Abstract
Although mpox is rare among children in the United States, pediatric cases are being reported during the 2022 multinational mpox outbreak. Vaccines and antiviral medications developed for other orthopoxviruses have recently become widely used to prevent and treat mpox in both children and adults in the United States. Although scientific literature regarding mpox in children and adolescents is scant, prior case reports can provide valuable information about the clinical features and potential complications of untreated clade II mpox in these age groups. In this review, we summarize the epidemiology and clinical features of mpox in children and adolescents and provide recommendations for clinicians regarding its diagnosis, management, and prevention. Robust, dedicated surveillance of pediatric exposures and cases in the current outbreak, including the use of vaccines and `therapeutics, are needed to guide clinical management and public health strategies.
In May 2022, an unprecedented global outbreak of mpox (formerly known as monkeypox) was recognized; since the beginning of the outbreak, over 80 000 cases have been identified in nonendemic countries including over 29 000 cases in the United States. Although the current multinational outbreak has primarily affected adults, there have been over 500 laboratory confirmed cases in children and adolescents worldwide,1 and many more children and adolescents exposed to mpox virus (MPXV).2
There are 2 well-described clades of MPXV with differing clinical features: clade I (formerly known as Congo Basin Clade) and clade II (formerly known as West African Clade). The majority of published literature, including most pediatric case reports, describes infection with clade I MPXV, which has been associated with greater severity and a higher case fatality rate than clade II.3 Because of its relevance to the current outbreak, this review emphasizes outbreak reports and pediatric case reports of infection with clade II MPXV.
As the outbreak evolves, pediatricians and other healthcare providers caring for children and adolescents should be prepared to recognize, diagnose, and treat mpox and its complications and assist with prevention efforts. This review article summarizes scientific literature on mpox in children and adolescents, provides recommendations for clinicians on diagnosis, management, and prevention of mpox, and discusses current research gaps.
EPIDEMIOLOGY
MPXV is an orthopoxvirus endemic to West and Central Africa that can infect humans and animals. Although the virus was originally named “monkeypox” following its identification in captive primates, small ground rodents are the most likely reservoir species. MPXV is most often transmitted through close skin-to-skin contact or mucosal contact with lesions, and less commonly, is believed to spread through respiratory secretions or transplacentally.4 People are most commonly infected through contact with infected humans or animals; fomite-facilitated transmission, such as through linens, towels, or other household items, can also occur.5 The incubation period for MPXV is 3 to 17 days.6 The clinical manifestations and severity of disease can vary depending on the route of acquisition.7
The first documented human case of mpox was reported in 1970 in the Democratic Republic of Congo.8 During 1970 and 1971, 6 cases were reported in 3 West African countries; 5 cases were among children.8 Following the discovery of mpox as a zoonosis that can infect humans, subsequent cases were reported in Central Africa during the 1970s and 1980s.9–12 Most cases were sporadic or occurred in small clusters following a common animal exposure, and children constituted a majority of cases.9,12,13 It was originally thought that there was limited capacity for extended person-to-person spread.14 More recently, during a 2003 outbreak of clade II MPXV in the United States, children and adolescents comprised 26% of confirmed or probable cases (12 of 47); illness was associated with exposure to pet prairie dogs that had been cohoused with imported small mammals from Africa.7
Smallpox vaccination provides crossprotection against mpox. As decades have elapsed since routine smallpox vaccination ended in 1972, adults have been rendered increasingly susceptible to MPXV; the median age of documented cases increased from 4 years in the 1970s to 21 years in the 2010s.3,15 It is unknown whether there are age-related physiologic differences in susceptibility to MPXV independent of vaccination status.
In contrast to prior outbreaks, children and adolescents less than 18 years of age have accounted for 0.3% of cases to date in the United States during the ongoing 2022 outbreak.16 In addition to lack of immunity among most adults, the relatively low proportion of MPXV infections among children in 2022 is likely attributable to a substantial shift in transmission: mpox is now spreading predominantly (though not exclusively) through close, intimate human-to-human contact. In particular, it has proliferated in sexual networks of gay, bisexual, and other men who have sex with men.17 Therefore, it is essential that pediatricians remain alert to the possibility of mpox when caring for adolescent or young adult males who have sex with male partners, though mpox can affect people of all ages, gender identities, and sexual orientations.
CLINICAL FEATURES
Mpox is characterized by a rash with lesions that are firm or rubbery, well-circumscribed, deep-seated, and often umbilicated in later stages. The rash progresses through macular, papular, vesicular, and pustular stages before scabbing over and desquamation (Fig 1).6 The rash can be localized or diffuse, can involve the palms of hands and soles of feet, and can present in different areas of the body at different times. Lesions are often painful or pruritic. Fever, chills, myalgias, or malaise may occur at any time during the illness. Lymphadenopathy is also commonly observed.
FIGURE 1.
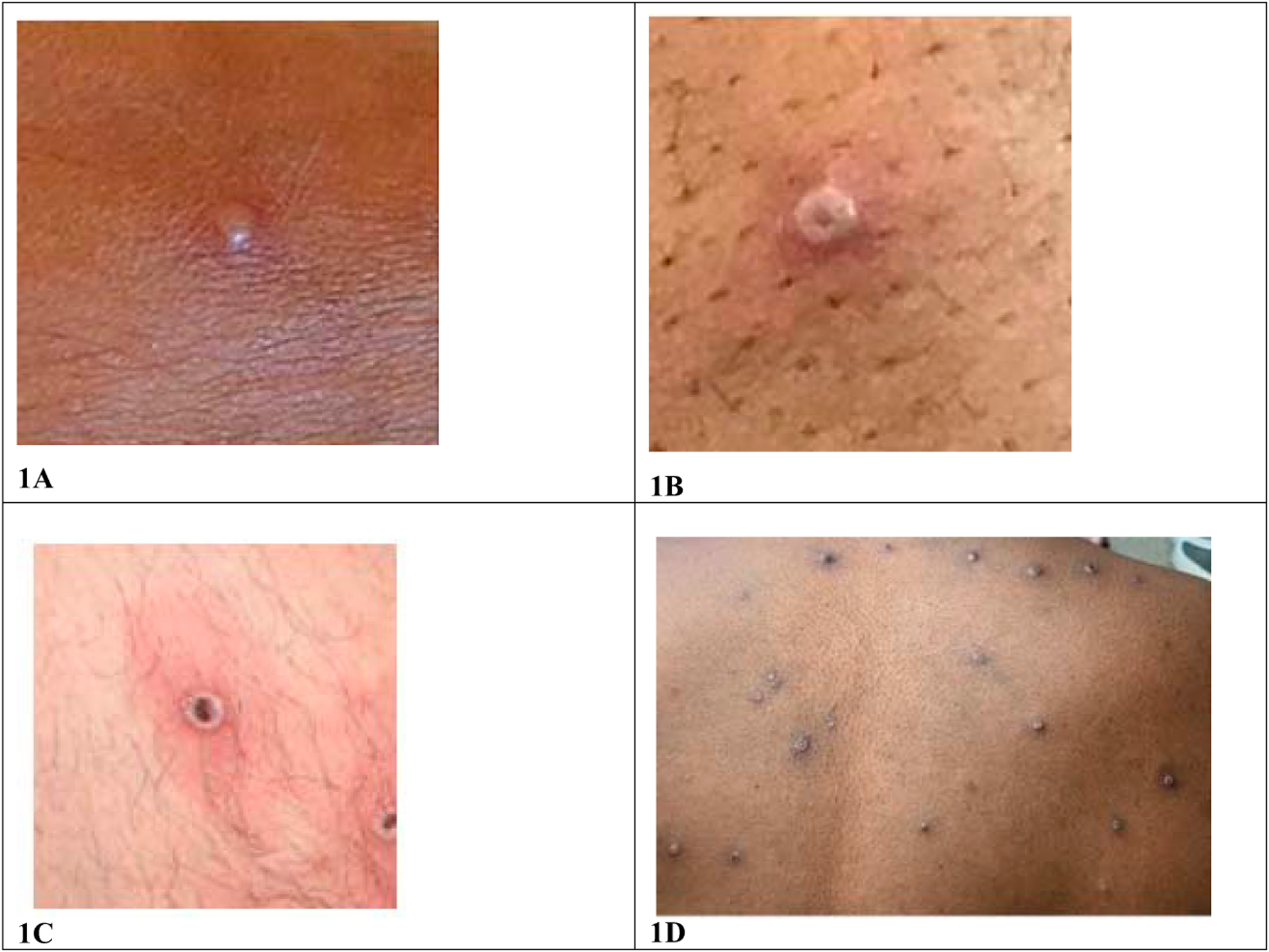
Mpox photos. Clinical photographs depicting an early vesicle (1A), umbilicated pustule (1B), ulcerated lesion (1C), and diffuse pustular rash (1D). 1A, 1B, 1C image credit: UK Health Security Agency. 1D image credit: NHS England High Consequence Infectious Diseases Network.
The severity, complications, and case fatality ratios (CFR) of mpox may differ in children compared with adults. Children are known to be at risk for severe complications from other orthopoxviruses, including variola virus and vaccinia virus.18–21 Historical reports of the severity of clade I mpox in children and adolescents suggest greater severity in children. Outcome data from a review of mpox cases caused by clade I MPXV in the Democratic Republic of Congo during 1980 to 1985 demonstrated greater severity in children less than 8 years of age, and particularly those ≤4 years of age, with a CFR of up to 15% for this group.11 However, a later analysis suggested that the mortality rate attributed to clade I mpox in the Democratic Republic of Congo is lower than previously reported because of likely underreporting of cases and misattribution of death from mpox.22,23 Ultimately, of 92 possible cases included in the latter analysis, 3 deaths occurred that were attributed to mpox; all 3 occurred in patients under 3 years of age.
Clinical Descriptions of Clade II Mpox Before 2022 Outbreak
Detailed descriptions of clade II mpox, its complications, and severity in children and adolescents are limited. For this review of clade II mpox before the current outbreak, we performed a literature search in multiple databases to identify articles in English that included a pediatric age-related term (eg, “NICU,” “preschool,” or “teenager”) and a term related to mpox or mpox vaccines or therapeutics (eg, “monkeypox,” “MPXV,” “JYNNEOS,” or “tecovirimat”). We summarized articles containing individual case-level data for patients less than 18 years of age with confirmed or probable mpox (Table 1). Cases were limited to those caused by clade II MPXV, determined by molecular genotyping or inferred based on location in West Africa. We included cases from outbreaks during 1970–1979 in Nigeria, Sierra Leone, Ivory Coast, and Liberia, the 2003 outbreak in the United States, an outbreak in Nigeria that began in 2017, and 1 additional 2019 case from Sierra Leone. These data are supplemented with previously unpublished case data from the 2003 US outbreak, provided by the US Centers for Disease Control and Prevention (CDC).
TABLE 1.
Demographic Characteristics, Clinical Features, and Outcomes of Patients <18 Years of Age With Clade II Mpox
| Country, Exposure, Year | Case Number | Age and Sex | Rash Characteristics | Additional Clinical Features | Hospitalization, Duration in Days | Vaccination Status | Laboratory Testing Results; Mpox Case Designationa | Outcome |
|---|---|---|---|---|---|---|---|---|
| Liberia, outbreak cause unknown, 1970b | 18,12,13 | 4 y M | Description: vesicles, 10 lesions; location: NA; distribution: generalized | Fever, mild severityc | Unknown | None | Serology +; Probable | Survived |
| Liberia, outbreak cause unknown, 1970b | 28,12,13 | 4 y F | Description: pustules, vesicles, <25 lesions; location: palms, soles, diffuse; distribution: generalized | Fever, sore throat, malaise, moderate severityc | Yes, duration unknown | None | Lesion-derived specimens positive for poxvirus by: EM, AG, CAM; Confirmed | Survived |
| Liberia, outbreak cause unknown, 1970b | 28,12,13 | 6 y F | Description: vesicles, 10 lesions; location: NA; distribution: generalized | Fever, mild severityc | Unknown | None | Serology +; Probable | Survived |
| Liberia, outbreak cause unknown, 1970b | 48,12,13 | 9 y M | Description: vesicles, pustules, >25 but <100 lesions; location: cornea, face, palms, arms or hands, trunk, back, soles, legs or feet, buttocks; distribution: generalized | Generalized facial swelling, tender submaxillary mass and abscess, corneal scarring, moderate severityc | Unknown | None | Lesion-derived specimens positive for poxvirus by: EM, CAM; serology +; Confirmed | Survived |
| Nigeria, outbreak cause unknown, 1971b | 58,12,13 | 4 y F | Description: macules, papules, pustules, >100 lesions; location: face, head or neck, palms, arms or hands, trunk, back, soles, legs or feet; distribution: generalized, centripetal | Fever, malaise, headache, sweating, prostration, toxic appearance, severe severityc | Yes, length unknown | None | Lesion-derived specimens positive for poxvirus by: EM, AG, CAM; serology +; Confirmed | Survived |
| Ivory Coast, outbreak cause unknown, 1971b | 612,13 | 5 y M | Description: >25 but <100 lesions; location: NA; description: NA | Moderate severityc | Unknown | None | Serology +; Probable | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 7 | 17 y M | Description: papules, vesicles, pustules, target lesions, desquamation, ulcers, eschar; location: head or neck, palms, arms or hands, trunk, legs or feet; distribution: generalized | Fever, sweats, chills, myalgia, headache, ear pain, sore throat, cervical lymphadenopathy, cough | Yes, 2 d | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 8 | 14 y M | Description: papules, vesicles, pustules, umbilicated; location: mucosa, mouth or pharynx, head or neck, palms, arms or hands, trunk, back, legs or feet; distribution: generalized, centrifugal | Fever, sweats, myalgia, headache, sore throat, cervical and inguinal lymphadenopathy, chest pain | No | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 966,67 | 10 y F | Description: vesicles, pustules, umbilicated, desquamation, dry or flaking; location: conjunctiva, mucosa or tongue, mouth or pharynx, head or neck, palms, arms or hands, trunk, back, soles, legs or feet; distribution: generalized | Fever, myalgia, nausea or vomiting, sore throat, cervical lymphadenopathy, retropharyngeal abscess that led to shortness of breath, hypoxia, tracheal airway compromise | Yes, 6 d | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 10 | 11 y M | Description: papules, vesicles, pustules, umbilicated; location: palms, arms or hands, trunk, back; distribution: generalized | None | No | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 11 | 12 y M | Description: NA; location: NA; description: NA | Unknown | Unknown | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 1266,68 | 6 y F | Description: papules, vesicles, pustules, umbilicated, dry or flaking; location: face, head or neck, palms, arms or hands, trunk, back, soles, legs or feet; distribution: generalized, centrifugal | Fever, chills, malaise, myalgia, back pain, headache, diarrhea, sore throat, tonsillar enlargement, cough, difficulty swallowing, decreased appetite, shortness of breath, cervical lymphadenopathy, respiratory distress, hypoxia, nuchal rigidity, encephalitis leading to altered mental status, lethargy, seizures, coma | Yes, 16 d | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 13 | 3 y M | Description: NA; location: NA; distribution: NA | Unknown | Unknown | None | IgG NP, IgM NP, PCR NP; Probable | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 14 | 8 y M | Description: NA; location: NA; distribution: NA | Unknown | Unknown | None | IgG NP, IgM NP, PCR NP; Probable | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 15 | 12 y M | Description: NA; location: NA; distribution: NA | Unknown | Unknown | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 16 | 4 y F | Description: NA; location: NA; distribution: NA | Unknown | Unknown | None | IgG NP, IgM NP, PCR NP; Probable | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 17 | 9 y F | Description: papules, vesicles, pustules, umbilicated, crusting; location: trunk, back, legs or feet; distribution: generalized, centripetal | Fever, headache, sore throat, cervical lymphadenopathy | No | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| United States, outbreak from prairie dog exposure, 2003d | 18 | 12 y M | Description: erythematous, papules, vesicles, pruritic; location: NA; distribution: NA | Headache, myalgias, lymphadenopathy, exudative pharyngitis; Comorbid condition: hemophilia A | No | Dryvax 24 d after initial exposure (continual exposure for 3 wks); symptoms began 7 d after vaccination | IgG +, IgM +, PCR +; Confirmed | Survived |
| Sierra Leone, case from wild animal exposure, 2014 | 1969 | 11 mo U | Description: papules, vesicles, pustules, umbilicated, pruritic; location: mouth, oral mucosa, face, palms, trunk, back, genital area; distribution: generalized | Sweats, fever, chills, vomiting, decreased appetite, cough | Yes, length unknown | None | IgG +, IgM +, PCR +; Confirmed | Survived |
| Nigeria, outbreak from monkey exposure, 2017 | 2070 | 11 y M | Description: papules, pustules, umbilicated, crusting, ulcers; location: oral or nasal mucosa, face, palms, trunk, soles; distribution: generalized | Fever, headache, malaise, sore throat, generalized lymphadenopathy | Yes, length unknown | None | Unknown; Probable | Survived |
| Nigeria, outbreak cause unknown, 2017–2018b | 2171 | 28 d F | Description: vesicles, pustules; location: NA; distribution: generalized | Bronchopneumonia, encephalitis, seizures | Yes, length unknown | None | Unknown; Probable | Died (8 d after symptom onset), suspected to be secondary to encephalitis |
| United Kingdom, outbreak associated with travel to Nigeria, 2021b | 2240,72 | 18 mo F | Description: vesicles, 30 lesions; location: face, trunk, arms or hands, legs or feet; distribution: generalized | Fever, lymphadenopathy | Yes, 22 d | None | PCR +; Confirmed | Survived |
AG, agar gel; CAM, isolation on chorioallantoic membrane (CAM) of embryonated eggs; EM, electron microscopy; F, female; IgG, Immunoglobulin G; IgM, Immunoglobulin M; M, male; NA, not available; NP, not performed; PCR, polymerase chain reaction.
Confirmed case 5 virologic evidence of MPXV infection (mpox-specific PCR, histopathology or EM); probable case 5 orthopoxvirus serology positive or a known epidemiologic link to a confirmed case, plus clinical criteria.
Clade II inferred by outbreak region; no molecular confirmation available.
Severity scoring was given by the authors of the manuscripts detailing the patient’s illness.
Some case information from this outbreak includes previously unpublished data from CDC.
We identified 22 confirmed or probable cases (Table 1). Of these, 13 (59%) were male, and median age was 7 years (range 28 days–17 years old). All patients developed a rash. Of the 22 patients, 17 (77%) had additional clinical data available. Consistent with descriptions of infections caused by clade II MPXV in adults, the rash was most commonly described as papular, vesicular, pustular, or umbilicated.24 Commonly reported locations of lesions included trunk, palms or soles, arms or hands, back, and legs or feet. All cases for which rash distribution was described reported a generalized rash. The most common additional features included fever, chills, or sweats in 12 (71%) of 17 cases, lymphadenopathy in 8 (47%) (cervical lymphadenopathy was most common), sore throat in 7 (41%), headache in 7 (41%), and myalgias in 4 (24%) cases.
One 12-year-old child with mpox had recently received Dryvax, a live vaccinia virus vaccine, as postexposure prophylaxis (PEP). Symptoms developed 7 days after vaccination. He did not require hospitalization. The role of vaccination in attenuating symptoms for this patient is uncertain. No others received vaccines or MPXV-directed therapeutics.
Hospitalization status was known in 13 (59%) cases; 9 (69%) of these were hospitalized. The reason for hospitalization is not known for all cases, but includes severe disease, quarantine, and travel for medical care. Two (9%) cases had ocular involvement. Five of 22 cases (23%) developed severe illness: a 6-year-old female who developed encephalitis leading to seizures and coma (case 12); a 9-year-old male who had a pustular corneal lesion with subsequent corneal scarring and a submaxillary abscess (case 4); a 4-year-old female who was hospitalized because of toxic appearance and inability to stand (case 5); a 10-year-old female who developed a retropharyngeal abscess that led to shortness of breath and airway compromise (case 9); and a 28-day-old female who developed bronchopneumonia and encephalitis that led to seizures and death (case 21).
Clinical Descriptions of Clade II Mpox During 2022 Outbreak
Pediatric and adolescent cases of mpox have been described in multiple countries in 2022. The severity and rate of complications appears to be less than in previous outbreaks of clade II mpox, which may be attributable to timely diagnosis, access to medical care, provision of antiviral treatment, or other factors. A summary of US pediatric cases in 2022 described 83 cases: 22% received antiviral treatment, 11% were hospitalized, none required ICU-level care and no deaths were reported; complications included conjunctivitis, pain, and secondary skin infections. In a report of 16 cases in Spain ranging from ages 7 months to 17 years, none received antiviral treatment, 1 had a minor complication (secondary skin infection), and none required hospitalization.25
Several clinical case reports of clade II mpox in infants have been published in 2022. A report from the United Kingdom described a diffuse rash and respiratory failure in a neonate infected with both MPXV and adenovirus.26 A report from Florida described an infant <2 months old who was hospitalized because of concern for secondary skin infection and mpox lesions near the eye, received tecovirimat, vaccinia immune globulin intravenous (VIGIV), and prophylactic ophthalmic trifluridine, and subsequently recovered fully.27 Finally, a 7-month-old infant in Spain had a mild course and recovered fully without treatment.28
Adult cases of mpox in the current outbreak have been characterized by relatively localized disease, often involving the genitals, though a variety of dermatologic and other manifestations have been reported. Reported complications of mpox in adults in the current outbreak have included proctitis, urethritis, phimosis, balanitis, secondary bacterial skin infections, conjunctivitis, and other ocular complications, and rarely, encephalomyelitis.24,29,30 Children or adolescents who acquire mpox during the current outbreak may also be at risk for these complications. In particular, sexually active adolescents with anogenital lesions are likely at risk for anal or genitourinary complications.
Severe manifestations of mpox, including death, have occurred in immunocompromised adults with HIV and clade II mpox in the current outbreak. Children and adolescents with immunocompromising conditions are at increased risk of severe mpox.
DIAGNOSIS
When mpox is being considered in children and adolescents, the differential diagnosis is likely to include other conditions that cause rashes, such as varicella (chickenpox); herpes zoster (shingles); hand, foot, and mouth disease; scabies; molluscum contagiosum; impetigo; measles; herpes simplex virus; syphilis; allergic skin rashes; drug eruptions; and a variety of congenital infections in neonates. In contrast to the lesions associated with varicella, which display regional pleomorphism, mpox lesions in 1 region of the body are typically at the same stage of evolution.31 Although the rash of mpox progresses over approximately 2 to 4 weeks, many other rash illnesses, including varicella, typically evolve more rapidly. The unique characteristics of lesions, gradual evolution, and painful or pruritic quality can aid in distinguishing the rash of mpox from other systemic rashes. In sexually active adolescents with genital lesions, other sexually transmitted infections (STIs), including syphilis, chancroid, herpes simplex virus, and lymphogranuloma venereum, should also be considered.
CDC maintains a list of epidemiologic criteria used for defining cases of mpox (Fig 2). When considering evaluation for mpox in a child or adolescent, assessment using current epidemiologic criteria is paramount; the presence of 1 or more of these criteria greatly increases the likelihood that a compatible rash illness is caused by MPXV.32
FIGURE 2.

Epidemiologic criteria. See https://www.cdc.gov/poxvirus/monkeypox/clinicians/case-definition.html for Case Definitions for Use in the 2022 Mpox Response.
Testing for MPXV should be performed if epidemiologic criteria are present or if there is a strong suspicion based on clinical features. The preferred modality is polymerase chain reaction (PCR) testing for orthopoxviruses, which is currently available at public health laboratories and some commercial laboratories. Sample collection should include vigorous swabbing, but unroofing or aspirating the lesion is not necessary nor recommended. Testing the crust is also possible.33,34 Caution is needed when interpreting single laboratory test findings in patients with low risk of mpox.32 Serologic testing for MPXV might be considered as an adjunct in patients who have not been vaccinated against orthopoxviruses if there is concern for false positive testing or if testing the rash is not possible.
Concurrent testing for other infections should be considered if there is clinical suspicion. False positive orthopoxvirus testing has been described in children who were ultimately diagnosed with other viral illnesses that can cause similar lesions, and coinfections with MPXV have been described.32,35–37
MANAGEMENT
Mpox Treatment
As of this writing, there are no medications to treat mpox approved by the US Food and Drug Administration (FDA), nor are there clinical trial data to guide therapeutic decisions. Investigational drugs can be considered to treat children and adolescents who have severe illness (eg, sepsis, encephalitis); those with involvement of anatomic areas which might result in serious sequelae, such as scarring or strictures; and those who may be at increased risk for severe illness, such as children less than 1 year of age, those with immunocompromising conditions, and those with eczema or any other condition leading to a break in the skin.
Tecovirimat is an antiviral medication originally developed to treat smallpox that is currently being used as the first-line medication to treat mpox under an Expanded Access Investigational New Drug protocol in the United States. Tecovirimat appears to be well tolerated in adults, though its efficacy is unknown; outcome data from the current outbreak are being collected, including data from randomized controlled trials. Tecovirimat has been given to infants, children, and adolescents in at least 18 cases in the United States, including 8 children under 5 years old, and to an infant as young as 10 days old in the United Kingdom.16,26 Dosing for infants and children under 13 kg is not yet established, and the challenge of achieving accurate dosing and adequate absorption with enteral tecovirimat must be weighed against the potential for nephrotoxicity from intravenous tecovirimat.38
The use of other therapeutics can be considered as adjuncts or alternatives to tecovirimat in consultation with specialists and public health authorities. VIGIV is licensed for treatment of vaccinia virus vaccine complications and has been used historically for this purpose; it has also been given to young infants in the current outbreak without reported adverse events.27 Other antiviral medications, including cidofovir and its prodrug, brincidofovir, have shown in vitro activity against orthopoxviruses, but their use is limited by potential renal and hepatic toxicity.39,40
The cases described in Table 1 highlight complications of clade II mpox that have been described in children. Treatment teams should be vigilant for signs of encephalitis in children with mpox. Lesions should be covered and kept clean to decrease risk of secondary skin infection, and care should be taken to prevent children from scratching or picking lesions or touching their eyes after touching lesions.41 Ocular infections with MPXV can lead to permanent corneal scarring and can be sight-threatening. Topical trifluridine can be used in consultation with ophthalmology both to treat ocular manifestations of mpox and as prophylaxis for lesions near the eye or on the eyelid, given the risk of autoinoculation. Lastly, pain at the sites of mpox lesions can be severe. Patients with mpox may benefit from supportive care and pain control that is implemented early in the illness.
Managing MPXV Exposures in Children and Adolescents
Vaccines developed primarily for the prevention of smallpox are being used in the current outbreak to prevent or lessen the severity of mpox following a known exposure to MPXV. Although no vaccine products are yet licensed to prevent mpox, postexposure vaccination is being provided to children and adolescents in many countries, including the United States. When PEP is provided after a known exposure, timely vaccination (ideally within 4 days) likely maximizes effectiveness.42
There are 2 vaccines that can be used as PEP: ACAM2000, which contains a live, replication-competent vaccinia virus vaccine, and JYNNEOS, which contains a nonreplicating vaccinia virus and is administered in a 2-dose series.43,44 The use of ACAM2000, which must occur under an Expanded Access Investigational New Drug protocol, is limited by numerous contraindications and precautions, including age less than 12 months, immune deficiency, history of eczema or other active exfoliative skin conditions, pregnancy, cardiac disease, and others. Because it is nonreplicating, JYNNEOS lacks these contraindications and can currently be provided via subcutaneous route to children and adolescents of all ages exposed to MPXV under an Emergency Use Authorization from the FDA.45 Notably, the intradermal route of administration available to adults is not authorized for those under 18 years of age.
There have been no clinical trials of JYNNEOS in children. FDA authorized JYNNEOS for emergency use in children citing safety and immune response data from adults as well as historical data from the use of live vaccinia virus vaccine in children.46 The safety of JYNNEOS in children is also supported by clinical trials in which the vaccine (Modified Vaccine Ankara) was used as a platform for other investigational vaccines, including those for tuberculosis, malaria, measles, and Ebola.47–50 These studies included children as young as 5 months. The first doses of JYNNEOS have been administered to over 1000 children and adolescents in the United States during the current outbreak without any serious adverse events identified by October 21, 2022.51
Limited published observational data regarding JYNNEOS effectiveness in adults suggest that a single dose provides some protection against mpox.52,53 Its effectiveness to prevent mpox in children before or after exposure is unknown. Limited animal data suggest that preexposure vaccination with JYNNEOS may be immunogenic even at a young age.54
There are also other modalities for prevention or attenuation of mpox infection in children, including VIGIV, which (despite a lack of efficacy data) may be preferred to vaccination in young infants for whom safety data for JYNNEOS is relatively lacking. Like other PEP modalities, VIGIV is likely most effective when administered as close to the time of exposure as possible. At least 2 infants in the United States have received VIGIV for PEP during the current 2022 outbreak.27, 32 Antiviral medications have not been studied for efficacy as PEP.
PREVENTION
Primary Prevention of Mpox in Children and Adolescents
Pediatric health providers have an important role in mpox prevention. Crucially, adolescent patients who are sexually active, particularly males having sex with other males, are likely to benefit from enhanced counseling about mpox, including behavioral prevention strategies, signs and symptoms of mpox, and vaccination as preexposure prophylaxis (PrEP).55,56 Where minor consent laws allow, or with parental consent, adolescents and young adults should be offered mpox PrEP if they are determined to be at increased risk for mpox, eg, gay, bisexual, or other MSM, transgender, or nonbinary people with more than one sexual partner in the last 6 months, or those diagnosed with an STI in the last 6 months; sexual partners of people with the above risks; and people who anticipate experiencing the above risks.57
Proactive screening of sexually active adolescents for STIs other than mpox should continue, and people who acquired mpox following sexual activity should be screened for other STIs such as syphilis, HIV, gonorrhea, and chlamydia, even if they do not have symptoms.58,59 Condom use has not been studied as a prevention strategy for mpox during the current outbreak, and alone, is unlikely to be effective for mpox prevention since condoms do not prevent all skin-to-skin contact with lesions. Condom use should nevertheless continue to be encouraged to prevent the transmission of other STIs.
Current US recommendations state that people with mpox should isolate from others for the duration of their illness, from the time symptoms begin until lesions have healed, the scabs have fallen off, and a new layer of skin has formed.60,61 People with mpox should avoid cuddling, hands-on caregiving, or bed-sharing with others who are uninfected, including children. There is also evidence from the current outbreak that substantial surface contamination in the home may occur, highlighting the possibility of transmission via fomites.62 Surface disinfection is encouraged in homes where people with mpox have been living, and clothing and other linens used by people with mpox should be laundered.63
Because of the risk of transmission through direct contact and the potential for severe illness in newborns, parents and caregivers in isolation for mpox should be advised to isolate from their newborns after delivery.64 If isolation is not possible, precautions should be taken to minimize exposure to lesions and respiratory secretions from the infected parent or caregiver. It is unknown whether MPXV is detectable in human milk or whether mpox can be acquired through breastfeeding or feeding of expressed human milk. The risk of parent-to-child MPXV transmission during childbirth is not well described, and specific prevention strategies during childbirth (such as cesarean delivery) have not been evaluated.
Infection Control in Clinical Settings
In addition to standard precautions, additional precautions should be implemented when treating or evaluating children and adolescents with known or suspected mpox. Patients should be placed in individual rooms (not common waiting areas), and any healthcare personnel entering the room should wear a gown, gloves, eye protection, and a National Institute for Occupational Safety and Health-approved particulate respirator equipped with N95 filters or higher. People with confirmed or suspected mpox should be placed in isolation; asymptomatic people who have been exposed do not need to be placed in isolation but should be monitored for symptoms.
DISCUSSION
Mpox has historically affected children disproportionately to adults. The opposite is true in the current outbreak; however, the growing number of pediatric cases worldwide serves as a reminder that the epidemiology of mpox is not static. Mpox can affect anyone. With the ease of global travel, very little residual population immunity from smallpox vaccination,65 the potential for MPXV to become established in mammalian reservoir hosts, and the ever-present threat of viral variants, children and adolescents remain at risk for mpox infection during the ongoing 2022 outbreak and future outbreaks.
Although the majority of adults and children with mpox in the 2022 outbreak have experienced self-limited disease and have not required hospitalization, our review of 22 untreated cases of clade II mpox in children and adolescents before the current outbreak revealed 5 cases of severe illness or complications (23%) and 1 death (5% CFR), all among patients without documented comorbidities. There are important limitations to the summary provided here, including missing data and potential bias toward diagnosis and publication of severe cases, but this case summary nonetheless highlights that pediatric patients, especially young infants, may be at risk for severe disease or complications from infection with clade II MPXV.
Continued robust, dedicated surveillance and documentation of pediatric cases—including exposure types, diagnosis, coinfections, and clinical course—are urgently needed to guide public health communication and interventions, such as preventive messaging, PEP strategies, and the use of therapeutics for mpox. Systems for collecting data on the outcomes of expanded-access and emergency-use therapeutics are especially important in infants and young children for whom dosing and pharmacokinetic data are relatively lacking. Additionally, pediatricians should work with public health authorities to develop research protocols to answer outstanding questions, such as whether MPXV can be transmitted in human milk. Mpox has been documented in Africa since the 1970s, but resources for its surveillance, treatment, and prevention have been scarce. Although there are differences in the epidemiology of clade I and clade II mpox infections, both can infect children and adolescents, and both have potential to cause substantial morbidity and mortality, including hospitalization, visual impairment, critical illness, and death. Vaccines and antiviral medications have become widely used to prevent and treat mpox for the first time during the 2022 outbreak, but much remains unknown about their efficacy in pediatric patients. These tools should be rigorously evaluated in the current outbreak and implemented not only in the United States, but globally, particularly in countries where mpox is endemic, to reduce the morbidity and mortality children and adolescents face from infection with MPXV throughout the world.
ACKNOWLEDGMENTS
We thank Martha Knuth, MLIS and Edith R. Lederman, MD.
FUNDING:
No external funding.
ABBREVIATIONS
- CDC
Centers for Disease Control and Prevention
- CFR
case fatality ratio
- DRC
Democratic Republic of Congo
- FDA
Food and Drug Administration
- MPXV
mpox virus
- PCR
polymerase chain reaction
- PEP
postexposure prophylaxis
- PrEP
preexposure prophylaxis
- STI
sexually transmitted infection
- VIGIV
vaccinia immune globulin intravenous
Footnotes
CONFLICT OF INTEREST DISCLOSURES: The authors have indicated they have no conflicts of interest to disclose. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Center for Disease Control and Prevention.
REFERENCES
- 1.World Health Organization. 2022 monkeypox outbreak: global trends. Available at: https://worldhealthorg.shinyapps.io/mpx_global/. Accessed November 1, 2022
- 2.Centers for Disease Control and Prevention. Monkeypox vaccine administration in the U.S. Available at: https://www.cdc.gov/poxvirus/monkeypox/response/2022/vaccines_data.html. Accessed November 1, 2022
- 3.Bunge EM, Hoet B, Chen L, et al. The changing epidemiology of human monkeypox-a potential threat? A systematic review. PLoS Negl Trop Dis. 2022;16(2):e0010141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mbala PK, Huggins JW, Riu-Rovira T, et al. Maternal and fetal outcomes among pregnant women with human monkeypox infection in the Democratic Republic of Congo. J Infect Dis. 2017;216(7):824–828 [DOI] [PubMed] [Google Scholar]
- 5.Hutson CL, Carroll DS, Gallardo-Romero N, et al. Monkeypox disease transmission in an experimental setting: prairie dog animal model. PLoS One. 2011;6(12): e28295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Centers for Disease Control and Prevention. Monkeypox: clinical recognition. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/clinical-recognition.html. Accessed August 30, 2022
- 7.Reynolds MG, Yorita KL, Kuehnert MJ, et al. Clinical manifestations of human monkeypox influenced by route of infection. J Infect Dis. 2006;194(6):773–780 [DOI] [PubMed] [Google Scholar]
- 8.Foster SO, Brink EW, Hutchins DL, et al. Human monkeypox. Bull World Health Organ. 1972;46(5):569–576 [PMC free article] [PubMed] [Google Scholar]
- 9.Jezek Z, Grab B, Paluku KM, Szczeniowski MV. Human monkeypox: disease pattern, incidence and attack rates in a rural area of northern Zaire. Trop Geogr Med. 1988;40(2):73–83 [PubMed] [Google Scholar]
- 10.Jezek Z, Marennikova SS, Mutumbo M, Nakano JH, Paluku KM, Szczeniowski M. Human monkeypox: a study of 2,510 contacts of 214 patients. J Infect Dis. 1986;154(4):551–555 [DOI] [PubMed] [Google Scholar]
- 11.Jezek Z, Szczeniowski M, Paluku KM, Mutombo M. Human monkeypox: clinical features of 282 patients. J Infect Dis. 1987;156(2):293–298 [DOI] [PubMed] [Google Scholar]
- 12.Breman JG, Kalisa-Ruti, Steniowski MV, Zanotto E, Gromyko AI, Arita I. Human monkeypox, 1970–79. Bull World Health Organ. 1980;58(2):165–182 [PMC free article] [PubMed] [Google Scholar]
- 13.Arita I, Henderson DA. Monkeypox and whitepox viruses in West and Central Africa. Bull World Health Organ. 1976; 53(4):347–353 [PMC free article] [PubMed] [Google Scholar]
- 14.Nolen LD, Osadebe L, Katomba J, et al. Extended human-to-human transmission during a monkeypox outbreak in the Democratic Republic of the Congo. Emerg Infect Dis. 2016;22(6):1014–1021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Centers for Disease Control and Prevention. Smallpox: vaccine basics Available at: https://www.cdc.gov/smallpox/vaccine-basics/index.html. Accessed August 15, 2022
- 16.Hennessee I, Shelus V, McArdle CE, et al. ; California Department of Public Health Monkeypox Pediatric Working Group; CDC Monkeypox Pediatric Working Group. Epidemiologic and clinical features of children and adolescents aged <18 years with monkeypox - United States, May 17-September 24, 2022. MMWR Morb Mortal Wkly Rep. 2022;71(44) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bragazzi NL, Kong JD, Mahroum N, et al. Epidemiological trends and clinical features of the ongoing monkeypox epidemic: a preliminary pooled data analysis and literature review [published online ahead of print June 12, 2022]. J Med Virol. doi: 10.1002/jmv. [DOI] [PubMed] [Google Scholar]
- 18.Centers for Disease Control and Prevention (CDC). Household transmission of vaccinia virus from contact with a military smallpox vaccinee–Illinois and Indiana, 2007. MMWR Morb Mortal Wkly Rep. 2007;56(19):478–481 [PubMed] [Google Scholar]
- 19.Fenner F, Henderson DA, Arita I, Jezek Z, Ladnvi ID. Smallpox and its Eradication. Geneva, Switzerland: World Health Organization; 1988 [Google Scholar]
- 20.Lane JM, Ruben FL, Neff JM, Millar JD. Complications of smallpox vaccination, 1968. N Engl J Med. 1969;281(22): 1201–1208 [DOI] [PubMed] [Google Scholar]
- 21.Vora S, Damon I, Fulginiti V, et al. Severe eczema vaccinatum in a household contact of a smallpox vaccinee. Clin Infect Dis. 2008;46(10):1555–1561 [DOI] [PubMed] [Google Scholar]
- 22.Mwanbal PT, Tshioko KF, Moudi A, et al. Human monkeypox in Kasai Oriental, Zaire (1996–1997). Euro Surveill. 1997; 2(5):33–35 [DOI] [PubMed] [Google Scholar]
- 23.World Health Organization. Monkeypox, Zaire. Wkly Epidemiol Rec. 1996;71:326 [Google Scholar]
- 24.Philpott D, Hughes CM, Alroy KA, et al. ; CDC Multinational Monkeypox Response Team. Epidemiologic and clinical characteristics of Monkeypox cases - United States, May 17-July 22, 2022. MMWR Morb Mortal Wkly Rep. 2022;71(32): 1018–1022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Aguilera-Alonso D, Alonso-Cadenas JA, Roguera-Sopena M, Lorusso N, Miguel LGS, Calvo C. Monkeypox virus infections in children in Spain during the first months of the 2022 outbreak. Lancet Child Adolesc Health. 2022;6(11):e22–e23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ramnarayan P, Mitting R, Whittaker E, et al. ; NHS England High Consequence Infectious Diseases (Airborne) Network. Neonatal monkeypox virus infection. N Engl J Med. 2022;387(17):1618–1620 [DOI] [PubMed] [Google Scholar]
- 27.Saunders KE, Van Horn AN, Medlin HK, et al. Monkeypox in a young infant - Florida, 2022. MMWR Morb Mortal Wkly Rep. 2022;71(38):1220–1221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.AIonso-Cadenas JA, Andina-Martínez D, García-García CJ, et al. Monkeypox disease in a breastfeeding infant [published online ahead of print, 2022 Sep 30]. Pediatr Dermatol. 2022;10.1111/pde.15145.doi: 10.1111/pde.15145 [DOI] [PubMed] [Google Scholar]
- 29.Cash-Goldwasser S, Labuda SM, McCormick DW, et al. ; CDC Monkeypox Clinical Escalations Team. Ocular monkeypox - United States, July-September 2022. MMWR Morb Mortal Wkly Rep. 2022;71(42):1343–1347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pastula DM, Copeland MJ, Hannan MC, et al. Two cases of monkeypox-associated encephalomyelitis - Colorado and the District of Columbia, July-August 2022. MMWR Morb Mortal Wkly Rep. 2022; 71(38):1212–1215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Patauner F, Gallo R, Durante-Mangoni E. Monkeypox infection: an update for the practicing physician. Eur J Intern Med. 2022;104:1–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Minhaj FS, Petras JK, Brown JA, et al. ; CDC Monkeypox Emergency Response Team; CDC Monkeypox Emergency Response Team. Orthopoxvirus testing challenges for persons in populations at low risk or without known epidemiologic link to monkeypox - United States, 2022. MMWR Morb Mortal Wkly Rep. 2022; 71(36):1155–1158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Centers for Disease Control and Prevention. Monkeypox: laboratory procedures Available at: https://www.cdc.gov/poxvirus/monkeypox/lab-personnel/lab-procedures.html. Accessed September 30, 2022
- 34.Centers for Disease Control and Prevention. Monkeypox: preparation and collection of specimens. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/prep-collection-specimens.html. Accessed September 30, 2022
- 35.Hughes CM, Liu L, Davidson WB, et al. A tale of two viruses: coinfections of monkeypox and varicella zoster virus in the Democratic Republic of Congo. Am J Trop Med Hyg. 2020;104(2):604–611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hoff NA, Morier DS, Kisalu NK, et al. Varicella coinfection in patients with active monkeypox in the Democratic Republic of the Congo. EcoHealth. 2017; 14(3):564–574 [DOI] [PubMed] [Google Scholar]
- 37.Bížová B, Veselý D, Trojánek M, Rob F. Coinfection of syphilis and monkeypox in HIV positive man in Prague, Czech Republic. Travel Med Infect Dis. 2022;49:102368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Expanded Access IND Protocol: Use of Tecovirimat (TPOXXV®) for Treatment of Human Non-Variola Orthopoxvirus Infections in Adults and Children. Available at: https://www.cdc.gov/poxvirus/monkeypox/pdf/Tecovirimat-IND-Protocol-CDC-IRB.pdf. Accessed November 1, 2022
- 39.Adler H, Gould S, Hine P, et al. ; NHS England High Consequence Infectious Diseases (Airborne) Network. Clinical features and management of human monkeypox: a retrospective observational study in the UK. Lancet Infect Dis. 2022;22(8):1153–1162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vora SB, Brothers AW, Englund JA. Renal toxicity in pediatric patients receiving cidofovir for the treatment of adenovirus infection. J Pediatric Infect Dis Soc. 2017;6(4):399–402 [DOI] [PubMed] [Google Scholar]
- 41.Centers for Disease Control and Prevention. Clinical considerations for monkeypox in children and adolescents. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/pediatric.html. Accessed September 1, 2022
- 42.Kecmanaovć M, Suvaković V. [The effect of vaccination status on the clinical form and outcome of the disease in smallpox patients (author’s transl)]. MMW Munch Med Wochenschr. 1975;117(3):87–92 [PubMed] [Google Scholar]
- 43.ACAM2000. (Smallpox (Vaccinia) Vaccine, Live) [package insert]. Gaithersburg, MD: Emergent Product Development Gaithers-burg Inc.; 2018 [Google Scholar]
- 44.JYNNEOS. (Smallpox and Monkeypox Vaccine, Live, Non-replicating) [package insert]. Kvistgaard, Denmark: Bavarian Nordic; 2022. [Google Scholar]
- 45.Centers for Disease Control and Prevention. JYNNEOS vaccine. Available at: https://www.cdc.gov/poxvirus/monkeypox/interim-considerations/jynneos-vaccine.html. Accessed August 30, 2022
- 46.U.S. Food and Drug Administration. Monkeypox update: FDA authorizes emergency use of JYNNEOS vaccine to increase vaccine supply. Available at: https://www.fda.gov/news-events/press-announcements/monkeypox-update-fda-authorizes-emergency-use-jynneos-vaccine-increase-vaccine-supply. Accessed August 30, 2022
- 47.Tameris MD, Hatherill M, Landry BS, et al. ; MVA85A 020 Trial Study Team. Safety and efficacy of MVA85A, a new tuberculosis vaccine, in infants previously vaccinated with BCG: a randomised, placebo-controlled phase 2b trial. Lancet. 2013; 381(9871):1021–1028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Anywaine Z, Barry H, Anzala O, et al. ; EBL2002 Study group. Safety and immunogenicity of 2-dose heterologous Ad26.-ZEBOV, MVA-BN-Filo Ebola vaccination in children and adolescents in Africa: a randomised, placebo-controlled, multi-centre phase II clinical trial. PLoS Med. 2022;19(1):e1003865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Afolabi MO, Ishola D, Manno D, et al. ; EBL3001 study group. Safety and immunogenicity of the two-dose heterologous Ad26.ZEBOV and MVA-BN-Filo Ebola vaccine regimen in children in Sierra Leone: a randomised, double-blind, controlled trial. Lancet Infect Dis. 2022;22(1):110–122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nordic B Study to evaluate safety, dose response and immunogenicity of the measles vaccine MVA mBN85B in healthy children aged 6 months to 6 years. Available at: https://www.clinicaltrials.gov/ct2/show/NCT00891007?cond=MVA-mBN85B&draw=2&rank=1. Accessed Jul 10, 2022
- 51.Duffy J, Marquez P, Moro P, et al. Safety Monitoring of JYNNEOS Vaccine During the 2022 Mpox Outbreak — United States, May 22–October 21, 2022. MMWR Morb Mortal Wkly Rep. 2022;71(49):1555–1559 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Arbel R, Wolff Sagy Y, Zucker R, et al. Effectiveness of a single-dose modified vaccinia Ankara in human monkeypox: an observational study [preprint]. Research Square. 2022 [Google Scholar]
- 53.Payne AB, Ray LC, Kugeler KJ, et al. Incidence of monkeypox among unvaccinated persons compared with persons receiving $1 JYNNEOS vaccine dose - 32 U.S. jurisdictions, July 31-September 3, 2022. MMWR Morb Mortal Wkly Rep. 2022;71(40):1278–1282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cheminay C, K00F6rner J, Bernig C, et al. A single vaccination with non-replicating MVA at birth induces both immediate and long-term protective immune responses. Vaccine. 2018;36(18):2427–2434 [DOI] [PubMed] [Google Scholar]
- 55.Centers for Disease Control and Prevention. Safer sex, social gatherings, and monkeypox. Available at: https://www.cdc.gov/poxvirus/monkeypox/prevention/sexual-health.html. Accessed August 30, 2022
- 56.Centers for Disease Control and Prevention. What you need to know about monkeypox if you are a teen or young adult. Available at: https://www.cdc.gov/poxvirus/monkeypox/community/teens-young-adults.html. Accessed August 30, 2022
- 57.Centers for Disease Control and Prevention. Vaccination. Available at: https://www.cdc.gov/poxvirus/monkeypox/interim-considerations/overview.html. Accessed November 18, 2022
- 58.Centers for Disease Control and Prevention. Sexually transmitted infections treatment guidelines, 2021 - adolescents. Available at: https://www.cdc.gov/std/treatment-guidelines/adolescents.htm. Accessed August 30, 2022
- 59.Curran KG, Eberly K, Russell OO, et al. ; Monkeypox, HIV, and STI Team. HIV and sexually transmitted infections among persons with monkeypox - eight U.S. jurisdictions, May 17-July 22, 2022. MMWR Morb Mortal Wkly Rep. 2022; 71(36):1141–1147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Centers for Disease Control and Prevention. Isolation and prevention practices for people with monkeypox. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/isolation-procedures.html. Accessed September 1, 2022
- 61.Centers for Disease Control and Prevention. Isolation and infection control at home. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/infection-control-home.html. Accessed August 1, 2022
- 62.Pfeiffer JA, Collingwood A, Rider LE, et al. High-contact object and surface contamination in a household of persons with monkeypox virus infection - Utah, June 2022. MMWR Morb Mortal Wkly Rep. 2022;71(34):1092–1094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.United States Environmental Protection Agency. Disinfectants for emerging viral pathogens (EVPs): list Q. Available at: https://www.epa.gov/pesticide-registration/disinfectants-emerging-viral-pathogens-evps-list-q. Accessed Aug 01, 2022
- 64.Centers for Disease Control and Prevention. Clinical considerations for monkeypox in people who are pregnant or breastfeeding. Available at: https://www.cdc.gov/poxvirus/monkeypox/clinicians/pregnancy.html. Accessed August 1, 2022
- 65.Reynolds MG, Carroll DS, Karem KL. Factors affecting the likelihood of monkeypox’s emergence and spread in the post-smallpox era. Curr Opin Virol. 2012;2(3):335–343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Huhn GD, Bauer AM, Yorita K, et al. Clinical characteristics of human monkeypox, and risk factors for severe disease. Clin Infect Dis. 2005;41(12):1742–1751 [DOI] [PubMed] [Google Scholar]
- 67.Anderson MG, Frenkel LD, Homann S, Guffey J. A case of severe monkeypox virus disease in an American child: emerging infections and changing professional values. Pediatr Infect Dis J. 2003;22(12): 1093–1096, discussion 1096–1098 [DOI] [PubMed] [Google Scholar]
- 68.Sejvar JJ, Chowdary Y, Schomogyi M, et al. Human monkeypox infection: a family cluster in the midwestern United States. J Infect Dis. 2004;190(10):1833–1840 [DOI] [PubMed] [Google Scholar]
- 69.Reynolds MG, Wauquier N, Li Y, et al. Human monkeypox in Sierra Leone after 44-year absence of reported cases. Emerg Infect Dis. 2019;25(5):1023–1025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kabuga AI, El Zowalaty ME. A review of the monkeypox virus and a recent outbreak of skin rash disease in Nigeria. J Med Virol. 2019;91(4):533–540 [DOI] [PubMed] [Google Scholar]
- 71.Ogoina D, Iroezindu M, James HI, et al. Clinical course and outcome of human monkeypox in Nigeria. Clin Infect Dis. 2020;71(8):e210–e214 [DOI] [PubMed] [Google Scholar]
- 72.Hobson G, Adamson J, Adler H, et al. Family cluster of three cases of monkeypox imported from Nigeria to the United Kingdom, May 2021. Euro Surveill. 2021;26(32):2100745. [DOI] [PMC free article] [PubMed] [Google Scholar]


