patients with suspected autoimmune OCD [16, 28] received a lumbar puncture to investigate an inflammatory cause of OCD. The CSF findings of OCD patients from our hospital were recently summarized in another publication, including patients from other wards and a larger retrospective interval [25, 28]. To discover potential novel anti-CNS antibodies, CSF–serum pairs of 12 patients were investigated on slices of unfixed murine brain tissue by indirect immunofluorescence [29, 30].
Instrument-based diagnostics
All patients were offered investigation with EEG (available from N = 60), extended MRI sequences of the neurocranium (available from N = 57), and ECG (available from N = 60). EEGs were examined by the respective ward clinician and retrospectively assessed for regional or focal slowing, intermittent generalized delta/theta activity (IRTA/IRDA), and epileptic activity. Independent component analysis (ICA) of EEGs with automatic calculation of IRDA/IRTA density was additionally performed as previously described [31]. MRI included the following sequences on a 3 Tesla scanner (MAGNETOM Prisma, Siemens Healthcare GmbH, Erlangen, Germany) in most patients: T1-weighted sequences with magnetization-prepared rapid gradient echo (MPRAGE) with isotropic 1-mm3 voxels for atrophy diagnostics, diffusion-weighted imaging (DWI) with axial 5-mm slices for stroke detection, fluid-attenuated inversion recovery (FLAIR) sequences with isotropic 1-mm3 voxels for the detection of signal alterations, and further innovative analyses such as diffusion tensor imaging (DTI) and pseudo-continuous arterial spin labeling. All MRIs were assessed and evaluated by experienced senior neuroradiologists. MRI abnormalities were categorized as white-/gray-matter alterations, atrophy, vascular changes, cysts, tumors, and anatomical variants, among others. An automated volume- and region-based approach (https://www.VEObrain.com) was used with the MPRAGE sequences for fully automated whole-brain volumetry for the detection of volume loss (VEOmorph, Freiburg, Germany). Selected cases (N = 7) with abnormalities in routine diagnostic work-up suggestive of an autoimmune cause were examined with cerebral [18F]fluorodeoxyglucose positron emission tomography (FDG-PET). Two patients received exome analysis due to suspected syndromal genesis.
Sociodemographic and psychometric data
During the admission routine, all important clinical and demographic data were documented. In addition, psychometric scales such as the Global Assessment of Functioning or Clinical Global Impression score were collected. In selected patients, further neuropsychological or psychometric assessments were performed.
Statistics
The retrospective data were collected and analyzed using SPSS (Version 28; IBM, New York, NY, USA). The analyses were mostly descriptive. The number of pathological values was based on established reference ranges.
Results
Sociodemographic and clinical findings
Most patients presented with a chronic course (>2 years) of the disease (78%), with an average of 14.8 years since OCD onset. On average it took six years from onset for OCD to be first diagnosed and patients lived on average 100 km away from the study center in Freiburg, Germany. A large proportion of patients were unmarried (72%), had at least a high school degree (52%), and were employed on a permanent basis (43%). Comorbid mental disorders were observed in nearly three-fourths of patients (74%), with one-quarter presenting with two comorbid diagnoses or more. Depression was by far the most frequent comorbidity (49%). Somatic comorbidities were registered in 82% of participants, with one-quarter of patients having endocrinological disorders. The three most common comorbidities were hypothyroidism (13%), arterial hypertension (8%), and Hashimoto’s thyroiditis (7%). Almost all patients were treated psychotherapeutically according to a cognitive-behavioral therapy protocol entailing exposure and reaction management (97%); in most cases (72%), pharmacological treatment was also provided with antidepressants. All data are summarized in Tables 1 and 2.
Table 1.
Description of the study cohort.
| Description of the study cohort | |||
| Sex | Age | ||
|
Female Male |
32 (52%) 29 (48%) |
Mean ± SD (range), in years | 32.67 ± 12.05 (18–69) |
| Diagnosis at admission | Origin/Country of birth | ||
| F42.2 | 50 (82%) | Germany | 54 (88%) |
| F42.1 | 6 (10%) | Europe | 4 (7%) |
| F42.0 | 5 (8%) | Unknown | 3 (5%) |
| Duration of illness (OCD) | Course of the disease | ||
| Mean ± SD (range), in years | 14.8 ± 11.8 (1–60), (N = 51) | Chronic course (>2 years) | 48 (78%) |
| First episode | 6 (10%) | ||
| Distance from home | Recurrent course | 6 (10%) | |
| Mean ± SD (range), in kilometers | 92.5 ± 100.4 (1–396), (N = 60) | Unknown | 1 (2%) |
| Inpatient/outpatient | Time between beginning of illness and diagnosis | ||
| Inpatient treatment | 52 (65%) | Mean ± SD (range), in years | 6.0 ± 6.4 (0–30), (N = 43) |
| Part-time inpatient | 2 (3%) | Earlier outpatient psychotherapies | |
| Inpatient, then only part-time | 7 (12%) | None | 10 (16%) |
| Earlier inpatient treatments | One | 28 (46%) | |
| None | 15 (24%) | Two | 16 (26%) |
| One | 15 (24%) | >Two | 3 (5%) |
| Two | 8 (13%) | Unknown | 4 (6%) |
| Three | 12 (20%) | ||
| Four and more | 8 (13%) | ||
| Unknown | 3 (5%) | ||
| Sociodemographic findings of OCD cohort | |||
| Civil status | Suicide attempts | ||
| Unmarried | 44 (72%) | None | 57 (93%) |
| Married | 10 (16%) | One | 2 (3%) |
| Divorced | 7 (12%) | Two | 2 (3%) |
| School education | |||
| Living situation | Low school degree | 5 (8%) | |
| Alone | 21 (34%) | Medium school degree | 14 (23%) |
| With partner/family | 17 (28%) | High school degree | 26 (42%) |
| With parents/siblings | 19 (31%) | University degree | 6 (10%) |
| Psychiatric transitional arrangement | 3 (5%) | No degree | 2 (3%) |
| Others/Unknown | 1 (2%) | Unknown | 8 (14%) |
| Occupation | Family history for psychiatric diseases | ||
| Employed | 26 (43%) | Positive | 37 (61%) |
| Unemployed | 15 (24%) | Negative | 17 (28%) |
| Disability pension | 2 (3%) | Unknown | 7 (11%) |
| Retirement pension | 2 (3%) | ||
| In-training/in studies/retraining | 12 (20%) | ||
| Others | 4 (7%) | ||
| Burden of acute events | Burden of long-term life circumstances | ||
| None | 17 (28%) | None | 5 (8%) |
| Mild | 8 (13%) | Mild | 9 (15%) |
| Intermediate | 17 (28%) | Intermediate | 24 (39%) |
| Severe | 15 (24%) | Severe | 14 (23%) |
| Extreme | 3 (5%) | Extreme | 5 (8%) |
| Unknown | 1 (2%) | Unknown | 4 (6%) |
| COMORBIDITIES | |||
| Mental disorder comorbidity | |||
| Number of psychiatric comorbidities | Affective disorders | ||
| Overall | 45 (74%) | Depression | 30 (49%) |
| • None | 16 (26%) | • mild | 5 (8%) |
| • One | 30 (49%) | • moderate | 12 (20%) |
| • Two | 8 (13%) | • severe | 11 (18%) |
| • >Two | 7 (12%) | • unknown | 2 (3%) |
| Psychotic disorders | 5 (8%) | Bipolar disorder | 0 (0%) |
| Paranoid hallucinatory | 1 (2%) | Personality disorders | 2 (3%) |
| Hebephrenic | 1 (2%) | Borderline personality disorder | 1 (2%) |
| Schizoaffective | 3 (5%) | Avoidant and dependent personality disorder | 1 (2%) |
| Neurodevelopmental disorders | 7 (11%) | ||
| ADHD | 2 (3%) | Anxiety disorders | 6 (10%) |
| Autism | 5 (8%) | Specific phobia | 6 (10%) |
| Tic disorders | 1 (2%) | Generalized anxiety disorder | 0 (0%) |
| Sleeping disorders | 1 (2%) | Panic disorder | 1 (2%) |
| Narcolepsy with cataplexy | 1 (2%) | Somatoform disorder | 3 (5%) |
| Substance abuse | 8 (13%) | Other disorders | 2 (4%) |
| Alcohol abuse | 6 (10%) | Trichotillomania | 1 (2%) |
| Illegal drug abuse | 3 (5%) | Dyslexia | 1 (2%) |
| Benzodiazepine | 1 (2%) | ||
| Somatic comorbidities | |||
| Somatic comorbidities | Somatic risk factors for psychiatric diseases | ||
| Overall | 50 (82%) | Pregnancy complications | 7 (11%) |
| • None | 11 (18%) | Febrile convulsions/seizures | 1 (2%) |
| • One | 18 (30%) | Traumatic brain injury | 6 (10%) |
| • Two | 12 (20%) | Previous meningitis/encephalitis | 0 (0%) |
| • Three | 10 (16%) | Severe infectious diseases | 1 (2%) |
| • Four and more | 10 (16%) | ||
| Neurological comorbidities | |||
| Neurological overall | 6 (10%) | Headache (e.g., migraine) | |
| Neuropsychiatric lupus erythematosusa | 1 (2%) | • Migraine without aura | 1 (2%) |
| Neurovascular | • Migraine not specified | 1 (2%) | |
| Cerebral microangiopathy | 2 (3%) | Circulation (e.g., syncope) | 1 (2%) |
| Progressive subcortical vascular encephalopathy | 1 (2%) | Traumatic injuries | 1 (2%) |
| Epilepsy (structural epilepsy) | 1 (2%) | Others | |
| Colloid cyst | 1 (2%) | ||
| Intracerebral cavernoma | 1 (2%) | ||
| Internistic comorbidity | |||
| Cardiological | 11 (18%) | Hematological | 4 (7%) |
| Arterial hypertension | 5 (8%) | Morbus Werlhof | 1 (2%) |
| Chronic venous insufficiency | 1 (2%) | Pseudothrombocytopenia | 1 (2%) |
| Aortic root dilation | 1 (2%) | Agranulocytosis | 1 (2%) |
| AV nodal reentry tachycardia | 1 (2%) | Nephrological | |
| Aortic valve sclerosis with mild to moderate aortic valve regurgitation | 1 (2%) | Chronic kidney disease stage 3 | 1 (2%) |
| Decent carotid bulb plaque | 1 (2%) | Endocrinological | 15 (25%) |
| Open foramen ovale | 1 (2%) | Hypothyroidism | 8 (13%) |
| Malignancies/tumors | 5 (8%) | Hashimoto’s thyroiditis | 4 (7%) |
| Diffuse large B-cell lymphoma | 1 (2%) | Hypertriglyceridaemia | 2 (3%) |
| Testicular carcinoma | 1 (2%) | Hyperprolactinaemia | 2 (3%) |
| Haemangioma in the hepatobilliary system and pancreas | 1 (2%) | Diabetes mellitus type 2 | 1 (2%) |
| Pituitary macroadenoma | 1 (2%) | Infectiological | 9 (15%) |
| Colon polyps | 1 (2%) | Dermatological | 3 (5%) |
| Gastroenterological | 7 (12%) | Infectious mononucleosis with EBV | 1 (2%) |
| Irritable Bowel Syndrome | 2 (3%) | Ophthalmic zoster | 2 (3%) |
| Gastroesophageal reflux disease | 2 (3%) | History of Lyme disease | 3 (5%) |
| Abdominal vasosclerosis | 1 (2%) | Pneumological | 5 (8%) |
| Others (gastritis, appendicitis, hemorrhoids) | 3 (5%) | Allergy/allergic asthma | 4 (7%) |
| Rheumatologic/autoimmune | 4 (7%) | Obstructive sleep apnea syndrome | 1 (2%) |
| Neurolupus | 1 (2%) | ||
| Spondylarthritis with HLA-B27 positivity | 1 (2%) | ||
| Fibromyalgia syndrome | 1 (2%) | ||
| Celiac disease | 1 (2%) | ||
| Other somatic comorbidities | |||
| Urogenital diseases | 5 (8%) | Gynaecological diseases | 3 (5%) |
| Urogenital infections (cystitis, urethritis) | 2 (4%) | Polycystic ovarian syndrome | 1 (2%) |
| Hypospadias | 1 (2%) | Premature ovarian failure | 1 (2%) |
| Varicocele | 1 (2%) | Pregnancy, puerperium, birth | 1 (2%) |
| Others (incontinence, narrow meatus) | 2 (4%) | Dermatologic diseases | 7 (12%) |
| Otorhinolaryngologic diseases | 3 (5%) | Eczema | 2 (4%) |
| Tinnitus aurium | 1 (2%) | Others (acne, furuncle, ictus reaction) | 5 (8%) |
| Hearing loss | 2 (4%) | Genetic syndromes | 4 (7%) |
| Muscle, skeleton and connective tissues disorders | 7 (12%) | Mayer–Rokitansky–Kuster–Hauser syndromea | 1 (2%) |
| Tendopathy | 3 (5%) | Duplication of chromosome 15q11-13a,b | 1 (2%) |
| Musculoskeletal injury | 3 (5%) | Turner syndrome | 1 (2%) |
| Drug-induced rhabdomyolysis | 1 (2%) | Triple-X syndrome | 1 (2%) |
| Coxarthrosis | 1 (2%) | Baraitser–Winter syndromeb | 1 (2%) |
Some parameters were not available from all patients, then the reduced sample size is always indicated in parentheses. ADHD Attention deficit hyperactivity disorder, AV atrioventricular, EBV Epstein-Barr virus, HLA human leukocyte antigen, N number of patients, OCD obsessive compulsive disorder, SD standard deviation.
aThe same patient suffered from both syndromes.
bNewly diagnosed.
Table 2.
Psychometric findings and therapy.
| Psychometric findings and therapy | |||
| GAF on admission | CGI on admission | ||
| Mean ± SD (range) | 48.4 ± 13.3 (22–81), (N = 61) | Borderline ill | 0 (0%) |
| Mildly ill | 0 (0%) | ||
| Moderately ill | 1 (2%) | ||
| Markedly ill | 15 (25%) | ||
| Severely ill | 38 (62%) | ||
| Extreme severely ill | 5 (8%) | ||
| Unknown | 2 (3%) | ||
| Treatment of inpatients | |||
| Overall psychopharmacological treatment on admission | Number of psychopharmaceutical classes per patient | ||
| Psychopharmacological treatment received | 44 (72%) | No treatment | 17 (28%) |
| No psychopharmacological treatment received | 17 (28%) | One class | 27 (44%) |
| Psychopharmacological treatment on admission in detail | Two classes | 15 (25%) | |
| SSRI | 32 (53%) | Three classes | 2 (3%) |
| Atypical neuroleptics | 13 (21%) | Psychotherapy concept treatment on the OCD ward overall | |
| Typical neuroleptics | 4 (7%) | No | 2 (3%) |
| Lithium | 2 (3%) | Yes | 59 (97%) |
| Anticonvulsants | 2 (3%) | ||
| Benzodiazepines | 2 (3%) | ||
| Stimulants | 2 (3%) | ||
| Clonidine | 1 (2%) | ||
| Clomipramine | 3 (5%) | ||
| Other antidepressants | 13 (21%) | ||
CGI clinical global impression, GAF global assessment of functioning, SD standard deviation, N numbers of patients, OCD obsessive compulsive disorder, SSRI Selective serotonin reuptake inhibitors.
Laboratory blood findings
In serum analysis, reduced leukocyte counts were identified in 10% of patients with OCD. Furthermore, increased calcium levels were found in 18% of participants, partial thromboplastin times (PTT) were decreased in 14%, GPT was increased in 12%, and increased total glycerides were detectable in 25% of the patients. Regarding vitamins, OCD patients presented with decreased vitamin D levels in 35% of cases—up to 75% when applying the optimal value of 30 ng/ml—and a folic acid deficiency was found in 24%. Signs for current Lyme disease in serology were identified in 6% of patients, whereby evidence of past Lyme disease was identified in 7%. Streptococcal antibodies were detected in 46% of the patients (anti-streptolysin in 38%, streptococcal-DNAseB in 23%). Rheumatological screening revealed aberrant rheumatic markers (CRP, immunoglobulins, complement system) in 34%, with elevated CRP levels in 16%. Furthermore, positive rheumatic serum autoantibodies were observed in 42% of patients, with 36% presenting with positive antinuclear antibodies (ANA) tested by indirect immunofluorescence (IIF) on human epithelial type 2 (HEp-2) cells (16% clearly positive and 20% borderline) and 10% with elevated anti-phospholipid antibodies. In addition, positive anti-thyroid antibodies were identified in 24%, with positive antibody findings for anti-thyroid peroxidase (TPO) in 19% and anti-thyroglobulin (TG) in 13%, but none against the thyroid-stimulating hormone receptor.
Well-characterized anti-neuronal serum antibodies against cell-surface (NMDA-R/LGI1/CASPR2/AMPA1/2-R/GABA-B-R/DPPX) and intracellular antigens (Yo/Hu/CV2[CRMP5]/Ri/Ma1/2/SOX1/Tr/Zic4/GAD65/amphiphysin) were negative in all OCD patients. Tissue-based assays on murine brain slices detected abnormal serum findings in five of 13 (38%) patients examined. This included one finding with binding against neuropil in the basal ganglia and brain stem [20] and one with a novel cytoplasmic pattern [32]. The other three patients had strong antinuclear binding with a perinuclear pattern. In addition, in two patients, a borderline finding with an anti-myelin-binding pattern was observed. All laboratory results are summarized in Table 3 and prevalences of laboratory, MRI and EEG alterations in OCD are presented in Fig. 2.
Table 3.
Laboratory findings.
| STANDARD LABORATORY ANALYSIS | ||
|---|---|---|
| Parameter (reference values) | Mean ± Standard deviation | Patients with increased/decreased values |
| Differential blood count | ||
| Leukocytes (4000–1040/µl) | 6330 ± 2000/µl (N = 61) | 3 ↑ (5%), 6 ↓ (10%), 52 normal (85%) |
| Platelets (150,000–400,000/µl) | 241,980 ± 55,340/µl (N = 61) | 4 ↓ (7%), 57 normal (93%) |
| Erythrocytes (♂: 4.5–5.8 mio/µl, ♀: 4.0–5.2 mio/µl) | 4.80 ± 0.47 mio/µl (N = 61) | 2 ↓ (7%), 59 normal (93%) |
| MCV (80.0–95.5 fl) | 87.57 ± 4.30 fl (N = 61) | 2 ↑ (3%), 1 ↓ (2%), 58 normal (95%) |
| MCH (27.6–32.8 pg) | 30.15 ± 1.46 pg (N = 61) | 4 ↑ (7%), 57 normal (93%) |
| MCHC (32.8–36.6 g/dl) | 34.45 ± 0.94 g/dl (N = 61) | 9 ↑ (15%), 52 normal (85%) |
| Hemoglobin (♂: 13.5–17.6 g/dl, ♀: 11.6–15.5 g/dl) | 14.47 ± 1.38 g/dl (N = 61) | 61 normal (100%) |
| Hematocrit (34.6–45.3%) | 41.97 ± 3.57% (N = 61) | 61 normal (100%) |
| Neutrophil granulocytes (40–75%) | 55.31 ± 10.05% (N = 59) | 1 ↑ (2%), 3 ↓ (5%), 55 normal (93%) |
| Lymphocytes (19–51%) | 32.55 ± 8.70% (N = 59) | 1 ↑ (2%), 3 ↓ (5%), 55 normal (93%) |
| Eosinophils (0–7%) | 2.43 ± 1.58% (N = 59) | 1 ↑ (2%), 58 normal (98%) |
| Basophile (0–2%) | 0.69 ± 0.33% (N = 59) | 59 normal (100%) |
| Monocytes (2–14%) | 8.77 ± 2.55% (N = 59) | 1 ↑ (2%), 58 normal (98%) |
| Neutrophil absol. (1.8–6.2 tsd/µl) | 3550 ± 1470 tsd/µl (N = 59) | 4 ↑ (7%), 3 ↓ (5%), 52 normal (88%) |
| Lymphocytes absol. (1200–3600/µl) | 1920 ± 520/µl (N = 59) | 5 ↓ (9%), 54 normal (91%) |
| Eosinophile absol. (30–440/µl) | 150 ± 110/µl (N = 59) | 2 ↓ (3%), 57 normal (97%) |
| Basophile absol. (10–80/µl) | 40 ± 20/µl (N = 59) | 59 normal (100%) |
| Monocytes absol. (250–850/µl) | 530 ± 170/µl (N = 59) | 1 ↑ (2%), 58 normal (98%) |
| Coagulation | ||
| Quick (70–130%) | 98.77 ± 12.70% (N = 56) | 56 normal (100%) |
| INR (0.85–1.15) | 1.02 ± 0.05 (N = 58) | 58 normal (100%) |
| PTT (25.1–37.7 s) | 29.38 ± 3.79 sec (N = 58) | 2 ↑ (3%), 8 ↓ (14%), 48 normal (83%) |
| Electrolytes | ||
| Sodium (136–145 mmol/l) | 139.85 ± 1.64 mmol/l (N = 61) | 1 ↓ (2%), 60 normal (98%) |
| Potassium (3.5–5.1 mmol/l) | 4.26 ± 0.30 mmol/l (N = 61) | 1 ↓ (2%), 60 normal (98%) |
| Calcium (2.15–2.5 mmol/l) | 2.41 ± 0.10 mmol/l (N = 61) | 11 ↑ (18%), 49 normal (82%) |
| Metabolic markers | ||
| Creatinine (0.51–0.95 mg/dl) | 0.86 ± 0.17 mg/dl (N = 61) | 2 ↑ (3%), 1 ↓ (2%), 58 normal (95%) |
| CK (<170 U/l) | 122.35 ± 126.273 U/l (N = 55) | 5 ↑ (9%), 50 normal (91%) |
| GOT (10–35 U/l) | 23.95 ± 7.55 U/l (N = 60) | 4 ↑ (7%), 56 normal (93%) |
| GPT (10–35 U/l) | 23.64 ± 11.19 U/l (N = 61) | 7 ↑ (12%), 2 ↓ (3%), 52 normal (85%) |
| Alkaline phosphatase (♂: 40–130 U/l, ♀: 35–105 U/l) | 72.15 ± 21.77 U/l (N = 54) | 2 ↑ (4%), 52 normal (96%) |
| Gamma-GT (<40 U/l) | 23.15 ± 24.70 U/l (N = 61) | 5 ↑ (8%), 56 normal (92%) |
| Lipase (13–60 U/l) | 33.07 ± 16.42 U/l (N = 57) | 2 ↑ (3%), 55 normal (97%) |
| HbA1C (3.4–6%) | 5.29 ± 0.29% (N = 54) | 1 ↑ (2%), 53 normal (98%) |
| Total triglycerides (<150 mg/dl) | 112.84 ± 58.59 mg/dl (N = 55) | 14 ↑ (25%), 41 normal (75%) |
| Thyroid hormones | ||
| TSH (0.27–4.20 µU/ml) | 1.80 ± 0.82 µU/ml (N = 60) | 2 ↓ (3%), 58 normal (97%) |
| Free T3 (3.1–6.8 pmol/l) | 5.14 ± 0.75 pmol/l (N = 58) | 58 normal (100%) |
| Free T4 (12–22 pmol/l) | 15.51 ± 2.59 pmol/l (N = 58) | 4 ↓ (7%), 54 normal (93%) |
| “Neurovitamins” | ||
| 25-OH-Vitamin D2/D3 (>20 ng/ml) | 24.48 ± 10.58 ng/ml (N = 57) | 20 ↓ (35%), 37 normal (65%) |
| •Optimal 25-OH-Vitamin D2/D3 (>30 ng/ml) | 43 ↓ (75%), 14 normal (25%) | |
| Folic acid (4.8–37.3 ng/ml) | 7.73 ± 4.66 ng/ml (N = 54) | 13 ↓ (24%), 41 normal (76%) |
| Vitamin B12 (197–771 pg/ml) | 432.36 ± 179.78 pg/ml (N = 55) | 2 ↑ (3%), 1 ↓ (2%), 52 normal (95%) |
| ADVANCED LABORATORY ANALYSIS | ||
|---|---|---|
| Parameter (Reference) | Patients with positive/negative/borderline findings or increased/decreased values | |
| Pathogen findings | ||
| Serology for Lyme disease | ||
| IgG-ELISA screening (up to 16 U) | 7 positive (13%), 46 negative (83%), 2 bord. (4%) (N = 52) | |
| IgM-ELISA screening (up to 5 U) | 3 positive (6%), 43 negative (83%), 6 bord. (11%) (N = 52) | |
| Western blot confirmation test (only performed if ELISA screening was conspicuous) |
- IgG: positive: 4 (7%), bord.: 1 (2%), negative: 4 (7%) - IgM: positive: 3 (6%), bord.: 1 (2%), negative: 5 (10%) - Both IgG and IgM positive: 2 (4%) |
|
| Serology for lues | ||
| Screening using Chemiluminescence Immunoassays (<0.9) | 52 normal (100%), (N = 52) | |
| Streptococcal antibodies | ||
| Anti-streptolysin (≥200 IU/ml positive) | 18 ↑ (38%), 27 normal (60%) | |
| Mean ± SD: 190.04 ± 131.84 (N = 48) | ||
| Anti-streptococcal-DNAseB U/ml (≥300 positive) | 11 ↑ (23%), 36 normal (77%) | |
| Mean ± SD: 240.58 ± 322.22 (N = 47) | ||
| Immunological findings | ||
| Rheumatic/immunological serum markers | ||
| C-reactive protein (<5 mg/l) | 9 ↑ (16%), 48 normal (84%) (N = 57) | |
| IgG (7–16 g/l) | 3 ↓ (6%), 52 normal (94%) (N = 55) | |
| Mean ± SD: 11.03 ± 2.14 (N = 55) | ||
| IgA (0.70–4 g/l) | 2 ↑ (4%), 3 ↓ (5%), 50 normal (91%) (N = 57) | |
| Mean ± SD: 2.05 ± 0.86 (N = 55) | ||
| IgM (0.40–2.30 g/l) | 1 ↑ (2%), 4 ↓ (7%), 52 normal (91%) (N = 57) | |
| Mean ± SD: 0.99 ± 0.50 (N = 55) | ||
| C3 (0.90–1.80 g/l) | 1 ↑ (2%), 3 ↓ (5%), 51 normal (93%) (N = 55) | |
| C4 (0.10-0.40 g/l) | 55 normal (100%) (N = 55) | |
| CH50 (65–115%) | 8 ↑ (15%), 1 ↓ (2%), 44 normal (83%) (N = 53) | |
| Rheumatic markers overall | 20 positive (34%), 38 negative (66%) (N = 58) | |
| Brain-associated systemic antibodies | ||
| Anti-TSHR antibodies (<1.75 IU/l) | 54 normal (100%) (N = 54) | |
| Anti-TPO antibodies (<34 IU/ml) | 10 ↑ (19%), 44 normal (81%) (N = 54) | |
| Anti-TG antibodies (<115 IU/ml) | 7 ↑ (13%), 47 normal (87%) (N = 54) | |
| Anti-GAD65 antibodies measured by RIA (< 1 U/ml, bord. 1–2 U/ml, positive >2 U/ml) | 1 positive (2%), 1 bord. (2%), 50 normal (96%) (N = 52) | |
| Lysoganglioside antibodies (GM1 IgG <10 normal, bord.: <15, positive >15) | 35 normal (100%) (N = 35) | |
| Anti-thyroid and diabetes antibodies overall | 13 positive (24%), 41 negative (76%) (N = 54) | |
| Rheumatic serum autoantibodies | ||
| Anti-phospholipid/ß2GP IgG antibodies (<14 GPL-U/ml) | 3 ↑ (6%), 50 normal (94%) (N = 53) | |
| Anti-phospholipid /ß2GP IgM antibodies (<10 MPL-U/ml) | 2 ↑ (4%), 52 normal (96%) (N = 53) | |
| ß2Glycoprotein1AKIgG | Positive 1 (2%), negative 50 (98%) (N = 51) | |
| ANA screening by indirect immunofluorescence (ANA-IIF) using Human Epithelial type 2 (Hep-2) cells | ||
| • against nucleus (1:50) | Positive 3a (5%), bord. 14 (25%), negative 38 (69%) (N = 55) | |
| • against nucleoli (1:50) | Positive 1 (2%), negative 54 (98%) (N = 55) | |
| • against chromosomes (1:50) | Positive 7a (13%), bord. 7 (13%), negative 41 (75%) (N = 55) | |
| • against cytoplasm (1:50) | Bord. 4 (7%), negative 51 (93%) (N = 55) | |
| ANA overall positive/borderline findings | Positive 9 (16%), bord. 11 (20%), negative 35 (64%) (N = 55) | |
| ENA-Screening (only performed if ANA-IIF screening was clearly positive) |
Positive 4 (7%), negative 6 (11%) (N = 10) Positive cases in detail: One patient with anti-Jo1 and anti-mitochondrial M2 abs as well as two patients with anti PCNA (proliferating cell nuclear antigen) abs. One patient had repeated anti-nucleosome abs. in the past due to lupus. One patient with anti DFS-70 abs. |
|
| Anti-dsDNA ELISA ( < 40 U/ml; only performed if ANA-IIF screening was clearly positive and suspicious clinical findings were present) | Negative 7 (100%) (N = 7) | |
| ANCA (IgG, 1:10) | Positive 0 (0%), negative 57 (100%) (N = 57) | |
| Rheumatic autoantibodies overall | Positive 23 (42%), negative 32 (58%) (N = 55) | |
| Well-characterized neuronal serum autoantibodies | ||
| Antibodies against intracellular onconeural antigens (Hu, Yo, Ri, cv2(CRMP5), Ma1/ -Ma2, SOX, Tr(DNER), Zic4, amphiphysin, GAD65) |
(N = 52) Positive 0 (0%) Negative 52 (100%) |
|
| Antibodies against neuronal cell surface antigens (LGI1, CASPR2, GABA-B, NMDA-R, AMPA 1/2) |
(N = 51) Positive 0 (0%) Negative 51 (100%) |
|
| Tissue-based assays (Prof. Prüss, Charité Berlin, Germany) in serum | ||
| (N = 13) | ||
| None/weak/unspecific (negative) | 6/13 (46%) | |
| Moderate/specific (borderline) | 2/13 (15%) | |
| Strong/specific (positive) | 5/13 (38%) | |
All results, which were altered for more than 10 percent of patients are in bold and italic, and with more than 15 percent only in bold.
↓ decreased, ↑ increased, abs antibodies, absol. absolute, ANA antinuclear antibodies, ANCA antineutrophil cytoplasmic antibody, bord. borderline, bord. borderline, C3 complement component 3, C4 complement component 4, CASPR2 Contactin-associated protein-like 2, CH50 50% hemolytic complement, CK creatine kinase, CRMP5 collapsing response-mediator protein-5, DFS dense fine speckled, DNA deoxyribonucleic acid, DNER delta/notch-like epidermal growth factor-related receptor, dsDNA double stranded deoxyribonucleic acid, ELISA Enzyme-linked immunosorbent assay, ELISA enzyme-linked immunosorbent assay, ENA extractable nuclear antigens, GABA-B gamma-aminobutyric acid receptor B, GAD glutamic acid decarboxylase, Gamma-GT Gamma-glutamyl transferase, GOT glutamic-oxaloacetic transaminase, GPT Glutamic-Pyruvic transaminase, HbA1c hemoglobin A1c, IgA immunoglobulin A, IgG immunoglobulin G, IgG immunoglobulin G, IgM immunoglobulin M, IgM immunoglobulin M, INR international normalized ratio, LG1 Leucine-rich glioma-inactivated 1, MCH Mean corpuscular hemoglobin, MCHC mean corpuscular hemoglobin concentration, MCV Mean corpuscular volume, MPA α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid, N numbers of patients, NMDA N-methyl-D-aspartate, PCNA proliferating cell nuclear antigen, PTT partial thromboplastin time, RIA radioimmunoassay, SD standard deviation, ß2GP ß2 glycoprotein, T3 triiodothyronine, T4 thyroxine, TG thyroglobulin, TPO thyroid peroxidase, TSH thyroid-stimulating hormone, TSHR thyroid-stimulating hormone-receptor, Zic4 zinc-finger protein 4.
aIncluding one patient with lupus and highly positive ANA-titers with repeated anti-nucleosomes abs. in the past before treatment.
Fig. 2. Prevalence of laboratory, MRI, and EEG alterations in OCD patients.
Elevated parameters are displayed in yellow/orange/red depending on level of elevation. Decreased parameters are displayd in blue tones. CH50 complement hemolytic activity, CSF Cerebrospinal fluid, DNAse Deoxyribonuclease, EEG Electroencephalogram, GM gray matter, MRI magnetic resonance imaging, OCB oligoclonal band, PTT Partial thromboplastin time, TG Thyroglobulin, TPO Thyroid peroxidase, WBC white blood cell.
Magnetic resonance imaging
Clinical assessment
MRI revealed abnormalities such as cerebral lesions, anatomic abnormalities, cysts, or tumors in 15 patients (26%). However, only 11 findings (19%) were rated as potentially relevant for mental disorders with specific gray or white matter changes. These findings included lesions in the right precentral gyrus, left tapetum, left thalamus, trigonum of a ventricle or other potential post-inflammatory localizations cortical, juxtacortical, and periventricular. No relevant atrophy was reported on visual assessment (Table 4).
Table 4.
Magnetic resonance imaging (MRI) findings in clinical assessment and using automated Veobrain analysis (available in most patients), as well as electroencephalography (EEG) results.
| Clinical MRI assessment (N = 57) | Number of patients with changes |
|---|---|
| White/gray matter changes | |
| Specific changes vs no specific changes | 11 (19%) vs. 46 (81%) |
| Non-specific white matter changes | 12 (21%) |
| (Post-)inflammatory white matter changes | 6 (11%) |
| Parenchymal defect | 2 (4%) |
| Atrophic changes | |
| No atrophy | 57 (100%) |
| Generalized/localized cortical atrophy | 0 (0%) |
| Ventricle enlargement | 0 (0%) |
| Vascular changes | |
| Macroangiopathic vascular alterations (post-ischemic changes) | 2 (4%) |
| Microhaemorrhage | 3 (5%) |
| Cysts, tumors, anatomical variants & other changes | |
| Cysts | |
| • Pineal cyst | 3 (5%) |
| • Arachnoid cyst | 2 (4%) |
| • Pars intermedia cyst | 1 (2%) |
| Tumors | |
| • Meningioma | 1 (2%) |
| Anatomical variants | |
| • Partial empty sella | 1 (2%) |
| • Virschow Robin spaces in loco typico | 1 (2%) |
| Overall MRI changes | |
| • Relevant abnormalities | 11 (19%) |
| • Any changes (incl. non-specific changes) | 15 (26%) |
| • No changes | 42 (74%) |
| Automated Veobrain analyses (N = 44) | Number of patients with changes | Mean ± SD percentage of voxel with z-score <−2 for gray matter decrease or >3 for CSF volume increase (only patients with changes) |
|---|---|---|
| CSF volume increase | ||
| Orbitofrontal | 2 (5%) | 10.22 ± 0.81 |
| (R:1/L:2) | (R:10.79 /L:9.46 ± 0.28) | |
| Frontal | 1 (2%); (R:0/L:1) | 8.74 |
| Parietal | 1 (2%) (R:1/L:0) | 7.28 |
| Temporal | 0 (0%) | |
| Mesiotemporal | 4 (9%) | 7.37 ± 2.88 |
| (R:3/L:3) | (R:8.43 ± 2.37/L:6.50 ± 2.03) | |
| Occipital | 3 (7%) | 7.16 ± 2.43 |
| (R:3/L:3) | (R:6.84 ± 3.34/L:7.56 ± 0.34) | |
| Cerebellum | 1 (2%) | 5.73 |
| Overall | 8 (18%) | |
| Gray matter volume decrease | ||
| Orbitofrontal | 7 (16%) | 13.92 ± 7.82 |
| (R:6/L:6) | (R: 15.54 ± 7.18/L:11.13 ± 6.66) | |
| Frontal | 7 (16%) | 8.33 ± 3.27 |
| (R:7/L:4) | (R:7.81 ± 2.50/L:8.16 ± 4.24) | |
| Parietal | 7 (16%) | 9.21 ± 3.06 |
| (R:4/L:6) | (R:9.10 ± 3.47/L:9.49 ± 2.57) | |
| Temporal | 6 (14%) | 7.55 ± 2.72 |
| (R:2/L:5) | (R: 9.44 ± 0.26/L:7.13 ± 2.82) | |
| Mesiotemporal | 3 (7%) | 18.36 ± 10.64 |
| (R:2/L:2) | (R:13.06 ± 7.57/L:20.93 ± 1139) | |
| Occipital | 8 (18%) | 8.54 ± 3.39 |
| (R:7/L:6) | (R:7.83 ± 3.57/L:9.24 ± 3.49) | |
| Central | 1 (2%) (R:0/L:1) | 10.75 |
| Cerebellum | 1 (2%) | 10.06 |
| Overall | 19 (43%) | |
| Overall changes in Veobrain analyses | 20/44 (45%) | |
| EEG findings (N = 60) | |
|---|---|
| Alterations | Number of patients with changes |
| Overall alterations | 4 (7%) |
| No alterations | 56 (93%) |
| Continuous generalized/reginal slow activity | 0 (0%) |
| Intermittent generalized slow activity | 3 (5%) |
| Intermittent regional slow activity | 1 (2%) |
| Epileptic pattern | 0 (0%) |
| Other alterations (e.g., anteriorization of the basic activity) | 0 (0%) |
| Automated IRDA/IRTA detection | Mean ± SD (range) |
| IRDA/IRTA rate before hyperventilation (per min.) | 1.12 ± 2.46 (0.00–17.30), (N = 60) |
| IRDA/IRTA rate after hyperventilation (per min.) | 1.26 ± 2.83 (0.00–18.82), (N = 53) |
| Difference in IRDA/IRTA rates before and after hyperventilation (per min.) | 0.69 ± 0.82 (0.00–3.2), (N = 53) |
| IRDA/IRTA rate overall (per min.) | 1.18 ± 2.51 (0.00–18.04), (N = 60) |
| Clinically relevant IRDA/IRTA-rate of more than 6 per minute | N = 2 (3%) |
CSF cerebrospinal fluid, EEG electroencephalography, IRDA/IRTA intermittent rhythmic delta and theta activity, L left, MRI magnetic resonance imaging, N number of patients, R right, SD standard deviation.
Automated MRI analysis
Fully automated analyses of 44 patients (only possible for patients with available MPRAGE sequence) showed alterations in 20 (45%) of them. Most prominently volume reduction, with a mean percentage of voxel with z-score < −2, of occipital (18%), but also orbitofrontal, frontal and parietal (each 16%) cortex were observed. A CSF volume increase, with percentage of voxel with z-score > 3, was detected mostly in mesiotemporal areas (9%). If affected, the mesiotemporal cortex with 18.36 and the orbitofrontal cortex with 13.92 deviated the most from the reference group. All four patients with genetic syndromes and OCD had orbitofrontal abnormalities.
EEG findings
EEG revealed changes in visual assessment in the form of slowing in 7% of all patients (Table 4). In the automatic IRDA/IRTA detection, two patients (3%) were observed with increased IRDA/IRTA rates (in-house ref.: rate of more than 6 per minute). These were already visually classified with mainly regional slow activity. The ECG findings are summarized in Supplementary Table S1.
Further analyses in selected cases
For CSF analysis, five of the 15 patients who underwent lumbar puncture presented abnormalities in the form of elevated WBC count (N = 3), elevated protein concentrations (N = 4), elevated IgG index (N = 1), and oligoclonal bands (OCBs) in CSF (N = 3). Of the 12 patients in which tissue-based assays were performed using CSF, five patients presented clearly positive antibody patterns. Two had specific neuronal antibody patterns—one patient each with a pattern against neuropil in basal ganglia and brain stem [33] and one patient with antibodies against the cilia of granule cells [34]. Three patients showed non-specific findings with strong antinuclear binding with partly perinuclear patterns in two of them. In addition, there was a borderline finding for one patient with an anti-myelin-binding pattern.
In the FDG-PET analysis of seven patients, only one showed alterations (see below; Table 5). Two patients were clinically suspected of having a previously undiagnosed genetic syndrome. An exome analysis was conspicuous in both patients (see below).
Table 5.
Additional analyses in a subgroup of patients.
| Cerebral spinal fluid (N = 15) and FDG-PET analysis (N = 7) | |
|---|---|
| WBC count (Mean ± SD, range) | 3.20 ± 3.91 (from 1 to 14/µl) |
| Increased WBC counta (ref. <5/µl) | ↑: 3a (20%) ↔: 12 (80%) |
| Protein concentration (Mean ± SD, range) | 380.40 ± 193.86 (from 159 to 927 mg/l) |
|
Increased total protein concentration (ref. <450 mg/l) |
↑: 4 (27%) |
| ↔: 11 (73%) | |
| Albumin quotients (Mean ± SD, range) | 5.42 ± 3.06 (from 2.90 to 14.30 × 10−3) |
| Increased albumin quotients (ref.: <40y.: <6.5 × 10−3; 40–60y.: <8 × 10−3; >60y.: <9.3 × 10−3) | ↑: 3 (20%) |
| ↔: 12 (80%) | |
| IgG-Index (Mean ± SD, range) | 0.54 ± 0.23 (from 0.41 to 1.37) |
| Number of patients with increased IgG indices (ref. <0.7) | ↑: 1 (7%) |
| ↔: 14 (93%) | |
| Isolated OCB in CSF | 3/15 (20%) |
| OCBs in CSF and Serum | 0/15 (0%) |
| Overall basic CSF alterations | 5 (33%) (N = 15) |
| Neuronal CSF antibodies | |
| Antibodies against neuronal cell surface antigens (LGI1, CASPR2, GABA-B, NMDA-R, AMPA 1/2) | Positive 0 (0%) (N = 15) |
| Negative 15 (100%) (N = 15) | |
| Tissue based assays (Prof. Prüss, Charité Berlin, Germany) | (N = 12) |
| None/weak/unspecific (negative) | 6/12 (50%) |
| Moderate/specific (borderline) | 1/12 (8%) |
| Strong/specific (positive) | 5/12 (42%) |
| FDG-PET | |
| Normal | 6 (86%) |
| Hypermetabolism | 0 (0%) |
| Hypometabolism | 1 (14%) |
AMPA α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid, CASPR2 Contactin-associated protein-like 2, CSF Cerebrospinal fluid, FDG-PET fluorodeoxyglucose - positron emission tomography, GABA-B gamma-aminobutyric acid receptor B, IgG Immunoglobulin G, LG1 Leucine-rich glioma-inactivated 1, n/N numbers of patients, NMDA N-methyl-D-aspartate, OCBs Oligoclonal bands, ref. reference, SD standard deviation, WBC white blood cell, ↑ increased, ↓ decreased, ↔ no change.
aIncluding one patient with neurolupus with pleocytosis and positive OCBs in the CSF in the past.
Organic OCD cases
Probable organic OCD was detectable in a total of five patients (8%) and possible organic OCD was detected in another five patients (8%).
Probable autoimmune OCD
The following autoimmune OCD cases were identified as probable organic froms: A female patient with pleocytosis, granule cell and endothelial cell antibodies as well as a reversible MRI lesion at the tapetum [34] (Patient 1), a patient with antibodies against basal ganglia structures in the CSF, autoimmune susceptibility (different autoantibodies in serum), and MRI changes with bilateral small white-matter lesions and a paraventricular defect along the left posterior horn, as well as a bilateral orbitofrontal volume reduction in automated MRI analyses [20] (Patient 2), and another autoimmune patient had pre-diagnosed neuropsychiatric lupus with inflammatory CSF changes and periventricular white-matter lesions (Patient 3). All three patients showed relevant improvement after initial high-dose steroid treatment over five days (in patient 1 decrease of Y-BOCS-scores from 29 to 0 after approx. 7 weeks; in patient 2 decrease of Y-BOCS-scores from 22 to 9 after approx. 8.5 weeks, in patient 3 unfortunately no Y-BOCS history was available).
Possible autoimmune OCD
Possible organic OCD was observed in a female patient with suspected Hashimoto’s encephalopathy (with highly elevated thyroid antibodies—titer level was associated with clinical symptom severity, postinflammatory MRI lesions, anti-(peri)nuclear antibodies in the tissue test of serum and CSF, as well as blood-CSF-barrier dysfunction (Patient 6; cf. [35]), as well as in one patient with slight pleocytosis in CSF, non-specific white-matter changes, slightly elevated ANAs, and borderline myelin-binding in serum using tissue-based assays (Patient 7). The only patient with alterations in the FDG-PET (with a slight asymmetry of the metabolism of the association cortices with a low frontotemporal emphasis and a z-score of −2) was also considered as possible autoimmune OCD due to additional positive OCBs in CSF, strong antinuclear binding in serum and CSF using tissue-bases assays, severe rhabdomyolysis under risperidone augmentation, as well as an occipital gray matter reduction and CSF increase in automated MRI analyses (Patient 8).
Probable genetic OCD
Genetic exome analysis was initiated in two patients with suspected genetic syndromes. Both patients had a probable syndromal cause related to newly diagnosed genetic syndromes (Patient 4: duplication of chromosome 15q11-13 and Mayer–Rokitansky–Küster–Hauser syndrome; Patient 5: Baraitser–Winter syndrome) and brain involvement (Patient 4: hearing loss, reduced intelligence quotient (IQ), bilateral orbitofrontal/frontal gray matter decrease in the automated MRI analyses; Patient 5: hearing loss, reduced IQ levels, small parenchymal brain defect right temporal after cavernomectomy, and a volume loss in the parietal/occipital cortex, left orbitofrontal, and the right mesiotemporal cortex) [36].
Possible genetic OCD
Furthermore, two patients with pre-diagnosed chromosome disorders involving the X chromosome (Patient 9: Triple X syndrome; Patient 10: Turner syndrome) and morphometric MRI alterations (bilateral (Patient 9), left (Patient 10) decreases in orbitofrontal gray-matter volume) were classified as possible organic cases [37].
All organic cases are summarized in detail in Table 6 and exemplary findings are depicted in Fig. 3.
Table 6.
Characteristics of probable and possible organic OCD patients.
| Case | Sex | Age | OCD symptoms | Somatic comorbidity | Immunological changes | CSF | Tissue-based assays | MRI and FDG-PET | Changes in Veobrain analyses | EEG (IRDA rate) |
|---|---|---|---|---|---|---|---|---|---|---|
| Probable organic OCD | ||||||||||
| Pat. (1) | F | Mid 20 | Mixed obsessional thoughts and acts | None | Elevated CRP (+) | Pleocytosis (10 cells), increased IgG index and elevated lactate, positive OCB in CSF | Abs against cilia of granule cells in CSF (not in serum) |
MRI: post-inflammatory lesion of left tapetum, pinealis cyst FDG-PET inconspicuous |
Inconspicuous | Inconspicuous (0/min) |
| Pat. (2) | M | ~60 | Mixed obsessional thoughts and acts | Arterial hypertension | ANA 1:400 with anti-Jo1 and anti-mitochondrial M2 abs, elevated CRP and anti-TPO abs | Inconspicuous | Abs against neuropil in basal ganglia (and brain stem) in CSF and serum |
MRI: bilateral small white matter lesions, paraventricular defect (left posterior horn) FDG-PET: inconspicuous |
Bilateral orbitofrontal CSF-volume reduction on both sides | Inconspicuous (0/min) |
| Pat. (3)a | M | Mid 20 | Mixed obsessional thoughts and acts, comorbid psychotic symptoms initially | Neuropsychiatric lupus | ANA 1:800 with anti-nucleosome specifity, anti-streptolysine and anti-streptococcal-DNAseB abs positive | Pleocytosis (14 cells), slightly increased total protein, positive OCB in CSF | Not investigated | MRI: multiple periventricular white matter lesions | Inconspicuous | Inconspicuous (0/min) |
| Pat. (4) | F | Mid 40 | Mixed obsessional thoughts and acts | Duplication of chromosome 15q11-13, Mayer–Rokitansky–Küster–Hauser syndrome | None | Not investigated | Not investigated | MRI: inconspicuous | GM-Volume reduction orbitofrontal on both sides, frontal on both sides, parietal right | Inconspicuous (0.36/min) |
| Pat. (5) | M | Mid 20 | Predominantly compulsive acts | Baraitser–Winter syndrome | Anti-streptolysine and anti-streptococcal-DNAseB abs positive | Not investigated | Not investigated | MRI: small parenchymal brain defect right temporal after cavernomectomy | GM-Volume reduction parietal on both sides, CSF-volume reduction orbitofrontal left, mesiotemporal right, occipital on both sides | Inconspicuous (1.53/min) |
| Possible organic OCD | ||||||||||
| Pat. (6) | F | ~40 | Mixed obsessional thoughts and acts | Arterial hypertension | Elevated anti-TPO and TG abs | Slight BBB-dysfunction (AQ 8.1) | Anti-nuclear binding with a perinuclear pattern (stronger in CSF than serum) | MRI: bilateral postinflammatory white matter lesions | Inconspicuous | Inconspicuous (0.40/min) |
| Pat. (7) | M | ~40 | Predominantly obsessional thoughts | None | ANA 1:200, elevated anti-TPO and TG abs | Pleocytosis (6 cells), BBB-dysfunction (AQ 14.3, TP 927 mg/l) | Borderline anti-myelin-binding in serum (+) | MRI: non-specific WM lesions | Volume loss in the OFC on both sides and in the left parietal cortex | Intermitted generalized slowing (0.62/min) |
| Pat. (8) | M | End 40 | Predominantly obsessional thoughts or ruminations | Rhabdomyolysis (CK max. 56870 U/l), latent hypothyroidism, arterial hypertension | Anti-streptolysine abs positive, elevated CH50 | Positive OCB in CSF | Strong anti-nuclear binding with a perinuclear pattern in CSF and serum |
MRI: inconspicuous FDG-PET slight asymmetry of association cortices metabolism (R < L) with a low frontotemporal emphasis (z-score −2) |
GM-Volume reduction occipital right, CSF-volume increase occipital left and in cerebellum |
Inconspicuous (0.11/min) |
| Pat. (9) | F | ~20 | Mixed obsessional thoughts and acts | Triple X syndrome | Elevated CRP (+) | Not investigated | Not investigated | MRI: small pinealis and pituitary pars intermedia cysts | GM-volume reduction orbitofrontal/frontal/ parietal/occipital on both sides, temporal right and central left | Intermitted generalized slowing (6.62/min) |
| Pat. (10) | F | Mid 20 | Predominantly compulsive acts | Turner syndrome, Hashimoto’s thyroiditis | Elevated anti-TG abs | Not investigated | Not investigated | MRI: inconspicuous | GM-Volume reduction orbitofrontal left and mesiotemporal left | Inconspicuous (0.11/min) |
ANA antinuclear antibodies, AQ albumin quotient, BBB blood–brain barrier, CK creatine kinase, CRP C-reactive protein, CSF cerebrospinal fluid, DNA deoxyribonucleic acid, EEG electroencephalogram, F female, FDG-PET fluorodeoxyglucose - positron emission tomography, GM gray matter, IRDA intermittent rhythmic delta activity, L left, M male, MRI Magnetic resonance imaging, OCB oligoclonal bands, OCD obsessive compulsive disorder, R right, TG thyroglobulin, TP total protein, TPO thyroid peroxidase.
aNeuropsychiatric lupus was diagnosed at an earlier stage.
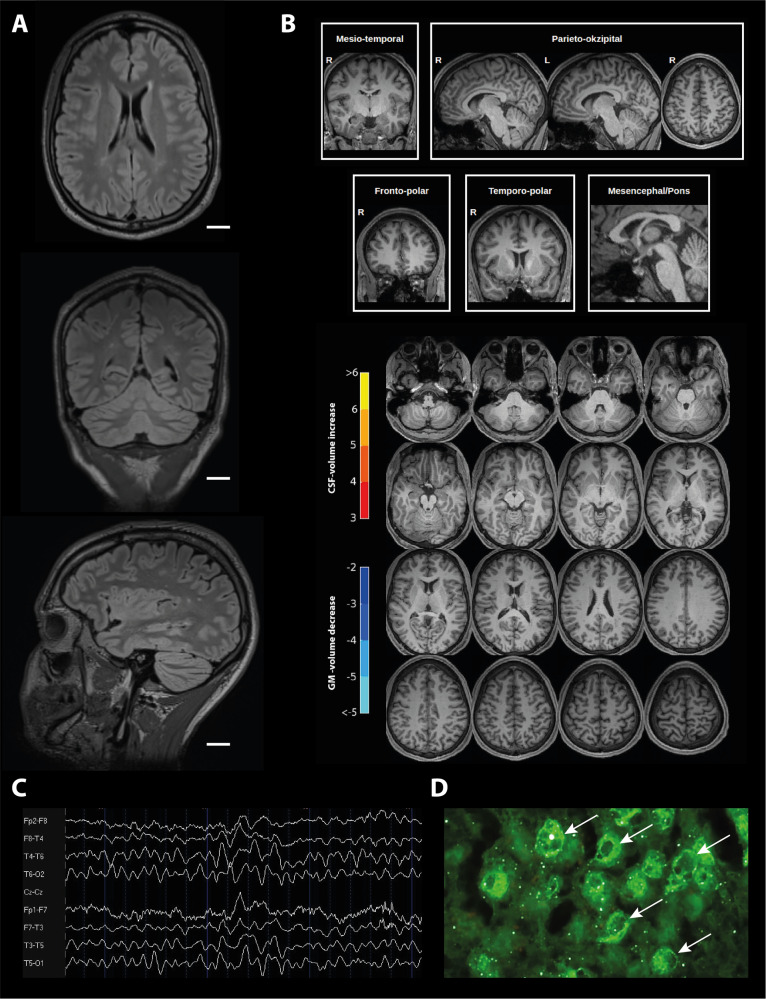
Fig. 3. Exemplary diagnostic findings as may be found in some patients with OCD.
A MRI with postinflammatory white matter lesions in FLAIR-sequences. B Automated Veobrain MRI analyses of the same patient shows normal findings without atrophic changes C EEG finding with intermittent generalized slowing. D Perinuclear staining (which were identified in serum and cerebrospinal fluid of some OCD patients) on unfixed murine brain slices (Prof. Prüß, Charité Berlin). FLAIR Fluid-attenuated inversion recovery, GM gray matter, FDG-PET fluorodeoxyglucose - positron emission tomography.
Discussion
The FDP-OCD screening detected probable or possible organic forms of OCD in 16%, as well as relevant diagnostic alterations in the overall patient sample.
Probable and possible organic OCD cases
Ten patients were considered to have organic OCD subtypes, whereby five patients (8%) were considered to have probable OCD (three patients with autoimmune causes and two patients with new genetic syndromes), and five patients (8%) were considered to have possible organic OCD (three with autoimmune causes and two patients with genetic syndromes).
Autoimmune-mediated OCD
Detection of autoimmune OCD is of particular importance because causal immunotherapies are available for treatment. According to recent international consensus criteria, autoimmune OCD is confirmed by the detection of specific neuronal antibodies in CSF and/or a response to immunotherapy [16]. This was the case for two patients in the present cohort (3%). Both patients had abnormalities in the immunological laboratory examinations and in the MRI; therefore, a CSF analysis was added. The first patient showed a specific pattern against granule cells that could result in glutamatergic dysregulation. This patient showed full remission under high-dose glucocorticoid treatment [34]. The second patient had specific antibodies against basal ganglia in combination with a volume reduction in the OFC, which could have induced a dysbalance of the cortico–striato–thalamo–cortical loops. This patient also showed significant improvement under glucocorticoid treatment [20]. A third patient had neuropsychiatric lupus erythematosus with isolated psychiatric manifestations (besides OCS also briefly psychotic symptoms) and clear diagnostic abnormalities with anti-nucleosome specificity and inflammatory CSF signals (pleocytosis, OCBs). This patient also received immunosuppressive therapy and initially showed clear improvement. These three cases (5%) impressively demonstrate the importance of an advanced diagnostic work-up and the potential of immunotherapy in some patients with autoimmune OCD (whereby placebo effects clearly cannot be excluded). As possible autoimmune OCD was considered one patient who had several borderline findings like slight pleocytosis in CSF, non-specific white-matter changes in MRI, borderline myelin-binding, and ANA, while another patient had conspicuous FDG-PET with positive OCBs in CSF and strong antinuclear binding in tissue-based assay. In addition, one patient had questionable Hashimoto’s encephalopathy [35]. Due to the borderline findings, immunotherapy was not initiated in these three cases. Currently, in clinical practice, decisions for immunotherapy are complex scenarios made on a case-by-case basis, especially in possible cases. Further research is needed to clarify if specific immunological causes are present in such cases and whether these patients profit from immunosuppressive or immunomodulatory therapies.
Genetic syndromes
Furthermore, genetic syndromes were assessed as possible and probable organic OCD. The duplication of chromosome 15q11-13 and Mayer–Rokitansky–Küster–Hauser syndrome in one patient and the Baraitser–Winter syndrome in another [36] were considered to be likely related to their OCS. Both patients showed other central nervous changes such as hearing loss, reduced IQ level, and matching MRI pathologies. Two patients with chromosome disorders involving the X chromosome (Turner syndrome and triple X syndrome) were assessed as having possible organic OCD [37]. Interestingly, all these patients showed orbitofrontal volume reduction in an automated MRI evaluation, which could be associated with dysfunction in the cortico–striato–thalamo–cortical loops [9]. Such well-defined genetic cases with brain involvement may also provide novel insights into OCD pathophysiology.
Diagnostic findings in the entire OCD patient group
Autoimmunity and antibodies
A high rate of endocrinological disease in 25% of cases was detected, with prominence of thyroid disorders with hypothyroidism in 13% and Hashimoto’s thyroiditis in 7%. Elevated anti-TPO in 19% and anti-TG antibodies in 13% were observed. Hereby elevated TPO antibodies were twice as common as in other reported control groups at 9.1% [38]. It has already been observed that OCD is more frequent in patients with Hashimoto’s thyroiditis [39] and other thyroid diseases [40], including papillary thyroid cancer [41]. The increased risk for Hashimoto’s thyroiditis in OCD was estimated to be 59% [42]. One patient was diagnosed with possible Hashimoto’s encephalopathy (cf. [35]). In such cases, therapeutic alternatives with immunotherapies might emerge [35].
However, other autoantibodies were also detected to an increased extent. Positive ANA findings were observed in 36% of the cases. In comparison, positive ANA findings of grades 1 to 4 have been found in only 16% of the general population in the United States of America [43]. However, only a few specific antibodies directed against extractable nuclear antigen (ENA) could be identified, with one patient having systemic lupus erythematosus with brain involvement. Prior data on ANA findings and OCS are limited to a few case presentations with suspected secondary OCD and positive ANAs [21, 44]. However, there is a known increased risk of any immune disease in OCD patients of 43%, with the highest risk of Sjögren’s syndrome at 94% [42]. Further research is needed to show whether there are specific ENA in OCD [33]. Well-characterized neuronal autoantibodies [45] could not be detected in the present cohort. In a subgroup of patients with suspected organic OCD, neuronal antibody binding using tissue-based assays on murine brain slices [29] was identified in two patients with a novel but specific pattern in CSF. This suggests that novel neuronal autoantibodies may play a role in a subgroup of patients with OCD. When considering organic causes of OCD, PANDAS is usually the first thing that comes to mind. Therefore, the investigation of streptococcal antibodies (anti-streptolysin and anti-streptococcal DNAseB) are of great interest in OCD. Elevated streptolysin antibodies were detected in 40% of patients in the cohort, which is more than in a healthy control group of Han Chinese (14%) [46]. Regarding the anti-streptococcal-DNAseB antibodies, the prevalence was comparable to the healthy adult Chinese population when applying the reference value of 480 IU/ml (6.4% in OCD patients of this cohort and 5.3% in healthy Chinese controls). Although streptococcal infections have been shown to increase the risk of OCD, it is unclear whether PANDAS or maybe unclassified milder forms are responsible [47]. In this cohort, none of the OCD patients with high anti-streptococcal antibody titers fulfilled the clinical criteria for PANDAS [48] in childhood and/or disease onset. Therefore, no therapeutic consequences followed from these findings.
Standard laboratory analysis and vitamin deficiencies
Increased serum calcium concentrations, which have previously been described in OCD [49], were noted. Interestingly, primary hyperparathyroidism with elevated calcium levels can present with OCS [50], and calcium-signaling pathways may play an important role in SSRI treatment response [51]. Elevated triglycerides were found in 25% of patients, which is significantly higher than in the general population (around 10%) [52]. In the literature to date, there are no known links between OCD severity and diet quality or nutrient intake [53]. In recent years, vitamin D has been discussed as a possible trigger and modifier of mental illness. Although most samples were collected in summer, 75% of OCD patients showed vitamin D deficiency at suboptimal levels. Reduced vitamin D levels have been described in children with OCD [54] or PANDAS [55], although more recent studies have found no significant differences in children with OCD [56]. In a recent meta-analysis of these studies, no association was found between OCD and vitamin D deficiency [57]. Nevertheless, vitamin D may have a disease-modifying effect on OCD [58]. Another vitamin, folic acid, was significantly decreased in 24% of the patients. In some studies, this was already reported for OCD [59]. Our patients with vitamin D and folic acid deficiencies were offered substitution therapy. A noteworthy reduction in vitamin B12 levels as reported in a meta-analysis [57] was not observed in this patient cohort.
MRI and EEG alterations
MRI showed abnormalities in 39% of patients, with 19% potentially relevant to OCD in the form of white-matter lesions due to post-inflammatory processes. While no atrophy was visually clearly apparent, automated analysis revealed either a decrease in gray-matter volume, an increase in CSF volume (indirect signs of discrete volume loss) or both in 45% of the examined MRIs. These findings do not necessarily indicate a loss of volume or atrophy, but may also be due to a neurodevelopmental process caused by e.g., genetic syndromes. Frequently, orbitofrontal or frontal areas in general were affected, which is consistent with alterations of the well-known cortico–striato–thalamo–cortical circuits in OCD [9]. The orbitofrontal cortex in particular showed increased functional/metabolic activity in functional MRI and PET studies, as well as a decreased density in postmortem studies in OCD [60–62]. However, parietal and occipital areas were also partially affected in the automated MRI analysis, which has also been previously reported for OCD and could possibly be associated with the impairment of the fronto–posterior tracts in OCD [9, 63]. Automated MRI approaches could have additional diagnostic value in the diagnostic work-up of patients with OCD. Evidence of epileptic activity was not found in the EEGs, and EEG slowing was relative rare (7%). No patient was thought to have an epileptic genesis, and signs for “paraepileptic changes”, that could potentially have therapeutic consequences [64], were also rare.
Limitations
Due to the retrospective approach in a naturalistic setting, not all results were available for all patients and only cases with suspected organic OCD received additional diagnostics including lumbar punctures and FGD-PET. In addition, the neuropsychological screening could have been broader. Altogether, the patient group could have been larger and in the absence of a control group, only comparisons with reference values and published prevalence of abnormalities in the general population were possible. Finally, the classification into probable or possible organic OCD forms was based mainly on preliminary criteria [16].
Conclusions
A relevant number of organic OCD forms (in 16%) were identified, which in 5% of all patients lead to successful treatment with immunotherapies. The frequent presence of autoantibodies such as anti-TPO or ANA further support the possible influences of autoimmune processes in OCD [16]. Further findings may well have modulatory effects on the course of the disease (e.g., substitution of folic acid deficiency). In addition, these findings might have a positive influence on the disease concept and self-image of affected patients and their relatives. The establishment of diagnostic regimens such as FDP-OCD should be evaluated in larger prospective and controlled studies. This could allow for a specific treatment of a small subgroup of patients with identified organic OCD reducing treatment resistance.
Supplementary information
Acknowledgements
DE and LTvE are members of the Immuno-NeuroPsychiatry Network of the European College of Neuropsychopharmacology (ECNP). MAS is a member of the Obsessive Compulsive and Related Disorders (OCRN) network of the ECNP. KR was funded by the Berta-Ottenstein-Programme for Clinician Scientists, Faculty of Medicine, University of Freiburg. LTvE and KR were supported by the KKS Foundation.
Author contributions
All authors were critically involved in the theoretical discussion and composition of the manuscript. All authors read and approved the final version of the manuscript.
Funding
The article processing charge was funded by the Baden-Wuerttemberg Ministry of Science, Research and Art and the University of Freiburg in the funding programme Open Access Publishing. Open Access funding enabled and organized by Projekt DEAL.
Data availability
All necessary data can be found in the article.
Competing interests
HU: Speaker fees from Bayer AG, Bracco, Eisai, Stryker, and UCB Pharma. KD: Steering Committee Neurosciences, Janssen. Speaker fees from Janssen. LTvE: Advisory boards, lectures, or travel grants within the last three years: Roche, Eli Lilly, Janssen-Cilag, Novartis, Shire, UCB, GSK, Servier, Janssen and Cyberonics. All other authors have no potential conflicts of interest to declare.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s41398-023-02368-8.
References
- 1.Adam Y, Meinlschmidt G, Gloster AT, Lieb R. Obsessive-compulsive disorder in the community: 12-month prevalence, comorbidity and impairment. Soc Psychiatry Psychiatr Epidemiol. 2012;47:339–49. doi: 10.1007/s00127-010-0337-5. [DOI] [PubMed] [Google Scholar]
- 2.Angst J, Gamma A, Endrass J, Goodwin R, Ajdacic V, Eich D, et al. Obsessive-compulsive severity spectrum in the community: prevalence, comorbidity, and course. Eur Arch Psychiatry Clin Neurosci. 2004;254:156–64. doi: 10.1007/s00406-004-0459-4. [DOI] [PubMed] [Google Scholar]
- 3.Jacobi F, Höfler M, Strehle J, Mack S, Gerschler A, Scholl L, et al. Psychische Störungen inder Allgemeinbevölkerung: Studie zur Gesundheit Erwachsener in Deutschland und ihrZusatzmodul Psychische Gesundheit (DEGS1-MH) Nervenarzt. 2014;85:77–87. doi: 10.1007/s00115-013-3961-y. [DOI] [PubMed] [Google Scholar]
- 4.Subramaniam M, Soh P, Vaingankar JA, Picco L, Chong SA. Quality of life in obsessive-compulsive disorder: impact of the disorder and of treatment. CNS Drugs. 2013;27:367–83. doi: 10.1007/s40263-013-0056-z. [DOI] [PubMed] [Google Scholar]
- 5.Robbins TW, Vaghi MM, Banca P. Obsessive-compulsive disorder: puzzles and prospects. Neuron. 2019;102:27–47. doi: 10.1016/j.neuron.2019.01.046. [DOI] [PubMed] [Google Scholar]
- 6.Gerentes M, Pelissolo A, Rajagopal K, Tamouza R, Hamdani N. Obsessive-compulsive disorder: autoimmunity and neuroinflammation. Curr Psychiatry Rep. 2019;21:78. doi: 10.1007/s11920-019-1062-8. [DOI] [PubMed] [Google Scholar]
- 7.Real E, Labad J, Alonso P, Segalàs C, Jiménez-Murcia S, Bueno B, et al. Stressful life events at onset of obsessive-compulsive disorder are associated with a distinct clinical pattern. Depress Anxiety. 2011;28:367–76. doi: 10.1002/da.20792. [DOI] [PubMed] [Google Scholar]
- 8.Pauls DL, Abramovitch A, Rauch SL, Geller DA. Obsessive-compulsive disorder: an integrative genetic and neurobiological perspective. Nat Rev Neurosci. 2014;15:410–24. doi: 10.1038/nrn3746. [DOI] [PubMed] [Google Scholar]
- 9.Stein DJ, Costa DLC, Lochner C, Miguel EC, Reddy YCJ, Shavitt RG, et al. Obsessive-compulsive disorder. Nat Rev Dis Prim. 2019;5:52. doi: 10.1038/s41572-019-0102-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schiele MA, Thiel C, Deckert J, Zaudig M, Berberich G, Domschke K. Monoamine oxidase A hypomethylation in obsessive-compulsive disorder: reversibility by successful psychotherapy? Int J Neuropsychopharmacol. 2020;23:319–23. doi: 10.1093/ijnp/pyaa016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Endres D, Domschke K, Schiele MA. Neurobiology of obsessive-compulsive disorder. Nervenarzt. 2022;93:670–7. doi: 10.1007/s00115-022-01331-0. [DOI] [PubMed] [Google Scholar]
- 12.Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Publishing; 2013.
- 13.World Health Organization. International statistical classification of diseases and related health problems (11th ed.); 2019/2021 https://icd.who.int/browse11.
- 14.Gamazo-Garrán P, Soutullo CA, Ortuño F. Obsessive-compulsive disorder secondary to brain dysgerminoma in an adolescent boy: a positron emission tomography case report. J Child Adolesc Psychopharmacol. 2002;12:259–63. doi: 10.1089/104454602760386950. [DOI] [PubMed] [Google Scholar]
- 15.Hugo F, van Heerden B, Zungu-Dirwayi N, Stein DJ. Functional brain imaging in obsessive-compulsive disorder secondary to neurological lesions. Depress Anxiety. 1999;10:129–36. doi: 10.1002/(SICI)1520-6394(1999)10:3<129::AID-DA7>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 16.Endres D, Pollak TA, Bechter K, Denzel D, Pitsch K, Nickel K, et al. Immunological causes of obsessive-compulsive disorder: is it time for the concept of an “autoimmune OCD” subtype? Transl Psychiatry. 2022;12:5. doi: 10.1038/s41398-021-01700-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Swedo SE. Sydenham’s chorea. A model for childhood autoimmune neuropsychiatric disorders. JAMA. 1994;272:1788–91. doi: 10.1001/jama.1994.03520220082035. [DOI] [PubMed] [Google Scholar]
- 18.Swedo SE, Leonard HL, Mittleman BB, Allen AJ, Rapoport JL, Dow SP, et al. Identification of children with pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections by a marker associated with rheumatic fever. Am J Psychiatry. 1997;154:110–2. doi: 10.1176/ajp.154.1.110. [DOI] [PubMed] [Google Scholar]
- 19.Esposito S, Bianchini S, Baggi E, Fattizzo M, Rigante D. Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections: an overview. Eur J Clin Microbiol Infect Dis. 2014;33:2105–9. doi: 10.1007/s10096-014-2185-9. [DOI] [PubMed] [Google Scholar]
- 20.Endres D, Mertens L, Berger B, Reisert M, Runge K, Nickel K, et al. Autoimmune Obsessive-Compulsive Disorder with Novel Anti-Basal Ganglia Antibodies. Psychother Psychosom. 2022;91:214–6. doi: 10.1159/000522136. [DOI] [PubMed] [Google Scholar]
- 21.Lüngen EM, Maier V, Venhoff N, Salzer U, Dersch R, Berger B, et al. Systemic lupus erythematosus with isolated psychiatric symptoms and antinuclear antibody detection in the cerebrospinal fluid. Front Psychiatry. 2019;10:391. doi: 10.3389/fpsyt.2019.00226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang L-Y, Chen S-F, Chiang J-H, Hsu C-Y, Shen Y-C. Systemic autoimmune diseases are associated with an increased risk of obsessive-compulsive disorder: a nationwide population-based cohort study. Soc Psychiatry Psychiatr Epidemiol. 2019;54:507–16. doi: 10.1007/s00127-018-1622-y. [DOI] [PubMed] [Google Scholar]
- 23.Endres D, Matysik M, Feige B, Venhoff N, Schweizer T, Michel M, et al. Diagnosing organic causes of schizophrenia spectrum disorders: findings from a one-year cohort of the Freiburg diagnostic protocol in psychosis (FDPP) Diagnostics. 2020;10:691. doi: 10.3390/diagnostics10090691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.International statistical classification of diseases and related health problems (Geneva, 2004). [PubMed]
- 25.Endres D, Meixensberger S, Dersch R, Feige B, Stich O, Venhoff N, et al. Cerebrospinal fluid, antineuronal autoantibody, EEG, and MRI findings from 992 patients with schizophreniform and affective psychosis. Transl Psychiatry. 2020;10:279. doi: 10.1038/s41398-020-00967-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Endres D, Zedtwitz K, von, Matteit I, Bünger I, Foverskov-Rasmussen H, Runge K, et al. Spectrum of novel anti-central nervous system autoantibodies in the cerebrospinal fluid of 119 patients with schizophreniform and affective disorders. Biol Psychiatry. 2022;92:261–74. doi: 10.1016/j.biopsych.2022.02.010. [DOI] [PubMed] [Google Scholar]
- 27.Venhoff N, Thiel J, Rizzi M, Venhoff A, Rauer S, Endres D, et al. The MRZ-reaction and specific autoantibody detection for differentiation of ANA-positive multiple sclerosis from rheumatic diseases with cerebral involvement. Front Immunol. 2019;10:514. doi: 10.3389/fimmu.2019.00514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pankratz B, Zedtwitz K von, Runge K, Denzel D, Nickel K, Schlump A, et al. Cerebrospinal fluid findings in adult patients with obsessive-compulsive disorder: a retrospective analysis of 54 samples. World J Biol Psychiatry. 2022;1–26 10.1080/15622975.2022.2104457. [DOI] [PubMed]
- 29.Kreye J, Reincke SM, Kornau H-C, Sánchez-Sendin E, Corman VM, Liu H, et al. A therapeutic non-self-reactive SARS-CoV-2 antibody protects from lung pathology in a COVID-19 hamster model. Cell. 2020;183:1058–1069.e19. doi: 10.1016/j.cell.2020.09.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kreye J, Wright SK, van Casteren A, Stöffler L, Machule M-L, Reincke SM, et al. Encephalitis patient-derived monoclonal GABAA receptor antibodies cause epileptic seizures. J Exp Med. 2021;218:e20210012. doi: 10.1084/jem.20210012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Endres D, Maier S, Feige B, Mokhtar NB, Nickel K, Goll P, et al. Increased rates of intermittent rhythmic delta and theta activity in the electroencephalographies of adult patients with attention-deficit hyperactivity disorder. Epilepsy Behav. 2017;75:60–65. doi: 10.1016/j.yebeh.2017.06.039. [DOI] [PubMed] [Google Scholar]
- 32.Endres D, Pankratz B, Thiem S, Runge K, Schlump A, Feige B, et al. Novel anti-cytoplasmic antibodies in cerebrospinal fluid and serum of patients with chronic severe mental disorders. World J Biol Psychiatry. 2022;23:794–801. doi: 10.1080/15622975.2022.2042599. [DOI] [PubMed] [Google Scholar]
- 33.Endres D, Schiele MA, Frye BC, Schlump A, Feige B, Nickel K, et al. Obsessive-compulsive disorder with inflammatory cerebrospinal fluid changes and intrathecal antinuclear antibody staining. Biol Psychiatry. 2022. 10.1016/j.biopsych.2022.06.037. [DOI] [PubMed]
- 34.Endres D, Pankratz B, Robinson T, Pitsch K, Göbel T, Runge K, et al. Autoimmune obsessive-compulsive disorder with novel anti-CNS autoantibodies in cerebrospinal fluid. Mol Psychiatry. 2022. 10.1038/s41380-022-01688-3. [DOI] [PMC free article] [PubMed]
- 35.Graus F, Titulaer MJ, Balu R, Benseler S, Bien CG, Cellucci T, et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016;15:391–404. doi: 10.1016/S1474-4422(15)00401-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Göbel T, Berninger L, Schlump A, Feige B, Runge K, Nickel K, et al. Obsessive-compulsive symptoms in ACTG1-associated Baraitser-Winter cerebrofrontofacial syndrome. J Neural Transm. 2022;129:1387–91. doi: 10.1007/s00702-022-02544-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Matteit I, Schlump A, Reisert M, Zedtwitz KV, Runge K, Nickel K, et al. Obsessive-compulsive symptoms in two patients with chromosomal disorders involving the X chromosome. World J Biol Psychiatry 2022;1–6 10.1080/15622975.2022.2147997. [DOI] [PubMed]
- 38.Snijders GJLJ, Witte LD, de, van den Berk D, van der Laan C, Regeer E, Begemann MJH, et al. No association between anti-thyroidperoxidase antibodies and bipolar disorder: a study in the Dutch Bipolar Cohort and a meta-analysis. Psychoneuroendocrinology. 2020;112:104518. doi: 10.1016/j.psyneuen.2019.104518. [DOI] [PubMed] [Google Scholar]
- 39.Giynas Ayhan M, Uguz F, Askin R, Gonen MS. The prevalence of depression and anxiety disorders in patients with euthyroid Hashimoto’s thyroiditis: a comparative study. Gen Hosp Psychiatry. 2014;36:95–98. doi: 10.1016/j.genhosppsych.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 40.Placidi GP, Boldrini M, Patronelli A, Fiore E, Chiovato L, Perugi G, et al. Prevalence of psychiatric disorders in thyroid diseased patients. Neuropsychobiology. 1998;38:222–5. doi: 10.1159/000026545. [DOI] [PubMed] [Google Scholar]
- 41.Çayköylü A, Uğurlu GK, Yenilmez DO, Çayköylü HH, Uğurlu M. Subthreshold obsessive-compulsive symptoms in 3 patients with papillary thyroid carcinoma. Prim Care Companion CNS Disord. 2020;22:19l02463. doi: 10.4088/PCC.19l02463. [DOI] [PubMed] [Google Scholar]
- 42.Mataix-Cols D, Frans E, Pérez-Vigil A, Kuja-Halkola R, Gromark C, Isomura K, et al. A total-population multigenerational family clustering study of autoimmune diseases in obsessive-compulsive disorder and Tourette’s/chronic tic disorders. Mol Psychiatry. 2018;23:1652–8. doi: 10.1038/mp.2017.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dinse GE, Parks CG, Weinberg CR, Co CA, Wilkerson J, Zeldin DC, et al. Increasing prevalence of antinuclear antibodies in the United States. Arthritis Rheumatol. 2020;72:1026–35. doi: 10.1002/art.41214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Freire de Carvalho J, Machado Ribeiro F. Sjögren syndrome associated with obsessive-compulsive disorder. Eur Rev Med Pharmacol Sci. 2020;24:11801–3. doi: 10.26355/eurrev_202011_23836. [DOI] [PubMed] [Google Scholar]
- 45.Prüss H. Autoantibodies in neurological disease. Nat Rev Immunol. 2021. 10.1038/s41577-021-00543-w. [DOI] [PMC free article] [PubMed]
- 46.Ding Q, Li J, Xiao F, Zhang C, Dong X, Han F. Anti-streptococcal antibodies in Chinese patients with type-1 narcolepsy. Sleep Med. 2020;72:37–40. doi: 10.1016/j.sleep.2020.03.019. [DOI] [PubMed] [Google Scholar]
- 47.Orlovska S, Vestergaard CH, Bech BH, Nordentoft M, Vestergaard M, Benros ME. Association of streptococcal throat infection with mental disorders: testing key aspects of the PANDAS hypothesis in a nationwide study. JAMA Psychiatry. 2017;74:740–6. doi: 10.1001/jamapsychiatry.2017.0995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Swedo SE, Leonard HL, Garvey M, Mittleman B, Allen AJ, Perlmutter S, et al. Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections: clinical description of the first 50 cases. Am J Psychiatry. 1998;155:264–71. doi: 10.1176/ajp.155.2.264. [DOI] [PubMed] [Google Scholar]
- 49.Shohag H, Ullah A, Qusar S, Rahman M, Hasnat A. Alterations of serum zinc, copper, manganese, iron, calcium, and magnesium concentrations and the complexity of interelement relations in patients with obsessive-compulsive disorder. Biol Trace Elem Res. 2012;148:275–80. doi: 10.1007/s12011-012-9371-3. [DOI] [PubMed] [Google Scholar]
- 50.Chiba Y, Satoh K, Ueda S, Kanazawa N, Tamura Y, Horiuchi T. Marked improvement of psychiatric symptoms after parathyroidectomy in elderly primary hyperparathyroidism. Endocr J. 2007;54:379–83. doi: 10.1507/endocrj.K06-152. [DOI] [PubMed] [Google Scholar]
- 51.Umehara H, Numata S, Tajima A, Nishi A, Nakataki M, Imoto I, et al. Calcium signaling pathway is associated with the long-term clinical response to selective serotonin reuptake inhibitors (SSRI) and SSRI with antipsychotics in patients with obsessive-compulsive disorder. PLoS ONE. 2016;11:e0157232. doi: 10.1371/journal.pone.0157232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Laufs U, Parhofer KG, Ginsberg HN, Hegele RA. Clinical review on triglycerides. Eur Heart J. 2020;41:99–109c. doi: 10.1093/eurheartj/ehz785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nguyen TP, Cribb L, Ng CH, Byrne GJ, Castle D, Brakoulias V, et al. Dietary quality and nutrient intake in adults with obsessive-compulsive disorder. BJPsych Open. 2021;7:e218. doi: 10.1192/bjo.2021.1039. [DOI] [Google Scholar]
- 54.Esnafoğlu E, Yaman E. Vitamin B12, folic acid, homocysteine and vitamin D levels in children and adolescents with obsessive compulsive disorder. Psychiatry Res. 2017;254:232–7. doi: 10.1016/j.psychres.2017.04.032. [DOI] [PubMed] [Google Scholar]
- 55.Çelik G, TAŞ D, Tahiroğlu A, AVCI A, YÜKSEL B, ÇAM P. Vitamin D deficiency in obsessive-compulsive disorder patients with pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections: a case control study. Noro Psikiyatr Ars. 2016;53:33–37. doi: 10.5152/npa.2015.8763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yazici KU, Percinel Yazici I, Ustundag B. Vitamin D levels in children and adolescents with obsessive compulsive disorder. Nord J Psychiatry. 2018;72:173–8. doi: 10.1080/08039488.2017.1406985. [DOI] [PubMed] [Google Scholar]
- 57.Balandeh E, Karimian M, Behjati M, Mohammadi AH. Serum vitamins and homocysteine levels in obsessive-compulsive disorder: a systematic review and meta-analysis. Neuropsychobiology. 2021;80:502–15. doi: 10.1159/000514075. [DOI] [PubMed] [Google Scholar]
- 58.Di Michele F, Siracusano A, Talamo A, Niolu C. N-acetyl cysteine and vitamin D supplementation in treatment resistant obsessive-compulsive disorder patients: a general review. Curr Pharm Des. 2018;24:1832–8. doi: 10.2174/1381612824666180417124919. [DOI] [PubMed] [Google Scholar]
- 59.Atmaca M, Tezcan E, Kuloglu M, Kirtas O, Ustundag B. Serum folate and homocysteine levels in patients with obsessive-compulsive disorder. Psychiatry Clin Neurosci. 2005;59:616–20. doi: 10.1111/j.1440-1819.2005.01425.x. [DOI] [PubMed] [Google Scholar]
- 60.Oliveira KCD, Grinberg LT, Hoexter MQ, Brentani H, Suemoto CK, Nery FG, et al. Layer-specific reduced neuronal density in the orbitofrontal cortex of older adults with obsessive-compulsive disorder. Brain Struct Funct. 2019;224:191–203. doi: 10.1007/s00429-018-1752-8. [DOI] [PubMed] [Google Scholar]
- 61.Nakao T, Okada K, Kanba S. Neurobiological model of obsessive-compulsive disorder: evidence from recent neuropsychological and neuroimaging findings. Psychiatry Clin Neurosci. 2014;68:587–605. doi: 10.1111/pcn.12195. [DOI] [PubMed] [Google Scholar]
- 62.Caykoylu A, Kabadayi Sahin E, Ugurlu M. Could the thyroid gland dominate the brain in obsessive-compulsive disorder? Neuroendocrinology. 2022. 10.1159/000524627. [DOI] [PubMed]
- 63.Piras F, Piras F, Abe Y, Agarwal SM, Anticevic A, Ameis S,, et al. White matter microstructure and its relation to clinical features of obsessive-compulsive disorder: findings from the ENIGMA OCD Working Group. Transl Psychiatry. 2021;11:173. doi: 10.1038/s41398-021-01276-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tebartz van Elst L, Krishnamoorthy ES, Schulze-Bonhage A, Altenmüller D-M, Richter H, Ebert D, et al. Local area network inhibition: a model of a potentially important paraepileptic pathomechanism in neuropsychiatric disorders. Epilepsy Behav. 2011;22:231–9. doi: 10.1016/j.yebeh.2011.06.016. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All necessary data can be found in the article.