Abstract
Pathological behaviours towards drugs and food rewards have underlying commonalities. Risk taking has a fourfold pattern varying as a function of probability and valence leading to the non-linearity of probability weighting with overweighting of small probabilities and underweighting of large probabilities. Here we assess these influences on risk taking in patients with pathological behaviours towards drug and food rewards and examine structural neural correlates of non-linearity of probability weighting in healthy volunteers. In the anticipation of rewards, subjects with binge eating disorder show greater risk taking, similar to substance use disorders. Methamphetamine dependent subjects had greater non-linearity of probability weighting along with impaired subjective discrimination of probability and reward magnitude. Ex-smokers also had lower risk taking to rewards compared to non-smokers. In the anticipation of losses, obesity without binge eating had a similar pattern to other substance use disorders. Obese subjects with binge eating also have impaired discrimination of subjective value similar to that of the methamphetamine dependent subjects. Non-linearity of probability weighting was associated with lower grey matter volume in dorsolateral and ventromedial prefrontal cortex and orbitofrontal cortex in healthy volunteers. Our findings support a distinct subtype of binge eating disorder in obesity with similarities with substance use disorders to risk taking in the reward domain. The results dovetail with the current approach of defining mechanistically based dimensional approaches rather than categorical approaches to psychiatric disorders. The relationship to risk probability and valence may underlie the propensity towards pathological behaviours towards different types of rewards.
Introduction
Risk evaluation underlies everyday decisions from the mundane to the more substantial: should I walk today, switch careers? Risk attitude varies as a function of probability and valence in a non-linear fashion thus varying as a function of context. The pathological use of exogenous drugs or natural food rewards are behaviours characterized by the evaluation of risk. The likelihood of obtaining reward is weighted against the likelihood of punishment in the form of social or financial losses or poor health. Here we assess the relationship between risk and the influence of probability, valence (i.e. gain or loss) and magnitude (value) across pathological behaviours towards drug and food rewards.
Prospect Theory and experimental evidence demonstrate a fourfold pattern of risk attitude that varies as a function of the non-linearity of probability weighting and valence (Kahneman and Tversky, 2000) (Figure S1). An outcome value is weighted by a decision weight, w(p), i.e., the subjective belief of the objective probability p. Probability influences risk attitudes. This is because outcomes with lower probabilities are over-weighted and thus perceived as more probable, leading to risk seeking for gains (e.g. buying lotteries) and risk aversion for losses of low probability (e.g. buying insurance). On the other hand, higher probabilities are under-weighted, causing risk aversion for gains and risk seeking for losses of high probability. This non-linearity of probability weighting has been associated with anterior cingulate activity (Paulus and Frank, 2006) and D1 receptor activity (Takahashi et al., 2010). Risk evaluation is also affected by subjective value, which has a concave function, reflecting diminishing subjective discrimination (marginal sensitivity) (Figure S1). Thus, a £10 increment at a higher magnitude, £1000 and £1010, has less subjective value than the same increment at a lower magnitude, £10 and £20.
We focus on risk-taking attitude across disorders of pathological choice involving natural (obese subjects with and without binge eating disorder) and drug rewards (abstinent alcohol and stimulant dependence). Although losses in the form of social and health cost are difficult to model, the secondary reinforcer of money can act as a proxy. The rodent model of sucrose binge eating has many similarities with models of substance use disorders (Avena et al., 2008) suggesting that the pattern of food intake, namely binge eating, is a crucial subtype and may differentiate obese subjects with and without binge eating. Although preclinical evidence supports similarities between obesity and substance use disorders in animal models, evidence from human studies is limited (Ziauddeen et al., 2012).
Risk taking abnormalities in disorders of drug and food reward are known to occur under conditions of risk (known probabilities) and uncertainty (unknown probabilities). For example, stimulant dependent subjects and subjects with binge eating or obesity have impaired decisions using the Iowa Gambling Task, which addresses risk taking under uncertainty (Danner et al., 2012; Le Berre et al., 2012). Chronic amphetamine and alcohol dependent users show suboptimal decisions under risk with explicit probabilities, on the Cambridge Gamble Task (Rogers et al., 1999; Chamberlain et al., 2007; Lawrence et al., 2009). Furthermore, subjects with alcohol use disorders and women with binge eating disorder also have impaired decision making on the Game of Dice Task (Svaldi et al., 2010), with known explicit rules and probabilities. While these tasks assess choices under ambiguous or risky conditions with feedback, they do not address the influences of valence, probability or value differences.
Here we use an adaptation of a risk task (Paulus and Frank, 2006; Takahashi et al., 2010) that assesses risk attitude focusing on the effects of valence, probability and value, without feedback. We hypothesize that these dimensions influence risk-taking in all disorders and that binge eating shows similar deficits to other substance use disorders. In particular, we expect greater risk seeking in the reward domain and that loss has divergent influence on risk-taking across all disorders. We further investigate the individual differences of non-linearity of probability weighting (α) with grey matter volume in healthy volunteers.
Methods
Recruitment
Abstinent subjects with alcohol dependence (EtOH, N=30), obese subjects (>30 Body Mass Index, BMI) with binge eating disorder (BED, N=30), obese controls without BED (N=30) and abstinent methamphetamine dependent subjects (Meth, N=23) were recruited for the study. For each subject in each patient group, two age- and gender-matched healthy volunteers (HV) were recruited. For the Voxel-based morphometry (VBM) study, a different sample of 30 HV (mean age 24.15 (SD 2.91); 17 males) underwent scanning. Further recruitment strategy and questionnaires completed have been reported elsewhere (Voon et al., 2013) and are described in Supplementary materials.
Task
Subjects chose between a Risky choice (jar with explicit probability of red and blue balls) and a Sure choice (Figure 1A). Subjects were told that if they chose the Risky choice, the computer would randomly select a ball from the jar. If the ball was red, the subject would win (or lose) the specified amount indicated above the jar. If they chose the Sure choice, they would win (or lose) the full amount indicated. Choices were presented with 4 probabilities (P=0.1 0.3 0.5 0.9) and 4 expected values (EV=£10, £50, £100, £500) for each probability, giving 16 prospects. The order of the probabilities and EVs were randomized. Reward and loss conditions were run separately and the order counterbalanced across subjects.
Figure 1.
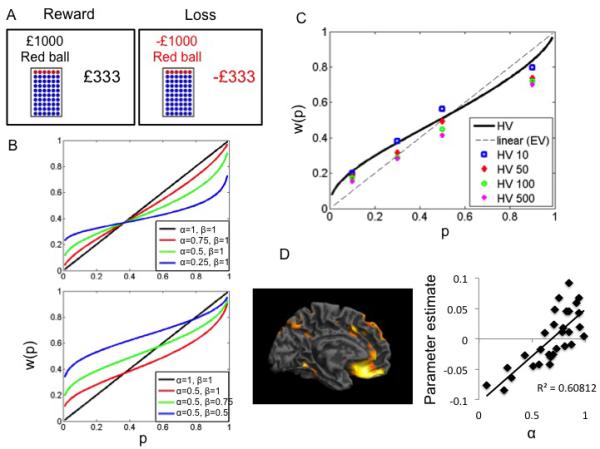
A. Risk task. Subjects choose between a Gamble and Sure choice in both Reward and Loss conditions across 4 probabilities (0.1 0.3 0.5 0.9) and 4 expected values (£10 £50 £100 £500). A staircase procedure was used to estimate the gamble’s certainty equivalents (amount of sure payoff in subject indifferent between sure payoff and gamble). B. Examples of functions with different non-linear (α) and convexity (β) values. C. Probability weighting, w(p), data of healthy volunteers in reward condition as a function of objective probability, p. The dotted line represents certainty equivalence = expected value. D. Regression analysis of grey matter volume and α. The regression represents left ventromedial prefrontal cortex as a function of α.
For each prospect the certainty equivalent (CE, the amount of certain money that would be accepted instead of a gamble) was computed. This was determined in a step-wise manner depending on the previous choices (Paulus and Frank, 2006; Takahashi et al., 2010). The value (V) of the Risky choice (amount indicated over the jar) was calculated as V=EV/P (e.g. for P=0.1, EV=£100, V=£1000) (Figure 1). The CE range for each prospect was determined by defining the range of values between 0 and V of the Risky choice (e.g. 0 to £1000) (described in detail in Figure S2). In trials 1 and 2, the amount of the Sure choice was the one-third and two-third cut point values. The interval for the next 2 trials included only the interval accepted by the subject in the first 2 trials. For example, if the subject rejected the lower and middle third, the upper third was used as the range for trials 3 and 4. The amount of the Sure choice was then the one-third and two-third cut point values of this upper third range. The same process was repeated for trials 5 and 6 and the average of these final choices was used to determine the CE. In the instructions, subjects were given an example trial of 6 choices to demonstrate the step-wise method and changes in the Sure magnitude. There was no time limit for each trial. The equivalent in US dollars were used for subjects tested in the United States. Subjects were told that they would not find out the outcome after each trial but that at the end of the study, the computer randomly selected one of the trials and they would either receive or lose a proportion of the amount won or lost. The task was coded in e-PRIME 2.0.
The weighted probability, w(p), was calculated according to prospect theory as:
where v (x,p) is the subjective value of amount x at probability p (i.e. the CE) and w is the decision weight of the objective probability p.
The estimation of the non-linearity of probability weighting was calculated using the two-parameter function derived by Prelec (Prelec, 1998). The one-parameter function derived by Prelec is described as follows:
with 0 < α < 1. Using the one-parameter function, the w(p) function is an inverted S-shaped function with a fixed inflection point at p = 1/e = 0.37 (where p = p(w)). The α parameter indicates the degree of non-linearity with α approaching 0 indicating greater non-linearity and α approaching 1 approximating the linear expected value. As we were focusing on pathological groups in which the inflection point may not conform to 1/e, we estimated probability weighting using a two-parameter function (Prelec, 1998) in which the inflection point was not constrained to the 1/e fixed value:
β indicates the net convexity of the curve. Where β=1 is consistent with the one-parameter function where the inflection point crosses at p=1/e. As β approaches 0, the inflection point at which risk seeking shifts to risk aversion increases occurring at p>1/e indicating greater risk seeking across a wider range of probabilities. Examples of functions with different α and β values are shown in Figure 1. α and β and the fit R2 were calculated for each EV. R2 outliers (> 3 SD from group mean) were removed from the analysis.
Statistical analysis
The w(p) was analyzed as a mixed measures ANOVA with Group as a between-subjects factor, and Valence, Probability and Value as a within-subjects factor. We focused on the main effects and the interaction effects involving the Group factor. We also conducted a post-hoc analysis comparing Group differences as a function of probability using Tukey test if there was a Group × Probability × Valence interaction to assess the influence of probability.
To assess the relationship with Value, we conducted two separate mixed measures ANOVA since Value was positive in the reward domain and negative in the loss domain. We also conducted a post-hoc analysis using Tukey test if there was a Group × Value interaction to assess the influence of Value.
The w(p) for each value for the reward condition were fit with the two parameter model and α and β analyzed as a mixed measures ANOVA with Group as a between-subjects factor and value as a within-subjects factor. For all analyses, P<0.05 was considered significant.
We also assessed the relationship of w(p) for reward and loss collapsed across probability and value to clinical measures for each subject group using Pearson correlation. For the BED and Obese subjects, the corrected Bonferroni value was P<0.0125 (4 comparisons) and for EtOH and Meth subjects was P<0.008 (6 comparisons).
MRI data acquisition and analysis details are reported in supplementary materials.
Results
Subject characteristics
Subject characteristics are reported in Supplementary materials (S1 and S2). Three HV, 1 BED, 1 EtOH and 2 Meth subjects were removed from the analysis as outliers.
For each subject group, the following analyses are divided into: (i) decision weight w(p) across probabilities (Table S3, Figure 2); (ii) w(p) separately for reward and loss across values (Table S4, Figure 3); (iii) nonlinearity, α, and convexity, β Table S5, Figure 5).
Figure 2.
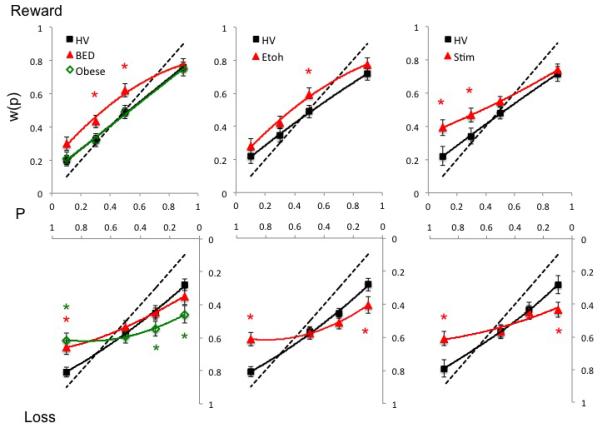
Probability weighting in pathological disorders in reward and loss conditions. The graphs compare probability weighting, w(p), in Obese subjects with binge eating disorder (BED) and without BED (Obese controls), abstinent alcohol dependent (EtOH) and abstinent methamphetamine dependent (Meth) versus their own age- and gender-matched controls for reward (top) and loss (bottom) conditions. The dotted line represents certainty equivalence = expected value. Error bars represent standard error of the mean. *posthoc analysis results following interaction effect of Group × Valence × Probability. Details of significant differences are described in the text and Table S3. Abbreviations: P = probability.
Figure 3.
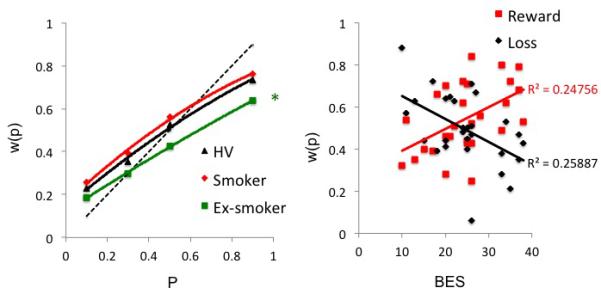
Left. Probability weighting as a function of current smokers, ex-smokers and non-smokers in healthy volunteers, and Obese subjects with and without binge eating disorder. *P<0.05 Group difference between ex-smokers and non-smokers. Right. Relationship to clinical variables. Obese subjects with binge eating disorder: relationship between binge eating severity (BES) and probability weighting, w(p), in the Reward and Loss domains.
Figure 4.
Probability weighting as a function of reward value. The graphs compare probability weighting, w(p), and value in the reward domain in Obese subjects with binge eating disorder (BED) and without BED (Obese controls), abstinent alcohol dependent (EtOH) and abstinent methamphetamine dependent (Meth) subjects versus their own age- and gender-matched controls. Error bars represent standard error of the mean. *Group × Value interaction. Details of significant differences are described in the text and Table S4.
Figure 5.
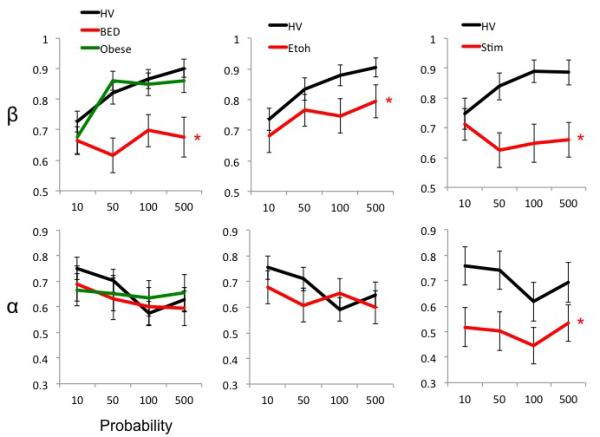
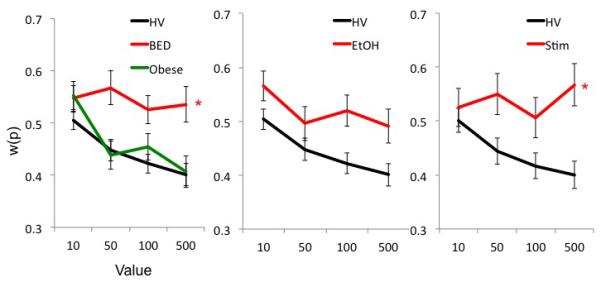
Convexity (β) and non-linearity (α). The graphs compare convexity (β; top) and non-linearity (α; bottom) in Obese subjects with binge eating disorder (BED) and without BED (Obese controls), abstinent alcohol dependent (EtOH) and abstinent methamphetamine dependent (Meth) subjects versus their own ageand gender-matched controls. Error bars represent standard error of the mean. *Main Group effect. Details of significant differences are described in the text and Table S5.
Obese subjects with BED
Decision weight, w(p): probability
A main Group effect revealed that BED overweighted probabilities compared to HV. The GroupxValence interaction showed greater risk taking in Reward (mean difference −0.100 (SEM 0.03) (95%CI: −0.161- −0.039) p=0.002) but not Loss (0.020 (SEM 0.034) (95%CI: −0.047- 0.087) p=0.553). There was a main Probability and Value effect and GroupxProbability and GroupxProbabilityxValence interactions. On posthoc analysis, BED made more risky choices to moderate probabilities (P=0.30 and 0.50) in Reward and more risky choices to high probabilities (P=0.90) in Loss.
Decision weight, w(p): value
Reward: there was a main Group, Probability and Value effect. There was a GroupxValue interaction in which there was a Value effect in HV (F(3,86)=12.036, p<0.001) but not in BED (F(3,86)=1.199, p=0.315). Loss: there was a main Probability and Value effect only.
Reward non-linearity (α) and convexity (β)
For β, there was a main Group effect: BED made more risky choices (lower β) compared to HV (R2: HV 0.57 (0.39), BED 0.51 (0.33), t=0.51, p=0.59). There was a main Probability effect. For α, there was a main Probability effect.
Obese controls without BED
Decision weight, w(p): probability
There was a main Probability, Value and Valence effect and a GroupxProbability and GroupxProbabilityxValence effect. On posthoc analysis, during Loss, Obese controls made fewer risky choices to low probabilities (P=0.10 and 0.30) and more risky choices to high probabilities (P=0.90).
Decision weight, w(p): value
There was a main Probability and Value effect. During loss, there was a main Probability and Value effect.
Reward non-linearity (α) and convexity (β)
For β and α, there were main Probability effect but not of Group or an interaction. (R2: HV 0.56 (0.40), Obese 0.47 (0.41), t=1.154, p=0.253)
Abstinent alcohol dependent subjects
Decision weight, w(p): probability
A main Group effect revealed that EtOH subjects overweighted probabilities compared to HV in Reward (mean difference −0.074 (SEM 0.033) (95%CI: −0.139 - − 0.010) p=0.025) but not in Loss (mean difference −0.015 (SEM 0.032) (95%CI: −0.078 – 0.048) p=0.639). There was a main Probability and Value effect and GroupxProbability and GroupxProbabilityxValence interaction. On posthoc analysis, EtOH subjects made more risky choices to moderate probabilities (P=0.50) to Reward and to high probability (P=0.90) to Loss along with fewer risky choices to low probability (P=0.10) to Loss.
Decision weight, w(p): value
Reward: there was a main Group, Probability and Value effect. Loss: there was a main Probability effect.
Reward non-linearity (α) and convexity (β)
For β, there was a main Group effect: EtOH made more risky choices (lower β) compared to HV (R2: HV 0.53 (0.40), EtOH 0.56 (0.26), t=−0.35, p=0.72). There was a main Value effect. For α, there were no main or interaction effects.
Abstinent stimulant dependent subjects
Decision weight, w(p): probability
A main Group effect demonstrated that Meth subjects overweighted probabilities compared to HV in Reward (mean difference −0.97 (SEM 0.038) (95%CI: −0.172 - − 0.022) p=0.012) and not in Loss (mean difference =0.029 (SEM 0.035) (95%CI: −0.099 – 0.041) p=0.413). There was a main Probability effect and GroupxProbability and GroupxValue interaction. On posthoc analysis, Meth subjects made more risky choices to low probability (P=0.10 and 0.30) in Reward and to high probability (P=0.90) in Loss along with fewer risky choices to low probability (P=0.10) in Loss.
Decision weight, w(p): value
Reward: there was a main Group, Probability and Value and GroupxValue interaction effect in which Value was significantly different in HV (F(3,65)=7.207, p<0.001) but not in Meth (F(3,65)=1.560, p=0.206). Loss: there was a main Group and Probability effect only.
Reward non-linearity (α) and convexity (β)
For β, there was a main Group effect: Meth made more risky choices (lower β) compared to HV (R2: HV 0.52 (0.40), Meth 0.40 (0.37), t=1.31, p=0.19). There was an interaction effect. For α, there was a Group effect: Meth had greater non-linearity (lower α) compared to HV. There was a main effect of Value.
Nicotine
Although the study was not intended to address nicotine effects, analyses were conducted on an exploratory basis focusing on w(p) for probability (Supplementary materials). Ex-smokers (N=25) in the combined group of Obese, BED and HV, made fewer risky choices than non-smokers (N=82) (F(1,103)=4.276, p=0.041) with no differences between current smokers (N=24) and non-smokers (N=82) (F(1,102)=1.471, p=0.228) (Figure 3).
Relationship to clinical measures
We also assessed the relationship of w(p) to clinical measures. For BED only, BES scores were positively correlated with w(p) for reward (Pearson correlation coefficient=0.498, p=0.007) and negatively correlated for loss (−0.509, p=0.006) (Bonferroni corrected P>0.0125) (Figure 3). There were no correlations with BMI. For EtOH, there was a negative correlation trend between weeks abstinent and w(p) for reward (−0.417, p=0.024) but not loss (0.036, p=0.850). There were no significant correlations with duration or total units (Bonferroni corrected P>0.008). For Meth there was a negative correlation trend with number of years of heavy use and w(p) for reward (−0.487, p=0.022) but not loss (0.143, p=0.523) (Bonferroni corrected P>0.008). There were no significant correlations between w(p) and BDI for any group (p>0.05).
Structural correlates of non-linearity
In 30 healthy volunteers, grey matter volume was positively correlated with α in the right dorsolateral prefrontal cortex (dlpfc) (reported in MNI coordinates x y z in mm = 33 50 36, Cluster size = 18, Z=5.18, whole brain FWE corrected P=0.009, BA 9), left ventromedial prefrontal cortex (vmpfc) (x y z in mm: −4 44 −18, Cluster size = 33, Z=4.94, whole brain FWE corrected P=0.02, BA 11, Figure 1D) and right medial orbitofrontal cortex (mOFC) (x y z in mm: 15 30 −18, Cluster size = 17, Z=4.98, whole brain FWE corrected P=0.02, BA 11).
Discussion
We demonstrate that risk attitude across disorders of addiction varies according to the currently manipulated dimensions of valence (gain and loss), probability and value (magnitude). In the reward domain, obese subjects with binge eating have enhanced risk seeking similar to that of substance use disorders, with the degree of risk seeking in the reward domain correlating with binge eating severity. In contrast, in the loss domain, obese subjects without binge eating have a similar risk attitude to substance use disorders with greater risk aversion to unlikely large losses and greater risk seeking to likely small losses. On the influence of value, obese subjects with binge eating also have a similar impairment to that of the Meth subjects, particularly in their discrimination of subjective value. These findings further explain the abnormalities in risk-taking observed in obese individuals (Pignatti 2006), and more importantly highlight the similarities in disorders characterized by natural and drug rewards. In terms of probability, methamphetamine dependent subjects have greater risk seeking to unlikely but large rewards along with greater non-linearity of probability weighting (lower α) and BED and alcohol dependent subjects have greater risk seeking to more likely but smaller rewards. Although not a direct correlate, this differential attitude towards reward probability may underlie choice tendencies towards the degree of risk or ‘high’ of illegal or legal drugs or natural rewards. These tendencies may combine with enhanced ‘waiting’ impulsivity in the alcohol and methamphetamine dependent subjects (Voon et al., 2014a) to drive risky decisions in pathological choices towards natural and drug rewards.
The relationship between obesity and disorders of substance addiction has recently been of great interest. In keeping with the preclinical literature on sugar bingeing in rodents, binge eating, rather than obesity per se, may have closer overlaps with substance use disorders. In rodents, sugar bingeing reflects addictive-like properties including enhanced responding for sugar after abstinence, amphetamine cross-sensitization and nucleus accumbens dopamine release (Avena et al., 2008). In humans, food stimulation in binge eating disorder subjects is also associated with greater striatal dopamine release (Wang et al., 2011). Thus, our data on risk attitude in the reward domain dovetails with these observations suggesting similarities in enhanced risk seeking between binge eating and substance dependence.
In the context of loss, obese subjects without BED and alcohol and stimulant dependent subjects are all more risk averse to low probability losses and more risk seeking to high probability losses. These results might explain the coexistence of conflicting risk attitudes influenced by context: risky addictive behaviours to avoid likely small negative outcomes and simultaneously, risk aversion in the anticipation of unlikely large negative outcomes. Our finding of enhanced sensitivity to loss anticipation dovetails with heightened sensitivity to negative cues (such as negative mood, stress or withdrawal) which may act as a negatively-reinforcing motivator in substance use disorders and compulsive overeating (Koob and Volkow, 2010). As expected in healthy volunteers, normal responses to loss are demonstrated by risk aversion to low probability losses and risk seeking to higher probability losses. Our findings in the pathological groups suggest an enhancement of this underlying tendency.
We show that enhanced non-linearity of probability weighting (lower α) in healthy volunteers is associated with lower grey matter volume in left vmpfc, right mofc and right dlpfc, consistent with regions implicated in the representation and evaluation of risk and probability (Fellows and Farah, 2007; Tom et al., 2007; Rangel et al., 2008). Greater non-linearity of probability weighting is believed to be associated with impaired discrimination of different probabilities (Takahashi et al., 2010) which dovetails with our findings of decreased grey matter volume in the vmpfc and ofc. The curvature of the weighting function has been postulated to be related to the psychophysics of diminishing sensitivity or affective influences such as hope or fear related to the gamble (Trepel et al., 2005). We further show that Meth subjects have greater non-linearity of probability weighting (lower α, i.e., over-weighting of low probabilities and under-weighting of high probabilities) to anticipated risky rewards. Methamphetamine dependence has been associated with decreased ofc volume (Nakama et al., 2011) possibly related to comorbid nicotine use (Morales et al., 2012). Abstinent stimulant use disorder subjects have also been shown to have decreased mofc volumes (Franklin et al., 2002), associated with impaired decisions in a modified gambling task (Tanabe et al., 2009). Although we did not show abnormalities in α in BED subjects, ofc volume differences have also been reported (Schafer et al., 2010; Voon et al., 2014b).
Subjective value was assessed by comparing the w(p) across the highest and lowest values within each group. As expected, healthy volunteers exhibited a decrease in subjective value equivalent to diminishing marginal sensitivity with increasing objective value (Kahneman and Tversky, 2000). BED and Meth subjects did not demonstrate any differences in subjective value with increasing objective value. This can be interpreted as decreased sensitivity towards changes in magnitude which coincides with reports of decreased sensitivity to changes in value in stimulant users. Psychostimulant-dependent subjects show decreased subjective discrimination of monetary reward gradients (Goldstein et al., 2007a), and decreased reaction times and lateral orbitofrontal responsivity to monetary reward outcomes (Goldstein et al., 2007b).
Although exploratory, we show that ex-smokers had lower risk taking in the reward domain than non-smokers. These findings suggest either that the lack of nicotine is associated with lower risk taking or more likely, that the ability to stop smoking may be related to lower risk taking tendencies. In the case of obese ex-smokers, lower risk taking for rewards may also reflect the shift from exogenous nicotine drug use towards endogenous natural rewards.
There were several limitations to the study. Relative to healthy volunteers, Meth, BED and EtOH subjects had higher depression scores as commonly reported in studies on substance use disorders and in relation to this current cohort (Voon et al., 2013). However we show that IQ and depression scores are unrelated to the current measures. As this is a cross-sectional study, we cannot address the question of whether risk attitude is a state or trait related factor. Unlike subjects with substance use disorders, the obese subjects were not ‘abstinent’ or food restricted which may affect motivational states. The influence of food restriction is baseline dependent such that it decreases risk aversion in those who are risk seeking and increases risk seeking in those who are risk averse (Levy et al., 2013). How food restriction might affect obese subjects should be assessed. We use a similar secondary reinforcer across all groups to allow for comparisons. The use of primary reinforcers are indicated to assess any differential influence on risk taking.
We show that risk taking is heterogeneous and pathological choices can be influenced by factors of valence, probability and magnitude. Our data uniquely highlights the overlaps between the behavior of binge eating and substance use disorders in the reward domain and obesity and substance use disorders in the loss domain. BED subjects similar to stimulant disorder subjects were also impaired in discrimination of subjective value. These characterizations are of particular importance as we have recently shown that an expected risk factor of ‘waiting’ impulsivity is not disturbed in obese patients with and without BED (Voon et al., 2014a). Our findings support mechanistically based dimensional rather than categorical approaches to psychiatric classifications (Insel et al., 2010) and suggest valence-specific modification of risk perception as a potential therapeutic avenue.
Supplementary Material
Acknowledgements
The study was funded by a Wellcome Trust Fellowship grant to Valerie Voon (093705/Z/10/Z). We would like to thank the subjects who participated in the study in Cambridge and staff and clients at the PRIDE Institute of Minnesota for their invaluable assistance with the study.
Funding and Disclosure: The study was funded by a Wellcome Trust Fellowship grant to Valerie Voon (093705/Z/10/Z). VV and NAH are Wellcome Trust (WT) intermediate Clinical Fellow. YW is supported by the Fyssen Fondation. The BCNI is supported by a WT and MRC grant. JG has received grants from the National Institute of Drug Abuse and the National Center for Responsible Gaming. ETB is employed part-time by the University of Cambridge and part-time by GSK PLC and is a shareholder of GSK. TWR is a consultant for Cambridge Cognition, Eli Lilly, GSK, Merck, Sharpe and Dohme, Lundbeck, Teva and Shire Pharmaceuticls. He is or has been in receipt of research grants from Lundbeck, Eli Lilly and GSK and is an editor for Springer-Verlag (Psychopharmacology). JWD is a consultant for Boehringer Ingelheim and has received grants from this company. JEG has received research grant support from NIDA, NCRG, Psyadon Pharmaceuticals and Transcept Pharmaceuticals. He has also received royalties from American Psychiatric Publishing Inc, Oxford University Press, Norton, and McGraw Hill Publishers. LSM, MAI, KD, VR, LRNS, BLO, JW declare no competing financial interests.
References
- Avena NM, Rada P, Hoebel BG. Evidence for sugar addiction: behavioral and neurochemical effects of intermittent, excessive sugar intake. Neuroscience and biobehavioral reviews. 2008;32:20–39. doi: 10.1016/j.neubiorev.2007.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chamberlain SR, Fineberg NA, Menzies LA, Blackwell AD, Bullmore ET, Robbins TW, Sahakian BJ. Impaired cognitive flexibility and motor inhibition in unaffected first-degree relatives of patients with obsessivecompulsive disorder. Am J Psychiatry. 2007;164:335–338. doi: 10.1176/appi.ajp.164.2.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danner UN, Ouwehand C, van Haastert NL, Hornsveld H, de Ridder DT. Decision-making impairments in women with binge eating disorder in comparison with obese and normal weight women. European eating disorders review. 2012;20:e56–62. doi: 10.1002/erv.1098. [DOI] [PubMed] [Google Scholar]
- Fellows LK, Farah MJ. The role of ventromedial prefrontal cortex in decision making: judgment under uncertainty or judgment per se? Cereb Cortex. 2007;17:2669–2674. doi: 10.1093/cercor/bhl176. [DOI] [PubMed] [Google Scholar]
- Franklin TR, Acton PD, Maldjian JA, Gray JD, Croft JR, Dackis CA, O’Brien CP, Childress AR. Decreased gray matter concentration in the insular, orbitofrontal, cingulate, and temporal cortices of cocaine patients. Biological psychiatry. 2002;51:134–142. doi: 10.1016/s0006-3223(01)01269-0. [DOI] [PubMed] [Google Scholar]
- Goldstein RZ, Tomasi D, Alia-Klein N, Cottone LA, Zhang L, Telang F, Volkow ND. Subjective sensitivity to monetary gradients is associated with frontolimbic activation to reward in cocaine abusers. Drug and alcohol dependence. 2007a;87:233–240. doi: 10.1016/j.drugalcdep.2006.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein RZ, Alia-Klein N, Tomasi D, Zhang L, Cottone LA, Maloney T, Telang F, Caparelli EC, Chang L, Ernst T, Samaras D, Squires NK, Volkow ND. Is decreased prefrontal cortical sensitivity to monetary reward associated with impaired motivation and self-control in cocaine addiction? Am J Psychiatry. 2007b;164:43–51. doi: 10.1176/appi.ajp.164.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Insel T, Cuthbert B, Garvey M, Heinssen R, Pine DS, Quinn K, Sanislow C, Wang P. Research domain criteria (RDoC): toward a new classification framework for research on mental disorders. Am J Psychiatry. 2010;167:748–751. doi: 10.1176/appi.ajp.2010.09091379. [DOI] [PubMed] [Google Scholar]
- Kahneman D, Tversky A. Choice, Values and Frames. Cambridge University Press; Cambridge: 2000. [Google Scholar]
- Koob GF, Volkow ND. Neurocircuitry of addiction. Neuropsychopharmacology. 2010;35:217–238. doi: 10.1038/npp.2009.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawrence AJ, Luty J, Bogdan NA, Sahakian BJ, Clark L. Problem gamblers share deficits in impulsive decision-making with alcohol-dependent individuals. Addiction. 2009;104:1006–1015. doi: 10.1111/j.1360-0443.2009.02533.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Berre AP, Rauchs G, La Joie R, Mezenge F, Boudehent C, Vabret F, Segobin S, Viader F, Allain P, Eustache F, Pitel AL, Beaunieux H. Impaired decision-making and brain shrinkage in alcoholism. European psychiatry. 2012 doi: 10.1016/j.eurpsy.2012.10.002. [DOI] [PubMed] [Google Scholar]
- Levy DJ, Thavikulwat AC, Glimcher PW. State dependent valuation: the effect of deprivation on risk preferences. PloS one. 2013;8:e53978. doi: 10.1371/journal.pone.0053978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morales AM, Lee B, Hellemann G, O’Neill J, London ED. Gray-matter volume in methamphetamine dependence: cigarette smoking and changes with abstinence from methamphetamine. Drug and alcohol dependence. 2012;125:230–238. doi: 10.1016/j.drugalcdep.2012.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller A, Brandl C, Kiunke W, Georgiadou E, Horbach T, Kohler H, de Zwaan M. Food-independent tendency to disadvantageous decisions in obese individuals with regular binge eating. Comprehensive psychiatry. 2014;55:64–70. doi: 10.1016/j.comppsych.2013.08.010. [DOI] [PubMed] [Google Scholar]
- Nakama H, Chang L, Fein G, Shimotsu R, Jiang CS, Ernst T. Methamphetamine users show greater than normal age-related cortical gray matter loss. Addiction. 2011;106:1474–1483. doi: 10.1111/j.1360-0443.2011.03433.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulus MP, Frank LR. Anterior cingulate activity modulates nonlinear decision weight function of uncertain prospects. NeuroImage. 2006;30:668–677. doi: 10.1016/j.neuroimage.2005.09.061. [DOI] [PubMed] [Google Scholar]
- Prelec D. The probability weighting function. Econometrica. 1998;66:497–527. [Google Scholar]
- Rangel A, Camerer C, Montague PR. A framework for studying the neurobiology of value-based decision making. Nature reviews Neuroscience. 2008;9:545–556. doi: 10.1038/nrn2357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers RD, Everitt BJ, Baldacchino A, Blackshaw AJ, Swainson R, Wynne K, Baker NB, Hunter J, Carthy T, Booker E, London M, Deakin JF, Sahakian BJ, Robbins TW. Dissociable deficits in the decision-making cognition of chronic amphetamine abusers, opiate abusers, patients with focal damage to prefrontal cortex, and tryptophan-depleted normal volunteers: evidence for monoaminergic mechanisms. Neuropsychopharmacology. 1999;20:322–339. doi: 10.1016/S0893-133X(98)00091-8. [DOI] [PubMed] [Google Scholar]
- Schafer A, Vaitl D, Schienle A. Regional grey matter volume abnormalities in bulimia nervosa and binge-eating disorder. NeuroImage. 2010;50:639–643. doi: 10.1016/j.neuroimage.2009.12.063. [DOI] [PubMed] [Google Scholar]
- Svaldi J, Brand M, Tuschen-Caffier B. Decision-making impairments in women with binge eating disorder. Appetite. 2010;54:84–92. doi: 10.1016/j.appet.2009.09.010. [DOI] [PubMed] [Google Scholar]
- Takahashi H, Matsui H, Camerer C, Takano H, Kodaka F, Ideno T, Okubo S, Takemura K, Arakawa R, Eguchi Y, Murai T, Okubo Y, Kato M, Ito H, Suhara T. Dopamine D(1) receptors and nonlinear probability weighting in risky choice. J Neurosci. 2010;30:16567–16572. doi: 10.1523/JNEUROSCI.3933-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanabe J, Tregellas JR, Dalwani M, Thompson L, Owens E, Crowley T, Banich M. Medial orbitofrontal cortex gray matter is reduced in abstinent substance-dependent individuals. Biological psychiatry. 2009;65:160–164. doi: 10.1016/j.biopsych.2008.07.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tom SM, Fox CR, Trepel C, Poldrack RA. The neural basis of loss aversion in decision-making under risk. Science. 2007;315:515–518. doi: 10.1126/science.1134239. [DOI] [PubMed] [Google Scholar]
- Trepel C, Fox CR, Poldrack RA. Prospect theory on the brain? Toward a cognitive neuroscience of decision under risk. Brain research Cognitive brain research. 2005;23:34–50. doi: 10.1016/j.cogbrainres.2005.01.016. [DOI] [PubMed] [Google Scholar]
- Voon V, Irvine MA, Derbyshire K, Worbe Y, Lange I, Abbott S, Morein-Zamir S, Dudley R, Caprioli D, Harrison NA, Wood J, Dalley JW, Bullmore ET, Grant JE, Robbins TW. Measuring “Waiting” Impulsivity in Substance Addictions and Binge Eating Disorder in a Novel Analogue of Rodent Serial Reaction Time Task. Biological psychiatry. 2013 doi: 10.1016/j.biopsych.2013.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voon V, Irvine MA, Derbyshire K, Worbe Y, Lange I, Abbott S, Morein-Zamir S, Dudley R, Caprioli D, Harrison NA, Wood J, Dalley JW, Bullmore ET, Grant JE, Robbins TW. Measuring “waiting” impulsivity in substance addictions and binge eating disorder in a novel analogue of rodent serial reaction time task. Biological psychiatry. 2014a;75:148–155. doi: 10.1016/j.biopsych.2013.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voon V, Derbyshire K, Ruck C, Irvine MA, Worbe Y, Enander J, Schreiber LR, Gillan C, Fineberg NA, Sahakian BJ, Robbins TW, Harrison NA, Wood J, Daw ND, Dayan P, Grant JE, Bullmore ET. Disorders of compulsivity: a common bias towards learning habits. Molecular psychiatry. 2014b doi: 10.1038/mp.2014.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang GJ, Geliebter A, Volkow ND, Telang FW, Logan J, Jayne MC, Galanti K, Selig PA, Han H, Zhu W, Wong CT, Fowler JS. Enhanced striatal dopamine release during food stimulation in binge eating disorder. Obesity. 2011;19:1601–1608. doi: 10.1038/oby.2011.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziauddeen H, Farooqi IS, Fletcher PC. Obesity and the brain: how convincing is the addiction model? Nature reviews Neuroscience. 2012;13:279–286. doi: 10.1038/nrn3212. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


