Abstract
Introduction: Antiplatelet therapy has been associated with fewer exacerbations and reduced respiratory symptoms in chronic obstructive pulmonary disease (COPD). Whether platelet activation is associated with respiratory symptoms in COPD is unknown.
Methods: Former smokers with spirometry-confirmed COPD had urine 11-dehydro-thromboxane B2 (11dTxB2), plasma soluble CD40L (sCD40L), and soluble P-selectin (sP-selectin) repeatedly measured during a 6- to 9-month study period. Multivariate mixed-effects models adjusted for demographics, clinical characteristics, and medication use evaluated the association of each biomarker with respiratory symptoms, health status, and quality of life.
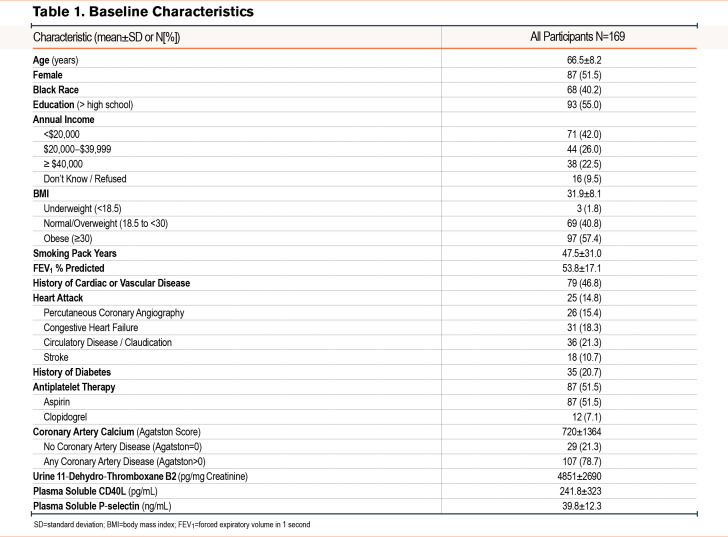
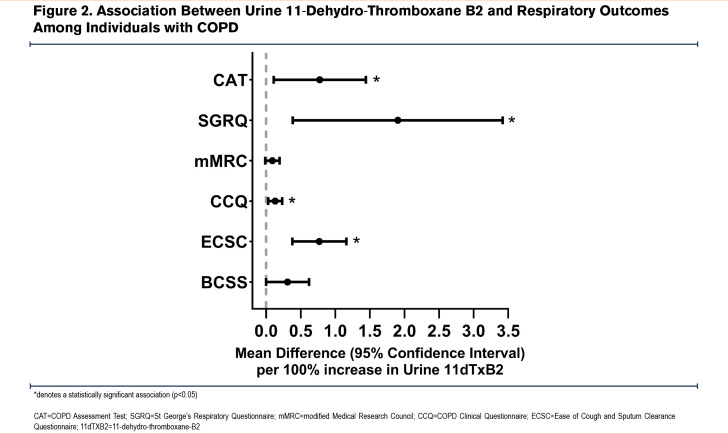
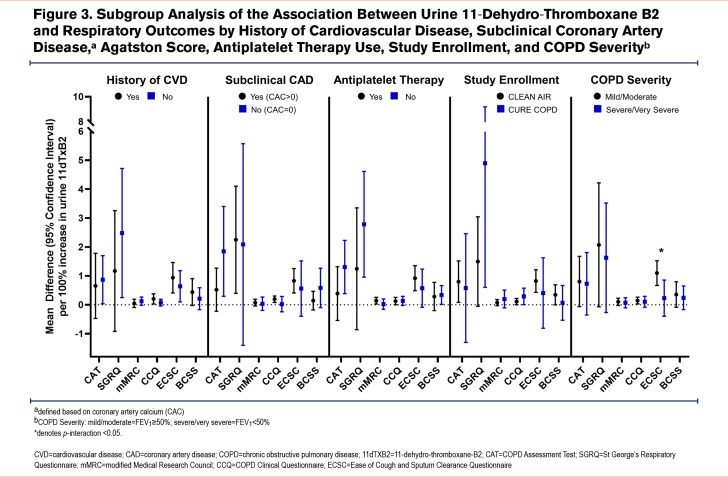
Results: Among 169 participants (average age 66.5±8.2 years, 51.5% female, 47.5±31 pack years, forced expiratory volume in 1 second percent predicted 53.8±17.1), a 100% increase in 11dTxB2 was associated with worse respiratory symptoms reflected by higher scores on the COPD Assessment Test (β 0.77, 95% confidence interval [CI]: 0.11–1.4) and Ease of Cough and Sputum Clearance Questionnaire β 0.77, 95%CI: 0.38–1.2, worse health status (Clinical COPD Questionnaire β 0.13, 95%CI: 0.03-0.23) and worse quality of life (St George’s Respiratory Questionnaire β 1.9, 95%CI: 0.39-3.4). No statistically significant associations were observed for sCD40L or sP-selectin. There was no consistent statistically significant effect modification of the relationship between urine 11dTxB2 and respiratory outcomes by history of cardiovascular disease, subclinical coronary artery disease, antiplatelet therapy, or COPD severity.
Conclusions: In stable moderate-severe COPD, elevated urinary11dTxB2, a metabolite of the platelet activation product thromboxane A2, was associated with worse respiratory symptoms, health status, and quality of life.
Keywords: chronic obstructive pulmonary disease, biomarkers, aspirin, platelet activation
Introduction
Extrapulmonary manifestations of chronic obstructive pulmonary disease (COPD) include heightened systemic inflammation, defective immune response, and increased risk for cardiovascular disease (CVD) and thrombosis.1-5 In addition to their well-known roles in CVD and thrombosis, activated platelets are critical for trafficking immune cells and release cytokines and chemokines that perpetuate systemic inflammation or act directly on the respiratory system.6-9 Several studies have reported on abnormal platelet indices in COPD, such as elevated platelet count and mean platelet volume, as potential surrogates of platelet activation and have shown adverse outcomes in both stable and exacerbated COPD.10-14 Limited studies have demonstrated increased platelet activation in individuals with stable COPD compared with healthy controls with even further elevation during acute exacerbations.15-17 Furthermore, persistently elevated platelet reactivity despite anti-platelet therapy after percutaneous coronary intervention was significantly more common among individuals with COPD compared with matched controls.18 Whether higher levels of platelet activation among individuals with COPD impacts respiratory morbidity and outcomes remains unknown.
Activated platelets release thromboxane A2 (TxA2), which is a prostanoid produced by the enzymatic action of cyclooxygenase-1 (COX-1) on arachidonic acid. TxA2 is itself a strong platelet activator, causing feedback activation and recruitment of additional platelets to vascular sites.19 In the lung, TxA2 acts on airway smooth muscles, causing bronchoconstriction, airway hyper-responsiveness, and remodeling.20,21 Direct measurement of TxA2 is difficult and inaccurate given its extremely short half-life (≤30 seconds), however, a stable urinary metabolite, 11-dehydro-thromboxane-B2 (11dTxB2), offers an opportunity to indirectly measure platelet activation that occurs in vivo. In observational studies of individuals with COPD, aspirin, which acetylates and permanently inactivates COX-1 in platelets, has been associated with lower all-cause mortality, lower incidence rate of acute exacerbations of COPD (AECOPDs), less severe AECOPDs, better quality of life, less dyspnea, and slower progression of subclinical emphysema.10,22-28
Activated platelets also release additional molecules that are stored in α and dense granules. Measuring these molecules is another method to determine the degree of in vivo platelet activation.29 CD40 ligand (CD40L) and P-selectin are a granule constituents that become expressed on the platelet surface after activation, and both can be secreted as soluble biologically active molecules that can be measured in plasma (sCD40L and sP-selectin). When expressed on the surface of activated platelets, CD40L activates the endothelium to secrete chemokines and express adhesion molecules while P-selectin recruits leukocytes via its counter-receptor, P-selectin glycoprotein ligand-1 (PSGL-1).6,7,30,31
The aim of this study was to evaluate the association of platelet activation markers — urine 11dTxB2, plasma sCD40L, and plasma sP-selectin— with respiratory symptoms in a cohort of individuals with stable COPD. The a priori hypothesis was that elevated levels of platelet activation markers would be associated with increased respiratory symptoms, worse health status, and worse quality of life.
Methods
Participants
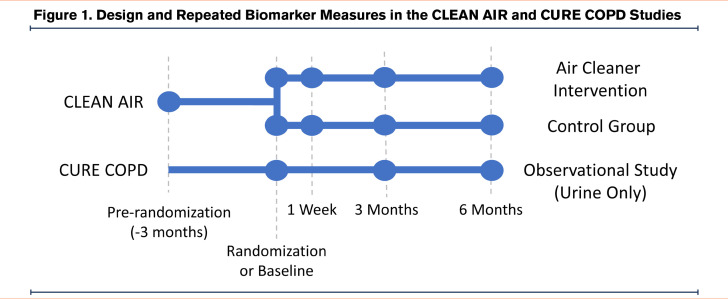
This is a secondary analysis of data from the Clinical Trial of Air Cleaners to Improve Indoor Air Quality and COPD Health (CLEAN AIR) study (ClinicalTrials.gov #NCT02236858) and Comparing Urban and Rural Effects of Poverty on COPD (CURE COPD) study. Both studies enrolled former smokers (≥10 pack-year history) with stable moderate-severe COPD (forced expiratory volume in 1 second [FEV1] to forced vital capacity [FVC] ≤70% and FEV1<80% predicted). CLEAN AIR was a double-blind clinical trial that randomized participants to active air cleaners with HEPA and carbon filters or a sham and is described in detail elsewhere.32 CURE COPD was an observational study evaluating the interactive effects of indoor air pollution and diet. For 39 participants who enrolled in both studies, only data from CLEAN AIR was included in the analysis. CLEAN AIR participants had 5 visits over a 9-month period (3 months prior to randomization, at randomization, then at 1 week, 3 months, and 6 months post-randomization) and CURE COPD participants had 3 visits over a 6-month period (baseline, 3 months, and 6 months) (Figure 1). Each study was approved by the Johns Hopkins University Institutional Review Board and all participants provided written informed consent.
Measurements
Urine samples were collected at each study visit and ethylenediaminetetraacetic acid (EDTA) plasma was additionally collected in CLEAN AIR. A complete blood count was analyzed in the clinical laboratory immediately following sample collection. Urine and plasma samples were processed within 2 hours of collection, stored at -80⁰C, and analyzed in a single batch at the Johns Hopkins Institute for Clinical and Translational Research (JH ICTR) Core Laboratory. The primary biomarker of interest was urine 11dTxB2 which was measured using a commercially available enzyme-linked immunoassay (ELISA) kit (Enzo Life Sciences, Lyon, France;pg/mL). The secondary biomarkers of interest were likewise measured by ELISA: plasma sP-selectin (R&D Systems, Minneapolis, Minnesota;ng/mL) and plasma sCD40L (R&D Systems, Minneapolis, Minnesota;pg/mL). Urine 11dTxB2 was adjusted by dividing the measured value by urine creatinine which was also measured by ELISA (Quidel Corporation, San Diego, CA;mg/mL) at the JH ICTR Core Laboratory.
Medical history was obtained at baseline using structured questionnaires. Although antiplatelet therapy use was not explicitly solicited, participants were asked to bring all medications they used in the last 3 months to each study visit. Research staff transcribed a list of medications at each study visit which were parsed for generic and trade names of antiplatelet therapies (aspirin, clopidogrel, prasugrel, ticagrelor). A participant was determined to be on a specific antiplatelet therapy during the entire study period if the medication appeared on their medication list at least once during the study. Spirometry was performed according to the American Thoracic Society protocol (Koko spirometer; nSpire) at each visit and post-bronchodilator percent predicted values calculated.33 Non-contrast non-electrocardiography-gated computed tomography (CT) scans of the chest were performed once during the study period, reconstructed at contiguous 0.75 mm and 3 mm thickness slices. Coronary artery calcium (CAC) score (Agatston score) was quantified by a single fellowship-trained cardiothoracic radiologist (C.T.L.) on inspiratory CT scans using the calcium scoring tool on either Carestream Vue PACS (version 12.2.2.0105) or Syngo.Via (version VB40B). Non-gated CT scans have demonstrated good agreement and reliability for quantification of CAC relative to electrocardiography-gated CT scans, although they tend to systematically underestimate CAC burden.34,35
Outcomes
Clinical outcomes ascertained at each visit included respiratory symptoms (COPD Assessment Test [CAT],36 modified Medical Research Council questionnaire [mMRC],37 Ease of Cough and Sputum Clearance questionnaire [ECSC],38 and Breathlessness, Cough, and Sputum Scale [BCSS]),39 and health status (Clinical COPD Questionnaire [CCQ]).40 Quality of life (St George’s Respiratory Questionnaire [SGRQ])41 was ascertained at all visits except the 1-week follow-up in CLEAN AIR. The total score of all outcomes were evaluated as continuous variables and the minimal clinically important difference has been established42 for most: CAT (2 units),43 SGRQ (4 units),44 CCQ (0.3–0.5 points),45 BCSS (small improvement=0.3 units, moderate improvement=0.6 units, substantial improvement >1 unit).39
Statistical Analysis
Baseline characteristics were described and compared using chi-squared, Fisher’s exact test, or t-test between participants in the 2 studies. A comparison of the baseline distribution of each biomarker was performed using the Wilcoxon rank sum test for aspirin use, history of CVD, and evidence of sub-clinical coronary artery disease (CAD) using presence of CAC on CT scan (Agatston score=0 versus >0).46 To estimate the association of each biomarker with the outcomes of interest, a generalized linear mixed model with random intercept for participants was used. Biomarkers were modeled continuously and log-transformed, and the linearity assumption was tested using restricted cubic splines. In the case where non-linearity was suggested, the biomarker was modeled categorically as quartiles (i.e., sP-selectin). Multivariate models were adjusted for factors that may impact respiratory outcomes or degree of systemic platelet activation, including study (CURE versus CLEAN AIR intervention versus CLEAN AIR control), time (in months), and its interaction with study group, age, gender, race (Black versus White or other), educational attainment (≤high school versus >high school), body mass index, lung function (percent predicted FEV1), pack-year smoking history, CVD history, diabetes history, and use of antiplatelet therapy (aspirin or clopidogrel). CVD history was defined as self-reported baseline history of congestive heart failure, stroke, heart attack, percutaneous coronary angiography, or circulatory disease. Disaggregation of within- and between-person effects was explored.47 Sensitivity analyses were conducted including adding platelet count and MPV as covariates to biomarker models and limiting the analysis of urine 11dTxB2 to participants and study visits with plasma samples available to investigate whether any discrepancy in findings between urine and plasma biomarkers are attributable to selection bias. Effect modification by history of CVD, presence of CAC, antiplatelet therapy use, study enrollment, and COPD disease severity (categorized as mild/moderate [FEV1 ≥50%] and severe/very severe [FEV1 <50%]) were explored for the relationship between urine 11dTxB2 and outcomes using an interaction term for urine 11dTxB2, and the effect modifier of interest and subgroup analysis results are presented. Additionally, effect modification by history of CVD and presence of CAC was explored in a subset of participants not on antiplatelet therapy. Analysis of effect modification for sCD40L and sP-selectin is not presented due to small sample size of the subgroups. All analyses were conducted using SAS 9.4 (Carey, North Carolina) and all statistical comparisons including interactions were considered significant at p<0.05.
Results
There were 192 unique participants after excluding CURE COPD data for dual enrolled participants. Individuals were additionally excluded for missing biomarker data (19, 9.9%) or baseline spirometry that did not meet acceptability and repeatability criteria33 (4, 2.1%) resulting in a final sample of 169 participants, of whom 113 (66.9%) were enrolled in CLEAN AIR (57 intervention, 56 control) and 56 (33.1%) were enrolled in CURE COPD. Baseline characteristics are presented in Table 1. Compared to participants in CLEAN AIR, CURE COPD participants were older, more likely to be Black and reported fewer pack years smoked (Supplemental Table 1 in the online supplement). In sum, 16.3% of visits had missing urine samples and 11.9% had missing plasma samples (collected in the CLEAN AIR study only), with a greater proportion at 3- and 6-month follow-up, resulting in a total of 630 urine and 505 plasma samples (Supplemental Table 2 in the online supplement). The distribution of 11dTxB2, sCD40L, and sP-selectin at each visit is also presented in Supplemental Table 2 in the online supplement. Among participants who had more than 1 measurement, the median within-person difference (maximum-minimum/minimum) was 95% for 11dTxB2 (N=154) and 132% for sCD40L (N=112) and thus, results are reported per 100% change (doubling) of the biomarker. Baseline 11dTxB2 was higher among CLEAN AIR participants compared with CURE COPD participants (5500±2748 versus 3503±1991 pg/mg Cr; p<0.0001). Half of the participants (N=87) were aspirin users of whom 12 (13.8%) were concomitantly using clopidogrel while none reported using prasugrel, ticagrelor, or clopidogrel monotherapy. Individuals who reported antiplatelet therapy use had significantly lower baseline 11dTxB2 compared with antiplatelet therapy non-users with no difference in sCD40L or sP-selectin between the 2 groups (Supplemental Table 2 in the online supplement). SGRQ, which was not ascertained at the 1-week follow-up, was missing in 7.5% of visits (available N=573), BCSS was missing in 12% (available N=644), while the remainder of the questionnaires had approximately 7% missing (available N=679–685).
In multivariate analysis (Figure 2), a 100% increase in 11dTxB2 was associated with worse symptoms (CAT score mean difference [β] 0.77, 95% confidence interval [CI]: 0.11, 1.4; ECSC β 0.77, 95%CI: 0.38, 1.2), worse health status (CCQ score β 0.13, 95%CI: 0.03, 0.23), and worse quality of life (SGRQ score β 1.9, 95%CI: 0.39, 3.4). The association of 11dTxB2 with mMRC (β 0.1, 95%CI: -0.007, 0.20) and BCSS (β 0.31, 95%CI -0.001, 0.62) did not reach statistical significance. The observed associations appear to be related to within-person effect (Supplemental Table 3 in the online supplement). No statistically significant associations were observed for sCD40L (Supplemental Figure 1 in the online supplement) or sP-selectin (Supplemental Figure 2 in the online supplement). sP-selectin was modeled categorically as quartiles due to the evidence of non-linear relationships with a few of the outcomes. Additional adjustment of the biomarker models with platelet count and mean platelet volume did not substantially impact the findings (results not shown).
Antiplatelet therapy use was higher among individuals who reported a history of CVD (63.3%) compared with those without a history of CVD (41.1%, p=0.004) and higher prevalence among those with CAC compared to those without CAC (55.1% versus 41.4%; p=0.2). There was no significant difference in the distribution of any of the biomarkers between participants with history of CVD compared to those without CVD or those with CAC compared to those without CAC although median urine 11dTxB2 was higher among participants without CVD history or CAC while sCD40L and sP-selectin were higher among participants with CVD history or CAC (Supplemental Table 2 in the online supplement). There was no consistently, statistically significant effect modification of the association between urine 11dTxB2 and respiratory outcomes by history of CVD, presence of CAC, antiplatelet therapy use, study enrollment, or COPD severity and subgroup analyses did not reveal any consistent trends (Figure 3). Among aspirin non-users, median levels of all biomarkers were higher among participants without CVD and participants with CAC>0 (Supplemental Table 4 in the online supplement). When limited to aspirin non-users, results were consistent with stronger association of urine 11dTxB2 with health status (CCQ; pinteraction=0.03) and quality of life (SGRQ; pinteraction=0.06) among individuals with CAC>0 (Supplemental Figure 3 in the online supplement). A sensitivity analysis of urine 11dTxB2 limited to participants and visits where plasma samples were available demonstrated consistent results for all respiratory outcomes, though estimates were attenuated and no longer statistically significant for CCQ and SGRQ (Supplemental Figure 4 in the online supplement).
Discussion
Among individuals with stable moderate-severe COPD, elevated urine 11dTxB2, a biomarker of platelet activation, was consistently associated with significantly worse respiratory symptoms, health status, and quality of life independent of comorbid cardiovascular disease. This was largely driven by within-person differences in biomarker and respiratory symptoms. There were no associations between the plasma biomarkers (sCD40L and sP-selectin) and respiratory outcomes. To our knowledge this is the first study investigating the association of systemic biomarkers of platelet activation and respiratory symptoms in COPD and suggests a role of platelet activation through the TxA2 pathway in respiratory morbidity among individuals with COPD.
Of the 3 platelet biomarkers investigated, 11dTxB2 most closely reflects systemic platelet activation. Urine 11dTxB2 is one of several metabolites of TxA2 which is generated and released from the action of COX on arachidonic acid when platelets are activated. Urine 11dTxB2 is commonly used as a non-invasive measure of in vivo platelet activation,48,49 although, its concentration may also reflect vascular, renal, and immune cell production, or differences in excretion.50 In contrast, P-selectin and CD40L are stored in alpha granules that are transported to the surface upon platelet activation. A portion of the stored P-selectin and CD40L is retained on the platelet surface to facilitate interactions with leukocytes and endothelium while the remainder is secreted. There is evidence that the majority of circulating sP-selectin is of platelet origin and that sCD40L is correlated with platelet-monocyte aggregates, a sensitive marker of platelet activation.51-53 However, sP-selectin may originate from endothelial cells or be resorbed by platelets for recycling while sCD40L may have been cleaved from the surface of immune cells.54,55 The non-specific origin and complex mechanisms of sP-selectin and sCD40L complicates interpretation of those results. The discrepant results between urine 11dTxB2 and the plasma platelet activation markers may alternatively be attributable to in vitro platelet activation during acquisition, processing, and storage of plasma samples. Since the parent study that contributed plasma samples was not designed for the purpose of evaluating markers of platelet activation,32 precautions to minimize in vitro platelet activation were not protocolized. Such precautions include drawing the sample from the antecubital fossa using a large needle, removing the tourniquet prior to sample collection, discarding several mL of blood before acquisition of the plasma sample, and minimizing the time between collection and processing of the sample.56 Conversely, use of EDTA as the plasma anticoagulant would have inhibited sCD40L release that may have become activated after collection or during sample storage.57
This study did not find any significant effect modification of the association between 11dTxB2 and respiratory outcomes by self-reported history of CVD or subclinical CAD as determined by presence of coronary artery calcification. Thus, the association between activated platelets and worse respiratory symptoms was present among individuals with and without underlying CVD or CAD. Respiratory outcomes in COPD have been difficult to distinguish from respiratory manifestations of cardiac symptoms which may complicate interpretation of platelet activation markers that have been associated with cardiovascular disease. Presence of an association between 11dTxB2 and respiratory outcomes regardless of clinical or subclinical CVD reduces the possibility that patient-reported respiratory symptoms are manifestations of CVD, though further study is warranted.
The findings of this study build upon a previous study showing reduced respiratory symptoms, better quality of life, and fewer exacerbations among individuals with COPD reporting daily aspirin use compared to aspirin non-users.28 Other studies have reported that aspirin use was associated with reduced all-cause mortality in stable COPD and following COPD exacerbation,10,25 less severe COPD exacerbations,24 and slower progression of subclinical emphysema.27 Aspirin is an antiplatelet agent that specifically antagonizes TxA2 synthesis, the pathway found to be associated with worse respiratory morbidity in this study. TxA2 or its synthetic analog, have been shown to cause bronchoconstriction in both humans and animal models, potentially mediated by airway muscarinic receptors, a target of first line COPD therapy.20,58 In clinical trials of asthma patients, seratrodast, a TxA2 receptor antagonist, decreased airway responsiveness to methacholine, improved symptoms, and increased peak expiratory flow.59,60 It is currently recommended as add-on therapy in the treatment of asthma in Japan.61 A crossover trial of 15 individuals with emphysema due to alpha-1 antitrypsin deficiency did not show any significant difference in microvascular blood flow with low-dose aspirin compared to placebo.62 Future clinical trials evaluating the impact of aspirin or thromboxane A2 receptor antagonists on platelet activation suppression and respiratory symptoms in more heterogenous COPD samples are warranted.
This study has limitations. The relatively small sample size reduces the power to detect between-person effects, which may have conservatively biased estimates, in this study that drew its power from repeated measurements and identification of within-person effects. Associations between 11dTxB2 and clinical outcomes did not reach the minimal clinically important differences. Furthermore, there may be residual confounding as a consequence of this study including secondary analysis of clinical trial data, although, the study intervention arm was included as a covariate. Comorbidities were self-reported and antiplatelet therapy including dose and adherence were not specifically ascertained which may result in misclassification due to recall bias. Additionally, there were few individuals on P2Y12-antagonist medications and none using it as monotherapy which precluded investigation of that therapeutic class of antiplatelet medications independently of aspirin. Finally, although aspirin use has been associated with reduced incidence of COPD exacerbations,28 due to the short follow-up period and paucity of events, this study was unable to assess the association between platelet activation biomarkers and COPD exacerbations.
In conclusion, elevated urinary 11dTxB2, a marker of COX-mediated platelet activation, was associated with worse respiratory symptoms and quality of life independent of reported cardiovascular disease or coronary artery calcification; however, this was not reflected by sP-selectin and sCD40L. Future studies employing more specific measures of platelet activation or evaluating the efficacy of aspirin or a targeted thromboxane inhibitor in COPD are warranted.
Abbreviations
Abbreviations: 11dTxB2=11-dehydro-thromboxane B2; AECOPD= acute exacerbation of COPD; BCSS=Breathlessness, Cough, and Sputum Scale; CAC=coronary artery calcium; CAD=coronary artery disease; CAT=COPD Assessment Test; CCQ=Clinical COPD Questionnaire; CD40L=CD40 Ligend; CI=confidence interval; COPD=chronic obstructive pulmonary disease; COX-1=cyclooxygerase-1; CT=computed tomography; CVC=cardiovascular disease; ECSC=Ease of Cough and Sputum Clearance Questionnaire; EDTA=ethylene diaminetetraacetic acid; ELISA=enzyme-linked immunoassay; FEV1=forced expiratory volume in 1 second; FVC=forced vital capacity; JH ICTR=Johns Hopkins Institute for Clinical and Translational Research, mMRC=modified Medical Research Council; PSGL-1=P-selection glycoprotein ligand-1; SCD40L=soluble CD40L; SGRQ=St George’s Respiratory Questionnaire; sP-selectin=soluble P-selectin; TxA2=thromboxane A-2
Funding Statement
The Clinical Trial of Air Cleaners to Improve Indoor air Quality and COPD Health (CLEAN AIR) was supported by the National Institute of Environmental Health Sciences (NIEHS) R01ES022607. CLEAN AIR HEART was supported by NIEHS R21ES025840. The Comparing Urban and Rural Effects of Poverty on COPD (CURE COPD) study was supported by National Institute on Minority Health Disparities P50MD010431. AF was supported by the National Heart Lung and Blood Institute (NHLBI) T32HL007534, NIEHS F32ES028576, and NHLBI K23HL151758. NP was supported by K23HL123594. Trial Registration: ClinicalTrials.gov Identifier #NCT02236858
References
- 1.Agustí A,Edwards LD,Rennard SI,et al. Persistent systemic inflammation is associated with poor clinical outcomes in COPD: a novel phenotype. PLoS One. 2012;7(5):e37483. doi: https://doi.org/10.1371/journal.pone.0037483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bhat TA,Panzica L,Kalathil SG,Thanavala Y. Immune dysfunction in patients with chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2015;12 (Suppl 2):S169-175. doi: https://doi.org/10.1513/AnnalsATS.201503-126AW [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rabe KF,Hurst JR,Suissa S. Cardiovascular disease and COPD: dangerous liaisons? Eur Resp Rev. 2018;27(149):180057. doi: https://doi.org/10.1183/16000617.0057-2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kunisaki KM,Dransfield MT,Anderson JA,et al. Exacerbations of chronic obstructive pulmonary disease and cardiac events. A post hoc cohort analysis from the SUMMIT randomized clinical trial. Am J Respir Crit Care Med. 2018;198(1):51-57. doi: https://doi.org/10.1164/rccm.201711-2239OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Børvik T,Brækkan SK,Enga K,et al. COPD and risk of venous thromboembolism and mortality in a general population. Eur Respir J. 2016;47(2):473-481. doi: https://doi.org/10.1183/13993003.00402-2015 [DOI] [PubMed] [Google Scholar]
- 6.Larsen E,Celi A,Gilbert GE,et al. PADGEM protein: a receptor that mediates the interaction of activated platelets with neutrophils and monocytes. Cell. 1989;59(2):305-312. doi: https://doi.org/10.1016/0092-8674(89)90292-4 [DOI] [PubMed] [Google Scholar]
- 7.Pitchford SC,Yano H,Lever R,et al. Platelets are essential for leukocyte recruitment in allergic inflammation. J Allergy Clin Immunol. 2003;112(1):109-118.doi: https://doi.org/10.1067/mai.2003.1514 [DOI] [PubMed] [Google Scholar]
- 8.Rendu F,Brohard-Bohn B. The platelet release reaction: granules' constituents, secretion and functions. Platelets. 2001;12(5):261-273. doi: https://doi.org/10.1080/09537100120068170 [DOI] [PubMed] [Google Scholar]
- 9.Vieira-de-Abreu A,Campbell RA,Weyrich AS,Zimmerman GA. Platelets: versatile effector cells in hemostasis, inflammation, and the immune continuum. Semin Immunopathol. 2012;34(1):5-30.doi: https://doi.org/10.1007/s00281-011-0286-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Harrison MT,Short P,Williamson PA,Singanayagam A,Chalmers JD,Schembri S. Thrombocytosis is associated with increased short-and long-term mortality after exacerbation of chronic obstructive pulmonary disease: a role for antiplatelet therapy? Thorax. 2014;69(7):609-615. doi: https://doi.org/10.1136/thoraxjnl-2013-203996 [DOI] [PubMed] [Google Scholar]
- 11.Fawzy A,Putcha N,Paulin LM,et al. Association of thrombocytosis with COPD morbidity: the SPIROMICS and COPDGene cohorts. Respir Res. 2018;19(1):20. doi: https://doi.org/10.1186/s12931-018-0717-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Garcia-Pachon E,Baeza-Martinez C,Ruiz-Alcaraz S,Grau-Delgado J. Prediction of three-month readmission based on haematological parameters in patients with severe COPD exacerbation. Adv Respir Med. 2021;89(5):501-504. doi: https://doi.org/10.5603/ARM.a2021.0076 [DOI] [PubMed] [Google Scholar]
- 13.El-Gazzar AG,Kamel MH,Elbahnasy OKM,El-Naggar MES. Prognostic value of platelet and neutrophil to lymphocyte ratio in COPD patients. Expert Rev Respir Med. 2020;14(1):111-116. doi: https://doi.org/10.1080/17476348.2019.1675517 [DOI] [PubMed] [Google Scholar]
- 14.Huebner ST,Henny S,Giezendanner S,et al. Prediction of acute COPD exacerbation in the Swiss multicenter COPD cohort study (TOPDOCS) by clinical parameters, medication use, and immunological biomarkers. Respiration. 2022;101(5) :1-14. doi: https://doi.org/10.1159/000520196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maclay JD,McAllister DA,Johnston S,et al. Increased platelet activation in patients with stable and acute exacerbation of COPD. Thorax. 2011;66(9):769-774. doi: https://doi.org/10.1136/thx.2010.157529 [DOI] [PubMed] [Google Scholar]
- 16.Muñoz-Esquerre M,Ferreiro JL,Huertas D,et al. Impact of acute exacerbations on platelet reactivity in chronic obstructive pulmonary disease patients. Int J Chron Obstruct Pulmon Dis. 2017;13:141-148. doi: https://doi.org/10.2147/COPD.S152660 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.van der Vorm LN,Li L,Huskens D,et al. Acute exacerbations of COPD are associated with a prothrombotic state through platelet-monocyte complexes, endothelial activation and increased thrombin generation. Respir Med. 2020;171:106094. doi: https://doi.org/10.1016/j.rmed.2020.106094 [DOI] [PubMed] [Google Scholar]
- 18.Campo G,Pavasini R,Pollina A,Tebaldi M,Ferrari R. On-treatment platelet reactivity in patients with chronic obstructive pulmonary disease undergoing percutaneous coronary intervention. Thorax. 2014;69(1):80-81. doi: https://doi.org/10.1136/thoraxjnl-2013-203608 [DOI] [PubMed] [Google Scholar]
- 19.Offermanns S. Activation of platelet function through G protein-coupled receptors. Circ Res. 2006;99(12):1293-1304. doi: https://doi.org/10.1161/01.RES.0000251742.71301.16 [DOI] [PubMed] [Google Scholar]
- 20.Jones GL,Saroea HG,Watson RM,O'Byrne PM. Effect of an inhaled thromboxane mimetic (U46619) on airway function in human subjects. Am Rev Respir Dis. 1992;145(6):1270-1274. doi: https://doi.org/10.1164/ajrccm/145.6.1270 [DOI] [PubMed] [Google Scholar]
- 21.Barnes PJ,Chung KF,Page CP. Inflammatory mediators of asthma: an update. Pharmacol Rev. 1998;50(4):515-596. doi: https://doi.org/10.1124/pr.56.4.2 [PubMed] [Google Scholar]
- 22.Ekström MP,Hermansson AB,Ström KE. Effects of cardiovascular drugs on mortality in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2013;187(7):715-720.doi: https://doi.org/10.1164/rccm.201208-1565OC [DOI] [PubMed] [Google Scholar]
- 23.Short PM,Lipworth SIW,Elder DHJ,Schembri S,Lipworth BJ. Effect of beta blockers in treatment of chronic obstructive pulmonary disease: a retrospective cohort study. BMJ. 2011;342:d2549. doi: https://doi.org/10.1136/bmj.d2549 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goto T,Faridi MK,Camargo CA,Hasegawa K. The association of aspirin use with severity of acute exacerbation of chronic obstructive pulmonary disease: a retrospective cohort study. NPJ Prim Care Respir Med. 2018;28(1):7. doi: https://doi.org/10.1038/s41533-018-0074-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pavasini R,Biscaglia S,d'Ascenzo F,et al. Antiplatelet treatment reduces all-cause mortality in COPD patients: a systematic review and meta-analysis. COPD. 2016;13(4):509-514. doi: https://doi.org/10.3109/15412555.2015.1099620 [DOI] [PubMed] [Google Scholar]
- 26.Ellingsen J,Johansson G,Larsson K,et al. Impact of comorbidities and commonly used drugs on mortality in COPD - real-world data from a primary care setting. Int J Chron Obstruct Pulmon Dis. 2020;15:235-245. doi: https://doi.org/10.2147/COPD.S231296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aaron CP,Schwartz JE,Hoffman EA,et al. A longitudinal cohort study of aspirin use and progression of emphysema-like lung characteristics on CT imaging. Chest. 2018;154(1):41-50. doi: https://doi.org/10.1016/j.chest.2017.11.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fawzy A,Putcha N,Aaron CP,et al. Aspirin use and respiratory morbidity in COPD: a propensity score-matched analysis in Subpopulations and Intermediate Outcome Measures in COPD Study. Chest. 2019;155(3):519-527.doi: https://doi.org/10.1016/j.chest.2018.11.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kamath S,Blann AD,Lip GY. Platelet activation: assessment and quantification. Eur Heart J. 2001;22(17):1561-1571. doi: https://doi.org/10.1053/euhj.2000.2515 [DOI] [PubMed] [Google Scholar]
- 30.Henn V,Slupsky JR,Gräfe M,et al. CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature. 1998;391(6667):591-594. doi: https://doi.org/10.1038/35393 [DOI] [PubMed] [Google Scholar]
- 31.Frenette PS,Johnson RC,Hynes RO,Wagner DD. Platelets roll on stimulated endothelium in vivo: an interaction mediated by endothelial P-selectin. Proc Natl Acad Sci U S A. 1995;92(16):7450-7454. doi: https://doi.org/10.1073/pnas.92.16.7450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hansel NN,Putcha N,Woo H,et al. Randomized Clinical Trial of Air Cleaners to Improve Indoor Air Quality and COPD Health: results of the CLEAN AIR STUDY. Am J Respir Crit Care Med. 2022;205(4):421-430.doi: https://doi.org/10.1164/rccm.202103-0604OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.American Thoracic Society. Standardization of spirometry, 1994 Update. Am J Respir Crit Care Med. 1995;152(3):1107-1136. doi: https://doi.org/10.1164/ajrccm.152.3.7663792 [DOI] [PubMed] [Google Scholar]
- 34.Takx RAP,Jong PA,de Leiner T,et al. Automated coronary artery calcification scoring in non-gated chest CT: agreement and reliability. PLoS One. 2014;9(3):e91239. doi: https://doi.org/10.1371/journal.pone.0091239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shin JM,Kim TH,Kim JY,Park CH. Coronary artery calcium scoring on non-gated, non-contrast chest computed tomography (CT) using wide-detector, high-pitch and fast gantry rotation: comparison with dedicated calcium scoring CT. J Thorac Dis. 2020;12(10):5783-5793. doi: https://doi.org/10.21037/jtd-20-1371 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jones PW,Harding G,Berry P,Wiklund I,Chen WH,Leidy NK. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34(3):648-654. doi: https://doi.org/10.1183/09031936.00102509 [DOI] [PubMed] [Google Scholar]
- 37.Bestall J,Paul E,Garrod R,Garnham R,Jones P,Wedzicha J. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax. 1999;54(7):581-586. doi: https://doi.org/10.1136/thx.54.7.581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rubin BK,Ramirez O,Ohar JA. Iodinated glycerol has no effect on pulmonary function, symptom score, or sputum properties in patients with stable chronic bronchitis. Chest. 1996;109(2):348-352.doi: https://doi.org/10.1378/chest.109.2.348 [DOI] [PubMed] [Google Scholar]
- 39.Leidy NK,Rennard SI,Schmier J,Jones MKC,Goldman M. The breathlessness, cough, and sputum scale: the development of empirically based guidelines for interpretation. Chest. 2003;124(6):2182-2191. doi: https://doi.org/10.1378/chest.124.6.2182 [DOI] [PubMed] [Google Scholar]
- 40.van der Molen T,Willemse BWM,Schokker S,ten Hacken NHT,Postma DS,Juniper EF. Development, validity and responsiveness of the Clinical COPD Questionnaire. Health Qual Life Outcomes. 2003;1:13.doi: https://doi.org/10.1186/1477-7525-1-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jones PW,Quirk FH,Baveystock CM,Littlejohns P. A self-complete measure of health status for chronic airflow limitation. The St George's Respiratory Questionnaire. Am Rev Respir Dis. 1992;145(6):1321-1327. doi: https://doi.org/10.1164/ajrccm/145.6.1321 [DOI] [PubMed] [Google Scholar]
- 42.Cazzola M,Hanania NA,MacNee W,Rüdell K,Hackford C,Tamimi N. A review of the most common patient-reported outcomes in COPD - revisiting current knowledge and estimating future challenges. Int J Chron Obstruct Pulmon Dis. 2015;10:725-738. doi: https://doi.org/10.2147/COPD.S77368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kon SSC,Canavan JL,Jones SE,et al. Minimum clinically important difference for the COPD Assessment Test: a prospective analysis. Lancet Respir Med. 2014;2(3):195-203. doi: https://doi.org/10.1016/S2213-2600(14)70001-3 [DOI] [PubMed] [Google Scholar]
- 44.Jones PW. St. George's Respiratory Questionnaire: MCID. COPD. 2005;2(1):75-79.doi: https://doi.org/10.1081/COPD-200050513 [DOI] [PubMed] [Google Scholar]
- 45.Smid DE,Franssen FME,Houben-Wilke S,et al. Responsiveness and MCID estimates for CAT, CCQ, and HADS in patients with COPD undergoing pulmonary rehabilitation: a prospective analysis. J Am Med Dir Assoc. 2017;18(1):53-58. doi: https://doi.org/10.1016/j.jamda.2016.08.002 [DOI] [PubMed] [Google Scholar]
- 46.Sarwar A,Shaw LJ,Shapiro MD,et al. Diagnostic and prognostic value of absence of coronary artery calcification. JACC Cardiovasc Imaging. 2009;2(6):675-688. doi: https://doi.org/10.1016/j.jcmg.2008.12.031 [DOI] [PubMed] [Google Scholar]
- 47.Howard AL. Leveraging time-varying covariates to test within- and between-person effects and interactions in the multilevel linear model. Emerg Adulthood. 2015;3(6):400-412. doi: https://doi.org/10.1177/2167696815592726 [Google Scholar]
- 48.Davì G,Catalano I,Averna M,et al. Thromboxane biosynthesis and platelet function in type II diabetes mellitus. N Engl J Med. 1990;322(25):1769-1774. doi: https://doi.org/10.1056/NEJM199006213222503 [DOI] [PubMed] [Google Scholar]
- 49.Eldor A,Lellouche F,Goldfarb A,Rachmilewitz EA,Maclouf J. In vivo platelet activation in β-thalassemia major reflected by increased platelet-thromboxane urinary metabolites. Blood. 1991;77(8):1749-1753. doi: https://doi.org/10.1182/blood.V77.8.1749.1749 [PubMed] [Google Scholar]
- 50.Patrono C,Rocca B. Measurement of thromboxane biosynthesis in health and disease. Front Pharmacol. 2019;10:1244. doi: https://doi.org/10.3389/fphar.2019.01244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fijnheer R,Frijns CJ,Korteweg J,et al. The origin of P-selectin as a circulating plasma protein. Thromb Haemost. 1997;77(6):1081-1085. doi: https://doi.org/10.1055/s-0038-1656116 [PubMed] [Google Scholar]
- 52.Heeschen C,Dimmeler S,Hamm CW,et al. Soluble CD40 ligand in acute coronary syndromes. New Engl J Med. 2003;348(12):1104-1111. doi: https://doi.org/10.1056/NEJMoa022600 [DOI] [PubMed] [Google Scholar]
- 53.Michelson AD,Barnard MR,Krueger LA,Valeri CR,Furman MI. Circulating monocyte-platelet aggregates are a more sensitive marker of in vivo platelet activation than platelet surface P-selectin: studies in baboons, human coronary intervention, and human acute myocardial infarction. Circulation. 2001;104(13):1533-1537. doi: https://doi.org/10.1161/hc3801.095588 [DOI] [PubMed] [Google Scholar]
- 54.Gurney D,Lip GYH,Blann AD. A reliable plasma marker of platelet activation: does it exist? Am J Hematol. 2002;70(2):139-144. doi: https://doi.org/10.1002/ajh.10097 [DOI] [PubMed] [Google Scholar]
- 55.Pietravalle F,Lecoanet-Henchoz S,Blasey H,et al. Human native soluble CD40L is a biologically active trimer, processed inside microsomes (∗). J Biol Chem. 1996;271(11):5965-5967. doi: https://doi.org/10.1074/jbc.271.11.5965 [DOI] [PubMed] [Google Scholar]
- 56.Gresele P,Lopez JA,Kleiman NS,Page CPeds. Platelets in Thrombotic and Non-thrombotic Disorders. Pathophysiology, Pharmacology and Rherapeutics: An Update. Springer International Publishing:2017. doi: https://doi.org/10.1007/978-3-319-47462-5 [Google Scholar]
- 57.Furman MI,Krueger LA,Linden MD,Barnard MR,Frelinger AL,Michelson AD. Release of soluble CD40L from platelets is regulated by glycoprotein IIb/IIIa and actin polymerization. J Am Coll Cardiol. 2004;43(12):2319-2325. doi: https://doi.org/10.1016/j.jacc.2003.12.055 [DOI] [PubMed] [Google Scholar]
- 58.Allen IC,Hartney JM,Coffman TM,Penn RB,Wess J,Koller BH. Thromboxane A2 induces airway constriction through an M3 muscarinic acetylcholine receptor-dependent mechanism. Am J Physiol Lung Cell Mol Physiol. 2006;290(3):L526-L533. doi: https://doi.org/10.1152/ajplung.00340.2005 [DOI] [PubMed] [Google Scholar]
- 59.Aizawa H,Inoue H,Nakano H,et al. Effects of thromboxane A2 antagonist on airway hyperresponsiveness, exhaled nitric oxide, and induced sputum eosinophils in asthmatics. Prostaglandins, Leukot Essent Fatty Acids. 1998;59(3):185-190. doi: https://doi.org/10.1016/S0952-3278(98)90061-8 [DOI] [PubMed] [Google Scholar]
- 60.Hoshino M,Sim J,Shimizu K,Nakayama H,Koya A. Effect of AA-2414, a thromboxane A2 receptor antagonist, on airway inflammation in subjects with asthma. J Allergy Clin Immunol. 1999;103(6):1054-1061. doi: https://doi.org/10.1016/S0091-6749(99)70179-X [DOI] [PubMed] [Google Scholar]
- 61.Nakamura Y,Tamaoki J,Nagase H,et al. Japanese guidelines for adult asthma 2020. Allergol Int. 2020;69(4):519-548. doi: https://doi.org/10.1016/j.alit.2020.08.001 [DOI] [PubMed] [Google Scholar]
- 62.Pistenmaa C l.,Dashnaw S,Hoffman E a.,et al. Pulmonary perfusion in alpha-1 antitrypsin deficiency: a randomized crossover study. Am J Respir Crit Care Med. 2021;203:A2269. doi: https://doi.org/10.1164/ajrccm-conference.2021.203.1_MeetingAbstracts.A2269 [Google Scholar]