Abstract
Background
Metronomic oral vinorelbine (Navelbine®) was shown in clinical trials to yield sustainable antitumor activity possibly through anti-angiogenic mechanisms. We investigated the effects of protracted low dose vinorelbine (VRL) on Human Umbilical Vein Endothelial Cells (HUVEC) and compared it with a conventional chemotherapy model.
Methods
HUVEC cultures were treated with different concentrations of VRL (0.001nM to 1mM) for 4, 24 and 96h. The effects of different drug concentrations on cell growth, cell cycle, apoptosis and expression of angiogenesis modulating genes IL-8, COX-2, CD36 and PPARgamma were assessed using the metronomic or conventional chemotherapy model. Apoptosis and cell cycle effects were assessed by flow cytometry. Gene expression was measured at the transcript level by qRT-PCR, protein expression by immunoblotting and proteins secreted in the cell medium by ELISA. Activation of the NF-kB pathway was investigated by immunoblot analysis of cytosolic and nuclear protein extracts.
Results
The half-maximal inhibitory concentrations (IC50) of VRL at 96h were four orders lower compared to those after a 24h exposure (1.23nM vs. 32mM for VRL). Drug concentrations at high nanomolar levels and above, which are relevant to conventional pulsatile dosing of VRL, induced a dose-dependent, and NF-κB related, increase of pro-angiogenic IL-8 and COX-2 and a decrease of the TSP-1 receptor CD36 and PPARgamma at mRNA and protein levels. In contrast, the opposite was evident with protracted picomolar to low nanomolar concentrations (metronomic dosing).
Conclusions
Our data provide experimental support for metronomic vinorelbine by showing that protracted low dose outperforms pulsed high dose administration in inducing anti-angiogenic effects on proliferating human endothelial cells.
Keywords: metronomic chemotherapy, vinorelbine, PPARgamma, IL-8, NF-κB, HUVEC
Background
Cytotoxic anticancer drugs are conventionally administered in a pulsatile way at maximum tolerated doses on the aim to induce the highest possible apoptosis of cancer cells. However, at these doses they also affect healthy proliferating tissues and require treatment-free intervals to allow recovery from toxicities. These treatment gaps are known to facilitate repair of damaged endothelial and tumor cells and often to render most solid cancers resistant to these agents [1,2].
Metronomic chemotherapy is a novel dose-scheduling strategy for cytotoxic drug administration in cancer. It refers to frequent, regular administration of conventional chemotherapy at relatively sub-toxic doses, with no prolonged break periods [1,3,4]. In contrast to conventional chemotherapy, the primary target of metronomic chemotherapy is cancer vasculature, which plays a critical role in malignant tumor development and progression [1,5-7]. In this context, it was initially considered and eventually shown that optimal anti-angiogenic cancer therapy may steadily inhibit the proliferation and function of activated endothelial cells and therefore tackle cancer progression on a chronic basis [8].
Among several classes of cytotoxics, microtubule-targeted drugs are considered a reasonable choice for use in the setting of metronomic low-dose chemotherapy. They are known to act by inhibiting microtubule formation and function during mitosis, but they also have a potency to inhibit the function of endothelial cells at very low concentrations, a characteristic that is of major importance in metronomic chemotherapy [8,9]. Vinorelbine (VRL) is a semisynthetic vinca alkaloid metabolized in the liver, of which the main metabolite 4-O-deacetylvinorelbine (DVRL) has been identified in plasma of patients [10,11]. The availability of an oral formulation of vinorelbine (Navelbine® Soft Caps) led to the clinical investigation of this drug as monotherapy at a metronomic dosing schedule, with promising results [10,12]. In this setting, we investigated functional and molecular effects of VRL and its metabolite DVRL against proliferating endothelial cells (HUVEC) in an in vitro simulation study of conventional and metronomic chemotherapy. Under the influence of protracted and short exposure to VRL, we studied cell growth and apoptosis and the effects on angiogenesis signalling mediators.
The aim of the current study was to discover whether the protracted exposure of endothelial cells to VRL at low nanomolar and subnanomolar concentrations outperforms short exposure at concentrations in the range of high nanomolar to micromolar concentrations in inducing anti-angiogenic effects at cellular and molecular levels. We also sought to investigate whether and to what extend experimental evidence matches the clinical experience with metronomic oral VRL that yielded sustainable antitumor activity without overt toxicity [10,12].
Materials and Methods
Test compounds
Purified tartrate vinorelbine (VRL) and its active metabolite, 4-O-deacetylvinorelbine (DVRL), were kindly provided by the Institute de Recherché Pierre Fabre (Castres, France). Stock solutions were prepared (1mM, in conditioned medium) followed by filtration (0.2μm, Whatman, USA). Shortly before adding the research compounds to the cultured cells, each chemical was freshly diluted with conditioned medium to the desired concentrations.
Cell culture and Treatment
HUVEC were isolated from human umbilical cord veins, obtained from the Department of Gynecology of the University Hospital of Ioannina, were digested with 0.1% type-I collagenase at 37°C for 12min and grew in M199 (GIBCO) medium supplemented with 20% FBS, 30mg/ml endothelial cell growth supplement (ECGS; BD Biosciences, USA), heparin (10KU/ml; SIGMA), L-glutamine and streptomycin/ penicillin. Cells were used between the third and the sixth passage. HUVEC were plated to sub-confluence in plates coated with collagen (BD Biosciences, USA) and treated with VRL for 4, 24 and 96h. In the 96h experiment, in order to simulate the metronomic dosing schedule we replaced the drug-enriched medium every 24h. The concentrations used in the metronomic model (0.001nM-100nM) were based on the results of pharmacokinetic analysis of two clinical studies in conjunction with IC50 value of VRL on HUVECs. In particular, pharmacokinetic analysis of clinical trials with metronomic oral vinorelbine shows that the steady state trough levels of VRL averaged at 2-3 nM [12]. Therefore up to these levels, the concentrations considered metronomic. For this reason, we selected 10nM for nuclear and cytosolic protein extraction as an estimated value of Cmax of metronomic drug administration. In addition, concentrations between 100nM-1μΜ simulate peak plasma levels of the conventional chemotherapy protocol, as specified by Marty et al [13]. Accordingly, we used a range of concentration between 0.1nM- 10μΜ to simulate the conventional bolus administration protocol, which included HUVEC’s exposure to VRL/DVRL for 4h with a wash out period with drug-free medium for the consecutive 92h. At the end of exposure, both control and drug-treated cells were collected. Total RNA and protein fractions were extracted with Nucleospin kit (Macherey-Nagel, Germany) according to the manufacture’s protocol and stored at −80°C. Medium supernatants were also collected, centrifuged at 1500rpm/min and frozen at −80°C for ELISA and pharmacokinetic analysis.
Pharmacokinetics analysis
Conditioned medium obtained from control and drug-treated cells from the 24h incubation with VRL/DVRL and the 96h metronomic experiment (daily sampling, for 4 consecutive days) was shipped on dry ice to the Institute de Recherché Pierre Fabre (Castres, France), where they were analyzed. Concentrations of VRL and DVRL were quantified using a sensitive liquid chromatography tandem mass spectrometry method previously reported [14,15]. The method has been used and validated for assessment of drug levels in human biological samples. For in vitro systems a partial in-house validation has been done at Centre de Recherche et Développement Pierre Fabre (personal communication, data not published).
Proliferation Assay
Proliferation inhibition testing was performed on HUVEC seeded in collagen-coated 96-well plates. Cells were harvested from cultures (at 80% confluence) by trypsinization, were counted and were plated at optimal seeding densities of 1000cells/well. Cells were attached 24h later and then, the medium was replaced with growth medium containing material to be tested and the cells were incubated at 37°C, 5% CO2 for 24 and 96h. To maintain a constant concentration of the drugs during the protracted experimental period of 96h, the medium was carefully removed every 24h and fresh solutions were added. Cell numbers were determined by using a Cell Titter 96® AQueous One Solution Cell Proliferation (MTS) Assay (Promega, Madison, WI, USA). Briefly, 20μl of MTS solution was added to each well and the absorbance was recorded 2h later. Results were expressed as the percentage of control cell proliferation and half-maximal inhibitory concentrations (IC50) were assessed by using the Prism4 software (GraphPad Software, Inc. La Jolla, CA, USA).
Apoptosis/Cell cycle analysis
To determine the proportion of the apoptotic cells, cells were treated with the appropriate concentrations of the experimental drugs at the conditions described above. Then, 5×105 cells were diluted at 400μl binding buffer of Annexin (included in Annexin V-FITC kit, Assay Designs, DK-700). Part of the suspension (96μl) was incubated with 1μl Annexin V-FITC solution and 2.5μl Propidium Iodine (250μg/ml) on ice for 15min in the dark, diluted to 250μl binding buffer, and apoptotic cells were counted with flow cytometry (Partec CyFlow ML, Germany).
To analyze cell cycle, both control and drug-treated cells were trypsinized, collected and fixed with cold 70% ethanol for 12h at 4°C. After fixation, cells were centrifuged at 1500rpm/min, treated with 2ml Cystain DNA 1 step/DAPI (PARTEC, Germany) at room temperature for 5min, and analyzed using UV excitation (Partec CyFlow ML, Germany).
qRT-PCR
To determine the mRNA expression of the angiogenesis modulating genes CD36, COX-2, IL-8, PPARgamma and TSP-1 we performed real time Reverse Transcription-Polymerase Chain Reaction (qRT-PCR) in a CFX96 Real-Time System (C1000 Thermal Cycler, BIO-RAD, USA) with IQ Supermix (BIO-RAD, USA) using specific primers/probe sets (Hs00354519_m1, Hs01573471_m1, Hs00174103_m1, Hs01115513_m1 and Hs00170236_m1, respectively; Taqman Gene Expression Assays, Applied Biosystems, USA).
For cDNA synthesis, 9μl of total RNA (1μg RNA) was reverse transcribed in 20μl of reaction mixture containing 10mM dNTP, 5x reaction buffer, random hexamer primer [pd(N)6], 40units/μl of ribonuclease inhibitor and 20units/μl reverse transcriptase (Fermentas, EU). The reaction mix was incubated at 70°C for 3min, chilled in ice for 5min, incubated at 37°C for 1h and the reaction stopped with incubation at 70°C for 10min. qRT-PCR was performed with the following amplification settings: 50°C for 2min and 95°C for 10min for the AmpliTaq activation, then 40 cycles of 15sec denaturation at 95°C, 15sec annealing at 60°C, 1min extension at 60°C and melting.
Enzyme-linked immunosorbent assay (ELISA)
Quantitative sandwich enzyme immunoassay technique was used to quantitate secreted IL-8 and TSP-1 (R&D Systems, MN, USA) in the conditioned medium obtained from control and drug-treated cells from the 96h metronomic experiment. The concentration of proteins in unknown samples was determined by comparing the absorbance of the samples to the standard curve, according to the manufactures protocol.
Western blotting
Total cellular proteins were extracted and protein concentration was determined using the suggested method for protein quantification in protein loading buffer PLB-TCEP as described NucleoSpin RNA/Protein isolation kit. Samples containing 10μg of protein were analyzed by 10% SDS-PAGE, transferred to nitrocellulose transfer membranes (Protran, Schleicher & Schuell, Germany) and subjected to immunoblot analysis. Antibodies used in this study were: PPARgamma (sc-7273) and CD36 (sc-70642) from Santa Cruz Biotechnology (Germany), COX-2 (07-693; Upstate, NY, USA) and a mouse monoclonal to β-actin (CloneAC15, A5441; Sigma). Signals were revealed using horseradish peroxidase (HRP)-conjugated secondary antibodies (Santa Cruz Biotechnology, Germany) and developed with an ECL detection kit (GE HealthCare). Protein signals were normalized to the corresponding b-actin stain signal.
Nuclear and cytosolic protein extraction
Nuclear and cytoplasmic extracts from HUVEC were prepared as previously described [16]. Briefly, HUVEC cells plated into 10cm dishes were grown to 80% confluence and treated with 10nM of VRL for different time-periods (0.5, 1, 3, and 16h). Cells were collected and suspended in cold hypotonic buffer; following centrifugation, the cells were re-suspended and lysed in hypotonic buffer containing NP-40 (0.1%). Samples were then centrifuged and the cytoplasmic proteins was collected and stored (−80°C). The pellets were suspended in 50μl ice-cold high salt extraction buffer and incubated on ice for 30min. The nuclear proteins were collected after centrifugation at 13000rpm for 30min at 4°C and stored at −80°C for further analysis.
Protein concentration was determined by the Bio-Rad Assay (Bio-Rad Laboratories, Hercules, CA, USA). Cytoplasmic or nuclear extracts (10μg) were resolved by 10% SDS-polyacrylamide gel electrophoresis, transferred to nitrocellulose membranes and probed with rabbit polyclonal anti-NF-kB RelA/p65 (sc-372) and goat polyclonal anti-NF-κB p50 (sc-1190) as first antibodies and lamin B (sc-6216) or β-actin as second antibodies.
Statistics
For statistical analysis we used the GraphPad Prism4 software (GraphPad Software, Inc. La Jolla, CA, USA). Comparisons between groups were analyzed using Student’s t-test. A probability value of less than 0.05 was considered statistically significant.
Results
1. Stability of VRL concentrations in tissue culture
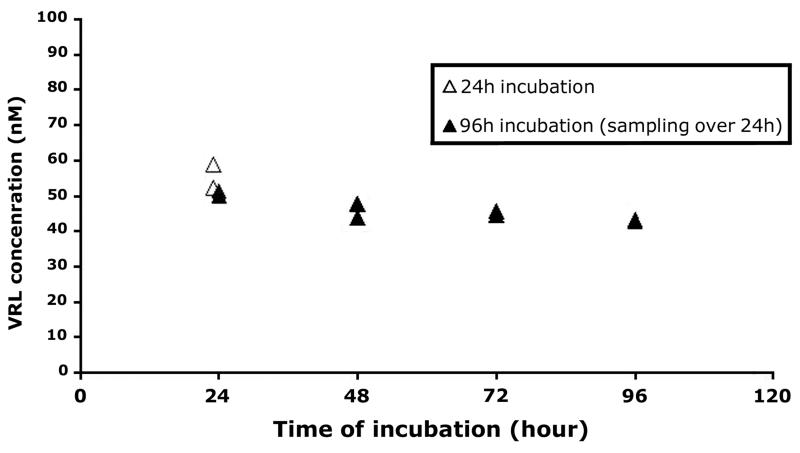
Drug analysis showed dose-related linearity for VRL levels in our in vitro system (R2=0.98; Figure S1). In addition, in our metronomic model the levels of VRL and DVRL in culture medium remained constant over the 96h exposure period (Figure 1 and supplemental digital content 2). For the active metabolite DVRL, mass spectrometry analysis revealed similar dose-related linearity to the parent compound (R2=0.9025; Figure S1) providing evidence that DVRL active metabolite can be synthesized through VRL in HUVEC over a period of 24h without accumulation. Overall, at the 96h period the conditioned medium had constant concentration of both drugs, which meant in vitro simulation of metronomic exposure.
Figure 1. VRL/DVRL concentrations after incubation of 100nM of substrate in HUVEC.
Cells exposed to VRL/DVRL (100nM) for 24h as well as for the metronomic protocol of 96h with daily medium change. Samples collected at the end of the 24h period.
2. Effect of VRL on endothelial cell proliferation
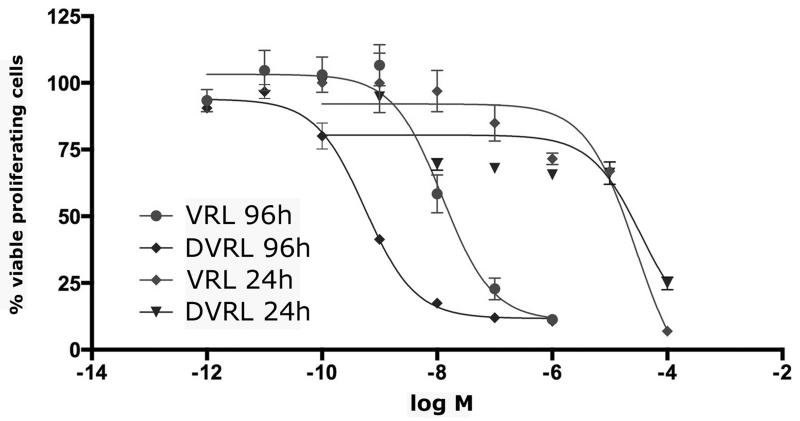
The half-maximal inhibitory concentration (IC50), at 96h, was four orders of magnitude lower compared to 24h-exposure (1.23nM vs. 32μM for the VRL and 0.55nM vs. 78μM for DVRL). Moreover, we found DVRL to be more active than the VRL when considering endothelial cell growth (Figure 2).
Figure 2. Inhibition of endothelial cell proliferation, by VRL and DVRL.
HUVEC were exposed to the indicated concentrations of VRL/DVRL for either 24 or 96h. Viable cells were determined by using an MTS assay. Results are expressed as the % percentage of viable control cells and half-maximal inhibitory concentrations (IC50).
3. VRL effects on cell cycle distribution and apoptosis of HUVEC
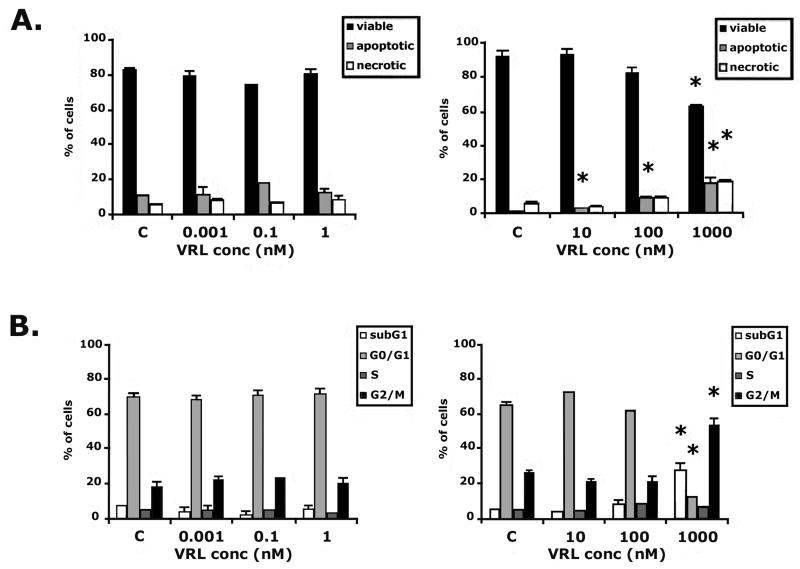
HUVEC were treated with VRL by the metronomic protocol in the range from 0.001nM to 1nM for 96h and in the conventional protocol exposed at concentrations from 10nM to 1μM for 4h followed by a wash-out period of 92h. VRL treatment in the metronomic model (0.001 to 1nM) had no significant effects on cell cycle progression and apoptosis but short term exposure at conventional concentrations induced both early and late apoptosis and necrosis; the dose of 1μM significantly increased the percentage of cells at sub-G1, decreased the number of viable cells and induced their accumulation in G2-M cell cycle phase (p=0.001; Figure 3).
Figure 3. VRL on apoptosis and cell cycle.
HUVEC were treated with VRL for 96h (0.001, 0.1 and 1nM; metronomic model) or for 4h with a wash-out period of 92h (10 to 1000nM; conventional chemotherapy). Percentages of viable, total apoptotic and necrotic cells (A) or cells in cell cycle phases (B) in metronomic (left) vs. conventional (right) chemotherapy were recorded. *p<0.05 compared with control group.
4. Low dose continuous exposure to VRL influences the expression of angiogenesis modulators at mRNA level
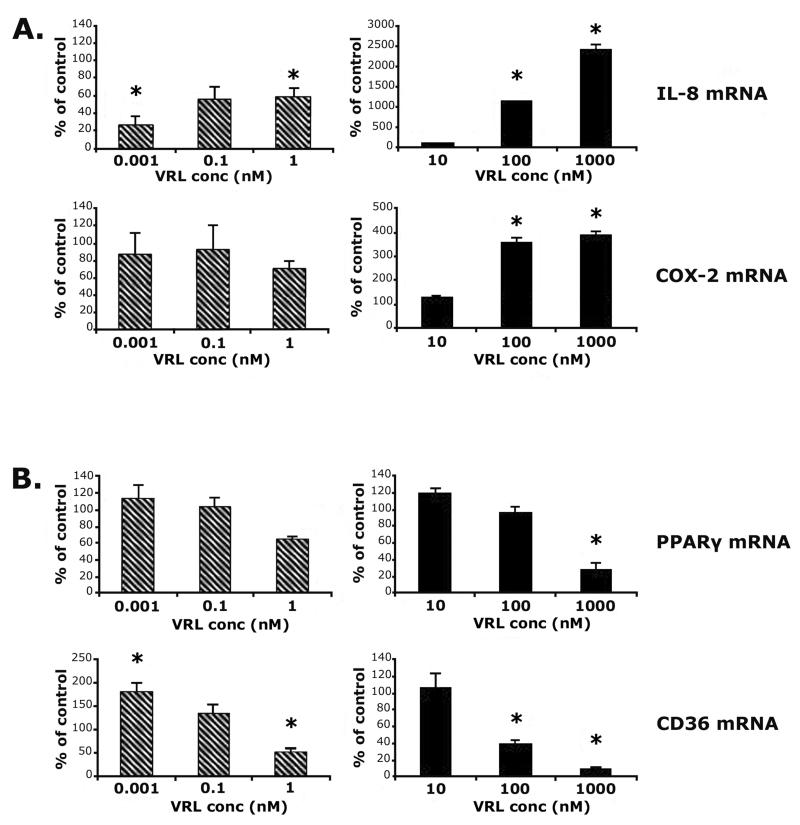
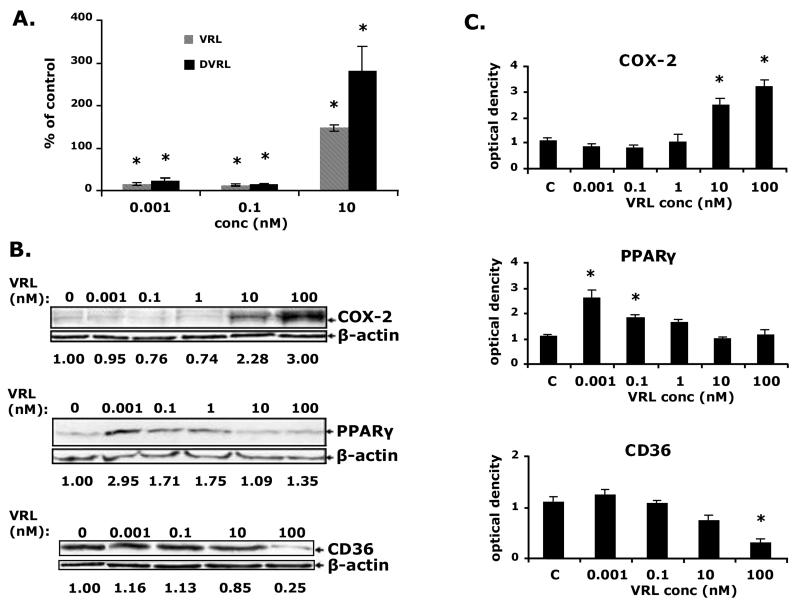
Quantitative RT-PCR was carried out to investigate whether VRL could influence synthesis of transcripts of angiogenesis modulators. Metronomic concentrations of VRL led to dose-dependent decreases of pro-angiogenic (IL-8) and increases of anti-angiogenic genes (PPARgamma and CD36; Figure 4). Protracted exposure to low concentrations of VRL (0.001, 0.1 or 1nM) suppressed mRNA levels of IL-8 (p<0.05), but did not affect COX-2 (Figure 4A). However, expression of both genes were induced when cells were exposed to VRL concentrations above 10nM, using the conventional chemotherapy protocol (Figure 4A).
Figure 4. VRL on angiogenic markers.
Angiogenic (A) and anti-angiogenic (B) markers after applying the metronomic (left) or the conventional (right) chemotherapy in vitro model.*p<0.05 compared with control group.
For PPARgamma, low concentrations VRL (0.001 and 0.1nM) had no effect on basal levels but, the lowest tested dose of 0.001nM, induced the expression of CD36 (p<0.05) at the metronomic schedule of 96h exposure (Figure 4B). In contrast, concentrations higher than 10nM caused a significant reduction of mRNA of anti-angiogenic molecules PPARgamma (p<0.05 at 1μM) and CD36 (p<0.05 at 100nM; Figure 4B).
5. Decrease of IL-8 and increase of PPARgamma secreted proteins at metronomic exposure of HUVEC to VRL
Similar effects were seen on protein level following exposure of HUVEC to metronomic concentrations of VRL. At metronomic schedule, 0.001nM and 0.1nM of VRL diminished the secreted IL-8 protein (p<0.05; Figure 5A), and increased the PPARgamma protein levels (3 times the control levels; Figure 5B). Protein expression of COX-2, CD36 was similar to control. However, higher concentrations of VRL (10 and 100nM) either had no effect on anti-angiogenic proteins e.g. PPARgamma or lowered CD36 (p<0.05, figure 5A). This range of concentration also increased angiogenic COX-2 protein expression (Figure 5B) and secreted IL-8 protein concentrations (Figure 5A). As far as DVRL is concerned, we observed that DVRL had the same activity profile as VRL for IL8 (figure 5A).
Figure 5. VRL/DVRL on angiogenic and anti-angiogenic proteins in the protracted administration model.
A) IL-8 and TSP-1 protein levels were determined by ELISA B)protein levels of COX-2, PPARgamma and CD36 were determined by western blotting using respective antibodies;last blot shows only the non-glycosylated but full-lenth CD36 protein (55kDa). Numbers under the blotts indicate normalized to b-actin values of respective proteins. Representative figures of 2 independent experiments are shown. *p<0.05 compared with control group.
6. Involvement of NF-κB in VRL-induced IL-8 and COX-2 transcription
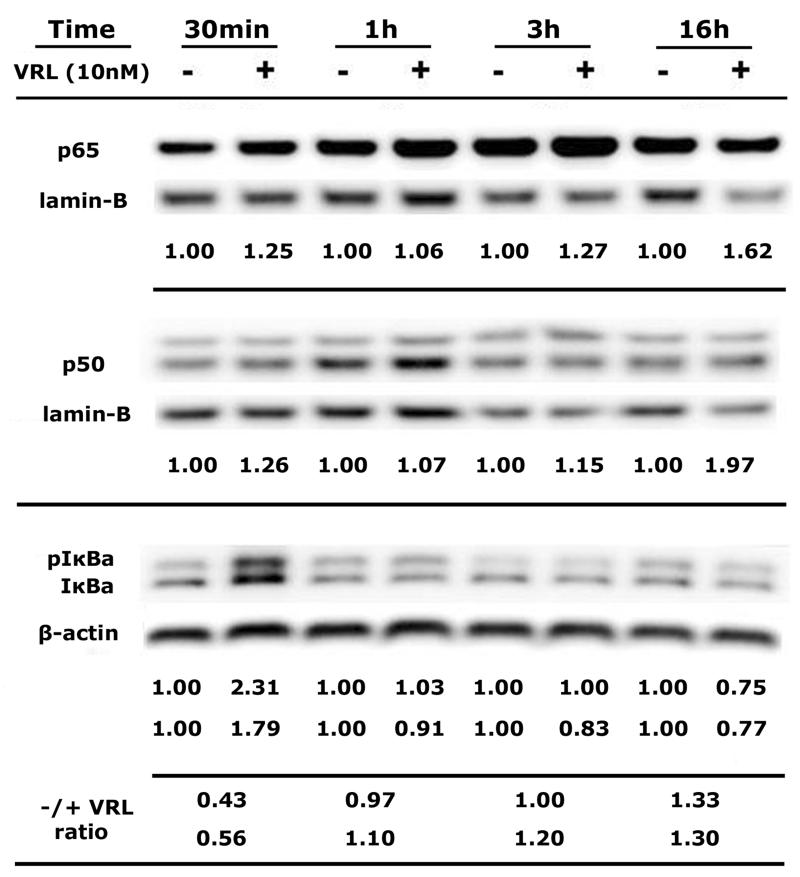
To determine the potential involvement of transcriptional factor NF-κB in VRL-induced IL-8 and COX-2 signalling pathways, cytosolic and nuclear extracts were isolated from HUVEC after a time-course of 0.5 to 16h exposure to 10nM VRL, which considered an estimated value of Cmax of metronomic VRL in the clinical setting. (Figure 6). Western blot analysis was performed for NF-κB subunits of p65 and p50 and for IκBa; expression was calculated after normalization to lamin-B or β-actin. Protein expression of both NF-κB subunits were increased in the nucleus even at 30min and remained elevated until 16h after adding VRL. The translocation of p65 and p50 to the nucleus accompanied by elevated levels of phosphorylated and non-phosphorylated IκBa [18] at the first 30min after VRL treatment, an effect that minimized at 1h; and followed by degradation of pIκBa and IκBa as ratio values for both forms (before and after VRL) became greater than 1.0, over time (Figure 6).
Figure 6. Activation of NF-kB pathway by VRL.
Both the p65 and p50 sub-units determined in nuclear extracts of HUVEC (normalized to lamin-B) and IkBa proteins in cytosol (normalized to b-actin). Presented values express the proteins levels of NF-kB units normalized to respective house keeping protein, before and after VRL treatment; for IkBa, values are for pIkBa and IkBa, respectively. Ratio before and after VRL treatment at each time point, for pIkBa (upper line) and IkBa (lower line). Representative figures of 2 independent experiments are shown.
Discussion
Metronomic oral vinorelbine was been shown in clinical studies to yield sustainable antitumor activity in a number of metastatic cancers, possibly through an anti-angiogenic mechanism [10,12,17]. We sought to investigate the cellular and molecular effects of metronomic VRL and DVRL on HUVEC, in an attempt to elucidate underlying mechanisms of its anti-angiogenic activity. We found that metronomic exposure of HUVEC to VRL/DVRL halts growth of endothelial cells at low nanomolar concentrations and induces molecular effects that favor the expression of anti-angiogenic modulators and suppress the expression of pro-angiogenic modulators.
Metronomic chemotherapy is by concept an endothelial cell–targeted antiangiogenic therapy, which exploits the high turnover rate and remarkable sensitivity of endothelial cells to cytotoxic agents [7]. For this reason, we selected HUVEC cultures, which are considered an acceptable model of proliferating endothelial cells. Regarding the selection of most appropriate drug concentrations, we took into consideration the pharmacokinetic analysis data of clinical studies [10, 12]. In this context, we considered determination of drug levels in our in vitro study was important to ensure that conclusions could be relevant to the clinic. We found that the levels of VRL in culture medium remained constant over the 96h exposure period in our model, which is in line with clinical data that showed no evidence of accumulation over time. We understand as limitations of this work, the fact that we opted not to include tumor cells in this study for focusing on the effects of metronomic vinorelbine on endothelial cells, and the relatively limited number of angiogenesis modulators selected for investigation, which however are among the most common, and some are therapeutic targets on their own [18-20].
One of the important steps in anti-angiogenesis is the inhibition of endothelial cell proliferation. In our in vitro metronomic model, the half-maximal inhibitory concentration (IC50) of vinorelbine was 1,23nM. This concentration is lower and possibly not representative of clinically observed metronomic trough drug levels in the clinical studies [10,12]. Interestingly, these drug concentrations did not have an obvious effect on apoptosis or the cell cycle. In contrast, in the study arm that was compatible with conventional chemotherapy the IC50 was much higher (~30μΜ) and these concentrations produced G2-M cell cycle arrest and induced apoptosis.
Regarding angiogenesis modulators, we found that metronomic and conventional chemotherapy model influenced the basal levels of IL-8 and PPARgamma in dose-dependent manner, but in the opposite way. Characteristically, metronomic VRL was found to decrease both mRNA and secreted protein of IL-8 while conventional treatment produced a marked increase of IL-8 mRNA. Keeping IL-8 at low levels is potentially important in cancer therapy. Characteristically, over-expression of IL-8 has been associated with tumor progression and metastasis [21] and in a clinical trial of metronomic oral VRL, patients who benefited from this therapy tended to have low levels of IL-8 [10]. A pathway closely related to IL-8 is that of COX-2, an enzyme that stimulates angiogenesis. Low metronomic concentrations of VRL did not affect the expression of COX-2, but high concentrations for 96h of treatment activated protein production of COX-2. On the other hand, metronomic concentrations of VRL that reduced IL-8 levels caused induction of protein expression of the anti-angiogenic PPARgamma. Concentrations of VRL above 10nM, which increased angiogenic molecules (IL-8 and COX-2), minimized the production of PPARgamma protein. The anti-angiogenic TSP-1 receptor CD36 was not affected by metronomic treatment of HUVEC whereas conventional treatment decreased CD36 at both mRNA and protein level.
Previous studies indicate that PPARgamma, COX-2 and IL-8 signalling pathways are regulated by NF-κB [19,22]. A recent study showed the involvement of NF-κB in VRL induced oxidative injury but with doses higher than those used in typical chemotherapy [23]. In our study, VRL at a concentration of 10nM caused instant rapid activation of the NF-κB pathway as shown by p65/p50 nuclear translocation and IκBa release, which indicates potential involvement of NF-κB in affecting IL-8 and COX-2 up-regulation and PPARgamma deactivation by VRL concentrations just above the steady state trough levels of metronomic dosing.
Conclusion
Protracted exposure of endothelial cells to vinorelbine at very low concentrations, that simulate steady state serum levels achieved with chronic metronomic administration of oral vinorelbine, has shown superiority compared to short high dose exposure which simulates conventional pulse dosing of vinorelbine, in inducing anti-angiogenic effects at cellular and molecular levels. This study provides experimental evidence that supports recent favorable clinical findings of metronomic oral vinorelbine and suggests potential therapeutic combinations that could be of clinical interest.
Supplementary Material
Figure S1: Pooled concentrations of VRL and DVRL in HUVEC cells, over 96h incubation. Samples collected at the end of 24h period of cell exposure to 10, 100 and 10000nM of VRL. Data from 12, 12 and 4 samples from each treatment group, respectively.
Acknowledgements
We thank Eleftheria Hatzimichael for her contribution in the writing of this article; we also thank Pierre Fabre Hellas for providing us both vinorelbine and 4-O-deacetylvinorelbine. This work is supported by HESMO (Hellenic Society of Medical Oncology; E.M.)
Abbreviations
- HUVEC
Human Umbilical Vein Endothelial Cells
- VRL
vinorelbine
- DVRL
4-O-deacetylvinorelbine
Footnotes
Competing interests
The authors declare that they have no competing interests.
References
- 1.Shaked Y, Kerbel RS. Antiangiogenic strategies on defense: on the possibility of blocking rebounds by the tumor vasculature after chemotherapy. Cancer Res. 2007;67(15):7055–8. doi: 10.1158/0008-5472.CAN-07-0905. [DOI] [PubMed] [Google Scholar]
- 2.Frei E, III, Canellos GP. Dose: a critical factor in cancer chemotherapy. Am J Med. 1980;69(4):585–94. doi: 10.1016/0002-9343(80)90472-6. [DOI] [PubMed] [Google Scholar]
- 3.Hanahan D, Bergers G, Bergsland E. Less is more, regularly: metronomic dosing of cytotoxic drugs can target tumor angiogenesis in mice. J Clin Invest. 2000;105(8):1045–7. doi: 10.1172/JCI9872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hatzimichael E, Briasoulis E. Metronomic chemotherapy beyond misconceptions. Haematologica. 2013;98(11):145. doi: 10.3324/haematol.2013.096917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Folkman J. Angiogenesis: an organizing principle for drug discovery? Nat Rev Drug Discov. 2007;6(4):273–86. doi: 10.1038/nrd2115. [DOI] [PubMed] [Google Scholar]
- 6.Harris AL. Antiangiogenesis for cancer therapy. Lancet. 1997;349(Suppl 2):SII13–SII15. doi: 10.1016/s0140-6736(97)90014-3. [DOI] [PubMed] [Google Scholar]
- 7.Kerbel RS, Kamen BA. The anti-angiogenic basis of metronomic chemotherapy. Nat Rev Cancer. 2004;4(6):423–36. doi: 10.1038/nrc1369. [DOI] [PubMed] [Google Scholar]
- 8.Ganguly A, Yang H, Zhang H, Cabral F, Patel KD. Microtubule dynamics control tail retraction in migrating vascular endothelial cells. Mol Cancer Ther. 2013;12(12):2837–46. doi: 10.1158/1535-7163.MCT-13-0401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schwartz EL. Antivascular actions of microtubule-binding drugs. Clin Cancer Res. 2009;15(8):2594–601. doi: 10.1158/1078-0432.CCR-08-2710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Briasoulis E, Pappas P, Puozzo C, Tolis C, Fountzilas G, Dafni U, et al. Dose-ranging study of metronomic oral vinorelbine in patients with advanced refractory cancer. Clin Cancer Res. 2009;15(20):6454–61. doi: 10.1158/1078-0432.CCR-09-0970. [DOI] [PubMed] [Google Scholar]
- 11.Wargin WA, Lucas VS. The clinical pharmacokinetics of vinorelbine (Navelbine) Semin Oncol. 1994;21(5 Suppl 10):21–7. [PubMed] [Google Scholar]
- 12.Briasoulis E, Aravantinos G, Kouvatseas G, Pappas P, Biziota E, Sainis I, et al. Dose selection trial of metronomic oral vinorelbine monotherapy in patients with metastatic cancer: a hellenic cooperative oncology group clinical translational study. BMC Cancer. 2013;13(1):263. doi: 10.1186/1471-2407-13-263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marty M, Fumoleau P, Adenis A, Rousseau Y, Merrouche Y, Robinet G, et al. Oral vinorelbine pharmacokinetics and absolute bioavailability study in patients with solid tumors. Ann Oncol. 2001;12(11):1643–9. doi: 10.1023/a:1013180903805. [DOI] [PubMed] [Google Scholar]
- 14.Van Heugen JC, De GJ, Zorza G, Puozzo C. New sensitive liquid chromatography method coupled with tandem mass spectrometric detection for the clinical analysis of vinorelbine and its metabolites in blood, plasma, urine and faeces. J Chromatogr A. 2001;926(1):11–20. doi: 10.1016/s0021-9673(01)00993-1. [DOI] [PubMed] [Google Scholar]
- 15.Puozzo C, Ung HL, Zorza G. A high performance liquid chromatography method for vinorelbine and 4-O-deacetyl vinorelbine: a decade of routine analysis in human blood. J Pharm Biomed Anal. 2007;44(1):144–9. doi: 10.1016/j.jpba.2007.01.011. [DOI] [PubMed] [Google Scholar]
- 16.Sfikas A, Batsi C, Tselikou E, Vartholomatos G, Monokrousos N, Pappas P, et al. The canonical NF-kappaB pathway differentially protects normal and human tumor cells from ROS-induced DNA damage. Cell Signal. 2012;24(11):2007–23. doi: 10.1016/j.cellsig.2012.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Addeo R, Sgambato A, Cennamo G, Montella L, Faiola V, Abbruzzese A, et al. Low-dose metronomic oral administration of vinorelbine in the first-line treatment of elderly patients with metastatic breast cancer. Clin Breast Cancer. 2010;10(4):301–6. doi: 10.3816/CBC.2010.n.039. [DOI] [PubMed] [Google Scholar]
- 18.Perkins ND. The diverse and complex roles of NF-kappaB subunits in cancer. Nat Rev Cancer. 2012;12(2):121–32. doi: 10.1038/nrc3204. [DOI] [PubMed] [Google Scholar]
- 19.Waugh DJ, Wilson C. The interleukin-8 pathway in cancer. Clin Cancer Res. 2008;14(21):6735–41. doi: 10.1158/1078-0432.CCR-07-4843. [DOI] [PubMed] [Google Scholar]
- 20.Huang S, Robinson JB, Deguzman A, Bucana CD, Fidler IJ. Blockade of nuclear factor-kappaB signaling inhibits angiogenesis and tumorigenicity of human ovarian cancer cells by suppressing expression of vascular endothelial growth factor and interleukin 8. Cancer Res. 2000;60(19):5334–9. [PubMed] [Google Scholar]
- 21.Lee YS, Choi I, Ning Y, Kim NY, Khatchadourian V, Yang D, et al. Interleukin-8 and its receptor CXCR2 in the tumour microenvironment promote colon cancer growth, progression and metastasis. Br J Cancer. 2012;106(11):1833–41. doi: 10.1038/bjc.2012.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kaplan J, Cook JA, O'Connor M, Zingarelli B. Peroxisome proliferator-activated receptor gamma is required for the inhibitory effect of ciglitazone but not 15-deoxy-Delta 12,14-prostaglandin J2 on the NFkappaB pathway in human endothelial cells. Shock. 2007;28(6):722–6. doi: 10.1097/SHK.0b013e318055683a. [DOI] [PubMed] [Google Scholar]
- 23.Tsai KL, Chiu TH, Tsai MH, Chen HY, Ou HC. Vinorelbine-induced oxidative injury in human endothelial cells mediated by AMPK/PKC/NADPH/NF-kappaB pathways. Cell Biochem Biophys. 2012;62(3):467–79. doi: 10.1007/s12013-011-9333-y. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1: Pooled concentrations of VRL and DVRL in HUVEC cells, over 96h incubation. Samples collected at the end of 24h period of cell exposure to 10, 100 and 10000nM of VRL. Data from 12, 12 and 4 samples from each treatment group, respectively.