Abstract
The COVID-19 pandemic and the ensuing barriers to the collection and transport of donor cells, it is often necessary to collect and cryopreserve grafts before initiation of transplant conditioning. The effect on transplant outcomes in non-malignant disease is unknown. This analysis examined the effect of cryopreservation of related and unrelated donor grafts for transplantation for severe aplastic anemia in the US during 2013-2019. Included are 52 recipients of cryopreserved grafts who were matched for age, donor type, and graft type to 194 recipients who received non-cryopreserved grafts. Marginal Cox regression models were built to study the effect of cryopreservation and other risk factors associated with outcomes. We recorded higher 1-year rates of graft failure (HR 2.26, 95% CI 1.17 – 4.35, p=0.01) and of 1-year overall mortality (HR 3.13, 95% CI 1.60 – 6.11, p=0.0008) after transplantation of cryopreserved compared to non-cryopreserved grafts with adjustment for sex, performance score, comorbidity, cytomegalovirus serostatus, and ABO blood group match). Acute and chronic GVHD did not differ between groups. Adjusted probabilities of 1-year survival were 73% (95% 60 – 84) and 91% (95% CI 86 – 94) with cryopreserved and non-cryopreserved grafts, respectively. These data support the use of non-cryopreserved grafts, when possible, for severe aplastic anemia.
INTRODUCTION
The emergence of coronavirus disease 2019 (COVID-19) as a global pandemic triggered an unprecedented worldwide health-care crisis. It also impacted the world economy and disrupted travel across international borders and within countries. These travel restrictions combined with potentially reduced HCT donor availability (due to infection, quarantine, and constraints on travel to collection centers) and complex allograft processing logistics (donor assessment, collection, on-schedule delivery for fresh infusion) directly impacted the ability to infuse fresh donor-cells into intended recipients on the scheduled day of transplantation. Consequently, the American Society for Transplantation and Cellular Therapy (ASTCT)1 and the National Marrow Donor Program/Be The Match (NMDP)2 issued strong recommendations that unrelated donor products should be delivered and cryopreserved at transplant centers before initiation of patient conditioning. NMDP now requires that grafts be delivered and cryopreserved at the transplant center before initiation of transplant-conditioning regimen for any patient intended to undergo unrelated donor hematopoietic cell transplantation (HCT), in the absence of unique considerations.2 Many transplant centers also instituted a similar practice for related donor HCT since related donors face many of the same issues as unrelated donors. The use of cryopreserved grafts introduces more flexibility and has occurred sporadically for several decades, although to our knowledge the practice has been ad hoc.3 Several reports examined the effect of transplantation of cryopreserved grafts for hematologic malignancy, including one done very recently by the Center for International Blood and Marrow Transplant Research (CIBMTR), in response to the need for information during the COVID-19 pandemic. None showed a difference in survival.4-7 To our knowledge, there are no reports of outcomes after transplantation of cryopreserved related or unrelated donor graft for non-malignant hematologic diseases. Thus, the current analysis was undertaken to inform clinical practice for transplantation for severe aplastic anemia, a common non-malignant indication for HCT.
METHODS
Patients
Patients with severe aplastic anemia were identified from the database of the CIBMTR and transplanted between 2013 and 2019 in the United States. Donors included HLA-matched siblings, haploidentical relatives, HLA-matched and HLA-mismatched unrelated adults who donated bone marrow or peripheral blood. Recipients of cord blood transplants were excluded as all units are cryopreserved. Patients were followed longitudinally until death or loss to follow-up. Patients or their legal guardians provided written informed consent for the study. The Institutional Review Board of the NMDP approved this study.
Endpoints
The primary outcome was 1-year survival. Death from any cause was considered an event and surviving patients were censored at 1-year or earlier for follow-up less than 1 year. Neutrophil recovery was defined as the first of 3 consecutive days with absolute neutrophil count (ANC) ≥ 0.5 x 109 /L, and platelet recovery, ≥ 20 x 109/L without transfusion for 7 days. Graft failure was defined as a failure to achieve ANC ≥0.5 x 109/L or ANC decline to <0.5 x 109/L without recovery after having achieved ANC ≥0.5 x 109/L, or myeloid donor chimerism (<5%), or second transplant.8 Other outcomes studied were grade II-IV acute and chronic GVHD, graded using standard criteria.9,10
Statistical Analysis
Fifty-two patients (cases) who were transplanted with a cryopreserved graft were matched on age (≤17, 18 – 39 and ≥40 years),11,12 donor type (HLA-matched sibling, haploidentical relative, HLA-matched or HLA-mismatched unrelated donor)12,13 and graft type (bone marrow or peripheral blood)14,15 to 195 controls from a pool of 979 patients transplanted during the same period with non-cryopreserved grafts. Forty-five cases were matched to 4 controls, 2 were matched to 3 controls, 4 were matched to 2 controls and 1 was matched to 1 control.
To study the effect of cryopreserved compared to non-cryopreserved grafts, (matched-pairs) marginal Cox regression models were built and adjusted for sex, cytomegalovirus serostatus, performance score, comorbidity score, and donor-recipient ABO blood group match.16 All variables met the assumptions for proportional hazards. Results are expressed as hazard ratio (HR) with 95% confidence interval (CI). Adjusted probabilities for outcomes of interest were generated from the marginal Cox model.17,18 The level of significance was p-value ≤0.01 (two-sided), in consideration of the multiple comparisons. Analyses were done using SAS version 9.4 (Cary, NC).
RESULTS
Patients and Transplant Characteristics
The characteristics of the treatment groups matched for age, donor type and graft type, are shown in Table 1.11-15 Females were more likely to receive cryopreserved grafts but other characteristics such as recipient cytomegalovirus serostatus, performance score, comorbidity index and donor-recipient ABO blood group match, transplant conditioning regimen and GVHD prophylaxis were similar between treatment groups. Although the total nucleated cell doses (TNC) of harvested bone marrow were similar between the groups, the TNC dose infused differed with recipients of cryopreserved bone marrow grafts receiving significantly lower cell doses (Table 1). The difference between cell dose at harvest and infusion was statistically significant (paired t-test, p=0.0008). CD34 doses for peripheral blood grafts were not significantly different between cryopreserved and non-cryopreserved grafts (Table 1). The median follow-up of surviving cases and controls was 35 months (range 6 – 74) and 26 months (range 5 – 76), respectively.
Table 1.
Patient and Transplant Characteristics
| Controls (non- cryopreserved graft) |
Cases (cryopreserved graft) |
p-value | |
|---|---|---|---|
| Number | 195 | 52 | |
| Age, median (range) | 22 (4-67) | 21 (5-64) | 0.96 |
| 1 – 17 years | 77 (40%) | 21 (40%) | 0.95 |
| 18 – 39 years | 72 (37%) | 18 (35%) | |
| ≥ 40 years | 46 (24%) | 13 (25%) | |
| Sex, male/female | 115 (59%)/80 (41%) | 22 (42%)/30 (58%) | 0.03 |
| Performance score | 0.72 | ||
| 90 – 100 | 128 (66%) | 31 (60%) | |
| ≤80 | 61 (31%) | 19 (37%) | |
| Not reported | 6 ( 3%) | 2 ( 4%) | |
| Co-morbidity score | 0.55 | ||
| ≤2 | 136 (70%) | 34 (65%) | |
| ≥3 | 59 (30%) | 18 (35%) | |
| Cytomegalovirus serostatus | 0.13 | ||
| Negative | 58 (30%) | 10 (19%) | |
| Positive | 137 (70%) | 42 (81%) | |
| Donor type* | 0.97 | ||
| HLA-matched sibling | 79 (41%) | 21 (41%) | |
| HLA-haploidentical | 25 (13%) | 8 (15%) | |
| HLA-matched unrelated | 64 (33%) | 16 (31%) | |
| HLA-mismatched unrelated | 27 (14%) | 7 (14%) | |
| Donor-recipient ABO match | 0.86 | ||
| Matched | 63 (32%) | 19 (37%) | |
| Minor mismatch | 22 (11%) | 6 (11%) | |
| Major mismatch | 31 (16%) | 6 (11%) | |
| Not reported | 79 (41%) | 21 (41%) | |
| Conditioning regimen** | 0.15 | ||
| Cy + ATG | 59 (30%) | 11 (21%) | |
| Flu + Cy + ATG | 18 (9%) | 6 (12%) | |
| Bu/Cy ± ATG | 4 (2%) | 3 (6%) | |
| Flu + TBI (200 cGy) | 1 (<1%) | 1 (2%) | |
| Cy + ATG + TBI (200 cGy) | 38 (19%) | 8 (15%) | |
| Cy + ATG + TBI (1000 cGy) | 0 | 1 (2%) | |
| Flu + Cy + ATG + TBI (200 cGy) | 55 (28%) | 13 (25%) | |
| Flu + Bu ± ATG | 8 (4%) | 5 (10%) | |
| Flu + melphalan + thiotepa + ATG | 2 (1%) | 2 (4%) | |
| Flu + melphalan | 10 (5%) | 2 (4%) | |
| Graft type | 0.56 | ||
| Bone marrow | 132 (68%) | 33 (64%) | |
| Peripheral blood | 63 (32%) | 19 (36%) | |
| Bone marrow | |||
| Total nucleated cells (x108/kg) Median (IQR) | |||
| Pre-cryopreservation | Not applicable | 3.83 (2.70 – 5.07) N = 19 of 33 |
|
| Infusion | 3.40 (2.45 – 4.57) N = 109 of 132 |
2.63 (1.49 – 3.05) N = 23 of 33 |
0.004 |
| Peripheral blood | |||
| CD34+ (x 106/kg) Median (IQR) |
|||
| Pre-cryopreservation | Not applicable | 7.90 (7.14 – 8.74) N = 15 of 19 |
|
| Infusion | 6.63 (4.78 – 10.97) N = 62 of 63 |
5.38 (3.78 – 10.97) N = 15 of 19 |
0.45 |
| GVHD prophylaxis | 0.07 | ||
| Ex vivo T cell depletion or CD34+ | 18 (10%) | 4 (8%) | |
| Post-transplant Cy + other | 22 (11%) | 6 (12%) | |
| Calcineurin inhibitor + MMF | 21 (11%) | 14 (27%) | |
| Calcineurin inhibitor + MTX | 110 (56%) | 25 (48%) | |
| Calcineurin inhibitor + other | 21 (11%) | 2 (4%) | |
| Other agents | 3 (2%) | 1 (2%) | |
| Interval, diagnosis to transplant | 0.28 | ||
| ≤ 3 months# | 44 (23%) | 8 (15%) | |
| 3 – 6 months╪ | 41 (21%) | 15 (29%) | |
| 7 – 12 months║ | 42 (22%) | 15 (29%) | |
| >12 months# | 68 (35%) | 14 (27%) | |
| Transplant period | 0.16 | ||
| 2013 – 2015 | 103 (53%) | 20 (39%) | |
| 2016 – 2019 | 92 (47%) | 32 (61%) |
Abbreviations:
ABO = blood group A, B, O
Cy = cyclophosphamide; ATG = anti-thymocyte globulin; Flu = fludarabine; TBI = total body irradiation
IQR = inter quartile range
MMF = mycophenolate mofetil; MTX = methotrexate
Donor age, median (range):
Haploidentical: Controls: 32 years (10-65); Cases: 36 years (14-65)
Unrelated: Controls: 27 years (18-59); Cases: 30 years (19-43)
Cyclophosphamide dosing:
Cy + ATG
Cases: Cy dose=200 mg/kg (n=11)
Controls: Cy dose= 200 mg/kg (n=56), Cy dose=120 mg/kg (n=3)
Flu + Cy + ATG
Cases: Cy dose=120 mg/kg (n=5), Cy dose 60 mg/kg (n=1)
Controls Cy dose= 120 mg/kg (n=15), Cy dose 60 mg/kg (n=3)
Bu + Cy
Cases: Cy dose=200 mg/kg (n=1), Cy dose=120 mg/kg (n=2)
Controls Cy dose=200 mg/kg (n=2), Cy dose=120 mg/kg (n=2)
Cy + ATG + TBI (200 cGy)
Cases: Cy dose=200 mg/kg (n=6), Cy dose 29 mg/kg Cases (n=2)
Controls: Cy dose=200 mg/kg (n=22), Cy dose 120 mg/kg (n=1), Cy dose=100 mg/kg (n=4), Cy dose=50 mg/kg (n=2), Cy dose 29 mg/kg (n=8), Cy dose unknown (n=1)
Cy + ATG + TBI (1000 cGy)
Cases: Cy dose=120 mg/kg (n=1)
Flu + Cy + ATG + TBI (200 cGy)
Cases: Cy dose=100 mg/kg (n=4), Cy dose=50 mg/kg (n=4), Cy dose=29 mg/kg (n=5)
Controls: Cy dose=100 mg/kg (n=19), Cy dose=50 mg/kg (n=15), Cy dose=29 mg/kg (n=19)
Interval between diagnosis and transplant
77% were HLA-matched sibling and 23% were HLA-matched or mismatched unrelated donor transplants
55% were HLA-matched sibling, 30% were HLA-matched or mismatched unrelated donor and 14% were HLA-haploidentical transplants
23% were HLA-matched sibling, 59% were HLA-matched or mismatched unrelated donor and 19% were HLA-haploidentical transplants
20% were HLA-matched sibling, 63% were HLA-matched or mismatched unrelated donor and 17% were HLA-haploidentical transplants
Hematopoietic recovery
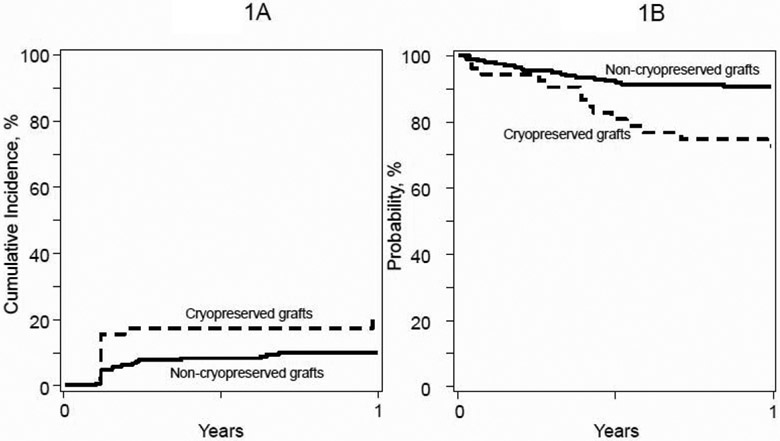
We did not record statistically significant differences in day-28 neutrophil recovery between those who received cryopreserved and fresh grafts, 83% (95% CI 71 – 92), and 91% (95% CI 86 – 94), p=0.17, respectively. The corresponding incidences of day-100 platelet recovery were 91% (95% CI 79 – 98) and 90% (95% CI 86 – 94), p=0.89. In multivariate analysis, the rate of neutrophil (HR 0.76, 95% CI 0.54 – 1.08, p=0.13) and platelet (HR 0.77, 95% CI 0.57 – 1.04, p=0.08) recovery was lower with transplantation of cryopreserved grafts but the difference did not reach statistical significance. However, the risk of 1-year graft failure was higher after transplantation of cryopreserved compared to non-cryopreserved graft (HR 2.26, 95% CI 1.17 − 4.35, p=0.01), Figure 1A. Graft failure was primary for 7 patients who received cryopreserved and for 8 patients who received non-cryopreserved grafts. Three patients who received cryopreserved and for 11 patients who received non-cryopreserved grafts were reported to have developed secondary graft failure. The likelihood of hematopoietic recovery and risk for graft failure were adjusted for sex, recipient cytomegalovirus serostatus, performance score, comorbidity index, and blood group ABO match.
Figure 1.
Graft failure and overall survival
A. The 1-year graft failure for cryopreserved and non-cryopreserved transplants was 19% (95% CI 10 – 31) and 10% (95% CI 6 – 14), respectively
B. The 1-year overall survival for cryopreserved and non-cryopreserved transplants was 73% (95% CI 60 – 84) and 91% (95% CI 86 – 94), respectively
Acute and chronic graft-versus-host disease
We did not observe differences in grade II-IV acute GVHD (HR 0.93, 95% CI 0.41 – 2.13, p=0.87) and chronic GVHD (HR 0.79, 95% CI 0.41 – 1.50, p=0.46) by treatment group. The day-100 incidence of grade II-IV acute GVHD after transplantation of cryopreserved and non-cryopreserved grafts were 12% (95% CI 5 – 22) and 13% (95% CI 8 – 18), p=0.94, respectively. The corresponding incidence of 1-year chronic GVHD was 23% (95% CI 12 – 37) and 28% (95% CI 21 – 35), p=0.49.
Overall survival
One-year mortality was higher after transplantation of cryopreserved compared to non-cryopreserved grafts (HR 3.31, 95% CI 1.60 – 6.11, p=0.0008) after adjusting for sex, recipient cytomegalovirus serostatus, performance score, comorbidity index and blood group ABO match. The adjusted 1-year probabilities of overall survival were 73% (95% 60 – 84) with cryopreserved and 91% (95% CI 86 – 94) with non-cryo-reserved grafts, Figure 1B. We also evaluated mortality risks without censoring at 1-year post-transplantation and observed similar HRs of mortality after transplantation of cryopreserved products. A subset analysis limited to 19 cryopreserved peripheral blood transplants and 63 controls also showed higher graft failure (HR 2.98, 95% CI 0.92 – 9.64, p=0.06) and higher mortality (HR 3.84, 95% CI 1.44 – 10.21, p=0.007) with cryopreservation. Seventeen patients (17 of 52, 33%) died after transplantation of cryopreserved grafts; primary disease was reported as the predominant cause of death (13 of 17, 76%). Other causes of death included GVHD (n=2), infection (n=1) and hemorrhage (n=1). Thirty-three patients (33 of 194, 17%) died after transplantation of non-cryopreserved grafts; primary disease was also reported as the predominant cause of death (24 of 33, 73%). Other causes of death included infection (n=3), interstitial pneumonitis (n=2), organ failure (n=2) and hemorrhage (n=3).
DISCUSSION
The current analysis was undertaken to examine whether there are differences in survival or other transplant outcomes after transplantation of cryopreserved bone marrow or peripheral blood for severe aplastic anemia. Recipients of cryopreserved grafts were matched to recipients of non-cryopreserved grafts for age at transplantation, donor type/donor-recipient HLA-match and graft type, factors that are consistently associated with transplant outcomes for this disease.11-15 The analyses also considered the effect of other potential risk factors on transplant-outcomes. After carefully controlled analyses, we observed higher graft failure and mortality rates after transplantation of cryopreserved compared to non-cryopreserved grafts. Thus, our findings favor the transplantation of non-cryopreserved grafts for severe aplastic anemia.
Transplant conditioning-regimen for severe aplastic anemia varies by type of donor.19 Other reports have shown an effect of conditioning-regimen for survival after HLA-matched sibling transplants.19 None of the patients in the current analysis received Cy alone or Cy with Flu, the conditioning regimens that are associated with higher graft failure and mortality rates.19 The cell dose of the graft has also been associated with graft failure.20 It is recommended that bone marrow grafts contain a minimum of 3 x 108/kg TNC to avoid graft failure.20 These data derive from an analysis of non-cryopreserved bone marrow grafts. Data on infused bone marrow TNC was available for only 70% (23 of 33) cryopreserved and 83% (109 of 132) and non-cryopreserved grafts. Despite this limitation, we observed significantly lower TNC infused after cryopreservation of bone marrow grafts. Although 68% of patients receiving cryopreserved bone marrow grafts had ≥3 x 108/kg TNC harvested, only 26% had that number infused. This loss of cells may have resulted in the observed differences in outcomes. The difference between TNC at harvest and infusion implies cryopreservation/thaw process is associated with cell loss. However, there may be other unmeasured or unknown factors that also resulted in the observed differences in outcome. We do not have data on cell function at any time point. An earlier report on the functional assay of cryopreserved bone marrow suggests preservation of cell function although that report included only 7 grafts.21 An analysis of non-cryopreserved bone marrow cellular subsets for unrelated donor transplantations failed to show an effect of graft composition on hematopoietic recovery or survival although that report included only 7 patients with aplastic anemia.22
All cryopreserved peripheral blood grafts in the current analysis had CD34+ doses greater than 2 x 106/kg, the recommended minimum dose for severe aplastic anemia.23 A subset analysis limited to recipients of peripheral blood was consistent with the findings of the main analysis. Cryopreserved CD34+ cells from peripheral blood are reported to have significant loss of membrane integrity, viability, and CFU potential that collectively could contribute to the adverse effect of transplantation of cryopreserved peripheral blood in our study.23 We hypothesize that several factors led to the poor outcomes after transplantation of cryopreserved grafts. Optimizing cell dose is desirable but a controlled study that examines for changes in graft composition with cryopreservation/thaw that is specific for aplastic anemia is needed. A detailed analysis of the composition and function of cryopreserved grafts is beyond the scope of this study. We did not observe statistically significant differences in neutrophil and platelet recovery despite lower rates of recovery after transplantation of cryopreserved grafts. We hypothesize the absence of significant differences is attributed the modest number of patients in the current analysis. We do not know the indication for using a cryopreserved graft for the patients included in this analysis. The data on the interval between diagnosis and transplant was not different between the treatment groups. Further, the timing of transplantation by donor type is also consistent with accepted clinical practice guidelines. HLA-matched sibling transplants were mostly offered within 6 months from diagnosis and alternative donor transplants later after failure of at least one course of immunosuppressive therapy.24 Recipients of cryopreserved and non-cryopreserved grafts were matched for graft type (bone marrow or peripheral blood). Subset analyses limited to peripheral blood transplants confirmed higher graft failure and mortality consistent with the main analysis and suggest a greater effect than with bone marrow grafts.
These findings differ from findings in studies of patients receiving cryopreserved grafts for hematologic malignancies. Patients with malignancy often come to transplant after multiple chemotherapy and immune-suppressive therapies and also usually receive more intensive pretransplant conditioning than patients with aplastic anemia. For these reasons, and perhaps because of difference in the nature of the underlying diseases, the risk of graft failure is in general lower after HCT for malignant disease than the risk after HCT aplastic anemia and may be less affected by any alterations in cell dose or function induced by cryopreservation.
In summary, the data support the use of non-cryopreserved bone marrow or peripheral blood for transplantation for severe aplastic anemia. If this is not possible, it may be prudent to delay transplantation until it is. These transplants are often not deemed urgent and every effort must be made to provide the best available supportive care for the patient until the transplant center can ensure the availability of a non-cryopreserved graft. If a delay is not possible, careful assessment of the risk of cryopreservation versus the risk of not receiving the graft when it is needed is necessary. The NMDP/Be The Match considers the diagnosis of aplastic anemia a valid reason to try to deliver a fresh graft for unrelated donor transplants for severe aplastic anemia.
Acknowledgments
Funding Source
The Center for International Blood and Marrow Transplant Research is supported primarily by Public Health Service Grant/Cooperative Agreement 5U24-CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI) and the National Institute of Allergy and Infectious Diseases (NIAID); contract HHSH250201200016C with Health Resources and Services Administration (HRSA/DHHS); grants N00014-15-1-0848 and N00014-16-1-2020 from the Office of Naval Research. The views expressed in this article do not reflect the official policy or position of the National Institute of Health, the Department of the Navy, the Department of Defense, Health Resources and Services Administration or any other agency of the U.S. Government.
Footnotes
Conflict of interest
Dr. Hamadani receives research support from Takeda Pharmaceutical Company; Otsuka Pharmaceutical; Spectrum Pharmaceuticals; Astellas Pharma. Consultancy: Incyte Corporation; ADC Therapeutics; Celgene Corporation; Pharmacyclics, Magenta Therapeutics, Omeros, AbGenomics, Verastem, TeneoBio.
Speaker’s Bureau: Sanofi Genzyme, AstraZeneca.
Dr. D’Souza receives research support from Takeda Pharmaceutical Company; Sanofi Genzyme, AstraZeneca; TeneoBio; Prothena; EDO; Mundipharma Consulting fees: Prothena, Pfizer, Akcea, Imbrium, Janssen
The other authors declare no financial conflicts of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.ASTCT RESOURCES FOR COVID-19. https://www.astct.org
- 2.National Marrow Donor Program (Be The Match). https://network.bethematchclinical.org/news/nmdp/be-the-match-response-to-covid-19/
- 3.Frey NV, Lazarus HM, Goldstein SC. Has allogeneic stem cell cryopreservation been given the 'cold shoulder'? An analysis of the pros and cons of using frozen versus fresh stem cell products in allogeneic stem cell transplantation. Bone Marrow Transplant. 2006; 38:399–405. [DOI] [PubMed] [Google Scholar]
- 4.Stockschlader M, Hassan HT, Krog C, et al. Long-term follow-up of leukaemia patients after related cryopreserved allogeneic bone marrow transplantation. Br J Haematol. 1997; 96:382–386. [DOI] [PubMed] [Google Scholar]
- 5.Kim DH, Jamal N, Saragosa R, et al. Similar outcomes of cryopreserved allogeneic peripheral stem cell transplants (PBSCT) compared to fresh allografts. Biol Blood Marrow Transplant. 2007; 13:1233–1243. [DOI] [PubMed] [Google Scholar]
- 6.Medd P, Nagra S, Hollyman D, Craddock C, Malladi R. Cryopreservation of allogeneic PBSC from related and unrelated donors is associated with delayed platelet engraftment but has no impact on survival. Bone Marrow Transplant. 2013; 48:243–248. [DOI] [PubMed] [Google Scholar]
- 7.Hamadani M, Zhang M-J, Tang X-Y, et al. Graft cryopreservation does not impact overall survival allogeneic hematopoietic cell transplantation using post-transplant cyclophosphamide for GVHD prophylaxis. Biol Blood Marrow Transplant. 2020. (https://doi.Org/10.1016/j.bbmt.2020.04.001) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Olsson R, Remberger M, Schaffer M, et al. Graft failure in the modern era of allogeneic hematopoietic SCT. Bone Marrow Transplant. 2013; 48: 537–543 [DOI] [PubMed] [Google Scholar]
- 9.Przepiorka D, Weisdorf D, Martin P, et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant. 1995; 15: 825–828 [PubMed] [Google Scholar]
- 10.Atkinson K, Horowitz MM, Gale RP, Lee MB, Rimm AA, Bortin MM. Committee of the International Bone Marrow Transplant Registry. Consensus among bone marrow transplanters for diagnosis, grading and treatment of chronic graft versus host disease. Bone Marrow Transplant. 1989; 4: 247–254 [PubMed] [Google Scholar]
- 11.Gupta V, Eapen M, Brazauskas R, et al. Impact of age on outcomes after bone marrow transplantation for acquired aplastic anemia using HLA-matched sibling donors. Haematologica. 2010; 95: 2119–2125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bacigalupo A How I treat acquired aplastic anemia. Blood. 2017; 129:1428–1436 [DOI] [PubMed] [Google Scholar]
- 13.Horan J, Wang T, Haagenson M, et al. Evaluation of HLA matching in unrelated hematopoietic cell transplantation for nonmalignant disorders. Blood. 2012; 120:2918–2924 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schrezenmeier H, Passweg JR, Marsh JC et al. Worse outcome and more chronic GVHD with peripheral blood progenitor cells than bone marrow in HLA-matched sibling donor transplants for young patients with severe acquired aplastic anemia. Blood. 2007; 110:1397–1400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Eapen M, Le Rademacher J, Antin JH et al. Effect of stem cell source on outcomes after unrelated donor transplantation in severe aplastic anemia. Blood. 2011; 118:2618–2621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cox DR. Regression models and life tables. J R Stat Soc. 1972; 34:187 – 220 [Google Scholar]
- 17.Zhang X, Loberiza FR, Klein JP, Zhang MJ. A SAS macro for estimation of direct adjusted survival curves based on a stratified Cox regression model. Comput Methods Programs Biomed. 2007; 88:95 – 101 [DOI] [PubMed] [Google Scholar]
- 18.Zhang X, Zhang MJ. SAS macros for estimation of direct adjusted cumulative incidence curves under proportional subdistribution hazards models. Comput Methods Programs Biomed. 2011; 101: 87–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bejanyan N, Kim S, Hebert KM, et al. Choice of conditioning regimens for bone marrow transplantation in severe aplastic anemia. Blood Adv. 3:3123–3131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Marsh JC, Ball SE, Cavenagh J, et al. Guidelines for the diagnosis and management of aplastic anemia. Br J Haematol. 2009; 147:43–70 [DOI] [PubMed] [Google Scholar]
- 21.Islam MS, Anoop P, Dutta-Nemdharry P et al. Implications of CD34+ cell dose on clinical and hematological outcome of allo-SCT for acquired aplastic anemia. Bone Marrow Transplant. 2010; 45:886–894 [DOI] [PubMed] [Google Scholar]
- 22.Collins NH, Gee AP, Durett AG, et al. Effect of the composition of unrelated donor bone marrow and peripheral blood progenitor cell grafts on transplantation outcomes. Biol Blood Marrow Transplant. 2010; 16:253–262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lioznov M, Dellbrugger C, Sputlet A, et al. Transportation and cryopreservation may impact hematopoietic stem cell function and engraftment of allogeneic peripheral blood stem cells and not bone marrow. Bone Marrow Transplant. 2008; 42:121–128 [DOI] [PubMed] [Google Scholar]
- 24.Young NS. Aplastic anemia. N Engl J Med. 2018; 379:1643–1656 [DOI] [PMC free article] [PubMed] [Google Scholar]