Abstract
The American College of Radiology Breast Imaging Reporting and Data System (BI-RADS) classification has been used for the diagnosis of breast masses for several decades and constantly updated, but the terminology used to describe breast ultrasound findings is still evolving and a great amount of large sample data is necessary to verify and improve ultrasound BI-RADS. The objective of the present study was to explore the value of ultrasound Breast Imaging Reporting and Data System (US BI-RADS) classification in the preoperative evaluation of the US-guided Mammotome-assisted minimally invasive resection of breast masses. A total of 1,028 patients with 1,341 breast masses from a single hospital were selected for retrospective analysis. All patients underwent minimally invasive resection using a US-guided Mammotome device, and postoperative pathological examinations were performed for all samples. The preoperative US BI-RADS classification and postoperative pathological examination results were compared and analyzed. A receiver operating characteristic (ROC) curve was used to analyze the preoperative evaluation efficacy of the US BI-RADS classification in US-guided Mammotome-assisted minimally invasive breast mass resection. Among the 1,341 breast masses that underwent resection, 1,307 were benign and 34 were malignant. The specificity, sensitivity, accuracy, positive predictive value and negative predictive value of the US BI-RADS classification in the preoperative diagnosis of malignant breast masses were 83.47, 100.00, 83.89, 13.60 and 100.00%, respectively, and the area under the ROC curve was 0.917. It may be concluded that the US BI-RADS classification has a good preoperative diagnostic performance and can provide an accurate assessment prior to Mammotome-assisted minimally invasive resection. It may help surgeons to make reasonable decisions for subsequent therapy and therefore is worthy of further clinical use.
Keywords: Breast masses, ultrasound Breast Imaging Reporting and Data System, Mammotome minimally invasive resection
Introduction
Breast masses are one of the most common diseases in women. According to the 2020 GLOBOCAN cancer estimates, female breast cancer is the fifth leading cause of cancer mortality worldwide with 685,000 deaths; it also ranks first for incidence among women in the vast majority of countries (1). Therefore, an increasing number of surgical procedures are performed on patients with breast masses that may be breast cancer. With the continuous development of minimally invasive breast technology and focus on the aesthetic needs of patients, minimally invasive resection using the ultrasound (US)-guided Mammotome system has been widely used for the accurate biopsy of suspicious lesions or removal of benign breast masses (2,3), which is particularly efficient for multifocal, tiny and impalpable breast lesions. It has the advantages of being minimally invasive, resulting in tiny scars, having a high accuracy and good performance, and being safe while providing real-time visualization (4,5). Despite these advantages, it is nonetheless an invasive procedure and varying degrees of breast tissue injury can occur. Therefore, the precise preoperative assessment of breast masses is imperative when performing Mammotome minimally invasive resection, as it helps to avoid unnecessary biopsy or surgery.
The Breast Imaging Reporting and Data System (BI-RADS) (6) is a tool developed by the American College of Radiology (ACR). It has unified and standardized the risk stratification of breast masses in terms of breast lesion characteristics observed in different types of imaging, including mammography, US and magnetic resonance imaging. This standardized system is used for the identification of pathology based on various medical imaging methods, which facilitates reproducibility and consistency and decreases the subjectivity of breast mass diagnosis. It provides a means of communication between radiologists and surgeons via a lexicon of feature descriptors, structured reports based on assessment categories and management recommendations, and a framework for collecting and auditing data (7).
Although the US BI-RADS classification unifies the descriptive terms and risk stratification of the lesions, the terminology used to describe breast US findings is evolving, and the diversity of this terminology may cause confusion. Therefore, its practical application efficacy varies and an abundance of large-sample data is required to verify and improve US BI-RADS, adapt to changes in the practice of breast imaging and improve its practical use for interpreting physicians. Although the US BI-RADS classification has been used for the diagnosis of breast masses for several decades, few studies have combined minimally invasive breast surgery with US BI-RADS classification. In the present study, ultrasonography was used to guide the Mammotome minimally invasive system and to assess and classify the breast lesions. Minimally invasive breast surgery performed using the US-guided Mammotome system was combined with US BI-RADS in a large sample of patients with breast masses. The preoperative US BI-RADS classification and postoperative pathology results were retrospectively analyzed to explore the effectiveness of applying this classification in the preoperative evaluation of breast masses.
Materials and methods
Patients and breast masses
The study was approved by the Institutional Review Committee of Tongji Hospital of Huazhong University of Science and Technology (Wuhan, China). All patients gave their consent for the presentation of their data in this publication. They had preoperative conversations with the surgeons and provided written informed consent to participate in the study. A total of 1,028 patients with 1,341 breast masses who underwent minimally invasive resection with a US-guided Mammotome device from September 2018 to February 2019 were selected. All patients were female, ranging from 16 to 75 years in age, with a median age of 40 years. The maximum diameter of the breast masses ranged from 0.3 to 3.2 cm, with a median of 1.5 cm. The exclusion criteria were as follows: i) The mass was close to the nipple, and breast-feeding or avoidance of breast duct damage was necessary; ii) the mass was close to the marginal area of the breast, with minimal normal breast tissue around the mass; iii) the mass was too close to the skin and the risk of cutting off the skin was too high; iv) patients with bleeding tendency, blood coagulation disorder and associated disorders; v) patients with serious systemic diseases who were unable to tolerate surgery; and vi) masses with previous intervention affecting the judgment of US images.
Instruments and methods. Breast ultrasonography and body marking
All breast ultrasonography was performed with a Mindray DC-8 color Doppler US diagnostic system (Mindray Medical International Ltd.), equipped with L12-3E linear-array transducers. Generally, the patients assumed a supine position and completely exposed the mass-containing breast. However, if necessary, the patients were placed in a semi-lateral position that was suitable for minimally invasive resection using the Mammotome device. Conventional US images of the breast lesions were acquired and saved, including B-mode and color Doppler flow mode images. The lesion position and B-mode US characteristics, including size, shape, echo pattern, margin, growth orientation, posterior features, calcifications, presence of architectural distortion and duct changes were recorded. Vascularity was classified into three patterns, namely absent, internal vascularity and vessels in the rim, by color Doppler flow according to US BI-RADS. At the end of the examination, the location of the breast lesion was marked on the skin.
Minimally invasive resection using a US-guided Mammotome
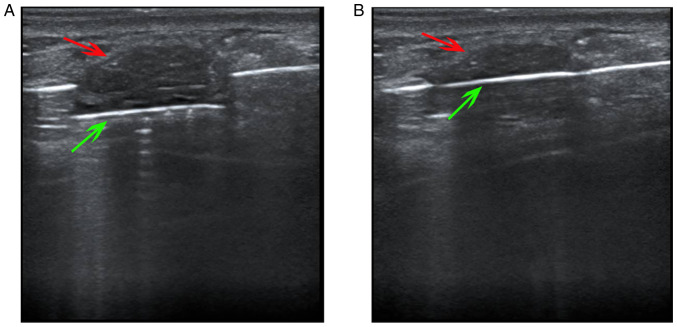
All minimally invasive surgical procedures were conducted using a vacuum-assisted Mammotome biopsy system (Devicor Medical Products, Inc.) with the following components: 8G Mammotome rotary cutter, control handle, vacuum suction pump and associated software (Mammotome EX SCMSW5). While undergoing routine sterilization, the patient was placed in a supine or semi-lateral position with their ipsilateral arm lifted up and then draped with a surgical towel. A moderate anesthetic (local anesthesia, 1% lidocaine ≤200 mg.) was administered subcutaneously and underneath the posterior breast space in the surgical area. A ~3-mm incision was made in the predetermined location, which allowed for the proper insertion of the 8G Mammotome needle. The needle was placed underneath the deep surface of the breast mass by US guidance at an appropriate angle so that the breast mass was just inside the groove of the needle (Fig. 1). Repeated rotary cutting was performed to remove the aspirated lesion tissue until no residual lesions were detected in the US images. After completion of the resection, hemostasis was performed in the surgical area to stop bleeding. Compression bandages were applied to all patients for 72 h following the procedure.
Figure 1.
US-guided Mammotome-assisted minimally invasive resection of a breast mass. (A) The needle was positioned underneath the deep surface of the breast mass with US guidance, and the rotary cutter opened so that the breast mass was just in the groove of needle. (B) The rotary cutter was closed to remove the aspirated lesion tissue. Red arrows indicate breast mass, and green arrows indicate rotary cutter. US, ultrasound.
US BI-RADS classification. Breast mass classification was based on the latest edition of the US BI-RADS recommendations of the ACR (8). Two physicians with >10 years of breast US experience determined the US BI-RADS classification. If the analysis results were inconsistent, the two physicians discussed the results together until a consensus was reached. According to the US BI-RADS management recommendations, category 3 lesions should have a short (6-month) follow-up interval or continued surveillance, while category 4 lesions require biopsy for tissue diagnosis. As there is a marked difference in the treatment of category 3 and 4 lesions by clinicians, category 3 lesions were defined as benign and lesions of category 4 and above were defined as malignant in the present study.
Statistical analysis
SPSS19.0 (IBM Corp.) statistical analysis software was used to analyze the diagnostic efficacy of US BI-RADS classification in breast masses that underwent minimally invasive resection using the Mammotome system. To detect statistical differences in lesion characteristics, χ2-test was used for shape while Fisher's exact test was used for other characteristics. The specificity, sensitivity, accuracy, positive predictive value and negative predictive value were calculated by comparison with pathology results. The receiver operating characteristic (ROC) curve for the US BI-RADS classification in the diagnosis of breast masses subjected to minimally invasive surgery was constructed, and the area under curve (AUC) was calculated.
Results
Lesion position and US characteristics
The position and main ultrasonographic features of benign and malignant lesions were analyzed, and the results are shown in Table I. Significant statistical differences in shape, orientation, margin, posterior features, calcifications and architectural distortion were detected between benign and malignant lesions (P<0.001). However, no significant difference in position, echo pattern, duct changes and vascularity was found.
Table I.
Position and ultrasound characteristics of benign and malignant breast masses removed by ultrasound-guided Mammotome-assisted minimally invasive resection.
| Lesion characteristics | Benign, n | Malignant, n | P-value |
|---|---|---|---|
| Position | 0.182 | ||
| Upper outer quadrant | 667 | 12 | |
| Upper inner quadrant | 307 | 9 | |
| Lower outer quadrant | 175 | 8 | |
| Lower inner quadrant | 158 | 5 | |
| Shape | <0.001 | ||
| Oval | 987 | 10 | |
| Round | 124 | 9 | |
| Irregular | 196 | 15 | |
| Orientation | <0.001 | ||
| Parallel | 1,197 | 20 | |
| Not parallel | 110 | 14 | |
| Margin | <0.001 | ||
| Circumscribed | 1,201 | 11 | |
| Not circumscribed | 106 | 23 | |
| Echo pattern | 1.000 | ||
| Hypoechoic | 1,286 | 34 | |
| Isoechoic | 9 | 0 | |
| Complex cystic and solid | 12 | 0 | |
| Posterior features | <0.001 | ||
| No posterior features | 901 | 19 | |
| Enhancement | 369 | 0 | |
| Shadowing | 29 | 15 | |
| Combined pattern | 8 | 0 | |
| Calcifications | <0.001 | ||
| Microcalcifications | 0 | 13 | |
| No microcalcifications | 1,307 | 21 | |
| Architectural distortion | <0.001 | ||
| Yes | 0 | 9 | |
| No | 1,307 | 25 | |
| Duct changes | 0.207 | ||
| Yes | 8 | 1 | |
| No | 1,299 | 33 | |
| Vascularity | 0.162 | ||
| Absent | 536 | 11 | |
| Internal vascularity | 277 | 12 | |
| Vessels in the rim | 494 | 11 |
Pathology results
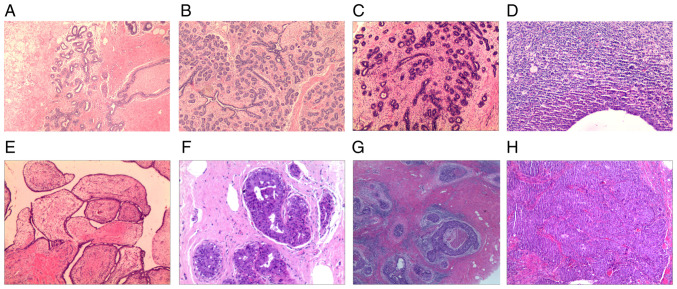
Among the 1,028 patients with 1,341 breast masses who underwent minimally invasive resection, there were 1,307 benign lesions, including adenosis, fibroadenosis, sclerosing adenosis, fibrocystic breast disease, adenosis with fibroadenomatous nodules, fibroadenoma, intraductal papilloma, benign phyllodes tumor, inflammatory lesions and other benign lesions. There were 34 malignant lesions, including ductal carcinoma in situ and invasive carcinoma. The detailed pathology results of this study are summarized in Table II and typical pathological images are displayed in Fig. 2.
Table II.
Pathological results of breast masses removed by ultrasound-guided Mammotome-assisted minimally invasive resection.
| Pathology | n (%) |
|---|---|
| Adenosis | 343 (25.58) |
| Fibroadenosis | 57 (4.25) |
| Sclerosing adenosis | 6 (0.45) |
| Fibrocystic breast disease | 5 (0.37) |
| Adenosis with fibroadenomatous nodules | 709 (52.87) |
| Fibroadenoma | 138 (10.29) |
| Intraductal papilloma | 29 (2.16) |
| Benign phyllodes tumor | 5 (0.37) |
| Inflammatory lesion | 3 (0.22) |
| Other benign lesions | 12 (0.89) |
| Invasive carcinoma | 23 (1.72) |
| Ductal carcinoma in situ | 11 (0.82) |
Figure 2.
Representative pathology images. (A) Breast adenosis (magnification, x40), (B) breast adenosis with fibroadenomatous nodules (magnification, x40), (C) breast fibroadenoma (magnification, x40), (D) breast inflammatory lesion (magnification, x100), (E) breast benign phyllodes tumor (magnification, x40), (F) intraductal papilloma (magnification, x100), (G) ductal carcinoma in situ (magnification, x40) and (H) invasive carcinoma (magnification, x40). Hematoxylin and eosin staining.
Comparative analysis of US BI-RADS classification and pathology results
Taking pathology results as the gold standard, according to the US BI-RADS classification, there were 1,091 category 3 masses, of which 1091 (100.00%) were benign and 0 (0.00%) were malignant. There were also 188 category 4a masses with 181 benign (96.28%) and 7 malignant (3.72%), 50 category 4b masses with 35 benign (70.00%) and 15 malignant (30.00%), 10 category 4c masses with 0 benign (0.00%) and 10 malignant (100.00%), and 2 category 5 masses with 0 benign (0.00%) and 2 malignant (100.00%) (Table III).
Table III.
Comparative analysis of US BI-RADS classification and pathological results of breast masses removed by ultrasound-guided Mammotome minimally invasive resection
| US BI-RADS classification, n (%) | ||||||
|---|---|---|---|---|---|---|
| Pathology results | Category 3 | Category 4a | Category 4b | Category 4c | Category 5 | Total, n (%) |
| Benign | 1,091 (100.00) | 181 (96.28) | 35 (70.00) | 0 (0.00) | 0 (0.00) | 1,307 (97.46) |
| Malignant | 0 (0.00) | 7 (3.72) | 15 (30.00) | 10 (100.00) | 2 (100.00) | 34 (2.54) |
| Total | 1,091 (81.36) | 188 (14.02) | 50 (3.73) | 10 (0.75) | 2 (0.15) | 1,341 |
US, ultrasound; BI-RADS, Breast Imaging Reporting and Data System.
Diagnostic efficacy of US BI-RADS classification in patients with breast masses undergoing Mammotome-assisted resection
The specificity, sensitivity, accuracy, positive predictive value and negative predictive value of the US BI-RADS classification in the diagnosis of the patients were 83.47, 100.00, 83.89, 13.60 and 100.00%, respectively (Table IV). The AUC calculated from the corresponding ROC curve was 0.917 (Fig. 3).
Table IV.
Diagnostic efficacy of the ultrasound Breast Imaging Reporting and Data System in breast masses undergoing Mammotome-assisted minimally invasive resection.
| Statistic | Result, % |
|---|---|
| Specificity | 83.47 |
| Sensitivity | 100.00 |
| Accuracy | 83.89 |
| PPV | 13.60 |
| NPV | 100.00 |
PPV, positive predictive value; NPV, negative predictive value.
Figure 3.
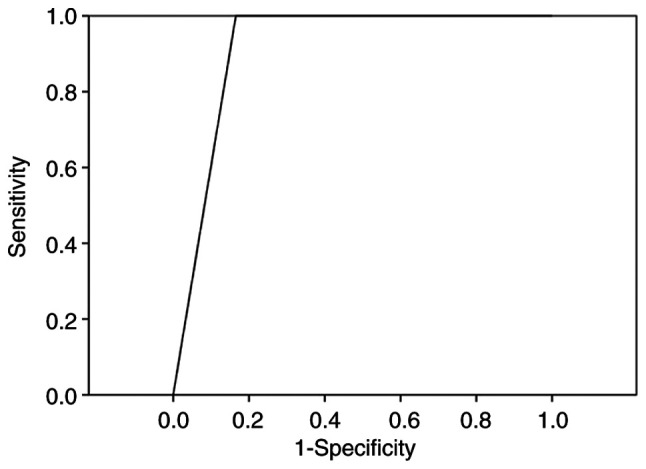
ROC curve of ultrasound Breast Imaging Reporting and Data System assessment in the diagnosis of breast masses undergoing Mammotome-assisted minimally invasive surgery. The area under the curve calculated from the ROC curve was 0.917. ROC, receiver operating characteristic.
Discussion
With the increasing prevalence of breast disease screening and health consciousness of the population, the early detection rate of breast masses has increased significantly. The risk of breast cancer has also increased markedly for several reasons, including lifestyle and diet changes, as well as genetic, environmental and drug-associated factors (9). Subsequently, greater numbers of patients experience psychological anxiety, such as carcinophobia, due to the presence of breast masses, and choose to find out the pathological nature of their lesions and undergo surgical resection. Thus, it is very important for the surgeon to provide a reasonable therapy plan. A meta-analysis (10) comparing Mammotome vacuum-assisted biopsy with open excision for benign breast lesions indicated that the procedure using a hand-held Mammotome device was advantageous with respect to the size of the skin incision, intraoperative blood loss, surgery and healing times, scar size, wound infection and cosmetic breast deformity, with additional advantages including real-time and dynamic observation, high accuracy, minimal pain during the procedure, high patient satisfaction, a smaller incision and improved aesthetic appearance when compared with that achieved using conventional open surgery (11,12). Among the 1,341 breast masses in the present study, 1,328 lesions were completely removed under US guidance via the Mammotome minimally invasive system; there were only 13 lesions that could not be completely removed due to hard calcifications in the lesions that abraded the Mammotome needle, causing the surgeon to switch to a conventional open surgical approach.
The BI-RADS has been designed by the ACR to standardize the reporting of breast imaging findings, reduce uncertainty in the interpretation of images and the recommendations based on them, and facilitate outcome monitoring. The US BI-RADS classification provides consistent and standardized terminology for the assessment of breast masses using feature descriptors including mass, shape, orientation, margin, internal echo pattern, posterior echo features, calcification, associated features and special cases (8). Several feature descriptors were analyzed in the present study, and some US features were found to overlap between benign and malignant lesions. There were also statistically significant differences in shape, orientation, margin, posterior features, microcalcifications and architectural distortion; however, the position, echo pattern, duct changes and vascularity did not significantly differ. These findings indicate that the accurate assessment of breast masses requires the analysis of multiple feature descriptors rather than any single characteristic. In the present study, there were 1,091 category 3 masses, all of which were benign, and the negative predictive value was 100.00%. This is consistent with the 0-2% likelihood of cancer for US BI-RADS category 3 lesions, indicating that the US BI-RADS system had high diagnostic accuracy for category 3 lesions in the present study. Furthermore, the sensitivity was 100.00% and all the pathological malignant lesions were classified as category 4 or above, indicating that no pathological malignant tumors were misdiagnosed using US BI-RADS and the diagnostic efficacy of US BI-RADS in the diagnosis of true positive lesions was good. The specificity and accuracy were 83.47 and 83.89%, respectively, and the AUC was 0.917, which further validated the good preoperative diagnostic performance of US BI-RADS. However, the positive predictive value was only 13.60% due to false positives in the category 4a and 4b lesions in the present study. This upgraded classification may be due to lesion features such as irregular shape, different sized calcifications and posterior shadowing. The low positive predictive value and the presence of false positive cases can be compensated for by combining US BI-RADS with elastography or contrast-enhanced US (13-17). Furthermore, given its high accuracy, cost effectiveness, low complication rate and convenience, core needle biopsy is the most common method used for impalpable and palpable lesions (18), and most suspicious lesions undergo an initial core needle biopsy first. Therefore, in the present study category 4c and 5 lesions were rare and the majority of lesions that underwent minimally invasive resection using a Mammotome device were category 3. The diagnosis of typical benign category 3 lesions using the US BI-RADS classification was found to be relatively easy and accurate, and provides satisfactory diagnostic efficiency according to the results of the present study.
In summary, the present study demonstrates that the US BI-RADS classification has good preoperative diagnostic effectiveness for breast masses undergoing US-guided Mammotome-assisted minimally invasive resection, particularly for category 3 lesions and true positive lesions. Therefore, it can effectively help surgeons to make reasonable decisions for subsequent therapy and is recommended for further clinical use.
Acknowledgements
Not applicable.
Funding Statement
Funding: The present study was supported by Hubei Province Health and Family Planning Scientific Research Project (grant no. WJ2017M081).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
YP and HW organized and designed the study. HW performed US guidance work during the study and wrote the manuscript. YZ recorded and collected the raw data. QW and HW confirm the authenticity of all the raw data and performed the data analysis. YP and HW performed the statistical analysis and reviewed the study. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The study was approved by the Institutional Review Committee of Tongji Hospital of Huazhong University of Science and Technology (approval no. TJ-IRB2021916). All patients provided written informed consent to participate.
Patient consent for publication
All patients gave their consent for the presentation of their data and materials in this publication.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Fang M, Liu G, Luo G, Wu T. Feasibility and safety of image-guided vacuum-assisted breast biopsy: A PRISMA-compliant systematic review and meta-analysis of 20 000 population from 36 longitudinal studies. Int Wound J. 2019;16:1506–1512. doi: 10.1111/iwj.13224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Li SJ, Hao XP, Hua B, Wang JD, Fan ZM. Clinical practice guidelines for ultrasound-guided vacuum-assisted breast biopsy: Chinese Society of Breast Surgery (CSBrS) practice guidelines 2021. Chin Med J (Engl) 2021;134:1390–1392. doi: 10.1097/CM9.0000000000001508. Chinese Society of Breast Surgery. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li X, Gao H, Xu M, Wu Y, Gao D. Breast papillary lesions diagnosed and treated using ultrasound-guided vacuum-assisted excision. BMC Surg. 2020;20(204) doi: 10.1186/s12893-020-00869-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pistolese CA, Castrignano A, Ricci F, Meucci R, Croce G, Mondillo M, Collura A, Perretta T, Floris R. Ultrasound-Guided vacuum-assisted biopsy in small breast: A cost-saving solution. Clin Breast Cancer. 2019;19:e352–e357. doi: 10.1016/j.clbc.2018.12.002. [DOI] [PubMed] [Google Scholar]
- 6.D'Orsi CJ, Mendelson EB, Morris EA. ACR BI-RADS® Atlas, Breast Imaging Reporting and Data System. American College of Radiology, Reston, VA, 2013. [Google Scholar]
- 7.Spak DA, Plaxco JS, Santiago L, Dryden MJ, Dogan BE. BI-RADS(R) fifth edition: A summary of changes. Diagn Interv Imaging. 2017;98:179–190. doi: 10.1016/j.diii.2017.01.001. [DOI] [PubMed] [Google Scholar]
- 8.Mendelson EB, Berg WA. ACR BI-RADS® Ultrasound. In: ACR BI-RADS® Atlas, Breast Imaging Reporting and Data System. American College of Radiology, Reston, VA, 2013. [Google Scholar]
- 9.Lukasiewicz S, Czeczelewski M, Forma A, Baj J, Sitarz R, Stanislawek A. Breast cancer-epidemiology, risk factors, classification, prognostic markers, and current treatment strategies-an updated review. Cancers (Basel) 2021;13(4287) doi: 10.3390/cancers13174287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ding B, Chen D, Li X, Zhang H, Zhao Y. Meta analysis of efficacy and safety between mammotome minimally invasive operation and open excision for benign breast tumor. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2013;38:291–300. doi: 10.3969/j.issn.1672-7347.2013.03.013. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 11.Papathemelis T, Heim S, Lux MP, Erhardt I, Scharl A, Scharl S. Minimally invasive breast fibroadenoma excision using an ultrasound-guided vacuum-assisted biopsy device. Geburtshilfe Frauenheilkd. 2017;77:176–181. doi: 10.1055/s-0043-100387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jiang Y, Lan H, Ye Q, Jin K, Zhu M, Hu X, Teng L, Cao F, Lin X. Mammotome(R) biopsy system for the resection of breast lesions: Clinical experience in two high-volume teaching hospitals. Exp Ther Med. 2013;6:759–764. doi: 10.3892/etm.2013.1191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cantisani V, David E, Barr RG, Radzina M, de Soccio V, Elia D, De Felice C, Pediconi F, Gigli S, Occhiato R, et al. US-Elastography for breast lesion characterization: Prospective comparison of US BIRADS, strain elastography and shear wave elastography. Ultraschall Med. 2021;42:533–540. doi: 10.1055/a-1134-4937. [DOI] [PubMed] [Google Scholar]
- 14.Zheng X, Li F, Xuan ZD, Wang Y, Zhang L. Combination of shear wave elastography and BI-RADS in identification of solid breast masses. BMC Med Imaging. 2021;21(183) doi: 10.1186/s12880-021-00702-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ding Z, Liu W, He N, Ma X, Fu L, Ye L. Value of ultrasound elastography combined with contrast-enhanced ultrasound and micro-flow imaging in differential diagnosis of benign and malignant breast lesions. Am J Transl Res. 2021;13:13941–13949. [PMC free article] [PubMed] [Google Scholar]
- 16.Cheng M, Tong W, Luo J, Li M, Liang J, Pan F, Pan J, Zheng Y, Xie X. Value of contrast-enhanced ultrasound in the diagnosis of breast US-BI-RADS 3 and 4 lesions with calcifications. Clin Radiol. 2020;75:934–941. doi: 10.1016/j.crad.2020.07.017. [DOI] [PubMed] [Google Scholar]
- 17.Janu E, Krikavova L, Little J, Dvorak K, Brancikova D, Jandakova E, Pavlik T, Kovalcikova P, Kazda T, Valek V. Prospective evaluation of contrast-enhanced ultrasound of breast BI-RADS 3-5 lesions. BMC Med Imaging. 2020;20(66) doi: 10.1186/s12880-020-00467-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Park HL, Kim KY, Park JS, Shin JE, Kim HR, Yang B, Kim JY, Shim JY, Shin EA, Noh SM. Clinicopathological analysis of ultrasound-guided vacuum-assisted breast biopsy for the diagnosis and treatment of breast disease. Anticancer Res. 2018;38:2455–2462. doi: 10.21873/anticanres.12499. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.