Abstract
Aims
Patient decision aids have previously demonstrated an improvement in the quality of the informed consent process. This study assessed the effectiveness of detailed written patient information, compared to standard verbal consent, in improving postoperative recall in adult orthopaedic trauma patients.
Methods
This randomized controlled feasibility trial was conducted at two teaching hospitals within the South Eastern Sydney Local Health District. Adult patients (age ≥ 18 years) pending orthopaedic trauma surgery between March 2021 and September 2021 were recruited and randomized to detailed or standard methods of informed consent using a random sequence concealed in sealed, opaque envelopes. The detailed group received procedure-specific written information in addition to the standard verbal consent. The primary outcome was total recall, using a seven-point interview-administered recall questionnaire at 72 hours postoperatively. Points were awarded if the participant correctly recalled details of potential complications (maximum three points), implants used (maximum three points), and postoperative instructions (maximum one point). Secondary outcomes included the anxiety subscale of the Hospital and Anxiety Depression Scale (HADS-A) and visual analogue scale (VAS) for pain collected at 24 hours preoperatively and 72 hours postoperatively. Additionally, the Patient Satisfaction Questionnaire Short Form (PSQ-18) measured satisfaction at 72 hours postoperatively.
Results
A total of 60 patients were randomized, 32 to the standard group and 28 to the detailed group. Patients in the detailed group had significantly higher total recall score compared to the standard group (mean difference 1.29 points (95% confidence interval (CI) 0.51 to 2.08); p = 0.002). There were no differences in HADS-A (mean difference 0.39 (95% CI -2.11 to 2.88); p = 0.757), VAS pain (mean difference 5.71 (95% CI -22.25 to 11.11); p = 0.499), or PSQ-18 (mean difference 0.499; 95% CI -1.6 to 3.42; p = 0.392).
Conclusion
Detailed written tools are useful in improving postoperative recall in adult orthopaedic trauma patients.
Cite this article: Bone Jt Open 2023;4(2):104–109.
Keywords: Orthopaedic trauma, Informed consent, Detailed written tool, Patient recall, Orthopaedic Trauma, randomized controlled trial, anxiety, Hospital and Anxiety Depression Scale, Orthopaedic trauma surgeries, visual analogue scale (VAS) for pain, VAS pain, t-test, Anesthesiologists, orthopaedic operations
Introduction
Informed consent is a legal prerequisite for clinical treatment in healthcare. Orthopaedic trauma surgeries are common and complex procedures with well-documented risks. Patients often receive large amounts of information about the likely course of their treatment, to allow them to make informed decisions. However, there are concerns regarding the quality of consent in the context of orthopaedic trauma,1 and a higher proportion of trauma patients (up to 57%) have no recollection of potential complications and postoperative instructions when compared to elective patients (26%).2
Atrey et al3 reviewed successful litigation cases in the UK and found that a “poor consent process” was a common theme in successful litigations. They specifically noted cases where recognized complications of common orthopaedic operations were not disclosed. Litigation costs due to poor consent had an average payment of USD $136,178 per case.
Patient decision aids (PtDAs) are important educational interventions used to promote shared decision-making and provide more patient-centred care.4 Studies have supported the use of written information to supplement informed consent. PtDAs such as leaflets, web-based information, and booklets have improved retention of important surgical information in the elective orthopaedic surgery setting.5-7 However, little is known about the effectiveness of these communication methods in the emergency and trauma setting. The primary aim of this study is to assess the effectiveness of a procedure-specific detailed written tool in improving postoperative recall of information in adult orthopaedic trauma patients. Secondary outcomes include patient perioperative anxiety using the anxiety subscale of the Hospital and Anxiety Depression Scale (HADS-A)8 and perioperative pain at 24 hours preoperatively and 72 hours postoperatively. Postoperative satisfaction using the Patient Satisfaction Questionnaire Short-Form (PSQ-18)9 was measured at 72 hours postoperatively. The recruitment rate of eligible patients was measured at the end of the trial.
Methods
Study design
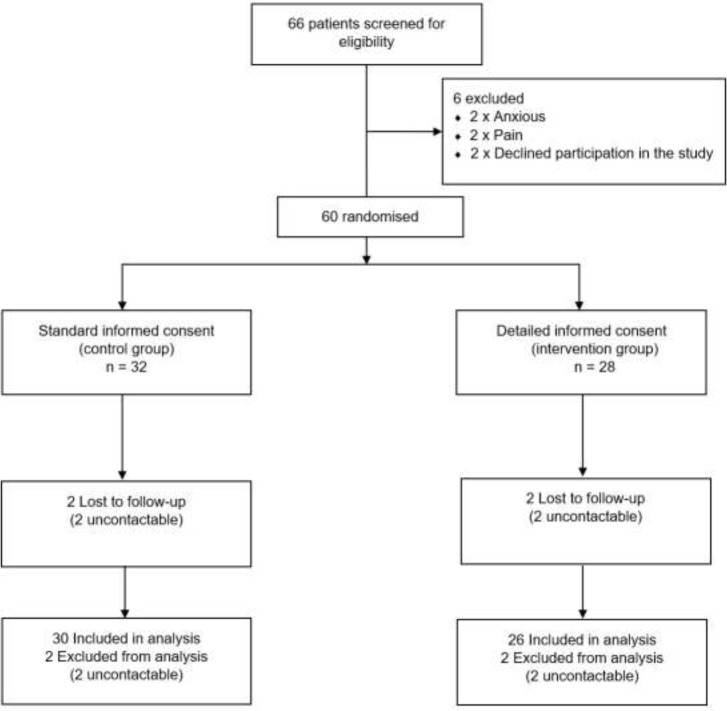
This study was a multicentre, randomized controlled parallel feasibility trial with two arms: standard informed consent method and detailed written informed consent method (Figure 1). Participants were recruited over a period of seven months (March 2021 to September 2021) at two teaching hospitals within the South Eastern Sydney Local Health District (SESLHD): St George Hospital and The Sutherland Hospital. The study protocol was approved by the SESLHD Human Research and Ethics Committee (2019/ETH13394). The trial was registered with anzctr.org.au (ACTRN12621000852853).
Fig. 1.
Consolidated Standards of Reporting Trials flow diagram of study design and patient enrolment methods.
Participants
Patients were eligible if they presented with a confirmed distal radius, ankle, femoral shaft, tibial shaft, distal femur, or proximal tibia fracture requiring surgery, were aged > 18 years, were able to speak and read the English language, and had capacity to make healthcare decisions independently. Patients were deemed ineligible if they were medically diagnosed with cognitive impairment or were otherwise incapacitated, had limited literacy, or had revision surgery.
Randomization and blinding
A researcher (AMA) screened patients and obtained verbal consent for participation in the trial, prior to any information being provided about their expected treatment. Using simple randomization, the computer-generated 1:1 ratio sequence was concealed in consecutively numbered, sealed, opaque envelopes, which were opened by the researcher when a participant consented to the trial. The patient was then informed about their treatment using their allocated method by a member of the surgical team, which included both trainee and consultant surgeons. Patients were asked to complete a consent form for their surgery and a participant information form for the study. Participants and researchers were not blinded to allocation.
Standard method (control group)
The standard method of informed consent was the usual method of providing information to patients at the two hospitals. A member of the surgical team provided a verbal explanation of the procedure including surgical complications, potential benefits, details of the injury, and the proposed surgical management. The patient was then provided with an opportunity to ask any questions about the surgery.
Detailed method (intervention group)
The intervention incorporated all the usual verbal information required by the standard method of informed consent. Additionally, it included the use of written information containing details and diagrams of the injury and proposed surgery. The tool was adapted from the Queensland Health orthopaedic consent templates for the included fractures,10 and supplemented with figures and diagrams for each condition, adapted from the Orthopaedic Trauma Association, an international education organization for patients and surgeons.11 The treating surgeon would elaborate on the written information consent support tool with the patient, allowing ample time for the patient to read the form and comprehend the information. The patient was then provided with the opportunity to ask questions regarding the procedure and the treating surgeon addressed any concerns. The patient was given a copy of the information to keep.
Data collection
Patient demographic data were collected at baseline and included age, sex, first language, highest level of education, type of procedure undertaken, and American Society of Anesthesiologists grade (ASA).12 All data were collected on the ward at the participating centres.
From 1 March 2021 to 31 September 2021, 66 patients were screened for eligibility. Six participants were excluded: two patients were in severe pain, two reported feelings of anxiety, and two declined to participate in the study. Overall, 60 patients participated in the trial. Therefore, the recruitment rate for this trial was 90.91%. Of these 60, 32 were randomized to the standard method of informed consent and 28 were randomized to the detailed method and 30 (94%) and 26 (93%) were followed up in the standard and detailed groups, respectively. The Consolidated Standards of Reporting Trials13 flow diagram for recruitment is presented in Figure 1.
Participant demographics and surgical procedures included in the study are presented in Table I. The mean age was 53 years (standard deviation (SD) 17.95). The most undertaken procedures were distal radius (40%; n = 24) and ankle fracture (38.3%; n = 23) fixations. Baseline demographic data were similar between groups.
Table I.
Baseline demographic data of study participants.
| Variable | Standard method | Detailed method | p-value |
|---|---|---|---|
| Mean age, yrs (SD) | 53.9 (18.7) | 52.6 (17.4) | 0.793* |
| Sex, n (%) | 0.782† | ||
| Male | 16 (50) | 13 (46.4) | |
| Female | 16 (50) | 15 (53.6) | |
| English first language, n (%) | 0.929† | ||
| Yes | 26 (81.3) | 23 (82.1) | |
| No | 6 (18.7) | 5 (17.9) | |
| Highest level of education, n (%) | 0.600† | ||
| Incomplete year 12 | 8 (25) | 7 (25) | |
| Complete year 12 | 13 (40.6) | 8 (28.6) | |
| Bachelor’s degree or higher | 11 (34.4) | 12 (42.9) | |
| Procedure, n (%) | 0.527† | ||
| Ankle fracture fixation | 13 (40.6) | 10 (35.7) | |
| Distal radius fracture fixation | 12 (37.5) | 12 (42.9) | |
| Femur fracture fixation | 2 (6.3) | 0 (0) | |
| Tibia fracture fixation | 5 (17.9) | 6 (21.4) | |
| ASA grade, n (%) | 0.283† | ||
| I | 13 (40.6) | 6 (21.4) | |
| II | 13 (40.6) | 15 (53.6) | |
| III | 5 (15.6) | 7 (25) | |
| IV | 1 (3.1) | 0 (0) | |
| Mean time to follow-up, days (SD) | 3.2 (0.4) | 3.81 (1.7) | 0.069* |
Independent-samples t-test.
Chi-squared test.
ASA, American Society of Anesthesiologists; SD, standard deviation.
The primary outcome measure was patient recall at 72 hours postoperatively using a seven-point interviewer-administered questionnaire. The questionnaire asked the participant details of potential complications (maximum three points), implants used (maximum three points), and postoperative instructions (maximum one point). A total score was calculated and was used as an overall measure of patient recall and had good face validity.
Secondary outcomes included perioperative anxiety using the HADS-A measured at 24 hours preoperatively and 72 hours postoperatively (minimum 0, maximum 21); postoperative satisfaction using the PSQ-18 measured at 72 hours postoperatively (minimum 0, maximum 90); perioperative pain using the VAS14 measured at 24 hours preoperatively and 72 hours postoperatively (minimum 0, maximum 10); and recruitment rate estimated at the conclusion of the trial. The HADS-A and VAS pain change was calculated by subtracting the postoperative score from the preoperative score. The recruitment rate was defined as the percentage of patients recruited into the study from those that were screened. A rate of 75% of eligible participants was needed for the study to be considered feasible.
Sample size
No information was available for the primary outcome (recall of information) to allow a sample size calculation. Instead, a convenience sample was recruited over a planned seven-month period across the two hospitals.
Statistical analysis
The analysis was conducted using the intention-to-treat (ITT) principle. Means, SDs, and 95% confidence intervals (CIs) were used to present all continuous variables. The independent-samples t-test was used to compare all continuous data between both groups. The chi-squared test was used for categorical data analysis. A p-value ≤ 0.05 was considered statistically significant. Statistical analysis was conducted using SPSS v. 26 software (IBM, USA).
Results
A summary of the outcomes are presented in Table II. The detailed group had a significantly higher total recall score compared to the standard group (mean difference 1.29 points (95% CI 0.51 to 2.08); p = 0.002, independent-samples t-test). There was no significant difference in change in anxiety between the two groups (mean difference 0.39 (95% CI -2.11 to 2.88); p = 0.757, independent-samples t-test). No significant differences in patient satisfaction were found between groups (mean difference 2.09 (95% CI -2.78 to 6.94); p = 0.392, independent-samples t-test). Similarly, the differences in perioperative pain were insignificant (mean difference 5.71 (95% CI -22.25 to 11.11); p = 0.499, independent-samples t-test).
Table II.
Primary and secondary outcomes.
| Variable | Standard method | Detailed method | Difference (95% CI) | p-value* |
|---|---|---|---|---|
| Mean total postoperative recall (SD) | 4.28 (1.68) | 5.58 (1.18) | 1.3 (0.51 to 2.08) | 0.002 |
| Mean Δ HADS anxiety subscale (SD) | -1.18 (4.68) | -0.79 (4.21) | 0.39 (-2.11 to 2.88) | 0.757 |
| Mean Δ VAS pain (SD) | -20.41 (29.36) | -26.12 (32.22) | 5.71 (-22.25 to 11.11) | 0.499 |
| Mean PSQ-18 total satisfaction (SD) | 76.57 (10.13) | 78.65 (7.57) | 2.09 (-2.78 to 6.94) | 0.392 |
Independent-samples t-test
CI, confidence interval; HADS-A, Hospital and Anxiety Depression scale; PSQ-18, Patient Satisfaction Questionnaire Short-Form; SD, standard deviation; VAS, visual analogue scale.
Discussion
We conducted a randomized controlled trial which demonstrated that patients consented using a detailed written tool had a greater postoperative recall than the patients using the standard method. The trial further found insignificant differences in the HADS-A, PSQ-18, and VAS pain scores between the detailed and standard groups. The trial proved to be feasible by achieving a recruitment rate of approximately 90.9%, with 93.3% of patients followed up.
These findings are similar to those reported by Smith et al15 and Langdon et al,5 where the supplementation of written information significantly improved recall for orthopaedic patients. In contrast to these studies, others have found that written information does not improve recall.16-18 It is plausible that the benefit of written information may be dependent on the simplicity of the document. Kim et al19 found that those consented with a simplified consent document had higher levels of understanding relative to those consented with a standard consent document. The simplified document contained short sentences, bullet points, and diagrams. The detailed written tools in our study were similarly formatted. The detailed tools in our study included illustrations to aid understanding and reinforce information provided in the text. Stewart et al20 have previously demonstrated that patients consented using visual aids had a significantly higher recall of complications than those through the standard care.
Poor recall has medicolegal implications. Alleged complications or adverse outcomes have formed the basis of a large proportion of malpractice suits in orthopaedic surgery.21-23 These findings allude to a failed or inadequate consent process. As evidenced in our study, patients consented using a detailed tool had an increased awareness of complications associated with their surgery. Hence, using a detailed written tool to comprehensively educate patients about complications may be an important consideration to prevent potential lawsuits from arising.24 However, medicolegal claims are likely to be lodged years after the operation itself,25 so a future trial assessing patient recall at this timepoint may be beneficial.
The use of written information is a feasible option to augment shared decision-making in a trauma setting. In Australia, the median time for a patient to receive initial care after a traumatic injury is one hour and 29 minutes, and orthopaedic trauma is generally performed on semiurgent operating lists within normal working hours.26 This gives patients sufficient time to read a written information sheet and understand important information regarding their surgery.
Patients with improved recall have demonstrated greater treatment adherence and fewer complications.27,28 Hence, these findings are clinically relevant as better recall at discharge may improve self-care knowledge and complication management.
Our findings suggest that consenting a patient using a detailed written tool did not overwhelm them or prompt anxiety. These findings were in line with those reported by Fraval et al,6 who found that exposing patients to written information regarding a procedure did not increase anxiety or apprehension for adult patients undergoing orthopaedic surgery. However, it is important to acknowledge that this study was underpowered to detect a change in anxiety across time due to the small number of participants in the trial. According to our data, a minimal clinically important difference for the HADS-A to guide future trials was 2.2 points and required 140 patients in a trial assuming p ≤ 0.05, a power of 80%, and a 10% attrition rate.
We acknowledge the limitations of this study. First, the treating surgeons and researchers were not blinded. Second, we could not control for information provided by other members of the healthcare team, such as nursing staff and physiotherapists, particularly regarding postoperative care instructions. However, it is unlikely this information was provided differently to each intervention group. Third, we had no data to guide an a priori sample size calculation for the recall of information. However, CIs for our primary outcome are consistent with a substantial difference in intervention groups. Finally, the trial is likely underpowered to detect differences in other important outcomes, such as postoperative complications or non-compliance rates.
The detailed written tools are useful adjuncts to improve postoperative recall in adult orthopaedic trauma patients. The tool did not significantly influence anxiety, satisfaction, or perceived pain levels. A future trial with a larger sample size powered for important outcomes is needed to support the findings of this study.
Take home message
- Better recall at discharge may improve self-care knowledge and complication management.
- Consenting a patient using a detailed written tool did not overwhelm them or prompt anxiety.
Author contributions
A. M. Aslam: Project administration, Investigation, Data curation, Formal analysis, Writing – original draft, Writing – review & editing.
J. Kennedy: Investigation, Data curation, Formal analysis, Writing – original draft, Writing – review & editing.
H. Seghol: Data curation, Writing – original draft, Writing – review & editing.
N. Khisty: Investigation, Data curation, Writing – original draft, Writing – review & editing.
T. A. Nicols: Data curation, Writing – original draft, Writing – review & editing.
S. Adie: Conceptualization, Methodology, Supervision, Writing – original draft, Writing – review & editing.
Funding statement
The authors received no financial or material support for the research, authorship, and/or publication of this article.
Trial registration number
The trial was registered with anzctr.org.au (ACTRN12621000852853).
Ethical review statement
The study protocol was approved by the SESLHD Human Research and Ethics Committee (2019/ETH13394).
Open access funding
The authors confirm that the open access fee was self-funded.
© 2023 Authors et al. This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial No Derivatives (CC BY-NC-ND 4.0) licence, which permits the copying and redistribution of the work only, and provided the original author and source are credited. See https://creativecommons.org/licenses/by-nc-nd/4.0/
Contributor Information
Amjad M. Aslam, Email: amjadaslam9@gmail.com.
James Kennedy, Email: jken8805@gmail.com.
Haider Seghol, Email: haider.sehgol@health.nsw.gov.au.
Nikhil Khisty, Email: nikhilkhisty@gmail.com.
Thomas A. Nicols, Email: tomnicols@me.com.
Sam Adie, Email: sam.adie@gmail.com.
References
- 1. Singh S, Mayahi R. Consent in orthopaedic surgery. Ann R Coll Surg Engl. 2004;86(5):339–341. doi: 10.1308/147870804353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Sahin N, Oztürk A, Ozkan Y, Demirhan Erdemir A. What do patients recall from informed consent given before orthopedic surgery? Acta Orthop Traumatol Turc. 2010;44(6):469–475. doi: 10.3944/AOTT.2010.2396. [DOI] [PubMed] [Google Scholar]
- 3. Atrey A, Gupte CM, Corbett SA. Review of successful litigation against English health trusts in the treatment of adults with orthopaedic pathology: clinical governance lessons learned. J Bone Joint Surg Am. 2010;92-A(18):e36. doi: 10.2106/JBJS.J.00277. [DOI] [PubMed] [Google Scholar]
- 4. Mangla M, Cha TD, Dorrwachter JM, et al. Increasing the use of patient decision aids in orthopaedic care: results of a quality improvement project. BMJ Qual Saf. 2018;27(5):347–354. doi: 10.1136/bmjqs-2017-007019. [DOI] [PubMed] [Google Scholar]
- 5. Langdon IJ, Hardin R, Learmonth ID. Informed consent for total hip arthroplasty: does a written information sheet improve recall by patients? Ann R Coll Surg Engl. 2002;84(6):404–408. doi: 10.1308/003588402760978201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Fraval A, Chandrananth J, Chong YM, Coventry LS, Tran P. Erratum to: Internet based patient education improves informed consent for elective orthopaedic surgery: a randomized controlled trial. BMC Musculoskelet Disord. 2015;16(1):283. doi: 10.1186/s12891-015-0660-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Mauffrey C, Prempeh EM, John J, Vasario G. The influence of written information during the consenting process on patients’ recall of operative risks. A prospective randomised study. Int Orthop. 2008;32(4):425–429. doi: 10.1007/s00264-007-0361-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67(6):361–370. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 9.Marshall GN, Hays RD. The Patient Satisfaction Questionnaire Short-Form (PSQ-18) Santa Monica, California, USA: RAND Corporation; 1994. [Google Scholar]
- 10.No authors listed Consent forms and patient information sheets. Queensland Health. 2022. [16 January 2023]. https://www.health.qld.gov.au/consent/html/for_clinicians date last. accessed.
- 11.No authors listed About OTA. Orthopaedic Trauma Association. 2022. [16 January 2022]. https://ota.org/about-ota date last. accessed.
- 12. Saklad M. Grading of patients for surgical procedures. Anesthesiology. 1941;2(3):281–284. [Google Scholar]
- 13. Schulz KF, Altman DG, Moher D, CONSORT Group CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. BMJ. 2010;340:c332. doi: 10.1136/bmj.c332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Delgado DA, Lambert BS, Boutris N, et al. Validation of digital visual analog scale pain scoring with a traditional paper-based visual analog scale in adults. J Am Acad Orthop Surg Glob Res Rev. 2018;2(3):e088. doi: 10.5435/JAAOSGlobal-D-17-00088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Smith HK, Manjaly JG, Yousri T, et al. Informed consent in trauma: does written information improve patient recall of risks? A prospective randomised study. Injury. 2012;43(9):1534–1538. doi: 10.1016/j.injury.2011.06.419. [DOI] [PubMed] [Google Scholar]
- 16. Wong AL, Martin J, Tang D, et al. The effect of written information on recall of surgical risks of carpal tunnel release surgery: a randomized controlled study. Plast Reconstr Surg. 2016;138(6):1011e–1018e. doi: 10.1097/PRS.0000000000002771. [DOI] [PubMed] [Google Scholar]
- 17. Pesudovs K, Luscombe CK, Coster DJ. Recall from informed consent counselling for cataract surgery. J Law Med. 2006;13(4):496–504. [PubMed] [Google Scholar]
- 18. Turner P, Williams C. Informed consent: patients listen and read, but what information do they retain? N Z Med J. 2002;115(1164):U218. [PubMed] [Google Scholar]
- 19. Kim EJ, Kim SH. Simplification improves understanding of informed consent information in clinical trials regardless of health literacy level. Clin Trials. 2015;12(3):232–236. doi: 10.1177/1740774515571139. [DOI] [PubMed] [Google Scholar]
- 20. Stewart JA, Wood L, Wiener J, et al. Visual teaching aids improve patient understanding and reduce anxiety prior to a colectomy. Am J Surg. 2021;222(4):780–785. doi: 10.1016/j.amjsurg.2021.01.029. [DOI] [PubMed] [Google Scholar]
- 21. Mouton J, Gauthé R, Ould-Slimane M, Bertiaux S, Putman S, Dujardin F. Litigation in orthopedic surgery: what can we do to prevent it? Systematic analysis of 126 legal actions involving four university hospitals in France. Orthop Traumatol Surg Res. 2018;104(1):5–9. doi: 10.1016/j.otsr.2017.11.002. [DOI] [PubMed] [Google Scholar]
- 22. Ahmed SA, DeFroda SF, Naqvi SJ, et al. Malpractice litigation following traumatic fracture. J Bone Joint Surg Am. 2019;101-A(7):e27. doi: 10.2106/JBJS.18.00853. [DOI] [PubMed] [Google Scholar]
- 23. Ring J, Talbot C, Cross C, Hinduja K. NHSLA litigation in hip fractures: Lessons learnt from NHSLA data. Injury. 2017;48(8):1853–1857. doi: 10.1016/j.injury.2017.06.009. [DOI] [PubMed] [Google Scholar]
- 24. Barry MJ, Wescott PH, Reifler EJ, Chang Y, Moulton BW. Reactions of potential jurors to a hypothetical malpractice suit: alleging failure to perform a prostate-specific antigen test. J Law Med Ethics. 2008;36(2):396–402. doi: 10.1111/j.1748-720X.2008.00283.x. [DOI] [PubMed] [Google Scholar]
- 25. Mukherjee S, Pringle C, Crocker M. A nine-year review of medicolegal claims in neurosurgery. Ann R Coll Surg Engl. 2014;96(4):266–270. doi: 10.1308/003588414X13814021679834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.No authors listed Annual report: 1 July 2019 to 30 June 2020. Australia New Zealand Trauma Registry. [16 January 2023]. https://static1.squarespace.com/static/5b761ed3f93fd491065f7839/t/614808ed200a41594dcdcdbf/1632110845316/ATR_Annual+Report_19-20_FA_2web.pdf date last. accessed.
- 27. Dong L, Lee JY, Harvey AG. Do improved patient recall and the provision of memory support enhance treatment adherence? J Behav Ther Exp Psychiatry. 2017;54:219–228. doi: 10.1016/j.jbtep.2016.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Blay N, Donoghue J. The effect of pre-admission education on domiciliary recovery following laparoscopic cholecystectomy. Aust J Adv Nurs. 2005;22(4):14–19. [PubMed] [Google Scholar]